Abstract
The development of neutralizing antibodies (inhibitors) after factor VIII (FVIII) infusions is a serious complication that affects approximately one-quarter of hemophilia A patients who have access to replacement therapy. To investigate the differentiation of naive T cells into FVIII-specific helper T cells that promote B-cell activation and antibody secretion, HLA-DRA-DRB1*0101-restricted T-cell clones that respond to a specific epitope in FVIII were isolated from a mild hemophilia A subject (the proband) 19 weeks and 21 months after his development of a high-titer inhibitor. Clones responding to the same epitope were also isolated from his multiply infused brother, who has not developed a clinically significant inhibitor. The 19-week proband clones were T helper (TH)17/TH1- or TH1/TH2-polarized, whereas all 8 clones isolated 21 months postinhibitor development were TH2-polarized cells. In contrast, all 6 clones from the brother who did not develop an inhibitor were TH1-polarized, indicating that tolerance to FVIII can be maintained even with circulating TH1-polarized cells that respond vigorously to in vitro FVIII stimulation. This is the first evidence that TH17/TH1-polarized cells play a role in hemophilic immune responses to FVIII. Furthermore, this is the first report of successful isolation and expansion of antigen-specific human TH17/TH1 clones using standard culture conditions.
Introduction
Anti–factor VIII (FVIII) inhibitory antibodies (referred to clinically as inhibitors) develop in 20% to 35% of severe and 3% to 13% of mild/moderate severity hemophilia A patients who receive FVIII replacement therapy, and they are associated with significant mortality, morbidity, and lower quality of life.1–3 FVIII-specific antibodies are dependent on CD4+ T helper (TH) cells,4 which respond to FVIII-derived peptides bound to major histocompatibility complex class II receptors on antigen-presenting cells when these class II-peptide complexes bind with sufficient avidity to their T-cell receptors, causing proliferation and cytokine release. CD4+ T cells may be divided into 4 subsets that secrete distinct cytokines, as follows: TH1, TH2, inducible regulatory T cells, and the recently defined TH17 cell lineage.5,6 These subsets have distinct functions in up- and down-regulating immune responses and in orchestrating antibody class switching.7–10 CD4+ T cells from hemophilia A subjects with inhibitors have been found to secrete interferon (IFN)–γ, indicating the presence of TH1-polarized cells, and interleukin (IL)–4, indicating the presence of TH2-polarized cells, upon FVIII stimulation.11 In contrast, FVIII-stimulated T cells from some hemophilia A subjects without inhibitors secreted both the proinflammatory cytokine IFN-γ and the anti-inflammatory cytokine transforming growth factor (TGF)–β, with the latter indicating the induction of inducible regulatory T cells.11 TH17-polarized cells are associated with chronic inflammation and with various autoimmune disorders,12,13 but there has as yet been no report of their possible role in hemophilia A immune responses. T-cell clones that respond to distinct epitopes in the FVIII A2, C1, and C2 domains have been isolated from several hemophilic subjects,14–17 and these monoclonal CD4+ cell lines are proving useful in mechanistic investigations of inhibitor development.
T-cell clones isolated from 2 brothers with mild hemophilia A due to the missense substitution A2201P, including clones from serial samples obtained after inhibitor development, showed highly similar HLA-DRA-DRB1*0101-restricted T-cell responses to an epitope in FVIII that included the missense substitution site.15,16 The objective of the present study was to investigate the phenotypes of these clones by characterizing cytokine production, chemokine receptor expression, and transcription factor usage, as phenotypic changes over time and differences between these subjects should offer clues as to why only one of them developed a clinically significant inhibitor. Thirteen T-cell clones were isolated from the proband's blood at 2 time points, and 6 clones were obtained from a single sample obtained from his brother. Interestingly, TH17/TH1-polarized clones were isolated from the sample obtained 19 weeks after initial detection of the proband's inhibitor, whereas clones isolated at the 21-month time point all had a distinct TH2 profile, suggesting that the TH17 cell lineage played a role only in earlier stages of this person's anti-FVIII immune response. All 6 clones isolated from the noninhibitor subject were TH1 polarized, a phenotype previously seen in hemophilic subjects with and without an inhibitor, as well as in healthy, nonhemophilic controls.11,14,17
Methods
Human samples and inhibitor titers
Blood samples from hemophilic brothers with the FVIII missense substitution A2201P, who shared the HLA-DRA-DRB1*0101 allele, were obtained after written, informed consent, according to a protocol approved by the University of Washington Human Subjects Review Committee and the Declaration of Helsinki. In a previous study,16 the proband and his (half-)brother were designated IV-1 and IV-2, reflecting their assigned designations in the family pedigree. They are referred to in this study as subjects 17A (the proband) and 32A (his brother), respectively, because these clinical study identifiers were also used to label T-cell clones isolated from their blood. The naming convention for the clones consists of the subject number, the amount of time elapsed since initial inhibitor development (if applicable), and the sequential number of the clone isolated from a particular blood sample. The clinical status of the proband and his brother was described previously.15,16 The proband did not receive any additional FVIII infusions during the course of the present study. His inhibitor titer at 19 weeks was 29 BU/mL, and at 21 months it had fallen to 5 BU/mL.
