Summary
One of the most prominent features at the mid-blastula transition (MBT) observed in most embryos is a pause in cell cycle regulated by the nucleocytoplasmic (N/C) ratio. By using chromosome rearrangements to manipulate the DNA content of embryos, we determined that the threshold for this cell cycle pause in Drosophila is about 70% of the DNA content normally present at cycle 14. Embryos with DNA contents around this value show intermediate cell cycle behaviors. Some pause at cycle 14, some at cycle 15, and some form patches arrested in different mitotic cycles. A second feature at MBT is a massive increase in zygotic transcription and a parallel degradation of maternally supplied RNAs. To determine whether these changes in gene expression are governed by the same N/C ratio that controls cell cycle pause, we compared gene expression in haploid and diploid Drosophila embryos. We find that most maternal RNA degradation and most new transcription correlate with absolute time or developmental stage, and are timed independently of the N/C ratio. We identify a class of zygotically active genes whose expression depends on the N/C ratio and which are only expressed at cycle 15 in haploids. In embryos with patchy cell cycle behavior due to threshold DNA contents, the expression of these genes correlates tightly with the boundaries of the mitotic patches, suggesting either that the mechanism that pauses the mitotic cycle is the same as the one that measures the N/C ratio, or that it is tightly coupled to the mechanism controlling zygotic transcription of N/C ratio genes at the MBT.
Keywords: Drosophila, Mid-blastula transition, Nucleocytoplasmic ratio, Transcription, Cell cycle
INTRODUCTION
The development of most metazoan embryos is characterized by a series of rapid, synchronous cell divisions, followed by a dramatic slowing of the cell cycle, the onset of asynchronous cleavages and the initiation of morphogenetic movements (O'Farrell et al., 2004). The alteration in cell cycle behavior occurs coordinately with both the onset of bulk transcription from the zygotic genome and the degradation of maternal RNAs required for early development prior to the activation of the zygotic genome (McKnight and Miller, 1976; Zalokar, 1976). Despite the near universality of this developmental switch, known as the mid-blastula transition (MBT), the molecular mechanisms that regulate the MBT and coordinate alterations in the cell cycle timing with the activation of zygotic transcription and the degradation of maternal messages remain largely unknown.
In most organisms, the MBT occurs after a defined number of cell cycles and thus at a fixed time after fertilization. However, previous work has shown that the transition is not controlled by the absolute time after fertilization or by a cell cycle counting mechanism. Instead, the ratio of nuclear content to cytoplasmic volume (the N/C ratio) controls the timing of the MBT and the onset of zygotic transcription in organisms as diverse as Drosophila and Xenopus. In Xenopus embryos, where wholesale zygotic transcription normally begins after 12 cycles of DNA replication and mitosis, manipulations that artificially increase the DNA content cause premature activation of the zygotic genome (Newport and Kirschner, 1982a; Newport and Kirschner, 1982b). Similarly, in Drosophila embryos, which normally undergo 13 rapid cleavage divisions, reduction of the DNA content by half in haploid embryos results in an extra cell cycle, with a corresponding delay of the MBT (Edgar et al., 1986). These observations have led to a model in which the exponential increase in DNA content during cleavage stages results in the titration and inactivation of a hypothetical cytoplasmic factor that inhibits onset of the MBT before the embryo attains the correct N/C ratio. However, it is unknown whether the control of the MBT requires the presence of specific genomic intervals and/or certain DNA sequences, or, conversely, depends upon total DNA content irrespective of specific sequences. Moreover, proper development requires that every cell in the embryo responds robustly and simultaneously to the N/C ratio, but no prior study has addressed how the N/C ratio ensures a robust response or to what degree the embryo is susceptible to variations in N/C values.
The burst of zygotic transcription seen at the MBT is correlated with the elimination of maternal RNAs (Mathavan et al., 2005; Pilot et al., 2006). In zebrafish, zygotic activation and maternal degradation are coupled through the transcription of specific microRNAs that promote maternal RNA deadenylation and clearance (Giraldez et al., 2006). A similar role for zygotically expressed miRNAs has been postulated for maternal RNA degradation during Drosophila MBT (Bushati et al., 2008). However, no mechanistic links have yet been established between the alterations in cell cycle and the switch from maternally to zygotically driven developmental processes. It is unclear whether these changes in gene expression are controlled by the same processes that alter cell cycle downstream of the N/C measurement. Previous studies identified a small number of genes whose transcriptional activation appears to be controlled by the N/C ratio, as well as genes whose expression appears independent of that ratio (Grosshans et al., 2003; Grosshans and Wieschaus, 2000; Yasuda and Schubiger, 1991). No studies have characterized global transcriptional activation in the context of an altered N/C ratio, and thus it is uncertain how tightly general transcriptional activation is coupled to cell cycle control of the MBT.
Here, we address these questions using an array of genetic tools available in Drosophila. By using compound chromosome rearrangements (Merrill et al., 1988), we found that cell cycle behavior does not require the presence of any specific genomic interval. We determined that the coordinated, robust initiation of MBT requires an N/C ratio above about 70% of that found in a normal embryo at cycle 14. Embryos whose DNA content is near this 70% threshold display patches of nuclei paused at either cycle 14 or cycle 15. Next, we compared the transcriptomes of MBT-delayed haploid embryos with those of wild-type diploid embryos. We found that most zygotic transcription, as well as the accompanying maternal RNA degradation, does not rely on the N/C ratio, but instead occurs at a strict time interval following fertilization, indicating the presence of a timing mechanism that operates independently of the N/C ratio. We also identified a small class of 88 zygotically active genes whose expression does depend on the N/C ratio. We found that in embryos with DNA content near the 70% threshold, the expression of this class of genes correlates precisely with boundaries of cell cycle behavior, arguing that the mechanism that reads the N/C ratio regulates both cell cycle and the temporal expression of this gene class. By contrast, N/C ratio-independent genes were expressed in their correct spatial patterns irrespective of cell cycle behavior, providing further support for the existence of two independent mechanisms modulating the onset of zygotic gene expression near the MBT.
MATERIALS AND METHODS
Fly strains and genetics
w; Histone 2A-GFP was used as wild type; the H2A-GFP transgene was crossed into the maternal haploid strain to obtain mh/FM7; H2A-GFP. w ssm[185b]/FM7c was a gift from Kami Ahmad (Harvard University, USA). The compound II chromosome RM(2L); RM(2R) (=C(2)v) and compound III chromosome RM(3L); RM(3R) (=C(3)se) are maintained at Princeton, the compound II C(2) EN and compound III C(3) EN chromosomes were obtained from the Bloomington Stock Center (stock numbers 2974 and 1117), as were the compound stocks with free fragments (stock numbers 2682: y[1];F(3L)2, h2; C(3R)RM-P3, sr and 2707: y; C(2L)RM-P2,dpov1; F(2R)1, bw). To introduce the Histone 2A-GFP marker into the compound stocks, the transgene was first jumped from its original location on the third chromosome to the X chromosome, and then either the third chromosomal or X-chromosomal transgene was crossed into the compound lines as the rare survivors of nondisjunction in the female.
