Abstract
Context
The anterior cingulate cortex (ACC) and insula are important neural substrates for the integration of cognitive, emotional, and physiological information, as well as the coordination of responses to anticipated stimuli. Increased neural activation within these structures has been observed in individuals with anxiety and depressive disorders. Selective serotonin reuptake inhibitors (SSRIs) are among the most effective and frequently prescribed anxiolytic agents, yet it is not known whether ACC or insula underlie the effects of these drugs. We examined whether subchronic administration of an SSRI to healthy volunteers attenuate activation in ACC or insula during anticipation, an important emotional process underlying anxiety. Support for this hypothesis would help to understand where and by what process SSRIs may exert beneficial effects as anxiolytics and would provide further mechanistic evidence for functional magnetic resonance imaging (fMRI) as a biomarker for the development of anxiolytics.
Participants and Design
15 volunteers participated in a double-blind, placebo-controlled, randomized cross-over study. Participants completed a pleasant and aversive picture cued anticipation task during fMRI after taking either escitalopram (10 mg) or placebo for 21 days.
Main Outcome Measure
Percent BOLD signal change during SSRI administration.
Results
Escitalopram significantly decreased activation in bilateral posterior and middle insula during the anticipation condition irrespective of stimulus valence and in medial prefrontal and ACC during anticipation of aversive versus pleasant images.
Conclusion
Reduced insular and ACC activation during anticipation may be integral to the therapeutic efficacy of SSRIs and provide a mechanistic approach for the use of pharmacofMRI in the identification of novel pharmacotherapeutic agents.
Keywords: SSRI, escitalopram, insula, fMRI, anticipation
Introduction
Increased emotionality associated with the anticipation of future events is a key feature of anxiety disorders 1, 2. Related evidence indicates that heightened anticipatory anxiety is associated with deleterious psychophysiological stress responses 3, 4. Serotonin may well be an important neurotransmitter in this, and other, affective processes 5, as well as playing a role in modulating both psychological 6–9 and physiological 10–12 aspects of anticipatory anxiety. Serotonin receptors are widely expressed within the amygdala 13, the ventral anterior cingulate (ACC) and insula 14, neural substrates that play a critical role in regulating psychological well-being and physiological homeostasis 15, 16.
Functional neuroimaging studies have shown that the medial prefrontal gyrus (MPFG), ACC 17–19 and insula 19, 20 are activated during anticipation of an electric shock or a noxious thermal stimulus, and during anticipation of feedback in a decision-making task 21. Previously 22, we examined anticipation of aversive images (i.e., spiders and snakes) in healthy volunteers and found anticipation-related activation within the right insula. Furthermore, using that task, we observed greater insula activity in subjects with high trait anxiety 23, as well as in patients with PTSD (Simmons et al., submitted). In a similar study, Nitschke and colleagues displayed aversive and non-aversive pictures to healthy volunteers and found anticipation-related activation in ventral and dorsal ACC, bilateral insula, and bilateral amygdala 24. In addition, Bermpohl and colleagues have found that the dorsal MPFG/ACC are particularly sensitive to expectancy while other regions such as the insula and amygdala are more sensitive to emotion intensity of the stimulus25, 26. The dorsal ACC has strong connections with the insula and these areas are often described as being part of a primary “default mode” network 27.
The insula, a part of the extended limbic system, can be subdivided into anterior agranular (Ia), central/middle dysgranular (Id) and posterior granular (Ig) subregions based on function and cytoarchitectural structure 28, 29. The anterior insula has efferent connections with ventral frontal brain regions such as the ACC and orbital frontal cortex (OFC), as well as with periamygdaliod areas. Its middle region has strong connections with the amygdala body, OFC and secondary somatosensory areas 28, 30, 31. The posterior insula has afferent projections from the frontal cortex, the temporopolar cortex, and secondary somatosensory area 29. Recent literature has suggested that the rostral parts of the insula (Ia and anterior Id) motivates action while the caudal insula (Ig and posterior Id) is involved in monitoring the physiological condition of the body 32, 33. In comparison, the ACC—particularly the ventral subdivision—plays a similar role in emotional and physiological processing (Brodmann Area, BA 24a). Its ventral region has projections to the anterior insula 28, 29, 34–36 and the amygdala, 34, 37–40 exerting top-down regulation on these structures 38, 40. The ventral ACC is involved in fear conditioning 40–42, in the pathophysiology of anxiety disorders 41, 43, self-relevant cognition 44–52, and error processing 49, 53–55. Given the importance of the various subdivisions of the ACC in the integration of physiological and psychological processes, changes in their activity are potentially useful neural biomarkers for the efficacy of pharmacotherapies 56.
Altered anticipatory processing is a key feature of many of the anxiety disorders, reflected by greater activation in the insula 19, 23, 24, 57, medial frontal gyrus 23, 24, 57–59, and amygdala 4, 24, 57. Although anticipation may be less pertinent to depression than to anxiety, the high comorbidity of anxiety and depressive symptoms60 suggests that elevating anticipatory symptoms may be relevant to both conditions. SSRIs are among the first-line treatments for anxiety and depression 5, 13, 61–68. Although SSRIs block synaptic neuronal reuptake of secreted serotonin 69, 70, current theories posit that the antidepressant (and, possibly, the anxiolytic) actions of SSRIs involve effects that extend beyond serotonin reuptake 65, 66, 71. The effects of SSRIs in affective disorders may be the result of modulation by serotonin pathways of the cortical and subcortical circuitry involved in the processing of emotional stimuli 72. The acute effects of SSRIs are sometimes opposite the chronic effects in that an early elevation of anxiety symptoms is often followed by an anxiolytic effect if treatment is continued 13, 73. Acute oral 74 and intravenous 75 administration of an SSRI (i.e., citalopram) has been shown to increase the processing of anxiety-related stimuli in healthy volunteers. Acute SSRI administration has been associated with decreased activation during affective image processing 76 and during a go-nogo task 77, whereas more prolonged administration has more consistently been associated with attenuation of the recognition of fearful stimuli 78 and amygdala activation 73. Thus the role of SSRIs in brain may be highly dependent on the task used to probe the brain and the length of drug administration. Although effects of SSRIs on emotion processing networks are under intense investigation, much is still unknown about how these substances work to normalize abnormal cognitive and emotional processes.
In an effort to better understand the mechanism (and brain localization) of SSRI treatments, fMRI techniques have recently been applied to measure their effect on neural processing 73, 79, 80. We have previously shown that acute administration of a benzodiazepine anxiolytic (lorazepam) attenuates activity of the amygdala, ACC, and insula during risk-taking decision making 81 and emotional face processing 56. Acute doses of SSRI treatment can increase anxiety whereas prolonged administration can attenuate amygdala activation 73 and decrease anxious distress. Although several studies have used pharmaco-fMRI to assess the neural correlates of certain antidepressant agents 72, 73, 77, 82–86, to our knowledge, this is the first study to implement a sub-chronic, placebo-control cross-over (i.e., within subjects) design using a cohort of healthy volunteers to assess the effects of SSRIs on emotion anticipation. Considering the pivotal role of the insula and ACC in subjective feeling states and interoceptive awareness 32, 87 and their implication in the pathophysiology of anxiety disorders 87–89, we hypothesized that subchronic administration (3 weeks) of the SSRI escitalopram would be associated with attenuated activation in the ACC and insula during an emotional anticipation task. Confirmation of this hypothesis would provide further evidence of the utility of pharmacofMRI as a tool to identify the neural substrates important for anxiety and depression 90. Once neural substrates are established, changes in their activity can be used as biomarkers for the measurement of efficacy of novel anxiolytics or antidepressants.
