Elevated thyroid hormones in rabbits resulted in significant decreases in the size and number of EOM myofibers. Concomitantly, there was a decreased number of activated satellite cells and neonatal- and developmental-positive myofibers, correlating with known effects of thyroid hormone on nonocular skeletal muscle.
Abstract
Purpose.
Human extraocular muscles (EOM) are preferentially susceptible to thyroid eye disease. Although the specific cause of this autoimmune disorder is unknown, it is often associated with elevated thyroid hormone levels. Thus, the effect of elevated thyroid hormone levels on cross-sectional area, myofiber size, satellite cells, and myosin heavy chain (MyHC) isoform expression was examined in adult rabbit EOMs, to determine how elevated thyroid hormone alters EOM biology.
Methods.
After 1 month of elevated thyroid hormone levels, the EOMs were removed and prepared for histologic examination. Total muscle mass, myofiber size, patterns of MyHC isoform expression, and the number of satellite cells were determined.
Results.
Elevated thyroid hormone levels significantly decreased muscle mass, total number of myofibers, and mean cross-sectional area of the myofibers. Alterations in MyHC isoform expression were extremely complex, but several basic patterns emerged. The percentages of neonatal- and developmental-positive myofibers decreased in almost all EOM regions examined, and the percentages of slow-positive myofibers significantly increased. In contrast to normal EOMs, which retain a population of activated satellite cells throughout life, elevated thyroid hormone levels resulted in the virtual disappearance of MyoD-positive cells and a decrease in Pax7-positive cells.
Conclusions.
The reductions in EOM size, number of fibers expressing developmental and neonatal MyHC, and number of MyoD- and Pax7-positive satellite cells suggest that elevated thyroid hormone levels decrease the ongoing myofiber remodeling normally seen in the EOM. These catabolic changes have important implications for maintenance of function in the EOMs.
Extraocular muscles (EOM) differ from other skeletal muscles in their anatomy, physiology, and even more important, in their propensity for involvement or sparing in skeletal muscle diseases.1 In particular, the EOM are unique in their exclusive involvement in thyroid eye disease (TED), an autoimmune disease of the eye muscles that results in their enlargement in the acute phase and, in the chronic phase, muscle fiber replacement and contraction or tightening of the muscles. TED in humans is often associated with thyroid hormone endocrinopathy, and this inflammatory orbitopathy results in proptosis, strabismus, and eyelid retraction.2,3 Unfortunately, there is no satisfactory animal model for the autoimmune process that leads to TED, in part because the pathophysiology is complex and not well understood. TED does not always occur in cases of elevated thyroid hormone, but its frequent association—eye signs in 50% of patients with Graves' disease—lends credibility and relevance to the study of the specific effects of hyperthyroidism on the EOMs. Although elucidation of the effects of elevated thyroid hormone cannot explain the mechanism of this complex autoimmune disease, characterization of the changes in the EOM muscle as a result of altered thyroid hormones may suggest important directions for future research.
Thyroid hormone has well-described catabolic effects on adult skeletal muscle, and sustained elevations in blood thyroid hormone levels result in muscle weakness and wasting.4 The direct effects of elevated thyroid hormone are complex. Increased rates of protein degradation occur,5 specifically reducing myosin heavy chain isoform content, but oxidative modifications that impact the ability of the contractile proteins to generate force also are seen.6 Significant decreases occur in mitochondrial uncoupling proteins 2 and 3 as well.7 Elevated thyroid hormone levels affect muscle proliferative cells, increasing both the rate of myoblast withdrawal from the cell cycle8 and their rate of terminal differentiation.9,10 In the adult, the EOMs retain a population of activated satellite cells,11 with resultant continuous myofiber remodeling that involves both myonuclear addition and removal.12 As elevated thyroid hormones significantly alter these properties in development, the effects of increased thyroid hormone on satellite cell content, MyHC isoform, and muscle size in adult EOMs were examined.
In limb skeletal muscle, four MHC isoforms are commonly expressed: three produce fast contractile properties, type IIA, -IIB, and -IIX, and one produces slow contractile properties, type I. Thyroid hormone status has a significant impact on differential expression of MyHC isoforms in adult mammalian limb skeletal muscle.13,14 In fact, thyroid hormone status appears to predominate over other potential regulators of MyHC expression, such as electrical stimulation. For example, in muscles composed of mostly type I or slow MyHC isoform myofibers, high levels of thyroid hormone upregulate the expression of type II MyHC mRNA, whereas chronically low levels of thyroid hormone upregulate type I MyHC expression,15,16 which in turn results in alterations in contraction velocity (Vmax).17,18 Elevated thyroid hormone levels also result in increased percentages of hybrid fibers, which coexpress multiple MyHC isoforms in single fibers.19
Compared with limb skeletal muscle, EOMs have a more complex composition with regard to MyHC isoform expression, expressing nine distinct isoforms, including two normally found only in developing or regenerating muscle, neonatal, and developmental MyHC.20,21 The neonatal and developmental MyHC isoforms normally are downregulated in limb skeletal muscles during late stages of development. Adding to this complexity is the fact that many of these individual MyHC isoforms are coexpressed in single myofibers.22–26 EOMs normally express an increased number of thyroid hormone receptors compared with nonocular skeletal muscles.27 Elevated thyroid hormone accelerates the MyHC transition from neonatal to adult isoforms.28 Thus, elevation of thyroid hormone levels could result in significantly more complex MyHC expression changes than in normal limb muscle. To our knowledge, the effects of elevated thyroid hormones on EOM myosin composition, satellite cell activity, and muscle size in populations of myofibers from mature eye muscles have not been studied. In this study, we evaluated the effects of elevated thyroid hormone levels on the size and number of EOM fibers; the overall size of EOM muscles; and the percentage of myofibers expressing fast, slow, neonatal, and/or developmental MyHC in adult rabbits, as well as the effect on the Pax7- and MyoD-positive populations of satellite cells. These effects were compared to the properties of normal adult EOMs.
