Abstract
Alzheimer’s disease is characterized by numerous pathological abnormalities, including amyloid β (Aβ) deposition in the brain parenchyma and vasculature. In addition, intracellular Aβ accumulation may affect neuronal viability and function. In this study, we evaluated the effects of different forms of Aβ on cognitive decline by analyzing the behavioral induction of the learning-related gene Arc/Arg3.1 in three different transgenic mouse models of cerebral amyloidosis (APPPS1, APPDutch, and APP23). Following a controlled spatial exploration paradigm, reductions in both the number of Arc-activated neurons and the levels of Arc mRNA were seen in the neocortices of depositing mice from all transgenic lines (deficits ranging from 14 to 26%), indicating an impairment in neuronal encoding and network activation. Young APPDutch and APP23 mice exhibited intracellular, granular Aβ staining that was most prominent in the large pyramidal cells of cortical layer V; these animals also had reductions in levels of Arc. In the dentate gyrus, striking reductions (up to 58% in aged APPPS1 mice) in the number of Arc-activated cells were found. Single-cell analyses revealed both the proximity to fibrillar amyloid in aged mice, and the transient presence of intracellular granular Aβ in young mice, as independent factors that contribute to reduced Arc levels. These results provide evidence that two independent Aβ pathologies converge in their impact on cognitive function in Alzheimer’s disease.
Alzheimer’s disease (AD) is the most common form of dementia in the elderly, with abnormal accumulation of the amyloid β (Aβ) peptide being one of the hallmarks of the disease. The correlation between parenchymal amyloid deposition (plaques) and cognitive decline is, however, controversial. Although the number of plaques does not closely correspond to the degree of dementia,1,2 some studies point toward a detrimental effect on neuronal connectivity and function.3,4,5,6 The majority of AD cases show further Aβ deposition in the cerebral vasculature,7,8,9 referred to as cerebral amyloid angiopathy (CAA). Although CAA is a clear risk factor for hemorrhagic stroke,10,11,12 indirectly leading to brain damage and cognitive impairments,9,13,14,15 its direct impact on cognition is more difficult to characterize. A detrimental role of intraneuronal AB accumulation has been indicated prior to the onset of extracellular deposition (see reviews16,17). Intraneuronal Aβ has been reported in brains of subjects with mild cognitive impairment, AD,18,19 and in patients with Down syndrome,20,21 and has also been described for several mouse models of cerebral Aβ amyloidoses.22,23,24,25 Negative effects have been illustrated by several in vitro studies,26,27 and coincidence of intraneuronal Aβ appearance with cognitive deficits has been shown in a triple transgenic mouse model of AD.28
Here we took advantage of three mouse models replicating distinct aspects of AD pathology to investigate the impact extracellular amyloid (vascular and parenchymal) and factors such as intracellular Aβ accumulation, have on cognitive impairment. APPPS1 mice show predominantly parenchymal amyloid with a high Aβ42 to Aβ40 ratio due to the L166P PS1 mutation in combination with the K670N/M671L “Swedish” double mutation. Both transgenes (as in all models studied) are driven by the neuron-specific Thy-1 minigene promoter.29 In comparison, APPDutch mice exhibit almost exclusively vascular amyloid, due to the overexpression of human amyloid precursor protein (APP) bearing the E693Q “Dutch” mutation. This mutation was identified as the cause of hereditary cerebral hemorrhage with amyloidosis-Dutch type, a rare autosomal dominant disorder, characterized by severe CAA, recurrent strokes, and dementia.30 APPDutch mice develop extensive vascular Aβ deposition initially within the walls of leptomeningeal vessels followed by cortical vessels.31 The third analyzed model, APP23 mice, overexpresses human APP with the “Swedish” mutation and exhibits both amyloid plaques and deposition within the vasculature.32
Within these mouse models, we investigated the behavioral induction of activity-regulated cytoskeleton-associated protein (Arc)/Arg3.1, as a marker for the functional status of individual neurons and memory systems. Arc is an effector immediate early gene implicated in synaptic plasticity, and is known to be necessary for memory consolidation and learning.33,34,35,36 The specificity and characteristic time course of Arc mRNA induction can be used to monitor neural circuit activation following behavior episodes, as initially demonstrated by Guzowski and colleagues37 and expanded in subsequent studies.38,39,40,41 Recent evidence has demonstrated a role of Arc in alpha-amino-3-hydroxyl-5-methyl-4-isoxazole-propionate (AMPA) receptor trafficking, and thus it’s involvement in the molecular processes of synaptic plasticity.42,43,44
Although a reduction of Arc and other plasticity-related genes has been previously demonstrated in aging rats45,46 and AD mouse models,47,48,49 we show for the first time that particular pathological events in AD transiently impact neuronal function.
Materials and Methods
Transgenic Mice
Young and aged male hemizygous transgenic (tg) mice from the strains APPPS1,29 APPDutch,31 and APP23,32 were used and age-matched non-transgenic (ntg) mice served as controls. APPPS1 mice co-express human APP carrying the K670N/M671L “Swedish” double mutation and hPS1 L166P with a threefold overexpression of human APP over endogenous mouse APP. APPDutch mice overexpress human APP bearing the E693Q “Dutch” mutation (by about fivefold). APP23 mice carry a human APP harboring the “Swedish” double mutation (sevenfold overexpression). All mouse lines express the transgene under the control of a neuron-specific mThy-1 promoter element;50 were either generated on (APPS1 and APPDutch) or backcrossed to (APP23) the same source strain (C57BL6/J); and aside from the mutations listed above, use the same APP751 construct. Young APPPS1 mice and ntg controls were 2 to 3 months old; aged APPPS1 and controls were analyzed at the age of 8 to 9 months (each group: n = 5). Young APPDutch mice (n = 6) and controls (n = 4) were 3 to 4 months old, and aged APPDutch animals (n = 8) and controls (n = 7) were between 25 and 30 months old. Young APP23 mice (n = 3) and controls (n = 5) were 3 to 4 months old, and aged APP23 mice (n = 6) and controls (n = 7) were 16 to 25 months old. Four ntg animals (4 to 7 months old) were included as a homecage control group (see below). Mice were maintained on a 12 hour light/dark cycle with food and water ad libitum and were moved from group to single housing 4 days before the behavioral experiment. All procedures with animals were performed in compliance with protocols approved by the local animal use committee and university regulations.
Behavior Exposure to Novel Environments
Mice of all groups were exposed to two novel environments (termed environments A and B), following a protocol known to induce Arc expression in hippocampal and neocortical neurons.37 For each of the 3 days before the experiment, the animals were handled for 5 to 10 minutes to familiarize them with the experimenter and to handling in general. Environment A was a 42 to 46 cm square cardboard box with 15 cm-high walls. Environment B was a 52 to 60 cm square laminated table-top without walls. Each environment contained unique local and distal cues in separate parts of the same room, and was cleaned thoroughly between exposures. Mice were exposed for 5 minutes to A, and then transferred to their home cage for 30 minutes, followed by 5 minutes in B. The experimenter, who was blinded to genotype, watched the exploration trial quietly taking records of the exploratory behavior. Environments were theoretically divided in four quadrants and the number of visits in each quadrant was noted. To ensure an adequate and equivalent exploratory behavior, mice had by definition to explore each quadrant at least three times and were gently placed in another quadrant by the experimenter if they did not. A move by experimenter was also performed if mice stayed at the same spatial location without any exploratory behavior (eg, head movement/rearing) for longer than 30 seconds. Analysis of the exploratory behavior, recorded as ‘sum of total visits’ revealed no significant difference between transgenic mice and age-matched controls (P values from 0.12 to 0.55). Animals were sacrificed as described below immediately following the second environmental exposure. Animals of the homecage group were directly transferred from their homecages and sacrificed.
Tissue Preparation
Animals were deeply anesthetized with isoflurane (induction average <30 seconds) and perfused transcardially for 1 minute with cold PBS, followed by 12 minutes with cold 4% paraformaldehyde in PBS. Brains were prepared and post-fixed in 4% paraformaldehyde in PBS at 4°C overnight, followed by cryoprotection overnight in 10% and 20% glycerol in 0.1 M phosphate buffer at 4°C. Brains were then snap-frozen by immersion in a 2-methyl-butane bath (<−20°C) surrounded by dry ice for 20 minutes. Tissue was stored at −80°C until sectioning on a freezing/sliding microtome. Forty μm-thick coronal sections were taken through the extent of the entire forebrain. Sections were stored in cryoprotectant solution (30% glycerol and 30% ethylene glycol in 0.1 M phosphate buffer) at −80°C until staining, when they were allowed to thaw and washed thoroughly in PBS. For each experiment, sections from transgenic and control animals were mounted in random positions on the same slides.
