Abstract
Historically, drug research targeted to pain treatment has focused on trying to prevent the propagation of action potentials in the periphery from reaching the brain rather than pinpointing the molecular basis underlying the initial detection of the nociceptive stimulus: the receptor itself. This has now changed, given that many receptors of nociceptive stimuli have been identified and/or cloned. Transient Receptor Potential (TRP) channels have been implicated in several physiological processes such as mechanical, chemical and thermal stimuli detection. Ten years after the cloning of TRPV1, compelling data has been gathered on the role of this channel in inflammatory and neuropathic states. TRPV1 activation in nociceptive neurons, where it is normally expressed, triggers the release of neuropeptides and transmitters resulting in the generation of action potentials that will be sent to higher CNS areas where they will often be perceived as pain. Its activation also will evoke the peripheral release of pro-inflammatory compounds that may sensitize other neurons to physical, thermal or chemical stimuli. For these reasons as well as because its continuous activation causes analgesia, TRPV1 has become a viable drug target for clinical use in the management of pain. This review will provide a general picture of the physiological and pathophysiological roles of the TRPV1 channel and of its structural, pharmacological and biophysical properties. Finally, it will provide the reader with an overall view of the status of the discovery of potential therapeutic agents for the management of chronic and neuropathic pain.
Keywords: ion channels, TRPV1, pain, disease, analgesia, neuropatic inflammation
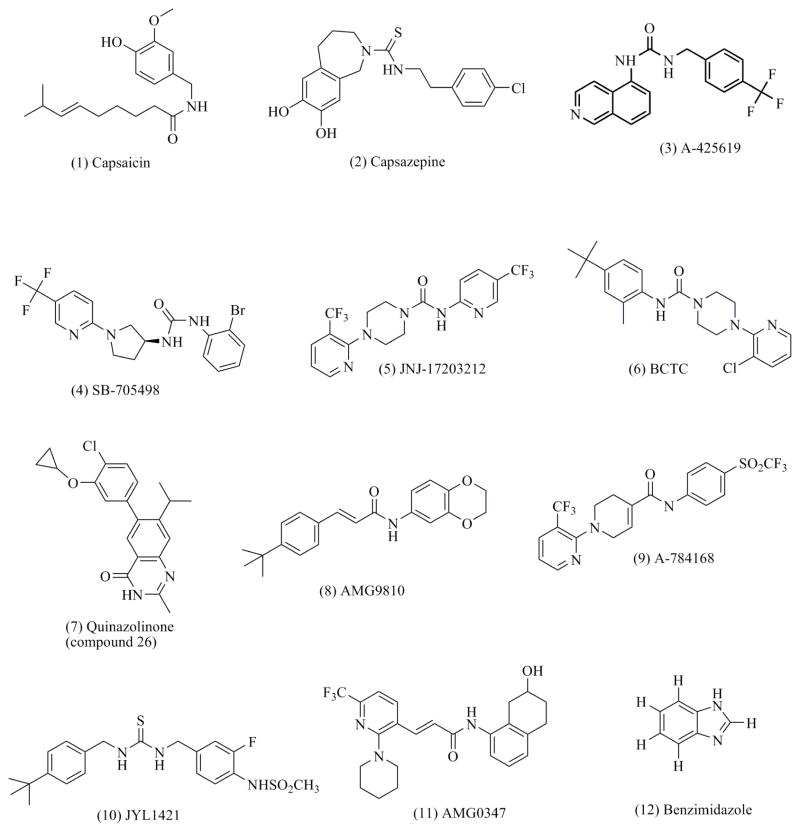
TRP ion channels were first described in 1989 in Drosophila melanogaster [1]. However, it was not until 1997 when TRPV1, one of the members of the family of TRP channels, was cloned and shown to respond to a variety of stimuli such as capsaicin (Fig. (3), compound (1) ), the primary pungent ingredient of hot chilli peppers, to low pH and high temperatures [2–4]. Since then, the field of ion channel-study has witnessed a surge in research relative to the physiology of TRP channels. Subsequently it has been found that TRP channels are responsive to mechanical, thermal, chemical (ie. acid, lipids), osmotic pressure gradients, and many other stimuli coming from the extra and intracellular milieu [5–10].
Figure 3.
Chemical structures of capsaicin and selected TRPV1 antagonists. Antagonists such as A-425619 (1-isoquinolin-5-yl-3-(4-trifluoromethyl-benzyl)-urea) [175,241,242], SB-705498 (N-(2-bromophenyl)-N′-[((R)-1-(5-trifluromethyl-2-pyridyl)pyrrolidin-3-yl)]urea)[178,243], BCTC (N-(4-tertiarybutylphenyl)-4-(3-cholorphyridin-2-yl)tetrahydropryazine-1(2H)-carbox-amide)[244], quinazolinone compound 26 [245] and AMG 9810 ([(E)-3-(4-t-Butylphenyl)-N-(2,3-dihydrobenzo[b] dioxin-6-yl)acrylamide) [246] (Fig. (3), compounds (3), (4), (6), (7) and (8), respectively) have implicated TRPV1 as an important mediator of nociceptive responses to mechanical stimuli under inflammatory conditions [17].
Presently, the TRP channel family contains seven divisions: TRPC, TRPV, TRPM, TRPA, TRPN, TRPP, and TRPML [6, 10–12]. The TRPV (‘Vanilloid’) subfamily presently comprises six members (TRPV1–TRPV6). The most studied member of this subfamily is TRPV1. TRPV1 has been implicated in a wide variety of cellular and physiological processes, including noxious physical and chemical stimuli detection, making it a promising target for pain-relieving drugs. For example, neurons containing the TRPV1 channel can be rendered insensitive to further painful stimuli through receptor desensitization in response to some agonists, which can result in a generalized lack of responsiveness of this protein to further noxious stimuli [13–15].
During the last several years, potent orally bio-available TRPV1 antagonists have been identified and synthesized. These discoveries have further authenticated TRPV1 as a target for disease conditions, including inflammatory pain. To date, TRPV1 antagonists have been reported to partially reverse inflammation as well as skin incision-induced thermal hyperalgesia. Several reviews have discussed the topic of pain treatment through the TRPV1 channel [16, 17, 18]; however this review will give a more generalized picture of the TRPV1, from its biophysical properties to its physiological roles, in order to establish that a great deal of knowledge regarding this channel protein is still missing.
Moreover, to understand how painful processes are mediated through the activation of TRPV1, we consider it essential to provide information concerning the basic characteristics of this receptor as well as many of the regulators of its activity. Thus, we will focus on the current status of TRPV1 research and its implications for the physiology of nociception and pain, and on the advances made in the field of TRPV1-targeted pain-relieving drugs.
1) Tissue distribution, general properties and splice variants of TRPV1
TRPV1 is expressed in all sensory ganglia (DRG, TG, Vagal) and in small sensory C-and Aδ fibers, which may contain various neuropeptides including substance P (SP) and/or Calcitonin Gene-Related Peptide (CGRP) [2, 19–27]. TRPV1 is also found at the Central Nervous System (CNS) and in non-neuronal tissues such as keratinocytes, mast cells, hair follicles, smooth muscle, bladder, liver, kidney, spleen and lungs [28–37].
The TRPV1 channel is predicted to have six transmembrane domains and a short, pore-forming hydrophobic stretch between the fifth and sixth transmembrane domains. It is activated by capsaicin[2], noxious heat (>43°C), low pH (5.2) [2, 3], voltage [38, 39], various lipids [4, 19, 40–45] and other pungent compounds such as zingerone, piperine and those found in garlic and onion, such as allicin[46, 47].
TRP channels contain six transmembrane domains that assemble as homo- or hetero-tetramers in order to form cation selective channels[41, 48] Fig. (1). Similar to other six-transmembrane domain channels, TRPV1 probably forms a tetrameric quaternary structure [49, 50], where each subunit contributes to the ion-conducting pore and the selectivity filter. Although all known TRP channels are cation selective, their permeability for different monovalent and divalent cations varies among their subtypes[5, 51, 52].
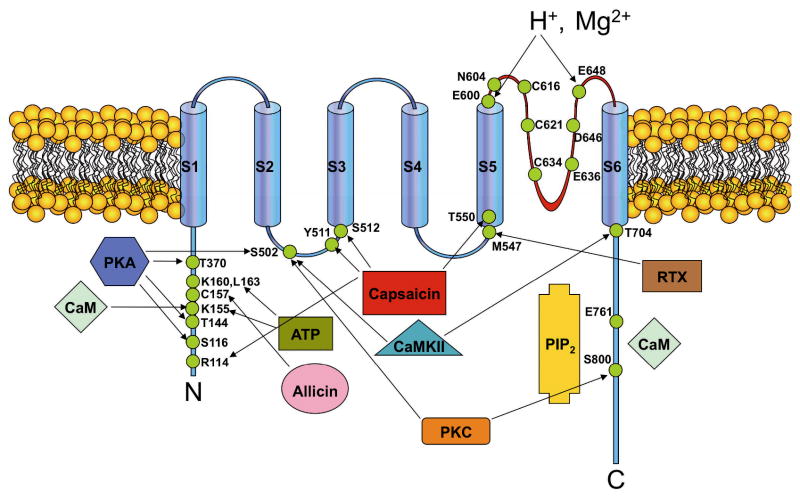
Figure 1. TRPV1 residues involved in ligand- and modulator interactions.
Some of the residues responsible for the actions of some TRPV1 agonists or modulators have been identified. TRPV1 has multiple phosphorylation sites. cAMP-dependent protein kinase (PKA) directly phosphorylates the TRPV1 channel at several sites. Residues Ser 116 and Thr 370 are phosphorylated by PKA and have been implicated in desensitization, while residues Thr 144, Thr 370, and Ser 502 have been implicated in sensitization of heat-evoked TRPV1 responses when phosphorylated by PKA. Ser 502 is also the target for protein kinase C (PKC) and calcium-calmodulin dependent kinase II (CaMKII). PKC can phosphorylate the channel at a second residue, Ser 800, while CaMKII phosphorylates Thr 704. The region formed by amino acids 777–820 and both charged aminoacids R701 and K710 of TRPV1 are responsible for some of the actions of PIP2 on the channel. Cysteine 157, at the N-terminus, reacts to cysteine-modifying agents such as allicin, a pungent compound present in garlic and onion, which cause TRPV1 channel activation. Several residues responsible for the actions of capsaicin and other vanilloids have been identified. Glu761 in the C-terminus and Arg 114 at the N-terminus have been postulated as agonist recognition sites. Ser 512 is important for capsaicin-mediated activation of the channel, while Thr 550 and Tyr 511 are necessary for maintaining capsaicin sensitivity. Met 547 is responsible for RXT binding and sensitivity, and is also involved in some of the vanilloid’s actions. Glu 600 serves as an important regulator site for proton potentiation of TRPV1 activity, while Glu 648 seems to be involved in direct proton-evoked activation of TRPV1. Both Glu 648 and Glu 600 are responsible for the Gd3+ activating effects on the channel. Additionally Glu648, together with Asp646, are responsible for polyamine actions on TRPV1. The three pore-cysteines, but especially C621, are important for TRPV1 modulation by extracellular reducing agents. Two calmodulin binding sites in the TRPV1 channel have been identified, one in the N-terminus and one in the C-terminus of the protein. Calmodulin bound to calcium in the N-terminus of the channel causes desensitization. Glu 636 is a key molecular determinant of the TRPV1 pore region, and is responsible for TRPV1 channel block by ruthenium red. Asp 604 is a site for N-glycosylation.
Ion permeation is controlled by allosteric interactions among the subunits and by an activation gate which, as for voltage-gated potassium channels, is most probably located in the innermost region of the S6 segment [47, 53]. In this regard TRPV1 channels also exhibit voltage-dependent behaviour [54].
Splice variants of the TRPV1 channel have been reported in several species. For example, the human TRPV1b splice variant, which lacks exon 7 corresponding to 60 aminoacids in the N-terminal region of the channel, can be found in DRG neurons and in the CNS. It was first reported that TRPV1b could be activated by heat, but not by capsaicin or low pH [55, 56]. However, in a more recent study it was reported that this splice variant is unresponsive to vanilloid agonists, heat and protons and can inhibit channel function by associating with canonical hTRPV1 channels, functioning as a dominant-negative variant, which suggests that it constitutes an endogenous TRPV1 modulator.
Another known TRPV1 splice variant is the rat TRPV1(VAR), thought to be a truncated form of TRPV1, is present at high levels in renal papillary lysates and seems to be non-functional by itself. Interestingly, TRPV1(VAR) is able to modulate TRPV1 function in opposing ways depending on the expression system [57].
The vanilloid receptor 5′-splice variant (VR.5′sv) is another rat TRPV1 splice variant, which lacks the majority of the intracellular N-terminal region and ankyrin repeat elements and does not form functional ion channels. VR.5′sv is expressed in capsaicin-responsive tissues such as brain, DRG and peripheral mononuclear cells, and when associated with TRPV1, it has been found to inhibit its activity through a dominant-negative mechanism[58, 59].
The TRPV1α murine splice variant forms a Ca2+-permeable channel which can be activated by the same ligands known to stimulate TRPV1. In contrast, the TRPV1β murine splice variant is not functional by itself but co-expression with TRPV1α inhibits the function of TRPV1α[60]. It has been suggested that TRPV1β is a naturally occurring dominant-negative regulator of the responses of sensory neurons to noxious stimuli [59,60].
2) TRPV1 activators (agonists)
2.a) Capsaicin and its analogues
Capsaicin and resiniferatoxin (RTX), a highly irritant diterpene related to the phorbol esters, are well established activators of TRPV1[61, 62], with RTX being almost 20-fold more potent than capsaicin (Kd of ~ 40 and 710 nM, respectively) [63]. Other natural TRPV1 agonists are anandamide which also activates CB1 receptors, 12-hydroperoxy-eicosatetraenoic acid (12-HPETE) and N-arachidonoyl dopamine (NADA) [64–67]. Piperine from black pepper, eugenol from cloves and zingerone from horseradish have also been shown to activate TRPV1 receptors [68, 69]. Additionally, gingerols, present in raw ginger, and shogaols, which are dehydration products of gingerols present in steamed ginger, both of which posses a vanillyl moiety, also activate TRPV1 [70–73]
Since both capsaicin and its analogues are lipophilic, they are able to cross the cell membrane and act on binding sites present on the intracellular surface of TRPV1 [74]. In the rodent TRPV1, residues in the N-terminus (Arg114) and in the C-terminus (Glu761) are agonist recognition sites [75]. Moreover, residue Tyr511, located at TM4, was necessary for capsaicin-mediated activation of the TRPV1, and Met547 was essential for RTX sensitivity [76]. In addition, it was proposed that Thr550 interacts with the vanillyl moiety of capsaicin, while Tyr511 is responsible for hydrophobic interactions with the aliphatic domain of capsaicin and other vanilloid agonists [77]. Other residues, such as Trp549 and Ser512 are also important for capsaicin sensitivity [76].
2.b) Allicin and camphor
Recently, controversy has arisen over whether pungent compounds derived from plants of the Allium genus, such as garlic and onion, are able to activate TRPV1. It has been proposed that the TRPA1 channel, which is co-expressed in many of the same neurons as TRPV1, is the sole target for the actions of allicin [78–80]. However, other groups have shown that TRPV1 is also a target for the actions of this compound [46, 47, 81,82]
In contrast to what happens with TRPA1, where channel activation by allicin requires the presence of polyphosphates [83], activation of TRPV1 by garlic and onion extracts as well as by allicin occurs in excised membrane patches through modification of a single cysteine, C157, in the N-terminus of the protein[47].
The crystal structure of the N-terminal region of TRPV1 has been recently solved and is formed in part by six ankyrin repeats [84]. In addition, this region forms a multi-ligand binding domain which mediates its response to ATP, PIP2 and calmodulin [84]. C157 is located on inner helix 2 of the ANK-2 repeat, a region in which other regulatory ligands such as ATP bind. For instance, sites K155, K160 and L163 in the inner helix 2 of the N-terminus, depicted in Fig. (1), form the ATP-binding site in the TRPV1 channel [84].
Camphor is a naturally occurring compound that is used as a topical analgesic, activates heterologously-expressed TRPV1 channels and potentiates currents in DRG neurons, albeit at higher doses than capsaicin. Camphor acts at a site different than capsaicin, since camphor-mediated activation was insensitive to the capsaicin antagonist, capsazepine (Fig. (3), compound (2)) and also occurred in a capsaicin-insensitive point-mutant. Additionally, camphor desensitizes the channel, through a vanilloid-independent mechanism, more rapidly and completely than capsaicin, thereby demonstrating how this compound may function as an analgesic[85].
2.c) Protons and nitric oxide
Acidic extracellular pH augments pain sensation during inflammation or ischemia. In Aδ and C-fibers, the activation of acid-sensing ion channels (ASICs) and TRPV1 are associated with a variety of pain-related conditions including cancer and arthritis [86, 87]. TRPV1 is activated by lowering the extracellular pH [88, 89]. In addition, it sensitizes the responses to capsaicin and, more importantly, to heat, so that the channel can open at moderately high pH at room temperature [3, 68, 90, 91].
Several biological processes are controlled by pleiotropic cell signaling molecules such as nitric oxide (NO). NO signal transduction can occur through protein S-nitrosylation and this S-nitrosylation is capable of conveying physiological redox-based cellular signals [92, 93]. TRPV1 is activated by NO through the modification of cysteines in the primary sequence of the protein. Two cysteines located at the N-terminal side of the putative pore-forming region, in the linker region located between the fifth and sixth transmembrane domains S5 and S6, are partly responsible for the activating effects of NO on the channel protein. These data suggest a role for TRPV1 as a sensor integrating NO signals [94]. The enzyme responsible for NO synthesis, NO synthase (NOS), is activated by intracellular calcium [95]. TRPV1 activation by NO might then result in a feedback regulation mechanism between channel activation, calcium entry and NO production [95]. This might result in enhanced NO production under conditions where NO synthesis is initially stimulated, e.g. under hypoxic conditions.
2.d) Heat and voltage
TRPV1 functions as a molecular thermometer. At a holding potential where normally no channel openings are observed, the inward current abruptly increases when the temperature is stepped to a transition temperature of 43°C [2]. This increase in temperature not only produces a sensation of pain through direct activation of TRPV1, but it also produces neurogenic inflammation through the efferent release of pro-inflammatory neuropeptides [96]. The presence of TRPV1 in free nerve terminals in the skin allows us to detect nociceptive temperatures (> 43°C). However, these channels are exposed to a plethora of regulators that potentiate the channel’s response to temperature.
