Abstract
Trypanosomiasis is one of the major parasitic diseases for which control is still far from reality. The vaccination approaches by using dominant surface proteins have not been successful, mainly due to antigenic variation of the parasite surface coat. On the other hand, the chemotherapeutic drugs in current use for the treatment of this disease are toxic and problems of resistance are increasing (see Kennedy (2004) and Legros et al. (2002)). Therefore, alternative approaches in both treatment and vaccination against trypanosomiasis are needed at this time. To be able to design and develop such alternatives, the biology of this parasite and the host response against the pathogen need to be studied. These two aspects of this disease with few examples of alternative approaches are discussed here.
1. Introduction
Trypanosomiasis is a fatal disease of both human and livestock. Besides death, it causes a heavy economic loss mainly in Africa. The etiological agent of the disease is a unicellular flagellated protozoan parasite of the genus Trypanosoma. Trypanosomes (20–30 μm × 1.5–3.5 μm) are blood-borne unicellular protozoan parasites dwelling in various body and tissue fluids. These parasites are motile due to the undulatory motion of their flagellum. The parasite is known for more than a century, but still the control of the disease remains elusive. Trypanosomes are the causative agent of human African trypanosomiasis (HAT), also known as “sleeping sickness”. The term “sleeping sickness” describes the deregulation of the sleep-wake cycle and the intrasleep cycle which is observed in the late stage of the disease [1].
Sleeping sickness is caused by Trypanosoma brucei rhodesiense in Eastern and Southern Africa and by Trypanosoma brucei gambiense in Western and Central Africa. Both protozoan species are morphologically indistinguishable, but have drastically different epidemiological features. Both forms of the sleeping sickness affect the central nervous system. The typical East African form of trypanosomiasis is characterized by a rapid and acute development of the disease, and untreated patients can die within weeks or months of the infection, whereas the West African form of this disease is more chronic that can last for several years. According to WHO reports, African sleeping sickness is the third most important contributor to the global burden of the parasitic diseases after malaria and schistosomiasis, if the Disability Adjusted Life Year (DALY) figures (i.e., loss of healthy life years by premature mortality and disability) are considered [2]. More than 60 million people are at risk of infection with human African trypanosomiasis, with about 45,000 new cases reported annually. It is estimated that at least 300,000–500,000 people are presently infected. However, less than 4 million people are under surveillance and as such, it is estimated that less than 10% of the new cases are diagnosed and treated [3].
Several species of hematophagous glossina, commonly known as tsetse flies (Glossina spp.) are the vectors of the African trypanosomes and are responsible for cyclical transmission of the parasitic protozoan between numerous vertebrate hosts. The vector is distributed over a wide range of habitats covering about 10 million square kilometer potential grazing lands in 37 countries which are rendered unsuitable for livestock breeding and farming in Africa [4].
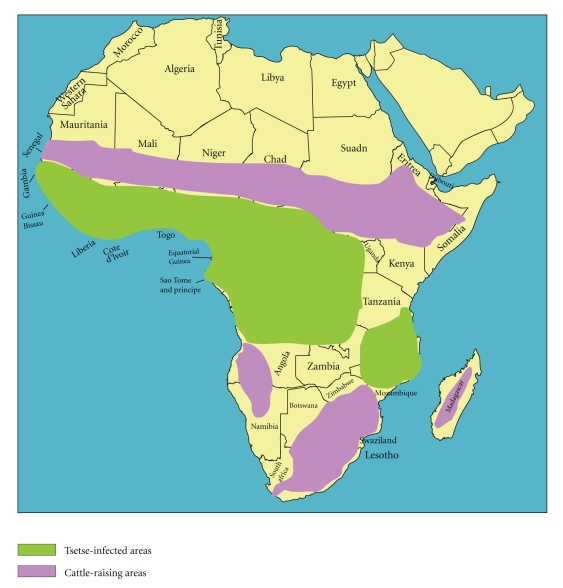
Trypanosome infections in livestock are known as Nagana and Surra. Animal trypanosomiasis, caused by a wider number of trypanosome species and carried with higher prevalence by a greater number of glossina species, is invariably the greater epidemic across the African continent with direct economic consequences. In general, trypanosome infections that threaten livestock have a 100 to 150-fold higher prevalence than the HAT [5]. Historically, the impact of animal trypanosomiasis has been so profound, that it has influenced the migration routes of cattle-owning tribes that were forced to avoid the G. morsitans “fly-belts” [6] (Figure 1), as well as the movements of early European and Arab settlers who depended on horses and oxen in Africa [7].
Figure 1.
Distribution of tsetse and cattle raising area in Africa http://pathmicro.med.sc.edu/lecture/trypanosomiasis.htm.
Although trypanosomiasis is often referred to as African trypanosomiasis, certain trypanosomes do cause infections outside this continent. T. evansi, the causative agent of “surra” occurs not only in Africa, but also in Central and South America, the Middle East, and Asia. It causes a disease in camels, horses, cattle, pigs, buffaloes, and dogs. In Southeast Asia, T. evansi infection is a disease of economic importance since it affects the health of buffalo, cattle, and swine [8]. The acute stage symptoms of this disease include abortion, central nervous system disorders, and even death, while in the chronic condition; working capacity and productivity of the animals are affected. Even though it is generally considered as a livestock disease, there are now recent reports of the human T. evansi trypanosomiasis in India [9, 10].
2. General Features of Trypanosomes
2.1. Classification of Trypanosome
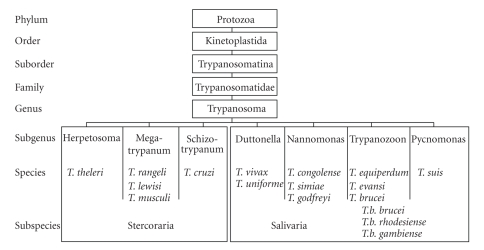
The protozoal parasite trypanosomes are grouped in the order “kinetoplastida” because of the presence of a kinetoplast (discussed later). Based on the mode of transmission by their insect vector, the genus Trypanosoma is divided in two main groups: stercoraria and salivaria [11, 12] (Figure 2). The development of stercoraria parasites takes place in the intestinal track of the invertebrate vector and the infection to the vertebrate is via feces. T. cruzi, the causative agent of Chagas' disease, is an example of the stercoraria group. On the other hand, salivarian parasites colonize the stomach of their invertebrate vector, but never pass to the intestinal track. Rather, they migrate towards the salivary gland of the vector where the infectious form for vertebrate host develops. Infection of the vertebrate occurs via saliva when the vector bites in order to take the blood meal. The African parasites, T. brucei, T. congolense, T. evansi, and T. equiperdum all belong to the salavarian group. T. brucei has three subgenera; while T. b. brucei is the causative agent of Nagana, a cattle disease in Africa, T. b. rhodesiense and T. b. gambiense are the causative agents of the sleeping sickness in human. The other two species T. congolense and T. vivax are the major causative agents of animal trypanosomiasis in Africa. T. equiperdum is the causative agent of an equine venereal disease that is called “dourine” where the parasites are transmitted during coitus. T. evansi causes a livestock disease called “surra”.
Figure 2.
Classification of trypanosomes.
2.2. Life Cycle
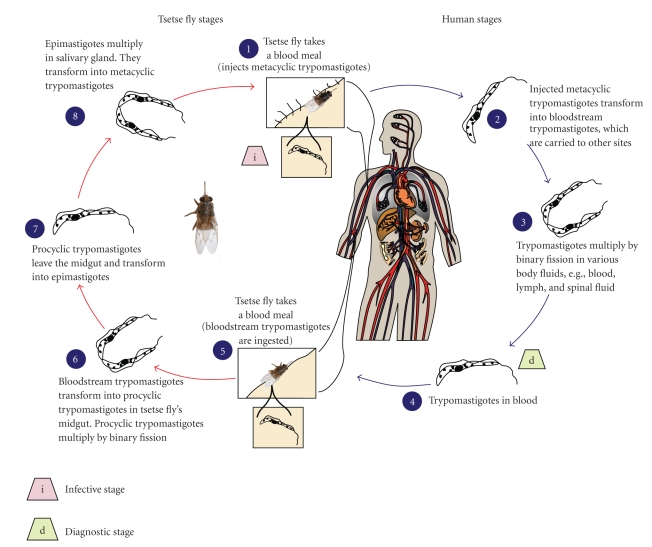
Trypanosomes are the excellent examples of organisms that display an extreme adaptation to their environment, in many cases because they must evade the immune response of the host. African trypanosomes are transmitted between mammalian hosts by tsetse flies. However, in each host, the parasites undergo many life cycle stages involving forms with discrete morphologies, patterns of gene expression, and proliferation status. In each case, these developmental changes are programmed precisely [13]. Infection in the mammalian host begins when the infective stage, known as the metacyclic stage, is injected intradermally by the tsetse fly (Figure 3). The organisms rapidly transform into the blood-stage trypomastigotes (long, slender forms), and divide by binary fission in the interstitial spaces at the site of the bite. The buildup of metabolic wastes and cell debris leads to the formation of a “chancre”. In the mammalian host, the metacyclic parasites rapidly undergo cell cycle reentry and morphological changes, and exchange the restricted repertoire for antigenic variation that is the characteristics of the metacyclic forms with a more elaborate system of the bloodstream forms [14]. Once established in the mammalian host, the bloodstream parasite is heterogeneous [15, 16] comprising the proliferative slender forms during the ascending phase of parasitemia and the nonproliferative stumpy forms at the peak of parasitemia [17]. The transition from the morphological extremes (i.e., the slender versus stumpy forms) involves a progression from proliferation to cell cycle arrest, accompanied by a series of biological and morphological transformations [18, 19]. Once stumpy forms develop during the course of parasitemia, the population is preadapted for transition to the procyclic forms, which occupy and proliferate in the midgut of the tsetse. The key features of the stumpy formation are the cell cycle arrest, the elaboration of some mitochondrial activities, and a relative resistance to lysis by antibodies [20, 21] or to the proteolytic environment that might be encountered in the midgut of tsetse [21, 22]. When the vector-fly (tsetse) bites to an infected individual, it takes the parasites with the blood meal. The parasites undergo metabolic changes in the midgut of the fly. They lose their surface coat, which consists of about 107 molecules of the Variant Specific Surface Glycoprotein (VSG), and transform into the proliferative procyclic forms. In this form, they express their own surface proteins called the Procyclic Acidic Repetitive Proteins (PARPs, or procyclins). The defining events of the differentiation from the bloodstream forms to the procyclic forms are the loss of VSG and gain of the procyclins. VSG loss occurs very rapidly and involves the combined action of glycosyl-phosphatidyl-inositol-specific phospholipase C (GPI-PLC) and a proteolytic cleavage of the VSG via a zinc metalloprotease [23–26]. The transformation to procyclic form also changes the energy generation from being exclusively based on glycolysis in the bloodstream to a mitochondrion-based respiratory system, which requires a structural elaboration and the metabolic activation of organelle [27]. For successful transmission, the parasite undergoes two stages of differentiation in the fly: first, establishment in the midgut and then maturation in the mouthparts or the salivary gland. It is generally thought that during normal development in the fly, there are no intracellular stages and the parasites do not cross an epithelial barrier to enter the fly. After proliferation in the tsetse midgut, the parasite migrates to the salivary gland. The epimastigote forms generated there attach to the gland through elaboration of the flagellar membrane. After further multiplication, the parasite undergoes division arrest, re-acquires a VSG coat, and is released into the salivary gland lumen, in preparation for an inoculation into a new mammalian host [27].
Figure 3.
Life cycle of African trypanosomes, http://www.dpd.cdc.gov/dpdx/HTML/TrypanosomiasisAfrican.htm.
If the tsetse flies ingest more than one strain of trypanosome, there is the possibility of genetic exchange between the two strains, generating an increase in the genetic diversity in an organism that may not have a true sexual cycle. Indeed, it was shown by laboratory crosses that genetic exchange in the African trypanosome is possible [28–33]. Precisely at which stage of the life cycle this genetic exchange takes place is equivocal, suggesting at the midgut stage [34], in the salivary gland of the fly [35], and possibly at the proventriculus and foregut stage [36]. Though there are conflicting results for the stage at which this exchange takes place, it is shown that this is not a compulsory process. The mechanism of genetic exchange in T. brucei is still unclear though it appears to be a true sexual process involving meiosis [35]. However, no haploid stage has been observed and the intermediates in the process are still a matter of conjecture. The frequency of sex in trypanosomes in nature is also a matter for speculation and controversy, with conflicting results arising from population genetics [37, 38].
In contrast to tsetse transmitted trypanosomes, T. evansi is transmitted mechanically by the blood sucking insects, in Asia especially by the horseflies (Tabanus spp.) and the stable flies (Stomoxys spp.), and in Africa the tsetse fly, like other biting flies, can act as mechanical vector. In South and Central America, in addition to blood sucking flies, T. evansi can also be transmitted by the vampire bats (Dosmodus rotundus). Besides mechanical transmission by insects and vampire bats, T. evansi can be transmitted through milk or during coitus [39]. Developmental stages were not observed in any of the vectors mentioned above. A procyclic or insect stage does not exist in T. evansi which is attributed to the lack of maxi circles in the kinetoplast DNA (discussed later) [40].
3. Special Features of Trypanosomes
Kinetoplasts are eukaryotes and hence exhibit conventional features such as the presence of a nucleus delimited by a nuclear membrane and organelles such as the endoplasmic reticulum, the Golgi apparatus, the endo/exocytosis system, the mitochondrion, and so forth [41]. However, many of these organelles exhibit specific and sometimes extreme features often found only in the kinetoplastids. As trypanosomes have a dual host life cycle, they have some specific adaptation characteristics at different levels, that is, DNA, RNA, and cellular organization. This includes the presence of a kinetoplast DNA, flagellum and flagellar pocket, unique gene regulation, RNA-editing, and the presence of glycosomes.
3.1. Kinetoplast DNA
Being a eukaryote, trypanosomes contain mitochondria, however, there is only a single mitochondrion per cell, which is extremely large and elongated, and its whole DNA content is condensed in a substructure called the kinetoplast [42]. The mitochondrial DNA makes up 10–20% of the total cellular DNA with an unusual network in the kinetoplast which is called kinetoplast DNA (kDNA) [43]. The kDNA is a planer network composed of several thousand topologically interlocked DNA circles. The network contains two types of circles of different sizes: minicircles of about 1 kb and maxicircles of 22 kb [44, 45]. Maxicircles number in dozen per cell with conserved sequences and code for ribosomal RNA and some mitochondrial proteins [46]. Many of the maxicircle transcripts are cryptic and require editing to form a functional mRNA. Editing specificity is controlled by the minicircle-encoded guide RNA (gRNA) that serve as a template [47]. The minicircles are in high copy number (5000–10000 per cell) and their sequences vary between each other [48]. No transfer RNA (tRNA) genes are found neither in the maxi-circle nor the minicircles [49, 50]. So all the tRNAs necessary for protein translation of mitochondrial origin may be nuclear-encoded and imported from the cytosol into the mitochondrion [49, 51]. Unlike other trypanosomes, the kDNA of T. evansi does not contain maxicircle [40].
3.2. The Flagellum and Flagellar Pocket
Trypanosomatids possess a single flagellum that exits from a flagellar pocket, a specialized invagination of the plasma membrane where the entirety of endocytosis/exocytosis traffic takes place. In Trypanosoma spp., the flagellum is attached along the cell body for most of its length, with the exception of the distal tip [42]. The site of attachment defines a specialized region of the flagellum and of the cell body called the flagellum attachment zone (FAZ). The flagellum could accomplish several functions. Firstly, it is involved in the cell motility; trypanosomes are actively motile cells, swimming at average speeds of 10–30 μm per second in the culture medium [52]. The flagellar motility is required for the viability of the bloodstream trypanosomes [53]. A striking feature is the fact that trypanosomes swim with their flagellum leading, that is, the flagellum drags the cell behind it. This is related to the way wave forms are initiated: from tip to base, and not from base to tip as seen in the majority of flagellated organisms [42]. Secondly, the flagellum is involved in the attachment of the parasite to the host surfaces. Thirdly, the flagellum plays a role in the morphogenesis and the cell division [54, 55]. The flagellar pocket is a flask-shaped invagination of the plasma membrane where the flagellum emerges out from the plasma membrane [56]. This pocket constitutes a highly differentiated region that facilitates internalization of host macromolecules, while restricting host access to the exposed, endocytic receptors of the parasites [57]. The contribution of the flagellar pocket to protein trafficking, immune evasion, and other processes has been recently reviewed [58].
3.3. RNA Editing
Most trypanosomatid mitochondrial mRNAs undergo RNA editing by which the precursor mRNA (pre-mRNA) sequences are changed, often extensively, by the insertion and less frequently the deletion of uridine nucleotides (Us) [59]. The pre-mRNAs are encoded in the maxicircles, whereas the minicircles encode gRNA that specify the editing. The editing in T. brucei is catalyzed by compositionally and functionally distinct multiprotein complexes, called editosomes [60]. In this editing process up to half of the nucleotides can be added. This is a posttranscriptional process responsible for correcting the coding sequences of the mRNAs. Recently it had been shown that the structural conformation of gRNA and the thermodynamic stability of the gRNA-mRNA binary complex are very important for the editing to proceed [61]. It has been shown by comparing the rRNA synthesis in the bloodstream and the insect life-stages that the mitochondrial gene expression levels are controlled not at the transcriptional level, but rather by a mechanism which likely modulates the stability of the mature RNAs [62]. Thus it is speculated that editing may play a central role in controlling different mitochondrial functions during the development cycle of the trypanosomes.
3.4. The Nuclear Genome
The nuclear chromosomes of T. brucei can be grouped into three classes according to their sizes: 11 pairs of megabase chromosomes (1 to 6 Mb) that contain the house keeping genes and represent about 80% of the nuclear DNA content, a few intermediate-sized chromosomes (200 to 900 kb) and an undetermined number of minichromosomes (in the range of 100 that are 50 to 150 kb) which comprise about 10% of the nuclear DNA [63, 64]. The sequence of the 11 megabase-sized chromosomes of Trypanosoma brucei contains 9068 predicted genes, including approximately 900 pseudogenes and approximately 1700 T. brucei-specific genes. A large subtelomeric arrays contain an archive of 806 VSG genes used by the parasite to evade the mammalian immune system. Most VSG genes are pseudogenes, which may be used to generate expressed mosaic genes by ectopic recombination [65].
