Abstract
Mice lacking estrogen receptor alpha in the pituitary gonadotroph (PitEsr1KO) were generated to determine the physiologic role of pituitary estrogen signaling in the reproductive axis. PitEsr1KO female mice are subfertile or infertile and have elevated levels of serum luteinizing hormone (LH) and LH beta subunit gene expression, reflecting a lack of estrogen negative feedback effect on the gonadotroph. While serum LH values are elevated in PitEsr1KO mice, the degree of elevation is much less than that observed in ESR1-null mice, indicating that the hypothalamus must also have an important role in estrogen negative feedback. PitEsr1KO mice also demonstrate a defect in estrogen positive feedback, as surge LH values and estrous cyclicity are absent in these mice. Although sex steroid feedback in the reproductive axis is thought to involve discrete anatomic regions that mediate either a positive or negative estrogen effect, PitEsr1KO mice demonstrate novel evidence that localizes both estrogen positive feedback and estrogen negative feedback to the gonadotroph, which suggests that they may be mechanistically related.
Keywords: anterior pituitary, Cre/loxP, estradiol, estradiol receptor, gonadotropin-releasing hormone, luteinizing hormone, ovary, ovulatory cycle
Deletion of the ER alpha in the pituitary gonadotroph results in impaired regulation of both estrogen negative and positive feedback.
INTRODUCTION
Neuroendocrine control of ovulation is mediated by pulsatile secretion of the decapeptide gonadotropin-releasing hormone (GnRH), which binds to receptors on gonadotrophs. This results in synthesis and secretion of luteinizing hormone (LH), which then directs gonadal steroidogenesis. Estradiol (E2) in turn exerts negative feedback action at the level of the hypothalamus and pituitary to alter GnRH and LH synthesis and secretion. In rodent models, estrogen secretion results in an abrupt release of GnRH in proestrus and an LH surge that triggers ovulation. Thus, both positive and negative feedback actions of estrogen on GnRH and LH secretion are important for the physiologic control of reproduction.
After detailed investigations of animal models, controversies still exist as to the effect of estrogen on the central reproductive axis, the mechanism of the effect of estrogen (direct vs. indirect), the level or levels of estrogen regulation (hypothalamic, pituitary, or both), and which isoforms of estrogen receptor (ER) (ESR1 [also known as ERα] and/or ESR2 [also known as ERβ]) mediate signaling. Esr1 and Esr2 knockout (KO) mouse models have been developed to define the physiologic effects of E2 signaling through its cognate receptors. Esr1 KO mice display significant gonadal defects and impaired feedback regulation of the neuroendocrine axis marked by elevated levels of LH and E2 [1, 2]. Compared with wild-type (WT) mice, the phenotype of Esr2 KO mice was reported to be much milder, with subfertility and mild elevations in LH, until a recent study by Antal et al. reported infertility [3–6]. These models have defined ESR1 as the predominant receptor isoform that conveys negative feedback regulation to the central reproductive axis [7] and support a predominant role for ESR1 in feedback regulation of the central reproductive axis. Cell-specific KO of Esr1 has recently been used to define more precisely the level of E2 regulation of the reproductive axis. Using a neuron-specific Esr1 KO mouse model, estrogen positive feedback has been shown to require brain ESR1 [8].
To determine the cell-specific effects of ESR1 at the level of the gonadotroph, our laboratory developed a new Esr1 floxed mouse model in which exon 3, which encodes the DNA-binding domain of ESR1, was flanked by loxP sites. We have shown that deletion of exon 3 using this model results in a lack of any detectable ESR1 protein [9]. These floxed animals were then bred to animals in which Cre recombinase expression is under the control of the mouse common glycoprotein α subunit promoter. This promoter has been shown to target the gonadotroph and direct the expression of Cre recombinase to the anterior pituitary [10, 11]. The resultant mice, referred to as PitEsr1KO, demonstrate a loss of ESR1, specifically in the anterior pituitary.
The PitEsr1KO females had irregular estrous cycles punctuated by prolonged periods of disorganized cycling. This resulted in either infertility or subfertility marked by a decrease in the number of litters and size of the litters. The serum LH and E2 values were elevated compared with those in control littermates; however, the elevation in both values was lower than that in mice with a complete KO of Esr1. In addition, the degree of elevation tracked with the severity of the reproductive defect, with infertile female mice having a greater elevation in serum LH vs. subfertile female mice. The ovaries of PitEsr1KO mice were larger, with disrupted architecture, and displayed few or no corpora lutea vs. WT animals. In addition to the defect in estrogen negative feedback, PitEsr1KO female mice have evidence of blunted or absent LH surge activity. These findings demonstrate for the first time (to our knowledge) a close relationship between estrogen negative and positive feedback in the pituitary and suggest that these regulatory pathways may not be dissociable in vivo.
MATERIALS AND METHODS
Transgene Construction
To generate the Cga-Cre transgene (Fig. 1A), a 2.1-kilobase (kb) fragment containing a nuclear localization signal, the bacteriophage P1 gene Cre recombinase, and the beta-actin polyadenylation signal was excised from pML78. This fragment was subcloned downstream of the Cga promoter (−5000 to +43) in pGEM7Zf+. A 7.0-kb Cga-Cre fragment was generated by digestion of the Cga-Cre plasmid, which reduced the Cga promoter to 4.5 kb.
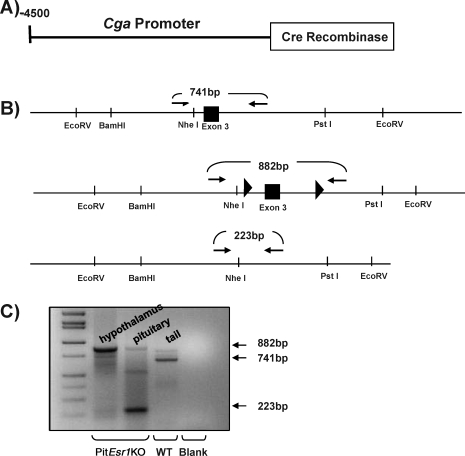
FIG. 1.
Construct used to target the Esr1 locus. A) Schematic of the transgene used to produce the Tg(Cga-cre)1Awo. A 4500-bp murine common α promoter fragment was fused to the Cre recombinase gene. B) The targeting construct used to generate Esr1 floxed (Esr1tm1.2Awo/tm1.2Awo) mice. The loxP sites were inserted in intronic regions surrounding exon 3, and a self-excising neomycin cassette, which was used for clonal selection, was inserted just downstream of the 3′ loxP site in intron 3. This was removed in the F1 generation. C) To display the specificity of the conditional KO, DNA was extracted from pituitary and hypothalamic tissue of PitEsr1KO mice. An agarose gel was run of PCR products performed with primers specifically designed to amplify the mouse Esr1 floxed gene. A 223-bp band is shown in the pituitary of the floxed Esr1 KO. An 882-bp floxed band is shown in the hypothalamus of the PitEsr1KO mouse. Genomic DNA extracted from the tail of a WT mouse was used as a positive control for the size of the WT control (741 bp). A PCR reaction containing no DNA was used as a negative control (Blank).