As discussed below, most of these channel regulators are produced in response to inflammatory conditions or due to tissue damage. Thus, channel activation might occur at normal physiological temperatures under certain cellular conditions, such as inflammation and ischemia, leading to pain.
Until recently it was not clear how or where heat acts to gate the TRPV1 channel. It had been proposed that the distal half of the TRPV1 C-terminus is involved in thermal sensitivity [97]; nonetheless, no mutation had been shown to abrogate thermal sensitivity. Recent studies have shown the “temperature sensor” of at least TRPV1 and TRPM8, another member of the TRP superfamily of channels, to be located at the C-terminus of the protein. Swapping of the C-terminus temperature-sensing “module” of TRPV1 into TRPM8 and vice versa, confers the ability to activate at the temperature at which the donor channel does. That is, TRPV1 with a TRPM8 C-terminus activates at low temperatures (around 25°C) and TRPM8 with a TRPV1 C-terminus activates at high temperatures (around 45°C) [98].
TRPV1 is weakly voltage-dependent, with a shallow g-V relation, a small gating charge associated with channel activation (z) of 0.6–0.8, as compared to voltage-activated potassium channels (z of ~ 12) and a voltage of half maximal activation (V1/2) of around 150mV at 17°C. However, the V1/2 of activation for TRPV1 is highly temperature-dependent, displaying dramatic shifts to more negative potentials upon heating [54, 99]. That is, the sensitivity of this thermoreceptor also depends on the membrane potential and thus would be expected to differ among various cell types. The voltage-sensor in TRPV1 remains unknown, and inspection of the amino acid sequence of the channel reveals the presence of only one positively charged amino acid in the putative TM4. The weak voltage-dependence of the channel likely arises from the scarcity of basic residues in the voltage-sensor domain.
The coupling of voltage- and temperature- gating of TRPV1 channels has been extensively discussed and at least two models have been proposed to account for the temperature-activation of TRPV1 channels. One model explains the temperature-sensitivity of both TRPV1 and TRPM8 through effects of temperature on voltage-dependent gating, so that temperature and voltage-dependent activation are totally dependent on each other [54, 99]. This model assumes a two-state scheme in which temperature changes result in large shifts in the V1/2 of activationdue to the small gating charge of the channel. The direction of the shift is determined by the sign of the entropy difference between the open and closed states, which is positive for TRPV1 channels [54, 99].
The second model, proposed also for the TRPM8 and TRPV1 channels, assumes modular channel architecture with different allosterically coupled domains responsible for temperature- or voltage- activation. This model implies the existence of multiple open and closed states, and the possibility of the channel opening in response to changes in the temperature, which are independent of voltage and vice versa. Here, the large temperature-sensitivity of the channel would not result from the small gating charge, but from the large enthalpy difference between closed and open channels [63, 98].
Alternatively, other TRPV1 channel agonists, such as capsaicin, also shift the channel activation curve to more hyperpolarized potentials. Capsaicin activation seems to be allosterically coupled to voltage- and probably to temperature-activation, since the channel can open in the absence of capsaicin at room temperature (23°C) at depolarized potentials and the curves of open probability vs capsaicin concentration have all the features of a cooperative activation mechanism [53].
2.e.) Artificial sweeteners, inorganic cations, polyamines and spider toxins
TRPV1 receptors or splice variants have been found in taste receptor cells and in nerve terminals throughout the oral cavity. It was recently found that TRPV1 activation could be involved in the artificial sweetener aftertaste or even contribute to the poorly understood metallic taste sensation [100]. Artificial sweeteners not only activate TRPV1 receptors both in heterologous expression systems and in dissociated primary sensory neurons but they also sensitize these channels to acid and heat. Moreover, TRPV1 receptors are activated by CuSO4, ZnSO4, and FeSO4, three salts known to produce a metallic taste sensation[100]. Furthermore, extracellular Na+, Mg2+, and Ca2+ ions sensitize the channel’s response to capsaicin and other related compounds such as anandamide (AEA) and N-arachidonoyl dopamine (NADA) and concentrations of divalent cations >10 mM (e.g. Ca2+, Mg2+) directly gate the receptor[101]. Two glutamates, E600 and E648, formerly identified as proton-binding residues, whose schematized location is shown in Fig. (1), are believed responsible for these effects[101].
Multivalent cations like polyamines are molecules known to increase inflammation and pain signalling and their levels are raised during infection, trauma, and cancer [102–104]. For example, intrathecal administration of sperminein rodents produces nocifensive behaviors such as licking, scratching, and biting. A recent study has determined that cationic polyamines regulate TRPV1 activity. That is, extracellular application of polyamines such as spermine and spermidine directly activate TRPV1 both in heterologous expression systems and sensory neurons [105].
Bites and stings from venomous creatures are well known to produce pain and inflammation. Although many molecules responsible for the effects of these venoms have been widely characterized, the mechanisms underlying the painful processes produced by poisons have remained rather obscure. Recently, several venoms from spiders and scorpions were examined and a fraction of the venom of a tarantula from the West Indies, Psalmopoeus cambridgei, activated TRPV1 [106]. The fraction responsible for the activating effects observed contained three cysteine knot (ICK) peptides, now termed vanillotoxins. The mechanism by which vanillotoxins activate TRPV1 remains to be clarified.
The venom from the spider Agelenopsis aperta, a North American funnel web spider, is a potent inhibitor of TRPV1. Two acylpolyamine toxins, AG489 and AG505, inhibit TRPV1 from the extracellular side of the membrane [107]. Four amino acid mutations located at the TM5-TM6 linker dramatically decreased toxin affinity, consistent with the notion that this region forms the outer vestibule of TRPV1 channels and that AG489 is a pore blocker [107].
3) TRPV1 blockers and the effects of oxidizing and reducing agents
Recently, it was shown that the activity of nociceptors can be selectively suppressed by the membrane-impermeant local anesthetic and lidocaine derivative, QX-314 [108]. Binshtok et al. (2007) demonstrated that this voltage gated sodium-channel blocker, which blocks sodium channels from the intracellular face of the membrane, can be targeted to nociceptors by promoting the entrance of these compounds through the TRPV1 pore. Applying QX-314 in the presence of the TRPV1 agonist capsaicin, allowed QX-314 to diffuse into nociceptors expressing TRPV1 and block voltage–gated sodium channels, thus inhibiting their excitability. Moreover, injection of QX-314 together with capsaicin into rat hindpaws produced a long-lived increase in mechanical and thermal nociceptive thresholds [108], proving this to be an efficient method for reducing pain that originates in the periphery.
Another TRPV1 pore blocker has been recently identified. The quaternary ammonium tetrabutylammonium (TBA) blocks TRPV1 with high affinity from the intracellular side of the membrane. As in voltage-gated potassium channels, TBA acts as a voltage-dependent pore blocker [53]. Kinetics of block were consistent with a state-dependent blocking mechanism, with TBA interfering with closing of an activation gate. This study suggested, for the first time, that the activation gate of TRPV1 may be located cytoplasmically, similar to what has been observed in potassium channels [53].
The lanthanide, gadolinium (Gd3+), is a known blocker of several types of cation selective channels, including some members of the TRP superfamily [109–112]. Depending on its concentration, Gd3+ promotes interesting effects on TRPV1 channels. At low concentrations (near 10 μM), it activates and potentiates the rat TRPV1 channel -whereas at higher concentrations (>300 μM) it blocks them.
TRPV1 has 18 cysteines in its primary sequence [2]. This has led several groups to investigate the role of reducing and oxidizing compounds on TRPV1 activity. The reducing agents dithiothreitol (DTT) and glutathione lower the temperature threshold for TRPV1 activation and potentiate capsaicin-induced currents [113, 114]. Site-directed mutagenesis experiments in the pore loop have identified Cys621 as the residue responsible for the extracellular modulation of TRPV1 by reducing agents. Moreover, the oxidizing agents diamide and chloramine-T also facilitated thermally-induced TRPV1-mediated currents [114]. Alkylating agents such as N-ethylmaleimide also strongly and irreversibly affect heat-evoked responses from TRPV1, lowering the thermal-activation threshold in a DTT-dependent manner [114]. From these data it follows that TRPV1 is targeted by redox-active substances that directly modulate channel activity, and that channel potentiation might occur under altered redox states in a tissue, e.g. during ischemia and/or inflammation, presumably leading to allodynia.
4) Desensitization and tachyphylaxis: antinociception
The phenomenon of desensitization by vanilloids in sensory neurons was first described in 1949 by Nicholas Jancsó [115–118]. This desensitization, or the refractory state where there is loss of activity, occurs at the level of the receptors, that is, at the level of TRPV1 channels. In 1961 Jancsó and colleagues showed that 4, 8, and finally 15mg of capsaicin administered to adult rats (approximately 80 mg/kg s.c.) over a period of 1 to 3 days is sufficient to render the animals fully insensitive to chemically evoked pain for up to 3 months [118]. There are two types of desensitization described for TRPV1 channels: acute desensitization, characterized by a rapid loss of activity of the receptor with an agonist bound to it, and tachyphylaxis, evidenced by a gradually diminishing response to repeated agonist administrations [19].
Acute desensitization of TRPV1 reflects an agonist-induced conformational change, which results in the closing of the channel pore. This process is dependent upon the presence of intracellular calcium and can be inhibited by intracellular calcium chelators[2, 119]. Studies have shown that acute desensitization arises from the interaction of the channel with calcium-calmodulin (CaM), where CaM acts as a Ca2+ sensor for TRPV1 thereby decreasing channel activity in response to increases in intracellular Ca2+ concentration. When capsaicin binds to TRPV1 the channels open and Ca2+ enters the cell. Ca2+ then binds to CaM, producing desensitization by either biasing gating toward the closed state or inducing a new closed state, without altering unitary conductance or channel number [120].
Tachyphylaxis, on the other hand, involves the cycling of TRPV1 between resting and active states through numerous nonconducting intermediate states [121]. This is why tachyphylaxis has been viewed as the recovery of TRPV1 from the intermediate states to the resting state where the channels can be activated again by agonist binding, a process where calcium and many other factors such as ATP and PIP2 might also play a role [84, 122, 123]
5) TRPV1 modulators
The following section will focus on the actions of modulators of TRPV1 activity. Fig. (2) depicts a summary of some of the pathways used by TRPV1 modulators to regulate its activity and promote inflammatory or painful responses while the structural regions of TRPV1 that interact with its agonists and modulators are depicted in Fig. (1).
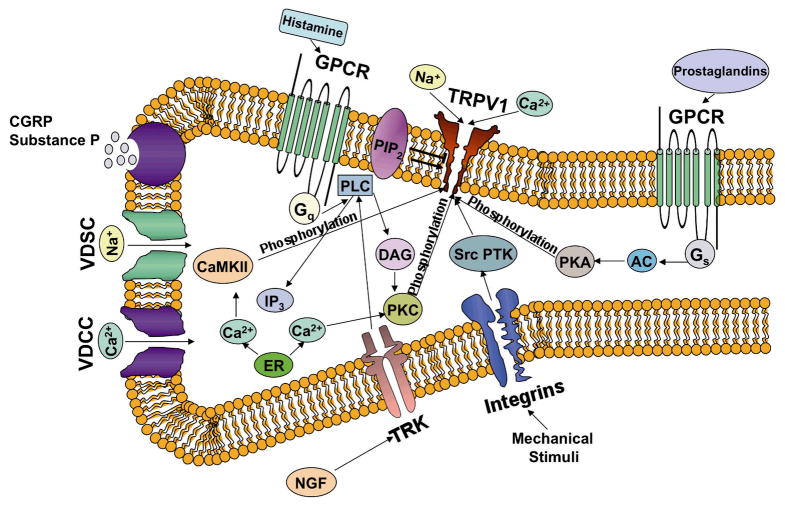
Figure 2. TRPV1 regulation by intracellular signaling pathways.
The TRPV1 channel is coupled to many intracellular signaling cascades related to inflammatory processes that regulate channel activity. Multiple G-protein coupled receptors (GPCRs) are activated by pro-inflammatory agents including histamine and prostaglandins. The activation of receptors coupled to Gs proteins leads to adenylate-cyclase (AC) stimulation, cAMP production and concomitant cAMP-dependent protein kinase (PKA) activation. PKA directly phosphorylates the TRPV1 channel modulating its activity. Activation of GPCRs coupled to Gq proteins leads to phospholipase-C (PLC) stimulation, which degrades plasma membrane-associated PIP2 into 1,2-diacylglycerol (DAG) and (1,4,5)-inositol triphosphate (IP3). Increases in IP3 lead to Ca2+ release from intracellular stores such as the endoplasmic reticulum (ER). Both Ca2+ and DAG activate protein kinase C (PKC), which also phosphorylates the TRPV1 channel and regulates its function. PIP2 regulates the TRPV1 channel in a complex manner, with both positive and negative effects depending on the state of the receptor. TRK receptor activation by extracellular signals such as nerve growth factor (NGF) also leads to PLC activation. Intracellular calcium elevation through TRPV1 or voltage-dependent calcium channel (VDCC) activation or through calcium release from intracellular calcium stores due to pro-inflammatory mediators also regulates channel function: calcium binds to calmodulin which is associated with TRPV1 and promotes channel desensitization. Calcium-calmodulin dependent kinase II (CaMKII) directly phosphorylates the channel. Mechanical stimuli lead to integrin-dependent src-Protein tyrosine kinase (srcPTK) activation and direct action on the TRPV1 channel. Additionally, cell depolarization due to voltage-dependent calcium or sodium (VDSC) channel activation directly gates the channel. All regulators depicted in the figure are positive regulators of the channel except for the bimodal action of PIP2.
The processes of phosphorylation and dephosphorylation are crucial for TRPV1 function. This is exemplified by the role of the phosphatase, calcineurin, which inhibits TRPV1desensitization [124], and by the actions of calmodulin dependent kinase CaMKII, which regulates TRPV1 activity through phosphorylation of two residues: Ser 502 and Thr 704 [44].
In nociceptive neurons, activation of phospholipase C (PLC)-coupled receptors by pro-inflammatory agents such as ATP, nerve growth factor (NGF), bradykinin, or chemokines sensitizes TRPV1 to heat, acid and capsaicin [40, 125–127]. This phenomenon underliesthe increased sensitivity to painful stimuliafter tissue injury or inflammation.
TRPV1’s activity is also modulated by the regulatory lipid, phosphatidylinositol-4,5-bisphosphate (PIP2) via activation of phospholipases like PLC. One early study showed that PIP2 synthesis is necessaryfor the recovery of TRPV1 currents from desensitization [128]. Nonetheless, there is controversy as to whether PtdIns(4,5)P2 (PIP2) increases or decreases the open probability of the channel. On the one hand, PIP2 was said to bias the channels in the closed state and relief from inhibition could be obtained by the activation of PLC [40]. This idea was based on indirect experiments where the effects of phosphoinositideswere not directly tested in excised patches [40].
On the other hand, in excised patches it was found that PtdIns(4,5)P2, its precursor PtdIns(4)P (PIP), and other phosphoinositides activate TRPV1 [129, 130] and positively charged aminoacids R701 and K701 in the TRPV1 sequence are responsible for the direct activating actions of PIP2 [131].
In another study the controversy regarding the role of PIP2 may have been resolved. Using HEK293 cells, the authors found that after exposing TRPV1 to high capsaicin concentrations, the ensuing Ca2+ influx activates PLC, which results in the depletion of PtdIns(4,5)P2 and PtdIns(4)P, which reduces channel activity, leading to desensitization[129]. Inhibition of PLC activity resulted in a lack of desensitization. It was also shown in excised patches that PtdIns(4)P, the precursor of PtdIns(4,5)P2, activated TRPV1 and inhibited desensitization, and, in addition, that PtdIns(4,5)P2 had an inhibitory effect on the channel, but only at low capsaicin concentrations. This inhibitory effect could only be detected in intact cells and not in excised patches, indicating that this effect might be indirect. In this study, the authors conclude that the balance between the inhibitory and activating effects of PtdIns(4,5)P2 depends on the stimulation level ofthe channel, since during sensitization PLC-coupled agonists induce a moderate depletion of PtdIns(4,5)P2, removing its inhibitory effect, but not producing low enough lipid levels to inhibit channel activity. In contrast, high capsaicin concentrations induce a severedepletion of PtdIns(4,5)P2 that limits channel activity and leads todesensitization [129], proving TRPV1 regulation by lipids to be rather complex.
In this regard, it has been shown that phosphoinositide 3-kinase interacts directly with TRPV1 and that this complex facilitates TRPV1 trafficking to the plasma-membrane. This trafficking is observed in response to nerve growth factor (NGF), a mechanism that might be responsible for NGF and other related pro-algesic agents’ ability to induce hyperalgesia [130, 132].
Other membrane-derived lipids also regulate TRPV1. For example, oleylethanolamide (OEA), a natural analogue of the endogenous cannabinoid anandamide, anandamide itself, and some lipoxygenase products all modulate TRPV1 function [64, 133, 134]. TRPV1 is also activated by the metabolic products of lipoxygenases (LOXs), such as 12- and 15-HPETEs (hydroperoxyeicosatetraenoic acids) and 5- and 15-HETEs (hydroxyeicosatetraenoic acids) [64].
Recently, omega-3 (n-3) fatty acids, which exhibit analgesic properties, have been shown to interact directly with TRPV1 [135]. These fatty acids activate TRPV1 in a phosphorylation-dependent manner and enhance its responses to extracellular protons. Interestingly, these lipids competitively inhibit the responses of vanilloid agonists. This differential regulation of TRPV1 by n-3 fatty acids might be advantageous for the development of a therapy for painful conditions [135].
Other inflammatory agents which activate TRPV1 through intracellular pathways include prostaglandins, histamine, bradykinin and serotonin [40, 125, 136–144]. TRPV1 channel activation results in nociceptor activation, with concomitant physiological consequences.
The effects of inflammatory mediators on TRPV1 arise from a variety of intracellular signals. Tyrosine kinases and G protein-coupled receptors are capable of modulating TRPV1’s response to heat, enabling the channel to open even at a normal body temperature [125, 140]. For example, 12-HPETE formation by means of bradykinin action results in TRPV1 activation [145]. Some effects of inflammatory agents on TRPV1 depend on channel phosphorylation through protein kinase C (PKC) [139, 140, 146] or cAMP-dependent protein kinase (PKA) [147–149].