T-cell clones
Isolation of 5 T-cell clones from subject 17A 19 weeks after initial detection of his inhibitor, and of 6 clones from subject 32A, was previously described.15,16 Eight additional T-cell clones that recognized the synthetic peptide FVIII2194-2213 were isolated from subject 17A 21 months after initial inhibitor detection using the same method. Clones were expanded by stimulation with irradiated peripheral blood mononuclear cells (PBMCs) from an HLA-mismatched person plus phytohemagglutinin (REMEL) in the presence of human IL-2 (Hemagen Diagnostics; a detailed protocol is given in supplemental methods; available on the Blood website; see the Supplemental Materials link at the top of the online article).
Antigen-specific T-cell proliferation and cytokine secretion
T-cell proliferation assays were performed, as described.16 Supernatants were collected 48 hours after peptide stimulation and were stored at −80°C until enzyme-linked immunosorbent assays (ELISAs) were performed. Supernatants from triplicate wells were pooled and analyzed by ELISA to measure IFN-γ, IL-4, IL-17A, and TGF-β1. Sandwich ELISAs were performed using standard methods (supplemental methods).
Transcription factor mRNA expression
Total RNA was isolated from 2 million resting T cells using an RNeasy Mini Kit (QIAGEN). RNA samples were treated with DNase using a TURBO DNA-free kit (Applied Biosystems). cDNA was generated using a high-capacity cDNA reverse-transcription kit (Applied Biosystems). Transcripts were quantified on a 7900HT Fast Real-Time PCR System using predesigned TaqMan gene expression assays, according to the manufacturer's instructions (Applied Biosystems). Probes with the following Applied Biosystems assay identification numbers were used: TBX21, Hs00203436_m1; GATA3, Hs00231122_m1; RORC, Hs01076112_ml. mRNA levels were normalized to 18S rRNA levels, as quantified with the 18S probe/primer TaqMan assay kit (Applied Biosystems). Mean cycle threshold values (n = 3) were determined from amplification plots using Sequence Detection System Software Version 2.3 (Applied Biosystems). Relative mRNA expression levels were determined from a standard curve of serial RNA dilutions from clone 32A-14 (TBX21 mRNA, GATA3 mRNA, and 18S rRNA standard curves) and 17A-19WK-11 (RORC mRNA standard curve).
Chemokine receptor expression
Resting T-cell clones were stained with antibodies, according to the manufacturer's instructions, and flow cytometry was performed on a FACSCalibur (BD Biosciences). Antibodies were from R&D Systems: monoclonal anti–human CXC chemokine receptor (CXCR)3-carboxyfluorescein (CFS) IgG1, mouse IgG1 isotype control-CFS, monoclonal anti–human C-C chemokine receptor (CCR)4-CFS IgG2B, mouse IgG2B isotype control CFS, monoclonal anti–human CCR6 allophycocyanin, and mouse IgG2B isotype control allophycocyanin. Mean fluorescence intensities (MFI) were determined using FlowJo software.
