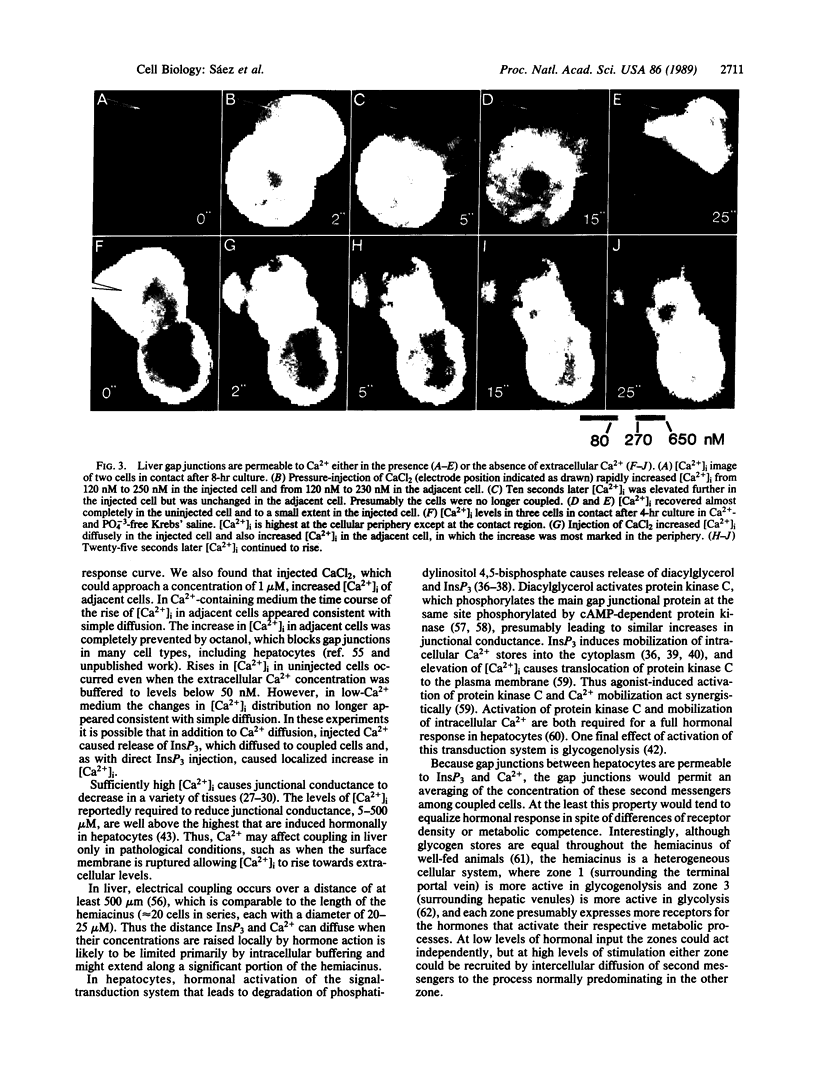
Abstract
Hepatocytes are well coupled by gap junctions, which allow the diffusion of small molecules between cells. Although gap junctions in many tissues are permeable to molecules larger than cAMP and in several preparations gap junctions pass cAMP itself, little direct evidence supports permeation by other second-messenger species. Ca2+, perhaps the smallest second messenger, would be expected to cross gap junctions, but the issue is complicated because gap-junction channels are closed when intracellular free Ca2+ concentration, [Ca2+]i, is elevated to micromolar levels or above. Inositol 1,4,5-trisphosphate (InsP3), a second messenger that can evoke Ca2+ release, might also reduce junctional permeability by this mechanism. We report here evidence for transjunctional flux of Ca2+ and InsP3 in freshly isolated pairs or small clusters of rat hepatocytes. The Ca2+ indicator fura-2 was used to monitor transjunctional diffusion of Ca2+ directly or to detect passage of InsP3 by localized Ca2+ release. Fura-2 injected as the free acid passed between cells. Injection of InsP3 or CaCl2 immediately increased [Ca2+]i in the injected cell (peak values less than 1 microM), and [Ca2+]i increased rapidly in contacting cells (within seconds). The initial rise in [Ca2+]i induced by InsP3 was greater at discrete regions in the cytoplasm of both injected and uninjected cells and was inconsistent with simple diffusion of Ca2+. In the coupled cells the regions of greatest increase were not necessarily near the contact zone. In contrast, the rise induced in [Ca2+]i by CaCl2 injection when cells were bathed in normal Ca2+ was always more diffuse than with InsP3 injection, and in cells coupled to a cell injected with CaCl2 the earliest and maximal increases occurred at the region of cell contact. This difference in distribution indicates that injected InsP3 (or an active metabolite, but not Ca2+) diffused between cells to cause localized release of Ca2+ from intracellular stores. Ca2+ injection induced a rise in [Ca2+]i in coupled cells even when cells were maintained in Ca2+-free saline, suggesting that changes in [Ca2+]i seen in adjacent cells were due to transjunctional diffusion from the injected cell and not to uptake from the extracellular solution. However, in Ca2+-free saline, [Ca2+]i distribution was nonuniform, indicating that Ca2+-releasing mechanisms contribute to the observed changes. No increase in [Ca2+]i was seen in adjacent cells when Ca2+ was injected after treatment with the uncoupling agent octanol (500 microM), which itself did not change [Ca2+]i. These data provide evidence that the second messengers Ca2+ and InsP3 can be transmitted from cell to cell through gap junctions, a process that may have an important role in tissue function.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Atwater I., Rosario L., Rojas E. Properties of the Ca-activated K+ channel in pancreatic beta-cells. Cell Calcium. 1983 Dec;4(5-6):451–461. doi: 10.1016/0143-4160(83)90021-0. [DOI] [PubMed] [Google Scholar]
- Azarnia R., Dahl G., Loewenstein W. R. Cell junction and cycle AMP: III. Promotion of junctional membrane permeability and junctional membrane particles in a junction-deficient cell type. J Membr Biol. 1981;63(1-2):133–146. doi: 10.1007/BF01969454. [DOI] [PubMed] [Google Scholar]
