Abstract
The precise role of Leishmania glycoconjugate molecules including phosphoglycans (PGs) and lipophosphoglycan (LPG) on host cellular responses is still poorly defined. Here, we investigated the interaction of Leishmania major LPG2 null mutant (lpg2−), which lacks both PGs and LPG, with dendritic cells (DCs) and the subsequent early immune response in infected mice. Surprisingly, the absence of phosphoglycans did not influence expression pattern of major histocompatibility complex class II (MHC II), CD40, CD80, and CD86 on DCs in vitro and in vivo. However, lpg2− L. major induced significantly higher production of interleukin-12p40 (IL-12p40) by infected bone marrow-derived DCs (BMDCs) than wild-type (WT) parasites in vitro. Furthermore, the production of IL-12p40 by draining lymph node cells from lpg2− mutant-infected mice was higher than those from WT L. major-infected mice. In model antigen presentation experiments, DCs from lpg2− mutant-infected mice induced more gamma interferon (IFN-γ) and IL-2 production by Leishmania-specific T cells than those from WT-infected mice. Lymphocytes isolated from mice infected for 3 days with lpg2− parasites produce similar levels of IFN-γ, but significantly less IL-4 and IL-10 than WT controls. Decreased IL-4 production was also seen in another general PG-deficient mutant lacking the Golgi UDP-galactose transporters (lpg5A− lpg5B−), but not with the lpg1− mutant lacking only LPG, thereby implicating PGs generally in the reduction of IL-4 production. Thus, Leishmania PGs influence host early immune response by modulating DC functions in a way that inhibits antigen presentation and promotes early IL-4 response, and their absence may impact the balance between Th1 and Th2 responses.
Leishmania species are the causative agents of the zoonotic disease leishmaniasis, which is prevalent in six continents and considered endemic in 88 countries. According to the WHO, more than 12 million people are currently suffering from leishmaniasis and a population of over 350 million are at risk of contracting the disease (14, 15). Human cutaneous leishmaniasis is caused by members of the Leishmania major subspecies and is the most common form of the disease. Various forms of pentavalent antimonial components are used for treatment of human leishmaniasis, but treatment failures and drug resistances are common (47, 48). Therefore, there is a critical need for new drugs and drug targets, as well as vaccines.
The outer membrane of Leishmania major is covered by a dense glycocalyx consisting predominantly of lipophosphoglycan (LPG) and other phosphoglycan (PG)-containing molecules, including proteophosphoglycans (PPG), and a heterogeneous group of glycoinositolphospholipids (6, 21). These molecules are thought to be important virulence factors and play key roles in entry, survival, and proliferation of parasites inside host cells (11, 33, 49, 50). In vitro and in vivo studies using purified parasite molecules have identified LPG as a multifunctional virulence factor required for the establishment of infections (6, 43). The roles ascribed to LPG by in vitro testing include inhibition of complement lysis, inhibition of phagolysosome fusion, modulation of macrophage signal transduction pathways, and downregulation of the expression of inducible nitric oxide synthase and synthesis of interleukin-12 (IL-12) (6, 10, 13, 35, 49, 50). However, because of close structural similarities of the glycoconjugate molecules (6, 21), there is a strong potential for cross-activity of shared domains between LPG and other molecules. Moreover, these studies explore LPG in an artificial context, rather than in the natural membranous context during parasite infection (6).
To overcome these limitations, we and others have used targeted gene inactivation to generate mutants which specifically lack the expression of one or more glycoconjugates (7, 20, 43, 45, 55). This allows dissection of the role of these molecules in virulence in the context of the parasite in situ and their influence on the host immune response as well. For example, L. major mutants generated by deleting the LPG1 gene encoding the putative galactofuranosyl transferase involved in the biosynthesis of the LPG core glycan specifically lack LPG (18). The lpg1− L. major mutant shows deficiencies in the initial steps of the infectious cycle, including complement resistance, phagolysosomal fusion, and macrophage survival (43, 44). However, those parasites that go on to survive can form amastigotes that are still virulent in vivo (43), consistent with the observation that amastigotes do not express LPG. In contrast, lpg2− parasites, generated by deleting the LPG2 gene encoding the transporter required for GDP mannose uptake into the Golgi lumen, lack both LPG and other PG-containing molecules (27) and other potential LPG2-dependent metabolites (8). These parasites are highly attenuated in vivo, showing deficiencies in the early steps of promastigote survival, and fail to induce any overt pathology in infected mice (45).
Based on the analysis of known glycoconjugates, the virulence defect of lpg2− L. major has been attributed to its clear PG deficiency by most researchers. However, our recent studies of another PG-deficient L. major mutant, obtained through inactivation of the UDP-Gal transporters encoded by the LPG5A and LPG5B genes (lpg5A− lpg5B−), question this assumption. Unexpectedly, the lpg5A− lpg5B− mutant resembled the lpg1− rather than the lpg2− parasites, by displaying a virulence defect when tested as promastigotes but showing normal virulence as amastigotes (7, 8). This suggests that a loss of LPG2-dependent glycoconjugates, other than PGs, may underlie the virulence defect of lpg2− L. major. These data likewise raise the possibility that PG deficiency alone may not be responsible for alterations in the immune response seen in lpg2− infections (51).
We previously showed that vaccination with lpg2− L. major protects mice against virulent wild-type (WT) challenge (51). In that report, we speculated the protected effect of lpg2− parasites could be related to their differential interaction with cells of the immune system (including dendritic cells [DCs]) early after infection. Here we have investigated the early host innate and adaptive immune responses to lpg2− parasites and compared them to those induced by WT, lpg2− plus LPG2 gene add-back (lpg2 AB), and lpg1− parasites. Our results show that the outcome of the lpg2− parasite interaction with dendritic cells (DCs) and the host early immune response against these mutants are qualitatively and quantitatively different from those of WT L. major. Specifically, we show that although lpg2− L. major infection does not upregulate the expression of costimulatory molecules on DCs, it significantly enhanced their production of IL-12, thereby altering the host early immune response such that the ratio of IL-4 and gamma interferon (IFN-γ) production by the responding T cells is altered. Thus, in response to infection by lpg2− L. major, the antigen-presenting abilities of DCs are skewed toward a more effective Th1 response.
MATERIALS AND METHODS
Mice.
Female 6- to 8-wk-old BALB/c mice were purchased from GMC, Central Animal Care Services, University of Manitoba, or from The Jackson Laboratory. BALB/c ovalbumin (OVA) T-cell receptor (TCR) transgenic mice (DO11.10) were kindly provided by Xi Yang, Department of Medical Microbiology, University of Manitoba. Mice were maintained in a specific-pathogen-free environment at the Central Animal Care Services and were used according to the guidelines stipulated by the Canadian Council on Animal Care.
Parasite strains and infection protocol.
lpg1−, lpg2−, lpg2 AB, and lpg5A− lpg5B− mutants were derivatives of the WT L. major strain LV39 clone 5 (Rho/SU/59/P) and were made by homologous gene targeting as described previously (7, 8, 43, 45). Parasites were cultured at 26°C in M199 medium (HyClone, Logan, UT) supplemented with 10% heat-inactivated fetal bovine serum (FBS) (Cansera, Mississauga, Ontario, Canada), 2 mM l-glutamine, 100 U/ml penicillin, and 100 μg/ml streptomycin (Invitrogen Life Technologies, Burlington, Ontario, Canada). For the lpg1−, lpg2−, and lpg5A− lpg5B− lines, selective drugs were added to the culture media as appropriate for the selective markers used in their construction (puromycin, 15 μg/ml; G418, 15 μg/ml; hygromycin B, 20 μg/ml; blasticidin S, 15 μg/ml).
For infection of mice, stationary-phase promastigotes (7 days postpassage) were labeled with CFSE [5 (6-)-carboxyfluorescein diacetate succinimidyl ester] dye (see below), washed three times in cold phosphate-buffered saline (PBS), and resuspended in PBS at 108/ml and 50 μl containing 5 × 106 parasites was injected into the right hind footpad. In some experiments, mice were infected intradermally in the ear with 5 × 106 parasites resuspended in 10 μl PBS.
DC isolation from infected mice.
