Abstract
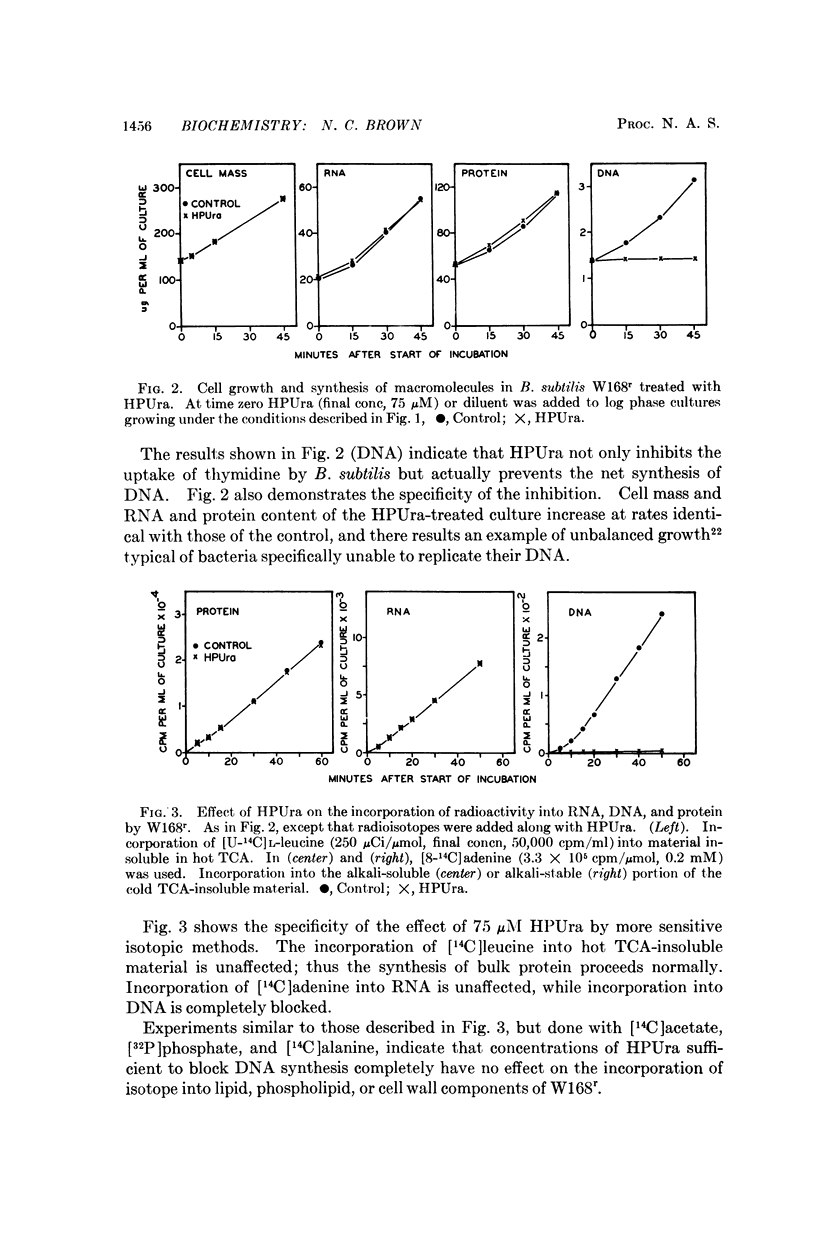
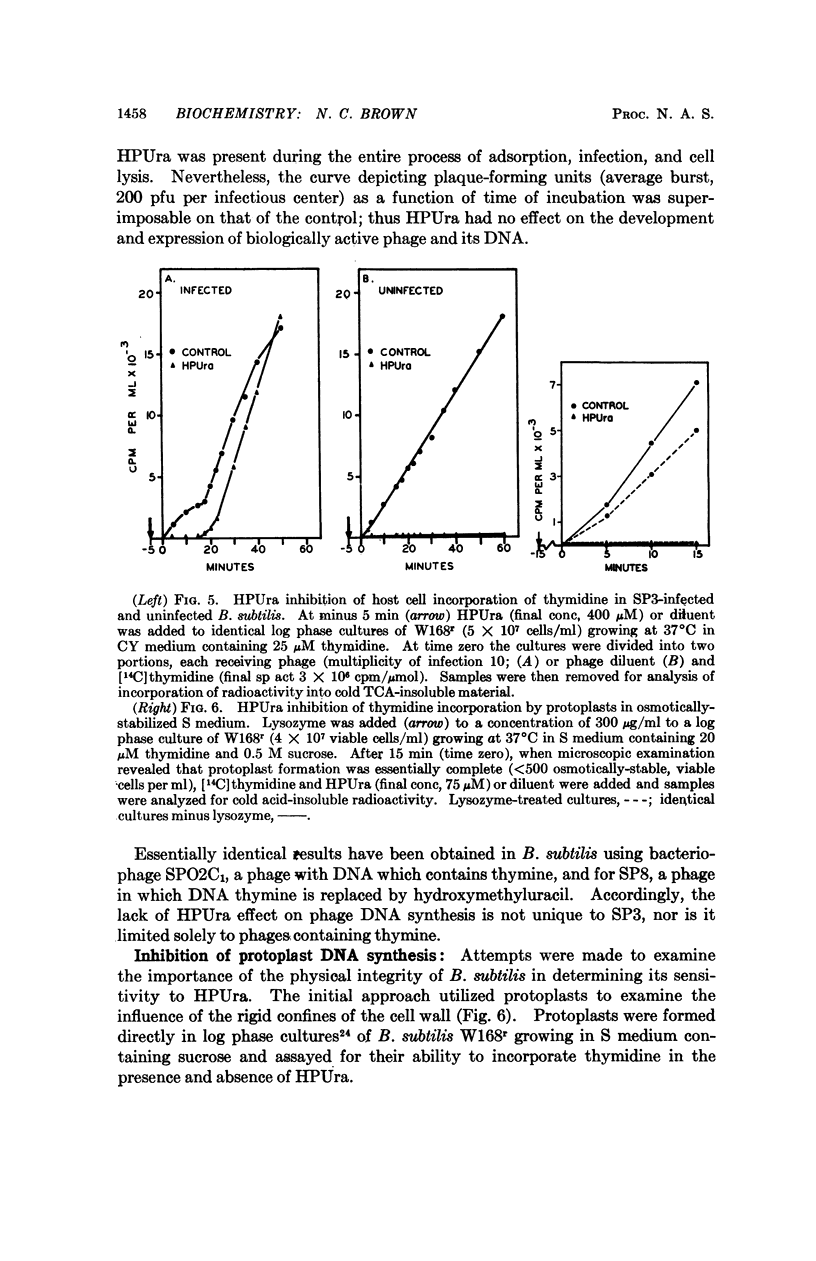
The azopyrimidine, 6-(p-hydroxyphenylazo)-uracil, inhibits the replication of bacterial DNA selectively, completely, and reversibly, and has no apparent effects on the metabolism of other cellular macromolecules thus far examined. The mechanism of its action has been investigated in uninfected and phage-infected Bacillus subtilis, and the compound appears to be specific for a host function. In cells infected with virulent phage the synthesis of phage DNA proceeds normally, while residual host DNA synthesis is completely blocked. The drug-sensitive host site retains its sensitivity even after partial disruption of the cell by lysozyme treatment.
Full text
PDF





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- BURTON K. A study of the conditions and mechanism of the diphenylamine reaction for the colorimetric estimation of deoxyribonucleic acid. Biochem J. 1956 Feb;62(2):315–323. doi: 10.1042/bj0620315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown N. C., Handschumacher R. E. Inhibition of the synthesis of deoxyribonucleic acid in bacteria by 6-(p-hydroxyphenylazo)-2,4-dihydroxypyrimidine. I. Metabolic studies in Streptococcus fecalis. J Biol Chem. 1966 Jul 10;241(13):3083–3089. [PubMed] [Google Scholar]
- Cairns J., Denhardt D. T. Effect of cyanide and carbon monoxide on the replication of bacterial DNA in vivo. J Mol Biol. 1968 Sep 28;36(3):335–342. doi: 10.1016/0022-2836(68)90159-9. [DOI] [PubMed] [Google Scholar]
- Cohen S. S., Barner H. D. STUDIES ON UNBALANCED GROWTH IN ESCHERICHIA COLI. Proc Natl Acad Sci U S A. 1954 Oct;40(10):885–893. doi: 10.1073/pnas.40.10.885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cook T. M., Brown K. G., Boyle J. V., Goss W. A. Bactericidal action of nalidixic acid on Bacillus subtilis. J Bacteriol. 1966 Nov;92(5):1510–1514. doi: 10.1128/jb.92.5.1510-1514.1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Lucia P., Cairns J. Isolation of an E. coli strain with a mutation affecting DNA polymerase. Nature. 1969 Dec 20;224(5225):1164–1166. doi: 10.1038/2241164a0. [DOI] [PubMed] [Google Scholar]
