Abstract
The Polycomb group protein enhancer of zeste homolog 2 (EZH2), which has roles during development of numerous tissues, is a critical regulator of cell type identity. Overexpression of EZH2 has been detected in invasive breast carcinoma tissue samples and is observed in human breast tissue samples of morphologically normal lobules up to 12 years before the development of breast cancer. The function of EZH2 during preneoplastic progression in the mammary gland is unknown. To investigate the role of EZH2 in the mammary gland, we targeted the expression of EZH2 to mammary epithelial cells using the mouse mammary tumor virus long terminal repeat. EZH2 overexpression resulted in aberrant terminal end bud architecture. By the age of 4 months, 100% of female mouse mammary tumor virus-EZH2 virgin mice developed intraductal epithelial hyperplasia resembling the human counterpart accompanied by premature differentiation of ductal epithelial cells and up-regulation of the luminal marker GATA-3. In addition, remodeling of the mammary gland after parturition was impaired and EZH2 overexpression caused delayed involution. Mechanistically, we found that EZH2 physically interacts with β-catenin, inducing β-catenin nuclear accumulation in mammary epithelial cells and activating Wnt/β-catenin signaling. The biological significance of these data to human hyperplasias is demonstrated by EZH2 up-regulation and colocalization with β-catenin in human intraductal epithelial hyperplasia, the earliest histologically identifiable precursor of breast carcinoma.
Breast cancer progresses through pathological stages starting with a deregulated epithelial intraductal proliferation, progressing into atypical hyperplasia, carcinoma in situ, and invasive carcinoma, which may develop metastasis.1 Compelling pathological and epidemiological studies have established that women with proliferative epithelial lesions have an increased risk for developing breast cancer.2 Intraductal epithelial hyperplasia is composed of differentiated cells that proliferate in a disorganized fashion inside breast ducts.1 The precise steps involved in the progression from intraductal epithelial hyperplasia to ductal carcinoma in situ and invasive carcinoma have not been elucidated.
Cell differentiation depends on expression and repression of specific genes through multiple cell divisions. The differential gene expression patterns that define a cell type need to be maintained during DNA replication and at mitosis so that the daughter cells retain the differentiated cell type of the parental cell. The Polycomb group proteins act in conjunction with the Trithorax group proteins to maintain repressed or active transcription states of developmentally regulated genes.3,4,5 Enhancer of zeste homolog 2 (EZH2) is an essential component of the Polycomb repressive complex.6,7 Recent studies have shown that EZH2 is also capable of transcriptional activation of genes, especially those involved in cell proliferation.3,4 EZH2 plays a fundamental role in cell differentiation during development,8 and its deregulation can give rise to preneoplastic lesions and carcinogenesis in various organs.3,9
Our laboratory has demonstrated that EZH2 protein is elevated in a group of invasive carcinomas of the breast and that it is an independent predictor of survival in women with breast cancer.10 Invasive carcinomas with high EZH2 expression are significantly more likely to recur and metastasize than tumors with low EZH2 expression. The importance of EZH2 up-regulation as an early event in mammary tumorigenesis is highlighted by the observation that low levels of EZH2 overexpression are detected in morphologically normal lobules up to 12 years before breast cancer develops and that the expression of EZH2 increases with increasing severity of epithelial atypias in human breast tissues.11
Although these studies in human breast samples strongly suggest that EZH2 may be involved in the progression from normal epithelium to hyperplasia, which increases the risk of breast cancer, direct evidence supporting this contention is lacking. To gain further insight into the causal relationship between EZH2 expression, mammary gland development, and preneoplastic progression, we have established mice carrying mouse mammary tumor virus (MMTV) long terminal repeat (LTR) Simian virus 40-driven EZH2 overexpression in the mammary gland. We discovered that specific overexpression of EZH2 in mammary epithelial cells led to intraductal epithelial hyperplasia and hyperbranching of mammary ducts with up-regulation and nuclear accumulation of β-catenin. We found that EZH2 physically interacts with β-catenin in vivo in intraductal hyperplastic cells of EZH2 transgenic mice and in human breast tissue samples. These data implicate EZH2 in the pathobiology of mammary intraductal hyperplasia.
Materials and Methods
Generation and Identification of Transgenic Mice
All procedures were conducted in accordance with the NIH Guide for the Care and Use of Laboratory Animals (http://www.nap.edu/openbook.php?record_id=5140, last accessed July 27, 2009) and were approved by the Institutional Animal Care and Use Committee of the University of Michigan. MMTV-EZH2 transgenic mice were developed by cloning a cDNA containing the mouse EZH2 into the EcoR1 site of plasmid p206, harboring the MMTV LTR and the polyadenylation sequence of the Simian virus 40 early region (kind gift of Dr. William Muller). The expression cassette was then prepared and injected into one-cell zygotes of FBV/N mice with the assistance of the University of Michigan Transgenic Animal Core. Five founders were obtained from the transgenic core. To identify transgenic animals, genomic DNA was isolated from 0.5 cm clippings of mouse tails and PCR-amplified using a primer that amplifies a portion of EZH2 and of the MMTV vector (forward primer: 5′-GACCACAGGATAGGCATCTTTGC′-3; reverse primer: 5′-GTCAGCAGTAGCCTCATCATC-3′) to generate a transgene-specific amplification product. PCR conditions included a 1-minute annealing step at 60°C and a 1-minute extension at 72°C for 35 cycles.