Generation, Genotyping, and Breeding of Transgenic Mice
Purified DNA was microinjected into zygotes from C57BL/6J × SJL parents. Embryos were transferred to postcoitum pseudopregnant CD1 females. Genomic DNA was prepared from tail biopsies of all progeny born and screened for the presence of the transgene. Mice with the Cga-Cre transgene (Tg(Cga-cre)1Awo) were identified by PCR using oligonucleotides that amplify a 150-base pair (bp) product spanning the junction of the Cga promoter and the Cre coding sequence (5′-ACATTGTTCCCCTCAGATCG-3′ and 5′-ATGTGAGCGAGTAACAACCCGTCGGATTCT-3′). Reactions proceeded for 30 cycles of denaturation at 92°C for 15 sec, annealing at 60°C for 45 sec, and extension at 72°C for 1 min, with a final extension at 72°C for 10 min. All reactions were performed under standard conditions using approximately 100–200 ng of genomic DNA, 0.5 pmol/μl primers, 2.5 mM MgCl2, and 0.02 U/μl Taq DNA polymerase per reaction. Transgenic founders and their progeny were bred to C57BL/6J mice to establish and maintain lines. Three lines were generated with the Cga-Cre transgene.
Generation and Genotyping of “Floxed” Esr1 Mice by Homologous Recombination
The cloned mouse Esr1 fragment was obtained from BAC clones of the Esr1 locus obtained from the Microarray and Genomics Facility at Roswell Park Cancer Institute (Buffalo, NY) and used to generate a targeting construct in which exon 3 of the mouse ESR1 gene was flanked by loxP sites, or “floxed” [12]. Homologous recombination was determined by using an EcoRI digestion of embryonic stem (ES) cell DNA. After ES cell selection was achieved, the Neo cassette was removed from the mutant allele in vivo. To that effect, a self-excising ACN [tACE-Cre/Neo] cassette was used that included a testes-specific promoter tACE [angiotensin-converting enzyme] driving the expression of Cre recombinase. The tACE-Cre fragment is adjacent to a Neo marker gene [13], and the entire tACE-Cre/Neo cassette is flanked by two loxP sites. Retaining a Neo cassette in vivo has been shown to have unpredictable consequences on the phenotype of mice, including generation of a hypomorphic allele with a wide range of phenotypic abnormalities [14, 15] and misregulation of adjacent genes [16]. The tACE present in the ACN cassette has been shown to specifically activate Cre recombinase activity in the testes (sperm cells) but not in other tissues [13]. Thus, self-excision of the ACN cassette in the testis of chimeric male mice resulted in sperm cells transmitting the mutant allele that contains the floxed ESR1 gene, lacking the ACN cassette, to the F1 progeny.
The ES cells heterozygous for the allele containing the integrated homologous recombinant were injected into WT mouse blastocysts. The resultant mice were chimeric for WT host-derived and recombinant ES-derived cells. Male chimeric mice transmitted one of two alleles, WT or homologous recombinant, through their sperm cells. Two types of deletions are expected to occur in the testes of chimeric males: type 1 deletion, in which both the ACN and exon 3 fragments are deleted (Esr1tm1.1Awo), and type 2 deletion, in which only the ACN cassette (and not exon 3) is deleted (Esr1tm1.2Awo). In the type 2 deletion, the mutant allele will be marked by two loxP sites flanking exon 3 of the Esr1 gene and will not contain the ACN cassette.
Southern blot analysis of tail DNA obtained from the Fl progeny was performed to identify heterozygous Fl progeny with either a type 1 or type 2 deletion. F1 mice heterozygous for the type 1 deletion were bred. Generalized homozygous Esr1 KO mice served as controls in these experiments. Fl mice heterozygous for the type 2 deletion were selected through Southern blot and PCR analysis. Heterozygote floxed Esr1 mice (Esr1+/tm1.2Awo) were bred to homozygosity to generate homozygous floxed Esr1 mice (Esr1tm1.2Awo/tm1.2Awo). The F2 Esr1tm1.2Awo/tm1.2Awo animals were backcrossed with the pure C57BL/6 strain to generate a more homogeneous background in which to perform the analyses. The floxed Esr1 mice were genotyped using primers flanking both loxP sites (sense 5′-AGGCTTTGTCTCGCTTTCC-3′ and antisense 5′-CCAAGGAGAACAGAGAGACTTACTAG-3′).
Generation of PitEsr1KO Mice Using a Cell Type-Specific Cre/loxP Binary System
Heterozygote pituitary KO mice (Tg(Cga-cre)1Awo, Esr1+/tm1.2Awo) mice were bred to Esr1tm1.2Awo/tm1.2Awo animals, generating pituitary Esr1 KO (PitEsr1KO) mice. To confirm a KO of Esr1 in the pituitary, PCR analysis on pituitary DNA obtained from PitEsr1KO mice was performed using the genotyping oligonucleotide primers that flank exon 3 and the loxP sites (as already described).
Reagents
Most of the common chemicals used were obtained from Sigma Chemical Co. (St. Louis, MO). The LH beta (LHB) polyclonal antibody was obtained from the National Hormone and Peptide Program, National Institutes of Health (Bethesda, MD), and anti-ESR1 rabbit antibody was obtained from Millipore (Billerica, MA). Estrous cycling was performed using a Diff-Quick stain kit from IMEB Inc. (San Marcos, CA).
Animal Care
All animal procedures were performed according to the Johns Hopkins University protocol approved by the animal care and use committee. All mice used in the experiments had mixed CD1/129SVJ genetic backgrounds.
Fertility Testing
Six-wk-old female mice of varying genotypes (WT, Esr1+/+; heterozygote KO, Tg(Cga-cre)1Awo, Esr1+/tm1.2Awo; and PitEsr1KO, Tg(Cga-cre)1Awo, Esr1tm1.2Awo/tm1.2Awo) were housed with WT males for a total of 6 mo. Females were monitored daily for vaginal plugs. The date of birth and size of litters for each female were recorded. Pups were removed after delivery.
Ovarian Function and Histology
Vaginal lavage was performed to document estrous cyclicity. Ovariectomy was performed as previously described [17], and the mice were allowed to recover for 10 days. Animals were anesthetized with ketamine/xylazine. Animals were perfused using 4% cold paraformaldehyde. The ovary and pituitary were then transferred to 10% formalin. Brains were transferred to 30% sucrose solution in 0.1 M phosphate buffer to dehydrate. On the next day, ovaries and pituitaries were washed twice with 1× PBS and transferred to 70% ethanol overnight at 4°C. After dehydration, the ovary and pituitary were processed for paraffin embedding, and 6-μm sections were cut using a microtome for hematoxylin-eosin (H&E) staining (ovary) and for immunostaining (pituitary).
