Abstract
We have investigated the phase behavior of self-assembled lyotropic liquid crystals (LC) formed by ternary mixtures of oleoylethanolamide (OEA), water and arginine. OEA, a natural analog of the endogenous cannabinoid anandamide involved in the peripheral regulation of feeding, was selected as a main component due to its capacity to induce efficient decreases in food intake and gains in body mass. Arginine was selected as representative hydrophilic amino acid and added to the OEA-water mixture at different concentrations. The phase diagrams were determined by combining cross-polarized optical microscopy and small angle x-ray scattering. First, the phase diagram for the OEA-water system was determined. It was shown that these two compounds give rise to reverse Ia3d double gyroid and reverse Pn3m double diamond cubic phases existing in bulk over a large window of temperature and composition, and that for water content beyond 25% Pn3m coexisted with excess water. Successively, the influence of arginine as guest molecule in the water channels of the reverse LC was investigated. For the sake of comparison, results for the OEA-water-arginine system were compared with analog series of OEA-water-glucose. The results showed that, at a fixed water content and temperature, the phase behavior of the liquid crystalline phases is strongly dependent on arginine concentration. In more detail, arginine could be encapsulated in the bulk OEA-water LC up to 2.0% wt, whereas transitions from Ia3d to Pn3m cubic phase were observed with increasing arginine concentration. Interestingly, upon an increase of water concentration beyond 20–25%, Pn3m phase started to coexist with excess water releasing the arginine in external water solution. Quantitative measurements of arginine content inside the LC water channels and in the excess external water solution revealed a complete release of the amino acid, demonstrating that the investigated lyotropic liquid crystalline systems can be used as ideal vehicles for the delivery of functional hydrophilic active molecules in aqueous environment.
Introduction
In recent years, self-assembled lyotropic liquid crystalline (LC) phases of lipids and water, as well as their aqueous colloidal dispersions, have gained increasing interest, due to their potential in different fields of applications, such as food technology (1–3), encapsulation and crystallization of proteins (4–10), polysaccharides (11), cholesterol (12), administration of drugs (13–15) and the formulation of new delivery systems (13–16). Among the lipids studied, monoglycerides are the most widely investigated systems. The common types of liquid crystalline phases encountered in binary monoglyceride/water systems are the isotropic micellar fluid (L2), the lamellar phase (Lam), the reverse hexagonal columnar phase (HII), and some types of reverse bicontinuous cubic structures, namely the double diamond (Pn3m), and the double gyroid (Ia3d) (17–19). Because the structure of LC systems is a key factor in their applications, it is of great importance to investigate the effects of guest molecules on their morphologies and the way these molecules may partition within hydrophilic or hydrophobic domains of the LC structures. The polarity of the guest molecules will be the first factor determining changes in the topology of LC phases. For example, polar guest molecules located in the aqueous domains will alter the water solution properties and can either modify the equilibrium hydration level of the monoglyceride polar headgroups or participate directly in cooperative hydrogen bonding with the monoglyceride headgroups (20–22). The research groups of Saturni and Mariani (21,22) have investigated the liquid crystalline behavior of lipid-water systems in presence of large excess water, modified by trehalose saccharide as examples of hydrophilic guest molecules. They found that the introduction of trehalose sugar molecules in the water regions caused a strong reduction of the lattice parameter of the Pn3m cubic phases. Mezzenga et al. have confirmed these findings (11). Furthermore, by using a dextran polysaccharide series of different molecular weights in bulk monoglyceride-water systems, the same authors have further demonstrated that the size of hydrophilic molecules also plays a role by inducing, for example, order-to-order transitions among bicontinuous cubic phases of different group spaces (11). On the other hand, the effect of nonpolar additives, which preferably partition into the hydrophobic region of the LC systems, will essentially tend to cause lipid hydrocarbon tails to swell, release their packing frustration and increase the water/lipid curvature favoring the formation of reversed phases (20,23,24).
One of the major limitations in using monoglyceride-water systems as possible delivery vehicles for active ingredients and components in food applications is the introduction of new fat components in the resulting formulations. As a result, the overall amount of monoglyceride in the composition has to be maintained at a low amount, which consequently hinders the maximum quantity of target active components to be delivered.
The main objective of this work is to investigate the lyotropic liquid crystalline phase behavior of a new system based on water and a special type of monoglyceride, oleoylethanolamide (OEA), and to evaluate this system's possible potential as a delivery vehicle for amino acids and other hydrophilic molecules in aqueous environments. OEA is a natural analog of the endogenous cannabinoid anandamide and it is naturally present in chocolate. It is produced in cells in a stimulus-dependent manner and is rapidly eliminated by enzymatic hydrolysis, suggesting a function in cellular signaling (25,26). Administration of OEA causes a potent and persistent decrease in food intake and a gain in body mass. OEA is a lipid mediator involved in the peripheral regulation of feeding (25). In what follows, we first want to assess whether thermodynamic conditions exist for using OEA as a carrier for hydrophilic functional molecules in conditions of high hydration—those highly diluted concentration regimes where OEA can coexist with excess water and can be administrated in the form of structured colloidal dispersions. The effect of arginine and glucose as polar guest molecules influencing the type of the liquid crystalline phases and their capacity to be encapsulated within OEA-water systems is an integrant part of the study. Fig. 1 illustrates schematically the main concepts toward the use of OEA-water systems as delivery vehicles for amino acids and other hydrophilic molecules.
Figure 1.
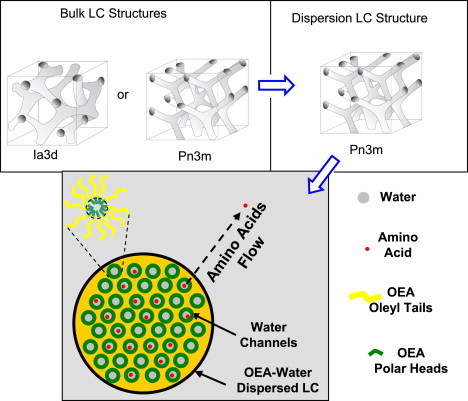
Schematic concepts toward the possible use of OEA-water liquid crystals in excess water as vehicle to deliver amino acids and other hydrophilic guest molecules.