Materials and Methods
New Zealand White rabbits were purchased from Birchwood Valley Farms and housed in University of Minnesota animal facilities. All experiments complied with the published guidelines of the National Institutes of Health and the ARVO Statement for Use of Animals in Ophthalmic and Vision Research and were approved by the University Animal Care and Use Committee at the University of Minnesota. Hyperthyroidism was induced in six rabbits by IP injection of 3,3′,5-triiodothyronine (T3) at a dose of 0.2 mg/kg body weight every other day for 1 month.29–31 Serum thyroid hormone levels were determined at 1 week, 2 weeks, and 1 month. Blood samples were analyzed for blood thyroid hormone levels by the clinical laboratory in the Department of Veterinary Medicine at the University of Minnesota Veterinary School. All animals were considered to have hyperthyroidism if their blood T3 levels increased by 50%. All animals met this criterion with significantly elevated levels of T3 and T4 compared with baseline and control rabbits. The rabbits were given access to food ad libitum and were monitored daily for signs of hyperthyroidism including weight, food intake, water intake, diarrhea, and irritability. Weight loss occurred within the first 2 weeks and then stabilized, dropping from an average initial weight of 2.23 to 1.37 kg, as previously published.32
The rabbits were killed after 1 month by an overdose of barbiturates given via the ear vein. The extraocular muscles were dissected from the orbits, frozen, and processed for histologic examination. In accordance with other studies,24 the four rectus muscles were chosen for quantitative analysis: the superior rectus (SR), medial rectus (MR), inferior rectus (IR), and lateral rectus (LR). Immunohistochemistry was performed on serial frozen sections of EOMs to visualize pan-fast, slow, neonatal, and developmental MyHC isoforms (Vector Laboratories, Burlingame, CA). After incubation in blocking serum, the sections were incubated in the primary antibody for 1 hour at a dilution of 1:40 for the antibodies to fast and slow MyHC isoforms and 1:20 for the antibodies to neonatal and developmental MyHC isoforms. The sections were rinsed and incubated using the reagents of an ABC kit (Vectastain Elite, Vector Laboratories) containing biotin-avidin-peroxidase complexes. The reacted tissue sections were processed by using the heavy metal–intensified diaminobenzidine procedure. Sections were also immunostained for the presence of satellite cells positive for either Pax7 or MyoD, one of the myogenic regulatory factors, using the procedure described earlier with the following modifications. For Pax7, the sections were fixed in cold acetone, rinsed in PBS, and incubated in a primary antibody to Pax7 at a concentration of 1:500 (Hybridoma Bank, University of Iowa, Iowa City, IA). For MyoD, the cross sections were fixed in 4% paraformaldehyde, rinsed in PBS, and incubated with a primary antibody to MyoD at a concentration of 1:50 (Vector Laboratories). For the control muscles, both primary antibody incubation times were 1 hour. For the hyperthyroid muscles, sections were incubated overnight to maximize the possibility of staining even cells that contained small amounts of MyoD or Pax7. This method allowed the visualization of a few Pax7-positive cells that were not seen with a 1-hour primary incubation period. Muscle sections from six age-matched control rabbits were similarly processed and analyzed to assess whether the muscle from the thyroid hormone-treated rabbits were significantly different.
Because of the complexity of MyHC isoform expression in different regions and layers of individual EOM,24,25 and the complex coexpression patterns of MyHC isoforms within single myofibers,22,25,26 we chose to study populations of myofibers, rather than changes in a small number of single, identified myofibers. This method would elucidate trends in MyHC isoform expression within the muscles as a whole. At least three cross sections from the midbelly region and the distal EOM near the tendon from each of the four rectus muscles were analyzed for individual myofiber cross-sectional area, total muscle cross-sectional area, percentages of myofibers positive for each of the specific MyHC isoforms examined in this study, and the number of MyoD-positive satellite cells. A minimum of four fields, or between 200 and 400 myofibers, were analyzed on each cross section from each of the six experimental and six control rabbits studied. Fiber cross-sectional areas, total muscle cross-sectional areas, and counts of myofibers positive for each of the four MyHC isoforms in control and hyperthyroid EOMs were determined with the aid of a morphometry program (Nova Prime; Bioquant, Nashville, TN). Because of the known changes in MyHC isoform composition along the muscle length in rabbits,24 care was taken to quantify muscle sections from areas the same distance from the tendon end of each muscle. The percentage positive for each of the four isoforms examined was determined based on the total number of myofibers analyzed for each specimen, and these were compared with the percentage positive for each of the four antibodies in mature rabbit EOMs from untreated controls. All data were analyzed for statistical significance with an unpaired Student's t-test (Prism and Statmate software; Graphpad, San Diego, CA). An F-test was used to verify that the variances of the control and experimental groups were not significantly different. Data were considered significantly different if P < 0.05. Error bars represent SE of the means.
Results
Grossly, the EOM from the rabbits with elevated thyroid hormone were thinner compared with normal control rabbit EOM. Both total muscle cross-sectional area and total number of myofibers in the EOMs from rabbits with elevated thyroid hormone levels were significantly smaller than those of euthyroid rabbits (Fig. 1). There was a significant difference in mean individual myofiber cross-sectional areas of myofibers positive for fast and developmental MyHC isoforms, whereas slow myofibers actually increased in mean cross-sectional area compared with the control muscles (Fig. 2). No significant change was seen in the cross-sectional areas of neonatal MyHC-positive myofibers. When the distal ends of the EOM from the rabbits with elevated thyroid hormone levels were examined, the inner orbital layer (intermediate layer) was either substantially thinned or not visible in the sections analyzed (data not shown). This finding was consistent and particularly pronounced in the lateral and superior rectus muscles. It is unclear whether it was due to changes in patterns of MyHC isoform expression or to the decreased number of myofibers. It should be noted that there was no evidence of inflammatory cell infiltrate or fiber necrosis in any of the muscle sections examined.
Figure 1.
