Abstract
Decoy receptors, such as the human tumor necrosis factor receptor (TNFR), are potential new therapies for brain disorders. However, decoy receptors are large molecule drugs that are not transported across the blood-brain barrier (BBB). To enable BBB transport of a TNFR decoy receptor, the human TNFR-II extracellular domain was re-engineered as a fusion protein with a chimeric monoclonal antibody (MAb) against the human insulin receptor (HIR). The HIRMAb acts as a molecular Trojan horse to ferry the TNFR therapeutic decoy receptor across the BBB. The HIRMAb-TNFR fusion protein was expressed in stably transfected CHO cells, and was analyzed with electrophoresis, Western blotting, size exclusion chromatography, and binding assays for the HIR and TNFα. The HIRMAb-TNFR fusion protein was radiolabeled by trititation, in parallel with the radio-iodination of recombinant TNFR:Fc fusion protein, and the proteins were co-injected in the adult Rhesus monkey. The TNFR:Fc fusion protein did not cross the primate BBB in vivo, but the uptake of the HIRMAb-TNFR fusion protein was high and 3% of the injected dose was taken up by the primate brain. The TNFR was selectively targeted to brain, relative to peripheral organs, following fusion to the HIRMAb. This study demonstrates that decoy receptors may be re-engineered as IgG fusion proteins with a BBB molecular Trojan horse that selectively targets the brain, and enables penetration of the BBB in vivo. IgG-decoy receptor fusion proteins represent a new class of human neurotherapeutics.
Keywords: blood-brain barrier, delivery systems, decoy receptor, tumor necrosis factor-alpha
1. Introduction
The pathologic effects of the cytotoxic cytokine, tumor necrosis factor (TNF)-α, are suppressed by the administration of the extracellular domain (ECD) of the tumor necrosis factor receptor (TNFR) type II, which is fused to the amino terminus of the Fc region of human IgG1 (Peppel, et al, 1991). This fusion protein, designated TNFR:Fc, sequesters TNFα, thereby blocking the effects of the cytokine. The TNFR decoy receptor is a FDA approved biopharmaceutical used to suppress inflammatory reactions in non-brain tissues (Valesini et al, 2007).
TNFα also plays a pathologic role in disorders of the central nervous system (CNS), including acute disorders such as stroke (Nawashiro et al, 1997), traumatic brain injury (Knoblach et al, 1999), or spinal cord injury (Marchand et al, 2009), and in chronic brain conditions such as neurodegeneration (Tweedie et al, 2007) or depression (Himmerich et al, 2008). However, the TNFR decoy receptor cannot be developed as a pharmaceutical for the brain, because this large molecule drug does not cross the blood-brain barrier (BBB). Large molecule drugs can penetrate the BBB and be developed as new drugs for the brain following the re-engineering of the drug as a fusion protein with a BBB molecular Trojan horse (Pardridge, 2008). The most active BBB Trojan horse is a monoclonal antibody (MAb) against the human insulin receptor (HIR) (Pardridge et al, 1995), and the HIRMAb has been genetically engineered for administration in humans (Boado et al, 2007a). Prior work described the engineering of a fusion protein of the HIRMAb and the ECD of the human TNFR-II, and this fusion protein is designated the HIRMAb-TNFR fusion protein (Hui et al, 2009). The ECD of the human TNFR-II decoy receptor was fused to the carboxyl terminus of the CH3 region of the heavy chain of the HIRMAb. The fusion protein was expressed transiently in COS cells, and the bi-functionality of the COS-derived HIRMAb-TNFR fusion protein was demonstrated with in vitro assays measuring fusion protein binding both to the HIR and to human TNFα.
The purpose of this investigation was to confirm the hypothesis that (a) the TNFR decoy receptor does not cross the BBB, and (b) that the decoy receptor could penetrate the brain in the form of the HIRMAb-TNFR fusion protein. The brain uptake study was performed in the adult Rhesus monkey, since the HIRMAb cross reacts with the insulin receptor of Old World primates, but does not recognize the insulin receptor of lower species (Pardridge et al, 1995). To execute these studies, a stably transfected Chinese hamster ovary (CHO) line producing the HIRMAb-TNFR in serum free medium was engineered. The results demonstrate that the TNFR:Fc fusion protein does not cross the BBB, and that there is a marked increase in brain penetration of the TNFR following fusion to the BBB molecular Trojan horse. The uptake of the fusion proteins in non-brain organs in the primate is also measured, which shows that fusion of the decoy receptor to the HIRMAb results in a selective targeting of the pharmaceutical to the brain.
2. Materials and Methods
2.1 Engineering of tandem vector and production of CHO line
The cDNA encoding the human TNFR ECD was fused to the 3′ end of the cDNA encoding the heavy chain (HC) of the chimeric HIRMAb, using methods described previously (Boado et al, 2007b). A tandem vector (TV) was engineered in which the expression cassettes encoding this fusion HC, as well as the HIRMAb light chain (LC), and the murine DHFR, are all contained on a single strand of DNA (Boado et al, 2007b). The sequence of the TV was confirmed by bi-directional DNA sequencing performed at Eurofins MWG Operon (Huntsville, AL) using custom sequencing oligodeoxynucleotides synthesized at Midland Certified Reagent Co. (Midland, TX). The TV was linearized and DG44 CHO cells were electroporated, followed by selection in hypoxanthine-thymine deficient medium and amplification with graded increases in methotrexate (MTX) up to 80 nM in serum free medium (SFM). The CHO line underwent 2 successive rounds of 1 cell/well dilutional cloning, and positive clones were selected by measurement of medium human IgG concentrations by enzyme-linked immunosorbent assay (ELISA). The CHO line was stable through multiple generations, and produced medium IgG levels of 10-20 mg/L in shake flasks at a cell density of 1-2 million cells/mL.
2.2 Protein A chromatography
The CHO cells were propagated in 1 L bottles, until 2.4L of conditioned SFM was collected. The medium was ultra-filtered with a 0.2 um Sartopore-2 sterile-filter unit (Sartorius Stedim Biotech, Goettingen, Germany), and applied to a 25 mL protein A Sepharose 4 Fast Flow (GE Life Sciences, Chicago, IL) column equilibrated in 25 mM Tris/25 mM NaCl/5 mM EDTA/pH=7.1. Following application of the sample, the column was washed with 25 mM Tris/1 M NaCl/5 mM EDTA/pH=7.1, and the fusion protein was eluted with 0.1 M sodium acetate/pH=3.7. The acid eluate was pooled, Tris was added to 0.05 M, NaCl was added to 0.15 M, the pH was increased to pH=6.5, and the solution was stored sterile-filtered at 4C.
