Abstract
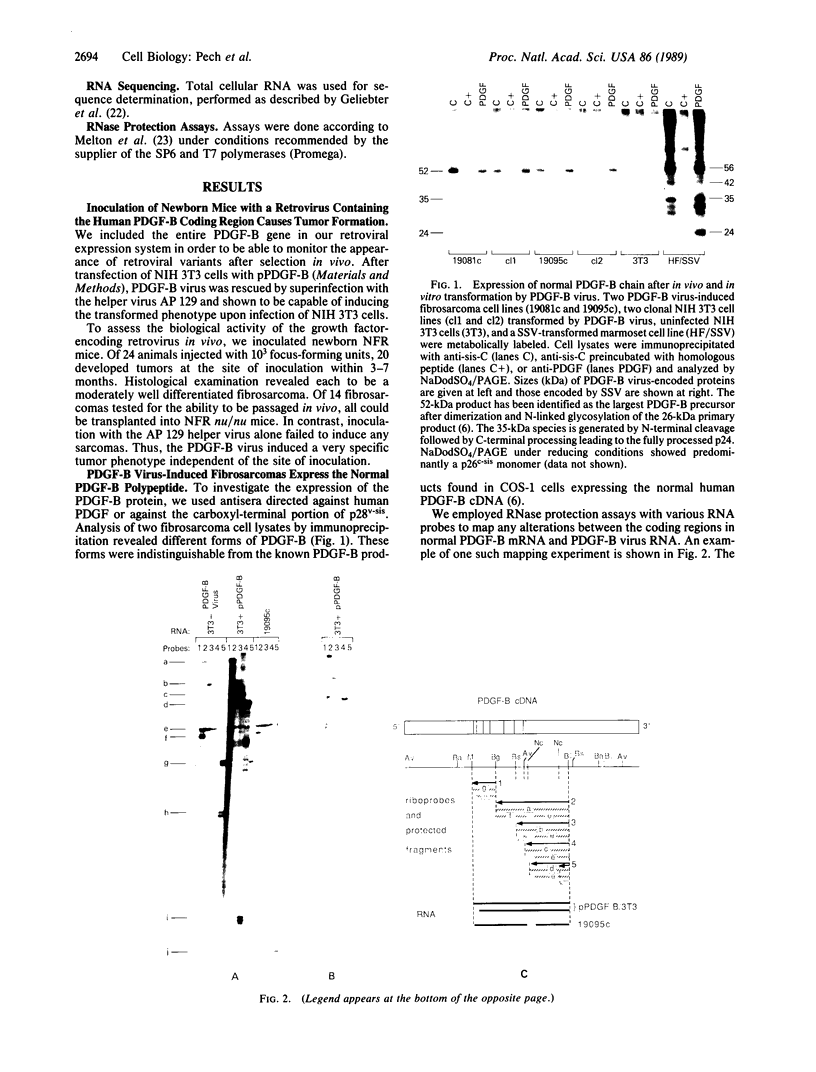
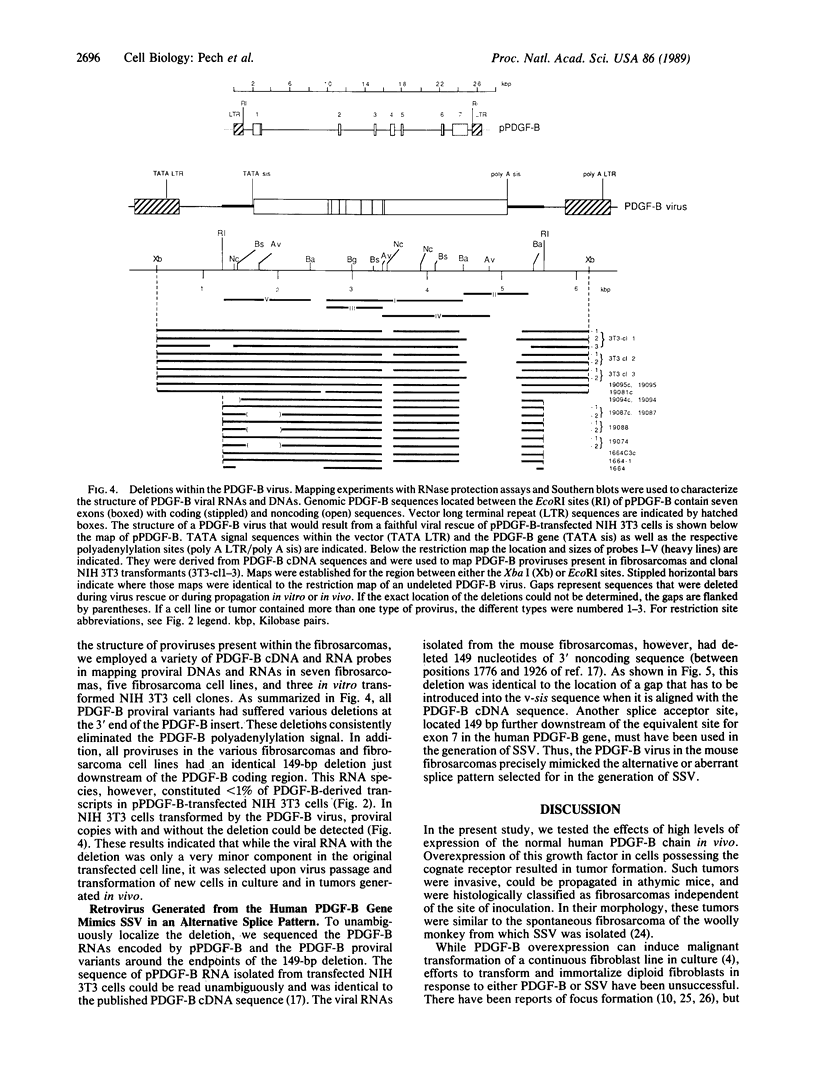
A retrovirus containing the entire human platelet-derived growth factor B-chain (PDGF-B) gene was constructed in order to investigate the in vivo biological activity of its encoded growth factor. When this virus was introduced into newborn mice, it reproducibly generated fibrosarcomas at the site of inoculation. Proviruses in each fibrosarcoma analyzed had lost 149 nucleotides downstream of the PDGF-B coding region. This deletion originated from an alternative or aberrant splice event that occurred within exon 7 of the PDGF-B gene and mimicked the v-sis oncogene. Thus, deletion of this region may be necessary for efficient retrovirus replication or for more potent transforming function. Evidence that the normal growth factor coding sequence was unaltered derived from RNase protection studies and immunoprecipitation analysis. Tumors were generally polyclonal but demonstrated clonal subpopulations. Moreover, tumor-derived cell lines became monoclonal within a few tissue culture passages and rapidly formed tumors in vivo. These findings argue that overexpression of the normal human PDGF-B gene product under retrovirus control can induce the fully malignant phenotype.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bishop J. M. The molecular genetics of cancer. Science. 1987 Jan 16;235(4786):305–311. doi: 10.1126/science.3541204. [DOI] [PubMed] [Google Scholar]
- Chiu I. M., Reddy E. P., Givol D., Robbins K. C., Tronick S. R., Aaronson S. A. Nucleotide sequence analysis identifies the human c-sis proto-oncogene as a structural gene for platelet-derived growth factor. Cell. 1984 May;37(1):123–129. doi: 10.1016/0092-8674(84)90307-6. [DOI] [PubMed] [Google Scholar]
- Doolittle R. F., Hunkapiller M. W., Hood L. E., Devare S. G., Robbins K. C., Aaronson S. A., Antoniades H. N. Simian sarcoma virus onc gene, v-sis, is derived from the gene (or genes) encoding a platelet-derived growth factor. Science. 1983 Jul 15;221(4607):275–277. doi: 10.1126/science.6304883. [DOI] [PubMed] [Google Scholar]