Results
Cytokine secretion and T-cell proliferation
Cytokine secretion and proliferation by T-cell clones isolated from the proband, subject 17A, 19 weeks and 21 months after inhibitor detection, and by clones obtained from his brother, subject 32A, 2 years after his last FVIII infusion, were evaluated. Isolation, tetramer staining, and proliferation assays of some of these clones were described previously.15,16 A total of 19 clones was stimulated with 0.1, 1.0, and 10 μM FVIII2194-2213: 5 from subject 17A at the 19-week time point, 8 from subject 17A at the 21-month time point, and 6 from subject 32A. All clones showed dose-dependent proliferation in response to peptide FVIII2194-2213 (Figure 1A). IFN-γ, IL-4, and IL-17 secretion levels identified 4 distinct cytokine secretion profiles (Figure 1B-D). Two profiles were seen for the 19-week 17A clones, a third profile was seen for the 21-month 17A clones, and a fourth profile was seen for all 32A clones. Clones 17A-19WK-5 and 17A-19WK-10 secreted IL-4 and low amounts of IFN-γ (< 300 pg/mL at 10 μM peptide). Clones 17A-19WK-11, -22, and -33 showed dose-dependent secretion of IL-17 and IFN-γ, although the IFN-γ levels were low. All 8 of the 21-month 17A clones secreted predominantly IL-4, and all 6 clones from 32A secreted high levels of IFN-γ and IL-4. T-cell clones cultured with no peptide or with the irrelevant peptide FVIII519-538 all showed low cytokine production (< 145 pg/mL IFN-γ, < 35 pg/mL IL-4, < 9 pg/mL IL-17; data not shown). TGF-β1 was not detected in any of the supernatants (data not shown). To verify that IL-17 secretion was not an artifact resulting from culturing cells under conditions that could influence their polarization, IL-17 secretion from clones 17A-19WK-11, 17A-19WK-22, and 17A-19WK-33 was assayed using cells that had been frozen after early passages of the original clonal culture (Figure 2). The cytokine profile was similar to that seen in Figure 1, although somewhat higher levels of both IFN-γ and IL-17 were detected.
Figure 1.
Proliferation and cytokine secretion of T-cell clones isolated from subject 17A 19 weeks and 21 months after inhibitor development and from subject 32A at one time point. Resting T-cell clones were stimulated with FVIII2194-2213 (0.1, 1.0, and 10 μM) presented on irradiated PBMCs from an unrelated DRB1*0101 donor. Proliferation (n = 3) was measured by addition of [3H]thymidine at 48 hours, and cells were harvested 18 hours later (A). Cell supernatants were collected after 48 hours to measure IFN-γ (B), IL-4 (C), and IL-17 (D) by ELISA. The proliferation and cytokine levels of cells stimulated with buffer as a negative control were subtracted. Results are presented as stacked bar graphs. (E-F) The total amounts of cytokines secreted after stimulation with FVIII2194-2213 at the 3 concentrations indicated in panel A were summed, and ratios of these total levels were calculated. In all panels, clones are grouped according to their cytokine secretion profiles.
Figure 2.
Cytokine secretion profiles for early lots of IL-17–secreting clones. Resting T-cell clones (first seed lot for 17A-19WK-11 and second seed lots for 17A-19WK-22 and -33) were stimulated with FVIII2194-2213 (0.1, 1.0, and 10 μM) presented on irradiated PBMCs from an unrelated DRB1*0101 donor. Cell supernatants were collected from triplicate wells after 48 hours to measure IFN-γ, IL-4, and IL-17 by ELISA. The cytokine levels of cells stimulated with buffer as a negative control were subtracted.
IFN-γ, IL-4, and IL-17 secretion levels are shown as ratios to reflect T-cell polarization relative to “pure” TH1, TH2, or TH17 cytokine responses (Figure 1E-F). Cytokine ratios were calculated after stimulation with 0.1, 1.0, and 10 μM FVIII2194-2213. Clones 17A-19WK-5 and 17A-19WK-10 were either TH1/TH2 polarized or TH2 polarized, and clones 17A-19WK-11, 17A-19WK-22, and 17A-19WK-33 were TH17/TH1-polarized, whereas at 21 months after initial inhibitor development only TH2-polarized clones were isolated. Calculated ratios were variable for 17A-21MO clones, as IFN-γ levels were at or slightly above background levels. The clones from the tolerant subject 32A secreted approximately 10 times as much IFN-γ as IL-4, consistent with TH1 polarization, although dose-dependent IL-4 secretion was still significant.