- Bennett M. V., Spira M. E., Spray D. C. Permeability of gap junctions between embryonic cells of Fundulus: a reevaluation. Dev Biol. 1978 Jul;65(1):114–125. doi: 10.1016/0012-1606(78)90184-7. [DOI] [PubMed] [Google Scholar]
- Berridge M. J., Galione A. Cytosolic calcium oscillators. FASEB J. 1988 Dec;2(15):3074–3082. doi: 10.1096/fasebj.2.15.2847949. [DOI] [PubMed] [Google Scholar]
- Berridge M. J., Irvine R. F. Inositol trisphosphate, a novel second messenger in cellular signal transduction. Nature. 1984 Nov 22;312(5992):315–321. doi: 10.1038/312315a0. [DOI] [PubMed] [Google Scholar]
- Berry M. N., Friend D. S. High-yield preparation of isolated rat liver parenchymal cells: a biochemical and fine structural study. J Cell Biol. 1969 Dec;43(3):506–520. doi: 10.1083/jcb.43.3.506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Biegon R. P., Atkinson M. M., Liu T. F., Kam E. Y., Sheridan J. D. Permeance of Novikoff hepatoma gap junctions: quantitative video analysis of dye transfer. J Membr Biol. 1987;96(3):225–233. doi: 10.1007/BF01869304. [DOI] [PubMed] [Google Scholar]
- Burgess G. M., Irvine R. F., Berridge M. J., McKinney J. S., Putney J. W., Jr Actions of inositol phosphates on Ca2+ pools in guinea-pig hepatocytes. Biochem J. 1984 Dec 15;224(3):741–746. doi: 10.1042/bj2240741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burt J. M., Spray D. C. Inotropic agents modulate gap junctional conductance between cardiac myocytes. Am J Physiol. 1988 Jun;254(6 Pt 2):H1206–H1210. doi: 10.1152/ajpheart.1988.254.6.H1206. [DOI] [PubMed] [Google Scholar]
- Charest R., Blackmore P. F., Berthon B., Exton J. H. Changes in free cytosolic Ca2+ in hepatocytes following alpha 1-adrenergic stimulation. Studies on Quin-2-loaded hepatocytes. J Biol Chem. 1983 Jul 25;258(14):8769–8773. [PubMed] [Google Scholar]
- Connor J. A., Cornwall M. C., Williams G. H. Spatially resolved cytosolic calcium response to angiotensin II and potassium in rat glomerulosa cells measured by digital imaging techniques. J Biol Chem. 1987 Feb 25;262(6):2919–2927. [PubMed] [Google Scholar]
- Connor J. A. Digital imaging of free calcium changes and of spatial gradients in growing processes in single, mammalian central nervous system cells. Proc Natl Acad Sci U S A. 1986 Aug;83(16):6179–6183. doi: 10.1073/pnas.83.16.6179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Creba J. A., Downes C. P., Hawkins P. T., Brewster G., Michell R. H., Kirk C. J. Rapid breakdown of phosphatidylinositol 4-phosphate and phosphatidylinositol 4,5-bisphosphate in rat hepatocytes stimulated by vasopressin and other Ca2+-mobilizing hormones. Biochem J. 1983 Jun 15;212(3):733–747. doi: 10.1042/bj2120733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dahl G., Isenberg G. Decoupling of heart muscle cells: correlation with increased cytoplasmic calcium activity and with changes of nexus ultrastructure. J Membr Biol. 1980 Mar 31;53(1):63–75. doi: 10.1007/BF01871173. [DOI] [PubMed] [Google Scholar]
- De Mello W. C. Effect of intracellular injection of calcium and strontium on cell communication in heart. J Physiol. 1975 Sep;250(2):231–245. doi: 10.1113/jphysiol.1975.sp011051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dunlap K., Takeda K., Brehm P. Activation of a calcium-dependent photoprotein by chemical signalling through gap junctions. Nature. 1987 Jan 1;325(6099):60–62. doi: 10.1038/325060a0. [DOI] [PubMed] [Google Scholar]