Time-lapse fluorescence microscopy
Two- to 3-hour embryos were collected at room temperature from apple-juice agar plates, dechorinated and placed onto a slide with carbon-hydro oil 27 (Sigma). Fluorescent time-lapse images were taken using a 20× objective on CARVI spinning disc microscope at 20 second intervals. The movies were compressed to 10 frames/second.
Microarray and data analysis
Embryos were identified under an Epifluorescence microscope, timed from telophase of the previous cycle to 15 and 40 minutes later, hand-selected and frozen by dry-ice chilled heptane (Sigma). Triplicates of about 50 embryos each at the desired age were collected for each time point. RNA extraction and microarray procedures were performed according to the standard Affymetrix protocols. Eighteen Drosophila 2.0 Affymetrix chips were used. Chip signal was normalized by the Loess method and log-transformed before all data analyses. For hierarchical clustering, genes were centered and normalized in Gene Cluster 3.0 before clustering. Complete linkage was used with Euclidean distance. Microarray data reported herein have been deposited at the NCBI Gene Expression Omnibus (http://www.ncbi.nlm.nih.gov/geo/) with the accession number GSE14287.
Multi-dimensional scaling (MDS) is a dimensionality reduction technique that takes as input a matrix of distances between gene expression profiles, e.g. across multiple time points, and returns an arrangement of points in a two-dimensional space, so that each point represents a gene, and the distances between points in the two dimensions are as close as possible to the original distances (Cox and Cox, 2001). MDS comes in multiple variants, and in this paper, we use Kruskal's non-metric multi-dimensional scaling, implemented by the isoMDS() function in the R statistical software.
Quantitative RT-PCR
Embryos were collected the same way as in the microarray experiment. RNA extraction was performed following the TRIzol protocol (Invitrogen) and cDNA was synthesized using the SuperScript III First-Strand Synthesis System for RT-PCR (Invitrogen). Quantitative PCR was performed using an ABI 7900 Real Time PCR System. Triplicates were done for each genotype and each time point. Primers used were: nos-F, AGCCGATCATCACCATGGA; nos-R, CTTGGCTAGGCGGAACGAT; Hsp83-F, ACATGGAGGAGGTCGATTAAGC; Hsp83-R, TGCGAGTGATAGAATGAATTTTGG; bcd-F, AGTTGCCGCCACAATTCC; bcd-R, GCTCTTGTCCAGACCCTTCAAA; stg-F, AATCGGCGACTTCAGCAAA; stg-R, CGATGACGACCCTCCATCA; twe-F, TGCCAGCACCACCGTTCT; twe-R, GGCGGGCTCCATTCATATT; tub56D-F, GCATGGACGAGATGGAGTTCA; and tub56D-R, CTCGGACACCAGATCGTTCAT.
In situ hybridization
Two- to 4-hour embryos were collected, dechorinated, and fixed with 4% paraformaldehyde. In most in situ hybridization experiments, standard digoxigenin-labeled RNA probes were detected using alkaline phosphatase-conjugated secondary antibodies. Probes against different genes were made by PCR and in vitro transcription (Invitrogen transcription kit). Primers for each probe were designed by PRIMER3.0 (Rozen and Skaletsky, 2000). During the final wash, Hoechst 33342 was applied for DNA staining. Embryos were mounted in Aqua-Polymount (Polysciences). For fluorescent in situ (FISH), mouse-anti-digoxigenin (1:200) primary antibody (Roche) was used followed by anti-mouse Alexa Fluor 488 (Invitrogen). Nascent transcripts were visualized using a Leica SP5 confocal microscope.
RESULTS
Nonspecific alteration of genomic content modifies the N/C ratio and controls mitotic behavior
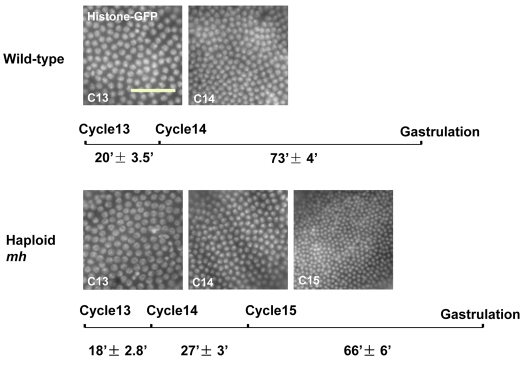
To compare the temporal dynamics of mitotic division in haploid embryos to normal diploids, we used homozygous maternal haploid (mh) females, which produce only haploid embryos, and monitored transgenically expressed Histone-GFP by time-lapse fluorescence microscopy. Whereas diploid and haploid embryos possess similar cell cycle duration and lengthening of interphase 13, haploid embryos failed to pause after the thirteenth division, and instead underwent an extra round of synchronous mitosis about 27 minutes into cycle 14 interphase (Fig. 1). Consistent with previous results (Edgar et al., 1986), in haploid embryos cellularization was initiated but aborted at cycle 14 and then completed at cycle 15. The subsequent cycle 15 interphase was comparable in length to the diploid cycle 14, although the nuclei showed an irregular distribution during the nuclear elongation phase and cellularization, presumably because of their increased density (see Fig. S1A in the supplementary material).
Fig. 1.
N/C ratio-regulated development of haploid embryos. Nuclear density of embryos at different stages visualized by Histone-GFP. Scale bar: 50 μm. The duration of cycles 13, 14 and 15 is based on time-lapse movies of eight wild-type and eight haploid embryos.
The additional mitosis allows mh embryos to reach the N/C ratio of wild-type embryos at cycle 14, confirming a role for the N/C ratio in timing the cell cycle pause at the Drosophila MBT. However, the results do not distinguish whether specific chromosomal loci are required to induce MBT, or alternatively if total DNA content, irrespective of specific sequence, determines the N/C ratio. To distinguish between these two possibilities, we used a set of chromosomal rearrangements [C(1)DX/Y, C(2)v and C(3)se] that allowed us to replace one arm of a given chromosome with a duplicate copy of the opposite arm of the same chromosome. Because the left and right arms of each chromosome are similar in size (Fig. 2A), embryos produced from such rearrangements lack one chromosomal arm, but possess a total DNA content similar to the wild-type one (see Fig. S1B in the supplementary material). We found that nearly all such embryos followed a normal cell cycle pattern, regardless of the removal of any portion of the genome (see Table 1). The occasional extra divisions observed in C(3)se stocks could be attributed to tribbles and fruhstart on chromosome III, which have been shown to have small but reproducible effects on the cell cycle at MBT (Grosshans et al., 2003; Grosshans and Wieschaus, 2000). These results support the view that the N/C ratio is not determined by any individual zygotic locus.
