Abstract
NKT cells are positively selected on cortical thymocytes expressing the non-classical MHC class I CD1d molecules. However, it is less clear how NKT cells are negatively selected in the thymus. In this study, we investigated the role of MHC class II expression in NKT cell development. Transgenic mice expressing MHC class II on thymocytes and peripheral T cells had a marked reduction in invariant NKT (iNKT) cells. Reduced numbers of iNKT cells correlated with the absence of in vivo production of cytokines in response to the iNKT cell agonist α–galactosylceramide. Using mixed bone marrow chimeras, we found that MHC class II expressing thymocytes suppressed the development of iNKT cells in trans in a CD4-dependent manner. Our observations have significant implications for human iNKT cell development since human thymocytes express MHC class II, which can lead to inefficient selection of iNKT cells.
Keywords: iNKT cells, MHC class II, transgeneic mice, CD1d, thymocyte development
INTRODUCTION
NKT cells are a specialized subset of T cells that co-express NK cell markers, such as NK1.1. They originate in the thymus and are positively selected by CD1d on cortical DP thymocytes1,2, 3. There are at least two sub-populations of NKT cells based on their TCR diversity1. The major subset is called invariant NKT (iNKT) cells because their TCR repertoire is very limited with the majority of them carrying the invariant Vα14-Jα18 αchain paired mostly with one of the three Vβ chains (Vβ8.2, Vβ7, and Vβ2) in mice. The other subset has a diverse TCR repertoire and represents the non-invariant NKT (non-iNKT) cells.
iNKT cells can be negatively selected when iNKT TCR receives too strong signal2–4. The addition of iNKT cell agonist α-galactosylceramide (α-GalCer) to fetal thymic organ culture leads to iNKT cell deletion5, 6 and the transgenic expression of CD8α and CD8β chains in T cells also results in a reduction of iNKT cells7. Transgenic mice overexpressing the NK activating receptor LY49D have reduced iNKT cell development8. Furthermore, over-expression of CD1d in dendritic cells decreased the number of iNKT cells6, 9.
Unlike murine NKT cells, the number of Vα24+ iNKT cells in humans is highly variable among individuals and they represent only a small portion of total NKT cells, whereas non-iNKT cells are the major population1, 10–14. Human thymocytes can express not only CD1 but also MHC class II15. However, it is not known whether MHC class II on thymocytes plays a role during NKT cell development because murine thymocytes do not express MHC class II. In this study, we showed that MHC class II-expressing thymocytes suppress the development of iNKT cells partly due to the interaction of MHC class II with CD4.
RESULTS AND DISCUSSION
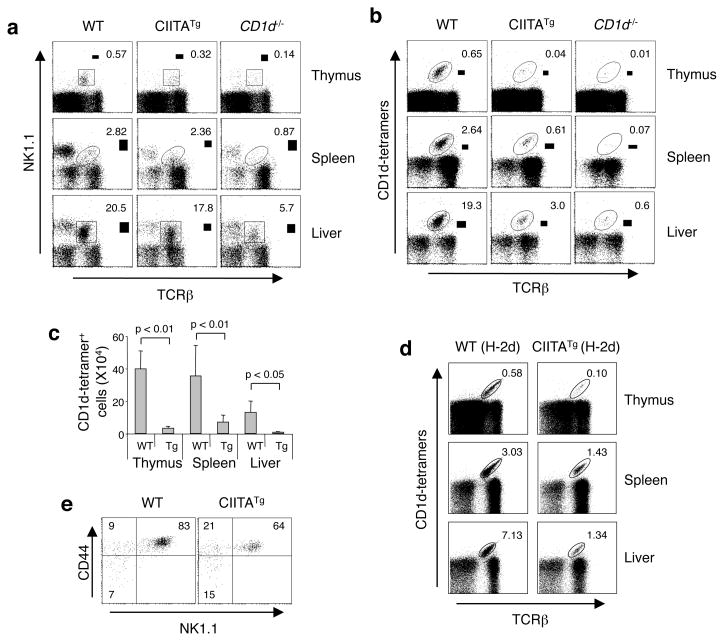