Fluorescent in Situ Hybridization
Riboprobes were generated from nearly full-length mouse Arc cDNA inserted into pBluescript II SK plasmid (kindly provided by Drs. J. Guzowski and P. Worley). Sense and antisense digoxigenin (DIG)-labeled probes were transcribed from the linearized plasmid using DIG RNA Labeling Mix (Roche Diagnostics, Mannheim, Germany), and T3 or T7 RNA polymerases respectively (Stratagene, La Jolla, CA), and purified on mini Quick Spin RNA Columns (Roche Diagnostics). To improve tissue penetration, probes were alkaline hydrolyzed for 30 minutes at 60°C. Probe concentration was estimated by spotting serial dilutions of probe and DIG-labeled Control RNA (Roche Diagnostics) on membranes, immunodetection with anti-DIG Fab fragments conjugated to alkaline phosphatase (Roche Diagnostics), and colorization with nitro blue tetrazolium/5-bromo-4-chloro-3-indolyl phosphate (Roche Diagnostics). All solutions used for in situ hybridization were prepared RNase-free. Sections were mounted onto slides (SuperFrost Plus; R. Langenbrinck Labor- und Medizintechnik, Teningen, Germany) in a systematic random order/location, assuring a balanced mixture of tg and ntg groups and therefore minimizing potential experimental/staining effects. Mounted slides were dried for 1 hour at 30°C, and post-fixed for 20 minutes in 4% paraformaldehyde in PBS at 4°C, followed by two washes in PBS. Sections were permeabilized with 0.3% Triton-X100 in PBS and 30 μg/ml proteinase K (Roche Diagnostics) in Tris EDTA (TE) [10 mM Tris-HCl and 1 mM EDTA, pH 7.5] at 30°C with interleaved PBS washes. Proteinase K treatment was stopped by 0.2% glycine in PBS with 50 mM EDTA followed by two PBS washes. Tissue was then equilibrated in 0.1 M triethanolamine and acetylated with 0.25% acetic anhydride in 0.1 M triethanolamine. After three washes in 2× SSC [0.3 M NaCl and 0.03 M sodium citrate, pH adjusted to 7.0 with citric acid], sections were dehydrated through graded alcohols and defatted in chloroform. Tissue was rehydrated through graded alcohols and dried. For hybridization, the probe RNA was diluted to 180 ng/ml in hybridization buffer [50% deionized formamide, 2× SSC, 1× Denhardt’s solution (1% bovine serum albumin, 1% polyvinylpyrolidone, and 1% Ficoll), 10% dextran sulfate, 0.5 mg/ml wheat germ tRNA, and 0.5 mg/ml salmon sperm DNA] and denatured at 80°C for 5 minutes before application to tissue. Sections were then sealed with a coverslip and incubated 16 to 20 hours at 59°C in a humid chamber. Coverslips were removed and slides were washed twice in 2× SSC/1 mM EDTA. Afterward sections were treated with 10 μg/ml RNase A (Sigma-Aldrich, Steinheim, Germany) in TE/0.5 M NaCl and washed twice with 2× SSC/1 mM EDTA. Stringency washes were performed in 0.1× SSC/1 mM EDTA at 59°C and 0.5× SSC at room temperature. Endogenous peroxidase activity was quenched with 1% H2O2 in PBS, followed by subsequent washes in PBS, washing buffer [100 mM maleic acid, 150 mM sodium chloride, 0.3% Tween-20, pH adjusted to 7.5] and tris-buffered saline (TBS) (84 mM Tris-HCl, 16 mM Trisbase, and 154 mM NaCl). Sections were blocked with 10% bovine serum albumin in TBS and incubated with anti-DIG Fab fragments conjugated to horseradish peroxidase (Roche Diagnostics) diluted 1:400 in 10% bovine serum albumin in TBS for 2 hours at 4°C for detection. After three washes with washing buffer and a second block with the provided tyramide blocking buffer (Perkin Elmer, Rottgau-Jügesheim, Germany) for 30 minutes at room temperature, the conjugate was detected with Cyanine-3 Tyramide Working Solution (TSAPlus Fluorescence Systems, Perkin Elmer). Developed sections were washed with TE and PBS, counterstained with 4′6-diamidino-2-phenylindole (DAPI; Sigma-Aldrich) and allowed to dry before coverslipping with VectaShield (Vector Laboratories, Burlingame, CA).
An adjusted fluorescence in situ hybridization (FISH) protocol was used for the intracellular approach designed for better preservation of endogenous proteins. Summarized pepsin was used instead of proteinase K (end concentration of 100 μg/ml, from Sigma-Aldrich), as was a formamide-free hybridization buffer [1× SSC, 6 M urea (Sigma-Aldrich), 0.2% N-Lauroylsarcosine (Fluka), 0.5% Tween20, 1× Denhardt’s solution, 10% dextran sulfate, 0.75% Chaps (Sigma-Aldrich), 0.4 M dithiothreitol] with an adapted probe concentration of 400 ng/ml. Hybridization was performed at 53°C and stringency washes were accordingly adjusted.
Immunohistochemistry
For estimation of the amount of parenchymal and vascular amyloid, immunostaining against Aβ was performed with rabbit polyclonal antibody NT12, raised against Aβ 1-4031 (gifts of Drs. M. Staufenbiel and P. Paganetti), using standard immunoperoxidase procedures with Elite ABC kit (Vector Laboratories) and Vector SG (Vector Laboratories) as a substrate. For FISH/immuno double-staining, immunostaining followed the FISH protocol and preceded the counterstaining of cell nuclei. Alexa Fluor 633 (Invitrogen, Karlsruhe, Germany) goat anti-rabbit IgG secondary antibody was used to visualize NT12. For the intracellular experiments the primary antibodies NT12, rabbit polyclonal A4CT, specific to the C-terminal 100 amino acids of APP (courtesy of Dr. K. Beyreuther, Heidelberg, Germany), and mouse monoclonal 22C11, directed against amino acids 66 to 81 of the N-terminus of APP (Millipore, Schwalbach, Germany), were used in combination with Alexa Fluor 488 or 568 (Invitrogen) secondary antibodies. DRAQ5 (Biostatus Limited, Leicestershire, UK) was applied for counterstaining of cell nuclei and Mowiol (Kremer Pigmente, Aichstetten, Germany; preparation see Carl Roth GmbH, Karlsruhe, Germany) was used as the mounting medium.
Hemosiderin
To detect the occurrence of microhemorrhages, ferric iron in hemosiderin was visualized using the standard Berlin Blue method51 in a systematic set of every 20th section from the aged APPDutch mice and controls (8 to 10 sections/mouse).
Radioactive in Situ Hybridization (Arc and Synaptophysin)
All solutions and tissue pretreatment were as described for non-radioactive in situ hybridization. Relative probe concentration was measured by using LumaPlate-96 (Perkin Elmer) and the Scintillation Counter 1450 MicroBetaPlus (Wallac, Turku, Finland). RNA probes: Arc: 35S-labeled sense and antisense RNA probes were generated from nearly full-length Arc cDNA using [α-S35]-rUTP (1000 Ci/mmol, Hartmann Analytic, Braunschweig, Germany) and T3 or T7 RNA polymerases, respectively. RNA probes were DNaseI (Qiagen, Hilden, Germany) digested and purified on mini Quick Spin RNA Columns (Roche Diagnostics). Synaptophysin: the following synthetic 45mer oligo DNA probes were used, corresponding to the mouse synaptophysin sequence: Synaptophysin antisense: TCCGGTGTAGCTGCCGCACGTAGCAAAGGCGAAGATGGCAAAGAC; synaptophysin sense: GTCTTTGCCATCTTCGCCTTTGCTACGTGCGGCAGCTACACCGGA (biomers.net GmbH, Ulm, Germany). The oligonucleotide probes were labeled with terminal deoxynucleotidyl transferase (Fermentas, St. Leon-Rot, Germany) and [α-S35]dATP (1000 Ci/mmol, Hartmann Analytic). Hybridization: Arc: Probe RNA was diluted according to relative concentration measurements, denatured at 80°C for 5 minutes, and applied to the tissue. Sections were coverslipped and incubated 16 to 20 hours at 59°C in a humid chamber. Excess hybridization buffer was drained, and slides were washed twice in 2× SSC plus 1 mM EDTA. Sections were then treated with 10 μg/ml RNase A in TE/0.5 M NaCl and washed twice with 2× SSC plus 1 mM EDTA followed by 1× SSC/1 mM EDTA. Tissue was then subjected to several stringency washes in 0.1× SSC/1 mM EDTA at 59°C and 0.5× SSC and 0.1× SSC at room temperature. Synaptophysin: Oligo probe DNA was diluted in hybridization buffer, denatured at 80°C for 5 minutes, and applied to the tissue. Sections were coverslipped and incubated 16 to 20 hours at 37°C in a humid chamber. Excess hybridization buffer was drained, and slides were washed in 2× SSC at room temperature and in 2× SSC, 1× SSC, and 0.25× SSC at 37°C. Arc and synaptophysin: Sections were dehydrated in 50% and 70% ethanol and allowed to dry before exposure to Kodak BioMax MR film (Sigma-Aldrich). Quantification: Films were developed with a Fujifilm FPM800A (FUJIFILM Europe GmbH, Düsseldorf, Germany), dried and scanned at 800 pixels per inch. Image J software (http://rsb.info.nih.gov/ij/) was used to trace and quantify the mean optical density of the neocortex from every 20th section, from which the mean background signal of the according film was subtracted before calculating the overall mean per mouse. Individual section outliers were apparent in a few cases due to edge effects (>2SD of the respective group), and were excluded before calculation of the mean.