Prostaglandins, such as PGE2, increase cAMP levels and therefore activate PKA, which directly phosphorylates the channel [147]. Residues located in the N-terminus of TRPV1 (Ser 116 and Thr 370) are phosphorylated by PKA and have been implicated in desensitization [147–149] while residues Thr 144, Thr 370, and Ser 502 have been implicated in sensitization of heat-evoked TRPV1 responses when phosphorylated by PKA [150]. This latter effect suggests a role for PKA in the development of thermal hyperalgesia [150]. Interestingly, this effect is suppressed by morphine acting through peripheral opioid receptors [151].
The activation of PKC and the subsequent phosphorylation of TRPV1 potentiates capsaicin, acid, and thermal responses in TRPV1 channels. This phosphorylation occurs at two target Ser residues (Ser 502 and Ser 800) [45, 152] which are also implicated: (1) in potentiation of endovanilloid/endocannabinoid NADA-induced TRPV1 activation [66], (2) rephosphorylation of TRPV1 after desensitization in the presence of Ca2+ [153] and (3) OEA-induced TRPV1 activation [133]. Moreover, PKC is also at least partly involved in the trafficking of the channel to the plasma membrane through SNARE-dependent exocytosis [154]. The N-terminal region of TRPV1 is able to interact with the vesicular proteins snapin and synaptotagmin IX, which inhibit PKC-dependent TRPV1 potentiation [154].
Molecules such as phorbol esters have also been implicated in TRPV1 activation. For instance, phorbol 12-myristate 13-acetate (PMA), a PKC-activating phorbol, decreases binding of [3H] RTX to TRPV1 [40] through interaction with Tyr 704 in the C-terminus[45].
Recently, in neurons it was found that TRPV1 interacts through the N- and C-terminal regions with the tubulin-cystoskeleton that acts to regulate growth-cone motility and cytoskeletal dynamics. As long as it is membrane-associated, the C-terminal portion of the protein can stabilize tubulin, which can induce filopodia formation independently of the rest of the channel. This suggests a role for some of the apparently non-functional TRPV1 splice variants, which apart from regulating the functional channel, might have a role in cytoskeletal-dynamics regulation. Tubulin destabilization is induced upon channel activation in a partially Ca2+-independent manner resulting in rapid growth-cone collapse [155–159]. Cytoskeletal destabilization through the TRPV1 channel might be a mechanism for pain-chronification (i.e., the pathophysiological process of developing chronic T-cell driven inflammation after acute (macrophage) driven inflammation) [160].
6) TRPV1 in disease and pain
We have presented evidence that multiple signals that originate from inflammatory processes converge to activate TRPV1, whose activation in sensory neurons has the final consequence of pain perception. In the following section we will show that, TRPV1 plays a role in a wide range of pathologies, proving this channel protein to be a formidable potential therapeutic target for pain-management drugs. In this section we will also elaborate on some of the advances made in this respect.
Neurogenic inflammation is characterized by edema, thermal and mechanical hyperalgesia, vasodilatation and inflammatory pain caused by overstimulation of peripheral nociceptor terminals subsequent to injury [161]. Overstimulation of these terminals gives rise to an increased release of neurotransmitters and pro-inflammatory peptides from central and peripheral nociceptor terminals and, in the case of tissue injury, to a release of protons from damaged cells. Indeed, inflammatory diseases such as bowel disease, asthma, allergic dermatitis, pancreatitis and vulvodynia include neurogenic components caused by the release of neuropeptides such as substance P (SP), calcitonin gene-related peptide (CGRP) and neuropeptide Y (NPY) [96, 162–164]. Other molecules, such as nerve growth factor (NGF), protons, ATP, histamine, cytokines and chemokines act as proalgesic, proinflammatory mediators [165, 166]. In addition, TRPV1 is also modulated by leukotriene B4 (LTB4) and other metabolites of arachidonic acid, and this contributes to the development of neurogenic inflammation [167]. To this point, following injury, increased TRPV1-immunoreactive fiber innervation has been observed in inflamed tissues such as: gastrointestinal tract, human skin and vulva [161, 168–172]. This has led several groups to propose that upregulation of TRPV1 can contribute to the pathogenesis of various diseases such as inflammatory bowel disease, gastroesophageal reflux disease, irritable bowel syndrome, prurigo nodularis and vulvar allodynia [168–171]. Increased expression of TRPV1 also correlates with inflammatory hyperalgesia [173].
In models of pathological nociception and thermal hyperalgesia, a selective TRPV1 blocker, A-425619 (Fig. (3), compound (3) ), produces antinociceptive effects [174, 175]. In the capsaicin-induced secondary hyperalgesia model in the rat [176] the oral TRPV1antagonist SB-705498 (Fig. (3), compound (4) ), acts to reduce hyperalgesia-and allodynia [177, 178]. Furthermore, this compound has also been tested in humans, in which the effects of SB-705498 on heat-evoked pain and skin sensitization induced by capsaicin or UVB irradiation were assessed. It was found that the drug increased heat pain tolerance at the site of UVB-evoked inflammation [177, 178]. From the above, it is clear that there is great potential for TRPV1 antagonists in the treatment of painful conditions. On the other hand, the use of potent analgesics, which act through the induction of desensitization of TRPV1, has also proven to be an efficient method of antihyperalgesia. This is the case for the cannabinoid receptor (CB1) receptor agonist WIN55 which promotes TRPV1 desensitization via a calcium calcineurin-dependent mechanism [179].
6.a) Digestive tract
In the stomach and the duodenum, one of the most important roles of TRPV1-expressing sensory nerves is the preservation of the integrity of the tissues exposed to aggressive compounds, such as protons and activated enzymes [180]. Tissue protection by capsaicin-sensitive primary afferents seems to occur through multiple mechanisms. For example, capsaicin can either induce an increase in blood flow to a tissue or hyperemia through vasorelaxation produced by calcitonin gene-related peptide release from capsaicin-sensitive primary sensory fibres [181],. Alternatively, capsaicin-induced CGRP release can promote activation of cyclooxygenase-1 enzymes leading to the production of prostaglandin E2 [182]. In turn, this latter compound activates secretory cells, which produce the protective mucus layer [183]. Two TRPV1 activators, protons (pH4.0) and alcohol (10%) induce cell damage, while activators such as the vanilloids, capsaicin (10−9–10−6 M) and resiniferatoxin (10−12–10−9 M) concentration-dependently prevent the proton and alcohol-evoked effects [184].
6.b) Respiratory system
TRPV1 is expressed in C-fibers originating from the nodose and intracranial jugular ganglia, which innervate the respiratory tract [185, 186]. TRPV1 is also expressed in lung epithelial cells and bronchial smooth muscle [187]. Activation of these fibers leads to bronchoconstriction, mucus secretion, bradycardia and hypotension, in addition to cough and airway irritation[188–190]. Moreover, the nerve terminals of these fibers often contain neuropeptides such as tachykinins (TKs) and CGRP, which are released upon nerve stimulation and lead to bronchoconstriction and inflammatory cell chemotaxis [191, 192].
Using the trpv1−/− mice it was shown that TRPV1 is obligatory for vagal C-fiber activation by capsaicin and anandamide, and that the channel plays a regulatory role in the effects caused by bradykinin and acid [193]. In humans, capsaicin can evoke the cough reflex [194–197] and this response is exaggerated in patients with asthma or chronic obstructive pulmonary disease [198]. Pre-treatment of animal allergic models with capsaicin inhibits several of the effects normally observed in the presence of allergen [199, 200]. Similar effects of capsaicin have been observed in a mouse model of non-atopic asthma [201], indicating a connection between TRPV1 channel activation and asthma. TRPV1 agonists or antagonists might then be useful in the treatment of these conditions; however, there are currently no drugs for the treatment of pulmonary diseases targeted to the TRPV1 channel that have been tested in humans [202].
6.c) Bladder
An important role for TRPV1 in bladder disease has also been identified. In fact, due to their desensitizing effects, capsaicin and resiniferatoxin have been effective in the treatment of overactive (irritable) bladder symptoms [168, 203–210]. TRPV1 knockout mice show differences in their response to bladder injury when compared to their wild-type counterparts. For instance, trpv1 knockout mice do not develop bladder overactivity during acute bladder inflammation, pointing to a role for TRPV1 in bladder inflammatory states [211]. A role for TRPV1 in bladder overactivity is also supported by clinical observations. In patients suffering from neurogenic detrusor overactivity (NDO), TRPV1 immunoreactivity in the urothelium and the number of nerve fibers expressing TRPV1 are increased [212]. For those patients who benefited from intravesical resiniferatoxin (RTX) therapy, TRPV1 urothelial immunoreactivity decreased after treatment. In addition, in biopsies from the same patients, suburothelial TRPV1-expressing nerve fibers were reduced in number following therapy with RTX. Apparently, successfultherapy using RTX leads to a reduced TRPV1 expression in both urothelial and neuronal cells [212].
6.d) Diseases of the Basal Ganglia
There are several studies showing that TRPV1 plays a role in dopaminergic mechanisms associated with schizophrenia and Parkinson’s disease. In this regard, N-oleoyldopamine, an endogenous ligand for the TRPV1, increases the firing rate of dopaminergic neurons of the midbrain ventral tegmental area (VTA). In addition, capsaicin –evoked dopamine (DA) release was inhibited by application of TRPV1 antagonists such as iodo-resiniferatoxin (I-RTX) [65]. In regard to TRPV1’s effects in the basal ganglia exposure of mesencephalic dopaminergic neurons to capsaicin triggers cell death, while exposure to TRPV1 antagonists prevents these effects [213]. Moreover, schizophrenic patients tend to display reduced pain sensitivity [214, 215] and a diminished skin flare response to niacin [216–218], suggesting that there are defects in TRPV1-expressingafferent nerve fibers.
6.e) Cardiovascular
TRPV1 is expressed in cardiac spinal sympathetic sensory fibers [138]. During cardiac ischemia these fibers are essential for the sympathoexcitatory reflex, which is associated with increased blood pressure and chest pain [219]. During ischemia, there is bradykinin-induced activation of sensory nerve endings in the heart [138]. The activation of TRPV1 under conditions of acidosis and ischemia provides the organism with a mechanism, which relays painful information to the brain. On the other hand, the release of agents such as SP, neurokinin A (NKA) and CGRP by the nerve fiber itself has beneficial effects, which help antagonize the negative effects of ischemia and acidosis, resulting in a cardioprotective role for TRPV1. Among these beneficial effects we find vasodilatation, reduction in Ca2+ accumulation, lipid antiperoxidation, cellular membrane stabilization and anti-arrythmic effects [220, 221]. It should be noted that TRPV1 is implicated in the cardioprotective effect associated with alcohol consumption, where ethanol causes coronary artery dilation and release of CGRP from perivascular sensory nerve terminals [222].
A role for anandamide- and capsaicin-induced desensitization in vasoconstriction has been proposed [223], establishing a possible connection between TRPV1 and hypertension. The proposed mechanism for this effect is a reduced release of the potent vasodilators CGRP and SP [224]. Anandamide can also act as a TRPV1 receptor agonist in the trigeminovascular system where it promotes channel activation leading to CGRP release and excessive vasodilation. In fact, it is possible that TRPV1-mediated CGRP release is related to migraine, since TRPV1 expressed in nociceptive afferent fibers of the encephalic dura mater contributes to dural vasodilation [225]. A recent surge of evidence has shown TRPV1 expression in astrocytes, microglia and pericytes in the brain [226]. The TRPV1 channel might have an additional role in regulating vascular tone and blood-brain-barrier permeability under inflammatory conditions in the brain.
6.f) Diabetes
Studies using the Zucker diabetic rat model of type 2 diabetes show that doses of capsaicin and RTX which desensitize TRPV1 result in improved glucose tolerance through enhancement of insulin secretion and decreased plasma insulin levels [227]. It follows that TRPV1-expressing cells could be involved in glucose regulation [227]. Other studies using non-obese diabetic (NOD) mice that are genetically prone to develop type 1 (insulin-dependent) diabetes have implicated TRPV1 in the development of diabetes. These specific mice carry a hypofunctional TRPV1 mutant (TRPV1NOD) localized to the Idd4.1 diabetes-risk locus [228]. In this animal model ablation of TRPV1-expressing neurons which innervate the pancreas through neonatal capsaicin treatment averts the insulitis and pancreatic β-cell destruction that normally occurs in these animals.
6.g) Itch
A role for TRPV1 in itch has been suggested. The itch-selective sensory afferents respond to capsaicin, suggesting that TRPV1 might be expressed on the “pruriceptor subpopulation” of mechanoinsensitive fibers [30, 144]. In humans, changes in skin temperature and in pH can effectively modulate itch sensation [229], and contrary to common belief, raising skin temperature alleviated histamine-induced itch [230]. Therefore, TRPV1 may function as a “central integrator” molecule in the itch pathway[229].
6.h) Bone Cancer
Recent advances have been made in the treatment of pain caused by bone sarcomas, where TRPV1 seems to play an important role [231, 232]. Bone cancer leads to osteoclast activation, which promotes acidosis and concomitant TRPV1 activation in sensory fibers [86]. In a murine in vivo model of bone cancer pain, treatment of mice with TRPV1 antagonists such as JNJ-17203212 (Fig. (3), compound (5)) resulted in a marked decrease in movement-evoked nocifensive behaviour [231]. Additionally, recent findings indicate that TRPV1 expression is increased in bone cancer [233]. Taken together, all the evidence points to a role of TRPV1 in bone cancer pain. Clearly, future studies are needed to solidify this finding.
6.i) Osteoarthritis
In in vivo models of osteoarthritis, over expression of TRPV1 and increased CGRP release occur [234]. In wild-type mice, injection of Complete Freund’s Adjuvant (CFA) promoted marked arthritic changes in tibiotarsal joints while in TRPV1 knock-out animals, such treatments had reduced effects. Swelling of the knee-joint by injection of CFA was greatly reduced in TRPV1-defficient mice when compared to wild-type animals [235].
7) TRPV1 antagonists and agonists: the road to pain relief
Sufficient evidence has been presented regarding the importance of the TRPV1 channel in different pain-producing diseases and some of the advances made in TRPV1-directed therapies have been mentioned. This last section will provide the reader with a general picture of our current understanding of the road to pain relief in TRPV1-targeted drug research. Selected structures and possible functions of some of the TRPV1 agonists and antagonists are summarized in Fig. (3) and Table 1.
Table 1.
Potential uses of TRPV1 agonists and antagonists in pain relief.
| Compound | Agonist | Antagonist | Possible applications |
|---|---|---|---|
| Capsaicin | √ | - | Arthritic pain relief |
| WL-1002 | √ | - | Topical relief of osteoarthritis pain. |
| WL-1001 | √ | - | Relief of post-herpetic neuralgia of the trigeminal nerve and in migraine prophylaxis. |
| Capsazepine | - | √ | Induces bronchorelaxation. Treatment of asthma. |
| A-425619 | - | √ | Inflammatory, postoperative and arthritic pain |
| BCTC | - | √ | Inhibition of capsaicin induced cough |
| Quinazolinone compound 26 | - | √ | Asthma treatment |
| AMG 0347 | - | √ | Inhibits skin incision-induced pain |
| AMG 9810 | - | √ | Antihyperalgesic properties in a rat model of inflammatory pain (abdominal allodynia). |
| AMG 8163 | - | √ | Hyperthermia |
| A-784168 | - | √ | Inhibits both thermal hyperalgesia and mechanical allodynia |
| Benzimidazole | - | √ | Achieved significant reversal of CFA-induced thermal hyperalgesia |
| JNJ-17203212 | - | √ | Inhibits bone cancer pain |
| SB-705498 | - | √ | Acute treatment of migraine. Produces reduced capsaicin-evoked flare and acute heat-evoked pain on non-sensitized skin |
| GRC 6127 | - | √ | Reverses certain types of mechanical hyperalgesia in the rat. |
After the cloning of TRPV1, pharmaceutical companies have made the search for TRPV1 antagonists into standard discovery programs. For example, capsazepine (Fig. (3), compound (2)), a relatively non-specific TRPV1 inhibitor, has been extensively used as a tool in pharmacological studies such as evaluating the role of TRPV1 in inflammatory pain processes [232, 236–240].
The anti-hyperalgesic effects of some TRPV1 antagonists have been evaluated in several in vivo pain models. In these studies, the findings were that certain antagonists, including capsazepine, A-425619, SB-705498, JNJ-17203212 (4-(3-trifluoromethyl-pyridin-2-yl)-piperazine-1-carboxylic acid ( 5-trifluoromethyl-pyridin-2-yl)-amide), BCTC, a quinazolone termed compound 26, A-784168 (N-1H-indazol-4-yl-N′-[(1R)-5-piperidin-1-yl-2,3-dihydro-1H-inden-1-yl]urea) and JYL1421( N-(4-tert-butylbenzyl)-N′-[3-fluoro-4-(methylsulfonylamino)benzyl]thiourea), (Fig. (3), compounds (2), (3), (4), (5), (6), (7), (9) and (10), respectively) were modestly effective in reversing the nociceptive behaviours associated with neuropathic pain, bone cancer pain, osteoarthritic pain, etc [161, 174, 175, 177, 178, 232, 236, 240, 241, 244, 245, 247–264].
The ability of antagonists to block several modes of TRPV1-activation seems to be essential for these compounds to act on nociceptive (pain) and/or inflammatory processes. The compounds A-425619, BCTC and AMG9810 (Fig. (3), compounds (3), (6) and (8), respectively), which inhibit acid-, vanilloid- and heat-activation of the TRPV1 in rats also inhibit and reduce inflammation-related hyperalgesia [175, 241, 244, 246, 253, 265]. It was recently found that both the antagonists AMG 517 (N-(4-[6-(4-trifluoromethyl-phenyl)-pyrimidin-4-yloxy]-benzothiazol-2-yl)-acetamide I) and AMG8163 (tert-butyl-2-(6-([2-(acetylamino)-1,3-benzothiazol-4-yl]oxy)pyrimidin-4-yl)-5 (trifluoromethyl)phenylcarbamate) completely antagonize capsaicin, proton, and heat activation of TRPV1 in vitro and block capsaicin-induced flinch in rats. Together, these results suggest that the antagonists capable of blocking many forms of TRPV1-activation are those which will achieve anti-hyperalgesic effects.
TRPV1 antagonists produce some serious side effects. For instance, in rodents JNJ-17203212 (Fig. (3), compound (5) ), AMG0347 (Fig. (3), compound (11) ), AMG9810 (Fig. (3), compound (8) ) and AMG8163 cause significant hyperthermia [266, 267]. This effect seems to be regulated by centrally-expressed TRPV1 receptors since JYL1421 (Fig. (3), compound (10) ), a peripherally restricted antagonist of TRPV1, does not cause hyperthermia [267, 268].