Immunohistochemistry of the Pituitary
Six-micrometer coronal sections were deparaffinized and serially rehydrated in ethanol. After antigen retrieval, gonadotrophs were labeled using an LHB-specific antibody, and ESR1-expressing cells were labeled using an ESR1-specific antibody (rabbit antiserum C1355; Millipore), followed by Cy3 and fluorescein isothiocyanate-conjugated secondary antibodies (Jackson Laboratories, Bar Harbor, ME) [18].
Hormone Determinations
Blood was collected from the mandibular vein either from a terminal bleed or in survival studies in mice lightly anesthetized with isoflurane. Serum was obtained after centrifugation for radioimmunoassay (RIA) of LH and 17-β-E2 by the Northwestern University Radioimmunoassay Core Facility under the direction of Dr. John Levine. National Institute of Diabetes and Digestive and Kidney Diseases (Bethesda, MD) antiserum and standards (rLH-RP-3 standard/rLH-S-11 antibody) were used for LH measurements. The LH assay sensitivity was 0.2 ng/ml. An E2 double-antibody RIA kit (Diagnostic Products Corp., Los Angeles, CA) containing antibodies and standards was used for E2 RIAs. The E2 assay sensitivity was 2.0 pg/ml. Total thyroxine (T4) was measured using an ELISA kit (Alpha Diagnostic International, San Antonio, TX).
For the Luminex (Luminex Corporation, Austin, TX) system analysis, serum obtained from cycling WT and PitEsr1KO mice was stored at −20°C until the assay was performed. Because of the small samples of blood obtained from the mice during the study, only single measurements were performed using 8 μl of serum per individual time point for each animal. The LH was analyzed using xMap technology (Millipore) with the rat pituitary panel. A standard curve was generated using 5-fold serial dilutions of the LH/follicle-stimulating hormone (FSH) standard cocktail provided by the vendor. Standards and samples were incubated with the antibody-coated beads on a microplate shaker overnight at 4°C and washed three times using a vacuum manifold apparatus. Detection antibody was then added to the wells and incubated on a microplate shaker at room temperature for 30 min. Streptavidin-phycoerythrin solution was then added, and an additional 30-min incubation at room temperature was performed using the microplate shaker. Plates were then washed three times, and sheath fluid was added to each well. Beads were resuspended on the microplate shaker for 5 min. Plates were then read on the Luminex 200IS system using xPonent software (Luminex Corporation). Data were analyzed using five-parameter logistic curve fitting. The limit of detection for the LH assay was 80 pg/ml, and the intraassay coefficient of variation was between 9% and 6.3%.
Quantitative Real-Time PCR
Total RNA was obtained from pituitary and hypothalamic tissue using Trizol (Invitrogen, San Diego, CA) extractions as previously described [19, 20]. Two micrograms of RNA was reverse transcribed (iScript cDNA synthesis kit; BioRad, Hercules, CA) to produce cDNA. The cDNA obtained from 50 ng of total RNA was used in each reaction. Twenty-five-microliter PCR reactions were performed using the IQ SybrGreen Supermix (BioRad). Reactions were measured using the MyiQ quantitative real-time PCR machine (BioRad). The following primer sets were used: mouse Gnrh1 (sense 5′-CCCTTTGACTTTCACATCC-3′ and antisense 5′-GGGTTCTGCCATTTGATCCAC-3′), Esr1 (sense 5′-CTTCAGTGCCAACAGCCT-3′ and antisense 5′-GACAGTCTCTCTCGGCCAT-3′), Esr2 (sense 5′-GCCAACCTCCTGATGCTTCTTT-3′ and antisense 5′-TTGTACCCTCGAAGCGTGTGA-3′), Lhb (sense 5′-CAGTCTGCATCACCTTCACCA-3′ and antisense 5′-GGTAGGTGCACACTGGCTGA-3′), and a ribosomal 18S (Rn18s) control (sense 5′-TGGTTGATCCTGCCAGTAG-3′ and antisense 5′-CGACCAAAGGAACCATAACT-3′). The PCR conditions were optimized to generate >95% PCR efficiency, and only those reactions between 95% and 105% efficiency were included in subsequent analyses. The cycle threshold (Ct) was obtained for each sample. A corrected Ct (delta Ct) was calculated by subtracting the RN18S Ct from the unknown sample Ct for each sample. Relative differences from the control sample were then calculated using the following formula: fold change = 2 ˆ (control delta Ct minus sample delta Ct). The PCR products were also analyzed by gel electrophoresis.
Statistical Analysis
All results are expressed as the mean ± SEM. Statistical significance was assessed by one-way or two-way ANOVA using the GraphPad Prism 5 program (San Diego, CA), with P < 0.05 considered significant. Post hoc analysis was conducted using Tukey multiple comparison test or Bonferroni post test.
RESULTS
Generation of PitEsr1KO Mice
PitEsr1KO mice were obtained by breeding Esr1 floxed mice (Esr1tm1.2Awo/tm1.2Awo) to mice expressing Cre recombinase in the anterior pituitary (Tg(Cga-cre)1Awo) (Fig. 1). Mice were born in the expected mendelian frequency and were of normal size and weight. Esr1tm1.2Awo/tm1.2Awo and Tg(Cga-cre)1Awo animals were identified using published methods [21]. The resulting mice were tested for the presence of a pituitary-specific KO using a PCR strategy. Figure 1C shows the results of a PCR analysis of DNA from several tissues in these animals. Note that PitEsr1KO mice had a smaller band (223 bp) in pituitary DNA due to the deletion of the third exon of Esr1 in this tissue. A weak-intensity floxed band (882 bp) was also observed in this PCR reaction, presumably representing the intact floxed allele in non-Cga-expressing pituitary cells. However, the smaller band was not observed in hypothalamic tissue or tail DNA from the PitEsr1KO and WT mice, which contained bands consistent with the floxed and WT alleles, respectively (Fig. 1C), or in cortex, cerebellum, liver, or gonads (data not shown). These data indicate the specificity of the recombination event. Although this analysis demonstrated the tissue-specific KO, it was unable to show the extent of ESR1 in the pituitary because of the heterogeneous nature of cells in the mRNA sample.
Absence of ESR1 in Gonadotrophs of PitEsr1KO Mice
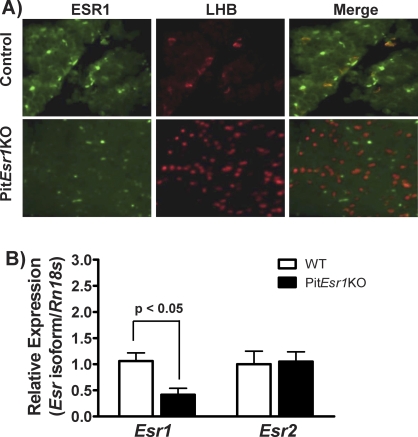
Immunohistochemistry for LHB and ESR1 in the pituitary of control mice demonstrates colocalization of LHB and ESR1 (Fig. 2A, merged image, upper right panel). These results demonstrate that about 20% of cells could be identified as gonadotrophs, of which about 50% contain ESR1 [22]. There was an increase in the number of LH-staining cells in WT vs. PitEsr1KO mice (Fig. 2A, center panels). As expected, ESR1 is absent from the gonadotrophs of PitEsr1KO mice (Fig. 2A, merged image, lower right panel), while it is present in non-gonadotroph cells of the pituitary (Fig. 2A, lower left panel), thereby establishing the specificity of the assay. Examination of >30 pituitary sections failed to reveal any colocalization of LHB and ESR1 in two subfertile or two infertile PitEsr1KO mice. These findings validate the deletion of Esr1 in pituitary gonadotrophs. Esr1 mRNA levels were measured using quantitative RT-PCR in both WT and PitEsr1KO mice, as shown in Figure 2B. There was a statistically significant reduction in Esr1 mRNA levels in the pituitary of PitEsr1KO vs. control mice consistent with the immunohistochemistry results.
