Summary
Ureteric bud (UB) branching during kidney development determines the final number of nephrons. Although hepatocyte growth factor and its receptor Met have been shown to stimulate branching morphogenesis in explanted embryonic kidneys, loss of Met expression is lethal during early embryogenesis without obvious kidney abnormalities. Metfl/fl;HoxB7-Cre mice, which lack Met expression selectively in the UB, were generated and found to have a reduction in final nephron number. These mice have increased Egf receptor expression in both the embryonic and adult kidney, and exogenous Egf can partially rescue the branching defect seen in kidney explants. Metfl/fl;HoxB7-Cre;wa-2/wa-2 mice, which lack normal Egfr and Met signaling, exhibit small kidneys with a marked decrease in UB branching at E14.5 as well as a reduction in final glomerular number. These mice developed progressive interstitial fibrosis surrounding collecting ducts with kidney failure and death by 3-4 weeks of age. Thus, in support of previous in vitro findings, Met and the Egf receptor can act cooperatively to regulate UB branching and mediate maintenance of the normal adult collecting duct.
Keywords: Kidney, Met receptor, Ureteric bud, Branching, Mouse
INTRODUCTION
Kidney development occurs via a series of carefully orchestrated interactions between the ureteric bud and the adjacent metanephric mesenchyme. The ureteric bud branches to form the collecting system of the kidney (collecting duct through ureter), whereas the more proximal parts of the nephron (glomerulus through the connecting segment) develop via mesenchymal-epithelial transformation of induced mesenchyme at the tips of ureteric bud branches (Kobayashi et al., 2008; Saxen, 1987). The result of this interplay is that the final nephron number is determined by the signals that regulate ureteric bud branching. In vitro studies have demonstrated that a number of factors may play a role in either stimulating (Gdnf, Fgf2, Fgf7, Fgf10, Hgf, Egf and pleiotropin) or inhibiting (Bmp4, activin A and semiphorin) this process (Bates, 2007; Sainio et al., 1997; Sakurai et al., 2001; Tufro et al., 2007). However, in vivo studies using knockout technologies have, to date, supported only a few of these as key regulators of UB branching (reviewed by Costantini, 2006).
The process of UB branching requires the proliferation and migration of cells at the UB tip, as well as alterations in tissue morphology that create the branch point. This process has been partially modeled in vitro by first growing renal epithelial cells into cysts, and then stimulating them with factors that induce branching morphogenesis to form tubules sprouting from the cysts (Pollack et al., 1998). One of the factors that has been most heavily studied in these in vitro models of branching morphogenesis is Hgf (Montesano et al., 1991). Hgf binds to the Met tyrosine kinase receptor and activates downstream signaling via Erk, PI 3-kinase, Pkc, Plc, Src, Fak and Jak/Stat that are crucial for the cytoskeletal remodeling, focal adhesion turnover and cell-cell junction reshaping that is necessary for branching morphogenesis (Ishibe et al., 2006; Ishibe et al., 2004; Ishibe et al., 2003; Karihaloo et al., 2005; Liu et al., 2002; O'Brien et al., 2004; Ponzetto et al., 1994; Rosario and Birchmeier, 2004; Weidner et al., 1993).
In the developing kidney, Hgf is expressed by the metanephric mesenchyme whereas the Met receptor is present on both UB and mesenchymal cells (Woolf et al., 1995). Furthermore, in embryonic kidney explant studies, the addition of neutralizing antibodies that block endogenously produced Hgf inhibited explant growth and UB branching (Santos et al., 1994; Woolf et al., 1995). Based on these findings, it was predicted that Hgf signaling would play a significant role in the regulation of UB branching. However, loss of either Hgf or Met expression in the mouse led to death by embryonic day 12-13 due to liver and placental abnormalities (Bladt et al., 1995; Schmidt et al., 1995; Uehara et al., 1995). In these early embryos, limb bud development was significantly impaired but early kidney development, including initial UB branching, appeared to occur normally.
As UB branching continues through E18 (Saxen and Sariola, 1987), we used a conditional knockout approach to more accurately study the role of Hgf-Met signaling in the development of the kidney collecting system, and specifically in the regulation of final nephron number. Selective loss of Met receptor expression in the collecting system of the kidney was achieved using a Cre-loxP approach. The collecting system morphology was not demonstrably abnormal in these mice, but there was a 35% reduction in nephron number at 12 weeks of age and glomerular hypertrophy by 1 year of age. Examination of the collecting ducts revealed that there was sustained upregulation and activation of the Egf receptor, and addition of Egf to explanted kidneys from E12.5 Metfl/fl;HoxB7-Cre mice rescued the decrease in ex vivo UB branching that was observed. Metfl/fl;HoxB7-Cre:wa-2/wa-2 mice lacking both Met and Egfr signaling in the collecting duct demonstrated a marked decrease in UB branching, small kidneys, renal failure and early death.
MATERIALS AND METHODS
Reagents
Antibodies to Met and Aqp2 were obtained from Santa Cruz Biotechnology (San Diego, CA, USA). Anti-Egf and E-cadherin antibodies were obtained from BD Biosciences (San Jose, CA, USA). Anti-pEgfr 992 and 1068 and pErk5 antibodies were obtained from Cell Signaling Technology (Boston, MA, USA). Dolichos biflorus agglutinin (DBA) was obtained from Vector Laboratories (Burlingame, CA, USA).
Creation and genotyping of conditional Met knock-out mice
The Metfl/fl mouse was developed on the 129SV/C57Bl/6 background as described (Huh et al., 2004). HoxB7-Cre mice on the C57Bl/6 background were purchased from Jackson Laboratory (Bar Harbor, ME, USA). Tail genotyping was performed using the Met forward primer (F) 5′-tta ggc aat gag gtg tcc cac-3′ and reverse primer (R) 5′-cca ggt ggc ttc aaa ttc taa gg-3′. To detect deletion of Met in the collecting duct, primer 5′-cag ccg tca gac aat tgg cac-3′ and primer 5′-cca ggt ggc ttc aaa ttc taa gg-3′ were used. The expected sizes of wild-type allele, floxed allele and deleted allele were 380 bp, 300 bp and 650 bp, respectively. To determine Cre expression, CreF 5′-ccgggctgccacgaccaa-3′ and CreR 5′-ggcgcggcaacaacattttt-3′ primers were used, generating a 400 bp fragment. In all experiments, homozygous littermates from the same breeding pair were used as controls. Waved 2 (wa-2) mice on a C57BL/6JEiC3H/HeSnJ/CD-1 background were purchased from Jackson Laboratory. Genotyping of wa-2 mice was performed using wa-2F (5′-ata acc tga cac ttg tca gag tac-3′) and wa-2 R (5′-ttt gca atc tgc aca cac cag ttg-3′) primers followed by digestion with FokI. The expected sizes for wild-type and wa-2 alleles are 326 bp and 160 bp, respectively. All mouse experiments were performed under approval of the Yale IACUC.
Protein isolation and western analysis
Mice were anesthetized by intraperitoneal injection of ketamine (100 mg/kg) and xylazine (20 mg/kg), kidneys extracted and renal papilla surgically removed. The papilla was homogenized in saline with EDTA-free protease inhibitor (Complete; Roche, Indianapolis, IN, USA) using a Dounce homogenizer and centrifuged at 100,000 g. The pellet was resuspended in 100 μl of PBS and 40 μg of protein/sample separated by SDS-PAGE, electrophoretically transferred to Immobilon-P membranes (Millipore, Billerica, MA, USA), immunoblotted with the appropriate antibody and visualized by enhanced chemiluminescence (ECL; Amersham Biosciences, Pittsburgh, PA, USA).
