Abstract
Galanin coexists with acetylcholine in medial septal neurons projecting to the ventral hippocampus, a projection thought to modulate memory functions. Neurochemical lesions of the nucleus basalis-medial septal area in rats impaired choice accuracy on a delayed alternation t-maze task. Acetylcholine (7.5 or 10 micrograms intraventricularly or 1 micrograms micro-injected into the ventral hippocampus) significantly improved performance in the lesioned rats. Atropine (5 mg/kg intraperitoneally or 10 micrograms intraventricularly), but not mecamylamine (3 mg/kg intraperitoneally or 20 micrograms intraventricularly), blocked this action of acetylcholine, suggesting involvement of a muscarinic receptor. Galanin (100-500 ng intraventricularly or 200 ng into the ventral hippocampus) attenuated the ability of acetylcholine to reverse the deficit in working memory in the lesioned rats. The antagonistic interaction between galanin and acetylcholine suggests that endogenous galanin may inhibit cholinergic function in memory processes, particularly in pathologies such as Alzheimer disease that involve degeneration of basal forebrain neurons.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aigner T. G., Mitchell S. J., Aggleton J. P., DeLong M. R., Struble R. G., Price D. L., Wenk G. L., Mishkin M. Effects of scopolamine and physostigmine on recognition memory in monkeys with ibotenic-acid lesions of the nucleus basalis of Meynert. Psychopharmacology (Berl) 1987;92(3):292–300. doi: 10.1007/BF00210833. [DOI] [PubMed] [Google Scholar]
- Bartus R. T., Flicker C., Dean R. L., Fisher S., Pontecorvo M., Figueiredo J. Behavioral and biochemical effects of nucleus basalis magnocellularis lesions: implications and possible relevance to understanding or treating Alzheimer's disease. Prog Brain Res. 1986;70:345–361. doi: 10.1016/s0079-6123(08)64315-0. [DOI] [PubMed] [Google Scholar]
- Berrettini W. H., Kaye W. H., Sunderland T., May C., Gwirtsman H. E., Mellow A., Albright A. Galanin immunoreactivity in human CSF: studies in eating disorders and Alzheimer's disease. Neuropsychobiology. 1988;19(2):64–68. doi: 10.1159/000118436. [DOI] [PubMed] [Google Scholar]
- Ch'ng J. L., Christofides N. D., Anand P., Gibson S. J., Allen Y. S., Su H. C., Tatemoto K., Morrison J. F., Polak J. M., Bloom S. R. Distribution of galanin immunoreactivity in the central nervous system and the responses of galanin-containing neuronal pathways to injury. Neuroscience. 1985 Oct;16(2):343–354. doi: 10.1016/0306-4522(85)90007-7. [DOI] [PubMed] [Google Scholar]
- Chan-Palay V. Galanin hyperinnervates surviving neurons of the human basal nucleus of Meynert in dementias of Alzheimer's and Parkinson's disease: a hypothesis for the role of galanin in accentuating cholinergic dysfunction in dementia. J Comp Neurol. 1988 Jul 22;273(4):543–557. doi: 10.1002/cne.902730409. [DOI] [PubMed] [Google Scholar]
