Abstract
Forced expression of gap junction proteins, connexins, enables gap junction-deficient cell lines to propagate intercellular calcium waves. Here, we show that ATP secretion from the poorly coupled cell lines, C6 glioma, HeLa, and U373 glioblastoma, is potentiated 5- to 15-fold by connexin expression. ATP release required purinergic receptor-activated intracellular Ca2+ mobilization and was inhibited by Cl− channel blockers. Calcium wave propagation also was reduced by purinergic receptor antagonists and by Cl− channel blockers but insensitive to gap junction inhibitors. These observations suggest that cell-to-cell signaling associated with connexin expression results from enhanced ATP release and not, as previously believed, from an increase in intercellular coupling.
Astrocytic calcium signaling has received considerable interest, during the last few years, because of its possible role in brain function (1). Astrocytic calcium waves modulate neuronal calcium levels and thereby synaptic activity (2–7). Originally, it was believed that interastrocytic signaling was mediated by diffusion of inositol trisphosphate (IP3) and/or Ca2+ across gap junctions, but recent observations suggest that an extracellular pathway may exist: Astrocytic calcium waves can cross cell-free areas and evoke calcium increments in astrocytes physically isolated from the stimulated cell (8–9). In other cell types, including rat basophilic leukemia cells, hepatocytes, and neuroepithelioma cells, ATP has been identified as the diffusable factor that mediates intercellular calcium signaling (10–11). These cells release ATP, or a related compound, which stimulates surrounding purinoceptors leading to further ATP release through an IP3–protein-dependent pathway. An array of components that interfere with this pathway, including receptor blockage and hydrolysis of ATP, reduce calcium wave propagation (10–12). Yet, the majority of cell types that propagate calcium waves are tightly coupled by gap junctions. Furthermore, gap junction-deficient cell lines, including C6 glioma, HeLa cells, and HEK293 cells (13–15), acquire the ability to propagate calcium waves only after induced expression of connexins. To account for these seemingly divergent observations, that expression of connexin is a prerequisite for calcium signaling and that wave propagation is sensitive to the manipulation of extracellular ATP, we postulate here that ATP release is regulated by gap junctions. We have reexamined the mechanisms of astrocytic communication and demonstrate that forced expression of connexins potentiate cellular ability to release ATP 5- to 15-fold.
MATERIALS AND METHODS
Culture and Transfections.
Cortical astrocytes from 1 day postnatal rat were prepared and maintained as previously described (16). Cultures were grown 2–6 weeks in vitro before use. C6 glioma cells (American Type Culture Collection) were grown in DMEM/F12 supplemented with 10% fetal bovine serum and antibiotics (17). cDNAs for connexin 43 (Cx43) and Cx32 were ligated into expression vectors pcDNA1 and pBEHpac18 containing the genes for geneticin and puromycin resistance, respectively, (ref. 14; manufacturer’s instruction and stable transfectants were selected with 2 mg/ml geneticin or 2 mg/ml puromycin). One Cx43 clone (Cx43–13) was cultured as earlier shown (17). Expression of Cx43 and Cx32 were assayed by immunolabeling with polyclonal antibodies (kindly provided by B. Nicholson, State University of New York, Buffalo, and D. Paul, Harvard Medical School, Boston) and functional dye transfer (18). Three independent clones of C6 cells, overexpressing either Cx43, Cx32, or null-transfected controls were used for all assays with essentially the same results. HeLa cells overexpressing Cx43, Cx32, Cx26, or HeLa wild-type (Wt/g) were kindly provided by K. Willecke, Bonn University, Germany (14). Another HeLa wild-type clone (Wt/u) was donated by P. Seghal, New York Medical College, New York. Subconfluent human glioblastoma U373-MG cells (ATCC) were transfected with the expression vector pBEH18 containing Cx32 cDNA or expression vector pSV containing β-galactosidase cDNA by using SuperFect (Qiagen, Chatsworth, CA) and following manufacturer’s specifications. Two days after transfection, ATP release was quantified in both set of transfectants.
Fluorescence Recovery After Photobleach (FRAP).
Cultures were incubated with 2 μM dicarboxydichlorofluorescein diacetate (CDCF; 19) for 5 min and post-incubated in the absence of CDCF for another 30 min. Excitation of CDCF was provided by the 488 nm line of a 25 mW krypton/argon laser attached to the confocal microscope (MRC1000, Bio-Rad). Emission was long pass-filtered (515 nm) and detected with the confocal aperture set to its maximal opening (7 mm). After obtaining a baseline fluorescence image of the culture, the area of laser scanning was reduced to include only one target cell. Complete or almost complete photobleaching occurred after three to five scans each lasting 1 sec at full laser power. Subsequently, the microscope settings were returned to recording configuration, and refill of the cell was monitored for 2 min (16, 20). The extent of astrocytic coupling was examined before (control) and after addition of inhibitors in the same culture in a minimum of four independent cultures (n = 4–20, most >8).
Ca2+ Imaging.