Kidney immunofluorescence and histology
Mice were anesthetized by intraperitoneal injection of ketamine and xylazine followed by perfusion fixation with 40 ml of 4% PFA. The kidneys were frozen, sectioned at 4 μm, and subjected to antigen retrieval (Retrievagen; BD Biosciences, San Jose, CA, USA) followed by blocking with 1% bovine serum albumin for 1 hour. Immunostaining was performed with the appropriate primary antibody overnight at 4°C, followed by Alexaflour donkey anti-rabbit 488 or donkey anti-goat 594 secondary antibodies for visualization. 4,6,-diamidino-2-phenylindole (DAPI) was included in the mounting medium as a counterstain (Vector Laboratories, Burlingame, CA, USA). For kidney histology, mice were perfusion fixed as above followed by Hematoxylin and Eosin or Trichrome staining performed by the Yale Pathology Department. E17 kidneys were harvested, fixed with 4% PFA and cryosections stained with anti-Egfr antibody and DBA lectin.
Calculation of glomerular size
Hematoxylin and Eosin stained sections from kidneys of 12-week-old and 1-year-old male mice were used to calculate the relative glomerular cross-sectional area. Quantitation of 15 randomly chosen juxtamedullary glomeruli was performed in a blinded fashion by two independent investigators on sections from two separate mice for each age and genotype. Image J software was used to determine the relative area in square pixels. To determine total glomeruli/kidney cross-section, all glomeruli in 15 sagittal sections from Met+/+, Metfl/fl;HoxB7-Cre and Metflfl;HoxB7-Cre;wa-2/wa-2 mice were counted and averaged.
Glomeruli isolation for quantitation of nephron number
Mice were anesthetized by intraperitoneal injection of ketamine and xylazine then perfused through the heart with 7.5×107 dynabeads diluted in 40 ml Hanks Buffered Saline Solution. The kidneys were removed and digested in collagenase (1 mg/ml) containing 100 U/ml deoxyribonuclease I in HBSS at 37°C for 30 minutes with gentle agitation. The collagenase-digested tissue was pressed through a 100 μm cell strainer using a pestle and washed with 5 ml HBSS. The cell suspension was centrifuged at 200 g for 5 minutes. The supernatant was discarded and the cell pellet resuspended in 1 ml HBSS. The suspension containing the dynabeads was harvested with a magnet and washed three times with HBSS and aliquots counted under the microscope in a blinded fashion.
Kidney explant culture
E12.5 embryos were harvested from pregnant Metfl/+;HoxB7-Cre females mated with Metfl/+;HoxB7-Cre males. To ensure that the gestational age was identical, only embryos from the same female were directly compared. The embryos were microdissected and kidneys cultured on a Transwell clear polyester filter (0.4 mm; Costar) with L-15 Leibovitz Medium for 2 days at 37°C with or without Egf (20 ng/ml; Sigma Chemical Company, St Louis, MO, USA). The embryonic kidneys were then incubated for 30 minutes with 0.050% saponin followed by 0.050% saponin with 0.1% gelatin overnight. Kidneys were then incubated with FITC-conjugated Dolichos biflorus (DBA) (Vector, Burlingame, CA, USA) at a 1:40 dilution for 24 hours at 37°C in the saponin/gelatin mixture, washed with saponin three times and then visualized using Nikon Epiflourescence Microscopy.
Terminal ureteric bud branches were quantified for each explant in a blinded fashion. Kidney size in square pixels was determined using Image J software in a blinded fashion. Embryos were genotyped as above using tail tissue.
Quantitative PCR
Kidneys were obtained on E14.5 from Metfl/fl;HoxB7-Cre and Met+/+;HoxB7-Cre littermates and total RNA isolated using the RNeasy kit (QIAGEN, Valencia, CA). Total RNA was extracted using the RNeasy Kit (QIAGEN) and 1 μg of RNA was reverse transcribed using random hexamer primers according to the manufacturer's instructions (SuperScript II, Invitrogen). qPCR was conducted using power SYBR green mix (Applied Biosystems) with a 7300 AB Real-time PCR machine (Applied Biosystems). The primers used for PCR were selected for an efficiency of 90-100%, details can be provided on request. Results for each factor were normalized to Gapdh expression from the same PCR reaction (dCt) and then the expression in Metfl/fl;HoxB7-Cre kidneys plotted relative to expression in control Met+/+;HoxB7-Cre kidneys (2-ddCt). A value of 1 would indicate equal expression in both genotypes.
Clinical chemistry and statistics
Plasma and urine electrolytes were analyzed using the Yale University School of Medicine Core Mouse Metabolic Phenotyping Center. All data are expressed as mean±s.e.m. Statistical significance was determined using the Student's t-test.
RESULTS
Collecting duct-specific deletion of the Met receptor
Metfl/fl mice, in which exon 16 of the Met gene is flanked by loxP sites (Huh et al., 2004), were mated with HoxB7-Cre mice that express the Cre recombinase in the Wolffian duct and ureteric bud-derived structures (Yu et al., 2002) (Fig. 1A). Offspring that were heterozygous for the floxed Met allele (Metfl/+;HoxB7-Cre) were mated to generate Metfl/fl;HoxB7-Cre and Met+/+;HoxB7-Cre offspring, confirmed by DNA genotyping of the tail (Fig. 1B). Metfl/fl;HoxB7-Cre mice were born in the expected Mendelian frequency, comprising 18% of the offspring (12/68). PCR analysis of DNA from the renal papilla (comprised primarily of collecting duct and thin limb segments), liver and heart revealed that exon 16 of Met had been deleted in cells from the renal papilla but not in other organs (Fig. 1C).
Fig. 1.

Collecting duct knockout of Met expression. (A) Schematic of the Metfl/fl allele showing the location of the loxP sites flanking exon 16 and the primers (A,B,C) used for genotyping. (B) PCR of tail DNA using primers B and C reveals the expected band at 300 bp in wild-type mice, 380 bp in Metfl/fl mice, and both bands in heterozygotes. All three contain the Cre recombinase gene. (C) PCR of DNA from the heart (H), liver (L) and kidney papilla (KP) of Metfl/fl;HoxB7-Cre mice using primers A and C reveals the selective excision of the floxed allele in the kidney to generate the smaller MetΔ16/Δ16 fragment (M, DNA markers). A faint band is present at the expected size of the Metfl/fl allele in the papilla, presumably owing to the small number of thin limb cells present in this tissue. (D) Western analysis of renal papilla from Metfl/fl;HoxB7-Cre and Met+/+;HoxB7-Cre mice. Aqp2 was used as a loading control and immortalized IMCD cells were included as a positive control. (E) Immunostaining of the renal papilla from Metfl/fl;HoxB7-Cre and Met+/+;HoxB7-Cre mice with α-Met and α-Aqp2 reveals a strong Met signal in the collecting duct cells of wild-type mice that is markedly diminished in the Metfl/fl;HoxB7-Cre mice.
Western blot analysis of renal papilla confirmed that Met protein expression was markedly diminished in this region of the Metfl/fl;HoxB7-Cre mice (Fig. 1D), as had been previously reported for Met expression in hepatocytes of the Metfl/fl;AlbCre mouse (Huh et al., 2004). Immunofluorescent staining of kidney cryosections from Met+/+;HoxB7-Cre mice revealed that Met is most highly expressed both apically and basolaterally in collecting duct cells of the papilla (Fig. 1E) and in cells of the proximal tubule (data not shown), consistent with previous reports (Konda et al., 2004; Liu et al., 1996). Metfl/fl;HoxB7-Cre mice exhibited a marked decrease in Met protein expression in the collecting duct cells.