Methods
Subjects
Sixteen healthy, nonsmoking females provided written informed consent and were paid for their participation in this study, which was approved by the University of California San Diego School of Medicine institutional review board. One subject was excluded because her urine escitalopram level was undetectable during the period of time when she was to have been taking escitalopram, suggesting non-adherence to the protocol. The remaining 15 subjects were females of ages 19 to 27 years (mean ± SD, 22.3 ± 2.3 years) with 11 to 17 years of education (mean ± SD, 15.5 ± 1.8 years). Participants did not have medical or psychiatric disorders as determined by medical history and diagnoses according to the Structured Clinical Interview for Diagnostic and Statistical Manual of Mental Disorders, Revised Fourth Edition 91. Subjects had no history of drug or alcohol abuse and no history of previously taking benzodiazepines, SSRIs, monoamine oxidase inhibitors (MAOIs), or neuroleptics. All participants had a negative urine drug screen at baseline. EKG and routine laboratory blood tests, which included a CBC, electrolytes, and liver function tests, were within normal limits. Subjects were instructed to maintain their regular bedtimes and wake times for 1 week before and throughout the study period.
Study design
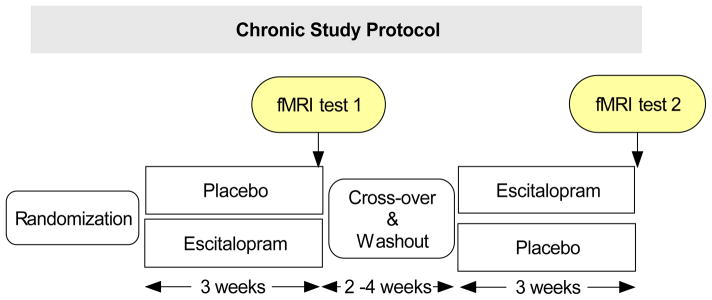
This study used a randomized, cross-over, double blind design (see Figure 1). Once it was determined that a subject was eligible for the study, and informed written consent was obtained, the subject was randomized to receive either escitalopram (5 mg/d for the first 3 days, then 10 mg/d for another 18 days) or placebo, administered in identical, capsular form. Subjects were instructed to take the medication each morning throughout each 21 day arm of the study. In between arms, there was a 14–28 day tapered wash-out period, during which the medication was reduced from 10 mg/d to 5 mg/d for 3 days, and then discontinued. The study physician (MPP) also met with subjects weekly in order to address any concerns and to ensure that compliance with the medication was maintained.
Figure 1.
Study design
At the end of each 21-day medication arm (prior to taper), subjects were scheduled for an fMRI visit. During this visit, and prior to the scanning session, subjects completed several self-report questionnaires, including the State-Trait Anxiety Inventory (STAI-S) 92, Beck Depression Inventory (BDI) 93, Social Interaction Anxiety Scale SIAS 94, and the Brief Symptom Inventory BSI 95 to evaluate their psychological state at that time and provided a urine sample for escitalopram measurement.
Task
The task combined a continuous performance task (CPT), similar to a task described previously 96, with the interspersed presentation of aversive affective stimuli. During the CPT, subjects were asked to press a LEFT mouse button whenever they saw a blue circle and a RIGHT mouse button whenever they saw a blue square on the screen. Stimuli were presented at a visual angle of 4 degrees at a rate of 0.5 Hz. Simultaneously, a 250 msec long 500 Hz tone was presented every 2 seconds. Subjects were instructed prior to the task that a switch from a blue to a green circle or square accompanied by a 250 Hz tone would indicate that a positive image was going to appear on the screen. In contrast, a switch from blue to red stimuli together with a 1000 Hz tone signaled an impending negative image. The picture stimuli were comprised of 17 positive (i.e., pleasant) and 17 negative (i.e., unpleasant, or aversive) images taken from the International Affective Picture System (IAPS) 97, which consisted of superficial physical injuries, assaults, traffic accidents or other common traumatic events. The anticipation periods during the task (red and green shapes) lasted 6 seconds and the image presentation lasted 2 seconds. The baseline CPT task was interspersed for variable duration averaging about 8 seconds in between these task components. The total duration of the task was 580 seconds. No response from subjects was required when a picture stimulus was presented on the screen.
Response accuracy and response latency were obtained for the CPT, anticipation of a positive image (API), and anticipation of a negative image (ANI). To examine the behavioral effect of anticipation, we examined the difference between behavioral measures during the API and ANI.
Image Acquisition
During the task, one fMRI run sensitive to blood oxygenation level-dependent (BOLD) contrast was collected for each subject using a Signa EXCITE (GE Healthcare, Milwaukee) 3.0T scanner (T2 * weighted echo planar imaging, TR = 2000 ms, TE = 32 ms, FOV = 250 × 250 mm3, 64 × 64 matrix, 30 2.6mm axial slices with a 1.4mm gap, 290 scans). fMRI acquisitions were time-locked to the onset of each trial. During the same experimental session, a high resolution T1-weighted image (SPGR, TI = 450, TR = 8 ms, TE = 4 ms, flip angle = 12°, FOV = 250 × 250, ~1 mm3 voxels) was obtained for anatomical reference.
Data were preprocessed and analyzed with the Analysis of Functional NeuroImages (AFNI) software package 98. Preprocessed time series data for each individual were analyzed using a multiple regression model. Regressors of interest included four orthogonal regressors that were constructed to quantify the neural substrates contributing to the different components of the task: 1) the API, capturing the anticipation of a positive image, 2) the ANI, capturing the anticipation of a negative image, 3) the positive image (PI) phase, which assesses the processing of positive stimuli, and 4) the negative image (NI) phase, which assesses the processing of negative stimuli. In addition, six nuisance regressors were entered into the linear regression model: three movement-related regressors used to account for residual motion (in the roll, pitch, and yaw direction), a white matter mask to control for physiological noise 99, and regressors for baseline and linear trends used to eliminate slow signal drifts. The CPT task (blue shapes) provided the baseline condition and was accounted for by the baseline regressor. Percent signal change was calculated by dividing the regressor of interest by the baseline regressor. Subsequently, simple contrasts were constructed on an individual subject level for all anticipation (ANI+API) and differential anticipation (DA) of negative versus positive (ANI−API). A Gaussian filter with full width- half maximum 6 mm was applied to the voxel-wise percent signal change data to account for individual variations in the anatomical landmarks. Data of each subject were normalized to Talairach coordinates.
Voxel-wise percent signal change data for whole brain were entered into a paired samples t-test for drug effects during anticipation and image presentation between SSRI dosing and placebo dosing. A threshold adjustment method based on Monte-Carlo simulations was used to guard against identifying false positive areas of activation 100. A prior voxel-wise probability of p< 0.05 in a cluster of 1440 μL resulted in whole brain corrected probability of p <0.05. Finally, the average percent signal difference was extracted from regions of activation that were found to survive this threshold/cluster method and the t-values were calculated with and without education as a covariate. All analyses for the behavioral data were carried out with SPSS 12.0 101.
In addition, a region of interest (ROI) based analysis was performed on several apriori areas of interest: the bilateral insula, bilateral amygdala, ventral ACC, and dorsal ACC. These corrected voxel probabilities are based on Monte Carlo simulations via AFNI’s program AlphaSim, using the filtered data and the a-priori defined regions of interest. Stereotactic coordinates of the ROIs were based on standardized Talairach atlas locations 102. This resulted in minimum clusters sizes of 128 μL for the amygdala ROIs and 256 μL for all remaining ROIs. While the cluster significance is p<.05 for the ROIs, the corrected voxel-wise probabilities are as follows: amygdala p < 0.012, insular cortex p < 0.00007, ventral medial prefrontal cortex p < 0.00014, and dorsal medial prefrontal cortex p < 0.00014.
Correlational analyses were also conducted for the placebo minus escitalopram effects for particular contrasts of interest, including imaging, behavioral, and self-report data.