3.5. Glycosomes
The bloodstream form of trypanosomes has adapted its life in an abundance of glucose and relies entirely on glycolysis and substrate level phosphorylation for their energy production [66, 67]. The first seven to nine enzymes of the glycolytic pathway are present in the glycosomes, the peroxisome-related small globular organelles [68], found in all kinetoplastida. Expression of either phosphoglycerate kinase or triosephosphate isomerase in the cytosol inhibits parasite growth, suggesting that correct localization of the glycolytic enzymes is important [69, 70]. Various results, including the metabolic modeling, suggest that in bloodstream T. brucei the glycosome plays a vital role in the regulation of glycolysis [71–73]. The glycosomal membrane of T. brucei is impermeable to several metabolites [72], implying the presence of specific glycosomal metabolite receptors. Indeed, with the proteomic data it has been shown that there are certain receptors/carrier proteins in the glycosomal membrane [74]. Because the glycosomes of the bloodstream T. brucei together make up-to about 4% of the total cellular volume, and the enzymes are present at relatively high concentrations within the organelle, it was postulated that their confinement to a small volume overcomes a diffusion limitation of the metabolites between the glycolytic enzymes [66]. This led to the more general notion that glycosomes would enable the trypanosomatids to maintain their high glycolytic flux. However, several arguments have been put forward that render such an explanation unlikely [71]. Even though the glycosomal protein content is dominated by the glycolytic enzymes, representing up to 90%, the glycosomes are not only involved in the glycolysis but are predicted to carry out also the gluconeogenesis, reactions of the hexose-monophosphate pathway, purine salvage and pyrimidine biosynthesis, β-oxidation of fatty acids, fatty acid elongation, and the biosynthesis of ether lipid [75].
4. Unique Features of Trypanosomes
4.1. Antigenic Variation and Immune Evasion
Trypanosomes growing in the bloodstream of mammalian host need mechanisms to circumvent the host immune response. Antigenic variation is one of the most spectacular adaptive mechanisms exhibited by the African trypanosomes. The bloodstream form of trypanosomes is entirely covered with a monolayer made of 107 copies of the VSG, which is a major antigen of the parasite whose antigenicity is in continuous changes. During the ascending phase of the parasitemia, the majority of parasites are of the same antigenic type (called homotype). The host immune system recognizes this homotype and makes antibodies against it. As the parasites of the major variable antigenic type (VAT) are eliminated the parasitemia goes in descending phase but at the same time, the parasites expressing the heterotype or the minor VATs are multiplying and one of them overgrows others. As a result this one becomes the new homotype, leading to a new wave of parasitemia and resulting in a long-lasting chronic infection. So expression of the VSG is central in the antigenic variation process and eventually for exhausting the host immune system in the benefit of the parasite. For the immune evasion, trypanosomes have also developed another method; macromolecular trafficking mechanism [76] whereby the VSG-complexed with antibody are sorted and endocytosed [77]. This mechanism most probably protects the parasites from the complement-mediated killing and as such the trypanosomes escape from the host immune system. For the purpose of escaping from the host immune system, trypanosomes have more mechanisms such as capping of the surface bound immune factors, restriction of the invariant receptors in the flagellar pocket, rendering them inaccessible to the host immune effectors [58].
4.1.1. VSG Expression
The trypanosome genome contains hundreds of VSG genes (VSG) of which very few (7%) are fully functional (encoding all recognizable features of known functional VSG), whereas the majority (66%) are full-length pseudogenes (with frame shifts and/or in-frame stop codons) [65]. Most of these VSGs are clustered in the subtelomeric arrays. Transcription of the VSG occurs in one of the telomeres of the large chromosomes, which contain the VSG expression sites (VSG ESs) [78]. These expression sites are polycistronic transcription units having expression site associated genes (ESAGs) upstream of the VSG. These polycistronic mRNAs are matured by polyadenylation and addition of a spliced leader sequence by a process called transsplicing. Among the different expression sites only one is active at a given time. Thus only one of the VSG molecules is present within the trypanosome surface coat, resulting in the homogeneous display of an identical surface coat. Transcription starts simultaneously in all VSG ESs, but only in the “active” one there is complete transcription and all the others are aborted [79, 80]. In the rapidly dividing long slender form of trypanosomes, the active expression site was found to be present in a specialized region called expression site body (ESB) [81] and no similar structure was detected for the silent expression sites.
4.1.2. Mechanism of Antigenic Variation
There are several studies trying to unravel the different systems involved in the antigenic variation (reviewed by Pays et al. [80, 82, 83]. A first mechanism for the parasite to perform a VSG switch is to change the expression site. By switching off the active expression site [84] and activating a silent expression site, a VSG switch takes place. However, as the VSG genes are transcribed as polycistronic units, at the same time an ESAG switch will occur. This type of VSG switching is called in situ activation. This process could be one of the mechanisms which makes it possible for the trypanosomes to survive in various conditions and as such extending the host range [85]. A second system of the antigenic variation occurs possibly via the VSG gene rearrangements including a reciprocal recombination [86] and a gene conversion [87]. In this system, the active site is not changed (and the ESAGs remain the same) and only the VSG gene undergoes a modification. During the reciprocal recombination, the whole transcribed VSG gene in one telomere is replaced by another VSG gene present on a silent telomere [88]. This system occurs by crossing over in the 70 bp repeat region flanking every VSG gene on the promoter site of the gene. For the gene conversion, an actively transcribed VSG gene or at least a part encoding for the surface epitopes is replaced by a copy of another VSG gene or a pseudogene, present on a silent telomere or another location in the genome. This event occurs more frequently than the reciprocal recombination [86].
4.2. Resistance to Normal Human Serum
Humans and some other primates [89, 90] are resistant to most of the trypanosomes because of the presence of a trypanolytic factor in their serum. The trypanolytic factor of the human serum is discussed below. Two subspecies of T. brucei, T. b. gambiense and T. b. rhodesiense, are resistant to the human serum trypanolytic factor and thus are able to infect human causing HAT or “sleeping sickness”.
The resistance to human serum, at least in case of T. b. rhodesiense, is linked to an antigenic variation. It had been shown that a gene called serum resistance-associated (SRA) gene is only expressed in the resistant clone of T. b. rhodesiense and not in the sensitive clone of the same parasite [91–93]. The SRA was necessary and enough for the resistance against normal human serum (NHS) [94]. It was found that the SRA is associated, as an ESAG, to a specific VSG ES, termed R-ES, which is selected in the human serum by antigenic variation [94]. The expression of SRA by T. b. rhodesiense is not a permanent phenomenon as it looses the resistance to NHS in the absence of the trypanolytic factor of the human serum, for example, after several passage in mice [95] or the T. b rhodesiense which are found in nonprimate animals [96]. The SRA contains all the characteristics of a VSG except that the region coding for the surface-exposed epitope is missing because of an in-frame deletion [97–100]. So SRA is a kind of truncated VSG. As in the VSGs, the SRA contains the N-terminal α-helices [100], which in case of VSG are involved in a coil-coil interaction with the adjacent VSG to make the dimer [101], whereas this helix of the SRA is responsible for neutralizing the human serum trypanolytic factor by coil-coil interaction [102].
4.2.1. Trypanolytic Factor of the Normal Human Serum (NHS)
It has been known already for a century, reported by Laveran and Mesnil between 1902 and 1912, (cited in [98, 103], that the sera from humans and other primates such as baboon and mangabey can kill trypanosomes, although there were differences in the killing activities within these sera. Recently it has been shown that gorilla serum also has trypanolytic activity but the serum from chimpanzees, which are evolutionarily most close to the human [104], does not have trypanolytic capacity [89]. Further characterization of trypanolytic factor (TLF) in the human serum showed that the factor is associated with high-density lipoprotein (HDL) [105, 106], and endocytosis of these HDL particles by the trypanosome is necessary for the lysis [107, 108]. The trypanosomal receptor for the HDL is yet to be defined, but it is most likely to be a lipoprotein scavenger receptor [109]. Two TLF complexes have been shown to be present in the human serum; (i) TLF1, a 500 kDa high-density lipoprotein complex composed of apolipoprotein A-I (apoA-I), haptoglobin-related protein (Hpr), apoliprotein L-I (APOL1), human cathelicidin antimicrobial peptide (hCAP18), GPI-specific phospholipase D (GPI-PLD), apolipoprotein A-II, and paraoxanase [106, 110], and (ii) TLF2, a 1000 kDa lipid poor immunocomplex composed of apoA-I, Hpr, APOL1, hCAP18, GPI-PLD, and IgM [111–113]. Among these components, Hpr has been thought for a long time to be the active trypanolytic component of TLF, because (i) it was considered to be the component recognized by parasite surface [114], (ii) anti-haptoglobin antibodies, which cross-react to the HPR, could inhibit the TLF mediated trypanolysis [110, 115], and (iii) it is not expressed in chimpanzees serum which lacks the trypanolytic capacity [90]. But now there are more and more confirmative evidences showing that APOL1 is the trypanolytic factor of normal human serum [89, 98, 102, 103, 116]. There are still ongoing discussions on which component of TLF is the most important for the trypanolytic activity. Some have suggested that there is a synergistic effect of Hpr and APOL1 for which HDL provides the platform [117]. Others have shown three human apolipoproteins (Hpr, APOA1, and APOL1) acting cooperatively for maximal killing capacity; however; the truncated APOL1 did not function in transgenic animal [118]. Furthermore, recently it has been shown that baboon contains orthologs of APOL1 and Hpr and when these two genes were expressed together with APOA-1; there was full protection against both animal and human infective T. b. rhodesiense infection [119]. In addition, TLF can also ameliorate infection by the intracellular parasite Leishmania [120, 121].
In humans, the APOL family has six members (APOL1 to APOL6) which all are clustered in the chromosome 22 [122–124]. APOL1 is only found in the sera from humans and gorilla which have the trypanolytic capacity but not in the chimpanzee serum which lacks the trypanolytic potential [89]. There are growing evidences that support APOL1 to be the active trypanolytic component of TLF: (i) APOL1 depleted NHS lost the trypanolytic capacity, (ii) this lost function could be rescued by adding a physiological concentration of both natural and importantly the recombinant APOL1, (iii) physiological concentrations of APOL1 lysed trypanosomes in fetal calf serum (FCS), showing that the addition of only APOL1 is enough to have trypanolytic activity in FCS, and (iv) APOL1 was lytic only to the NHS sensitive strain and not to the NHS resistant strain, showing that killing by purified APOL1 is not due to the toxicity acquired in the purification process [98, 102, 103]. APOL1 contains a signal sequence (amino acid 1-27) and the secreted protein could be divided in three domains in relation with trypanolysis: a pore-forming domain, a membrane-addressing domain, and an SRA-interacting domain [116]. The pore-forming domain, which spans from Met60 to Trp235, has structural and functional similarities with the pore-forming domain of bacterial colicins. Next to the pore-forming domain there is the membrane-addressing domain, Ala238 to Pro304, which is predicted to bind to HDL particles only in neutral pH such as blood. At the C-terminal of APOL1, there is an α-helix which interacts with the SRA of T. b. rhodesiense and this interaction leads to a loss of trypanolytic capability of the APOL1. The pore-forming and the membrane addressing domains are necessary for the trypanolytic capacity whereas the SRA-interacting domain is dispensable. In fact, removal of this C-terminal domain, by which the SRA can no longer interact, makes the APOL1 even lytic to NHS-resistant T. b. rhodesiense [98]. Therefore, this truncated molecule can be envisaged as a possible therapeutic molecule of human origin.
4.2.2. Mechanism of Trypanolysis by APOL1
The mechanism of trypanolysis by APOL1 has been proposed by Pays et al. [93] and Baral et al. [103]. The APOL1 which is associated with HDL particles are internalized by the blood-stream form of trypanosomes via HDL-receptor mediated endocytosis in the flagellar pocket. The endocytic pathway leads the APOL1-loaded HDL to the endosomes and then to the lysosomes. The acidic pH of the lysosome induces a conformational change in the pH sensitive membrane-addressing domain of the APOL1. This conformational change would cause a dissociation of the APOL1 from the HDL and leads to association with the lysosomal membrane. At this stage the pore-forming domain would be able to form ionic-pore in the lysosomal membrane causing an influx of chloride ions (Cl−) from the cytoplasma to the lysosomal lumen. There would be a compensatory influx of Cl− to the cytoplasma via ion channels in the plasma membrane. In the plasma membrane the anionic influx can be accompanied by some other cationic influx as well [112] as Hpr is suggested to permeabilize both anionic and zwitterionic membranes [125]. The ionic influx leads to the movement of water to the lysosome causing an osmotic swelling of this organelle. This uncontrolled swelling of the lysosome can cause an increased intracellular pressure which might cause the plasma membrane damage and ultimately kill the parasite. The SRA protein expressed by T. b. rhodesiense interacts with the C-terminal domain of APOL1 in the endosomes and/or lysosomes which prevents the APOL1 being able to form the pore, and as such makes the parasite resistant to the APOL1/ NHS.
5. The Surface Proteins of the Trypanosomes
Trypanosomes are covered by their stage-specific surface proteins. In the bloodstream form they are covered by the VSG and in the procyclic insect form they are covered by another protein called procyclins. The procyclins are GPI-anchored glycoproteins with either five or six pentapeptide repeats (GPEET procyclin) or up to 30 glutamic acid-prolin dipeptide repeats (EP procyclin) [126, 127].
5.1. Variant-Specific Surface Glycoprotein (VSG)
The VSG is the most abundant surface protein in the bloodstream form of the trypanosomes. It forms a dense surface coat of 12-15 nm over the entire surface of the trypanosome [128] and accounts for about 10% of the total protein content of the bloodstream form of the parasite and more than 95% of the externally disposed cell surface protein [128]. Nearly the entire cell surface of the parasite and the flagellum is covered by the VSG. The bloodstream form of the trypanosome is coated with a continuous layer of approximately five million densely packed identical homodimers of VSGs [128, 129] that provide the parasite a defense barrier against both innate and specific immune effectors of the host [130].
5.1.1. Structure of VSG
VSGs are antigenically distinct due to extensive differences in primary sequence. Despite the differences in the primary structure, it is believed that the VSGs have a conserved tertiary structure which could explain how arrangement of the VSGs with different primary sequences can perform the same apparent function of producing a monolayer barrier that prevents binding of the host complement components or other lytic components that are present in the serum of the host [131]. The main feature of the VSG tertiary structure is the formation of two long alpha helices per monomer that are perpendicular to the cell surface and define the elongated shape of the VSG [132]. Due to the elongated shape and densely packed composition, only a very limited number of amino acids are accessible to the extra cellular environment which might be hostile. The mature VSG polypeptide has 400–500 amino acid residues (most having between 420 and 460) consisting of two or three domains, namely, N- and C-terminal domains [133]. The majority of the sequence forms a single N-terminal domain of 350–400 residues and the remainder one or two smaller C-terminal domains of 40–80 residues each [133–135]. Other than cysteine residues, there is little conservation of primary sequence within the N-terminal domain whereas the C-terminal domain has a greater degree of primary sequence identity [135]. The C-terminal domain is attached to the membrane of the parasite by a glycosyl-phosphatidyl-inositol (GPI) anchor [136]. Both attachment of the GPI anchor and the N-glycosylation of the VSG occur in the endoplasmic reticulum during the transportation of the protein towards the surface. The attachment of the GPI is essential for further transport of the VSG [137]. An N-terminal part of the VSG, containing about two thirds of the mature polypeptide can be cleaved off by exogenous protease cleavage.
The three dimensional structure of this N-terminal domain of T. brucei VSG has been resolved at high resolution [138]. The dimer was found to be ±100 Å long and having an asymmetrical cross section being 60 Å × 40 Å at the bottom, 30 Å × 20 Å in the middle and 45 Å × 45 Å near the top in which the top of the molecule is normally exposed to the external environment. This top part represents the hyper-variable part of the VSG molecule and is stabilized by two-conserved disulfide bridges [139]. The N-terminal domain of VSG ends with an α-helix which is followed by a single C-terminal domain which is attached to the parasite membrane with the GPI anchor.
5.1.2. The GPI Anchor
Each VSG and related molecules like the ESAG6 is attached to the bloodstream form T. brucei cell surface by a GPI anchor [136, 140, 141]. GPI anchors influence the trafficking of the VSG in the early and late secretory pathway [142], and play an important role in the expression of the VSG on the surface of the parasite [143].
5.1.3. Structure of the GPI Anchor
The first GPI-anchor structure determination, as well as the first description of the mechanism of GPI biosynthesis were both established using trypanosome VSG [136, 144–146]. This GPI-anchor is preassembled as a GPI precursor with the following structure: NH2CH2CH2-PO4H-6Manα1-2Manα1-6Manα1-4GlcNα1-6myoinositol-1-PO4H-3(sn-1,2-di-Myristoylglycerol, [147]. This core is attached to the mature C-terminal amino acid in exchange for a hydrophobic C-terminal GPI-addition signal peptide [140]. The VSG-linked GPI anchors are subsequently substituted with unique galactose side-chain residues, not present in other mammalian GPI anchored proteins [136]. Another unique feature of the trypanosome GPI anchor is that it contains exclusively myristate (a 14-carbon, saturated fatty acid) as lipid moiety [148]. As analogues of myristate are selectively toxic for the parasites, the GPI biosynthesis pathway can be a potential candidate for treatment of trypanosomiasis [149, 150].
Trypanosomes contain an endogenous phospholipase-C (PLC) known as the GPI-PLC that is capable of hydrolyzing the GPI-anchor of the membrane-bound form VSG (mfVSG) [151]. This PLC is located primarily in the membrane of the flagellar pocket and is possibly part of the VSG-membrane recycling system [152]. It is highly expressed in the bloodstream form parasites (30 000 copies per cell) and severely down-regulated in the procyclic form [153, 154]. The GPI-PLC expressed by one parasite only targets its own plasma membrane and as such is unable to release the VSG of another parasite [155]. As a consequence the death of one parasite would not harm others. The GPI-PLC under stress condition cleaves off the VSG leaving the dimyristoyl glycerol (DMG) in the membrane and the glycosil inositol phosphate (GIP) fraction on the released soluble VSG (sVSG) [156–158]. This conversion can be detected immunologically as it results in the exposure of the cryptic cross-reacting determinant (CRD) [158–160] . A low rate of the sVSG release from trypanosomes has been observed in the cultures of the bloodstream forms and clearly demonstrated not to result from the lysis of a subset of the population [161]. In addition, release of the sVSG can be induced under stress conditions that do not lyse the parasites [162]. Both of these observations suggest that GPI-PLC acts on the mfVSG in living trypanosomes and not just on cell lysis. The VSG and the GPI-PLC show the same developmentally regulated expression, being found in the blood-stream form and not in the procyclic form [154]. The ability of the GPI-PLC to catalyze the shedding of the VSG coat in vitro, and the contemporal expression of the two proteins, has led to the models that suggest an important role for the enzyme in the developmental changes that involve alterations in the expression of cell surface proteins [163]. However, by analyzing a GPI-PLC null mutant trypanosome it was shown that the GPI-PLC is not essential and is not necessary for antigenic variation, though it influences parasitemia in mice [164]. Moreover, beside the GPI-PLC activity, there is a zinc metalloproteases (MSP-B) activity which also causes shedding of pre-existed VSG and which even could be playing a quantitatively major role than the GPI-PLC [25, 26].
5.2. Other Surface Proteins
Even though trypanosomes are covered mainly by their stage specific major surface proteins, some other surface proteins are also present which might be located either beneath the surface coat of the VSG/procyclin or within the flagellar pocket. Some of these are invariant surface proteins, others are receptors and transporters [165].