FIG. 2.
ESR1 expression is abolished in LH-expressing cells in the pituitary of female PitEsr1KO mice. A) Double immunofluorescence for ESR1 (green), LHB (red), or both (yellow) in the pituitaries of control (top row) or PitEsr1KO (bottom row) mice. No LH-expressing cells were found to coexpress ESR1 in the PitEsr1KO mice (bottom right panel). Original magnification ×100. B) Total RNA was extracted from pituitary tissue of PitEsr1KO mice. The RNA from the pituitary of WT mice was used as a control. Quantitative real-time PCR was performed using primers specifically designed to amplify mouse Esr1 and mouse Esr2. Data are expressed as the mean ± SEM (n = 6).
Pituitary ESR2 Expression Was Not Affected by the Lack of ESR1
Because ESR2 has also been shown to be expressed in the pituitary [22–24], Esr2 mRNA levels were determined using quantitative RT-PCR in both WT and PitEsr1KO mice (Fig. 2B). There was no difference in Esr2 mRNA levels in the pituitary between PitEsr1KO and control mice.
Infertility in Female PitEsr1KO Mice
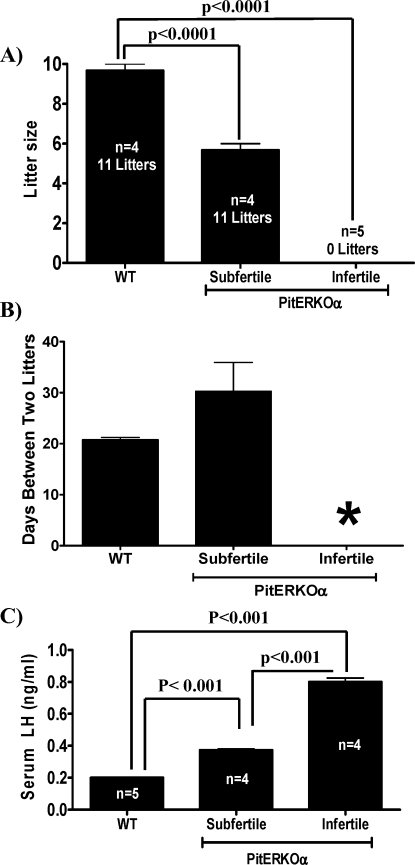
PitEsr1KO female mice and age- and sex-matched control mice were tested for fertility with proven fertile control male mice. After 6 mo of breeding, PitEsr1KO female mice revealed two distinct phenotypes, infertile and subfertile. Five of nine PitEsr1KO females were infertile, and the remaining four females were subfertile (Fig. 3A). Subfertile mice had a significantly decreased litter size (5.9 pups/litter vs. 9.8 pups/litter in control animals) and an increased number of days between litters (31 days vs. 21 days in control animals) (Fig. 3B). The difference in days between litters for the subfertile PitEsr1KO mice did not achieve statistical significance because of a large variability of intervals, ranging from 42 days to 21 days. Vaginal plugs were frequently detected in infertile PitEsr1KO females, suggesting that the infertility was not caused by any impairment of sexual behavior in the female KO mice. Control mice display fertility parameters similar to those of WT mice in the colony. Male PitEsr1KO mice had normal fertility as judged by test matings with known fertile females (data not shown).
FIG. 3.
PitEsr1KO mice exhibit impaired fertility. A) Litter size was assessed in WT control mice and PitEsr1KO mice. Data from subfertile and infertile PitEsr1KO are shown. B) Time between litters of WT control mice and PitEsr1KO mice. The asterisk indicates that no litters were observed in PitEsr1KO mice. C) The LH levels are elevated in PitEsr1KO female mice. The LH levels (in ng/ml) are indicated for WT controls and subfertile and infertile PitEsr1KO female mice. The number of mice analyzed for each group is indicated. Significance relative to the other groups is indicated by brackets.
Disrupted Ovarian Histology in PitEsr1KO Mice
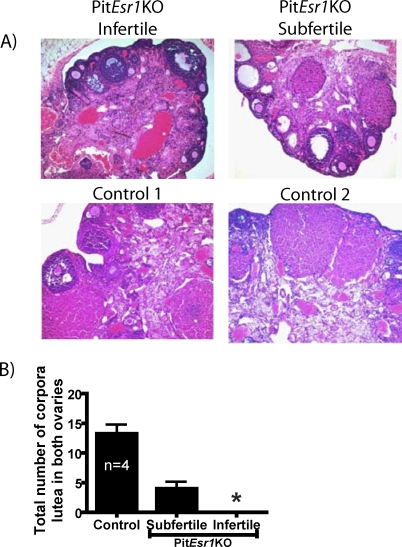
Hematoxylin-eosin staining of ovarian sections from WT control mice showed follicles in different stages of development and the presence of multiple corpora lutea (Fig. 4A). In contrast, ovarian sections from subfertile and infertile PitEsr1KO mice had few and no corpora lutea, respectively; both displayed an increase in interstitial space compared with that in control mice. Also seen were atretic follicles and primary and secondary follicles that failed to reach to the periphery of the ovary. As shown in Figure 4B, the average number of corpora lutea was significantly less in subfertile PitEsr1KO vs. control mice (four vs. 14). Among 10 ovaries that were evaluated, a single corpus luteum was found in the ovary of an infertile female PitEsr1KO mouse.
FIG. 4.
Mouse ovarian morphology is disrupted in PitEsr1KO mice. A) The H&E-stained ovarian sections are shown from infertile and subfertile PitEsr1KO mice and from two representative WT controls. PitEsr1KO ovaries exhibit increased numbers of atretic follicles and cysts and reduced numbers of corpora lutea compared with those in WT ovaries. Original magnification ×100. B) Counts of corpora lutea in paired ovaries of PitEsr1KO and WT control females. The asterisk indicates that only one corpus luteum was observed in a PitEsr1KO ovary.
