Abstract
Objectives
Increasing HDL levels is a potential strategy for the treatment of atherosclerosis.
Methods and results
ITX5061, a molecule initially characterized as a p38 MAPK inhibitor, increased HDL-C levels by 20 % in a human population of hypertriglyceridemic subjects with low HDL levels. ITX5061 also moderately increased apoA-I but did not affect VLDL/LDL cholesterol or plasma triglyceride concentrations. ITX5061 increased HDL-C in WT and human apoA-I transgenic mice and kinetic experiments showed that ITX5061 decreased the fractional catabolic rate of HDL-CE and reduced its hepatic uptake. In transfected cells, ITX5061 inhibited SR-BI-dependent uptake of HDL-CE. Moreover, ITX5061 failed to increase HDL-C levels in SR-BI−/− mice. To assess effects on atherosclerosis, ITX5061 was given to atherogenic diet-fed Ldlr+/− mice with or without CETP expression for 18 weeks. In both the control and CETP-expressing groups, ITX5061 treated mice displayed reductions of early atherosclerotic lesions in the aortic arch (−40 %, p<0.05), and a non-significant trend to reduced lesion area in the proximal aorta.
Conclusions
Our data indicate that ITX5061 increases HDL-C levels by inhibition of SR-BI activity. This suggests that pharmacological inhibition of SR-BI has the potential to raise HDL-C and apoA-I levels without adverse effects on VLDL/LDL cholesterol levels in humans.
Keywords: Scavenger Receptor B-I, High Density Lipoproteins, Inhibitors, p38 MAPK, atherosclerosis
Introduction
Plasma high-density lipoprotein (HDL) levels are inversely correlated with atherosclerotic cardiovascular disease and raising HDL levels by lifestyle changes or pharmacological interventions is an emerging strategy that might help to reduce the residual burden of disease in patients treated with Low-density lipoprotein (LDL) lowering approaches 1, 2. While increasing synthesis or infusion of apolipoprotein A-I (apoA-I) or HDL reduces atherosclerosis in animals and humans, it is not clear that all approaches to raising HDL will reduce atherosclerosis.
The scavenger receptor B-I (SR-BI) is a major factor regulating HDL catabolism. SR-BI binds HDL and mediates the selective uptake of HDL-cholesteryl ester (CE) in the liver and steroidogenic tissues. 3. While the role of SR-BI in HDL metabolism in mice and rats has been clearly shown, much less information is available on the function of SR-BI in humans. Primary human hepatocytes do demonstrate selective uptake of HDL-CE 4 but the quantitative importance of this pathway has not been shown. In humans, cholesteryl ester transfer protein (CETP), a factor lacking in mice and rats, plays a major role in HDL-CE catabolism. In the present study, we demonstrate that ITX5061, a molecule initially characterized as a p38 mitogen-actiated protein kinase (MAPK) inhibitor, caused an increase in HDL cholesterol (HDL-C) and apoA-I levels in humans. Studies in mice and cultured cells indicated that ITX5061 is an inhibitor of the SR-BI activity. Interestingly, ITX5061 did not cause increases in very-low-density lipoprotein (VLDL) and LDL cholesterol and reduced atherosclerosis in the aortic arch in LDL receptor (Ldlr) heterozygous mice fed an atherogenic diet.
Materials and methods
A complete Materials and Methods section is available in the online supplemental materials (online at http://atvb.ahajournals.org).
Molecule
Structure of ITX5061 is shown in figure S1. The empirical formula of ITX5061 is C30H38ClN3O7S. The molecular weight of ITX5061 (as an HCl salt) is 620.2.
Human data
KC706-C06 study was conducted from 6 November 2006 to 8 May 2007. It was a multicenter, randomized, double-blind, placebo-controlled study. Its primary objective was to evaluate the ability of ITX5061 to increase HDL-C in patients with low HDL-C and elevated triglycerides (TG) at Baseline.
Animals
C57Bl/6 Wild Type (WT) mice, C57Bl/6 SR-BI deficient mice (SR-BI−/−) 5, and C57Bl/6 transgenic mice expressing human apoA-I under the control of its natural flanking regions (HuAITg) 6 were used in the present study. Atherosclerosis studies were conducted in F1 hybrid C57BL/6 × DBA/1 Ldlr+/− mice fed the Paigen diet, which is high-fat/cholesterol/bile salt diet containing 1.25% cholesterol, 7.5% cocoa butter, and 0.5% cholic acid (TD 88051; Harlan Teklad, Madison, WI). Animal protocols were approved by the Institutional Animal Care and Use Committee of Columbia University.
