Abstract
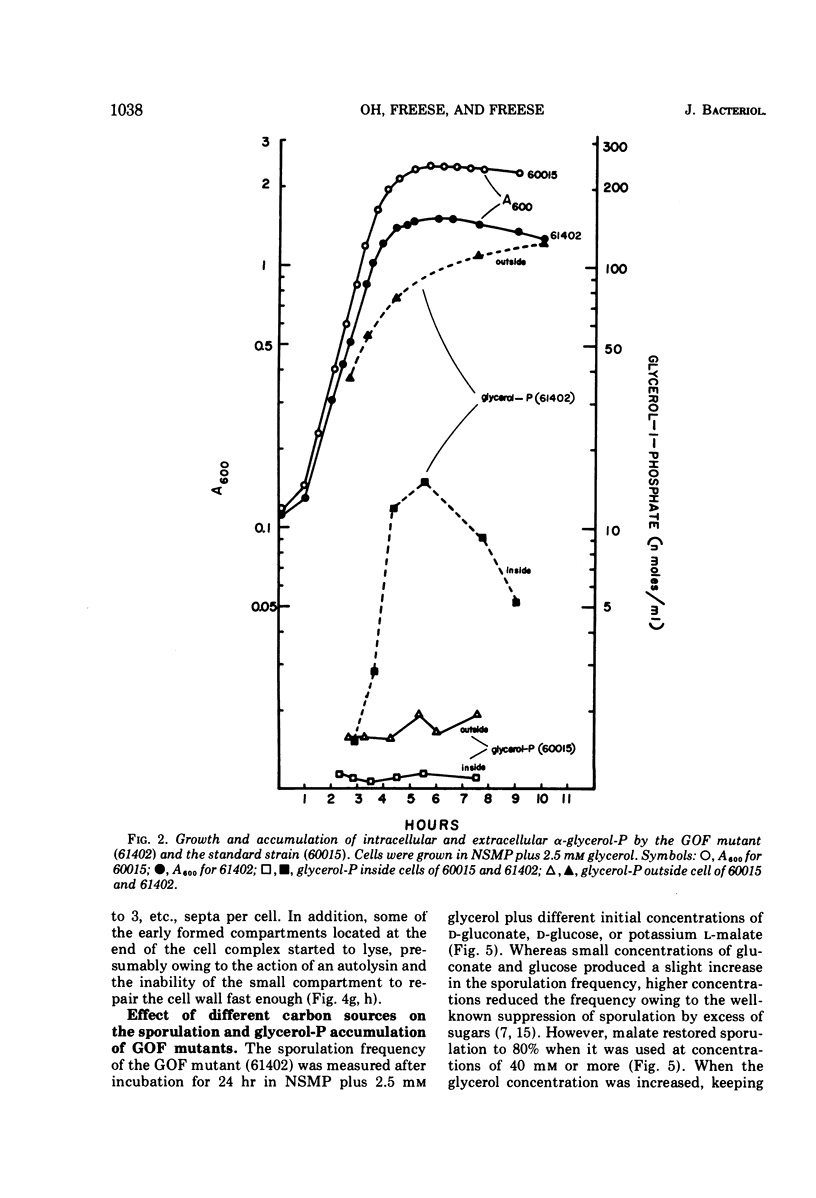
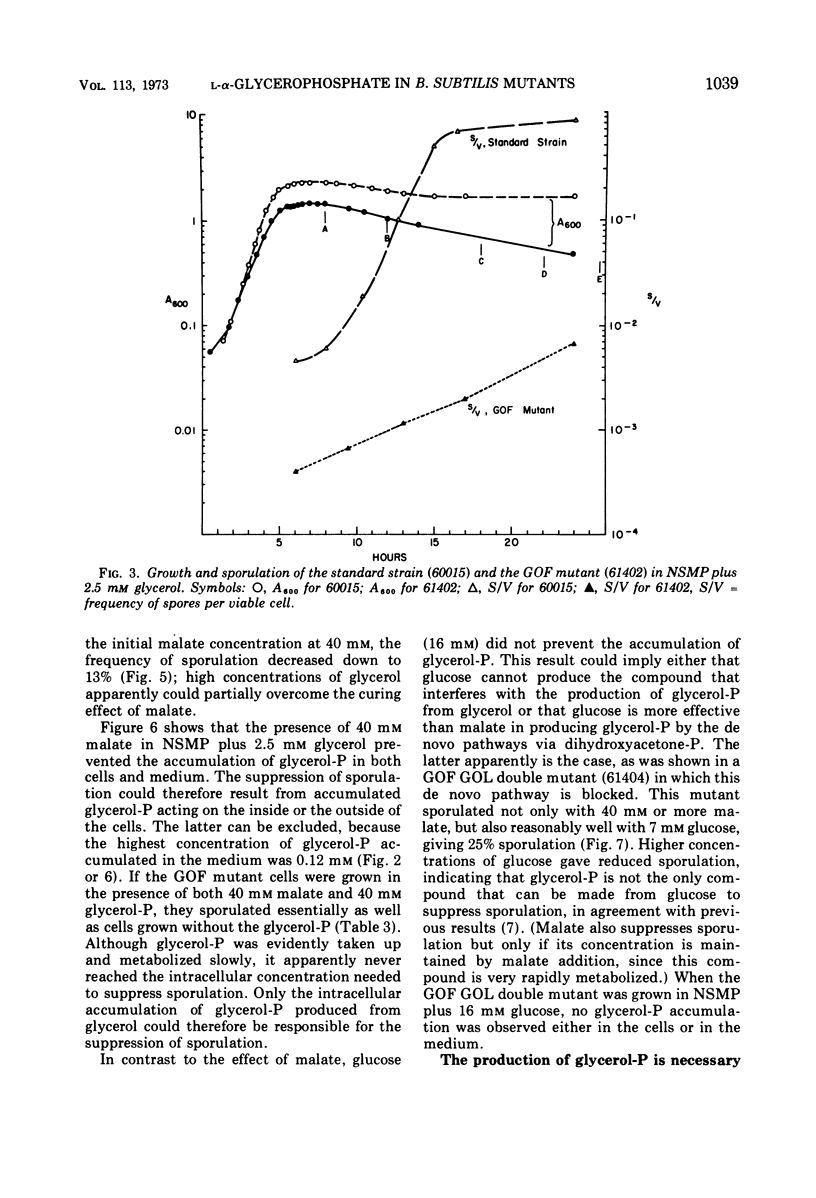
Accumulation of l-α-glycerophosphate, in cells of Bacillus subtilis mutants lacking the nicotinamide adenine dinucleotide-independent glycerophosphate dehydrogenase activity, suppresses both growth and sporulation. After growth has stopped, the cells slowly develop one and later more asymmetric septa that are thicker than normal prespore septa and apparently contain too much cell wall material to allow further membrane development into forespores or spores. l-Malate prevents accumulation of glycerophosphate and restores sporulation of the mutant. Glucose or gluconate cannot resotre sporulation, because they still effect glycerophosphate accumulation via de novo synthesis. If that accumulation is blocked in a double mutant, which is unable to make glycerophosphate from or to metabolize it into Embden-Meyerhof compounds, then nonsuppressing amounts of glucose or gluconate can restore sporulation.
Full text
PDF









Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Berman M., Zwaig N., Lin E. C. Suppression of a pleiotropic mutant affecting glycerol dissimilation. Biochem Biophys Res Commun. 1970 Jan 23;38(2):272–278. doi: 10.1016/0006-291x(70)90708-4. [DOI] [PubMed] [Google Scholar]
- Fortnagel P., Freese E. Analysis of sporulation mutants. II. Mutants blocked in the citric acid cycle. J Bacteriol. 1968 Apr;95(4):1431–1438. doi: 10.1128/jb.95.4.1431-1438.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freese E. B., Cole R. M., Klofat W., Freese E. Growth, sporulation, and enzyme defects of glucosamine mutants of Bacillus subtilis. J Bacteriol. 1970 Mar;101(3):1046–1062. doi: 10.1128/jb.101.3.1046-1062.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freese E., Klofat W., Galliers E. Commitment to sporulation and induction of glucose-phosphoenolpyruvate-transferase. Biochim Biophys Acta. 1970 Nov 24;222(2):265–289. doi: 10.1016/0304-4165(70)90115-7. [DOI] [PubMed] [Google Scholar]