2.3 Analytical assays
The homogeneity of the fusion proteins was evaluated with sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) under reducing conditions using a 10% Tris gel (Ready Gel, Biorad, Richmond, CA). Western blot analysis was performed with a goat anti-human IgG (H+L) antibody (Vector Labs, Burlingame, CA), and a mouse monoclonal antibody against human TNFR-II (Santa Cruz Biotechnology, Santa Cruz, CA). Size exclusion chromatography (SEC) high performance liquid chromatography (HPLC) of the protein A purified HIRMAb-TNFR fusion protein was performed with two 7.8 mm × 30 cm TSK-GEL G3000SWXL columns (Tosoh Bioscience, Tokyo, Japan) in series, under isocratic conditions at a flow rate of 0.5 ml/min with Perkin-Elmer Series 200 pump. The absorbance at 280 nm was detected with a Shimadzu SPD-10A UV-VIS detector and a Shimadzu CR-8 chart recorder. The elution of molecular weight (MW) standards (GE Healthcare, Buckinghamshire, UK), blue dextran-2000, aldolase, and ovalbumin was measured under the same elution conditions.
2.4 Potency assays
HIR binding ELISA
The binding of the HIRMAb-TNFR fusion protein to the extracellular domain (ECD) of the HIR was determined by ELISA using CHO-derived HIR ECD affinity purified by lectin chromatography, as described previously (Coloma et al, 2000). The capture reagent is the HIR ECD and the detection reagent is a biotinylated goat anti-human IgG (H+L) antibody from Vector Labs.
TNFα radio-receptor assay
The saturable binding of human TNFα to the HIRMAb-TNFR fusion protein was determined with a radio-receptor assay (RRA) described previously (Hui et al, 2009). The capture reagent is a mouse anti-human IgG1 Fc antibody (Invitrogen, Carlsbad, CA), and the detection reagent was [125I]-TNFα (Perkin Elmer, Boston, MA). The half-saturation constant, ED50, of TNFα binding to the HIRMAb-TNFR fusion protein was determined by non-linear regression analysis using the BMDP2007e software.
2.5 Human cell bio-assay
The suppression of TNFα cytotoxic activity in human WEHI-13VAR cells (CRL-2148, American Type Culture Collection, Manassas, VA) by 1 nM concentrations of either the HIRMAb-TNFR fusion protein or the TNFR:Fc fusion protein was determined with a bio-assay, as described previously (Hui et al, 2009). TNFα (1 to 100 pg/Ml) causes cytotoxicity in these cells following treatment with 1 ug/mL of actinomycin D. Cell viability was measured with thiazolyl blue tetrazolium bromide (Sigma Chemical Co., St. Louis, MO).
2.6 Radio-labeling of proteins
[125I]-Bolton-Hunter reagent was purchased from American Radiolabeled Chemicals (St. Louis, MO). The TNFR:Fc fusion protein (#726-R2) was purchased from R&D Systems (Minneapolis, MN), and shown to be homogenous by SDS-PAGE. The TNFR:Fc was radio-labeled with fresh Bolton-Hunter reagent to a specific activity of 11.5 uCi/ug and a trichloroacetic acid (TCA) precipitability of >99% following purification with a 1.0×28 cm column of Sephadex G-25 and elution with 0.01 M NaH2PO4/0.15 M NaCl/pH=7.4/0.05% Tween-20 (PBST). The TCA precipitation of the labeled TNFR:Fc fusion protein remained >99% at 24 hours after iodination, and the TNFR:Fc fusion protein was administered to the primate within 24 hrs of radio-labeling. [3H]-Nsuccinimidyl propionate (NSP) was purchased from American Radiolabeled Chemicals. The HIRMAb-TNFR fusion protein was radio-labeled with fresh NSP to a specific activity of 3.0 uCi/ug and a TCA precipitability of 95% following purification with a 1.0×28 cm column of Sephadex G-25 and elution with 0.02 M Tris/0.15 M NaCl/pH=6.5 (TBS). The solution was buffer exchanged with TBS and an Ultra-15 microconcentrator (Millipore, Bedford, MA), which increased the TCA precipitability to 99%. The 3H-labeled HIRMAbTNFR fusion protein was labeled in advance of the primate study and stored at -70C.
2.7 Primate injection study and capillary depletion study
An adult female Rhesus monkey, 4.1 kg, was obtained from Covance (Alice, TX). The animal was injected intravenously (IV) with 1806 uCi of [3H]-HIRMAb-TNFR fusion protein, 428 uCi of [125I]-TNFR:Fc fusion protein in 3.1 mL of TBS by bolus injection over 30 seconds in the left femoral vein. The dose of HIRMAb-TNFR fusion protein was 0.15 mg/kg. The animal was initially anesthetized with intramuscular ketamine, and anesthesia was maintained by 1% isoflurane by inhalation. All procedures were carried out in accordance with the Guide for the Care and Use of Laboratory Animals as adopted and promulgated by the U.S. National Institutes of Health. Following intravenous drug administration, femoral venous plasma was obtained at 1, 2.5, 5, 15, 30, 60, and 120 min for determination of 3H and 125I radioactivity. The animal was euthanized, and samples of major organs (heart, liver, spleen, lung, skeletal muscle, and omental fat) were removed, weighed, and processed for determination of radioactivity. The cranium was opened and the brain was removed. Samples of frontal cortical gray matter, frontal cortical white matter, cerebellar gray matter, and cerebellar white matter were removed for radioactivity determination.
Samples (~2 gram) of frontal cortex were removed for capillary depletion analysis, as described previously (Triguero et al, 1990). The brain was homogenized in 8 mL cold PBS in a tissue grinder. The homogenate was supplemented with 9.4 mL cold 40% dextran (70 kDa, Sigma Chemical Co.), and an aliquot of the homogenate was taken for radioactivity measurement. The homogenate was centrifuged at 3200 g at 4C for 10 min in a fixed angle rotor. The brain microvasculature quantitatively sediments as the pellet (Triguero et al, 1990), and the post-vascular supernatant is a measure of capillary depleted brain parenchyma. The vascular pellet and supernatant were counted for 3H and 125I radioactivity in parallel with the homogenate. The volume of distribution (VD) was determined for each of the 3 fractions from the ratio of total 125I or 3H radioactivity in the fraction divided by the total 125I or 3H radioactivity in the 120 min terminal plasma.