To study the effect of MHC class II expression on NKT cell development, we utilized mice that express MHC class II in thymocytes and peripheral T cells by introducing the MHC class II transactivator (CIITA) gene as a transgene (CIITATg)16. CIITATg mice had reduced thymic NK1.1+TCRβ+ NKT cells but not in the periphery (Figure 1a). We next examined iNKT cells using CD1d tetramers loaded with α–GalCer (hereafter referred to as tetramers) that specifically identify iNKT cells. As shown in Figure 1b and 1c, CIITATg mice exhibited a marked reduction in iNKT cells both in the thymus and the peripheral tissue, indicating CIITA expression in T cells led to a selective failure of iNKT development and a corresponding expansion of non-iNKT cells. The reduction of iNKT cell number was MHC haplotype independent (Figure 1d). However, unlike CD1d−/− mice that had no iNKT cells, residual iNKT cells were present in CIITATg mice (Figure 1b). These residual iNKT cells in the CIITATg thymus seemed to have undergone proper developmental stages and become mature iNKT cells as defined by CD44 and NK1.1 expression, albeit a slight increase and decrease in percentages of immature and the most mature iNKT cells, respectively (Fig. 1e). The decrease in iNKT cell number in the periphery resulted in very little circulating IL-4 or IFN-γ upon in vivo activation (Supplementary Figure 1a). The CD1d levels on CIITATg DP thymocytes (Supplementary Figure 1b) were comparable to WT cells.
Figure 1.
Reduction of iNKT cells in CIITATg mice. Expression of TCRβ and NK1.1 markers (a) or TCRβ and tetramer (b) on indicated cell types from WT (CIITATg littermates), CIITATg, and CD1d−/− mice. The number in the dot plots represents the percentage of NK1.1+TCRβ+ NKT cells (a) or iNKT cells (b). Mice used were 10–11 weeks old and similar results were obtained from at least four mice per group. (c) Absolute numbers of iNKT cells. Values are mean ± SD of iNKT cells present in thymi (n=10), spleens (n=6), and livers (n=4). Mice were 2–3 months old. (d) Representative iNKT cell staining of 3 pairs of WT (CIITATg littermates) and CIITATg on the H-2d background. (e) Analysis of NK1.1 and CD44 expression on thymic iNKT cells. Tetramer+HSAlo cells were gated and percentages of cells in each quadrant are shown. Results are representative of four pairs of WT and CIITATg littermates. Mice were 10 weeks old.
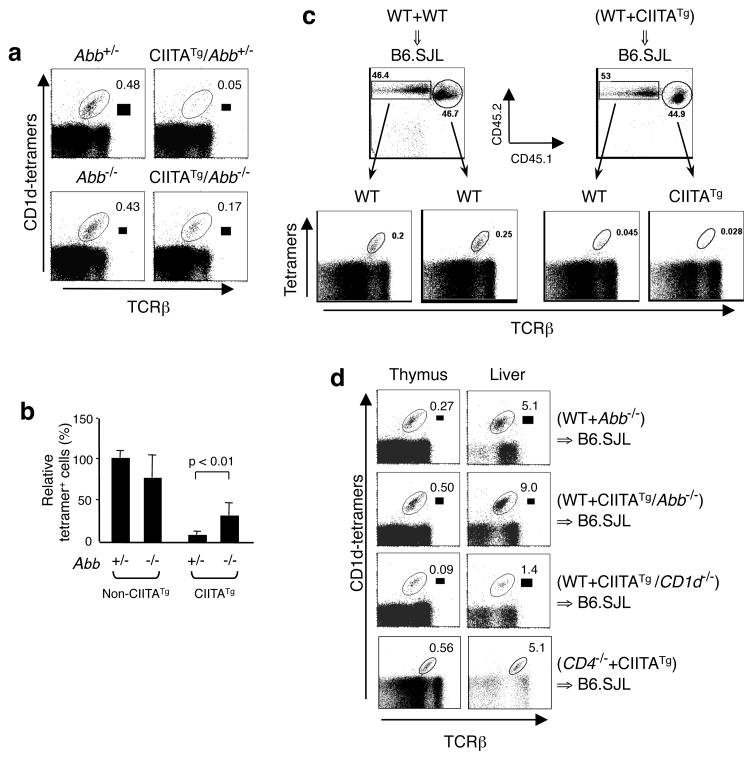
Because CIITA can turn on the expression of multiple genes involved in MHC class II antigen presentation, such as the invariant chain (Ii)17, we wanted to determine whether the expression of MHC class II or a gene product regulated by CIITA is responsible for the observed inefficient iNKT cell development. To do this, we generated CIITATg mice lacking MHC class II by crossing them with MHC class II deficient Aβ −/− mice (CIITATg /Aβ−/−). In these mice, developing thymocytes express the CIITA