Abstract
OBJECTIVE
To examine the efficacy and safety of rimonabant, a selective cannabinoid receptor type-1 antagonist, in patients with type 2 diabetes receiving insulin monotherapy.
RESEARCH DESIGN AND METHODS
Patients (n = 368; A1C ≥7%) were randomized to 20 mg/day rimonabant or placebo in this 48-week, double-blind, placebo-controlled multicenter trial. Change in baseline A1C to week 48 (primary outcome) and changes in body weight, waist circumference, and lipid levels (secondary outcomes) were assessed.
RESULTS
Rimonabant significantly reduced baseline A1C versus placebo (−0.89 vs. −0.24%; P < 0.0001), and significantly greater improvements were observed in cardiometabolic risk factors. More rimonabant patients achieved >10% reduction in mean total daily insulin dose versus placebo (P = 0.0012), and fewer required rescue medication (P < 0.0001). Hypoglycemia, nausea, dizziness, anxiety, and depression were more frequent with rimonabant.
CONCLUSIONS
Rimonabant improved glycemic control and cardiometabolic risk factors in patients with type 2 diabetes receiving insulin.
The increasing prevalence of obesity is contributing to a type 2 diabetes epidemic (1). Current standard of care of type 2 diabetes targets control of lipid and blood pressure levels, as well as glucose control (1–3). Most classes of antidiabetes agents are associated with weight gain, spurring research into therapeutic agents that improve both weight and glycemia, along with favorable effects on other comorbidities (4).
The endocannabinoid system contributes to energy homeostasis and lipid and glucose metabolism regulation (5). Treatment with the selective cannabinoid type 1 (CB1) receptor antagonist rimonabant improves multiple cardiometabolic risk factors in overweight/obese patients (6–10) as well as glycemic control in patients with drug-naïve type 2 diabetes (SERENADE study) (11) or disease suboptimally controlled on sulfonylurea/metformin (RIO-Diabetes study) (8).
The ARPEGGIO study evaluated once-daily 20 mg rimonabant on glycemic control in patients with type 2 diabetes inadequately controlled on insulin monotherapy (a population considered therapeutically challenging). It should be noted that the clinical development of rimonabant has stopped and the compound withdrawn from the market.
RESEARCH DESIGN AND METHODS
Eligible patients were aged ≥18 years, with type 2 diabetes, screening A1C ≥7%, and receiving insulin monotherapy for ≥3 months (≥30 units/day for ≥4 weeks). Type 1 diabetes was excluded with C-peptide <1.0 ng/dl. Patients with a history of depression and/or past/current antidepressant treatment were included. Institutional review boards and independent ethics committees at each center approved the protocol. The study was conducted in accordance with the Declaration of Helsinki. Patients provided written informed consent.
Following a 14-day screening period, patients were randomized (1:1; stratified by A1C level) to placebo or 20 mg rimonabant (oral, once daily before breakfast) for 48 weeks. Patients were advised to follow a controlled diet and increase physical activity. The total daily insulin dose (TDID) was to be maintained at a stable level (<10% increase/decrease of baseline dose). Use of rescue medication (increased insulin dose, oral medication) was at the investigator's discretion if the patient met certain criteria; such patients remained in the study.
Primary end point was the change from baseline to study end (day 336) in A1C. Secondary end points included changes in glycemic parameters (fasting plasma glucose, patients meeting A1C <7 and <6.5% targets, introduction of rescue medication, and change in mean TDID [MTDID]), proportion of patients with decreased TDID (>10% of baseline dose), lipid parameters (HDL cholesterol, triglycerides, LDL cholesterol, total cholesterol, and total-to-HDL cholesterol ratio), body weight, and waist circumference. In addition to standard safety assessments, a scripted neurological and psychiatric questionnaire was completed at each visit (see online appendix [available at http://care.diabetesjournals.org/cgi/content/full/dc09-0455/DC1]).