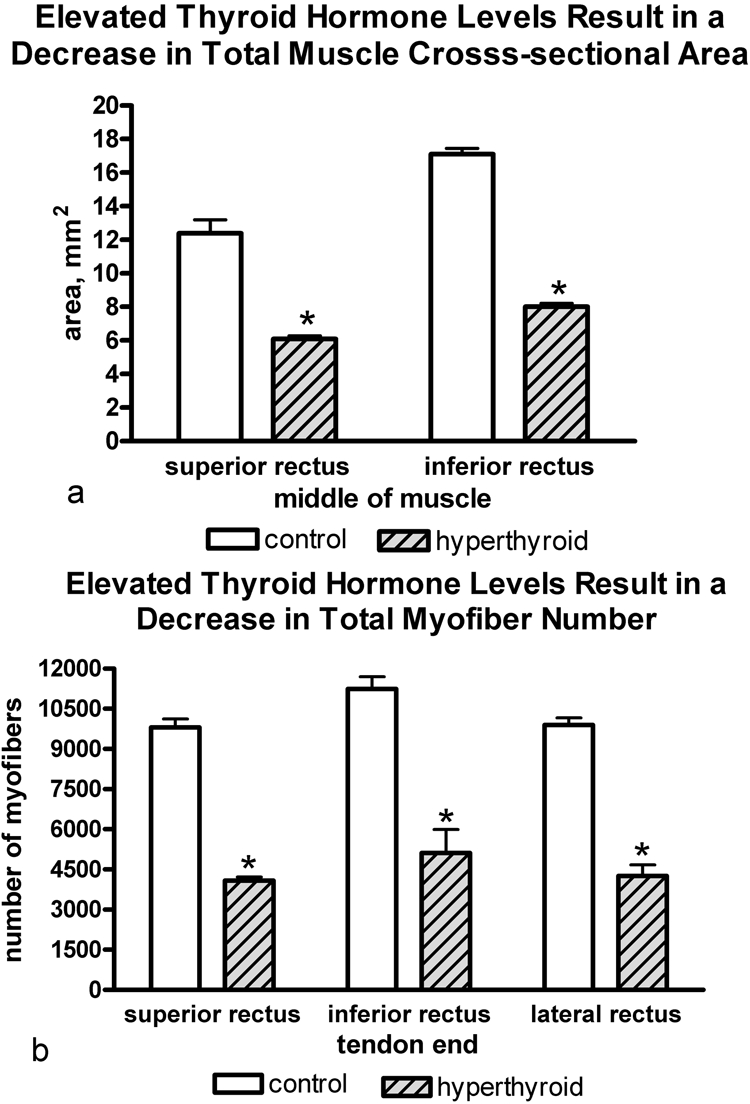
(a) There was a significant decrease in overall muscle mass as indicated by midbelly cross-sectional areas in the hyperthyroid EOMs in all rectus muscles studied (SR and IR shown here) compared with control EOMs. Individual myofiber cross-sectional areas were unchanged. (b) There was a decrease in the total number of myofibers in all three muscles compared with control EOMs. *Significant difference from controls. Data are the mean ± SEM.
Figure 2.
Mean cross-sectional areas for (a) fast, (b) slow, (c) developmental, and (d) neonatal MyHC-positive myofibers from medial rectus muscles of rabbits exposed to elevated blood thyroid hormone levels compared with age-matched controls. *Significant difference from control sections. Data are the mean ± SEM.
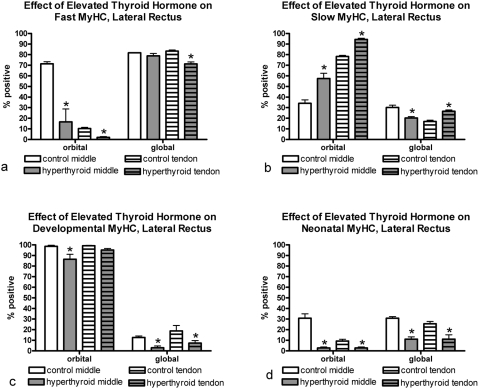
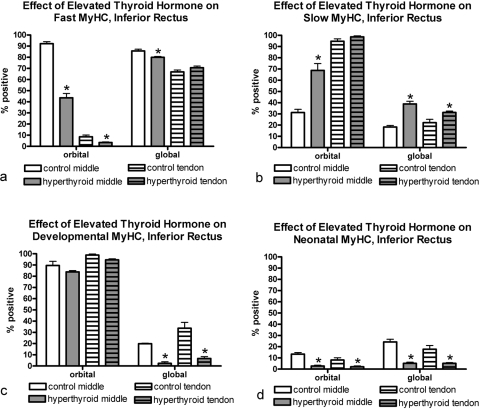
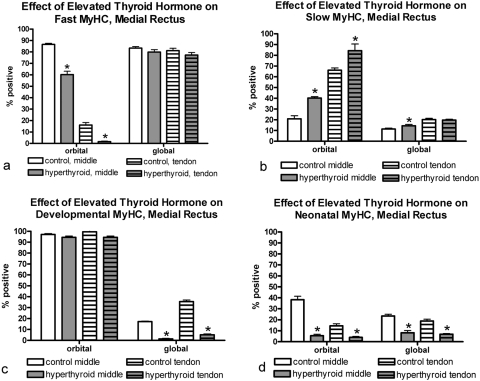
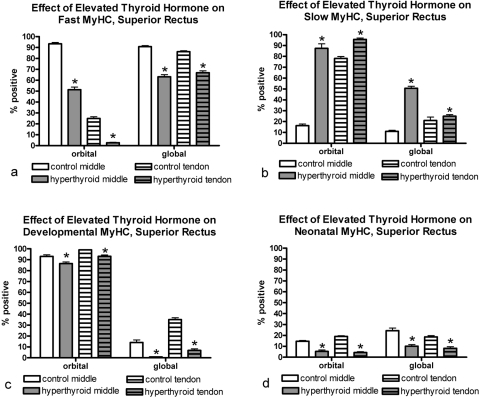
Elevated thyroid hormone levels resulted in complex changes in MyHC composition in the four rectus muscles examined compared with age-matched control subjects (Figs. 3–7). Specifically, there was a decrease in the percentage of myofibers positive for the fast MyHC isoform in the orbital layer of all muscles examined and a large decrease in percentage of fast myofibers in the global layer of only the superior rectus muscle (Fig. 4). Several of the muscles showed a decreased number of fast MyHC-positive myofibers in the tendon region, most pronounced in the superior and lateral rectus muscles. Elevated thyroid hormone levels resulted in an increase in the percentage of myofibers positive for slow myosin in both the orbital and global regions from each of the four EOMs examined (Figs. 4–7), although the difference was not always statistically significant. Note that percentages of fast and slow myofibers do not add up to 100%; it appears that elevated thyroid hormone levels resulted in an increased number of hybrid myofibers.