RNA Expression Analysis
To identify mice expressing the MMTV-EZH2 transgene, total mammary gland RNA was isolated using TRIzol (Invitrogen, Carlsbad, CA). Reverse transcription was performed using a reverse transcription system (Promega, San Luis Obispo, CA). Transgene expression was determined by RNase protection analysis, using a riboprobe specific for EZH2 (forward: 5′-TGAGGGTAGTCGACAGAAGTC-3′; reverse: 5′-GAAGGCACGGAAGCTCATGTG-3′). For normalization, glyceraldehyde-3-phosphate dehydrogenase was used as an internal control. In addition to EZH2, real-time RT-PCR was performed to investigate the levels of GATA-3 (forward: 5′-CGCTCCTTGCTACTCAGGTGAT-3′; reverse: 5′-GACCACACTGCACACTGATTC-3′) and β-catenin (forward: 5′-GCACACGAATGGATCACAAGA-3′; reverse: 5′-GCAAAGTCAGCACCACTACAGAT-3′).
Protein Extraction and Western Blot Analysis
Inguinal mammary gland samples were removed, homogenized in lysis buffer (50 mmol/L Tris, pH 7.4, 250 mmol/L NaCl, 5 mmol/L EDTA. 1% Nonidet P-40, and 0.1% SDS; protease inhibitor 1:100) and centrifuged at 16,000 × g (10 minutes, 4°C). Protein concentrations were determined using the Bio-Rad protein assay kit. Primary antibodies used included anti-EZH2 (612667, BD Biosciences, San Jose, CA), anti-GATA-3 (sc-268, Santa Cruz Biotechnology, Santa Cruz, CA), anti-β-catenin (610153, BD Biosciences), and anti-cyclin D1 (sc-8396, Santa Cruz Biotechnology). Verification of equal loading was done by staining the membranes with Ponceau S (Sigma-Aldrich, St. Louis, MO). This procedure was more accurate than quantification of cytoskeletal proteins because the mammary tissues exhibited variable expression of these proteins, which did not correlate with the measured protein concentrations or Ponceau S techniques, as has been reported by others.12,13,14 Semiquantitative protein expression levels were determined by densitometry using ImageJ 1.38x software.
Nuclear Fraction and Immunoprecipitation
Nuclear extraction was performed using NE-PER Nuclear and Cytoplasmic Extraction Reagents (78833, Pierce Chemical, Rockford, IL). Inguinal mammary gland samples were homogenized directly in CERI (500 μl of CERI to 50 mg of tissue) and incubated on ice for 10 minutes. Then 5.5 μl of ice-cold CERII per 100 μl of CERI was added, and the solution was mixed for 5 seconds on the highest setting and incubated on ice for 1 minute. The samples were centrifuged for 5 minutes at 16,000 × g, and the supernatant (cytoplasmic extract) was immediately transferred to a prechilled tube. The insoluble fraction was resuspended in 100 μl of ice-cold NER and centrifuged for 15 seconds. The sample was returned to ice and centrifugation was continued for 15 seconds every 10 minutes, for a total of 40 minutes. Final centrifugation was done at 16,000 × g for 10 minutes. The supernatant (nuclear extract) fraction was immediately transferred to a prechilled tube, and all extracts were stored at −80°C.
Immunoprecipitation was performed using protein G PLUS-agarose immunoprecipitation reagent (sc-2002, Santa Cruz Biotechnology). Mammary gland lysates (200 μg) of nuclear extracts were precleared by adding 1.0 μg of normal mouse IgG, together with 20 μl of resuspended volume of Protein G PLUS-Agarose. Anti-mouse antibodies (Amersham, Piscataway, NJ) were used as secondary antibodies. Blots were developed using ECL reagent and treated as indicated above for Western blotting. Samples were then incubated at 4°C for 30 minutes and centrifuged at 1000 × g for 5 minutes at 4°C. The supernatant was transferred to a new tube and incubated for 1 hour at 4°C with 2 μg of anti-EZH2 or anti-β-catenin antibodies and control normal mouse IgG. Subsequently, 20 μl of resuspended volume of Protein G PLUS-Agarose was added, and incubated overnight at 4°C on a rocker. Immunoprecipitates were collected by centrifugation at 1000 × g for 5 minutes at 4°C, and the supernatant was discarded. The pellet was washed 4 times with 1.0 ml of PBS, each time repeating the centrifugation step and resuspending the pellet in 40 μl of 1× electrophoresis sample buffer before the immunoprecipitation protein was subjected to immunoblotting. Anti-mouse antibodies (Amersham) were used as secondary antibodies. Blots were developed using ECL reagent and treated as indicated above for Western blotting.
Histological and Whole-Mount Analyses
Mouse mammary gland tissue samples were collected for whole-mount preparation or histological analysis. Mammary gland whole mounts were prepared by mounting the inguinal mammary fat pads on glass slides and processed as described.15 In brief, glands were excised, dehydrated, stained with alum carmine, and stored in methyl salicylate (Sigma-Aldrich). Mammary gland tissue samples for histological analysis were fixed overnight in 10% neutral buffered formalin (Sigma-Aldrich), processed through graded alcohols, cleared in xylene, and embedded in paraffin. Tissues were sectioned at 5 μm and placed on charged slides. Sections were then stained with hematoxylin and eosin.
Immunohistochemical and Immunofluorescence Analyses
Paraffin-embedded sections of the mammary glands were stained with hematoxylin and eosin or subjected to immunohistochemical analysis. Slides were deparaffinized in xylene and rehydrated through graded alcohols. We applied antibodies against EZH2 (1:75, BD Biosciences, San Jose, CA), GATA-3 (1:30, Santa Cruz Biotechnology, Santacruz, CA), E-cadherin (1:4500, BD Biosciences), cytokeratin-18 (1:100, Abcam, Cambridge, MA), proliferating cell nuclear antigen (1:100, DakoCytomation, Carpinteria, CA), p63 (1:100, NeoMarkers, Fremont, CA), β-catenin (1:400, BD Biosciences), and CD68 (1:600, AbD Serotec, Raleigh, NC). Staining was visualized with the DAB+ Kit (DakoCytomation) with the exception of EZH2, which was visualized with Permanent Red (DakoCytomation). Slides were counterstained in hematoxylin, blued in running tap water, dehydrated through graded alcohols, cleared in xylene, and mounted with Permount.