The therapeutic value of many TRPV1 agonists arises from their ability to reduce electrical activity of TRPV1-containing nerves. Activation of TRPV1 by its agonists leads to membrane depolarization, which in turn results in sodium and calcium channel activation. Then, acute reduction in neuronal activity occurs, which arises from voltage-dependent inactivation of sodium channels, while longer-term inhibition of activity occurs in response to the associated rise in intracellular Ca2+ and associated calcium dependent processes [269]. In this regard, several studies have demonstrated that RTX-application inhibits the activity of capsaicin-responsive sensory neurons [270, 271].
Topical creams and oral compounds containing capsaicin have been used to treat pain [272]. However, the administration of agonists causes acute pain and discomfort which has led to limited use in patients, so that new and less pungent TRPV1 agonists with the same desensitizing effects of capsaicin need to be developed.
Capsaicin patches with a high concentration of trans-capsaicin (NGX-4010) applied directly to the skin have been reported useful in trial studies of patients with post-herpetic neuralgia (see webpage for Neurogesx labs at www.neurogesx.com/ngx_4010). Nonetheless, the use of RTX and other capsaicin-based agonists has not proven successful in the management of some kinds of pain, suggesting that only in some cases may the TRPV1-agonist approach prove useful.
Clinical laboratories have produced TRPV1 agonists such as WL-1002, a topical agent which might be used to reduce osteoarthritic pain and WL-1001, a nasal spray which could be used for post-herpetic neuralgia of the trigeminal nerve and in migraine prophylaxis (nasal spray, see webpage for Winston Labs www.winstonlaboratories.com).
Aminoglycoside antibiotics such as neomycin induce analgesia in various animal models [273]. Until recently the underlying mechanism for the analgesic effects of neomycin was unknown. It has now been shown in DRG neurons that neomycin acts as a potent non-competitive blocker of TRPV1 by lowering the open probability (Po) at both negative and positive potentials [273].
On the basis of so much evidence, it is clear that TRPV1 plays a key role in the physiology of pain, not only integrating several pain-related molecular and physical signals but also mediating a response at several levels of action, from systemic effects through nerve depolarization and signal transmission to the brain, to local effects by stimulating neuropeptide release, altering intracellular signal cascades or regulating cytoskeletal dynamics, among many other actions. Its widespread expression in many organs and tissues, including the brain, and its apparently differing functions according to the tissue or organ where it is expressed add to the complexity of the system.
To date, extensive evidence linking the TRPV1 channel to several states of disease exhibiting pain-related symptoms has been obtained. Hence, TRPV1’s privileged position in the pain-pathway makes it a very profitable target for pain-management drugs in general, and several advances have been made thus far. Several TRPV1 antagonists with therapeutic potential have been developed and some positive results have been obtained in laboratory trials. Also several channel agonists that take advantage of the desensitizing properties of the channel are the focus of extensive research, which has already given rise to interesting results. In any event, success in finding a viable therapy targeting the TRPV1 channel will depend on experimental studies aimed at obtaining detailed knowledge of the channel protein itself and of the physiological importance of this channel in the tissues in which it is expressed. To date the clinical data hint at the possibility that TRPV1 antagonists might prove to be practical therapeutic options for conditions such as diabetes, bladder disease, migraine, respiratory afflictions, and pain related to several types of diseases. Based on the studies considered in this review, it seems likely that several advances with therapeutic applicability will be made in the future.
Acknowledgments
TR was supported in part by grant CONACyT No. 58038 and DGAPA–UNAM IN200308. SAS was supported in part by grants NIH Grants GM27278, DC-01065 and by grants from Philip Morris USA and Philip Morris International Inc.
References
- 1.Montell C, Rubin GM. Molecular characterization of the Drosophila trp locus: a putative integral membrane protein required for phototransduction. Neuron. 1989;2:1313–1323. doi: 10.1016/0896-6273(89)90069-x. [DOI] [PubMed] [Google Scholar]
- 2.Caterina MJ, Schumacher MA, Tominaga M, Rosen TA, Levine JD, Julius D. The capsaicin receptor: a heat-activated ion channel in the pain pathway. Nature. 1997;389:816–824. doi: 10.1038/39807. [DOI] [PubMed] [Google Scholar]
- 3.Tominaga M, Caterina MJ, Malmberg AB, Rosen TA, Gilbert H, Skinner K, Raumann BE, Basbaum AI, Julius D. The cloned capsaicin receptor integrates multiple pain-producing stimuli. Neuron. 1998;21:531–543. doi: 10.1016/s0896-6273(00)80564-4. [DOI] [PubMed] [Google Scholar]
- 4.Caterina MJ, Julius D. The vanilloid receptor: a molecular gateway to the pain pathway. Annu Rev Neurosci. 2001;24:487–517. doi: 10.1146/annurev.neuro.24.1.487. [DOI] [PubMed] [Google Scholar]
- 5.Ramsey IS, Delling M, Clapham DE. An introduction to TRP channels. Annu Rev Physiol. 2006;68:619–647. doi: 10.1146/annurev.physiol.68.040204.100431. [DOI] [PubMed] [Google Scholar]
- 6.Nilius B, Voets T. TRP channels: a TR(I)P through a world of multifunctional cation channels. Pflugers Arch. 2005;451:1–10. doi: 10.1007/s00424-005-1462-y. [DOI] [PubMed] [Google Scholar]
- 7.Pedersen SF, Owsianik G, Nilius B. TRP channels: an overview. Cell Calcium. 2005;38:233–252. doi: 10.1016/j.ceca.2005.06.028. [DOI] [PubMed] [Google Scholar]
- 8.Montell C, Birnbaumer L, Flockerzi V. The TRP channels, a remarkably functional family. Cell. 2002;108:595–598. doi: 10.1016/s0092-8674(02)00670-0. [DOI] [PubMed] [Google Scholar]
- 9.Montell C. The TRP superfamily of cation channels. Sci STKE. 2005:re3. doi: 10.1126/stke.2722005re3. [DOI] [PubMed] [Google Scholar]
- 10.Moran MM, Xu H, Clapham DE. TRP ion channels in the nervous system. Curr Opin Neurobiol. 2004;14:362–369. doi: 10.1016/j.conb.2004.05.003. [DOI] [PubMed] [Google Scholar]
- 11.Montell C, Birnbaumer L, Flockerzi V, Bindels RJ, Bruford EA, Caterina MJ, Clapham DE, Harteneck C, Heller S, Julius D, Kojima I, Mori Y, Penner R, Prawitt D, Scharenberg AM, Schultz G, Shimizu N, Zhu MX. A unified nomenclature for the superfamily of TRP cation channels. Mol Cell. 2002;9:229–231. doi: 10.1016/s1097-2765(02)00448-3. [DOI] [PubMed] [Google Scholar]
- 12.Clapham DE, Runnels LW, Strubing C. The TRP ion channel family. Nat Rev Neurosci. 2001;2:387–396. doi: 10.1038/35077544. [DOI] [PubMed] [Google Scholar]
- 13.Dray A, Bettaney J, Forster P. Actions of capsaicin on peripheral nociceptors of the neonatal rat spinal cord-tail in vitro: dependence of extracellular ions and independence of second messengers. Br J Pharmacol. 1990;101:727–733. doi: 10.1111/j.1476-5381.1990.tb14148.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Dray A, Bettaney J, Rueff A, Walpole C, Wrigglesworth R. NE-19550 and NE-21610, antinociceptive capsaicin analogues: studies on nociceptive fibres of the neonatal rat tail in vitro. Eur J Pharmacol. 1990;181:289–293. doi: 10.1016/0014-2999(90)90091-j. [DOI] [PubMed] [Google Scholar]
- 15.Dray A, Bettaney J, Forster P. Capsaicin desensitization of peripheral nociceptive fibres does not impair sensitivity to other noxious stimuli. Neurosci Lett. 1989;99:50–54. doi: 10.1016/0304-3940(89)90263-2. [DOI] [PubMed] [Google Scholar]
- 16.Cortright DN, Krause JE, Broom DC. TRP channels and pain. Biochim Biophys Acta. 2007;1772:978–988. doi: 10.1016/j.bbadis.2007.03.003. [DOI] [PubMed] [Google Scholar]
- 17.Szallasi A, Cortright DN, Blum CA, Eid SR. The vanilloid receptor TRPV1: 10 years from channel cloning to antagonist proof-of-concept. Nat Rev Drug Discov. 2007;6:357–372. doi: 10.1038/nrd2280. [DOI] [PubMed] [Google Scholar]
- 18.Levine JD, Alessandri-Haber N. TRP channels: targets for the relief of pain. Biochim Biophys Acta. 2007;1772:989–1003. doi: 10.1016/j.bbadis.2007.01.008. [DOI] [PubMed] [Google Scholar]
- 19.Szallasi A, Blumberg PM. Vanilloid (Capsaicin) receptors and mechanisms. Pharmacol Rev. 1999;51:159–212. [PubMed] [Google Scholar]
- 20.Szallasi A, Nilsson S, Farkas-Szallasi T, Blumberg PM, Hokfelt T, Lundberg JM. Vanilloid (capsaicin) receptors in the rat: distribution in the brain, regional differences in the spinal cord, axonal transport to the periphery, and depletion by systemic vanilloid treatment. Brain Res. 1995;703:175–183. doi: 10.1016/0006-8993(95)01094-7. [DOI] [PubMed] [Google Scholar]
- 21.Szallasi A, Blumberg PM. Characterization of vanilloid receptors in the dorsal horn of pig spinal cord. Brain Res. 1991;547:335–338. doi: 10.1016/0006-8993(91)90982-2. [DOI] [PubMed] [Google Scholar]
- 22.Szolcsanyi J, Szallasi A, Szallasi Z, Joo F, Blumberg PM. Resiniferatoxin. An ultrapotent neurotoxin of capsaicin-sensitive primary afferent neurons. Ann N Y Acad Sci. 1991;632:473–475. doi: 10.1111/j.1749-6632.1991.tb33161.x. [DOI] [PubMed] [Google Scholar]
- 23.Szolcsanyi J, Szallasi A, Szallasi Z, Joo F, Blumberg PM. Resiniferatoxin: an ultrapotent selective modulator of capsaicin-sensitive primary afferent neurons. J Pharmacol Exp Ther. 1990;255:923–928. [PubMed] [Google Scholar]
- 24.Fischer MJ, Reeh PW, Sauer SK. Proton-induced calcitonin gene-related peptide release from rat sciatic nerve axons, in vitro, involving TRPV1. Eur J Neurosci. 2003;18:803–810. doi: 10.1046/j.1460-9568.2003.02811.x. [DOI] [PubMed] [Google Scholar]
- 25.Bernardini N, Neuhuber W, Reeh PW, Sauer SK. Morphological evidence for functional capsaicin receptor expression and calcitonin gene-related peptide exocytosis in isolated peripheral nerve axons of the mouse. Neuroscience. 2004;126:585–590. doi: 10.1016/j.neuroscience.2004.03.017. [DOI] [PubMed] [Google Scholar]
- 26.Price TJ, Flores CM. Critical evaluation of the colocalization between calcitonin gene-related peptide, substance P, transient receptor potential vanilloid subfamily type 1 immunoreactivities, and isolectin B4 binding in primary afferent neurons of the rat and mouse. J Pain. 2007;8:263–272. doi: 10.1016/j.jpain.2006.09.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Price TJ, Louria MD, Candelario-Soto D, Dussor GO, Jeske NA, Patwardhan AM, Diogenes A, Trott AA, Hargreaves KM, Flores CM. Treatment of trigeminal ganglion neurons in vitro with NGF, GDNF or BDNF: effects on neuronal survival, neurochemical properties and TRPV1-mediated neuropeptide secretion. BMC Neurosci. 2005;6:4. doi: 10.1186/1471-2202-6-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Mezey E, Toth ZE, Cortright DN, Arzubi MK, Krause JE, Elde R, Guo A, Blumberg PM, Szallasi A. Distribution of mRNA for vanilloid receptor subtype 1 (VR1), and VR1-like immunoreactivity, in the central nervous system of the rat and human. Proc Natl Acad Sci U S A. 2000;97:3655–3660. doi: 10.1073/pnas.060496197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Inoue K, Koizumi S, Fuziwara S, Denda S, Inoue K, Denda M. Functional vanilloid receptors in cultured normal human epidermal keratinocytes. Biochem Biophys Res Commun. 2002;291:124–129. doi: 10.1006/bbrc.2002.6393. [DOI] [PubMed] [Google Scholar]
- 30.Stander S, Moormann C, Schumacher M, Buddenkotte J, Artuc M, Shpacovitch V, Brzoska T, Lippert U, Henz BM, Luger TA, Metze D, Steinhoff M. Expression of vanilloid receptor subtype 1 in cutaneous sensory nerve fibers, mast cells, and epithelial cells of appendage structures. Exp Dermatol. 2004;13:129–139. doi: 10.1111/j.0906-6705.2004.0178.x. [DOI] [PubMed] [Google Scholar]
- 31.Biro T, Bodo E, Telek A, Geczy T, Tychsen B, Kovacs L, Paus R. Hair cycle control by vanilloid receptor-1 (TRPV1): evidence from TRPV1 knockout mice. J Invest Dermatol. 2006;126:1909–1912. doi: 10.1038/sj.jid.5700321. [DOI] [PubMed] [Google Scholar]
- 32.Bodo E, Biro T, Telek A, Czifra G, Griger Z, Toth BI, Mescalchin A, Ito T, Bettermann A, Kovacs L, Paus R. A hot new twist to hair biology: involvement of vanilloid receptor-1 (VR1/TRPV1) signaling in human hair growth control. Am J Pathol. 2005;166:985–998. doi: 10.1016/S0002-9440(10)62320-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Bodo E, Kovacs I, Telek A, Varga A, Paus R, Kovacs L, Biro T. Vanilloid receptor-1 (VR1) is widely expressed on various epithelial and mesenchymal cell types of human skin. J Invest Dermatol. 2004;123:410–413. doi: 10.1111/j.0022-202X.2004.23209.x. [DOI] [PubMed] [Google Scholar]
- 34.Szallasi A, Conte B, Goso C, Blumberg PM, Manzini S. Characterization of a peripheral vanilloid (capsaicin) receptor in the urinary bladder of the rat. Life Sci. 1993;52:PL221–226. doi: 10.1016/0024-3205(93)90051-4. [DOI] [PubMed] [Google Scholar]
- 35.Siegmund SV, Uchinami H, Osawa Y, Brenner DA, Schwabe RF. Anandamide induces necrosis in primary hepatic stellate cells. Hepatology. 2005;41:1085–1095. doi: 10.1002/hep.20667. [DOI] [PubMed] [Google Scholar]
- 36.Russell JA, Lai-Fook SJ. Reflex bronchoconstriction induced by capsaicin in the dog. J Appl Physiol. 1979;47:961–967. doi: 10.1152/jappl.1979.47.5.961. [DOI] [PubMed] [Google Scholar]
- 37.Lundberg JM, Martling CR, Saria A. Substance P and capsaicin-induced contraction of human bronchi. Acta Physiol Scand. 1983;119:49–53. doi: 10.1111/j.1748-1716.1983.tb07304.x. [DOI] [PubMed] [Google Scholar]
- 38.Gunthorpe MJ, Harries MH, Prinjha RK, Davis JB, Randall A. Voltage- and time-dependent properties of the recombinant rat vanilloid receptor (rVR1) J Physiol. 2000;525(Pt 3):747–759. doi: 10.1111/j.1469-7793.2000.t01-1-00747.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Piper AS, Yeats JC, Bevan S, Docherty RJ. A study of the voltage dependence of capsaicin-activated membrane currents in rat sensory neurones before and after acute desensitization. J Physiol. 1999;518 (Pt 3):721–733. doi: 10.1111/j.1469-7793.1999.0721p.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Chuang HH, Prescott ED, Kong H, Shields S, Jordt SE, Basbaum AI, Chao MV, Julius D. Bradykinin and nerve growth factor release the capsaicin receptor from PtdIns(4,5)P2-mediated inhibition. Nature. 2001;411:957–962. doi: 10.1038/35082088. [DOI] [PubMed] [Google Scholar]
- 41.Clapham DE. TRP channels as cellular sensors. Nature. 2003;426:517–524. doi: 10.1038/nature02196. [DOI] [PubMed] [Google Scholar]
- 42.Cortright DN, Szallasi A. Biochemical pharmacology of the vanilloid receptor TRPV1. An update. Eur J Biochem. 2004;271:1814–1819. doi: 10.1111/j.1432-1033.2004.04082.x. [DOI] [PubMed] [Google Scholar]
- 43.Prescott ED, Julius D. A modular PIP2 binding site as a determinant of capsaicin receptor sensitivity. Science. 2003;300:1284–1288. doi: 10.1126/science.1083646. [DOI] [PubMed] [Google Scholar]
- 44.Jung J, Shin JS, Lee SY, Hwang SW, Koo J, Cho H, Oh U. Phosphorylation of vanilloid receptor 1 by Ca2+/calmodulin-dependent kinase II regulates its vanilloid binding. J Biol Chem. 2004;279:7048–7054. doi: 10.1074/jbc.M311448200. [DOI] [PubMed] [Google Scholar]