Experimental Methods
Materials
OEA was extracted and purified as described previously by Rodríguez de Fonseca et al. (25,27). L(+)-arginine (Mw = 174.20 g/mole), purity grade ∼99%, was purchased from Aldrich, Switzerland. Its solubility in water was determined to be 15.91 wt % at 20°C (see the Supporting Material). The density of arginine saturated solution at 20°C is 1.041 g/ml. Limonene was selected as a typical hydrophobic plasticizing compound for the OEA tails. Therefore, 5.0 wt % limonene with respect to OEA, was mixed with the OEA; the resulting 95:5 wt % binary mixture of OEA:Limonene was considered, for simplicity, as one single component throughout the whole work and will simply be referred as OEA in what follows. All samples were used as received. Scheme 1 sketches the chemical structures of the various compounds used in the study.
Scheme 1.
Chemical structures of the compounds used in preparation of the different liquid crystalline structures.
Sample preparation
Arginine solutions of various concentrations ranging from 0 to 10 wt % of arginine in water were prepared by dissolving arginine in high-performace liquid chromatography-grade water (pH 7.0) at room temperature under continuous stirring. The aqueous arginine solution was then added to OEA at different weight ratios ranging from 10 to 50 wt %. The resulting mixtures were then placed in glass vials (15 ml volume) with sealed caps and left in a water bath at 85°C for 10 min. The vials were then subjected to a vortex to shake the samples. This step was repeated twice to assure complete and homogeneous mixing of the components. Finally, the vials containing homogeneous samples were quenched at 4°C to allow the study of truly equilibrium morphologies from room temperature upward, in agreement with the procedure previously described by Caffrey et al. (28) to prevent formation of metastable states. Because the isoeletric point (pI) of Arginine is 10.8, at the pH used for the formulation (pH 7), Arginine is positively charged. For the preparation of small angle x-ray scattering (SAXS) samples, quartz capillaries tubes (∼7 cm length, 1 mm thick) were immersed in the 15 ml glass vials (∼12 cm length) at temperatures ∼80–85°C (10–15°C higher than the temperature at which the L2 + water is formed) and they were filled by simple capillarity. They were then cooled to room temperature, removed from the vials and sealed by rapid-cure epoxy. Before measurements were taken, they underwent the same quenching treatment used for the vials.
Cross-polarized optical microscopy (CPOM) and turbidity
CPOM was used as a quick and easy inspection method to determine the phase boundaries of LC phases showing birefringent behavior (crystalline OEA, Lamellar). Samples contained in sealed glass vials were placed in a homemade setup consisting of a water bath in between two polarizers crossed at 90° and were illuminated by a halogen light source from behind one polarizer. The temperature was increased at 2°C steps allowing detection of birefringence in the temperature range from 30 to 80°C. For each isothermal step, samples were allowed to equilibrate in the isothermal water bath for at least 30 min, securing the achievement of LC thermodynamic conditions.
Turbidity relies on the scattering of visible light by large (∼μm) interfaces and was used to detect the coexistence of water and a specific mesophase. The same temperature steps used by CPOM were employed for turbidity screening.
SAXS
SAXS experiments were carried out to further refine the phase diagrams tentatively constructed by cross-polarized microscopy, to afford a distinction among the different types of bicontinuous cubic phases (and other possible isotropic nonbirefringent phases) and to determine the main lattice parameters of the observed LC structures.
SAXS diffractograms were acquired on a SAXSess instrument by Anton Paar (Gratz, Austria) equipped with a line collimation set-up. The system used a Cu Kα radiation source in a sealed tube (λ = 1.542 nm). The beam was attenuated by a semi-transparent nickel foil beam stop. The sample holder was temperature controlled in the range comprised from −4 to 90°C. Measurements were performed using capillaries of 1 mm in diameter, in the q-range of 7.7x10−2 to 28.5 nm−1. Acquisition time was typically 30–40 min. To reduce the noise and to cancel out the effect of LC mono-domains contribution in the final SAXS diffractograms, the capillaries were slightly rotated every 10 min. The time allowed for equilibrating samples was 45 min for temperatures between 42 and 55°C and 30 min for temperatures above 55°C.
All scattering signals were treated with SAXSquant software (Anton Paar).
UV spectroscopy
An UV spectrometer (evolution 100, Thermo Electron) was used to measure absorption in water solutions. First, a wavelength scan was performed to determine the suitable wavelength of measurements (λ = 200 nm). Then, a series of arginine solutions ranging from 0.0005 to 0.0035 wt % were prepared to construct a calibration curve at 200 nm, where the absorbance increases linearly with arginine concentration. The arginine solution from cubic phases in excess water was isolated by centrifugation and then diluted (1000 times) to allow measurements in the linear regime and resulting measurements were then corrected to account for dilution. The final value of arginine concentration in water was determined by interpolation from the calibration curve. The determined concentrations were the average values of two independent experiments (the difference in absorbance in each pair of experiments is ≤ 0.012).
NIR Fourier transform spectroscopy
Near IR spectra were performed using a spectrometer (FT-NIR MPA, Bruker, Ettlingen, Germany) to detect possible interactions between OEA and Arginine. Measurements were carried out in the wavelength range from 1000 to 2500 nm at room temperature. The thicknesses of the prepared samples were of the order of 5.0 mm.
Results
Phase diagrams and topologies of LC structures
Fig. 2 depicts the SAXS scattering spectra with indexed peaks at 42.0°C for the mixtures of OEA-water at 20 wt % water and with 0.0, 2.5, 5.0, 7.5 and 10.0 wt % arginine and 5.0 wt % glucose. Fig. 2 also reports the diffractogram recorded for an OEA-water mixture with 50 wt % water and with 5.0 wt % arginine, that is, in excess water conditions. It can be clearly observed from the diffratograms that samples prepared by mixing OEA with solutions having a lower arginine content (0.0, 2.5) exhibit a bulk Ia3d cubic morphology, whereas coexistence of Ia3d and Pn3m cubic morphologies can be clearly resolved in the sample containing 5.0 wt % of arginine (see Table 1 for the attribution of peak sequences in Ia3d and Pn3m phases). Single Pn3m cubic phase is observed in samples containing 7.5 and 10.0 wt % arginine, whereas in the presence of excess water, the Pn3m cubic phase is the only LC phase detected, which is consistent with literature data on monolyceride-water systems (21,22).