Cultures were loaded with 10 μM fluo-3 acetoxymethyl ester (fluo-3 AM, Molecular Probes) for 1 hr at 37°C. Sequences of images were acquired at the confocal microscope and recorded on a optical disc recorder. Relative changes in fluorescence were calculated and normalized against baseline fluorescence by ΔF/F. Background counts were subtracted from all measurements. Experiments were carried out at room temperature in a 1:1 mixture of DMEM and F12 (Ham). Single cells were mechanically stimulated with a glass micropipette as earlier described (21). The wave radius was measured as the farthest distance traveled by the wave in any direction from the stimulated cell. The percentage change in wave radius was calculated as the mean of wave radius in presence of inhibitor divided by the mean of wave radius in matched control (vehicle treated) cultures. A minimum of five waves in four independent cultures was evaluated (total ≥20). The radius of calcium waves was within the range of 200–300 μm during control conditions.
ATP Quantification.
ATP determinations were carried out by using a bioluminescent ATP assay kit (Sigma) and a Chrono-Log luminometer (22). Cells were grown in 24-well tissue culture plates. Each well was half-washed with DMEM/F12 six times and incubated for 20 min in 400 μl of DMEM. Samples of the supernatant were collected immediately before and 10 min after exposure to 100 μM UTP. Stimulated ATP release was calculated as the difference between the two samples and normalized as a function of cell number. A maximum of vehicle of 0.4% (dimethyl sulfoxide, dimethylformamide, methanol, or ethanol) was added to UTP-exposed control culture in the concentration used to dissolve inhibitors. A minimum of three wells in three independent experiment was evaluated (n ≥ 9, most >20). Number of cells in each well was determined after dissociation on a hemocytometer. C6 cells contained 75 ± 10 μg of protein/105 cells, HeLa cells contained 33 ± 7 μg of protein/105 cells, and U373-MG 65 ± 6 μg of protein/105 cells. Cx expression did not alter protein content (data not shown). Lactate dehydrogenase activity was below detection in the supernatant after washing and after treatment with UTP (22). The 2-methylthioATP (2MeSATP)- and ADP-evoked ATP release were comparable with UTP-stimulated ATP release, but at 100 μM, both 2MeSATP and ADP exhibited an elevated ATP background. The 2MeSATP was purchased from RBI, whereas all other analogs were from Sigma.
RESULTS
Gap Junction Blockers Fail to Reduce Ca2+ Signaling at Uncoupling Concentrations.
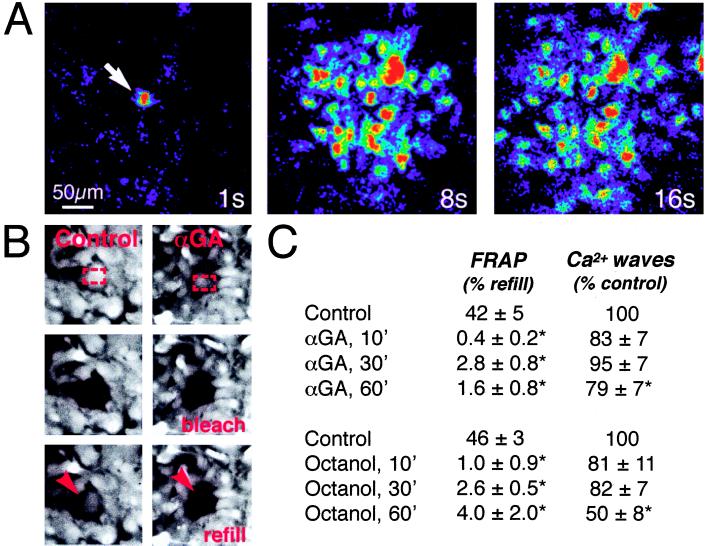
A key observation in the study of astrocytic signaling is that gap junction inhibitors reduce or block wave propagation. However, the relationship of gap junction function per se on calcium signaling has not been studied (23). This issue is of particular relevance, since currently available inhibitors are nonspecific and may affect a variety of channels and transport systems. We tested the effects of two commonly used gap junction inhibitors, 18α-glycyrrhetinic acid (αGA), an aglycone with mineralocorticoid properties (24) and the long-chain alcohol, octanol (25) in cultured cortical rat astrocytes. Within 10 min, both αGA and octanol reduced intercellular diffusion of the junctional tracer, CDCF, using the FRAP technique (20). Fluorescence refill was lowered to 0–4%, or to levels comparable with, or lower than, refill among communication-deficient cell lines (see below) (Fig. 1). However, the mean radius of calcium wave propagation was not significantly reduced by identical treatment of sister cultures. Longer exposure time (Fig. 1) or higher concentrations (data not shown) of both αGA and octanol were necessary to reduce the extent of calcium signaling. Calcium waves could also propagate across cell-free areas in accordance with an earlier report (8; our data not shown). These observations suggest that astrocytic calcium signaling is insensitive to gap junction inhibition at doses which effectively reduce coupling. However, because coupling was evaluated with the FRAP method, we cannot exclude that octanol or αGA decreased intercellular diffusion of relatively large molecules, such as CDCF, disproportionally more than smaller molecules, like Ca2+.