- Crawley J. N., Stivers J. A., Blumstein L. K., Paul S. M. Cholecystokinin potentiates dopamine-mediated behaviors: evidence for modulation specific to a site of coexistence. J Neurosci. 1985 Aug;5(8):1972–1983. doi: 10.1523/JNEUROSCI.05-08-01972.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dunnett S. B., Whishaw I. Q., Jones G. H., Bunch S. T. Behavioural, biochemical and histochemical effects of different neurotoxic amino acids injected into nucleus basalis magnocellularis of rats. Neuroscience. 1987 Feb;20(2):653–669. doi: 10.1016/0306-4522(87)90117-5. [DOI] [PubMed] [Google Scholar]
- Fisone G., Wu C. F., Consolo S., Nordström O., Brynne N., Bartfai T., Melander T., Hökfelt T. Galanin inhibits acetylcholine release in the ventral hippocampus of the rat: histochemical, autoradiographic, in vivo, and in vitro studies. Proc Natl Acad Sci U S A. 1987 Oct;84(20):7339–7343. doi: 10.1073/pnas.84.20.7339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flood J. F., Smith G. E., Cherkin A. Memory retention: potentiation of cholinergic drug combinations in mice. Neurobiol Aging. 1983 Spring;4(1):37–43. doi: 10.1016/0197-4580(83)90052-0. [DOI] [PubMed] [Google Scholar]
- Gage F. H., Björklund A. Enhanced graft survival in the hippocampus following selective denervation. Neuroscience. 1986;17(1):89–98. doi: 10.1016/0306-4522(86)90227-7. [DOI] [PubMed] [Google Scholar]
- Hagan J. J., Salamone J. D., Simpson J., Iversen S. D., Morris R. G. Place navigation in rats is impaired by lesions of medial septum and diagonal band but not nucleus basalis magnocellularis. Behav Brain Res. 1988 Jan;27(1):9–20. doi: 10.1016/0166-4328(88)90105-2. [DOI] [PubMed] [Google Scholar]
- Haroutunian V., Barnes E., Davis K. L. Cholinergic modulation of memory in rats. Psychopharmacology (Berl) 1985;87(3):266–271. doi: 10.1007/BF00432705. [DOI] [PubMed] [Google Scholar]
- Hepler D. J., Olton D. S., Wenk G. L., Coyle J. T. Lesions in nucleus basalis magnocellularis and medial septal area of rats produce qualitatively similar memory impairments. J Neurosci. 1985 Apr;5(4):866–873. doi: 10.1523/JNEUROSCI.05-04-00866.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hepler D. J., Wenk G. L., Cribbs B. L., Olton D. S., Coyle J. T. Memory impairments following basal forebrain lesions. Brain Res. 1985 Oct 28;346(1):8–14. doi: 10.1016/0006-8993(85)91088-1. [DOI] [PubMed] [Google Scholar]
- Hökfelt T., Millhorn D., Seroogy K., Tsuruo Y., Ceccatelli S., Lindh B., Meister B., Melander T., Schalling M., Bartfai T. Coexistence of peptides with classical neurotransmitters. Experientia. 1987 Jul 15;43(7):768–780. doi: 10.1007/BF01945354. [DOI] [PubMed] [Google Scholar]