- Flagg-Newton J. L., Dahl G., Loewenstein W. R. Cell junction and cyclic AMP: 1. Upregulation of junctional membrane permeability and junctional membrane particles by administration of cyclic nucleotide or phosphodiesterase inhibitor. J Membr Biol. 1981;63(1-2):105–121. doi: 10.1007/BF01969452. [DOI] [PubMed] [Google Scholar]
- Fletcher W. H., Greenan J. R. Receptor mediated action without receptor occupancy. Endocrinology. 1985 Apr;116(4):1660–1662. doi: 10.1210/endo-116-4-1660. [DOI] [PubMed] [Google Scholar]
- Garrison J. C., Johnsen D. E., Campanile C. P. Evidence for the role of phosphorylase kinase, protein kinase C, and other Ca2+-sensitive protein kinases in the response of hepatocytes to angiotensin II and vasopressin. J Biol Chem. 1984 Mar 10;259(5):3283–3292. [PubMed] [Google Scholar]
- Graf J., Petersen O. H. Cell membrane potential and resistance in liver. J Physiol. 1978 Nov;284:105–126. doi: 10.1113/jphysiol.1978.sp012530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grynkiewicz G., Poenie M., Tsien R. Y. A new generation of Ca2+ indicators with greatly improved fluorescence properties. J Biol Chem. 1985 Mar 25;260(6):3440–3450. [PubMed] [Google Scholar]
- Hallcher L. M., Sherman W. R. The effects of lithium ion and other agents on the activity of myo-inositol-1-phosphatase from bovine brain. J Biol Chem. 1980 Nov 25;255(22):10896–10901. [PubMed] [Google Scholar]
- Hax W. M., van Venrooij G. E., Vossenberg J. B. Cell communication: a cyclic AMP mediated phenomenon. J Membr Biol. 1974;19(3):253–266. doi: 10.1007/BF01869981. [DOI] [PubMed] [Google Scholar]
- Johnson R. G., Sheridan J. D. Junctions between cancer cells in culture: ultrastructure and permeability. Science. 1971 Nov 12;174(4010):717–719. doi: 10.1126/science.174.4010.717. [DOI] [PubMed] [Google Scholar]
- Joseph S. K., Thomas A. P., Williams R. J., Irvine R. F., Williamson J. R. myo-Inositol 1,4,5-trisphosphate. A second messenger for the hormonal mobilization of intracellular Ca2+ in liver. J Biol Chem. 1984 Mar 10;259(5):3077–3081. [PubMed] [Google Scholar]
- Jungermann K., Katz N. Functional hepatocellular heterogeneity. Hepatology. 1982 May-Jun;2(3):385–395. doi: 10.1002/hep.1840020316. [DOI] [PubMed] [Google Scholar]
- Kessler J. A., Spray D. C., Saez J. C., Bennett M. V. Determination of synaptic phenotype: insulin and cAMP independently initiate development of electrotonic coupling between cultured sympathetic neurons. Proc Natl Acad Sci U S A. 1984 Oct;81(19):6235–6239. doi: 10.1073/pnas.81.19.6235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kohen E., Kohen C., Thorell B., Mintz D. H., Rabinovitch A. Intercellular communication in pancreatic islet monolayer cultures: a microfluorometric study. Science. 1979 May 25;204(4395):862–865. doi: 10.1126/science.35828. [DOI] [PubMed] [Google Scholar]
- Larson D. M., Sheridan J. D. Intercellular junctions and transfer of small molecules in primary vascular endothelial cultures. J Cell Biol. 1982 Jan;92(1):183–191. doi: 10.1083/jcb.92.1.183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lawrence T. S., Beers W. H., Gilula N. B. Transmission of hormonal stimulation by cell-to-cell communication. Nature. 1978 Apr 6;272(5653):501–506. doi: 10.1038/272501a0. [DOI] [PubMed] [Google Scholar]
