Abstract
In response to osmolarity, Salmonella enterica serotype Typhi (S. Typhi) regulates genes required for Vi capsular antigen expression oppositely to those required for motility and invasion. Previous studies suggest that osmoregulation of motility, invasion and capsule expression is mediated through the RcsC/RcsD/RcsB phosphorelay system. Here we performed gene expression profiling and functional studies to determine the role of TviA, an auxiliary protein of the RcsB response regulator, in controlling virulence gene expression in S. Typhi. TviA repressed expression of genes encoding flagella and the invasion associated type III secretion system (T3SS-1) through repression of the flagellar regulators flhDC and fliZ, resulting in reduced invasion, reduced motility and reduced expression of FliC. Both RcsB and TviA repressed expression of flhDC, but only TviA altered flhDC expression in response to osmolarity. Introduction of tviA into S. enterica serotype Typhimurium rendered flhDC transcription sensitive to changes in osmolarity. These data suggest that the auxiliary TviA protein integrates a new regulatory input into the RcsB regulon of S. Typhi, thereby altering expression of genes encoding flagella, the Vi antigen and T3SS-1 in response to osmolarity.
Keywords: Typhoid fever, RcsCDB phosphorelay, flagellar and invasion gene regulation
INTRODUCTION
Salmonella enterica serotype Typhi (S. Typhi) causes a severe systemic infection termed Typhoid fever. In contrast, infection with the closely related S. enterica serotype Typhimurium (S. Typhimurium) leads to gastroenteritis in immunocompetent individuals, a localized infection of the terminal ileum and colon. Tissue invasion by S. Typhimurium, but not by S. Typhi, results in a strong exudative intestinal inflammation (reviewed in (Tsolis et al., 2008)). These observations indicate that S. Typhi must have acquired new virulence genes and/or altered expression of preexisting virulence factors to alter its clinical presentation.
One DNA region that is present in S. Typhi but absent from the S. Typhimurium chromosome is a stretch of 134 kb termed Salmonella pathogenicity island (SPI)-7 (Parkhill et al., 2001, McClelland et al., 2001, Deng et al., 2003). The presence of tRNA genes upstream and downstream of SPI-7 suggest that this region was acquired by a horizontal gene transfer event. Encoded within SPI-7 is the viaB locus, a 14kb DNA region, containing genes required for the regulation (tviA), the biosynthesis (tviBCDE), and the export (vexABCDE) of the Vi capsular antigen (Johnson & Baron, 1969, Virlogeux et al., 1995). SPI-7 is genetically unstable (Bueno et al., 2004, Nair et al., 2004), thereby giving rise to Vi antigen negative S. Typhi isolates. Comparison of Vi positive and Vi negative S. Typhi isolates suggests that SPI-7 significantly alters virulence properties, including phagocytosis (Looney & Steigbigel, 1986) and pro-inflammatory responses (Sharma & Qadri, 2004, Hirose et al., 1997). Recently, the viaB locus was identified as the DNA region within SPI-7 responsible for reducing inflammatory responses during S. Typhi infection (Raffatellu et al., 2005, Raffatellu et al., 2007, Wilson et al., 2008, Winter et al., 2008). These observations underline that acquisition of the viaB locus was a critical event in the evolution of the S. Typhi lineage.
Acquisition of the viaB locus introduced a novel regulatory protein, TviA, an activator of the tviABCDEvexABCDE operon (Virlogeux et al., 1995, Virlogeux et al., 1996). TviA activates its own transcription and is thought to serve as an auxiliary protein for RcsB (Virlogeux et al., 1996, Majdalani & Gottesman, 2005), the response regulator of the RcsC/RcsD/RcsB phosphorelay system. RcsB is required for transcription of the tviA gene (Pickard et al., 1994, Virlogeux et al., 1995, Virlogeux et al., 1996). The Rcs phosphorelay system controls capsular biosynthesis in several members of the Enterobacteriaceae and consist of a hybrid membrane bound signal transducer (RcsC and RcsD) and a cytosolic response regulator (RcsB). Auxiliary proteins of RcsB allow additional signal input by altering the activity of this response regulator (reviewed in (Majdalani & Gottesman, 2005)).
Expression of flagella and the invasion associated type III secretion system (T3SS-1) is reduced under low osmolarity in S. Typhi compared to S. Typhimurium (Arricau et al., 1998). Since the Rcs phosphorelay system controls expression of the corresponding genes, it has been proposed that S. Typhi specific osmoregulation is mediated by the Rcs system (Arricau et al., 1998). However, the role of the horizontally acquired regulator TviA in altering gene expression in S. Typhi compared to other serotypes has received little attention. The goal of this study was to investigate what genes outside the viaB locus are regulated by TviA and how the acquisition of this new auxiliary protein altered behavior of the preexisting RcsB regulatory network.
RESULTS
Genes regulated by RcsB and TviA identified by gene expression profiling
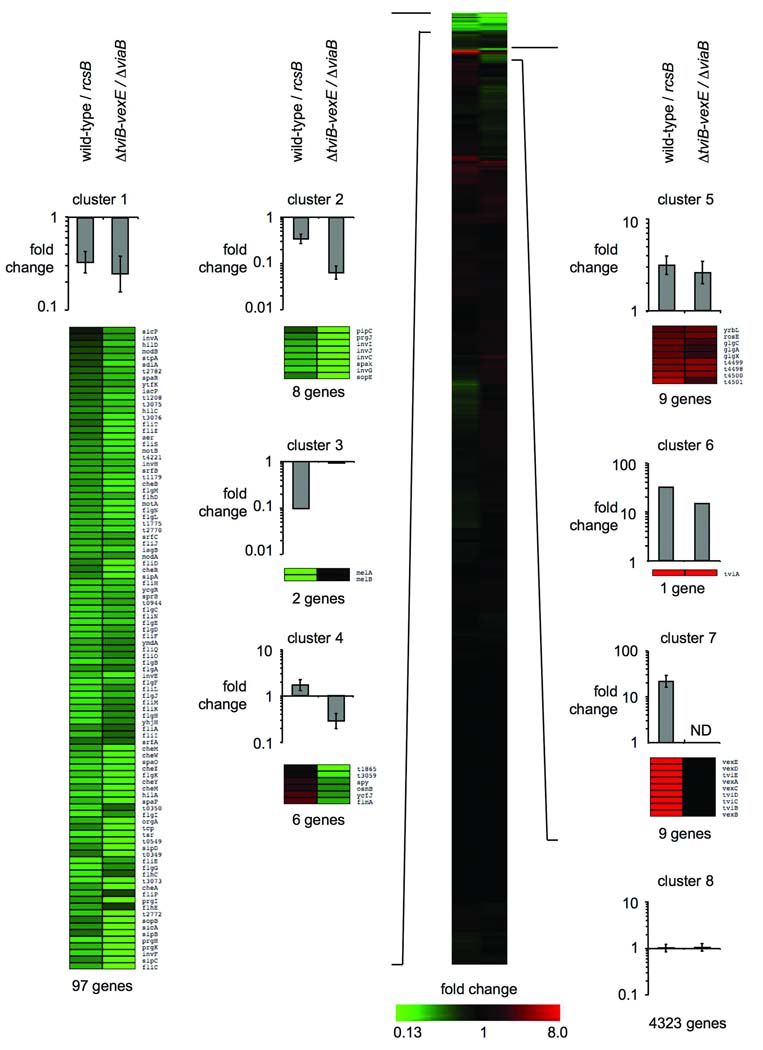
Previous reports have demonstrated that the Rcs phosphorelay system and the TviA auxiliary protein are involved in the transcriptional control of virulence factors in S. Typhi (Virlogeux et al., 1996, Arricau et al., 1998, Winter et al., 2008). To determine the extent of regulatory overlap between the response regulator RcsB and the auxiliary protein TviA, gene expression profiling was performed on S. Typhi strains grown in low salt media, which either carried or lacked the rcsB or the tviA gene (supplementary table 1). Low salt conditions are known to induce expression of tviA and the viaB operon (Pickard et al., 1994, Raffatellu et al., 2005). To assess TviA regulation in the absence of secondary effects potentially arising from expression of the Vi antigen, we compared gene expression profiles of a ΔtviB-vexE mutant (SW74) with those observed in a ΔviaB mutant (STY2) (Fig. 1).
Figure 1. Cluster analysis of RcsB and TviA regulated gene expression levels in S. Typhi.
Gene expression profiling was performed on bacterial strains (wild-type strain, rcsB mutant, ΔviaB mutant or ΔtviB-vexE mutant) grown under low osmolarity condition (SOB broth). The CAST algorithm was used to identify similarities in gene expression between samples. Bars above the heat maps represent geometric means of fold change ± standard deviation for cluster of genes repressed (clusters 1–4) or activated (clusters 5–7) by RcsB and/or TviA or genes with no change in gene expression (cluster 8). The number of genes within each cluster is indicated below each heat map. ND: none detected.
Since RcsB is an activator of critical importance for tviA transcription (Virlogeux et al., 1996, Virlogeux et al., 1995) (Fig. 1, cluster 6; Fig. 2A), but TviA does not regulate rcsB transcriptionally (Fig. 2B), we expected that TviA regulated genes would be either identical to or would constitute a subset of RcsB regulated genes. Cluster analysis of gene expression profiles revealed that both RcsB and TviA influence the transcription of similar regulatory circuits: RcsB and TviA repressed transcription of genes encoding components of the T3SS-1, genes involved in chemotaxis, flagellar regulation and flagellar biosynthesis (Fig. 1, clusters 1 and 2). Transcription of genes involved in carbohydrate anabolism (glgA, glgC and glgX) and genes encoding an arginine deiminase system were positively regulated by RcsB and TviA (Fig. 1, cluster 5). In addition, RcsB also activated transcription of the viaB operon (Fig. 1, clusters 6 and 7). As expected, no mRNA of the tviBCDEvexABCDE genes was detected in extracts from the ΔtviB-vexE mutant (SW74) and the ΔviaB mutant (STY2) employed in the gene expression experiment (Fig. 1, cluster 7). In subsequent studies, we focused on the major regulatory networks identified by our analysis, namely regulation of the flagella regulon and the T3SS-1 by RcsB and TviA.
Figure 2. Influence of RcsB and TviA on transcription of tviA and rcsB in S. Typhi.