5.2.1. Invariant Surface Glycoproteins (ISGs)
Due to the low abundance of ISGs, it is not very easy to identify minor surface proteins in the trypanosome. However, by use of different techniques some invariant surface glycoproteins (ISGs) have been identified, such as ISG 60, ISG 65 and ISG 75 [130, 166], ISG 64, and ISG 70 [167]. The ISGs 65 and 75 both are predicted to be composed of large extracellular domains, a single transmembrane domain, a small C-terminal intracellular domain and are not accessible for antibodies on live parasites. Using fixed parasites, it was shown that both of them were distributed over the entire cell surface. The ISG65 and not the ISG75 has been shown to elicit an antibody response in the chronically infected mice [130]. Another invariant protein with a single copy gene, ISG 100, has been described. It is present in the flagellar pocket and is associated with the endo/exocytosis compartments. This suggests that it might play a role in the pathway for the endocytosis or it may have a structural role in the compartments involved in intracellular trafficking of T. brucei [168].
5.2.2. Surface Receptors
For the uptake of different molecules from the host, trypanosomes use different receptors. Not many receptors have been characterized in the trypanosomes, so far. One of the best characterized receptors in the trypanosomes is the transferrin receptor. African trypanosomes grow in the bloodstream of different mammals where they take up their nutrients required for growth. Iron is one of the crucial molecules for the trypanosome survival [169]. Iron requirement of the trypanosomes is fulfilled by a receptor-mediated uptake of the host transferrin (Tf) [170–172]. Unlike the mammalian transferrin receptor (Tf-R) which is a homo-dimer transmembrane protein distributed over the surface of the cell and binding two Tf molecules [173], the trypanosomal Tf-R, located in the flagellar pocket membrane, is a hetero-dimer constructed from two very similar VSG-like N-terminal domains [174] and binds only one molecule of Tf [170]. The trypanosomal Tf-R differs in primary structure, subunit organization and mode of anchorage from its human counterpart [175]. Another important receptor might be the lipoprotein scavenger receptor which is involved in uptake of the LDL, HDL as well as the human trypanolytic factor (TLF) [109]. In addition to the scavenger receptor another LDL receptor has been described to be present in the flagellar pocket [176, 177]. There are reports that small molecules are taken up by the trypanosomes via transporters like the glucose transporters [178] to transport glucose, the nucleoside transporters with diverse substrate specificities and distinct patterns of expression during the trypanosome life cycle [179].
6. Immune Responses during Infection with African Trypanosomes
The immune response of vertebrates consists of two arms: the innate immune response which has a low specificity and the adaptive immune response which is antigen specific. The immunology of infections by the African trypanosomes is a complex process and has been recently reviewed [180, 181]. Being an extracellular parasite, the African trypanosome encounters both the innate as well as the adaptive immune response from the host.
6.1. Innate Immune Response against African Trypanosomes
Once in the bloodstream of the mammalian host, the trypanosomes encounter the innate host immune system as the first barrier. As already mentioned, human and some other primates have trypanolytic factors in their serum that aid the primary defense mechanism.
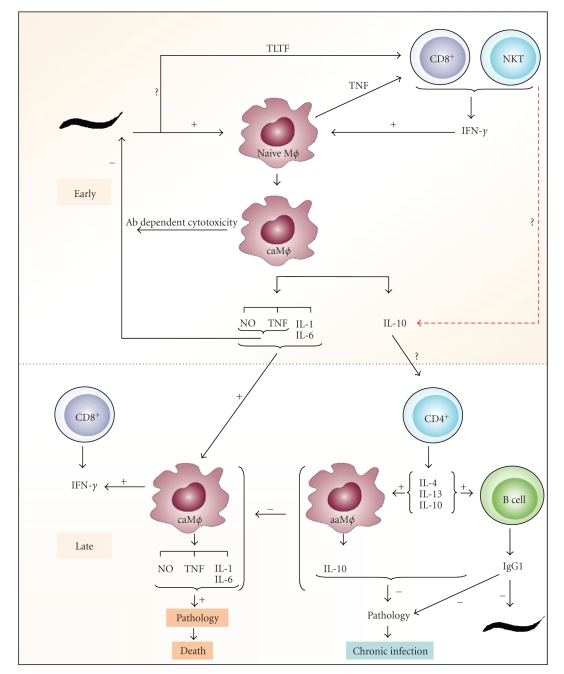
In a cellular innate immune response, different host cells are activated by different trypanosomal factors, initiating an acute inflammatory response [182, 183] (Figure 4). Among many molecules, the trypanosomal DNA that might be released from the dead trypanosomes has been shown to activate macrophages in a process called classical activation, to secrete proinflammatory molecules like TNF, IL-12 and NO [184, 185]. In this regard, the involvement of toll like receptors (TLR) and in particular the TLR9, in parasitemia control [186], would suggest that the DNA from trypanosomes plays a role in disease progression. The GPI anchor of the VSG also interacts with the macrophages (via a putative receptor which is still elusive) and induces secretion of pro-inflammatory cytokines [187–189]. So, the first response of the host immune system consists of classically activated macrophages (caMϕ) secreting pro-inflammatory molecules such as TNF, IL-1, IL-6, NO (219–221). The caMϕs can phagocytose antibody-opsonised parasites [190] as well as secrete trypanotoxic molecules such as TNF and NO [191–194] that are involved in the control of the first peak of parasitemia.
Figure 4.
Trypanosome-host interaction (from [208]). Model for induction classical (caMϕ) and alternative activation (caMϕ) of macrophages during trypanosome infection; TLTF: trypanosome-derived lymphocyte-triggering factor.
6.2. Adaptive Immune Response
The initial inflammatory response is beneficial to the host at the early stage of the infection, but a sustained inflammation can cause pathology. Hence, it is essential for the host to reduce the inflammation which is obtained by down regulating the caMϕ and their pro- inflammatory cytokines. Production of type II cytokines such as IL-4, IL-10 and IL-13 which can modulate the macrophages to become more anti-inflammatory type alternatively activated macrophages (aaMϕ) are involved in a longer survival of the host (Figure 4). So a type I inflammatory response at the beginning of the infection and a shift to the type II immune response in the late stage of the infection are correlated with the capacity of the host to control the parasite and the pathology respectively. In a murine model, it has been shown that the VSG-specific cytokine responses associated with the resistance to the murine African trypanosomosis are infection-stage dependent, with the type-I cytokine responses being critical during the early stage of infection while the type-II cytokine responses to be more important during the late and chronic phases of the disease [195]. Several studies suggest that the cytokine responses influence the outcome of African trypanosomiasis [196–199]. However, the precise role of the individual cytokines is still equivocal and may be dependent on the parasite strain, the mouse model or both. In this context the role of IFN-γ [196] for resistance against T. b. rhodesiense and the role of IFN-γ and NO together with the antibody response have been shown to be crucial in the control of T. congolense infection [200]. However, in the T. evansi model even though TNF, IFN and NO levels are elevated in the early stage of infection, none of these molecules seem to be important for the parasitemia control as well as the survival of the host [201]. In another trypanosome model, T. borelli, a blood parasite of carp, NO hinders antibody clearance from the surface of the parasite and increases susceptibility to the complement lysis [202]. Moreover, Magez et al. [203, 204] demonstrated that TNF plays a key role in both parasitemia controls as well in the development of pathology in T. brucei infections. Concerning the role of type II cytokines, some have shown that CD4+ T cell regulated IL-4 production was crucial for controlling T. b. gambiense infections in mice [198] and a role for IL-4 in resistance to bovine trypanosomiasis was also proposed [199] while others [196] reported that IL-4 knockout mice do not show any alteration in the parasite control. Namangala et al. [205] showed that during the chronic stage of infection a Th2 cytokines production as well as a IgG1 antibody response to the trypanosome antigens are linked to the longer survival of the host in T. brucei infection model. Moreover, the levels of IL-10 and IL-6 in the brain have been shown to be associated with the protection from neuroinflammatory pathology of HAT [206, 207].
6.2.1. Humoral Responses
During the trypanosome infection a dominant humoral response of the host is expected, since the location of the parasite is extra-cellular. Both the murine and bovine trypanosomiasis is characterized by a polyclonal B cell activation as evidenced by an increased number of B cells and a significant elevation in plasma Igs [209–211]. Because of the polyclonal B cell activation, a significant component of the resultant antibody is either polyspecific or auto reactive [212–214]. Although the VSG molecules are highly immunogenic for all mouse strains upon immunization, dramatic differences in the ability of animals to mount the VSG-specific B cell response occur after infection [215]. It is shown in different independent studies that specific antibodies directed against the trypanosome VSG mediate the destruction and clearance of parasites in successive parasitemic waves and hence contribute to antibody-mediated trypanotolerance [197, 213, 216, 217]. Animals immunized with the irradiated trypanosomes or the VSG are successfully protected against a challenge with the homologous parasites [218, 219]. The antibodies directed against the specific surface-exposed epitopes of the VSG coat opsonize the parasites and the immune complexes are efficiently phagocytosed and destroyed, mainly in the liver, by the macrophages (Kupffer cells) [190, 220, 221].
During African trypanosomiasis, the VSG-specific B cell responses can occur in a T-cell independent manner [222]. However, the T-cells improve the B-cell responses, mainly by secreting cytokines mediating antibody class switching. In this context an increased IL-4 mRNA level and a concomitant increase in the IgG1 antibodies against the VSG was observed in the trypanotolerant N'dama cattle infected with T. congolense but not in the trypanosusceptible Boran cattle [199]. In animal trypanosomiasis, trypanotolerance is a combination of the humoral response needed for parasite control as well as the ability to control the immunopathology (described below) which is the cause for loss of productivity. Schofield et al. [223] described a rapid major histocompatibility complex (MHC)-unrestricted antibody response to the diverse pathogens including the trypanosomes. These authors demonstrated the CD1d-restricted IgG formation in response to Plasmodium and Trypanosoma GPI anchored antigens mediated by IL-4 producing CD4+, NK1.1+ helper T-cells (NKT cells) and proposed that this may represent a general mechanism for a rapid response to the GPI-anchored surface antigens and the parasite control.
Although the trypanosome-specific antibodies are produced in the early stage of infection and may be protective as they mediate parasite clearance [224–228], remove immune complexes [229], and possibly neutralize the parasite products, yet a significant proportion of the antibodies is either polyspecific or auto-reactive [209, 213, 214, 230, 231]. Moreover, later in the infection, the B-cells become suppressed or exhausted, resulting in a total absence of IgG responses and a strongly reduced IgM response [232]. Using B-cell (μMT) and IgM-deficient mice, it has been shown that in the murine experimental T. brucei trypanosomiasis, B-cells were crucial for periodic peak parasitemia clearance, whereas the IgM antibody played a limited role [233]. However, in the T. evansi infection model, the IgM has been shown to play an important role in the control of the disease [201] suggesting the role of different antibodies can vary with different trypanosome strains.
6.3. Immunosuppression
One of the striking features of the trypanosome infections is the dramatic suppression of the immune responses, which might result in a high susceptibility to opportunistic infections. The generalized immune suppression has been reported to affect a large variety of both the humoral (B cell) and the cellular (T-cell and macrophage) immune functions [234], consequently leading to occurrence of the trypanosome-induced immunopathology [235–237]. Although the existence of the immunosuppression has been known for long time, the unresolved question was whether the immunosuppression was mediated by the macrophages or the T cells. There are suggestions that both cells might be involved [238].
Suppressive macrophages elicited by the T. brucei infection play a central role in the immunosuppression observed in this infection [239–241]. The immunosuppression is characterized by an inhibition of the T cell proliferation due to down regulation of both IL-2 production and expression of IL-2 receptor [235, 240]. Prostaglandins and nitric oxide (NO) impair mitogen-induced T-cell proliferation in the spleen, peritoneal cavity, and lymph nodes of T. brucei infected mice but only during the early stage of infection [241–243]. At the early stage of the infection, the involvement of TNF and IFN-γ in the inhibition of T-cell proliferation seems to be involved in an up-regulation of prostaglandins and NO synthesis [204, 244]. In addition, TNF promotes the development of suppressive cells by inducing IFN-γ production in the lymph nodes of T. brucei infected mice [244]. Moreover, T. brucei infection also impairs the MHC class II antigen presenting capacity of the classically activated macrophages [245] resulting in a reduced T-cell activation. But at the late stage of infection, inhibition of T-cell proliferation in the lymph nodes occurs through NO/prostaglandin independent pathway, whereby IFN-γ released by CD8+ T-cell plays a crucial role [242, 246]. There are reports showing at the late stage of infection, macrophages displaying an anti-inflammatory cytokine production, which might modulate in several aspect of the immune system as the infection progresses [247, 248]. Factors like IL-10 secreted by the macrophages of the infected animals are shown to inhibit antigen presentation [249] and contributing to the impairment of T-cell activation. However, the mechanisms of suppression by the alternatively activated macrophages elicited at later stage of the African trypanosome infections are not fully understood. Regulatory T cells (Tregs) have also been shown to limit the production of IFN-γ by CD4+ and CD8+ T cells and also down regulate the activation of macrophages [250, 251]. Furthermore, these Tregs are suggested to suppress the NKT cell [238]. Presentation of glycolipids to the NKT cells in the context of CD1d have been suggested in the trypanosome infection [238] or a GPI treatment [252].
6.4. Immunopathology
As mentioned above, uncontrolled type I immune reaction of the host leads to a pathological condition. The major pathological complication associated with the human trypanosomiasis is the neurological disorder which is finally manifested as ‘sleeping sickness'. However the pathological symptoms observed in experimental trypanosomiasis are mainly loss of body weight, fever, reduced locomotory activity, splenomegaly, and liver damages. One of the common pathological features observed in human, bovine as well as the experimental murine trypanosomiasis is the loss of red blood cell count, that is, anemia. Here, the degree of anemia might be considered as an indicator of the disease severity [253]. At least in case of the bovine trypanosomiasis, one aspect of the trypanotolerance is the measurement of the ability to control the infection-associated anemia and subsequently the loss of productivity of the host [254]. Anemia during trypanosomiasis might be due to either loss of RBC, for example, the cytokine-activated macrophages (M1 cells) are suggested to be responsible for the enhanced phagocytosis of parasites as well as the RBCs [255] or due to inability to mount a vigorous compensatory erythropoietic response [255, 256].
During trypanosome infections, TNF is involved both in parasitemia control and infection associated pathology [204] such as anemia, neurological disorders, fever and cachexia during both human and animal trypanosomiasis [234, 257]. In view of this dual role, lots of works reveal how trypanosomal components induce TNF. In this regard, VSG was identified as major TNF inducing component in trypanosome-soluble extract. Both sVSG and mfVSG were shown to manifest similar TNF inducing capacities but by a detailed analysis it was indicated that they are working in a different way. The GIP moiety of the VSG via the GIP associated galactose side chain is responsible for direct induction of TNF. Yet, the mfVSG, but not the sVSG, stimulates macrophages toward IL-1 secretion and acquisition of the LPS-responsiveness and is, as such, involved in indirect TNF production. Thus, the VSG has two distinct macrophage activating components [258]. TNF can signal for cellular activities through 2 different receptors; TNF-R1 (CD120a) and TNF-R2 (CD120b). It is suggested, that TNF-R2 signaling in trypanosomiasis mediates infection-associated pathology, whereas TNF-R1 signaling has no impact on infection [259].
It was also shown that serum TNF levels correlate with the severity of neuropathological symptoms in the human sleeping sickness [260]; however, some studies found no correlation between the TNF serum level and pathology of the HAT [261, 262]. In the same line of research, there are reports that demonstrate the enhanced expression of TNF mRNA in the brain of T. brucei-infected mice [263, 264] and the correlation in trypanosome-infected cattle between TNF production by monocytes and the severity of diseases-associated anemia [265]. Hence, the accumulated knowledge about trypanosome-elicited production of TNF indicates that while this cytokine might be beneficial during the early stage of infection through its role in parasite clearance, the overall pathology-inducing aspect overrules in a negative way. As outlined before, due to highly sophisticated antigenic variation, an antiparasitic vaccination seems to be very difficult. However, the knowledge of the immunopathology gives a basis for a design of an antidisease vaccination.
7. Current and Alternative Control Strategies for Trypanosomiasis
The control of trypanosomiasis can be approached from three different aspects: control the vector, vaccinate the host as a preventive measure, or treat the infected host. All three approaches have their own benefits but at the same time suffer from some difficulties.
There are different vector-control methods currently available; use of insecticide (through the sequential aerosol spraying technique, insecticide-treated targets or insecticide treated animals), use of traps, and the sterile insect technique (reviewed [266]). Technical improvements to make these methods cheaper, more efficient and less time consuming are still needed [267]. In an attempt to develop a cost effective control method for Glosina fiscipes, a perspect for developing the odour baits based on the kairomones present in lizard odour and pig odour have been suggested [268]. In all these and other studies it has been agreed that more precise and possibly new method of vector control are still in demand. One of the problems associated with the use of chemical insecticides as a vector control is the evolution of resistance against that chemical. As an alternative approach, one may think about an “evolution-proof” insecticide as has been discussed for the malaria control [269].
The current treatment of HAT is based on four main drugs, namely suramin, pentamidine, melarsoprol, and eflornithine (difluoromethylornithine or DFMO), with nifurtimox undergoing evaluation [270, 271]. Most of these drugs were developed in the first half of the twentieth century and there has been no new registered drug since 1981. The candidate drug called DB 289, a dimidine derivative has finished clinical trial II [270–272]. Early-stage disease is treated with an intravenous injection of suramin in rhodesiense disease and with an intramuscular pentamidine in gambiense disease. The arsenical melarsoprol is the only effective drug for the late-stage disease in both forms of the HAT, as the drug crosses the blood brain barrier [272]. Nifurtimox taken orally for 1 to 2 months and DFMO with an administration scheme spread over five weeks including 14 days of intravenous injection can be other alternatives. However, the DFMO is not commonly available and is considered too expensive for routine use. Moreover, the DMFO is known to be active only against T. b. gambiense [273].
The major problems of therapy are the frequent and often serious adverse events due to drug toxicity, relapses, and the long duration of treatment. The worst adverse events are encephalopathic syndromes, which occur in 5–10% of patients, and result in the death in 10–50% of those in whom the encephalopathy develops. Other severe adverse reactions reported are polyneuropathies (up to 10%), exfoliative dermatitis, fever, headache, diarrhea, maculopapular eruptions, pruritus, and abdominal and chest pain [274, 275]. There are reports for a shorter treatment regime of melarsoprol [276, 277] or a combination of melarsoprol with suramin [278]. Drug resistance in trypanosomes appears to be increasing in the field and is now hindering efforts to control the HAT. Failures of the melarsoprol treatment, the preferred treatment during the encephalitic stage of T. b. rhodesiense infections, have reached alarming levels. The unacceptable toxicity of the currently available drugs for HAT underpins the urgency of developing more effective and safer drugs [270].
Control of African bovine trypanosomiasis mainly relies in endemic areas on chemotherapy and chemoprophylaxis using three trypanocidal compounds; isometamidium, homidium and diminazine. Suramin and quinapyramine are also in practice since long time in animal trypanosomiasis treatment. All of these drugs have been in widespread use for about 40 years and resistance has been reported in many parts [279–282]. Resistance to the Berenil (a drug commonly used for treatment of livestock trypanosomiasis [283]) has also been reported in several foci [284].
