Abstract
Molecular breast imaging (MBI) is a new nuclear medicine technique that utilizes small semiconductor-based γ-cameras in a mammographic configuration to provide high-resolution functional images of the breast. Current studies with MBI have used Tc-99m sestamibi, which is an approved agent for breast imaging. The procedure is relatively simple to perform. Imaging can be performed within 5 min postinjection, with the breast lightly compressed between the two detectors. Images of each breast are acquired in the craniocaudal and mediolateral oblique projections facilitating comparison with mammography. Key studies have confirmed that MBI has a high sensitivity for the detection of small breast lesions. In patients with suspected breast cancer, MBI has an overall sensitivity of 90%, with a sensitivity of 82% for lesions less than 10 mm in size. Sensitivity was lowest for tumors less than 5 mm in size. Tumor detection does not appear to be dependent on tumor type, but rather on tumor size. Studies using MBI and breast-specific γ-imaging have shown that these methods have comparable sensitivity to breast MRI. A large clinical trial compared MBI with screening mammography in over 1000 women with mammographically dense breast tissue and increased risk of breast cancer and showed that MBI detected two-to three-times more cancers than mammography. In addition, MBI appears to have slightly better specificity than mammography in this trial. MBI provides high-resolution functional images of the breast and its potential applications range from evaluation of the extent of disease to a role as an adjunct screening technique in certain high-risk populations. MBI is highly complementary to existing anatomical techniques, such as mammography, tomosynthesis and ultrasound.
Keywords: breast cancer, breast MRI, mammography, molecular breast imaging, screening, Tc-99m sestamibi
Breast cancer is one of the most common cancers in women, with over 180,000 new cases estimated in 2009. Many studies have shown that early detection of breast cancer and improved diagnosis of the extent of disease enable more appropriate treatment and treatment at an earlier stage, and help minimize the morbidity and mortality of this disease. Mammography is currently the gold standard for early breast cancer detection and several analyses have shown that regular breast screening with mammography can significantly reduce the mortality rate due to breast cancer [1]. While the reported overall sensitivity of screening mammography is high (ranging from 71 to 96% [2,3]), the sensitivity is significantly reduced in certain subsets of women, particularly in women with radiographically dense breasts and those at increased risk of breast cancer [4,5]. Sensitivities of 33–59% have been reported for annual screening mammograms in women at increased risk [4]. Estimates of film mammographic sensitivity in women with extremely dense breasts range from 48 to 63% [6,7]. The introduction of digital mammography has led to only modest improvements in sensitivity in the subset of women who are under 50 years of age, pre- or peri-menopausal and have dense breasts [8]. Other modalities for breast imaging, such as ultrasound and MRI, have been extensively evaluated to determine their role in these subsets of women at increased risk. A study by the American College of Radiology Imaging Network (ACRIN) found that the combination of whole-breast screening, ultrasound and mammography in women with dense breasts and an elevated risk of breast cancer yielded a sensitivity of 77.5 versus 50% for mammography or ultrasound alone [9]. However, the addition of ultrasound substantially increased the number of false-positive findings. Further concerns include cost and generalizability, given that the ultrasound examinations were performed by radiologists who underwent specialized training, at an average time of 19 min per scan. To date, the most promising alternative has been contrast-enhanced breast MRI [5] and the American Cancer Society (ACS) has recently recommended its use for annual screening in women at high risk of breast cancer [10]. However, in doing so, the ACS also recognized the variable specificity of this modality, which can range from 50 to 90%. In addition, in the current economic climate, the high cost of breast MRI is likely to be a limiting factor in the widespread adoption of this imaging technique.
Recognizing the limitations of mammography, considerable effort has been expended over the last 5 years in either enhancing current imaging techniques (e.g., digital tomosynthesis [11] and 3D whole-breast ultrasound [12]) or in the development of new imaging modalities, such as breast CT [13], vibro-acoustography [14] or positron emission mammography (PEM) [15]. The purpose of this review is to present an update on new developments in breast imaging in the field of nuclear medicine with a focus on a new technique called molecular breast imaging (MBI).
Technical aspects of breast imaging in nuclear medicine
Scintimammography
The breast imaging techniques described below using small γ-cameras have as their origin an older technique called scintimammography. This technique used conventional γ-cameras to image the uptake of a tumor-seeking radiopharmaceutical, Tc-99m sestamibi, in breast tumors, and was extensively evaluated in the mid to late 1990s. While results showed good overall sensitivity and specificity, the technique was found to have poor sensitivity for the detection of small breast tumors [16,17]. In particular, its sensitivity was only approximately 50% for tumors less than 15 mm in diameter. At that time, the reason for the poor performance of scintimammography was thought to be partly due to the poor uptake of sestamibi in some tumors, and partly due to the limited resolving power of conventional γ-cameras in imaging the breast.
