Mitochondria form a highly dynamic filamentous network. The elongated shape of the individual mitochondrion, visualized by fluorescence microscopy, contrasts with the classic text book representation that shows the mitochondrion as an elliptical short rod-like structure. The networks are in constant motion and undergo continuous remodeling through the fusion of mitochondria with each other and the fission of filamentous mitochondria into shorter fragments (1–3). Both processes are mediated by a few core fusion/fission proteins. Tethering and fusion of the outer mitochondrial membrane is mediated by the related GTPases Mitofusin1 and 2, whereas OPA1 catalyzes the fusion of the inner mitochondrial membrane. Fis1 catalyzes the assembly of the dynamin-related protein Drp1 into oligomeric complexes on the outer mitochondrial membrane to drive mitochondrial scission. Mutations in OPA1 and Mfn2 cause, respectively, autosomal dominant optic atrophy and peripheral neuropathy (Charcot-Marie-Tooth disease) associated with insulin resistance in type 2 diabetes (1,2,4,5).
The ability of mitochondria to undergo fusion/fission dynamics is intimately linked with mitochondrial function. Mitochondria may use this mechanism for even distribution of metabolites, lipids, and proteins. Fission is also used to mediate the removal of nonfunctional or aged mitochondria to be targeted for autophagy as also shown for β-cells (6). Fusion/fission proteins have attracted attention for their close link with mitochondrial respiration/energy metabolism, impact on cristae structure, and mitochondria-linked cell death (3,4,7,8).
This research topic is of particular interest in the pancreatic β-cell given the essential role of mitochondria in metabolism-secretion coupling (9). In this cell type, mitochondria form a very dense meshwork (6,8,10). In primary rodent and human β-cells, the average length of mitochondria estimated by fluorescence microscopy varies dramatically (1–10 μm) (8). Interestingly, mitochondria in β-cells of islets from Zucker diabetic fatty rats are fragmented, suggesting an imbalance in the regulation of mitochondrial fusion/fission (10). These findings uncovered a first possible link among mitochondrial morphology, β-cell dysfunction, and type 2 diabetes.
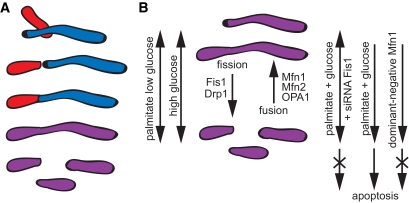
The study of Molina et al. (11) in this issue of Diabetes has further characterized mitochondrial morphology and dynamics in the pancreatic β-cell. The authors demonstrate that palmitate causes mitochondrial fragmentation and impairs network dynamics, an effect aggravated by elevated glucose concentrations (11). Two-photon laser microscopy of matrix-targeted photoactivatable green fluorescent protein (PAGFPmt) was used to label individual mitochondria. Surprisingly, the large majority of the mitochondria analyzed with this method were short (76% less than 2 μm). Previous findings using the same methodology reported highly elongated mitochondria in several cell types (12). Standard morphometric methods by Molina et al. show that ∼60% of the cells have length-to-width ratios of 2 or more, and 30% have elongated mitochondria with length-to-width ratios larger than 4. Taken together, findings with the two different methods would argue that matrix boundaries may be shorter than the actual mitochondrial length either because structures that appear as single filaments are composed of aligned mitochondrial rods or because matrix proteins are separated by nonfused inner mitochondrial membranes (Fig. 1A). In the primary β-cell mitochondrial fusion is rapidly followed by fission, offering a kinetic explanation for the rather short mitochondrial rods (6). Short mitochondria are apparently sufficient to execute the essential function of energy metabolism and metabolism-secretion coupling. This is also supported by the finding that DnMfn1 causes dramatic fragmentation of the mitochondrial network but has no impact on glucose-dependent ATP synthesis, insulin granule exocytosis, or induction of cell death (8) (Fig. 1B).
FIG. 1.