Results
ITX5061 increases HDL levels in humans
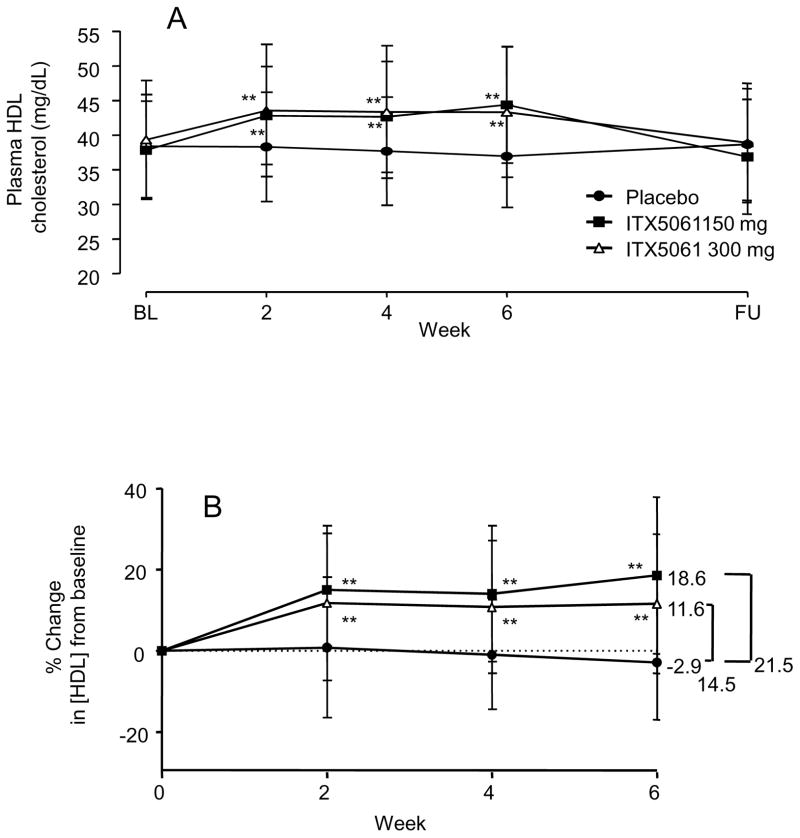
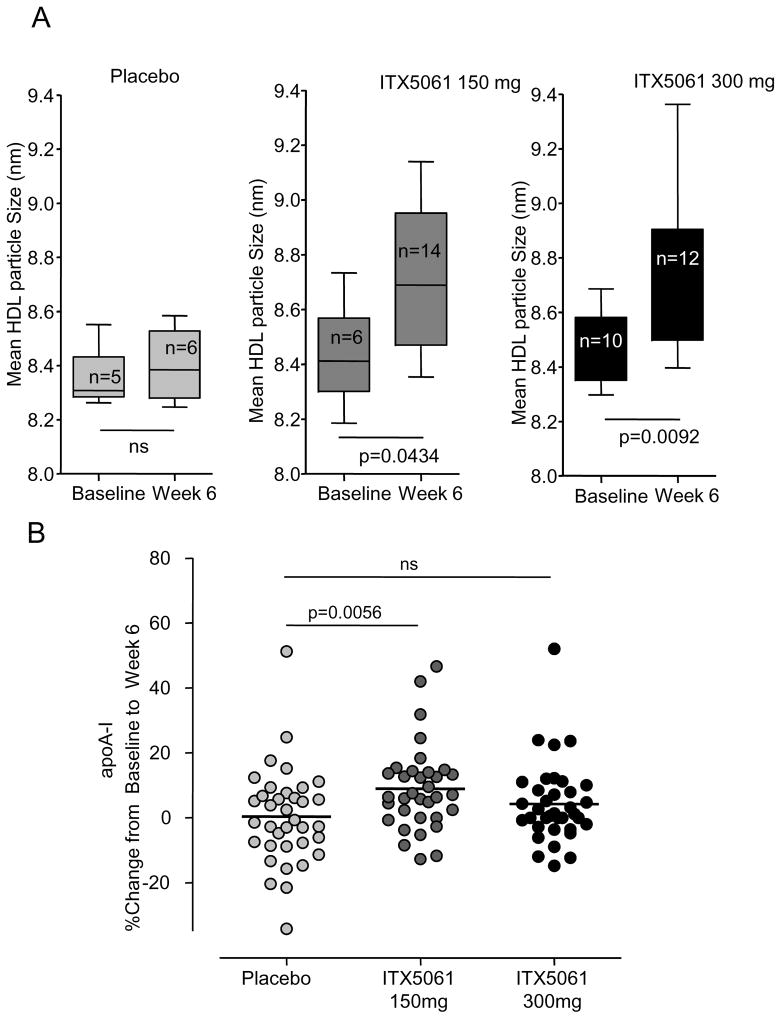
ITX5061 was initially characterized as a type II (non competitive) inhibitor of p38 MAPK (Fig S2), and was noted to cause increases in HDL-C levels in humans. In order to systematically investigate effects on plasma lipoprotein levels, ITX5061 was administered at two different doses in a population of subjects with low HDL-C (men <45 mg/dl and women <55 mg/dl) and increased TG concentrations (TG ranging from 150 to 400 mg/dl). Baseline characteristics are given in table SI. Subjects received either placebo, ITX5061 at 150 mg or 300 mg daily. ITX5061 treatment resulted in an increase in HDL-C concentration that was similar in the two ITX5061 treated groups (approx. 20 % increases). The rise in HDL-C was observed throughout the treatment period and was reversed after cessation of the drug (Fig 1). The mean HDL particle size was significantly increased in ITX5061 treated subjects (P<0.05) (Fig 2A). No significant changes in other lipid parameters (LDL-C and TG) were observed, although a transient, non-significant, increase in TG was observed after 2 weeks of treatment (Fig S3). ApoA-I protein levels were moderately increased in the 150 mg group (+ 10%, P<0.05) but were not statistically different in the 300 mg group (Fig 2B). Adverse events were similar in all groups except for one patient in the 150 mg group and 4 patients in the 300 mg group who had reversible increases in transaminase levels. These findings indicate a selective increase in HDL-C and apoA-I in subjects treated with the 150 mg dose of ITX5061. Further increases in these parameters were not seen at the 300 mg dose.
Figure 1. Changes in lipid parameters over time during ITX5061 administration.
Patients received placebo (n=37), ITX5061 at 150mg (n=38) and ITX5061 at 300 mg (n=40) daily. Lipid parameters were determined at indicated time points. (A): Absolute change in HDL-C concentration. (B): Mean percent changes in HDL-C from baseline. Values are mean ± S.D. **P<0.05, ***P<0.01, P-values are against placebo, same time point, Kruskall-Wallis, Mann-Whitney test. .
Figure 2.
(A) HDL size distribution in ITX5061 treated patients. HDL size was determined after 6 weeks of treatment using NMR. P-value based on Mann Whitney test. (B) Effect of ITX5061 treatment on apoA-I concentration. ApoA-I concentration was determined at the indicated time point. P-value based on Mann-Whitney test.