One of the primary factors that limited the resolution of images acquired with conventional γ-cameras was the large dead space at the edge of the camera requiring the patient to be imaged prone with the camera positioned laterally. This imaging position increased the distance between the breast and the camera, and resulted in considerable loss of resolution. Over the last 5–7 years significant progress has been made in the development of small compact γ-camera systems based on either multicrystal arrays of cesium iodide or sodium iodide, or alternatively utilizing semiconductor materials, such as cadmium zinc telluride (CZT) [18,19]. The total active area of these detectors is typically 20 × 20 cm or 20 × 15 cm. These small detectors overcome one of the primary limitations of conventional systems for breast imaging by reducing the dead space at the edge of the detector field of view from 8–10 cm down to 5–8 mm. The compact size and lack of dead space now allows these detectors to be used in a similar geometry to conventional mammography.
Breast-specific γ-imaging
Currently the only commercially available small compact γ-camera for breast imaging is the Dilon 6800 system, (Dilon Technologies, Newport News, VA, USA). This detector contains a 20 × 15 cm array of 3 × 3 mm sodium iodide crystals. The technique called breast-specific γ-imaging (BSGI) involves light compression of the breast between the detector head and a compression paddle. Results to date have demonstrated significantly better sensitivity for the detection of small breast lesions than those previously obtained with scintimammography [20,21].
Positron-emission mammography
As with conventional scintimammography, which has insufficient resolution for the detection of small breast tumors with Tc-99m sestamibi, PET using 2-deoxy-2-[18F] fluoro-d-glucose (FDG) also appears to have insufficient resolution for the detection of small tumors (under 10 mm), with a sensitivity of approximately 57% [22]. As a consequence, a new technology known as PEM has been developed that uses detectors comprising multicrystal arrays of either bismuth germanate or lutetium orthosilicate [23,24]. With PEM, two high-resolution detector heads are placed on opposite sides of a compressed breast, in a similar orientation to mammography. Tomographic slices through the breast can be obtained. The close proximity of the detectors to the breast results in an in-plane resolution of 1.5 mm compared with 4–6 mm for a conventional PET scanner. Preliminary results have shown comparable sensitivity to MRI for the detection of breast cancer with excellent specificity of higher than 90% [25].
CZT-based molecular breast imaging system
Small multicrystal γ-cameras that are used to image Tc-99m typically achieve energy resolutions of 12–20%, whereas CZT detectors can achieve energy resolution of 3–4%. This improvement in energy resolution translates into less scatter in the images and improved image contrast. In addition, current CZT detectors can be constructed with pixel sizes as small as 1.6 × 1.6 mm, with the possibility of even smaller pixel sizes in the future. Because the breast can be positioned in direct contact with the surface of the detector, image resolution is now highly dependent on the intrinsic resolving power of the detector. Multicrystal systems are typically limited to a minimum pixel size of approximately 3 mm. While these differences in intrinsic resolution are of little consequence in conventional nuclear medicine, they are fundamental to improvements in the resolving power of small detectors optimized for imaging the breast. A comparison of these various detector technologies has shown that CZT offers improved detection of sub-10 mm lesions over multicrystal detectors owing to these improvements in intrinsic spatial resolution and energy resolution [26]. The disadvantage of this material is that it is still relatively new and more expensive than the established detector materials.
For these reasons, several years ago we choose to utilize CZT-based detectors for the construction of a prototype breast imaging system. We labeled the technique MBI as the term scintimammography was not applicable to a nonscintillating technology. We opted for a design that employed two opposing CZT detectors with the breast lightly compressed between them. This configuration ensures that a breast lesion can never be more than half the breast thickness from a detector and improves the sensitivity of the technique for the detection of small breast tumors. Figure 1 shows one of the current dual-detector MBI systems in use at Mayo Clinic mounted on a modified mammographic gantry. This system utilizes two LumaGem 3200 detectors (Gamma Medica – Ideas, Northridge, CA, USA). The second unit utilizes two Alcyone detectors (GE Healthcare, Haifa, Israel) also mounted on a modified mammographic gantry. This configuration allows standard mammographic views (craniocaudal [CC]and mediolateral oblique [MLO]) to be obtained and facilitates easy comparison between images acquired with MBI and mammography. In addition to improving tumor detection, the use of a dual-detector configuration permits quantitative analysis of tumor uptake [27] and has been critical for implementation of some of the dose-reduction techniques that are discussed later.
Figure 1. Dual-head molecular breast imaging (MBI) system comprising two cadmium zinc telluride detectors mounted on a modified mammographic gantry.
To perform MBI, the breast is lightly compressed between the two detectors.