Fig. 2.
Titration of N/C ratio by modifying DNA content affects mitotic cycles. (A) DNA quantity of each chromosomal arm expressed as a percentage of the entire genome. (B) Manipulation of DNA quantity using compound stocks. Embryos from C(3)EN are shown. C(2)EN is similar. (C) Embryos with different amounts of DNA pause at different mitotic cycles; higher magnification views are shown on the right. Some form mitotic patches with different nuclear densities (see Table 1). Arrow indicates the boundary of the mitotic patch in the cycle 13/cycle 14 embryo; the patch with the lower cycle 13 density is to the right, the asterisk indicates the region with the higher cycle 14 nuclear density. Scale bar for higher magnification images: 50 μm.
Table 1.
Titration of N/C ratio by modifying DNA content with compound stocks
| DNA content (%) | Cross/Stock | Embryo genotype | Cycle 13 (%) | Cycle 14 (%) | Cycle 15 (%) | n |
|---|---|---|---|---|---|---|
| 50 | mh | mh/mh | 0 | 2 | 98 (5) | 187 |
| 50 | ssm | ssm/ssm | 0 | 1 | 99 (0) | 204 |
| 60 | C(3)EN(chr3-) | chr3- | 0 | 0 | 100 (2) | 77 |
| 66 | C(2)EN(chr2-) | chr2- | 0 | 0 | 100 (6) | 74 |
| 69 | C(3R);F(3L) × C(3)EN | chr3R- (1×chr3L) | 0 | 71 | 29 (29) | 98 |
| 75 | C(2L);F(2R) × C(2)EN | chr2L- (1×chr2R) | 0 | 96 | 4 (4) | 61 |
| 76 | C(1)/C(1,Y) | X- | 0 | 88 | 12 (12) | 177 |
| 84 or 82 | c2v | chr2L- or chr2R- | 0 | 97 | 3 (3) | 176 |
| 98 or 102 | c2v | chr2L- (4×chr2R) or chr2R- (4×chr2L) | ||||
| 116 or 118 | c2v | 4×chr2L or 4×chr2R | 0 | 100 | 0 | 143 |
| 82 or 78 | c3se | chr3L- or chr3R- | 0 | 84 | 16 (16*) | 171 |
| 98 or 102 | c3se | chr3L- (4×chr3R) or chr3R- (4×chr3L) | ||||
| 118 or 122 | c3se | 4×chr3L or 4×chr3R | 0 | 99 | 1 (1) | 109 |
| 100 | C(1)DX × w/Y | Y/Y(X-) | 0 | 99 | 1 (1) | 200 |
| 100 | OreR | +/+ | 0 | 100 | 0 | 309 |
| 134 | C(2)EN | chr2+ | 15 (10) | 85 | 0 | 84 |
| 140 | C(3)EN | chr3+ | 13 (2) | 87 | 0 | 189 |
Embryos paused in interphase (judged by nuclear elongation and cellularization) were counted and the percentage calculated. The percentage in parentheses indicates the patchy embryos of the corresponding cycle.
The increased percentage of patchy embryos in c3se is probably due to tribbles or fruhstart on chromosome III.
To test whether total DNA accounts for N/C ratio-dependent behavior, we used compound stocks (C(2)EN or C(3)EN) in which the entire diploid content of a particular autosome has been fused to the same centromere. Such stocks produce embryos that lack all copies of a specific chromosome (Fig. 2B) while retaining normal diploid complement of the others. The total DNA content per nucleus in such embryos is reduced to 66% of the wild-type content for C(2)EN and 60% for C(3)EN. Even though such embryos are diploid for the majority of their genome, they showed haploid cell cycle behavior, and underwent an additional fifteenth mitosis (Table 1). This indicates that the presence of two copies of any single gene does not ensure a diploid-like N/C ratio at cycle 14 and, coupled with the normal behavior of C(2)v and C(3)se embryos, demonstrates that nonspecific alterations in total DNA content modulate the N/C ratio and control cell cycle behavior.
A robust, coordinated mitotic response requires an N/C ratio above 70% of that of cycle 14 wild-type embryos
Embryos lacking either chromosome II or chromosome III have 10% more DNA than do true haploids, yet this increase in nuclear content is not sufficient to suppress an additional mitotic cycle. To determine the minimum DNA content necessary to induce cell cycle pause, we used additional chromosomal rearrangements to generate embryos with DNA content ranging from 60-85% of wild type (see Fig. S1B in the supplementary material; Table 1). Embryos with more than 75% of the normal DNA content behaved like wild-type embryos and paused at cycle 14, whereas embryos with less than 66% of normal DNA content generally underwent an additional mitotic division. By contrast, embryos whose DNA content was between 66% and 75% displayed an intermediate behavior, with many pausing at either cycle 14 or 15. A fraction of these embryos displayed intermixed regions of different nuclear densities, with some regions paused at cycle 14 and others at cycle 15 (Fig. 2C, Table 1). These results suggest that a DNA content above about 70% of that normally present at cycle 14 is required for the coordinated pause in mitotic progression.
If an N/C ratio near 70% of that seen in wild type at cycle 14 represents a threshold value needed to alter mitotic behavior, embryos reaching this value prematurely should show an inappropriately early pause in cell cycle progression. Therefore, we examined nuclear division and cellularization in embryos containing an extra diploid set of either the second or third chromosomes. Such embryos possess DNA contents of 134% and 140% compared with wild type, and at cycle 13 have already attained N/C ratios (67% and 70%) near the threshold for a cell cycle pause in wild type. We observed cellularized embryos (an indication of cycle pause) at a nuclear density similar to that at cycle 13, and embryos with mixed territories of nuclei in cycles 13 and 14. In the latter case, cellularization was more advanced in the patch with the lower nuclear density, suggesting that regions that pause prematurely initiate cellularization early (Fig. 2C; see Fig. S1C in the supplementary material). The mitotic behavior of all of these embryos confirms that total DNA content, rather than the number of copies of specific genes, dictates the N/C ratio.