Hormone Levels in PitEsr1KO Mice
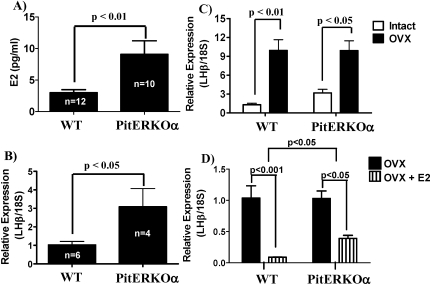
The mean nonsurge serum LH value in WT female mice with normal fertility was 0.2 ng/ml, whereas the mean values of subfertile and infertile PitEsr1KO mice were significantly higher (0.4 ng/ml and 0.8 ng/ml, respectively) (Fig. 3C). These values are somewhat lower than previously reported LH values obtained in the same assay [25]. The mean E2 level at 800 h in WT female mice with normal fertility was 2.5 pg/ml, whereas subfertile and infertile PitEsr1KO mice demonstrated significantly elevated E2 levels (Fig. 5A). Unlike serum LH values, there was no difference in the degree of E2 elevation between subfertile and infertile PitEsr1KO mice. Because the Cga promoter should also target the pituitary thyrotroph, we determined the T4 levels in these mice. Serum T4 levels were not significantly different from those of control mice in any of the PitEsr1KO animals (data not shown).
FIG. 5.
Negative feedback is impaired in the PitEsr1KO female mice. A) Serum E2 levels are elevated in PitEsr1KO female mice compared with those in WT controls. B) Total RNA was extracted from pituitary tissue of WT and PitEsr1KO mice. Quantitative real-time PCR was performed using primers specifically designed to amplify mouse Lhb and corrected for Rn18s content. C) Total RNA was extracted from pituitary tissue of WT and PitEsr1KO mice that were ovariectomized (OVX) or sham ovariectomized (Intact). Data are expressed as the mean ± SEM (n = 6–11). D) Total RNA was extracted from pituitary tissue of WT and PitEsr1KO mice that were ovariectomized (OVX) or ovariectomized and administered E2 (OVX+E2). Data are expressed as the mean ± SEM (n = 6–11). Brackets above adjacent bars indicate significant differences between the two groups. The bracket at the top of D denotes a significant difference in the response of the two genotypes to E2.
Estrogen Feedback Regulation of Lhb Subunit and Gnrh1 mRNA in PitEsr1KO Mice
Lhb and Gnrh1 mRNA levels were measured in infertile PitEsr1KO mice and compared with those in control animals. Consistent with a pituitary defect in estrogen negative feedback, basal Lhb mRNA levels were >3-fold elevated in PitEsr1KO mice vs. control mice (Fig. 5C, white bars). After gonadectomy, Lhb subunit mRNA levels in PitEsr1KO mice were elevated to the same degree as that seen in the WT mice; however, the fold change was reduced because of an elevation in baseline values. In contrast, hypothalamic Gnrh1 mRNA levels were not statistically different between PitEsr1KO mice and control mice (data not shown).
Lack of Estrous Cyclicity in PitEsr1KO Mice
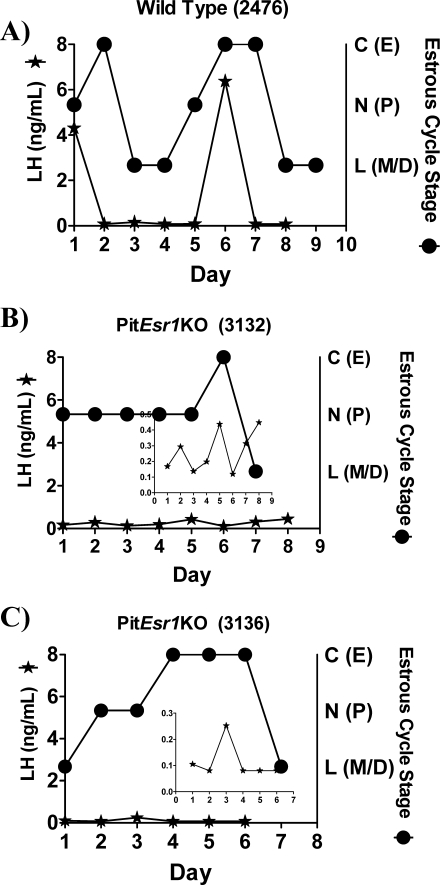
Over 8 consecutive days, the estrous cycle stage was evaluated in the morning by vaginal cytology, and afternoon blood samples were obtained for serial serum LH measurements in a multiplex assay system. Figure 6 shows data obtained from one representative control animal and two representative PitEsr1KO mice. In control mice, we observed 4- to 5-day estrous cycles as assessed by vaginal cytology, and these correlated with LH surge levels (Fig. 6A). Surge levels reached 7 ng/ml in control mice, and nonsurge levels were measured at approximately 80 pg/ml, which is the limit of detectability in this assay. In PitEsr1KO mice, disorganized estrous cyclicity was always observed, and it was at times difficult to definitively assess the stage. Basal LH levels were higher in PitEsr1KO mice, averaging about 0.2 ng/ml, which is roughly 4-fold higher than those in control mice, and no definitive surge LH values were obtained. Peak LH levels in PitEsr1KO mice reached approximately 0.4 ng/ml, or about 18-fold lower than those in control animals.
FIG. 6.
Estrous cycle stage and daily LH levels from WT and PitEsr1KO mice. The LH levels (left axis) and estrous cycle stage as assessed by vaginal cytology (right axis) are shown in a representative WT mouse (A) and two representative PitEsr1KO mice (B and C). The LH levels (in ng/ml) are represented by black stars. The insets in the PitEsr1KO mice graphs (B and C) show LH levels (in ng/ml) in a reduced scale so that the small changes in LH can be visualized. Estrous stage is indicated by black circles as either C (primarily cornified epithelium, estrous [E]), N (primarily nucleated epithelium, proestrous [P]), or L (primarily leukocytic cells, metestrous/diestrous [M/D]).
DISCUSSION
Neuroendocrine control of ovarian function is mediated by the pulsatile secretion of GnRH into the hypophysial portal vasculature [26, 27]. The GnRH decapeptide binds to GnRH receptors on the plasma membrane of gonadotrophs, which then directs the synthesis and secretion of the gonadotropins LH and FSH into the peripheral circulation to control ovarian steroidogenesis and folliculogenesis. Ovarian steroid secretion in turn exerts homeostatic feedback actions that alter GnRH and gonadotropin secretions. Throughout most of the rodent estrous cycle, E2 exerts negative feedback actions on GnRH and LH secretion until (on the afternoon of proestrus) elevated follicular E2 secretions evoke an abrupt release of a preovulatory GnRH surge and hence an LH surge, which triggers ovulation [28, 29]. Thus, both negative and positive feedback actions of E2 are critically important in the physiologic control of cyclic hormone secretions and ovulatory cyclicity.
Generation and study of both Esr1 and Esr2 KO mouse models have made important contributions to our understanding of estrogen action in the reproductive axis [2, 4]. Both male and female Esr1 KO mice are infertile, although the Esr2 KO mice in conflicting studies have been reported to exhibit normal fertility in males and subfertility in females [30–36] or infertility in both sexes [5]. Findings in physiologic studies [7, 37, 38] of Esr1 KO mouse models suggest that estrogen negative feedback occurs primarily through ESR1. From these models, it is unclear whether the level of feedback is hypothalamic, pituitary, or both, as ESR1 is absent from all tissues. Moreover, Esr1 KO mice have a severe gonadal phenotype that could be owing to either a direct effect of a loss of ESR1 from the ovary or an indirect effect due to an alteration of gonadotropin secretion. Hence, the role of ESR1 in estrogen feedback regulation of the pituitary gonadotroph is complicated by the potential sites of estrogen action and nature of feedback in the reproductive axis.