Plasma and tissue samples were analyzed for 125I radioactivity with a gamma counter (Wizard 1470, Perkin Elmer), and were analyzed for 3H radioactivity with a liquid scintillation counter (Tricarb 2100TR, Perkin Elmer, Downers Grove, IL). The 125I isotope emits radiation that is detected in the 3H channel (0-12 keV) of the liquid scintillation counter (LSC). Therefore, quench curves were produced using chloroform as the quench agent, to compute the efficiency of counting of 125I in the 3H window, as described previously (Boado and Pardridge, 2009a). All samples for 3H counting were solubilized in Soluene-350 and counted in the LSC in Opti-Fluor O (Perkin Elmer).
2.8 Pharmacokinetics and organ PS product
The 3H or 125I radioactivity in plasma, DPM/mL, was converted to % injected dose (ID)/mL, and the %ID/mL was fit to a bi-exponential equation,
The intercepts (A1, A2) and the slopes (k1, k2) were used to compute the median residence time (MRT), the central volume of distribution (Vc), the steady state volume of distribution (Vss), the area under the plasma concentration curve (AUC), and the systemic clearance (CL), as described previously (Pardridge, et al, 1995). Non-linear regression analysis used the AR subroutine of the BMDP Statistical Software (Statistical Solutions Ltd, Cork, Ireland). Data were weighted by 1/(%ID/mL)2.
The organ clearance (μ-L/min/g), also called the permeability-surface area (PS) product, is computed from the terminal organ uptake (%ID/g) and the 120 min plasma AUC (%IDmin/mL) as follows:
3. Results
A tandem vector was engineered, which contained the expression cassettes for the heavy chain fusion gene, the light chain gene, and the DHFR gene on a single strand of DNA. The 3 expression cassettes spanned 9,237 nucleotides (nt). The light chain was comprised of 234 amino acids (AA), which included a 20 AA signal peptide, a 108 AA variable region of the light chain of the HIRMAb light chain, and a 106 AA human kappa light chain constant (C)-region. The predicted molecular weight of the light chain is 23,398 Da with a predicted isoelectric point (pI) of 5.45.The fusion protein of the HIRMAb heavy chain and the TNFR ECD was comprised of 699 AA, which included a 19 AA signal peptide, a 113 AA variable region of the heavy chain of the HIRMAb, a 330 AA human IgG1 C-region, a 2 AA linker (Ser-Ser), and a 235 AA TNFR ECD, which corresponded to Leu23-Asp257 of the human TNFR-II (Genbank NP_001057). The predicted molecular weight of the fusion heavy chain, without glycosylation, is 73,900 Da with a predicted pI of 8.45. Predicted N-linked glycosylation sequences were present at a single site in the human IgG1 C-region, and at 2 sites within the TNFR ECD.
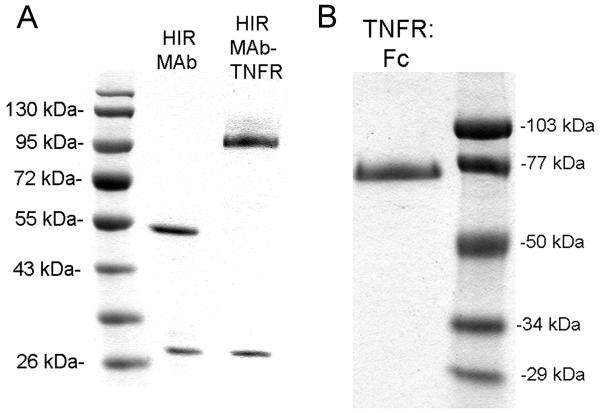
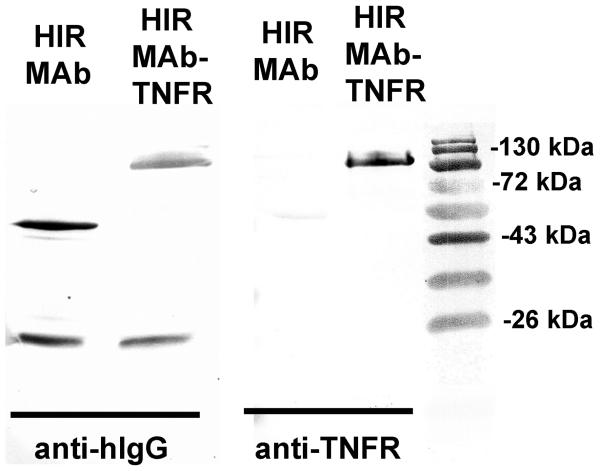
The CHO-derived, protein A-purified HIRMAb-TNFR fusion protein (Figure 1A), and the recombinant TNFR:Fc fusion protein (Figure 1B) were homogeneous on reducing SDS-PAGE. The molecular weight of the heavy chain of the HIRMAb-TNFR fusion protein was about 30 kDa larger than the molecular weight of the heavy chain of the HIRMAb without the fused TNFR (Figure 1A). The HIRMAb-TNFR fusion protein and the HIRMAb have identical light chains, whereas the TNFR:Fc fusion protein has no light chain (Figure 1). Western blot analysis with a primary antibody to either human IgG (Figure 2, left panel) or a primary antibody to human TNFR-II (Figure 2, right panel) demonstrated immunoreactivity of both the heavy and light chains of the HIRMAb or the HIRMAb-TNFR fusion protein with the anti-IgG antibody, and immunoreactivity only with the heavy chain of the HIRMAb-TNFR fusion protein with the anti-TNFR antibody (Figure 2). The HIRMAb-TNFR fusion protein migrated as a single peak with <2% aggregate on SEC HPLC (Methods).
Figure 1.
(A) SDS-PAGE under reducing conditions for the chimeric HIRMAb and the HIRMAb-TNFR fusion protein. (B) SDS-PAGE under reducing conditions for the TNFR:Fc fusion protein. Molecular weight standards are shown on the left and right sides of the gels.
Figure 2.
Western blot with either a primary antibody against human IgG (hIgG) (left panel) or against the human TNFR (right panel). Immunoreactivity is shown for the HIRMAb or the HIRMAb-TNFR fusion protein. Molecular weight standards are shown on the right side.