Results
Behavioral Analysis
Subchronic administration of escitalopram had no significant effect on task performance during the different task conditions (CPT, ANI, and API) as measured by response latency or accuracy (F(1,14)=2.303, p = ns; F(1,14)=0.007, p = ns, respectively). Escitalopram did not alter self report measures of various types of anxiety symptoms or depression (i.e., BDI, BSI, SIAS, STAIS; data not shown) in this group of healthy volunteers.
Brain Activation Analysis
Task Effect
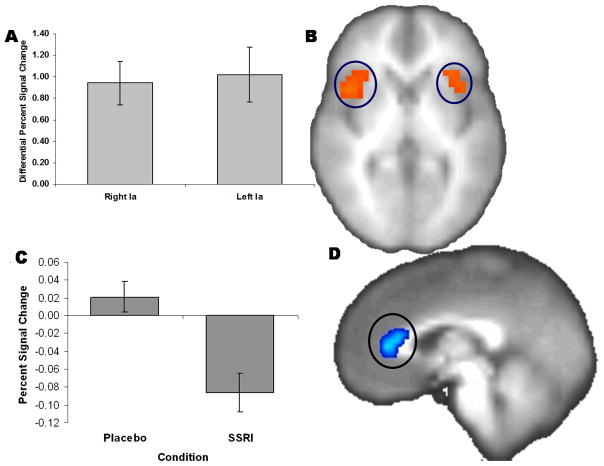
ROI analysis of the task related activation (combined placebo and SSRI) was observed for differential anticipation (DA; ANI−API) in the bilateral anterior insula (Ia) (right Ia: F(1,14)=4.645, p=0.001; left Ia: F(1,14)=4.005, p=0.001; see Figure 2) in the ROI analysis. The bilateral anterior insula regions did not differ significantly across conditions.
Figure 2.
Task activation: greater activation (% signal change) in the bilateral anterior insula for negative anticipation minus positive anticipation [A] shown at . Condition activation: deactivation (% signal change in ventral anterior cingulate [C]) in negative minus positive anticipation during escitalopram versus placebo conditions shown at x=0 [D].
SSRI Effect
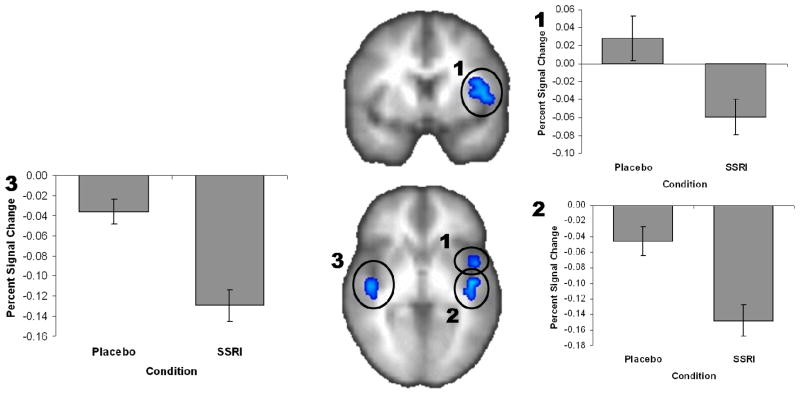
There was a main effect of subchronic administration of escitalopram, which was seen as a relative deactivation during differential anticipation (ANI−API) in the ventral ACC (2624μl, x=5, y=32, z=−12; F(1,14)=3.259, p=0.005; see Figure 2). Moreover, individuals after escitalopram administration relative to the placebo condition showed relative deactivation for all anticipation (ANI+API) trials in the right posterior insula (1344μl, x=42, y=−19, z=3; F(1,14)=4.496, p=0.001), left inferior posterior insula (1088μl, x=−42, y=−17, z=2; F(1,14)=2.855, p=0.05), left superior posterior insula (832μl, x=−41, y=−13, z=25; F(1,14)=3.028, p=0.01), and left middle insula (1216μl, x=−40, y=6, z=−9; F(1,14)=3.011, p=0.01) (see Figure 3).
Figure 3.
Deactivation (% signal change) in positive and negative anticipation during escitalopram versus placebo conditions in the (1) left middle (2) left posterior, and (3) right posterior insula.
Correlations
There were no significant correlations between the change in response latency or accuracy, any self-report measures (i.e., BDI, BSI, SIAS, STAIS), or the change in the degree of activation in the insula or cingulate during the task across drug conditions (data not shown).
Discussion
This experiment yielded three main findings. First, sub-chronic administration of “therapeutic doses” of the SSRI escitalopram resulted in significant relative deactivation of the ventral ACC during anticipation of negative compared to positive visual stimuli. Second, escitalopram reduced middle to posterior bilateral insula activation during anticipation regardless of the valence of the stimulus. Third, we confirmed previous observations22, 23 that bilateral anterior insula is important for anticipation of negative (aversive) compared to positive (pleasant) visual stimuli. Taken together, these results show that escitalopram influences anticipatory processing by modulating insula and ventral ACC activity during emotion processing. This is a compelling hypothetical mechanism by which SSRIs may act as anxiolytics. Therapeutic effects of SSRIs may involve modulation of cues that signal expected emotional states such that they contribute less significantly to emotion processing. These observations add to the growing literature that pharmacofMRI may be useful in revealing effects of well-established anxiolytics and antidepressants in the brain and could thus be a useful tool in the development of novel therapeutics 90.
The current study replicated the relative increases in the bilateral insula during anticipation seen in our prior work 22, 23. The insula has been suggested to play a key role in evaluating the impact that environmental stimuli may have on the interoceptive body state 15, 88. Activity in this region relates to anxiety during risk-taking decision making 103, is elevated in individuals with specific phobia when viewing fearful faces 104, is increased during anticipation of emotion face processing in those with high trait anxiety 23, 105, relates to anticipatory anxiety in those with social phobia 57, and is associated with increased perfusion in patients with panic disorder 106. Taken in combination, these studies suggest that altered insula activity may be a common denominator that could be used as a biomarker for treatment effects. The anterior subdivision of the insula has been highlighted as an important region for the integration of physiological and psychological self 32, 88, and is of particular importance in down-modulating the posterior insula 28. In the present study, activity in the posterior insula was reduced by the administration of escitalopram, suggesting that SSRIs may contribute to central reduction in physiological reactivity during emotional anticipation. Escitalopram did not affect processing within the anterior insula, which is important for the integration of cognitive, affective, and physiological processes. In comparison, escitalopram attenuated the more posterior aspects of the insula, which are important for the physiological representation of potentially aversive emotional experiences. Therefore, escitalopram (and, by inference, other SSRIs) may have a more subtle “bottom up” effect, i.e., modulating the physiological associations of anticipatory stimuli, rather than a “top-down” modulation, i.e., modulating the cognitive attributes of anticipatory stimuli, which is consistent with models proposed by Mayberg and colleagues 107, 108. In fact the model proposed by Mayberg focus3w on the subgenual cingulate, directly inferior to the region found in this study, as being in the critical path for the treatment effects of SSRIs due to the serotonin density of this region 107–111. Slight discrepancy in location of the effects of SSRI in the current study may be due in part to the selection of an anticipatory task to probe brain functioning. As a cautionary note, however, it is important to point out that activation differences between the anticipation and baseline condition in posterior insula regions were mostly negative. This may be due to uncorrected physiological effects such as breathing 99 or alternatively there may be a dampening of somatic information during anticipation112.
We also found a significant attenuation of the ventral ACC during anticipation of negative versus positive visual stimuli. This region is often linked with self-focus and emotional evaluation 44–52, as well as anticipatory processing 19, 24, 59, 113. Thus, the attenuating effects of escitalopram in this region may reflect decreases in self-focus during the anticipation of aversive stimuli. Numerous studies have found an anticipatory activation in the dorsal ACC particularly in contrast with uncued anticipatory phases 25, 26; considering the strong connections between the insula and dorsal ACC/MPFG 29 this relationship may play a part in top-down modulation of interoceptive processing.