Figure 2.
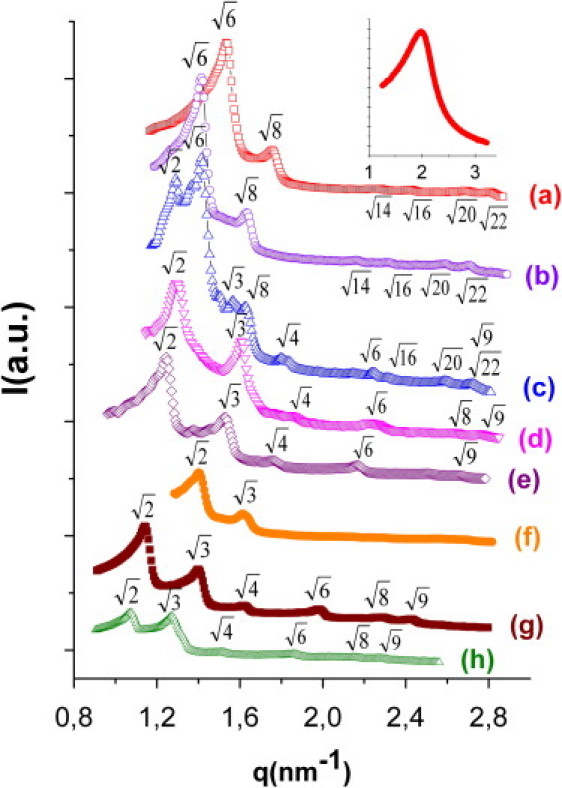
SAXS spectra for the lyotropic liquid crystalline structures observed at different composition ratios at 42°C. The spectra a, b, c, d and e correspond to constant water/lipid ratio of 20.0% wt and arginine concentrations of 0.0, 2.5, 5.0, 7.5 and 10.0% wt, respectively. The spectrum f represents water/lipid ratio of 20.0% wt, and glucose concentration of 5.0%. The spectra g and h represent water/lipid ratio of 50.0% wt and arginine concentration of 5.0 and 2.5% wt, respectively. The SAXS spectrum in the right top inset of the figure corresponds to the L2 phase sample having water/lipid ratio of 20.0% wt, 2.5% wt arginine and 70°C.
Table 1.
Miller indices (hkl) and Bragg peak ratio for Ia3d and Pn3m bicontinuous cubic phases
| Ia3d | Pn3m | ||
|---|---|---|---|
| (211) | √6 | (110) | √2 |
| (220) | √8 | (111) | √3 |
| (321) | √14 | (200) | √4 |
| (400) | √16 | (211) | √6 |
| (420) | √20 | (220) | √8 |
| (332) | √22 | (221) | √9 |
From diffractograms such as those reported in Fig. 2, the lattice parameter of the individual LC phases present in bulk, coexisting or present in excess with water, could be easily determined by:
| (1) |
where q is the scattering vector at which a maximum in scattering intensity is found and h, k and l are the corresponding Miller indexes of the group space corresponding to the LC structure observed.
The results are summarized as a function of temperature and arginine content for samples having 20 and 50 wt % content, respectively in Table S1 and Table S2 of the Supporting Material. In the Pn3m bulk phase of the various mixtures, the general tendency of the variation of lattice parameter with temperature is a monotonous decrease. This is consistent with results reported by Caffrey et al. (28) and Mezzenga et al. (19) in other types of monoglyceride-water binary systems and was previously rationalized by the increased flexibility and rearrangement of the lipid hydrophobic tails with temperature as well as the progressive dehydration of polar heads. Trends for the lattice parameter of the Ia3d with temperature are less straightforward, possibly because of the extended coexistence region of this phase together with the Pn3m. More interestingly, the effect of both arginine and glucose content is reflected by a systematic decrease in the lattice parameter for all the LC structures considered, where the decrease is proportional to the concentration of arginine and glucose, and with glucose having a more pronounced effect than arginine. Finally, as expected, increasing water content led, in all cases, to increased lattice parameters.
By combining SAXS, turbidity measurements and CPOM analysis, it was possible to efficiently and rapidly construct the phase diagrams for the various OEA-water-arginine mixtures, as previously reported for other lipid/water mixtures (19). More specifically, CPOM allowed us to screen for the presence of anisotropic phases, (such as lamellar or hexagonal) and crystalline OEA states, which all show birefringence versus optically inert isotropic mesophases, such as the bicontinuous cubic phases; turbidity enabled detecting coexistence of specific phases with water and SAXS allowed to confirm the attribution of individual phase diagram regions to a specific crystalline group space, or combination thereof for the coexistence regions.