After 3 days of infection, mice were sacrificed and CD11c+ DCs were isolated from the draining popliteal lymph nodes (dLNs) of mice infected with CFSE-labeled WT, lpg2−, or lpg2 AB L. major as described previously (4) with some modifications. Briefly, pooled dLNs from six to eight mice were cut into small pieces and digested at 37°C with 1 mg/ml collagenase/dispase (Roche Scientific, Laval, Quebec, Canada) and 50 μg/ml DNase I (Sigma Aldrich, Mississauga, Ontario, Canada) in 1 ml RPMI (Invitrogen) medium containing 2% fetal bovine serum (Cansera). Frequent mixing at room temperature for 20 min was sufficient to disperse the cells, after which 600 μl of 0.1 M EDTA (pH 7.2) was added and stirred continuously for another 5 min to break up DC-T-cell conjugates. Undigested fibrous materials were removed by filtration through a 70-μm-pore cell strainer (Falcon, VWR, Edmonton, Alberta, Canada). The digested lymph node cell suspensions were counted, labeled with anti-CD11c antibody-coated microbeads, and passed through an autoMACS separator (Miltenyi Biotec, Auburn, CA) according to the manufacturer's suggested protocols. The purity of the isolated DCs was between 90 and 95%.
BMDC generation, in vitro infection and stimulation.
Bone marrow cells were isolated from the femur and tibia of WT BALB/c mice. Briefly, after depletion of erythrocytes with ACK lysis buffer (150 μM NH4Cl, 1 μM KHCO3, 0.1 μM Na2EDTA, pH 7.2 to 7.4), the cells were seeded in petri dishes at 2 × 105/ml and differentiated in the presence of recombinant mouse granulocyte-macrophage colony-stimulating factor (20 ng/ml; Peprotech, Indianapolis, IN) at 37°C in a CO2 incubator. The culture media were changed twice (on day 3 and 6), and on day 7, the nonadherent cells (DCs) were collected for in vitro experiments. The purity of the cells was between 85 and 92% (CD11c+ cells).
For infection, bone marrow-derived DCs (BMDCs) were incubated with unlabeled or CFSE-labeled parasites for 5 h at a BMDC/parasite ratio of 1:10. The free parasites were washed away (three washes with complete medium), and infected cells were cultured at 37°C for 24 h. In some experiments, infected BMDCs were stimulated with different concentrations of agonistic stimuli, including anti-CD40 monoclonal antibody (MAb), IFN-γ, and lipopolysaccharide (LPS).
CFSE labeling protocol.
The CFSE labeling protocol has been described previously (26). Briefly, single-cell suspensions from the dLNs were counted and stained with CFSE at 1.25 μM at room temperature in the dark with continuous rocking. After 5 min, staining was quenched with heat-inactivated FBS and the cells were washed, counted, resuspended in complete medium, and used for in vitro cultures. For labeling parasites with CFSE, the stationary parasites were washed three times in PBS, resuspended at 40 × 106/ml in PBS, and mixed with an equal volume of 1.6 μM CFSE for 5 min at room temperature.
DC and T-cell coculture experiments.
T cells were purified from the spleens of DO11.10 mice by positive selection using CD90.2-coated microbeads and the autoMACS separator system according to the manufacturer's suggested protocols (Miltenyi) and labeled with CFSE. The infected BMDCs were pulsed with full-length OVA protein (100 μg/ml) overnight and then cocultured with CFSE-labeled DO11.10 cells at different T-cell/DC ratios (ranging from 5:1 to 1,000:1) for 4 days, and cell proliferation and intracellular IFN-γ production were assessed by flow cytometry. In some experiments, infected DCs were cocultured with a LACK-specific CD4+ T-cell hybridoma expressing I-Ad (28) (LMR7.5; kindly provided by Nicholas Glaichenhaus, Institut National de la Sante et de la Recherche Medicale, Valbonne, France). After 3 days, proliferation and cytokine production were assessed.
In vitro recall response and intracellular cytokine staining.
At various times after infection, dLNs were harvested and made into single-cell suspensions. The cells were washed, resuspended at 4 × 106 cells/ml in complete medium (Dulbecco's modified Eagle's medium supplemented with 10% heat-inactivated FBS, 2 mM glutamine, 100 U/ml penicillin, and 100 μg/ml streptomycin), plated at 1 ml/well in 24-well tissue culture plates (Falcon), and stimulated with soluble leishmanial antigen (SLA; 50 μg/ml) (42). After 72 h, the culture supernatants were collected and stored at −20°C until assayed for cytokines by enzyme-linked immunosorbent assay (ELISA). Some cells were used for intracellular cytokine staining, as previously described (54). Briefly, cells were stimulated with 20 ng/ml phorbol myristate acetate, 1 μM ionomycin, and 10 μg/ml brefeldin A (all from Sigma) for 4 to 6 h before surface staining. Surface-stained cells were washed and fixed with 2% paraformaldehyde and then permeabilized with 0.1% saponin (Sigma) in staining buffer and stained with specific fluorochrome-conjugated MAbs against IFN-γ, IL-4, and IL-10 (all from BD Bioscience, Mississauga, Ontario, Canada). Samples were acquired on either a FACSCalibur or a FACSCanto II flow cytometer (BD Bioscience) and analyzed using CellQuest Pro software (BD Bioscience) or FlowJo software (TreeStar, Ashland, OR).
Cytokine ELISAs.
IL-4, IL-10, IL-12, and IFN-γ concentrations in culture supernatant fluids were determined by sandwich ELISA using the following antibody pairs from BioLegend (San Diego, CA): IL-12p40, C15.6 and C17.8; IL-10, JES5-16E3 and JES5-2A5; IFN-γ, R4-6A2 and XMG1.2; and IL-4, 11B11 and BVD6-24G2. The sensitivities of the ELISA are as follows: IL-4, 15 pg/ml; IL-10, 15 pg/ml; IL-12, 64 pg/ml, and IFN-γ, 30 pg/ml.
Real-time PCR assay.
Three days postinfection, total RNA was extracted from dLN cells using RNA Stat-60 (Tel-Test, Friendswood, TX), according to the manufacturer's specifications and reverse transcribed into cDNA using the RT transcription kit (Gibco-BRL, Gaithersburg, MD). Real-time PCRs were performed with the Smart Cycler II system (Cepheid, Sunnyvale, CA). PCR cycling conditions included 2 min at 50°C, 10 min at 95°C, and then 40 cycles of 5 s at 95°C and 1 min at 60°C. The quantitative PCR was performed according to the manufacturer's protocol using the predeveloped TaqMan assay reagents for murine IL-4, IL-10, IL-12p40, and IFN-γ genes and the SYBR green master mix (SuperArray, Frederick, MD). Differences in starting amount of RNA or cDNA were corrected by normalizing with β-actin RNA (housekeeping gene). Data were analyzed with the Smart Cycler software v2.0 (Cepheid). The results are expressed as the relative increase in gene expression in infected animals over the uninfected ones.
Statistics.
Results are shown as means ± standard errors. Results from each group were compared using Student's t test. A P value of <0.05 was considered significant.
RESULTS
WT and lpg2− L. major induce similar pattern of expression of costimulatory molecules but differential IL-12 production in BMDCs.