MBI scan procedure
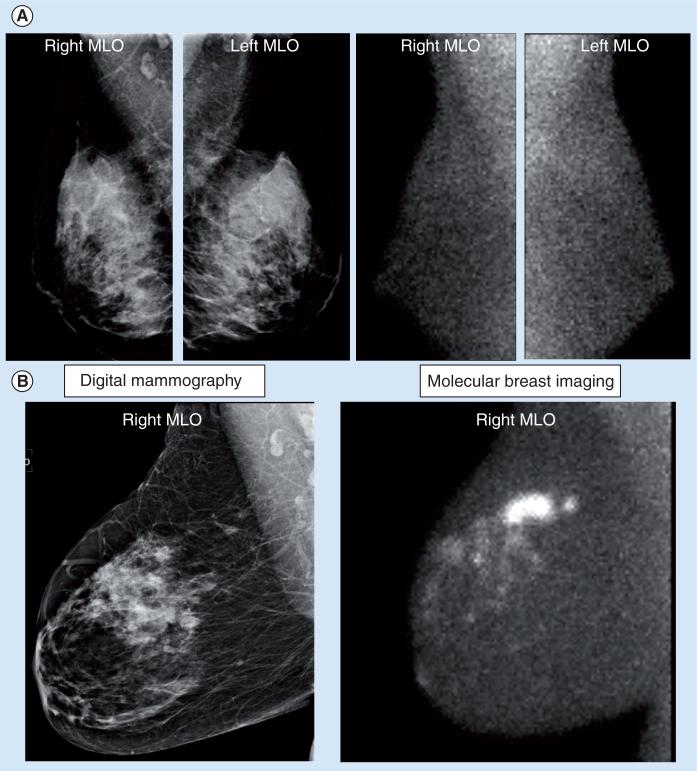
Current research studies on MBI have utilized a single injection of 20 mCi Tc-99m sestamibi with imaging of the breast commencing approximately 5 min postinjection. The breast is lightly compressed between the two detectors, using a compression force approximately a third that of mammography (~15 lb force). Typically, two 10-min images are obtained of each breast in the CC and MLO projections, giving a total imaging time of 40 min. Although the procedure time is longer than a mammogram, patients have indicated that the absence of pain associated with high compression outweighed the disadvantage of the longer imaging time. Other than the use of a lighter compression force, the procedural aspects of MBI are very comparable with conventional mammography and images are easily compared with conventional mammograms. Imaging is performed by nuclear medicine technologists trained in mammographic positioning techniques. Figure 2 shows examples of MBI studies in a normal subject and in a patient with confirmed invasive ductal carcinoma.
Figure 2. (A) Right and left MLO views obtained by digital mammography and molecular breast imaging (MBI) in a normal subject. (B) Right MLO views obtained by digital mammography and molecular breast imaging.
Mammogram showed a 1.1 cm indeterminate nodule in the medial right breast approximately 10 cm from the nipple. The MBI scan indicated a larger more extensive lesion, with multiple smaller adjacent lesions. Pathology confirmed the presence of extensive multifocal disease (invasive ductal carcinoma and ductal carcinoma in situ).
MLO: Mediolateral oblique.
Clinical MBI trials to date
Detection of small breast tumors
One of the first objectives that needed to be accomplished with MBI was a clear demonstration that MBI could reliably detect small breast lesions, that is, lesions smaller than 15 mm in diameter. The study was designed to select patients with small positive lesions on mammography and then determine whether or not MBI could likewise detect these lesions. Table 1 shows data from a study of 150 patients with Breast Imaging-Reporting and Data System (BI-RADS) 4–5 lesions identified on mammography that were estimated to be less than 20 mm in size [28]. MBI was performed prior to biopsy of the lesions and images were read by a panel of radiologists blinded to any other imaging data or clinical history. This study found that MBI had an overall sensitivity of 90%, with a sensitivity of 82% for all lesions less than 10 mm in size. Sensitivity was lowest for tumors less than 5 mm in size. One unanticipated benefit of MBI in this study was the detection of multifocal or multicentric disease in approximately 10% of patients initially considered to have a single lesion on the mammogram. This study found that tumor detection did not appear to be dependent on tumor type, but rather on tumor size. One very positive finding from this study was that Tc-99m sestamibi appeared to be an excellent tumor imaging agent and the failure to recognize its potential value in previous scintimammographic studies was primarily due to the inadequacy of conventional γ-camera technology for breast imaging.
Table 1.
Average sensitivity of molecular breast imaging as a function of tumor size during blinded review by three readers
| Tumor diameter (mm) | Tumors (n) | Average sensitivity (three readers) |
|---|---|---|
| 0–5 | 16 | 0.67 |
| 6–10 | 45 | 0.87 |
| 11–15 | 30 | 0.97 |
| 16–20 | 19 | 0.95 |
| > 20 | 18 | 1.00 |
| ≤ 10 | 61 | 0.82 |
| All tumors | 128 | 0.87 |
Adapted from [28].
Detection of DCIS & atypical breast lesions
While no study at the Mayo Clinic has yet directly evaluated the ability of MBI to detect ductal carcinoma in situ (DCIS), the combined results from various published studies to date showed no difference in the sensitivity of MBI for the detection of solid tumors (sensitivity: 91%), versus ductal carcinoma in situ (sensitivity: 94%) [28–30]. In addition, recent studies using a small multicrystal γ-camera showed a high sensitivity (>90%) for the detection of DCIS [20,21].