Figure 1.
Uncoupling agents fail to reduce astrocytic calcium signaling despite a marked reduction in gap junction coupling. (A) Representative experiment of the propagation of a calcium wave in an astrocytic culture exposed to the gap junction inhibitor, αGA (10 μM) for 10 min. Culture was loaded with fluo-3 and imaged by confocal microscopy. Sequence of images was collected 1, 8, and 16 sec after focal mechanical stimulation (arrow). The color scale is the same as in Fig. 2 and indicates relative changes in fluo-3 signal (ΔF/F) (B) FRAP in matched sister cultures treated with αGA (Right) or vehicle (Left). Both cultures were loaded with the gap junction permeant fluorescence tracer CDCF (2 μM). Red rectangles outline area of photobleach (Top), Middle panels display culture immediately after bleach, and red arrows indicate refill of fluorescence 2 min later (Bottom). αGA reduced refill to 2%, whereas fluorescence recovered to 38% of prebleach level within 2 min in the control culture. (C) Data summarizing effects of αGA and octanol (0.5 mM) upon gap junction coupling (FRAP) and calcium signaling. The extent of astrocytic coupling and calcium signaling was examined before (control) and after addition of inhibitors. Gap junction coupling was calculated as percentage refill of fluorescence signal 2 min after photobleach. Radius of calcium waves was in the range of 200–300 μm during control conditions. ∗, statistically significant difference from control at P < 0.05 by t test; n = 5–15.
Astrocytic Ca2+ Waves Are Mediated by Autocrine Activation of Purinergic Receptors.
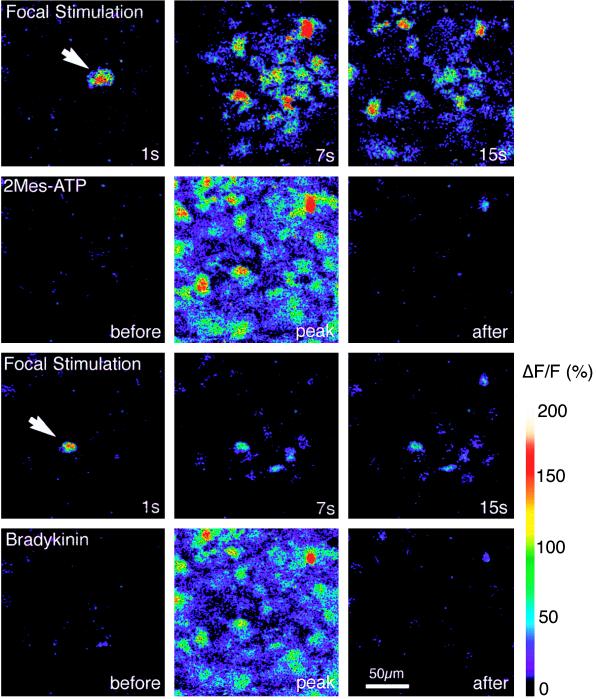
We next examined the effect of purinergic receptor blockers and observed that the P2 receptor antagonists, suramin and reactive blue (25–27), decreased propagation of Ca2+ waves (Table 1), without affecting either the extent of coupling or cellular ability to increase Ca2+ upon focal stimulation (result not shown). In contrast, pyridoxal phosphate-6-azophenyl-2′,4′-disulfonic acid (PPADS), a P2X antagonist (28, 29), did not significantly reduce wave activity (Table 1). Because P2Y receptors are characterized by rapid desensitization (26–30), the effect of receptor desensitization on Ca2+ wave propagation was studied. Cultures that consistently propagated calcium waves (Fig. 2) were exposed to either ATP or 2MeSATP. Both polyphosphates evoked a significant increase in intracellular calcium concentration (Ca2+i), which consisted of a peak increment followed by a slow return to resting or near resting Ca2+i levels. When calcium waves were triggered 5–10 min later in the continuing presence of ATP or 2MeSATP, Ca2+i increased in the stimulated cell but failed to propagate effectively (Fig. 2). Further, exposure to bradykinin (25 μM), a neuropeptide that releases Ca2+ through an IP3-dependent pathway, evoked a robust Ca2+i response in the cells that failed to propagate calcium waves, indicating that the inhibitory effect of ATP did not result from exhaustion of IP3-sensitive calcium stores (Fig. 2). Addition of the ATPase apyrase to the extracellular medium was also an effective inhibitor of calcium signaling (Table 1). Together, these observations support the notion that calcium waves among cultured astrocytes are mediated by release of nucleotides that activate purinergic receptors on neighboring cells, leading to an autocatalytic propagation of calcium signaling. Although these observations indicate that calcium wave propagation is mediated by purinergic receptor activation, and that calcium signaling can occur in the absence of cell coupling, they leave unexplained the necessity of connexins in wave propagation.