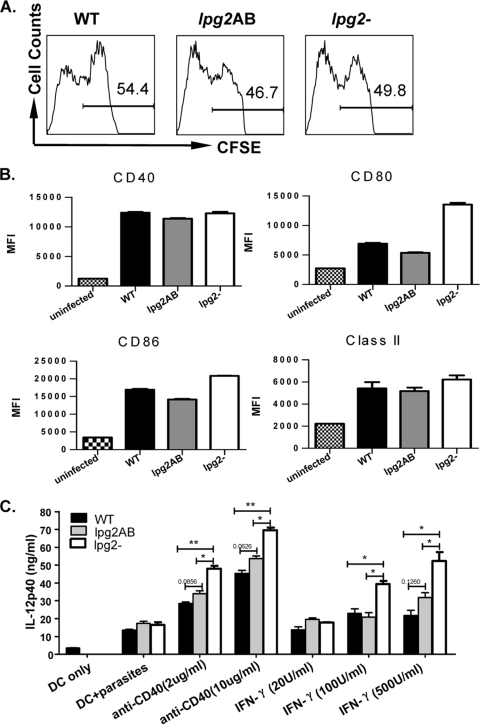
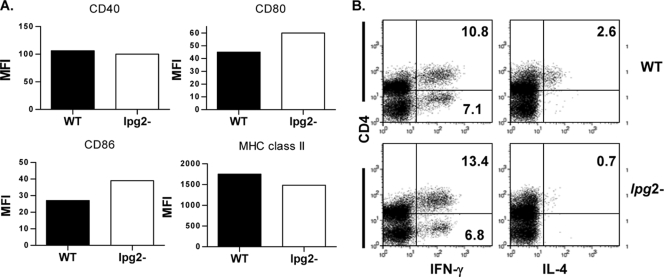
We previously showed that similar to WT, lpg2− L. major parasites can invade macrophages but are progressively destroyed beyond 24 h (45). DCs, which are among the first cells infected by Leishmania (3, 40), are the most efficient antigen-presenting cells and hence are essential for inducing an efficient anti-Leishmania adaptive immune response (41). To investigate the possible effect of LPG2-dependent molecules on DC function, we generated BMDCs and determined their expression of costimulatory molecules after infection with WT and lpg2− L. major. In our initial experiments, at all times tested, there was no difference in the expression (percent expression and mean fluorescence intensities [MFI]), of major histocompatibility complex (MHC) class II, CD40, CD80, and CD86 molecules by BMDCs infected with WT or lpg2− organisms (data not shown). However, because the infectivity of the DCs in our system was low (∼47 to 54% for WT, lpg2 AB, and lpg2− parasites) (Fig. 1A), we considered the possibility that moderate changes in expression of these molecules by infected cells may be masked by normal or enhanced expressions in uninfected cells. Therefore, we infected the cells with CFSE-labeled parasites and analyzed the expression of costimulatory molecules on only infected (CFSEhigh) DCs. The percentages of WT-, lpg2 AB-, or lpg2−-infected (CFSEhigh) DCs were comparable (Fig. 1A), suggesting that the loss of cell surface glycoconjugates did not affect uptake of lpg2− parasites by DCs. By focusing only on infected DCs, there was still no difference in expression (MFI) of these molecules by WT- and lpg2− L. major-infected BMDCs (Fig. 1B). These data suggest that expression of LPG2-dependent glycoconjugates does not greatly affect the expression of costimulatory molecules on DCs.
FIG. 1.
L. major glycoconjugate molecules do not affect the expression of costimulatory molecules but influence IL-12 production by DCs. (A) BMDCs were infected with CFSE-labeled WT, lpg2 AB and lpg2-deficient (lpg2−) L. major stationary-phase promastigotes (BMDC/parasite ratio, 1:10), and the rate of infection was determined after 24 h by gating on infected cells (CD11c+ CFSE+ cells). The expression of MHC class II, CD40, CD80, and CD86 on infected (CFSE+) or on total (CD11c+) DCs was determined by flow cytometry. Data are presented as percent MFI of gated infected cells (B). Some uninfected and infected BMDCs were stimulated with various concentrations of anti-CD40 and IFN-γ for 24 h, and the culture supernatant fluids were collected and assayed for IL-12p40 by ELISA (C). Data presented are representative of three independent experiments with similar results. *, P < 0.05; **, P < 0.01.
IL-12 produced by DCs also plays a critical role in the development of CD4+ Th1 cells and resistance to L. major infection in mice (24, 31, 34, 41). Thus, we measured the production of IL-12p40 by BMDCs after 24 h of infection. lpg2− L. major-infected BMDCs produced significantly (P < 0.05 to 0.01) more IL-12p40 than those infected with WT parasites following stimulation with several IL-12-inducing agonists, including the anti-CD40 MAb, IFN-γ, and LPS (Fig. 1C) (data not shown). Cells infected with lpg2 AB parasites showed a pattern of cytokine production similar to those infected with WT parasites (Fig. 1C). Interestingly, we were unable to detect significant levels of IL-12p70 subunit by ELISA despite the high levels of IL-12p40 subunit, as has been previously reported (2, 17). Taken together, these results indicate that although infection with lpg2− L. major did not lead to significant alteration in the expression of costimulatory molecules on DCs, they are capable of positively enhancing their secretion of IL-12p40, a critical Th1-inducing cytokine.
Freshly isolated (ex vivo) DCs from WT- and lpg2− mutant-infected mice express similar levels of costimulatory molecules.
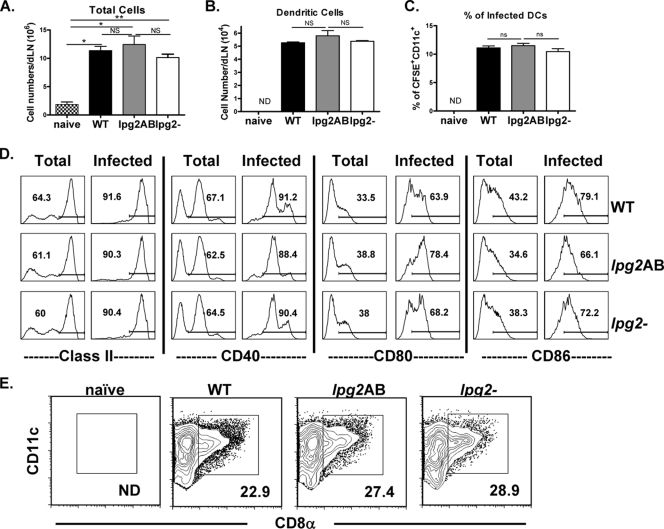
Since we were unable to demonstrate differential effects of WT and lpg2− parasites on the expression of costimulatory molecules on infected DCs in vitro, we asked whether similar effects occurred in vivo. We infected mice in the footpad with CFSE-labeled WT, lpg2 AB, and lpg2− L. major parasites and after 3 days purified DCs from pooled popliteal lymph nodes draining the infection site (dLNs) and analyzed their numbers, subsets, and expression of costimulatory molecules. Labeling with CFSE allowed us to determine the percentages of infected DCs and also to focus our assessment of costimulatory molecule expression on only these infected cells. As expected, infection with both parasite lines led to significant increase in the number of cells recovered from the dLN (Fig. 2A). However, there was no significant difference in the total numbers of cells (Fig. 2A), CD11c+ (Fig. 2B) or CFSEhigh (i.e., infected) (Fig. 2C) DCs recovered from the dLNs of mice infected with either parasite line, suggesting that the deficiency of PGs does not impair the early inflammatory response, migration, and/or expansion of cells (including DCs) into the dLNs. Consistent with the in vitro findings, we found no difference in the expression of costimulatory molecules (MHC II, CD40, CD80, and CD86) by directly ex vivo total or infected (CFSEhigh) DCs isolated from mice infected with WT, lpg2 AB, or lpg2− parasites (Fig. 2D). Because the relative expression of CD8α on DCs may affect their ability to present Leishmania antigens and activate Leishmania-specific T cells (19, 40), we determined whether infection with lpg2− affected the expression of this subpopulation of DCs. There was no difference in percentage or MFI (Fig. 2E) (data not shown) of CD8α-positive DCs isolated from dLNs of lpg2− and WT L. major-infected mice. Together with the in vitro data, these results show that infection with lpg2− L. major does not significantly alter the expression of costimulatory molecules on DCs relative to that seen with WT L. major.
FIG. 2.
The glycoconjugate molecules of Leishmania major do not modulate DC activation in vivo. BALB/c mice infected in the footpad with CFSE-labeled WT, lpg2 AB, or lpg2− (lpg2 knockout) L. major parasites were sacrificed at 3 days, the dLNs were collected and then digested with collagenase, and the total number of cells was enumerated by direct cell count using a hemocytometer (A). DCs were isolated from pooled dLN cells by positive selection using autoMACS column, and stained routinely with fluorochrome-labeled anti-CD11c MAb and other various cell surface markers (CD40, CD80, CD86, and MHC II). The stained cells were analyzed by flow cytometry to enumerate the total number of DCs (i.e., CD11c+ cells in panel B) or percentage of infected cells (i.e., CFSEhigh CD11c+ cells in panel C). The expression of MHC class II, CD40, CD80, and CD86 molecules (D) and CD8α molecules (E) on both infected (CFSEhigh) and total DCs were analyzed after gating on CD11c+ cells. Data presented are representative of three (A to C) and two (D) independent experiments with similar results. ND, not done. *, P < 0.05; **, P < 0.01.