transgene but not MHC class II. The absence of MHC class II in CIITATg/Abb−/− mice significantly improved the generation of iNKT cells (Figure 2a and 2b), suggesting that MHC class II participates in development of iNKT cells. The incomplete restoration implies the possible involvement of other CIITA-regulated components in iNKT cell development. One putative candidate is Ii that is known to associate with CD1d in endosomes and enhances CD1d antigen presentation of dendritic cells and B cells to iNKT cells18. Further study is required to determine whether Ii plays a suppressive role in iNKT cell development.
Figure 2.
MHC class II inhibits iNKT cell development. (a) MHC class II expression in thymocytes is necessary for the inhibition of iNKT cell generation. Total thymocytes from the indicated mice were analyzed for TCRβ and tetramer reactivity. WT and CIITATg mice on the Abb+/− and Abb−/− background were littermates. Results are representative of at least 4 experiments. (b) Relative percentages of thymic iNKT cells in at least 8 mice of Abb+/−, CIITATg/Abb+/−, CIITATg /Abb−/−, or Abb−/−. The value of the percentage of iNKT cells in WT mice was set at 100%. Mice were 2–3 months old. (c) Suppression of iNKT cell development by MHC class II-expressing thymoyctes in trans. BM cells from WT (CD45.2) and WT (CIITATg littermates; CD45.1/CD45.2) or WT (CD45.2) together with CIITATg (CD45.1/CD45.2) were co-injected into B6.SJL (CD45.1) recipients. Shown at the top is the composition of total thymocytes in reconstituted mice. TCRβ versus tetramer profiles of thymocytes are shown at the bottom. Chimeras were analyzed 10–12 weeks post BM transplantation. (d) MHC class II and CD4 expression is required for the disappearance of iNKT cells in mixed BM chimeric mice as indicated. BM were prepared from WT (CD45.1/2), Abb−/− (CD45.2), CIITATg/Abb−/− (CD45.2), CIITATg/CD1d−/− (CD45.2), CIITATg (CD45.1/2), and CD4−/− (CD45.2) mice and transferred to B6.SJL mice. The number in the dot plots represents the percentage of iNKT cells developed from WT BM cells except the bottom group that shows CD4−/− iNKT cells. Chimeras were analyzed 2–3 months post BM transplantation.
To test whether MHC class II-expressing thymocytes fail to develop into iNKT cells in a cell autonomous manner, we constructed mixed bone marrow (BM) chimeric mice. Different CD45 markers were used to distinguish cells derived from the two different BM sources and the host. In WT+WT→B6.SJL mice, each WT BM population generated a sizable iNKT cell population (Figure 2c, left group; Supplementary Figure 2). However, in the presence of CIITATg BM, WT BM cells failed to generate a comparable proportion of iNKT cells, implying that MHC class II expressing thymocytes suppress neighboring thymocytes to progress to iNKT cells in trans (Figure 2c, right group; Supplementary Figure 2).