Table 1.
Inhibition of calcium waves in primary cultures of cortical astrocytes
| Compound | Wave radius, % of control |
|---|---|
| ATP, 10 μM, prior exposure | 5 ± 4* |
| 2MeSATP, 10 μM, prior exposure | 11 ± 8* |
| Suramin, 100 μM | 18 ± 10* |
| Reactive blue, 30 μM | 0 |
| PPADS, 50 μM | 87 ± 17 |
| Apyrase, 40 units/ml | 22 ± 4* |
| NPPB, 100 μM | 53 ± 5* |
| SITS, 3 mM | 56 ± 5* |
| Furosemide, 5 mM | 57 ± 7* |
Extent of astrocytic calcium signaling was examined in control (vehicle treated) and in matched cultures exposed to several compounds. The wave radius was measured as the farthest distance traveled by the wave in any direction from the stimulated cell. The percentage change in wave radius was calculated as the mean of wave radius in presence of inhibitor divided by the mean of wave radius during control condition (mean ± SEM). Radius of calcium waves was in the range of 200–300 μm during control conditions. SITS, 4-acetamido-4′-isothiocyanatostilbene-2,2′-disulfonic acid; PPADS, pyridoxal phosphate-6-azophenyl-2′,4′-disulfonic acid tetrasodium salt; NPPB, 5-nitro-2-(3-phenylpropylamino)benzoic acid.
Statistically significant difference from control at P < 0.05 by t test; n = 5–35.
Figure 2.
Desensitization of purinergic receptors inhibits the extent of calcium signaling in cultured astrocytes. First row shows mechanical-induced astrocytic calcium wave (white arrow). Sequence of images (1, 7, 15 sec after stimulation) depicts a typical calcium wave under control conditions. Second row shows the same field of astrocytes mobilize Ca2+i transiently in response to 2MeSATP (10 μM). Third row shows mechanical stimulation of a cell neighboring the cell stimulated in the first block (white arrow) failed to induce a propagating Ca2+ wave after exposure to 2MeSATP. Ca2+i increases are limited to a few cells surrounding the stimulated cell. Stimulation was performed when Ca2+i had returned to resting level after the exposure to 2MeSATP (5–10 min). Wave radius was on an average reduced to 11 ± 8 (n = 8) after exposure to 2MeSATP. Fourth row shows subsequent challenge with bradykinin (25 μM) demonstrating that intracellular calcium stores were not depleted by 2MeSATP exposure and that the cells remained viable.
Ca2+ Signaling in Cx-Expressing C6 Cells Is also Mediated by ATP.
On the basis that wild-type C6 glioma cells lack gap junctions and acquire the ability to propagate calcium waves only after forced expression of connexin proteins (13), we tested the ATP requirement for calcium signaling among Cx-transfected C6 cells. Gap junction coupled C6 clones were generated through stable transfection with cDNA for the connexins genes, Cx43 and Cx32, placed under the control of the strong constitutive cytomegalovirus or simian virus 40 promoter, respectively. The resultant Cx43+ and Cx32+ transfectants were extensively coupled (refill of CDCF fluorescence after photobleach; 43 ± 6% and 45 ± 3%, respectively) and propagated waves (mean wave radius >150 μm) that were sensitive to both suramin (15 ± 6% of control) and prior desensitization by ATP (17 ± 7% of control). Wild-type or null-transfectant C6 cells were minimally coupled (4 ± 2% refill of CDCF after photobleach) and were unable to propagate Ca2+ waves (mean wave radius <50 μm). As in astrocytes, calcium signaling in Cx43+ or Cx32+ transfectants was also insensitive to uncoupling agents but inhibited by thapsigargin (5 ± 6% of control) or preloading with the calcium chelator 1,2-bis(2-aminophenoxy)ethane-N,N,N′,N′-tetraacetate acetoxymethyl ester (BAPTA; 9 ± 7% of control) (21). Their similar pharmacological characteristics suggest that calcium signaling in astrocytes and Cx-transfected C6 cells are mediated by identical signaling cascades.
Both Primary and Neoplastic Glial Cells Express P2Y Purinergic Receptors.
Currently, pharmacological distinction between P2Y and P2X is primarily based on potency rank of agonists (26–29). By measuring the peak amplitude of ΔF/F in fluo-3-loaded cultures exposed to ATP analogs (10 μM) and normalized to ATP responses (10 μM) in matched cultures, we observed a similar rank order of potency for both primary astrocytes and C6 cells: 2MeSATP = 2Cl-ATP = ATP = UTP < ADP < AMP < αβmeATP = βγmeATP. This pharmacological profile support the notion that P2Y receptors, but not P2X receptors, are expressed by both primary and neoplastic astrocytes cells as previously suggested (29, 30). The EC50 of astrocytic Ca2+i responses occurred at 1.3 ± 0.2 μM ATP and at 2.3 ± 0.9 μM ATP in C6 cells. Cx expression was not associated with a significant change of EC50 to ATP exposure (P > 0.67).