DCs from lpg2− L. major-infected mice are more efficient antigen-presenting cells than those from WT L. major-infected mice.
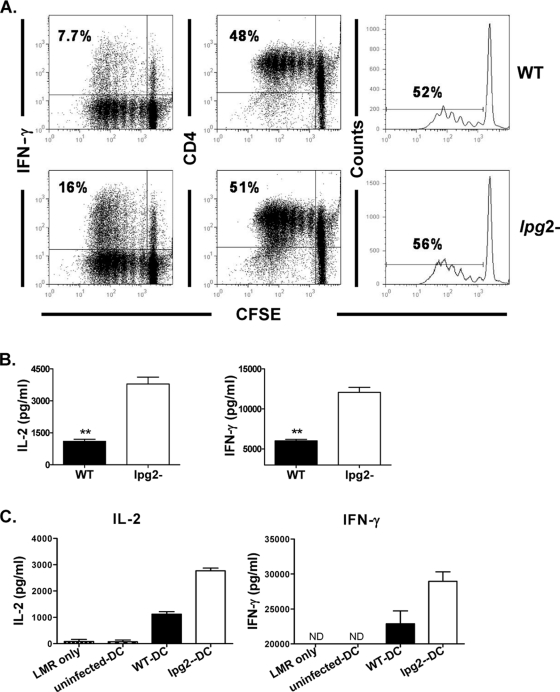
IL-12 is a critical cytokine that skews the development of naïve CD4+ T cells into Th1 cells. To determine if the higher IL-12 production by lpg2−-infected DCs affects their ability to activate naïve T cells, we pulsed freshly isolated DCs from 3-day lpg2−- and WT L. major-infected mice with whole-length OVA protein overnight and then cocultured them in vitro with CFSE-labeled OVA-specific (DO11.10) T cells for 4 days. As shown in Fig. 3A and B, DCs from mice infected with lpg2− L. major stimulated OVA-specific T cells to produce larger amounts of IL-2 and IFN-γ than those from WT-infected controls. Interestingly and consistent with lack of differences in the expression of costimulatory molecules (Fig. 1B and 2D), there was no significant difference in antigen-specific proliferation of OVA-specific T cells following presentation with OVA by DCs from mice infected with either parasite (Fig. 3A).
FIG. 3.
More efficient induction of IL-2 and IFN-γ responses by DCs from lpg2− L. major-infected mice. DCs isolated from BALB/c mice infected for 3 days with WT or lpg2− L. major were pulsed overnight with full-length OVA protein (100 μg/ml) and then cocultured with purified CFSE-labeled CD4+ T cells from DO11.10 transgenic mice. After 4 days, proliferation and IFN-γ production were determined by flow cytometry (A). The culture supernatant fluids were collected and assayed for IL-2 and IFN-γ by ELISA (B). Some purified DCs were cocultured with a LACK-specific T-cell hybridoma (LMR7.5) for 72 h, and the culture supernatant fluids were assayed for IL-2 and IFN-γ by ELISA (C). The data presented are representative of three (A and B) and two (C) independent experiments (n = 4 to 5 mice/group in panels A and B) with similar results. ND, not done. *, P < 0.05; **, P < 0.01.
To determine the relevance of the enhanced IFN-γ production by DO11.10 T cells following OVA presentation by freshly isolated DCs from lpg2−-infected mice, we compared the ability of freshly isolated DCs from WT- or lpg2−-infected mice to activate the Leishmania-specific T-cell hybridoma, LMR7.5, in vitro (9). LMR7.5 cells cocultured with DCs from lpg2−-infected mice produced more IL-2 and IFN-γ than those stimulated with cells from WT-infected mice (Fig. 3C). Taken together, these results indicate that the absence of glycoconjugate molecules influences the expression of costimulatory molecules and in the process modulates the antigen-presenting functions of DCs.
L. major glycoconjugates regulate the host early immune response.
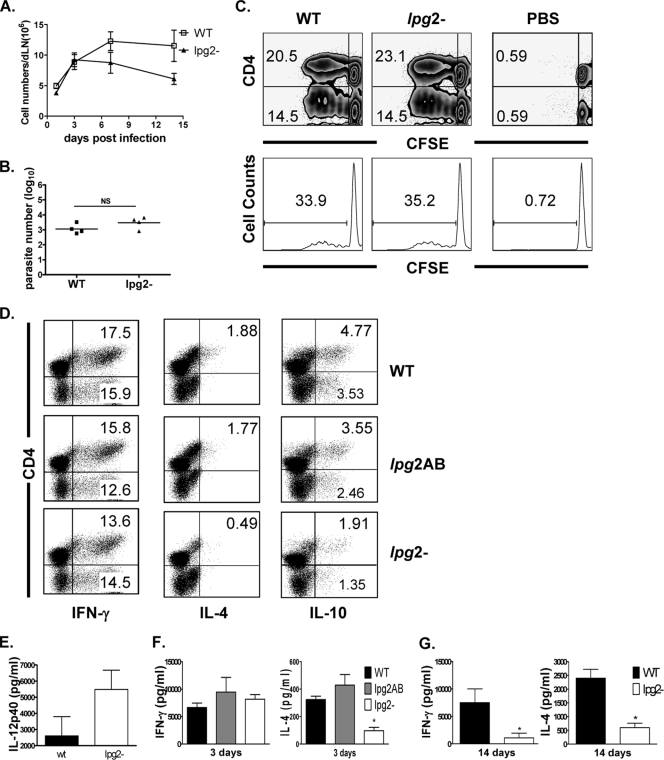
We previously showed that despite parasite persistence, the effector T-cell response in mice infected with lpg2− parasites for 4 weeks is weak (51). This suggests that the early immune response in lpg2−-infected mice may be defective. Here we examined the early immune response induced by lpg2−, lpg2 AB, and WT parasites in the first few days postinfection. The numbers of cells in the dLNs of mice infected with lpg2− parasites increased at 24 h, reaching a peak at 3 days postinfection, and were comparable to those seen in mice infected with WT. However, from day 7 postinfection, the cell numbers began to decline in lpg2−-infected mice, while the number of cells in dLNs from WT-infected mice was sustained (Fig. 4A). Interestingly and consistent with similar infectivity seen in DCs (Fig. 2C), similar numbers of WT and lpg2− parasites were recovered at the site of infection on days 3 and 7 postinfection (Fig. 4B) (data not shown), suggesting that lpg2− parasites were not rapidly destroyed by the innate immune system.
FIG. 4.
Modulation of the host early immune response by L. major glycoconjugate molecules. BALB/c mice infected in the footpad with WT, lpg2 AB, and lpg2− L. major were sacrificed at the indicated days, and the number of cells in the dLN was enumerated by direct cell count (A). Parasite burden in the infected footpad was determined by limiting dilution (B). Some cells were labeled with CFSE dye and stimulated in vitro with SLA (50 μg/ml) for 3 days, and proliferation was assessed by flow cytometry (C). After 3 days of culture, some cells were stained for intracellular production/expression of IFN-γ, IL-4, and IL-10 and analyzed by flow cytometry (D). The cell culture supernatants were collected and assayed for cytokines (IL-12p40, IL-4, and IFN-γ) by ELISA (E to G). The data presented are representative of three to five independent experiments (n = 3 to 5 mice/group) with similar results. *, P < 0.05.