Figure 3.
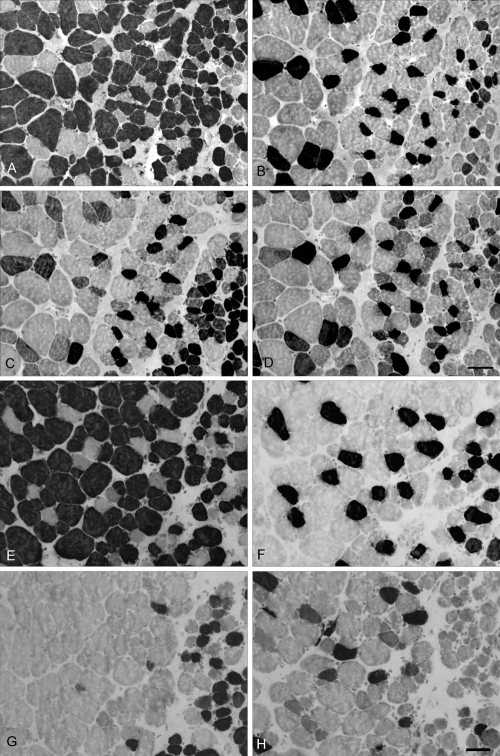
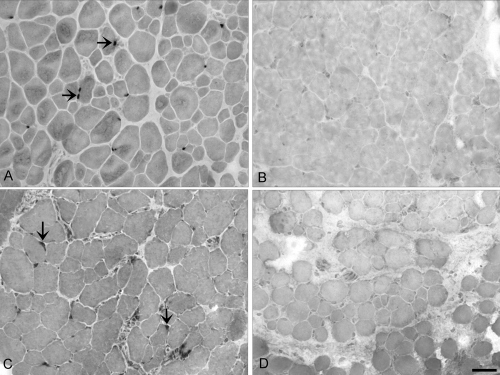
Extraocular lateral rectus muscle sections: (A–D) control; (E–H) hyperthyroid rabbits immunostained for the presence of (A, E) fast, (B, F) slow, (C, G) developmental, and (D, H) neonatal MyHC isoforms. Positive myofibers appear black. Note that the global layer is to the left in all images and a portion of the orbital layer can be seen at right. These sections are intermediate between the midbelly and tendon ends. Note the decrease in myofibers positive for developmental and neonatal MyHC isoforms in the muscles from the hyperthyroid rabbit. Bar, 20 μm.
Figure 4.
Quantification of the percentage of myofibers in the lateral rectus muscle from hyperthyroid and control sections positive for (a) fast, (b) slow, (c) developmental, and (d) neonatal MyHC isoforms. *Significant difference from control sections. Data are the mean ± SEM.
Figure 5.
Quantification of the percentage of myofibers in the inferior rectus muscle from hyperthyroid and control sections positive for (a) fast, (b) slow, (c) developmental, and (d) neonatal MyHC isoforms. *Significant difference from control sections. Data are the mean ± SEM.
Figure 6.
Quantification of the percentage of myofibers in the medial rectus muscle from hyperthyroid and control sections positive for (a) fast, (b) slow, (c) developmental, and (d) neonatal MyHC isoforms. *Significant difference from control sections. Data are the mean ± SEM.
Figure 7.
Quantification of the percentage of myofibers in the superior rectus muscle from hyperthyroid and control sections positive for (a) fast, (b) slow, (c) developmental, and (d) neonatal MyHC isoforms. *Significant difference from control sections. Data are the mean ± SEM.
The changes in expression patterns of the immature MyHC isoforms were relatively similar for the four rectus muscles (Figs. 3–7). Each showed a significant decrease in the percentage of myofibers positive for the neonatal MyHC isoform in all layers in both regions of the muscle. The percentage of myofibers positive for the developmental MyHC isoform decreased in the global layers of all muscles examined. Whereas the superior rectus muscles showed a significant reduction in the percentage of developmental positive myofibers in the orbital layer, the other three rectus muscles demonstrated only a trend toward a decreased number in the orbital layer.
Elevated thyroid hormone levels resulted in a significant reduction in the number of pax7-positive cells, even after substantially increasing antibody incubation times (Figs. 8, 9). In addition, the virtual disappearance of MyoD-positive cells in the EOM cross-sections in both layers of all regions of the hyperthyroid muscles was seen (Fig. 8).
Figure 8.
Extraocular inferior rectus muscle sections from (A, B) control and (C, D) hyperthyroid rabbits immunostained for the presence Pax7 (A, B) and MyoD (C, D). Arrows: positive nuclei. Note the complete absence of positive cells in the hyperthyroid muscle samples. Bar, 20 μm.
Figure 9.

Quantification of the number of pax7-positive satellite cells in the control and hyperthyroid rectus muscles.
Discussion
Elevated thyroid hormone levels resulted in significant decreases in single myofiber and total muscle cross-sectional areas and a decrease number of myofibers in the EOM in rabbits, a phenomenon observed in orbicularis oculi muscle from rabbits with hyperthyroidism.32 It is particularly important to point out that no alterations in connective tissue were seen in the thyroid hormone–treated EOMs, similar to what we saw in the eyelid muscles. Loss of satellite cells positive for both Pax7, the general satellite cell marker, and MyoD, a marker of activated satellite cells, was seen. A complex pattern of changes in the four MyHC isoforms examined emerged. Generally, increases in the percentage of slow MyHC isoforms fibers were seen, and decreases were seen in the percentages of fast, developmental, and neonatal MyHC isoform expressing fibers. It appears that as little as 1 month of elevated thyroid hormone levels can result in a complex set of MyHC isoform alterations in the EOM myofiber populations, in both the orbital and global layers along the length of the muscle from insertion toward the midbelly region. Further studies should address changes in the more EOM-specific MyHC isoforms, as this type of detailed analysis was beyond the scope of the present study. These changes are summarized and graphically depicted in Figure 10. In the hyperthyroid extraocular muscle, the total muscle cross-sectional area and individual myofiber cross-sectional areas are reduced, there is a reduction in expression of immature MyHC isoforms, in this case neonatal MyHC is depicted, and a decrease in cells positive for the MyoD and Pax7 markers.