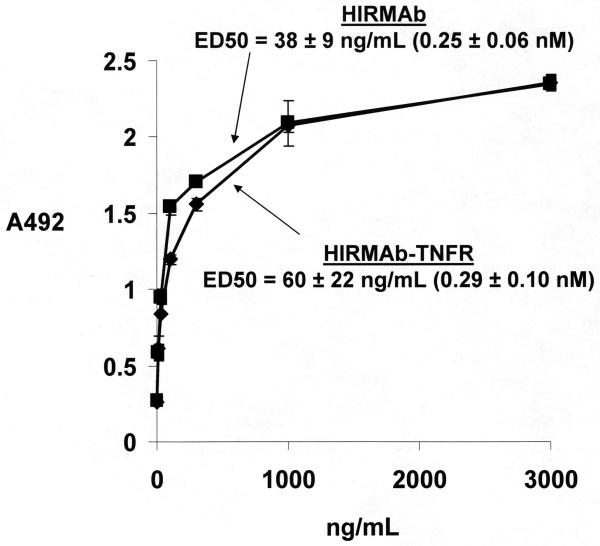
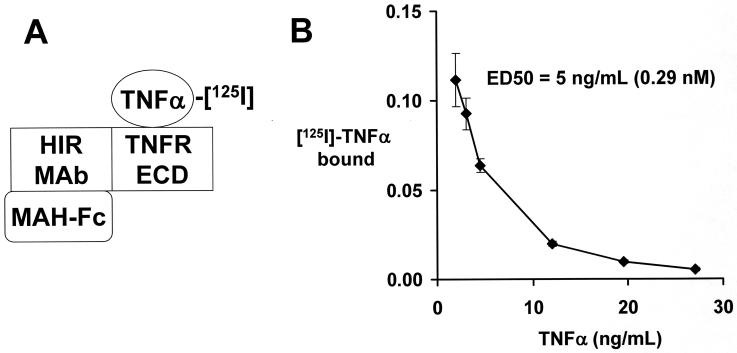
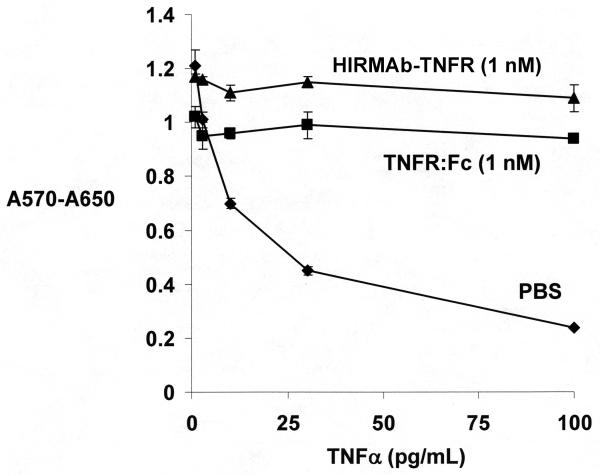
The HIRMAb and the HIRMAb-TNFR fusion protein both yielded saturable binding to the HIR with an ED50 of 0.25 ± 0.06 nM and 0.29 ± 0.10 nM, respectively (Figure 3). The design of the TNFα RRA is shown in Figure 4A. The ED50 of TNFα binding to the HIRMAb-TNFR fusion protein is 0.29 nM (Figure 4B). Both the CHO cell-derived HIRMAb-TNFR fusion protein and the TNFR:Fc fusion protein suppressed the cytotoxic actions of TNFα in a human cell bio-assay (Figure 5).
Figure 3.
Binding of either the HIRMAb or the HIRMAb-TNFR fusion protein to the HIR is saturable. The ED50 was determined by non-linear regression analysis. Data are mean ± SE (n=3 replicates per point). The molecular weights of the HIRMAb and the HIRMAb-TNFR fusion protein are 150 kDa and 210 kDa, respectively.
Figure 4.
(A) A radio-receptor assay is used to quantitate the saturable binding of TNFα to the HIRMAb-TNFR fusion protein. A mouse anti-human (MAH) IgG1 Fc is plated, which binds the Fc region of the HIRMAb-TNFR fusion protein. The TNFR extracellular domain (ECD) region of the fusion protein binds the [125I]-TNFα, which is displaced by unlabeled TNFα. (B) The saturable binding was analyzed by a non-linear regression analysis to yield the concentration, ED50, that yields 50% inhibition of TNFα binding to the HIRMAb-TNFR fusion protein.
Figure 5.
TNFα causes cytotoxicity in actinomycin D-treated human WEHI-13VAR cells with an ED50 of about 10 pg/mL. In the presence of either 1 nM TNFR:Fc or 1 nM HIRMAb-TNFR, there is no cytotoxicity caused by the high concentrations of TNFα.
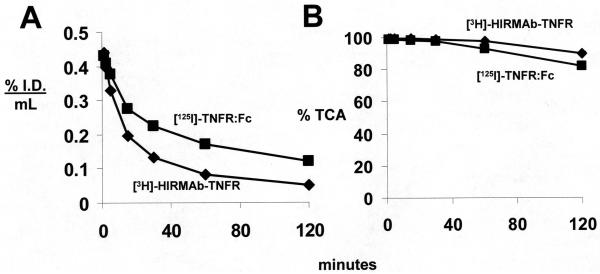
The HIRMAb-TNFR fusion protein was radiolabeled with the [3H] and the TNFR:Fc fusion protein was radiolabeled with [125I], and the proteins were co-injected IV into an adult Rhesus monkey. The clearance of the plasma radioactivity is shown in Figure 6A, and the plasma radioactivity that was precipitable with TCA is shown in Figure 6B. The plasma clearance profiles (Figure 6A) were fit to a bi-exponential function (Methods) for estimation of the PK parameters, which are shown in Table 1 for each fusion protein. The uptake of the fusion proteins by brain and peripheral organs was measured as a % ID/100 gram tissue, and these values are given in Table 2. The brain volume of distribution (VD) of the fusion proteins was measured with the capillary depletion method and the VD values for the homogenate, the vascular pellet, and the post-vascular supernatant are given in Table 3.
Figure 6.
(A) The plasma concentration of [125I]-TNFR:Fc fusion protein and [3H]-HIRMAb-TNFR fusion protein is plotted vs the time after a single intravenous injection of the proteins in the adult Rhesus monkey. Data are expressed as % injected dose (I.D.)/mL. (B) The % of plasma radioactivity that is precipitable by 10% trichloroacetic acid (TCA) is plotted vs. the time after injection for both proteins. Data are mean ± SE (n=3 replicates per point).