(A) The S. Typhi wild-type strain (Ty2) and the isogenic ΔrcsB mutant (SW513), were grown in low osmolarity medium (tryptone yeast extract broth) under aerobic conditions. RNA was extracted and transcription of tviA was determined by real time qRT-PCR. Bars represent the geometric mean of mRNA levels (in percent of wild-type strain levels) ± standard error. (B) The S. Typhi wild-type strain (Ty2), an isogenic ΔviaB mutant (STY2), a rcsB::lacZYA mutant (SW237), and a ΔviaB rcsB::lacZYA (SW239) were grown in low osmolarity medium (SOB broth) under aerobic conditions and β-galactosidase activity was measured. Bars represent the geometric mean of three independent experiments ± standard error. ND: none detected. ns: not statistically significant.
Repression of the flagellar regulon by TviA
Biosynthesis of the flagellum is controlled in a hierarchical fashion (reviewed in (Chilcott & Hughes, 2000, Macnab, 2004)). Expression of the early genes flhD and flhC is regulated by various signaling pathways and is an important control point in flagellar biogenesis. The flhDC encode the master regulator FlhDC, which activates transcription from class two promoters, facilitating expression of middle genes encoding the alternative sigma factor FliA, the anti-sigma factor FlgM, the regulator FliZ, as well as genes required for hook-basal body formation. Secretion of FlgM through the completed hook-basal body frees FliA to induce expression of late genes, such as fliC, encoding the major component of the filament (Hughes et al., 1993).
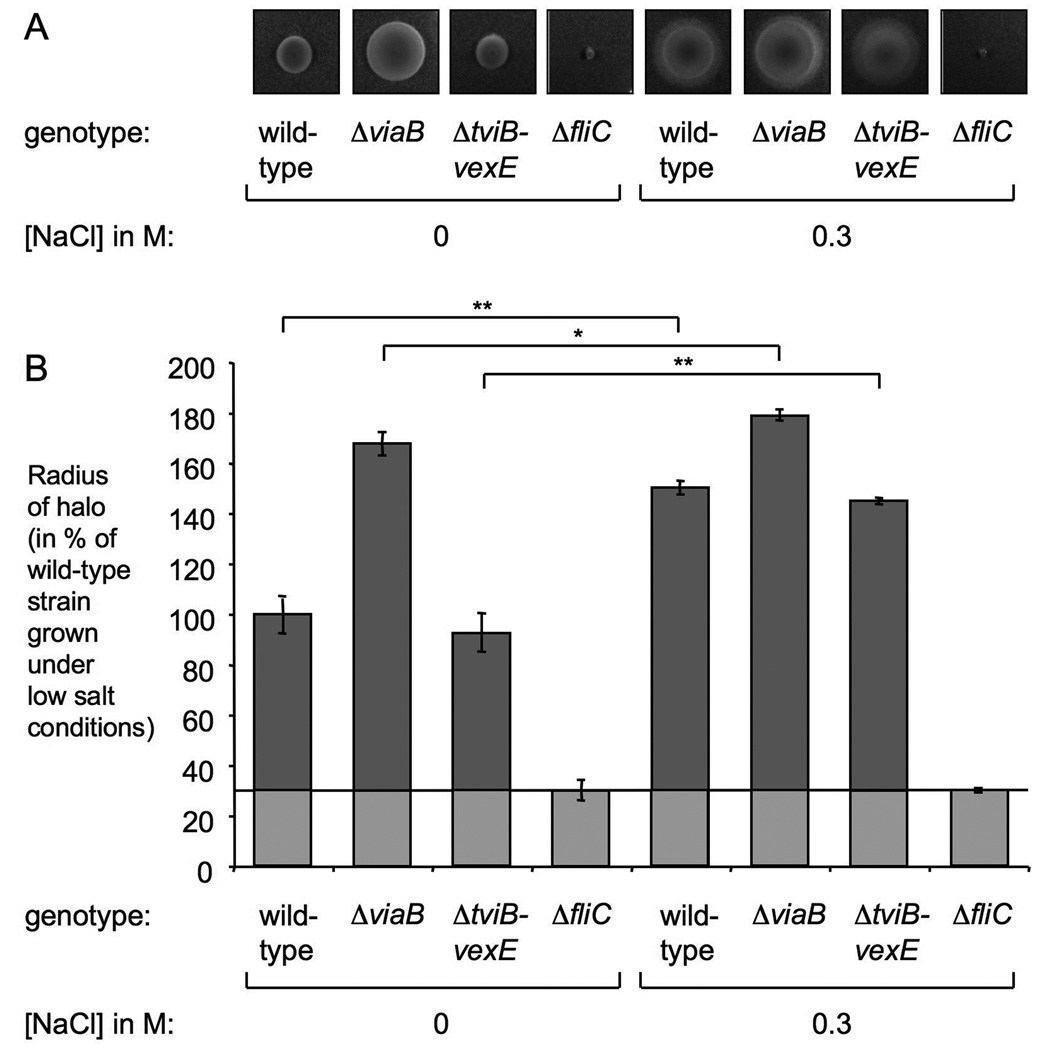
To confirm the findings of the gene expression profiling experiments, relative expression levels of selected target genes by the S. Typhi wild-type strain (Ty2), the ΔviaB mutant (STY2), the ΔtviB-vexE mutant (SW74), and the ΔrcsB mutant (SW513) were determined by real-time quantitative reverse transcription polymerase chain reaction (real-time qRT-PCR). Consistent with our previous findings (Winter et al., 2008), both RcsB and TviA repressed transcription of flhC low osmolarity conditions (Fig. 3A and Fig 4A). The mRNA levels of fliZ and fliC were significantly increased (P < 0.05) in the ΔviaB mutant when compared to the wild-type strain or the ΔtviB-vexE mutant (Fig. 4B and C). Likewise, expression of TviA reduced the amount of FliC protein in whole cell lysate as determined by Western Blot (Fig. 4D). Repression of the flagellar regulon by TviA under low osmolarity conditions resulted in decreased motility (Fig 5A and B).
Figure 3. RcsB represses flhC and hilA transcription in S. Typhi.
The S. Typhi wild-type strain (Ty2), ΔrcsB mutant (SW513), flhDC mutant (SW491), and flhC ΔrcsB mutant (SW612) were grown in low osmolarity medium (SOB broth) under aerobic conditions. RNA was extracted and transcription of flhC (A) and hilA (B) was determined by real time qRT-PCR. Bars represent the geometric mean of mRNA levels (in percent of wild-type strain levels) ± standard error. Statistical significance between data sets is indicated by asterisks: * (P < 0.05) or ** (P 0.01). ns: not statistically significant.
Figure 4. TviA represses early, middle and late genes of the flagellar regulon in S. Typhi.
(A–C) The S. Typhi wild-type strain (Ty2), ΔviaB mutant (STY2),ΔtviB-vexE mutant (SW74), fliZ mutant (SW531), fliZ ΔviaB mutant (SW532), fliZ ΔtviB-vexE mutant (SW533), flhDC mutant (SW491), flhDC ΔviaB mutant (SW492), and flhDC ΔtviB-vexE mutant (SW477) were grown in low osmolarity medium (SOB broth) under aerobic conditions. RNA was extracted and transcription of flhC (early gene) (A), fliZ (middle gene) (B), and fliC (late gene) (C) was determined by real time qRT-PCR. Bars represent the geometric mean of mRNA levels (in percent of wild-type strain levels) ± standard error of three independent experiments. Statistical significance between data sets is indicated by asterisks: * (P < 0.05) or ** (P < 0.01). ns: not statistically significant. (D) The S Typhi wild-type strain (Ty2), ΔviaB mutant (STY2),ΔtviB-vexE mutant (SW74), and the ΔviaB ΔfliC mutant (SW483) were grown in low osmolarity medium (tryptone yeast extract broth) and flagellin expression was determined by Western Blot using FliC-specific antiserum (H antiserum d, αHd). To ensure equal loading of samples, expression of GroEL was determined by Western Blot (αGroEL). Approximate positions of standard proteins are indicated.
Figure 5. TviA represses S. Typhi motility under conditions of low osmolarity.
(A) Motility plates containing 0.3 % agar were inoculated with the S. Typhi wild-type strain (Ty2), ΔviaB mutant (STY2), ΔtviB-vexE mutant (SW74), and the ΔfliC mutant (SW359). NaCl was added at a concentration of 300 mM to increase the osmolarity of the medium as indicated. (B) Measurements of the halo size produced by bacterial strains on motility plates. Bars represent the geometric means from three independent experiments. Statistical significance is indicated by asterisks: * (P < 0.05) or ** (P < 0.01).
TviA reduces expression of the T3SS-1 by repressing flhDC and fliZ transcription
Expression of the T3SS-1 apparatus and T3SS-1 effector proteins is under central control of the activator HilA (Bajaj et al., 1995). RcsB and TviA significantly (P < 0.05) repressed expression of hilA (Fig. 3B and Fig 6A). Transcription of hilE, a gene encoding a repressor of SPI-1 expression (Baxter et al., 2003, Boddicker et al., 2003), was not controlled by TviA (Fig. 6C). RcsB can bind the flhDC promoter downstream of the transcriptional start site, thereby repressing expression (Francez-Charlot et al., 2003), however no such interaction site was identified in the hilA promoter region, indicating that RcsB-mediated repression of hilA expression may be indirect. The flagellar regulator FliZ influences expression of the T3SS-1 by activating hilD, a gene encoding a positive regulator of hilA expression in S. Typhimurium (Kage et al., 2008, Ellermeier et al., 2005, Lucas et al., 2000). To test whether TviA represses invasion gene expression by limiting expression of flhDC and fliZ, the effect of TviA on relative mRNA levels of hilA and invG was determined each in flhDC and fliZ mutant backgrounds (Fig. 6A and B). Consistent with their function as positive regulators of invasion gene expression, lack of either the fliZ or flhDC genes reduced hilA and invG mRNA levels significantly (P < 0.05). While deletion of the tviA gene significantly (P < 0.05) increased hilA and invG transcription in the wild-type background, this derepression was not observed when either the fliZ or the flhDC genes were not functional. Consistent with the notion that FliZ activates T3SS-1 expression through HilD, transcription of hilD was induced in a tviA defective strain (Fig. 1A, cluster 1). Collectively, these data indicated that repression of invasion genes by TviA is mediated by the flhDC – fliZ – hilD – hilA – axis. Similarly, repression of hilA by RcsB was dependent on flhDC (Fig. 3B).
Figure 6. Repression of SPI-1 encoded invasion gene transcription by TviA is mediated through flhDC and fliZ in S. Typhi.