Uptake of sestamibi has been observed in a number of high-risk breast lesions, such as lobular carcinoma in situ, atypical ductal hyperplasia and atypical lobular hyperplasia. The role of MBI, PEM and BSGI in the evaluation of patients with high-risk lesions is not known at this time, but is under active investigation.
Comparison of MBI & breast MRI
Data comparing MBI with breast MRI are limited. Preliminary results in a small study of 54 cancers indicated comparable sensitivity (98% for breast MRI and 94% for MBI) [31]. Given the superior resolving power of breast MRI, MRI may demonstrate better sensitivity than MBI for the detection of very small breast lesions (i.e., those in the 2–4-mm range). However, the high resolution of MRI also contributes to the relatively low specificity in breast imaging. An ongoing prospective study comparing MBI and MRI directly will determine the relative sensitivity and specificity of these two imaging modalities. Preliminary studies with PEM and BSGI have also shown comparable sensitivity between these technologies and MRI [21,24].
Comparison of MBI & screening mammography
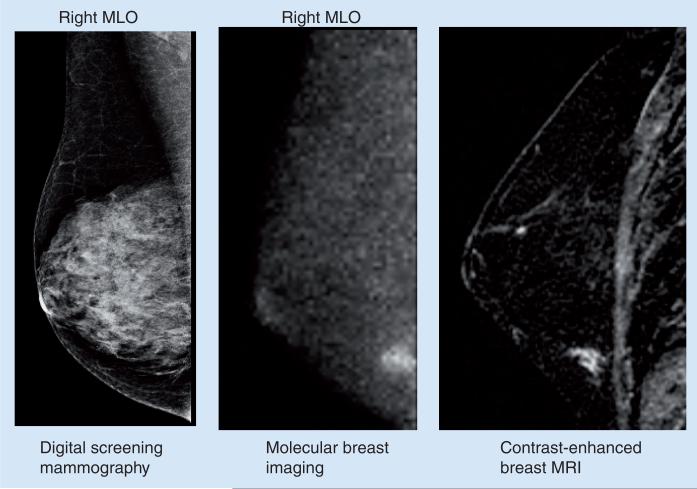
One of the most interesting studies to date with MBI has compared this technique with screening mammography in asymptomatic women with mammographically dense breast tissue and additional risk factors for breast cancer [32]. A total of 1007 women were recruited into this study over a 2.5-year period. All women had a screening mammogram and an MBI study performed within 21 days of each other. Breast status was assessed 12 months after enrollment: any cancer detected in the interim or on the subsequent annual mammogram that was not detected on the initial mammogram and MBI was considered as a false negative. In 904 patients with verified cancer status at 12-month follow-up, MBI detected ten out of 12 cancers, while mammography only detected three out of 12 cancers. Cancers missed by MBI included one case of DCIS 2–3 mm in size detected on the 12-month follow-up mammogram, and one case of DCIS approximately 5 mm in size detected at the initial screening mammogram. While the number of cancers is small, these results are very promising and would indicate that MBI has two- to three-times the sensitivity of mammography. One concern with MBI had been the possibility of a large number of false-positive cases. However, the recall rates were very comparable between the two modalities and were slightly lower for MBI (7.6%; 69/904) compared with mammography (9.2%; 83/904). Of the 904 patients, 893 patients had negative findings (no cancer or atypia), 810 patients had true negative findings on mammography and 830 patients had true negative findings on MBI, giving specificities of 90.71 and 93.0% for mammography and MBI, respectively. Figure 3 shows an example of a tumor detected by MBI but missed on screening mammography. The results with MBI in this high-risk population are similar to those reported for breast MRI in a comparable high-risk population [4].
Figure 3. Right MLO views obtained by digital mammography, molecular breast imaging and contrast-enhanced MRI.
Screening mammogram was interpreted as negative for disease. Molecular breast imaging indicated a small lesion in the lower quadrant of the right breast. Lesion was also seen on contrast enhanced breast MRI and was confirmed at surgery as a 9 mm ductal carcinoma in situ.
MLO: Mediolateral oblique.
MBI causes of false-positive results
There are a number of factors that can result in misleading results or can impact image interpretation. Tc-99m sestamibi is known to be taken up in cells with increased mitochondrial activity. Some benign processes, such as fibroadenomas, fat necrosis, benign papillomas and phyllodes tumors, can occasionally demonstrate increased uptake of the Tc-99m sestamibi. Correlation with the mammogram and clinical history is important for determining the significance of these positive scans. As mentioned earlier, high-risk breast lesions, such as atypical ductal hyperplasia, atypical lobular hyperplasia and atypical papillomas, will often demonstrate increased uptake of sestamibi, and further follow-up is required in these cases.