TH lineage markers
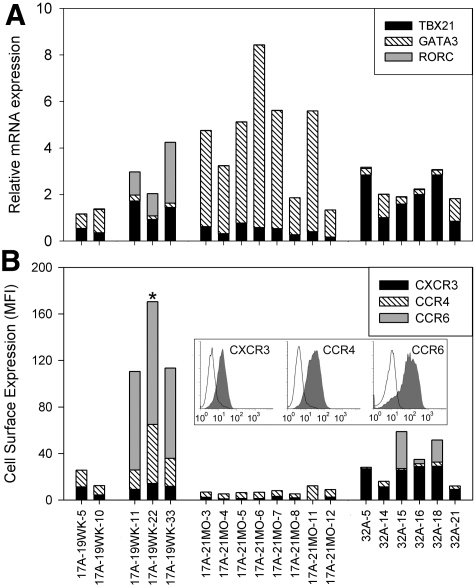
Cytokine secretion patterns indicated the T-cell clones had distinct TH lineages. To better characterize their phenotypes, mRNA expression levels of the lineage-specific transcription factors TBX21, GATA3, and RORγt, which are used by TH1, TH2, and TH17 cells, respectively,5,6 were measured using TaqMan gene expression assays (Figure 3A). The RORγt mRNA assay measured both isoforms expressed from the RORC gene. Cell surface expression of chemokine receptors CXCR3, CCR4, and CCR6, which are associated with TH1, TH2, and TH17 effector function, respectively,18–20 was also measured by flow cytometry (Figure 3B). Clones 17A-19WK-5 and 17A-19WK-10 expressed TBX21 and GATA3 mRNAs, as well as CXCR3 and CCR4 proteins, consistent with the TH1/TH2 lineage deduced from their cytokine profiles. Clones 17A-19WK-11, -22, and -33 expressed RORC and TBX21 mRNA, as well as high levels (MFI ∼100) of CCR6 protein and moderate levels of CXCR3 and CCR4, consistent with a TH17/TH1 lineage. The 17A-21MO clones expressed high levels of GATA3, low levels of TBX21, low levels of CCR4 protein, and CXCR3 expression just above background; despite the relatively low CCR4 expression, this profile was consistent with a TH2 lineage. The 32A clones exhibited some heterogeneity, with various amounts of TBX21 and GATA3 mRNA. Expression of CXCR3 was lower on clones 32A-14 and 32A-21, consistent with their lower level expression of TBX21. Three of these clones also expressed low to moderate levels of CCR6, which was observed on only a fraction of the cells. Histogram plots showed 2 distinct populations for these clones, only one of which expressed CCR6 (data not shown), even though more than 95% of these cells were stained by DR0101 tetramers loaded with peptide FVIII2194-2213. This indicated that the 2 distinct populations revealed by CCR6 staining did not lose clonality during expansion. Overall, phenotypes for clones 32A-5, 32A-15, 32A-16, and 32A-18 indicated a TH1 polarization, whereas clones 32A-14 and 32A-21 showed a profile indicating a mixed TH1/TH2 lineage.
Figure 3.
Transcription factor mRNA and chemokine receptor expression for FVIII-specific T-cell clones. (A) TBX21, GATA3, and RORC mRNA expression in T-cell clones was quantified by real-time quantitative polymerase chain reaction using TaqMan gene expression assays. Expression levels were normalized to 18S rRNA expression. Mean values are shown for TBX21, GATA3, and RORC mRNAs, plotted as stacked bar graphs. Cycle threshold values were determined in triplicate for 2 to 3 samples of each clone. The samples were generated by separate expansions, except for T-cell clones 17A-19WK-10, -22, -33, 17A-21MO-12, and 32A-21, which were evaluated only once because they expanded poorly compared with the other clones. (B) CXCR3, CCR4, and CCR6 cell surface expression on T-cell clones was quantified by flow cytometry. The MFI indicating the specific antibody staining minus the isotype staining was calculated. Mean values from 2 to 3 independent experiments are shown, plotted as stacked bar graphs. Clones are grouped according to their cytokine secretion profiles. One example of the chemokine receptor-staining data is shown in the inset as a conventional histogram overlay (chemokine receptor antibodies, filled histogram; isotype control antibodies, open histogram) with fluorescent intensity plotted on the x-axis (logarithmic scale from 100 to 103 fluorescent units) and cell counts plotted on the y-axis, normalized as percentage of maximum (linear scale from 0%-100%). * indicates that the inset corresponds to clone 17A-19WK-22.
Discussion
The cloning of T cells restricted by the same HLA-DR allele from 2 brothers with mild hemophilia A, only one of whom developed a high-titer inhibitor, presented an opportunity to explore characteristics of human T-cell responses to a clinically relevant FVIII epitope. T cells from both subjects responded to one epitope that coincides with the A2201P missense substitution site.16 Distinct patterns of cytokine secretion, transcription factor mRNA, and chemokine receptor expression were seen for clones isolated from the proband at 2 time points, approximately 5 and 21 months after he developed an inhibitor. Clones isolated from his brother, who did not have a clinically significant inhibitor, showed yet another profile. Our results indicated that the TH lineages of FVIII-responsive cells changed over time after an initial inhibitor response, and a separate TH lineage was found for clones isolated from the subject who has maintained B-cell tolerance to FVIII.