- 45.Bhave G, Hu HJ, Glauner KS, Zhu W, Wang H, Brasier DJ, Oxford GS, Gereau RWt. Protein kinase C phosphorylation sensitizes but does not activate the capsaicin receptor transient receptor potential vanilloid 1 (TRPV1) Proc Natl Acad Sci U S A. 2003;100:12480–12485. doi: 10.1073/pnas.2032100100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Macpherson LJ, Geierstanger BH, Viswanath V, Bandell M, Eid SR, Hwang S, Patapoutian A. The pungency of garlic: activation of TRPA1 and TRPV1 in response to allicin. Curr Biol. 2005;15:929–934. doi: 10.1016/j.cub.2005.04.018. [DOI] [PubMed] [Google Scholar]
- 47.Rosenbaum T, Castanares DT, Lopez-Valdes HE, Hiriart M. Nerve growth factor increases L-type calcium current in pancreatic beta cells in culture. J Membr Biol. 2002;186:177–184. doi: 10.1007/s00232-001-0143-9. [DOI] [PubMed] [Google Scholar]
- 48.Kedei N, Szabo T, Lile JD, Treanor JJ, Olah Z, Iadarola MJ, Blumberg PM. Analysis of the native quaternary structure of vanilloid receptor 1. J Biol Chem. 2001;276:28613–28619. doi: 10.1074/jbc.M103272200. [DOI] [PubMed] [Google Scholar]
- 49.Tominaga M, Tominaga T. Structure and function of TRPV1. Pflugers Arch. 2005;451:143–150. doi: 10.1007/s00424-005-1457-8. [DOI] [PubMed] [Google Scholar]
- 50.Ferrer-Montiel A, Garcia-Martinez C, Morenilla-Palao C, Garcia-Sanz N, Fernandez-Carvajal A, Fernandez-Ballester G, Planells-Cases R. Molecular architecture of the vanilloid receptor. Insights for drug design. Eur J Biochem. 2004;271:1820–1826. doi: 10.1111/j.1432-1033.2004.04083.x. [DOI] [PubMed] [Google Scholar]
- 51.Reaves BJ, Wolstenholme AJ. The TRP channel superfamily: insights into how structure, protein-lipid interactions and localization influence function. Biochem Soc Trans. 2007;35:77–80. doi: 10.1042/BST0350077. [DOI] [PubMed] [Google Scholar]
- 52.Owsianik G, Talavera K, Voets T, Nilius B. Permeation and selectivity of TRP channels. Annu Rev Physiol. 2006;68:685–717. doi: 10.1146/annurev.physiol.68.040204.101406. [DOI] [PubMed] [Google Scholar]
- 53.Oseguera AJ, Islas LD, Garcia-Villegas R, Rosenbaum T. On the mechanism of TBA block of the TRPV1 channel. Biophys J. 2007;92:3901–3914. doi: 10.1529/biophysj.106.102400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Voets T, Droogmans G, Wissenbach U, Janssens A, Flockerzi V, Nilius B. The principle of temperature-dependent gating in cold- and heat-sensitive TRP channels. Nature. 2004;430:748–754. doi: 10.1038/nature02732. [DOI] [PubMed] [Google Scholar]
- 55.Lu G, Henderson D, Liu L, Reinhart PH, Simon SA. TRPV1b, a functional human vanilloid receptor splice variant. Mol Pharmacol. 2005;67:1119–1127. doi: 10.1124/mol.104.009852. [DOI] [PubMed] [Google Scholar]
- 56.Vos MH, Neelands TR, McDonald HA, Choi W, Kroeger PE, Puttfarcken PS, Faltynek CR, Moreland RB, Han P. TRPV1b overexpression negatively regulates TRPV1 responsiveness to capsaicin, heat and low pH in HEK293 cells. J Neurochem. 2006;99:1088–1102. doi: 10.1111/j.1471-4159.2006.04145.x. [DOI] [PubMed] [Google Scholar]
- 57.Tian W, Fu Y, Wang DH, Cohen DM. Regulation of TRPV1 by a novel renally expressed rat TRPV1 splice variant. Am J Physiol Renal Physiol. 2006;290:F117–126. doi: 10.1152/ajprenal.00143.2005. [DOI] [PubMed] [Google Scholar]
- 58.Schumacher MA, Moff I, Sudanagunta SP, Levine JD. Molecular cloning of an N-terminal splice variant of the capsaicin receptor. Loss of N-terminal domain suggests functional divergence among capsaicin receptor subtypes. J Biol Chem. 2000;275:2756–2762. doi: 10.1074/jbc.275.4.2756. [DOI] [PubMed] [Google Scholar]
- 59.Eilers H, Lee SY, Hau CW, Logvinova A, Schumacher MA. The rat vanilloid receptor splice variant VR.5′sv blocks TRPV1 activation. Neuroreport. 2007;18:969–973. doi: 10.1097/WNR.0b013e328165d1a2. [DOI] [PubMed] [Google Scholar]
- 60.Wang C, Hu HZ, Colton CK, Wood JD, Zhu MX. An alternative splicing product of the murine trpv1 gene dominant negatively modulates the activity of TRPV1 channels. J Biol Chem. 2004;279:37423–37430. doi: 10.1074/jbc.M407205200. [DOI] [PubMed] [Google Scholar]
- 61.Szallasi A, Blumberg PM. Resiniferatoxin, a phorbol-related diterpene, acts as an ultrapotent analog of capsaicin, the irritant constituent in red pepper. Neuroscience. 1989;30:515–520. doi: 10.1016/0306-4522(89)90269-8. [DOI] [PubMed] [Google Scholar]
- 62.Szallasi A, Joo F, Blumberg PM. Duration of desensitization and ultrastructural changes in dorsal root ganglia in rats treated with resiniferatoxin, an ultrapotent capsaicin analog. Brain Res. 1989;503:68–72. doi: 10.1016/0006-8993(89)91705-8. [DOI] [PubMed] [Google Scholar]
- 63.Latorre R, Brauchi S, Orta G, Zaelzer C, Vargas G. ThermoTRP channels as modular proteins with allosteric gating. Cell Calcium. 2007;42:427–438. doi: 10.1016/j.ceca.2007.04.004. [DOI] [PubMed] [Google Scholar]
- 64.Hwang SW, Cho H, Kwak J, Lee SY, Kang CJ, Jung J, Cho S, Min KH, Suh YG, Kim D, Oh U. Direct activation of capsaicin receptors by products of lipoxygenases: endogenous capsaicin-like substances. Proc Natl Acad Sci U S A. 2000;97:6155–6160. doi: 10.1073/pnas.97.11.6155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Price TJ, Patwardhan A, Akopian AN, Hargreaves KM, Flores CM. Modulation of trigeminal sensory neuron activity by the dual cannabinoid-vanilloid agonists anandamide, N-arachidonoyl-dopamine and arachidonyl-2-chloroethylamide. Br J Pharmacol. 2004;141:1118–1130. doi: 10.1038/sj.bjp.0705711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Premkumar LS, Qi ZH, Van Buren J, Raisinghani M. Enhancement of potency and efficacy of NADA by PKC-mediated phosphorylation of vanilloid receptor. J Neurophysiol. 2004;91:1442–1449. doi: 10.1152/jn.00745.2003. [DOI] [PubMed] [Google Scholar]
- 67.Suh YG, Oh U. Activation and activators of TRPV1 and their pharmaceutical implication. Curr Pharm Des. 2005;11:2687–2698. doi: 10.2174/1381612054546789. [DOI] [PubMed] [Google Scholar]
- 68.Liu L, Simon SA. Similarities and differences in the currents activated by capsaicin, piperine, and zingerone in rat trigeminal ganglion cells. J Neurophysiol. 1996;76:1858–1869. doi: 10.1152/jn.1996.76.3.1858. [DOI] [PubMed] [Google Scholar]
- 69.McNamara FN, Randall A, Gunthorpe MJ. Effects of piperine, the pungent component of black pepper, at the human vanilloid receptor (TRPV1) Br J Pharmacol. 2005;144:781–790. doi: 10.1038/sj.bjp.0706040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Dedov VN, Tran VH, Duke CC, Connor M, Christie MJ, Mandadi S, Roufogalis BD. Gingerols: a novel class of vanilloid receptor (VR1) agonists. Br J Pharmacol. 2002;137:793–798. doi: 10.1038/sj.bjp.0704925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Witte DG, Cassar SC, Masters JN, Esbenshade T, Hancock AA. Use of a fluorescent imaging plate reader--based calcium assay to assess pharmacological differences between the human and rat vanilloid receptor. J Biomol Screen. 2002;7:466–475. doi: 10.1177/108705702237679. [DOI] [PubMed] [Google Scholar]
- 72.Iwasaki Y, Morita A, Iwasawa T, Kobata K, Sekiwa Y, Morimitsu Y, Kubota K, Watanabe T. A nonpungent component of steamed ginger--[10]-shogaol--increases adrenaline secretion via the activation of TRPV1. Nutr Neurosci. 2006;9:169–178. doi: 10.1080/110284150600955164. [DOI] [PubMed] [Google Scholar]
- 73.Yang BH, Piao ZG, Kim YB, Lee CH, Lee JK, Park K, Kim JS, Oh SB. Activation of vanilloid receptor 1 (VR1) by eugenol. J Dent Res. 2003;82:781–785. doi: 10.1177/154405910308201004. [DOI] [PubMed] [Google Scholar]
- 74.Jung J, Hwang SW, Kwak J, Lee SY, Kang CJ, Kim WB, Kim D, Oh U. Capsaicin binds to the intracellular domain of the capsaicin-activated ion channel. J Neurosci. 1999;19:529–538. doi: 10.1523/JNEUROSCI.19-02-00529.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Jung J, Lee SY, Hwang SW, Cho H, Shin J, Kang YS, Kim S, Oh U. Agonist recognition sites in the cytosolic tails of vanilloid receptor 1. J Biol Chem. 2002;277:44448–44454. doi: 10.1074/jbc.M207103200. [DOI] [PubMed] [Google Scholar]
- 76.Chou MZ, Mtui T, Gao YD, Kohler M, Middleton RE. Resiniferatoxin binds to the capsaicin receptor (TRPV1) near the extracellular side of the S4 transmembrane domain. Biochemistry. 2004;43:2501–2511. doi: 10.1021/bi035981h. [DOI] [PubMed] [Google Scholar]
- 77.Gavva NR, Klionsky L, Qu Y, Shi L, Tamir R, Edenson S, Zhang TJ, Viswanadhan VN, Toth A, Pearce LV, Vanderah TW, Porreca F, Blumberg PM, Lile J, Sun Y, Wild K, Louis JC, Treanor JJ. Molecular determinants of vanilloid sensitivity in TRPV1. J Biol Chem. 2004;279:20283–20295. doi: 10.1074/jbc.M312577200. [DOI] [PubMed] [Google Scholar]
- 78.Bautista DM, Movahed P, Hinman A, Axelsson HE, Sterner O, Hogestatt ED, Julius D, Jordt SE, Zygmunt PM. Pungent products from garlic activate the sensory ion channel TRPA1. Proc Natl Acad Sci U S A. 2005;102:12248–12252. doi: 10.1073/pnas.0505356102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Bautista DM, Jordt SE, Nikai T, Tsuruda PR, Read AJ, Poblete J, Yamoah EN, Basbaum AI, Julius D. TRPA1 mediates the inflammatory actions of environmental irritants and proalgesic agents. Cell. 2006;124:1269–1282. doi: 10.1016/j.cell.2006.02.023. [DOI] [PubMed] [Google Scholar]
- 80.Hinman A, Chuang HH, Bautista DM, Julius D. TRP channel activation by reversible covalent modification. Proc Natl Acad Sci U S A. 2006;103:19564–19568. doi: 10.1073/pnas.0609598103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Bandell M, Story GM, Hwang SW, Viswanath V, Eid SR, Petrus MJ, Earley TJ, Patapoutian A. Noxious cold ion channel TRPA1 is activated by pungent compounds and bradykinin. Neuron. 2004;41:849–857. doi: 10.1016/s0896-6273(04)00150-3. [DOI] [PubMed] [Google Scholar]
- 82.Macpherson LJ, Dubin AE, Evans MJ, Marr F, Schultz PG, Cravatt BF, Patapoutian A. Noxious compounds activate TRPA1 ion channels through covalent modification of cysteines. Nature. 2007;445:541–545. doi: 10.1038/nature05544. [DOI] [PubMed] [Google Scholar]
- 83.Kim D, Cavanaugh EJ. Requirement of a soluble intracellular factor for activation of transient receptor potential A1 by pungent chemicals: role of inorganic polyphosphates. J Neurosci. 2007;27:6500–6509. doi: 10.1523/JNEUROSCI.0623-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Lishko PV, Procko E, Jin X, Phelps CB, Gaudet R. The ankyrin repeats of TRPV1 bind multiple ligands and modulate channel sensitivity. Neuron. 2007;54:905–918. doi: 10.1016/j.neuron.2007.05.027. [DOI] [PubMed] [Google Scholar]
- 85.Xu H, Blair NT, Clapham DE. Camphor activates and strongly desensitizes the transient receptor potential vanilloid subtype 1 channel in a vanilloid-independent mechanism. J Neurosci. 2005;25:8924–8937. doi: 10.1523/JNEUROSCI.2574-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Nagae M, Hiraga T, Yoneda T. Acidic microenvironment created by osteoclasts causes bone pain associated with tumor colonization. J Bone Miner Metab. 2007;25:99–104. doi: 10.1007/s00774-006-0734-8. [DOI] [PubMed] [Google Scholar]
- 87.Ugawa S, Ueda T, Ishida Y, Nishigaki M, Shibata Y, Shimada S. Amiloride-blockable acid-sensing ion channels are leading acid sensors expressed in human nociceptors. J Clin Invest. 2002;110:1185–1190. doi: 10.1172/JCI15709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Jordt SE, Tominaga M, Julius D. Acid potentiation of the capsaicin receptor determined by a key extracellular site. Proc Natl Acad Sci U S A. 2000;97:8134–8139. doi: 10.1073/pnas.100129497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Welch JM, Simon SA, Reinhart PH. The activation mechanism of rat vanilloid receptor 1 by capsaicin involves the pore domain and differs from the activation by either acid or heat. Proc Natl Acad Sci U S A. 2000;97:13889–13894. doi: 10.1073/pnas.230146497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Ryu S, Liu B, Qin F. Low pH potentiates both capsaicin binding and channel gating of VR1 receptors. J Gen Physiol. 2003;122:45–61. doi: 10.1085/jgp.200308847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Reeh PW, Kress M. Molecular physiology of proton transduction in nociceptors. Curr Opin Pharmacol. 2001;1:45–51. doi: 10.1016/s1471-4892(01)00014-5. [DOI] [PubMed] [Google Scholar]
- 92.Jaffrey SR, Erdjument-Bromage H, Ferris CD, Tempst P, Snyder SH. Protein S-nitrosylation: a physiological signal for neuronal nitric oxide. Nat Cell Biol. 2001;3:193–197. doi: 10.1038/35055104. [DOI] [PubMed] [Google Scholar]
- 93.Hess DT, Matsumoto A, Kim SO, Marshall HE, Stamler JS. Protein S-nitrosylation: purview and parameters. Nat Rev Mol Cell Biol. 2005;6:150–166. doi: 10.1038/nrm1569. [DOI] [PubMed] [Google Scholar]
- 94.Yoshida T, Inoue R, Morii T, Takahashi N, Yamamoto S, Hara Y, Tominaga M, Shimizu S, Sato Y, Mori Y. Nitric oxide activates TRP channels by cysteine S-nitrosylation. Nat Chem Biol. 2006;2:596–607. doi: 10.1038/nchembio821. [DOI] [PubMed] [Google Scholar]
- 95.Moncada S, Higgs A, Furchgott R. International Union of Pharmacology Nomenclature in Nitric Oxide Research. Pharmacol Rev. 1997;49:137–142. [PubMed] [Google Scholar]
- 96.Holzer P. Local effector functions of capsaicin-sensitive sensory nerve endings: involvement of tachykinins, calcitonin gene-related peptide and other neuropeptides. Neuroscience. 1988;24:739–768. doi: 10.1016/0306-4522(88)90064-4. [DOI] [PubMed] [Google Scholar]
- 97.Vlachova V, Teisinger J, Susankova K, Lyfenko A, Ettrich R, Vyklicky L. Functional role of C-terminal cytoplasmic tail of rat vanilloid receptor 1. J Neurosci. 2003;23:1340–1350. doi: 10.1523/JNEUROSCI.23-04-01340.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Brauchi S, Orta G, Salazar M, Rosenmann E, Latorre R. A hot-sensing cold receptor: C-terminal domain determines thermosensation in transient receptor potential channels. J Neurosci. 2006;26:4835–4840. doi: 10.1523/JNEUROSCI.5080-05.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Nilius B, Talavera K, Owsianik G, Prenen J, Droogmans G, Voets T. Gating of TRP channels: a voltage connection? J Physiol. 2005;567:35–44. doi: 10.1113/jphysiol.2005.088377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Riera CE, Vogel H, Simon SA, le Coutre J. Artificial sweeteners and salts producing a metallic taste sensation activate TRPV1 receptors. Am J Physiol Regul Integr Comp Physiol. 2007;293:R626–634. doi: 10.1152/ajpregu.00286.2007. [DOI] [PubMed] [Google Scholar]
- 101.Ahern GP, Brooks IM, Miyares RL, Wang XB. Extracellular cations sensitize and gate capsaicin receptor TRPV1 modulating pain signaling. J Neurosci. 2005;25:5109–5116. doi: 10.1523/JNEUROSCI.0237-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Wallace HM. Targeting polyamine metabolism: a viable therapeutic/preventative solution for cancer? Expert Opin Pharmacother. 2007;8:2109–2116. doi: 10.1517/14656566.8.13.2109. [DOI] [PubMed] [Google Scholar]
- 103.Li J, Doyle KM, Tatlisumak T. Polyamines in the brain: distribution, biological interactions, and their potential therapeutic role in brain ischaemia. Curr Med Chem. 2007;14:1807–1813. doi: 10.2174/092986707781058841. [DOI] [PubMed] [Google Scholar]
- 104.Medina MA, Correa-Fiz F, Rodriguez-Caso C, Sanchez-Jimenez F. A comprehensive view of polyamine and histamine metabolism to the light of new technologies. J Cell Mol Med. 2005;9:854–864. doi: 10.1111/j.1582-4934.2005.tb00384.x. [DOI] [PubMed] [Google Scholar]