It should also be remembered that MBI represents a functional image of the breast tissue. We have found that in premenopausal women we occasionally observe nonlocalized patchy uptake of sestamibi in the breast, especially if the MBI study is performed within a few days following the peak of the luteal phase of the menstrual cycle. This phenomenon has only been observed in a small percentage of cases and more work is needed to elucidate the physiological basis for this uptake. This patchy uptake can potentially mask the presence of uptake in a tumor, particularly in diffuse lesions, such as DCIS.
False-positive findings on PEM with FDG have not been extensively reported; however, Berg et al. have noted uptake in a variety of benign process, such as fibroadenomas, fibrocystic changes and fat necrosis [25]. These false-positive findings are very similar to what we have observed for Tc-99m sestamibi with MBI.
Strengths & weaknesses of MBI
Molecular breast imaging has been studied in over 1500 patients at the Mayo Clinic during the last 6 years. In our experience, we have found the dedicated semiconductor-based MBI technology to offer optimal breast positioning analogous to mammography and superior spatial and energy resolution compared with conventional γ-cameras. From the body of work performed at Mayo and now underway at other laboratories, we believe that MBI has a potentially important role as an adjunct to mammography in both screening and diagnostic settings.
Because the uptake of Tc-99m sestamibi is independent of breast density, MBI may serve as a valuable imaging technique for women with mammographically dense breasts. Whether as a screening tool or in the evaluation of patients with known breast cancer, we have found that MBI consistently detects additional cancers in the breast that are occult on mammography, while maintaining a reasonable false-positive rate that is equal to or less than that of mammography. However, at present the ability of MBI to detect microcalcifications is unclear and, hence, MBI must be utilized in a similar manner to contrast-enhanced breast MRI (i.e., as an adjunct technique to mammography rather than an alternative).
In MBI, images obtained of the breast are analogous to those in mammography (CC and MCO), facilitating interpretation and comparison with mammography. One advantage of MBI over mammography is that heavy compression of breast tissues is not required. During MBI, light pain-free compression, approximately a third of the pressure of mammography, is applied, primarily to limit patient movement. The MBI procedure is generally well tolerated; an assessment of patient comfort resulted in lower pain scores with MBI than mammography [29]. An advantage of MBI over breast MRI is that it can be utilized in patients with contraindications to MRI, such as allergy to gadolinium, claustrophobia, cardiac pacemakers and implanted defibrillator, cochlear implants, metal such as aneurysm clips, and renal insufficiency. Interpretation of MBI images is also much less complex and time intensive than interpretation of MRI, as bilateral dual-head MBI generates eight images in comparison with upwards of 1500 images generated by bilateral breast MRI. The most striking difference between the two modalities is cost. In the USA, MRI is approximately ten to 15 times the cost of mammography. The cost of a MBI study in the USA has not been fully established but is estimated to be approximately twice that of mammography.
Currently, the main disadvantages of MBI are the long imaging time (4 × 10-min images) and the radiation dose associated with the injection of the Tc-99m sestamibi. Studies using multicrystal detectors have typically employed 25–30 mCi of Tc-99m sestamibi. With the dual-head CZT technology, we have utilized a dose of 20 mCi Tc-99m sestamibi. While this dose is low compared with doses used in many nuclear medicine procedures, it is high relative to that delivered by a mammogram. The effective dose from 20 mCi Tc-99m sestamibi is approximately 6.5 mSv [33]. By comparison, a screening mammogram has an effective dose of 0.7–1.0 mSv. Over the last year, considerable work has been performed at the Mayo Clinic to reduce the administered dose of Tc-99m sestamibi. Through optimization of the detector collimators, improved utilization of the energy spectra and innovative noise-reduction algorithms, it is anticipated that the administered dose can be reduced to less than 4 mCi, yielding effective doses of less than 1.3 mSv. This would make MBI comparable with screening mammography in terms of radiation exposure. This is a key step in the development and acceptance of MBI as a screening adjunct to mammography. A new study is currently underway at the Mayo Clinic to screen 1000 women with dense breast tissue on mammography who are at increased risk of breast cancer, using a low-dose MBI technique.
As molecular imaging of the breast becomes better established it is likely that a variety of specialized devices combining function (PET and SPECT) with anatomy (CT and breast tomosynthesis) will move from the physics laboratory into clinical trials. While these units may not be applicable to the screening of breast cancer, they will offer improved spatial resolution over what can be achieved with current MBI technology and, in combination with anatomical information, will probably result in their use as secondary diagnostic devices in clinical practice.
Alternative radiopharmaceuticals
To date, all studies with MBI have utilized Tc-99m sestamibi. However, there are a large number of other radiopharmaceuticals that hold tremendous promise for breast imaging. Studies are in progress to evaluate a new Tc-99m-labeled angiogenesis agent [34] as a tool for assessing the efficacy of neoadjuvant therapy. New Tc-99m-labeled analogs of glucose are also under evaluation for lung cancer [35], and should have comparable value in breast cancer. In addition, there are several existing older radiopharmaceuticals that need to be re-evaluated with the newer technology. Tc-99m sestamibi has proven to be significantly better for breast imaging than originally thought. It is likely that other compounds, such as Tc-99m DMSA (V) [36] and Tc-99m tetrofosmin [37], may also prove more useful than indicated from earlier clinical studies with scintimammography.