To further substantiate the role of MHC class II in iNKT cell development, we co-transferred WT BM cells together with CIITATg/Abb−/− BM to B6.SJL recipients. If MHC class II expression itself is responsible for deleting thymocytes, iNKT cell development should be restored in WT+CIITATg/Abb−/− →B6.SJL mice. Indeed, iNKT cells originated from WT BM were present at a normal level (compare the 2nd row in Figure 2d to the right group in Figure 2c; Supplementary Figure 2). In the control WT+Abb−/−→B6.SJL chimeras, iNKT cells were also generated (Figure 2d, top row; Supplementary Figure 2). Previously, over-expressing CD1d on DC that also express MHC class II lead to negative selection of iNKT cells6, 9. Therefore, we asked whether co-expression of CD1d together with MHC class II on the same cells is necessary to delete iNKT cells. iNKT cells derived from WT BM were dramatically decreased in the presence of CIITATg/CD1d−/− thymocytes indicating un unlikely role of CD1d in deletion of iNKT cells (Figure 2d, 3rd row; Supplementary Figure 2). Lastly, MHC class II on the thymocyte surface might function as a co-receptor for CD4 molecules, enhancing the avidity of TCR on NKT progenitors that are being selected. If so, the strength of signaling could reach beyond the threshold for survival of cells. Indeed, in the CD4−/−+CIITATg→B6.SJL chimeras, CD4−/− thymocytes were developed to iNKT cells normally in the presence of MHC class II expressing thymocytes (Figure 2d, bottom row; Supplementary Figure 2), suggesting that the interaction of MHC class II with CD4 is at least partly responsible for the disappearance of iNKT cells. Thus, in addition to its role in iNKT cell activation19, 20, CD4 seems to participate in the regulation of iNKT cell development.
Taken all together, we have demonstrated, for the first time, that MHC class II plays an important role during iNKT cell development. Similar to CIITATg mice, humans have much fewer iNKT cells than non-iNKT cells compared to mice. Perhaps, this is related to the fact that human fetal and neonatal thymocytes express MHC class II, which likely plays an inhibitory role in iNKT cell generation. The final tuning of the strength of the signal delivered by CD1d and/or MHC class II may be critical for survival or death of developing thymocytes to progress toward iNKT cells.
METHODS
Mice
Mice carrying the human type III CIITA transgene (CIITATg), CIITATg mice on the MHC class II deficient Abb−/− background (CIITATg/Abb−/−), and CD1d−/− on the C57BL/6 background were described previously15, 16, 21. CIITATg and CD1d−/− mice were backcrossed with C57BL/6 (B6) mice at least 6 to 8 times, respectively. CIITATg mice on the Balb/c background were backcrossed 6 times. CD4−/− mice and B6 mice were purchased from the Jackson Laboratory (Bar Harbor, ME) and C57BL/6.SJL (B6.SJL) mice from Taconic (Germantown, NY). Non-CIITATg littermates from heterozygous CIITATg breeding were used as wild type (WT) controls. CIITATg and WT mice were also bred to carry CD45.1 and CD45.2 congenic markers. All mice were housed in the animal facility at the Indiana University School of Medicine (IUSM) or the University of Michigan Medical School under SPF conditions and used at 6–12 weeks of age. All animal experiments were performed under protocols approved by the institutions.
Mixed bone marrow chimeras
Preparation of mature lymphocyte-depleted BM cells and BM transfers were done as described previously15. To generate mixed BM chimeras, BM cells from two different types of donor mice were mixed at a ratio of 1:1 and each lethally-irradiated recipient mouse (950 rad) was infused with 2–5×105 cells via tail vein injection.