To our knowledge, the clones described in this study are the first evidence that FVIII-specific TH17/TH1 cells are present in an inhibitor response. The detection of TH17/TH1 cells at 19 weeks, but not 21 months, suggests that TH17/TH1 lineage cells play a role in the early months of anti-FVIII antibody production, but that they were not involved in maintenance of long-term B-cell responses to FVIII. Interestingly, a study of T-cell responses in 2 unrelated subjects with mild hemophilia A due to missense substitution R593C, both of whom had a longstanding low-titer inhibitor after an earlier peak response, characterized FVIII-specific T-cell clones, and they did not secrete IL-17.17 Because these blood samples were obtained years after initial inhibitor development, this result is consistent with the notion that TH17/TH1-polarized cells may play a role in earlier stages of the immune response. Additional studies analyzing responses of T cells isolated from serial samples after an inhibitor response are needed to verify and properly interpret the discovery of this proinflammatory T-cell lineage in the present study.
TH17 cells play an important role in host defense against infection and in tissue inflammation by inducing the release of proinflammatory and neutrophil-mobilizing cytokines.12,13,21 The proband developed an inhibitor after receiving FVIII to support tonsillectomy/adenectomy, so TH17 cells having specificities for other antigens were most likely already present when he received the initial infusions with wild-type FVIII. IL-21 produced by TH17 cells is a powerful positive-feedback stimulant of TH17 cell differentiation.5,6,13 Thus, the cytokine environment may have promoted differentiation of naive T cells toward a TH17 polarization. Inflammation is a widely acknowledged risk factor for inhibitor development; therefore, it is reasonable to hypothesize that the TH17-polarized cells may contribute to the development of anti-FVIII antibodies.
TH17 and TH17/TH1 cells, once formed, have the capacity to stimulate antibody production and regulate humoral immune responses.22–27 In various mouse models of allergy and autoimmunity, IL-17 deficiency decreases germinal center B-cell development and antigen-specific antibody production, whereas high levels of IL-17 promote germinal center and antigen-specific antibody production.22–24,26,27 Human TH17 and TH17/TH1 clones have been shown to induce CD19+ B cells to produce IgM, IgG, and IgA, but not IgE when stimulated with anti-CD3 antibody.25 Human TH17 cells, but not TH1 and TH2 cells, secrete high levels of the B-cell chemoattractant CXC chemokine ligand 13.28
This is the first description of antigen-specific, HLA-restricted, TH17-polarized human clones isolated using culture conditions optimized for TH1- and TH2-polarized cells. Takagi et al28 produced human TH17 alloreactive and Candida albicans-specific clones by culturing CD4+ cells under TH17-polarizing conditions and cloning by limiting dilution. Streeck et al29 isolated TH17 and TH17/TH1 clones by sorting IL-17–secreting cells labeled with an IL-17 capture complex. In the present study, TH17/TH1 clones were isolated from CD4+ T cells by tetramer staining, followed by single-cell sorting and expansion in the presence of IL-2 and human serum. Their isolation under these conditions was somewhat surprising, because it has been reported that IL-2 inhibits TH17 polarization of murine T cells.30 Although 2 of the 3 TH17/TH1 clones (17A-19WK-22 and 17A-19WK-33) expanded less robustly in the presence of IL-2, expansion of clone 17A-19WK-11 was comparable with that of the TH2 and TH1/TH2 lineages described in this study (data not shown). The TH17/TH1 clones, however, had a greater tendency to form aggregates in culture (data not shown). Various culture conditions are now being tested to determine optimal conditions for expansion and maintenance of human TH17/TH1 clones.
The identification of FVIII-specific TH2 and TH1/TH2 cells in an inhibitor subject, and of TH1 cells in a hemophilic subject without an inhibitor, is consistent with an earlier report describing polyclonal CD4+ T-cell responses to FVIII by Hu et al.11 In another study, hemophilia A mice that were treated with anti-CD3 to prevent inhibitors showed a shift in antigen-specific cytokine production from a mixed TH1/TH2 response toward a TH1 polarization.31 Results of these recent studies and of the present investigation indicate that TH1 responses to FVIII can persist in hemophilic mice and humans without precipitating development of a high-titer inhibitor.