Expert commentary
Molecular breast imaging is a new way of providing high-resolution functional images of the breast. The potential applications of this technology range from evaluation of the extent of disease to a role as an adjunct screening technique in certain populations. We believe that it is highly complementary to existing anatomical techniques, such as mammography, tomosynthesis and ultrasound, and will have an important role to play in the diagnosis of breast cancer.
Five-year view
Over the next 1–2 years, we expect to see the development of low-dose high-resolution MBI units that provide a low-cost imaging alternative to MRI for screening in populations not well served by mammography and for problem solving in difficult clinical cases. In the current economic climate, the cost differential between MBI and breast MBI may accelerate this development. Interesting possibilities for the future include the merging of MBI with digital tomosynthesis and the development of dedicated SPECT/CT and PEM/CT devices for breast imaging. These types of system would provide coregistered functional and anatomical information on the breast, in the same way that PET/CT does today in oncology. This should also facilitate the biopsy of lesions positive on MBI but occult on mammography. Newer radiopharmaceuticals should enable MBI to accurately predict response to chemotherapy or neoadjuvant therapy and allow the physician to individualize patient treatment.
Key issues.
Molecular breast imaging (MBI) is a new nuclear medicine technique that employs small semiconductor γ-cameras for high-resolution imaging of the breast.
Imaging technique is similar to mammography but only uses a third the compression force.
Current studies employ Tc-99m sestamibi and provide functional images of the breast that are complementary to mammography.
Early results indicate that MBI has comparable sensitivity to breast MRI, but at a fraction of the cost per procedure.
In a screening study of women with dense breast tissue at increased risk of breast cancer, MBI detected three times as many tumors as mammography while maintaining a lower false positive rate.
Recent work has been aimed at reducing the radiation dose from MBI. The goal is to reduce the effective dose to the level of screening mammography.
MBI is complementary to anatomical techniques such as mammography, tomosynthesis and ultrasound and will have an important role to play in the diagnosis of breast cancer.
Financial & competing interests disclosure
This work was funded in part by grants from the Susan G Komen Foundation, NIH (CA 128407) and the Department of Defense (W81XWH-07–01–0548). Michael O'Connor has provisional patents filed on tumor quantification and dose reduction techniques for molecular breast imaging and has received a research grant from GE Healthcare. Carrie Hruska has provisional patents filed on tumor quantification and dose reduction techniques for molecular breast imaging. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
No writing assistance was utilized in the production of this manuscript.
Contributor Information
Michael O'Connor, Department of Radiology, Mayo Clinic, Rochester, MN 55905, USA Tel.: +1 507 284 7083 Fax: +1 507 266 4461 mkoconnor@mayo.edu.
Deborah Rhodes, Department of Internal Medicine, Mayo Clinic, Rochester, MN 55905, USA Tel.: +1 507 284 7083 Fax: +1 507 284 0909 rhodes.deborah@mayo.edu.
Carrie Hruska, Department of Radiology, Mayo Clinic, Rochester, MN 55905, USA Tel.: +1 507 284 7083 Fax: +1 507 266 4461 hruska.carrie@mayo.edu.
References
Papers of special note have been highlighted as:
• of interest
•• of considerable interest
- 1.Berry DA, Cronin KA, Plevritis SK, et al. Effect of screening and adjuvant therapy on mortality from breast cancer. N. Engl. J. Med. 2005;353:1784–1792. doi: 10.1056/NEJMoa050518. [DOI] [PubMed] [Google Scholar]
- 2.Mushlin AI, Kouides RW, Shapiro DE. Estimating the accuracy of screening mammography: a meta-analysis. Am. J. Prev. Med. 1998;14(2):143–153. doi: 10.1016/s0749-3797(97)00019-6. [DOI] [PubMed] [Google Scholar]
- 3.Humphrey LL, Helfand M, Chan BKS, Woolf SH. Breast cancer screening: a summary of the evidence for the USA Preventive Services Task Force. Ann. Intern. Med. 2002;137:347–360. doi: 10.7326/0003-4819-137-5_part_1-200209030-00012. [DOI] [PubMed] [Google Scholar]