- 105.Ahern GP, Wang X, Miyares RL. Polyamines are potent ligands for the capsaicin receptor TRPV1. J Biol Chem. 2006;281:8991–8995. doi: 10.1074/jbc.M513429200. [DOI] [PubMed] [Google Scholar]
- 106.Siemens J, Zhou S, Piskorowski R, Nikai T, Lumpkin EA, Basbaum AI, King D, Julius D. Spider toxins activate the capsaicin receptor to produce inflammatory pain. Nature. 2006;444:208–212. doi: 10.1038/nature05285. [DOI] [PubMed] [Google Scholar]
- 107.Kitaguchi T, Swartz KJ. An inhibitor of TRPV1 channels isolated from funnel Web spider venom. Biochemistry. 2005;44:15544–15549. doi: 10.1021/bi051494l. [DOI] [PubMed] [Google Scholar]
- 108.Binshtok AM, Bean BP, Woolf CJ. Inhibition of nociceptors by TRPV1-mediated entry of impermeant sodium channel blockers. Nature. 2007;449:607–610. doi: 10.1038/nature06191. [DOI] [PubMed] [Google Scholar]
- 109.Halaszovich CR, Zitt C, Jungling E, Luckhoff A. Inhibition of TRP3 channels by lanthanides. Block from the cytosolic side of the plasma membrane. J Biol Chem. 2000;275:37423–37428. doi: 10.1074/jbc.M007010200. [DOI] [PubMed] [Google Scholar]
- 110.Hamill OP, McBride DW., Jr The pharmacology of mechanogated membrane ion channels. Pharmacol Rev. 1996;48:231–252. [PubMed] [Google Scholar]
- 111.Cho H, Shin J, Shin CY, Lee SY, Oh U. Mechanosensitive ion channels in cultured sensory neurons of neonatal rats. J Neurosci. 2002;22:1238–1247. doi: 10.1523/JNEUROSCI.22-04-01238.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Yang XC, Sachs F. Block of stretch-activated ion channels in Xenopus oocytes by gadolinium and calcium ions. Science. 1989;243:1068–1071. doi: 10.1126/science.2466333. [DOI] [PubMed] [Google Scholar]
- 113.Vyklicky L, Lyfenko A, Susankova K, Teisinger J, Vlachova V. Reducing agent dithiothreitol facilitates activity of the capsaicin receptor VR-1. Neuroscience. 2002;111:435–441. doi: 10.1016/s0306-4522(02)00051-9. [DOI] [PubMed] [Google Scholar]
- 114.Susankova K, Tousova K, Vyklicky L, Teisinger J, Vlachova V. Reducing and oxidizing agents sensitize heat-activated vanilloid receptor (TRPV1) current. Mol Pharmacol. 2006;70:383–394. doi: 10.1124/mol.106.023069. [DOI] [PubMed] [Google Scholar]
- 115.Jancso N. Pharmacological analysis of the function and receptor structure of the pain-sensitive nerve endings. Acta Physiol Hung. 1957;11:11–14. [PubMed] [Google Scholar]
- 116.Porszasz J, Jancso N. Studies on the action potentials of sensory nerves in animals desensitized with capsaicine. Acta Physiol Acad Sci Hung. 1959;16:299–306. [PubMed] [Google Scholar]
- 117.Jancso-Gabor A, Szolcsanyi J, Jancso N. Stimulation and desensitization of the hypothalamic heat-sensitive structures by capsaicin in rats. J Physiol. 1970;208:449–459. doi: 10.1113/jphysiol.1970.sp009130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Jancso N, Jancso-Gabor A, Takats I. Pain and inflammation induced by nicotine, acetylcholine and structurally related compounds and their prevention by desensitizing agents. Acta Physiol Acad Sci Hung. 1961;19:113–132. [PubMed] [Google Scholar]
- 119.Schwarz S, Greffrath W, Busselberg D, Treede RD. Inactivation and tachyphylaxis of heat-evoked inward currents in nociceptive primary sensory neurones of rats. J Physiol. 2000;528:539–549. doi: 10.1111/j.1469-7793.2000.00539.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Rosenbaum T, Gordon-Shaag A, Munari M, Gordon SE. Ca2+/calmodulin modulates TRPV1 activation by capsaicin. J Gen Physiol. 2004;123:53–62. doi: 10.1085/jgp.200308906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Liu L, Simon SA. Capsaicin-induced currents with distinct desensitization and Ca2+ dependence in rat trigeminal ganglion cells. J Neurophysiol. 1996;75:1503–1514. doi: 10.1152/jn.1996.75.4.1503. [DOI] [PubMed] [Google Scholar]
- 122.Liu L, Simon SA. The influence of removing extracellular Ca2+ in the desensitization responses to capsaicin, zingerone and olvanil in rat trigeminal ganglion neurons. Brain Res. 1998;809:246–252. doi: 10.1016/s0006-8993(98)00853-1. [DOI] [PubMed] [Google Scholar]
- 123.Koplas PA, Rosenberg RL, Oxford GS. The role of calcium in the desensitization of capsaicin responses in rat dorsal root ganglion neurons. J Neurosci. 1997;17:3525–3537. doi: 10.1523/JNEUROSCI.17-10-03525.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Docherty RJ, Yeats JC, Bevan S, Boddeke HW. Inhibition of calcineurin inhibits the desensitization of capsaicin-evoked currents in cultured dorsal root ganglion neurones from adult rats. Pflugers Arch. 1996;431:828–837. doi: 10.1007/s004240050074. [DOI] [PubMed] [Google Scholar]
- 125.Tominaga M, Wada M, Masu M. Potentiation of capsaicin receptor activity by metabotropic ATP receptors as a possible mechanism for ATP-evoked pain and hyperalgesia. Proc Natl Acad Sci U S A. 2001;98:6951–6956. doi: 10.1073/pnas.111025298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Moriyama T, Iida T, Kobayashi K, Higashi T, Fukuoka T, Tsumura H, Leon C, Suzuki N, Inoue K, Gachet C, Noguchi K, Tominaga M. Possible involvement of P2Y2 metabotropic receptors in ATP-induced transient receptor potential vanilloid receptor 1-mediated thermal hypersensitivity. J Neurosci. 2003;23:6058–6062. doi: 10.1523/JNEUROSCI.23-14-06058.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Zhang N, Inan S, Cowan A, Sun R, Wang JM, Rogers TJ, Caterina M, Oppenheim JJ. A proinflammatory chemokine, CCL3, sensitizes the heat- and capsaicin-gated ion channel TRPV1. Proc Natl Acad Sci U S A. 2005;102:4536–4541. doi: 10.1073/pnas.0406030102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Liu B, Zhang C, Qin F. Functional recovery from desensitization of vanilloid receptor TRPV1 requires resynthesis of phosphatidylinositol 4,5-bisphosphate. J Neurosci. 2005;25:4835–4843. doi: 10.1523/JNEUROSCI.1296-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Lukacs V, Thyagarajan B, Varnai P, Balla A, Balla T, Rohacs T. Dual regulation of TRPV1 by phosphoinositides. J Neurosci. 2007;27:7070–7080. doi: 10.1523/JNEUROSCI.1866-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Stein AT, Ufret-Vincenty CA, Hua L, Santana LF, Gordon SE. Phosphoinositide 3-kinase binds to TRPV1 and mediates NGF-stimulated TRPV1 trafficking to the plasma membrane. J Gen Physiol. 2006;128:509–522. doi: 10.1085/jgp.200609576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Brauchi S, Orta G, Mascayano C, Salazar M, Raddatz N, Urbina H, Rosenmann E, Gonzalez-Nilo F, Latorre R. Dissection of the components for PIP2 activation and thermosensation in TRP channels. Proc Natl Acad Sci U S A. 2007;104:10246–10251. doi: 10.1073/pnas.0703420104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Zhang X, Huang J, McNaughton PA. NGF rapidly increases membrane expression of TRPV1 heat-gated ion channels. Embo J. 2005;24:4211–4223. doi: 10.1038/sj.emboj.7600893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Ahern GP. Activation of TRPV1 by the satiety factor oleoylethanolamide. J Biol Chem. 2003;278:30429–30434. doi: 10.1074/jbc.M305051200. [DOI] [PubMed] [Google Scholar]
- 134.Zygmunt PM, Petersson J, Andersson DA, Chuang H, Sorgard M, Di Marzo V, Julius D, Hogestatt ED. Vanilloid receptors on sensory nerves mediate the vasodilator action of anandamide. Nature. 1999;400:452–457. doi: 10.1038/22761. [DOI] [PubMed] [Google Scholar]
- 135.Matta JA, Miyares RL, Ahern GP. TRPV1 is a novel target for omega-3 polyunsaturated fatty acids. J Physiol. 2007;578:397–411. doi: 10.1113/jphysiol.2006.121988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Rukwied R, Chizh BA, Lorenz U, Obreja O, Margarit S, Schley M, Schmelz M. Potentiation of nociceptive responses to low pH injections in humans by prostaglandin E2. J Pain. 2007;8:443–451. doi: 10.1016/j.jpain.2006.12.004. [DOI] [PubMed] [Google Scholar]
- 137.Moriyama T, Higashi T, Togashi K, Iida T, Segi E, Sugimoto Y, Tominaga T, Narumiya S, Tominaga M. Sensitization of TRPV1 by EP1 and IP reveals peripheral nociceptive mechanism of prostaglandins. Mol Pain. 2005;1:3. doi: 10.1186/1744-8069-1-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Wu ZZ, Pan HL. Role of TRPV1 and intracellular Ca2+ in excitation of cardiac sensory neurons by bradykinin. Am J Physiol Regul Integr Comp Physiol. 2007;293:R276–283. doi: 10.1152/ajpregu.00094.2007. [DOI] [PubMed] [Google Scholar]
- 139.Premkumar LS, Ahern GP. Induction of vanilloid receptor channel activity by protein kinase C. Nature. 2000;408:985–990. doi: 10.1038/35050121. [DOI] [PubMed] [Google Scholar]
- 140.Vellani V, Mapplebeck S, Moriondo A, Davis JB, McNaughton PA. Protein kinase C activation potentiates gating of the vanilloid receptor VR1 by capsaicin, protons, heat and anandamide. J Physiol. 2001;534:813–825. doi: 10.1111/j.1469-7793.2001.00813.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Ohta T, Ikemi Y, Murakami M, Imagawa T, Otsuguro K, Ito S. Potentiation of transient receptor potential V1 functions by the activation of metabotropic 5-HT receptors in rat primary sensory neurons. J Physiol. 2006;576:809–822. doi: 10.1113/jphysiol.2006.112250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Sugiuar T, Bielefeldt K, Gebhart GF. TRPV1 function in mouse colon sensory neurons is enhanced by metabotropic 5-hydroxytryptamine receptor activation. J Neurosci. 2004;24:9521–9530. doi: 10.1523/JNEUROSCI.2639-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Shim WS, Tak MH, Lee MH, Kim M, Kim M, Koo JY, Lee CH, Kim M, Oh U. TRPV1 mediates histamine-induced itching via the activation of phospholipase A2 and 12-lipoxygenase. J Neurosci. 2007;27:2331–2337. doi: 10.1523/JNEUROSCI.4643-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Kim BM, Lee SH, Shim WS, Oh U. Histamine-induced Ca(2+) influx via the PLA(2)/lipoxygenase/TRPV1 pathway in rat sensory neurons. Neurosci Lett. 2004;361:159–162. doi: 10.1016/j.neulet.2004.01.019. [DOI] [PubMed] [Google Scholar]
- 145.Shin J, Cho H, Hwang SW, Jung J, Shin CY, Lee SY, Kim SH, Lee MG, Choi YH, Kim J, Haber NA, Reichling DB, Khasar S, Levine JD, Oh U. Bradykinin-12-lipoxygenase-VR1 signaling pathway for inflammatory hyperalgesia. Proc Natl Acad Sci U S A. 2002;99:10150–10155. doi: 10.1073/pnas.152002699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Cesare P, Moriondo A, Vellani V, McNaughton PA. Ion channels gated by heat. Proc Natl Acad Sci U S A. 1999;96:7658–7663. doi: 10.1073/pnas.96.14.7658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Bhave G, Zhu W, Wang H, Brasier DJ, Oxford GS, Gereau RWt. cAMP-dependent protein kinase regulates desensitization of the capsaicin receptor (VR1) by direct phosphorylation. Neuron. 2002;35:721–731. doi: 10.1016/s0896-6273(02)00802-4. [DOI] [PubMed] [Google Scholar]
- 148.Mohapatra DP, Nau C. Desensitization of capsaicin-activated currents in the vanilloid receptor TRPV1 is decreased by the cyclic AMP-dependent protein kinase pathway. J Biol Chem. 2003;278:50080–50090. doi: 10.1074/jbc.M306619200. [DOI] [PubMed] [Google Scholar]
- 149.Mohapatra DP, Wang SY, Wang GK, Nau C. A tyrosine residue in TM6 of the Vanilloid Receptor TRPV1 involved in desensitization and calcium permeability of capsaicin-activated currents. Mol Cell Neurosci. 2003;23:314–324. doi: 10.1016/s1044-7431(03)00054-x. [DOI] [PubMed] [Google Scholar]
- 150.Rathee PK, Distler C, Obreja O, Neuhuber W, Wang GK, Wang SY, Nau C, Kress M. PKA/AKAP/VR-1 module: A common link of Gs-mediated signaling to thermal hyperalgesia. J Neurosci. 2002;22:4740–4745. doi: 10.1523/JNEUROSCI.22-11-04740.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Vetter I, Wyse BD, Monteith GR, Roberts-Thomson SJ, Cabot PJ. The mu opioid agonist morphine modulates potentiation of capsaicin-evoked TRPV1 responses through a cyclic AMP-dependent protein kinase A pathway. Mol Pain. 2006;2:22. doi: 10.1186/1744-8069-2-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Numazaki M, Tominaga M. Nociception and TRP Channels. Curr Drug Targets CNS Neurol Disord. 2004;3:479–485. doi: 10.2174/1568007043336789. [DOI] [PubMed] [Google Scholar]
- 153.Mandadi S, Numazaki M, Tominaga M, Bhat MB, Armati PJ, Roufogalis BD. Activation of protein kinase C reverses capsaicin-induced calcium-dependent desensitization of TRPV1 ion channels. Cell Calcium. 2004;35:471–478. doi: 10.1016/j.ceca.2003.11.003. [DOI] [PubMed] [Google Scholar]
- 154.Morenilla-Palao C, Planells-Cases R, Garcia-Sanz N, Ferrer-Montiel A. Regulated exocytosis contributes to protein kinase C potentiation of vanilloid receptor activity. J Biol Chem. 2004;279:25665–25672. doi: 10.1074/jbc.M311515200. [DOI] [PubMed] [Google Scholar]
- 155.Goswami C, Dreger M, Jahnel R, Bogen O, Gillen C, Hucho F. Identification and characterization of a Ca2+-sensitive interaction of the vanilloid receptor TRPV1 with tubulin. J Neurochem. 2004;91:1092–1103. doi: 10.1111/j.1471-4159.2004.02795.x. [DOI] [PubMed] [Google Scholar]
- 156.Goswami C, Dreger M, Otto H, Schwappach B, Hucho F. Rapid disassembly of dynamic microtubules upon activation of the capsaicin receptor TRPV1. J Neurochem. 2006;96:254–266. doi: 10.1111/j.1471-4159.2005.03551.x. [DOI] [PubMed] [Google Scholar]
- 157.Goswami C, Schmidt H, Hucho F. TRPV1 at nerve endings regulates growth cone morphology and movement through cytoskeleton reorganization. Febs J. 2007;274:760–772. doi: 10.1111/j.1742-4658.2006.05621.x. [DOI] [PubMed] [Google Scholar]
- 158.Goswami C, Hucho TB, Hucho F. Identification and characterisation of novel tubulin-binding motifs located within the C-terminus of TRPV1. J Neurochem. 2007;101:250–262. doi: 10.1111/j.1471-4159.2006.04338.x. [DOI] [PubMed] [Google Scholar]
- 159.Goswami C, Hucho T. TRPV1 expression-dependent initiation and regulation of filopodia. J Neurochem. 2007 doi: 10.1111/j.1471-4159.2007.04846.x. [DOI] [PubMed] [Google Scholar]
- 160.Dina OA, McCarter GC, de Coupade C, Levine JD. Role of the sensory neuron cytoskeleton in second messenger signaling for inflammatory pain. Neuron. 2003;39:613–624. doi: 10.1016/s0896-6273(03)00473-2. [DOI] [PubMed] [Google Scholar]
- 161.Szallasi A, Cruz F, Geppetti P. TRPV1: a therapeutic target for novel analgesic drugs? Trends Mol Med. 2006;12:545–554. doi: 10.1016/j.molmed.2006.09.001. [DOI] [PubMed] [Google Scholar]
- 162.Richardson JD, Vasko MR. Cellular mechanisms of neurogenic inflammation. J Pharmacol Exp Ther. 2002;302:839–845. doi: 10.1124/jpet.102.032797. [DOI] [PubMed] [Google Scholar]
- 163.Brain SD. Sensory neuropeptides: their role in inflammation and wound healing. Immunopharmacology. 1997;37:133–152. doi: 10.1016/s0162-3109(97)00055-6. [DOI] [PubMed] [Google Scholar]
- 164.Szolcsanyi J. Capsaicin-sensitive sensory nerve terminals with local and systemic efferent functions: facts and scopes of an unorthodox neuroregulatory mechanism. Prog Brain Res. 1996;113:343–359. doi: 10.1016/s0079-6123(08)61097-3. [DOI] [PubMed] [Google Scholar]
- 165.Nilius B, Voets T, Peters J. TRP channels in disease. Sci STKE. 2005:re8. doi: 10.1126/stke.2952005re8. [DOI] [PubMed] [Google Scholar]
- 166.Nilius B, Owsianik G, Voets T, Peters JA. Transient receptor potential cation channels in disease. Physiol Rev. 2007;87:165–217. doi: 10.1152/physrev.00021.2006. [DOI] [PubMed] [Google Scholar]
- 167.McHugh D, McMaster RS, Pertwee RG, Roy S, Mahadevan A, Razdan RK, Ross RA. Novel compounds that interact with both leukotriene B4 receptors and vanilloid TRPV1 receptors. J Pharmacol Exp Ther. 2006;316:955–965. doi: 10.1124/jpet.105.095992. [DOI] [PubMed] [Google Scholar]