Flow cytometry
All antibodies used for flow cytometry were purchased from BD Biosciences (San Diego, CA). Cells were pre-incubated with the anti-FcγR mAb 2.4G2 to block non-specific antibody binding before stained with the following FITC-, PE-, PerCP-, CyChrome-, APC-or biotin-conjugated antibodies: TCRβ (H57), CD4 (L3T4), B220 (RA3-6B2), NK1.1 (PK136), CD1d (1B1), CD45.1 (A20), CD45.2 (104). Flourochome-conjugated streptavidin was used to visualize staining by biotinylated primary antibodies. APC-conjugated CD1d-α– GalCer tetramers were generously provided by Dr. Albert Bendalac from University of Chicago and the NIH Tetramer Facility. Events were acquired on a FACSCalibur or FACSCanto (BD Biosciences) flow cytometer and the data were analyzed using the CELLQuest Pro or FlowJo software.
In vivo Cytokine Production Assay
Mice were injected via i.v with 2 μg α– GalCer22 or vehicle in 100 μl of PBS. After 2 hours, mice were sacrificed and serum IL-4 and IFN-γ were detected using ELISA kits (BD Biosciences).
Statistic analysis
The nonparametric Wilcoxon Rank Sum Test was performed to calculate statistical significance. P values less than 0.05 were considered statistically significant.
Supplementary Material
Acknowledgments
We thank Dr. Barry Katz (Indiana University School of Medicine) for helping us with the statistical analyses. This study was supported in part by the NIH grant AI66642.
References
- 1.Bendelac A, Savage PB, Teyton L. The biology of NKT cells. Annu Rev Immunol. 2007;25:297–336. doi: 10.1146/annurev.immunol.25.022106.141711. [DOI] [PubMed] [Google Scholar]
- 2.Kronenberg M. Toward an understanding of NKT cell biology: progress and paradoxes. Annu Rev Immunol. 2005;23:877–900. doi: 10.1146/annurev.immunol.23.021704.115742. [DOI] [PubMed] [Google Scholar]
- 3.Godfrey DI, Berzins SP. Control points in NKT-cell development. Nat Rev Immunol. 2007;7:505–518. doi: 10.1038/nri2116. [DOI] [PubMed] [Google Scholar]
- 4.Gapin L, Matsuda JL, Surh CD, Kronenberg M. NKT cells derive from double-positive thymocytes that are positively selected by CD1d. Nat Immunol. 2001;2:971–978. doi: 10.1038/ni710. [DOI] [PubMed] [Google Scholar]
- 5.Pellicci DG, Uldrich AP, Kyparissoudis K, Crowe NY, Brooks AG, Hammond KJ, Sidobre S, et al. Intrathymic NKT cell development is blocked by the presence of alpha-galactosylceramide. Eur J Immunol. 2003;33:1816–1823. doi: 10.1002/eji.200323894. [DOI] [PubMed] [Google Scholar]
- 6.Chun T, Page MJ, Gapin L, Matsuda JL, Xu H, Nguyen H, Kang HS, et al. CD1d-expressing dendritic cells but not thymic epithelial cells can mediate negative selection of NKT cells. J Exp Med. 2003;197:907–918. doi: 10.1084/jem.20021366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bendelac A, Killeen N, Littman DR, Schwartz RH. A subset of CD4+ thymocytes selected by MHC class I molecules. Science. 1994;263:1774–1778. doi: 10.1126/science.7907820. [DOI] [PubMed] [Google Scholar]
- 8.Voyle RB, Beermann F, Lees RK, Schumann J, Zimmer J, Held W, MacDonald HR. Ligand-dependent inhibition of CD1d-restricted NKT cell development in mice transgenic for the activating receptor Ly49D. J Exp Med. 2003;197:919–925. doi: 10.1084/jem.20021615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Schumann J, Pittoni P, Tonti E, Macdonald HR, Dellabona P, Casorati G. Targeted expression of human CD1d in transgenic mice reveals independent roles for thymocytes and thymic APCs in positive and negative selection of Valpha14i NKT cells. J Immunol. 2005;175:7303–7310. doi: 10.4049/jimmunol.175.11.7303. [DOI] [PubMed] [Google Scholar]