Figure 10.
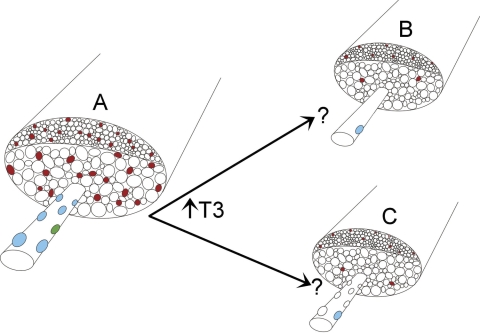
Depiction of the changes that occur in the extraocular muscles after exposure to elevated thyroid hormone. Relative to the normal muscle depicted on the left, the orbital and global layers, superiorly and inferiorly placed on the drawing, show decreased overall muscle cross-sectional area, decreased individual myofiber cross-sectional areas, and decreased number of fibers. In addition, immature MyHC isoform expression decreases, with neonatal MyHC isoform changes depicted here. There is a loss of cells expressing Pax7 and MyoD. Two alternative hypotheses for the disappearance of pax7-positive myogenic precursor cells (satellite cells) are proposed. Elevated thyroid hormone causes a decrease in the number of these cells, due to fusion into existing fibers or apoptosis or to a loss of Pax7 expression. Left: a normal extraocular muscle. (A) One fiber, with several satellite cells, is depicted: Pax7-positive (blue)and MyoD-positive (green). Right: two possible explanations for the loss of Pax7-positive cells. Either (B) all but a very few disappear, or (C) the cells remain but they no longer express Pax7.
Although measurements of muscle force were not determined in the present study, alteration in myofiber size, number of myofibers, and MyHC isoform composition would have a significant impact on force development. In animal studies, elevated thyroid hormone levels cause significant reduction in force production in both diaphragm and soleus muscles, fast and slow muscles, respectively.6,33 When the effect of elevated thyroid hormone on EOM force was assessed in patients with both active and chronic, mild and severe TED, the muscles from all patients showed abnormal tension, even those with no clinical signs of eye movement disorders.34 The authors hypothesize that this abnormality had to be due to direct EOM changes in these patients, and this hypothesis is supported by our current analysis. Although magnetic resonance imaging studies demonstrate significant enlargement of one or more of the EOM in patients with TED, these patients have already developed eye signs. Enlargement of the muscles is due to inflammatory processes within the muscle, and to our knowledge, the muscle itself in these patients has not been studied. This problem will be difficult to solve, as hyperthyroid humans could not reasonably have biopsies of eye muscles taken, nor is EOM surgery a part of normal treatment for patients in whom TED develops. Ongoing experiments in the laboratory are focused on whether and when the EOMs return to normal size after the rabbits return to control levels of thyroid hormone. Our prediction, based on studies of hyperthyroid patients in whom muscle strength eventually returns to normal levels after they are maintained for 6 months in the normal euthyroid state, is that the same will occur in rabbit EOMs.35
Decreases in the number of myofibers, mean myofiber cross-sectional area, and total muscle cross-sectional area are not surprising, given the well-known catabolic effects of thyroid hormone on skeletal muscle in general. In the EOM, we propose that the catabolic effects of increased myofibrillar protein degradation caused by elevation in thyroid hormone are exacerbated by the normal process of myofiber remodeling that occurs throughout life, which is not present in normal adult limb skeletal muscle.11,12 Elevating thyroid hormone levels in animals during normal limb skeletal muscle development results in an accelerated process of differentiation.8–10,36 As would be predicted by the premature withdrawal from the cell cycle, premature myoblast fusion, and accelerated differentiation in developing limb skeletal muscle exposed to elevated thyroid hormone levels,8 increased thyroid hormone levels in EOMs resulted in smaller muscles presumably because of the same effects of thyroid hormone on the satellite cell populations within the EOM. Based on immunostaining alone, it is unclear whether there is an overall reduction in the total number of satellite cells in these muscles, as indicated by the decrease in Pax7-positive cells, or there is a decrease in the expression of Pax7 within individual cells (Fig. 10). The net result, however, of either scenario is that less muscle regeneration/repair would be possible, and thus a smaller muscle would be predicted.
The decrease in the percentage of EOM myofibers expressing the neonatal and developmental MyHC isoforms in most of the rectus muscles examined mirrors the accelerated loss of these immature isoforms in developing skeletal muscle under conditions of elevated thyroid hormone.28 Thus, the overall decrease in the percentage of EOM myofibers expressing these immature MyHC isoforms is compatible with current knowledge about the effects of elevated thyroid hormone on nonocular skeletal muscle differentiation processes and rates of differentiation.
Elevated levels of thyroid hormones are known to hinder muscle repair and cause skeletal muscle atrophy in adult nonocular skeletal muscle.37 These processes would explain the reduction in overall muscle cross-sectional area seen in the present study. There are three processes that can be proposed to explain the decreased myofiber size and overall myofiber number. First, it is known that the EOMs continue to express various myogenic growth factors, including IGF, in the adult.38 Previous studies demonstrated that the addition of exogenous IGF results in increased skeletal muscle mass.39,40 Since IGF is regulated by thyroid hormone, reduction of this myogenic growth factor would result in reduced muscle mass.41 This hypothesis is currently under investigation. Second, a significant reduction in the number of satellite cells positive for the myogenic regulatory factor MyoD, a marker of activated satellite cells,42 was seen. This means that elevated thyroid hormone levels appear to decrease the process that controls myofiber remodeling in adult rabbit EOMs.12,43 Inhibition of ongoing myofiber remodeling would be predicted to result in decreased muscle mass, as seen in the present study. Other laboratories have demonstrated that thyroid hormone specifically inhibits satellite cell proliferation in vitro,8,44 which again supports the present results. Finally, elevated levels of thyroid hormone can stimulate apoptosis in muscle cells.5,45,46 Of interest, the basal level of cell turnover in normal thyroid glands is regulated by apoptosis, which in turn is altered in several thyroid diseases.47 In light of these studies, similar changes in rates of cell turnover in the EOM are not surprising. Studies of the effect of thyroid hormone on apoptosis in the EOMs are ongoing. However, as the effects of both these processes would be to decrease muscle mass, the overall decrease in myofiber size and number would be expected. Elevated thyroid hormone levels cause many other changes in cells. In particular, it is known that the EOMs are particularly rich in mitochondria and that these mitochondria display metabolic differences compared with normal limb muscle.48 As elevated thyroid hormones alter mitochondrial uncoupling proteins,7 this and other aspects of the unique metabolism of EOMs would certainly play a role in alterations in these muscles under these conditions.49 Based on the appearance of the muscles, as seen in Figure 3, no fibrosis was observed. Thus, the satellite cells did not change their differentiation pathway toward a fibroblast fate, nor is it likely that the myofibers themselves would have transdifferentiated into connective tissue.