Cx Expression Enhanced ATP Release.
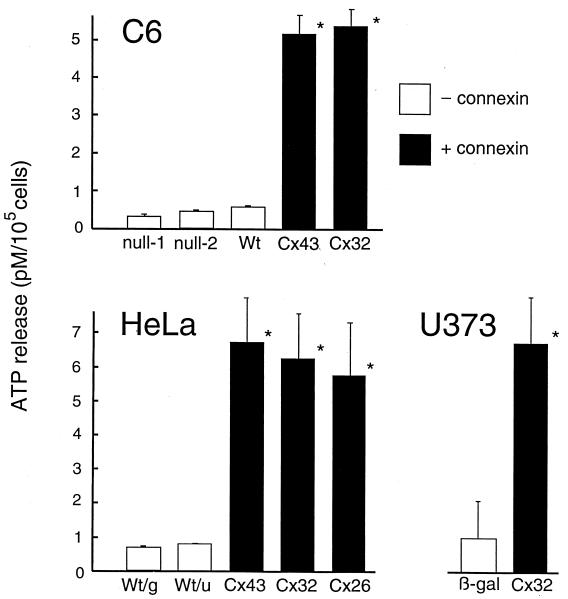
Because calcium waves propagate through cell free zones (ref. 8; our observations), and yet require connexins, we postulated that the connexin protein might play a role in calcium wave propagation over and above that of mediating physical coupling. One possibility is that connexins facilitate ATP release. To test this idea, ATP release from our C6 clones was compared by using the luciferin–luciferase assay (22). Stimulation of purinergic receptors by UTP consistently evoked a 10-fold higher ATP release in Cx expressing C6 clones compared with null-transfectants or wild-type C6 clones (Fig. 3). ATP release also was increased 10-fold after transfection of HeLa cells with either Cx43, Cx32, or Cx26 compared with poorly coupled wild-type clones (Fig. 3). Furthermore, ATP release was enhanced in a human glioblastoma cell line, U373-MG, that was transiently transfected to express Cx32. Thus, Cx expression facilitates ATP release after purinergic receptor stimulation in several cell lines independently. In each cell line, the effect was specifically related to purinergic activation: Exposure to calcium ionophore, bradykinin, or endothelin were all relatively inefficient in eliciting ATP release (Table 2), even though each of these compounds readily elicited substantial Ca2+i elevations (data not shown).
Figure 3.
Increased ATP release as a consequence of connexin expression in C6 glioma cells, HeLa cells, and the human-derived glioblastoma cell line, U373-MG. ATP release was evoked by exposure to 100 μM UTP and supernatant collected 10 min later. ATP release was quantified by using the luciferase–luciferin assay. Null-1 and null-2 are null-transfected C6 control cells, Wt represents wild-type C6 cells; Cx43, Cx32, and Cx26 are stable transfected clones overexpressing connexins. Results from C6 expressing Cx43 and Cx32 represent the mean of three independent clones. Wt/g and Wt/u are HeLa wild-type cells from Germany and U.S. U373 MG cells were transiently transfected with either cDNA for either Cx32 or β-galactosidase (β-gal). Cx expression enhanced ATP release 5- to 15-fold in all cell lines studied.
Table 2.
ATP release in C6 cells overexpressing Cx43, Cx32, or null-transfected controls
| Compounds/conditions | Cx43+ cells | Cx32+ cells | Control cells |
|---|---|---|---|
| Purinergic receptor antagonism | |||
| Suramin (100 μM) | BD | BD | BD |
| Reactive blue (30 μM) | BD | BD | BD |
| PPADS (50 μM) | 121 ± 14 | 105 ± 7 | 89 ± 17 |
| Elevation of Ca2+i | |||
| Ca2+-ionophore (40 μM) | 22 ± 3* | 21 ± 8* | 27 ± 12* |
| Bradykinin (25 μM) | 18 ± 5* | 24 ± 13* | 29 ± 4* |
| Endothelin (0.1 μM) | 16 ± 9* | 11 ± 7* | 14 ± 5* |
| Ionic composition | |||
| Cl−-free (gluconate substitute) | 3380 ± 1600* | 1530 ± 700* | 148 ± 17* |
| Na+-free (choline substitute) | 100 ± 15 | 63 ± 4* | 77 ± 23 |
| Ca2+-free | 1280 ± 390* | 870 ± 117* | 139 ± 15 |
| High K+ (100 mM) | 97 ± 10 | 132 ± 17 | 107 ± 9 |
| Hypoosmotic (220 mOs) | 173 ± 19* | 192 ± 17* | 147 ± 11* |
| Hyperosmotic (420 Osm) | 103 ± 12 | 108 ± 1 | 108 ± 14 |
| Ca2+ signaling inhibition | |||
| Thapsigargin, pretreatment (4 μM) | BD | BD | BD |
| BAPTA (10 μM) | 11 ± 7* | 27 ± 12* | BD |
| Cl− channel or transport inhibition† | |||
| SITS (3 mM) | 10 ± 2* | 1 ± 1* | BD |
| NPPB (100 μM) | 31 ± 5* | 30 ± 8* | BD |
| Furosemide (5 mM) | 25 ± 11* | 28 ± 13* | BD |
| Vesicular release | |||
| Latrotoxin (3 nM) | 2 ± 3* | 5 ± 6* | BD |
The fractional change in ATP release was calculated as the mean of ATP released in presence of compounds divided by the mean of ATP in matched cultures (Cx43, Cx32, or control) exposed to vehicle (mean ± SEM). BD, ATP release below detection. SITS, 4-acetamido-4′-isothiocyanatostilbene-2,2′-disulfonic acid; PPADS, pyridoxal phosphate-6-azophenyl-2′,4′-disulfonic acid tetrasodium; NPPB, 5-nitro-2-(3-phenylpropylamino)benzoic acid; BAPTA, 1,2-bis(2-aminophenoxy)ethane-N,N,N′,N′-tetraacetate.