- 168.Birder LA. TRPs in bladder diseases. Biochim Biophys Acta. 2007;1772:879–884. doi: 10.1016/j.bbadis.2007.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Gopinath P, Wan E, Holdcroft A, Facer P, Davis JB, Smith GD, Bountra C, Anand P. Increased capsaicin receptor TRPV1 in skin nerve fibres and related vanilloid receptors TRPV3 and TRPV4 in keratinocytes in human breast pain. BMC Womens Health. 2005;5:2. doi: 10.1186/1472-6874-5-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Tympanidis P, Casula MA, Yiangou Y, Terenghi G, Dowd P, Anand P. Increased vanilloid receptor VR1 innervation in vulvodynia. Eur J Pain. 2004;8:129–133. doi: 10.1016/S1090-3801(03)00085-5. [DOI] [PubMed] [Google Scholar]
- 171.Yiangou Y, Facer P, Dyer NH, Chan CL, Knowles C, Williams NS, Anand P. Vanilloid receptor 1 immunoreactivity in inflamed human bowel. Lancet. 2001;357:1338–1339. doi: 10.1016/s0140-6736(00)04503-7. [DOI] [PubMed] [Google Scholar]
- 172.Biggs JE, Yates JM, Loescher AR, Clayton NM, Boissonade FM, Robinson PP. Changes in vanilloid receptor 1 (TRPV1) expression following lingual nerve injury. Eur J Pain. 2007;11:192–201. doi: 10.1016/j.ejpain.2006.02.004. [DOI] [PubMed] [Google Scholar]
- 173.Shimosato G, Amaya F, Ueda M, Tanaka Y, Decosterd I, Tanaka M. Peripheral inflammation induces up-regulation of TRPV2 expression in rat DRG. Pain. 2005;119:225–232. doi: 10.1016/j.pain.2005.10.002. [DOI] [PubMed] [Google Scholar]
- 174.McGaraughty S, Chu KL, Faltynek CR, Jarvis MF. Systemic and site-specific effects of A-425619, a selective TRPV1 receptor antagonist, on wide dynamic range neurons in CFA-treated and uninjured rats. J Neurophysiol. 2006;95:18–25. doi: 10.1152/jn.00560.2005. [DOI] [PubMed] [Google Scholar]
- 175.Honore P, Wismer CT, Mikusa J, Zhu CZ, Zhong C, Gauvin DM, Gomtsyan A, El Kouhen R, Lee CH, Marsh K, Sullivan JP, Faltynek CR, Jarvis MF. A-425619 [1-isoquinolin-5-yl-3-(4-trifluoromethyl-benzyl)-urea], a novel transient receptor potential type V1 receptor antagonist, relieves pathophysiological pain associated with inflammation and tissue injury in rats. J Pharmacol Exp Ther. 2005;314:410–421. doi: 10.1124/jpet.105.083915. [DOI] [PubMed] [Google Scholar]
- 176.Bingham S, Beswick PJ, Bountra C, Brown T, Campbell IB, Chessell IP, Clayton N, Collins SD, Davey PT, Goodland H, Gray N, Haslam C, Hatcher JP, Hunter AJ, Lucas F, Murkitt G, Naylor A, Pickup E, Sargent B, Summerfield SG, Stevens A, Stratton SC, Wiseman J. The cyclooxygenase-2 inhibitor GW406381X [2-(4-ethoxyphenyl)-3-[4-(methylsulfonyl)phenyl]-pyrazolo[1,5-b]pyridazine ] is effective in animal models of neuropathic pain and central sensitization. J Pharmacol Exp Ther. 2005;312:1161–1169. doi: 10.1124/jpet.104.075267. [DOI] [PubMed] [Google Scholar]
- 177.Chizh BA, O’Donnell MB, Napolitano A, Wang J, Brooke AC, Aylott MC, Bullman JN, Gray EJ, Lai RY, Williams PM, Appleby JM. The effects of the TRPV1 antagonist SB-705498 on TRPV1 receptor-mediated activity and inflammatory hyperalgesia in humans. Pain. 2007 doi: 10.1016/j.pain.2007.06.006. [DOI] [PubMed] [Google Scholar]
- 178.Rami HK, Thompson M, Stemp G, Fell S, Jerman JC, Stevens AJ, Smart D, Sargent B, Sanderson D, Randall AD, Gunthorpe MJ, Davis JB. Discovery of SB-705498: a potent, selective and orally bioavailable TRPV1 antagonist suitable for clinical development. Bioorg Med Chem Lett. 2006;16:3287–3291. doi: 10.1016/j.bmcl.2006.03.030. [DOI] [PubMed] [Google Scholar]
- 179.Evans RM, Scott RH, Ross RA. Chronic exposure of sensory neurones to increased levels of nerve growth factor modulates CB(1)/TRPV1 receptor crosstalk. Br J Pharmacol. 2007 doi: 10.1038/sj.bjp.0707411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Holzer P. Sensory neurone responses to mucosal noxae in the upper gut: relevance to mucosal integrity and gastrointestinal pain. Neurogastroenterol Motil. 2002;14:459–475. doi: 10.1046/j.1365-2982.2002.00353.x. [DOI] [PubMed] [Google Scholar]
- 181.Holzer P, Guth PH. Neuropeptide control of rat gastric mucosal blood flow. Increase by calcitonin gene-related peptide and vasoactive intestinal polypeptide, but not substance P and neurokinin A. Circ Res. 1991;68:100–105. doi: 10.1161/01.res.68.1.100. [DOI] [PubMed] [Google Scholar]
- 182.Saeki T, Ohno T, Kamata K, Arai K, Mizuguchi S, Katori M, Saigenji K, Majima M. Mild irritant prevents ethanol-induced gastric mucosal microcirculatory disturbances through actions of calcitonin gene-related peptide and PGI2 in rats. Am J Physiol Gastrointest Liver Physiol. 2004;286:G68–75. doi: 10.1152/ajpgi.00538.2002. [DOI] [PubMed] [Google Scholar]
- 183.Harada N, Okajima K, Uchiba M, Katsuragi T. Contribution of capsaicin-sensitive sensory neurons to stress-induced increases in gastric tissue levels of prostaglandins in rats. Am J Physiol Gastrointest Liver Physiol. 2003;285:G1214–1224. doi: 10.1152/ajpgi.00364.2002. [DOI] [PubMed] [Google Scholar]
- 184.Kato S, Aihara E, Nakamura A, Xin H, Matsui H, Kohama K, Takeuchi K. Expression of vanilloid receptors in rat gastric epithelial cells: role in cellular protection. Biochem Pharmacol. 2003;66:1115–1121. doi: 10.1016/s0006-2952(03)00461-1. [DOI] [PubMed] [Google Scholar]
- 185.Ni D, Gu Q, Hu HZ, Gao N, Zhu MX, Lee LY. Thermal sensitivity of isolated vagal pulmonary sensory neurons: role of transient receptor potential vanilloid receptors. Am J Physiol Regul Integr Comp Physiol. 2006;291:R541–550. doi: 10.1152/ajpregu.00016.2006. [DOI] [PubMed] [Google Scholar]
- 186.Watanabe N, Horie S, Michael GJ, Spina D, Page CP, Priestley JV. Immunohistochemical localization of vanilloid receptor subtype 1 (TRPV1) in the guinea pig respiratory system. Pulm Pharmacol Ther. 2005;18:187–197. doi: 10.1016/j.pupt.2004.12.002. [DOI] [PubMed] [Google Scholar]
- 187.Yang XR, Lin MJ, McIntosh LS, Sham JS. Functional expression of transient receptor potential melastatin- and vanilloid-related channels in pulmonary arterial and aortic smooth muscle. Am J Physiol Lung Cell Mol Physiol. 2006;290:L1267–1276. doi: 10.1152/ajplung.00515.2005. [DOI] [PubMed] [Google Scholar]
- 188.Lee LY, Pisarri TE. Afferent properties and reflex functions of bronchopulmonary C-fibers. Respir Physiol. 2001;125:47–65. doi: 10.1016/s0034-5687(00)00204-8. [DOI] [PubMed] [Google Scholar]
- 189.Coleridge JC, Coleridge HM. Afferent vagal C fibre innervation of the lungs and airways and its functional significance. Rev Physiol Biochem Pharmacol. 1984;99:1–110. doi: 10.1007/BFb0027715. [DOI] [PubMed] [Google Scholar]
- 190.Taylor-Clark T, Undem BJ. Transduction mechanisms in airway sensory nerves. J Appl Physiol. 2006;101:950–959. doi: 10.1152/japplphysiol.00222.2006. [DOI] [PubMed] [Google Scholar]
- 191.Joos GF, Germonpre PR, Pauwels RA. Role of tachykinins in asthma. Allergy. 2000;55:321–337. doi: 10.1034/j.1398-9995.2000.00112.x. [DOI] [PubMed] [Google Scholar]
- 192.Lundberg JM, Saria A. Polypeptide-containing neurons in airway smooth muscle. Annu Rev Physiol. 1987;49:557–572. doi: 10.1146/annurev.ph.49.030187.003013. [DOI] [PubMed] [Google Scholar]
- 193.Kollarik M, Undem BJ. Activation of bronchopulmonary vagal afferent nerves with bradykinin, acid and vanilloid receptor agonists in wild-type and TRPV1-/-mice. J Physiol. 2004;555:115–123. doi: 10.1113/jphysiol.2003.054890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Lee LY, Kwong K, Lin YS, Gu Q. Hypersensitivity of bronchopulmonary C-fibers induced by airway mucosal inflammation: cellular mechanisms. Pulm Pharmacol Ther. 2002;15:199–204. doi: 10.1006/pupt.2002.0338. [DOI] [PubMed] [Google Scholar]
- 195.Canning BJ, Farmer DG, Mori N. Mechanistic studies of acid-evoked coughing in anesthetized guinea pigs. Am J Physiol Regul Integr Comp Physiol. 2006;291:R454–463. doi: 10.1152/ajpregu.00862.2005. [DOI] [PubMed] [Google Scholar]
- 196.Lalloo UG, Fox AJ, Belvisi MG, Chung KF, Barnes PJ. Capsazepine inhibits cough induced by capsaicin and citric acid but not by hypertonic saline in guinea pigs. J Appl Physiol. 1995;79:1082–1087. doi: 10.1152/jappl.1995.79.4.1082. [DOI] [PubMed] [Google Scholar]
- 197.Winning AJ, Hamilton RD, Shea SA, Guz A. Respiratory and cardiovascular effects of central and peripheral intravenous injections of capsaicin in man: evidence for pulmonary chemosensitivity. Clin Sci (Lond) 1986;71:519–526. doi: 10.1042/cs0710519. [DOI] [PubMed] [Google Scholar]
- 198.Geppetti P, Materazzi S, Nicoletti P. The transient receptor potential vanilloid 1: role in airway inflammation and disease. Eur J Pharmacol. 2006;533:207–214. doi: 10.1016/j.ejphar.2005.12.063. [DOI] [PubMed] [Google Scholar]
- 199.Manzini S, Maggi CA, Geppetti P, Bacciarelli C. Capsaicin desensitization protects from antigen-induced bronchospasm in conscious guinea-pigs. Eur J Pharmacol. 1987;138:307–308. doi: 10.1016/0014-2999(87)90451-1. [DOI] [PubMed] [Google Scholar]
- 200.Herd CM, Gozzard N, Page CP. Capsaicin pre-treatment prevents the development of antigen-induced airway hyperresponsiveness in neonatally immunised rabbits. Eur J Pharmacol. 1995;282:111–119. doi: 10.1016/0014-2999(95)00291-r. [DOI] [PubMed] [Google Scholar]
- 201.Buckley TL, Nijkamp FP. Airways hyperreactivity and cellular accumulation in a delayed-type hypersensitivity reaction in the mouse. Modulation by capsaicin-sensitive nerves. Am J Respir Crit Care Med. 1994;149:400–407. doi: 10.1164/ajrccm.149.2.8306037. [DOI] [PubMed] [Google Scholar]
- 202.Jia Y, Lee LY. Role of TRPV receptors in respiratory diseases. Biochim Biophys Acta. 2007;1772:915–927. doi: 10.1016/j.bbadis.2007.01.013. [DOI] [PubMed] [Google Scholar]
- 203.Bley KR. Recent developments in transient receptor potential vanilloid receptor 1 agonist-based therapies. Expert Opin Investig Drugs. 2004;13:1445–1456. doi: 10.1517/13543784.13.11.1445. [DOI] [PubMed] [Google Scholar]
- 204.Avelino A, Cruz F. TRPV1 (vanilloid receptor) in the urinary tract: expression, function and clinical applications. Naunyn Schmiedebergs Arch Pharmacol. 2006;373:287–299. doi: 10.1007/s00210-006-0073-2. [DOI] [PubMed] [Google Scholar]
- 205.Sellers DJ, McKay N. Developments in the pharmacotherapy of the overactive bladder. Curr Opin Urol. 2007;17:223–230. doi: 10.1097/MOU.0b013e3281299033. [DOI] [PubMed] [Google Scholar]
- 206.Cruz F, Dinis P. Resiniferatoxin and botulinum toxin type A for treatment of lower urinary tract symptoms. Neurourol Urodyn. 2007 doi: 10.1002/nau.20479. [DOI] [PubMed] [Google Scholar]
- 207.Holzer P. TRPV1 and the gut: from a tasty receptor for a painful vanilloid to a key player in hyperalgesia. Eur J Pharmacol. 2004;500:231–241. doi: 10.1016/j.ejphar.2004.07.028. [DOI] [PubMed] [Google Scholar]
- 208.Greffrath W. The capsaicin receptor. “TRPing” transduction for painful stimuli. Schmerz. 2006;20:219–225. doi: 10.1007/s00482-005-0440-9. [DOI] [PubMed] [Google Scholar]
- 209.Hicks GA. TRP channels as therapeutic targets: hot property, or time to cool down? Neurogastroenterol Motil. 2006;18:590–594. doi: 10.1111/j.1365-2982.2006.00823.x. [DOI] [PubMed] [Google Scholar]
- 210.Liu L, Mansfield KJ, Kristiana I, Vaux KJ, Millard RJ, Burcher E. The molecular basis of urgency: regional difference of vanilloid receptor expression in the human urinary bladder. Neurourol Urodyn. 2007;26:433–438. doi: 10.1002/nau.20326. discussion 439; discussion 451–433. [DOI] [PubMed] [Google Scholar]
- 211.Birder LA, Nakamura Y, Kiss S, Nealen ML, Barrick S, Kanai AJ, Wang E, Ruiz G, De Groat WC, Apodaca G, Watkins S, Caterina MJ. Altered urinary bladder function in mice lacking the vanilloid receptor TRPV1. Nat Neurosci. 2002;5:856–860. doi: 10.1038/nn902. [DOI] [PubMed] [Google Scholar]
- 212.Apostolidis A, Brady CM, Yiangou Y, Davis J, Fowler CJ, Anand P. Capsaicin receptor TRPV1 in urothelium of neurogenic human bladders and effect of intravesical resiniferatoxin. Urology. 2005;65:400–405. doi: 10.1016/j.urology.2004.10.007. [DOI] [PubMed] [Google Scholar]
- 213.Kim SR, Lee DY, Chung ES, Oh UT, Kim SU, Jin BK. Transient receptor potential vanilloid subtype 1 mediates cell death of mesencephalic dopaminergic neurons in vivo and in vitro. J Neurosci. 2005;25:662–671. doi: 10.1523/JNEUROSCI.4166-04.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Kudoh A, Ishihara H, Matsuki A. Current perception thresholds and postoperative pain in schizophrenic patients. Reg Anesth Pain Med. 2000;25:475–479. doi: 10.1053/rapm.2000.7617. [DOI] [PubMed] [Google Scholar]
- 215.Blumensohn R, Ringler D, Eli I. Pain perception in patients with schizophrenia. J Nerv Ment Dis. 2002;190:481–483. doi: 10.1097/00005053-200207000-00011. [DOI] [PubMed] [Google Scholar]
- 216.Waldo MC. Co-distribution of sensory gating and impaired niacin flush response in the parents of schizophrenics. Schizophr Res. 1999;40:49–53. doi: 10.1016/s0920-9964(99)00031-6. [DOI] [PubMed] [Google Scholar]
- 217.Messamore E. Relationship between the niacin skin flush response and essential fatty acids in schizophrenia. Prostaglandins Leukot Essent Fatty Acids. 2003;69:413–419. doi: 10.1016/j.plefa.2003.08.013. [DOI] [PubMed] [Google Scholar]
- 218.Messamore E, Hoffman WF, Janowsky A. The niacin skin flush abnormality in schizophrenia: a quantitative dose-response study. Schizophr Res. 2003;62:251–258. doi: 10.1016/s0920-9964(02)00311-0. [DOI] [PubMed] [Google Scholar]
- 219.Wu M, Komori N, Qin C, Farber JP, Linderoth B, Foreman RD. Sensory fibers containing vanilloid receptor-1 (VR-1) mediate spinal cord stimulation-induced vasodilation. Brain Res. 2006;1107:177–184. doi: 10.1016/j.brainres.2006.05.087. [DOI] [PubMed] [Google Scholar]
- 220.Deng PY, Li YJ. Calcitonin gene-related peptide and hypertension. Peptides. 2005;26:1676–1685. doi: 10.1016/j.peptides.2005.02.002. [DOI] [PubMed] [Google Scholar]
- 221.Wang DH. The vanilloid receptor and hypertension. Acta Pharmacol Sin. 2005;26:286–294. doi: 10.1111/j.1745-7254.2005.00057.x. [DOI] [PubMed] [Google Scholar]
- 222.Breyne J, Vanheel B. Methanandamide hyperpolarizes gastric arteries by stimulation of TRPV1 receptors on perivascular CGRP containing nerves. J Cardiovasc Pharmacol. 2006;47:303–309. doi: 10.1097/01.fjc.0000205053.53946.10. [DOI] [PubMed] [Google Scholar]
- 223.O’Sullivan SE, Kendall DA, Randall MD. The effects of Delta9-tetrahydrocannabinol in rat mesenteric vasculature, and its interactions with the endocannabinoid anandamide. Br J Pharmacol. 2005;145:514–526. doi: 10.1038/sj.bjp.0706218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Wang Y, Wang DH. Neural control of blood pressure: focusing on capsaicin-sensitive sensory nerves. Cardiovasc Hematol Disord Drug Targets. 2007;7:37–46. doi: 10.2174/187152907780059100. [DOI] [PubMed] [Google Scholar]
- 225.Dux M, Santha P, Jancso G. Capsaicin-sensitive neurogenic sensory vasodilatation in the dura mater of the rat. J Physiol. 2003;552:859–867. doi: 10.1113/jphysiol.2003.050633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Toth A, Boczan J, Kedei N, Lizanecz E, Bagi Z, Papp Z, Edes I, Csiba L, Blumberg PM. Expression and distribution of vanilloid receptor 1 (TRPV1) in the adult rat brain. Brain Res Mol Brain Res. 2005;135:162–168. doi: 10.1016/j.molbrainres.2004.12.003. [DOI] [PubMed] [Google Scholar]