The resistance to drugs for the African trypanosomes has been shown to be associated with reduced drug uptake by the parasites stressing the importance of drug transporter (reviewed by [285]). Reduction of net drug uptake can be caused by either a decreased drug import or an increased drug export. Either mechanism implies mutations in the transporters since most trypanocides do not freely diffuse through the plasma membrane. In this context, the adenine nucleoside transporter (P2-transporter) has been found to be the transporter for arsenical as well as pentamidine [286, 287]. That provides an explanation for the frequent occurrence of cross-resistance between arsenicals and dimidines in T. brucei spp. [288]. A trypanosome gene (TbAT1) product exhibits P2-like transport activity but this is not the only transporter for the trypanocidal drugs, indicating that drug resistance involves the loss of more than one transporter [289]. Besides loss of import to the parasite, increased export of the drug also can be responsible for the drug resistance. In this aspect, over expression of one exporter, TbMRPA (T. brucei multidrug-resistance associated protein) caused 10-fold resistance to the melarsoprol in vitro [290, 291] but such over expression in patients with treatment failure with melarsoprol has not been reported yet. Similarly, over expression of a gene TeDR40 has been shown to correlate with Berenil resistance observed in T. evansi [292]. Besides, alterations in drug import and export, there can be other alternative mechanisms like failure to undergo apoptosis may also contribute to the drug resistance. Different resistance mechanisms are not mutually exclusive. On the contrary, drug resistance is often multifactorial.
Due to the facts that (i) there are very few drugs available for the treatment of trypanosomiasis, (ii) the drugs being used cause severe toxic effects, and (iii) there are increasing field cases of drug resistance and there is a clear demand for alternative treatment schemes. Furthermore, due to antigenic variation and immunosuppression, the conventional vaccinations strategies are not able to give promising results. So, also in this regard alternative approaches are necessary.
Even a century after its first description, the trypanosomiasis is still one of the major parasitic disease for which control is far from reality. Most antitrypanosome drugs are taken up by the parasite via specific transporters (e.g., P2 transporter). As a result, mutation on the gene encoding that transporter can lead to multiple drug resistance [286, 287]. Therefore when new drug strategies for trypanosomiasis are designed, they should rely on novel molecular and biochemical pathways. The human trypanolytic factor, APOL1, has been exploited as an alternative approach for the treatment of HAT. APOL1 lyses trypanosomes except the ones which are causing HAT [102]. In case of T. rhodesiense the resistance to the trypanolytic capacity of APOL-1 is due to the expression of serum resistance-associated (SRA) protein [94] which neutralizes APOL1 through the interaction with the C-terminal of this lipoprotein. Deletion of the SRA interacting domain of APOL1 results in the generation of a new molecule, that is, Tr-APOL1 that is lytic to both NHS-sensitive as well as resistant T. rhodesiense. Hence, Tr-APOL1 represents a possible drug against all T. rhodesiense parasites. However, to avoid the possible competition with the native APOL1 in the serum, specific trypanosomal targeting of Tr-APOL1 is essential. One such approach where the Tr-APOL1 has been conjugated with a single domain camel VHH that targets the oligomannose moiety of VSG has been reported [293]. The results in that study showed that treatment with this conjugated Tr-APOL1 cured mice infected with either NHS-resistant T. rhodesiense or NHS-sensitive T. brucei. The treatment also had beneficial effect when used in the chronic stage of the trypanosomiasis, although in this case a complete elimination of the parasite was not obtained [103]. The results thus may suggest that, Tr-APOL1 could be developed as an alternative drug for treatment of the early stage HAT. Due to the fact that APOL1 is a human self-antigen, and since VHH is highly homologous to human VH, the authors of this paper suggest very minimal, if any, immune response against the conjugated protein, when administered to HAT patients. Furthermore, it has been shown that the full length baboon APOL1 (which does not interact with SRA) is protective against both the animal-infective and human-infective T. rhodesiense in an experimental mouse model, and, as such, it has been suggested to create transgenic livestock that would be resistant to animal and human-infective trypanosomes, which is envisaged to result in the reduction of the livestock trypanosomiasis and zoonotic transmission of human infective trypanosomes [119].
Beside treatment, effective vaccination strategy is a second approach for the control of any infectious diseases. In the case of trypanosomiasis, all conventional anti-parasitic vaccination efforts undertaken so far, that used dominant surface protein, have failed due to the antigenic variation of the trypanosomes surface coat. Therefore, an alternative strategy of the vaccination is demanding. As alternative vaccination approaches, different molecules such as trypanosomal cystein protease (congopain) [294], trypanosomal tubulin [295, 296] or trypanosomal GPI have been attempted. The GPI-anchor of the VSG as one of the major parasitic components causing the inflammatory response associated to the infection has been identified [258]. In one of the studies, this information has been used to evaluate GPI-based vaccination as an alternative strategy with antidisease potential [252]. Using liposomes as slow delivery system, the GPI administered prior to the infection had been shown to result in a better control of the parasitemia and a longer lifespan of the infected mice. The treated animals were better protected from various pathological conditions including anemia which is considered as one of the major pathological parameters of the trypanosomiasis [254]. These results are related to the fact that the treatment oriented the classically activated inflammatory macrophages, to more counter-inflammatory alternatively activated macrophages [208], subsequently resulting in reduced TNF production and reduced pathology. With the GPI-based treatment, though there were positive effects on the infection-induced pathology as well as survival, but the animals were not cleared from their parasites. Therefore, this strategy is more an anti-disease approach rather than an anti-parasitic strategy. While this solution would not be advisable for the HAT where an antiparasitic treatment would be needed, the persistence of a low level infection that would prevent the severe disease might be preferred in the livestock field conditions, where the host has controlled geographical movement and is under continuous threat of re-infection by the infected tsetse flies. The needs, possibilities and requirements of further knowledge for the way to develop an anti-disease control strategy for trypanosomiasis has been recently highlighted [297].
8. Conclusion
Both the prophylactic as well as the therapeutic aspects of trypanosomiasis need alternative approaches as currently there is no vaccine and the drugs that are in use have several short comings. Some such alternative approaches have been initiated but still more detail and comprehensive strategies should be envisaged. These approaches should be based on the strong background of understanding the biology of the parasite as well as the host-pathogen interaction needs to be further studied. One final point of consideration for such alternative approach would be its economical viability since the disease affects the poorest of the poor of the world.
Acknowledgment
The author thanks Dr. Stefan Magez and Dr. Patrick De Baetselier for their help at the initial stage of the manuscript and Tom Devecseri for the figures.
References
- 1.Buguet A, Bisser S, Josenando T, Chapotot F, Cespuglio R. Sleep structure: a new diagnostic tool for stage determination in sleeping sickness. Acta Tropica. 2005;93(1):107–117. doi: 10.1016/j.actatropica.2004.10.001. [DOI] [PubMed] [Google Scholar]
- 2.Cattand P, Jannin J, Lucas P. Sleeping sickness surveillance: an essential step towards elimination. Tropical Medicine and International Health. 2001;6(5):348–361. doi: 10.1046/j.1365-3156.2001.00669.x. [DOI] [PubMed] [Google Scholar]
- 3.WHO. Control and Surveillance of African Trypanosomiasis, Report of WHO Expert Comittee. Geneva, Switzerland: World Health Organization; 1998. (WHO Technical Report Series, no. 881). [PubMed] [Google Scholar]
- 4.Kuzoe FAS. Current situation of African trypanosomiasis. Acta Tropica. 1993;54(3-4):153–162. doi: 10.1016/0001-706x(93)90089-t. [DOI] [PubMed] [Google Scholar]
- 5.Jordan AM. Tsetse flies as vectors of trypanosomes. Veterinary Parasitology. 1976;2(1):143–152. [Google Scholar]
- 6.Ford J. The influence of tsetse flies on the distribution of African cattle. In: Proceedings of the 1st Federal Scientific Congress; 1960; Salisbury, Md, USA. pp. 357–365. [Google Scholar]
- 7.McKelvey JJ. Man against Tsetse: Struggle for Africa. London, UK: Cornell University Press; 1973. [Google Scholar]
- 8.Wuyts N, Chokesajjawatee N, Panyim S. A simplified and highly sensitive detection of Trypanosoma evansi by DNA amplification. The Southeast Asian Journal of Tropical Medicine and Public Health. 1994;25(2):266–271. [PubMed] [Google Scholar]
- 9.Joshi PP, Shegokar VR, Powar RM, et al. Human trypanosomiasis caused by Trypanosoma evansi in India: the first case report. American Journal of Tropical Medicine and Hygiene. 2005;73(3):491–495. [PubMed] [Google Scholar]
- 10.WHO. A new form of human trypanosomiasis in India. Description of the first human case in the world caused by Trypanosoma evansi. Weekly Epidemiological Record. 2005;80(7):62–63. [PubMed] [Google Scholar]
- 11.Hoare CA. The classification of mammalian trypanosomes. Ergebnisse der Mikrobiologie, Immunitatsforschung und Experimentellen Therapie. 1966;39:43–57. doi: 10.1007/978-3-662-38353-7_3. [DOI] [PubMed] [Google Scholar]
- 12.Wéry M. Protozoologie Médical. Bruxelles, Belgium: De Boek; 1995. Protozoaires Flagellés (Trypanosomatida): parasite du sang et des tissues. Généralités; pp. 83–89. [Google Scholar]
- 13.Matthews KR. Developments in the differentiation of Trypanosoma brucei. Parasitology Today. 1999;15(2):76–80. doi: 10.1016/s0169-4758(98)01381-7. [DOI] [PubMed] [Google Scholar]
- 14.David Barry J, McCulloch R. Antigenic variation in trypanosomes: enhanced phenotypic variation in a eukaryotic parasite. Advances in Parasitology. 2001;49:2–70. doi: 10.1016/s0065-308x(01)49037-3. [DOI] [PubMed] [Google Scholar]
- 15.Vickerman K. Developmental cycles and biology of pathogenic trypanosomes. British Medical Bulletin. 1985;41(2):105–114. doi: 10.1093/oxfordjournals.bmb.a072036. [DOI] [PubMed] [Google Scholar]
- 16.Vickerman K. Polymorphism and mitochondrial activity in sleeping sickness trypanosomes. Nature. 1965;208(5012):762–766. doi: 10.1038/208762a0. [DOI] [PubMed] [Google Scholar]
- 17.Matthews KR, Ellis JR, Paterou A. Molecular regulation of the life cycle of African trypanosomes. Trends in Parasitology. 2004;20(1):40–47. doi: 10.1016/j.pt.2003.10.016. [DOI] [PubMed] [Google Scholar]
- 18.Vassella E, Reuner B, Yutzy B, Boshart M. Differentiation of African trypanosomes is controlled by a density sensing mechanism which signals cell cycle arrest via the cAMP pathway. Journal of Cell Science. 1997;110(21):2661–2671. doi: 10.1242/jcs.110.21.2661. [DOI] [PubMed] [Google Scholar]
- 19.Tyler KM, Matthews KR, Gull K. The bloodstream differentiation-division of Trypanosoma brucei studied using mitochondrial markers. Proceedings of the Royal Society B. 1997;264(1387):1481–1490. doi: 10.1098/rspb.1997.0205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.McLintock LML, Turner CMR, Vickerman K. Comparison of the effects of immune killing mechanisms on Trypanosoma brucei parasites of slender and stumpy morphology. Parasite Immunology. 1993;15(8):475–480. doi: 10.1111/j.1365-3024.1993.tb00633.x. [DOI] [PubMed] [Google Scholar]
- 21.Redpath MB, Windle H, Nolan D, Pays E, Voorheis HP, Carrington M. ESAG11, a new VSG expression site-associated gene from Trypanosoma brucei. Molecular and Biochemical Parasitology. 2000;111(1):223–228. doi: 10.1016/s0166-6851(00)00305-4. [DOI] [PubMed] [Google Scholar]
- 22.Sbicego S, Vassella E, Kurath U, Blum B, Roditi I. The use of transgenic Trypanosoma brucei to identify compounds inducing the differentiation of bloodstream forms to procyclic forms. Molecular and Biochemical Parasitology. 1999;104(2):311–322. doi: 10.1016/s0166-6851(99)00157-7. [DOI] [PubMed] [Google Scholar]
- 23.Ziegelbauer K, Stahl B, Karas M, Stierhof Y-D, Overath P. Proteolytic release of cell surface proteins during differentiation of Trypanosoma brucei. Biochemistry. 1993;32(14):3737–3742. doi: 10.1021/bi00065a028. [DOI] [PubMed] [Google Scholar]
- 24.Ziegelbauer K, Overath P. Surface antigen change during differentiation of Trypanosoma brucei. Biochemical Society Transactions. 1990;18(5):731–733. doi: 10.1042/bst0180731. [DOI] [PubMed] [Google Scholar]
- 25.Gruszynski AE, DeMaster A, Hooper NM, Bangs JD. Surface coat remodeling during differentiation of Trypanosoma brucei. Journal of Biological Chemistry. 2003;278(27):24665–24672. doi: 10.1074/jbc.M301497200. [DOI] [PubMed] [Google Scholar]
- 26.Gruszynski AE, van Deursen FJ, Albareda MC, et al. Regulation of surface coat exchange by differentiating African trypanosomes. Molecular and Biochemical Parasitology. 2006;147(2):211–223. doi: 10.1016/j.molbiopara.2006.02.013. [DOI] [PubMed] [Google Scholar]
- 27.Matthews KR. The developmental cell biology of Trypanosoma brucei. Journal of Cell Science. 2005;118(2):283–290. doi: 10.1242/jcs.01649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Sternberg J, Turner CMR, Wells JM, Ranford-Cartwright LC, Le Page RWF, Tait A. Gene exchange in African trypanosomes: frequency and allelic segregation. Molecular and Biochemical Parasitology. 1989;34(3):269–279. doi: 10.1016/0166-6851(89)90056-x. [DOI] [PubMed] [Google Scholar]
- 29.Sternberg J, Tait A, Haley S, et al. Gene exchange in African trypanosomes: characterisation of a new hybrid genotype. Molecular and Biochemical Parasitology. 1988;27(2-3):191–200. doi: 10.1016/0166-6851(88)90038-2. [DOI] [PubMed] [Google Scholar]
- 30.Sternberg J, Tait A. Genetic exchange in African trypanosomes. Trends in Genetics. 1990;6(10):317–322. doi: 10.1016/0168-9525(90)90252-2. [DOI] [PubMed] [Google Scholar]
- 31.Turner CMR, Aslam N, Smith E, Buchanan N, Tait A. The effects of genetic exchange on variable antigen expression in Trypanosoma brucei. Parasitology. 1991;103(3):379–386. doi: 10.1017/s0031182000059898. [DOI] [PubMed] [Google Scholar]
- 32.Turner CMR, Sternberg J, Buchanan N, Smith E, Hide G, Tait A. Evidence that the mechanism of gene exchange in Trypanosoma brucei involves meiosis and syngamy. Parasitology. 1990;101(3):377–386. doi: 10.1017/s0031182000060571. [DOI] [PubMed] [Google Scholar]
- 33.Jenni L, Marti S, Schweizer J. Hybrid formation between African trypanosomes during cyclical transmission. Nature. 1986;322(6075):173–175. doi: 10.1038/322173a0. [DOI] [PubMed] [Google Scholar]
- 34.Schweizer J, Jenni L. Hybrid formation in the life cycle of Trypanosoma (T) brucei: detection of hybrid trypanosomes in a midgut-derived isolate. Acta Tropica. 1991;48(4):319–321. doi: 10.1016/0001-706x(91)90020-k. [DOI] [PubMed] [Google Scholar]
- 35.Bingle LEH, Eastlake JL, Bailey M, Gibson WC. A novel GFP approach for the analysis of genetic exchange in trypanosomes allowing the in situ detection of mating events. Microbiology. 2001;147(12):3231–3240. doi: 10.1099/00221287-147-12-3231. [DOI] [PubMed] [Google Scholar]
- 36.Van Den Abbeele J, Claes Y, Van Bockstaele D, Le Ray D, Coosemans M. Trypanosoma brucei spp. development in the tsetse fly: characterization of the post-mesocyclic stages in the foregut and proboscis. Parasitology. 1999;118(5):469–478. doi: 10.1017/s0031182099004217. [DOI] [PubMed] [Google Scholar]
- 37.Gibson W. Sex and evolution in trypanosomes. International Journal for Parasitology. 2001;31(5-6):643–647. doi: 10.1016/s0020-7519(01)00138-2. [DOI] [PubMed] [Google Scholar]
- 38.Gibson W, Stevens J. Genetic exchange in the trypanosomatidae. Advances in Parasitology. 1999;43:1–46. doi: 10.1016/s0065-308x(08)60240-7. [DOI] [PubMed] [Google Scholar]
- 39.Brun R, Hecker H, Lun Z-R. Trypanosoma evansi and T. equiperdum: distribution, biology, treatment and phylogenetic relationship (a review) Veterinary Parasitology. 1998;79(2):95–107. doi: 10.1016/s0304-4017(98)00146-0. [DOI] [PubMed] [Google Scholar]