Significant difference from control at P < 0.05 by t test; n = 8–35.
ATP Release and Calcium Wave Activity Are Modulated in Parallel.
ATP release has, in several systems, been associated with activation of a Cl− conductance (27, 29, 31, 32). To establish whether ATP efflux is linked to Cl− channel function, we evaluated the effect of the chloride channel inhibitor 5-nitro-2-(3-phenylpropylamino)benzoic acid (NPPB) on ATP release (32). NPPB, and two other inhibitors, 4-acetamido-4′-isothiocyanatostilbene-2,2′-disulfonic acid (SITS) and furosemide (33–34), decreased or blocked ATP release (Table 2) and reduced astrocytic Ca2+ signaling (Table 1). Pretreatment with thapsigargin or 1,2-bis(2-aminophenoxy)ethane-N,N,N′,N′-tetraacetate (BAPTA), both potent inhibitors of Ca2+ wave propagation, completely blocked stimulated ATP release without affecting basal release, further suggesting the role of ATP in Ca2+ signaling. ATP release was reduced by 50% in cultures exposed for prolonged time to gap junction inhibitors (data not shown). Replacement of Cl− (with gluconate) or removal of Ca2+ in the bathing medium were both associated with a 10- to 103-fold increase in ATP release, whereas other ionic substitutions had minimal effect. Similarly, removal of either chloride or Ca2+ also significantly enhanced astrocytic Ca2+ signaling. The maximal radius of calcium wave propagation in absence of extracellular Ca2+ was increased to 148 ± 14% (n = 11, P < 0.05, Student t test) compared with matched sister cultures. Substitution of chloride with gluconic acid increased the relative expansion of calcium waves by 181 ± 14% (n = 10, P < 0.05). Together, these data suggest that, upon activation of purinergic (P2Y) receptors, ATP is secreted by a mechanism sensitive to extracellular Cl− and Ca2+ and blocked by agents that inhibit Cl− channels.
DISCUSSION
On the basis of these results, we propose that interastrocytic calcium signaling is mediated by purinergic ATP receptors. Moreover, purinergic-receptor mediated ATP release is controlled by connexins. The observations do not exclude that coupling by gap junctions play a role in interastrocytic signaling during conditions not studied here or in other cell types. Our observations are consistent with ATP anion flow through a conductive pathway but does not exclude the existence of an ATP transport mechanism. Involvement of Cx-hemichannel in ATP efflux has intuitive appeal, because the pore diameter of gap junctions is sufficiently large to allow passage of polyphosphates and because ATP release is tightly linked to Cx expression. Furthermore, the facilitation of ATP release in Ca2+-deficient medium is consistent with that removal of extracellular Ca2+ promotes opening of Cx hemichannels (35, 36). In this regard, α-latrotoxin, a spider venom that mediates vesicular release did not evoke ATP release suggesting a nonvesicular pathway of ATP secretion (Table 2; ref. 37). The mechanism of ATP release may share features with the cystic fibrosis transmembrane conductance regulator or other members of the ATP-binding cassette family of proteins, such as the multidrug-resistant gene product P-glycoprotein (38–39). This family of transmembrane proteins are involved in the regulation of both Cl− and ATP conductance. The mechanism by which the cystic fibrosis transmembrane conductance regulator releases ATP, whether through the cystic fibrosis transmembrane conductance regulator itself, through Cl− channels, or by a separate mechanism, remains highly controversial (40–41). Further studies also will be required to define the cellular mechanisms whereby connexins regulate ATP release and calcium signaling. Nevertheless, our findings explain the requirement of connexin expression in purine-dependent Ca2+ signaling. As such, in addition to connexins, purinergic receptors may be a new target in conditions associated with abnormal astrocytic signaling, including spreading depression, seizure disorders, and secondary expansion of ischemic injury (42–44).