Our findings are based on results from healthy volunteers who did not report significant subjective changes during subchronic escitalopram administration. Nevertheless, imaging of healthy volunteers is an important step in proof of concept in drug discovery 114. In particular, the use of a relatively homogenous, healthy population may allow for the use of smaller groups to detect neural effects of a compound. It should be noted that healthy volunteers may show brain changes without behavioral changes 114. Although escitalopram is an approved, marketed drug, proving that sub-chronic doses of SSRIs act on specific neural pathways can provide biomarkers for efficacy in similar drugs entering phase I or II (i.e., safety/efficacy studies). In comparison, the advantage of using patient samples may be the ability to determine the relationship between the neural substrate effects of potential therapeutics and subjective or objective changes in disorder symptoms.
In terms of the mechanistic actions of SSRIs, these findings suggest that direct or indirect serotonergic modulation of insular cortex, among other regions, results in relative deactivation of affective neural substrates during anticipation. Specifically, the mechanism of action could be explained as a reduction of the affective/physiological reactivity to anticipation that may then result in decreased feelings of anxiety and/or depression. Reduction of self-focused attention during negative anticipation may relate to less concern about the internal body state. Given the importance of somatic reactivity in both anxiety and anticipatory processing 3, 4, 115, this mechanism may be considered as a potentially effective way to modulate affect through particular pharmacological interventions. This model would help explain why SSRIs are effective at modification of mood only in conditions of distress, such as anxiety, and in the current study are only seen when the individual is momentarily provoked by an affective anticipatory task that can induce physiological or homeostatic distress.
The current study has several limitations. First, as noted above, this study was conducted with healthy volunteers and generalizability to patient samples still need to be established. In particular, we did not observe changes in subjective ratings on scales measuring anxiety or depression. This range restriction in the emotional state of healthy volunteers may explain the lack of significant correlations between change in psychological measures and change in BOLD signal in functional ROIs during SSRI treatment. Because physiologic reactivity to anticipatory anxiety is greater in anxious individuals 4, 115, future studies with patients suffering from anxiety disorders may reveal even larger BOLD changes during SSRI treatment. Also the neural expression of escitalopram appears to be task dependant both in the mechanism and strength of the effect 116, so these findings should not be over-generalized in its effects.
In summary, our results suggest that treatment with escitalopram results in attenuation of the neural response to affective anticipation in brain regions responsible for the integration of physiological and affective well-being. Specifically, sub-chronic SSRI treatment may reduce the anticipatory reactivity to emotional—especially aversive—stimuli. These findings can have important implications for the development of pharmacological interventions to treat anxiety disorders, suggesting that BOLD signal in the insula and ACC during anticipatory anxiety may be a useful biomarker for measuring psychopharmacological effects of extant and novel anxiolytic agents.
Acknowledgments
Funding: Supported by grants from NIMH (MH65413, MH075792, and MH64122)
References
- 1.Eysenck MW. Anxiety and Cognition: a Unified Theory. Hove, UK: Psychology Press; 1997. [Google Scholar]
- 2.Bradley BP, Mogg K, Lee SC. Attentional biases for negative information in induced and naturally occurring dysphoria. Behaviour research and therapy. 1997;35(10):911–927. doi: 10.1016/s0005-7967(97)00053-3. [DOI] [PubMed] [Google Scholar]
- 3.Masaoka Y, Homma I. The effect of anticipatory anxiety on breathing and metabolism in humans. Respir Physiol. 2001;128(2):171–177. doi: 10.1016/s0034-5687(01)00278-x. [DOI] [PubMed] [Google Scholar]
- 4.Masaoka Y, Onaka Y, Shimizu Y, Sakurai S, Homma I. State Anxiety Dependent on Perspiration during Mental Stress and Deep Inspiration. J Physiol Sci. 2007;57(2):121–126. doi: 10.2170/physiolsci.RP000607. [DOI] [PubMed] [Google Scholar]
- 5.Deakin JF. The role of serotonin in panic, anxiety and depression. International clinical psychopharmacology. 1998;13 (Suppl 4):S1–5. doi: 10.1097/00004850-199804004-00001. [DOI] [PubMed] [Google Scholar]
- 6.Brown SM, Hariri AR. Neuroimaging studies of serotonin gene polymorphisms: exploring the interplay of genes, brain, and behavior. Cogn Affect Behav Neurosci. 2006;6(1):44–52. doi: 10.3758/cabn.6.1.44. [DOI] [PubMed] [Google Scholar]
- 7.Graeff FG, Guimaraes FS, DeAndrade T, Deakin JFW. Role of 5-HT in stress, anxiety, and depression. Pharmacology Biochemistry and Behavior. 1996;54:129–141. doi: 10.1016/0091-3057(95)02135-3. [DOI] [PubMed] [Google Scholar]
- 8.Hariri AR, Brown SM. Serotonin. The American journal of psychiatry. 2006;163(1):12. doi: 10.1176/appi.ajp.163.1.12. [DOI] [PubMed] [Google Scholar]
- 9.Hariri AR, Drabant EM, Weinberger DR. Imaging genetics: perspectives from studies of genetically driven variation in serotonin function and corticolimbic affective processing. Biol Psychiatry. 2006;59(10):888–897. doi: 10.1016/j.biopsych.2005.11.005. [DOI] [PubMed] [Google Scholar]
- 10.Feldman JL, Mitchell GS, Nattie EE. Breathing. Annual Review of Neuroscience. 2003;26:239–266. doi: 10.1146/annurev.neuro.26.041002.131103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hood SD, Hince DA, Robinson H, Cirillo M, Christmas D, Kaye JM. Serotonin regulation of the human stress response. Psychoneuroendocrinology. 2006;31(9):1087–1097. doi: 10.1016/j.psyneuen.2006.07.001. [DOI] [PubMed] [Google Scholar]
- 12.Richter DW, Manzke T, Wilken B, Ponimaskin E. Serotonin receptors: guardians of stable breathing. Trends Mol Med. 2003;9(12):542–548. doi: 10.1016/j.molmed.2003.10.010. [DOI] [PubMed] [Google Scholar]
- 13.Kent JM, Coplan JD, Gorman JM. Clinical utility of the selective serotonin reuptake inhibitors in the spectrum of anxiety. Biol Psychiatry. 1998;44(9):812–824. doi: 10.1016/s0006-3223(98)00210-8. [DOI] [PubMed] [Google Scholar]