The alterations of muscle size and myosin isoform expression were relatively uniform among the four rectus muscles despite their significant differential involvement in TED in humans.50 Thus, it appears that these particular characteristics would not contribute to the differential susceptibility of individual EOM to TED, where the medial and inferior rectus muscles are affected more often.51 However, as any type of increased cell loss can trigger release of inflammatory mediators,52 it may be that the myofiber loss plays a role in initiation of the inflammatory cascade in this disease.
The effects of elevated thyroid hormone levels on fast and slow MyHC isoform composition have been studied in nonocular adult skeletal muscles. Most skeletal muscles, when subjected to elevated thyroid hormone, shift their overall MyHC composition in the opposite direction from their normal state; thus, in the soleus, a muscle predominantly containing slow MyHC-positive myofibers, elevated thyroid hormone levels result in a shift toward an increasing number of myofibers positive for fast myosin.16,53 In the plantaris, a muscle normally containing predominantly fast MyHC-positive myofibers, elevated thyroid hormone results in a shift toward an increased number of slow myofibers.54 In orbicularis oculi from hyperthyroid rabbits, which is also a muscle expressing predominantly fast-positive myofibers, similarly there is a significant increase in the percentage of myofibers expressing the slow MyHC.32 It is unclear what controls these patterns of fast and slow MyHC transitions under hyperthyroid conditions. One factor may be the number of thyroid hormone receptors on the nuclei within each muscle type, as the EOMs express an increased number of thyroid hormone receptors compared with limb skeletal muscles27; however, it is unknown whether receptor expression varies between fiber types. Thus, the effects of thyroid hormone on metabolism and MyHC phenotype appear to be extremely complex and fiber specific.53,54
The rabbit model used in the present study addresses changes in the EOMs caused by increased systemic thyroid hormone levels. Human TED is a complex autoimmune inflammatory response in the EOM, and it must be stressed that it is not the same as simple hyperthyroidism. However, it is evident from our study that hyperthyroidism itself leads to significant changes in extraocular muscle metabolism, such that the muscles are reduced in size, have altered populations of satellite cells (the muscle regenerative cell), as well as altered expression of MHC isoforms in populations of myofibers in the EOMs. As clinically relevant eye signs are seen in at least 50% of patients with Graves' disease,54 changes within the EOM with increased thyroid hormone levels may suggest new testable hypotheses. It may be that TED is a result of a set of predisposing factors in patients, but it only develops if some type of precipitating factor is added as occurs with blepharospasm.55 Additionally, muscle catabolism may expose nuclear antigens to the immune system, suggesting new directions for future studies. It is hoped that a greater understanding of the changes in the EOM as a result of increased thyroid hormone may suggest alternative strategies for manipulating the muscles in the acute phase of patients who develop this potentially blinding disease.
Footnotes
Supported by Grants EY11374 and EY15313 from the National Eye Institute, the Minnesota Lions and Lionesses, the Lew Wasserman Mid-Career Merit Award from Research to Prevent Blindness (RPB), and an unrestricted grant to the Department of Ophthalmology from RPB Inc.
Disclosure: A.R. Harrison, None; M.S. Lee, None; L.K. McLoon, None
References
- 1.Porter JD, Baker RS. Muscles of a different color: the unusual properties of the extraocular muscles may predispose or protect them in neurogenic and myogenic disease. Neurology 1996; 46: 30–37 [DOI] [PubMed] [Google Scholar]
- 2.Bartley GB. The epidemiologic characteristics and clinical course of ophthalmopathy associated with autoimmune thyroid disease in Olmstead county, Minnesota. Trans Am Ophthalmol Soc 1994; 92: 477–588 [PMC free article] [PubMed] [Google Scholar]
- 3.Bahn RS, Dutton CM, Natt N, et al. Thyrotropin receptor expression in Graves' orbital adipose/connective tissues: potential autoantigen in Graves' ophthalmopathy. J Clin Endocrinol Metab 1998; 83: 998–1002 [DOI] [PubMed] [Google Scholar]
- 4.Ramsay ID. Muscle dysfunction in hyperthyroidism. Lancet 1966; 2: 931–934 [DOI] [PubMed] [Google Scholar]
- 5.Nakashima K, Ohtsuka A, Hayashi K. Comparison of the effects of thyroxine and triiodothyronine on protein turnover and apoptosis in primary chick muscle cell cultures. Biochem Biophys Res Commun 1998; 251: 442–448 [DOI] [PubMed] [Google Scholar]
- 6.Yamada T, Mishima T, Sakamoto M, Sugiyama M, Matsunaga S, Wada M. Oxidation of myosin heavy chain and reduction in force production in hyperthyroid rat soleus. J Appl Physiol 2006; 100: 1520–1526 [DOI] [PubMed] [Google Scholar]
- 7.Brennan MD, Coenen-Schimke JM, Bigelow ML, Nai KS. Changes in skeletal muscle protein metabolism and myosin heavy chain isoform messenger ribonucleic acid abundance after treatment of hyperthyroidism. J Clin Endocrinol Metab 2006; 91: 4650–4656 [DOI] [PubMed] [Google Scholar]