Table 1.
Pharmacokinetic parameters
| parameter | units | [125I]-TNFR:Fc Fusion protein |
[3H]-HIRMAb-TNFR Fusion protein |
|---|---|---|---|
| A1 | %ID/mL | 0.211 ± 0.010 | 0.319 ± 0.015 |
| A2 | %ID/mL | 0.239 ± 0.010 | 0.146 ± 0.011 |
| k1 | min-1 | 0.082 ± 0.009 | 0.099 ± 0.011 |
| k2 | min-1 | 0.0057 ± 0.0004 | 0.0091 ± 0.0008 |
| MRT | min | 166 ± 12 | 93 ± 8 |
| Vc | mL/kg | 54 ± 1 | 52 ± 2 |
| Vss | mL/kg | 91 ± 3 | 118 ± 6 |
| AUC|120 | %IDmin/mL | 23.3 ± 0.2 | 13.9 ± 0.2 |
| AUCss | %IDmin/mL | 44.5 ± 1.9 | 19.3 ± 0.7 |
| CL | mL/min/kg | 0.55 ± 0.02 | 1.28 ± 0.04 |
Estimated from the plasma clearance data in Figure 6.
Table 2.
Organ uptake of [125I]-TNFR:Fc and [3H]-HIRMAb-TNFR in the Rhesus monkey
| organ | [125I]-TNFR:Fc Fusion protein |
[3H]-HIRMAb-TNFR Fusion protein |
|---|---|---|
| Frontal gray | 0.230 ± 0.057 | 3.00 ± 0.07 |
| Frontal white | 0.070 ± 0.007 | 1.49 ± 0.19 |
| Cerebellar gray | 0.168 ± 0.009 | 2.41 ± 0.07 |
| Cerebellar white | 0.100 ± 0.004 | 2.23 ± 0.22 |
| heart | 1.06 ± 0.03 | 1.03 ± 0.08 |
| liver | 21.6± 0.2 | 30.3 ± 1.9 |
| spleen | 8.4 ± 0.2 | 26.6 ± 1.7 |
| lung | 3.96 ± 0.24 | 3.96 ± 0.57 |
| Skeletal muscle | 0.223 ± 0.013 | 0.17 ± 0.02 |
| fat | 0.279 ± 0.013 | 0.19 ± 0.01 |
Data are % ID/100 grams; mean ± SE (n=3).
Table 3.
Capillary depletion analysis of HIRMAb-TNFR and TNFR:Fc distribution in brain
| Parameter | TNFR:Fc | HIRMAb-TNFR |
|---|---|---|
| Homogenate VD | 13 ± 3 | 354 ± 21 |
| Post-vascular supernatant VD | 8.3 ± 0.2 | 208 ± 23 |
| Brain capillary pellet VD | 0.4 ± 0.1 | 28 ± 5 |
| TCA precipitation (%) | 71 ± 2 | 93 ± 1 |
Mean ± SE (n=3). VD=volume of distribution (uL/g); TCA=trichloroacetic acid.
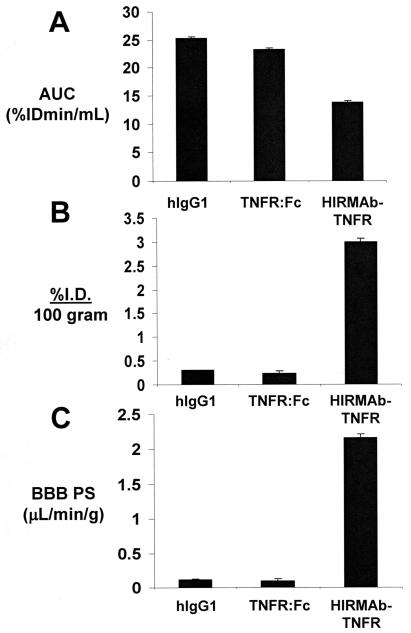
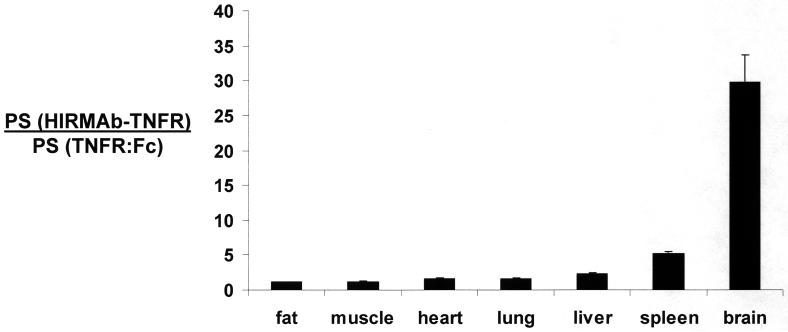
The BBB PS products for the HIRMAb-TNFR and TNFR:Fc fusion proteins were computed from the 2 hour plasma AUC (Figure 7A) and the brain uptake or %ID/100g (Figure 7B), and the PS products are given in Figure 7C. For comparison, the data in Figure 7 also display the AUC, the %ID/100g, and the BBB PS product for a vascular space marker, human IgG1. The PS products were similarly computed for the HIRMAb-TNFR and TNFR:Fc fusion proteins in peripheral organs and these data are given in Table 4. The ratio of the PS product for the HIRMAb-TNFR fusion protein relative to the PS product for the TNFR:Fc fusion protein in each organ is plotted in Figure 8.
Figure 7.
The plasma area under the concentration curve or AUC (A), the brain uptake or % injected dose (ID) per 100 gram brain (B), and the BBB permeability-surface area (PS) product (C), are plotted for the TNFR:Fc fusion protein, for the HIRMAb-TNFR fusion protein, and a brain plasma volume marker, human IgG1 (hIgG1). The IgG1 data are from Boado and Pardridge (2009a). All measurements were made at 2 hours after intravenous administration of the protein. Data are mean ± SE (n=3 replicates per point).
Table 4.