- 40.Borst P, Fase-Fowler F, Gibson WC. Kinetoplast DNA of Trypanosoma evansi. Molecular and Biochemical Parasitology. 1987;23(1):31–38. doi: 10.1016/0166-6851(87)90184-8. [DOI] [PubMed] [Google Scholar]
- 41.Clayton C, Hausler T, Blattner J. Protein trafficking in kinetoplastid protozoa. Microbiological Reviews. 1995;59(3):325–344. doi: 10.1128/mr.59.3.325-344.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Bastin P, Pullen TJ, Moreira-Leite FF, Gull K. Inside and outside of the trypanosome flagellum: a multifunctional organelle. Microbes and Infection. 2000;2(15):1865–1874. doi: 10.1016/s1286-4579(00)01344-7. [DOI] [PubMed] [Google Scholar]
- 43.Hajduk SL, Siqueira AM, Vickerman K. Kinetoplast DNA of Bodo caudatus: a noncatenated structure. Molecular and Cellular Biology. 1986;6(12):4372–4378. doi: 10.1128/mcb.6.12.4372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Simpson L. The mitochondrial genome of kinetoplastid protozoa: genomic organization, transcription, replication, and evolution. Annual Review of Microbiology. 1987;41:363–382. doi: 10.1146/annurev.mi.41.100187.002051. [DOI] [PubMed] [Google Scholar]
- 45.Simpson L, Neckelmann N, de la Cruz VF, et al. Comparison of the maxicircle (mitochondrial) genomes of Leishmania tarentolae and Trypanosoma brucei at the level of nucleotide sequence. Journal of Biological Chemistry. 1987;262(13):6182–6196. [PubMed] [Google Scholar]
- 46.Motyka SA, et al. Overexpression of a cytochrome B5 reductase-like protein causes kinetoplast DNA loss in Trypanosoma brucei. The Journal of Biological Chemistry. 2006;281:18499–18506. doi: 10.1074/jbc.M602880200. [DOI] [PubMed] [Google Scholar]
- 47.Liu B, Liu Y, Motyka SA, Agbo EEC, Englund PT. Fellowship of the rings: the replication of kinetoplast DNA. Trends in Parasitology. 2005;21(8):363–369. doi: 10.1016/j.pt.2005.06.008. [DOI] [PubMed] [Google Scholar]
- 48.Sturm NR, Simpson L. Kinetoplast DNA minicircles encode guide RNAs for editing of cytochrome oxidase subunit III mRNA. Cell. 1990;61(5):879–884. doi: 10.1016/0092-8674(90)90198-n. [DOI] [PubMed] [Google Scholar]
- 49.Mottram JC, Bell SD, Nelson RG, Barry JD. tRNAs of Trypanosoma brucei: unusual gene organization and mitochondrial importation. Journal of Biological Chemistry. 1991;266(27):18313–18317. [PubMed] [Google Scholar]
- 50.de Vries BF, Mulder E, Brakenhoff JPJ, Sloof P, Benne R. The variable region of the Trypanosoma brucei kinetoplast maxicircle: sequence and transcript analysis of a repetitive and a non-repetitive fragment. Molecular and Biochemical Parasitology. 1988;27(1):71–82. doi: 10.1016/0166-6851(88)90026-6. [DOI] [PubMed] [Google Scholar]
- 51.Hancock K, Jahduk SL. The mitochondrial tRNAs of Trypanosoma brucei are nuclear encoded. Journal of Biological Chemistry. 1990;265(31):19208–19215. [PubMed] [Google Scholar]
- 52.Bastin P, Pullen TJ, Sherwin T, Gull K. Protein transport and flagellum assembly dynamics revealed by analysis of the paralysed trypanosome mutant snl-1. Journal of Cell Science. 1999;112(21):3769–3777. doi: 10.1242/jcs.112.21.3769. [DOI] [PubMed] [Google Scholar]
- 53.Broadhead R, Dawe HR, Farr H, et al. Flagellar motility is required for the viability of the bloodstream trypanosome. Nature. 2006;440(7081):224–227. doi: 10.1038/nature04541. [DOI] [PubMed] [Google Scholar]
- 54.Kohl L, Robinson D, Bastin P. Novel roles for the flagellum in cell morphogenesis and cytokinesis of trypanosomes. EMBO Journal. 2003;22(20):5336–5346. doi: 10.1093/emboj/cdg518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Kohl L, Bastin P. The flagellum of trypanosomes. International Review of Cytology. 2005;244:227–285. doi: 10.1016/S0074-7696(05)44006-1. [DOI] [PubMed] [Google Scholar]
- 56.Overath P, Engstler M. Endocytosis, membrane recycling and sorting of GPI-anchored proteins: Trypanosoma brucei as a model system. Molecular Microbiology. 2004;53(3):735–744. doi: 10.1111/j.1365-2958.2004.04224.x. [DOI] [PubMed] [Google Scholar]
- 57.Webster P, Russell DG. The flagellar pocket of trypanosomatids. Parasitology Today. 1993;9(6):201–206. doi: 10.1016/0169-4758(93)90008-4. [DOI] [PubMed] [Google Scholar]
- 58.Field MC, Carrington M. The trypanosome flagellar pocket. Nature Reviews Microbiology. 2009;7(11):775–786. doi: 10.1038/nrmicro2221. [DOI] [PubMed] [Google Scholar]
- 59.Stuart KD, Schnaufer A, Ernst NL, Panigrahi AK. Complex management: RNA editing in trypanosomes. Trends in Biochemical Sciences. 2005;30(2):97–105. doi: 10.1016/j.tibs.2004.12.006. [DOI] [PubMed] [Google Scholar]
- 60.Panigrahi AK, Ernst NL, Domingo GJ, Fleck M, Salavati R, Stuart KD. Compositionally and functionally distinct editosomes in Trypanosoma brucei. RNA. 2006;12(6):1038–1049. doi: 10.1261/rna.45506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Golden DE, Hajduk SL. The importance of RNA structure in RNA editing and a potential proofreading mechanism for correct guide RNA:pre-mRNA binary complex formation. Journal of Molecular Biology. 2006;359(3):585–596. doi: 10.1016/j.jmb.2006.03.041. [DOI] [PubMed] [Google Scholar]
- 62.Michelotti EF, Harris ME, Adler B, Torri AF, Hajduk SL. Trypanosoma brucei mitochondrial ribosomal RNA synthesis, processing and developmentally regulated expression. Molecular and Biochemical Parasitology. 1992;54(1):31–41. doi: 10.1016/0166-6851(92)90092-x. [DOI] [PubMed] [Google Scholar]
- 63.Wickstead B, Ersfeld K, Gull K. The small chromosomes of Trypanosama brucei involved in antigenic variation are constructed around repetititve palindromes. Genome Research. 2004;14(6):1014–1024. doi: 10.1101/gr.2227704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Bringaud F, Biteau N, Melville SE, et al. A new, expressed multigene family containing a hot spot for insertion of retroelements is associated with polymorphic subtelomeric regions of Trypanosoma brucei. Eukaryotic Cell. 2002;1(1):137–151. doi: 10.1128/EC.1.1.137-151.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Berriman M, Ghedin E, Hertz-Fowler C, et al. The genome of the African trypanosome Trypanosoma brucei. Science. 2005;309(5733):416–422. doi: 10.1126/science.1112642. [DOI] [PubMed] [Google Scholar]
- 66.Opperdoes FR. Compartmentation of carbohydrate metabolism in trypanosomes. Annual Review of Microbiology. 1987;41:127–151. doi: 10.1146/annurev.mi.41.100187.001015. [DOI] [PubMed] [Google Scholar]
- 67.Vickerman K, Tetley L, Hendry KAK, Turner CMR. Biology of African trypanosomes in the tsetse fly. Biology of the Cell. 1988;64(2):109–119. doi: 10.1016/0248-4900(88)90070-6. [DOI] [PubMed] [Google Scholar]
- 68.Opperdoes FR, Baudhuin P, Coppens I. Purification, morphometric analysis, and characterization of the glycosomes (microbodies) of the protozoan hemoflagellate Trypanosoma brucei. Journal of Cell Biology. 1984;98(4):1178–1184. doi: 10.1083/jcb.98.4.1178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Blattner J, Helfert S, Michels P, Clayton C. Compartmentation of phosphoglycerate kinase in Trypanosoma brucei plays a critical role in parasite energy metabolism. Proceedings of the National Academy of Sciences of the United States of America. 1998;95(20):11596–11600. doi: 10.1073/pnas.95.20.11596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Helfert S, Estévez AM, Bakker B, Michels P, Clayton C. Roles of triosephosphate isomerase and aerobic metabolism in Trypanosoma brucei. Biochemical Journal. 2001;357(1):117–125. doi: 10.1042/0264-6021:3570117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Michels PAM, Hannaert V, Bringaud F. Metabolic aspects of glycosomes in trypanosomatidae—new data and views. Parasitology Today. 2000;16(11):482–489. doi: 10.1016/s0169-4758(00)01810-x. [DOI] [PubMed] [Google Scholar]
- 72.Bakker BM, Mensonides FIC, Teusink B, Van Hoek P, Michels PAM, Westerhoff HV. Compartmentation protects trypanosomes from the dangerous design of glycolysis. Proceedings of the National Academy of Sciences of the United States of America. 2000;97(5):2087–2092. doi: 10.1073/pnas.030539197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Guerra-Giraldez C, Quijada L, Clayton CE. Compartmentation of enzymes in a microbody, the glycosome, is essential in Trypanosoma brucei. Journal of Cell Science. 2002;115(13):2651–2658. doi: 10.1242/jcs.115.13.2651. [DOI] [PubMed] [Google Scholar]
- 74.Colasante C, Ellis M, Ruppert T, Voncken F. Comparative proteomics of glycosomes from bloodstream form and procyclic culture form Trypanosoma brucei brucei. Proteomics. 2006;6(11):3275–3293. doi: 10.1002/pmic.200500668. [DOI] [PubMed] [Google Scholar]
- 75.Opperdoes FR, Szikora J-P. In silico prediction of the glycosomal enzymes of Leishmania major and trypanosomes. Molecular and Biochemical Parasitology. 2006;147(2):193–206. doi: 10.1016/j.molbiopara.2006.02.010. [DOI] [PubMed] [Google Scholar]
- 76.Field MC, Lumb JH, Adung’a VO, Jones NG, Engstler M. Macromolecular trafficking and immune evasion in African trypanosomes. International Review of Cell and Molecular Biology. 2009;278:1–67. doi: 10.1016/S1937-6448(09)78001-3. [DOI] [PubMed] [Google Scholar]
- 77.Engstler M, Pfohl T, Herminghaus S, et al. Hydrodynamic flow-mediated protein sorting on the cell surface of trypanosomes. Cell. 2007;131(3):505–515. doi: 10.1016/j.cell.2007.08.046. [DOI] [PubMed] [Google Scholar]
- 78.Pays E, Lips S, Nolan D, Vanhamme L, Pérez-Morga D. The VSG expression sites of Trypanosoma brucei: multipurpose tools for the adaptation of the parasite to mammalian hosts. Molecular and Biochemical Parasitology. 2001;114(1):1–16. doi: 10.1016/s0166-6851(01)00242-0. [DOI] [PubMed] [Google Scholar]
- 79.Vanhamme L, Poelvoorde P, Pays A, Tebabi P, Van Xong H, Pays E. Differential RNA elongation controls the variant surface glycoprotein gene expression sites of Trypanosoma brucei. Molecular Microbiology. 2000;36(2):328–340. doi: 10.1046/j.1365-2958.2000.01844.x. [DOI] [PubMed] [Google Scholar]
- 80.Pays E, Vanhamme L, Pérez-Morga D. Antigenic variation in Trypanosoma brucei: facts, challenges and mysteries. Current Opinion in Microbiology. 2004;7(4):369–374. doi: 10.1016/j.mib.2004.05.001. [DOI] [PubMed] [Google Scholar]
- 81.Navarro M, Gull K. A poll transcriptional body associated with VSG mono-allelic expression in Trypanosoma brucei. Nature. 2001;414(6865):759–763. doi: 10.1038/414759a. [DOI] [PubMed] [Google Scholar]
- 82.Pays E. Regulation of antigen gene expression in Trypanosoma brucei. Trends in Parasitology. 2005;21(11):517–520. doi: 10.1016/j.pt.2005.08.016. [DOI] [PubMed] [Google Scholar]
- 83.Pays E. The variant surface glycoprotein as a tool for adaptation in African trypanosomes. Microbes and Infection. 2006;8(3):930–937. doi: 10.1016/j.micinf.2005.10.002. [DOI] [PubMed] [Google Scholar]
- 84.Borst P, Ulbert S. Control of VSG gene expression sites. Molecular and Biochemical Parasitology. 2001;114(1):17–27. doi: 10.1016/s0166-6851(01)00243-2. [DOI] [PubMed] [Google Scholar]
- 85.Bitter W, Gerrits H, Kieft R, Borst P. The role of transferrin-receptor variation in the host range of Trypanosoma brucei. Nature. 1998;391(6666):499–502. doi: 10.1038/35166. [DOI] [PubMed] [Google Scholar]
- 86.Pays E, Guyaux M, Aerts D. Telomeric reciprocal recombination as a possible mechanism for antigenic variation in trypanosomes. Nature. 1985;316(6028):562–564. doi: 10.1038/316562a0. [DOI] [PubMed] [Google Scholar]
- 87.Pays E, Van Assel S, Laurent M, et al. Gene conversion as a mechanism for antigenic variation in Trypanosomes. Cell. 1983;34(2):371–381. doi: 10.1016/0092-8674(83)90371-9. [DOI] [PubMed] [Google Scholar]
- 88.Rudenko G, McCulloch R, Dirks-Mulder A, Borst P. Telomere exchange can be an important mechanism of variant surface glycoprotein gene switching in Trypanosoma brucei. Molecular and Biochemical Parasitology. 1996;80(1):65–75. doi: 10.1016/0166-6851(96)02669-2. [DOI] [PubMed] [Google Scholar]
- 89.Poelvoorde P, Vanhamme L, Van Den Abbeele J, Switzer WM, Pays E. Distribution of apolipoprotein L-I and trypanosome lytic activity among primate sera. Molecular and Biochemical Parasitology. 2004;134(1):155–157. doi: 10.1016/j.molbiopara.2003.11.006. [DOI] [PubMed] [Google Scholar]
- 90.Lugli EB, Pouliot M, Portela MDPM, Loomis MR, Raper J. Characterization of primate trypanosome lytic factors. Molecular and Biochemical Parasitology. 2004;138(1):9–20. doi: 10.1016/j.molbiopara.2004.07.004. [DOI] [PubMed] [Google Scholar]
- 91.De Greef C, Chimfwembe E, Kihang’a Wabacha J, Bajyana Songa E, Hamers R. Only the serum-resistant bloodstream forms of Trypanosoma brucei rhodesiense express the serum resistance associated (SRA) protein. Annales de la Societe Belge de Medecine Tropicale. 1992;72(supplement 1):13–21. [PubMed] [Google Scholar]
- 92.De Greef C, Imberechts H, Matthyssens G, Van Meirvenne N, Hamers R. A gene expressed only in serum-resistant variants of Trypanosoma brucei rhodesiense. Molecular and Biochemical Parasitology. 1989;36(2):169–176. doi: 10.1016/0166-6851(89)90189-8. [DOI] [PubMed] [Google Scholar]
- 93.Pays E, Vanhollebeke B, Vanhamme L, Paturiaux-Hanocq F, Nolan DP, Pérez-Morga D. The trypanolytic factor of human serum. Nature Reviews Microbiology. 2006;4(6):477–486. doi: 10.1038/nrmicro1428. [DOI] [PubMed] [Google Scholar]
- 94.Xong HV, Vanhamme L, Chamekh M, et al. A VSG expression site-associated gene confers resistance to human serum in Trypanosoma rhodesiense. Cell. 1998;95(6):839–846. doi: 10.1016/s0092-8674(00)81706-7. [DOI] [PubMed] [Google Scholar]
- 95.van Meirvenne N, Magnus E, Janssens PG. The effect of normal human serum on trypanosomes of distinct antigenic type (ETat 1 to 12) isolated from a strain of Trypanosoma brucei rhodesiense. Annales de la Societe Belge de Medecine Tropicale. 1976;56(1):55–63. [PubMed] [Google Scholar]
- 96.Welburn SC, Picozzi K, Fèvre EM, et al. Identification of human-infective trypanosomes in animal reservoir of sleeping sickness in Uganda by means of serum-resistance-associated (SRA) gene. The Lancet. 2001;358(9298):2017–2019. doi: 10.1016/s0140-6736(01)07096-9. [DOI] [PubMed] [Google Scholar]
- 97.De Greef C, Hamers R. The serum resistance-associated (SRA) gene of Trypanosoma brucei rhodesiense encodes a variant surface glycoprotein-like protein. Molecular and Biochemical Parasitology. 1994;68(2):277–284. doi: 10.1016/0166-6851(94)90172-4. [DOI] [PubMed] [Google Scholar]
- 98.Vanhamme L, Pays E. The trypanosome lytic factor of human serum and the molecular basis of sleeping sickness. International Journal for Parasitology. 2004;34(8):887–898. doi: 10.1016/j.ijpara.2004.04.008. [DOI] [PubMed] [Google Scholar]
- 99.Vanhamme L, Renauld H, Lecordier L, Poelvoorde P, Van Den Abbeele J, Pays E. The Trypanosoma brucei reference strain TREU927/4 contains T. brucei rhodesiense-specific SRA sequences, but displays a distinct phenotype of relative resistance to human serum. Molecular and Biochemical Parasitology. 2004;135(1):39–47. doi: 10.1016/j.molbiopara.2004.01.004. [DOI] [PubMed] [Google Scholar]
- 100.Campillo N, Carrington M. The origin of the serum resistance associated (SRA) gene and a model of the structure of the SRA polypeptide from Trypanosoma brucei rhodesiense. Molecular and Biochemical Parasitology. 2003;127(1):79–84. doi: 10.1016/s0166-6851(02)00306-7. [DOI] [PubMed] [Google Scholar]
- 101.Blum ML, Down JA, Gurnett AM, Carrington M, Turner MJ, Wiley DC. A structural motif in the variant surface glycoproteins of Trypanosoma brucei. Nature. 1993;362(6421):603–609. doi: 10.1038/362603a0. [DOI] [PubMed] [Google Scholar]
- 102.Vanhamme L, Paturiaux-Hanocq F, Poelvoorde P, et al. Apolipoprotein L-I is the trypanosome lytic factor of human serum. Nature. 2003;422(6927):83–87. doi: 10.1038/nature01461. [DOI] [PubMed] [Google Scholar]
- 103.Baral TN, Magez S, Stijlemans B, et al. Experimental therapy of African trypanosomiasis with a nanobody-conjugated human trypanolytic factor. Nature Medicine. 2006;12(5):580–584. doi: 10.1038/nm1395. [DOI] [PubMed] [Google Scholar]
- 104.Ruvolo M, Pan D, Zehr S, Goldberg T, Disotell TR, Von Dornum M. Gene trees and hominoid phylogeny. Proceedings of the National Academy of Sciences of the United States of America. 1994;91(19):8900–8904. doi: 10.1073/pnas.91.19.8900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Rifkin MR. Role of phospholipids in the cytotoxic action of high density lipoprotein on trypanosomes. Journal of Lipid Research. 1991;32(4):639–647. [PubMed] [Google Scholar]
- 106.Hajduk SL, Moore DR, Vasudevacharya J, et al. Lysis of Trypanosoma brucei by a toxic subspecies of human high density lipoprotein. Journal of Biological Chemistry. 1989;264(9):5210–5217. [PubMed] [Google Scholar]
- 107.Hager KM, Pierce MA, Moore DR, Tytler EM, Esko JD, Hajduk SL. Endocytosis of a cytotoxic human high density lipoprotein results in disruption of acidic intracellular vesicles and subsequent killing of African trypanosomes. Journal of Cell Biology. 1994;126(1):155–167. doi: 10.1083/jcb.126.1.155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Hager KM, Hajduk SL. Mechanism of resistance of African trypanosomes to cytotoxic human HDL. Nature. 1997;385(6619):823–826. doi: 10.1038/385823a0. [DOI] [PubMed] [Google Scholar]