Adult Metfl/fl;HoxB7-Cre mice exhibit decreased nephron number and glomerular hypertrophy
Metfl/fl;HoxB7-Cre mice appeared to grow and behave normally. At 12 weeks of age there were no significant differences between Metfl/fl;HoxB7-Cre and Met+/+;HoxB7-Cre littermates with regards to body weight, plasma electrolytes or urinary electrolytes (Table 1). The kidneys of Metfl/fl;HoxB7-Cre mice appeared grossly normal and renal histology revealed no obvious abnormalities in cortical architecture or collecting duct morphology (see Fig. S1 in the supplementary material), with no increase in interstitial fibrosis detected on trichrome stained sections (data not shown). Of note, the glomeruli demonstrated a slight increase in mesangial cellularity without evidence for sclerosis or mesangiolysis.
Table 1.
Physiology of Met mutant mice
| Met+/+; HoxB7-Cre | Metfl/fl; HoxB7-Cre | |
|---|---|---|
| Blood urea nitrogen (mg/dl) | 20.8±2.2 | 19.9±3.3 |
| Plasma Na+ (mM) | 149±2.6 | 148.3±1.5 |
| Plasma K+ (mM) | 3.2±0.4 | 3.2±0.7 |
| Plasma Cl− (mM) | 106.7±4.8 | 105.7±1.8 |
| Urine Na+ (mM) | 34.5±10.2 | 39.7±16.2 |
| Urine K+ (mM) | 141.7±47.2 | 123.7+55.8 |
| Urine Cl− (mM) | 153.5±66.9 | 131.3±59.7 |
| Body weight (female, g) | 18.7±0.1 | 18.5±0.8 |
| Body weight (male, g) | 26.2±2.0 | 27.9±1.2 |
Values are mean ± s.e.m., n=3 for urine and plasma electrolytes for each genotype and n=6 for male and female body weight at 12 weeks of age for each genotype.
None of the differences is significant.
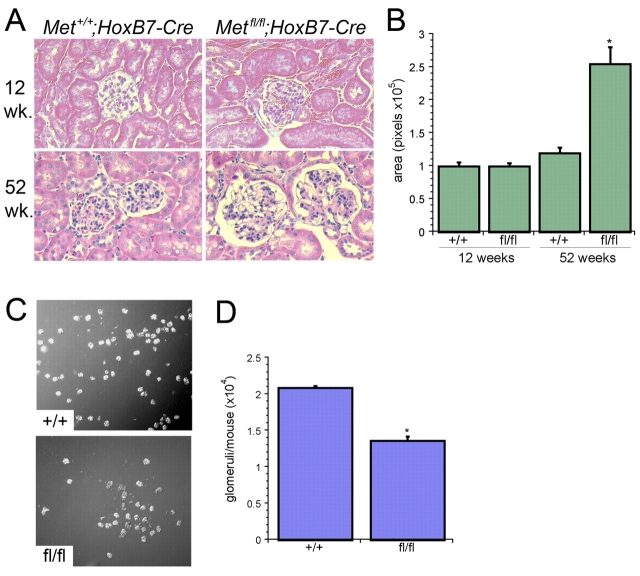
Hematoxylin and Eosin stained sections of kidneys from Metfl/fl;HoxB7-Cre mice at 1 year of age revealed that the glomeruli appeared larger than those from Met+/+;HoxB7-Cre littermates (Fig. 2A). Quantification of average glomerular surface area confirmed that juxta-medullary glomeruli from 1-year-old Metfl/fl;HoxB7-Cre mice were larger than those from either 1-year-old Met+/+;HoxB7-Cre littermates or 12-week-old Metfl/fl;HoxB7-Cre mice (Fig. 2B). In these older Metfl/fl;HoxB7-Cre mice, there was no evidence of either an increase in glomerular or interstitial fibrosis or an increase in albuminuria (data not shown).
Fig. 2.
Metfl/fl;HoxB7-Cre mice have reduced nephron number. (A) Representative images of glomeruli at 12 and 52 weeks from Metfl/fl;HoxB7-Cre and Met+/+;HoxB7-Cre mice, as indicated. (B) Quantification of glomerular surface area (in pixels) from kidney sections as in A (n=15 glomeruli/genotype, *P<0.001 versus Met+/+;HoxB7-Cre glomeruli). (C) Representative image of isolated glomeruli from Metfl/fl;HoxB7-Cre and Met+/+;HoxB7-Cre mice. (D) Quantification of total glomeruli/mouse in Metfl/fl;HoxB7-Cre and Met+/+;HoxB7-Cre mouse (n=5 mice/group, *P<0.001).
Glomerular hypertrophy can occur in the setting of a reduction in total nephron number owing to sustained hyperfiltration (Cullen-McEwen et al., 2003; Hostetter et al., 1981; Novick et al., 1991). Glomeruli from 12-week-old Metfl/fl;HoxB7-Cre mice were quantified using magnetic bead isolation as described by Takemoto et al. (Takemoto et al., 2002) (Fig. 2C). Kidneys from Met+/+;HoxB7-Cre mice contained 20,912±252 glomeruli/mouse [consistent with previous reports for wild-type mice (Takemoto et al., 2002)], whereas Metfl/fl;HoxB7-Cre mice had an average of only 13,660±600 glomeruli/mouse (Fig. 2D).
UB branching is reduced in explants of Metfl/fl;HoxB7-Cre mice
As final nephron number is determined by ureteric bud branching, we examined the possibility that UB branching is reduced in Metfl/fl;HoxB7-Cre. Metfl/+;HoxB7-Cre heterozygous mice were mated and embryonic kidneys harvested at E12.5. Consistent with previous reports (Bladt et al., 1995), E12.5 kidneys from Metfl/fl;HoxB7-Cre embryos were indistinguishable from that seen in Met+/+;HoxB7-Cre littermates (Fig. 3A, quantified in Fig. 3B,C), demonstrating that early UB branching is intact in the Metfl/fl;HoxB7-Cre mice. Examination of kidneys harvested at E14.5 also revealed no difference in size (Fig. 3B), although accurate quantification of UB branching could not be performed at this later time point. However, when E12.5 kidneys were explanted and cultured for 48 hours in the absence of exogenous growth factors, kidneys from Metfl/fl;HoxB7-Cre embryos grew less well and exhibited significantly less ureteric bud branches when compared with Met+/+;HoxB7-Cre littermates (Fig. 3A bottom panels, quantified in Fig. 3D,E).
Fig. 3.
Metfl/fl;HoxB7-Cre kidney explants have decreased ureteric bud branching. (A) Representative images of E12.5 kidneys from Metfl/fl;HoxB7-Cre and Met+/+;HoxB7-Cre mice (top panels) and E12.5 kidneys grown for 2 days in explant culture (bottom panels), as indicated, and stained with Dolichos biflorus (DBA). (B) Quantification of kidney surface area (in pixels) from E12.5 (n=7) and E14.5 (n=5) kidneys. (C) Quantification of terminal UB branches from E12.5 kidneys stained as in A (n=7). (D) Quantification of surface area of E12.5 kidneys grown as in A for 2 days in explant culture with or without Egf (n=10, *P<0.03 versus +/+ and P<0.001 versus fl/fl+Egf). (E) Quantification of terminal UB branches from E12.5 kidney explants grown as in A for 2 days in culture with or without Egf (n=12, *P<0.02 versus +/+ and P<0.01 versus fl/fl+Egf). (F) Representative image of E12.5 Metfl/fl;HoxB7-Cre explant as in A cultured in the presence of Egf. (G) Real-time PCR for mRNA levels of the indicated factors in E14.5 Metfl/fl;HoxB7-Cre kidneys expressed relative to Met+/+;HoxB7-Cre E14.5 kidneys. n=3 for each factor. *P<0.05 versus Met+/+;HoxB7-Cre kidneys.