Next, we determined the early antigen-specific cytokine production by dLNs following stimulation with SLA. Although the levels of antigen-specific proliferation of dLN cells from lpg2− and WT L. major-infected mice were comparable at 3 days postinfection (Fig. 4C), similar to that seen by ex vivo DC stimulation of DO11.10 T cells (Fig. 3A), cytokine production was dramatically different. Intracellular cytokine staining showed that the numbers of IFN-γ-producing cells were comparable, whereas the numbers of IL-4-producing (and to a lesser degree IL-10-producing) cells in the dLNs from mice infected with lpg2− were lower than those from WT-infected mice (Fig. 4D). Interestingly, most of the IL-4-producing cells also coproduced IL-10 (data not shown), an important disease-promoting cytokine in experimental cutaneous leishmaniasis. As expected and consistent with our previous report (51), the pattern of cytokine production by dLN cells from mice infected with lpg2− parasites complemented with the LPG2 gene (lpg2 AB) was similar to those infected with WT parasites (Fig. 4D), indicating that decreased IL-4 response by lpg2− parasites is related to deficiency of PGs and not a result of the genetic manipulation processes. This pattern of intracellular cytokine response (normal IFN-γ and low IL-4 in lpg2−-infected mice) was also confirmed by ELISA (Fig. 4F and G). Furthermore, cells from mice infected with lpg2− parasites produced more IL-12p40 than those from WT L. major-infected mice (Fig. 4E). By day 14 postinfection, the production of IFN-γ and IL-4 was dramatically reduced in lpg2−-infected mice (Fig. 4F and G). This sharp decline in cytokine production parallels the decline in parasite numbers in the footpads of lpg2−-infected mice (data not shown), suggesting that parasite number is directly correlated with the magnitude of immune response in infected mice.
As the route of infection with L. major influences the nature of the immune response (5, 37), we investigated whether modulation of DC function and early host immune response by lpg2− L. major is a general phenomenon that is independent of route of infection. Therefore, we assessed the early immune response in mice infected intradermally in the ear with lpg2− and WT L. major. As shown in Fig. 5A and B, the pattern of DC activation and early cytokine response was similar to that observed after subcutaneous (footpad) infection, such that cells from lpg2−-infected mice expressed equivalent (MHC II, CD40, and CD86) or slightly higher (CD80) levels of costimulatory molecules and produced less IL-4 than those from WT-infected mice. Taken together, these results show that infection with lpg2− parasites alters the host early immune response toward a more robust Th1 phenotype.
FIG. 5.
The immunomodulatory effect of L. major glycoconjugate molecules is independent of site of infection. BALB/c mice infected in the ear (intradermal) with 5 million WT or lpg2− L. major parasites were sacrificed after 3 days, and CD11c+ DCs were purified from pooled dLN cells; stained for surface expression of MHC class II, CD40, CD80, and CD86 molecules; and analyzed by flow cytometry (A). Some unfractionated dLN cells were stimulated with SLA for 3 days, and the percentage of IFN-γ- and IL-4-producing cells was determined by intracellular cytokine staining (B). The data presented are representative of two independent experiments (n = 4 to 5 mice/group) with similar results.
Deficiency of PGs may contribute to the differences in the early immune response in mice infected with lpg2− L. major.
As summarized in the introduction, lpg2− parasites lack all PGs, including those attached to LPG and PPGs, which exist in both secreted and membrane-bound forms throughout the parasite infectious cycle. However, recent data showed that PG deficiency is not responsible for the amastigote virulence defects of the lpg2− mutant (8), likewise raising the possibility that PG deficiency alone may not be responsible for alterations in the early immune response seen in lpg2− infections. To further probe the nature of the molecule(s) responsible for the LPG2-dependent effects on DCs in vitro and in vivo (seen above), we made use of additional L. major mutants deficient in various PGs through inactivation of other LPG genes and differing in virulence from WT or lpg2− parasites.
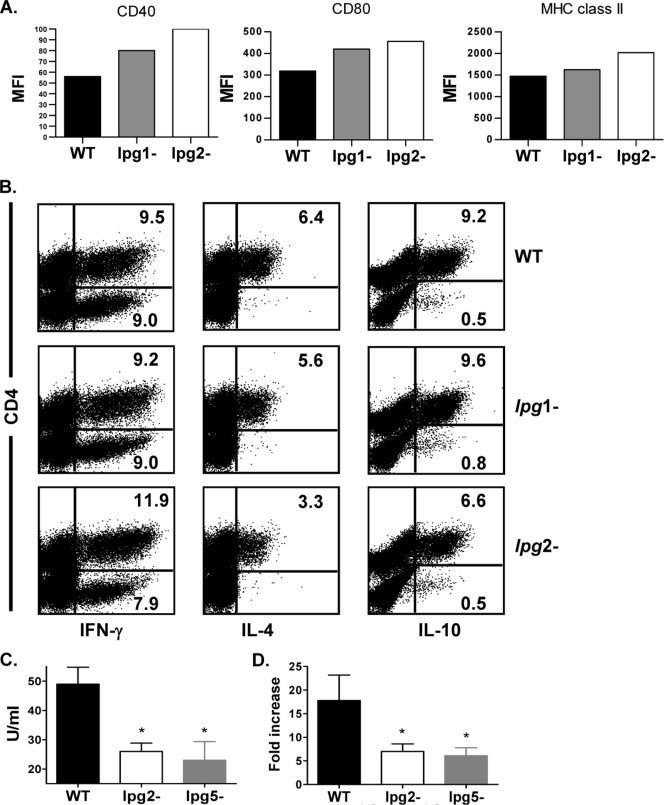
lpg1−-deficient parasites specifically lack LPG but maintain WT levels of other PGs such as PPG (43). In vivo, parasites involved in lpg1− infection show defects in the initial phases of macrophage establishment, but parasites that survive go on to form fully virulent amastigotes are able to cause progressive lesions in susceptible BALB/c mice (43). As shown in Fig. 6A, there was no significant alteration in the expression of costimulatory molecules on DCs from mice infected for 3 days with WT, lpg1−, or lpg2− L. major. In contrast, whereas the early cytokine responses induced by lpg1− parasites were similar to those of WT parasites (no decrease in IL-4 and IL-10 responses), the frequencies of IL-4- and IL-10-producing cells from mice infected with lpg2− parasites were reduced (Fig. 6B), consistent with previous results (Fig. 4D and 5B). Since the effects of lpg1− parasites in vitro and in vivo were similar to those of WT parasites, it was not necessary to investigate the effects of lpg1 AB parasites in these studies.
FIG. 6.
Differences in the early immune response in mice infected with lpg2− L. major are unrelated to absence of LPG. BALB/c mice were infected in the footpad with 5 million WT, lpg1−, and lpg2− L. major stationary-phase promastigotes. After 3 days, mice were sacrificed and CD11c+ cells (DCs) were isolated from pooled dLN cells; stained for surface expression of MHC class II, CD40, and CD80 molecules; and analyzed by flow cytometer (A). Some unfractionated dLN cells were also stimulated with SLA for 3 days, and the percentage of IFN-γ-, IL-4-, and IL-10-producing cells was determined by intracellular cytokine staining (B). In some experiments, dLN cells from mice infected with WT, lpg2−, and lpg5A− lpg5B− L. major for 3 days were cultured for 72 h, and the culture supernatant fluid was assayed for IL-4 by ELSA (C). Total cellular RNA was isolated from some dLN cells and quantified by real-time PCR (D). Data presented are representative of two independent experiments (n = 3 to 5 mice/group) with similar results. *, P < 0.05.
To confirm whether the decreased IL-4 response following infection with lpg2− L. major was due to lack of PGs or related to PG-associated loss of virulence, we examined IL-4 production in infections by the lpg5A− lpg5B− L. major mutant (lpg5−), which lacks all PGs due to inactivation of the parasite's Golgi UDP-Gal transporters (7), yet show virulent characteristics similar to lpg1− L. major (7, 8). As shown in Fig. 6C and D, both IL-4 message and protein were suppressed two- to threefold in mice infected with lpg2− and lpg5− L. major, suggesting that the absence of PG molecules and not lack of virulence was primarily responsible for the decrease in IL-4 response in mice infected with lpg2− L. major.
DISCUSSION
The primary aim of this study was to determine whether there are differences in the early immune responses to WT and lpg2− L. major that could help account for the protection observed in mice vaccinated with lpg2− parasites (51). We investigated the early immune response and DC functions following infection with WT and lpg2− L. major. First we focused on the expression of costimulatory molecules and antigen presentation by DCs. We found that WT and lpg2− L. major induced comparable expression of costimulatory molecules on infected DCs in vitro and in vivo. After 3 days, infection with both WT and lpg2− parasites did not lead to appreciable changes in the expression of MHC class II and other costimulatory molecules, including CD40, CD80, and CD86 on total DCs isolated from the dLNs of infected mice. However, these molecules were upregulated equally on the minor population (∼10%) of infected DCs, suggesting that LPG2-dependent PG-containing molecules do not alter the expression of costimulatory molecule expression on DCs. Interestingly, infection with the specifically LPG-deficient lpg1− mutant also failed to induce any significant changes in the expression of costimulatory molecules on DCs (Fig. 6A). Quantitative studies show that in promastigotes, LPG constitutes well over 90% of the parasite PGs in the L. major line studied here (43). The present study suggests that neither LPG- nor PG-containing molecules play any significant role in altering the expression of costimulatory molecules on DCs.