- 10.Doherty DG, Norris S, Madrigal-Estebas L, McEntee G, Traynor O, Hegarty JE, O’Farrelly C. The human liver contains multiple populations of NK cells, T cells, and CD3+CD56+ natural T cells with distinct cytotoxic activities and Th1, Th2, and Th0 cytokine secretion patterns. J Immunol. 1999;163:2314–2321. [PubMed] [Google Scholar]
- 11.Karadimitris A, Gadola S, Altamirano M, Brown D, Woolfson A, Klenerman P, Chen JL, et al. Human CD1d-glycolipid tetramers generated by in vitro oxidative refolding chromatography. Proc Natl Acad Sci USA. 2001;98:3294–3298. doi: 10.1073/pnas.051604498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lee PT, Benlagha K, Teyton L, Bendelac A. Distinct functional lineages of human V(alpha)24 natural killer T cells. J Exp Med. 2002;195:637–641. doi: 10.1084/jem.20011908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kita H, Naidenko OV, Kronenberg M, Ansari AA, Rogers P, He XS, Koning F, et al. Quantitation and phenotypic analysis of natural killer T cells in primary biliary cirrhosis using a human CD1d tetramer. Gastroenterology. 2002;123:1031–1043. doi: 10.1053/gast.2002.36020. [DOI] [PubMed] [Google Scholar]
- 14.Gumperz JE, Miyake S, Yamamura T, Brenner MB. Functionally distinct subsets of CD1d-restricted natural killer T cells revealed by CD1d tetramer staining. J Exp Med. 2002;195:625–636. doi: 10.1084/jem.20011786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Li W, Kim MG, Gourley TS, McCarthy BP, Sant’angelo DB, Chang CH. An Alternate Pathway for CD4 T Cell Development: Thymocyte-Expressed MHC Class II Selects a Distinct T Cell Population. Immunity. 2005;23:375–386. doi: 10.1016/j.immuni.2005.09.002. [DOI] [PubMed] [Google Scholar]
- 16.Patel DR, Li W, Park JS, Sofi MH, Gourley TS, Hangoc G, Kaplan MH, et al. Constitutive expression of CIITA directs CD4 T cells to produce Th2 cytokines in the thymus. Cell Immunol. 2005;233:30–40. doi: 10.1016/j.cellimm.2005.03.006. [DOI] [PubMed] [Google Scholar]
- 17.Chang CH, Flavell RA. Class II transactivator regulates the expression of multiple genes involved in antigen presentation. J Exp Med. 1995;181:765–767. doi: 10.1084/jem.181.2.765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Jayawardena-Wolf J, Benlagha K, Chiu YH, Mehr R, Bendelac A. CD1d endosomal trafficking is independently regulated by an intrinsic CD1d-encoded tyrosine motif and by the invariant chain. Immunity. 2001;15:897–908. doi: 10.1016/s1074-7613(01)00240-0. [DOI] [PubMed] [Google Scholar]
- 19.Thedrez A, de Lalla C, Allain S, Zaccagnino L, Sidobre S, Garavaglia C, Borsellino G, et al. CD4 engagement by CD1d potentiates activation of CD4+ invariant NKT cells. Blood. 2007;110:251–258. doi: 10.1182/blood-2007-01-066217. [DOI] [PubMed] [Google Scholar]
- 20.Chen X, Wang X, Besra GS, Gumperz JE. Modulation of CD1d-restricted NKT cell responses by CD4. J Leukoc Biol. 2007;82:1455–1465. doi: 10.1189/jlb.0307163. [DOI] [PubMed] [Google Scholar]
- 21.Mendiratta SK, Martin WD, Hong S, Boesteanu A, Joyce S, Van Kaer L. CD1d1 mutant mice are deficient in natural T cells that promptly produce IL-4. Immunity. 1997;6:469–477. doi: 10.1016/s1074-7613(00)80290-3. [DOI] [PubMed] [Google Scholar]
- 22.Du W, Kulkarni SS, Gervay-Hague J. Efficient, one-pot syntheses of biologically active alpha-linked glycolipids. Chem Commun (Camb) 2007;23:2336–2338. doi: 10.1039/b702551c. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.