ITX5061 increases HDL-C and size in HuAITg mice
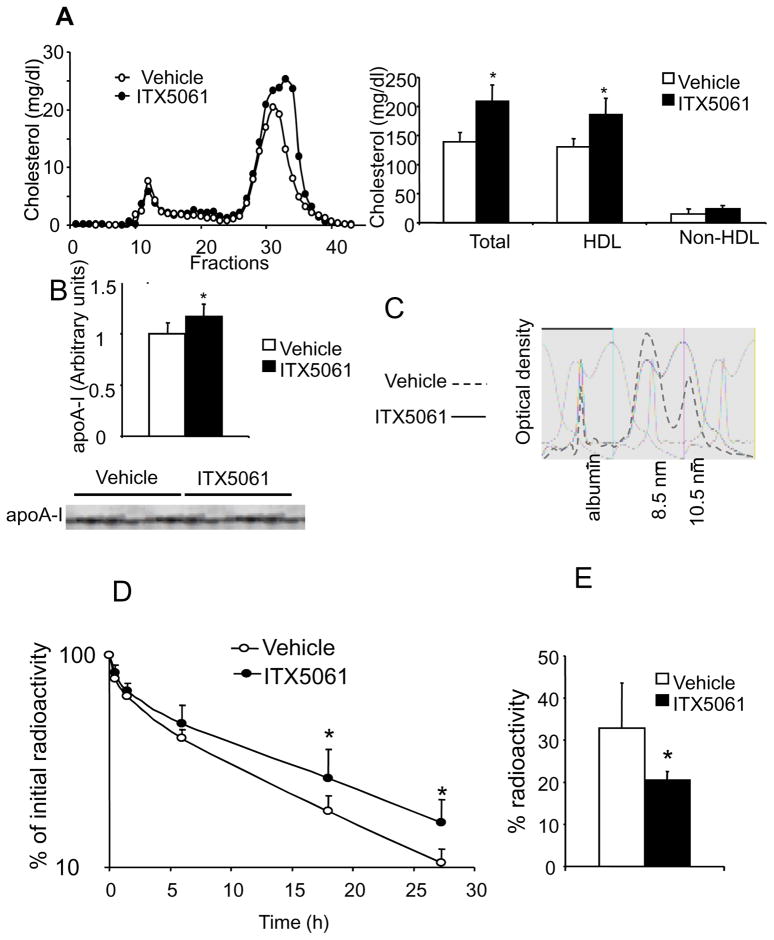
To investigate the mechanism of HDL-C raising by ITX5061, further studies were conducted in mice. HuAITg mice were treated with ITX5061 (30 mg/kg/day) or vehicle for one week. This resulted in a 50% increase in HDL-C levels compared to baseline, but no change in non-HDL-C levels (Fig 3A). As determined by scanning SDS-PAGE gels, apoA-I levels were moderately (+15 %) but significantly increased in ITX5061-treated HuAITg mice, compared to mice received vehicle (Fig 3B). Consistent with the more prominent increase in HDL-C than apoA-I, ITX5061 treatment induced a shift towards larger sized HDL as shown by native-PAGE (Fig 3C). In order to assess whether ITX5061 could affect expression of genes involved in HDL biosynthesis or catabolism in the liver, relative mRNA levels of apoA-I, ABCA1 and SR-BI were determined in ITX5061 and vehicle treated animals. No changes in mRNA levels were found for any of these transcripts (Fig S4) suggesting that ITX5061 raised HDL independent of these transcript levels.
Figure 3.
(A) Plasma lipid levels and cholesterol distribution in HuAITg mice with or without ITX5061. HuAITg mice were treated for one week with ITX5061 or vehicle at 30 mg/kg/day. 100 μl of pooled plasma from 5 distinct mouse were used for FPLC analysis. Plasma HDL-C was determined after ultracentrifugation. * indicates a significant difference from mice receiving vehicle. (P<0.05 in both cases; Mann-Whitney test). (B) apoAI concentrations in HuAITg mice with or without ITX5061. * indicates a significant difference from mice receiving vehicle. (P<0.05; Mann-Whitney test). (C) HDL size distribution in HuAITg mice with or without ITX5061. HDL size was determined by native-PAGE. Profiles were determined in individual plasma samples. One presentative profile in each group is shown. (D) Plasma kinetic of HDL labeled with [3H]CE in HuAITg mice with or without ITX5061. Values are the fraction of the injected dose remaining at each time point. The curves were fitted using a biexponential equation. Data are given as mean ± S.D.; n = 4 mice/group for each point. *: P<0.05 Vs vehicle, same time point, Mann-Whitney test. (E) HDL-C uptake. Values are expressed as the percentage of total injected radioactivity retrieved in the liver. *: P<0.05 Vs vehicle, Mann-Whitney test.
ITX5061 decreases HDL-CE catabolism and hepatic uptake
To gain more insight into the mechanisms by which ITX5061 increases HDL-C, a kinetic study was performed. HDL labelled with [3H] CE was injected via the tail vein into HuAITg mice pre-treated or not with ITX5061. As shown in Fig 3D, ITX5061 significantly decreased HDL-CE catabolism with an FCR of 1.86±0.40 pools/d vs 2.47±0.26 pools/d in the control group (P<0.05), while calculated production rates were identical in both groups (129±24 μg/g/d vs 129±16 μg/g/d). Moreover, accumulation of [3H] CE in the liver was significantly lower in ITX5061-treated mice indicating that increased HDL-CE levels were due to reduced uptake by the liver (Fig 3E).
ITX5061 does not increase HDL levels in SR-BI−/− mice and reduces selective uptake via SR-BI in cell culture
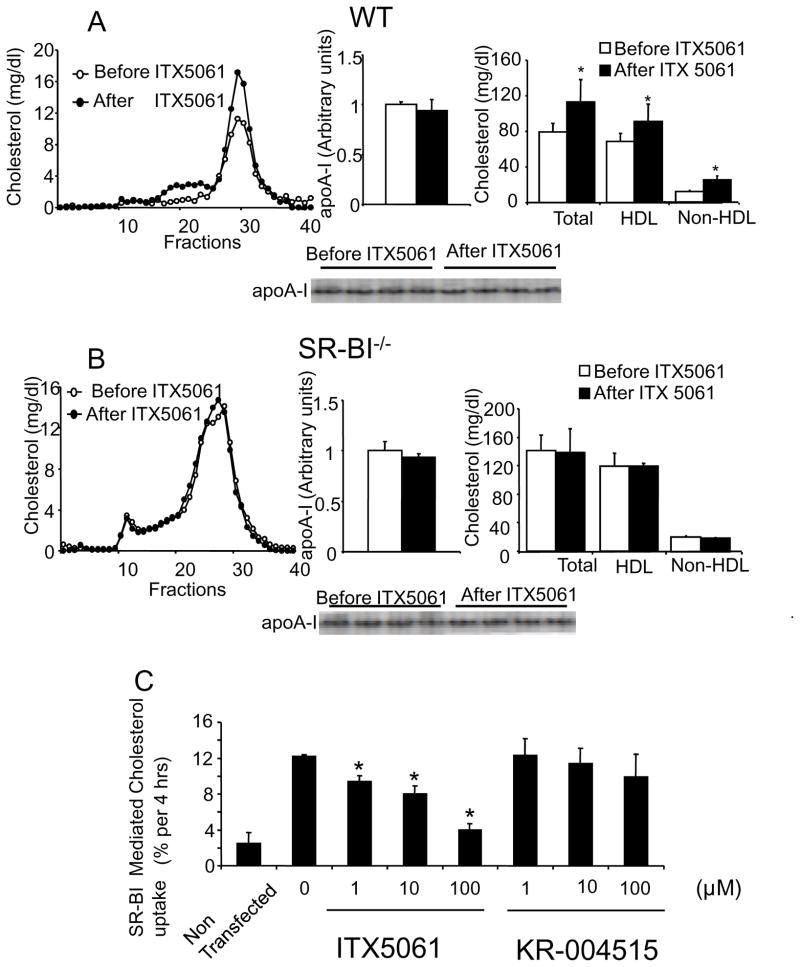
Since SR-BI has a major role in the clearance of HDL-C and CE in the liver, we determined whether ITX5061 could still raise HDL levels in SR-BI−/− mice. WT and SR-BI−/− mice were treated for one week by daily administration of ITX5061 at 30mg/kg/day. While ITX5061 increased HDL-C in WT mice its effects were abolished in SR-BI−/− mice suggesting that the mechanism of action was dependent on SR-BI (Fig 4B). A significant increase in non HDL cholesterol levels was also observed in ITX5061-treated WT mice. Analysis of apolipoproteins by SDS-PAGE in fractions corresponding to the LDL region of similar profiles indicated they consisted primarily of apoB lipoproteins with only trace amounts of apoA-1 (data not shown). Interestingly despite increases in HDL-C, no increase in apoA-I was observed in ITX5061-treated WT mice (Fig 4A). We also confirmed by Western blot that ITX5061 did not affect the protein levels of apoA-I, ABCA1 and SR-BI in the liver (figure S4B)
Figure 4. Plasma lipids, apoA-I concentrations and cholesterol distribution in WT and SR-BI−/− mice with or without ITX5061.