- 14.Lanzenberger RR, Mitterhauser M, Spindelegger C, Wadsak W, Klein N, Mien LK, Holik A, Attarbaschi T, Mossaheb N, Sacher J, Geiss-Granadia T, Kletter K, Kasper S, Tauscher J. Reduced serotonin-1A receptor binding in social anxiety disorder. Biol Psychiatry. 2007;61(9):1081–1089. doi: 10.1016/j.biopsych.2006.05.022. [DOI] [PubMed] [Google Scholar]
- 15.Craig AD. Interoception: the sense of the physiological condition of the body. Curr Opin Neurobiol. 2003;13(4):500–505. doi: 10.1016/s0959-4388(03)00090-4. [DOI] [PubMed] [Google Scholar]
- 16.Critchley HD, Mathias CJ, Josephs O, O’Doherty J, Zanini S, Dewar BK, Cipolotti L, Shallice T, Dolan RJ. Human cingulate cortex and autonomic control: converging neuroimaging and clinical evidence. Brain. 2003;126(Pt 10):2139–2152. doi: 10.1093/brain/awg216. [DOI] [PubMed] [Google Scholar]
- 17.Sawamoto N, Honda M, Okada T, Hanakawa T, Kanda M, Fukuyama H, Konishi J, Shibasaki H. Expectation of pain enhances responses to nonpainful somatosensory stimulation in the anterior cingulate cortex and parietal operculum/posterior insula: an event-related functional magnetic resonance imaging study. J Neurosci. 2000;20(19):7438–7445. doi: 10.1523/JNEUROSCI.20-19-07438.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Simpson JR, Jr, Drevets WC, Snyder AZ, Gusnard DA, Raichle ME. Emotion-induced changes in human medial prefrontal cortex: II. During anticipatory anxiety. Proc Natl Acad Sci USA. 2001;98(2):688–693. doi: 10.1073/pnas.98.2.688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Chua P, Krams M, Toni I, Passingham R, Dolan R. A functional anatomy of anticipatory anxiety. Neuroimage. 1999;9(6 Pt 1):563–571. doi: 10.1006/nimg.1999.0407. [DOI] [PubMed] [Google Scholar]
- 20.Ploghaus A, Tracey I, Gati JS, Clare S, Menon RS, Matthews PM, Rawlins JN. Dissociating pain from its anticipation in the human brain. Science (New York, NY) 1999;284(5422):1979–1981. doi: 10.1126/science.284.5422.1979. [DOI] [PubMed] [Google Scholar]
- 21.Critchley HD, Mathias CJ, Dolan RJ. Neural activity in the human brain relating to uncertainty and arousal during anticipation. Neuron. 2001;29(2):537–545. doi: 10.1016/s0896-6273(01)00225-2. [DOI] [PubMed] [Google Scholar]
- 22.Simmons A, Matthews SC, Stein MB, Paulus MP. Anticipation of emotionally aversive visual stimuli activates right insula. Neuroreport. 2004;15(14):2261–2265. doi: 10.1097/00001756-200410050-00024. [DOI] [PubMed] [Google Scholar]
- 23.Simmons A, Strigo I, Matthews SC, Paulus MP, Stein MB. Anticipation of aversive visual stimuli is associated with increased insula activation in anxiety-prone subjects. Biol Psychiatry. 2006;60(4):402–409. doi: 10.1016/j.biopsych.2006.04.038. [DOI] [PubMed] [Google Scholar]
- 24.Nitschke JB, Sarinopoulos I, Mackiewicz KL, Schaefer HS, Davidson RJ. Functional neuroanatomy of aversion and its anticipation. Neuro Image. 2006;29(1):106–116. doi: 10.1016/j.neuroimage.2005.06.068. [DOI] [PubMed] [Google Scholar]
- 25.Bermpohl F, Pascual-Leone A, Amedi A, Merabet LB, Fregni F, Gaab N, Alsop D, Schlaug G, Northoff G. Dissociable networks for the expectancy and perception of emotional stimuli in the human brain. Neuro Image. 2006;30(2):588–600. doi: 10.1016/j.neuroimage.2005.09.040. [DOI] [PubMed] [Google Scholar]
- 26.Bermpohl F, Pascual-Leone A, Amedi A, Merabet LB, Fregni F, Gaab N, Alsop D, Schlaug G, Northoff G. Attentional modulation of emotional stimulus processing: an fMRI study using emotional expectancy. Human brain mapping. 2006;27(8):662–677. doi: 10.1002/hbm.20209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Fox MD, Snyder AZ, Vincent JL, Corbetta M, Van Essen DC, Raichle ME. The human brain is intrinsically organized into dynamic, anticorrelated functional networks. Proc Natl Acad Sci USA. 2005;102(27):9673–9678. doi: 10.1073/pnas.0504136102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Augustine JR. Circuitary and functional aspects of the insular lobe in primates including humans. Brain Res Rev. 1996;22:229–244. doi: 10.1016/s0165-0173(96)00011-2. [DOI] [PubMed] [Google Scholar]
- 29.Dupont S, Bouilleret V, Hasboun D, Semah F, Baulac M. Functional anatomy of the insula: new insights from imaging. Surg Radiol Anat. 2003;25(2):113–119. doi: 10.1007/s00276-003-0103-4. [DOI] [PubMed] [Google Scholar]
- 30.Bermudez-Rattoni F, Ramirez-Lugo L, Gutierrez R, Miranda MI. Molecular signals into the insular cortex and amygdala during aversive gustatory memory formation. Cell Mol Neurobiol. 2004;24(1):25–36. doi: 10.1023/B:CEMN.0000012722.45805.c8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Devinsky O, Morrell MJ, Vogt BA. Contributions of Anterior Cingulate Cortex to Behavior. Brain. 1995;118:279–306. doi: 10.1093/brain/118.1.279. [DOI] [PubMed] [Google Scholar]
- 32.Craig AD. How do you feel? Interoception: the sense of the physiological condition of the body. Nat Rev Neurosci. 2002;3(8):655–666. doi: 10.1038/nrn894. [DOI] [PubMed] [Google Scholar]
- 33.Craig ADB. Forebrain emotional asymmetry: a neuroanatomical basis? Trends Cogn Sci. 2005;9(12):566–571. doi: 10.1016/j.tics.2005.10.005. [DOI] [PubMed] [Google Scholar]
- 34.Ongur D, An X, Price JL. Prefrontal cortical projections to the hypothalamus in macaque monkeys. J Comp Neurol. 1998;401(4):480–505. [PubMed] [Google Scholar]
- 35.Ongur D, Ferry AT, Price JL. Architectonic subdivision of the human orbital and medial prefrontal cortex. J Comp Neurol. 2003;460(3):425–449. doi: 10.1002/cne.10609. [DOI] [PubMed] [Google Scholar]
- 36.Shi CJ, Cassell MD. Cortical, thalamic, and amygdaloid connections of the anterior and posterior insular cortices. J Comp Neurol. 1998;399(4):440–468. doi: 10.1002/(sici)1096-9861(19981005)399:4<440::aid-cne2>3.0.co;2-1. [DOI] [PubMed] [Google Scholar]
- 37.An X, Bandler R, Ongur D, Price JL. Prefrontal cortical projections to longitudinal columns in the midbrain periaqueductal gray in macaque monkeys. J Comp Neurol. 1998;401(4):455–479. [PubMed] [Google Scholar]
- 38.Likhtik E, Pelletier JG, Paz R, Pare D. Prefrontal control of the amygdala. J Neurosci. 2005;25(32):7429–7437. doi: 10.1523/JNEUROSCI.2314-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Phan KL, Taylor SF, Welsh RC, Decker LR, Noll DC, Nichols TE, Britton JC, Liberzon I. Activation of the medial prefrontal cortex and extended amygdala by individual ratings of emotional arousal: a fMRI study. Biol Psychiatry. 2003;53(3):211–215. doi: 10.1016/s0006-3223(02)01485-3. [DOI] [PubMed] [Google Scholar]