Organ PS products for TNFR:Fc and HIRMAb-TNFR fusion proteins
| organ | PS product (μL/min/g) | |
|---|---|---|
| TNFR:Fc | HIRMAb-TNFR | |
| Cerebral gray | 0.098 ± 0.020 | 2.2 ± 0.1 |
| Cerebral white | 0.030 ± 0.003 | 1.1 ± 0.2 |
| Cerebellar gray | 0.072 ± 0.003 | 1.7 ± 0.1 |
| Cerebellar white | 0.043 ± 0.002 | 1.6 ± 0.2 |
| Heart | 0.45 ± 0.02 | 0.72 ± 0.06 |
| Liver | 9.3 ± 0.1 | 21.8 ± 1.4 |
| Spleen | 3.6 ± 0.1 | 19.1 ± 0.8 |
| Lung | 1.7 ± 0.4 | 2.8 ± 0.4 |
| Skeletal muscle | 0.094 ± 0.004 | 0.12 ± 0.01 |
| Fat | 0.12 ± 0.01 | 0.14 ± 0.01 |
Data are mean ± SE (n=3).
Figure 8.
Ratio of the organ PS product for the HIRMAb-TNFR fusion protein, relative to the organ PS product for the TNFR:Fc fusion protein, is plotted for each organ. Data are mean ± SE (n=3 replicates per point). The ratio for brain is the mean of the values for frontal gray matter, frontal white matter, cerebellar gray matter, and cerebellar white matter, which varied between 22-37.
4. Discussion
The results of this study are consistent with the following conclusions. First, an IgG-TNFR fusion protein has been engineered and a high expressing CHO cell line cultured in serum free medium has been cloned. Second the CHO-derived, protein A purified HIRMAb-TNFR fusion protein conforms to expected specifications with respect to SDS-PAGE, IgG and TNFR Western blotting, SEC HPLC, high affinity binding both to the HIR and to human TNFα, and suppression of TNFα cytotoxic action in cultured human cells (Figures 1-5). Third, the brain uptake of the HIRMAb-TNFR fusion protein, 3.0 ± 0.1 % I.D/100 gram brain, is high compared to the brain uptake of the TNFR:Fc fusion protein, 0.23 ± 0.06 % I.D/100 gram (Table 2). Fourth, normalization of the organ uptake (%ID/gram) by the 2 hour plasma AUC allows for computation of the organ PS product for brain (Figure 7) and peripheral organs (Table 4); this analysis demonstrates the brain targeting properties of the HIRMAb Trojan horse, as the ratio of the PS product for the HIRMAb-TNFR fusion protein, relative to the TNFR:Fc fusion protein, is 30 for brain, but near unity for peripheral organs (Figure 8).
The structure of the HIRMAb-TNFR fusion protein departs from all prior structures of decoy receptor-IgG fusion proteins, where the decoy receptor is uniformly fused to the amino terminus of the human IgG Fc fragment (Peppel et al, 1991; Holash et al, 2002; Yepes et al, 2005; Plant et al, 2007). Fusion of the decoy receptor to the amino terminus of the human Fc serves to stabilize the decoy receptor in a dimeric configuration and to prolong the blood residence time of the decoy receptor pharmaceutical. However, the only receptor targeted by a decoy receptor:Fc fusion protein is the Fc receptor. Consequently, such Fc fusion proteins do not cross the BBB, as demonstrated in this study. While the neonatal Fc (FcRn) receptor is expressed at the BBB (Schlachetzki et al, 2002), the BBB Fc receptor only mediates the reverse transcytosis of IgG molecules from brain to blood (Zhang and Pardridge, 2001), and does not mediate the influx of IgG molecules from blood to brain. In contrast to the design of the typical decoy receptor:Fc fusion protein, the HIRMAb-TNFR fusion protein incorporates the fusion of the amino terminus of the decoy receptor to the carboxyl terminus of the CH3 region of the heavy chain of the HIRMAb (Hui et al, 2009). This design places the TNFR ECD in a dimeric configuration, which mimics the native state of the receptor, which crystallizes as a dimer (Chan et al, 2000). Fusion of the decoy receptor to the amino terminus of the HIRMAb heavy chain would most likely impair binding of the antibody to the HIR, as demonstrated previously for a HIRMAb-enzyme fusion protein (Boado and Pardridge, 2009b). Therefore, the design of the HIRMAb-TNFR fusion protein allows for retention of both functionalities of the fusion protein, i.e., high affinity binding to the HIR (Figure 3), for mediation of BBB transport, and high affinity binding to human TNFα (Figure 4), for suppression of the cytotoxic effects of this cytokine (Figure 5).
Binding of the HIRMAb-TNFR fusion protein to the BBB insulin receptor should trigger transport of the fusion protein across the BBB. BBB transport was evaluated in the adult Rhesus monkey in the present study with the HIRMAb-TNFR fusion protein that was radio-labeled by tritation with 3H-N-succinimidyl propionate (Methods). This reagent labels the ε-amino group of lysine residues by a non-oxidative chemical reaction. Previously, HIRMAb fusion proteins were radio-iodinated with 125I-idodine and chloramine T (Boado et al, 2007a; Boado and Pardridge, 2009a). However, the chloramine T reaction is an oxidative process that can damage the protein. An index of protein stability is the amount of plasma radioactivity that is TCA precipitable at the terminal time point. In the present study, >90% of the 120 minute plasma radioactivity was TCA precipitable for the [3H]-HIRMAb-TNFR fusion protein (Figure 6B). This high degree of stability is comparable to that reported originally for the murine HIRMAb (Pardridge et al, 1995). Accordingly, the brain uptake (Table 2) and the BBB PS product (Figure 7) of the HIRMAb-TNFR fusion protein are comparable to the same parameters reported previously for the murine HIRMAb (Pardridge et al, 1995).