WT (A) and SR-BI−/− mice (B) were treated for one week with ITX5061 or vehicle at 30 mg/kg/d. 100 μl of a pooled plasma from 4 distinct mouse were used for FPLC analysis. * indicates a significant difference from mice before treatment (P<0.05 in both cases; Wilcoxon test). (C) Effect of ITX5061 on SR-BI mediated cholesterol uptake. HEK 293 cells were transfected with SR-BI cDNA or a mock vector and HDL-C uptake was determined as describe in materials and methods (*: P<0.05 Vs 0 μM; Mann-Whitney test). Values obtained at 4°C were subtracted.
To test the SR-BI inhibitory potential of ITX5061 in vitro, HEK 293 cells over expressing SR-BI were incubated with [3H]CE labelled HDL. As shown in Fig 4C, ITX5061 significantly decreased HDL uptake at 1 μM concentration while a structurally related p38 MAPK inhibitor (KR-004515) had no effect at the same concentration.
ITX5061 increases HDL-C and reduces atherosclerosis in Ldlr+/− mice fed the Paigen diet
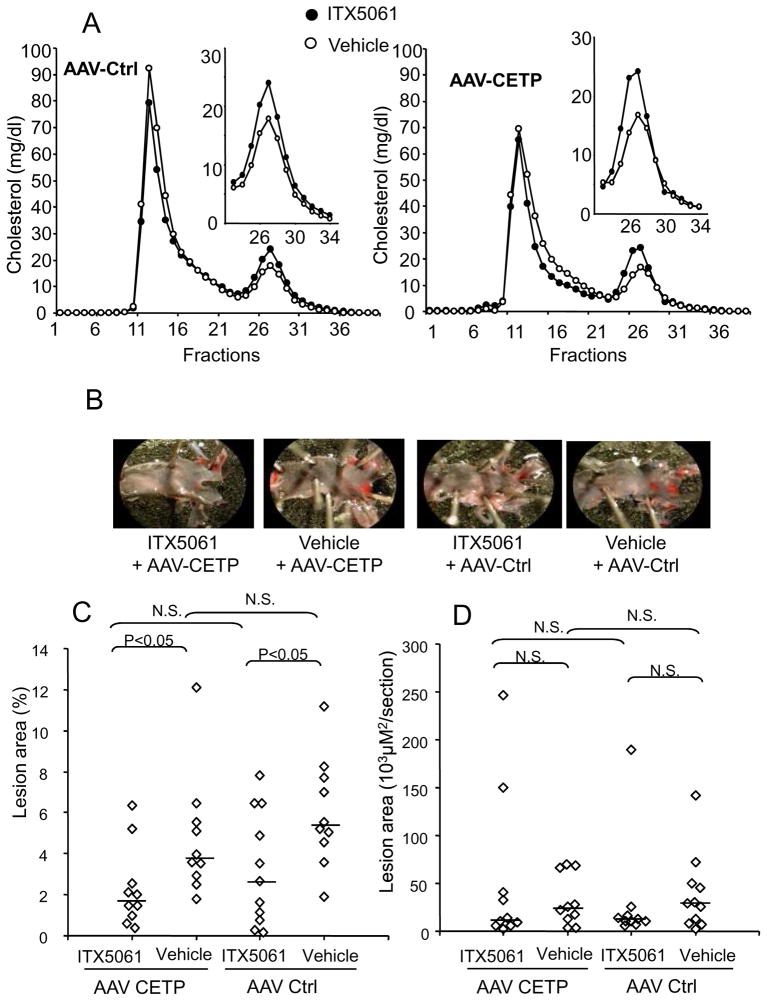
Most prior studies of the effects of SR-BI deficiency or inhibition on atherogenesis have used Ldlr−/− or apoE−/− mice 7–9. We wished to determine effects of ITX5061 on atherogenesis in a setting where the clearance of VLDL/LDL particles by apoE- or Ldlr-dependent pathways, is not completely impaired and with the potential availability of a compensatory pathway of reverse cholesterol transport via CETP. Indeed, it has been shown recently that CETP expression was able to reverse the atherogenic phenotype in SR-BI−/− mice10. Thus, to determine the effects of ITX5061 on atherogenesis, Ldlr+/− mice, with or without expression of CETP, were fed the Paigen diet containing ITX5061 (0.037 %) for 18 weeks. CETP expression was induced by injection of AAV-CETP11 and was monitored two weeks later. Plasma CETP levels were slightly lower than obtained in a human plasma sample in mice injected with AAV-CETP but within the physiological range for humans (Fig S5). Plasma lipid parameters were monitored after 2 and 18 weeks of treatment. No differences in total cholesterol or TG levels were observed between ITX5061-treated or control mice. However, a 30 % increase in HDL-C concentrations was observed in ITX5061 treated groups as determined by FPLC (Fig 5A) and ultracentrifugation (Table SII). At human-like levels, CETP expression has only little effects on plasma lipid levels in WT mice as originally reported 12. As expected, animals fed the Paigen diet developed moderately elevated transaminase levels13 but there were no hyperbilirubinemia and there were no differences between the 4 groups (Table SII)
Figure 5.