It has been found that knockout of a single gene may lead to the upregulated expression of related or redundant pathways, resulting in phenotypic compensation. To address this possibility, Metfl/fl;HoxB7-Cre and Met+/+;HoxB7-Cre embryonic kidneys were harvested at E14.5 and total RNA isolated followed by quantitative PCR to define expression levels of a number of factors known to potentially regulate UB branching. These experiments revealed that the expression of two factors that positively regulate branching were increased in the Metfl/fl;HoxB7-Cre kidneys: Egfr and Fgf2 (Fig. 3G). There were also modest reductions in the expression of negative regulators of branching such as Bmp4 and 7 that did not reach statistical significance. Expression of Fgf1, Fgf7, Fgf10, Fgfr2, Gdnf, heregulin (Hrg) and pleiotrophin (Ptn) were unaltered.
Egf can stimulate in vitro morphogenic responses similar to those described for Hgf (Sakurai and Nigam, 1998; Sakurai et al., 1997b). As we have previously found that immortalized cells derived from kidneys of E12.5 Met-/- embryos express the Egfr and exhibit branching morphogenesis in response to Egf stimulation (Kjelsberg et al., 1997), we examined the possibility that increased Egfr signaling in Metfl/fl;HoxB7-Cre embryonic kidneys might be providing a compensatory UB growth and branching stimulus that accounts for the relatively modest loss of final nephron number in the adult Metfl/fl;HoxB7-Cre mouse. To determine whether the addition of Egf could rescue the defect in UB branching seen in the explanted Metfl/fl;HoxB7-Cre kidneys, some explants were treated with Egf (20 ng/ml) in addition to the defined medium. Both Metfl/fl;HoxB7-Cre and Met+/+;HoxB7-Cre explants responded to exogenous Egf with increased UB branching and increased kidney size (Fig. 3F, quantified in Fig. 3D,E), resulting in Metfl/fl;HoxB7-Cre explants in the presence of Egf being indistinguishable from Met+/+;HoxB7-Cre control kidneys.
Increased Egf receptor expression and activation in kidneys from Metfl/fl;HoxB7-Cre mice.
The observation that EGF could rescue the decreased branching morphogenesis seen in explanted Metfl/fl;HoxB7-Cre kidneys led us to determine whether or not this pathway was upregulated in vivo in the absence of Hgf-Met signaling. Immunostaining of E17 kidneys from Metfl/fl;HoxB7-Cre mice revealed increased Egfr expression in multiple cells including the UB (arrows) and renal vesicles (arrowhead) as compared with wild-type mice (Fig. 4A). This staining was seen on basolateral as well as apical surfaces of the cells. Western analysis of papilla isolated from 6-week-old mice confirmed an increase in Egf receptor expression in the Metfl/fl;HoxB7-Cre mice (Fig. 4B).
Fig. 4.
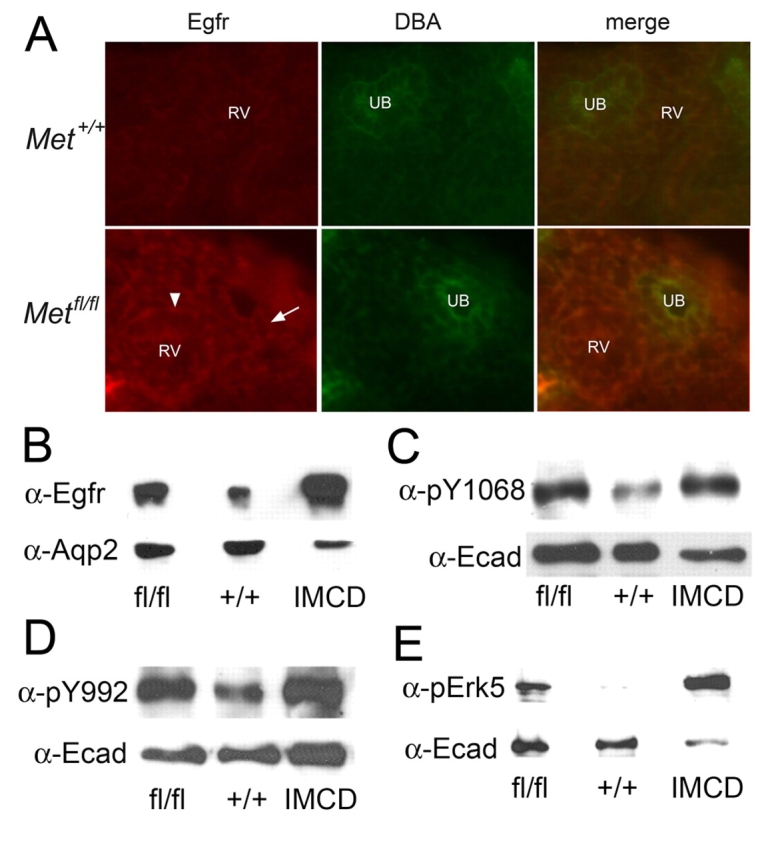
Increased expression and activation of Egf receptor in UB-derived cells of Metfl/fl;HoxB7-Cre mice. (A) Immunostaining of E17 explant cryosections from Met+/+;HoxB7-Cre and Metfl/fl;HoxB7-Cre mice with α-Egfr and DBA. Cells from the DBA-positive UB tips (arrow) as well as the adjacent renal vesicle (arrowhead, RV) and metanephric mesenchyme show increased Egfr fluorescence. (B-E) Western blot analyses of renal papilla obtained from 6-week-old Met+/+;HoxB7-Cre and Metfl/fl;HoxB7-Cre mice. (B) Immunoblotted with α-Egfr and α-aquaporin-2, (C) α-pEgfr (Y1068) and α-E-cadherin, (D) α-pEgfr (Y992) and α-E-cadherin, (E) α-pErk5 and α-E-cadherin.
Immunoblotting with antibodies that detect phosphorylation of the Egfr at the autophosphorylation sites Y992 and Y1068 revealed increased Egf receptor phosphorylation in Metfl/fl;HoxB7-Cre kidneys (Fig. 4C,D). Of note, there was no detectable difference in phosphorylation at tyrosine 845 (data not shown). The Y1068 site has been shown to be heavily phosphorylated following receptor activation with Egf or heparin-binding Egf (HB-EGF) and to mediate Grb2 binding and Erk1/2 activation (Ward et al., 1996; Wu et al., 2004; Yamauchi et al., 1998). A second MAPK family member, Erk5 (also known as Bmk1), is activated downstream of Egfr activation and has been shown to positively regulate both proliferative and morphogenic actions of Egf (Karihaloo et al., 2001; Kato et al., 1998). Immunoblotting with an antibody that detects the phosphorylated form of Erk5 confirmed that this downstream effector is activated in papillary cells from Metfl/fl;HoxB7-Cre kidneys (Fig. 4E). These results demonstrate that the Egfr is upregulated and activated in the absence of normal Met signaling by UB-derived structures, and that this activation is maintained even after development is complete.
Loss of Egfr signaling in Metfl/fl;HoxB7-Cre mice results in decreased UB branching and renal failure
Cumulatively, these observations suggest that upregulation of Egfr signaling might act to partially compensate for the lack of Hgf-Met signaling in Metfl/fl;HoxB7-Cre mice, resulting in the relative preservation of UB branching and final nephron number observed in these animals. To test this possibility, Metfl/fl;HoxB7-Cre mice were mated with waved-2 (wa-2) mice that have a spontaneous point mutation in Egfr that decreases receptor activation (Fowler et al., 1995; Luetteke et al., 1994). These mice exhibit decreased lactation, curly (waved) hair and small size, but are otherwise phenotypically normal with normal kidney morphology and function (Wang et al., 2003). Consistent with data from liver homogenates of wa-2 mice (Luetteke et al., 1994), stimulation with Egf failed to induce significant Egfr phosphorylation in kidneys from these mice (data not shown).