A: Scheme illustrating how mitochondrial matrix components (red and blue) spread during fusion/fission (purple). The red-colored mitochondria are perceived as short rods using PAGFPmt, appearing elongated when classic morphometric analysis is applied. B: Impact of nutrients on the balance of mitochondrial fusion/fission as executed by the core protein machinery. Apoptosis is observed when fragmentation is caused by glucolipotoxic conditions.
The individual mitochondrial units in β-cells, although short, are in very active communication with the rest of the mitochondrial network (11). Molina et al. follow the dilution of PAGFPmt with an elegant readout for the sharing of mitochondrial components mediated by mitochondrial fusion/fission. Within 50 min, the PAGFPmt activated in a small fraction of the total pool of mitochondria distributed throughout the network. Possibly the main finding of this study is that a combination of fatty acid and high glucose concentrations, conditions of glucolipotoxicity (13), almost abolished fusion/fission activity, also resulting in pronounced mitochondrial fragmentation (Fig. 1B). It is noteworthy that palmitate alone shifted the balance toward fragmentation without affecting the sharing of PAGFPmt in the network. The inhibition of mitochondrial fusion/fission was already observed 4 h after high-fat glucose treatment. The time course suggests moment-to-moment signaling to the mitochondria rather than altered gene expression. Palmitate could promote mitochondrial fragmentation by inducing endoplasmic reticulum (ER) stress, which indeed occurs rapidly. Palmitate depletes the ER calcium stores over a similar time course (14), which results in elevated cytosolic calcium. Subsequently, calcium activates the phosphatase calcineurin, which together with the ER stress–induced C/EBP homologous protein (CHOP) initiates apoptosis (14,15). Drp1 dephosphorylation by calcineurin is a likely link between the ER-mediated calcium rise and mitochondrial fragmentation as demonstrated for other cells (16). The dephosphorylated Drp1 is recruited to the mitochondrial outer membrane and binds to Fis1 to mediate mitochondrial scission. In agreement with such a mechanism, Molina et al. report that siRNA-mediated suppression of Fis1 prevents Drp1 recruitment and mitochondrial fragmentation induced by high-fat glucose treatment. Of interest, inhibition of calcineurin activation partially prevented palmitate-evoked apoptosis (15). Fis1 downregulation also counteracts the effect of high fat on apoptosis but not its suppression of glucose-stimulated insulin secretion in clonal β-cells (11). The antiapoptotic effect after inhibition of fission is consistent with findings in neurons (7) and probably occurs via inactivation of the proapoptotic Bcl-2 family members Bax and Bak (2). However, induction of fission per se impairs neither cell viability nor insulin secretion (8). We can conclude that mitochondrial dysfunction in response to fatty acid stress is not limited to the regulation of mitochondrial dynamics.
In general, the connection between substrate conditions and mitochondrial dynamics is of central interest because this is a means of linking nutrient availability to mitochondrial energy metabolism. Further understanding of this link should result in the identification of pharmacological targets for the protection of β-cell function and β-cell mass in glucose-intolerant and type 2 diabetic subjects.
Acknowledgments
The authors acknowledge funding by the Swiss National Foundation (310000-116750/1) and EuroDia (LSHM-CT-2006-518153), a European Community–funded project under framework program six.
No potential conflicts of interest relevant to this article were reported.
Footnotes
See accompanying original article, p. 2303.