For immunofluorescence, paraffin-embedded tissue sections of five biopsy samples of human breast tissues containing hyperplasia and normal breast from reduction mammoplasties were sectioned at 5 μm and placed on charged slides. Slides were deparaffinized in xylene and rehydrated through graded alcohols. Heat-induced epitope retrieval with target retrieval, pH 6.0 (DakoCytomation) was performed in a microwave oven. Slides were incubated in Image-it FX signal enhancer (Invitrogen) for 30 minutes at room temperature. Rabbit monoclonal anti-β-catenin (1247-1, Epitomics, Burlingame, CA) diluted 1:600 and mouse monoclonal anti-EZH2 (612667, BD Biosciences) diluted 1:300 were combined together in a cocktail and applied simultaneously to the slide for 1.5 hours at room temperature in the dark. Antibodies were detected simultaneously at room temperature for 1 hour in the dark with a cocktail containing goat anti-mouse AF568 (Invitrogen) and donkey anti-rabbit AF488 (Invitrogen), both diluted 1:2000. Slides were rinsed in distilled water and cover-slipped with Prolong Gold antifade reagent with 4,6-diamidino-2-phenylindole (Invitrogen).
Terminal Deoxynucleotidyl Transferase-Mediated dUTP Nick-End Labeling Assay
Sections of mammary glands from EZH2 transgenic and wild-type mice at involution days 1, 3, 4, 7, and 14 (five mammary glands per group per time point) were subjected to terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling (TUNEL) staining. This was performed by using the DeadEnd Fluorometric TUNEL system (TB235, Promega). After staining, the sections were mounted with FluorSave mounting reagent (Calbiochem, San Diego, CA) containing 1 μg/ml 4,6-diamidino-2-phenylindole and observed by fluorescence microscopy (IX-70, Olympus, Melville, NY). The number of TUNEL-positive epithelial cells in 10 microscopic fields at 40× were counted per slide. The number of TUNEL-positive cells was normalized to the total number of epithelial cells in each microscopic field.
Results
Mammary Gland Tissue Targeted Overexpression of EZH2 Causes Aberrant Terminal End Buds and Promotes Ductal Branching
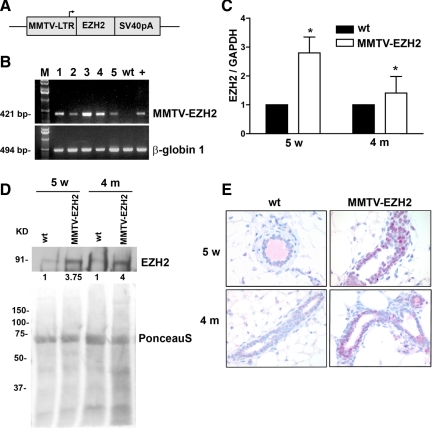
To assess the effects of elevated EZH2 expression in the development of preneoplastic alterations in mammary epithelial cells in vivo, one-cell mouse zygotes of the FBV/N strain were microinjected with an expression cassette in which the full-length cDNA of mouse EZH2 was placed under the transcriptional control of the MMTV LTR (Figure 1A). Five founder mice were obtained (Figure 1B). Guided by our studies in human breast tissues showing that various levels of EZH2 overexpression in morphologically normal breast epithelial cells are associated with an increased risk for development of invasive carcinoma, we chose a transgenic line with intermediate EZH2 copy number for subsequent experiments (Figure 1B, transgenic line 1).
Figure 1.
Targeted expression of EZH2 to the mammary gland of transgenic mice. A: Expression cassette used for the generation of MMTV-EZH2 mice. The mouse EZH2 cDNA followed by Simian virus 40 polyadenylation fragment (SV40pA) was inserted downstream of MMTV LTR, which was used to drive selective targeted expression to the mammary gland. B: Genotyping of the EZH2 transgenic mice by PCR analysis confirmed the presence of MMTV-EZH2 in five lines of transgenic mice (lanes labeled 1 to 5). The wild-type (wt) lane represents the negative control. The + lane is the one-copy standard positive control. β-Globin 1 serves as a loading control. C: Real-time PCR analysis shows elevated EZH2 mRNA levels in MMTV-EZH2 mammary glands at puberty (5 weeks [5w]) and in virgin mice at 4 months (4 m) of age compared with wild-type mice (n = 5 mice/group). Samples were prepared in quadruplicate. and the experiment was repeated three times. Student’s t-test, *P < 0.05. Error bars represent SD. D: Immunoblot analysis of EZH2 expression in mammary glands of MMTV-EZH2 transgenic mice and wild-type mice (n = 5 mice/group) at puberty (5 weeks) and virgin mice at 4 months. Ponceau S staining is the loading control. The number beneath each band indicates the fold change in intensity of the corresponding band relative to the control. E: Immunohistochemical analysis shows elevated levels of EZH2 protein in the nuclei of mammary epithelial cells of MMTV-EZH2 mice compared with rare expression of EZH2 in wild-type mice at 5 weeks and 4 months of age virgin mice. Original magnification, ×400.
Real-time PCR and Western blot analysis of mammary glands showed that MMTV-EZH2 mice exhibit increased EZH2 mRNA and protein expression in the mammary epithelium at different ages compared with the low endogenous levels of EZH2 in wild-type mice (Figure 1, C and D). Immunohistochemical staining verified that EZH2 protein was elevated and restricted to the nuclei of mammary epithelial cells of MMTV-EZH2 compared with low or absent EZH2 expression in wild-type glands (Figure 1E).