Fluorescent Microscopy and Stereology
Stained slides were analyzed with a Zeiss Axioplan 2 fluorescent microscope (Carl Zeiss MicroImaging GmbH, Jena, Germany). Image stacks were collected using a Zeiss Axiocam MRm digital camera and a Zeiss 63x/1.4 oil-immersion Plan-Apochromat objective from every 20th or 40th section (consistent within corresponding groups) through the entire neocortex (left hemisphere; anatomical borders as previously described52,53). A systematic-random sampling approach was used, with Z-stacks (10 optical sections, 1-μm spacing) taken every 800 μm spaced in a grid equally in XY space. For aged APPDutch animals and their respective controls, both hemispheres were analyzed, and therefore images were captured at a larger grid size (1131 μm). These sampling parameters yielded an average of 72 Z-stacks/subject. Illumination and contrast values were kept constant for sections/slides within each experiment. Neurons were identified based on DAPI counterstaining, with larger, diffusely stained, and rounded nuclei considered as presumptive neurons and included in the analysis, while the remainder of the cells, presumably glia cells, had much smaller irregularly shaped nuclei, and stained strongly with DAPI (these were never Arc-positive). In pilot studies, staining with the neuron-specific marker NeuN (Millipore) was compared with DAPI and revealed a close correspondence using these morphological criteria, and DAPI was used subsequently because NeuN staining did not provide reliable contrast following FISH. By using a customized ImageJ macro and the DAPI channel, only neuronal nuclei coming into focus while scanning through the Z stacks (4 μm depth; skipping the first Z level) were included in the analysis. With this approach, an optical disector technique, sampling errors (due to partial cells) are minimized and variations in cell volumes do not influence sampling frequencies.54 In a second step, overlapping channels were used to define all counted nuclei as being Arc-negative, or as showing intranuclear foci, cytoplasmic staining or both (adapted from 37). The total of Arc mRNA-positive neurons refers to the sum of intranuclear foci-, cytoplasmic- and double-positive cells. In cases with high Arc signal intensity, an additional Z-stack with lower illumination time was obtained, which served as a reference for correct categorization. The values are reported as percentage of Arc mRNA-positive neurons per total counted neurons for each mouse. Heavily-labeled Arc mRNA-expressing granule cells of the dentate gyrus were counted in every 20th section of all animals with a 20× objective by Z-scanning through the section manually (for aged APPDutch animals and controls both hemispheres were counted and divided by two).
Confocal Microscopy and Intracellular Analysis
Images were taken with a Zeiss LSM 510 META (Axiovert 200M) confocal microscope equipped with three lasers (488, 543, and 633 nm). The neocortex of one section per animal was systematic randomly sampled (1 hemisphere: 800 μm × 800 μm; both hemispheres: (2 × 566 μm) × (2 × 566 μm)) using a Zeiss Plan-Neofluor 40×/0.75 air objective. Settings were kept constant for different sessions and laser powers were measured with a PT 9610 laser power analyzer from Gigahertz-Optik (Puchheim, Germany) to control for changes in intensities. RGB images were analyzed with ImageJ using Sync Measure 3D and a customized macro allowing for simultaneous measurement of the Arc mRNA (Cy3) and the immunohistochemistry signal of NT12 (Alexa488). Regions of Interest of circular fixed sizes were specified for the different staining types and placed subsequently around all neuronal nuclei to include associated cytoplasmic label and immediately adjacent immunolabeling. Only neurons within a disector counting frame of 120 μm × 120 μm were quantified. From these all neurons showing lipofuscin, more frequent in the aged groups (APPPS1/ntg: 0.7%/0.3%; APPDutch/ntg: 13.5%/10.7%; APP23/ntg: 6.0%/7.5%), were categorized as such and excluded from statistical analyses because this endogenous fluorescent signal cannot be reliably separated from our other fluorophores. Categorization subtypes for Aβ are described in results. Mean pixel intensities were recorded for the separate channels within each Region of Interest. Numbers of mice for these analyses were the same as above except for the aged APPDutch mice (n = 6) and their respective controls (n = 3).
Estimation of Plaque Load and Vascular Amyloid
Estimation of amyloid plaque load (diffuse amyloid was not quantified) was performed on random sets of every 20th NT12-immunostained section throughout the neocortex using an area fraction technique, as described previously.53,55 Frequency and severity grade of vascular pathology in the aged APPDutch animals were estimated by two independent investigators examining sections from each animal at three matched anatomical levels.56
Statistics
Effects of the individual tg groups were tested by normalization to the corresponding ntg controls (mean set at 100%), which had been behaviorally and experimentally matched, thus correcting for potential differences in tissue processing and staining intensities. As neuron number estimates would not be subject to such variation, these were compared using total numbers. Individual tg groups and matching controls were compared using the Student’s t-test. For intracellular analyses, initial effect screening was done with analysis of variance, with Fisher’s protected least significant difference test used subsequently to compare groups as indicated. Because this data were not normally distributed and we were interested in comparing frequency distributions, differences in the extent of Arc activation between pathological subtypes were log (ln) transformed and tested using nonparametric statistics (rank-sums with Mann-Whitney). Mean values are given ± SEM.
Results
Age-Dependent Pathology
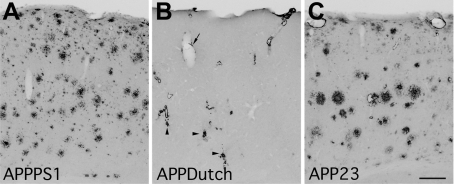
The basic pathology of these tg lines was consistent with previous descriptions, and only a brief summary is provided for the mice used in these studies. Aged APPPS1 mice (8 to 9 months) showed robust Aβ pathology in both cortical (Figure 1A) and hippocampal structures.29 Stereological quantification of the neocortical plaque load revealed an amyloid load of 10.3 ± 0.7%. Aged APPDutch mice (25 to 30 months) exhibited pathology in leptomeningeal and cortical vessels of all severity grades (smooth immunoreactivity within the vessel wall; granular immunoreactivity in and around the vessel wall; and fibrils extending from the vessel into the neuropil) (Figure 1B), also similar to that previously described.12,31 In half of the animals some minor vascular pathology was also apparent in the hippocampus. Staining for microhemorrhages (hemosiderin-positive cells) in a systematic set of sections revealed a mean of one instance per mouse. Aged APP23 mice (16 to 25 months) had pathology similar to previous descriptions32 with a group amyloid load of 5.0 ± 1.6% and a substantial amount of vascular amyloid56 (Figure 1C). Young APPPS1 mice were studied at the age of 2.5 to 3 months, with these mice exhibiting an amyloid load of 1.1 ± 0.5%. Plaques in the APPPS1 model tended to be smaller and more numerous than in mice with a similar plaque load from the APP23 line (Figure 1).
Figure 1.
Amyloid deposition in mouse models with distinct aspects of Alzheimer’s disease pathology. A: APPPS1 mice exhibit almost exclusively parenchymal amyloid, with plaques distributed throughout the neocortex. Shown is a 9-month-old mouse with a neocortical plaque load of 9.6%. B: APPDutch mice exhibit almost exclusively vascular amyloid, with pathology varying from smooth immunoreactivity in the vessel wall (arrow) to fibrils radiating from the vessel into the neuropil (arrowheads). An example of a more severely affected 29-month-old mouse is shown. C: APP23 mice develop both parenchymal and vascular pathologies. A 25-month-old mouse with neocortical plaque load of 8.5% is depicted. Scale bar: 200 μm (A–C).
Analysis of the percentage of Arc-expressing neurons (described below) also involved stereological sampling of neocortical neurons (identified morphologically via DAPI staining, see methods). Across lines, an overall average of 15.4 million DAPI-positive neurons was observed (both hemispheres), which remained stable with age in ntg mice (decrease <0.3 × 106; ns). While young tg mice of all lines did not differ from their age-matched controls, depositing mice of all lines revealed a modest decrease in total DAPI-positive neocortical neuron number compared with age-matched control mice (mean difference −3.3 × 106; or viewed as percent control, 81%, 87%, and 85% for the aged APPPS1, APPDutch, and APP23 lines, respectively; all P values <0.05). Note that the stereological Arc results reported below are activation percentage of existing neurons, and thus represent an additional effect.
Intracellular Granular Aβ
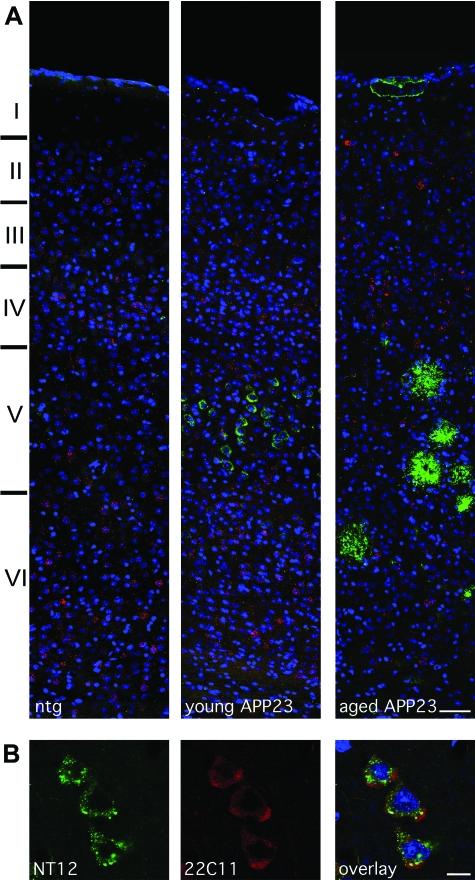
Young APPDutch and APP23 mice (3 to 4 months of age for both groups) did not show any amyloid deposition, but exhibited intracellular immunoreactivity with the Aβ-specific antibody NT12, most prominently in the large pyramidal cells of cortical layer V (Figure 2A), here referred to as intracellular granular Aβ. These granular structures were observed within the soma in different amounts and sizes, with the larger of these having an amorphous structure (Figure 2B). Notably they were positive specifically for the Aβ antibody NT12, and negative for the antibodies A4CT and 22C11, which are specific to the C- and N-terminus of APP respectively (Figure 2B).