(A) FPLC analysis of plasma cholesterol in male Ldlr+/− mice with or without ITX5061. 100 μl of pooled plasma were used for FPLC analysis. (B) Representative Oil Red O –stained aortas Ldlr+/− mice were fed the Paigen diet containing or not ITX5061 for 18 weeks. Aortas were stained with Oil Red O. (C) Quantification of atherosclerosis lesions in Ldlr+/− mice. Oil Red O-stained lesions were quantified and expressed as a percent of total aortic area. Horizontal bars indicate the group medians. Statistical analysis by Kruskall-Wallis-Mann Whitney test (D) Atherosclerotic lesion development in the proximal aorta. Quantification of proximal aortic root lesion area was performed by morphometric analysis of H&E stained sections. Values represent the average of 5 sections per mouse. Horizontal bars indicate the group medians Statistical analysis by Kruskall-Wallis-Mann Whitney test
Following 18 weeks of ITX5061 administration, atherosclerotic lesion areas were first quantified by en face analysis of aortic arches. Data are reported as the percentage of the atherosclerotic lesions stained by Oil Red O on aortic surface. Fig 5B shows representative pictures of aortic arches from female mice with or without treatment by ITX5061. Lesions were observed primarily at branch points and sometimes along the lesser curvature. A significant 40% reduction of atherosclerotic lesions was observed in ITX5061-treated mice. Similar reductions in atherosclerosis were seen in ITX5061-treated mice injected with AAV-CETP or AAV-Ctrl (Fig 5C). No difference in lesion area was observed when comparing AAV-Ctrl groups and AAV-CETP groups either in control or ITX-5061 treated mice. Analysis of the lesions was also performed in the aortic valves (figure 5D). Although there was a trend towards reduced lesion area in both groups treated with ITX5061, the differences were not statistically significant.
Discussion
Our findings indicate that ITX5061, initially characterized as a p38 MAPK inhibitor, is able to increase HDL-C levels in humans and rodents and strongly suggest that this occurs at least in part through inhibition of SR-BI activity. Importantly, while previous studies have shown that modulation of SR-BI activity could affect apoB-containing lipoprotein levels, ITX5061 did not increase VLDL/LDL cholesterol or plasma TG concentrations in humans and Ldlr+/− mice. Finally, although many studies have documented markedly increased atherosclerosis in various mouse models deficient in SR-BI, we observed that ITX5061 had no significant effect on atherosclerotic lesions in the aortic valves and reduced early atherosclerotic lesions in the aortic arch of Ldlr+/− mice fed the Paigen diet for 18 weeks.
Several lines of evidence suggest that the primary mode of action of ITX5061 is via inhibition of SR-BI activity. First, similar to what is observed in SR-BI−/− mice, ITX5061 increased HDL-C more than apoA-I and led to formation of large sized HDL enriched in cholesterol. Second, ITX5061 raised HDL-C by decreasing HDL-CE plasma catabolism and uptake by liver. Third, ITX5061 effects on HDL-C were abolished in SR-BI−/− mice. Fourth, in vitro, ITX5061 inhibited SR-BI mediated HDL-CE uptake while other structurally related compounds without HDL raising ability did not. While ITX5061 was initially developed as a p38 MAPK inhibitor it seems unlikely that this property contributed to increased HDL since structurally similar p38 MAPK inhibitors did not increase HDL levels. Nevertheless, the exact mechanism of SR-BI inhibition by ITX5061 remains to be determined. Other SR-BI inhibitors described to date decreased HDL-CE selective uptake and increased the binding of HDL to SR-BI suggesting the formation of inactive HDL-SR-BI complexes 14, 15.
The effects of SR-BI deficiency/inhibition on lipoprotein profiles are well documented in mice 5, 16, 17. SR-BI−/− mice display a marked increase in HDL-C concentration due to accumulation of very large size apoE- and cholesterol- and CE- enriched HDL. In the absence of apoE or Ldlr, SR-BI deficiency also increases VLDL/LDL concentration indicating a role for SR-BI as a back up receptor for these lipoproteins 16, 18–20. In contrast to mice, much less is known about the effects of SR-BI deficiency or inhibition on lipoprotein profiles in humans. Several non functional SR-BI polymorphisms have been associated with changes in HDL and LDL cholesterol levels perhaps suggesting a role of SR-BI in humans 21–24. No genetic deficiencies for SR-BI have been described to date 25, 26. Only one preliminary report on administration of SR-BI inhibitory molecules in humans has been presented. HDL376, a molecule with SR-BI inhibitory properties was able to increase HDL-C levels in humans (+20 %) and also in non human primates 27, 28. Our results suggest that the use of SR-BI inhibitors in humans has the potential to increase HDL-C and to a lesser extent apoA-I without detrimental effects on apoB-containing lipoprotein concentration, leading to a potentially less atherogenic lipoprotein profile. Interestingly, ITX5061 increased apoA-I as well as HDL-C in humans and in HuAITg mice, but in wild type mice only increased HDL-C levels. This suggests that there could be a difference in effects of SR-BI deficiency on HDL particles containing human apoA-I versus mouse apoA-I. Nevertheless, the increase of apo A-I was only moderate as compared to HDL-C, consistent with the SR-BI inhibition mechanism. The lack of increase VLDL and LDL cholesterol in humans treated with ITX5061 likely reflects the effect of partial SR-BI deficiency in a setting where apoE and Ldlr are still functional.
Following the discovery of dramatically increased HDL levels in SR-BI−/− mice 5, there was considerable initial interest in the potential to use inhibition of SR-BI as an anti-atherogenic strategy. However, this idea was discarded in the face of compelling evidence that SR-BI deficiency increased atherosclerosis in several different mouse models, sometimes in a dramatic fashion, while SR-BI overexpression had the opposite effects 7–9, 16, 29–32. Besides genetic models of SR-BI overexpression or deficiency, pharmacological SR-BI inhibitors such as BLTs, HDL376 and R-138329 have been characterized 14, 15, 27, 33, 34. The effects of the latter compound on atherosclerosis have been investigated in apoE−/− mice and moderate pro-atherogenic effects were observed but only at high concentration 35. The increase in atherosclerosis seen in SR-BI−/− mice has been attributed to the decrease in reverse cholesterol transport that has been clearly demonstrated in these mice 36. In favor of this hypothesis, CETP expression was able to reverse the atherogenic phenotype in SR-BI−/− mice10. However, in many of these studies 7, 8, 16 changes in HDL levels were associated with parallel changes in levels of VLDL and LDL, tending to confound the analysis of the mechanisms of atherogenesis. We did not find a pro-atherogenic effect of CETP on atherosclerosis in our model, either with or without ITX5061 treatment. This is possibly due to the low level of CETP expression at which we chose to work. CETP was previouly shown to be moderately pro-atherogenic in Ldlr−/− that displayed much higher plasma CETP levels37.