- 8.Marchal S, Cassar-Malek I, Pons F, Wrutniak C, Cabello G. Triiodothyronine influences quail myoblast proliferation and differentiation. Biol Cell 1993; 78: 191–197 [DOI] [PubMed] [Google Scholar]
- 9.Carnac G, Albagli-Curiel O, Vandromme M, et al. 3,5,3′-triiodothyronine positively regulates both MyoD1 gene transcription and terminal differentiation in C2 myoblasts. Mol Endocrinol 1992; 6: 1185–1194 [DOI] [PubMed] [Google Scholar]
- 10.Marchal S, Cassar-Malek I, Magaud JP, Rouault JP, Wrutniak C, Cabello G. Stimulation of avian myoblast differentiation by triiodothyronine: possible involvement of the cAMP pathway. Exp Cell Res 1995; 220: 1–10 [DOI] [PubMed] [Google Scholar]
- 11.McLoon LK, Wirtschafter JD. Continuous myonuclear addition to single extraocular myofibers of uninjured adult rabbits. Muscle Nerve 2002; 25: 348–358 [DOI] [PubMed] [Google Scholar]
- 12.McLoon LK, Rowe J, Wirtschafter JD, McCormick KM. Continuous myofiber remodeling in uninjured extraocular myofibers: myonuclear turnover and evidence for apoptosis. Muscle Nerve 2004; 29: 707–715 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gambke B, Lyons GE, Haselgrove J, Kelly A, Rubinstein NA. Thyroidal and neural control of myosin transitions during development of rat fast and slow muscles. FEBS Lett 1983; 156: 335–339 [DOI] [PubMed] [Google Scholar]
- 14.Lyons G, Kelly A, Rubinstein N. Neural and thyroid control of IIb myosin heavy chain and its mRNA during development. Basic Appl Myol 1993; 3: 37–46 [Google Scholar]
- 15.Izumo S, Nadal-Ginard B, Mahdavi V. All members of the MHC multigene family respond to thyroid hormone in a highly tissue-specific manner. Science 1986; 231: 597–600 [DOI] [PubMed] [Google Scholar]
- 16.Yu F, Degens H, Li X, Larsson L. Gender and age-related differences in the regulatory influence of thyroid hormone on the contractility and myosin composition of single rat soleus muscle fibres. Pflugers Arch 1998; 437: 21–30 [DOI] [PubMed] [Google Scholar]
- 17.Caiozzo VJ, Herrick RE, Balwin KM. Influence of hyperthyroidism on maximal shortening velocity and myosin isoform distribution in skeletal muscles. Am J Physiol 1991; 261: C285–C295 [DOI] [PubMed] [Google Scholar]
- 18.Caiozzo VJ, Herrick RE, Balwin KM. Response of slow and fast muscle to hypothyroidism: maximal shortening velocity and myosin isoforms. Am J Physiol 1992; 263: C86–C94 [DOI] [PubMed] [Google Scholar]
- 19.Caiozzo VJ, Baker MH, McCue SA, Baldwin KM. Single fiber and whole muscle analyses of MyHC isoform plasticity: an interaction between thyroid hormone and mechanical unloading. Am J Physiol Cell Physiol 1997; 273: C944–C952 [DOI] [PubMed] [Google Scholar]
- 20.Wieczorek DF, Periasamy M, Butler-Browne GS, Whalen RG, Nadal-Ginard B. Co-expression of multiple myosin heavy chain genes, in addition to a tissue-specific one, in extraocular muscles. J Cell Biol 1985; 101: 618–629 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Asmussen G, Traub I, Pette D. Electrophoretic analysis of myosin heavy chain isoform patterns in extraocular muscles of the rat. FEBS Lett 1993; 335: 243–245 [DOI] [PubMed] [Google Scholar]
- 22.Jacoby J, Ko K, Weiss C, Rushbrook J. Systemic variation in myosin expression along extraocular muscle fibers of the adult rat. J Muscle Res Cell Motil 1989; 11: 25–40 [DOI] [PubMed] [Google Scholar]
- 23.Jacoby J, Ko K. Sarcoplasmic reticulum fast Ca2+-pump and myosin heavy chain expression in extraocular muscles. Invest Ophthalmol Vis Sci 1993; 4: 2848–2858 [PubMed] [Google Scholar]
- 24.McLoon LK, Rios L, Wirtschafter JD. Complex three-dimensional patterns of myosin isoform expression: differences between and within specific extraocular muscles. J Muscle Res Cell Motil 1999; 20: 771–783 [DOI] [PubMed] [Google Scholar]
- 25.Rubinstein N, Hoh J. The distribution of myosin heavy chain isoforms among rat extraocular muscle fiber types. Invest Ophthalmol Vis Sci 2000; 41: 3391–3398 [PubMed] [Google Scholar]
- 26.Kjellgren D, Thornell LE, Andersen J, Pedrosa-Domellof F. Myosin heavy chain isoforms in human extraocular muscles. Invest Ophthalmol Vis Sci 2003; 44: 1419–1425 [DOI] [PubMed] [Google Scholar]
- 27.Schmidt ED, Schmidt EDL, van der Gaag R, et al. Distribution of the nuclear thyroid-hormone receptor in extraocular and skeletal muscles. J Endocrinol 1992; 133: 67–74 [DOI] [PubMed] [Google Scholar]
- 28.D'Albis A, Lenfant-Guyot M, Janmot C, Chanoine C, Weinman J, Callien CL. Regulation by thyroid hormones of terminal differentiation in the skeletal dorsal muscle. I. Neonate mouse. Dev Biol 1987; 123: 25–32 [DOI] [PubMed] [Google Scholar]
- 29.Nichol C, Johnston IA. Energy metabolism of fast- and slow-twitch skeletal muscle in the rat: thyroid hormone induced changes. J Comp Physiol 1981; 142: 465–472 [Google Scholar]
- 30.Nwoye L, Mommaerts W, Simpson D, et al. Evidence for direct action of thyroid hormone in specifying muscle properties. Am J Physiol 1982; 242: R401–R408 [DOI] [PubMed] [Google Scholar]