- Duesberg P. H. Cancer genes: rare recombinants instead of activated oncogenes (a review). Proc Natl Acad Sci U S A. 1987 Apr;84(8):2117–2124. doi: 10.1073/pnas.84.8.2117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eva A., Robbins K. C., Andersen P. R., Srinivasan A., Tronick S. R., Reddy E. P., Ellmore N. W., Galen A. T., Lautenberger J. A., Papas T. S. Cellular genes analogous to retroviral onc genes are transcribed in human tumour cells. Nature. 1982 Jan 14;295(5845):116–119. doi: 10.1038/295116a0. [DOI] [PubMed] [Google Scholar]
- Gardner M. B., Officer J. E., Rongey R. W., Charman H. P., Hartley J. W., Estes J. D., Huebner R. J. C-type RNA tumor virus in wild house mice (Mus musculus). Bibl Haematol. 1973;39:335–344. doi: 10.1159/000427860. [DOI] [PubMed] [Google Scholar]
- Gazit A., Igarashi H., Chiu I. M., Srinivasan A., Yaniv A., Tronick S. R., Robbins K. C., Aaronson S. A. Expression of the normal human sis/PDGF-2 coding sequence induces cellular transformation. Cell. 1984 Nov;39(1):89–97. doi: 10.1016/0092-8674(84)90194-6. [DOI] [PubMed] [Google Scholar]
- Gazit A., Pierce J. H., Kraus M. H., Di Fiore P. P., Pennington C. Y., Aaronson S. A. Mammalian cell transformation by a murine retrovirus vector containing the avian erythroblastosis virus erbB gene. J Virol. 1986 Oct;60(1):19–28. doi: 10.1128/jvi.60.1.19-28.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geliebter J., Zeff R. A., Melvold R. W., Nathenson S. G. Mitotic recombination in germ cells generated two major histocompatibility complex mutant genes shown to be identical by RNA sequence analysis: Kbm9 and Kbm6. Proc Natl Acad Sci U S A. 1986 May;83(10):3371–3375. doi: 10.1073/pnas.83.10.3371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Igarashi H., Rao C. D., Siroff M., Leal F., Robbins K. C., Aaronson S. A. Detection of PDGF-2 homodimers in human tumor cells. Oncogene. 1987 Mar;1(1):79–85. [PubMed] [Google Scholar]
- Jainchill J. L., Aaronson S. A., Todaro G. J. Murine sarcoma and leukemia viruses: assay using clonal lines of contact-inhibited mouse cells. J Virol. 1969 Nov;4(5):549–553. doi: 10.1128/jvi.4.5.549-553.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnsson A., Betsholtz C., Heldin C. H., Westermark B. The phenotypic characteristics of simian sarcoma virus-transformed human fibroblasts suggest that the v-sis gene product acts solely as a PDGF receptor agonist in cell transformation. EMBO J. 1986 Jul;5(7):1535–1541. doi: 10.1002/j.1460-2075.1986.tb04394.