In the present study we observed that ITX5061 treatment resulted in a 40 % reduction in atherosclerosis in the aortic arch after 18 weeks of treatment in Ldlr+/− mice fed the Paigen diet. The antiatherogenic effect of the molecule was observed in the control and in the CETP expressing group. Our results suggest therefore that partial SR-BI inhibition could have some beneficial effects per se whether an alternative pathway for HDL cholesterol uptake or transfer is provided by CETP or not.
Several points might account for differences between our results and those from previous studies. First we used a model of Ldlr+/− mice where the clearance of apoB lipoproteins is not completely impaired leading only to moderate hypercholesterolemia. Secondly, SR-BI inhibition was not complete in our experimental conditions, only a 30–40 % increase in HDL levels was observed as compared with typical +125 % increases in SR-BI−/− animals. Since it has been shown that SR-BI deficiency has different effects in liver and in vascular wall, selective inhibition of SR-BI by ITX5061 in different tissues could also explain some of the differences38. Third, while most studies used Western-type diet, we used a cholic acid containing diet, which have different effects on atherogenesis. Although this diet has been extensively employed in atherosclerosis studies 39, it is also recognized to have significant limitations. Indeed, cholate that is present in the Paigen diet induces liver inflammation and fibrosis and these effects contribute to the peculiar atherogenicity of the diet 40. These concerns limit the usefulness of this model in extrapolating results to other settings such as in humans. Nonetheless, in the same dietary model marked overexpression of SR-BI was anti-atherogenic in conjunction with both HDL and VLDL/LDL lowering. Fourth, since ITX5061 is also a potent p38 MAPK inhibitor, it is possible that this off-target effect contributes, at least partially, to the reduction of atherosclerotic lesions in ITX5061-treated mice. However recent data do not support a role for p38 MAPK in regulating atherosclerotic lesion size. A p38 MAPK inhibitor decreased inflammation in atherosclerotic plaques but did not affect lesion size in apoE−/− mice infused with angiotensin II 41. Similarly, p38 MAPK deficiency in macrophages increased necrotic cores in advanced lesions in Western-type diet-fed apoE−/−mice but did not change lesion size 42. Therefore, although it seems unlikely that p38 MAPK inhibition contributed significantly to the reduction of atherosclerosis in our model, we cannot rule this out. In conclusion, inhibition of hepatic SR-BI in a setting that leads to increases in HDL without concomitant increases in VLDL and LDL cholesterol levels could potentially have an anti-atherogenic effect.
Supplementary Material
Acknowledgments
We thank Carrie Welch and Rong Li for determination of the lesions in aortic roots. David Masson was supported by a grant form the Philippe foundation.
Footnotes
Disclosures: Christopher J. Larson, Stephen G. Miller, Bernard D. King were formerly employed by Kemia, Inc.
References
- 1.Singh IM, Shishehbor MH, Ansell BJ. High-density lipoprotein as a therapeutic target: a systematic review. Jama. 2007;298:786–798. doi: 10.1001/jama.298.7.786. [DOI] [PubMed] [Google Scholar]
- 2.Linsel-Nitschke P, Tall AR. HDL as a target in the treatment of atherosclerotic cardiovascular disease. Nat Rev Drug Discov. 2005;4:193–205. doi: 10.1038/nrd1658. [DOI] [PubMed] [Google Scholar]
- 3.Acton S, Rigotti A, Landschulz KT, Xu S, Hobbs HH, Krieger M. Identification of scavenger receptor SR-BI as a high density lipoprotein receptor. Science. 1996;271:518–520. doi: 10.1126/science.271.5248.518. [DOI] [PubMed] [Google Scholar]
- 4.Rinninger F, Brundert M, Jackle S, Galle PR, Busch C, Izbicki JR, Rogiers X, Henne-Bruns D, Kremer B, Broelsch CE, Greten H. Selective uptake of high-density lipoprotein-associated cholesteryl esters by human hepatocytes in primary culture. Hepatology. 1994;19:1100–1114. [PubMed] [Google Scholar]
- 5.Rigotti A, Trigatti BL, Penman M, Rayburn H, Herz J, Krieger M. A targeted mutation in the murine gene encoding the high density lipoprotein (HDL) receptor scavenger receptor class B type I reveals its key role in HDL metabolism. Proc Natl Acad Sci U S A. 1997;94:12610–12615. doi: 10.1073/pnas.94.23.12610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rubin EM, Ishida BY, Clift SM, Krauss RM. Expression of human apolipoprotein A-I in transgenic mice results in reduced plasma levels of murine apolipoprotein A-I and the appearance of two new high density lipoprotein size subclasses. Proc Natl Acad Sci U S A. 1991;88:434–438. doi: 10.1073/pnas.88.2.434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Arai T, Wang N, Bezouevski M, Welch C, Tall AR. Decreased atherosclerosis in heterozygous low density lipoprotein receptor-deficient mice expressing the scavenger receptor BI transgene. J Biol Chem. 1999;274:2366–2371. doi: 10.1074/jbc.274.4.2366. [DOI] [PubMed] [Google Scholar]
- 8.Braun A, Trigatti BL, Post MJ, Sato K, Simons M, Edelberg JM, Rosenberg RD, Schrenzel M, Krieger M. Loss of SR-BI expression leads to the early onset of occlusive atherosclerotic coronary artery disease, spontaneous myocardial infarctions, severe cardiac dysfunction, and premature death in apolipoprotein E-deficient mice. Circ Res. 2002;90:270–276. doi: 10.1161/hh0302.104462. [DOI] [PubMed] [Google Scholar]