In some embryos with N/C ratios near the 70% threshold value, we observed patches of different nuclear densities. These patches were of different sizes and showed relatively sharp boundaries, with no co-mingling of nuclei in discrepant cell cycles. We note that if the N/C ratio controlled an embryo-wide or global-level mechanism for enacting alterations in the cell cycle, all nuclei in a given embryo would pause after the same number of divisions. Conversely, if cell cycle decisions were made by each nucleus independently, we would expect a salt-and-pepper-like pattern of mixing of nuclei in discrepant cell cycles. The large mitotic patches we observed suggest the presence of a community effect, in which neighboring nuclei influence one another's mitotic behavior. Therefore, we propose that the response to the N/C ratio occurs in a locally nonautonomous manner, such that an N/C value of greater than 70% of that seen in cycle 14 wild-type embryos is required to ensure a robust, synchronized response from every nucleus of the embryo.
Degradation of maternal RNA occurs independently of N/C ratio
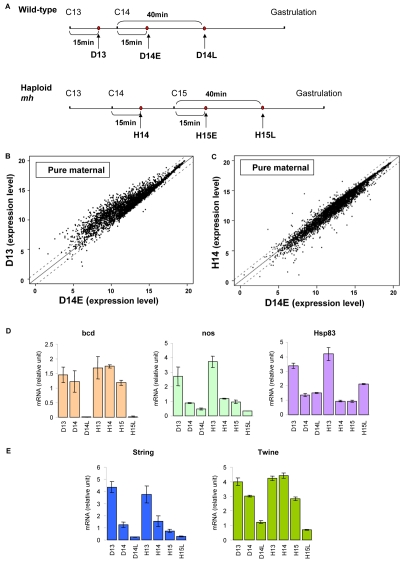
We next investigated whether the N/C threshold controls the switch from maternal to zygotic gene expression. We compared transcript levels in wild-type and haploid embryos using cDNA microarrays. For the diploid transcriptional profile, we prepared cDNA from hand selected wild-type embryos at cycle 13 interphase (15 minutes after the nuclear division of cycle 12), and at early and late cycle 14 interphase (15 minutes and 40 minutes after cycle 13 division). We compared this with cDNA from haploids that were matched to wild type with respect to N/C ratio, i.e. at cycle 14 (15 minutes after cycle 13 mitosis), and at early and late cycle 15 (15 minutes and 40 minutes after the nuclear division of cycle 14; Fig. 3A). In all experiments the cell cycle progression was monitored in each embryo by the Histone 2A-GFP pattern. Gene expression profiles were acquired using Affymetrix microarray, each with three biological replicates that displayed little variation (see Fig. S2 in the supplementary material). Previously, our laboratory identified ∼4000 purely maternal (no zygotic transcription) and ∼300 purely zygotic (no maternal RNA) transcripts (De Renzis et al., 2007) that form the categories used for data analysis.
Fig. 3.
Time-dependent maternal RNA degradation. (A) Collection scheme of wild-type and haploid embryos for microarray profiling, with collection time points indicated by red dots. (B) Scatter plot of purely maternal RNAs at diploid cycle 13 versus early diploid cycle 14. (C) Scatter plot of purely maternal RNAs (given in Log to the base 2 by Loess normalization) at haploid cycle 14 versus early diploid cycle 14. (D) Expression pattern of maternal RNAs in wild-type and maternal haploid embryos at different stages (Semotok and Lipshitz, 2007), confirmed by quantitative RT-PCR. mRNA levels are expressed as relative values after normalizing to β-tubulin56D. (E) Degradation trend of maternal string (stg) and twine (twe) using quantitative RT-PCR at different stages in wild-type and maternal haploid embryos. Error bars show s.d. D, diploid; H, maternal haploid embryos; D14L, diploid late cycle 14; H15L, haploid late cycle 15.
We first examined the purely maternal RNAs by comparing their expression at cycle 13 and early cycle 14 in the diploid embryos. Expression of about 30% of the maternal RNA species fell more than 2-fold during this 20-minute period (Fig. 3B), in patterns that were consistent with previous reports for individual RNAs used as markers for specific degradation pathways (Semotok and Lipshitz, 2007). For example, we saw a significant degradation of maternal nanos and Hsp83 between cycle 13 and 14, and a destabilization of bcd RNA late in cycle 14, which was independently confirmed by quantitative RT-PCR of samples from hand-collected diploid and haploid embryos staged visually using Histone 2A-GFP (Fig. 3D). When diploids and haploids at the same developmental stage (cycle 14) were compared, mRNA levels were similar (Fig. 3C), even though haploids had not yet achieved the N/C ratio needed to initiate the cell cycle pause. This suggests that maternal RNA degradation depends on absolute developmental timing, rather than the N/C ratio. To confirm this time dependence, we also used quantitative RT-PCR to measure levels of string (stg), a homolog of cdc25 previously shown to undergo significant degradation at the onset of MBT (Fig. 3E). We found that the temporal regulation of maternal stg was not altered by DNA content: stg levels at cycle 13 were similar in haploids and diploids, and fell about 3-fold in both genotypes at the onset of cycle 14. By late cycle 14, stg RNA was essentially undetectable. Interestingly, we noted that another cdc25 homolog, twine, did not follow the pattern of time-dependent maternal message degradation; its elimination correlated instead with the N/C ratio (Fig. 3E). Because the Cdc25 proteins are essential for the G2/M transition in the cell cycle, the time and stage dependence of maternal RNA degradation might be relevant for the mechanisms that induce the cell cycle pause occurring at cycle 14 (Edgar and Datar, 1996).
Two broad classes of zygotically expressed genes are activated at cycle 14
To determine the extent to which the activation of zygotic transcription correlates with the N/C ratio, we compared the expression of 290 purely zygotic genes (De Renzis et al., 2007) in haploid and diploid embryos. When grouped by hierarchical clustering based on temporal dynamics of expression, these genes fell into two large clusters that accounted for 75% of the 290 genes (Fig. 4A). In the first cluster of 88 genes, the gene expression level at cycle 13 in diploid embryos (D13) was similar to that at cycle 14 in haploid embryos (H14), and the gene expression level at early cycle 14 in diploids (D14E) was similar to that at cycle 15 in haploids (H15). The expression of these genes was delayed for one cell cycle in haploid embryos and thus correlated with the N/C ratio. We refer to these as N/C ratio-dependent genes. However, the majority of the zygotic genes (127 genes) fell in a second cluster in which expression levels at early cycle 14 in haploid embryos (H14) were very similar to those at cycle 14 in diploid embryos (D14E) and much higher than those of cycle 13 diploid embryos (D13). Transcription of these genes began at the same developmental stage irrespective of the N/C ratio, similar to the timing of maternal transcript degradation. Therefore, we designated this class the time-dependent genes. Although both N/C ratio-dependent and -independent groups show further heterogeneities that suggest additional levels of control, the unbiased hierarchical clustering supports the existence of two relatively distinct timing mechanisms associated with the MBT. The remaining 25% of the zygotically active genes showed more complicated expression patterns and many in fact represent a cohort of genes expressed prior to the MBT in cycle 11/12 embryos (Bosch et al., 2006; De Renzis et al., 2007; Liang et al., 2008) (data not shown).