Male and female Metfl/fl;HoxB7-Cre;wa-2/+ mice were viable and fertile. These mice were mated and pregnant females sacrificed for embryo harvest at E14.5. Kidneys from Metfl/fl;HoxB7-Cre;wa-2/wa-2 embryos were found to be substantially smaller with markedly reduced UB branches when compared with Metfl/fl;HoxB7-Cre;Egfr+/+ littermates (Fig. 5A,B). To determine whether these kidneys could respond to exogenous Egf, embryos were harvested on E12.5 and kidneys maintained in explant culture for 2 days with or without Egf. Kidneys from Metfl/fl;HoxB7-Cre;wa-2/wa-2 embryos grown in control media exhibited less surface area and less ureteric bud branching when compared with Metfl/fl;HoxB7-Cre;Egfr+/+ kidneys (Fig. 5C, quantified in Fig. 5D,F), and treatment with Egf failed to rescue kidney growth or ureteric bud branching in the explanted Metfl/fl;HoxB7-Cre;wa-2/wa-2 kidneys (Fig. 5E, quantified in Fig. 5D,F).
Fig. 5.
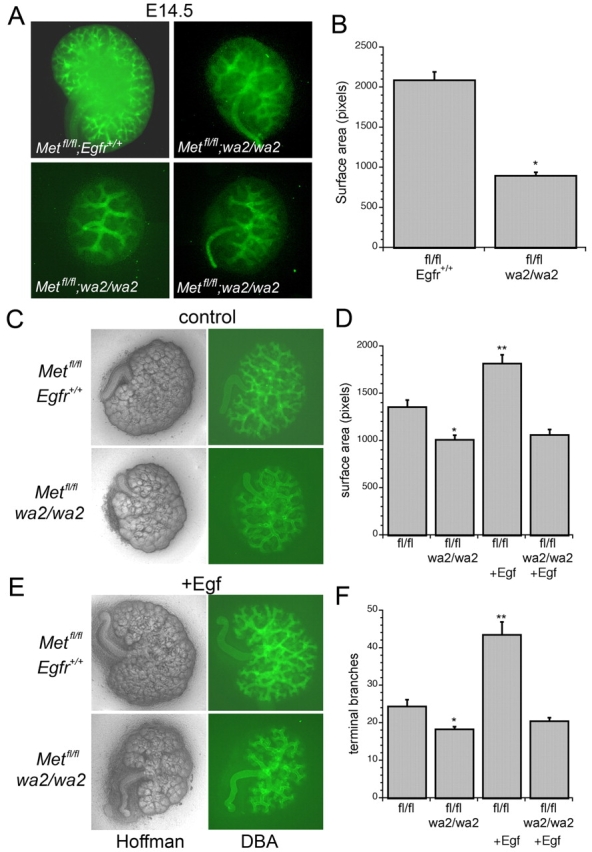
Embryonic kidneys from Metfl/fl;HoxB7-Cre:wa-2/wa-2 mice have decreased UB branching and fail to respond to exogenous Egf. (A) Representative images of E14.5 kidneys from a Metfl/fl;HoxB7-Cre;Egfr+/+ embryo and three separate Metfl/fl;HoxB7-Cre;wa-2/wa-2 littermates stained with DBA to show UB branches. (B) Quantitation of surface area from images of E14.5 kidneys (n=4, *P<0.005). (C) Representative images of E12.5 kidney explants from Metfl/fl;HoxB7-Cre;Egfr+/+ and Metfl/fl;HoxB7-Cre;wa-2/wa-2 mice. Hoffman images on left and corresponding DBA stain on right. (D) Quantification of surface area from explanted kidneys as in C (n=6, *P<0.03 vs. fl/fl; **P<0.003 vs. fl/fl;wa2/wa2+Egf). (E) Kidneys as in C treated with or without Egf (20ng/μl). (F) Quantification of terminal branch number from explanted kidneys as in A (n=5, *P<0.004 versus fl/fl; **P<0.001 versus fl/fl;wa2/wa2+Egf).
Although compound heterozygotes were born at the expected Mendelian frequency, only seven viable pups were obtained with the Metfl/fl;HoxB7-Cre;wa-2/wa-2 genotype rather than the predicted 19 (Fig. 6A). Metfl/fl;HoxB7-Cre;wa-2/wa-2 mice were small in size compared with heterozygous littermates at 3 weeks (7.9±0.7 g versus 12.8±0.5 g, P<0.001). Kidneys from these mice were small compared with either wild-type or wa-2 mice (Fig. 6B, Met+/+;Egfr+/+=0.036 g; Met+/+:wa-2/wa-2=0.032 g; Metfl/fl;HoxB7-Cre;wa-2/wa-2=0.011 g) and contained substantially fewer glomeruli/cross-section than did wild-type, wa-2 or Metfl/fl;HoxB7-Cre kidneys (Fig. 6C). Kidneys from Metfl/fl;HoxB7-Cre;wa-2/+ heterozygotes exhibited a loss of the upregulated Egfr activation seen in the Metfl/fl;HoxB7-Cre;Egfr+/+ kidneys and an intermediate phenotype in regards to both kidney size and glomerular number (Fig. 6B,C).
Fig. 6.
Metfl/fl;HoxB7-Cre:wa-2/wa-2 mice have reduced glomerular number and papillary fibrosis. (A) PCR analysis of genomic DNA from Egfr+/+, wa-2/wa-2 and heterozygous mice. (B) Upper panel shows representative kidneys from Metfl/fl;HoxB7-Cre;Egfr+/+, Metfl/fl;HoxB7-Cre;wa-2/+ and Metfl/fl;HoxB7-Cre;wa-2/wa-2 mice at 21 days. The lower panels show western analysis from renal papillas of these same genotypes immunoblotted with antibodies to detect the activated Egfr (α-pY1068) and a loading control (Ecad). (C) Quantification of glomeruli/section in Met+/+;HoxB7-Cre;Egfr+/+ (WT), wa-2/wa-2, Metfl/fl;HoxB7-Cre, Metfl/fl;HoxB7-Cre;wa-2/+ and Metfl/fl;HoxB7-Cre;wa-2/wa-2 mice (n=4 kidneys/genotype, *P<0.04 versus WT; **P<0.04 versus Metfl/fl;wa2/wa2; ***P<0.03 versus Metfl/fl). (D) Representative images of trichrome stained sections of the renal papilla from 3-week-old Metfl/fl;HoxB7-Cre;Egfr+/+ and Metfl/fl;HoxB7-Cre;wa-2/wa-2 mice. (E) Serum BUN values from 3-week-old Metfl/fl;HoxB7-Cre;Egfr+/+, Met+/+;wa-2/wa-2 and Metfl/fl;HoxB7-Cre;wa-2/wa-2 mice (n=6, *P<0.002).
Examination of kidney histology showed abnormal collecting duct morphology with significant interstitial fibrosis in the papilla and outer medulla of Metfl/fl;HoxB7-Cre;wa-2/wa-2 mice, but not in the cortex (Fig. 6D). Despite the reduction in nephron numbers, glomerulosclerosis was minimal at this early age (data not shown). Serum BUN levels were significantly elevated compared with either Metfl/fl;HoxB7-Cre;Egfr+/+ or Met+/+;HoxB7-Cre;wa-2/wa-2 littermates (Fig. 6E), and five out of seven mice died prior to 1 month of age. These data demonstrate that loss of Egfr signaling in the Metfl/fl;HoxB7-Cre mouse leads to a severe decline in UB branching and that these factors act cooperatively to maintain normal tubule architecture and prevent fibrosis in the mature collecting duct.