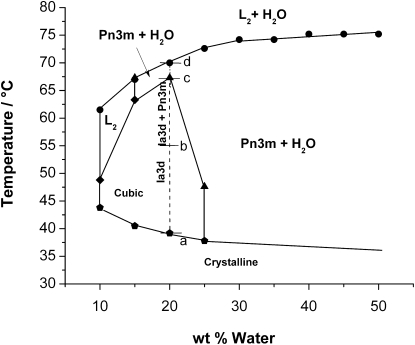
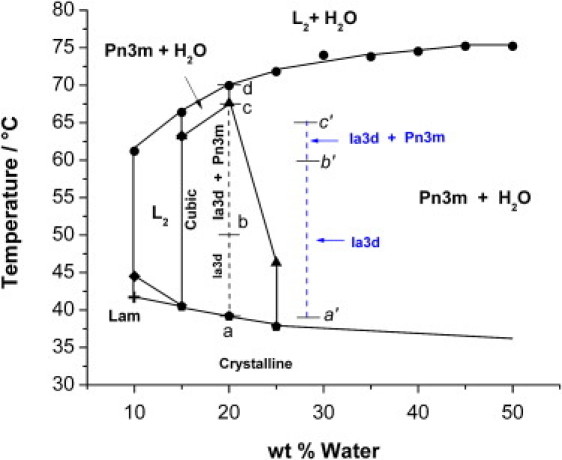
Fig. 3 shows the phase diagram of the pure OEA-water binary system. A large region corresponding to bicontinuous cubic phases is observed between 10 and 25% water content and temperatures ranging between 37°C and 67°C. With the support of SAXS analysis at fixed composition of 20% water, Ia3d reverse bicontinuous cubic phase is identified between 40°C and 55°C, whereas between 55°C and 67°C coexistence of Ia3d and Pn3m cubic phases is observed. The Ia3d-Pn3m transition occurring upon a temperature increase is in agreement with the behavior shown by monoolein-water (28) and monolinolein-water systems (19). Between 67°C and 70°C, Pn3m coexists with water, as argued by combining SAXS data and the turbidity aspect of the samples. Finally, beyond 70°C, the sample still remains turbid, but the corresponding SAXS diffractogram shows a single broaded peak characteristic of reversed micellar isotropic fluid (L2), indicating that L2 phase coexists with excess water. Therefore, the present binary system does not exhibit a reversed hexagonal phase at higher temperatures, as it is the case for the most common types of monoglycerides. Pure L2 phase exists in a narrow window between 47°C and 67°C and 10 and 15% water. Finally, between 37°C and 70°C and for water content exceeding 25%, the Pn3m coexists at thermodynamic equilibrium with excess water.
Figure 3.
Phase diagram of water-OEA lipid system as obtained by combining CPOM and SAXS. The dashed line on the phase diagram depicts the SAXS results corresponding to water/lipid composition ratio of 20.0 wt %. The small letters (a, b, c, and d) are given to distinguish the temperature intervals of the different cubic phases. The abbreviation (L2) refers to the reversed micellar isotropic fluid.
Figs. 4–7 show the effect of increasing arginine content on the phase diagram. The large bicontinuous cubic phase region characteristic of OEA-water mixture shrinks substantially with the increase of arginine content. Furthermore, the Ia3d phase is rapidly destabilized by the presence of arginine, in favor of the Pn3m phase. For example, the maximum temperature at which the Ia3d can be observed is: 67.5°C, 66°C and 60°C for 0.0, 2.5 and 5.0% arginine content, respectively, whereas Ia3d phase is completely suppressed starting from 7.5% arginine content or more. Furthermore, 100% bulk Ia3d reversed bicontinuous cubic phase is observed only for a 2.5% maximum arginine content; at 5% arginine, the Ia3d is always observed in coexistence with Pn3m phase. Since Ia3d is in general greatly stabilized by water hydrating the monoglyceride polar heads (29), the results discussed above suggest that the major effect of arginine in the water channels is to dehydrate the lipid polar head.
Figure 4.
Phase diagrams of water-OEA lipid system as obtained by combining CPOM and SAXS at 2.5% wt arginine. The blue dashed lines correspond to the SAXS results of water-OEA lipid system in presence of glucose solution at constant water/lipid ratio of 20.0 wt %, and concentration of glucose of 2.5% wt. The small letters a′, b′, and c′ across the blue dashed lines are given to distinguish the temperature intervals of the different cubic phases of water-OEA lipid system in presence of glucose solution. The abbreviation “Lam” refers the presence of a lamellar phase.
Figure 5.
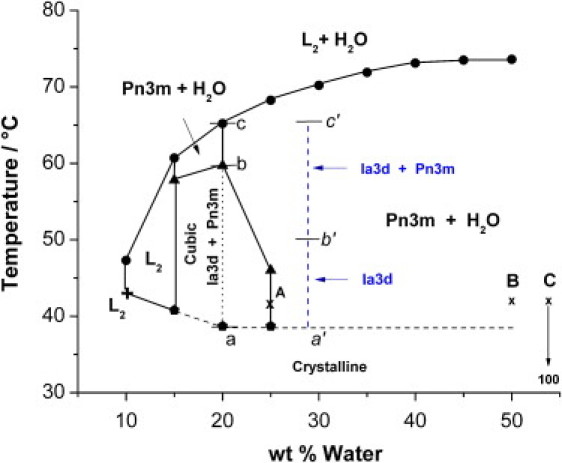
Phase diagrams of water-OEA lipid system as obtained by combining CPOM and SAXS at 5.0% wt arginine. The blue dashed lines correspond to the SAXS results of water-OEA lipid system in presence of glucose solution at constant water/lipid ratio of 20.0 wt %, and concentration of glucose of 5.0% wt. The small letters a′, b′, and c′ across the blue dashed lines are given to distinguish the temperature intervals of the different cubic phases of water-OEA lipid system in presence of glucose solution. The three selected points A, B, and C located at 42.0°C in the phase diagram correspond to water/lipid ratios of 25.0, 50.0, and 100.0%, respectively (details are presented in the Discussion section).
Figure 6.
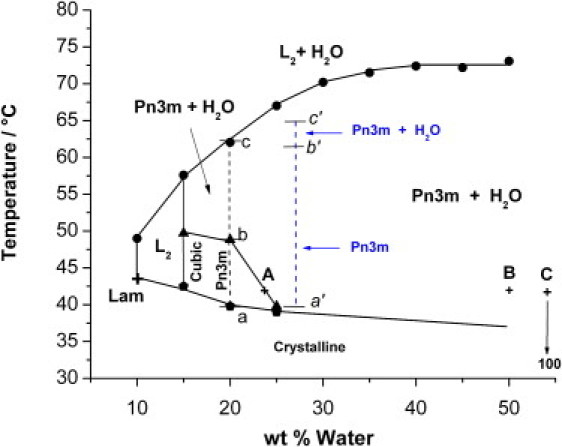
Phase diagrams of water-OEA lipid system as obtained by combining CPOM and SAXS at 7.5% wt arginine. The blue dashed lines correspond to the SAXS results of water-OEA lipid system in presence of glucose solution at constant water/lipid ratio of 20.0 wt %, and concentration of glucose of 7.5% wt. The small letters a′, b′, and c′ across the blue dashed lines are given to distinguish the temperature intervals of the different cubic phases of water-OEA lipid system in presence of glucose solution. The abbreviation “Lam” refers to the presence of a lamellar phase. The three selected points A, B, and C located at 42.0°C in the phase diagram correspond to water/lipid ratios of 25.0, 50.0, and 100.0%, respectively (details are presented in Discussion section).