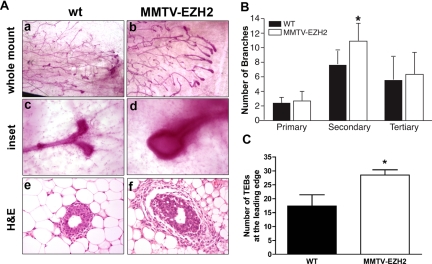
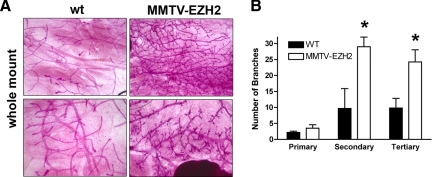
To investigate whether mammary epithelium-specific overexpression of EZH2 could perturb mammary gland development, we performed whole-mount analyses on the mammary glands of virgin female transgenic mice at various ages (Figure 2A). At 5 weeks of age, MMTV-EZH2 mammary glands had increased secondary ductal branches as well as more numerous and larger terminal end buds (TEBs) (Figure 2, B and C). Whereas TEBs of wild-type mice showed smooth contours and bifurcated dichotomously, TEBs of MMTV-EZH2 mice were hypertrophic and hyperplastic (Figure 2A). By 4 months of age all virgin MMTV-EZH2 transgenic mice had a phenotype consisting of ductal hyperplasia, with significantly increased numbers of secondary and tertiary ductal branches (Figure 3, A and B).
Figure 2.
EZH2 overexpression disrupts TEB architecture and branching in 5-week-old nulliparous mice. A: Whole mounts of inguinal mammary glands stained with alum carmine from 5-week-old virgin mice showing aberrant budding and hypercellular TEBs of MMTV-EZH2 mice compared with wild-type (wt) mice (a–d). Higher magnification (×400) showing that whereas wild-type TEBs are small and rounded and undergo dichotomous branching, MMTV-EZH2 TEBs are enlarged and hypercellular and fail to dichotomously branch (c and d). H&E-stained sections show that MMTV-EZH2 TEBs are larger than wild-type TEBs (e and f). B: Bar graph of branch point analysis reveals that MMTV-EZH2 mice have increased secondary branches compared with wild-type virgin mice at 5 weeks of age (n = 5 in each group, Student’s t-test, *P < 0.002, error bars = SD). C: Bar graph showing increased number of TEBs at the leading edge in MMTV-EZH2 mice compared with wild-type mice (each group n = 5, Student’s t-test, * P < 0.05, error bars = SD).
Figure 3.
The MMTV-EZH2 transgene induces precocious hyperplasia in nulliparous female transgenic mice. A: Whole mount of mammary glands from 4-month-old nulliparous female wild-type (wt) and MMTV-EZH2 mice showing ductal hyperplasia in the MMTV-EZH2 glands. Note the increased numbers of secondary and tertiary ductal branches and larger TEBs in MMTV-EZH2 mice. B: MMTV-EZH2 mice have increased secondary and tertiary branches compared with wild-type virgin mice at 4 months of age (n = 5 in each group, Student’s t-test, *P < 0.001 for both, error bars = SD).
EZH2 Induces Hyperplasia of Differentiated Luminal Epithelial Cells
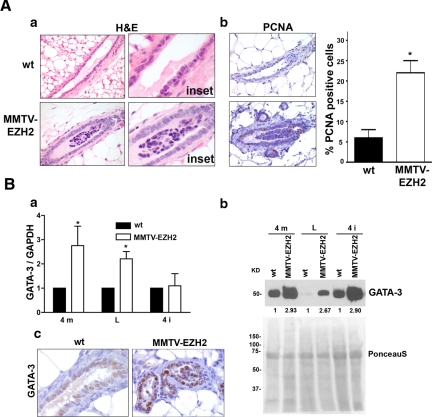
We next examined the ability of EZH2 overexpression to cause intraductal epithelial hyperplasia. Histological assessment of mature ducts of virgin 4-month-old mice revealed epithelial intraductal hyperplasia with formation of micropapillary structures, which contrasted with the single-layer polarized columnar epithelium of wild-type mice (Figure 4, A,a). EZH2 overexpression led to increased proliferating cell nuclear antigen-positive epithelial cells (Figure 4, A, b). The hyperplasia described in MMTV-EZH2 transgenic mice resembles the epithelial hyperplasia of the usual type that occurs in the human breast.
Figure 4.
EZH2 overexpression induces intraductal epithelial hyperplasia. A, a: H&E-stained mammary gland tissue sections of virgin MMTV-EZH2 and wild-type (wt) mice at 4 months of age. Mammary specific expression of the MMTV-EZH2 transgene induces a multilayered and disorganized ductal epithelium with formation of epithelial tufts. b: Proliferating cell nuclear antigen (PCNA) immunostaining of 4-month old MMTV-EZH2 and wild-type (wt) mammary glands showing that EZH2 up-regulation increases the proliferation of epithelial cells. Error bars indicate mean ± SD, Student’s t-test, *P < 0.001. B, a: Real-time PCR analysis shows elevated GATA-3 mRNA levels in MMTV-EZH2 mammary glands compared with wild-type mice. 4 m, 4 month; L, lactating; 4 I, involution day 4. Samples were prepared in quadruplicate, and the experiment was repeated three times. Student’s t-test, *P < 0.05, error bars = SD. b: Immunoblot analysis of GATA-3 expression level in MMTV-EZH2 transgenic and wild-type mice mammary tissues. Ponceau S staining is used as the loading control. The number underneath each band indicates the fold change in intensity of the corresponding band relative to the control. c: Immunohistochemical staining for GATA-3 in mature ducts of 4-month-old virgin wild-type and MMTV-EZH2 mice. Note that GATA-3 is up-regulated in the nuclei of epithelial cells of EZH2 transgenic mice compared with those of wild-type mice.