- 31.d'Albis A, Couteaux R, Janmot C, Mira CJ. Opposite regulations by androgenic and thyroid hormones of VI myosin expression. FEBS Lett 1993; 318: 53–56 [DOI] [PubMed] [Google Scholar]
- 32.Harrison A, McLoon LK. Effect of hyperthyroidism on the orbicularis oculi muscle in rabbits. Ophthalmic Plast Reconst Surg 2002; 18: 289–294 [DOI] [PubMed] [Google Scholar]
- 33.Simonides WS, van Hardeveld C. Thyroid hormone as a determinant of metabolic and contractile phenotype of skeletal muscle. Thyroid 2008; 18: 205–216 [DOI] [PubMed] [Google Scholar]
- 34.Tian S, Bolzani R, Banassi M, Lennerstrand G. Eye muscle force development in thyroid-associated ophthalmopathy in different stages of disease. Acta Ophthalmol Scand 2007; 85: 431–437 [DOI] [PubMed] [Google Scholar]
- 35.Olson BR, Klein I, Benner R, Burdett R, Trzepacz P, Levey GS. Hyperthyroid myopathy and the response to treatment. Thyroid 1991; 137–141 [DOI] [PubMed] [Google Scholar]
- 36.Butler-Browne GS, Barbet JP, Thornell LE. Myosin heavy and light chain expression during human skeletal muscle development and precocious muscle maturation induced by thyroid hormone. Anat Embryol 1990; 181: 513–522 [DOI] [PubMed] [Google Scholar]
- 37.Yamada T, Mishima T, Sakamoto M, Sugiyama M, Matsunaga S, Wada M. Myofibrillar protein oxidation and contractile dysfunction in hyperthyroid rat diaphragm. J Appl Physiol 2007; 102: 1850–1855 [DOI] [PubMed] [Google Scholar]
- 38.McLoon LK, Christiansen SP. Increasing extraocular muscle strength with insulin-like growth factor II. Invest Ophthalmol Vis Sci 2003; 44: 3866–3872 [DOI] [PubMed] [Google Scholar]
- 39.McKoy G, Ashley W, Mander J, et al. Expression of insulin growth factor-1 splice variants and structural genes in rabbit skeletal muscle induced by stretch and stimulation. J Physiol 1999; 516: 583–592 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Goldspink G. Changes in muscle mass and phenotype and the expression of autocrine and systemic growth factors by muscle in response to stretch and overload. J Anat 1999; 194: 323–334 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Thelen MH, Muller A, Zuidwijk MJ, van der Linden GC, Simonides WS, van Hardeveld C. Differential regulation of the expression of fast-type sarcoplasmic-reticulum Ca(2+)-ATPase by thyroid hormone and insulin-like growth factor-I in the L6 mouse cell line. Biochem J 1994; 15: 467–474 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Eppley ZA, Kim J, Russell B. A myogenic regulatory gene, gmf1, is expressed by adult myonuclei after injury. Am J Physiol 1993; 265: C397–C405 [DOI] [PubMed] [Google Scholar]
- 43.McLoon LK, Wirtschafter J. Activated satellite cells in extraocular muscles of normal adult monkeys and humans. Invest Ophthalmol Vis Sci 2003; 44: 1927–1932 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Cassar-Malek I, Langlois N, Picard B, Geay Y. Regulation of bovine satellite cell proliferation and differentiation by insulin and triiodothyronine. Domest Anim Endocrinol 2007; 33: 91–106 [DOI] [PubMed] [Google Scholar]
- 45.Nishikawa A, Hayashi H. Spatial, temporal and hormonal regulation of programmed muscle cell death during metamorphosis of the frog Xenopus laevis. Differentiation 1995; 59: 207–214 [DOI] [PubMed] [Google Scholar]
- 46.Yaoita Y, Nakajima K. Induction of apoptosis and CPP32 expression by thyroid hormone in a myoblastic cell line derived from tadpole tail. J Biol Chem 1997; 21: 5122–5127 [DOI] [PubMed] [Google Scholar]
- 47.Arscott PL, Baker JR. Apoptosis and thyroiditis. Clin Immunol Immunopathol 1998; 87: 207–217 [DOI] [PubMed] [Google Scholar]
- 48.Patel SP, Gamboa JL, McMullen CA, Rabchevsky A, Andrade FH. Lower respiratory capacity in extraocular muscle mitochondria: evidence for intrinsic differences in mitochondrial composition and function. Invest Ophthalmol Vis Sci 2009; 60: 180–186 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Andrade FH, McMullen CA, Rumbaut RE. Mitochondria are fast Ca2+ sinks in rat extraocular muscles: a novel regulatory influence on contractile function and metabolism. Invest Ophthalmol Vis Sci 2005; 46: 4541–4547 [DOI] [PubMed] [Google Scholar]
- 50.Buckley EG. Eye movement disorders. In: Wright KW. ed. Textbook of Ophthalmology Baltimore: Williams and Wilkins; 1997; 177–195 [Google Scholar]
- 51.Feldon SE, Weiner JM. Clinical significance of extraocular muscle volumes in Graves' ophthalmopathy: a quantitative computed tomography study. Arch Ophthalmol 1982; 100: 1266–1269 [DOI] [PubMed] [Google Scholar]
- 52.Borgerson KL, Bretz JD, Baker JR. The role of Fas-mediated apoptosis in thyroid autoimmune disease. Autoimmunity 1999; 30: 251–264 [DOI] [PubMed] [Google Scholar]
- 53.Caiozzo VJ, Swoap S, Tao M, et al. Single fiber analyses of type IIA MHC distribution in hyper- and hypothyroid soleus. Am J Physiol 1993; 265: C842–C849 [DOI] [PubMed] [Google Scholar]
- 54.Haddad F, Arnold C, Zeng M, Baldwin K. Interaction of thyroid state and denervation on skeletal myosin heavy chain expression. Muscle Nerve 1997; 20: 1487–1496 [DOI] [PubMed] [Google Scholar]
- 55.Evinger C, Bao JB, Powers AS, et al. Dry eye, blinking, and blepharospasm. Mov Disord 2002; 2( suppl): S75–S78 [DOI] [PMC free article] [PubMed] [Google Scholar]