- 4••.Sardanelli F, Podo F, D'Agnolo G, et al. Multicenter comparative multimodality surveillance of women at genetic-familial high risk for breast cancer (HIBCRIT study): interim results. Radiology. 2007;242:698–715. doi: 10.1148/radiol.2423051965. [•• of considerable interestLarge multicenter study that highlights the limitations of mammography in women at high risk of breast cancer] [DOI] [PubMed] [Google Scholar]
- 5.Lehman CD. Role of MRI in screening women at high risk for breast cancer. J. Magn. Reson. Imaging. 2006;24:964–970. doi: 10.1002/jmri.20752. [DOI] [PubMed] [Google Scholar]
- 6.Leconte I, Feger C, Galant C, et al. Mammography and subsequent whole-breast sonography of nonpalpable breast cancers: the importance of radiologic breast density. Am. J. Roentgenol. 2003;180:1675–1679. doi: 10.2214/ajr.180.6.1801675. [DOI] [PubMed] [Google Scholar]
- 7.Kolb TM, Lichy J, Newhouse JH. Comparison of the performance of screening mammography, physical examination, and breast ultrasound and evaluation of factors that influence them: an analysis of 27,825 patient evaluations. Radiology. 2002;225:165–175. doi: 10.1148/radiol.2251011667. [DOI] [PubMed] [Google Scholar]
- 8•.Pisano ED, Gatsonis C, Hendrick E, et al. Diagnostic performance of digital versus film mammography for breast-cancer screening. N. Engl. J. Med. 2005;353(17):1773–1783. doi: 10.1056/NEJMoa052911. [• of interestA 50,000 patient study comparing digital and film mammography] [DOI] [PubMed] [Google Scholar]
- 9•.Berg WA, Blume JD, Cormack JB, et al. For the ACRIN 6666 investigators combined screening with ultrasound and mammography versus mammography alone in women at elevated risk of breast cancer. JAMA. 2008;299(18):2151–2163. doi: 10.1001/jama.299.18.2151. [• of interestACRIN multicenter study comparing ultrasound to mammography.] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10•.Saslow D, Boetes C, Burke W, et al. American cancer society guidelines for breast screening with MRI as an adjunct to mammography. CA. Cancer J. Clin. 2007;57:75–89. doi: 10.3322/canjclin.57.2.75. [• of interestProvides overview of strengths and limitations of breast MRI] [DOI] [PubMed] [Google Scholar]
- 11.Rafferty EA. Digital mammography: novel applications. Radiol. Clin. North Am. 2007;45(5):831–843. doi: 10.1016/j.rcl.2007.06.005. [DOI] [PubMed] [Google Scholar]
- 12.Weismann CF, Datz L. Diagnostic algorithm: how to make use of new 2D, 3D and 4D ultrasound technologies in breast imaging. Eur. J. Radiol. 2007;64(2):250–257. doi: 10.1016/j.ejrad.2007.07.025. [DOI] [PubMed] [Google Scholar]
- 13.Karellas A, Vedantham S. Breast cancer imaging: a perspective for the next decade. Med. Phys. 2008;35(11):4878–4897. doi: 10.1118/1.2986144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Alizad A, Whaley DH, Greenleaf JF, Fatemi M. Critical issues in breast imaging by vibro-acoustography. Ultrasonics. 2006;44(Suppl 1):e217–e220. doi: 10.1016/j.ultras.2006.06.021. [DOI] [PubMed] [Google Scholar]
- 15.Raylman RR, Majewski S, Smith MF, et al. The positron emission mammography/tomography breast imaging and biopsy system (PEM/PET): design, construction and phantom-based measurements. Phys. Med. Biol. 2008;53(3):637–653. doi: 10.1088/0031-9155/53/3/009. [DOI] [PubMed] [Google Scholar]
- 16.Taillefer R. The role of 99mTc sestamibi and other conventional radiopharmaceuticals in breast cancer diagnosis. Semin. Nucl. Med. 1999;29:16–40. doi: 10.1016/s0001-2998(99)80027-0. [DOI] [PubMed] [Google Scholar]
- 17.Waxman AD. The role of (99m) Tc methoxyisobutylisonitrile in imaging breast cancer. Semin. Nucl. Med. 1997;27:40–54. doi: 10.1016/s0001-2998(97)80035-9. [DOI] [PubMed] [Google Scholar]
- 18.Brem RF, Schoonjans JM, Kieper DA, Majewski S, Goodman S, Civelek C. High resolution scintimammography: a pilot study. J. Nucl. Med. 2002;43:909–915. [PubMed] [Google Scholar]
- 19.Coover LR, Caravaglia G, Kuhn P. Scintimammography with dedicated breast camera detects and localizes occult carcinoma. J. Nucl. Med. 2004;45:553–558. [PubMed] [Google Scholar]
- 20.Brem RF, Floerke AC, Rapelyea JA, Teal C, Kelly T, Mathur V. Breast-specific γ imaging as an adjunct imaging modality for the diagnosis of breast cancer. Radiology. 2008;247:651–657. doi: 10.1148/radiol.2473061678. [DOI] [PubMed] [Google Scholar]