Fig. 4.
Identification of N/C ratio- and time-dependent genes. (A) Hierarchical clustering of purely zygotic genes; only the two largest clusters are shown. Genes tested by in situ hybridization are labeled. (B) Multi-dimensional scaling plot for both N/C ratio- (green) and time- (red) dependent genes. Genes tested by in situ hybridization are circled.
To validate the different properties of the two major clusters, we used multiple-dimension scaling (MDS), which reduces the dimensionality of the data set but preserves the relative distance of individual data points. Time- and N/C ratio-dependent genes remained well separated by this method (Fig. 4B). To verify the temporal and spatial expression pattern of the two classes of purely zygotic genes, we performed in situ hybridization for selected N/C ratio- and time-dependent genes on wild-type diploid embryos and on haploids produced from mh or another maternal effect mutation, sesame (ssm; Hira - FlyBase) (Loppin and Couble, 2001; Loppin and Couble, 2005). All tested time-dependent genes were zygotically activated at cycle 14 in both haploids and diploids, regardless of the N/C ratio. By contrast, N/C ratio-dependent genes showed very similar expression patterns in wild-type diploids and mh haploids, but with a one-cycle delay in the haploid embryos (Fig. 5; see Figs S3, S4 in the supplementary material). Remarkably, N/C ratio-dependent genes preserved their normal spatial and temporal expression patterns in haploids. Genes such as odd skipped (odd) and spalt major (salm), which are activated in stripes of discrete width over a well-defined temporal progression in wild type, maintained their correct spatial domains and were expressed in their proper temporal order in haploids, despite the occurrence of an extra division and the presence of twice the normal number of nuclei (Fig. 5A,C). Based on these results, we conclude that two different regulatory mechanisms control the expression of purely zygotic genes around the MBT. Moreover, haploid embryos appear to maintain a large degree of patterning information in spite of the temporal decoupling of two regulatory mechanisms.
Fig. 5.
Confirmation of N/C ratio-dependent expression. (A) In situ hybridization for the N/C ratio-dependent gene spalt major (salm) and the time-dependent gene insulin-like peptide 4 (ilp4) on both wild-type and mh haploid embryos at different cycles. Corresponding RNA expression levels from the microarray experiment are shown in the panel above the figures. (B,C) In situ hybridization for the N/C-ratio dependent genes (B) fruhstart (frs) and (C) odd skipped (odd) on both wild-type and ssm haploid embryos. Nuclear density is indicated by Hoechst staining below. Scale bar: 50 μm.
Titration of the N/C ratio specifically affects zygotic transcription of N/C ratio-dependent genes
The delayed expression of N/C ratio-dependent genes in haploid embryos suggests that control of their transcription relies on total DNA content, similar to the onset of the mitotic pause. Alternatively, the apparent delay of zygotic transcription could be simply due to reduced zygotic transcription resulting from the reduction in copy number of genes in haploid embryos. To discriminate between these possibilities, we examined the expression of N/C ratio-dependent genes in embryos from C(2)EN or C(3)EN compound stocks that possess altered DNA content from the modulation of either chromosome II or III copy number, but have the normal diploid number of others. We found that the activation of odd, located on chromosome II, was still entirely dependent on the N/C ratio when the ratio was manipulated by changing the numbers of chromosome III: expression was delayed in embryos with a reduced amount of DNA (∼60% of total DNA equivalent), and premature in embryos with extra chromosome copies (∼140% of total DNA equivalent; Fig. 6A). Similar results were obtained with other N/C ratio-dependent genes, such as fruhstart, short of gastrulation, giant and spalt major (see Fig. S5A-D in the supplementary material). We conclude that expression of the N/C ratio-dependent gene class relies on overall DNA content and not the presence of specific genomic regions, just as we observed for the control of nuclear divisions.
Fig. 6.
Coupling of transcription of N/C ratio-dependent genes to mitotic cycles. (A) In situ hybridization for the N/C ratio-dependent gene odd on embryos from C(3)EN stocks. Wild-type expression of seven stripes was observed in embryos with wild-type DNA. The fusion of the stripes in C(3)EN embryos is due to the absence of other patterning genes, such as knirps, on Chromosome III. huckebein (hkb) expression at the two poles of the embryo was used to mark the presence of Chromosome III. (B) In situ hybridization for the N/C ratio-dependent gene odd-paired (opa) and the time-dependent gene snail (sna) on patchy embryos. Arrows indicate the boundary of mitotic patches; asterisk indicates the region with higher nuclear density. (C) RNA FISH for the N/C ratio-dependent gene opa on a C(2)EN embryo with patches in early and late interphase. DNA is stained with Hoechst and pseudo-colored in red. Scale bars: 50 μm.
The above results indicate a correlation between cell cycle behavior and the onset of expression of the N/C ratio-dependent genes. However, these findings do not address whether the control of these two events derives from a shared mechanism that interprets the N/C ratio, or conversely if separate processes measure the N/C ratio and independently control transcription and cell cycle behavior. To address this, we took advantage of the patchy behavior found in embryos with DNA content at the threshold for the N/C ratio. We reasoned that if transcriptional and cell cycle responses represented a shared assessment of N/C ratio, the expression domains of N/C ratio-dependent genes would always correlate with nuclear density. However, if the cell cycle and transcriptional decisions were mechanistically unlinked, the patchy mitotic behavior would not always align with any patchy or variable transcriptional responses observed with probes for the N/C ratio-dependent genes.
The examination of expression patterns in patchy embryos strongly supported a tight coupling of the two decisions. For N/C ratio-dependent class genes, such as opa, odd and frs, the expression pattern correlated precisely with the boundary of the nuclear density, i.e. transcription was delayed in the area that failed to pause in the cell cycle (Fig. 6B; see Fig. S6 in the supplementary material). Consistent with this model, nascent transcripts visualized by FISH showed delayed expression in the compartment with a higher nuclear density, but were detected in low-density regions in which the N/C ratio was perceived as being sufficient to induce a cell cycle pause (Fig. 6C). By contrast, and as expected from the above results, nuclear density had no effect on the expression of genes in the time-dependent class (ilp4 and sna): the expression patterns were not disrupted by the discontinuity of the nuclear density (Fig. 6B; see also Fig. S6 in the supplementary material), arguing against the possibility that the extra mitotic division in the higher nuclear density region nonspecifically interferes with transcription. On the basis of these findings, we propose a model of MBT with two separate layers of temporal control: one branch relies on the time interval following fertilization to activate both maternal RNA degradation and the majority of gene transcription, while a parallel, independent mechanism measures the N/C ratio and subsequently regulates both mitotic cycle and N/C ratio-controlled gene transcription. The degree to which these branches and the associated behaviors reflect truly independent mechanisms requires the testing of additional probes to exclude more trivial explanations, such as differences in transcript stability or gene size.