- 227.Gram DX, Ahren B, Nagy I, Olsen UB, Brand CL, Sundler F, Tabanera R, Svendsen O, Carr RD, Santha P, Wierup N, Hansen AJ. Capsaicin-sensitive sensory fibers in the islets of Langerhans contribute to defective insulin secretion in Zucker diabetic rat, an animal model for some aspects of human type 2 diabetes. Eur J Neurosci. 2007;25:213–223. doi: 10.1111/j.1460-9568.2006.05261.x. [DOI] [PubMed] [Google Scholar]
- 228.Razavi R, Chan Y, Afifiyan FN, Liu XJ, Wan X, Yantha J, Tsui H, Tang L, Tsai S, Santamaria P, Driver JP, Serreze D, Salter MW, Dosch HM. TRPV1+ sensory neurons control beta cell stress and islet inflammation in autoimmune diabetes. Cell. 2006;127:1123–1135. doi: 10.1016/j.cell.2006.10.038. [DOI] [PubMed] [Google Scholar]
- 229.Biro T, Toth BI, Marincsak R, Dobrosi N, Geczy T, Paus R. TRP channels as novel players in the pathogenesis and therapy of itch. Biochim Biophys Acta. 2007;1772:1004–1021. doi: 10.1016/j.bbadis.2007.03.002. [DOI] [PubMed] [Google Scholar]
- 230.Yosipovitch G, Fast K, Bernhard JD. Noxious heat and scratching decrease histamine-induced itch and skin blood flow. J Invest Dermatol. 2005;125:1268–1272. doi: 10.1111/j.0022-202X.2005.23942.x. [DOI] [PubMed] [Google Scholar]
- 231.Ghilardi JR, Rohrich H, Lindsay TH, Sevcik MA, Schwei MJ, Kubota K, Halvorson KG, Poblete J, Chaplan SR, Dubin AE, Carruthers NI, Swanson D, Kuskowski M, Flores CM, Julius D, Mantyh PW. Selective blockade of the capsaicin receptor TRPV1 attenuates bone cancer pain. J Neurosci. 2005;25:3126–3131. doi: 10.1523/JNEUROSCI.3815-04.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232.Menendez L, Juarez L, Garcia E, Garcia-Suarez O, Hidalgo A, Baamonde A. Analgesic effects of capsazepine and resiniferatoxin on bone cancer pain in mice. Neurosci Lett. 2006;393:70–73. doi: 10.1016/j.neulet.2005.09.046. [DOI] [PubMed] [Google Scholar]
- 233.Niiyama Y, Kawamata T, Yamamoto J, Omote K, Namiki A. Bone cancer increases transient receptor potential vanilloid subfamily 1 expression within distinct subpopulations of dorsal root ganglion neurons. Neuroscience. 2007;148:560–572. doi: 10.1016/j.neuroscience.2007.05.049. [DOI] [PubMed] [Google Scholar]
- 234.Fernihough J, Gentry C, Bevan S, Winter J. Regulation of calcitonin gene-related peptide and TRPV1 in a rat model of osteoarthritis. Neurosci Lett. 2005;388:75–80. doi: 10.1016/j.neulet.2005.06.044. [DOI] [PubMed] [Google Scholar]
- 235.Szabo A, Helyes Z, Sandor K, Bite A, Pinter E, Nemeth J, Banvolgyi A, Bolcskei K, Elekes K, Szolcsanyi J. Role of transient receptor potential vanilloid 1 receptors in adjuvant-induced chronic arthritis: in vivo study using gene-deficient mice. J Pharmacol Exp Ther. 2005;314:111–119. doi: 10.1124/jpet.104.082487. [DOI] [PubMed] [Google Scholar]
- 236.Walker KM, Urban L, Medhurst SJ, Patel S, Panesar M, Fox AJ, McIntyre P. The VR1 antagonist capsazepine reverses mechanical hyperalgesia in models of inflammatory and neuropathic pain. J Pharmacol Exp Ther. 2003;304:56–62. doi: 10.1124/jpet.102.042010. [DOI] [PubMed] [Google Scholar]
- 237.Trevisani M, Gazzieri D, Benvenuti F, Campi B, Dinh QT, Groneberg DA, Rigoni M, Emonds-Alt X, Creminon C, Fischer A, Geppetti P, Harrison S. Ethanol causes inflammation in the airways by a neurogenic and TRPV1-dependent mechanism. J Pharmacol Exp Ther. 2004;309:1167–1173. doi: 10.1124/jpet.103.064162. [DOI] [PubMed] [Google Scholar]
- 238.Zhang F, Yang H, Wang Z, Mergler S, Liu H, Kawakita T, Tachado SD, Pan Z, Capo-Aponte JE, Pleyer U, Koziel H, Kao WW, Reinach PS. Transient receptor potential vanilloid 1 activation induces inflammatory cytokine release in corneal epithelium through MAPK signaling. J Cell Physiol. 2007 doi: 10.1002/jcp.21141. [DOI] [PubMed] [Google Scholar]
- 239.Bhatia M, Zhi L, Zhang H, Ng SW, Moore PK. Role of substance P in hydrogen sulfide-induced pulmonary inflammation in mice. Am J Physiol Lung Cell Mol Physiol. 2006;291:L896–904. doi: 10.1152/ajplung.00053.2006. [DOI] [PubMed] [Google Scholar]
- 240.Sawynok J, Reid A, Meisner J. Pain behaviors produced by capsaicin: influence of inflammatory mediators and nerve injury. J Pain. 2006;7:134–141. doi: 10.1016/j.jpain.2005.09.013. [DOI] [PubMed] [Google Scholar]
- 241.El Kouhen R, Surowy CS, Bianchi BR, Neelands TR, McDonald HA, Niforatos W, Gomtsyan A, Lee CH, Honore P, Sullivan JP, Jarvis MF, Faltynek CR. A-425619 [1-isoquinolin-5-yl-3-(4-trifluoromethyl-benzyl)-urea], a novel and selective transient receptor potential type V1 receptor antagonist, blocks channel activation by vanilloids, heat, and acid. J Pharmacol Exp Ther. 2005;314:400–409. doi: 10.1124/jpet.105.084103. [DOI] [PubMed] [Google Scholar]
- 242.Gomtsyan A, Bayburt EK, Schmidt RG, Zheng GZ, Perner RJ, Didomenico S, Koenig JR, Turner S, Jinkerson T, Drizin I, Hannick SM, Macri BS, McDonald HA, Honore P, Wismer CT, Marsh KC, Wetter J, Stewart KD, Oie T, Jarvis MF, Surowy CS, Faltynek CR, Lee CH. Novel transient receptor potential vanilloid 1 receptor antagonists for the treatment of pain: structure-activity relationships for ureas with quinoline, isoquinoline, quinazoline, phthalazine, quinoxaline, and cinnoline moieties. J Med Chem. 2005;48:744–752. doi: 10.1021/jm0492958. [DOI] [PubMed] [Google Scholar]
- 243.Westaway SM, Chung YK, Davis JB, Holland V, Jerman JC, Medhurst SJ, Rami HK, Stemp G, Stevens AJ, Thompson M, Winborn KY, Wright J. N-Tetrahydroquinolinyl, N-quinolinyl and N-isoquinolinyl biaryl carboxamides as antagonists of TRPV1. Bioorg Med Chem Lett. 2006;16:4533–4536. doi: 10.1016/j.bmcl.2006.06.026. [DOI] [PubMed] [Google Scholar]
- 244.Pomonis JD, Harrison JE, Mark L, Bristol DR, Valenzano KJ, Walker K. N-(4-Tertiarybutylphenyl)-4-(3-cholorphyridin-2-yl)tetrahydropyrazine -1(2H)-carbox-amide (BCTC), a novel, orally effective vanilloid receptor 1 antagonist with analgesic properties: II. in vivo characterization in rat models of inflammatory and neuropathic pain. J Pharmacol Exp Ther. 2003;306:387–393. doi: 10.1124/jpet.102.046268. [DOI] [PubMed] [Google Scholar]
- 245.Culshaw AJ, Bevan S, Christiansen M, Copp P, Davis A, Davis C, Dyson A, Dziadulewicz EK, Edwards L, Eggelte H, Fox A, Gentry C, Groarke A, Hallett A, Hart TW, Hughes GA, Knights S, Kotsonis P, Lee W, Lyothier I, McBryde A, McIntyre P, Paloumbis G, Panesar M, Patel S, Seiler MP, Yaqoob M, Zimmermann K. Identification and biological characterization of 6-aryl-7-isopropylquinazolinones as novel TRPV1 antagonists that are effective in models of chronic pain. J Med Chem. 2006;49:471–474. doi: 10.1021/jm051058x. [DOI] [PubMed] [Google Scholar]
- 246.Gavva NR, Tamir R, Qu Y, Klionsky L, Zhang TJ, Immke D, Wang J, Zhu D, Vanderah TW, Porreca F, Doherty EM, Norman MH, Wild KD, Bannon AW, Louis JC, Treanor JJ. AMG 9810 [(E)-3-(4-t-butylphenyl)-N-(2,3-dihydrobenzo[b][1,4] dioxin-6-yl)acrylamide], a novel vanilloid receptor 1 (TRPV1) antagonist with antihyperalgesic properties. J Pharmacol Exp Ther. 2005;313:474–484. doi: 10.1124/jpet.104.079855. [DOI] [PubMed] [Google Scholar]
- 247.Joshi SK, Hernandez G, Mikusa JP, Zhu CZ, Zhong C, Salyers A, Wismer CT, Chandran P, Decker MW, Honore P. Comparison of antinociceptive actions of standard analgesics in attenuating capsaicin and nerve-injury-induced mechanical hypersensitivity. Neuroscience. 2006;143:587–596. doi: 10.1016/j.neuroscience.2006.08.005. [DOI] [PubMed] [Google Scholar]
- 248.Cui M, Honore P, Zhong C, Gauvin D, Mikusa J, Hernandez G, Chandran P, Gomtsyan A, Brown B, Bayburt EK, Marsh K, Bianchi B, McDonald H, Niforatos W, Neelands TR, Moreland RB, Decker MW, Lee CH, Sullivan JP, Faltynek CR. TRPV1 receptors in the CNS play a key role in broad-spectrum analgesia of TRPV1 antagonists. J Neurosci. 2006;26:9385–9393. doi: 10.1523/JNEUROSCI.1246-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 249.Kimball ES, Wallace NH, Schneider CR, D’Andrea MR, Hornby PJ. Vanilloid receptor 1 antagonists attenuate disease severity in dextran sulphate sodium-induced colitis in mice. Neurogastroenterol Motil. 2004;16:811–818. doi: 10.1111/j.1365-2982.2004.00549.x. [DOI] [PubMed] [Google Scholar]
- 250.Miranda A, Nordstrom E, Mannem A, Smith C, Banerjee B, Sengupta JN. The role of transient receptor potential vanilloid 1 in mechanical and chemical visceral hyperalgesia following experimental colitis. Neuroscience. 2007 doi: 10.1016/j.neuroscience.2007.05.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251.Jakab B, Helyes Z, Varga A, Bolcskei K, Szabo A, Sandor K, Elekes K, Borzsei R, Keszthelyi D, Pinter E, Petho G, Nemeth J, Szolcsanyi J. Pharmacological characterization of the TRPV1 receptor antagonist JYL1421 (SC0030) in vitro and in vivo in the rat. Eur J Pharmacol. 2005;517:35–44. doi: 10.1016/j.ejphar.2005.05.002. [DOI] [PubMed] [Google Scholar]
- 252.Saitoh C, Kitada C, Uchida W, Chancellor MB, de Groat WC, Yoshimura N. The differential contractile responses to capsaicin and anandamide in muscle strips isolated from the rat urinary bladder. Eur J Pharmacol. 2007;570:182–187. doi: 10.1016/j.ejphar.2007.05.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 253.Kanai Y, Hara T, Imai A, Sakakibara A. Differential involvement of TRPV1 receptors at the central and peripheral nerves in CFA-induced mechanical and thermal hyperalgesia. J Pharm Pharmacol. 2007;59:733–738. doi: 10.1211/jpp.59.5.0015. [DOI] [PubMed] [Google Scholar]
- 254.McLeod RL, Fernandez X, Correll CC, Phelps TP, Jia Y, Wang X, Hey JA. TRPV1 antagonists attenuate antigen-provoked cough in ovalbumin sensitized guinea pigs. Cough. 2006;2:10. doi: 10.1186/1745-9974-2-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 255.Kanai Y, Nakazato E, Fujiuchi A, Hara T, Imai A. Involvement of an increased spinal TRPV1 sensitization through its up-regulation in mechanical allodynia of CCI rats. Neuropharmacology. 2005;49:977–984. doi: 10.1016/j.neuropharm.2005.05.003. [DOI] [PubMed] [Google Scholar]
- 256.Borsani E, Labanca M, Bianchi R, Rodella LF. AM404 decreases Fos-immunoreactivity in the spinal cord in a model of inflammatory pain. Brain Res. 2007;1152:87–94. doi: 10.1016/j.brainres.2007.03.071. [DOI] [PubMed] [Google Scholar]
- 257.Tang L, Chen Y, Chen Z, Blumberg PM, Kozikowski AP, Wang ZJ. Antinociceptive pharmacology of N-(4-chlorobenzyl)-N′-(4-hydroxy-3-iodo-5-methoxybenzyl) thiourea, a high-affinity competitive antagonist of the transient receptor potential vanilloid 1 receptor. J Pharmacol Exp Ther. 2007;321:791–798. doi: 10.1124/jpet.106.117572. [DOI] [PubMed] [Google Scholar]
- 258.Jones RC, 3rd, Xu L, Gebhart GF. The mechanosensitivity of mouse colon afferent fibers and their sensitization by inflammatory mediators require transient receptor potential vanilloid 1 and acid-sensing ion channel 3. J Neurosci. 2005;25:10981–10989. doi: 10.1523/JNEUROSCI.0703-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 259.Singh Tahim A, Santha P, Nagy I. Inflammatory mediators convert anandamide into a potent activator of the vanilloid type 1 transient receptor potential receptor in nociceptive primary sensory neurons. Neuroscience. 2005;136:539–548. doi: 10.1016/j.neuroscience.2005.08.005. [DOI] [PubMed] [Google Scholar]
- 260.Rigoni M, Trevisani M, Gazzieri D, Nadaletto R, Tognetto M, Creminon C, Davis JB, Campi B, Amadesi S, Geppetti P, Harrison S. Neurogenic responses mediated by vanilloid receptor-1 (TRPV1) are blocked by the high affinity antagonist, iodo-resiniferatoxin. Br J Pharmacol. 2003;138:977–985. doi: 10.1038/sj.bjp.0705110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 261.Southall MD, Li T, Gharibova LS, Pei Y, Nicol GD, Travers JB. Activation of epidermal vanilloid receptor-1 induces release of proinflammatory mediators in human keratinocytes. J Pharmacol Exp Ther. 2003;304:217–222. doi: 10.1124/jpet.102.040675. [DOI] [PubMed] [Google Scholar]
- 262.Perkins MN, Campbell EA. Capsazepine reversal of the antinociceptive action of capsaicin in vivo. Br J Pharmacol. 1992;107:329–333. doi: 10.1111/j.1476-5381.1992.tb12746.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 263.Bianchi BR, El Kouhen R, Neelands TR, Lee CH, Gomtsyan A, Raja SN, Vaidyanathan SN, Surber B, McDonald HA, Surowy CS, Faltynek CR, Moreland RB, Jarvis MF, Puttfarcken PS. [3H]A-778317 [1-((R)-5-tert-butyl-indan-1-yl)-3-isoquinolin-5-yl-urea]: A Novel, Stereoselective, High-Affinity Antagonist is a Useful Radioligand For The Human TRPV1 Receptor. J Pharmacol Exp Ther. 2007 doi: 10.1124/jpet.107.124305. [DOI] [PubMed] [Google Scholar]
- 264.Gunthorpe MJ, Hannan SL, Smart D, Jerman JC, Arpino S, Smith GD, Brough S, Wright J, Egerton J, Lappin SC, Holland VA, Winborn K, Thompson M, Rami HK, Randall A, Davis JB. Characterization of SB-705498, a potent and selective vanilloid receptor-1 (VR1/TRPV1) antagonist that inhibits the capsaicin-, acid-, and heat-mediated activation of the receptor. J Pharmacol Exp Ther. 2007;321:1183–1192. doi: 10.1124/jpet.106.116657. [DOI] [PubMed] [Google Scholar]
- 265.Christoph T, Grunweller A, Mika J, Schafer MK, Wade EJ, Weihe E, Erdmann VA, Frank R, Gillen C, Kurreck J. Silencing of vanilloid receptor TRPV1 by RNAi reduces neuropathic and visceral pain in vivo. Biochem Biophys Res Commun. 2006;350:238–243. doi: 10.1016/j.bbrc.2006.09.037. [DOI] [PubMed] [Google Scholar]
- 266.Swanson DM, Dubin AE, Shah C, Nasser N, Chang L, Dax SL, Jetter M, Breitenbucher JG, Liu C, Mazur C, Lord B, Gonzales L, Hoey K, Rizzolio M, Bogenstaetter M, Codd EE, Lee DH, Zhang SP, Chaplan SR, Carruthers NI. Identification and biological evaluation of 4-(3-trifluoromethylpyridin-2-yl)piperazine-1-carboxylic acid (5-trifluoromethylpyridin-2-yl)amide, a high affinity TRPV1 (VR1) vanilloid receptor antagonist. J Med Chem. 2005;48:1857–1872. doi: 10.1021/jm0495071. [DOI] [PubMed] [Google Scholar]
- 267.Immke DC, Gavva NR. The TRPV1 receptor and nociception. Semin Cell Dev Biol. 2006;17:582–591. doi: 10.1016/j.semcdb.2006.09.004. [DOI] [PubMed] [Google Scholar]
- 268.Gavva NR, Bannon AW, Surapaneni S, Hovland DN, Jr, Lehto SG, Gore A, Juan T, Deng H, Han B, Klionsky L, Kuang R, Le A, Tamir R, Wang J, Youngblood B, Zhu D, Norman MH, Magal E, Treanor JJ, Louis JC. The vanilloid receptor TRPV1 is tonically activated in vivo and involved in body temperature regulation. J Neurosci. 2007;27:3366–3374. doi: 10.1523/JNEUROSCI.4833-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 269.Cholewinski A, Burgess GM, Bevan S. The role of calcium in capsaicin-induced desensitization in rat cultured dorsal root ganglion neurons. Neuroscience. 1993;55:1015–1023. doi: 10.1016/0306-4522(93)90315-7. [DOI] [PubMed] [Google Scholar]
- 270.Szallasi A, Blumberg PM. Vanilloid receptors: new insights enhance potential as a therapeutic target. Pain. 1996;68:195–208. doi: 10.1016/s0304-3959(96)03202-2. [DOI] [PubMed] [Google Scholar]
- 271.Szallasi A, Szabo T, Biro T, Modarres S, Blumberg PM, Krause JE, Cortright DN, Appendino G. Resiniferatoxin-type phorboid vanilloids display capsaicin-like selectivity at native vanilloid receptors on rat DRG neurons and at the cloned vanilloid receptor VR1. Br J Pharmacol. 1999;128:428–434. doi: 10.1038/sj.bjp.0702810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 272.Sawynok J. Topical analgesics in neuropathic pain. Curr Pharm Des. 2005;11:2995–3004. doi: 10.2174/1381612054865019. [DOI] [PubMed] [Google Scholar]
- 273.Raisinghani M, Premkumar LS. Block of native and cloned vanilloid receptor 1 (TRPV1) by aminoglycoside antibiotics. Pain. 2005;113:123–133. doi: 10.1016/j.pain.2004.09.042. [DOI] [PubMed] [Google Scholar]