- 40.Quirk GJ, Likhtik E, Pelletier JG, Pare D. Stimulation of medial prefrontal cortex decreases the responsiveness of central amygdala output neurons. J Neurosci. 2003;23(25):8800–8807. doi: 10.1523/JNEUROSCI.23-25-08800.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Phan KL, Britton JC, Taylor SF, Fig LM, Liberzon I. Corticolimbic blood flow during nontraumatic emotional processing in posttraumatic stress disorder. Arch Gen Psychiatry. 2006;63(2):184–192. doi: 10.1001/archpsyc.63.2.184. [DOI] [PubMed] [Google Scholar]
- 42.Sotres-Bayon F, Cain CK, LeDoux JE. Brain mechanisms of fear extinction: historical perspectives on the contribution of prefrontal cortex. Biol Psychiatry. 2006;60(4):329–336. doi: 10.1016/j.biopsych.2005.10.012. [DOI] [PubMed] [Google Scholar]
- 43.Britton JC, Phan KL, Taylor SF, Fig LM, Liberzon I. Corticolimbic blood flow in posttraumatic stress disorder during script-driven imagery. Biol Psychiatry. 2005;57(8):832–840. doi: 10.1016/j.biopsych.2004.12.025. [DOI] [PubMed] [Google Scholar]
- 44.Beauregard M, Levesque J, Bourgouin P. Neural correlates of conscious self-regulation of emotion. J Neurosci. 2001;21(18):RC165. doi: 10.1523/JNEUROSCI.21-18-j0001.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Gusnard DA, Akbudak E, Shulman GL, Raichle ME. Medial prefrontal cortex and self-referential mental activity: relation to a default mode of brain function. Proc Natl Acad Sci USA. 2001;98(7):4259–4264. doi: 10.1073/pnas.071043098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Heatherton TF, Wyland CL, Macrae CN, Demos KE, Denny BT, Kelley WM. Medial prefrontal activity differentiates self from close others. Scan. 2006;1 (1):18–25. doi: 10.1093/scan/nsl001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Johnson SC, Schmitz TW, Kawahara-Baccus TN, Rowley HA, Alexander AL, Lee J, Davidson RJ. The cerebral response during subjective choice with and without self-reference. J Cogn Neurosci. 2005;17(12):1897–1906. doi: 10.1162/089892905775008607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Keenan JP, Wheeler MA, Gallup GG, Jr, Pascual-Leone A. Self-recognition and the right prefrontal cortex. Trends Cogn Sci. 2000;4(9):338–344. doi: 10.1016/s1364-6613(00)01521-7. [DOI] [PubMed] [Google Scholar]
- 49.Mars RB, Coles MG, Grol MJ, Holroyd CB, Nieuwenhuis S, Hulstijn W, Toni I. Neural dynamics of error processing in medial frontal cortex. Neuro Image. 2005;28(4):1007–1013. doi: 10.1016/j.neuroimage.2005.06.041. [DOI] [PubMed] [Google Scholar]
- 50.Northoff G, Bermpohl F. Cortical midline structures and the self. Trends Cogn Sci. 2004;8(3):102–107. doi: 10.1016/j.tics.2004.01.004. [DOI] [PubMed] [Google Scholar]
- 51.Wicker B, Ruby P, Royet JP, Fonlupt P. A relation between rest and the self in the brain? Brain Res Brain Res Rev. 2003;43(2):224–230. doi: 10.1016/j.brainresrev.2003.08.003. [DOI] [PubMed] [Google Scholar]
- 52.Amodio DM, Frith CD. Meeting of minds: the medial frontal cortex and social cognition. Nat Rev Neurosci. 2006;7(4):268–277. doi: 10.1038/nrn1884. [DOI] [PubMed] [Google Scholar]
- 53.Bush G, Luu P, Posner MI. Cognitive and emotional influences in anterior cingulate cortex. Trends Cogn Sci. 2000;4(6):215–222. doi: 10.1016/s1364-6613(00)01483-2. [DOI] [PubMed] [Google Scholar]
- 54.Whalen PJ, Bush G, McNally RJ, Wilhelm S, McInerney SC, Jenike MA, Rauch SL. The emotional counting Stroop paradigm: a functional magnetic resonance imaging probe of the anterior cingulate affective division. Biol Psychiatry. 1998;44(12):1219–1228. doi: 10.1016/s0006-3223(98)00251-0. [DOI] [PubMed] [Google Scholar]
- 55.Bremner JD, Vermetten E, Vythilingam M, Afzal N, Schmahl C, Elzinga B, Charney DS. Neural correlates of the classic color and emotional stroop in women with abuse-related posttraumatic stress disorder. Biol Psychiatry. 2004;55(6):612–620. doi: 10.1016/j.biopsych.2003.10.001. [DOI] [PubMed] [Google Scholar]
- 56.Paulus MP, Feinstein JS, Castillo G, Simmons AN, Stein MB. Dose-dependent decrease of activation in bilateral amygdala and insula by lorazepam during emotion processing. Arch Gen Psychiatry. 2005;62(3):282–288. doi: 10.1001/archpsyc.62.3.282. [DOI] [PubMed] [Google Scholar]
- 57.Lorberbaum JP, Kose S, Johnson MR, Arana GW, Sullivan LK, Hamner MB, Ballenger JC, Lydiard RB, Brodrick PS, Bohning DE, George MS. Neural correlates of speech anticipatory anxiety in generalized social phobia. Neuroreport. 2004;15(18):2701–2705. [PubMed] [Google Scholar]
- 58.Kalisch R, Wiech K, Critchley HD, Dolan RJ. Levels of appraisal: a medial prefrontal role in high-level appraisal of emotional material. Neuro Image. 2006;30(4):1458–1466. doi: 10.1016/j.neuroimage.2005.11.011. [DOI] [PubMed] [Google Scholar]
- 59.Bystritsky A, Pontillo D, Powers M, Sabb FW, Craske MG, Bookheimer SY. Functional MRI changes during panic anticipation and imagery exposure. Neuroreport. 2001;12(18):3953–3957. doi: 10.1097/00001756-200112210-00020. [DOI] [PubMed] [Google Scholar]
- 60.Mineka S, Watson D, Clark LA. Comorbidity of anxiety and unipolar mood disorders. Annu Rev Psychol. 1998;49:377–412. doi: 10.1146/annurev.psych.49.1.377. [DOI] [PubMed] [Google Scholar]
- 61.Burke WJ, Gergel I, Bose A. Fixed-dose trial of the single isomer SSRI escitalopram in depressed outpatients. The Journal of clinical psychiatry. 2002;63(4):331–336. doi: 10.4088/jcp.v63n0410. [DOI] [PubMed] [Google Scholar]
- 62.Taylor MJ, Freemantle N, Geddes JR, Bhagwagar Z. Early onset of selective serotonin reuptake inhibitor antidepressant action: systematic review and meta-analysis. Archives of general psychiatry. 2006;63(11):1217–1223. doi: 10.1001/archpsyc.63.11.1217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.van der Linden GJ, Stein DJ, van Balkom AJ. The efficacy of the selective serotonin reuptake inhibitors for social anxiety disorder (social phobia): a meta-analysis of randomized controlled trials. International clinical psychopharmacology. 2000;15 (Suppl 2):S15–23. doi: 10.1097/00004850-200008002-00004. [DOI] [PubMed] [Google Scholar]
- 64.Wade A, Michael Lemming O, Bang Hedegaard K. Escitalopram 10 mg/day is effective and well tolerated in a placebo-controlled study in depression in primary care. International clinical psychopharmacology. 2002;17(3):95–102. doi: 10.1097/00004850-200205000-00001. [DOI] [PubMed] [Google Scholar]
- 65.Waugh J, Goa KL. Escitalopram: a review of its use in the management of major depressive and anxiety disorders. CNS Drugs. 2003;17(5):343–362. doi: 10.2165/00023210-200317050-00004. [DOI] [PubMed] [Google Scholar]
- 66.Vaswani M, Linda FK, Ramesh S. Role of selective serotonin reuptake inhibitors in psychiatric disorders: a comprehensive review. Prog Neuropsychopharmacol Biol Psychiatry. 2003;27(1):85–102. doi: 10.1016/s0278-5846(02)00338-x. [DOI] [PubMed] [Google Scholar]