The Cre/loxP system in transgenic mice makes it possible to examine the effects of gene deletion in a specific cell type or tissue [39–43]. Two studies [8, 44] have used this technology to study the effects of Esr1 deletion in the brain and pituitary. In the first study [8], Esr1 was deleted from neurons using the Camk2a promoter, resulting in a defect in estrogen-mediated positive feedback, which the authors concluded was indirectly mediated by neurons that contact the GnRH neuron. Although the authors ascertained that estrogen positive feedback was located in the hypothalamus, it was not possible to determine whether ESR1 also had a role in hypothalamic negative feedback because of the design of the study. In contrast, Esr1 was deleted from the anterior pituitary in a second study [44]. These authors determined that PitEsr1KO mice had impaired estrous cyclicity and infertility, suggesting that pituitary Esr1 KO might have impaired estrogen positive feedback. Curiously, these mice had apparently normal numbers of corpora lutea and serum LH levels. Thus, whether estrogen positive feedback is located at the level of the pituitary is unclear from this study, and the locus of estrogen negative feedback remains to be defined. The complete infertility observed in that study differs from our observation of infertility and subfertility. This may be owing to several causes. Although similarly constructed, the floxed Esr1 and Cga-Cre mice used in the present study were of different origin from those used by Gieske et al. [44]. In addition, fertility assessment by Gieske et al. was performed for 3 mo vs. 6 mo in the present study. It is possible that subfertility may have been observed if Gieske et al. had extended their fertility assessment. Table 1 summarizes the female reproductive phenotypes of the complete Esr1 KO, brain Esr1 KO, and pituitary Esr1 KO models. In addition, it emphasizes the differences observed in our study vs. the study by Gieske et al.
TABLE 1.
Summary of Esr1 KO models and their effect on the reproductive axis.*
Given these conflicting results, we report herein on the generation and characterization of a mouse model lacking Esr1 in the pituitary gonadotroph (PitEsr1KO). We developed a floxed Esr1 mouse in which exon 3 was flanked by loxP elements. Production of a complete Esr1 KO mouse using a beta-actin Cre mouse demonstrated a complete lack of ESR1 immunoreactivity and almost completely abolished expression of ESR1 target genes [9]. PitEsr1KO mice were generated by crossing the floxed Esr1 mouse with a mouse expressing Cre recombinase under the control of a 4.5-kb mouse common glycoprotein α subunit promoter (CGA) (Fig. 1). This promoter fragment has been previously shown to confer high levels of hormonally regulated expression in the pituitary gonadotroph [10, 11]. Study of the PitEsr1KO mouse model revealed both subfertility and infertility in female mice (Fig. 3); male mice had normal fertility. Differences in fertility were not due to the extent of ESR1 loss in the anterior pituitary, as immunohistochemistry revealed a complete lack of ER in pituitary gonadotrophs regardless of the fertility phenotype (Fig. 2A). About 20% of the pituitary cells, either from WT or PitEsr1KO mice, were immunopositive for LHB, which is similar to the numbers reported in the literature for rodents [22–24, 45]. Somewhat greater numbers of LHB immunopositive cells were observed in pituitaries obtained from PitEsr1KO mice, although this difference was not statistically significant (data not shown). As expected based on the deletion of Esr1 from the gonadotrophs, the amount of Esr1 mRNA was decreased in pituitaries harvested from PitEsr1KO mice (Fig. 2B). It is unlikely that a compensatory increase in gonadotroph ESR2 could explain the differences in fertility, as ESR2 was found in few (<10%) gonadotrophs [22], and no difference in total pituitary Esr2 mRNA was observed between PitEsr1KO and WT mice (Fig. 2B). Differences in fertility corresponded to the ovarian phenotype, in which a reduced number of corpora lutea was noted in subfertile female mice (Fig. 4), and only one corpus luteum was found in the infertile mice. A few hemorrhagic cystic follicles were also noted in infertile PitEsr1KO mice but not to the extent found in complete Esr1KO mice. These data suggest that follicular development is partially arrested in subfertile and completely arrested in infertile PitEsr1KO mice, in contrast to previous findings [33].
Results in our PitEsr1KO mouse model differ from those in the study by Gieske et al. [44] in several other aspects. First, we demonstrate the mechanism by which pituitary ESR1 loss causes infertility; we definitively show that PitEsr1KO mice lack LH surge activity. Using a novel experimental design, we measured daily afternoon LH values from small-sample blood volumes (using a multiplex ligand assay system) and directly correlated LH measurements with the cycle stage. Data from WT mice demonstrate clear evidence of LH surge activity, which was totally absent in the PitEsr1KO mice (Fig. 6, B and C). The insets in Figure 6, B and C, suggest that the GnRH neuron may still exhibit pulsatile secretion in PitEsr1KO mice, as indicated by differences in daily LH secretion, albeit at low levels. Thus, in contrast to previous findings [8], estrogen positive feedback is also clearly located in part at the pituitary level.
Second, we demonstrate a defect in E2 negative feedback in PitEsr1KO mice. Nonsurge serum LH levels were significantly elevated in both subfertile and infertile PitEsr1KO mice (Fig. 3C), although infertile animals had a greater elevation in serum levels. Basal serum E2 levels (Fig. 5A) and pituitary LHB mRNA levels (Fig. 5B) were also elevated consistent with the serum LH levels. However, hypothalamic Gnrh1 mRNA levels were unchanged, despite the mild elevation in E2 levels in PitEsr1KO mice (data not shown). Although serum LH values were not as elevated as those found in complete Esr1 KO mice, they were nonetheless significantly elevated (2- to 4-fold) in PitEsr1KO female mice compared with those in WT animals. Clearly, other pathways may regulate the central reproductive axis in response to estrogen [34, 35]. When PitEsr1KO mice were ovariectomized and administered estrogen, Lhb mRNA levels were incompletely suppressed (Fig. 5D), which is also consistent with a defect in estrogen negative feedback regulation in PitEsr1KO mice. Thus, PitEsr1KO mice demonstrate a defect in estrogen negative feedback at the level of the gonadotroph that is associated with absent LH surges and impaired or absent fertility. Given that both estrogen positive and negative feedback is disrupted in the PitEsr1KO mice, these feedback mechanisms cannot be dissociated in the pituitary.