The S. Typhi wild-type strain (Ty2), ΔviaB mutant (STY2), ΔtviB-vexE mutant (SW74), fliZ mutant (SW531), fliZ ΔviaB mutant (SW532), fliZ ΔtviB-vexE mutant (SW533), flhDC mutant (SW491), flhDC ΔviaB mutant (SW492), and flhDC ΔtviB-vexE mutant (SW477) were grown in low osmolarity medium (SOB broth) under aerobic conditions. RNA was extracted and transcription of hilA (A), invG (B), and hilE (C) was determined by real time qRT-PCR. Bars represent the geometric mean of mRNA levels (in percent of wild-type strain levels) ± standard error. Asterisks indicate statistical significance between data sets: * (P < 0.05) or ** (P < 0.01). ns: not statistically significant.
TviA, but not the Vi capsular antigen, represses invasion of human intestinal epithelial cells
Expression of T3SS-1 as well as flagella mediated motility are major factors contributing to invasion of non-phagocytic cells, such as intestinal epithelial cells (Jones et al., 1981, Galan & Curtiss, 1989). Consistent with these previous observations, mutational inactivation of invA, a structural gene of the T3SS-1, led to a significant decrease in invasion (P < 0.01; 6-fold) when human colonic carcinoma (T84) cells were infected with S. Typhi (Fig. 7A). Lack of motility (ΔfliC mutant) reduced invasion even further (P < 0.01; 340-fold) when compared to the S. Typhi wild-type strain.
Figure 7. TviA regulates invasion of human epithelial cells by modulating both motility and SPI-1 gene expression in S. Typhi.
(A) Motility and T3SS-1 contribute to invasion of the T84 epithelial cell line by S. Typhi. S. Typhi wild-type strain (Ty2), invA mutant (SW222), ΔfliC mutant (SW359), and ΔfliC ΔinvA mutant (SW550) were grown under low osmolarity conditions (tryptone yeast extract broth) and monolayers of T84 cell were infected at a multiplicity of infection of 10:1 for 1 h. Recovered bacterial numbers were standardized to the number of the bacteria in the inoculum. (B) TviA modulates invasion of T84 cells by S. Typhi. S. Typhi wild-type strain (Ty2), ΔviaB mutant (STY2), ΔtviB-vexE mutant (SW74), invA mutant (SW222), ΔviaB invA mutant (SW224), ΔfliC mutant (SW359), and ΔfliC viaB mutant (SW483) were grown under low osmolarity conditions (tryptone yeast extract broth) and monolayers of T84 cell were infected as described above.
Bars represent the geometric mean of three independent experiments ± standard error. Statistical significance between data sets is indicated by asterisks: * (P < 0.05) or ** (P < 0.01). ns: not statistically significant. pWSK: pWSK29
Non-encapsulated strains of S. Typhi, e.g. strains that lack or display reduced expression of the viaB operon, have been shown to be hyperinvasive in vitro (Arricau et al., 1998, Zhao et al., 2001). Extracellular polysaccharides can interfere with the function of type III secretion systems (West et al., 2005) and it has been suggested that production of the Vi antigen can diminish invasiveness of S. Typhi (Bishop et al., 2008). Since TviA regulates motility (Fig. 5A and B) and the T3SS-1 (Fig. 6A and B), we wanted to determine whether the reduced invasiveness of S. Typhi was due to the production of the Vi capsular antigen or to a TviA-mediated repression of flagella and T3SS-1 genes. S. Typhi strains were grown in yeast extract medium (low salt) to allow for expression of the Vi antigen and invasiveness was determined by a gentamicin protection assay (Fig. 7B). Deletion of the Vi biosynthesis genes (ΔtviB-vexE mutant, SW74) did not significantly increase invasiveness (P > 0.05) when compared to the wild-type strain (Ty2). However, when the tviA was deleted in addition to the downstream Vi biosynthesis genes (ΔviaB mutant, SW347), invasion was increased significantly (P < 0.01; 5.3 fold) compared to the non-encapsulated ΔtviB-vexE (SW74) mutant indicating that TviA, and not the capsular Vi antigen, is predominantly responsible for the reduced invasion of S. Typhi.
To assess whether TviA-mediated repression of flagellar and/or T3SS-1 gene expression contributed to this phenotype, this experiment was repeated using a non-motile (ΔfliC) background strain or a T3SS-1 deficient (invA) strain background (Fig. 7B). In both the T3SS-1 expressing, non-motile ΔfliC strain background (SW359) and the motile, T3SS-1 deficient invA strain background (SW222), deletion of the viaB operon (ΔviaB ΔfliC, SW483; and ΔviaB invA, STY4, respectively) increased invasion compared to the respective isogenic parent strain (ΔfliC and invA mutant, respectively). Complementation of the ΔviaB mutation with a low copy plasmid expressing solely tviA from its native promoter (pTVIA1) (Winter et al., 2008) reduced invasiveness to levels comparable to the respective parent strain. These observations were consistent with the hypothesis that TviA repressed invasion by reducing expression of both flagella and the T3SS-1. We conclude that the viaB locus reduced invasion towards epithelial cells predominantly by a TviA-mediated repression of motility and invasion gene expression and not by expression of the Vi capsular polysaccharide.
TviA, but not RcsB, represses transcription of flhC in response to osmolarity
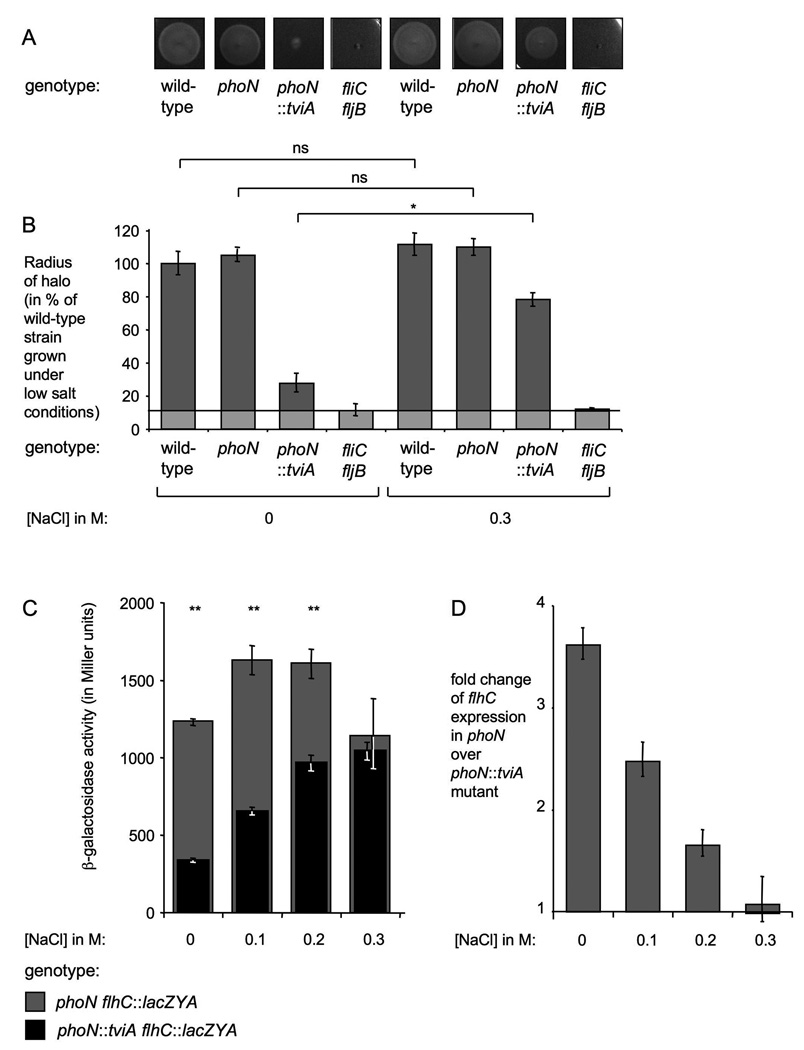
It has been previously suggested that the Rcs phosphorelay system regulates expression of flagellin and the T3SS-1 in response to osmolarity. Differences in gene expression between S. Typhimurium and S. Typhi were attributed to differences in signaling through the Rcs phosphorelay system (Arricau et al., 1998). However, the Rcs phosphorelay system, displays more than 99% sequence identity between the two serotypes (McClelland et al., 2001, Deng et al., 2003). We reasoned that the S. Typhi specific protein TviA may be a likely candidate for a regulator required for osmolarity dependent repression of flagellar and invasion gene expression. Since expression of the viaB (and therefore expression of tviA) is upregulated in response to low osmolarity (Pickard et al., 1994), TviA may only be available to serve as auxiliary protein for RcsB under conditions of low osmolarity. We therefore hypothesized that TviA, but not RcsB, mediates osmoregulation. To test this hypothesis, we measured flhC transcription in a ΔviaB and a ΔrcsB ΔviaB mutant background using a chromosomal flhC::lacZYA transcriptional reporter fusion. S. Typhi strains were grown in yeast extract broth and NaCl was added to adjust the osmolarity of the medium. An increase in osmolarity resulted in increased flhC transcription in the wild-type strain background (Fig. 8A, black bars). Deletion of the viaB operon resulted in increased overall flhC transcription, however derepression was more profound under low osmolarity conditions than under high salt conditions (Fig. 8A, dark grey bars). To further illustrate this point, we calculated the fold repression exerted by TviA at different salt concentrations by dividing the β-galactosidase activity of the ΔviaB flhC::lacZYA by the activity measured for the flhC::lacZYA mutant (Fig. 8C). The results illustrated that TviA repressed flhC transcription in response to decreased osmolarity.
Figure 8. Effect of TviA and RcsB on osmolarity dependent flhC transcription in S. Typhi.
(A) The flhC::lacZYA mutant (SW197, black bars), the ΔviaB flhC::lacZYA mutant (SW186, grey bars), and the ΔviaB ΔrcsB flhC::lacZYA mutant (SW452, white bars) were grown in tryptone yeast extract medium supplemented with the indicated concentrations of NaCl and β-galactosidase activity was measured. Fold repression mediated by RcsB (B) and TviA (in the presence of RcsB) (C) was calculated by dividing the β-galactosidase activity of the ΔviaB flhC::lacZYA mutant and the ΔviaB ΔrcsB flhC::lacZYA mutant by the corresponding value of the flhC::lacZYA and the ΔviaB flhC::lacZYA, respectively.
Bars represent the geometric mean of four independent experiments ± standard error.
To assess the contribution of RcsB to osmolarity dependent repression of flhC transcription in the absence of TviA, β-galactosidase activity of a ΔviaB flhC::lacZYA mutant was compared to a ΔviaB ΔrcsB flhC::lacZYA mutant (Fig. 8A and B). In the absence of TviA, RcsB repressed flhC expression by approximately 2-fold, however this repression was osmolarity-independent (Fig. 8B). We conclude that the horizontally acquired tviA gene allowed modulation of the flagellar regulon in response to osmolarity. RcsB is necessary for this trait since it is an important activator of tviA transcription, however RcsB alone was not sufficient to mediate osmoregulation.