- 109.Green HP, Portela MDPM, Jean ENSt, Lugli EB, Raper J. Evidence for a Trypanosoma brucei lipoprotein scavenger receptor. Journal of Biological Chemistry. 2003;278(1):422–427. doi: 10.1074/jbc.M207215200. [DOI] [PubMed] [Google Scholar]
- 110.Smith AB, Esko JD, Hajduk SL. Killing of trypanosomes by the human haptoglobin-related protein. Science. 1995;268(5208):284–286. doi: 10.1126/science.7716520. [DOI] [PubMed] [Google Scholar]
- 111.Raper J, Fung R, Ghiso J, Nussenzweig V, Tomlinson S. Characterization of a novel trypanosome lytic factor from human serum. Infection and Immunity. 1999;67(4):1910–1916. doi: 10.1128/iai.67.4.1910-1916.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Molina-Portela MDP, Lugli EB, Recio-Pinto E, Raper J. Trypanosome lytic factor, a subclass of high-density lipoprotein, forms cation-selective pores in membranes. Molecular and Biochemical Parasitology. 2005;144(2):218–226. doi: 10.1016/j.molbiopara.2005.08.018. [DOI] [PubMed] [Google Scholar]
- 113.Tomlinson S, Muranjan M, Nussenzweig V, Raper J. Haptoglobin-related protein and apolipoprotein AI are components of the two trypanolytic factors in human serum. Molecular and Biochemical Parasitology. 1997;86(1):117–120. [PubMed] [Google Scholar]
- 114.Drain J, Bishop JR, Hajduk SL. Haptoglobin-related protein mediates trypanosome lytic factor binding to trypanosomes. Journal of Biological Chemistry. 2001;276(32):30254–30260. doi: 10.1074/jbc.M010198200. [DOI] [PubMed] [Google Scholar]
- 115.Smith AB, Hajduk SL. Identification of haptoglobin as a natural inhibitor of trypanocidal activity in human serum. Proceedings of the National Academy of Sciences of the United States of America. 1995;92(22):10262–10266. doi: 10.1073/pnas.92.22.10262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Pérez-Morga D, Vanhollebeke B, Paturiaux-Hanocq F, et al. Apolipoprotein L-I promotes trypanosome lysis by forming pores in lysosomal membranes. Science. 2005;309(5733):469–472. doi: 10.1126/science.1114566. [DOI] [PubMed] [Google Scholar]
- 117.Shiflett AM, Bishop JR, Pahwa A, Hajduk SL. Human high density lipoproteins are platforms for the assembly of multi-component innate immune complexes. Journal of Biological Chemistry. 2005;280(38):32578–32585. doi: 10.1074/jbc.M503510200. [DOI] [PubMed] [Google Scholar]
- 118.Molina-Portela MP, Samanovic M, Raper J. Distinct roles of apolipoprotein components within the trypanosome lytic factor complex revealed in a novel transgenic mouse model. Journal of Experimental Medicine. 2008;205(8):1721–1728. doi: 10.1084/jem.20071463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Thomson R, Molina-Portela P, Mott H, Carrington M, Raper J. Hydrodynamic gene delivery of baboon trypanosome lytic factor eliminates both animal and human-infective African trypanosomes. Proceedings of the National Academy of Sciences of the United States of America. 2010;106(46):19509–19514. doi: 10.1073/pnas.0905669106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Samanovic M, Molina-Portela MP, Chessler A-DC, Burleigh BA, Raper J. Trypanosome lytic factor, an antimicrobial high-density lipoprotein, ameliorates Leishmania infection. PLoS Pathogens. 2009;5(1) doi: 10.1371/journal.ppat.1000276. Article ID e1000276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Thomson R, Samanovic M, Raper J. Activity of trypanosome lytic factor: a novel component of innate immunity. Future Microbiology. 2009;4(7):789–796. doi: 10.2217/FMB.09.57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Duchateau PN, Pullinger CR, Orellana RE, et al. Apolipoprotein L, a new human high density lipoprotein apolipoprotein expressed by the pancreas. Identification, cloning, characterization, and plasma distribution of apolipoprotein L. Journal of Biological Chemistry. 1997;272(41):25576–25582. doi: 10.1074/jbc.272.41.25576. [DOI] [PubMed] [Google Scholar]
- 123.Duchateau PN, Pullinger CR, Cho MH, Eng C, Kane JP. Apolipoprotein L gene family: tissue-specific expression, splicing, promoter regions; discovery of a new gene. Journal of Lipid Research. 2001;42(4):620–630. [PubMed] [Google Scholar]
- 124.Page NM, Butlin DJ, Lomthaisong K, Lowry PJ. The human apolipoprotein L gene cluster: identification, classification, and sites of distribution. Genomics. 2001;74(1):71–78. doi: 10.1006/geno.2001.6534. [DOI] [PubMed] [Google Scholar]
- 125.Harrington JM, Howell S, Hajduk SL. Membrane permeabilization by trypanosome lytic factor, a cytolytic human high density lipoprotein. Journal of Biological Chemistry. 2009;284(20):13505–13512. doi: 10.1074/jbc.M900151200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Roditi I, Clayton C. An unambiguous nomenclature for the major surface glycoproteins of the procyclic form of Trypanosoma brucei. Molecular and Biochemical Parasitology. 1999;103(1):99–100. doi: 10.1016/s0166-6851(99)00124-3. [DOI] [PubMed] [Google Scholar]
- 127.Urwyler S, Vassella E, Van Den Abbeele J, et al. Expression of procyclin mRNAs during cyclical transmission of Trypanosoma brucei. PLoS Pathogens. 2005;1(3) doi: 10.1371/journal.ppat.0010022. Article ID e22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Vickerman K, Luckins AG. Localization of variable antigens in the surface coat of Trypanosoma brucei using ferritin conjugated antibody. Nature. 1969;224(5224):1125–1126. doi: 10.1038/2241125a0. [DOI] [PubMed] [Google Scholar]
- 129.Cross GAM. Identification, purification and properties of clone specific glycoprotein antigens constituting the surface coat of Trypanosoma brucei. Parasitology. 1975;71(3):393–417. doi: 10.1017/s003118200004717x. [DOI] [PubMed] [Google Scholar]
- 130.Ziegelbauer K, Overath P. Organization of two invariant surface glycoproteins in the surface coat of Trypanosoma brucei. Infection and Immunity. 1993;61(11):4540–4545. doi: 10.1128/iai.61.11.4540-4545.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Ferrante A, Callison AC. Alternative pathway activation of complement by African trypanosomes lacking a glycoprotein coat. Parasite Immunology. 1983;5(5):491–498. doi: 10.1111/j.1365-3024.1983.tb00763.x. [DOI] [PubMed] [Google Scholar]
- 132.Carrington M, Boothroyd J. Implications of conserved structural motifs in disparate trypanosome surface proteins. Molecular and Biochemical Parasitology. 1996;81(2):119–126. doi: 10.1016/0166-6851(96)02706-5. [DOI] [PubMed] [Google Scholar]
- 133.Carrington M, Miller N, Blum M, Roditi I, Wiley D, Turner M. Variant specific glycoprotein of Trypanosoma brucei consists of two domains each having an independently conserved pattern of cysteine residues. Journal of Molecular Biology. 1991;221(3):823–835. doi: 10.1016/0022-2836(91)80178-w. [DOI] [PubMed] [Google Scholar]
- 134.Mehlert A, Richardson JM, Ferguson MAJ. Structure of the glycosylphosphatidylinositol membrane anchor glycan of a class-2 variant surface glycoprotein from Trypanosoma brucei. Journal of Molecular Biology. 1998;277(2):379–392. doi: 10.1006/jmbi.1997.1600. [DOI] [PubMed] [Google Scholar]
- 135.Hutchinson OC, Smith W, Jones NG, Chattopadhyay A, Welburn SC, Carrington M. VSG structure: similar N-terminal domains can form functional VSGs with different types of C-terminal domain. Molecular and Biochemical Parasitology. 2003;130(2):127–131. doi: 10.1016/s0166-6851(03)00144-0. [DOI] [PubMed] [Google Scholar]
- 136.Ferguson MAJ, Homans SW, Dwek RA, Rademacher TW. Glycosyl-phosphatidylinositol moiety that anchors Trypanosoma brucei variant surface glycoprotein to the membrane. Science. 1988;239(4841, part 1):753–759. doi: 10.1126/science.3340856. [DOI] [PubMed] [Google Scholar]
- 137.McDowell MA, Ransom DM, Bangs JD. Glycosylphosphatidylinositol-dependent secretory transport in Trypanosoma brucei. Biochemical Journal. 1998;335(3):681–689. doi: 10.1042/bj3350681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Freymann D, Down J, Carrington M, Roditi I, Turner M, Wiley D. 2.9 Å resolution structure of the N-terminal domain of a variant surface glycoprotein from Trypanosoma brucei. Journal of Molecular Biology. 1990;216(1):141–160. doi: 10.1016/S0022-2836(05)80066-X. [DOI] [PubMed] [Google Scholar]
- 139.Cross GA. Structure of the variant glycoproteins and surface coat of Trypanosoma brucei. Philosophical Transactions of the Royal Society B. 1984;307(1131):3–12. doi: 10.1098/rstb.1984.0104. [DOI] [PubMed] [Google Scholar]
- 140.Enghund PT. The structure and biosynthesis of glycosyl phosphatidylinositol protein anchors. Annual Review of Biochemistry. 1993;62:121–138. doi: 10.1146/annurev.bi.62.070193.001005. [DOI] [PubMed] [Google Scholar]
- 141.Ferguson MA, Homans SW, Dwek RA, Rademacher TW. The glycosylphosphatidylinositol membrane anchor of Trypanosoma brucei variant surface glycoprotein. Biochemical Society Transactions. 1988;16(3):265–268. doi: 10.1042/bst0160265. [DOI] [PubMed] [Google Scholar]
- 142.Triggs VP, Bangs JD. Glycosylphosphatidylinositol-dependent protein trafficking in bloodstream stage Trypanosoma brucei. Eukaryotic Cell. 2003;2(1):76–83. doi: 10.1128/EC.2.1.76-83.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Böhme U, Cross GAM. Mutational analysis of the variant surface glycoprotein GPI-anchor signal sequence in Trypanosoma brucei. Journal of Cell Science. 2002;115(4):805–816. doi: 10.1242/jcs.115.4.805. [DOI] [PubMed] [Google Scholar]
- 144.Masterson WJ, Doering TL, Hart GW, Englund PT. A novel pathway for glycan assembly: biosynthesis of the glycosyl-phosphatidylinositol anchor of the trypanosome variant surface glycoprotein. Cell. 1989;56(5):793–800. doi: 10.1016/0092-8674(89)90684-3. [DOI] [PubMed] [Google Scholar]
- 145.Ferguson MAJ, Williams AF. Cell-surface anchoring of proteins via glycosyl-phosphatidylinositol structures. Annual Review of Biochemistry. 1988;57:285–320. doi: 10.1146/annurev.bi.57.070188.001441. [DOI] [PubMed] [Google Scholar]
- 146.Ferguson MAJ. The structure, biosynthesis and functions of glycosylphosphatidylinositol anchors, and the contributions of trypanosome research. Journal of Cell Science. 1999;112(17):2799–2809. doi: 10.1242/jcs.112.17.2799. [DOI] [PubMed] [Google Scholar]
- 147.Paul KS, Jiang D, Morita YS, Englund PT. Fatty acid synthesis in African trypanosomes: a solution to the myristate mystery. Trends in Parasitology. 2001;17(8):381–387. doi: 10.1016/s1471-4922(01)01984-5. [DOI] [PubMed] [Google Scholar]
- 148.Ferguson MAJ, Cross GAM. Myristylation of the membrane form of a Trypanosoma brucei variant surface glycoprotein. The Journal of Biological Chemistry. 1984;259(5):3011–3015. [PubMed] [Google Scholar]
- 149.Doering TL, Raper J, Buxbaum LU, et al. An analog of myristic acid with selective toxicity for African trypanosomes. Science. 1991;252(5014):1851–1854. doi: 10.1126/science.1829548. [DOI] [PubMed] [Google Scholar]
- 150.Hong Y, Kinoshita T. Trypanosome glycosylphosphatidylinositol biosynthesis. Korean Journal of Parasitology. 2009;47(3):197–204. doi: 10.3347/kjp.2009.47.3.197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Carrington M, Carnall N, Crow MS, et al. The properties and function of the glycosylphosphatidylinositol-phospholipase C in Trypanosoma brucei. Molecular and Biochemical Parasitology. 1998;91(1):153–164. doi: 10.1016/s0166-6851(97)00190-4. [DOI] [PubMed] [Google Scholar]
- 152.Grab DJ, Webster P, Ito S, et al. Subcellular localization of a variable surface glycoprotein phosphatidylinositol-specific phospholipase-C in African trypanosomes. Journal of Cell Biology. 1987;105(2):737–746. doi: 10.1083/jcb.105.2.737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Mensa-Wilmot K, Hereld D, Englund PT. Genomic organization, chromosomal localization, and developmentally regulated expression of the glycosyl-phosphatidylinositol-specific phospholipase C of Trypanosoma brucei. Molecular and Cellular Biology. 1990;10(2):720–726. doi: 10.1128/mcb.10.2.720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Carrington M, Bulow R, Reinke H, et al. Sequence and expression of the glycosyl-phosphatidylinositol-specific phospholipase C of Trypanosoma brucei. Molecular and Biochemical Parasitology. 1989;33(3):289–296. doi: 10.1016/0166-6851(89)90091-1. [DOI] [PubMed] [Google Scholar]
- 155.De Almeida MLC, Geuskens M, Pays E. Cell lysis induces redistribution of the GPI-anchored variant surface glycoprotein on both faces of the plasma membrane of Trypanosoma brucei. Journal of Cell Science. 1999;112(part 23):4461–4473. doi: 10.1242/jcs.112.23.4461. [DOI] [PubMed] [Google Scholar]
- 156.Ferguson MAJ, Haldar K, Cross GAM. Trypanosoma brucei variant surface glycoprotein has a sn-1,2-dimyristyl glycerol membrane anchor at its COOH terminus. The Journal of Biological Chemistry. 1985;260(8):4963–4968. [PubMed] [Google Scholar]
- 157.Ferguson MAJ, Low MG, Cross GAM. Glycosyl-sn-1,2-dimyristylphosphatidylinositol is covalently linked to Trypanosoma brucei variant surface glycoprotein. The Journal of Biological Chemistry. 1985;260(27):14547–14555. [PubMed] [Google Scholar]
- 158.Cardoso de Almeida ML, Turner MJ. The membrane form of variant surface glycoproteins of Trypanosoma brucei. Nature. 1983;302(5906):349–352. doi: 10.1038/302349a0. [DOI] [PubMed] [Google Scholar]
- 159.Holder AA, Cross GAM. Glycopeptides from variant surface glycoproteins of Trypanosoma brucei. C-terminal location of antigenically cross-reacting carbohydrate moieties. Molecular and Biochemical Parasitology. 1981;2(3-4):135–150. doi: 10.1016/0166-6851(81)90095-5. [DOI] [PubMed] [Google Scholar]
- 160.Zamze SE, Ferguson MAJ, Collins R, Dwek RA, Rademacher TW. Characterization of the cross-reacting determinant (CRD) of the glycosyl-phosphatidylinositol membrane anchor of Trypanosoma brucei variant surface glycoprotein. European Journal of Biochemistry. 1988;176(3):527–534. doi: 10.1111/j.1432-1033.1988.tb14310.x. [DOI] [PubMed] [Google Scholar]
- 161.Bulow R, Nonnengasser C, Overath P. Release of the variant surface glycoprotein during differentiation of bloodstream to procyclic forms of Trypanosoma brucei. Molecular and Biochemical Parasitology. 1989;32(1):85–92. doi: 10.1016/0166-6851(89)90132-1. [DOI] [PubMed] [Google Scholar]
- 162.Rolin S, Hanocq-Quertier J, Paturiaux-Hanocq F, et al. Simultaneous but independent activation of adenylate cyclase and glycosylphosphatidylinositol-phospholipase C under stress conditions in Trypanosoma brucei. The Journal of Biological Chemistry. 1996;271(18):10844–10852. doi: 10.1074/jbc.271.18.10844. [DOI] [PubMed] [Google Scholar]
- 163.Carrington M, Walters D, Webb H. The biology of the glycosylphosphatidylinositol-specific phospholipase C of Trypanosoma brucei. Cell Biology International Reports. 1991;15(11):1101–1114. doi: 10.1016/0309-1651(91)90058-q. [DOI] [PubMed] [Google Scholar]
- 164.Webb H, Carnall N, Vanhamme L, et al. The GPI-phospholipase C of Trypanosoma brucei is non essential but influences parasitemia in mice. Journal of Cell Biology. 1997;139(1):103–114. doi: 10.1083/jcb.139.1.103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Overath P, Chaudhri M, Steverding D, Ziegelbauer K. Invariant surface proteins in bloodstream forms of Trypanosoma brucei. Parasitology Today. 1994;10(2):53–58. doi: 10.1016/0169-4758(94)90393-x. [DOI] [PubMed] [Google Scholar]
- 166.Ziegelbauer K, Overath P. Identification of invariant surface glycoproteins in the bloodstream stage of Trypanosoma brucei. The Journal of Biological Chemistry. 1992;267(15):10791–10796. [PubMed] [Google Scholar]
- 167.Jackson DG, Windle HJ, Voorheis HP. The identification, purification, and characterization of two invariant surface glycoproteins located beneath the surface coat barrier of bloodstream forms of Trypanosoma brucei. The Journal of Biological Chemistry. 1993;268(11):8085–8095. [PubMed] [Google Scholar]
- 168.Nolan DP, Jackson DG, Windle HJ, et al. Characterization of a novel, stage-specific, invariant surface protein in Trypanosoma brucei containing an internal, serine-rich, repetitive motif. The Journal of Biological Chemistry. 1997;272(46):29212–29221. doi: 10.1074/jbc.272.46.29212. [DOI] [PubMed] [Google Scholar]
- 169.Schell D, Borowy NK, Overath P. Transferrin is a growth factor for the bloodstream form of Trypanosoma brucei. Parasitology Research. 1991;77(7):558–560. doi: 10.1007/BF00931012. [DOI] [PubMed] [Google Scholar]
- 170.Steverding D, Stierhof YD, Fuchs H, Tauber R, Overath P. Transferrin-binding protein complex is the receptor for transferrin uptake in Trypanosoma brucei. Journal of Cell Biology. 1995;131(5):1173–1182. doi: 10.1083/jcb.131.5.1173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Steverding D. Bloodstream forms of Trypanosoma brucei require only small amounts of iron for growth. Parasitology Research. 1998;84(1):59–62. doi: 10.1007/s004360050357. [DOI] [PubMed] [Google Scholar]
- 172.Steverding D. The significance of transferrin receptor variation in Trypanosoma brucei. Trends in Parasitology. 2003;19(3):125–127. doi: 10.1016/s1471-4922(03)00006-0. [DOI] [PubMed] [Google Scholar]