Figure 2.
Intracellular granular Aβ is present predominantly in cortical layer V (shown here in a young APP23 mouse) and is localized subcellularly at locations distinct from APP. A: Layer distribution of Aβ and Arc mRNA expression in the neocortex (S1 somatosensory cortex shown). The left-most panel illustrates normal Arc mRNA expression in a nontransgenic animal. Note the Arc expression in the non-transgenic cortical layer V neurons. Intracellular granular Aβ staining is shown in a young APP23 mouse at the age of 3.5 months. With increasing amyloid deposition, a decline in intracellular granular Aβ was apparent and virtually zero positive neurons remained in an aged (25-month-old) depositing APP23 mouse. (red: Arc mRNA; green: Aβ; blue: DRAQ5). B: The intracellular granules are distinct in size, subcellular distribution, and staining intensity (NT12; green) when compared with the diffuse cytoplasmic labeling observed with staining for APP (22C11; red). An overlay with cell nuclei (DRAQ5; blue) is also shown. Scale bars: 50 μm (A); 10 μm (B).
Intracellular Aβ accumulation was a transient phenomenon, decreasing concurrently with expanding amyloid deposition. This observation is in line with other studies showing an inverse correlation of intra- and extracellular Aβ.21,57 Aged APPDutch (25 to 30 months) and aged APP23 mice at the age of 25 months showed virtually no persisting intracellular Aβ, while it was still present in aged APP23 mice at the age of 16 to 19 months (ie, in those with a lower plaque load: 2.6 ± 0.3%, compared with 9.9 ± 1.4% at 25 months). APPPS1 mice were negative for intracellular granular Aβ at both ages studied. Quantification of granular Aβ (see below) revealed prominent examples in a relatively small subset of neurons (young mice: APP23: 4.7%; APPDutch: 0.8%), although the incidence was much higher in cortical layer V, and smaller granules below the current detection threshold were almost certainly present.
Arc Induction and Decrease in Percentage of Arc mRNA-Positive Neocortical Neurons
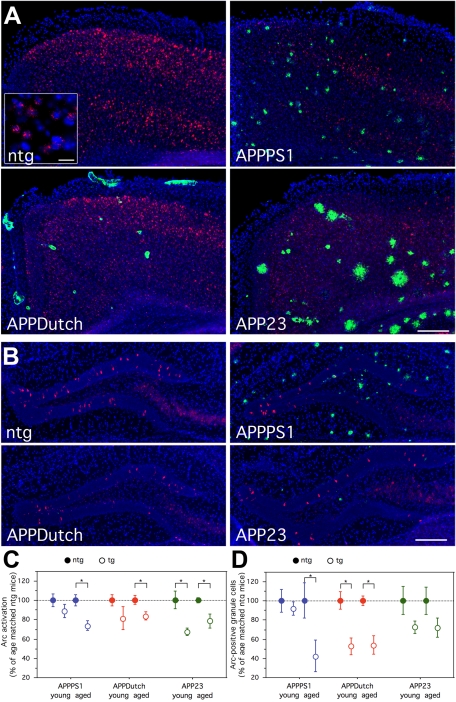
Young and aged mice and age-matched controls (see Materials and Methods, for ages of each group) were exposed twice to novel environments. Since our main aim was to identify potential deficits in neuronal encoding, behavior itself was controlled to assure an equivalent amount of exposure in each mouse, as described in the methods. Arc mRNA-positive neurons were quantified in the neocortex and the dentate gyrus, brain areas that show Arc induction following this behavioral protocol37,58 and are, to varying extents, affected by amyloid pathology. When neocortical Arc mRNA expression was compared qualitatively, a reduction of Arc mRNA signal was apparent in aged APPPS1, APPDutch, and APP23 mice, and the pattern of activated, Arc mRNA positive cells was disrupted around amyloid plaques (Figure 3A).
Figure 3.
Reduced Arc mRNA expression in mouse models of cerebral amyloidosis. A decrease in the number of Arc mRNA-expressing neurons in the neocortex and dentate gyrus following exploratory behavior was observed for parenchymal and vascular depositing mice as well as for young animals before amyloid deposition. A: Prominent Arc mRNA expression (red) is evident throughout all neocortical layers in a ntg control mouse (top-left; example shown at 29 months of age–see Materials and Methods for matched ages in all groups). In comparison, reduced Arc mRNA induction is evident within the neocortex of Aβ depositing mice (APPPS1 [9 mo], APPDutch [29 mo], and APP23 [25 mo]); (red: Arc mRNA; green: Aβ; blue: cell nuclei [DAPI]). Scale bar = 200 μm. The inlay shows Arc-positive neurons with cytoplasmic Arc mRNA and/or intranuclear foci, referring to the different activation time points of the cells. Scale bar = 20 μm. B: A small percentage of dentate gyrus granule cells express high levels of Arc mRNA in a ntg control mouse (top-left; 29 months old), whereas a reduction of Arc mRNA-expressing dentate gyrus granule cells is evident for aged APPPS1 and APPDutch mice. APP23 mice were less impaired (red: Arc mRNA; green: Aβ; blue: DAPI). Scale bar = 200 μm. C: Stereological quantification of neocortical Arc mRNA-positive neurons revealed changes in the percentage of Arc mRNA positive neurons in relation to age-matched ntg controls. D: Changes in total number of Arc mRNA-positive neurons within the dentate gyrus compared with age-matched wild-type controls. C and D: Number of mice per group (ntg/tg): young APPPS1 (5/5); aged APPPS1 (5/5); young APPDutch (4/6); aged APPDutch (7/8); young APP23 (5/3); aged APP23 (7/6).
Stereological quantification of Arc mRNA expressing neurons in the neocortex revealed a strong increase following behavior, with an overall average of 31% neocortical neurons being positive, compared with the homecage control group (6.0%). No reduction was observed with aging in the non-transgenic (ntg) mice. Compared with age-matched ntg mice, a decrease in the percentage of Arc mRNA-positive neurons within the neocortex was found for both parenchymal [APPS1: −26.3%, t(1,8) = 3.37; P < 0.01] and vascular [APPDutch: −16.4%, t(1,13) = 2.61; P < 0.05] depositing mice and for depositing APP23 mice having both pathologies [−21.4%, t(1,11) = 2.94; P < 0.05] (Figure 3C). A reduction was also observed in young APP23 mice [−32.5%, t(1,6) = 2.61; P < 0.05]. Although the percent change was similar to the aged group, young APPDutch did not differ significantly from their respective controls [−18.6%, t(1,8) = 1.23, n.s.].
Vulnerability of Dentate Gyrus
We next examined Arc mRNA expression in the dentate gyrus. Again, no significant changes were observed for young APPPS1 mice, whereas young APPDutch and APP23 mice showed a reduction similar to that of depositing mice of the same line (Figure 3, B and D). The total number of Arc mRNA-positive granule cells in one hemisphere was approximately 2950 for the aged ntg groups, while it was only ∼1240 and ∼1580 for the aged APPPS1 and APPDutch animals respectively, indicating a decrease of over 50%. Independent of age, APP23 mice were less impaired than other lines (both ages: −28%), with the difference not reaching statistical significance. Notably, the values for impaired lines fell below the baseline level of the HC group (∼2140 Arc mRNA-positive granule cells).
Decrease in Neocortical Arc mRNA Level
Similar results were obtained when the overall levels of neocortical Arc mRNA were compared via radioactive in situ hybridization, verifying and extending the stereological results. Representative pictures of Arc mRNA expression patterns in the neocortex and hippocampus are depicted for aged APPPS1, APPDutch, APP23, and a ntg animal in Figure 4A. Quantitative analysis of the mean neocortical Arc mRNA levels revealed a decrease of 14.8% in aged APPPS1 [t(1,8) = 2.53; P < 0.05], 14.4% in aged APPDutch [t(1,13) = 2.99; P = 0.01], and 15.9% in aged APP23 mice [t(1,11) = 3.88; P < 0.01], demonstrating a reduction in all depositing groups. In this analysis, significant results were also obtained for the young APPDutch animals [young APPDutch: −21.8%, t(1,8) = 3.49; P < 0.01], and the young APP23 mice had a trend toward reduced Arc levels [−13.3%, t(1,6) = 2.06; P = 0.08], while young APPPS1 mice were unaltered (Figure 4B).
Figure 4.