The selective transport of the HIRMAb-TNFR fusion protein across the primate BBB in vivo, relative to the TNFR:Fc fusion protein, is shown in Table 2, which gives the brain uptake of the proteins expressed as %ID/100 grams. The uptake data are expressed as 100 grams of tissue, because the weight of the Rhesus monkey brain is 100 grams (Davies and Morris, 1993). However, the brain uptake parameters in Table 2 are not direct measures of the relative brain penetration of the HIRMAb-TNFR and TNFR:Fc fusion proteins, and the following considerations should be made. First, the brain uptake, or %ID/g, is a function of the plasma AUC of the protein. Normalizing the %ID/g by the plasma AUC values in Table 1 allows for computation of the organ PS product, and the PS products are shown in Figure 7 for brain and in Table 4 for peripheral organs. Second, the brain uptake, or %ID/g, must be corrected for the organ blood volume. Organ uptake values for a given protein could reflect simply sequestration of the protein in the blood space of the organ, which can vary widely between tissues. The organ blood volume factor is normalized by determination of the PS product for a blood volume marker, such as human IgG1, which is the isotype control of the HIRMAb. The PS product for human IgG1 was measured in a previous study (Boado and Pardridge, 2009a), and the brain PS product for human IgG1 is shown in Figure 7. The equivalence of the BBB PS product for human IgG1 and the TNFR:Fc fusion protein is quantitative evidence that the TNFR:Fc fusion protein does not cross the BBB. Computation of a PS product for a brain blood volume marker, such as human IgG1 or the TNFR:Fc fusion protein, is an approximation since the actual PS product for either protein, after correction for the brain blood volume, is zero. Third, the high BBB PS product for the HIRMAb-TNFR fusion protein (Figure 7) could reflect sequestration of the fusion protein by the brain microvasculature, and not actual transcytosis across the BBB and penetration into brain parenchyma. For this reason, the capillary depletion analysis was performed. The high VD of the HIRMAb-TNFR fusion protein in the post-vascular supernatant, and low VD in the vascular pellet (Table 3), demonstrates that >90% of the HIRMAb-TNFR fusion protein taken up by brain has penetrated into the post-vascular parenchyma. The homogenate VD of the TNFR:Fc fusion protein. 13 ± 3 uL/g (Table 3), is equal to the brain blood volume (Boado and Pardridge, 2009a), which is further evidence that the TNFR:Fc fusion protein does not cross the BBB in the blood to brain direction. The global penetration of brain parenchyma in the primate by HIRMAb-based fusion proteins has been recently corroborated by both film and emulsion autoradiography of brain (Boado and Pardridge, 2009a).
The PS products of the HIRMAb-TNFR and TNFR:Fc fusion proteins were also computed for peripheral organs (Table 4). The ratio of the PS product for the HIRMAb-TNFR fusion protein, relative to the PS product for the TNFR:Fc fusion protein, for brain and peripheral organs is plotted in Figure 8. These data show that the ratio of the PS product for the HIRMAb-TNFR fusion protein, relative to the PS product for the TNFR:Fc fusion protein, is near unity for peripheral organs such as heart, lung, skeletal muscle, and fat, is modestly elevated 2- to 5-fold for organs such as liver or spleen, and is selectively elevated for brain (Figure 8). The PS product ratio in brain, 30, is an under-estimate, since the actual BBB PS product for the TNFR:Fc fusion protein is zero.
The pharmacokinetic and brain uptake data for the primate reported in this study allow for initial dosing considerations of therapeutic interventions with the HIRMAb-TNFR fusion protein. The brain uptake, 3.0 %ID/100 gram (Table 2), at an injection dose of 0.2 mg/kg (Methods), produces a brain concentration of the HIRMAb-TNFR fusion protein of 1.1 pmol/gram fusion protein, which is equivalent to 2.2 pmol/gram, since there are 2 TNFR moieties per individual fusion protein (Hui et al, 2009). The concentration of immunoreactive TNFα in normal brain is undetectable, but increases to 0.4 pmol/gram in traumatic brain injury (Shohami et al, 1994). Since the affinity of the HIRMAb-TNFR fusion protein for TNFα is high (Figure 4B), a low dose of the HIRMAb-TNFR fusion protein of 0.2 mg/kg will sequester most of the cerebral TNFα in brain in traumatic brain injury. Higher doses of the fusion protein would sequester essentially 100% of the TNFα in brain in pathologic conditions. Doses of HIRMAb fusion proteins as high as 20 mg/kg have been administered IV to Rhesus monkeys either acutely or chronically without side effects or changes in glycemic control in either plasma or cerebrospinal fluid (Pardridge and Boado, 2009; Boado et al, 2009).
In summary, the present study demonstrates that the re-engineering of a model decoy receptor pharmaceutical, the TNFR, as a fusion protein with a BBB molecular Trojan horse, the HIRMAb, produces a new chemical entity that rapidly penetrates the BBB in vivo. The cerebral concentrations of the HIRMAb-TNFR fusion protein are sufficient, at low systemic doses, to sequester all of the target cytokine in the brain in pathologic conditions. TNFα plays a pathologic role in acute brain disorders, such as stroke (Nawashiro et al, 1997), traumatic brain injury (Knoblach et al, 1999), or spinal cord injury (Marchand et al, 2009), and in chronic brain conditions, such as neurodegeneration (Tweedie et al, 2007) and depression (Himmerich et al, 2008). Moreover, other decoy receptors could be re-engineered as HIRMAb fusion proteins for brain conditions, such as the vascular endothelial growth factor receptor for brain cancer (Holash et al, 2002), the TNF-like weak inducer of apoptosis receptor for stroke (Yepes et al, 2005), or the lymphotoxin β receptor for multiple sclerosis (Plant et al, 2007). The results of this study emphasize the primary role played by brain drug targeting technology in the drug development of biopharmaceuticals for the human brain.