DISCUSSION
Using a conditional knockout approach to selectively eliminate Met expression in the ureteric bud, we obtained viable mice in which kidney development appeared grossly normal but resulted in a final nephron number that was only 70% of predicted. Based on prior studies showing that Hgf-Met signaling can stimulate epithelial branching morphogenesis, we pursued the possibility that this reduction in nephron number was due to a defect in ureteric bud branching. Consistent with the published studies of Met and Hgf knockout mice, UB branching was not significantly diminished in kidneys of Metfl/fl;HoxB7-Cre embryos at E12.5. The three-dimensional complexity of the ureteric bud made it technically difficult to accurately count terminal branches in vivo beyond this point, so an explant approach was used to assess later branching quantitatively. Explanted kidneys continue to undergo ureteric bud branching, nephrogenesis and growth, albeit at a reduced rate as compared with in vivo development [compare surface area of wild-type E14.5 kidneys (Fig. 3B) with that of wild-type explants at E12.5 + 2 days culture (Fig. 3D)]. In these experiments, we found that in the absence of exogenous growth factors the rate of branching and growth of E12.5 Metfl/fl;HoxB7-Cre kidney explants was significantly less than that of wild-type littermates.
Although the decreased UB branching seen in our explant experiments provides a plausible explanation for the reduction in nephron number seen in adult Metfl/fl;HoxB7-Cre mice, the number of UB branches in vivo at E12.5 was normal in these mice. The discrepancy between the in vitro and in vivo data has several possible explanations. As UB branching proceeds through E21 (Costantini, 2006), the decrease in final nephron number seen in the Metfl/fl;HoxB7-Cre mice may result from a selective loss of UB branching after E12.5. Alternatively, it is possible that a subtle defect in branching is present throughout development and that the cumulative effect of this defect is not quantitatively detectable at E12.5 without the examination of a significantly larger number of embryos than was performed in this study.
Our finding that a clear branching defect was detectable in explanted E12.5 Metfl/fl;HoxB7-Cre kidneys suggested to us that alternative signaling pathway(s) might be activated in vivo, which partially compensate for the loss of Hgf-Met signaling. Our examination of several candidate factors in the Metfl/fl;HoxB7-Cre embryonic kidneys revealed significant increases in two factors known to promote ureteric bud branching: the Egf receptor and Fgf2 (Qiao et al., 2001; Zhao et al., 2004). There was also a non-significant decrease in the expression of several factors known to inhibit branching [Bmp4 and Bmp7 (Bush et al., 2004; Luo et al., 1995)]. By contrast, there was no difference in mRNA expression of other well known UB branching regulators, including Fgf7, Fgf10, Gdnf and pleiotrophin (Ohuchi et al., 2000; Pichel et al., 1996; Qiao et al., 1999; Sakurai et al., 2001). Although only the levels of Egfr and Fgf-2 reached statistical significance, these trends suggest that several factors may be involved in compensating for the loss of Hgf signaling in these developing kidneys.
Several lines of evidence have supported the idea that Hgf and Egf can signal cooperatively to induce ureteric bud branching and tubulogenesis, at least in vitro. Early studies by Barros and co-workers using an embryonic kidney explant-renal tubular cell co-culture system revealed that both Hgf and Egf receptor ligands are made by the explanted kidney and induced tubulogenic responses in the co-cultured cells (Barros et al., 1995). Furthermore, Sakurai et al. found that cells derived from the metanephric mesenchyme secrete both Hgf and Egf receptor ligands, and that both factors were able to induce branching morphogenesis in ureteric bud-derived cells (Sakurai et al., 1997a). Consistent with this, Met-/- epithelial cells isolated from E12 kidneys exhibited cell migration and branching tubulogenesis in response to Egf and Tgfα (Kjelsberg et al., 1997; Sakurai et al., 1997b). It has also been suggested that Egf receptor ligands play a significant role in remodeling the collecting system during the later stages of kidney development. Examination of rat kidneys has demonstrated that apoptosis occurs as the first branches of the ureteric bud dilate in order to form the collecting system, and that treatment with Egf can substantially diminish this (Coles et al., 1993).
Our finding that Egfr expression is increased at both the mRNA and protein levels in Metfl/fl;HoxB7-Cre embryonic kidneys, coupled with the ability of exogenously added Egf to partially rescue the in vitro branching defect in these kidneys, suggests that Egf receptor signaling may be an important in vivo pathway to maintain UB branching during development of the Metfl/fl;HoxB7-Cre mouse. To more rigorously test this hypothesis, we generated Metfl/fl;HoxB7-Cre;wa-2/wa-2 embryos in which Egfr signaling is impaired in all cells and Met receptor signaling is selectively lost in the UB-derived cells. As opposed to the kidneys of parental Metfl/fl;HoxB7-Cre or wa-2/wa-2 mice, the E14.5 Metfl/fl;HoxB7-Cre;wa-2/wa-2 embryos demonstrated markedly decreased kidney size and ureteric bud branching in vivo, and an exaggerated loss of branching and growth during explant culture. Because very few of these animals survived beyond 3 weeks, we were unable to perform magnetic bead quantitation of nephron number. However, the number of glomeruli/kidney cross-section was reduced by 55% in fully developed kidneys from Metfl/fl;HoxB7-Cre;wa-2/wa-2 mice when compared with wild-type or wa-2 mice. The relative accuracy of this approach is suggested by our finding that Metfl/fl;HoxB7-Cre;Egfr+/+ kidneys demonstrated a 26% reduction in nephron number relative to wild-type mice, similar to the 35% reduction observed using the magnetic bead approach. Thus, these data support the conclusion that Egfr signaling in vivo acts to partially rescue ureteric bud branching defects in Metfl/fl;HoxB7-Cre mice, and that these two signaling pathways act cooperatively during normal development of the collecting system of the kidney. Based on our real-time PCR results, it is likely that Fgf signaling pathways are also altered as part of this compensatory response.
To our surprise, upregulated expression of the Egfr persisted in the collecting duct of adult Metfl/fl;HoxB7-Cre mice. Sufficient protein for western analysis was obtained from the adult papilla and revealed that the overexpressed Egfr was activated and that increased downstream signaling was present. Interestingly, collecting duct morphology and kidney function were normal for over 12 months in Metfl/fl;HoxB7-Cre mice, whereas Metfl/fl;HoxB7-Cre;wa-2/wa-2 mice demonstrated severe fibrosis of the renal papilla with renal failure and death by 3-4 weeks in the majority of animals. This observation is consistent with studies that have shown that Hgf-Met signaling can prevent fibrosis after unilateral ureteral obstruction (Yang et al., 2002), and that Egf signaling is required for maintenance of normal collecting duct architecture (Threadgill et al., 1995). Thus, similar to their role in UB development, Met signaling and Egfr signaling appear to play complementary roles in normal maintenance of the collecting duct.
Supplementary material
Supplementary material for this article is available at http://dev.biologists.org/cgi/content/full/136/2/337/DC1
This work was supported by an R01 award to L.G.C. (DK65109) and an American Heart Association Fellow-to-Faculty and American Society of Nephrology Gottschalk award to S.I. Deposited in PMC for release after 12 months.