- FARMER J. L., ROTHMAN F. TRANSFORMABLE THYMINE-REQUIRING MUTANT OF BACILLUS SUBTILS. J Bacteriol. 1965 Jan;89:262–263. doi: 10.1128/jb.89.1.262-263.1965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- GOLDBERG I. H., REICH E. ACTINOMYCIN INHIBITION OF RNA SYNTHESIS DIRECTED BY DNA. Fed Proc. 1964 Sep-Oct;23:958–964. [PubMed] [Google Scholar]
- Gage L. P., Fujita D. J. Effect of nalidixic acid on deoxyribonucleic acid synthesis in bacteriophage SPO1-infected Bacillus subtilis. J Bacteriol. 1969 Apr;98(1):96–103. doi: 10.1128/jb.98.1.96-103.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gellert M. Formation of covalent circles of lambda DNA by E. coli extracts. Proc Natl Acad Sci U S A. 1967 Jan;57(1):148–155. doi: 10.1073/pnas.57.1.148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knippers R., Strätling W. The DNA replicating capacity of isolated E. coli cell wall-membrane complexes. Nature. 1970 May 23;226(5247):713–717. doi: 10.1038/226713a0. [DOI] [PubMed] [Google Scholar]
- LEHMAN I. R., BESSMAN M. J., SIMMS E. S., KORNBERG A. Enzymatic synthesis of deoxyribonucleic acid. I. Preparation of substrates and partial purification of an enzyme from Escherichia coli. J Biol Chem. 1958 Jul;233(1):163–170. [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- MAALOE O., HANAWALT P. C. Thymine deficiency and the normal DNA replication cycle. I. J Mol Biol. 1961 Apr;3:144–155. doi: 10.1016/s0022-2836(61)80041-7. [DOI] [PubMed] [Google Scholar]
- Moses R. E., Richardson C. C. Replication and repair of DNA in cells of Escherichia coli treated with toluene. Proc Natl Acad Sci U S A. 1970 Oct;67(2):674–681. doi: 10.1073/pnas.67.2.674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- OKUBO S., STRAUSS B., STODOLSKY M. THE POSSIBLE ROLE OF RECOMBINATION IN THE INFECTION OF COMPETENT BACILLUS SUBTILIS BY BACTERIOPHAGE DEOXYRIBONUCLEIC ACID. Virology. 1964 Dec;24:552–562. doi: 10.1016/0042-6822(64)90207-7. [DOI] [PubMed] [Google Scholar]