REFERENCES
- 1.Chan DC: Mitochondrial fusion and fission in mammals. Annu Rev Cell Dev Biol 2006;22:79–99 [DOI] [PubMed] [Google Scholar]
- 2.Jourdain A, Martinou JC: Mitochondrial outer-membrane permeabilization and remodelling in apoptosis. Int J Biochem Cell Biol 9May2009. [Epub ahead of print] [DOI] [PubMed] [Google Scholar]
- 3.Benard G, Rossignol R: Ultrastructure of the mitochondrion and its bearing on function and bioenergetics. Antioxid Redox Signal 2008;10:1313–1342 [DOI] [PubMed] [Google Scholar]
- 4.Frezza C, Cipolat S, Martins de Brito O, Micaroni M, Beznoussenko GV, Rudka T, Bartoli D, Polishuck RS, Danial NN, De Strooper B, Scorrano L: OPA1 controls apoptotic cristae remodeling independently from mitochondrial fusion. Cell 2006;126:177–189 [DOI] [PubMed] [Google Scholar]
- 5.Zorzano A, Liesa M, Palacin M: Mitochondrial dynamics as a bridge between mitochondrial dysfunction and insulin resistance. Arch Physiol Biochem 2009;115:1–12 [DOI] [PubMed] [Google Scholar]
- 6.Twig G, Elorza A, Molina AJ, Mohamed H, Wikstrom JD, Walzer G, Stiles L, Haigh SE, Katz S, Las G, Alroy J, Wu M, Py BF, Yuan J, Deeney JT, Corkey BE, Shirihai OS: Fission and selective fusion govern mitochondrial segregation and elimination by autophagy. Embo J 2008;27:433–446 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Barsoum MJ, Yuan H, Gerencser AA, Liot G, Kushnareva Y, Graber S, Kovacs I, Lee WD, Waggoner J, Cui J, White AD, Bossy B, Martinou JC, Youle RJ, Lipton SA, Ellisman MH, Perkins GA, Bossy-Wetzel E: Nitric oxide-induced mitochondrial fission is regulated by dynamin-related GTPases in neurons. Embo J 2006;25:3900–3911 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Park KS, Wiederkehr A, Kirkpatrick C, Mattenberger Y, Martinou JC, Marchetti P, Demaurex N, Wollheim CB: Selective actions of mitochondrial fission/fusion genes on metabolism-secretion coupling in insulin-releasing cells. J Biol Chem 2008;283:33347–33356 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wiederkehr A, Wollheim CB: Impact of mitochondrial calcium on the coupling of metabolism to insulin secretion in the pancreatic beta-cell. Cell Calcium 2008;44:64–76 [DOI] [PubMed] [Google Scholar]
- 10.Bindokas VP, Kuznetsov A, Sreenan S, Polonsky KS, Roe MW, Philipson LH: Visualizing superoxide production in normal and diabetic rat islets of Langerhans. J Biol Chem 2003;278:9796–9801 [DOI] [PubMed] [Google Scholar]
- 11.Molina AJA, Wikstrom JD, Stiles L, Las G, Mohamed H, Elorza A, Walzer G, Twig G, Katz S, Corkey BE, Shirihai OS: Mitochondrial networking protects β-cells from nutrient-induced apoptosis. Diabetes 2009;58:2303–2315 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Karbowski M, Arnoult D, Chen H, Chan DC, Smith CL, Youle RJ: Quantitation of mitochondrial dynamics by photolabeling of individual organelles shows that mitochondrial fusion is blocked during the Bax activation phase of apoptosis. J Cell Biol 2004;164:493–499 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Poitout V, Robertson RP: Glucolipotoxicity: fuel excess and beta-cell dysfunction. Endocr Rev 2008;29:351–366 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cunha DA, Hekerman P, Ladriere L, Bazarra-Castro A, Ortis F, Wakeham MC, Moore F, Rasschaert J, Cardozo AK, Bellomo E, Overbergh L, Mathieu C, Lupi R, Hai T, Herchuelz A, Marchetti P, Rutter GA, Eizirik DL, Cnop M: Initiation and execution of lipotoxic ER stress in pancreatic beta-cells. J Cell Sci 2008;121:2308–2318 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Choi SE, Kim HE, Shin HC, Jang HJ, Lee KW, Kim Y, Kang SS, Chun J, Kang Y: Involvement of Ca2+-mediated apoptotic signals in palmitate-induced MIN6N8a beta cell death. Mol Cell Endocrinol 2007;272:50–62 [DOI] [PubMed] [Google Scholar]
- 16.Cereghetti GM, Stangherlin A, Martins de Brito O, Chang CR, Blackstone C, Bernardi P, Scorrano L: Dephosphorylation by calcineurin regulates translocation of Drp1 to mitochondria. Proc Natl Acad Sci U S A 2008;105:15803–15808 [DOI] [PMC free article] [PubMed] [Google Scholar]