- 173.Huebers HA, Finch CA. The physiology of transferrin and transferrin receptors. Physiological Reviews. 1987;67(2):520–582. doi: 10.1152/physrev.1987.67.2.520. [DOI] [PubMed] [Google Scholar]
- 174.Salmon D, Hanocq-Quertier J, Paturiaux-Hanocq F, et al. Characterization of the ligand-binding site of the transferrin receptor in Trypanosoma brucei demonstrates a structural relationship with the N-terminal domain of the variant surface glycoprotein. EMBO Journal. 1997;16(24):7272–7278. doi: 10.1093/emboj/16.24.7272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Steverding D. The transferrin receptor of Trypanosoma brucei. Parasitology International. 2000;48(3):191–198. doi: 10.1016/s1383-5769(99)00018-5. [DOI] [PubMed] [Google Scholar]
- 176.Coppens I, Bastin Ph, Opperdoes FR, Baudhuin P, Courtoy PJ. Trypanosoma brucei brucei: antigenic stability of its LDL receptor and immunological cross-reactivity with the LDL receptor of the mammalian host. Experimental Parasitology. 1992;74(1):77–86. doi: 10.1016/0014-4894(92)90141-v. [DOI] [PubMed] [Google Scholar]
- 177.Coppens I, Bastin P, Courtoy PJ, Baudhuin P, Opperdoes FR. A rapid method purifies a glycoprotein of Mr 145,000 as the LDL receptor of Trypanosoma brucei brucei. Biochemical and Biophysical Research Communications. 1991;178(1):185–191. doi: 10.1016/0006-291x(91)91797-g. [DOI] [PubMed] [Google Scholar]
- 178.Tetaud E, Barrett MP, Bringaud F, Baltz T. Kinetoplastid glucose transporters. Biochemical Journal. 1997;325(3):569–580. doi: 10.1042/bj3250569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Sanchez MA, Drutman S, Van Ampting M, Matthews K, Landfear SM. A novel purine nucleoside transporter whose expression is up-regulated in the short stumpy form of the Trypanosoma brucei life cycle. Molecular and Biochemical Parasitology. 2004;136(2):265–272. doi: 10.1016/j.molbiopara.2004.04.009. [DOI] [PubMed] [Google Scholar]
- 180.Vincendeau P, Bouteille B. Immunology and immunopathology of African trypanosomiasis. Anais da Academia Brasileira de Ciencias. 2006;78(4):645–665. doi: 10.1590/s0001-37652006000400004. [DOI] [PubMed] [Google Scholar]
- 181.Stijlemans B, Guilliams M, Raes G, Beschin A, Magez S, De Baetselier P. African trypanosomosis: from immune escape and immunopathology to immune intervention. Veterinary Parasitology. 2007;148(1):3–13. doi: 10.1016/j.vetpar.2007.05.005. [DOI] [PubMed] [Google Scholar]
- 182.Janeway CA, Jr., Medzhitov R. Innate immune recognition. Annual Review of Immunology. 2002;20:197–216. doi: 10.1146/annurev.immunol.20.083001.084359. [DOI] [PubMed] [Google Scholar]
- 183.Takeda K, Kaisho T, Akira S. Toll-like receptors. Annual Review of Immunology. 2003;21:335–376. doi: 10.1146/annurev.immunol.21.120601.141126. [DOI] [PubMed] [Google Scholar]
- 184.Shoda LKM, Kegerreis KA, Suarez CE, et al. DNA from protozoan parasites Babesia bovis, Trypanosoma cruzi, and T. brucei is mitogenic for B lymphocytes and stimulates macrophage expression of interleukin-12, tumor necrosis factor alpha, and nitric oxide. Infection and Immunity. 2001;69(4):2162–2171. doi: 10.1128/IAI.69.4.2162-2171.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Harris TH, Cooney NM, Mansfield JM, Paulnock DM. Signal transduction, gene transcription, and cytokine production triggered in macrophages by exposure to trypanosome DNA. Infection and Immunity. 2006;74(8):4530–4537. doi: 10.1128/IAI.01938-05. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Drennan MB, Stijlemans B, Van Den Abbeele J, et al. The induction of a type 1 immune response following a Trypanosoma brucei infection is MyD88 dependent. Journal of Immunology. 2005;175(4):2501–2509. doi: 10.4049/jimmunol.175.4.2501. [DOI] [PubMed] [Google Scholar]
- 187.Almeida IC, Camargo MM, Procópio DO, et al. Highly purified glycosylphosphatidylinositols from Trypanosoma cruzi are potent proinflammatory agents. EMBO Journal. 2000;19(7):1476–1485. doi: 10.1093/emboj/19.7.1476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Coller SP, Mansfield JM, Paulnock DM. Glycosylinositolphosphate soluble variant surface glycoprotein inhibits IFN-γ-induced nitric oxide production via reduction in STAT1 phosphorylation in African trypanosomiasis. Journal of Immunology. 2003;171(3):1466–1472. doi: 10.4049/jimmunol.171.3.1466. [DOI] [PubMed] [Google Scholar]
- 189.Tachado SD, Schofield L. Glycosylphosphatidylinositol toxin of Trypanosoma brucei regulates IL-1α and TNF-α expression in macrophages by protein tyrosine kinase mediated signal transduction. Biochemical and Biophysical Research Communications. 1994;205(2):984–991. doi: 10.1006/bbrc.1994.2763. [DOI] [PubMed] [Google Scholar]
- 190.Shi M, Wei G, Pan W, Tabel H. Trypanosoma congolense infections: antibody-mediated phagocytosis by Kupffer cells. Journal of Leukocyte Biology. 2004;76(2):399–405. doi: 10.1189/jlb.1003500. [DOI] [PubMed] [Google Scholar]
- 191.Mabbott NA, Sutherland IA, Sternberg JM. Trypanosoma brucei is protected from the cytostatic effects of nitric oxide under in vivo conditions. Parasitology Research. 1994;80(8):687–690. doi: 10.1007/BF00932954. [DOI] [PubMed] [Google Scholar]
- 192.Sternberg JM, Mabbott NA. Nitric oxide-mediated suppression of T cell responses during Trypanosoma brucei infection: soluble trypanosome products and interferon-γ are synergistic inducers of nitric oxide synthase. European Journal of Immunology. 1996;26(3):539–543. doi: 10.1002/eji.1830260306. [DOI] [PubMed] [Google Scholar]
- 193.Kaushik RS, Uzonna JE, Zhang Y, Gordon JR, Tabel H. Innate resistance to experimental African trypanosomiasis: differences in cytokine (TNF-α, IL-6, IL-10 and IL-12) production by bone marrow-derived macrophages from resistant and susceptible mice. Cytokine. 2000;12(7):1024–1034. doi: 10.1006/cyto.2000.0685. [DOI] [PubMed] [Google Scholar]
- 194.Magez S, Geuskens M, Beschin A, et al. Specific uptake of tumor necrosis factor-α is involved in growth control of Trypanosoma brucei. Journal of Cell Biology. 1997;137(3):715–727. doi: 10.1083/jcb.137.3.715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Namangala B, de Baetselier P, Beschin A. Both type-I and type-II responses contribute to murine trypanotolerance. Journal of Veterinary Medical Science. 2009;71(3):313–318. doi: 10.1292/jvms.71.313. [DOI] [PubMed] [Google Scholar]
- 196.Hertz CJ, Filutowicz H, Mansfield JM. Resistance to the African trypanosomes is IFN-γ dependent. Journal of Immunology. 1998;161(12):6775–6783. [PubMed] [Google Scholar]
- 197.Uzonna JE, Kaushik RS, Gordon JR, Tabel H. Cytokines and antibody responses during Trypanosoma congolense infections in two inbred mouse strains that differ in resistance. Parasite Immunology. 1999;21(2):57–71. doi: 10.1046/j.1365-3024.1999.00202.x. [DOI] [PubMed] [Google Scholar]
- 198.Inoue N, Inoue M, Kuriki K, et al. Interleukin 4 is a crucial cytokine in controlling Trypanosoma brucei gambiense infection in mice. Veterinary Parasitology. 1999;86(3):173–184. doi: 10.1016/s0304-4017(99)00143-0. [DOI] [PubMed] [Google Scholar]
- 199.Mertens B, Taylor K, Muriuki C, Rocchi M. Cytokine mRNA profiles in trypanotolerant and trypanosusceptible cattle infected with the protozoan parasite Trypanosoma congolense: protective role for interleukin-4? Journal of Interferon and Cytokine Research. 1999;19(1):59–65. doi: 10.1089/107999099314423. [DOI] [PubMed] [Google Scholar]
- 200.Magez S, Radwanska M, Drennan M, et al. Interferon-γ and nitric oxide in combination with antibodies are key protective host immune factors during Trypanosoma congolense Tc13 infections. Journal of Infectious Diseases. 2006;193(11):1575–1583. doi: 10.1086/503808. [DOI] [PubMed] [Google Scholar]
- 201.Baral TN, De Baetselier P, Brombacher F, Magez S. Control of Trypanosoma evansi infection is IgM mediated and does not require a type I inflammatory response. Journal of Infectious Diseases. 2007;195(10):1513–1520. doi: 10.1086/515577. [DOI] [PubMed] [Google Scholar]
- 202.Forlenza M, Nakao M, Wibowo I, et al. Nitric oxide hinders antibody clearance from the surface of Trypanoplasma borreli and increases susceptibility to complement-mediated lysis. Molecular Immunology. 2009;46(16):3188–3197. doi: 10.1016/j.molimm.2009.08.011. [DOI] [PubMed] [Google Scholar]
- 203.Magez S, Lucas R, Darji A, Bajyana Songa E, Hamers R, De Baetselier P. Murine tumour necrosis factor plays a protective role during the initial phase of the experimental infection with Trypanosoma brucei brucei. Parasite Immunology. 1993;15(11):635–641. doi: 10.1111/j.1365-3024.1993.tb00577.x. [DOI] [PubMed] [Google Scholar]
- 204.Magez S, Radwanska M, Beschin A, Sekikawa K, De Baetselier P. Tumor necrosis factor alpha is a key mediator in the regulation of experimental Trypanosoma brucei infections. Infection and Immunity. 1999;67(6):3128–3132. doi: 10.1128/iai.67.6.3128-3132.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Namangala B, De Baetselier P, Brijs L, et al. Attenuation of Trypanosoma brucei is associated with reduced immunosuppression and concomitant production of Th2 lymphokines. Journal of Infectious Diseases. 2000;181(3):1110–1120. doi: 10.1086/315322. [DOI] [PubMed] [Google Scholar]
- 206.Sternberg JM, Rodgers J, Bradley B, MacLean L, Murray M, Kennedy PGE. Meningoencephalitic African trypanosomiasis: brain IL-10 and IL-6 are associated with protection from neuro-inflammatory pathology. Journal of Neuroimmunology. 2005;167(1-2):81–89. doi: 10.1016/j.jneuroim.2005.06.017. [DOI] [PubMed] [Google Scholar]
- 207.Kennedy PGE. Cytokines in central nervous system trypanosomiasis: cause, effect or both? Transactions of the Royal Society of Tropical Medicine and Hygiene. 2009;103(3):213–214. doi: 10.1016/j.trstmh.2008.08.013. [DOI] [PubMed] [Google Scholar]
- 208.Baetselier P, Namangala B, Noël W, Brys L, Pays E, Beschin A. Alternative versus classical macrophage activation during experimental African trypanosomosis. International Journal for Parasitology. 2001;31(5-6):575–587. doi: 10.1016/s0020-7519(01)00170-9. [DOI] [PubMed] [Google Scholar]
- 209.Hudson KM, Byner C, Freeman J, Terry RJ. Immunodepression, high IgM levels and evasion of the immune response in murine trypanosomiasis. Nature. 1976;264(5583):256–258. doi: 10.1038/264256a0. [DOI] [PubMed] [Google Scholar]
- 210.Luckins AG, Mehlitz D. Evaluation of an indirect fluorescent antibody test, enzyme-linked immunosorbent assay and quantification of immunoglobulins in the diagnosis of bovine trypanosomiasis. Tropical Animal Health and Production. 1978;10(3):149–159. doi: 10.1007/BF02235328. [DOI] [PubMed] [Google Scholar]
- 211.Luckins AG, Mehlitz D. Observations on serum immunoglobulin levels in cattle infected with Trypanosoma brucei, T. vivax and T. congolense. Annals of Tropical Medicine and Parasitology. 1976;70(4):479–480. doi: 10.1080/00034983.1976.11687153. [DOI] [PubMed] [Google Scholar]
- 212.Buza J, Sileghem M, Gwakisa P, Naessens J. CD5+ B lymphocytes are the main source of antibodies reactive with non-parasite antigens in Trypanosoma congolense-infected cattle. Immunology. 1997;92(2):226–233. doi: 10.1046/j.1365-2567.1997.00330.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Williams DJL, Taylor K, Newson J, Gichuki B, Naessens J. The role of anti-variable surface glycoprotein antibody responses in bovine trypanotolerance. Parasite Immunology. 1996;18(4):209–218. doi: 10.1046/j.1365-3024.1996.d01-76.x. [DOI] [PubMed] [Google Scholar]
- 214.Kobayakawa T, Louis J, Izui S, Lambert PH. Autoimmune response to DNA, red blood cells, and thymocyte antigens in association with polyclonal antibody synthesis during experimental African trypanosomiasis. Journal of Immunology. 1979;122(1):296–301. [PubMed] [Google Scholar]
- 215.Schleifer KW, Filutowicz H, Schopf LR, Mansfield JM. Characterization of T helper cell responses to the trypanosome variant surface glycoprotein. Journal of Immunology. 1993;150(7):2910–2919. [PubMed] [Google Scholar]
- 216.Morrison WI, Murray M. The role of humoral immune responses in determining susceptibility of A/J and C57BL/6 mice to infection with Trypanosoma congolense. Parasite Immunology. 1985;7(1):63–79. doi: 10.1111/j.1365-3024.1985.tb00479.x. [DOI] [PubMed] [Google Scholar]
- 217.Taylor KA, Lutje V, Kennedy D, et al. Trypanosoma congolense: B-Lymphocyte responses differ between trypanotolerant and trypanosusceptible cattle. Experimental Parasitology. 1996;83(1):106–116. doi: 10.1006/expr.1996.0054. [DOI] [PubMed] [Google Scholar]
- 218.Morrison WI, Black SJ, Paris J. Protective immunity and specificity of antibody responses elicited in cattle by irradiated Trypanosoma brucei. Parasite Immunology. 1982;4(6):395–407. doi: 10.1111/j.1365-3024.1982.tb00451.x. [DOI] [PubMed] [Google Scholar]
- 219.Duxbury RE, Sadun EH, Wellde BT, Anderson JS, Muriithi IE. Immunization of cattle with x-irradiated African trypanosomes. Transactions of the Royal Society of Tropical Medicine and Hygiene. 1972;66(2):349–350. doi: 10.1016/0035-9203(72)90237-4. [DOI] [PubMed] [Google Scholar]
- 220.Mosser DM, Roberts JF. Trypanosoma brucei: recognition in vitro of two developmental forms by murine macrophages. Experimental Parasitology. 1982;54(3):310–316. doi: 10.1016/0014-4894(82)90040-6. [DOI] [PubMed] [Google Scholar]
- 221.Pan W, Ogunremi O, Wei G, Shi M, Tabel H. CR3 (CD11b/CD18) is the major macrophage receptor for IgM antibody-mediated phagocytosis of African trypanosomes: diverse effect on subsequent synthesis of tumor necrosis factor α and nitric oxide. Microbes and Infection. 2006;8(5):1209–1218. doi: 10.1016/j.micinf.2005.11.009. [DOI] [PubMed] [Google Scholar]
- 222.Reinitz DM, Mansfield JM. T-cell-independent and T-cell-dependent B-cell responses to exposed variant surface glycoprotein epitopes in trypanosome-infected mice. Infection and Immunity. 1990;58(7):2337–2342. doi: 10.1128/iai.58.7.2337-2342.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Schofield L, McConville MJ, Hansen D, et al. CD1d-restricted immunoglobulin G formation to GPI-anchored antigens mediated by NKT cells. Science. 1999;283(5399):225–229. doi: 10.1126/science.283.5399.225. [DOI] [PubMed] [Google Scholar]
- 224.Macaskill JA, Holmes PH, Whitelaw DD, et al. Immunological clearance of 75Se-labelled Trypanosoma brucei in mice. II. Mechanisms in immune animals. Immunology. 1980;40(4):629–635. [PMC free article] [PubMed] [Google Scholar]
- 225.Dempsey WL, Mansfield JM. Lymphocyte function in experimental African trypanosomiasis. V. Role of antibody and the mononuclear phagocyte system in variant-specific immunity. Journal of Immunology. 1983;130(1):405–411. [PubMed] [Google Scholar]
- 226.Levine RF, Mansfield JM. Genetics of resistance to the African trypanosomes: III. Variant-specific antibody responses of H-2-compatible resistant and susceptible mice. Journal of Immunology. 1984;133(3):1564–1569. [PubMed] [Google Scholar]
- 227.Black SJ, Sendashonga CN, Webster P. Regulation of parasite-specific antibody responses in resistant (C57BL/6) and susceptible (C3H/He) mice infected with Trypanosoma (trypanozoon) brucei brucei. Parasite Immunology. 1986;8(5):425–442. doi: 10.1111/j.1365-3024.1986.tb00859.x. [DOI] [PubMed] [Google Scholar]
- 228.Mahan SM, Hendershot L, Black SJ. Control of trypanodestructive antibody responses and parasitemia in mice infected with Trypanosoma (Duttonella) vivax. Infection and Immunity. 1986;54(1):213–221. doi: 10.1128/iai.54.1.213-221.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.Lambert PH, Berney M, Kazyumba G. Immune complexes in serum and in cerebrospinal fluid in African trypanosomiasis. Correlation with polyclonal B cell activation and with intracerebral immunoglobulin synthesis. Journal of Clinical Investigation. 1981;67(1):77–85. doi: 10.1172/JCI110035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Askonas BA, Corsini AC, Clayton CE, Ogilvie BM. Functional depletion of T- and B-memory cells and other lymphoid cell subpopulations during trypanosomiasis. Immunology. 1979;36(2):313–321. [PMC free article] [PubMed] [Google Scholar]
- 231.Ellis JA, Scott JR, Machugh ND. Peripheral blood leucocytes subpopulation dynamics during Trypanosoma congolense infection in Boran and N’Dama cattle: an analysis using monoclonal antibodies and flow cytometry. Parasite Immunology. 1987;9(3):363–378. doi: 10.1111/j.1365-3024.1987.tb00514.x. [DOI] [PubMed] [Google Scholar]
- 232.Hudson KM, Terry RJ. Immunodepression and the course of infection of a chronic Trypanosoma brucei infection in mice. Parasite Immunology. 1979;1(4):317–326. doi: 10.1111/j.1365-3024.1979.tb00717.x. [DOI] [PubMed] [Google Scholar]
- 233.Magez S, Schwegmann A, Atkinson R, et al. The role of B-cells and IgM antibodies in parasitemia, anemia, and VSG switching in Trypanosoma brucei-infected mice. PLoS Pathogens. 2008;4(8) doi: 10.1371/journal.ppat.1000122. Article ID e1000122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Taylor KA. Immune responses of cattle to African trypanosomes: protective or pathogenic? International Journal for Parasitology. 1998;28(2):219–240. doi: 10.1016/s0020-7519(97)00154-9. [DOI] [PubMed] [Google Scholar]