The proliferating cells in MMTV-EZH2 mammary glands exhibited a phenotype of differentiated luminal epithelial cells, because they expressed E-cadherin and cytokeratin-18 (Supplemental Figure S1, see http://ajp.amjpathol.org), as well as the luminal differentiation marker, GATA-3. As shown in Figure 4B, GATA-3 mRNA and protein levels were up-regulated in MMTV-EZH2 mice at different stages of life. Immunohistochemical staining shows elevated GATA-3 protein in the nuclei of hyperplastic epithelial cells of MMTV-EZH2 mice compared with wild-type mice (Figure 4, B, c).
EZH2 Overexpression Leads to Delayed Mammary Gland Involution
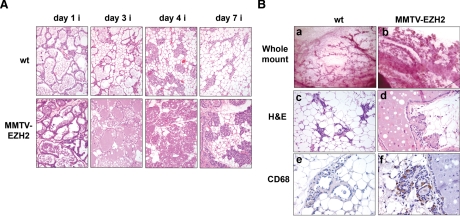
Although no defects in lactation were observed and pups nursed normally, EZH2-overexpressing mammary glands had defective involution. To make a valid comparison of mammary glands during the involution process, 4-month-old mothers during their first pregnancy were chosen. Pups were removed from EZH2-expressing and wild-type mothers directly after parturition, and the process of involution was studied at multiple time points as illustrated in Figure 5. Figure 5A shows the altered involution of MMTV-EZH2 mammary glands compared with wild-type glands. By day 7 of involution, mammary glands of wild-type mice showed collapsed alveolar structures and adipocytes became the most prominent cell type. In contrast, MMTV-EZH2 glands remained secretory with scant adipose cells (Figure 5A).
Figure 5.
Targeted overexpression of EZH2 impairs mammary gland involution. A: H&E-stained sections of the mammary glands of wild-type and MMTV-EZH2 mice at the indicated day of involution (i) (n = 5 mice/group/day were analyzed). Note the persistence of amorphous debris and secretory lobules in the mammary glands of MMTV-EZH2 mice at all phases of involution. B, a and b: Whole-mount carmine-stained mammary glands from EZH2 overexpressing mice at day 14 of involution retained epithelial clusters and enlarged ducts filled with membranous materials (arrows). c and d: H&E-stained sections of MMTV-EZH2 and wild-type mammary glands show that although wild-type glands underwent complete involution at day 14, MMTV-EZH2 retained abnormally dilated ducts with eosinophilic proteinaceous material. e and f: CD68 staining by immunohistochemistry reveals that MMTV-EZH2 mammary glands at day 14 of involution have multiple macrophages indicative of persistent involution, whereas there are no remaining macrophages in the wild-type gland.
The duct structure was also different in MMTV-EZH2 and wild-type mice. At day 14 of involution whole-mount staining of the mammary glands showed that MMTV-EZH2 glands had enlarged and distended ducts compared with those of wild-type mice (Figure 5B, a and b). At this time, histological analysis revealed that whereas wild-type mammary glands underwent complete remodeling to resemble the mature virgin gland, MMTV-EZH2 glands contained enlarged ducts filled with dense eosinophilic inspissated milk protein (Figure 5B, c and d). MMTV-EZH2 mammary glands retained foci of secretory alveolar structures with intracellular vesicles indicating continued secretory activity, and numerous macrophages (Figure 5B, e and f). No differences in apoptosis were observed between EZH2-overexpressing and wild-type involuting mammary glands as revealed by the TUNEL assay performed at involution days 1, 3, 4, 7, and 14 (Supplemental Figure S2, see http://ajp.amjpathol.org). These data demonstrate that sustained overexpression of EZH2 impairs the ability of the mammary glands to involute and regenerate after cessation of lactation.
EZH2 Interacts with β-Catenin in Vivo
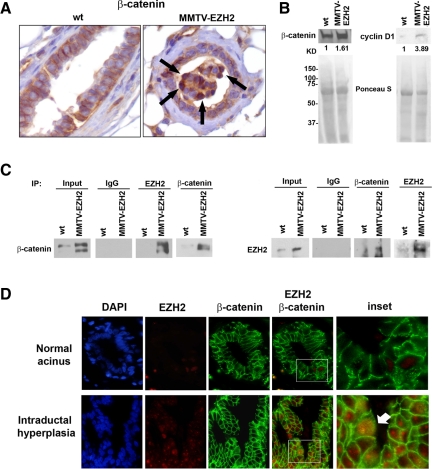
We noted that the intraductal epithelial hyperplasia induced by EZH2 overexpression was similar to that occurring in the human breast. This hyperplasia also had elements of those reported in Wnt transgenic mice.16,17,18 We hypothesized that EZH2 may promote mammary hyperplasia by activating Wnt/β-catenin signaling. Quantitative real-time RT-PCR revealed that MMTV-EZH2 mammary glands had increased β-catenin mRNA levels compared with those of wild-type glands (Supplemental Figure S3, see http://ajp.amjpathol.org). By immunohistochemical and immunoblot analyses EZH2 overexpression resulted in accumulation of β-catenin in the cytoplasm and nuclei of hyperplastic epithelial cells (Figure 6, A and B). In contrast, β-catenin protein was predominantly localized to the cellular membranes of wild-type glands (Figure 6A). Providing further support for the effect of EZH2 on β-catenin up-regulation and activation, the β-catenin target gene cyclin D1 was up-regulated in EZH2-overexpressing mammary glands compared with wild-type glands (Figure 6B). Of note, reciprocal immunoprecipitation using β-catenin or EZH2 antibodies revealed a direct association between the two proteins in MMTV-EZH2 mammary glands but not in wild-type glands (Figure 6C).