- Kesner R. P., Crutcher K. A., Measom M. O. Medial septal and nucleus basalis magnocellularis lesions produce order memory deficits in rats which mimic symptomatology of Alzheimer's disease. Neurobiol Aging. 1986 Jul-Aug;7(4):287–295. doi: 10.1016/0197-4580(86)90009-6. [DOI] [PubMed] [Google Scholar]
- Kyrkouli S. E., Stanley B. G., Leibowitz S. F. Galanin: stimulation of feeding induced by medial hypothalamic injection of this novel peptide. Eur J Pharmacol. 1986 Mar 11;122(1):159–160. doi: 10.1016/0014-2999(86)90175-5. [DOI] [PubMed] [Google Scholar]
- Mastropaolo J., Crawley J. N. Behavioral evidence for increased acetylcholine receptor sensitivity after nucleus basalis magnocellularis lesions in the rat. Eur J Pharmacol. 1988 Aug 24;153(2-3):301–304. doi: 10.1016/0014-2999(88)90619-x. [DOI] [PubMed] [Google Scholar]
- Melander T., Hökfelt T., Rökaeus A. Distribution of galaninlike immunoreactivity in the rat central nervous system. J Comp Neurol. 1986 Jun 22;248(4):475–517. doi: 10.1002/cne.902480404. [DOI] [PubMed] [Google Scholar]
- Melander T., Staines W. A. A galanin-like peptide coexists in putative cholinergic somata of the septum-basal forebrain complex and in acetylcholinesterase-containing fibers and varicosities within the hippocampus in the owl monkey (Aotus trivirgatus). Neurosci Lett. 1986 Jul 11;68(1):17–22. doi: 10.1016/0304-3940(86)90222-3. [DOI] [PubMed] [Google Scholar]
- Melander T., Staines W. A., Hökfelt T., Rökaeus A., Eckenstein F., Salvaterra P. M., Wainer B. H. Galanin-like immunoreactivity in cholinergic neurons of the septum-basal forebrain complex projecting to the hippocampus of the rat. Brain Res. 1985 Dec 23;360(1-2):130–138. doi: 10.1016/0006-8993(85)91228-4. [DOI] [PubMed] [Google Scholar]
- Melander T., Staines W. A., Rökaeus A. Galanin-like immunoreactivity in hippocampal afferents in the rat, with special reference to cholinergic and noradrenergic inputs. Neuroscience. 1986 Sep;19(1):223–240. doi: 10.1016/0306-4522(86)90017-5. [DOI] [PubMed] [Google Scholar]
- Miyamoto M., Shintani M., Nagaoka A., Nagawa Y. Lesioning of the rat basal forebrain leads to memory impairments in passive and active avoidance tasks. Brain Res. 1985 Feb 25;328(1):97–104. doi: 10.1016/0006-8993(85)91327-7. [DOI] [PubMed] [Google Scholar]
- Murray C. L., Fibiger H. C. Learning and memory deficits after lesions of the nucleus basalis magnocellularis: reversal by physostigmine. Neuroscience. 1985 Apr;14(4):1025–1032. doi: 10.1016/0306-4522(85)90273-8. [DOI] [PubMed] [Google Scholar]
- Murray C. L., Fibiger H. C. Pilocarpine and physostigmine attenuate spatial memory impairments produced by lesions of the nucleus basalis magnocellularis. Behav Neurosci. 1986 Feb;100(1):23–32. doi: 10.1037//0735-7044.100.1.23. [DOI] [PubMed] [Google Scholar]