- 67.Masand PS, Gupta S. Selective serotonin-reuptake inhibitors: an update. Harvard review of psychiatry. 1999;7(2):69–84. [PubMed] [Google Scholar]
- 68.Nutt DJ, Forshall S, Bell C, Rich A, Sandford J, Nash J, Argyropoulos S. Mechanisms of action of selective serotonin reuptake inhibitors in the treatment of psychiatric disorders. Eur Neuropsychopharmacol. 1999;9 (Suppl 3):S81–86. doi: 10.1016/s0924-977x(99)00030-9. [DOI] [PubMed] [Google Scholar]
- 69.Klein N, Sacher J, Geiss-Granadia T, Attarbaschi T, Mossaheb N, Lanzenberger R, Potzi C, Holik A, Spindelegger C, Asenbaum S, Dudczak R, Tauscher J, Kasper S. In vivo imaging of serotonin transporter occupancy by means of SPECT and [123I]ADAM in healthy subjects administered different doses of escitalopram or citalopram. Psychopharmacology. 2006;188(3):263–272. doi: 10.1007/s00213-006-0486-0. [DOI] [PubMed] [Google Scholar]
- 70.Klein N, Sacher J, Geiss-Granadia T, Mossaheb N, Attarbaschi T, Lanzenberger R, Spindelegger C, Holik A, Asenbaum S, Dudczak R, Tauscher J, Kasper S. Higher serotonin transporter occupancy after multiple dose administration of escitalopram compared to citalopram: an [123I]ADAM SPECT study. Psychopharmacology. 2007;191(2):333–339. doi: 10.1007/s00213-006-0666-y. [DOI] [PubMed] [Google Scholar]
- 71.Carrasco JL, Sandner C. Clinical effects of pharmacological variations in selective serotonin reuptake inhibitors: an overview. Int J Clin Pract. 2005;59(12):1428–1434. doi: 10.1111/j.1368-5031.2005.00681.x. [DOI] [PubMed] [Google Scholar]
- 72.Fu CH, Williams SC, Cleare AJ, Brammer MJ, Walsh ND, Kim J, Andrew CM, Pich EM, Williams PM, Reed LJ, Mitterschiffthaler MT, Suckling J, Bullmore ET. Attenuation of the neural response to sad faces in major depression by antidepressant treatment: a prospective, event-related functional magnetic resonance imaging study. Archives of general psychiatry. 2004;61(9):877–889. doi: 10.1001/archpsyc.61.9.877. [DOI] [PubMed] [Google Scholar]
- 73.Harmer CJ, Mackay CE, Reid CB, Cowen PJ, Goodwin GM. Antidepressant drug treatment modifies the neural processing of nonconscious threat cues. Biol Psychiatry. 2006;59(9):816–820. doi: 10.1016/j.biopsych.2005.10.015. [DOI] [PubMed] [Google Scholar]
- 74.Browning M, Reid C, Cowen PJ, Goodwin GM, Harmer C. A single dose of citalopram increases fear recognition in healthy subjects. Journal of psychopharmacology (Oxford, England) 2007 doi: 10.1177/0269881106074062. [DOI] [PubMed] [Google Scholar]
- 75.Bhagwagar Z, Hinz R, Taylor M, Fancy S, Cowen P, Grasby P. Increased 5-HT2A Receptor Binding in Euthymic, Medication-Free Patients Recovered From Depression: A Positron Emission Study With [11C]MDL 100,907. The American journal of psychiatry. 2006;163(9) doi: 10.1176/ajp.2006.163.9.1580. [DOI] [PubMed] [Google Scholar]
- 76.Takahashi H, Yahata N, Koeda M, Takano A, Asai K, Suhara T, Okubo Y. Effects of dopaminergic and serotonergic manipulation on emotional processing: a pharmacological fMRI study. Neuro Image. 2005;27(4):991–1001. doi: 10.1016/j.neuroimage.2005.05.039. [DOI] [PubMed] [Google Scholar]
- 77.Del-Ben CM, Deakin JF, McKie S, Delvai NA, Williams SR, Elliott R, Dolan M, Anderson IM. The effect of citalopram pretreatment on neuronal responses to neuropsychological tasks in normal volunteers: an FMRI study. Neuropsychopharmacology. 2005;30(9):1724–1734. doi: 10.1038/sj.npp.1300728. [DOI] [PubMed] [Google Scholar]
- 78.Harmer CJ, Shelley NC, Cowen PJ, Goodwin GM. Increased positive versus negative affective perception and memory in healthy volunteers following selective serotonin and norepinephrine reuptake inhibition. The American journal of psychiatry. 2004;161(7):1256–1263. doi: 10.1176/appi.ajp.161.7.1256. [DOI] [PubMed] [Google Scholar]
- 79.Fernandez M, Pissiota A, Frans O, von Knorring L, Fischer H, Fredrikson M. Brain function in a patient with torture related post-traumatic stress disorder before and after fluoxetine treatment: a positron emission tomography provocation study. Neurosci Lett. 2001;297(2):101–104. doi: 10.1016/s0304-3940(00)01674-8. [DOI] [PubMed] [Google Scholar]
- 80.Van der Linden G, van Heerden B, Warwick J, Wessels C, van Kradenburg J, Zungu-Dirwayi N, Stein DJ. Functional brain imaging and pharmacotherapy in social phobia: single photon emission computed tomography before and after treatment with the selective serotonin reuptake inhibitor citalopram. Prog Neuropsychopharmacol Biol Psychiatry. 2000;24(3):419–438. doi: 10.1016/s0278-5846(99)00109-8. [DOI] [PubMed] [Google Scholar]
- 81.Arce E, Miller DA, Feinstein JS, Stein MB, Paulus MP. Lorazepam dose-dependently decreases risk-taking related activation in limbic areas. Psychopharmacology. 2006;189(1):105–116. doi: 10.1007/s00213-006-0519-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Vollm B, Richardson P, McKie S, Elliott R, Deakin JF, Anderson IM. Serotonergic modulation of neuronal responses to behavioural inhibition and reinforcing stimuli: an fMRI study in healthy volunteers. The European journal of neuroscience. 2006;23(2):552–560. doi: 10.1111/j.1460-9568.2005.04571.x. [DOI] [PubMed] [Google Scholar]
- 83.Sheline YI, Barch DM, Donnelly JM, Ollinger JM, Snyder AZ, Mintun MA. Increased amygdala response to masked emotional faces in depressed subjects resolves with antidepressant treatment: an fMRI study. Biol Psychiatry. 2001;50(9):651–658. doi: 10.1016/s0006-3223(01)01263-x. [DOI] [PubMed] [Google Scholar]
- 84.Chen CH, Ridler K, Suckling J, Williams S, Fu CH, Merlo-Pich E, Bullmore E. Brain Imaging Correlates of Depressive Symptom Severity and Predictors of Symptom Improvement After Antidepressant Treatment. Biol Psychiatry. 2007 doi: 10.1016/j.biopsych.2006.09.018. [DOI] [PubMed] [Google Scholar]
- 85.Fu CH, Williams SC, Brammer MJ, Suckling J, Kim J, Cleare AJ, Walsh ND, Mitterschiffthaler MT, Andrew CM, Pich EM, Bullmore ET. Neural responses to happy facial expressions in major depression following antidepressant treatment. The American journal of psychiatry. 2007;164(4):599–607. doi: 10.1176/ajp.2007.164.4.599. [DOI] [PubMed] [Google Scholar]
- 86.Rose EJ, Simonotto E, Spencer EP, Ebmeier KP. The effects of escitalopram on working memory and brain activity in healthy adults during performance of the n-back task. Psychopharmacology. 2006;185(3):339–347. doi: 10.1007/s00213-006-0334-2. [DOI] [PubMed] [Google Scholar]
- 87.Critchley HD. The human cortex responds to an interoceptive challenge. Proc Natl Acad Sci USA. 2004;101(17):6333–6334. doi: 10.1073/pnas.0401510101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Paulus MP, Stein MB. An insular view of anxiety. Biol Psychiatry. 2006;60(4):383–387. doi: 10.1016/j.biopsych.2006.03.042. [DOI] [PubMed] [Google Scholar]