Figure 6.
EZH2 binds to β-catenin and induces β-catenin nuclear accumulation. A: Immunohistochemical staining for β-catenin protein in the mammary glands of 4 months virgin mice. Note the accumulation of β-catenin in the nucleus of hyperplastic epithelial cells of EZH2 transgenic mice (arrows) that contrasts with the crisp membrane β-catenin in wild-type glands (×400). B: Immunoblot analysis for β-catenin and cyclin D1 in MMTV-EZH2 and wild-type mammary glands. Nuclear and total protein lysates were prepared from 4-month-old virgin mice. MMTV-EZH2 glands show up-regulation of nuclear β-catenin and total cyclin D1 levels compared with wild-type glands. Ponceau S staining shows equal loading. The number beneath each band indicates the fold change in intensity of the corresponding band relative to the control. C: EZH2 interacts with β-catenin in mammary gland tissues from 4-month-old virgin mice. Immunoprecipitation (IP) of EZH2 or β-catenin followed by reciprocal EZH2 or β-catenin immunoblotting from nuclear extracted lysates of EZH2 transgenic and wild-type mammary glands. Inputs represented 10% of extracts. Immunoprecipitation with IgG was used as the negative control and immunoprecipitation with EZH2 and β-catenin served as the positive controls. D: Immunofluorescence of human breast tissue samples. EZH2 and β-catenin proteins colocalize in the nuclei of several hyperplastic epithelial cells but not in normal acini. Intraductal epithelial hyperplasia exhibits up-regulation of EZH2 in the nuclei as well as colocalization of EZH2 and β-catenin proteins in the nuclei of hyperplastic epithelial cells (arrow). EZH2 is red, β-catenin is green, colocalized proteins are yellow, and 4,6-diamidino-2-phenylindole (DAPI) stains the nuclei blue. Original magnification, ×600.
EZH2 and β-Catenin Colocalize in Human Intraductal Hyperplasia Tissue Samples
To determine whether the connection between EZH2 and the β-catenin signaling pathway has biological relevance, we assessed their expression by immunofluorescence on human breast tissue samples. Figure 6D shows that EZH2 protein is up-regulated in intraductal hyperplasia compared with normal non-hyperplastic acini. Whereas β-catenin is principally localized to the cellular membranes of epithelial cells in normal acini, we observed that several hyperplastic cells exhibited cytoplasmic and nuclear β-catenin accumulation, indicative of activation.19 EZH2 and β-catenin proteins colocalized in the nuclei of cells in hyperplastic lesions but not in normal acini. Collectively, these data show that EZH2 interacts directly with β-catenin in the nuclei of human epithelial hyperplastic cells in tissue samples, which may underlie the effect of EZH2 in the development of epithelial hyperplasia.
Discussion
To evaluate the causal relationship between EZH2 gene expression, breast tissue morphogenesis, and preneoplastic hyperplasia, we generated mice overexpressing EZH2 in their mammary glands. The initial morphological abnormalities exhibited by virgin MMTV-EZH2 mice were the appearance of increased numbers of enlarged and disorganized TEBs with an abnormal branching pattern, which contrasted with the delicate TEBs of wild-type mice that bifurcate dichotomously.
At 5 weeks of age, MMTV-EZH2 mice had increased ductal branching, which became more prominent by 4 months. In addition to exaggerated ductal branching, MMTV-EZH2 mammary glands exhibited intraductal epithelial hyperplasia recapitulating the histological features of usual intraductal epithelial hyperplasia in human breast tissue samples. These consisted of piled-up and disorganized proliferating epithelial cells forming tufts and micropapillae. Similar to human hyperplasia, the EZH2-induced hyperplasia was composed principally of differentiated luminal cells expressing high levels of the GATA-3 transcription factor, essential for specification and maintenance of the luminal cell fate in the mammary gland.20
We found that MMTV-EZH2 mammary glands had a delay in the process of involution compared with wild-type glands, which persisted up to 14 days after cessation of lactation. It is intriguing that the alterations in the involution process observed in MMTV-EZH2 mice were not associated with a decrease in apoptosis. This result is not surprising, however, based on previous studies by our group as well as those of other investigators, who failed to demonstrate an effect of EZH2 in apoptosis using cell line models.21 Given that EZH2 does not influence apoptosis during involution, EZH2 might influence the involution process by inducing premature cellular differentiation of epithelial cells in the mammary gland leading to the reduced plasticity needed to regenerate and remodel the mammary gland after cessation of lactation. This hypothesis is supported by our data showing that EZH2 overexpression results in hyperplasia of differentiated luminal epithelial cells expressing cytokeratin-18, E-cadherin, and GATA-3 proteins. The defective involution caused by EZH2 overexpression in the mammary gland provides a possible mechanism to explain the reported association between EZH2 protein up-regulation in human breast tissues and an increased breast cancer risk.11 This link is further strengthened by recent epidemiological studies showing that in humans lobular involution is associated with reduced risk of breast cancer and that aberrantly involuted lobules may predispose to breast carcinogenesis.22
In humans and mice, the development of carcinomas is preceded by an expansion of cells with the ability to initiate preneoplastic progression.23 Women with intraductal epithelial hyperplasia have a greater risk of developing breast cancer than women without this abnormality.2 We have found that EZH2 protein increases during hyperplasia and preneoplastic progression to atypical hyperplasia and carcinoma in human breast tissue samples.10,11 The mechanism by which EZH2 promotes ductal and epithelial hyperplasia is unknown. Our data suggest that EZH2 may induce mammary hyperplasia through up-regulation and activation of β-catenin signaling. The Wnt/β-catenin signaling pathway plays a central role in mammary gland growth and differentiation. Transgenic mice overexpressing several Wnt factors and an activated form of β-catenin develop lobuloalveolar hyperplasia followed by focal carcinomas.16,17,18 At this time, we have not observed mammary tumors in the MMTV-EZH2 transgenic mice. Further work is needed to determine with certainty whether EZH2 overexpression leads to mammary tumorigenesis and whether additional specific oncogenic events are necessary to develop carcinoma in the setting of EZH2 overexpression.