DISCUSSION
Multiple important transitions occur at the MBT: pause of cell cycle, accelerated degradation of maternal RNA, significant zygotic transcription, and cellularization. The simultaneous occurrence of these transitions poses an interesting question: are they independently timed with different mechanisms, or are they coordinated by some shared mechanism? Our data suggest the existence of a threshold mechanism that senses the ratio of total DNA content to cytoplasm and converts that ratio into an on-off cell cycle decision. The same N/C ratio appears to govern a significant fraction of the new transcription that occurs at cycle 14. The major change in transcription and almost all of the maternal RNA degradation that occur at the Drosophila MBT, however, appear to be regulated by absolute stage or developmental time, and are initiated independently of a particular N/C ratio.
Cell cycle control during MBT
By manipulating chromosomal genotypes in the embryo, we found that the DNA content threshold above which the embryo will stop mitosis is about 70% of the amount normally present at cycle 14 (Table 1). Setting the threshold approximately midway between the DNA present at cycle 13 and cycle 14 would achieve the maximal tolerance to fluctuations of cytoplasmic volume and thus ensure robust mitosis transition. The patchy distribution of nuclear densities and apparent cell cycle choices in embryos with near threshold values is intriguing (Fig. 2C). Because all nuclei in such embryos have similar DNA content and experience the same threshold N/C ratio, variations in its assessment between nuclei might have produced a salt-and-pepper-like distribution of cell cycle behaviors, if individual nuclei made autonomous choices. Instead, we see patches of coherent behavior large enough to require the coordination of neighboring nuclei. This apparent non-autonomy might be similar to that governing the rapid meta-synchronous mitotic divisions during the early cleavage stages. During those stages, divisions are timed by maternally deposited cyclins (Crest et al., 2007; Edgar et al., 1994; Ji et al., 2004; Stiffler et al., 1999; Su et al., 1998) and by the DNA replication checkpoint proteins that control entry into mitosis (Brodsky et al., 2000; Ji et al., 2004; Sibon et al., 1997; Takada et al., 2007). Local diffusion of such proteins might maintain synchrony during early mitosis and also account for the patchy pattern when nuclei reach a threshold N/C ratio. However, there are alternative hypotheses that might explain the patchiness of the response. For example, the cytoplasmic component of the N/C ratio might not be perfectly uniform in eggs. Non-uniformities might not matter in normal development but would have an impact when DNA contents are near threshold. It is also possible that the N/C ratio is read early and that decisions are inherited in coherent lineage-related patches. Further experiments will be required to distinguish between these possibilities.
Transcriptional activation at the MBT
The first 3 hours of embryonic development of Drosophila is characterized by a complex changing pattern of transcription (Pilot et al., 2006). Our transcriptional profiling uncovers two modes of zygotic activation at cycle 14: transcription dependent on the N/C ratio and transcription dependent on the absolute time/stage. Even though our hierarchical clustering automatically grouped genes into two large clusters with N/C ratio- and time-dependent properties, the expression patterns of individual genes within each cluster show considerable variation. This variation could reflect a regulation of subgroups in each cluster by additional specific transcription factor(s) on top of their N/C ratio- or time-dependent regulation. Alternatively, genes could be affected by both N/C ratio- and time-dependent regulation, but to differing extents. Consistent with either view, we found that the functions of both N/C ratio- and time-dependent genes are quite heterogeneous and overlapping. There are traditional gap genes in both N/C ratio (knirps, giant) and time (Kruppel) dependent classes, as well as genes affecting DV patterning and cytoskeleton.
A fraction of the time-dependent class is expressed earlier and significantly overlaps with a well-described group of genes first detected in cycle 11/12 (De Renzis et al., 2007). Although this overlap might imply a more gradual transcriptional activation during this period (Pritchard and Schubiger, 1996), our data suggests that the increase in expression may be more abrupt, as a more than 6-fold increase in transcript level from cycle 13 to early cycle 14 was observed. The molecular mechanisms responsible for this increased transcriptional activity might not yet be fully established at the beginning of cycle 14. When time-dependent genes are compared in haploid and diploid embryos, the associated RNA levels in diploids at early cycle 14 are only 1.3 times that of haploids. This value is significantly lower than the 2-fold difference in copy number (Ashburner and Bonner, 1979; Devlin et al., 1988; Driever and Nusslein-Volhard, 1988), and suggests that productivity per template is higher in haploids. This apparent hyperactivity in the haploid embryos has been lost by late cycle 14, when the diploid/haploid ratio (1.8) approaches the ratio of gene copy number. These observations suggest that the factors responsible for the burst in transcription at the beginning of cycle 14 are initially limiting, but that they continue to rise and reach a stable level towards the end of the cycle. The higher per template expression levels in haploid embryos might also explain why maternal RNA degradation that depends on zygotic transcription is still normal in haploids (see below)
The purely zygotic genes analyzed in our study account for only a small portion of the genes that are zygotically active at the MBT. Most zygotically active genes in Drosophila are also represented among maternal transcripts in the unfertilized egg (De Renzis et al., 2007). The persistence of these maternal transcripts obscures their zygotic expression in haploid and diploids, and we were not able to determine unambiguously whether the new zygotic RNAs fall into the time- or N/C ratio-dependent groups. However, we note that most of these maternal-zygotic genes show early cycle 14 diploid/haploid expression ratios that are close to the 1.3 value observed for time-dependent purely zygotic genes, which suggests that these genes are not only expressed at cycle 14 in haploids, but that their transcriptional activity per template is higher than in diploids. They share these features with purely zygotic genes that are time dependent.