- May W. S., Jr, Sahyoun N., Wolf M., Cuatrecasas P. Role of intracellular calcium mobilization in the regulation of protein kinase C-mediated membrane processes. Nature. 1985 Oct 10;317(6037):549–551. doi: 10.1038/317549a0. [DOI] [PubMed] [Google Scholar]
- Murphy E., Coll K., Rich T. L., Williamson J. R. Hormonal effects on calcium homeostasis in isolated hepatocytes. J Biol Chem. 1980 Jul 25;255(14):6600–6608. [PubMed] [Google Scholar]
- Phillips M. J., Oshio C., Miyairi M., Watanabe S., Smith C. R. What is actin doing in the liver cell? Hepatology. 1983 May-Jun;3(3):433–436. doi: 10.1002/hep.1840030325. [DOI] [PubMed] [Google Scholar]
- Piccolino M., Neyton J., Gerschenfeld H. M. Decrease of gap junction permeability induced by dopamine and cyclic adenosine 3':5'-monophosphate in horizontal cells of turtle retina. J Neurosci. 1984 Oct;4(10):2477–2488. doi: 10.1523/JNEUROSCI.04-10-02477.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pitts J. D., Simms J. W. Permeability of junctions between animal cells. Intercellular transfer of nucleotides but not of macromolecules. Exp Cell Res. 1977 Jan;104(1):153–163. doi: 10.1016/0014-4827(77)90078-7. [DOI] [PubMed] [Google Scholar]
- Politoff A., Pappas G. D., Bennett M. V. Cobalt ions cross an electrotonic synapse if cytoplasmic concentration is low. Brain Res. 1974 Aug 16;76(2):343–346. doi: 10.1016/0006-8993(74)90466-1. [DOI] [PubMed] [Google Scholar]
- Rieske E., Schubert P., Kreutzberg G. W. Transfer of radioactive material between electrically coupled neurons of the leech central nervous system. Brain Res. 1975 Feb 14;84(3):365–382. doi: 10.1016/0006-8993(75)90759-3. [DOI] [PubMed] [Google Scholar]
- Rose B., Loewenstein W. R. Permeability of cell junction depends on local cytoplasmic calcium activity. Nature. 1975 Mar 20;254(5497):250–252. doi: 10.1038/254250a0. [DOI] [PubMed] [Google Scholar]
- Saez J. C., Spray D. C., Nairn A. C., Hertzberg E., Greengard P., Bennett M. V. cAMP increases junctional conductance and stimulates phosphorylation of the 27-kDa principal gap junction polypeptide. Proc Natl Acad Sci U S A. 1986 Apr;83(8):2473–2477. doi: 10.1073/pnas.83.8.2473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sasse D., Katz N., Jungermann K. Functional heterogeneity of rat liver parenchyma and of isolated hepatocytes. FEBS Lett. 1975 Sep 1;57(1):83–88. doi: 10.1016/0014-5793(75)80157-8. [DOI] [PubMed] [Google Scholar]
- Segal S. S., Duling B. R. Flow control among microvessels coordinated by intercellular conduction. Science. 1986 Nov 14;234(4778):868–870. doi: 10.1126/science.3775368. [DOI] [PubMed] [Google Scholar]
- Simpson I., Rose B., Loewenstein W. R. Size limit of molecules permeating the junctional membrane channels. Science. 1977 Jan 21;195(4275):294–296. doi: 10.1126/science.831276. [DOI] [PubMed] [Google Scholar]
- Spray D. C., Bennett M. V. Physiology and pharmacology of gap junctions. Annu Rev Physiol. 1985;47:281–303. doi: 10.1146/annurev.ph.47.030185.001433. [DOI] [PubMed] [Google Scholar]