Consistent with our data, Shi et al24 recently reported that EZH2 interacts with β-catenin and activates the transcription of c-Myc and cyclin D1 using an in vitro system. However, the in vivo relationship between EZH2 and β-catenin and the biological relevance to human hyperplasias was unknown. We provide the first in vivo evidence that EZH2 binds to β-catenin and that it regulates β-catenin expression and nuclear accumulation in mammary epithelial cells. Detection of β-catenin protein in the nucleus and/or cytoplasm is a hallmark of active Wnt signaling.19 EZH2-overexpressing acini showed accumulation of β-catenin protein in the nuclei of several mammary epithelial cells, whereas β-catenin was localized to the epithelial cell membranes of wild-type glands. The direct interaction between EZH2 and β-catenin in the nuclei of hyperplastic cells resulted in up-regulation of cyclin D1. Our studies in human breast tissues show that normal human breast acini exhibit low to absent EZH2 protein and crisp membranous localization of β-catenin. Human epithelial hyperplasia shows up-regulation of EZH2 and nuclear accumulation of β-catenin. Furthermore, β-catenin and EZH2 co-localize exclusively in the nuclei of hyperplastic cells. These data provide a likely mechanistic explanation for the function of EZH2 in the development of intraductal epithelial hyperplasia in the human breast.10,11
In summary, targeted EZH2 overexpression to the mammary gland induces abnormal TEB development and precocious epithelial intraductal hyperplasia histopathologically similar to that of the human breast and with similarities to the hyperplasia triggered by Wnt/β-catenin overexpression. Mechanistically, our data establish the first in vivo link between EZH2 and β-catenin activation during mammary gland development and demonstrate its biological significance in the process of mammary epithelial hyperplasia.
Supplementary Material
Acknowledgments
We thank Dr. Gabriel Nuñez and members of the Kleer Laboratory for critical reading and helpful suggestions and Dr. William Muller (McGill University) for kindly providing the MMTV-LTR vector. We thank Robin Kunkel for assistance with art work. We also thank the Oberman family for the Harold A. Oberman Collegiate Professorship in Pathology, which supports Dr. Kleer’s work.
Footnotes
Address reprint requests to Dr. Celina G. Kleer, University of Michigan Medical School, Department of Pathology, 4217 Comprehensive Cancer Center, 1500 E. Medical Center Dr., Ann Arbor, MI 48109. E-mail: kleer@umich.edu.
Supported by NIH grants CA090876 (to C.G.K.), CA107469 (to C.G.K.), CA77612 (to S.D.M.), a grant from the Avon Foundation (to C.G.K.), and support from the Fashion Footwear Charitable Foundation of New York/QVC Presents Shoes on Sale™.
Supplemental material for this article can be found on http://ajp.amjpathol.org.
References
- Rosen PP. Rosen PP, editor. Philadelphia: Lippincott Williams & Wilkins,; Rosen’s Breast Pathology. 2008:pp 201–248. [Google Scholar]
- Hartmann LC, Sellers TA, Frost MH, Lingle WL, Degnim AC, Ghosh K, Vierkant RA, Maloney SD, Pankratz VS, Hillman DW, Suman VJ, Johnson J, Blake C, Tlsty T, Vachon CM, Melton LJ, 3rd, Visscher DW. Benign breast disease and the risk of breast cancer. N Engl J Med. 2005;353:229–237. doi: 10.1056/NEJMoa044383. [DOI] [PubMed] [Google Scholar]
- Laible G, Wolf A, Dorn R, Reuter G, Nislow C, Lebersorger A, Popkin D, Pillus L, Jenuwein T. Mammalian homologues of the Polycomb-group gene Enhancer of zeste mediate gene silencing in Drosophila heterochromatin and at S. cerevisiae telomeres. EMBO J. 1997;16:3219–3232. doi: 10.1093/emboj/16.11.3219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ringrose L, Paro R. Epigenetic regulation of cellular memory by the Polycomb and Trithorax group proteins. Annu Rev Genet. 2004;38:413–443. doi: 10.1146/annurev.genet.38.072902.091907. [DOI] [PubMed] [Google Scholar]
- Satijn DP, Otte AP. RING1 interacts with multiple Polycomb-group proteins and displays tumorigenic activity. Mol Cell Biol. 1999;19:57–68. doi: 10.1128/mcb.19.1.57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lund AH, van Lohuizen M. Polycomb complexes and silencing mechanisms. Curr Opin Cell Biol. 2004;16:239–246. doi: 10.1016/j.ceb.2004.03.010. [DOI] [PubMed] [Google Scholar]
- Otte AP, Kwaks TH. Gene repression by Polycomb group protein complexes: a distinct complex for every occasion? Curr Opin Genet Dev. 2003;13:448–454. doi: 10.1016/s0959-437x(03)00108-4. [DOI] [PubMed] [Google Scholar]