Maternal RNA degradation at the MBT
In Drosophila, the cis-acting elements that determine the stability of maternal RNAs are located in the 5′cap and the 3′UTR, as well as in the ORF itself (Semotok and Lipshitz, 2007). Many of the trans-acting factors that bind these elements are supplied as maternal RNAs and proteins that are activated at fertilization and result in a time-dependent `maternal degradation pathway' (Bashirullah et al., 1999). A second set of maternal RNAs is dependent on zygotic gene activity for their destabilization at the MBT (De Renzis et al., 2007; Edgar and Datar, 1996). The identity and nature of the required transcripts have not yet been determined, although recent experiments point to an important role for microRNAs in the process (Bushati et al., 2008; Giraldez et al., 2006). Our results indicate that most of the maternal RNA degradation occurring in Drosophila is a developmental time-regulated process that can be uncoupled from the N/C ratio. This time dependence is not inconsistent with the previously observed requirement for zygotic transcription, given the predominance of time-dependent transcription among the zygotically expressed genes. Instead, the observed time-dependent maternal RNA degradation could reflect the synergistic effects of both maternal and zygotic degradation mechanisms. Because our data came from a short developmental time window, cycle 13 to cycle 14, we could not distinguish which of the two pathways plays a major role during the MBT.
The maternal RNAs that encode the cdc25 homologs stg and twine merit special discussion. Stg is the major regulator of cell cycle progression at later stages and its degradation at cycle 14 correlates with the mitotic pause occurring at that time (Edgar and Datar, 1996). We find that in haploid embryos maternal stg RNA undergoes degradation in a manner very similar to that in diploids, even though the embryo subsequently undergoes an additional round of mitosis before the pausing at cycle 15. The time-dependent degradation of stg suggests that stg RNA might not control N/C ratio-dependent cell cycle behavior at the MBT. Stg protein might persist after its RNA has gone, and other factors might govern cell cycle pause. One obvious candidate would be the second cdc25 homolog twine, which has an N/C ratio-dependent degradation pattern. Zygotically active candidates that might contribute to the control include Tribbles and Fruhstart, which depend on the N/C ratio for their expression and are required for an efficient pause at cycle 14 (Grosshans et al., 2003; Grosshans and Wieschaus, 2000). How these regulators together with Stg coordinate cell cycle pause, and how stg and twine mRNAs are degraded by zygotic transcription require further investigation.
Failed coordination of MBT transcription might explain haploid lethality in flies
In all diploid multicellular animals in which haploid individuals can be generated, they are inevitably lethal. For inbred laboratory strains like Drosophila, this lethality cannot be attributed to previously existing recessive lethal mutations that have accumulated in the population and are only uncovered in the haploid state. The observation that haploid embryos undergo an additional division cycle suggested an alternative cause for haploid lethality, namely that, from the blastoderm stage onwards, haploid embryos would have twice the number of cells. Although spatial patterning is generally normal in such embryos, each primordium would be established with double the number of cells. This increased size and cell number might cause problems at later stages in development. Although this explanation might have some validity in other organisms, it seems less likely in Drosophila given the classical experiments in which Bicoid dosage was manipulated to produce embryos in which primordia were either increased or decreased in size and yet still developed into viable adults (Namba et al., 1997). The existence of two major modes of transcriptional regulation at the Drosophila MBT provides an alternative explanation for haploid lethality. In fly embryos, time-dependent and N/C ratio-dependent gene expression are normally coordinated to produce the transcriptional profile required during cellularization and gastrulation. In the haploid embryos with an altered N/C ratio, this coordination is lost. The resultant temporal shift in expression patterns might throw certain developmental steps out of sequence and result in specific defects at later stages in development. It will be interesting to see whether the transcriptional modes we observed in Drosophila are common to other organisms, and whether a temporal expression shift of the N/C-dependent group can explain the lethality universally observed in haploid embryos. An important next step will be the identification of the respective molecular mechanisms that control the two modes of transcription in Drosophila. This will allow us to determine whether the underlying mechanisms are conserved in MBT among species.
Supplementary material
Supplementary material for this article is available at http://dev.biologists.org/cgi/content/full/136/12/2101/DC1
Supplementary Material
We thank all members of the Wieschaus lab, Schupbach lab and Tavazoie lab for helpful discussions. We thank Shawn Little for manuscript revision and Adam Martin, Stefano De Renzis, Anna Sokac, Yu-Chiun Wang and Girish Deshpande for critical reading. We thank Stefano De Renzis for microarray experiments. We thank Donna Storton (microarray) and Joe Goodhouse (microscope facility) for technical support. We thank Kami Ahmad at Harvard and Bloomington Drosophila Stock Center for providing fly stocks. This work was supported by the Howard Hughes Medical Institute and by National Institute of Child Health and Human Development Grant 5R37HD15587 to E.F.W., by National Human Genome Research Institute Grant 5R01 hG3219-3 to S.T., and by NIH Grant P50 GM071508 to Princeton University. Deposited in PMC for release after 6 months.
References
- Ashburner, M. and Bonner, J. J. (1979). The induction of gene activity in Drosophila by heat shock. Cell 17, 241-254. [DOI] [PubMed] [Google Scholar]
- Bashirullah, A., Halsell, S. R., Cooperstock, R. L., Kloc, M., Karaiskakis, A., Fisher, W. W., Fu, W., Hamilton, J. K., Etkin, L. D. and Lipshitz, H. D. (1999). Joint action of two RNA degradation pathways controls the timing of maternal transcript elimination at the midblastula transition in Drosophila melanogaster. EMBO J. 18, 2610-2620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bosch, J. R. t., Benavides, J. A. and Cline, T. W. (2006). The TAGteam DNA motif controls the timing of Drosophila pre-blastoderm transcription. Development 133, 1967-1977. [DOI] [PubMed] [Google Scholar]
- Brodsky, M. H., Sekelsky, J. J., Tsang, G., Hawley, R. S. and Rubin, G. M. (2000). mus304 encodes a novel DNA damage checkpoint protein required during Drosophila development. Genes Dev. 14, 666-678. [PMC free article] [PubMed] [Google Scholar]
- Bushati, N., Stark, A., Brennecke, J. and Cohen, S. M. (2008). Temporal reciprocity of miRNAs and their targets during the maternal-to-zygotic transition in Drosophila. Curr. Biol. 18, 501-506. [DOI] [PubMed] [Google Scholar]
- Cox, T. F. and Cox, M. A. A. (2001). Multidimensional Scaling. London: Chapman & Hall.