TviA represses invasion of epithelial cells under low osmolarity conditions
S. Typhi grown under high salt conditions is more invasive towards epithelial cells (Tartera & Metcalf, 1993, Mills & Finlay, 1994). To test whether TviA contributes to osmoregulation of invasion, the S. Typhi wild-type strain (Ty2) and the ΔviaB mutant (SW347) were cultured in media with varying osmolarity and invasion of T84 cells was determined as described above. The wild-type strain was highly invasive at high osmolarity, but invasiveness was significantly decreased (P < 0.05) when grown in low salt broth (Fig. 9A). At high osmolarity, the ΔviaB mutant and the wild-type strain displayed comparable invasiveness (2.4 % vs 2.6 %, P = 0.85 ), however under low salt conditions, the ΔviaB mutant was recovered at significantly higher numbers than the wild-type strain (0.8 % vs 0.09 %, P < 0.05). To illustrate the repression mediated by TviA, invasiveness of the ΔviaB mutant was directly compared to that of the wild-type strain by dividing the percent of the ΔviaB mutant (SW347) recovered from epithelial cells by the corresponding number of the wild-type strain (Ty2) (Fig. 9B). When pre-grown in low osmolarity medium, TviA repressed invasion by a factor of 8.6. This repression was almost completely relieved under high salt conditions (1.1 fold), indicating that TviA regulates invasion of S. Typhi in response to osmolarity.
Figure 9. TviA-mediated osmoregulation of human epithelial cell invasion by S. Typhi.
Invasiveness of S. Typhi towards T84 cells was determined by a gentamicin protection assay as described in Fig. 7A. (A) The S. Typhi wild-type (Ty2, grey bars) and the ΔviaB mutant (SW347, white bars) were grown in tryptone yeast extract broth prior to infection of T84 cells. To adjust the osmolarity of the growth medium, NaCl was added at the indicated concentrations. (B) Fold repression of invasion was calculated by dividing the number of recovered bacteria of the ΔviaB mutant (SW347) by the number of the wild-type strain (Ty2).
Bars represent the geometric mean of three independent experiments ± standard error. Statistical significance is indicated by asterisks: ** (P < 0.01). ns: not significant.
Expression of tviA in S. Typhimurium
Our data suggest, that TviA enables the Rcs phosphorelay system to process osmolarity as an additional signal. The tviA encoding SPI-7 region was likely acquired in the S. Typhi lineage through horizontal gene transfer (Parkhill et al., 2001) and was integrated into the chromosome near the phoN gene. Therefore, we wanted to test whether transfer of the tviA gene into a non-typhoidal serotype, such as S. Typhimurium, would result in a S. Typhi-like osmoregulation of virulence gene expression. The phoN gene in S. Typhimurium was replaced with a copy of the S. Typhi tviA gene under control of its own promoter. Osmoregulation of motility and flhC transcription of the resulting strain (SW474) were compared to a phoN mutant (SW284) and the S. Typhimurium wild-type strain (IR715) (Fig. 10). A mutation in phoN (SW284) did not alter motility of S. Typhimurium whereas expression of tviA (phoN::tviA mutant, SW474) reduced motility significantly (P < 0.05) (Fig. 10A and B). Similar to S. Typhi, this repression was more profound at low osmolarity and was almost negated at elevated salt concentrations. Expression of flhC in S. Typhimurium strains was investigated by introducing a transcriptional β-galactosidase fusion (flhC::lacZYA) into their chromosome. Consistent with observations made in S. Typhi, flhC transcription was repressed in response to a decrease in osmolarity by introduction of the tviA gene into S. Typhimurium (Fig. 10C and D). In medium with low osmolarity, the magnitude of flhC repression in the tviA expressing S. Typhimurium strain (Fig. 10C) was comparable to that in the S. Typhi wild-type strain (Fig. 8A), indicating that the TviA protein was fully functional in S. Typhimurium.
Figure 10. Effect of tviA on osmoregulation of motility and the flagellar master regulator flhC in S. Typhimurium.
(A) Motility of the S. Typhimurium wild-type strain (IR715), phoN mutant (SW284),ΔphoN::tviA mutant (SW474), and the ΔfliC fljB mutant (SW473) was determined using 0.3 % agar plates. NaCl was added at a concentration of 300 mM as indicated to increase the osmolarity of the medium. (B) Measurements of the halo size produced by bacterial strains on motility plates. (C) The phoN flhC::lacZYA (SW317, black bars) and the phoN::tviA flhC::lacZYA (SW316, grey bars) were grown in tryptone yeast extract medium supplemented with the indicated concentrations of NaCl and β-galactosidase activity was measured. (D) Fold repression mediated by TviA was calculated as described in figure 8.
Bars represent the geometric mean of three independent experiments ± standard error. Statistical significance is indicated by asterisks: * (P < 0.05), ** (P < 0.01), ns: not significant.
The histidine kinase EnvZ is involved in TviA mediated osmoregulation
The osmosensing EnvZ/OmpR two component system activates the transcription of the viaB-operon (Pickard et al., 1994). One possible mechanism by which TviA allows osmoregulation of target genes is that RcsB responds to the availability of TviA, which is controlled on a transcriptional basis by the EnvZ/OmpR system. To test this hypothesis, a transcriptional β-galactosidase fusion to tviA was constructed in S. Typhi. Since TviA regulates its own transcription (Hashimoto et al., 1996), the transcriptional fusion was constructed such that the tviA coding sequence was not disrupted. As expected, a decrease in osmolarity resulted in significantly increased transcription of tviA in S. Typhi (Fig. 11A), thereby reducing the availability of TviA. Consistent with the hypothesis, that the EnvZ/OmpR system controls expression of tviA in response to osmolarity, disruption of the envZ gene in S. Typhi increased motility to levels comparable to the ΔviaB mutant (Fig. 11B and 11C). Likewise, introducing a envZ mutation into the tviA expressing S. Typhimurium strain abrogated the TviA-mediated repression of flagellin expression and motility in response to osmolarity (Fig. 12). These data are consistent with the idea that the EnvZ/OmpR system controls the availability of TviA in response to osmolarity.
Figure 11. EnvZ mediates osmorelulation of flagella through the availability of tviA in S. Typhi.
(A) Osmolarity dependent transcription of tviA in S. Typhi. The tviA::lacZYA mutant (SW620) was cultured in tryptone yeast extract medium supplemented with the indicated concentrations of NaCl and β-galactosidase activity was measured. Bars represent the geometric mean of three independent experiments ± standard error. Statistical significance is indicated by asterisks: * (P < 0.05), ** (P < 0.01), ns: not significant. (B and C) Effect of envZ on motility in S. Typhi. (B) Motility plates containing 0.3 % agar were inoculated with the S. Typhi wild-type strain (Ty2), ΔviaB mutant (STY2), envZ mutant (SW672), and the ΔfliC mutant (SW359). (C) Measurements of the halo size produced by bacterial strains on motility plates. Bars represent the geometric mean of three independent experiments ± standard error. Statistical significance is indicated by asterisks: * (P < 0.05), ns: not significant.
Figure 12. Introduction of tviA into S. Typhimurium renders flagella sensitive to EnvZ-mediated osmorelulation.
(A) Regulation of flagellin expression by TviA and EnvZ. The S. Typhimurium wild-type (IR715), the phoN::tviA mutant (SW474), the phoN::tviA envZ mutant (SW679), and the ΔfliC fljB mutant (SPN313) were grown in tryptone yeast extract broth supplement with the indicated concentration of NaCl and FliC as well as GroEL expression was determined by Western Blot using Salmonella H antiserum i (αHi) and anti-GroEL antiserum (αGroEL), respectively. Approximate positions of standard proteins are indicated. (B) Motility of the phoN mutant (SW284), the phoN::tviA mutant (SW474), the phoN::tviA envZ mutant (SW679), and the ΔfliC fljB mutant (SPN313). (C) Measurements of the halo size produced by bacterial strains on motility plates. Bars represent the geometric mean of three independent experiments ± standard error.
Collectively these data support the hypothesis that the function of the auxiliary TviA protein is to integrate osmolarity as an additional signal processed by the RcsB regulatory network. Our data support a model in which the EnvZ/OmpR system controls the availability of the auxiliary TviA protein in response to osmolarity (Fig. 13). In turn, gene regulation by the RcsB response regulator is sensitive to the availability of the auxiliary TviA protein. This new regulatory input results in altered expression of important virulence factors, including flagella, the Vi antigen and T3SS-1, in response to osmolarity.
Figure 13. Proposed model for a TviA-dependent integration of osmoregulation into the Rcs regulon of S. Typhi.
The Rcs phosphorelay system and the EnvZ/OmpR two-component system respond to envelope stress and osmolarity of the medium, respectively, by activating transcription of tviA. TviA acts as an auxiliary protein to the response regulator RcsB, thereby allowing integration of signals from both signaling pathways. RcsB and TviA repress transcription of flhDC and fliZ, thereby limiting expression of flagella and invasion genes under low osmolarity conditions. Please refer to text for details.
DISCUSSION
Homologues of the Rcs phosphorelay system are present in E. coli, Shigella spp., S. enterica, Yersinia spp., Klebsiella pneumoniae, Photorhabdus spp. and Erwinia spp. (Erickson & Detweiler, 2006), where they frequently regulate expression of exopolysaccharide biosynthesis (Majdalani & Gottesman, 2005). The RcsB response regulator can form functional homodimers, however, its activity can be enhanced by the formation of heterodimers with auxiliary proteins (Pristovsek et al., 2003). A well-characterized auxiliary protein in E. coli and S. Typhimurium is RcsA, which increases RcsB-dependent expression of cps genes involved in the biosynthesis of the exopolysaccharide colanic acid (Costa et al., 2003, Mouslim et al., 2003, Wehland & Bernhard, 2000). In S. Typhi, RcsB activates the biosynthesis of the Vi capsular polysaccharide in conjunction with the TviA auxiliary protein independent of RcsA (Virlogeux et al., 1996), indicating that TviA may have functionally replaced the auxiliary protein RcsA.