- 21.Brem RF, Fishman M, Rapelyea JA. Detection of ductal carcinoma in situ with mammography, breast specific γ imaging, and magnetic resonance imaging: a comparative study. Acad. Radiol. 2007;14:945–950. doi: 10.1016/j.acra.2007.04.004. [DOI] [PubMed] [Google Scholar]
- 22.Avril N, Rosé CA, Schelling M, et al. Breast imaging with positron emission tomography and fluorine-18 fluorodeoxyglucose: use and limitations. J. Clin. Oncol. 2000;18:3495–3502. doi: 10.1200/JCO.2000.18.20.3495. [DOI] [PubMed] [Google Scholar]
- 23.Raylman RR, Majewski S, Smith MF, et al. The positron emission mammography/tomography breast imaging and biopsy system (PEM/PET): design, construction and phantom-based measurements. Phys. Med. Biol. 2008;53:637–653. doi: 10.1088/0031-9155/53/3/009. [DOI] [PubMed] [Google Scholar]
- 24.Schilling K, Conti P, Adler L, Tafra L. The role of positron emission mammography in breast cancer imaging and management. Applied Radiol. 2008;37:26–36. [Google Scholar]
- 25.Berg WA, Weinberg IN, Narayanan D, et al. High-resolution fluorodeoxyglucose positron emission tomography with compression (“positron emission mammography”) is highly accurate in depicting primary breast cancer. Breast J. 2006;12:309–323. doi: 10.1111/j.1075-122X.2006.00269.x. [DOI] [PubMed] [Google Scholar]
- 26••.Hruska CB, O'Connor MK, Collins DA. Comparison of small field of view γ camera systems for scintimammography. Nucl. Med. Commun. 2005;26:441–445. doi: 10.1097/00006231-200505000-00008. [•• of considerable interestHead-to-head comparison of the various technologies available for small field of view γ cameras] [DOI] [PubMed] [Google Scholar]
- 27.Hruska CB, O'Connor MK. Development of a lesion quantification method for dual-head molecular breast imaging. Med. Phys. 2008;35:1365–1376. doi: 10.1118/1.2885371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28•.Hruska CB, Phillips SW, Whaley DH, Rhodes DJ, O'Connor MK. Molecular breast imaging: use of a dual-head dedicated γ camera for detection of small breast tumors. AJR Am J Roentgenol. 2008;191:1805–1815. doi: 10.2214/AJR.07.3693. [• of interestShows advantages of a dual-head system compared with a single-head system in detecting small breast lesions] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.O'Connor MK, Phillips SW, Hruska CB, Rhodes DJ, Collins DA. Molecular breast imaging: advantages and limitations of a scintimammographic technique in patients with small breast tumors. Breast J. 2007;13:3. doi: 10.1111/j.1524-4741.2006.00356.x. [DOI] [PubMed] [Google Scholar]
- 30.Hruska CB, Boughey JC, Phillips SW, et al. Molecular breast imaging: a review of the Mayo clinic experience. Am. J. Surg. 2008;196:470–476. doi: 10.1016/j.amjsurg.2008.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hruska CB, Phillips SW, Rhodes DJ, O'Connor MK. Concordance of molecular breast imaging and breast MRI findings: a retrospective review. Eur. J. Nucl. Med. Mol. Imag. 2008;35(Suppl 2):S200. [Google Scholar]
- 32••.Hruska CB, Rhodes DJ, Phillips SW, Whaley DH, Alabin TT, O'Connor MK. Molecular breast imaging for breast cancer screening in women with mammographically dense breasts and increased risk. Eur. J. Nucl. Med. Mol. Imag. 2008;35(Suppl 2):S200. [abstract] [•• of considerable interestAbstract only, but presents important data on advantages of molecular breast imaging over mammography in a screening environment] [Google Scholar]
- 33.Hesse B, Tägil K, Cuocolo A, et al. EANM/ESC Procedural guidelines for myocardial perfusion imaging in nuclear cardiology. Eur J. Nucl. Med. 2005;32:855–897. doi: 10.1007/s00259-005-1779-y. [DOI] [PubMed] [Google Scholar]
- 34.Bach-Gansmo T, Danielsson R, Saracco A, et al. Integrin receptor imaging of breast cancer: a proof-of-concept study to evaluate 99mTc-NC100692. J. Nucl. Med. 2006;47(9):1434–1439. [PubMed] [Google Scholar]
- 35.Bryant J, Yang D, Schechter N, et al. Evaluation of Tc-99m-EC-DG in lung cancer patients. J. Nucl. Med. 2007;48(Suppl 2):354P. [Google Scholar]
- 36.Papantoniou V, Tsiouris S, Koutsikos J, et al. Scintimammographic detection of usual ductal hyperplasia with increased proliferation rate at risk for malignancy. Nucl. Med. Commun. 2006;27:911–917. doi: 10.1097/01.mnm.0000243367.73141.85. [DOI] [PubMed] [Google Scholar]
- 37.Mansi L, Rambaldi PF, Procaccini E, et al. Scintimammography with technetium-99m tetrofosmin in the diagnosis of breast cancer and lymph node metastases. Eur. J. Nucl. Med. 1996;23:932–939. doi: 10.1007/BF01084367. [DOI] [PubMed] [Google Scholar]