RESULTS
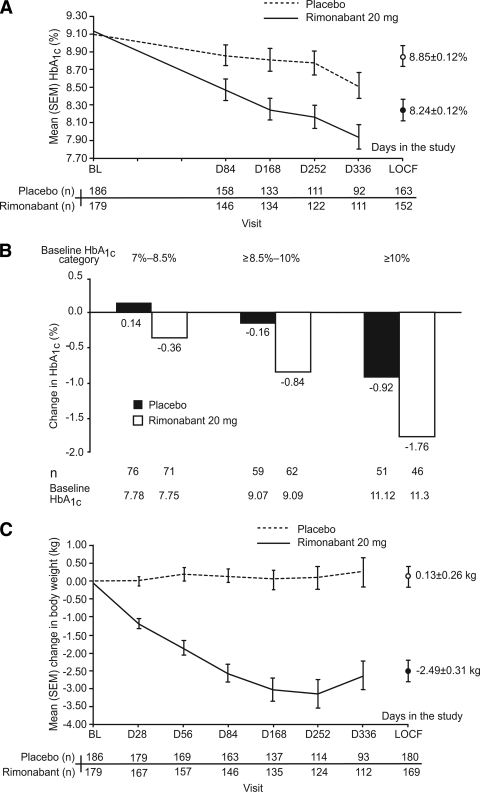
In total, 366 patients were randomized (n = 179 rimonabant; n = 187 placebo), and 284 completed treatment (134 [74.9%] rimonabant; 150 [80.2%] placebo). Demographic and baseline clinical characteristics were similar across groups (supplementary Table 1). At week 48, rimonabant produced significantly greater reductions in A1C and fasting plasma glucose levels versus placebo, and significantly more patients achieved target A1C levels (supplementary Table 2). A1C levels decreased continuously with rimonabant without plateau (Fig. 1A). Within both groups, higher baseline A1C levels correlated with greater absolute A1C reductions; between-group differences favored rimonabant within all baseline A1C categories (Fig. 1B). Rimonabant resulted in a significantly greater change in MTDID than placebo (mean difference −2.90; P = 0.0004), and more patients reduced MTDID by >10%. Significantly fewer patients receiving rimonabant required rescue medication. Days with at least one nonsymptomatic hypoglycemic event were greater with rimonabant than placebo (2.34 vs. 1.18 days, respectively; P = NS).
Figure 1.
A: Change in A1C over time. B: Change in A1C according to baseline level. C: change in body weight over time.
Significantly greater improvements with rimonabant versus placebo occurred in body weight (Fig. 1C), waist circumference, HDL cholesterol, triglycerides, and total-to-HDL cholesterol ratio. No between-group differences were observed for total or LDL cholesterol changes. The overall incidence of treatment-emergent adverse events (TEAEs) was similar between groups (supplementary Table 3); there were slightly fewer serious TEAEs with rimonabant versus placebo. TEAEs were mild to moderate in intensity, generally occurred early in treatment, and resolved without further corrective therapy. Metabolism/nutritional and psychiatric TEAEs occurred more frequently with rimonabant than placebo. Psychiatric disorders occurred in 31.3% of rimonabant-treated patients compared with 18.7% for placebo. Serious hypoglycemia was reported in four rimonabant patients versus two on placebo.
CONCLUSIONS
The ARPEGGIO study showed that 20 mg rimonabant significantly reduced A1C levels and improved multiple cardiometabolic risk factors in patients with type 2 diabetes receiving various types of insulin monotherapy, consistent with clinical trials of rimonabant in other type 2 diabetes populations (8,11).
Similar to previous studies and diabetes pharmacotherapy in general, A1C reductions were greatest in patients with highest baseline A1C (12). The ARPEGGIO study was characterized by a higher mean baseline A1C than patients in the SERENADE and RIO-Diabetes studies (8,11), and a larger fall in A1C may have been expected. The result may have been affected by weight loss (although significant was less in this study and may have been mitigated by larger increases in glysuria) and reduced insulin dosing in the rimonabant group secondary to hypoglycemia.
Weight loss was less than that observed in other studies (8,11). Weight gain is often associated with insulin therapy. In addition, patients in the ARPEGGIO study (mean diabetes duration 14 years) were characterized by advanced β-cell loss and variable insulin resistance. Rimonabant-induced weight loss occurs through central effects in reducing food intake and through peripheral metabolic pathways (altered substrate utilization, changes in adiponectin, and changes in energy expenditure) (13). This study showed that rimonabant facilitates weight loss in patients receiving insulin and may independently impact glucose, but the absolute change may be attenuated compared with nondiabetic patients or those receiving oral agents.
The safety profile of rimonabant was concordant with studies in similar patient populations (8,11). Patients with a history of psychiatric illness (depression/anxiety) were not excluded. Few serious psychiatric adverse events occurred (one case of suicidal ideation with rimonabant), consistent with previous studies involving patients with a psychiatric illness history (14). The ARPEGGIO study further supports the endocannabinoid system as a therapeutic target for overweight and glycemia. Further evaluation of this system is needed to achieve effective and well-tolerated agents.
Supplementary Material
Acknowledgments
This article was supported by sanofi-aventis.
P.A.H. has served on the advisory boards for sanofi-aventis, Pfizer, Novo Nordisk, and Merck; is a consultant for Orexigen, Sankyo, and Pfizer; is on the speakers bureau for Pfizer, Merck, and sanofi-aventis; has received research support only as part of multicenter clinical trials. A.A. has received honoraria from sanofi-aventis and has served on the advisory boards for MSD, Eli Lilly, Servier, and Novartis. L.E.L. has served on the advisory boards for sanofi-aventis, Roche, Pfizer, Eli Lilly, Novo Nordisk, and Novartis; is a board member for Eli Lilly and sanofi-aventis; has received research support from Roche, sanofi-aventis, and Novo Nordisk. U.C. is an employee of sanofi-aventis U.S. No other potential conflicts of interest relevant to this article were reported.