In S. Typhimurium, RcsB represses expression of the T3SS-1 (Lin et al., 2008) and flagella (Wang et al., 2007). Constitutive activation of the Rcs phosphorelay systems attenuates virulence in vivo (Mouslim et al., 2004). Interestingly, the Rcs phosphorelay system has been implicated in differences between S. Typhi and S. Typhimurium with regard to osmoregulation of flagellin and invasion gene expression (Arricau et al., 1998). The high conservation (99% sequence identity) of the Rcs phosphorelay systems of S. Typhi and S. (McClelland et al., 2001, Deng et al., 2003) prompted us to investigate the role of the TviA auxiliary protein in incorporating new regulatory inputs into RcsB gene regulation. Gene expression profiling indicated that both TviA and RcsB were required for repression of flagellin and invasion gene expression in S. Typhi. Our data further suggested that RcsB in conjunction with the TviA auxiliary protein represses motility and invasion in S. Typhi by repressing the flhDC genes. Consistent with this idea, microarray analysis of FlhDC regulated genes in S. Typhimurium (Frye et al., 2006) generate a profile very similar to that determined here for TviA and RcsB regulated genes. Interestingly, in S. Typhimurium, RcsB regulates flhDC transcription independent of the auxiliary protein RcsA (Cano et al., 2002). Invasion gene expression is linked to flagella biosynthesis through FliZ, a regulator within the flagellar regulatory cascade (Lucas et al., 2000). FliZ regulates invasion gene expression by controlling the availability of hilD, encoding an activator of hilA (Kage et al., 2008, Lin et al., 2008). Based on our observations, we propose that TviA represses invasion gene expression by repressing flhDC transcription, which in turn results in reduced fliZ expression (Fig. 13).
Growth in high osmolarity increases the invasiveness of S. Typhi for intestinal epithelial cells by increasing expression of flagella biosynthesis genes and T3SS-1 genes (Tartera & Metcalf, 1993, Zhao et al., 2001, Arricau et al., 1998), two factors important for cell entry (Jones et al., 1981, Galan & Curtiss, 1989). Here we show that expression of tviA is regulated by the EnvZ/OmpR system in response to osmolarity in S. Typhi. These data suggest that the EnvZ/OmpR system regulates motility and invasion in S. Typhi by regulating TviA, a repressor of flagellin and T3SS-1 gene expression. In contrast, the EnvZ/OmpR system does not contribute to regulation of T3SS-1 gene expression in S. Typhimurium (Galan & Curtiss, 1990). Osmoregulation of flhDC expression in S. Typhi was mediated through RcsB in conjunction with TviA, but not through RcsB alone. These data suggest that the RcsB protein of S. Typhi senses osmolarity through the availability of the TviA auxiliary protein, which in turn is controlled by the osmosensitive EnvZ/OmpR system (Fig. 13). The absence of the TviA auxiliary protein in S. renders flagella and invasion genes expression independent of EnvZ/OmpR. Experimental transfer of the tviA gene into this non-typhoidal Salmonella serotype placed the flhDC genes under control of the EnvZ/OmpR system. This example illustrates that horizontal gene transfer of a regulatory gene (tviA) in S. Typhi profoundly altered signal processing of a widely conserved regulatory unit (the RcsB regulon). Our data are consistent with the idea that acquisition of new genetic material (viaB locus), and altered regulation of pre-existing genes (flagella and invasion genes) were both important contributors to the evolution of host adaptation in the S. Typhi lineage.
Incorporation of osmolarity as a signal controlling expression of motility, invasion, and the Vi capsule may facilitate a rapid adjustment of virulence gene expression during the transition of S. Typhi from the environment of the intestinal lumen to its location in tissue. The hyperosmotic conditions of the intestinal lumen are predicted to result in repression of tviA and capsule biosynthesis genes, while flagella and T3SS-1 are expressed optimally to mediate epithelial invasion. The physiological salt concentration encountered in tissue is predicted to induce tviA expression, thereby activating Vi biosynthesis and repressing expression of T3SS-1 and flagellin. TviA-mediated repression of flagellin secretion reduces the host’s ability to detect S. Typhi through Toll-like receptor (TLR) 5 (Winter et al., 2008). Furthermore, expression of the Vi capsular antigen interferes with the recognition S. Typhi lipopolysaccharide by TLR4 (Wilson et al., 2008). Studies on intestinal inflammation in animal models suggest that these myeloid differentiation primary response protein 88 (MyD88)-dependent mechanisms contribute to a viaB-mediated reduction in neutrophil recruitment (Raffatellu et al., 2007, Haneda et al., 2009). Expression of the viaB locus in the gut mucosa may thus be responsible for the scarcity of neutrophils in intestinal infiltrates of typhoid fever patients. The picture emerging from these studies is that horizontal gene transfer and an altered regulation of existing genes acted in concert to alter the clinical presentation of infections with S. Typhi compared to infections with non-typhoidal Salmonella serotypes.
EXPERIMENTAL PROCEDURES
Bacterial strains, plasmids and culture conditions
Bacterial strains and plasmids used in this study are listed in supplementary table 2. Unless otherwise noted, strains were routinely grown at 37°C in LB broth (10 g/l tryptone, 5 g/l yeast extract, 10 g/l NaCl) or on LB agar plates. For optimal expression of TviA, strains were grown for 16 h in LB, diluted in either SOB broth (20 g/liter tryptone, 5 g/liter yeast extract, 10 mM NaCl, 2.5 mM KCl, 10 mM MgCl2) (Raffatellu et al., 2005) or tryptone yeast extract broth (10 g/l tryptone, 5 g/l yeast extract) and grown to mid-log phase at 37°C. When appropriate, antibiotics were added at the following concentrations: chloramphenicol (Cm) 0.03 mg/ml, carbenicillin 0.1 mg/ml, kanamycin (Kan) 0.05 mg/ml, and tetracycline (Tet) 0.01 mg/ml.
Construction of plasmids
Standard cloning techniques were performed as described previously (Sambrook et al., 1989). Primers used to amplify DNA fragments are listed in supplementary table 3. PCR products were cloned into pCR2.1 using the TOPO TA cloning kit (Invitrogen).
To achieve expression of rcsB from the native promoter upstream of rcsD (yojN), the rcsD and rcsB genes including the rcsD promoter region were PCR amplified from Ty2 and cloned into the low copy plasmid pWSK29 (Wang & Kushner, 1991) using BamHI and XhoI restriction sites. The resulting plasmid was digested with MluI and SmaI, treated with Klenow fragment of Polymerase I and religated to remove a majority of the rcsD coding sequence. The resulting plasmid was named pRCSB1.
To construct pSW77, an internal fragment of the rcsB coding sequence was amplified by PCR from Ty2 and cloned into pFUSE (Bäumler et al., 1996) using SmaI and XbaI. Integration of pSW77 into the chromosome by allelic exchange creates a lacZYA fusion to rcsB, concomitantly disrupting the rcsB gene.
The tviA promoter region and coding sequence was PCR amplified from the Ty2 chromosome and cloned in pSW85 using SmaI and SalI restriction sites, giving rise to pSW86. To obtain the plasmids pSW87 and pSW93, the Cmr cassette from pCMXX was cloned into pSW86 and pSW85, respectively, using SmaI restriction sites.
To construct pSW97, DNA regions upstream and downstream of rcsB were PCR amplified from Ty2. An equimolar mixture of the gel purified products served as the template for a PCR using the two non-cross annealing primers. The resulting DNA fragment comprising the linked rcsB flanking regions was cloned into pGP704 using EcoRI and BglII. The resulting plasmid was digested with EcoRI, treated with Klenow fragment of Polymerase I and ligated with a kanamycin resistance gene block obtained from EcoRV digested pUC4 KSAC (GE Healthcare). The resulting plasmid was named pSW97.
A plasmid containing a DNA fragment comprising an upstream and a downstream region of the viaB operon has been described previously (Raffatellu et al., 2005). This plasmid was digested with SphI and BglII, and treated with Klenow fragment of Polymerase I to obtain the linked upstream and downstream flanking regions of the viaB operon. This fragment was ligated into EcoRI digested and Klenow fragment of Polymerase I treated pRDH10 to create pSW129.
The tviA gene was amplified from Ty2 and cloned into the plasmid pFUSE using the restriction enzymes XbaI and SmaI to create pSW160.
Construction of mutants by allelic exchange
Suicide plasmids described in supplementary table 2 were propagated in DH5α λpir and introduced into Salmonella by conjugation using S17-1 λpir as donor strain. S. Typhimurium exconjugants were recovered using LB plates containing nalidixic acid and appropriate additional antibiotics. Simmons-Citrate agar (Difco) supplemented with 40 mg/l tryptophan, 40 mg/l cysteine and appropriate antibiotics was used to select for S. Typhi exconjugants. Integration of the plasmids in the chromosome at the appropriate location was verified by PCR.
The deletion of the fliC gene in S. Typhimurium has been described previously (Winter et al., 2009). pSPN29 was inserted in this location by allelic exchange to simplify the selection after P22 mediated transduction. The rcsB gene in IR715 and SW347 was deleted by allelic exchange using the suicide plasmid pSW97. One kanamycin resistant but carbenicillin sensitive exconjugant was termed SW429 and SW299, respectively. Transcriptional lacZYA fusions to rcsB in the Ty2 and STY2 backgrounds were generated by integration of the plasmid pSW77 into the chromosome. The resulting strains were named SW237 and SW239, respectively. SW620 was created by introducing the plasmid pSW160 into the Ty2 chromosome by homologous recombination. Conjugational transfer of pSW63 into SW299 and SW513 gave rise to SW452 and SW605, respectively. SW347 was created by introducing the plasmid pSW129 into STY2, initially selecting for single crossover events. Sucrose selection was performed to achieve deletion of the kanamycin resistance cassette, resulting in an unmarked deletion of the viaB operon. Strains SW284 and SW474 were constructed by transferring the plasmids pSW93 and pSW87 into IR715, selecting for Cm resistance. Integration into the chromosome was verified in carbenicillin sensitive exconjugants by PCR.
Generalized Transduction
Phage P22 HT int-105 was used for generalized transduction as described previously (Miller, 1972). A P22 lysate of TH3730 was used to transduce the P(flhDC)5451::Tn10dTc(del-25) mutation into Ty2, SW347, SW74, and SW513 thus creating SW491, SW492, SW477, and SW612 respectively. Strains SW531, SW532, and SW533 were created by transducing the fliZ5691::MudJ mutation from TH0049 into Ty2, SW347, and SW74, respectively. The ΔfliC(−25 to +1494)::pSPN29 mutation was separately introduced into Ty2 and SW347 by P22 transduction using a lysate of SPN305 selecting for Cm resistance. Sucrose selection was performed as described previously (Lawes & Maloy, 1995) to delete the fliC gene by allelic exchange, yielding SW359 and SW483, respectively. A P22 lysate of SPN452 was used to transduce ΔinvA::tetRA into SW359, generating SW550. To construct SW513, a P22 phage lysate of SW429 was used to transduce the ΔrcsB::Kanr mutation into Ty2. SW317 and SW316 were constructed by transducing the flhC5456::MudJ mutation of the strain TH4054 into SW284 and SW474 respectively. A lysate of CH1118 was used to transduce the envZ1005::MudJ into SW474 and Ty2 to create SW679 and SW672, respectively.