The TH lineages of the clones were determined by measuring cytokine secretion in response to antigen, and by measuring transcriptional regulators and surface chemokine receptor expression for resting clones. The mRNA expression levels of the lineage-specific transcriptional regulators TBX21, GATA3, and RORC correlated well with cytokine secretion patterns of the clones. The mRNA expression levels clearly marked the different cell types, particularly in the case of RORC, which was expressed only by clones that secreted IL-17 in response to antigen. The chemokine receptor expression patterns, in contrast, did not strictly correlate with cytokine secretion levels; in particular, CCR4 expression was not appreciably up-regulated on the TH2 clones and it was most pronounced on the TH17/TH1 cells. CCR6 was expressed on the clones that secreted IL-17, as expected, but it was also detected on 3 clones from the noninhibitor subject. Other studies have also noted that chemokine receptor expression is not strictly correlated with other TH lineage markers. Singh et al20 observed that CCR4 expression was up-regulated on human cord blood CD4+ T cells differentiated under TH17 conditions. Annunziato et al25 examined the expression of CCR4 and CCR6 mRNA in human T-cell clones and found that CCR6 expression was diagnostic of TH17 and TH17/TH1 clones, whereas CCR4 was expressed on cloned cells of various lineages. Acosta-Rodriguez et al32 showed that CCR6 is expressed on both IL-17 and IFN-γ-secreting human memory T cells from peripheral blood. The majority of these IL-17–secreting cells were CCR6+CCR4+, whereas IFN-γ–secreting cells were CCR6+CXCR3+ or CCR6−CXCR3+. Cells secreting both IL-17 and IFN-γ were either CCR6+CCR4+ or CCR6+CXCR3+. Overall, these studies suggest that CCR4 expression is not specific for TH2 cells, whereas CCR6 is generally expressed by IL-17–secreting cells, but may also be expressed by IFN-γ–secreting cells under certain conditions.
In conclusion, detailed phenotypic characterization of FVIII-specific T-cell clones isolated at earlier and later stages of an inhibitor response, and from a noninhibitor subject who nevertheless had a strong T-cell response to FVIII, indicated changes in T-cell lineages that accompany the progression of immune responses after FVIII stimulation. Additional time-course studies, including prospective studies following the differentiation of naive T cells to various TH lineages, will be necessary to confirm and extend the findings reported in this study. It will be particularly important to determine whether FVIII-specific TH17 cells are generally involved in initial stages of helper T-cell responses to FVIII replacement therapy, or whether they contribute to inhibitor development primarily when inflammation is present concurrent with FVIII administration.
Supplementary Material
Acknowledgments
We thank Charles Cooper for help with human subjects; Shelley Nakaya for DNA sequencing; Lonna Pells for assistance characterizing T-cell clones; Gerald T. Nepom for critical reading of the manuscript; and all subjects for blood donations.
This work was supported by a Bayer (Berlin) hemophilia award (to K.P.P.), a CSL Behring (King of Prussia, PA) hemophilia research award (to K.P.P.), National Institutes of Health (Bethesda, MD) Grant R01-HL 071093-01 (to A.R.T.), and National Institutes of Health Contract HHSN266200400028C (to W.W.K.).
Footnotes
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Authorship
Contribution: R.A.E. designed and performed the majority of the experiments, analyzed the data, and wrote the manuscript; E.A.J. isolated some T-cell clones, generated tetramers, and edited the manuscript; W.W.K. oversaw tetramer generation and edited the manuscript; A.R.T. oversaw the human subjects and edited the manuscript; and K.P.P. oversaw the study and wrote the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Kathleen P. Pratt, Puget Sound Blood Center, BRI Rm 3016, 921 Terry Ave, Seattle, WA 98104; e-mail: kathleenp@psbcresearch.org.