The decrease in total neocortical Arc mRNA levels was similar to the stereological results whereas synaptophysin mRNA expression is not reduced. A: Relative levels of Arc mRNA were detected via radioactive in situ hybridization. Representative pictures show the typical Arc activation pattern from aged animals of each line. No signal was observed with the sense Arc riboprobe. B: Quantification of the amount of neocortical Arc mRNA (mean intensity) in relation to age-matched ntg controls. C: Representative pictures illustrating neocortical synaptophysin mRNA expression. No signal was detected with the sense synaptophysin oligo probe. D: No significant changes in the relative levels of neocortical synaptophysin mRNA were obtained for any group in comparison with age-matched controls. B and D: Number of mice per group (ntg/tg): young APPPS1 (5/5); aged APPPS1 (5/5); young APPDutch (4/6); aged APPDutch (7/8); young APP23 (5/3); aged APP23 (7/6).
Stable Synaptophysin mRNA Expression
We then asked whether the observed reduction in total Arc mRNA expression is specific through quantification of the RNA for an unrelated protein, the synaptic vesicle membrane protein synaptophysin. Radioactive in situ hybridization failed to reveal a decrease in any of the groups (Figure 4, C and D).
Effects of Aβ-Single Cell Analyses
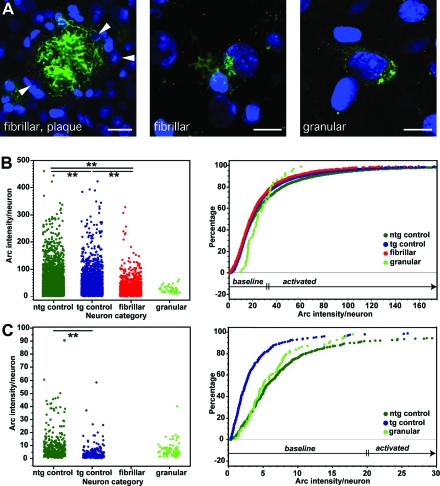
The main effects from the above analyses pointed to deficits related to the deposition of amyloid (eg, age-specific reduction in APPPS1 mice), but also to a prominent effect before the onset of traditional pathological features (deficits in the young APPDutch and APP23 mice). Another random stereological sampling scheme was thus designed to examine the relationship between Arc in individual neurons and the observed Aβ pathologies—intracellular granular and extracellular fibrillar Aβ. This relied on a modified FISH protocol combined with immunostaining for Aβ, measurement of fluorescence intensities at high power in circular areas each containing an individual neuronal nucleus and the immediately adjacent area including this neuron’s cytoplasm (Figure 5; see methods for details). Each neuron was then further characterized based on the presence of Aβ pathology using morphological criteria as described below, or if no abnormal Aβ immunostaining was present, as “control.” An overall effect of pathological subtype was observed [Figure 5B; F(3,11841) = 16.0; P < 0.001], with posthoc testing also revealing a difference for control neurons between ntg and tg mice (P < 0.001).
Figure 5.
Vicinity to fibrillar Aβ and the presence of intracellular granular Aβ both reduce Arc mRNA expression. A: Example pictures of neurons neighboring fibrillar Aβ, either in form of a plaque (left; white arrowheads indicate a subset of the affected neuronal nuclei) or diffuse fibrillar Aβ (center). Image on the right illustrates a neuron showing intracellular granular Aβ. Scale bars: 20 μm (left); 10 μm (middle and right). red: Arc mRNA; green: Aβ; blue: DRAQ5. B: Arc intensity measurement around single neuronal nuclei revealed an overall reduction in tg control neurons (ie, those negative for the separately analyzed Aβ pathologies) compared with ntg neurons from wild-type mice. A further reduction was found for fibrillar neurons in close vicinity to plaques/diffuse Aβ. Neurons with intracellular granular Aβ showed no significant mean difference, which is explained by the Arc intensity distribution (percentile plot, lime green curve) in the overall neocortical analysis (baseline shift). In total 29 ntg and 31 tg mice were analyzed. **P < 0.01 C: Targeted analysis of Arc mRNA expression in individual layer V cortical neurons of young APPDutch (n = 6) and APP23 (n = 3) mice and age-matched ntg mice (n = 4 and 5). A significant difference in mean Arc intensity levels was found for tg versus ntg control neurons. Activated neurons were almost completely absent for the population of neurons containing Aβ granules.
Arc mRNA Expression Reduced near Amyloid Fibrils
Individual neurons were classified as ‘fibrillar’ if NT12-positive extracellular amyloid fibrils were apparent in direct proximity to the neuron, whether or not those fibrils appeared to be associated with a dense amyloid deposit/plaque (Figure 5A). These criteria led to categorization of 6.8% of the neurons in the young APPPS1 mice as fibrillar, and for the aged APPPS1, APPDutch, and APP23 mice, the percentages were 80%, 0.8%, and 49% respectively. Note that the inclusion of diffuse amyloid led to substantially higher values than if only plaque-associated neurons were counted.
The overall effect of the presence of fibrillar Aβ across all tg lines was to reduce the average amount of Arc mRNA expression per neuron to 79% of ntg control levels (Figure 5B). Importantly, neurons associated with fibrillar amyloid also had significantly lower Arc levels than unaffected neurons in the tg lines (Figure 5B). Similar results were obtained when using a threshold Arc intensity level to compute the percentage of activated neurons (calculated to match the percentage activated from the above stereological results). In the APP23 aged group for instance, 31% of neurons were active in ntg controls; 26% for tg control neurons; and only 19% of fibrillar-associated neurons, F(3,4035) = 12.7; Fisher’s PLSD for all comparisons P < 0.01. These results show that fibrillar Aβ contributes to the observed Arc deficit in depositing mice.
Intracellular Granular Aβ Alters Arc Expression before Amyloid Pathology
Measurement of the amount of Arc mRNA per neuron or, according to the above described threshold, the percentage of Arc activated neurons did not reveal any significant mean differences for granular Aβ containing neurons compared with ntg or tg control neurons. However this was due to the unique Arc intensity distribution of granular-positive neurons (Figure 5B), which had higher baseline levels of Arc, but almost no neurons expressing Arc at higher levels.
Because of preferential layer V localization, we were concerned that sampling issues influence the observed difference in intensity distribution. We thus performed a targeted sampling of >1200 randomly selected neurons from sensorimotor cortex layer V in young mice from all tg lines and their age-matched controls (n = 28 mice). Neurons with intracellular granular Aβ represented 0%, 9.7%, and 50%, of the analyzed layer V neurons for the APPPS1, APPDutch, and APP23 mice, respectively. Consistent with the overall neocortical results, the mean Arc level was unchanged for young APPPS1 mice compared with their age-matched controls [0% difference; F(1479) = 0.00], but was significantly reduced for young APPDutch [to 41%; F(1381) = 25.6; P < 0.001], and young APP23 [to 66%; F(1354) = 11.45; P < 0.001] mice. Because young APPPS1 mice showed no intracellular granular Aβ, they were excluded from further statistical analyses.
An overall group effect of neuron type was found in layer V [F(2736) = 22.2; P < 0.001], with tg control neurons expressing only 44% of the Arc mRNA level of ntg controls (P < 0.001), and granular neurons 74% (P = 0.0505) (Figure 5C). The stronger reduction in tg control neurons can be explained by a decrease in low Arc baseline levels, while these levels were not different between granular and ntg control neurons (Figure 5C, percentile graph). However, the number of activated tg neurons (computed via a recalibrated threshold based on visual confirmation of activated neurons in this layer) was markedly reduced compared with ntg neurons [8.0% activation; main effect of staining subtype F(2736) = 8.12, P < 0.001] independent of whether these neurons contained Aβ granules (1.3% activation; compared with ntg, P < 0.05) or not (1.6% activation; compared with ntg, P < 0.001). In summary, Arc induction was markedly impaired in cortical layer V when granular Aβ was present (young APPDutch and APP23 mice), and unchanged when not (young APPPS1 mice).
Discussion
The pathogenesis of AD is clearly multifactorial, but this simple and well-known statement has been perhaps the biggest single impediment to understanding how the various brain changes contribute to cognitive decline. The generation of a wider array of mouse model systems replicating various elements of the disease process now facilitates research on distinct factors as potential culprits. By visualizing behavioral Arc induction and the specific pathologies within these models, we have been able to demonstrate the independent impact of both traditional amyloid pathologies and intracellular granular Aβ.
Sensitivity and specificity of cognitive measures are difficult issues in mice, 59,60 particularly when examining different strains, substrains, or ages.61 Notably all mice analyzed in this study had the same background (C57BL/6J), were age-matched, and expressed the transgene(s) under the same neuron-specific promoter (Thy1). In addition we used a controlled behavioral task, which by assuring equivalent exposure in all mice, allows subsequent comparison of encoding deficits. Although environmental exploration is necessarily basic, the associated spatial memory and involved neural systems are compromised in and relevant to AD and the models being studied. Following the same behavioral experience, we observed a reduction in total neocortical Arc activation, several-fold decreases in layer V activation dependent on pathological subtype, and a dentate gyrus activation below control baseline. These deficits in learning-associated plasticity were dependent on tg line and pathology type as discussed below.