Motility assays
For motility assays, plates containing 10 g/l tryptone and 0.3 % agar were inoculated with a single colony from a LB agar plate and incubated at 37°C for 24 h (S. Typhi) or 7 h (S. Typhimurium). Osmolarity was increased by adding NaCl at a final concentration of 300 mM. The area of the halo was determined (Labworks 4.6 software, UVP) and the diameter was calculated under the assumption that the curvature of the halo conforms to that of a circle. The experiments were performed in triplicate.
SDS-PAGE and Western Blot
S. Typhi and S. Typhimurium strains were grown for 4 h at 37°C in tryptone yeast extract broth. NaCl was added at the indicated concentrations. Based on culture turbidity (OD600), approximately 5 × 107 CFU were lysed in loading buffer. The lysate was resolved by SDS-PAGE (Ausubel et al., 1994) and transferred to polyvinylidene fluoride (PVDF) membrane (Millipore) using a semi-dry transfer system (Bio-Rad laboratories). Rabbit Salmonella H antiserum d (Difco), Salmonella H antiserum i (Difco), anti-GroEL antiserum (Sigma), and a horse radish peroxidase-conjugated goat anti-rabbit secondary antibody (Bio-Rad laboratories) were used to detect FliC and GroEL expression.
RNA extraction
S. Typhi strains were grown in 5 ml broth. 0.8 ml stop solution (5 % water saturated phenol in ethanol) was added and the suspension centrifuged at 7,700 g for 5 min at 4°C. The bacterial pellet was resuspended in 0.4 ml lysis solution (0.1 mg/ml lysozyme, 1mM EDTA 10mM, Tris/Cl pH 8.0) and the suspension incubated at 25°C for 1 h. Next, 40 µl 10 % SDS was added and the solution heated to 65°C. After 2 min, 0.44 ml 1 M sodium acetate and 0.9 ml hot (65°C) phenol was added to the sample. The emulsion was incubated at 65°C for 6 min, chilled on ice for 10 min and centrifuged at 20,000 g for 10 min at 4°C. The aqueous phase was transferred to a new tube and extracted with 0.9 ml chloroform. Phases were separated by centrifugation at 20,000 g for 5 min at 4°C. After transferring the upper aqueous phase to a new tube, the RNA was precipitated by adding 80 µl 1 mM EDTA 3 M sodium acetate pH 5.2 and 1 ml isopropanol. Samples were centrifuged for 30 min at 20,000 g at 4°C and the supernatant discarded. The RNA pellet was washed with 1 ml 80 % Ethanol and the samples were centrifuged for 5 min at 20,000 g at 4°C. RNA was air-dried and resuspended in RNase-free water. Traces of DNA in the RNA preparation were removed by DNase treatment (DNA-free DNase treatment, Applied Biosystems).
Gene expression profiling
For gene expression profiling experiments, overnight cultures of S. Typhi strains were diluted 1:50 in 5 ml SOB broth and grown statically until the turbidity reached an optical density of OD600 = 0.4 – 0.5. RNA was extracted from two cultures of the S. Typhi wild-type strain (Ty2), the rcsB mutant (SW237), the ΔviaB mutant (STY2), and the ΔtviB-vexE mutant (SW74), respectively. Biological replicates were separately used for cDNA synthesis and labeling.
Microarray hybridization and scanning steps were performed by the UC Davis ArrayCore Microarry facility as described previously (Seo et al., 2008) with the following modifications: cDNA was synthesized and differentially labeled with Alexa Flour 555 and 647, respectively, using the SuperScript Plus Indirect cDNA Labeling System (Invitrogen) and probes were hybridized to the array at 50°C for 16 h.
For raw data processing and analysis, the TM4 Microarray Software Suite (Saeed et al., 2003) was used. Raw expression data were normalized using a global Locfit/LOWESS algorithm (Quackenbush, 2002, Cleveland, 1979, Yang et al., 2002). Biological replicates were combined by averaging the fold change of each gene. Cluster analysis of gene expression data was perfomed using the Clustering Affinity Search Technique (CAST) algorithm (Ben-Dor et al., 1999, Promponas et al., 2000) with a threshold parameter of 0.85. Genes identified by cluster analysis are listed in supplementary table 1. Microarray data have been deposited at the Gene Expression Ominbus database under the accession number GSE15752.
RT-PCR and real time PCR
For real time quantitative reverse transcription polymerase chain reaction (real time qRT-PCR) experiments, S. Typhi strains were grown overnight in LB, diluted in 5 ml tryptone yeast extract broth to an OD600 of 0.4 – 0.6 and RNA was extracted. RT-PCR and real time PCR were performed as described previously (Winter et al., 2008). RNA (1 µg) was reverse transcribed in a 50 µl volume according to the recommendations of the manufacturer (TaqMan reverse transcription reagents; Applied Biosystems). 4 µl of cDNA was used as template for each real time PCR in a 25 µl total reaction volume. Real time PCR was performed using the primers listed in Table 1 as well as SYBR Green (Applied Biosystems) and the GeneAmp 7900 Sequence Detection System. Data was analyzed using the comparative Ct method (Applied Biosystems). Target gene transcription of each sample was normalized to the respective levels of RpoD (σ70) mRNA, encoded by the rpoD gene. Comparable results in gene expression changes were observed when target gene mRNA levels were standardized using gmk mRNA levels (data not shown). DNA contamination was assessed by performing real time PCR on RNA samples that were treated in a mock-RT-PCR (no addition of reverse transcriptase).
Table 1.
Primers used for real-time qRT-PCR in this study
| Target gene | Sequence | Reference |
|---|---|---|
| rpoD | 5’-ACATGGGTATTCAGGTAATGGAAGA-3’ | (Bohez et al., 2006) |
| 5’-CGGTGCTGGTGGTATTTTCA-3’ | ||
| gmk | 5’-TTGGCAGGGAGGCGTTT-3’ | (Bohez et al., 2006) |
| 5’-GCGCGAAGTGCCGTAGTAAT-3’ | ||
| flhC | 5’-CTGTAACTGCTGCGGTGGGAAC-3’ | This study |
| 5’-GGATGGCGGCTGGCATAAACTAC-3’ | ||
| fliZ | 5’-TGCTACTTGACGACGCTGCCTG-3’ | This study |
| 5’-ACGCCACCCACTCCTTCTGCTC-3’ | ||
| fliC | 5’-GTAACGCTAACGACGGTATC-3’ | This study |
| 5’-ATTTCAGCCTGGATGGAGTC-3’ | ||
| hilA | 5’-ATTAAGGCGACAGAGCTGGA-3’ | This study |
| 5’-GAATAGCAAACTCCCGACGA-3’ | ||
| hilE | 5’-TAACGGGAAAACGGTGAAAG-3’ | This study |
| 5’-GTGAAATTCGCACATTGGTG-3’ | ||
| invG | 5’-CGAGTGCTGCCCCGTTTTTC-3’ | This study |
| 5’-TCGTTGCCATCTTCAATGTCCAG-3’ | ||
| rcsB | 5’-AAATCGCCAAGAAGCTCAAC-3’ | This study |
| 5’-CTCTACGCCCAGTTTCATCA-3’ |
Gentamicin protection assay
Invasion assays were performed as described previously (Winter et al., 2008). The colorectal carcinoma cell line T84 (ATCC CCL-248) was obtained from the American Type Culture Collection. Cells were routinely grown in Dulbecco’s modified Eagle medium (DMEM)-F12 medium (Gibco), containing 1.2 g/l sodium bicarbonate, 2.5 mM L-glutamine, 15 mM HEPES, 0.5 mM sodium pyruvate (Gibco), and 10% fetal calf serum (FCS). For assays, cells were seeded in a 24 well plate at a density of approximately 1×105 cells per well, incubated for 24 h with complete growth media and 24 h with serum-free media.
S. Typhi strains were grown for 16 h at 37°C with aeration, diluted 1:25 or 1:50 in yeast extract broth and incubated for 3 h at 37°C with aeration until the turbidity (OD600) reached a value of 0.6–0.8. T84 epithelial cells were infected for 1 h at a MOI of 10. Cells were washed five times with Dulbecco’s phosphate-buffered saline (DPBS) (Gibco) and serum-free DMEM/F12 medium containing 0.1 mg/ml gentamicin (Gibco) was added. After 90 min, cells were washed three times with DPBS and intracellular bacteria were quantified by spreading serial 10-fold dilutions of T84 cell lysates (1% Triton-X-100) on LB agar plates to determine the number of colony forming units (cfu). Experiments were performed three or four times.
β-Galactosidase assay
β-Galactosidase activity was measured as described previously (Miller, 1972). Briefly, strains were grown overnight in LB broth, diluted 1:50 in tryptone yeast extract broth and incubated for 4 h at 37°C. If necessary, NaCl was added to the media to increase the osmolarity of the medium as indicated for each dataset in the figure legend. The bacterial suspension was diluted in Z-buffer (100 mM Na2HPO4, 10 mM KCl, 1 mM MgSO4, 50 mM β-mercaptoethanol pH 7.0) to a final volume of 1 ml and cells were permeated by adding 0.02 ml of 0.1 % SDS and 0.04 ml chloroform. Enzymatic activity was assayed at 30°C by adding 0.2 ml of 4 mg/ml 2-Nitrophenyl β-D-galactopyranoside (Sigma), The reaction was terminated by addition of 0.5 ml 1 M Na2CO3. To determine substrate turnover, the absorption at a wavelength of 420 nm and 550 nm was measured and activity was calculated as Miller units. Each experiment was repeated at least in triplicate.
Statistical analysis
For statistical analysis of ratios (i.e. increases in gene expression, or invasion data expressed as percentage of inoculum), values were transformed logarithmically to calculate geometric means and for further statistical analysis. A parametric test (Student's t-test) was used to calculate whether differences in fold changes of gene expression or invasion between treatment groups were statistically significant (P < 0.05). For data from tissue culture experiments and gene expression analysis, paired statistical analysis was used.
Supplementary Material
ACKNOWLEDGMENTS
We would like to thank Janice Pfeiff (UC Davis ArrayCore Microarry facility) for expert technical assistance. We are also grateful to Glenn Young, Briana Young, and Renée M. Tsolis for helpful discussion. We would also like to thank Kelly Hughes for providing bacterial strains. Salmonella DNA microarrays were obtained through NIAID’s Pathogen Functional Genomics Resource Center, managed and funded by Division of Microbiology and Infectious Disease, NIAID, NIH, DHHS and operated by the J. Craig Venter Institute. We are grateful to the Salmonella genetic stock centre (SGSC), University of Calgary, Canada for providing bacterial strains.