- 9.Kozarsky KF, Donahee MH, Glick JM, Krieger M, Rader DJ. Gene transfer and hepatic overexpression of the HDL receptor SR-BI reduces atherosclerosis in the cholesterol-fed LDL receptor-deficient mouse. Arterioscler Thromb Vasc Biol. 2000;20:721–727. doi: 10.1161/01.atv.20.3.721. [DOI] [PubMed] [Google Scholar]
- 10.Harder C, Lau P, Meng A, Whitman SC, McPherson R. Cholesteryl ester transfer protein (CETP) expression protects against diet induced atherosclerosis in SR-BI deficient mice. Arterioscler Thromb Vasc Biol. 2007;27:858–864. doi: 10.1161/01.ATV.0000259357.42089.dc. [DOI] [PubMed] [Google Scholar]
- 11.Tanigawa H, Billheimer JT, Tohyama J, Zhang Y, Rothblat G, Rader DJ. Expression of cholesteryl ester transfer protein in mice promotes macrophage reverse cholesterol transport. Circulation. 2007;116:1267–1273. doi: 10.1161/CIRCULATIONAHA.107.704254. [DOI] [PubMed] [Google Scholar]
- 12.Agellon LB, Walsh A, Hayek T, Moulin P, Jiang XC, Shelanski SA, Breslow JL, Tall AR. Reduced high density lipoprotein cholesterol in human cholesteryl ester transfer protein transgenic mice. J Biol Chem. 1991;266:10796–10801. [PubMed] [Google Scholar]
- 13.Desai MS, Mariscalco MM, Tawil A, Vallejo JG, Smith CW. Atherogenic diet-induced hepatitis is partially dependent on murine TLR4. J Leukoc Biol. 2008;83:1336–1344. doi: 10.1189/jlb.0607390. [DOI] [PubMed] [Google Scholar]
- 14.Nieland TJ, Shaw JT, Jaipuri FA, Maliga Z, Duffner JL, Koehler AN, Krieger M. Influence of HDL-cholesterol-elevating drugs on the in vitro activity of the HDL receptor SR-BI. J Lipid Res. 2007;48:1832–1845. doi: 10.1194/jlr.M700209-JLR200. [DOI] [PubMed] [Google Scholar]
- 15.Nieland TJ, Penman M, Dori L, Krieger M, Kirchhausen T. Discovery of chemical inhibitors of the selective transfer of lipids mediated by the HDL receptor SR-BI. Proc Natl Acad Sci U S A. 2002;99:15422–15427. doi: 10.1073/pnas.222421399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Huszar D, Varban ML, Rinninger F, Feeley R, Arai T, Fairchild-Huntress V, Donovan MJ, Tall AR. Increased LDL cholesterol and atherosclerosis in LDL receptor-deficient mice with attenuated expression of scavenger receptor B1. Arterioscler Thromb Vasc Biol. 2000;20:1068–1073. doi: 10.1161/01.atv.20.4.1068. [DOI] [PubMed] [Google Scholar]
- 17.Varban ML, Rinninger F, Wang N, Fairchild-Huntress V, Dunmore JH, Fang Q, Gosselin ML, Dixon KL, Deeds JD, Acton SL, Tall AR, Huszar D. Targeted mutation reveals a central role for SR-BI in hepatic selective uptake of high density lipoprotein cholesterol. Proc Natl Acad Sci U S A. 1998;95:4619–4624. doi: 10.1073/pnas.95.8.4619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Trigatti B, Rayburn H, Vinals M, Braun A, Miettinen H, Penman M, Hertz M, Schrenzel M, Amigo L, Rigotti A, Krieger M. Influence of the high density lipoprotein receptor SR-BI on reproductive and cardiovascular pathophysiology. Proc Natl Acad Sci U S A. 1999;96:9322–9327. doi: 10.1073/pnas.96.16.9322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Van Eck M, Hoekstra M, Out R, Bos IS, Kruijt JK, Hildebrand RB, Van Berkel TJ. Scavenger receptor BI facilitates the metabolism of VLDL lipoproteins in vivo. J Lipid Res. 2008;49:136–146. doi: 10.1194/jlr.M700355-JLR200. [DOI] [PubMed] [Google Scholar]
- 20.Calvo D, Gomez-Coronado D, Lasuncion MA, Vega MA. CLA-1 is an 85-kD plasma membrane glycoprotein that acts as a high-affinity receptor for both native (HDL, LDL, and VLDL) and modified (OxLDL and AcLDL) lipoproteins. Arterioscler Thromb Vasc Biol. 1997;17:2341–2349. doi: 10.1161/01.atv.17.11.2341. [DOI] [PubMed] [Google Scholar]
- 21.Morabia A, Ross BM, Costanza MC, Cayanis E, Flaherty MS, Alvin GB, Das K, James R, Yang AS, Evagrafov O, Gilliam TC. Population-based study of SR-BI genetic variation and lipid profile. Atherosclerosis. 2004;175:159–168. doi: 10.1016/j.atherosclerosis.2004.03.014. [DOI] [PubMed] [Google Scholar]
- 22.Hsu LA, Ko YL, Wu S, Teng MS, Peng TY, Chen CF, Chen CF, Lee YS. Association between a novel 11-base pair deletion mutation in the promoter region of the scavenger receptor class B type I gene and plasma HDL cholesterol levels in Taiwanese Chinese. Arterioscler Thromb Vasc Biol. 2003;23:1869–1874. doi: 10.1161/01.ATV.0000082525.84814.A9. [DOI] [PubMed] [Google Scholar]
- 23.Osgood D, Corella D, Demissie S, Cupples LA, Wilson PW, Meigs JB, Schaefer EJ, Coltell O, Ordovas JM. Genetic variation at the scavenger receptor class B type I gene locus determines plasma lipoprotein concentrations and particle size and interacts with type 2 diabetes: the framingham study. J Clin Endocrinol Metab. 2003;88:2869–2879. doi: 10.1210/jc.2002-021664. [DOI] [PubMed] [Google Scholar]
- 24.Acton S, Osgood D, Donoghue M, Corella D, Pocovi M, Cenarro A, Mozas P, Keilty J, Squazzo S, Woolf EA, Ordovas JM. Association of polymorphisms at the SR-BI gene locus with plasma lipid levels and body mass index in a white population. Arterioscler Thromb Vasc Biol. 1999;19:1734–1743. doi: 10.1161/01.atv.19.7.1734. [DOI] [PubMed] [Google Scholar]