Fibrillar Amyloid
The reduction in Arc mRNA expression in aged, depositing, APPPS1 mice (with young APPPS1 unchanged) argues for a role of the extracellular deposition of amyloid. If only considering their local impact, dense plaques would however be insufficient to explain the magnitude of deficit (APPPS1, plaques versus reduction in activated neurons: 10.3% vs. 26.3%; APP23: 5.0% vs. 21.4%). Direct study of neurons in the vicinity of amyloid fibrils strengthened this point, as impairment was observed independent of whether the fibrils are part of a compact plaque, vascular amyloid, or more diffuse fibrils. These results argue against the necessity for structural neurodegenerative changes as diffuse amyloid is not associated with microglia activation or neuron loss.62,63 Finally, the impact of Aβ on learning-related gene expression occurred independent of the ratio between Aβ 42/40, which changes substantially between the APPPS1 and APP23/APPDutch models, and whether the Aβ included a mutation (APPDutch) that led to its vascular rather than parenchymal localization. The combination of these results argues strongly that amyloid accumulation is a negative factor in cognition, rather than simply an epiphenomena.1,2
Intracellular Granular Aβ
Analysis across tg lines revealed the younger mice that exhibited granules (APPDutch and APP23) also exhibited a reduction in Arc. When analyzed directly, layer V where these neurons were prominent, showed a marked reduction in Arc expression and activation likelihood compared with ntg controls—substantially lower than the overall neocortical deficit. A reduction in Arc activation was observed for both granular and ‘control’ tg neurons and further study is required to isolate whether a high detection threshold influenced this result (ie, all layer V neurons could contain a small amount of granular Aβ). Alternatively, granules may represent a successful internal sequestration of Aβ, with these neurons thus able to maintain baseline Arc levels. Unlike with extracellular amyloid discussed above, where the spatial localization of the deficit may occur at a distance from the Aβ source, the somatic localization in layer V indicates that Aβ processing events within individual neurons lead to their inability to express Arc normally. This and the magnitude difference indicate that the reduction in layer V plasticity may however lead to downstream effects such as a loss of Arc in postsynaptic neurons or activated networks as a whole (see also dentate gyrus results).
Vascular Amyloid
The small subset of neurons in close contact with amyloid fibrils in the APPDutch model were not Arc-positive, but the extent of these cases was not enough to explain the overall deficit (16.4% reduction). The presence of hemorrhages is most certainly a factor complicating interpretation of CAA’s cognitive impact in other studies, but because of their relative infrequency in the mice used in this study, it is unlikely that they contributed. Of the remaining potential factors, it is unclear whether the deficit is downstream of vascular amyloid accumulation, represents a residual effect of intracellular factors, or a completely independent effect due to the presence of mutated Aβ. However, evidence for a direct impact of CAA is given by the similar pathogenesis to the APP23 model, where intracellular granular and fibrillar effects were not additive, indicating a transient effect of intracellular Aβ, thus leaving vascular changes as the primary culprit of the deficit in the aged group. In terms of therapeutics, this would suggest that clearing Aβ deposits from the brain into vascular compartments is not likely to improve cognition.
Neuron Loss
The observation of a modest reduction in the total number of neurons (identified morphologically using DAPI staining) specifically in the aged tg lines was somewhat unexpected, and we would generally suggest that this observation be confirmed via quantification using a more specific staining technique (eg, Nissl). However, if there were to be misidentification of glial cells as neurons in this study, the total number would be expected to increase in the tg lines, as substantially higher numbers of microglia are found following amyloid deposition.29,52 Instead, our results would be more consistent with a shift away from categorization of the observed DAPI-positive nuclei as neurons, and could represent a subset of neurons that were excluded due to smaller, darker, or more fragmented nuclear characteristics. Previous stereological analysis of Nissl-stained neurons in aged APP23 mice has demonstrated an inverse relationship to plaque load but no overall loss in neocortex compared with age-matched controls,52 and further analyses in the APPDutch and APPPS1 lines is necessary.
Timing and Relative Impact
The observed effects of fibrillar and intracellular granular Aβ appear to be independent and are not numerically additive. We observed a reduction in the presence of granules concomitant with increasing amyloid deposition. Although more evidence is required to show causality, this would lend credence to amyloid sequestration as a therapeutic strategy. Perhaps most promising in this model with independent timing of the two factors is that the first phase is reversible and would allow intervention following early detection. How smoothly the two factors integrate in time requires more study and our observation of a few residual puncta in mice at early/mid stages of amyloid deposition indicates the process may be gradual, would potentially lead to incomplete recovery, and might require treatment over a long interval.
As indicated above, in advanced stages of the disease, the impact of amyloid fibrils alone would certainly be sufficient to explain cognitive decline, particularly given the often widespread localization of diffuse amyloid. The early stages of the disease pose however a greater mystery. It may be that the answer lies more in region specificity—in all cases where a neocortical reduction was observed, the dentate gyrus was more severely affected, with the number of neurons active following behavior equivalent to control homecage levels. This reduction was apparent regardless of whether the underlying mechanism could be attributed to fibrillar Aβ or to intracellular granules. Although we cannot exclude direct effects of these two pathologies within the dentate, these results may suggest a funnel effect—as the widespread neocortical inputs converge on the dentate, plasticity within is reduced regardless of the original extrinsic cause. Dentate encoding is extremely sparse,58,64 evidenced here also by the very small percentage of Arc-positive neurons, and given a theoretical function of the dentate in pattern separation, a common deficit here could prevent accurate memory retrieval. Other soluble oligomeric forms of Aβ, which are difficult to delineate with immunocytochemistry alone, might also add to the observed effect, and thus further study is necessary, and could also shed light on the question of whether additional populations of neurons are affected in the young predepositing mice.
In summary, these results have reemphasized the negative impact of Aβ—whether within neurons or as extracellular fibrils, and demonstrated that the deficits occurring before the formation of amyloid plaques are transient and do not add to the later effect associated with fibrillary amyloid. Further study is necessary to isolate the intracellular events leading to a reduction in these neurons ability to express Arc, and to see how these events may be linked.
Acknowledgments
We thank David Milford and Birgitt Schoenfisch for analytical support.
Footnotes
Address reprint requests to Michael E. Calhoun, Department of Cellular Neurology, Hertie-Institute for Clinical Brain Research, University of Tübingen, Otfried-Müller-Strasse 27, D-72076 Tübingen, Germany. E-mail: michael.calhoun@uni-tuebingen.de.
Supported in part by grants from the Deutsche Forschungsgemeinschaft (CA 477-1/2) and Alzheimer’s Association (IIRG-05-13464) to M.E.C. and a fellowship to B.M.W.-B. of the German National Academic Foundation. Additional support was provided by grants to M. Jucker from the German National Genome Network (NGFNPlus).
References
- Arriagada PV, Growdon JH, Hedley-Whyte ET, Hyman BT. Neurofibrillary tangles but not senile plaques parallel duration and severity of Alzheimer’s disease. Neurology. 1992;42:631–639. doi: 10.1212/wnl.42.3.631. [DOI] [PubMed] [Google Scholar]
- Ingelsson M, Fukumoto H, Newell KL, Growdon JH, Hedley-Whyte ET, Frosch MP, Albert MS, Hyman BT, Irizarry MC. Early Abeta accumulation and progressive synaptic loss, gliosis, and tangle formation in AD brain. Neurology. 2004;62:925–931. doi: 10.1212/01.wnl.0000115115.98960.37. [DOI] [PubMed] [Google Scholar]