P.T. was supported by Faculty of Medicine, Chiang Mai University, Thailand. S.-P.N. was supported by the Floyd and Mary Schwall Fellowship, University of California, Davis, CA, USA. Work in A.J.B.’s laboratory is supported by Public Health Service Grants AI040124, AI044170, AI076246 and AI079173.
LITERATURE
- Arricau N, Hermant D, Waxin H, Ecobichon C, Duffey PS, Popoff MY. The RcsB-RcsC regulatory system of Salmonella typhi differentially modulates the expression of invasion proteins, flagellin and Vi antigen in response to osmolarity. Molecular microbiology. 1998;29:835–850. doi: 10.1046/j.1365-2958.1998.00976.x. [DOI] [PubMed] [Google Scholar]
- Ausubel FM, Brent R, Kingston RE, Moore DD, Seidman JG, Smith JA, Struhl K. Current protocols in molecular biology. J. Wiley & Sons; 1994. [Google Scholar]
- Bajaj V, Hwang C, Lee CA. hilA is a novel ompR/toxR family member that activates the expression of Salmonella typhimurium invasion genes. Molecular microbiology. 1995;18:715–727. doi: 10.1111/j.1365-2958.1995.mmi_18040715.x. [DOI] [PubMed] [Google Scholar]
- Bäumler AJ, Tsolis RM, van der Velden AWM, Stojiljkovic I, Anic S, Heffron F. Identification of a new iron regulated locus of Salmonella typhi. Gene. 1996;193:207–213. doi: 10.1016/s0378-1119(96)00560-4. [DOI] [PubMed] [Google Scholar]
- Baxter MA, Fahlen TF, Wilson RL, Jones BD. HilE interacts with HilD and negatively regulates hilA transcription and expression of the Salmonella enterica serovar Typhimurium invasive phenotype. Infection and immunity. 2003;71:1295–1305. doi: 10.1128/IAI.71.3.1295-1305.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ben-Dor A, Shamir R, Yakhini Z. Clustering gene expression patterns. J Comput Biol. 1999;6:281–297. doi: 10.1089/106652799318274. [DOI] [PubMed] [Google Scholar]
- Bishop A, House D, Perkins T, Baker S, Kingsley RA, Dougan G. Interaction of Salmonella enterica serovar Typhi with cultured epithelial cells: roles of surface structures in adhesion and invasion. Microbiology. 2008;154:1914–1926. doi: 10.1099/mic.0.2008/016998-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boddicker JD, Knosp BM, Jones BD. Transcription of the Salmonella invasion gene activator, hilA, requires HilD activation in the absence of negative regulators. J Bacteriol. 2003;185:525–533. doi: 10.1128/JB.185.2.525-533.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bohez L, Ducatelle R, Pasmans F, Botteldoorn N, Haesebrouck F, Van Immerseel F. Salmonella enterica serovar Enteritidis colonization of the chicken caecum requires the HilA regulatory protein. Vet Microbiol. 2006;116:202–210. doi: 10.1016/j.vetmic.2006.03.007. [DOI] [PubMed] [Google Scholar]
- Bueno SM, Santiviago CA, Murillo AA, Fuentes JA, Trombert AN, Rodas PI, Youderian P, Mora GC. Precise excision of the large pathogenicity island, SPI7, in Salmonella enterica serovar Typhi. J Bacteriol. 2004;186:3202–3213. doi: 10.1128/JB.186.10.3202-3213.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cano DA, Dominguez-Bernal G, Tierrez A, Garcia-Del Portillo F, Casadesus J. Regulation of capsule synthesis and cell motility in Salmonella enterica by the essential gene igaA. Genetics. 2002;162:1513–1523. doi: 10.1093/genetics/162.4.1513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chilcott GS, Hughes KT. Coupling of flagellar gene expression to flagellar assembly in Salmonella enterica serovar typhimurium and Escherichia coli. Microbiol Mol Biol Rev. 2000;64:694–708. doi: 10.1128/mmbr.64.4.694-708.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cleveland WS. Robust locally weighted regression and smoothing scatterplots. J. Amer. Stat. Assoc. 1979;74:829–836. [Google Scholar]
- Costa CS, Pettinari MJ, Mendez BS, Anton DN. Null mutations in the essential gene yrfF (mucM) are not lethal in rcsB, yojN or rcsC strains of Salmonella enterica serovar Typhimurium. FEMS Microbiol Lett. 2003;222:25–32. doi: 10.1016/S0378-1097(03)00221-0. [DOI] [PubMed] [Google Scholar]
- Deng W, Liou SR, Plunkett G, 3rd, Mayhew GF, Rose DJ, Burland V, Kodoyianni V, Schwartz DC, Blattner FR. Comparative genomics of Salmonella enterica serovar Typhi strains Ty2 and CT18. J Bacteriol. 2003;185:2330–2337. doi: 10.1128/JB.185.7.2330-2337.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ellermeier CD, Ellermeier JR, Slauch JM. HilD, HilC and RtsA constitute a feed forward loop that controls expression of the SPI1 type three secretion system regulator hilA in Salmonella enterica serovar Typhimurium. Molecular microbiology. 2005;57:691–705. doi: 10.1111/j.1365-2958.2005.04737.x. [DOI] [PubMed] [Google Scholar]
- Erickson KD, Detweiler CS. The Rcs phosphorelay system is specific to enteric pathogens/commensals and activates ydeI, a gene important for persistent Salmonella infection of mice. Molecular microbiology. 2006;62:883–894. doi: 10.1111/j.1365-2958.2006.05420.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Francez-Charlot A, Laugel B, Van Gemert A, Dubarry N, Wiorowski F, Castanie-Cornet MP, Gutierrez C, Cam K. RcsCDB His-Asp phosphorelay system negatively regulates the flhDC operon in Escherichia coli. Molecular microbiology. 2003;49:823–832. doi: 10.1046/j.1365-2958.2003.03601.x. [DOI] [PubMed] [Google Scholar]
- Frye J, Karlinsey JE, Felise HR, Marzolf B, Dowidar N, McClelland M, Hughes KT. Identification of new flagellar genes of Salmonella enterica serovar Typhimurium. J Bacteriol. 2006;188:2233–2243. doi: 10.1128/JB.188.6.2233-2243.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galan JE, Curtiss R., 3rd Cloning and molecular characterization of genes whose products allow Salmonella typhimurium to penetrate tissue culture cells. Proc Natl Acad Sci U S A. 1989;86:6383–6387. doi: 10.1073/pnas.86.16.6383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galan JE, Curtiss R., 3rd Expression of Salmonella typhimurium genes required for invasion is regulated by changes in DNA supercoiling. Infection and immunity. 1990;58:1879–1885. doi: 10.1128/iai.58.6.1879-1885.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haneda T, Winter SE, Butler BP, Wilson RP, Tukel C, Winter MG, Godinez I, Tsolis RM, Bäumler AJ. The capsule-encoding viaB locus reduces intestinal inflammation by a Salmonella pathogenicity island 1-independent mechanism. Infection and Immunity. 2009;77:2932–2942. doi: 10.1128/IAI.00172-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hashimoto Y, Khan AQ, Ezaki T. Positive autoregulation of vipR expression in ViaB region-encoded Vi antigen of Salmonella typhi. J Bacteriol. 1996;178:1430–1436. doi: 10.1128/jb.178.5.1430-1436.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hirose K, Ezaki T, Miyake M, Li T, Khan AQ, Kawamura Y, Yokoyama H, Takami T. Survival of Vi-capsulated and Vi-deleted Salmonella typhi strains in cultured macrophage expressing different levels of CD14 antigen. FEMS Microbiol Lett. 1997;147:259–265. doi: 10.1111/j.1574-6968.1997.tb10251.x. [DOI] [PubMed] [Google Scholar]
- Hughes KT, Gillen KL, Semon MJ, Karlinsey JE. Sensing structural intermediates in bacterial flagellar assembly by export of a negative regulator. Science (New York, N.Y. 1993;262:1277–1280. doi: 10.1126/science.8235660. [DOI] [PubMed] [Google Scholar]
- Johnson EM, Baron LS. Genetic transfer of the Vi antigen from Salmonella typhosa to Escherichia coli. J Bacteriol. 1969;99:358–359. doi: 10.1128/jb.99.1.358-359.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones GW, Richardson LA, Uhlman D. The invasion of HeLa cells by Salmonella typhimurium: reversible and irreversible bacterial attachment and the role of bacterial motility. J Gen Microbiol. 1981;127:351–360. doi: 10.1099/00221287-127-2-351. [DOI] [PubMed] [Google Scholar]
- Kage H, Takaya A, Ohya M, Yamamoto T. Coordinated regulation of expression of Salmonella pathogenicity island 1 and flagellar type III secretion systems by ATP-dependent ClpXP protease. J Bacteriol. 2008;190:2470–2478. doi: 10.1128/JB.01385-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lawes M, Maloy S. MudSacI, a transposon with strong selectable and counterselectable markers: use for rapid mapping of chromosomal mutations in Salmonella typhimurium. J Bacteriol. 1995;177:1383–1387. doi: 10.1128/jb.177.5.1383-1387.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin D, Rao CV, Slauch JM. The Salmonella SPI1 type three secretion system responds to periplasmic disulfide bond status via the flagellar apparatus and the RcsCDB system. J Bacteriol. 2008;190:87–97. doi: 10.1128/JB.01323-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Looney RJ, Steigbigel RT. Role of the Vi antigen of Salmonella typhi in resistance to host defense in vitro. J Lab Clin Med. 1986;108:506–516. [PubMed] [Google Scholar]
- Lucas RL, Lostroh CP, DiRusso CC, Spector MP, Wanner BL, Lee CA. Multiple factors independently regulate hilA and invasion gene expression in Salmonella enterica serovar typhimurium. J Bacteriol. 2000;182:1872–1882. doi: 10.1128/jb.182.7.1872-1882.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Macnab RM. Type III flagellar protein export and flagellar assembly. Biochim Biophys Acta. 2004;1694:207–217. doi: 10.1016/j.bbamcr.2004.04.005. [DOI] [PubMed] [Google Scholar]
- Majdalani N, Gottesman S. The Rcs phosphorelay: a complex signal transduction system. Annu Rev Microbiol. 2005;59:379–405. doi: 10.1146/annurev.micro.59.050405.101230. [DOI] [PubMed] [Google Scholar]