Figure 7.
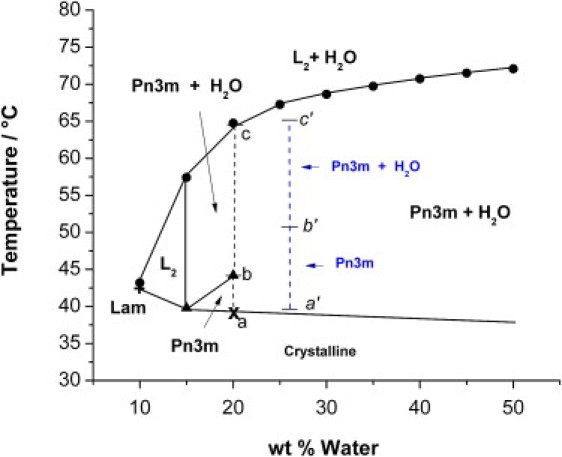
Phase diagrams of water-OEA lipid system as obtained by combining CPOM and SAXS at 10.0% wt arginine. The blue dashed lines correspond to the SAXS results of water-OEA lipid system in presence of glucose solution at constant water/lipid ratio of 20.0 wt %, and concentration of glucose of 10.0% wt. The small letters a′, b′, and c′ across the blue dashed lines are given to distinguish the temperature intervals of the different cubic phases of water-OEA lipid system in presence of glucose solution. The abbreviation ”Lam” refers to the presence of a lamellar phase.
These results are supported also by trends observed for the onset temperature at which Pn3m and water start to coexist. Indeed, at 20 wt % water, the temperature at which Pn3m starts to coexist with water decreases as a function of arginine concentration in the following order: 67.5, 67.0, 60.0, 47.5, and 43.0°C when 0.0, 2.5, 5.0, 7.5 and 10.0 wt % aqueous arginine solutions are used, respectively. The temperature at which the Pn3m melts into the isotropic L2 liquid follows similar trends and decreases gradually as a function of arginine content according to the following order: 70.0, 70.0, 65.0 and 62.0, when 0.0, 2.5, 5.0 and 7.5 wt % aqueous arginine solutions are used, respectively. The only exception is observed for the highest arginine containing sample (10.0 wt % aqueous arginine), where the onset of the L2 transition is observed at 64°C.
The shrinking of the bicontinuous cubic phases with arginine is also evident on the composition window at which these are observed whereas for the pure OEA-water system, the Ia3d bicontinuous region extends down to 10% water for all the ternary systems and the Ia3d cubic phase appears only starting at 15% water.
For the sake of comparison, the LC behavior of OEA with mixtures of aqueous glucose solutions has been also investigated. The samples studied have the same water/OEA ratio (20 wt %) and identical concentrations of glucose (2.5, 5.0, 7.5 and 10.0 wt %) as the OEA-water-arginine system. Because the molecular masses of arginine (174 g/mole) and glucose (180 g/mole) differ by ∼3%, identical concentrations by weight of glucose or arginine essentially have the same molar equivalent content. The order to order transitions revealed by SAXS results on these samples are represented by the dashed blue lines on the phase diagrams (Figs. 4–7). It can be clearly observed that at low glucose concentrations (2.5 and 5.0 wt %), the tendency to destabilize the Ia3d cubic phase is greatly reduced compared to the arginine homolog series, as this phase is maintained over a large temperature window still in the 5.0% glucose mixture. Furthermore, at higher glucose concentrations (7.5 and 10.0 wt %), where the Ia3d phase is suppressed also in the glucose series, the bulk Pn3m cubic phases exist in larger windows compared to homolog arginine series, since the Pn3m-water coexistence region begins in the former case systematically at higher temperatures. In addition, the glucose series shows a reduced tendency toward the coexistence of Pn3m and Ia3d cubic phases. Indeed, the temperature range of coexistence for Ia3d and Pn3m for samples mixed with 2.5 and 5.0 wt % glucose is 60.0–65.0°C and 50.0–65.0°C, respectively, as opposed to the respective 55.0–67.0°C and 39.0–60.0°C for the homolog 2.5 and 5.0 wt % arginine solutions, respectively. Finally, with the exception of 2.5 wt % concentration, the onset temperature of coexistence of Pn3m with water is greatly reduced when arginine is used, as opposed to glucose; this difference being as high as 15°C when 7.5% arginine or glucose are used.
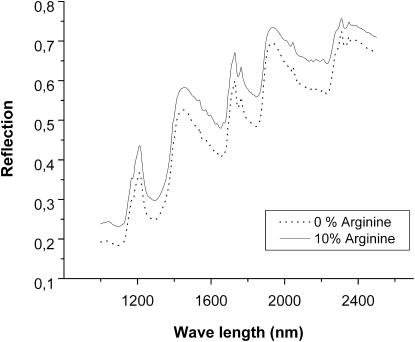
These results highlight the very different impact of arginine and glucose on OEA-water LC phases, where the arginine shrinks the bicontinuous cubic phase in a more remarkable way. This behavior has to be attributed to the different hydrophilic nature of the two guest molecules considered. First of all, at the pH considered in this study (7.0) arginine is positively charged, bearing two ammonium positive charges and one negative charge in the carboxyl group; it is therefore substantially more difficult to concentrate charged molecules (arginine) within the water channels of the liquid crystalline phases, as compared to a neutral molecule (glucose). Furthermore, the solubility in water of arginine is much less than that of glucose: although a maximum of 15.91% arginine can be dissolved in water, glucose and water constitute a fully miscible pair. Consequently, arginine needs to be hydrated by a larger amount of water compared to glucose, decreasing the effective amount of water molecules available for the hydration of OEA polar heads. This is consistent with the systematic destabilization of Ia3d in favor of the Pn3m phase. Finally, although glucose (6-hydroxyl groups per molecule) is expected to participate in direct hydrogen bonding with the OEA hydrophilic head, arginine is less likely to behave similarly. One additional indirect evidence of this possible scenario is provided by near-IR spectra of mixtures of OEA with pure water and arginine solution (10.0 wt %) presented in Fig. 8. Both spectra are nearly identical, suggesting that no preferable interactions of arginine with polar headgroups of OEA take place.