Thus, the physiologic role of the endogenous ESR1 signaling pathway in pituitary gonadotrophs has been defined using PitEsr1KO mice. PitEsr1KO female mice are subfertile or infertile and have elevated levels of serum LH and Lhb gene expression, reflecting the lack of estrogen negative feedback on the gonadotroph. Although serum LH values are elevated in PitEsr1KO mice, the degree of elevation is much less than that observed in Esr1 KO mice, indicating that the hypothalamus must also have an important role in estrogen negative feedback. Estrogen levels are also modestly elevated in PitEsr1KO mice, but Gnrh1 mRNA levels in the hypothalamus were not altered. Correlation between estrous cyclicity and serum LH values demonstrates that PitEsr1KO mice also have a defect in estrogen positive feedback, as surge LH values are not observed in these mice. Therefore, PitEsr1KO mice demonstrate a defect in both estrogen negative and positive feedback on the reproductive axis in the setting of preserved hypothalamic estrogen signaling. As in the pituitary, estrogen feedback in the central reproductive axis may be discrete neither in location nor in the direction of its effect.
Acknowledgments
The authors are indebted to Yayoi Shibusawa for technical assistance in the production of the floxed ERα mice. Technical assistance was also provided by Tameeka Williams, Stefanie Eames, Danielle Machado, and Elizabeth Sefton. Special thanks to Dan Diazcok and George Park for critical review of the manuscript.
Footnotes
1Supported by research grants from the National Institutes of Health (R01 HD44608 to A.W. and HD933067 and HD370246 to S.R.) and the Eunice Kennedy Shriver NICHD/NIH through cooperative agreement (U54 HD58820 to A.W. and S.R.) as part of the Specialized Cooperative Centers Program in Reproduction and Infertility Research.
These authors contributed equally to this work.
REFERENCES
- Couse JF, Curtis SW, Washburn TF, Eddy EM, Schomberg DW, Korach KS.Disruption of the mouse oestrogen receptor gene: resulting phenotypes and experimental findings. Biochem Soc Trans 1995; 23: 929–935.. [DOI] [PubMed] [Google Scholar]
- Lubahn DB, Moyer JS, Golding TS, Couse JF, Korach KS, Smithies O.Alteration of reproductive function but not prenatal sexual development after insertional disruption of the mouse estrogen receptor gene. Proc Natl Acad Sci U S A 1993; 90: 11162–11166.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dupont S, Krust A, Gansmuller A, Dierich A, Chambon P, Mark M.Effect of single and compound knockouts of estrogen receptors alpha (ERalpha) and beta (ERbeta) on mouse reproductive phenotypes. Development 2000; 127: 4277–4291.. [DOI] [PubMed] [Google Scholar]
- Krege JH, Hodgin JB, Couse JF, Enmark E, Warner M, Mahler JF, Sar M, Korach KS, Gustafsson JA, Smithies O.Generation and reproductive phenotypes of mice lacking estrogen receptor beta. Proc Natl Acad Sci U S A 1998; 95: 15677–15682.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Antal MC, Krust A, Chambon P, Mark M.Sterility and absence of histopathological defects in nonreproductive organs of a mouse ERbeta-null mutant. Proc Natl Acad Sci U S A 2008; 105: 2433–2438.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shughrue PJ, Askew GR, Dellovade TL, Merchenthaler I.Estrogen-binding sites and their functional capacity in estrogen receptor double knockout mouse brain. Endocrinology 2002; 143: 1643–1650.. [DOI] [PubMed] [Google Scholar]
- Couse JF, Yates MM, Walker VR, Korach KS.Characterization of the hypothalamic-pituitary-gonadal axis in estrogen receptor (ER) null mice reveals hypergonadism and endocrine sex reversal in females lacking ERalpha but not ERbeta. Mol Endocrinol 2003; 17: 1039–1053.. [DOI] [PubMed] [Google Scholar]
- Wintermantel TM, Campbell RE, Porteous R, Bock D, Grone HJ, Todman MG, Korach KS, Greiner E, Perez CA, Schutz G, Herbison AE.Definition of estrogen receptor pathway critical for estrogen positive feedback to gonadotropin-releasing hormone neurons and fertility. Neuron 2006; 52: 271–280.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen M, Wolfe A, Wang X, Chang C, Yeh S, Radovick S.Generation and characterization of a complete null ER mouse using Cre/LoxP technology. Mol Cell Biochem 2008; 321: 145–153.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burrows HL, Birkmeier TS, Seasholtz AF, Camper SA.Targeted ablation of cells in the pituitary primordia of transgenic mice. Mol Endocrinol 1996; 10: 1467–1477.. [DOI] [PubMed] [Google Scholar]
- Kendall SK, Gordon DF, Birkmeier TS, Petrey D, Sarapura VD, O'Shea S, Wood WM, Lloyd RV, Ridgeway EC, Camper SA.Enhancer-mediated high level expression of mouse pituitary glycoprotein hormone a-subunit transgene in thyrotropes, gonadotropes, and developing pituitary gland. Mol Endocrinol 1994; 8: 1420–1433.. [DOI] [PubMed] [Google Scholar]
- Lakso M, Sauer B, Mosinger BJ, Lee EJ, Manning RW, Yu SH, Mulder KL, Westphal H.Targeted oncogene activation by site-specific recombination in transgenic mice. Proc Natl Acad Sci U S A 1992; 89: 6232–6236.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bunting M, Bernstein KE, Greer JM, Capecchi MR, Thomas KR.Targeting genes for self-excision in the germ line. Genes Dev 1999; 13: 1524–1528.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colledge WH, Abella BS, Southern KW, Ratcliff R, Jiang C, Cheng SH, MacVinish LJ, Anderson JR, Cuthbert AW, Evans MJ.Generation and characterization of a delta F508 cystic fibrosis mouse model. Nat Genet 1995; 10: 445–452.. [DOI] [PubMed] [Google Scholar]
- Olson EN, Arnold HH, Rigby PW, Wold BJ.Know your neighbors: three phenotypes in null mutants of the myogenic bHLH gene MRF4. Cell 1996; 85: 1–4.. [DOI] [PubMed] [Google Scholar]
- Meyers EN, Lewandoski M, Martin GR.An Fgf8 mutant allelic series generated by Cre- and Flp-mediated recombination. Nat Genet 1998; 18: 136–141.. [DOI] [PubMed] [Google Scholar]
- Wolfe AM, Wray S, Westphal H, Radovick S.Cell-specific expression of the human gonadotropin-releasing hormone gene in transgenic animals. J Biol Chem 1995; 271: 20018–20023.. [DOI] [PubMed] [Google Scholar]
- Raetzman LT, Wheeler BS, Ross SA, Thomas PQ, Camper SA.Persistent expression of Notch2 delays gonadotrope differentiation. Mol Endocrinol 2006; 20: 2898–2908.. [DOI] [PubMed] [Google Scholar]