- McClelland M, Sanderson KE, Spieth J, Clifton SW, Latreille P, Courtney L, Porwollik S, Ali J, Dante M, Du F, Hou S, Layman D, Leonard S, Nguyen C, Scott K, Holmes A, Grewal N, Mulvaney E, Ryan E, Sun H, Florea L, Miller W, Stoneking T, Nhan M, Waterston R, Wilson RK. Complete genome sequence of Salmonella enterica serovar Typhimurium LT2. Nature. 2001;413:852–856. doi: 10.1038/35101614. [DOI] [PubMed] [Google Scholar]
- Miller JH. Experiments in Molecular Genetics. New York: Cold Spring Harbor Laboratory Press; 1972. [Google Scholar]
- Mills SD, Finlay BB. Comparison of Salmonella typhi and Salmonella typhimurium invasion, intracellular growth and localization in cultured human epithelial cells. Microbial pathogenesis. 1994;17:409–423. doi: 10.1006/mpat.1994.1086. [DOI] [PubMed] [Google Scholar]
- Mouslim C, Delgado M, Groisman EA. Activation of the RcsC/YojN/RcsB phosphorelay system attenuates Salmonella virulence. Molecular microbiology. 2004;54:386–395. doi: 10.1111/j.1365-2958.2004.04293.x. [DOI] [PubMed] [Google Scholar]
- Mouslim C, Latifi T, Groisman EA. Signal-dependent requirement for the co-activator protein RcsA in transcription of the RcsB-regulated ugd gene. The Journal of biological chemistry. 2003;278:50588–50595. doi: 10.1074/jbc.M309433200. [DOI] [PubMed] [Google Scholar]
- Nair S, Alokam S, Kothapalli S, Porwollik S, Proctor E, Choy C, McClelland M, Liu SL, Sanderson KE. Salmonella enterica serovar Typhi strains from which SPI7, a 134-kilobase island with genes for Vi exopolysaccharide and other functions, has been deleted. J Bacteriol. 2004;186:3214–3223. doi: 10.1128/JB.186.10.3214-3223.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parkhill J, Dougan G, James KD, Thomson NR, Pickard D, Wain J, Churcher C, Mungall KL, Bentley SD, Holden MT, Sebaihia M, Baker S, Basham D, Brooks K, Chillingworth T, Connerton P, Cronin A, Davis P, Davies RM, Dowd L, White N, Farrar J, Feltwell T, Hamlin N, Haque A, Hien TT, Holroyd S, Jagels K, Krogh A, Larsen TS, Leather S, Moule S, O'Gaora P, Parry C, Quail M, Rutherford K, Simmonds M, Skelton J, Stevens K, Whitehead S, Barrell BG. Complete genome sequence of a multiple drug resistant Salmonella enterica serovar Typhi CT18. Nature. 2001;413:848–852. doi: 10.1038/35101607. [DOI] [PubMed] [Google Scholar]
- Pickard D, Li J, Roberts M, Maskell D, Hone D, Levine M, Dougan G, Chatfield S. Characterization of defined ompR mutants of Salmonella typhi: ompR is involved in the regulation of Vi polysaccharide expression. Infection and immunity. 1994;62:3984–3993. doi: 10.1128/iai.62.9.3984-3993.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pristovsek P, Sengupta K, Lohr F, Schafer B, von Trebra MW, Ruterjans H, Bernhard F. Structural analysis of the DNA-binding domain of the Erwinia amylovora RcsB protein and its interaction with the RcsAB box. The Journal of biological chemistry. 2003;278:17752–17759. doi: 10.1074/jbc.M301328200. [DOI] [PubMed] [Google Scholar]
- Promponas VJ, Enright AJ, Tsoka S, Kreil DP, Leroy C, Hamodrakas S, Sander C, Ouzounis CA. CAST: an iterative algorithm for the complexity analysis of sequence tracts. Complexity analysis of sequence tracts. Bioinformatics. 2000;16:915–922. doi: 10.1093/bioinformatics/16.10.915. [DOI] [PubMed] [Google Scholar]
- Quackenbush J. Microarray data normalization and transformation. Nat Genet. 2002;32 Suppl:496–501. doi: 10.1038/ng1032. [DOI] [PubMed] [Google Scholar]
- Raffatellu M, Chessa D, Wilson RP, Dusold R, Rubino S, Bäumler AJ. The Vi capsular antigen of Salmonella enterica serotype Typhi reduces Toll-like receptor-dependent interleukin-8 expression in the intestinal mucosa. Infection and immunity. 2005;73:3367–3374. doi: 10.1128/IAI.73.6.3367-3374.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raffatellu M, Santos RL, Chessa D, Wilson RP, Winter SE, Rossetti CA, Lawhon SD, Chu H, Lau T, Bevins CL, Adams LG, Bäumler AJ. The capsule encoding the viaB locus reduces interleukin-17 expression and mucosal innate responses in the bovine intestinal mucosa during infection with Salmonella enterica serotype Typhi. Infection and immunity. 2007;75:4342–4350. doi: 10.1128/IAI.01571-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saeed AI, Sharov V, White J, Li J, Liang W, Bhagabati N, Braisted J, Klapa M, Currier T, Thiagarajan M, Sturn A, Snuffin M, Rezantsev A, Popov D, Ryltsov A, Kostukovich E, Borisovsky I, Liu Z, Vinsavich A, Trush V, Quackenbush J. TM4: a free, open-source system for microarray data management and analysis. Biotechniques. 2003;34:374–378. doi: 10.2144/03342mt01. [DOI] [PubMed] [Google Scholar]
- Sambrook J, Fritsch EF, Maniatis T. Molecular cloning. New York: Cold Spring Harbor Laboratory Press; 1989. [Google Scholar]
- Seo YS, Sriariyanun M, Wang L, Pfeiff J, Phetsom J, Lin Y, Jung KH, Chou HH, Bogdanove A, Ronald P. A two-genome microarray for the rice pathogens Xanthomonas oryzae pv. oryzae and X. oryzae pv. oryzicola and its use in the discovery of a difference in their regulation of hrp genes. BMC microbiology. 2008;8:99. doi: 10.1186/1471-2180-8-99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharma A, Qadri A. Vi polysaccharide of Salmonella typhi targets the prohibitin family of molecules in intestinal epithelial cells and suppresses early inflammatory responses. Proc Natl Acad Sci U S A. 2004;101:17492–17497. doi: 10.1073/pnas.0407536101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tartera C, Metcalf ES. Osmolarity and growth phase overlap in regulation of Salmonella typhi adherence to and invasion of human intestinal cells. Infection and immunity. 1993;61:3084–3089. doi: 10.1128/iai.61.7.3084-3089.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsolis RM, Young GM, Solnick JV, Bäumler AJ. From bench to bedside: stealth of enteroinvasive pathogens. Nat Rev Microbiol. 2008;6:883–892. doi: 10.1038/nrmicro2012. [DOI] [PubMed] [Google Scholar]
- Virlogeux I, Waxin H, Ecobichon C, Lee JO, Popoff MY. Characterization of the rcsA and rcsB genes from Salmonella typhi: rcsB through tviA is involved in regulation of Vi antigen synthesis. J Bacteriol. 1996;178:1691–1698. doi: 10.1128/jb.178.6.1691-1698.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Virlogeux I, Waxin H, Ecobichon C, Popoff MY. Role of the viaB locus in synthesis, transport and expression of Salmonella typhi Vi antigen. Microbiology. 1995;141(Pt 12):3039–3047. doi: 10.1099/13500872-141-12-3039. [DOI] [PubMed] [Google Scholar]
- Wang Q, Zhao Y, McClelland M, Harshey RM. The RcsCDB signaling system and swarming motility in Salmonella enterica serovar typhimurium: dual regulation of flagellar and SPI-2 virulence genes. J Bacteriol. 2007;189:8447–8457. doi: 10.1128/JB.01198-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang RF, Kushner SR. Construction of versatile low-copy-number vectors for cloning, sequencing and gene expression in Escherichia coli. Gene. 1991;100:195–199. [PubMed] [Google Scholar]
- Wehland M, Bernhard F. The RcsAB box. Characterization of a new operator essential for the regulation of exopolysaccharide biosynthesis in enteric bacteria. The Journal of biological chemistry. 2000;275:7013–7020. doi: 10.1074/jbc.275.10.7013. [DOI] [PubMed] [Google Scholar]
- West NP, Sansonetti P, Mounier J, Exley RM, Parsot C, Guadagnini S, Prevost MC, Prochnicka-Chalufour A, Delepierre M, Tanguy M, Tang CM. Optimization of virulence functions through glucosylation of Shigella LPS. Science (New York, N.Y. 2005;307:1313–1317. doi: 10.1126/science.1108472. [DOI] [PubMed] [Google Scholar]
- Wilson RP, Raffatellu M, Chessa D, Winter SE, Tukel C, Bäumler AJ. The Vi-capsule prevents Toll-like receptor 4 recognition of Salmonella. Cell Microbiol. 2008;10:876–890. doi: 10.1111/j.1462-5822.2007.01090.x. [DOI] [PubMed] [Google Scholar]
- Winter SE, Raffatellu M, Wilson RP, Russmann H, Bäumler AJ. The Salmonella enterica serotype Typhi regulator TviA reduces interleukin-8 production in intestinal epithelial cells by repressing flagellin secretion. Cell Microbiol. 2008;10:247–261. doi: 10.1111/j.1462-5822.2007.01037.x. [DOI] [PubMed] [Google Scholar]
- Winter SE, Thiennimitr P, Nuccio SP, Haneda T, Winter MG, Wilson RP, Russell JM, Henry T, Tran QT, Lawhon SD, Gomez G, Bevins CL, Russmann H, Monack DM, Adams LG, Bäumler AJ. Contribution of flagellin pattern recognition to intestinal inflammation during Salmonella enterica serotype Typhimurium infection. Infection and Immunity. 2009;77:1904–1916. doi: 10.1128/IAI.01341-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang IV, Chen E, Hasseman JP, Liang W, Frank BC, Wang S, Sharov V, Saeed AI, White J, Li J, Lee NH, Yeatman TJ, Quackenbush J. Within the fold: assessing differential expression measures and reproducibility in microarray assays. Genome Biol. 2002;3 doi: 10.1186/gb-2002-3-11-research0062. research0062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao L, Ezak T, Li ZY, Kawamura Y, Hirose K, Watanabe H. Vi-Suppressed wild strain Salmonella typhi cultured in high osmolarity is hyperinvasive toward epithelial cells and destructive of Peyer's patches. Microbiology and immunology. 2001;45:149–158. doi: 10.1111/j.1348-0421.2001.tb01283.x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.