- 89.Etkin A, Wager TD. Functional Neuroimaging of Anxiety: A Meta-Analysis of Emotional Processing in PTSD, Social Anxiety Disorder, and Specific Phobia. The American journal of psychiatry. 2007;164(10):1476–1488. doi: 10.1176/appi.ajp.2007.07030504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Paulus MP, Stein MB. Role of functional magnetic resonance imaging in drug discovery. Neuropsychology review. 2007;17(2):179–188. doi: 10.1007/s11065-007-9023-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.First MB, Spitzer RL, Gibbon M, Williams JBW. Structured Clinical Interview for DSM-IV-TR Axis I Disorders, Research Version, Non-patient Edition. (SCID-I/NP) New York Biometrics Research, New York State Psychiatric Institute; 2002. [Google Scholar]
- 92.Spielberger CD, Gorsuch RL, Lushene R, Vagg PR, Jacobs GA. Manual for the State-Trait Anxiety Inventory. Palo Alto, CA: Consulting Psychologists; 1983. [Google Scholar]
- 93.Beck AT, Ward CH, Medelson M, Mock J, Erbaugh J. An inventory for measuring depression. Archives of general psychiatry. 1961;4:561–571. doi: 10.1001/archpsyc.1961.01710120031004. [DOI] [PubMed] [Google Scholar]
- 94.Mattick RP, Clarke JC. Development and validation of measures of social phobia scrutiny fear and social interaction anxiety. Behaviour research and therapy. 1998;36(4):455–470. doi: 10.1016/s0005-7967(97)10031-6. [DOI] [PubMed] [Google Scholar]
- 95.Derogatis LR, Melisaratos N. The Brief Symptom Inventory: an introductory report. Psychol Med. 1983;13(3):595–605. [PubMed] [Google Scholar]
- 96.Huettel SA, Mack PB, McCarthy G. Perceiving patterns in random series: dynamic processing of sequence in prefrontal cortex. Nat Neurosci. 2002;5(5):485–490. doi: 10.1038/nn841. [DOI] [PubMed] [Google Scholar]
- 97.Lang PJ, Bradley MM, Cuthbert BN. Emotion and motivation: Measuring affective perception. Journal of Clinical Neurophysiology. 1998;15(5):397–408. doi: 10.1097/00004691-199809000-00004. [DOI] [PubMed] [Google Scholar]
- 98.Cox RW. AFNI: software for analysis and visualization of functional magnetic resonance neuroimages. Comput Biomed Res. 1996;29(3):162–173. doi: 10.1006/cbmr.1996.0014. [DOI] [PubMed] [Google Scholar]
- 99.Strigo I, Simmons A, Craig AD, Paulus MP. Society for Neuroscience. Atlanta, GA: 2006. Breathing and BOLD fMRI: watch out. [Google Scholar]
- 100.Forman SD, Cohen JD, Fitzgerald M, Eddy WF, Mintun MA, Noll DC. Improved assessment of significant activation in functional magnetic resonance imaging (fMRI): use of a cluster-size threshold. Magn Reson Med. 1995;33(5):636–647. doi: 10.1002/mrm.1910330508. [DOI] [PubMed] [Google Scholar]
- 101.SPSS for Windows [computer program]. Version Rel. 12.0.0. Chicago: SPSS Inc; 2001. [Google Scholar]
- 102.Talairach J, Tournoux P. Co-planar Stereotaxic Atlas of the Human Brain: 3-Dimensional Proportional System: An Approach to Cerebral Imaging. New York, NY: Thieme Medical Publishers, Inc; 1998. [Google Scholar]
- 103.Paulus MP, Rogalsky C, Simmons A, Feinstein JS, Stein MB. Increased activation in the right insula during risk-taking decision making is related to harm avoidance and neuroticism. Neuro Image. 2003;19(4):1439–1448. doi: 10.1016/s1053-8119(03)00251-9. [DOI] [PubMed] [Google Scholar]
- 104.Wright CI, Martis B, McMullin K, Shin LM, Rauch SL. Amygdala and insular responses to emotionally valenced human faces in small animal specific phobia. Biol Psychiatry. 2003;54(10):1067–1076. doi: 10.1016/s0006-3223(03)00548-1. [DOI] [PubMed] [Google Scholar]
- 105.Stein MB, Simmons AN, Feinstein JS, Paulus MP. Increased Amygdala and Insula Activation During Emotion Processing in Anxiety-Prone Subjects. Am J Psychiatry. 2006 doi: 10.1176/ajp.2007.164.2.318. [DOI] [PubMed] [Google Scholar]
- 106.Malizia AL, Cunningham VJ, Bell CJ, Liddle PF, Jones T, Nutt DJ. Decreased brain GABA(A)-benzodiazepine receptor binding in panic disorder: preliminary results from a quantitative PET study. Arch Gen Psychiatry. 1998;55(8):715–720. doi: 10.1001/archpsyc.55.8.715. [DOI] [PubMed] [Google Scholar]
- 107.Ressler KJ, Mayberg HS. Targeting abnormal neural circuits in mood and anxiety disorders: from the laboratory to the clinic. Nature neuroscience. 2007;10(9):1116–1124. doi: 10.1038/nn1944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Mayberg HS. Modulating limbic-cortical circuits in depression: targets of antidepressant treatments. Semin Clin Neuropsychiatry. 2002;7(4):255–268. doi: 10.1053/scnp.2002.35223. [DOI] [PubMed] [Google Scholar]
- 109.Kennedy SH, Evans KR, Kruger S, Mayberg HS, Meyer JH, McCann S, Arifuzzman AI, Houle S, Vaccarino FJ. Changes in regional brain glucose metabolism measured with positron emission tomography after paroxetine treatment of major depression. Am J Psychiatry. 2001;158(6):899–905. doi: 10.1176/appi.ajp.158.6.899. [DOI] [PubMed] [Google Scholar]
- 110.Meyer JH, Kapur S, Eisfeld B, Brown GM, Houle S, DaSilva J, Wilson AA, Rafi-Tari S, Mayberg HS, Kennedy SH. The effect of paroxetine on 5-HT(2A) receptors in depression: an [(18)F]setoperone PET imaging study. Am J Psychiatry. 2001;158(1):78–85. doi: 10.1176/appi.ajp.158.1.78. [DOI] [PubMed] [Google Scholar]
- 111.Goldapple K, Segal Z, Garson C, Lau M, Bieling P, Kennedy S, Mayberg H. Modulation of cortical-limbic pathways in major depression: treatment-specific effects of cognitive behavior therapy. Arch Gen Psychiatry. 2004;61(1):34–41. doi: 10.1001/archpsyc.61.1.34. [DOI] [PubMed] [Google Scholar]
- 112.Obrist PA, Webb RA, Sutterer JR. Heart rate and somatic changes during aversive conditioning and a simple reaction time task. Psychophysiology. 1969;5(6):696–723. doi: 10.1111/j.1469-8986.1969.tb02872.x. [DOI] [PubMed] [Google Scholar]
- 113.Porro CA, Baraldi P, Pagnoni G, Serafini M, Facchin P, Maieron M, Nichelli P. Does anticipation of pain affect cortical nociceptive systems? J Neurosci. 2002;22(8):3206–3214. doi: 10.1523/JNEUROSCI.22-08-03206.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Wise RG, Tracey I. The role of fMRI in drug discovery. J Magn Reson Imaging. 2006;23(6):862–876. doi: 10.1002/jmri.20584. [DOI] [PubMed] [Google Scholar]
- 115.Masaoka Y, Homma I. Anxiety and respiratory patterns: their relationship during mental stress and physical load. International Journal of Psychophysiology. 1997;27(2):153–159. doi: 10.1016/s0167-8760(97)00052-4. [DOI] [PubMed] [Google Scholar]
- 116.Arce E, Simmons AN, Lovero KL, Stein MB, Paulus MP. Escitalopram effects on insula and amygdala BOLD activation during emotional processing. Psychopharmacology. 2007 doi: 10.1007/s00213-007-1004-8. [DOI] [PMC free article] [PubMed] [Google Scholar]