References
- Barros, E. J., Santos, O. F., Matsumoto, K., Nakamura, T. and Nigam, S. K. (1995). Differential tubulogenic and branching morphogenetic activities of growth factors: implications for epithelial tissue development. Proc. Natl. Acad. Sci. USA 92, 4412-4416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bates, C. M. (2007). Role of fibroblast growth factor receptor signaling in kidney development. Pediatr. Nephrol. 22, 343-349. [DOI] [PubMed] [Google Scholar]
- Bladt, F., Riethmacher, D., Isenmann, S., Aguzzi, A. and Birchmeier, C. (1995). Essential role for the c-met receptor in the migration of myogenic precursor cells into the limb bud. Nature 376, 768-771. [DOI] [PubMed] [Google Scholar]
- Bush, K. T., Sakurai, H., Steer, D. L., Leonard, M. O., Sampogna, R. V., Meyer, T. N., Schwesinger, C., Qiao, J. and Nigam, S. K. (2004). TGF-beta superfamily members modulate growth, branching, shaping, and patterning of the ureteric bud. Dev. Biol. 266, 285-298. [DOI] [PubMed] [Google Scholar]
- Coles, H. S., Burne, J. F. and Raff, M. C. (1993). Large-scale normal cell death in the developing rat kidney and its reduction by epidermal growth factor. Development 118, 777-784. [DOI] [PubMed] [Google Scholar]
- Costantini, F. (2006). Renal branching morphogenesis: concepts, questions, and recent advances. Differentiation 74, 402-421. [DOI] [PubMed] [Google Scholar]
- Cullen-McEwen, L. A., Kett, M. M., Dowling, J., Anderson, W. P. and Bertram, J. F. (2003). Nephron number, renal function, and arterial pressure in aged GDNF heterozygous mice. Hypertension 41, 335-340. [DOI] [PubMed] [Google Scholar]
- Fowler, K. J., Walker, F., Alexander, W., Hibbs, M. L., Nice, E. C., Bohmer, R. M., Mann, G. B., Thumwood, C., Maglitto, R., Danks, J. A. et al. (1995). A mutation in the epidermal growth factor receptor in waved-2 mice has a profound effect on receptor biochemistry that results in impaired lactation. Proc. Natl. Acad. Sci. USA 92, 1465-1469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hostetter, T. H., Olson, J. L., Rennke, H. G., Venkatachalam, M. A. and Brenner, B. M. (1981). Hyperfiltration in remnant nephrons: a potentially adverse response to renal ablation. Am. J. Physiol. 241, F85-F93. [DOI] [PubMed] [Google Scholar]
- Huh, C. G., Factor, V. M., Sanchez, A., Uchida, K., Conner, E. A. and Thorgeirsson, S. S. (2004). Hepatocyte growth factor/c-met signaling pathway is required for efficient liver regeneration and repair. Proc. Natl. Acad. Sci. USA 101, 4477-4482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ishibe, S., Joly, D., Zhu, X. and Cantley, L. G. (2003). Phosphorylation-dependent paxillin-ERK association mediates hepatocyte growth factor-stimulated epithelial morphogenesis. Mol. Cell 12, 1275-1285. [DOI] [PubMed] [Google Scholar]
- Ishibe, S., Joly, D., Liu, Z. X. and Cantley, L. G. (2004). Paxillin serves as an ERK-regulated scaffold for coordinating FAK and Rac activation in epithelial morphogenesis. Mol. Cell 16, 257-267. [DOI] [PubMed] [Google Scholar]
- Ishibe, S., Haydu, J. E., Togawa, A., Marlier, A. and Cantley, L. G. (2006). Cell confluence regulates Hgf-stimulated cell morphogenesis in a {beta}-catenin dependent manner. Mol. Cell. Biol. 26, 9232-9243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karihaloo, A., O'Rourke, D. A., Nickel, C., Spokes, K. and Cantley, L. G. (2001). Differential MAPK pathways utilized for HGF- and EGF-dependent renal epithelial morphogenesis. J. Biol. Chem. 276, 9166-9173. [DOI] [PubMed] [Google Scholar]
- Karihaloo, A., Nickel, C. and Cantley, L. G. (2005). Signals which build a tubule. Nephron Exp. Nephrol. 100, e40-e45. [DOI] [PubMed] [Google Scholar]
- Kato, Y., Tapping, R. I., Huang, S., Watson, M. H., Ulevitch, R. J. and Lee, J. D. (1998). Bmk1/Erk5 is required for cell proliferation induced by epidermal growth factor. Nature 395, 713-716. [DOI] [PubMed] [Google Scholar]
- Kjelsberg, C., Sakurai, H., Spokes, K., Birchmeier, C., Drummond, I., Nigam, S. and Cantley, L. G. (1997). Met-/- kidneys express epithelial cells that chemotax and form tubules in response to EGF receptor ligands. Am. J. Physiol. 272, F222-F228. [DOI] [PubMed] [Google Scholar]
- Kobayashi, A., Valerius, M. T., Mugford, J. W., Carroll, T. J., Self, M., Oliver, G. and McMahon, A. P. (2008). Six2 defines and regulates a multipotent self-renewing nephron progenitor population throughout mammalian kidney development. Cell Stem Cell 3, 169-181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Konda, R., Sato, H., Hatafuku, F., Nozawa, T., Ioritani, N. and Fujioka, T. (2004). Expression of hepatocyte growth factor and its receptor C-met in acquired renal cystic disease associated with renal cell carcinoma. J. Urol. 171, 2166-2170. [DOI] [PubMed] [Google Scholar]
- Liu, Y., Tolbert, E. M., Sun, A. M. and Dworkin, L. D. (1996). In vivo and in vitro evidence for increased expression of HGF receptor in kidney of diabetic rat. Am. J. Physiol. 271, F1202-F1210. [DOI] [PubMed] [Google Scholar]
- Liu, Z. X., Yu, C. F., Nickel, C., Thomas, S. and Cantley, L. G. (2002). Hepatocyte growth factor induces ERK-dependent paxillin phosphorylation and regulates paxillin-focal adhesion kinase association. J. Biol. Chem. 277, 10452-10458. [DOI] [PubMed] [Google Scholar]
- Luetteke, N. C., Phillips, H. K., Qiu, T. H., Copeland, N. G., Earp, H. S., Jenkins, N. A. and Lee, D. C. (1994). The mouse waved-2 phenotype results from a point mutation in the EGF receptor tyrosine kinase. Genes Dev. 8, 399-413. [DOI] [PubMed] [Google Scholar]
- Luo, G., Hofmann, C., Bronckers, A. L., Sohocki, M., Bradley, A. and Karsenty, G. (1995). BMP-7 is an inducer of nephrogenesis, and is also required for eye development and skeletal patterning. Genes Dev. 9, 2808-2820. [DOI] [PubMed] [Google Scholar]
- Montesano, R., Matsumoto, K., Nakamura, T. and Orci, L. (1991). Identification of a fibroblast-derived epithelial morphogen as hepatocyte growth factor. Cell 67, 901-908. [DOI] [PubMed] [Google Scholar]
- Novick, A. C., Gephardt, G., Guz, B., Steinmuller, D. and Tubbs, R. R. (1991). Long-term follow-up after partial removal of a solitary kidney. N. Engl. J. Med. 325, 1058-1062. [DOI] [PubMed] [Google Scholar]
- O'Brien, L. E., Tang, K., Kats, E. S., Schutz-Geschwender, A., Lipschutz, J. H. and Mostov, K. E. (2004). ERK and MMPs sequentially regulate distinct stages of epithelial tubule development. Dev. Cell 7, 21-32. [DOI] [PubMed] [Google Scholar]
- Ohuchi, H., Hori, Y., Yamasaki, M., Harada, H., Sekine, K., Kato, S. and Itoh, N. (2000). FGF10 acts as a major ligand for FGF receptor 2 IIIb in mouse multi-organ development. Biochem. Biophys. Res. Commun. 277, 643-649. [DOI] [PubMed] [Google Scholar]
- Pichel, J. G., Shen, L., Sheng, H. Z., Granholm, A. C., Drago, J., Grinberg, A., Lee, E. J., Huang, S. P., Saarma, M., Hoffer, B. J. et al. (1996). Defects in enteric innervation and kidney development in mice lacking GDNF. Nature 382, 73-76. [DOI] [PubMed] [Google Scholar]