- Tsai J, Grutzendler J, Duff K, Gan WB. Fibrillar amyloid deposition leads to local synaptic abnormalities and breakage of neuronal branches. Nat Neurosci. 2004;7:1181–1183. doi: 10.1038/nn1335. [DOI] [PubMed] [Google Scholar]
- Brendza RP, Bacskai BJ, Cirrito JR, Simmons KA, Skoch JM, Klunk WE, Mathis CA, Bales KR, Paul SM, Hyman BT, Holtzman DM. Anti-Abeta antibody treatment promotes the rapid recovery of amyloid-associated neuritic dystrophy in PDAPP transgenic mice. J Clin Invest. 2005;115:428–433. doi: 10.1172/JCI23269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spires TL, Meyer-Luehmann M, Stern EA, McLean PJ, Skoch J, Nguyen PT, Bacskai BJ, Hyman BT. Dendritic spine abnormalities in amyloid precursor protein transgenic mice demonstrated by gene transfer and intravital multiphoton microscopy. J Neurosci. 2005;25:7278–7287. doi: 10.1523/JNEUROSCI.1879-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stern EA, Bacskai BJ, Hickey GA, Attenello FJ, Lombardo JA, Hyman BT. Cortical synaptic integration in vivo is disrupted by amyloid-beta plaques. J Neurosci. 2004;24:4535–4540. doi: 10.1523/JNEUROSCI.0462-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Humpel C, Marksteiner J. Cerebrovascular damage as a cause for Alzheimer’s disease. Curr Neurovasc Res. 2005;2:341–347. doi: 10.2174/156720205774322610. [DOI] [PubMed] [Google Scholar]
- Jellinger KA. Alzheimer disease and cerebrovascular pathology: an update. J Neural Transm. 2002;109:813–836. doi: 10.1007/s007020200068. [DOI] [PubMed] [Google Scholar]
- Attems J, Quass M, Jellinger KA, Lintner F. Topographical distribution of cerebral amyloid angiopathy and its effect on cognitive decline are influenced by Alzheimer disease pathology. J Neurol Sci. 2007;257:49–55. doi: 10.1016/j.jns.2007.01.013. [DOI] [PubMed] [Google Scholar]
- Vinters HV. Cerebral amyloid angiopathy. A critical review. Stroke. 1987;18:311–324. doi: 10.1161/01.str.18.2.311. [DOI] [PubMed] [Google Scholar]
- Itoh Y, Yamada M, Hayakawa M, Otomo E, Miyatake T. Cerebral amyloid angiopathy: a significant cause of cerebellar as well as lobar cerebral hemorrhage in the elderly. J Neurol Sci. 1993;116:135–141. doi: 10.1016/0022-510x(93)90317-r. [DOI] [PubMed] [Google Scholar]
- Winkler DT, Bondolfi L, Herzig MC, Jann L, Calhoun ME, Wiederhold KH, Tolnay M, Staufenbiel M, Jucker M. Spontaneous hemorrhagic stroke in a mouse model of cerebral amyloid angiopathy. J Neurosci. 2001;21:1619–1627. doi: 10.1523/JNEUROSCI.21-05-01619.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Natte R, Maat-Schieman ML, Haan J, Bornebroek M, Roos RA, van Duinen SG. Dementia in hereditary cerebral hemorrhage with amyloidosis-Dutch type is associated with cerebral amyloid angiopathy but is independent of plaques and neurofibrillary tangles. Ann Neurol. 2001;50:765–772. doi: 10.1002/ana.10040. [DOI] [PubMed] [Google Scholar]
- Greenberg SM, Gurol ME, Rosand J, Smith EE. Amyloid angiopathy-related vascular cognitive impairment. Stroke. 2004;35:2616–2619. doi: 10.1161/01.STR.0000143224.36527.44. [DOI] [PubMed] [Google Scholar]
- Thal DR, Ghebremedhin E, Orantes M, Wiestler OD. Vascular pathology in Alzheimer disease: correlation of cerebral amyloid angiopathy and arteriosclerosis/lipohyalinosis with cognitive decline. J Neuropathol Exp Neurol. 2003;62:1287–1301. doi: 10.1093/jnen/62.12.1287. [DOI] [PubMed] [Google Scholar]
- LaFerla FM, Green KN, Oddo S. Intracellular amyloid-beta in Alzheimer’s disease. Nat Rev Neurosci. 2007;8:499–509. doi: 10.1038/nrn2168. [DOI] [PubMed] [Google Scholar]
- Tseng BP, Kitazawa M, LaFerla FM. Amyloid beta-peptide: the inside story. Curr Alzheimer Res. 2004;1:231–239. doi: 10.2174/1567205043332045. [DOI] [PubMed] [Google Scholar]
- Gouras GK, Tsai J, Naslund J, Vincent B, Edgar M, Checler F, Greenfield JP, Haroutunian V, Buxbaum JD, Xu H, Greengard P, Relkin NR. Intraneuronal Abeta42 accumulation in human brain. Am J Pathol. 2000;156:15–20. doi: 10.1016/s0002-9440(10)64700-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mochizuki A, Tamaoka A, Shimohata A, Komatsuzaki Y, Shoji S. Abeta42-positive non-pyramidal neurons around amyloid plaques in Alzheimer’s disease. Lancet. 2000;355:42–43. doi: 10.1016/S0140-6736(99)04937-5. [DOI] [PubMed] [Google Scholar]
- Gyure KA, Durham R, Stewart WF, Smialek JE, Troncoso JC. Intraneuronal abeta-amyloid precedes development of amyloid plaques in Down syndrome. Arch Pathol Lab Med. 2001;125:489–492. doi: 10.5858/2001-125-0489-IAAPDO. [DOI] [PubMed] [Google Scholar]
- Mori C, Spooner ET, Wisniewsk KE, Wisniewski TM, Yamaguch H, Saido TC, Tolan DR, Selkoe DJ, Lemere CA. Intraneuronal Abeta42 accumulation in Down syndrome brain. Amyloid. 2002;9:88–102. [PubMed] [Google Scholar]
- Knobloch M, Konietzko U, Krebs DC, Nitsch RM. Intracellular Abeta and cognitive deficits precede beta-amyloid deposition in transgenic arcAbeta mice. Neurobiol Aging. 2007;28:1297–1306. doi: 10.1016/j.neurobiolaging.2006.06.019. [DOI] [PubMed] [Google Scholar]
- Oakley H, Cole SL, Logan S, Maus E, Shao P, Craft J, Guillozet-Bongaarts A, Ohno M, Disterhoft J, Van Eldik L, Berry R, Vassar R. Intraneuronal beta-amyloid aggregates, neurodegeneration, and neuron loss in transgenic mice with five familial Alzheimer’s disease mutations: potential factors in amyloid plaque formation. J Neurosci. 2006;26:10129–10140. doi: 10.1523/JNEUROSCI.1202-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oddo S, Caccamo A, Shepherd JD, Murphy MP, Golde TE, Kayed R, Metherate R, Mattson MP, Akbari Y, LaFerla FM. Triple-transgenic model of Alzheimer’s disease with plaques and tangles: intracellular Abeta and synaptic dysfunction. Neuron. 2003;39:409–421. doi: 10.1016/s0896-6273(03)00434-3. [DOI] [PubMed] [Google Scholar]
- Wirths O, Multhaup G, Czech C, Blanchard V, Moussaoui S, Tremp G, Pradier L, Beyreuther K, Bayer TA. Intraneuronal Abeta accumulation precedes plaque formation in beta-amyloid precursor protein and presenilin-1 double-transgenic mice. Neurosci Lett. 2001;306:116–120. doi: 10.1016/s0304-3940(01)01876-6. [DOI] [PubMed] [Google Scholar]
- Magrane J, Smith RC, Walsh K, Querfurth HW. Heat shock protein 70 participates in the neuroprotective response to intracellularly expressed beta-amyloid in neurons. J Neurosci. 2004;24:1700–1706. doi: 10.1523/JNEUROSCI.4330-03.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Y, McLaughlin R, Goodyer C, LeBlanc A. Selective cytotoxicity of intracellular amyloid beta peptide1–42 through p53 and Bax in cultured primary human neurons. J Cell Biol. 2002;156:519–529. doi: 10.1083/jcb.200110119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Billings LM, Oddo S, Green KN, McGaugh JL, LaFerla FM. Intraneuronal Abeta causes the onset of early Alzheimer’s disease-related cognitive deficits in transgenic mice. Neuron. 2005;45:675–688. doi: 10.1016/j.neuron.2005.01.040. [DOI] [PubMed] [Google Scholar]
- Radde R, Bolmont T, Kaeser SA, Coomaraswamy J, Lindau D, Stoltze L, Calhoun ME, Jaggi F, Wolburg H, Gengler S, Haass C, Ghetti B, Czech C, Holscher C, Mathews PM, Jucker M. Abeta42-driven cerebral amyloidosis in transgenic mice reveals early and robust pathology. EMBO Rep. 2006;7:940–946. doi: 10.1038/sj.embor.7400784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levy E, Carman MD, Fernandez-Madrid IJ, Power MD, Lieberburg I, van Duinen SG, Bots GT, Luyendijk W, Frangione B. Mutation of the Alzheimer’s disease amyloid gene in hereditary cerebral hemorrhage. Dutch type, Science. 1990;248:1124–1126. doi: 10.1126/science.2111584. [DOI] [PubMed] [Google Scholar]
- Herzig MC, Winkler DT, Burgermeister P, Pfeifer M, Kohler E, Schmidt SD, Danner S, Abramowski D, Sturchler-Pierrat C, Burki K, van Duinen SG, Maat-Schieman ML, Staufenbiel M, Mathews PM, Jucker M. Abeta is targeted to the vasculature in a mouse model of hereditary cerebral hemorrhage with amyloidosis. Nat Neurosci. 2004;7:954–960. doi: 10.1038/nn1302. [DOI] [PubMed] [Google Scholar]
- Sturchler-Pierrat C, Abramowski D, Duke M, Wiederhold KH, Mistl C, Rothacher S, Ledermann B, Burki K, Frey P, Paganetti PA, Waridel C, Calhoun ME, Jucker M, Probst A, Staufenbiel M, Sommer B. Two amyloid precursor protein transgenic mouse models with Alzheimer disease-like pathology. Proc Natl Acad Sci USA. 1997;94:13287–13292. doi: 10.1073/pnas.94.24.13287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lyford GL, Yamagata K, Kaufmann WE, Barnes CA, Sanders LK, Copeland NG, Gilbert DJ, Jenkins NA, Lanahan AA, Worley PF. Arc, a growth factor and activity-regulated gene, encodes a novel cytoskeleton-associated protein that is enriched in neuronal dendrites. Neuron. 1995;14:433–445. doi: 10.1016/0896-6273(95)90299-6. [DOI] [PubMed] [Google Scholar]