Figure 8.
NIR spectra of water-OEA system as a function of arginine concentration in water.
Discussion
Quantitative estimation of arginine concentration inside and outside the Pn3m liquid crystalline phases
Because the phase diagrams of the OEA-water-arginine systems all exhibit a large region of coexistence of Pn3m and water solutions, it becomes very interesting to determine the concentration of arginine within the free bulk water and inside the Pn3m coexisting phase. Indeed, bringing added insight into this point would allow us to determine whether the Pn3m-based dispersions, which can be formed by adding excess water to bulk Pn3m phase of OEA-water-arginine, can be used to efficiently deliver the arginine in the surrounding aqueous environment.
To establish this point unequivocally, four samples were prepared from water solutions with 5.0 and 7.5 wt % arginine and total amount of water of 50.0 and 70.0 wt %. The samples were then centrifuged (12,000 rpm) isothermally at 42°C for a period of 20 min; this allowed separating excess water from the mixture while preserving the Pn3m phase. Thereafter, the concentration of arginine could be estimated in the separated solutions by means of UV spectroscopy.
In all the experiments, the measured concentration of arginine in the free water was systematically found to exceed the concentration in the initial solution: for 50% water mixture, the measured arginine concentrations were 7.525 wt % and 10.402 wt % as opposed to initial 5.0% and 7.5% concentrations, respectively; for samples containing 70.0 wt % water, the measured arginine concentrations were 8.308 and 8.798 wt %, respectively. These data support a delivery of arginine from the Pn3m water channels to the external water phase, as would also be expected based on simple osmotic pressure considerations.
These data do not allow, however, quantifying directly the amount of transferred arginine, as this amount needs to be normalized with the respect to the quantity of water which is free and the amount of water which is encapsulated within Pn3m channels.
To do so, the weight fraction of Pn3m and coexisting free water has to be determined. In what follows, the typical procedure to estimate the absolute value of arginine transferred from the Pn3m channels to the coexisting water is discussed for the sample prepared by mixing OEA with arginine solution (5.0 wt %) and a water/lipid ratio of 50.0 wt.%.
Because compositions in the phase diagrams do not account on the x axis for arginine content, the mass of the lipid, water and arginine have to be renormalized first to a total of 100%, which is done in Table S3 using the amount of components typically used for the formulation of the various mixtures considered (see the Supporting Material).
The three selected points A, B, and C, located at 42.0°C in the phase diagram of Fig. 5 correspond to water/lipid ratios of 25.0, 50.0, and 100.0%, respectively.
Point A represents the first point on the phase diagram at which the Pn3m starts to coexist with water. Point C represents the last point in the phase diagram at which Pn3m and water-arginine solution coexist, that is 5.0% arginine, 95% water and 0% Pn3m. Point B represents the equilibrium composition between the Pn3m dispersed phase and the arginine-water solution for which a 7.5 wt % arginine content in free water is measured by UV spectroscopy.
At point B, using the composition law, and Table S3, the overall mass fraction of lipid , the mass fraction of Pn3m dispersion, , the mass fraction of free water-arginine solution coexisting with Pn3m, , and the total mass fraction of water-arginine solution (the free solution plus that contained within Pn3m channels), , are related by the following equations:
| (2) |
| (3) |
Solving Eqs. 2 and 3 leads to and . Therefore, for a total mass of the blend, M, considering the 7.5 wt% arginine concentration measured in the free water solution, a total 0.075·0.34·M = 0.026·M arginine is found in free water, rather than the 0.05·0.34·M = 0.017·M expected for a system maintaining the original arginine concentration.
This represents an excess of 0.009·M arginine which has been transferred from the channels of the Pn3m to the external water solution. It is interesting to compare this excess with the amount of arginine encapsulated in the Pn3m, and thus to evaluate the residual content of arginine in the Pn3m dispersions. This content can be evaluated directly from point A in Fig. 5 as 0.259·0.66·0.05·M = 0.009·M. It is then found that essentially all the arginine is transferred from the Pn3m phase to the surrounding water solution in excess, which is somewhat surprising since based on pure osmotic pressure considerations; one would expect similar concentrations in the water solutions within and outside water channels.
Analog calculations carried out for the 7.5% initial arginine concentration in the 50% water-to-lipid mixture, as well as for the 5% and 7.5% arginine initial concentrations in the 70% water-to-lipid mixtures lead to the same conclusions, that the complete amount of arginine is transferred from the Pn3m to the coexisting water. A possible explanation for this tendency to expel all the arginine, and which is in line with the observed effect of arginine on the entire phase diagrams, can be found in: i), the poor hydrophilic nature of arginine and ii), the fact that arginine is charged. Indeed, since only 15.91% of arginine can be dissolved in water, to prevent macroscopic phase separation and maintain all the arginine within the Pn3m channels, most of the water has to be made available to hydrate arginine, thus dehydrating the OEA polar heads. In such a scenario, the free unbound water within the Pn3m channels, available to decrease the arginine chemical potential, is expected to be small to nil. Consequently, the effective chemical potential of arginine inside the Pn3m channels is unaffordablely high, providing the driving force for its complete expulsion toward the surrounding coexisting water. The fact that arginine is also positively charged at pH 7.0 further increases the chemical potential of arginine within the Pn3m water channels, enhancing the driving force for migration (and thus dilution) toward the external water phase. Fig. 9, highlights the proposed mechanisms of arginine in the dehydration of OEA polar tails.
Figure 9.