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kozak M. Point mutations define a sequence flanking the AUG initiator codon that modulates translation by eukaryotic ribosomes. Cell. 1986 Jan 31;44(2):283–292. doi: 10.1016/0092-8674(86)90762-2. [DOI] [PubMed] [Google Scholar]
- Melton D. A., Krieg P. A., Rebagliati M. R., Maniatis T., Zinn K., Green M. R. Efficient in vitro synthesis of biologically active RNA and RNA hybridization probes from plasmids containing a bacteriophage SP6 promoter. Nucleic Acids Res. 1984 Sep 25;12(18):7035–7056. doi: 10.1093/nar/12.18.7035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parnes J. R., Seidman J. G. Structure of wild-type and mutant mouse beta 2-microglobulin genes. Cell. 1982 Jun;29(2):661–669. doi: 10.1016/0092-8674(82)90182-9. [DOI] [PubMed] [Google Scholar]
- Ponte P., Ng S. Y., Engel J., Gunning P., Kedes L. Evolutionary conservation in the untranslated regions of actin mRNAs: DNA sequence of a human beta-actin cDNA. Nucleic Acids Res. 1984 Feb 10;12(3):1687–1696. doi: 10.1093/nar/12.3.1687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rao C. D., Igarashi H., Chiu I. M., Robbins K. C., Aaronson S. A. Structure and sequence of the human c-sis/platelet-derived growth factor 2 (SIS/PDGF2) transcriptional unit. Proc Natl Acad Sci U S A. 1986 Apr;83(8):2392–2396. doi: 10.1073/pnas.83.8.2392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rao C. D., Igarashi H., Pech M. W., Robbins K. C., Aaronson S. A. Oncogenic potential of the human platelet-derived growth factor transcriptional unit. Cold Spring Harb Symp Quant Biol. 1986;51(Pt 2):959–966. doi: 10.1101/sqb.1986.051.01.109. [DOI] [PubMed] [Google Scholar]
- Rao C. D., Pech M., Robbins K. C., Aaronson S. A. The 5' untranslated sequence of the c-sis/platelet-derived growth factor 2 transcript is a potent translational inhibitor. Mol Cell Biol. 1988 Jan;8(1):284–292. doi: 10.1128/mcb.8.1.284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reddy E. P., Smith M. J., Srinivasan A. Nucleotide sequence of Abelson murine leukemia virus genome: structural