- LaJeunesse D, Shearn A. Trans-regulation of thoracic homeotic selector genes of the Antennapedia and bithorax complexes by the trithorax group genes: absent, small, and homeotic discs 1 and 2. Mech Dev. 1995;53:123–139. doi: 10.1016/0925-4773(95)00430-0. [DOI] [PubMed] [Google Scholar]
- Jacobs JJ, van Lohuizen M. Polycomb repression: from cellular memory to cellular proliferation and cancer. Biochim Biophys Acta. 2002;1602:151–161. doi: 10.1016/s0304-419x(02)00052-5. [DOI] [PubMed] [Google Scholar]
- Kleer CG, Cao Q, Varambally S, Shen R, Ota I, Tomlins SA, Ghosh D, Sewalt RG, Otte AP, Hayes DF, Sabel MS, Livant D, Weiss SJ, Rubin MA, Chinnaiyan AM. EZH2 is a marker of aggressive breast cancer and promotes neoplastic transformation of breast epithelial cells, Proc Natl Acad Sci USA. 2003;100:11606–11611. doi: 10.1073/pnas.1933744100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ding L, Erdmann C, Chinnaiyan AM, Merajver SD, Kleer CG. Identification of EZH2 as a molecular marker for a precancerous state in morphologically normal breast tissues. Cancer Res. 2006;66:4095–4099. doi: 10.1158/0008-5472.CAN-05-4300. [DOI] [PubMed] [Google Scholar]
- Adam L, Vadlamudi R, Kondapaka SB, Chernoff J, Mendelsohn J, Kumar R. Heregulin regulates cytoskeletal reorganization and cell migration through the p21-activated kinase-1 via phosphatidylinositol-3 kinase. J Biol Chem. 1998;273:28238–28246. doi: 10.1074/jbc.273.43.28238. [DOI] [PubMed] [Google Scholar]
- Landesman-Bollag E, Romieu-Mourez R, Song DH, Sonenshein GE, Cardiff RD, Seldin DC. Protein kinase CK2 in mammary gland tumorigenesis. Oncogene. 2001;20:3247–3257. doi: 10.1038/sj.onc.1204411. [DOI] [PubMed] [Google Scholar]
- Perfetti V, Dragani TA, Paulli M, Castelli G, Bellotti V, Gobbi PG, Merlini G. Gene expression of pyrogenic cytokines in Hodgkin’s disease lymph nodes. Haematologica. 1992;77:221–225. [PubMed] [Google Scholar]
- Jones FE, Jerry DJ, Guarino BC, Andrews GC, Stern DF. Heregulin induces in vivo proliferation and differentiation of mammary epithelium into secretory lobuloalveoli. Cell Growth Differ. 1996;7:1031–1038. [PubMed] [Google Scholar]
- Imbert A, Eelkema R, Jordan S, Feiner H, Cowin P. ΔN89 β-catenin induces precocious development, differentiation, and neoplasia in mammary gland. J Cell Biol. 2001;153:555–568. doi: 10.1083/jcb.153.3.555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lane TF, Leder P. Wnt-10b directs hypermorphic development and transformation in mammary glands of male and female mice. Oncogene. 1997;15:2133–2144. doi: 10.1038/sj.onc.1201593. [DOI] [PubMed] [Google Scholar]
- Tsukamoto AS, Grosschedl R, Guzman RC, Parslow T, Varmus HE. Expression of the int-1 gene in transgenic mice is associated with mammary gland hyperplasia and adenocarcinomas in male and female mice. Cell. 1988;55:619–625. doi: 10.1016/0092-8674(88)90220-6. [DOI] [PubMed] [Google Scholar]
- Wu R, Zhai Y, Fearon ER, Cho KR. Diverse mechanisms of β-catenin deregulation in ovarian endometrioid adenocarcinomas. Cancer Res. 2001;61:8247–8255. [PubMed] [Google Scholar]
- Kouros-Mehr H, Slorach EM, Sternlicht MD, Werb Z. GATA-3 maintains the differentiation of the luminal cell fate in the mammary gland. Cell. 2006;127:1041–1055. doi: 10.1016/j.cell.2006.09.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Varambally S, Dhanasekaran SM, Zhou M, Barrette TR, Kumar-Sinha C, Sanda MG, Ghosh D, Pienta KJ, Sewalt RG, Otte AP, Rubin MA, Chinnaiyan AM. The polycomb group protein EZH2 is involved in progression of prostate cancer. Nature. 2002;419:624–629. doi: 10.1038/nature01075. [DOI] [PubMed] [Google Scholar]
- Milanese TR, Hartmann LC, Sellers TA, Frost MH, Vierkant RA, Maloney SD, Pankratz VS, Degnim AC, Vachon CM, Reynolds CA, Thompson RA, Melton LJ, 3rd, Goode EL, Visscher DW. Age-related lobular involution and risk of breast cancer. J Natl Cancer Inst. 2006;98:1600–1607. doi: 10.1093/jnci/djj439. [DOI] [PubMed] [Google Scholar]
- Dontu G, Liu S, Wicha MS. Stem cells in mammary development and carcinogenesis: implications for prevention and treatment. Stem Cell Rev. 2005;1:207–213. doi: 10.1385/SCR:1:3:207. [DOI] [PubMed] [Google Scholar]
- Shi B, Liang J, Yang X, Wang Y, Zhao Y, Wu H, Sun L, Zhang Y, Chen Y, Li R, Zhang Y, Hong M, Shang Y. Integration of estrogen and Wnt signaling circuits by the polycomb group protein EZH2 in breast cancer cells. Mol Cell Biol. 2007;27:5105–5119. doi: 10.1128/MCB.00162-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.