- Crest, J., Oxnard, N., Ji, J. Y. and Schubiger, G. (2007). Onset of the DNA replication checkpoint in the early Drosophila embryo. Genetics 175, 567-584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Renzis, S., Elemento, O., Tavazoie, S. and Wieschaus, E. (2007). Unmasking activation of the zygotic genome using chromosomal deletions in the Drosophila embryo. PLoS Biol. 5, 1036-1051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Devlin, R. H., Holm, D. G. and Grigliatti, T. A. (1988). The Influence of whole-arm trisomy on gene expression in Drosophila. Genetics 118, 87-101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Driever, W. and Nusslein-Volhard, C. (1988). The bicoid protein determines position in the Drosophila embryo in a concentration-dependent manner. Cell 54, 95-104. [DOI] [PubMed] [Google Scholar]
- Edgar, B. and Datar, S. (1996). Zygotic degradation of two maternal Cdc25 mRNAs terminates Drosophila's early cell cycle program. Genes Dev. 10, 1966-1977. [DOI] [PubMed] [Google Scholar]
- Edgar, B. A., Kiehle, C. P. and Schubiger, G. (1986). Cell cycle control by the nucleo-cytoplasmic ratio in early Drosophila development. Cell 44, 365-372. [DOI] [PubMed] [Google Scholar]
- Edgar, B. A., Sprenger, F., Duronio, R. J., Leopold, P. and O'Farrell, P. H. (1994). Distinct molecular mechanism regulate cell cycle timing at successive stages of Drosophila embryogenesis. Genes Dev. 8, 440-452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giraldez, A. J., Mishima, Y., Rihel, J., Grocock, R. J., Van Dongen, S., Inoue, K., Enright, A. J. and Schier, A. F. (2006). Zebrafish MiR-430 promotes deadenylation and clearance of maternal mRNAs. Science 312, 75-79. [DOI] [PubMed] [Google Scholar]
- Grosshans, J. and Wieschaus, E. (2000). A genetic link between morphogenesis and cell division during formation of the ventral furrow in Drosophila. Cell 101, 523-531. [DOI] [PubMed] [Google Scholar]
- Grosshans, J., Muller, H. A. J. and Wieschaus, E. (2003). Control of cleavage cycles in Drosophila embryos by fruhstart. Dev. Cell 5, 285-294. [DOI] [PubMed] [Google Scholar]
- Ji, J. Y., Squirrell, J. M. and Schubiger, G. (2004). Both Cyclin B levels and DNA-replication checkpoint control the early embryonic mitoses in Drosophila. Development 131, 401-411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liang, H. L., Nien, C. Y., Liu, H. Y., Metzstein, M. M., Kirov, N. and Rushlow, C. (2008). The zinc-finger protein Zelda is a key activator of the early zygotic genome in Drosophila. Nature 456, 400-403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loppin, B. and Couble, P. (2001). Paternal chromosome incorporation into the zygote nucleus is controlled by maternal haploid in Drosophila. Dev. Biol. 231, 383-396. [DOI] [PubMed] [Google Scholar]
- Loppin, B. and Couble, P. (2005). The histone H3.3 chaperone HIRA is essential for chromatin assembly in the male pronucleus. Nature 437, 1386-1390. [DOI] [PubMed] [Google Scholar]
- Mathavan, S., Lee, S. G. P., Mak, A., Miller, L. D., Murthy, K. R. K., Govindarajan, K. R., Tong, Y., Wu, Y. L., Lam, S. H., Yang, H. et al. (2005). Transcriptome analysis of zebrafish embryogenesis using microarrays. PLoS Genet. 1, e29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McKnight, S. and Miller, J. O. (1976). Ultrastructural patterns of RNA synthesis during early embryogenesis of Drosophila melanogaster. Cell 8, 305-319. [DOI] [PubMed] [Google Scholar]
- Merrill, P., Sweeton, D. and Wieschaus, E. (1988). Requirements for autosomal gene activity during precellular stages of Drosophila melanogaster. Development 104, 495-509. [DOI] [PubMed] [Google Scholar]
- Namba, R., Pazdera, T., Cerrone, R. and Minden, J. (1997). Drosophila embryonic pattern repair: how embryos respond to bicoid dosage alteration. Development 124, 1393-1403. [DOI] [PubMed] [Google Scholar]
- Newport, J. and Kirschner, M. (1982a). A major developmental transition in early Xenopus embryos: I. characterization and timing of cellular changes at the midblastula stage. Cell 30, 675-686. [DOI] [PubMed] [Google Scholar]
- Newport, J. and Kirschner, M. (1982b). A major developmental transition in early Xenopus embryos: II. control of the onset of transcription. Cell 30, 687-696. [DOI] [PubMed] [Google Scholar]
- O'Farrell, P. H., Stumpff, J. and Tin Su, T. (2004). Embryonic cleavage cycles: how is a mouse like a fly? Curr. Biol. 14, R35-R45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pilot, F., Philippe, J. M., Lemmers, C., Chauvin, J. P. and Lecuit, T. (2006). Developmental control of nuclear morphogenesis and anchoring by charleston, identified in a functional genomic screen of Drosophila cellularisation. Development 133, 711-723. [DOI] [PubMed] [Google Scholar]
- Pritchard, D. and Schubiger, G. (1996). Activation of transcription in Drosophila embryos is a gradual process mediated by the nucleocytoplasmic ratio. Genes Dev. 10, 1131-1142. [DOI] [PubMed] [Google Scholar]
- Rozen, S. and Skaletsky, H. J. (2000). Primer3 on the WWW for general users and for biologist programmers. Totowa, NJ: Humana Press. [DOI] [PubMed]
- Semotok, J. L. and Lipshitz, H. D. (2007). Regulation and function of maternal mRNA destabilization during early Drosophila development. Differentiation 75, 482-506. [DOI] [PubMed] [Google Scholar]
- Sibon, O. C. M., Stevenson, V. A. and Theurkauf, W. E. (1997). DNA-replication checkpoint control at the Drosophila midblastula transition. Nature 388, 93-97. [DOI] [PubMed] [Google Scholar]
- Stiffler, L., Ji, J., Trautmann, S., Trusty, C. and Schubiger, G. (1999). Cyclin A and B functions in the early Drosophila embryo. Development 126, 5505-5513. [DOI] [PubMed] [Google Scholar]
- Su, T. T., Sprenger, F., DiGregorio, P. J., Campbell, S. D. and O'Farrell, P. H. (1998). Exit from mitosis in Drosophila syncytial embryos requires proteolysis and cyclin degradation, and is associated with localized dephosphorylation. Genes Dev. 12, 1495-1503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takada, S., Kwak, S., Koppetsch, B. S. and Theurkauf, W. E. (2007). grp (chk1) replication-checkpoint mutations and DNA damage trigger a Chk2-dependent block at the Drosophila midblastula transition. Development 134, 1737-1744. [DOI] [PubMed] [Google Scholar]
- Yasuda, G. and Schubiger, G. (1991). Temporal regulation of gene expression in the blastoderm Drosophila embryo. Genes Dev. 5, 1800-1812. [DOI] [PubMed] [Google Scholar]
- Zalokar, M. (1976). Autoradiographic study of protein and RNA formation during early development of Drosophila eggs. Dev. Biol. 49, 425-437. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.