Acknowledgments
We thank K. Lerea for help with ATP measurements and S. Goldman, J. Etlinger, and V. Fried for critical reading of the manuscript. We thank Qun Gao for expert technical assistance. This study was supported by the National Institute of Neurological Disorders and Stroke/National Institutes of Health Grants NS130007 and NS135011. M.N. is an Established Investigator sponsored by The American Heart Association.
ABBREVIATIONS
- IP3
inositol trisphosphate
- Cx
connexin
- Ca2+i
intracellular calcium concentration
- CDCF
dicarboxy-dichlorofluorescein diacetate
- 2MeSATP
2-methylthioATP
- FRAP
fluorescence recovery after photobleach
- αGA
18α-glycyrrhetinic acid
References
- 1.Smith S J. Curr Biol. 1994;4:807–810. doi: 10.1016/s0960-9822(00)00178-0. [DOI] [PubMed] [Google Scholar]
- 2.Nedergaard M. Science. 1994;263:1768–1771. doi: 10.1126/science.8134839. [DOI] [PubMed] [Google Scholar]
- 3.Parpura V, Basarsky T A, Liu F, Jeftinija K, Jeftinija S, Haydon P G. Nature (London) 1994;369:744–747. doi: 10.1038/369744a0. [DOI] [PubMed] [Google Scholar]
- 4.Pasti L, Volterra A, Pozzan T, Carmignoto G. J Neurosci. 1997;17:7817–7830. doi: 10.1523/JNEUROSCI.17-20-07817.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Araque A, Parpura V, Sanzgiri R P, Haydon P G. Eur J Neurosci. 1998;10:2129–2142. doi: 10.1046/j.1460-9568.1998.00221.x. [DOI] [PubMed] [Google Scholar]
- 6.Newman E A, Zahs K R. J Neurosci. 1998;18:4022–4028. doi: 10.1523/JNEUROSCI.18-11-04022.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kang J, Li J, Goldman S A, Nedergaard M. Nat Neurosci. 1998;1:683–692. doi: 10.1038/3684. [DOI] [PubMed] [Google Scholar]
- 8.Hassinger T D, Guthrie P B, Atkinson P B, Bennett M V L, Kater S B. Proc Natl Acad Sci USA. 1996;93:13268–13273. doi: 10.1073/pnas.93.23.13268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Cotrina M L, Lin J H-C, Nedergaard M. J Neurosci. 1998;18:8794–8804. doi: 10.1523/JNEUROSCI.18-21-08794.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Osipchuk Y, Cahalan M. Nature (London) 1992;359:241–244. doi: 10.1038/359241a0. [DOI] [PubMed] [Google Scholar]
- 11.Palmer R, Yule D, Shewach D, Williams J, Fisher S. Am J Physiol. 1996;271:C43–C53. doi: 10.1152/ajpcell.1996.271.1.C43. [DOI] [PubMed] [Google Scholar]
- 12.Schlosser S F, Burgstahler A D, Nathanson M H. Proc Natl Acad Sci USA. 1996;93:9948–9953. doi: 10.1073/pnas.93.18.9948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Charles A C, Naus C C G, Zhu D, Kidder G M, Dirkson E R, Sanderson M J. J Cell Biol. 1992;118:195–201. doi: 10.1083/jcb.118.1.195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Elfgang C, Eckert R, Lichten-Frate H, Butterweck A, Traub O, Klein R, Hulser D, Willecke K. J Cell Biol. 1995;129:805–817. doi: 10.1083/jcb.129.3.805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Toyofuku T, Yabuki M, Otsu K, Kuzuya T, Hori M, Tada M. J Biol Chem. 1998;273:1519–1528. doi: 10.1074/jbc.273.3.1519. [DOI] [PubMed] [Google Scholar]
- 16.Cotrina M L, Kang J, Lin J H-C, Bueno E, Hansen T W, He L, Liu Y, Nedergaard M. J Neurosci. 1998;18:2520–2537. doi: 10.1523/JNEUROSCI.18-07-02520.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zhu D, Caveney S, Kidder G, Naus C. Proc Natl Acad Sci USA. 1991;88:1883–1887. doi: 10.1073/pnas.88.5.1883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Goldberg S, Bechberger J, Naus C. BioTechniques. 1995;18:490–497. [PubMed] [Google Scholar]
- 19.Nedergaard M, Desai S, Pulsinelli W. Anal Biochem. 1990;187:109–114. doi: 10.1016/0003-2697(90)90425-9. [DOI] [PubMed] [Google Scholar]
- 20.Wade M H, Trosko J E, Schindler M. Science. 1986;232:525–528. doi: 10.1126/science.3961495. [DOI] [PubMed] [Google Scholar]