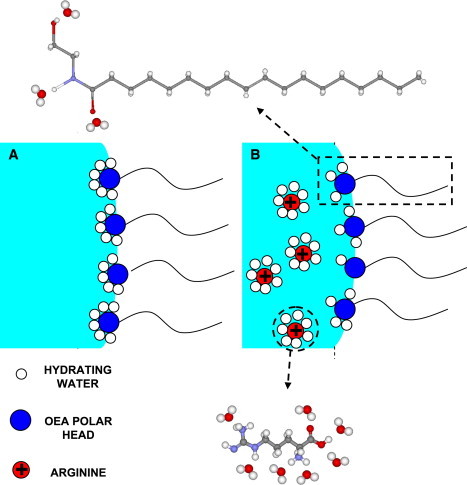
(A) Schematic diagram illustrating the mode of hydration of OEA polar heads by water molecules in absence of arginine. (B) A schematic diagram revealing the dehydration action of arginine molecules on the OEA polar heads, and the subsequent change in curvature of the OEA-water interface.
The OEA-water-arginine systems discussed in this work constitute an ideal candidate for the delivery of amino acids in aqueous environment. Indeed, not only the OEA-water system has unique nutritional behavior, but amino acids can also be efficiently encapsulated in these bulk systems to be completely released in water excess environment.
Although we have no information on how fast the release of arginine is achieved, it can speculatively be argued that this rate could be manipulated by the way Pn3m dispersions are stabilized in water. For example, manipulation of the pH can be envisaged as a tool to control the charges and thus the chemical potential of encapsulated hydrophilic guest molecules. This will have the direct consequence of decreasing their tendency to migrate to the external water phase, and thus, possibly also their release rate. These are only some of the possible open questions that need to be addressed by a comprehensive exhaustive study on the formulation of OEA-water mesophases as delivery systems. We are actively pursuing the investigation on these directions and we hope to be able to address these issues in future work.
Conclusion
We have investigated the phase behavior of lyotropic liquid crystals formed by self-assembled mixtures of oleoylethanolamide and arginine-water systems. Oleoylethanolamide, a natural analog of the endogenous cannabinoid anandamide with a unique metabolic role in the decrease of weight intake, was selected as a possible alternative to traditional monoglycerides and arginine was selected as a target amino acid. The phase diagrams and morphologies of oleoylethanolamide-water-arginine liquid crystalline structures were determined by combining CPOM and SAXS. The results show that the phase behavior and stability of the liquid crystalline phases are highly dependent on arginine concentration. Upon increasing arginine concentration, transitions from Ia3d cubic phase to Pn3m cubic phase were observed, together with remarkable shrinking of the whole phase diagram region in which bicontinuous cubic phases are stable. By comparing the effect of arginine with that of glucose as a low molecular weight hydrophilic compound, it was argued that arginine is an efficient dehydrating agent for the oleoylethanolamide polar head, resulting in the destabilization of Ia3d reversed cubic phases in favor of Pn3m cubic phases.
Due to an extended coexistence region of Pn3m cubic phase and water-arginine solutions, oleoylethanolamide-water-arginine liquid crystals were tested as possible delivery systems for amino acids in aqueous environment. A quantitative estimation of the variation in the content of arginine within the Pn3m channels between the case of pure Pn3m bulk phases and the Pn3m phase coexisting with excess water and arginine, indicated that all the amino acid is released in the surrounding environment when excess water is present, leaving the Pn3m formed by oleoylethanolamide and water, with virtually all the arginine expelled. The reason for such a behavior was found in: i), the relatively low hydrophilicity of arginine, which has tendency to dehydrate oleoylethanolamide polar heads and ii), the charged nature of arginine, which increases its chemical potential within Pn3m water channels.
These results demonstrate that oleoylethanolamide-water liquid crystals can efficiently encapsulate amino acids and are promising systems for a controlled release of these and other hydrophilic molecules in aqueous environment.
Acknowledgments
Delphine Tissot-favre, Frederic Destaillats, Jean Baptiste Bezelgues and Clémentine Thabuis at Nestlé Research Center are kindly acknowledged for providing the purified oleoylethanolamide. Nestlé Management is acknowledged for allowing the publication of this work. SZM is very thankful to Eidgenössischen StipendienKommission für Ausländische Studierende funding committee and Prof. W. Meier for his continuous help and support during the fellowship period.