References
- 1.Ehrenforth S, Kreuz W, Scharrer I, et al. Incidence of development of factor VIII and factor IX inhibitors in haemophiliacs. Lancet. 1992;339:594–598. doi: 10.1016/0140-6736(92)90874-3. [DOI] [PubMed] [Google Scholar]
- 2.Lusher JM, Arkin S, Abildgaard CF, Schwartz RS. Recombinant factor VIII for the treatment of previously untreated patients with hemophilia A: safety, efficacy, and development of inhibitors: Kogenate Previously Untreated Patient Study Group. N Engl J Med. 1993;328:453–459. doi: 10.1056/NEJM199302183280701. [DOI] [PubMed] [Google Scholar]
- 3.Hay CR. Factor VIII inhibitors in mild and moderate-severity haemophilia A. Haemophilia. 1998;4:558–563. doi: 10.1046/j.1365-2516.1998.440558.x. [DOI] [PubMed] [Google Scholar]
- 4.Bray GL, Kroner BL, Arkin S, et al. Loss of high-responder inhibitors in patients with severe hemophilia A and human immunodeficiency virus type 1 infection: a report from the Multi-Center Hemophilia Cohort Study. Am J Hematol. 1993;42:375–379. doi: 10.1002/ajh.2830420408. [DOI] [PubMed] [Google Scholar]
- 5.Zhu J, Paul WE. CD4 T cells: fates, functions, and faults. Blood. 2008;112:1557–1569. doi: 10.1182/blood-2008-05-078154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Dong C. TH17 cells in development: an updated view of their molecular identity and genetic programming. Nat Rev Immunol. 2008;8:337–348. doi: 10.1038/nri2295. [DOI] [PubMed] [Google Scholar]
- 7.Abbas AK, Murphy KM, Sher A. Functional diversity of helper T lymphocytes. Nature. 1996;383:787–793. doi: 10.1038/383787a0. [DOI] [PubMed] [Google Scholar]
- 8.Stavnezer J, Amemiya CT. Evolution of isotype switching. Semin Immunol. 2004;16:257–275. doi: 10.1016/j.smim.2004.08.005. [DOI] [PubMed] [Google Scholar]
- 9.Pène J, Gauchat JF, Lecart S, et al. Cutting edge: IL-21 is a switch factor for the production of IgG1 and IgG3 by human B cells. J Immunol. 2004;172:5154–5157. doi: 10.4049/jimmunol.172.9.5154. [DOI] [PubMed] [Google Scholar]
- 10.Avery DT, Bryant VL, Ma CS, de Waal Malefyt R, Tangye SG. IL-21-induced isotype switching to IgG and IgA by human naive B cells is differentially regulated by IL-4. J Immunol. 2008;181:1767–1779. doi: 10.4049/jimmunol.181.3.1767. [DOI] [PubMed] [Google Scholar]
- 11.Hu G, Guo D, Key NS, Conti-Fine BM. Cytokine production by CD4+ T cells specific for coagulation factor VIII in healthy subjects and haemophilia A patients. Thromb Haemost. 2007;97:788–794. [PubMed] [Google Scholar]
- 12.Fouser LA, Wright JF, Dunussi-Joannopoulos K, Collins M. Th17 cytokines and their emerging roles in inflammation and autoimmunity. Immunol Rev. 2008;226:87–102. doi: 10.1111/j.1600-065X.2008.00712.x. [DOI] [PubMed] [Google Scholar]
- 13.Awasthi A, Kuchroo VK. Th17 cells: from precursors to players in inflammation and infection. Int Immunol. 2009;21:489–498. doi: 10.1093/intimm/dxp021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Jacquemin M, Vantomme V, Buhot C, et al. CD4+ T-cell clones specific for wild-type factor VIII: a molecular mechanism responsible for a higher incidence of inhibitor formation in mild/moderate hemophilia A. Blood. 2003;101:1351–1358. doi: 10.1182/blood-2002-05-1369. [DOI] [PubMed] [Google Scholar]
- 15.James EA, Kwok WW, Ettinger RA, Thompson AR, Pratt KP. T-cell responses over time in a mild hemophilia A inhibitor subject: epitope identification and transient immunogenicity of the corresponding self-peptide. J Thromb Haemost. 2007;5:2399–2407. doi: 10.1111/j.1538-7836.2007.02762.x. [DOI] [PubMed] [Google Scholar]
- 16.Ettinger RA, James EA, Kwok WW, Thompson AR, Pratt KP. HLA-DR-restricted T-cell responses to factor VIII epitopes in a mild haemophilia A family with missense substitution A2201P. Haemophilia. doi: 10.1111/j.1365-2516.2008.01905.x. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.James E, van Haren S, Ettinger R, et al. HLA-DRB1*1101-restricted T cells from two unrelated mild hemophilia A inhibitor subjects with missense genotype R593C respond to the same minimal epitope [abstract]. Blood. 2008;112:441. Abstract 1212. [Google Scholar]