Functional antigen presentation studies showed that lpg2−-infected DCs exhibited an increased ability to present a test antigen (OVA) relative to those infected with WT parasites as manifested by increased production of IL-2 and IFN-γ by OVA-specific T cells. Since infection with lpg2− L. major did not alter the expression of costimulatory molecules on infected DCs, the results suggest that the increased Th1 response following lpg2− L. major infection may not be related to a more efficient T-cell activation resulting from interaction of more costimulatory molecules with their ligands, which leads to stronger signals on T cells. However, it is conceivable that altered T-cell response could also occur from altered antigen presentation that could have arisen through other LPG2-dependent effects such as transient delay in the formation of fusogenic phagolysosomes (10, 12, 13). However, because full-length OVA used in our studies should be taken up mostly by endocytosis, it is likely that altered phagolysosomal fusion in lpg2− mutant-infected cells may not have significantly affected the rate of antigen processing in this system. Therefore, the exact mechanism through which DCs from lpg2−-infected mice enhanced IFN-γ response without affecting proliferation remains unknown.
Previous reports showed that the LACK antigen is mostly released, processed, and presented to T cells by macrophages only following internalization of parasites with reduced infectivity that are subsequently killed (9, 38). This raises the possibility that the increased presentation of LACK observed by DCs from lpg2−-infected mice could reflect a superior killing of the mutant parasites. However, we were only able to recover about 1% of the infecting parasite dose (close to ∼5 × 104) (Fig. 4B) after 3 days postinfection with WT and lpg2− parasites, indicating that greater than 95% of the infecting parasites are killed within few hours or days of infection. This high and similar rate of early destruction of WT and lpg2− parasites suggests that levels of release of LACK in mice infected with WT and lpg2− parasites may be similar. Therefore, it is unlikely that the increased presentation of LACK and induction of higher IFN-γ and IL-2 production by lpg2− mutant-infected DCs over WT-infected cells are related to faster killing of lpg2− parasites after infection. Another important consideration is that differences between WT and lpg2− parasites could reflect differences in infectivity since unselected stationary-phase promastigotes used here may contain different numbers of infective metacylic promastigotes. However, given that our studies and analyses concentrated on the early phase of infection (3 days postinfection), when there was no difference in both the percentage of infected DCs and parasite burden in WT- and lpg2− mutant-infected mice (Fig. 2B and 4B), we believe the use of unselected parasites in these studies has not significantly affected the results and their interpretation.
A previous report showed that LPG of L. major negatively modulates the expression of costimulatory molecules on Langerhan cells (a subtype of DCs) and inhibits their migratory activities (36). However, since this report was based on studies using purified LPG, its biological relevance is difficult to assess. In contrast, a recent report suggests a critical role for Leishmania mexicana LPG in the upregulation of costimulatory molecules (CD40, MHC class II, and CD80) on DCs (1). Interestingly, the complementary add-backs were unable to restore the WT phenotype on infected cells. We found that like WT L. major, both lpg1− and lpg2− L. major infections of DCs altered the expression of costimulatory molecules to the same extent over basal levels seen in uninfected cells. These differences in observation may be related to the use of different Leishmania species (L. mexicana versus L. major). For instance, the ability of Leishmania to induce IL-12 production by DCs has been shown to vary depending on the parasite species (16, 29, 30). Furthermore, while lpg2− L. major parasites are highly attenuated (44, 45), lpg2− L. mexicana parasites retain amastigote virulence (20). It is conceivable that different species of Leishmania may use different mechanisms to subvert the host immune response. Interestingly, infection of DCs with lpg2− L. major significantly enhanced their production of IL-12p40 in response to various DC-stimulating agonists (Fig. 1C). Thus, while the glycoconjugate molecules of L. major do not influence DC expression of costimulatory molecules, they may act to prevent their effective antigen presentation capacity (in an as yet undetermined way), thereby affecting their effector Th1 cell-inducing abilities.
In addition to antigen processing and presentation, DCs also influence the nature of immune response by producing cytokines including IL-12. While in vitro infection of DCs with WT L. major resulted in minimal production of IL-12p40 over basal levels, infection with lpg2− L. major resulted in a more than 180% increase in IL-12p40 production (Fig. 1C). This difference in IL-12 production between WT and lpg2− L. major-infected DCs persisted following stimulation with various stimuli, including anti-CD40, IFN-γ, and LPS (Fig. 1C) (data not shown). Similar effects were seen in vivo, where IL-12p40 production by cells from the dLNs of mice infected with lpg2− L. major was higher than those from infected with WT parasites (Fig. 4F). Despite the high production of the IL-12p40 subunit, we were unable to detect measurable amounts of the bioactive IL-12p70, as has been reported by others (2, 17). Since the p40 subunit is common to both IL-12 and IL-23, we could not rule out the possibility that some of the molecules detected in this study are components of IL-23. Consistent with the DC findings, cells from the dLN of mice infected for 3 days with lpg2− parasites produced smaller amounts of macrophage-deactivating cytokines (IL-4/IL-10) than those from WT-infected mice (Fig. 4D, 5B, and 6B and C). Interestingly, infection with LPG-deficient lpg1− parasites did not lead to reduction in IL-4 production by dLN cells (Fig. 6B), whereas infection with PG-deficient lpg5A− lpg5B− parasites (which lack PGs similar to lpg2− parasites) also led to reduction of the early IL-4 response in the dLN (Fig. 6C and D). Surprisingly, the reduced IL-4 response in mice infected with lpg2− parasites was not associated with a concomitant increase in IFN-γ production, a cytokine that has been shown to suppress the production of IL-4 by T cells (32). Wang et al., (53) showed that IL-12 could directly downregulate IL-4 in the absence of IFN-γ. Thus, it is possible that higher IL-12 production by cells from lpg2−-infected mice directly suppress IL-4 response, in an IFN-γ-independent manner. These results suggest that the PGs play important role in regulating early IL-4 and IL-12p40 responses following infection with L. major. It should be emphasized that these data do not completely rule out a role for LPG in this process; however, if active, its role is fully redundant with that of other PGs unaffected in the lpg1− mutant. Furthermore, we do not know whether the reduced IL-4 levels in cultures of cells from lpg2− L. major-infected mice was related to suppression or inhibition of IL-4 production in the absence of PG-containing molecules.
The contributions of PGs and other LPG2-dependent molecules in modulating the host immune response, particularly in the induction of protective IFN-γ and pathogenic IL-4 responses, have not been investigated. The early immune response to L. major in mice is dominated by a unique population of Vβ4 Vα8 CD4+ T cells recognizing the immunodominant epitope of LACK protein (22, 23, 25, 46). These MHC class II-restricted cells rapidly expand in the dLNs, accumulate IL-4 transcripts within hours after infection, and secrete high levels of this cytokine in vivo and in vitro (22, 25). The reduction in IL-4 production in mice infected with lpg2− and lpg5A− lpg5B− parasites closely mimics the failure to produce an early IL-4 burst in mice made tolerant to LACK by transgenic expression (23). Thus, it is tempting to suggest that the PG-containing molecules of L. major influence the host immune response to LACK protein. Recently, LACK TCR transgenic (ABLE) mice on a BALB/c background (39) were developed and used to study the T-cell response to LACK protein (52). These mice will be valuable for studying the influence of PG-containing molecules on the induction of Vβ4 Vα8 CD4+ T cells and early IL-4 burst during L. major infection.