- 235.Darji A, Lucas R, Magez S, et al. Mechanisms underlying trypanosome-elicited immunosuppression. Annales de la Societe Belge de Medecine Tropicale. 1992;72(supplement 1):27–38. [PubMed] [Google Scholar]
- 236.Flynn JN, Sileghem M. The role of the macrophage in induction of immunosuppression in Trypanosoma congolense-infected cattle. Immunology; 1991. pp. 310–316. [PMC free article] [PubMed] [Google Scholar]
- 237.Sileghem M, Darji A, De Baetselier P. In vitro simulation of immunosuppresion caused by Trypanosoma brucei. Immunology. 1991;73(2):246–248. [PMC free article] [PubMed] [Google Scholar]
- 238.Tabel H, Wei G, Shi M. T cells and immunopathogenesis of experimental African trypanosomiasis. Immunological Reviews. 2008;225(1):128–139. doi: 10.1111/j.1600-065X.2008.00675.x. [DOI] [PubMed] [Google Scholar]
- 239.Borowy NK, Sternberg JM, Schreiber D, Nonnengasser C, Overath P. Suppressive macrophages occurring in murine Trypanosoma brucei infection inhibit T-cell responses in vivo and in vitro. Parasite Immunology. 1990;12(3):233–246. doi: 10.1111/j.1365-3024.1990.tb00951.x. [DOI] [PubMed] [Google Scholar]
- 240.Sileghem M, Darji A, Hamers R, Van de Winkel M, De Baetselier P. Dual role of macrophages in the suppression of interleukin 2 production and interleukin 2 receptor expression in trypanosome-infected mice. European Journal of Immunology. 1989;19(5):829–835. doi: 10.1002/eji.1830190508. [DOI] [PubMed] [Google Scholar]
- 241.Schleifer KW, Mansfield JM. Suppressor macrophages in African trypanosomiasis inhibit T cell proliferative responses by nitric oxide and prostaglandins. Journal of Immunology. 1993;151(10):5492–5503. [PubMed] [Google Scholar]
- 242.Beschin A, Brys L, Magez S, Radwanska M, De Baetselier P. Trypanosoma brucei infection elicits nitric oxide-dependent and nitric oxide-independent suppressive mechanisms. Journal of Leukocyte Biology. 1998;63(4):429–439. doi: 10.1002/jlb.63.4.429. [DOI] [PubMed] [Google Scholar]
- 243.Mabbott NA, Coulson PS, Smythies LE, Wilson RA, Sternberg JM. African trypanosome infections in mice that lack the interferon-γ receptor gene: nitric oxide-dependent and -independent suppression of T-cell proliferative responses and the development of anaemia. Immunology. 1998;94(4):476–480. doi: 10.1046/j.1365-2567.1998.00541.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Darji A, Beschin A, Sileghem M, Heremans H, Brys L, De Baetselier P. In vitro simulation of immunosuppression caused by Trypanosoma brucei: active involvement of gamma interferon and tumor necrosis factor in the pathway of suppression. Infection and Immunity. 1996;64(6):1937–1943. doi: 10.1128/iai.64.6.1937-1943.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245.Namangala B, Brys L, Magez S, De Baetselier P, Beschin A. Trypanosoma brucei brucei infection impairs MHC class II antigen presentation capacity of macrophages. Parasite Immunology. 2000;22(7):361–370. doi: 10.1046/j.1365-3024.2000.00314.x. [DOI] [PubMed] [Google Scholar]
- 246.Darji A, Sileghem M, Heremans H, Brys L, De Baetselier P. Inhibition of T-cell responsiveness during experimental infections with Trypanosoma brucei: active involvement of endogenous gamma interferon. Infection and Immunity. 1993;61(7):3098–3102. doi: 10.1128/iai.61.7.3098-3102.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 247.Raes G, De Baetselier P, Noël W, Beschin A, Brombacher F, Gholamreza HG. Differential expression of FIZZ1 and Ym1 in alternatively versus classically activated macrophages. Journal of Leukocyte Biology. 2002;71(4):597–602. [PubMed] [Google Scholar]
- 248.Namangala B, De Baetselier P, Noël W, Brys L, Beschin A. Alternative versus classical macrophage activation during experimental African trypanosomosis. Journal of Leukocyte Biology. 2001;69(3):387–396. [PubMed] [Google Scholar]
- 249.Ding L, Linsley PS, Huang LY, Germain RN, Shevach EM. IL-10 inhibits macrophage costimulatory activity by selectively inhibiting the up-regulation of B7 expression. Journal of Immunology. 1993;151(3):1224–1234. [PubMed] [Google Scholar]
- 250.Guilliams M, Oldenhove G, Noel W, et al. African trypanosomiasis: naturally occurring regulatory T cells favor trypanotolerance by limiting pathology associated with sustained type 1 inflammation. Journal of Immunology. 2007;179(5):2748–2757. doi: 10.4049/jimmunol.179.5.2748. [DOI] [PubMed] [Google Scholar]
- 251.Guilliams M, Bosschaerts T, Hérin M, et al. Experimental expansion of the regulatory T cell population increases resistance to African trypanosomiasis. Journal of Infectious Diseases. 2008;198(5):781–791. doi: 10.1086/590439. [DOI] [PubMed] [Google Scholar]
- 252.Stijlemans B, Baral TN, Guilliams M, et al. A glycosylphosphatidylinositol-based treatment alleviates trypanosomiasis-associated immunopathology. Journal of Immunology. 2007;179(6):4003–4014. doi: 10.4049/jimmunol.179.6.4003. [DOI] [PubMed] [Google Scholar]
- 253.D’Ieteren GDM, Authié E, Wissocq N, Murray M. Trypanotolerance, an option for sustainable livestock production in areas at risk from trypanosomosis. OIE Revue Scientifique et Technique. 1998;17(1):154–175. doi: 10.20506/rst.17.1.1088. [DOI] [PubMed] [Google Scholar]
- 254.Naessens J. Bovine trypanotolerance: a natural ability to prevent severe anaemia and haemophagocytic syndrome? International Journal for Parasitology. 2006;36(5):521–528. doi: 10.1016/j.ijpara.2006.02.012. [DOI] [PubMed] [Google Scholar]
- 255.Stijlemans B, Vankrunkelsven A, Brys L, Magez S, De Baetselier P. Role of iron homeostasis in trypanosomiasis-associated anemia. Immunobiology. 2008;213(9-10):823–835. doi: 10.1016/j.imbio.2008.07.023. [DOI] [PubMed] [Google Scholar]
- 256.Akinbamijo OO, Bennison JJ, Jaitner J, Dempfle L. Haematological changes in N’Dama and Gobra Zebu bulls during Trypanosoma congolense infection maintained under a controlled feeding regimen. Acta Tropica. 1998;69(3):181–192. doi: 10.1016/s0001-706x(97)00123-x. [DOI] [PubMed] [Google Scholar]
- 257.Taylor KA, Mertens B. Immune Response of Cattle Infected with African trypanosomes. Memorias do Instituto Oswaldo Cruz. 1999;94(2):239–244. doi: 10.1590/s0074-02761999000200022. [DOI] [PubMed] [Google Scholar]
- 258.Magez S, Stijlemans B, Radwanska M, Pays E, Ferguson MAJ, De Baetselier P. The glycosyl-inositol-phosphate and dimyristoylglycerol moieties of the glycosylphosphatidylinositol anchor of the trypanosome variant-specific surface glycoprotein are distinct macrophage-activating factors. Journal of Immunology. 1998;160(4):1949–1956. [PubMed] [Google Scholar]
- 259.Magez S, Truyens C, Merimi M, et al. P75 tumor necrosis factor-receptor shedding occurs as a protective host response during African trypanosomiasis. Journal of Infectious Diseases. 2004;189(3):527–539. doi: 10.1086/381151. [DOI] [PubMed] [Google Scholar]
- 260.Okomo-Assoumou MC, Daulouede S, Lemesre J-L, N’Zila-Mouanda A, Vincendeau P. Correlation of high serum levels of tumor necrosis factor-α with disease severity in human African trypanosomiasis. American Journal of Tropical Medicine and Hygiene. 1995;53(5):539–543. doi: 10.4269/ajtmh.1995.53.539. [DOI] [PubMed] [Google Scholar]
- 261.Lejon V, Lardon J, Kenis G, et al. Interleukin (IL)-6, IL-8 and IL-10 in serum and CSF of Trypanosoma brucei gambiense sleeping sickness patients before and after treatment. Transactions of the Royal Society of Tropical Medicine and Hygiene. 2002;96(3):329–333. doi: 10.1016/s0035-9203(02)90115-x. [DOI] [PubMed] [Google Scholar]
- 262.MacLean L, Odiit M, Sternberg JM. Nitric oxide and cytokine synthesis in human African trypanosomiasis. Journal of Infectious Diseases. 2001;184(8):1086–1090. doi: 10.1086/323479. [DOI] [PubMed] [Google Scholar]
- 263.Hunter CA, Gow JW, Kennedy PGE, Jennings FW, Murray M. Immunopathology of experimental African sleeping sickness: detection of cytokine mRNA in the brains of Trypanosoma brucei brucei-infected mice. Infection and Immunity. 1991;59(12):4636–4640. doi: 10.1128/iai.59.12.4636-4640.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 264.Hunter CA, Kennedy PGE. Immunopathology in central nervous system human African trypanosomiasis. Journal of Neuroimmunology. 1992;36(2-3):91–95. doi: 10.1016/0165-5728(92)90040-r. [DOI] [PubMed] [Google Scholar]
- 265.Sileghem M, Flynn JN, Logan-Henfrey L, Ellis J. Tumour necrosis factor production by monocytes from cattle infected with Trypanosoma (Duttonella) vivax and Trypanosoma (Nannomonas) congolense: possible association with severity of anaemia associated with the disease. Parasite Immunology. 1994;16(1):51–54. doi: 10.1111/j.1365-3024.1994.tb00304.x. [DOI] [PubMed] [Google Scholar]
- 266.Simarro PP, Jannin J, Cattand P. Eliminating human African trypanosomiasis: where do we stand and what comes next? PLoS Medicine. 2008;5(2, article e55) doi: 10.1371/journal.pmed.0050055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 267.Bouyer J, Stachurski F, Gouro AS, Lancelot R. Control of bovine trypanosomosis by restricted application of insecticides to cattle using footbaths. Veterinary Parasitology. 2009;161(3-4):187–193. doi: 10.1016/j.vetpar.2009.01.018. [DOI] [PubMed] [Google Scholar]
- 268.Omolo MO, Hassanali A, Mpiana S, et al. Prospects for developing odour baits to control Glossina fuscipes spp., the major vector of human African trypanosomiasis. PLoS Neglected Tropical Diseases. 2009;3(5, article e435) doi: 10.1371/journal.pntd.0000435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 269.Read AF, Lynch PA, Thomas MB. How to make evolution-proof insecticides for malaria control. PLoS Biology. 2009;7(4, article e1000058) doi: 10.1371/journal.pbio.1000058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 270.Kennedy PGE. Human African trypanosomiasis of the CNS: current issues and challenges. Journal of Clinical Investigation. 2004;113(4):496–504. doi: 10.1172/JCI21052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 271.Fairlamb AH. Chemotherapy of human African trypanosomiasis: current and future prospects. Trends in Parasitology. 2003;19(11):488–494. doi: 10.1016/j.pt.2003.09.002. [DOI] [PubMed] [Google Scholar]
- 272.Legros D, Ollivier G, Gastellu-Etchegorry M, et al. Treatment of human African trypanosomiasis—present situation and needs for research and development. Lancet Infectious Diseases. 2002;2(7):437–440. doi: 10.1016/s1473-3099(02)00321-3. [DOI] [PubMed] [Google Scholar]
- 273.Burri C, Brun R. Eflornithine for the treatment of human African trypanosomiasis. Parasitology Research. 2003;90(supplement 1):S49–S52. doi: 10.1007/s00436-002-0766-5. [DOI] [PubMed] [Google Scholar]
- 274.Burri C, Nkunku S, Merolle A, Smith T, Blum J, Brun R. Efficacy of new, concise schedule for melarsoprol in treatment of sleeping sickness caused by Trypanosoma brucei gambiense: a randomised trial. The Lancet. 2000;355(9213):1419–1425. doi: 10.1016/S0140-6736(00)02141-3. [DOI] [PubMed] [Google Scholar]
- 275.Pepin J, Milord F. The treatment of human African trypanosomiasis. Advances in Parasitology. 1994;33:1–47. doi: 10.1016/s0065-308x(08)60410-8. [DOI] [PubMed] [Google Scholar]
- 276.Schmid C, Richer M, Bilenge CMM, et al. Effectiveness of a 10-day melarsoprol schedule for the treatment of late-stage human African trypanosomiasis: confirmation from a multinational study (IMPAMEL II) Journal of Infectious Diseases. 2005;191(11):1922–1931. doi: 10.1086/429929. [DOI] [PubMed] [Google Scholar]
- 277.Schmid C, Nkunku S, Merolle A, Vounatsou P, Burri C. Efficacy of 10-day melarsoprol schedule 2 years after treatment for late-stage gambiense sleeping sickness. The Lancet. 2004;364(9436):789–790. doi: 10.1016/S0140-6736(04)16940-7. [DOI] [PubMed] [Google Scholar]
- 278.Jennings FW, Rodgers J, Bradley B, Gettinby G, Kennedy PGE, Murray M. Human African trypanosomiasis: potential therapeutic benefits of an alternative suramin and melarsoprol regimen. Parasitology International. 2002;51(4):381–388. doi: 10.1016/s1383-5769(02)00044-2. [DOI] [PubMed] [Google Scholar]
- 279.Eisler M, Brandt J, Bauer B, et al. Standardised tests in mice and cattle for the detection of drug resistance in tsetse-transmitted trypanosomes of African domestic cattle. Veterinary Parasitology. 2001;97(3):171–182. doi: 10.1016/s0304-4017(01)00415-0. [DOI] [PubMed] [Google Scholar]
- 280.Geerts S, Holmes PH, Diall O, Eisler MC. African bovine trypanosomiasis: the problem of drug resistance. Trends in Parasitology. 2001;17(1):25–28. doi: 10.1016/s1471-4922(00)01827-4. [DOI] [PubMed] [Google Scholar]
- 281.Delespaux V, Geysen D, Majiwa PAO, Geerts S. Identification of a genetic marker for isometamidium chloride resistance in Trypanosoma congolense. International Journal for Parasitology. 2005;35(2):235–243. doi: 10.1016/j.ijpara.2004.11.009. [DOI] [PubMed] [Google Scholar]
- 282.Peregrine AS. Chemotherapy and delivery systems: haemoparasites. Veterinary Parasitology. 1994;54(1–3):223–248. doi: 10.1016/0304-4017(94)90092-2. [DOI] [PubMed] [Google Scholar]
- 283.Van den Bossche P, Doran M, Connor RJ. An analysis of trypanocidal drug use in the Eastern Province of Zambia. Acta Tropica. 2000;75(2):247–258. doi: 10.1016/s0001-706x(00)00059-0. [DOI] [PubMed] [Google Scholar]
- 284.Bacchi CJ. Resistance to clinical drugs in African trypanosomes. Parasitology Today. 1993;9(5):190–193. doi: 10.1016/0169-4758(93)90145-6. [DOI] [PubMed] [Google Scholar]
- 285.Mäser P, Lüscher A, Kaminsky R. Drug transport and drug resistance in African trypanosomes. Drug Resistance Updates. 2003;6(5):281–290. doi: 10.1016/j.drup.2003.09.001. [DOI] [PubMed] [Google Scholar]
- 286.Carter NS, Berger BJ, Fairlamb AH. Uptake of diamidine drugs by the P2 nucleoside transporter in melarsen-sensitive and-resistant Trypanosoma brucei brucei. The Journal of Biological Chemistry. 1995;270(47):28153–28157. doi: 10.1074/jbc.270.47.28153. [DOI] [PubMed] [Google Scholar]
- 287.Fairlamb AH, Carter NS, Cunningham M, Smith K. Characterisation of melarsen-resistant Trypanosoma brucei brucei with respect to cross-resistance to other drugs and trypanothione metabolism. Molecular and Biochemical Parasitology. 1992;53(1-2):213–222. doi: 10.1016/0166-6851(92)90023-d. [DOI] [PubMed] [Google Scholar]
- 288.Pospichal H, Brun R, Kaminsky R, Jenni L. Induction of resistance to melarsenoxide cysteamine (Mel Cy) in Trypanosoma brucei brucei. Acta Tropica. 1994;58(3-4):187–197. doi: 10.1016/0001-706x(94)90013-2. [DOI] [PubMed] [Google Scholar]
- 289.Matovu E, Stewart ML, Geiser F, et al. Mechanisms of arsenical and diamidine uptake and resistance in Trypanosoma brucei. Eukaryotic Cell. 2003;2(5):1003–1008. doi: 10.1128/EC.2.5.1003-1008.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 290.Alibu VP, Richter C, Voncken F, et al. The role of Trypanosoma brucei MRPA in melarsoprol susceptibility. Molecular and Biochemical Parasitology. 2006;146(1):38–44. doi: 10.1016/j.molbiopara.2005.10.016. [DOI] [PubMed] [Google Scholar]
- 291.Shahi SK, Krauth-Siegel RL, Clayton CE. Overexpression of the putative thiol conjugate transporter TbMRPA causes melarsoprol resistance in Trypanosoma brucei. Molecular Microbiology. 2002;43(5):1129–1138. doi: 10.1046/j.1365-2958.2002.02831.x. [DOI] [PubMed] [Google Scholar]
- 292.Witola WH, Tsuda A, Inoue N, Ohashi K, Onuma M. Acquired resistance to berenil in a cloned isolate of Trypanosoma evansi is associated with upregulation of a novel gene, TeDR40. Parasitology. 2005;131(5):635–646. doi: 10.1017/S003118200500836X. [DOI] [PubMed] [Google Scholar]
- 293.Stijlemans B, Conrath K, Cortez-Retamozo V, et al. Efficient targeting of conserved cryptic epitopes of infectious agents by single domain antibodies: African trypanosomes as paradigm. The Journal of Biological Chemistry. 2004;279(2):1256–1261. doi: 10.1074/jbc.M307341200. [DOI] [PubMed] [Google Scholar]
- 294.Authié E, Boulangé A, Muteti D, Lalmanach G, Gauthier F, Musoke AJ. Immunisation of cattle with cysteine proteinases of Trypanosoma congolense: targetting the disease rather than the parasite. International Journal for Parasitology. 2001;31(13):1429–1433. doi: 10.1016/s0020-7519(01)00266-1. [DOI] [PubMed] [Google Scholar]
- 295.Lubega GW, Byarugaba DK, Prichard RK. Immunization with a tubulin-rich preparation from Trypanosoma brucei confers broad protection against African trypanosomosis. Experimental Parasitology. 2002;102(1):9–22. doi: 10.1016/s0014-4894(02)00140-6. [DOI] [PubMed] [Google Scholar]
- 296.Li S-Q, Fung M-C, Reid SA, Inoue N, Lun Z-R. Immunization with recombinant beta-tubulin from Trypanosoma evansi induced protection against T. evansi, T. equiperdum and T. b. brucei infection in mice. Parasite Immunology. 2007;29(4):191–199. doi: 10.1111/j.1365-3024.2006.00933.x. [DOI] [PubMed] [Google Scholar]
- 297.Antoine-Moussiaux N, Büscher P, Desmecht D. Host-parasite interactions in trypanosomiasis: on the way to an antidisease strategy. Infection and Immunity. 2009;77(4):1276–1284. doi: 10.1128/IAI.01185-08. [DOI] [PMC free article] [PubMed] [Google Scholar]