- Okazaki R., Okazaki T., Sakabe K., Sugimoto K., Sugino A. Mechanism of DNA chain growth. I. Possible discontinuity and unusual secondary structure of newly synthesized chains. Proc Natl Acad Sci U S A. 1968 Feb;59(2):598–605. doi: 10.1073/pnas.59.2.598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pène J. J. Host macromolecular synthesis in bacteriophage-infected Bacillus subtilis. Bacteriol Rev. 1968 Dec;32(4 Pt 1):379–386. [PMC free article] [PubMed] [Google Scholar]
- ROODYN D. B., MANDEL H. G. A simple membrane fractionation method for determining the distribution of radioactivity in chemical fractions of Bacillus cereus. Biochim Biophys Acta. 1960 Jun 17;41:80–88. doi: 10.1016/0006-3002(60)90371-1. [DOI] [PubMed] [Google Scholar]
- SEAMAN E., TARMY E., MARMUR J. INDUCIBLE PHAGES OF BACILLUS SUBTILIS. Biochemistry. 1964 May;3:607–613. doi: 10.1021/bi00893a001. [DOI] [PubMed] [Google Scholar]
- Sekiguchi M., Iida S. Mutants of Escherichia coli permeable to actinomycin. Proc Natl Acad Sci U S A. 1967 Dec;58(6):2315–2320. doi: 10.1073/pnas.58.6.2315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith D. W., Schaller H. E., Bonhoeffer F. J. DNA synthesis in vitro. Nature. 1970 May 23;226(5247):711–713. doi: 10.1038/226711a0. [DOI] [PubMed] [Google Scholar]
- Spizizen J. TRANSFORMATION OF BIOCHEMICALLY DEFICIENT STRAINS OF BACILLUS SUBTILIS BY DEOXYRIBONUCLEATE. Proc Natl Acad Sci U S A. 1958 Oct 15;44(10):1072–1078. doi: 10.1073/pnas.44.10.1072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trilling D. M., Aposhian H. V. A deoxyribonuclease found after infection of Bacillus subtilis with phage SP3. Proc Natl Acad Sci U S A. 1965 Aug;54(2):622–628. doi: 10.1073/pnas.54.2.622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wehrli W., Knüsel F., Schmid K., Staehelin M. Interaction of rifamycin with bacterial RNA polymerase. Proc Natl Acad Sci U S A. 1968 Oct;61(2):667–673. doi: 10.1073/pnas.61.2.667. [DOI] [PMC free article] [PubMed] [Google Scholar]