Overall, our results show that PGs and/or LPG2-dependent glycoconjugate molecules of L. major are responsible for the reduction in the production of IL-12p40 by infected DCs and the consequent induction of increased Th1 cell response. Thus, in addition to their role in subverting macrophage function, the glycoconjugates of Leishmania major influence the initial pathways that lead to effective T-cell activation, thereby ensuring optimal survival of the parasites in infected host. We speculate that reduction of IL-4 (and to some extent IL-10) response in mice infected with lpg2− L. major could lead to a more efficient IFN-γ-mediated activation of macrophages and DCs, leading to rapid early parasite control. The protection induced by lpg2− parasites is striking despite the absence of a large IFN-γ response (51). Our results suggest that one reason underlying this phenomenon is the possibility of a heightened early immune response to the lpg2− parasites due to the absence of PGs and LPG, in conjunction with the inability of this parasite to induce lesion pathology.
Acknowledgments
We thank Michael Mandel and Abdelilah Soussi-Gounni for critically reading the manuscript and making useful suggestions.
This work was supported by grants from the Canadian Institutes for Health Research (CIHR), Canadian Foundation for Innovation (CFI), and Manitoba Health Research Council (MHRC [to J.E.U.]) and National Institutes of Health grants NIH AI31078 (to S.M.B.) and NIH AI059396 (to P.S.). J.E.U. is a recipient of the CIHR New Investigator Award. D.L. is a recipient of the MHRC studentship.
The authors have no financial conflict of interest.
Editor: J. F. Urban, Jr.
Footnotes
Published ahead of print on 1 June 2009.
REFERENCES
- 1.Aebischer, T., C. L. Bennett, M. Pelizzola, C. Vizzardelli, N. Pavelka, M. Urbano, M. Capozzoli, A. Luchini, T. Ilg, F. Granucci, C. C. Blackburn, and P. Ricciardi-Castagnoli. 2005. A critical role for lipophosphoglycan in proinflammatory responses of dendritic cells to Leishmania mexicana. Eur. J. Immunol. 35476-486. [DOI] [PubMed] [Google Scholar]
- 2.Antunez, M. I., and R. L. Cardoni. 2001. Early IFN-gamma production is related to the presence of interleukin (IL)-18 and the absence of IL-13 in experimental Trypanosoma cruzi infections. Immunol. Lett. 79189-196. [DOI] [PubMed] [Google Scholar]
- 3.Ato, M., A. Maroof, S. Zubairi, H. Nakano, T. Kakiuchi, and P. M. Kaye. 2006. Loss of dendritic cell migration and impaired resistance to Leishmania donovani infection in mice deficient in CCL19 and CCL21. J. Immunol. 1765486-5493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Baldwin, T., S. Henri, J. Curtis, M. O'Keeffe, D. Vremec, K. Shortman, and E. Handman. 2004. Dendritic cell populations in Leishmania major-infected skin and draining lymph nodes. Infect. Immun. 721991-2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Belkaid, Y., E. Von Stebut, S. Mendez, R. Lira, E. Caler, S. Bertholet, M. C. Udey, and D. Sacks. 2002. CD8+ T cells are required for primary immunity in C57BL/6 mice following low-dose, intradermal challenge with Leishmania major. J. Immunol. 1683992-4000. [DOI] [PubMed] [Google Scholar]
- 6.Beverley, S. M., and S. J. Turco. 1998. Lipophosphoglycan (LPG) and the identification of virulence genes in the protozoan parasite Leishmania. Trends Microbiol. 635-40. [DOI] [PubMed] [Google Scholar]
- 7.Capul, A. A., T. Barron, D. E. Dobson, S. J. Turco, and S. M. Beverley. 2007. Two functionally divergent UDP-Gal nucleotide sugar transporters participate in phosphoglycan synthesis in Leishmania major. J. Biol. Chem. 28214006-14017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Capul, A. A., S. Hickerson, T. Barron, S. J. Turco, and S. M. Beverley. 2007. Comparisons of mutants lacking the Golgi UDP-galactose or GDP-mannose transporters establish that phosphoglycans are important for promastigote but not amastigote virulence in Leishmania major. Infect. Immun. 754629-4637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Courret, N., E. Prina, E. Mougneau, E. M. Saraiva, D. L. Sacks, N. Glaichenhaus, and J. C. Antoine. 1999. Presentation of the Leishmania antigen LACK by infected macrophages is dependent upon the virulence of the phagocytosed parasites. Eur. J. Immunol. 29762-773. [DOI] [PubMed] [Google Scholar]
- 10.Cunningham, A. C. 2002. Parasitic adaptive mechanisms in infection by leishmania. Exp. Mol. Pathol. 72132-141. [DOI] [PubMed] [Google Scholar]
- 11.Descoteaux, A., and S. J. Turco. 1999. Glycoconjugates in Leishmania infectivity. Biochim. Biophys. Acta 1455341-352. [DOI] [PubMed] [Google Scholar]
- 12.Desjardins, M., and A. Descoteaux. 1997. Inhibition of phagolysosomal biogenesis by the Leishmania lipophosphoglycan. J. Exp. Med. 1852061-2068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Desjardins, M., and A. Descoteaux. 1998. Survival strategies of Leishmania donovani in mammalian host macrophages. Res. Immunol. 149689-692. [DOI] [PubMed] [Google Scholar]
- 14.Desjeux, P. 2004. Leishmaniasis. Nat. Rev. Microbiol. 2692. [DOI] [PubMed] [Google Scholar]
- 15.Desjeux, P. 2004. Leishmaniasis: current situation and new perspectives. Comp. Immunol. Microbiol. Infect. Dis. 27305-318. [DOI] [PubMed] [Google Scholar]
- 16.Ghosh, M., and S. Bandyopadhyay. 2004. Interaction of Leishmania parasites with dendritic cells and its functional consequences. Immunobiology 209173-177. [DOI] [PubMed] [Google Scholar]
- 17.Hensler, H. R., G. Rappocciolo, C. R. Rinaldo, and F. J. Jenkins. 2009. Cytokine production by human herpesvirus 8-infected dendritic cells. J. Gen. Virol. 9079-83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Huang, C., and S. J. Turco. 1993. Defective galactofuranose addition in lipophosphoglycan biosynthesis in a mutant of Leishmania donovani. J. Biol. Chem. 26824060-24066. [PubMed] [Google Scholar]
- 19.Iezzi, G., A. Frohlich, B. Ernst, F. Ampenberger, S. Saeland, N. Glaichenhaus, and M. Kopf. 2006. Lymph node resident rather than skin-derived dendritic cells initiate specific T cell responses after Leishmania major infection. J. Immunol. 1771250-1256. [DOI] [PubMed] [Google Scholar]
- 20.Ilg, T., M. Demar, and D. Harbecke. 2001. Phosphoglycan repeat-deficient Leishmania mexicana parasites remain infectious to macrophages and mice. J. Biol. Chem. 2764988-4997. [DOI] [PubMed] [Google Scholar]
- 21.Ilg, T., E. Handman, and Y. D. Stierhof. 1999. Proteophosphoglycans from Leishmania promastigotes and amastigotes. Biochem. Soc. Trans. 27518-525. [DOI] [PubMed] [Google Scholar]
- 22.Julia, V., and N. Glaichenhaus. 1999. CD4+ T cells which react to the Leishmania major LACK antigen rapidly secrete interleukin-4 and are detrimental to the host in resistant B10.D2 mice. Infect. Immun. 673641-3644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Julia, V., M. Rassoulzadegan, and N. Glaichenhaus. 1996. Resistance to Leishmania major induced by tolerance to a single antigen. Science 274421-423. [DOI] [PubMed] [Google Scholar]
- 24.Konecny, P., A. J. Stagg, H. Jebbari, N. English, R. N. Davidson, and S. C. Knight. 1999. Murine dendritic cells internalize Leishmania major promastigotes, produce IL-12 p40 and stimulate primary T cell proliferation in vitro. Eur. J. Immunol. 291803-1811. [DOI] [PubMed] [Google Scholar]