Supporting Material
References
- 1.Larsson K. Aqueous dispersions of cubic lipid–water phases. Curr. Opin. Colloid Interface Sci. 2000;5:64–69. [Google Scholar]
- 2.Krog N.J. Food emulsifiers and their chemical and physical properties. In: Friberg S.E., Larsson K., editors. Food Emulsions. Marcel Dekker; New York: 1997. pp. 141–188. [Google Scholar]
- 3.Mezzenga R., Schurtenberger P., Burbidge A., Michel M. Understanding foods as soft materials. Nature Mater. 2005;4:729–740. doi: 10.1038/nmat1496. [DOI] [PubMed] [Google Scholar]
- 4.Landau E.M., Rummel G., Rosenbusch J.P., Cowan-Jacob S.W. Crystallization of a polar protein and small molecules from the aqueous compartment of lipidic cubic phases. J. Phys. Chem. B. 2005;101:1935–1937. [Google Scholar]
- 5.Caffrey M. A lipid's eye view of membrane protein crystallization in mesophases. Curr. Opin. Struct. Biol. 2000;10:486–497. doi: 10.1016/s0959-440x(00)00119-6. [DOI] [PubMed] [Google Scholar]
- 6.Caffrey M. Membrane protein crystallization. J. Struct. Biol. 2003;142:108–132. doi: 10.1016/s1047-8477(03)00043-1. [DOI] [PubMed] [Google Scholar]
- 7.Rummel G., Hardmeyer A., Widmer C., Chiu M.L., Nollert P. Lipidic cubic phases: new matrices for the three-dimensional crystallization of membrane proteins. J. Struct. Biol. 1998;121:82–91. doi: 10.1006/jsbi.1997.3952. [DOI] [PubMed] [Google Scholar]
- 8.Angelova A., Angelov B., Papahadjopoulos-Sternberg B., Ollivon M., Bourgaux C. Proteocubosomes: nanoporous vehicles with tertiary organized fluid interfaces. Langmuir. 2005;21:4138–4143. doi: 10.1021/la047745t. [DOI] [PubMed] [Google Scholar]
- 9.Misquitta L.V., Misquitta Y., Cherezov V., Slattery O., Mohan J.M. Membrane protein crystallization in lipidic mesophases with tailored bilayers. Structure. 2004;12:2113–2124. doi: 10.1016/j.str.2004.09.020. [DOI] [PubMed] [Google Scholar]
- 10.Cherezov V., Clogston J., Misquitta Y., Abdel-Gawad W., Caffrey M. Membrane protein crystallization in meso: Lipid type-tailoring of the cubic phase. Biophys. J. 2002;83:3393–3407. doi: 10.1016/S0006-3495(02)75339-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Mezzenga R., Grigorov M., Zhang Z., Servais C., Sagolowicz L. Polysaccharide-induced order-to-order transitions in lyotropic liquid crystals. Langmuir. 2005;21:6165–6169. doi: 10.1021/la050905w. [DOI] [PubMed] [Google Scholar]
- 12.Tenchov B.G., MacDonald R.C., Siegel D.P. Cubic phases in phosphatidylcholine-cholesterol mixtures: Cholesterol as membrane 'fusogen. Biophys. J. 2006;91:2508–2516. doi: 10.1529/biophysj.106.083766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Drummond C., Fong C. Surfactant self-assembly objects as novel drug delivery vehicles. Curr. Opin. Colloid Interface Sci. 1999;4:449–456. [Google Scholar]
- 14.Pouton C.W. Formulation of self-emulsifying drug delivery systems. Adv. Drug Deliv. Rev. 1997;25:47–58. [Google Scholar]
- 15.Engström S., Nordén T.P., Nyquist H. Cubic phases for studies of drug partition into lipid bilayers. Eur. J. Pharm. Sci. 1999;8:243–254. doi: 10.1016/s0928-0987(99)00012-3. [DOI] [PubMed] [Google Scholar]
- 16.Boyd B.J. Characterisation of drug release from cubosomes using the pressure ultrafiltration method. Int. J. Pharm. 2003;260:239–247. doi: 10.1016/s0378-5173(03)00262-x. [DOI] [PubMed] [Google Scholar]
- 17.Esposito E., Eblovi N., Rasi S., Drechsler M., Di Gregorio G.M., Menegatti E., Cortesi R. Lipid-based supramolecular systems for topical application: a preformulatory study. PharmSci. 2003;5 doi: 10.1208/ps050430. art. No. 30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Angelova A., Ollivon M., Campitelli A., Bourgaux C. Lipid cubic phases as stable nanochannel network structures for protein biochip development: X-ray diffraction study. Langmuir. 2003;19:6928–6935. [Google Scholar]
- 19.Mezzenga R., Meyer C., Servais C., Romoscanu A.I., Sagalowicz L. Shear rheology of lyotropic liquid crystals: a case study. Langmuir. 2005;21:3322–3333. doi: 10.1021/la046964b. [DOI] [PubMed] [Google Scholar]
- 20.Seddon J.M. Structure of the inverted hexagonal (HII) phase and non-lamellar phase transitions of lipids. Biochim. Biophys. Acta. 1990;1031:1–69. doi: 10.1016/0304-4157(90)90002-t. [DOI] [PubMed] [Google Scholar]
- 21.Saturni L., Rustichelli F., Di Gregorio G.M., Cordone L., Mariani P. Sugar-induced stabilization of the monoolein Pn3m bicontinuous cubic phase during dehydration. Phys. Rev. E Stat. Nonlin. Soft Matter Phys. 2001;64:040902. doi: 10.1103/PhysRevE.64.040902. [DOI] [PubMed] [Google Scholar]
- 22.Mariani P., Rustichelli F., Saturni L., Cordone L. Stabilization of the monoolein Pn 3 m cubic structure on trehalose glasses. Eur. Biophys. J. 1999;28:294–301. [Google Scholar]
- 23.Siegel D.P., Banschbach J., Yeagle P.L. Stabilization of HII phases by low levels of diglycerides and alkanes: an NMR, calorimetric, and x-ray diffraction study. Biochemistry. 1989;28:5010–5019. [Google Scholar]
- 24.Pouzot M., Mezzenga R., Leser M., Sagalowicz L., Guillot S. Structural and Rheological Investigation of Fd3m Inverse Micellar Cubic Phases. Langmuir. 2007;23:9618–9628. doi: 10.1021/la701206a. [DOI] [PubMed] [Google Scholar]
- 25.de Fonseca F.R., Navarro M., Gómez R., Escuredo L., Nava F. An anorexic lipid mediator regulated by feeding. Nature. 2001;414:209–212. doi: 10.1038/35102582. [DOI] [PubMed] [Google Scholar]
- 26.Thabuis C., Destaillats F., Tissot-Favre D., Martin J.-C. Oleoyl-ethanolamide (OEA): A bioactive lipid derived from oleic acid and phosphatidylethanol-amine. Lipid Technology. 2007;19:225–227. [Google Scholar]
- 27.Lo Verme J., Fu J., Astarita G., La Rana G., Russo R. The nuclear receptor peroxisome proliferator-activated receptor-α mediates the anti-inflammatory actions of palmitoylethanolamide. Mol. Pharmacol. 2005;67:15–19. doi: 10.1124/mol.104.006353. [DOI] [PubMed] [Google Scholar]
- 28.Qiu H., Caffrey M. The phase diagram of the monoolein/water system: metastability and equilibrium aspects. Biomaterials. 2000;21:223–234. doi: 10.1016/s0142-9612(99)00126-x. [DOI] [PubMed] [Google Scholar]
- 29.Lee W.B., Mezzenga R., Fredrickson G.H. Anomalous phase sequences in lyotropic liquid crystals. Phys. Rev. Lett. 2007;99:187801. doi: 10.1103/PhysRevLett.99.187801. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.