- 25.West M, Greason E, Kolmakova A, Jahangiri A, Asztalos B, Pollin TI, Rodriguez A. Scavenger Receptor Class B Type I Protein as an Independent Predictor of Hdl Cholesterol Levels in Subjects with Hyperalphalipoproteinemia. J Clin Endocrinol Metab. 2009 doi: 10.1210/jc.2008-1223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Roberts CG, Shen H, Mitchell BD, Damcott CM, Shuldiner AR, Rodriguez A. Variants in scavenger receptor class B type I gene are associated with HDL cholesterol levels in younger women. Hum Hered. 2007;64:107–113. doi: 10.1159/000101962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Coppola GM, Damon RE, Eskesen JB, France DS, Paterniti JR., Jr Biological evaluation of 1-alkyl-3-phenylthioureas as orally active HDL-elevating agents. Bioorg Med Chem Lett. 2006;16:113–117. doi: 10.1016/j.bmcl.2005.09.034. [DOI] [PubMed] [Google Scholar]
- 28.Scriabine A. HDL cholesterol: metabolic pathways and drug developments. Cardiovasc Drug Rev; Fifth Annual International Conference of the Knowledge Foundation; March 8–9, 2004; Cambridge, MA, USA. 2004. pp. 147–153. [DOI] [PubMed] [Google Scholar]
- 29.Trigatti BL, Krieger M, Rigotti A. Influence of the HDL receptor SR-BI on lipoprotein metabolism and atherosclerosis. Arterioscler Thromb Vasc Biol. 2003;23:1732–1738. doi: 10.1161/01.ATV.0000091363.28501.84. [DOI] [PubMed] [Google Scholar]
- 30.Covey SD, Krieger M, Wang W, Penman M, Trigatti BL. Scavenger receptor class B type I-mediated protection against atherosclerosis in LDL receptor-negative mice involves its expression in bone marrow-derived cells. Arterioscler Thromb Vasc Biol. 2003;23:1589–1594. doi: 10.1161/01.ATV.0000083343.19940.A0. [DOI] [PubMed] [Google Scholar]
- 31.Ueda Y, Gong E, Royer L, Cooper PN, Francone OL, Rubin EM. Relationship between expression levels and atherogenesis in scavenger receptor class B, type I transgenics. J Biol Chem. 2000;275:20368–20373. doi: 10.1074/jbc.M000730200. [DOI] [PubMed] [Google Scholar]
- 32.Huby T, Doucet C, Dachet C, Ouzilleau B, Ueda Y, Afzal V, Rubin E, Chapman MJ, Lesnik P. Knockdown expression and hepatic deficiency reveal an atheroprotective role for SR-BI in liver and peripheral tissues. J Clin Invest. 2006;116:2767–2776. doi: 10.1172/JCI26893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Nieland TJ, Shaw JT, Jaipuri FA, Duffner JL, Koehler AN, Banakos S, Zannis VI, Kirchhausen T, Krieger M. Identification of the molecular target of small molecule inhibitors of HDL receptor SR-BI activity. Biochemistry. 2008;47:460–472. doi: 10.1021/bi701277x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Nishizawa T, Kitayama K, Wakabayashi K, Yamada M, Uchiyama M, Abe K, Ubukata N, Inaba T, Oda T, Amemiya Y. A novel compound, R-138329, increases plasma HDL cholesterol via inhibition of scavenger receptor BI-mediated selective lipid uptake. Atherosclerosis. 2007;194:300–308. doi: 10.1016/j.atherosclerosis.2006.10.025. [DOI] [PubMed] [Google Scholar]
- 35.Kitayama K, Nishizawa T, Abe K, Wakabayashi K, Oda T, Inaba T, Amemiya Y. Blockade of scavenger receptor class B type I raises high density lipoprotein cholesterol levels but exacerbates atherosclerotic lesion formation in apolipoprotein E deficient mice. J Pharm Pharmacol. 2006;58:1629–1638. doi: 10.1211/jpp.58.12.0010. [DOI] [PubMed] [Google Scholar]
- 36.Zhang Y, Da Silva JR, Reilly M, Billheimer JT, Rothblat GH, Rader DJ. Hepatic expression of scavenger receptor class B type I (SR-BI) is a positive regulator of macrophage reverse cholesterol transport in vivo. J Clin Invest. 2005;115:2870–2874. doi: 10.1172/JCI25327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Plump AS, Masucci-Magoulas L, Bruce C, Bisgaier CL, Breslow JL, Tall AR. Increased atherosclerosis in ApoE and LDL receptor gene knock-out mice as a result of human cholesteryl ester transfer protein transgene expression. Arterioscler Thromb Vasc Biol. 1999;19:1105–1110. doi: 10.1161/01.atv.19.4.1105. [DOI] [PubMed] [Google Scholar]
- 38.Van Eck M, Twisk J, Hoekstra M, Van Rij BT, Van der Lans CA, Bos IS, Kruijt JK, Kuipers F, Van Berkel TJ. Differential effects of scavenger receptor BI deficiency on lipid metabolism in cells of the arterial wall and in the liver. J Biol Chem. 2003;278:23699–23705. doi: 10.1074/jbc.M211233200. [DOI] [PubMed] [Google Scholar]
- 39.Paigen B, Morrow A, Brandon C, Mitchell D, Holmes P. Variation in susceptibility to atherosclerosis among inbred strains of mice. Atherosclerosis. 1985;57:65–73. doi: 10.1016/0021-9150(85)90138-8. [DOI] [PubMed] [Google Scholar]
- 40.Vergnes L, Phan J, Strauss M, Tafuri S, Reue K. Cholesterol and cholate components of an atherogenic diet induce distinct stages of hepatic inflammatory gene expression. J Biol Chem. 2003;278:42774–42784. doi: 10.1074/jbc.M306022200. [DOI] [PubMed] [Google Scholar]
- 41.Morris JB, Olzinski AR, Bernard RE, Aravindhan K, Mirabile RC, Boyce R, Willette RN, Jucker BM. p38 MAPK inhibition reduces aortic ultrasmall superparamagnetic iron oxide uptake in a mouse model of atherosclerosis: MRI assessment. Arterioscler Thromb Vasc Biol. 2008;28:265–271. doi: 10.1161/ATVBAHA.107.151175. [DOI] [PubMed] [Google Scholar]
- 42.Seimon TA, Wang Y, Han S, Senokuchi T, Schrijvers DM, Kuriakose G, Tall AR, Tabas IA. Macrophage deficiency of p38alpha MAPK promotes apoptosis and plaque necrosis in advanced atherosclerotic lesions in mice. J Clin Invest. 2009;119:886–898. doi: 10.1172/JCI37262. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.