similarity of its transforming gene product to other onc gene products with tyrosine-specific kinase activity. Proc Natl Acad Sci U S A. 1983 Jun;80(12):3623–3627. doi: 10.1073/pnas.80.12.3623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Southern P. J., Berg P. Transformation of mammalian cells to antibiotic resistance with a bacterial gene under control of the SV40 early region promoter. J Mol Appl Genet. 1982;1(4):327–341. [PubMed] [Google Scholar]
- Srinivasan A., Dunn C. Y., Yuasa Y., Devare S. G., Reddy E. P., Aaronson S. A. Abelson murine leukemia virus: structural requirements for transforming gene function. Proc Natl Acad Sci U S A. 1982 Sep;79(18):5508–5512. doi: 10.1073/pnas.79.18.5508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stewart T. A., Pattengale P. K., Leder P. Spontaneous mammary adenocarcinomas in transgenic mice that carry and express MTV/myc fusion genes. Cell. 1984 Oct;38(3):627–637. doi: 10.1016/0092-8674(84)90257-5. [DOI] [PubMed] [Google Scholar]
- Temin H. M. Function of the retrovirus long terminal repeat. Cell. 1982 Jan;28(1):3–5. doi: 10.1016/0092-8674(82)90367-1. [DOI] [PubMed] [Google Scholar]
- Theilen G. H., Gould D., Fowler M., Dungworth D. L. C-type virus in tumor tissue of a woolly monkey (Lagothrix spp.) with fibrosarcoma. J Natl Cancer Inst. 1971 Oct;47(4):881–889. [PubMed] [Google Scholar]
- Waterfield M. D., Scrace G. T., Whittle N., Stroobant P., Johnsson A., Wasteson A., Westermark B., Heldin C. H., Huang J. S., Deuel T. F. Platelet-derived growth factor is structurally related to the putative transforming protein p28sis of simian sarcoma virus. Nature. 1983 Jul 7;304(5921):35–39. doi: 10.1038/304035a0. [DOI] [PubMed] [Google Scholar]
- Wigler M., Silverstein S., Lee L. S., Pellicer A., Cheng Y. c., Axel R. Transfer of purified herpes virus thymidine kinase gene to cultured mouse cells. Cell. 1977 May;11(1):223–232. doi: 10.1016/0092-8674(77)90333-6. [DOI] [PubMed] [Google Scholar]
- Wolfe L. G., Deinhardt F., Theilen G. H., Rabin H., Kawakami T., Bustad L. K. Induction of tumors in marmoset monkeys by simian sarcoma virus, type 1 (Lagothrix): a preliminary report. J Natl Cancer Inst. 1971 Nov;47(5):1115–1120. [PubMed] [Google Scholar]