- Divall SA, Radovick S, Wolfe A.Egr-1 binds the GnRH promoter to mediate the increase in gene expression by insulin. Mol Cell Endocrinol 2007; 270: 64–72.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim HH, Divall SA, Deneau RM, Wolfe A.Insulin regulation of GnRH gene expression through MAP kinase signaling pathways. Mol Cell Endocrinol 2005; 242: 42–49.. [DOI] [PubMed] [Google Scholar]
- Wolfe A, Divall S, Singh SP, Nikrodhanond AA, Baria AT, Le WW, Hoffman GE, Radovick S.Temporal and spatial regulation of CRE recombinase expression in gonadotrophin-releasing hormone neurones in the mouse. J Neuroendocrinol 2008; 20: 909–916.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mitchner NA, Garlick C, Ben-Jonathan N.Cellular distribution and gene regulation of estrogen receptors alpha and beta in the rat pituitary gland. Endocrinology 1998; 139: 3976–3983.. [DOI] [PubMed] [Google Scholar]
- Shughrue PJ, Lane MV, Scrimo PJ, Merchenthaler I.Comparative distribution of estrogen receptor-alpha (ER-alpha) and beta (ER-beta) mRNA in the rat pituitary, gonad, and reproductive tract. Steroids 1998; 63: 498–504.. [DOI] [PubMed] [Google Scholar]
- Wilson ME, Price RH, Jr, Handa RJ.Estrogen receptor-beta messenger ribonucleic acid expression in the pituitary gland. Endocrinology 1998; 139: 5151–5156.. [DOI] [PubMed] [Google Scholar]
- Glidewell-Kenney C, Hurley LA, Pfaff L, Weiss J, Levine JE, Jameson JL.Nonclassical estrogen receptor alpha signaling mediates negative feedback in the female mouse reproductive axis. Proc Natl Acad Sci U S A 2007; 104: 8173–8177.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levine JE, Kwok-Yuen FP, Ramirez VD, Jackson GL.Simultaneous measurement of luteinizing hormone-releasing hormone and luteinizing hormone release in unanesthetized, ovariectomized sheep. Endocrinology 1982; 111: 1449–1455.. [DOI] [PubMed] [Google Scholar]
- Levine JE, Ramirez VD.In vivo release of luteinizing hormone-releasing hormone estimated with push-pull cannulae from the mediobasal hypothalami of ovariectomized, steroid-primed rats. Endocrinology 1980; 107: 1782–1790.. [DOI] [PubMed] [Google Scholar]
- Meijs-Roelofs HM, Uilenbroek JT, De Greef WJ, De Jong FH, Kramer P.Gonadotrophin and steroid levels around the time of first ovulation in the rat. J Endocrinol 1975; 67: 275–282.. [DOI] [PubMed] [Google Scholar]
- Sarkar DK, Chiappa SA, Fink G, Sherwood NM.Gonadotropin-releasing hormone surge in pro-oestrous rats. Nature 1976; 264: 461–463.. [DOI] [PubMed] [Google Scholar]
- Kuiper GG, Enmark E, Pelto-Huikko M, Nilsson S, Gustafsson JA.Cloning of a novel receptor expressed in rat prostate and ovary. Proc Natl Acad Sci U S A 1996; 93: 5925–5930.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tremblay GB, Tremblay A, Copeland NG, Gilbert DJ, Jenkins NA, Labrie F, Giguere V.Cloning, chromosomal localization, and functional analysis of the murine estrogen receptor beta. Mol Endocrinol 1997; 11: 353–365.. [DOI] [PubMed] [Google Scholar]
- Mangelsdorf DJ, Thummel C, Beato M, Herrlich P, Schutz G, Umesono K, Blumberg B, Kastner P, Mark M, Chambon P, Evans RM.The nuclear receptor superfamily: the second decade. Cell 1995; 83: 835–839.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Korach KS, Couse JF, Curtis SW, Washburn TF, Lindzey J, Kimbro KS, Eddy EM, Migliaccio S, Snedeker SM, Lubahn DB, Schomberg DW, Smith EP.Estrogen receptor gene disruption: molecular characterization and experimental and clinical phenotypes. Recent Prog Horm Res 1996; 51: 159–186.. [PubMed] [Google Scholar]
- Ogawa S, Taylor JA, Lubahn DB, Korach KS, Pfaff DW.Reversal of sex roles in genetic female mice by disruption of estrogen receptor gene. Neuroendocrinology 1996; 64: 467–470.. [DOI] [PubMed] [Google Scholar]
- Ogawa S, Lubahn DB, Korach KS, Pfaff DW.Behavioral effects of estrogen receptor gene disruption in male mice. Proc Natl Acad Sci U S A 1997; 94: 1476–1481.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eddy EM, Washburn TF, Bunch DO, Goulding EH, Gladen BC, Lubahn DB, Korach KS.Targeted disruption of the estrogen receptor gene in male mice causes alteration of spermatogenesis and infertility. Endocrinology 1996; 137: 4796–4805.. [DOI] [PubMed] [Google Scholar]
- Wersinger SR, Haisenleder DJ, Lubahn DB, Rissman EF.Steroid feedback on gonadotropin release and pituitary gonadotropin subunit mRNA in mice lacking a functional estrogen receptor alpha. Endocrine 1999; 11: 137–143.. [DOI] [PubMed] [Google Scholar]
- Dorling AA, Todman MG, Korach KS, Herbison AE.Critical role for estrogen receptor alpha in negative feedback regulation of gonadotropin-releasing hormone mRNA expression in the female mouse. Neuroendocrinology 2003; 78: 204–209.. [DOI] [PubMed] [Google Scholar]
- Orban PC, Chui D, Marth JD.Tissue- and site-specific DNA recombination in transgenic mice. Proc Natl Acad Sci U S A 1992; 89: 6861–6865.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lewandoski M, Martin GR.Cre-mediated chromosome loss in mice. Nat Genet 1997; 17: 223–225.. [DOI] [PubMed] [Google Scholar]
- Cushman LJ, Burrows HL, Seasholtz AF, Lewandoski M, Muzyczka N, Camper SA.Cre-mediated recombination in the pituitary gland. Genesis 2000; 28: 167–174.. [DOI] [PubMed] [Google Scholar]
- Tsien JZ, Chen DF, Gerber D, Tom C, Mercer EH, Anderson DJ, Mayford M, Kandel ER, Tonegawa S.Subregion- and cell type-restricted gene knockout in mouse brain. Cell 1996; 87: 1317–1326.. [DOI] [PubMed] [Google Scholar]
- Ray MK, Fagan SP, Moldovan S, DeMayo FJ, Brunicardi FC.A mouse model for beta cell-specific ablation of target gene(s) using the Cre-loxP system. Biochem Biophys Res Commun 1998; 253: 65–69.. [DOI] [PubMed] [Google Scholar]
- Gieske MC, Kim HJ, Legan SJ, Koo Y, Krust A, Chambon P, Ko C.Pituitary gonadotroph estrogen receptor-alpha is necessary for fertility in females. Endocrinology 2008; 149: 20–27.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanchez-Criado JE, de Las Mulas JM, Bellido C, Aguilar R, Garrido-Gracia JC.Gonadotrope oestrogen receptor-alpha and -beta and progesterone receptor immunoreactivity after ovariectomy and exposure to oestradiol benzoate, tamoxifen or raloxifene in the rat: correlation with LH secretion. J Endocrinol 2005; 184: 59–68.. [DOI] [PubMed] [Google Scholar]