- Pollack, A. L., Runyan, R. B. and Mostov, K. E. (1998). Morphogenetic mechanisms of epithelial tubulogenesis: MDCK cell polarity is transiently rearranged without loss of cell-cell contact during scatter factor/hepatocyte growth factor-induced tubulogenesis. Dev. Biol. 204, 64-79. [DOI] [PubMed] [Google Scholar]
- Ponzetto, C., Bardelli, A., Zhen, Z., Maina, F., dalla Zonca, P., Giordano, S., Graziani, A., Panayotou, G. and Comoglio, P. M. (1994). A multifunctional docking site mediates signaling and transformation by the hepatocyte growth factor/scatter factor receptor family. Cell 77, 261-271. [DOI] [PubMed] [Google Scholar]
- Qiao, J., Uzzo, R., Obara-Ishihara, T., Degenstein, L., Fuchs, E. and Herzlinger, D. (1999). FGF-7 modulates ureteric bud growth and nephron number in the developing kidney. Development 126, 547-554. [DOI] [PubMed] [Google Scholar]
- Qiao, J., Bush, K. T., Steer, D. L., Stuart, R. O., Sakurai, H., Wachsman, W. and Nigam, S. K. (2001). Multiple fibroblast growth factors support growth of the ureteric bud but have different effects on branching morphogenesis. Mech. Dev. 109, 123-135. [DOI] [PubMed] [Google Scholar]
- Rosario, M. and Birchmeier, W. (2004). Making tubes: step by step. Dev. Cell 7, 3-5. [DOI] [PubMed] [Google Scholar]
- Sainio, K., Suvanto, P., Davies, J., Wartiovaara, J., Wartiovaara, K., Saarma, M., Arumae, U., Meng, X., Lindahl, M., Pachnis, V. et al. (1997). Glial-cell-line-derived neurotrophic factor is required for bud initiation from ureteric epithelium. Development 124, 4077-4087. [DOI] [PubMed] [Google Scholar]
- Sakurai, H. and Nigam, S. K. (1998). In vitro branching tubulogenesis: implications for developmental and cystic disorders, nephron number, renal repair, and nephron engineering. Kidney Int. 54, 14-26. [DOI] [PubMed] [Google Scholar]
- Sakurai, H., Barros, E. J., Tsukamoto, T., Barasch, J. and Nigam, S. K. (1997a). An in vitro tubulogenesis system using cell lines derived from the embryonic kidney shows dependence on multiple soluble growth factors. Proc. Natl. Acad. Sci. USA 94, 6279-6284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sakurai, H., Tsukamoto, T., Kjelsberg, C. A., Cantley, L. G. and Nigam, S. K. (1997b). EGF receptor ligands are a large fraction of in vitro branching morphogens secreted by embryonic kidney. Am. J. Physiol. 273, F463-F472. [DOI] [PubMed] [Google Scholar]
- Sakurai, H., Bush, K. T. and Nigam, S. K. (2001). Identification of pleiotrophin as a mesenchymal factor involved in ureteric bud branching morphogenesis. Development 128, 3283-3293. [DOI] [PubMed] [Google Scholar]
- Santos, O. F., Barros, E. J., Yang, X. M., Matsumoto, K., Nakamura, T., Park, M. and Nigam, S. K. (1994). Involvement of hepatocyte growth factor in kidney development. Dev. Biol. 163, 525-529. [DOI] [PubMed] [Google Scholar]
- Saxen, L. (1987). Organogenesis of the Kidney. Cambridge, UK: Cambridge University Press. Saxen, L. and Sariola, H. (1987). Early organogenesis of the kidney. Pediatr. Nephrol. 1, 385-392. [DOI] [PubMed] [Google Scholar]
- Schmidt, C., Bladt, F., Goedecke, S., Brinkmann, V., Zschiesche, W., Sharpe, M., Gherardi, E. and Birchmeier, C. (1995). Scatter factor/hepatocyte growth factor is essential for liver development. Nature 373, 699-702. [DOI] [PubMed] [Google Scholar]
- Takemoto, M., Asker, N., Gerhardt, H., Lundkvist, A., Johansson, B. R., Saito, Y. and Betsholtz, C. (2002). A new method for large scale isolation of kidney glomeruli from mice. Am. J. Pathol. 161, 799-805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Threadgill, D. W., Dlugosz, A. A., Hansen, L. A., Tennenbaum, T., Lichti, U., Yee, D., LaMantia, C., Mourton, T., Herrup, K., Harris, R. C. et al. (1995). Targeted disruption of mouse EGF receptor: effect of genetic background on mutant phenotype. Science 269, 230-234. [DOI] [PubMed] [Google Scholar]
- Tufro, A., Teichman, J., Woda, C. and Villegas, G. (2007). Semaphorin3a inhibits ureteric bud branching morphogenesis. Mech. Dev. 125, 558-568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uehara, Y., Minowa, O., Mori, C., Shiota, K., Kuno, J., Noda, T. and Kitamura, N. (1995). Placental defect and embryonic lethality in mice lacking hepatocyte growth factor/scatter factor. Nature 373, 702-705. [DOI] [PubMed] [Google Scholar]
- Wang, Z., Chen, J. K., Wang, S. W., Moeckel, G. and Harris, R. C. (2003). Importance of functional EGF receptors in recovery from acute nephrotoxic injury. J. Am. Soc. Nephrol. 14, 3147-3154. [DOI] [PubMed] [Google Scholar]
- Ward, C. W., Gough, K. H., Rashke, M., Wan, S. S., Tribbick, G. and Wang, J. (1996). Systematic mapping of potential binding sites for Shc and Grb2 SH2 domains on insulin receptor substrate-1 and the receptors for insulin, epidermal growth factor, platelet-derived growth factor, and fibroblast growth factor. J. Biol. Chem. 271, 5603-5609. [DOI] [PubMed] [Google Scholar]
- Weidner, K. M., Sachs, M. and Birchmeier, W. (1993). The Met receptor tyrosine kinase transduces motility, proliferation, and morphogenic signals of scatter factor/hepatocyte growth factor in epithelial cells. J. Cell Biol. 121, 145-154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woolf, A. S., Kolatsi-Joannou, M., Hardman, P., Andermarcher, E., Moorby, C., Fine, L. G., Jat, P. S., Noble, M. D. and Gherardi, E. (1995). Roles of hepatocyte growth factor/scatter factor and the met receptor in the early development of the metanephros. J. Cell Biol. 128, 171-184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu, W., Samet, J. M., Silbajoris, R., Dailey, L. A., Sheppard, D., Bromberg, P. A. and Graves, L. M. (2004). Heparin-binding epidermal growth factor cleavage mediates zinc-induced epidermal growth factor receptor phosphorylation. Am. J. Respir. Cell Mol. Biol. 30, 540-547. [DOI] [PubMed] [Google Scholar]
- Yamauchi, T., Ueki, K., Tobe, K., Tamemoto, H., Sekine, N., Wada, M., Honjo, M., Takahashi, M., Takahashi, T., Hirai, H. et al. (1998). Growth hormone-induced tyrosine phosphorylation of EGF receptor as an essential element leading to MAP kinase activation and gene expression. Endocr. J. 45Suppl., S27-S31. [DOI] [PubMed] [Google Scholar]
- Yang, J., Dai, C. and Liu, Y. (2002). Hepatocyte growth factor gene therapy and angiotensin II blockade synergistically attenuate renal interstitial fibrosis in mice. J. Am. Soc. Nephrol. 13, 2464-2477. [DOI] [PubMed] [Google Scholar]
- Yu, J., Carroll, T. J. and McMahon, A. P. (2002). Sonic hedgehog regulates proliferation and differentiation of mesenchymal cells in the mouse metanephric kidney. Development 129, 5301-5312. [DOI] [PubMed] [Google Scholar]
- Zhao, H., Kegg, H., Grady, S., Truong, H. T., Robinson, M. L., Baum, M. and Bates, C. M. (2004). Role of fibroblast growth factor receptors 1 and 2 in the ureteric bud. Dev. Biol. 276, 403-415. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.