- 25.Launois, P., I. Maillard, S. Pingel, K. G. Swihart, I. Xenarios, H. Acha-Orbea, H. Diggelmann, R. M. Locksley, H. R. MacDonald, and J. A. Louis. 1997. IL-4 rapidly produced by V beta 4 V alpha 8 CD4+ T cells instructs Th2 development and susceptibility to Leishmania major in BALB/c mice. Immunity 6541-549. [DOI] [PubMed] [Google Scholar]
- 26.Lyons, A. B., and C. R. Parish. 1994. Determination of lymphocyte division by flow cytometry. J. Immunol. Methods 171131-137. [DOI] [PubMed] [Google Scholar]
- 27.Ma, D., D. G. Russell, S. M. Beverley, and S. J. Turco. 1997. Golgi GDP-mannose uptake requires Leishmania LPG2. A member of a eukaryotic family of putative nucleotide-sugar transporters. J. Biol. Chem. 2723799-3805. [PubMed] [Google Scholar]
- 28.Malherbe, L., C. Filippi, V. Julia, G. Foucras, M. Moro, H. Appel, K. Wucherpfennig, J. C. Guery, and N. Glaichenhaus. 2000. Selective activation and expansion of high-affinity CD4+ T cells in resistant mice upon infection with Leishmania major. Immunity 13771-782. [DOI] [PubMed] [Google Scholar]
- 29.McDowell, M. A., M. Marovich, R. Lira, M. Braun, and D. Sacks. 2002. Leishmania priming of human dendritic cells for CD40 ligand-induced interleukin-12p70 secretion is strain and species dependent. Infect. Immun. 703994-4001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.McDowell, M. A., and D. L. Sacks. 1999. Inhibition of host cell signal transduction by Leishmania: observations relevant to the selective impairment of IL-12 responses. Curr. Opin. Microbiol. 2438-443. [DOI] [PubMed] [Google Scholar]
- 31.Moser, M., and K. M. Murphy. 2000. Dendritic cell regulation of TH1-TH2 development. Nat. Immunol. 1199-205. [DOI] [PubMed] [Google Scholar]
- 32.O'Garra, A., and K. Murphy. 1994. Role of cytokines in determining T-lymphocyte function. Curr. Opin. Immunol. 6458-466. [DOI] [PubMed] [Google Scholar]
- 33.Olivier, M., D. J. Gregory, and G. Forget. 2005. Subversion mechanisms by which Leishmania parasites can escape the host immune response: a signaling point of view. Clin. Microbiol. Rev. 18293-305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Park, A. Y., and P. Scott. 2001. Il-12: keeping cell-mediated immunity alive. Scand. J. Immunol. 53529-532. [DOI] [PubMed] [Google Scholar]
- 35.Piedrafita, D., L. Proudfoot, A. V. Nikolaev, D. Xu, W. Sands, G. J. Feng, E. Thomas, J. Brewer, M. A. Ferguson, J. Alexander, and F. Y. Liew. 1999. Regulation of macrophage IL-12 synthesis by Leishmania phosphoglycans. Eur. J. Immunol. 29235-244. [DOI] [PubMed] [Google Scholar]
- 36.Ponte-Sucre, A., D. Heise, and H. Moll. 2001. Leishmania major lipophosphoglycan modulates the phenotype and inhibits migration of murine Langerhans cells. Immunology 104462-467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Poulter, L. W., and C. R. Pandolph. 1982. Mechanisms of immunity to leishmaniasis. IV. Significance of lymphatic drainage from the site of infection. Clin. Exp. Immunol. 48396-402. [PMC free article] [PubMed] [Google Scholar]
- 38.Prina, E., T. Lang, N. Glaichenhaus, and J. C. Antoine. 1996. Presentation of the protective parasite antigen LACK by Leishmania-infected macrophages. J. Immunol. 1564318-4327. [PubMed] [Google Scholar]
- 39.Reiner, S. L., D. J. Fowell, N. H. Moskowitz, K. Swier, D. R. Brown, C. R. Brown, C. W. Turck, P. A. Scott, N. Killeen, and R. M. Locksley. 1998. Control of Leishmania major by a monoclonal alpha beta T cell repertoire. J. Immunol. 160884-889. [PubMed] [Google Scholar]
- 40.Ritter, U., A. Meissner, C. Scheidig, and H. Korner. 2004. CD8 alpha- and Langerin-negative dendritic cells, but not Langerhans cells, act as principal antigen-presenting cells in leishmaniasis. Eur. J. Immunol. 341542-1550. [DOI] [PubMed] [Google Scholar]
- 41.Scott, P., and C. A. Hunter. 2002. Dendritic cells and immunity to leishmaniasis and toxoplasmosis. Curr. Opin. Immunol. 14466-470. [DOI] [PubMed] [Google Scholar]
- 42.Scott, P., E. Pearce, P. Natovitz, and A. Sher. 1987. Vaccination against cutaneous leishmaniasis in a murine model. I. Induction of protective immunity with a soluble extract of promastigotes. J. Immunol. 139221-227. [PubMed] [Google Scholar]
- 43.Spath, G. F., L. Epstein, B. Leader, S. M. Singer, H. A. Avila, S. J. Turco, and S. M. Beverley. 2000. Lipophosphoglycan is a virulence factor distinct from related glycoconjugates in the protozoan parasite Leishmania major. Proc. Natl. Acad. Sci. USA 979258-9263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Spath, G. F., L. A. Garraway, S. J. Turco, and S. M. Beverley. 2003. The role(s) of lipophosphoglycan (LPG) in the establishment of Leishmania major infections in mammalian hosts. Proc. Natl. Acad. Sci. USA 1009536-9541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Spath, G. F., L. F. Lye, H. Segawa, D. L. Sacks, S. J. Turco, and S. M. Beverley. 2003. Persistence without pathology in phosphoglycan-deficient Leishmania major. Science 3011241-1243. [DOI] [PubMed] [Google Scholar]
- 46.Stetson, D. B., M. Mohrs, V. Mallet-Designe, L. Teyton, and R. M. Locksley. 2002. Rapid expansion and IL-4 expression by Leishmania-specific naive helper T cells in vivo. Immunity 17191-200. [DOI] [PubMed] [Google Scholar]
- 47.Sundar, S. 2001. Drug resistance in Indian visceral leishmaniasis. Trop. Med. Int. Health 6849-854. [DOI] [PubMed] [Google Scholar]
- 48.Sundar, S., and M. Rai. 2002. Advances in the treatment of leishmaniasis. Curr. Opin. Infect. Dis. 15593-598. [DOI] [PubMed] [Google Scholar]
- 49.Turco, S. J., and A. Descoteaux. 1992. The lipophosphoglycan of Leishmania parasites. Annu. Rev. Microbiol. 4665-94. [DOI] [PubMed] [Google Scholar]
- 50.Turco, S. J., G. F. Spath, and S. M. Beverley. 2001. Is lipophosphoglycan a virulence factor? A surprising diversity between Leishmania species. Trends Parasitol. 17223-226. [DOI] [PubMed] [Google Scholar]
- 51.Uzonna, J. E., G. F. Spath, S. M. Beverley, and P. Scott. 2004. Vaccination with phosphoglycan-deficient Leishmania major protects highly susceptible mice from virulent challenge without inducing a strong Th1 response. J. Immunol. 1723793-3797. [DOI] [PubMed] [Google Scholar]
- 52.Wang, Q., L. Malherbe, D. Zhang, K. Zingler, N. Glaichenhaus, and N. Killeen. 2001. CD4 promotes breadth in the TCR repertoire. J. Immunol. 1674311-4320. [DOI] [PubMed] [Google Scholar]
- 53.Wang, Z. E., S. Zheng, D. B. Corry, D. K. Dalton, R. A. Seder, S. L. Reiner, and R. M. Locksley. 1994. Interferon gamma-independent effects of interleukin 12 administered during acute or established infection due to Leishmania major. Proc. Natl. Acad. Sci. USA 9112932-12936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Zaph, C., J. Uzonna, S. M. Beverley, and P. Scott. 2004. Central memory T cells mediate long-term immunity to Leishmania major in the absence of persistent parasites. Nat. Med. 101104-1110. [DOI] [PubMed] [Google Scholar]
- 55.Zufferey, R., S. Allen, T. Barron, D. R. Sullivan, P. W. Denny, I. C. Almeida, D. F. Smith, S. J. Turco, M. A. Ferguson, and S. M. Beverley. 2003. Ether phospholipids and glycosylinositolphospholipids are not required for amastigote virulence or for inhibition of macrophage activation by Leishmania major. J. Biol. Chem. 27844708-44718. [DOI] [PubMed] [Google Scholar]