ACKNOWLEDGEMENTS
Winnie Tai and Phuong Tram provided technical assistance. This work was supported by NIH grant R43-NS066514.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Boado RJ, Pardridge WM. Comparison of blood-brain barrier transport of GDNF and an IgG-GDNF fusion protein in the Rhesus monkey. Drug Metab Dispos. 2009a;37:2299–2304. doi: 10.1124/dmd.109.028787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boado RJ, Pardridge WM. Genetic engineering of IgG-glucuronidase fusion proteins. J Drug Targeting. 2009b doi: 10.3109/10611860903353362. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boado RJ, Hui EK, Lu JZ, Pardridge WM. AGT-181: Expression in CHO cells and pharmacokinetics, safety, and plasma iduronidase enzyme activity in Rhesus monkeys. J Biotechnol. 2009;144:135–141. doi: 10.1016/j.jbiotec.2009.08.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boado RJ, Zhang Y, Zhang Y, Pardridge WM. Humanization of anti-human insulin receptor antibody for drug targeting across the human blood-brain barrier. Biotechnol Bioeng. 2007a;96:381–391. doi: 10.1002/bit.21120. [DOI] [PubMed] [Google Scholar]
- Boado RJ, Zhang Y, Zhang Y, Pardridge WM. Genetic engineering, expression, and activity of a fusion protein of a human neurotrophin and a molecular Trojan horse for delivery across the human blood-brain barrier. Biotechnol Bioeng. 2007b;97:1376–1386. doi: 10.1002/bit.21369. [DOI] [PubMed] [Google Scholar]
- Chan KF, Siegel MR, Lenardo JM. Signaling by the TNF receptor superfamily and T cell homeostasis. Immunity. 2000;13:419–22. doi: 10.1016/s1074-7613(00)00041-8. [DOI] [PubMed] [Google Scholar]
- Coloma MJ, Lee HJ, Kurihara A, Landaw EM, Boado RJ, Morrison SL, Pardridge WM. Transport across the primate blood-brain barrier of a genetically engineered chimeric monoclonal antibody to the human insulin receptor. Pharm Res. 2000;17:266–274. doi: 10.1023/a:1007592720793. [DOI] [PubMed] [Google Scholar]
- Davies B, Morris T. Physiological parameters in laboratory animals and humans. Pharm Res. 1993;10:1093–1095. doi: 10.1023/a:1018943613122. [DOI] [PubMed] [Google Scholar]
- Himmerich H, Fulda S, Linseisen J, Seiler H, Wolfram G, Himmerich S, Gedrich K, Kloiber S, Lucae S, Ising M, Uhr M, Holsboer F, Pollmacher T. Depression, comorbidities and the TNF-alpha system. Eur Psychiatry. 2008;23:421–9. doi: 10.1016/j.eurpsy.2008.03.013. [DOI] [PubMed] [Google Scholar]
- Holash J, Davis S, Papadopoulos N, Croll SD, Ho L, Russell M, Boland P, Leidich R, Hylton D, Burova E, Ioffe E, Huang T, Radziejewski C, Bailey K, Fandl JP, Daly T, Wiegand SJ, Yancopoulos GD, Rudge JS. VEGF-Trap: a VEGF blocker with potent antitumor effects. Proc Natl Acad Sci USA. 2002;99:11393–11398. doi: 10.1073/pnas.172398299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hui EK, Boado RJ, Pardridge WM. Tumor necrosis factor receptor-IgG fusion protein for targeted drug delivery across the human blood-brain barrier. Mol Pharm. 2009;6:1536–1543. doi: 10.1021/mp900103n. [DOI] [PubMed] [Google Scholar]
- Knoblach SM, Fan L, Faden AI. Early neuronal expression of tumor necrosis factor-alpha after experimental brain injury contributes to neurological impairment. J Neuroimmunol. 1999;95:115–125. doi: 10.1016/s0165-5728(98)00273-2. [DOI] [PubMed] [Google Scholar]
- Marchand F, Tsantoulas C, Singh D, Grist J, Clark AK, Bradbury EJ, McMahon SB. Effects of Etanercept and Minocycline in a rat model of spinal cord injury. Eur J Pain. 2009;13:673–681. doi: 10.1016/j.ejpain.2008.08.001. [DOI] [PubMed] [Google Scholar]
- Nawashiro H, Martin D, Hallenbeck JM. Neuroprotective effects of TNF binding protein in focal cerebral ischemia. Brain Res. 1997;778:265–271. doi: 10.1016/s0006-8993(97)00981-5. [DOI] [PubMed] [Google Scholar]
- Pardridge WM, Boado RJ. Pharmacokinetics and safety in rhesus monkeys of a monoclonal antibody-GDNF fusion protein for targeted blood-brain barrier delivery. Pharm Res. 2009;26:2227–2236. doi: 10.1007/s11095-009-9939-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pardridge WM. Re-engineering biopharmaceuticals for delivery to brain with molecular Trojan horses. Bioconjug Chem. 2008;19:1327–1338. doi: 10.1021/bc800148t. [DOI] [PubMed] [Google Scholar]
- Pardridge WM, Kang YS, Buciak JL, Yang J. Human insulin receptor monoclonal antibody undergoes high affinity binding to human brain capillaries in vitro and rapid transcytosis through the blood-brain barrier in vivo in the primate. Pharm Res. 1995;12:807–816. doi: 10.1023/a:1016244500596. [DOI] [PubMed] [Google Scholar]
- Peppel K, Crawford D, Beutler B. A tumor necrosis factor (TNF) receptor-IgG heavy chain chimeric protein as a bivalent antagonist of TNF activity. J Exp Med. 1991;174:1483–1489. doi: 10.1084/jem.174.6.1483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plant SR, Iocca HA, Wang Y, Thrash JC, O'Connor BP, Arnett HA, Fu YX, Carson MJ, Ting JP. Lymphotoxin beta receptor (Lt betaR): dual roles in demyelination and remyelination and successful therapeutic intervention using Lt betaR-Ig protein. J Neurosci. 2007;27:7429–7437. doi: 10.1523/JNEUROSCI.1307-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlachetzki F, Zhu C, Pardridge WM. Expression of the neonatal Fc receptor (FcRn) at the blood-brain barrier. J Neurochem. 2002;81:203–206. doi: 10.1046/j.1471-4159.2002.00840.x. [DOI] [PubMed] [Google Scholar]
- Shohami E, Novikov M, Bass R, Yamin A, Gallily R. Closed head injury triggers early production of TNF alpha and IL-6 by brain tissue. J Cereb Blood Flow Metab. 1994;14:615–619. doi: 10.1038/jcbfm.1994.76. [DOI] [PubMed] [Google Scholar]
- Triguero D, Buciak J, Pardridge WM. Capillary depletion method for quantification of blood-brain barrier transport of circulating peptides and plasma proteins. J Neurochem. 1990;54:1882–1888. doi: 10.1111/j.1471-4159.1990.tb04886.x. [DOI] [PubMed] [Google Scholar]
- Tweedie D, Sambamurti K, Greig NH. TNF-alpha inhibition as a treatment strategy for neurodegenerative disorders: new drug candidates and targets. Curr Alzheimer Res. 2007;4:378–385. doi: 10.2174/156720507781788873. [DOI] [PubMed] [Google Scholar]
- Valesini G, Iannuccelli C, Marocchi E, Pascoli L, Scalzi V, Di Franco M. Biological and clinical effects of anti-TNFalpha treatment. Autoimmun Rev. 2007;7:35–41. doi: 10.1016/j.autrev.2007.03.003. [DOI] [PubMed] [Google Scholar]
- Yepes M, Brown SA, Moore EG, Smith EP, Lawrence DA, Winkles JA. A soluble Fn14-Fc decoy receptor reduces infarct volume in a murine model of cerebral ischemia. Am J Pathol. 2005;166:511–520. doi: 10.1016/S0002-9440(10)62273-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Y, Pardridge WM. Mediated efflux of IgG molecules from brain to blood across the blood-brain barrier. J Neuroimmunol. 2001;114:168–172. doi: 10.1016/s0165-5728(01)00242-9. [DOI] [PubMed] [Google Scholar]