- Link W, Konietzko U, Kauselmann G, Krug M, Schwanke B, Frey U, Kuhl D. Somatodendritic expression of an immediate early gene is regulated by synaptic activity. Proc Natl Acad Sci USA. 1995;92:5734–5738. doi: 10.1073/pnas.92.12.5734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guzowski JF, Lyford GL, Stevenson GD, Houston FP, McGaugh JL, Worley PF, Barnes CA. Inhibition of activity-dependent arc protein expression in the rat hippocampus impairs the maintenance of long-term potentiation and the consolidation of long-term memory. J Neurosci. 2000;20:3993–4001. doi: 10.1523/JNEUROSCI.20-11-03993.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plath N, Ohana O, Dammermann B, Errington ML, Schmitz D, Gross C, Mao X, Engelsberg A, Mahlke C, Welzl H, Kobalz U, Stawrakakis A, Fernandez E, Waltereit R, Bick-Sander A, Therstappen E, Cooke SF, Blanquet V, Wurst W, Salmen B, Bosl MR, Lipp HP, Grant SG, Bliss TV, Wolfer DP, Kuhl D. Arc/Arg3.1 is essential for the consolidation of synaptic plasticity and memories. Neuron. 2006;52:437–444. doi: 10.1016/j.neuron.2006.08.024. [DOI] [PubMed] [Google Scholar]
- Guzowski JF, McNaughton BL, Barnes CA, Worley PF. Environment-specific expression of the immediate-early gene Arc in hippocampal neuronal ensembles. Nat Neurosci. 1999;2:1120–1124. doi: 10.1038/16046. [DOI] [PubMed] [Google Scholar]
- Burke SN, Chawla MK, Penner MR, Crowell BE, Worley PF, Barnes CA, McNaughton BL. Differential encoding of behavior and spatial context in deep and superficial layers of the neocortex. Neuron. 2005;45:667–674. doi: 10.1016/j.neuron.2005.01.042. [DOI] [PubMed] [Google Scholar]
- Tagawa Y, Kanold PO, Majdan M, Shatz CJ. Multiple periods of functional ocular dominance plasticity in mouse visual cortex. Nat Neurosci. 2005;8:380–388. doi: 10.1038/nn1410. [DOI] [PubMed] [Google Scholar]
- Wang KH, Majewska A, Schummers J, Farley B, Hu C, Sur M, Tonegawa S. In vivo two-photon imaging reveals a role of arc in enhancing orientation specificity in visual cortex. Cell. 2006;126:389–402. doi: 10.1016/j.cell.2006.06.038. [DOI] [PubMed] [Google Scholar]
- Zou Z, Buck LB. Combinatorial effects of odorant mixes in olfactory cortex. Science. 2006;311:1477–1481. doi: 10.1126/science.1124755. [DOI] [PubMed] [Google Scholar]
- Shepherd JD, Rumbaugh G, Wu J, Chowdhury S, Plath N, Kuhl D, Huganir RL, Worley PF. Arc/Arg3.1 mediates homeostatic synaptic scaling of AMPA receptors. Neuron. 2006;52:475–484. doi: 10.1016/j.neuron.2006.08.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chowdhury S, Shepherd JD, Okuno H, Lyford G, Petralia RS, Plath N, Kuhl D, Huganir RL, Worley PF. Arc/Arg3.1 interacts with the endocytic machinery to regulate AMPA receptor trafficking. Neuron. 2006;52:445–459. doi: 10.1016/j.neuron.2006.08.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rial Verde EM, Lee-Osbourne J, Worley PF, Malinow R, Cline HT. Increased expression of the immediate-early gene arc/arg3.1 reduces AMPA receptor-mediated synaptic transmission. Neuron. 2006;52:461–474. doi: 10.1016/j.neuron.2006.09.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blalock EM, Chen KC, Sharrow K, Herman JP, Porter NM, Foster TC, Landfield PW. Gene microarrays in hippocampal aging: statistical profiling identifies novel processes correlated with cognitive impairment. J Neurosci. 2003;23:3807–3819. doi: 10.1523/JNEUROSCI.23-09-03807.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Small SA, Chawla MK, Buonocore M, Rapp PR, Barnes CA. Imaging correlates of brain function in monkeys and rats isolates a hippocampal subregion differentially vulnerable to aging. Proc Natl Acad Sci USA. 2004;101:7181–7186. doi: 10.1073/pnas.0400285101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dickey CA, Loring JF, Montgomery J, Gordon MN, Eastman PS, Morgan D. Selectively reduced expression of synaptic plasticity-related genes in amyloid precursor protein + presenilin-1 transgenic mice. J Neurosci. 2003;23:5219–5226. doi: 10.1523/JNEUROSCI.23-12-05219.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dickey CA, Gordon MN, Mason JE, Wilson NJ, Diamond DM, Guzowski JF, Morgan D. Amyloid suppresses induction of genes critical for memory consolidation in APP + PS1 transgenic mice. J Neurochem. 2004;88:434–442. doi: 10.1111/j.1471-4159.2004.02185.x. [DOI] [PubMed] [Google Scholar]
- Palop JJ, Chin J, Bien-Ly N, Massaro C, Yeung BZ, Yu GQ, Mucke L. Vulnerability of dentate granule cells to disruption of arc expression in human amyloid precursor protein transgenic mice. J Neurosci. 2005;25:9686–9693. doi: 10.1523/JNEUROSCI.2829-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andra K, Abramowski D, Duke M, Probst A, Wiederhold KH, Burki K, Goedert M, Sommer B, Staufenbiel M. Expression of APP in transgenic mice: a comparison of neuron-specific promoters. Neurobiol Aging. 1996;17:183–190. doi: 10.1016/0197-4580(95)02066-7. [DOI] [PubMed] [Google Scholar]
- Gomori G. Microtechnical demonstration of iron. Am J Pathol. 1936;12:655–663. [PMC free article] [PubMed] [Google Scholar]
- Bondolfi L, Calhoun M, Ermini F, Kuhn HG, Wiederhold KH, Walker L, Staufenbiel M, Jucker M. Amyloid-associated neuron loss and gliogenesis in the neocortex of amyloid precursor protein transgenic mice. J Neurosci. 2002;22:515–522. doi: 10.1523/JNEUROSCI.22-02-00515.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calhoun ME, Wiederhold KH, Abramowski D, Phinney AL, Probst A, Sturchler-Pierrat C, Staufenbiel M, Sommer B, Jucker M. Neuron loss in APP transgenic mice. Nature. 1998;395:755–756. doi: 10.1038/27351. [DOI] [PubMed] [Google Scholar]
- West MJ, Slomianka L, Gundersen HJ. Unbiased stereological estimation of the total number of neurons in thesubdivisions of the rat hippocampus using the optical fractionator. Anat Rec. 1991;231:482–497. doi: 10.1002/ar.1092310411. [DOI] [PubMed] [Google Scholar]
- Gundersen HJ, Bendtsen TF, Korbo L, Marcussen N, Moller A, Nielsen K, Nyengaard JR, Pakkenberg B, Sorensen FB, Vesterby A, West MJ. Some new, simple and efficient stereological methods and their use in pathological research and diagnosis. Apmis. 1988;96:379–394. doi: 10.1111/j.1699-0463.1988.tb05320.x. [DOI] [PubMed] [Google Scholar]
- Calhoun ME, Burgermeister P, Phinney AL, Stalder M, Tolnay M, Wiederhold KH, Abramowski D, Sturchler-Pierrat C, Sommer B, Staufenbiel M, Jucker M. Neuronal overexpression of mutant amyloid precursor protein results in prominent deposition of cerebrovascular amyloid. Proc Natl Acad Sci USA. 1999;96:14088–14093. doi: 10.1073/pnas.96.24.14088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oddo S, Caccamo A, Tran L, Lambert MP, Glabe CG, Klein WL, LaFerla FM. Temporal profile of amyloid-beta (Abeta) oligomerization in an in vivo model of Alzheimer disease. A link between Abeta and tau pathology. J Biol Chem. 2006;281:1599–1604. doi: 10.1074/jbc.M507892200. [DOI] [PubMed] [Google Scholar]
- Chawla MK, Guzowski JF, Ramirez-Amaya V, Lipa P, Hoffman KL, Marriott LK, Worley PF, McNaughton BL, Barnes CA. Sparse, environmentally selective expression of Arc RNA in the upper blade of the rodent fascia dentata by brief spatial experience. Hippocampus. 2005;15:579–586. doi: 10.1002/hipo.20091. [DOI] [PubMed] [Google Scholar]
- Wolfer DP, Stagljar-Bozicevic M, Errington ML, Lipp HP. Spatial memory and learning in transgenic mice: fact or artifact? News Physiol Sci. 1998;13:118–123. doi: 10.1152/physiologyonline.1998.13.3.118. [DOI] [PubMed] [Google Scholar]
- Wahlsten D, Rustay NR, Metten P, Crabbe JC. In search of a better mouse test. Trends Neurosci. 2003;26:132–136. doi: 10.1016/S0166-2236(03)00033-X. [DOI] [PubMed] [Google Scholar]
- Ingram DK, Jucker M. Developing mouse models of aging: a consideration of strain differences in age-related behavioral and neural parameters. Neurobiol Aging. 1999;20:137–145. doi: 10.1016/s0197-4580(99)00033-0. [DOI] [PubMed] [Google Scholar]
- Radde R, Duma C, Goedert M, Jucker M. The value of incomplete mouse models of Alzheimer’s disease. Eur J Nucl Med Mol Imaging. 2008;35 Suppl 1:S70–S74. doi: 10.1007/s00259-007-0704-y. [DOI] [PubMed] [Google Scholar]
- Stalder M, Phinney A, Probst A, Sommer B, Staufenbiel M, Jucker M. Association of microglia with amyloid plaques in brains of APP23 transgenic mice. Am J Pathol. 1999;154:1673–1684. doi: 10.1016/S0002-9440(10)65423-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rolls ET, Kesner RP. A computational theory of hippocampal function, and empirical tests of the theory. Prog Neurobiol. 2006;79:1–48. doi: 10.1016/j.pneurobio.2006.04.005. [DOI] [PubMed] [Google Scholar]