- 21.Wang Z, Tymianski T, Jones O, Nedergaard M. J Neurosci. 1997;17:7359–7371. doi: 10.1523/JNEUROSCI.17-19-07359.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Nedergaard M, Goldman S, Desai S, Pulsinelli W. J Neurosci. 1991;11:2489–2497. doi: 10.1523/JNEUROSCI.11-08-02489.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sanderson M. Calcium Waves, Gradients and Oscillations, Ciba Foundation Symposium 188. Chichester: Wiley; 1995. pp. 175–194. [PubMed] [Google Scholar]
- 24.Davidson J S, Baumgarten I M. J Pharmacol Exp Ther. 1988;246:1104–1107. [PubMed] [Google Scholar]
- 25.Venance L, Stella N, Glowinski J, Giaume C. J Neurosci. 1997;17:1981–1992. doi: 10.1523/JNEUROSCI.17-06-01981.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Abbracchio M, Burnstock G. Pharmacol Ther. 1994;64:445–475. doi: 10.1016/0163-7258(94)00048-4. [DOI] [PubMed] [Google Scholar]
- 27.Wang Y, Roman R, Lidofsky S D, Fitz J G. Proc Natl Acad Sci USA. 1996;93:12020–12025. doi: 10.1073/pnas.93.21.12020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Gu J G, MacDermott A B. Nature (London) 1997;389:749–752. doi: 10.1038/39639. [DOI] [PubMed] [Google Scholar]
- 29.King B, Neary J, Zhu Q, Wang S, Norenberg M, Burnstock G. Neuroscience. 1997;74:1187–1196. doi: 10.1016/0306-4522(96)00209-6. [DOI] [PubMed] [Google Scholar]
- 30.Kastritsis C, Salm A, McCarthy K. J Neurochem. 1992;58:1277–1284. doi: 10.1111/j.1471-4159.1992.tb11339.x. [DOI] [PubMed] [Google Scholar]
- 31.Mitchell C, Carre D, McGlinn A, Stone R, Civan M. Proc Natl Acad Sci USA. 1998;95:7174–7178. doi: 10.1073/pnas.95.12.7174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Zeng W, Lee M, Muallem S. J Biol Chem. 1997;272:32956–32965. doi: 10.1074/jbc.272.52.32956. [DOI] [PubMed] [Google Scholar]
- 33.Nilius B, Eggermont J, Voets T, Droogmans G. Gen Pharmacol. 1996;27:1131–1140. doi: 10.1016/s0306-3623(96)00061-4. [DOI] [PubMed] [Google Scholar]
- 34.Kimelberg H, Goderie S, Higman S, Pang S, Waniewski R. J Neurosci. 1990;10:1583–1591. doi: 10.1523/JNEUROSCI.10-05-01583.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Li H, Liu T, Lazrak A, Peracchia C, Goldberg G, Lampe P, Johnson R. J Cell Biol. 1996;134:1019–1030. doi: 10.1083/jcb.134.4.1019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Hofer A, Dermietzel R. Glia. 1998;24:141–154. doi: 10.1002/(sici)1098-1136(199809)24:1<141::aid-glia13>3.0.co;2-r. [DOI] [PubMed] [Google Scholar]
- 37.Parpura V, Liu F, Brethorst S, Jeftinija K, Jeftinija S, Haydon P G. FEBS Lett. 1995;360:266–270. doi: 10.1016/0014-5793(95)00121-o. [DOI] [PubMed] [Google Scholar]
- 38.Abraham E, Prat A, Gerweck L, Seneveratne T, Arceci R, Kramer R, Guidotti G, Cantiello H. Proc Natl Acad Sci USA. 1993;90:312–316. doi: 10.1073/pnas.90.1.312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Schiebert E, Egan M, Hwang T-A, Fulmer S, Allen S, Cutting G, Guggino W. Cell. 1995;81:1063–1073. doi: 10.1016/s0092-8674(05)80011-x. [DOI] [PubMed] [Google Scholar]
- 40.Reddy M, Quinton P, Haws C, Wine J, Grygorczyk R, Tabcharani J, Hanrahan J, Gunderson K, Kopito R. Science. 1996;271:1876–1879. doi: 10.1126/science.271.5257.1876. [DOI] [PubMed] [Google Scholar]
- 41.Cantiello H, Jackson G, Grosman C, Prat A, Borkan S, Wang Y, Reisen I, O’Riordan C, Ausiello D. Am J Physiol. 1998;274:C799–C809. doi: 10.1152/ajpcell.1998.274.3.C799. [DOI] [PubMed] [Google Scholar]
- 42.Nedergaard M, Cooper A J L, Goldman S A. J Neurobiol. 1996;28:433–444. doi: 10.1002/neu.480280404. [DOI] [PubMed] [Google Scholar]
- 43.Heineman U, Eder C, Lab A. In: Neuroglia. Kettenman H, Ransom B R, editors. New York: Oxford Univ. Press; 1995. pp. 936–949. [Google Scholar]
- 44.Lin J H-C, Weigel H, Cotrina M L, Liu S, Bueno E, Hansen A J, Hansen T W, Nedergaard M. Nat Neurosci. 1998;1:494–500. doi: 10.1038/2210. [DOI] [PubMed] [Google Scholar]