- Spray D. C., Ginzberg R. D., Morales E. A., Gatmaitan Z., Arias I. M. Electrophysiological properties of gap junctions between dissociated pairs of rat hepatocytes. J Cell Biol. 1986 Jul;103(1):135–144. doi: 10.1083/jcb.103.1.135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spray D. C., Stern J. H., Harris A. L., Bennett M. V. Gap junctional conductance: comparison of sensitivities to H and Ca ions. Proc Natl Acad Sci U S A. 1982 Jan;79(2):441–445. doi: 10.1073/pnas.79.2.441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stewart W. W. Lucifer dyes--highly fluorescent dyes for biological tracing. Nature. 1981 Jul 2;292(5818):17–21. doi: 10.1038/292017a0. [DOI] [PubMed] [Google Scholar]
- Subak-Sharpe H., Bürk R. R., Pitts J. D. Metabolic co-operation between biochemically marked mammalian cells in tissue culture. J Cell Sci. 1969 Mar;4(2):353–367. doi: 10.1242/jcs.4.2.353. [DOI] [PubMed] [Google Scholar]
- Thomas A. P., Alexander J., Williamson J. R. Relationship between inositol polyphosphate production and the increase of cytosolic free Ca2+ induced by vasopressin in isolated hepatocytes. J Biol Chem. 1984 May 10;259(9):5574–5584. [PubMed] [Google Scholar]
- Traub O., Look J., Paul D., Willecke K. Cyclic adenosine monophosphate stimulates biosynthesis and phosphorylation of the 26 kDa gap junction protein in cultured mouse hepatocytes. Eur J Cell Biol. 1987 Feb;43(1):48–54. [PubMed] [Google Scholar]
- Tsien R. W., Weingart R. Inotropic effect of cyclic AMP in calf ventricular muscle studied by a cut end method. J Physiol. 1976 Aug;260(1):117–141. doi: 10.1113/jphysiol.1976.sp011507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsien R. Y. A non-disruptive technique for loading calcium buffers and indicators into cells. Nature. 1981 Apr 9;290(5806):527–528. doi: 10.1038/290527a0. [DOI] [PubMed] [Google Scholar]
- Verselis V., White R. L., Spray D. C., Bennett M. V. Gap junctional conductance and permeability are linearly related. Science. 1986 Oct 24;234(4775):461–464. doi: 10.1126/science.3489990. [DOI] [PubMed] [Google Scholar]
- Volpe P., Krause K. H., Hashimoto S., Zorzato F., Pozzan T., Meldolesi J., Lew D. P. "Calciosome," a cytoplasmic organelle: the inositol 1,4,5-trisphosphate-sensitive Ca2+ store of nonmuscle cells? Proc Natl Acad Sci U S A. 1988 Feb;85(4):1091–1095. doi: 10.1073/pnas.85.4.1091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wade M. H., Trosko J. E., Schindler M. A fluorescence photobleaching assay of gap junction-mediated communication between human cells. Science. 1986 Apr 25;232(4749):525–528. doi: 10.1126/science.3961495. [DOI] [PubMed] [Google Scholar]
- Watanabe S., Phillips M. J. Ca2+ causes active contraction of bile canaliculi: direct evidence from microinjection studies. Proc Natl Acad Sci U S A. 1984 Oct;81(19):6164–6168. doi: 10.1073/pnas.81.19.6164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williamson J. R., Cooper R. H., Joseph S. K., Thomas A. P. Inositol trisphosphate and diacylglycerol as intracellular second messengers in liver. Am J Physiol. 1985 Mar;248(3 Pt 1):C203–C216. doi: 10.1152/ajpcell.1985.248.3.C203. [DOI] [PubMed] [Google Scholar]
- Woods N. M., Cuthbertson K. S., Cobbold P. H. Repetitive transient rises in cytoplasmic free calcium in hormone-stimulated hepatocytes. Nature. 1986 Feb 13;319(6054):600–602. doi: 10.1038/319600a0. [DOI] [PubMed] [Google Scholar]
- Zimmerman A. L., Rose B. Permeability properties of cell-to-cell channels: kinetics of fluorescent tracer diffusion through a cell junction. J Membr Biol. 1985;84(3):269–283. doi: 10.1007/BF01871390. [DOI] [PubMed] [Google Scholar]