- 18.Bonecchi R, Bianchi G, Bordignon PP, et al. Differential expression of chemokine receptors and chemotactic responsiveness of type 1 T helper cells (Th1s) and Th2s. J Exp Med. 1998;187:129–134. doi: 10.1084/jem.187.1.129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sallusto F, Lenig D, Mackay CR, Lanzavecchia A. Flexible programs of chemokine receptor expression on human polarized T helper 1 and 2 lymphocytes. J Exp Med. 1998;187:875–883. doi: 10.1084/jem.187.6.875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Singh SP, Zhang HH, Foley JF, Hedrick MN, Farber JM. Human T cells that are able to produce IL-17 express the chemokine receptor CCR6. J Immunol. 2008;180:214–221. doi: 10.4049/jimmunol.180.1.214. [DOI] [PubMed] [Google Scholar]
- 21.Kolls JK, Linden A. Interleukin-17 family members and inflammation. Immunity. 2004;21:467–476. doi: 10.1016/j.immuni.2004.08.018. [DOI] [PubMed] [Google Scholar]
- 22.Nakae S, Komiyama Y, Nambu A, et al. Antigen-specific T cell sensitization is impaired in IL-17-deficient mice, causing suppression of allergic cellular and humoral responses. Immunity. 2002;17:375–387. doi: 10.1016/s1074-7613(02)00391-6. [DOI] [PubMed] [Google Scholar]
- 23.Nakae S, Nambu A, Sudo K, Iwakura Y. Suppression of immune induction of collagen-induced arthritis in IL-17-deficient mice. J Immunol. 2003;171:6173–6177. doi: 10.4049/jimmunol.171.11.6173. [DOI] [PubMed] [Google Scholar]
- 24.Komiyama Y, Nakae S, Matsuki T, et al. IL-17 plays an important role in the development of experimental autoimmune encephalomyelitis. J Immunol. 2006;177:566–573. doi: 10.4049/jimmunol.177.1.566. [DOI] [PubMed] [Google Scholar]
- 25.Annunziato F, Cosmi L, Santarlasci V, et al. Phenotypic and functional features of human Th17 cells. J Exp Med. 2007;204:1849–1861. doi: 10.1084/jem.20070663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Bai Y, Liu R, Huang D, et al. CCL2 recruitment of IL-6-producing CD11b+ monocytes to the draining lymph nodes during the initiation of Th17-dependent B cell-mediated autoimmunity. Eur J Immunol. 2008;38:1877–1888. doi: 10.1002/eji.200737973. [DOI] [PubMed] [Google Scholar]
- 27.Hsu HC, Yang P, Wang J, et al. Interleukin 17-producing T helper cells and interleukin 17 orchestrate autoreactive germinal center development in autoimmune BXD2 mice. Nat Immunol. 2008;9:166–175. doi: 10.1038/ni1552. [DOI] [PubMed] [Google Scholar]
- 28.Takagi R, Higashi T, Hashimoto K, et al. B cell chemoattractant CXCL13 is preferentially expressed by human Th17 cell clones. J Immunol. 2008;181:186–189. doi: 10.4049/jimmunol.181.1.186. [DOI] [PubMed] [Google Scholar]
- 29.Streeck H, Cohen KW, Jolin JS, et al. Rapid ex vivo isolation and long-term culture of human Th17 cells. J Immunol Methods. 2008;333:115–125. doi: 10.1016/j.jim.2008.01.018. [DOI] [PubMed] [Google Scholar]
- 30.Laurence A, Tato CM, Davidson TS, et al. Interleukin-2 signaling via STAT5 constrains T helper 17 cell generation. Immunity. 2007;26:371–381. doi: 10.1016/j.immuni.2007.02.009. [DOI] [PubMed] [Google Scholar]
- 31.Waters B, Qadura M, Burnett E, et al. Anti-CD3 prevents factor VIII inhibitor development in hemophilia A mice by a regulatory CD4+CD25+-dependent mechanism and by shifting cytokine production to favor a Th1 response. Blood. 2009;113:193–203. doi: 10.1182/blood-2008-04-151597. [DOI] [PubMed] [Google Scholar]
- 32.Acosta-Rodriguez EV, Rivino L, Geginat J, et al. Surface phenotype and antigenic specificity of human interleukin 17-producing T helper memory cells. Nat Immunol. 2007;8:639–646. doi: 10.1038/ni1467. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.