Footnotes
Clinical trial reg. no. NCT00288236, clinicaltrials.gov.
The costs of publication of this article were defrayed in part by the payment of page charges. This article must therefore be hereby marked “advertisement” in accordance with 18 U.S.C. Section 1734 solely to indicate this fact.
References
- 1. International Diabetes Federation. Clinical Guidelines Task Force: global guideline for type 2 diabetes: recommendations for standard, comprehensive, and minimal care. Diabet Med 2006;23:579–593 [DOI] [PubMed] [Google Scholar]
- 2. American Diabetes Association. Standards of medical care in diabetes, 2007. Diabetes Care 2007;30(Suppl. 1):S4–S41 [DOI] [PubMed] [Google Scholar]
- 3. Rodbard HW, Blonde L, Braithwaite SS, Brett EM, Cobin RH, Handelsman Y, Hellman R, Jellinger PS, Jovanovic LG, Levy P, Mechanick JI, Zangeneh F: American Association of Clinical Endocrinologists medical guidelines for clinical practice for the management of diabetes mellitus. Endocrine Practice 2007;13(Suppl. 1):3–68 [DOI] [PubMed] [Google Scholar]
- 4. Kelley DE, Bray GA, Pi-Sunyer FX, Klein S, Hill J, Miles J, Hollander P: Clinical efficacy of orlistat therapy in overweight and obese patients with insulin-treated type 2 diabetes. Diabetes Care 2002;25:1033–1041 [DOI] [PubMed] [Google Scholar]
- 5. Pagotto U, Marsicano G, Cota D, Lutz B, Pasquali R: The emerging role of the endocannabinoid system in endocrine regulation and energy balance. Endocr Rev 2006;27:73–100 [DOI] [PubMed] [Google Scholar]
- 6. Després JP, Van Gaal L, Golay A, Rissanen A: Rimonabant improves oral glucose tolerance in non-diabetic overweight/obese patients with/without comorbidities. 67th Scientific Sessions of the American Diabetes Association, 22–26 June 2007, Chicago, IL [abstract no. 345-OR] [Google Scholar]
- 7. Pi-Sunyer FX, Aronne LJ, Heshmati HM, Devin J, Rosenstock J: Effect of rimonabant, a cannabinoid-1 receptor blocker, on weight and cardiometabolic risk factors in overweight or obese patients: RIO-North America: a randomized controlled trial. JAMA 2006;295:761–775 [DOI] [PubMed] [Google Scholar]
- 8. Scheen AJ, Finer N, Hollander P, Jensen MD, Van Gaal LF: Efficacy and tolerability of rimonabant in overweight or obese patients with type 2 diabetes: a randomized controlled study. Lancet 2006;368:1660–1672 [DOI] [PubMed] [Google Scholar]
- 9. Van Gaal LF, Rissanen AM, Scheen AJ, Ziegler O, Rössner S: Effects of the cannabinoid-1 receptor blocker rimonabant on weight reduction and cardiovascular risk factors in overweight patients: 1-year experience from the RIO-Europe study. Lancet 2005;365:1389–1397 [DOI] [PubMed] [Google Scholar]
- 10. Van Gaal LF, Scheen AJ, Rissanen AM, Rössner S, Hanotin C, Ziegler O: the RIO-Europe Study Group. Long-term effect of CB1 blockade with rimonabant on cardiometabolic risk factors: 2-year results from the RIO-Europe Study. EHJ 2008;29:1761–1771 [DOI] [PubMed] [Google Scholar]
- 11. Rosenstock J, Hollander P, Chevalier S, Iranmanesh A: the SERENADE Study Group. The SERENADE Trial: effects of monotherapy with rimonabant, the first selective CB1 receptor antagonist, on glycemic control, body weight and lipid profile in drug-naïve type 2 diabetes. Diabetes Care 2008;31:2169–2176 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Bloomgarden ZT, Dodis R, Viscoli CM, Holmboe ES, Inzucchi SE: Lower baseline glycemia reduces apparent oral agent glucose-lowering efficacy: a meta-regression analysis. Diabetes Care 2006;29:2137–2139 [DOI] [PubMed] [Google Scholar]
- 13. Herling AW, Kilp S, Elvert R, Haschke G, Kramer W: Increased energy expenditure contributes more to the body weight-reducing effect of rimonabant than reduced food intake in candy-fed wistar rat. Endocrinology 2008;149:2557–2566 [DOI] [PubMed] [Google Scholar]
- 14. Christensen R, Kristensen PK, Bartels EM, Bliddal H, Astrup A: Efficacy and safety of the weight-loss drug rimonabant: a meta-analysis of randomized trials. Lancet 2007;370:1706–1713 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.