- Palazzi E., Fisone G., Hökfelt T., Bartfai T., Consolo S. Galanin inhibits the muscarinic stimulation of phosphoinositide turnover in rat ventral hippocampus. Eur J Pharmacol. 1988 Apr 13;148(3):479–480. doi: 10.1016/0014-2999(88)90133-1. [DOI] [PubMed] [Google Scholar]
- Palkovits M., Rökaeus A., Antoni F. A., Kiss A. Galanin in the hypothalamo-hypophyseal system. Neuroendocrinology. 1987 Nov;46(5):417–423. doi: 10.1159/000124855. [DOI] [PubMed] [Google Scholar]
- Salamone J. D., Beart P. M., Alpert J. E., Iversen S. D. Impairment in T-maze reinforced alternation performance following nucleus basalis magnocellularis lesions in rats. Behav Brain Res. 1984 Jul;13(1):63–70. doi: 10.1016/0166-4328(84)90030-5. [DOI] [PubMed] [Google Scholar]
- Skofitsch G., Jacobowitz D. M. Immunohistochemical mapping of galanin-like neurons in the rat central nervous system. Peptides. 1985 May-Jun;6(3):509–546. doi: 10.1016/0196-9781(85)90118-4. [DOI] [PubMed] [Google Scholar]
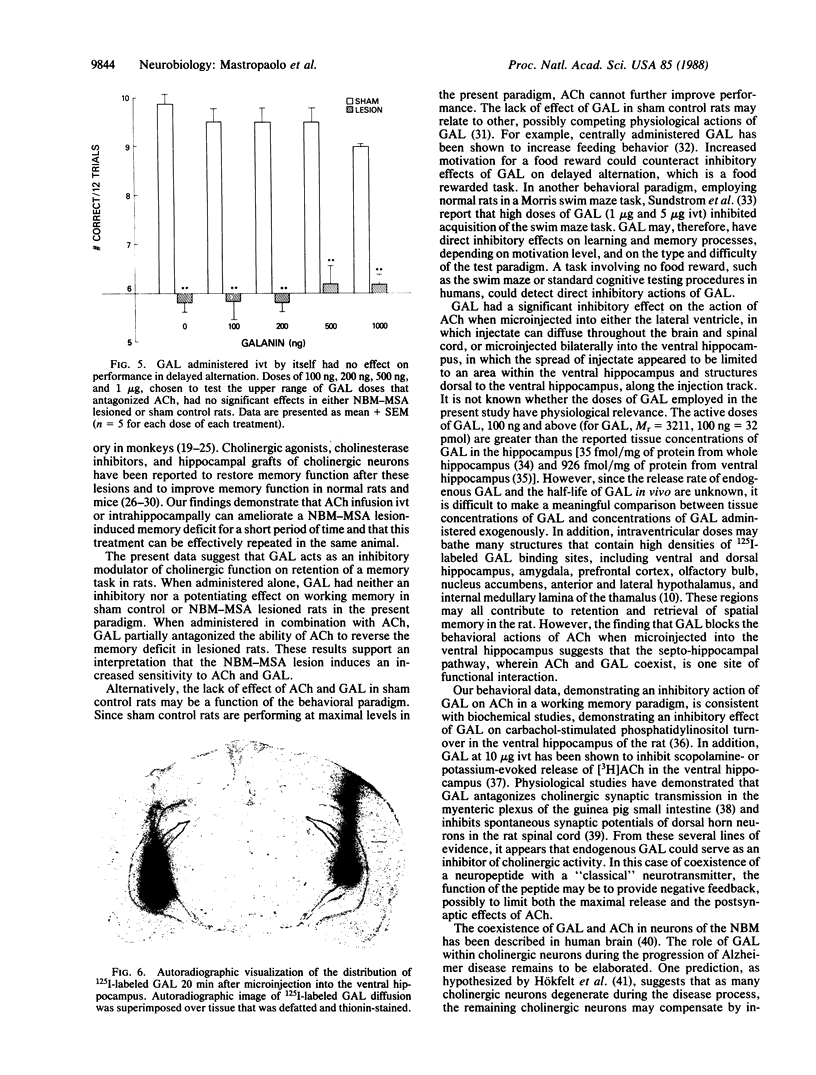
- Skofitsch G., Jacobowitz D. M. Quantitative distribution of galanin-like immunoreactivity in the rat central nervous system. Peptides. 1986 Jul-Aug;7(4):609–613. doi: 10.1016/0196-9781(86)90035-5. [DOI] [PubMed] [Google Scholar]
- Skofitsch G., Sills M. A., Jacobowitz D. M. Autoradiographic distribution of 125I-galanin binding sites in the rat central nervous system. Peptides. 1986 Nov-Dec;7(6):1029–1042. doi: 10.1016/0196-9781(86)90133-6. [DOI] [PubMed] [Google Scholar]
- Sundström E., Archer T., Melander T., Hökfelt T. Galanin impairs acquisition but not retrieval of spatial memory in rats studied in the Morris swim maze. Neurosci Lett. 1988 Jun 7;88(3):331–335. doi: 10.1016/0304-3940(88)90233-9. [DOI] [PubMed] [Google Scholar]
- Tamura K., Palmer J. M., Wood J. D. Galanin suppresses nicotinic synaptic transmission in the myenteric plexus of guinea-pig small intestine. Eur J Pharmacol. 1987 Apr 29;136(3):445–446. doi: 10.1016/0014-2999(87)90323-2. [DOI] [PubMed] [Google Scholar]
- Tatemoto K., Rökaeus A., Jörnvall H., McDonald T. J., Mutt V. Galanin - a novel biologically active peptide from porcine intestine. FEBS Lett. 1983 Nov 28;164(1):124–128. doi: 10.1016/0014-5793(83)80033-7. [DOI] [PubMed] [Google Scholar]
- Wenk G. L., Rökaeus A. Basal forebrain lesions differentially alter galanin levels and acetylcholinergic receptors in the hippocampus and neocortex. Brain Res. 1988 Sep 13;460(1):17–21. doi: 10.1016/0006-8993(88)90425-8. [DOI] [PubMed] [Google Scholar]
- Wilson S. H., Schrier B. K., Farber J. L., Thompson E. J., Rosenberg R. N., Blume A. J., Nirenberg M. W. Markers for gene expression in cultured cells from the nervous system. J Biol Chem. 1972 May 25;247(10):3159–3169. [PubMed] [Google Scholar]