- Freese E. Sporulation of bacilli, a model of cellular differentiation. Curr Top Dev Biol. 1972;7:85–124. doi: 10.1016/s0070-2153(08)60070-8. [DOI] [PubMed] [Google Scholar]
- KOCH J. P., HAYASHI S., LIN E. C. THE CONTROL OF DISSIMILATION OF GLYCEROL AND L-ALPHA-GLYCEROPHOSPHATE IN ESCHERICHIA COLI. J Biol Chem. 1964 Sep;239:3106–3108. [PubMed] [Google Scholar]
- LIN E. C., KOCH J. P., CHUSED T. M., JORGENSEN S. E. Utilization of L-alpha-glycerophosphate by Escherichia coli without hydrolysis. Proc Natl Acad Sci U S A. 1962 Dec 15;48:2145–2150. doi: 10.1073/pnas.48.12.2145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Mindich L. Membrane synthesis in Bacillus subtilis. I. Isolation and properties of strains bearing mutations in glycerol metabolism. J Mol Biol. 1970 Apr 28;49(2):415–432. doi: 10.1016/0022-2836(70)90254-8. [DOI] [PubMed] [Google Scholar]
- Ryter A., Schaeffer P., Ionesco H. Classification cytologique, par leur stade de blocage, des mutants de sporulation de Bacillus subtilis Marburg. Ann Inst Pasteur (Paris) 1966 Mar;110(3):305–315. [PubMed] [Google Scholar]
- Schaeffer P. Sporulation and the production of antibiotics, exoenzymes, and exotonins. Bacteriol Rev. 1969 Mar;33(1):48–71. doi: 10.1128/br.33.1.48-71.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waites W. M., Kay D., Dawes I. W., Wood D. A., Warren S. C., Mandelstam J. Sporulation in Bacillus subtilis. Correlation of biochemical events with morphological changes in asporogenous mutants. Biochem J. 1970 Jul;118(4):667–676. doi: 10.1042/bj1180667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- YOUNG I. E. CHARACTERISTICS OF AN ABORTIVELY DISPORIC VARIANT OF BACILLUS CEREUS. J Bacteriol. 1964 Jul;88:242–254. doi: 10.1128/jb.88.1.242-254.1964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamamoto T., Balassa G. Biochemical genetics of bacterial sporulation. II. Membrane development suring sporulation of B. subtilis and its mutants. Mol Gen Genet. 1969;106(1):1–13. doi: 10.1007/BF00332816. [DOI] [PubMed] [Google Scholar]



