Abstract
Rationale: Exhaled nitric oxide (FeNO) is a biomarker of airway inflammation in mild to moderate asthma. However, whether FeNO levels are informative regarding airway inflammation in patients with severe asthma, who are refractory to conventional treatment, is unknown. Here, we hypothesized that classification of severe asthma based on airway inflammation as defined by FeNO levels would identify a more reactive, at-risk asthma phenotype.
Methods: FeNO and major features of asthma, including airway inflammation, airflow limitation, hyperinflation, hyperresponsiveness, and atopy, were determined in 446 individuals with various degrees of asthma severity (175 severe, 271 nonsevere) and 49 healthy subjects enrolled in the Severe Asthma Research Program.
Measurements and Main Results: FeNO levels were similar among patients with severe and nonsevere asthma. The proportion of individuals with high FeNO levels (>35 ppb) was the same (40%) among groups despite greater corticosteroid therapy in severe asthma. All patients with asthma and high FeNO had more airway reactivity (maximal reversal in response to bronchodilator administration and by methacholine challenge), more evidence of allergic airway inflammation (sputum eosinophils), more evidence of atopy (positive skin tests, higher serum IgE and blood eosinophils), and more hyperinflation, but decreased awareness of their symptoms. High FeNO identified those patients with severe asthma characterized by the greatest airflow obstruction and hyperinflation and most frequent use of emergency care.
Conclusions: Grouping of asthma by FeNO provides an independent classification of asthma severity, and among patients with severe asthma identifies the most reactive and worrisome asthma phenotype.
Keywords: nitric oxide, severe asthma, phenotype, airway reactivity, exhaled breath
AT A GLANCE COMMENTARY.
Scientific Knowledge on the Subject
Exhaled nitric oxide (FeNO) is a biomarker of airway inflammation in mild to moderate asthma. However, whether FeNO levels are informative regarding airway inflammation in patients with severe asthma, who are refractory to conventional treatment, is unknown.
What This Study Adds to the Field
Here, we demonstrate that grouping of asthma by FeNO provides an independent classification of asthma severity, and among patients with severe asthma identifies the most reactive and worrisome asthma phenotype.
Despite progress that has been made in the understanding and treatment of mild and moderate asthma, severe asthma is poorly understood, refractory to established treatments, and accounts for a high proportion of the adverse financial impact, morbidity, and mortality of asthma in the United States (1–4). The underlying reasons why certain individuals with asthma have severe, refractory disease are poorly defined. Although sputum eosinophils have been shown to predict acute exacerbations in asthma (5, 6), sputum induction is not easy to do or widely available. Thus, there is a need for a noninvasive, easy-to-perform test to monitor patients with severe asthma and predict acute and often life-threatening asthma exacerbations, and thus allow for determination of whether or not therapy is adequate (1–4). As a free radical that reacts with oxidants and antioxidants, nitric oxide (NO) in exhaled breath (FeNO) reflects the redox state of the airway and has been proposed as a marker of airway inflammation and guide for antiinflammatory therapy in asthma (7). High levels of FeNO are well documented in nonsevere asthma (8–21) and decrease in response to treatment with corticosteroids (22–27). However, measures of FeNO in 50 patients with severe asthma in the European multicenter study of chronic severe asthma suggest that FeNO levels of patients with severe asthma, who are refractory to conventional treatments, may not be suppressed by corticosteroids (28). Although the mean FeNO levels of patients with severe asthma were similar to those of patients with nonsevere asthma, 22 (44%) of the subjects with severe asthma who were receiving high-dose oral corticosteroids had threefold higher FeNO than those receiving inhaled corticosteroids, which suggested that a substantial subpopulation of patients with severe asthma had persistent airway inflammation and possible relative corticosteroid resistance.
In this study, we hypothesized that classification of severe asthma based on airway inflammation as defined by FeNO levels would identify a more severe asthma phenotype. The present study was designed to assess alterations of FeNO in patients with severe asthma as compared with patients with nonsevere asthma and healthy control subjects, and the relationship between FeNO and asthma severity, airflow limitation, hyperinflation, hyperresponsiveness, and atopy. Although the average FeNO levels in severe and nonsevere asthma were previously reported to be similar (29), when asthma was classified on the basis of FeNO levels, a distinct asthma phenotype emerged. In general, patients with asthma and high FeNO levels tended to be younger and diagnosed with asthma at a younger age. They were more likely to be atopic and to have evidence of airway inflammation. Furthermore, patients with severe asthma and high FeNO levels had the greatest airway reactivity, the most hyperinflation, and the least awareness of their asthma symptoms. The findings provide evidence that FeNO levels are informative for classification of severe asthma phenotypes and allow identification of a particularly worrisome subgroup of patients with severe asthma. Some of the results of these studies have been previously reported in the form of an abstract (30).
METHODS
Detailed methods and statistical analyses are provided in the online supplement. A brief description is provided here.
Subject Enrollment and Characterization
All subjects were recruited by centers participating in the Severe Asthma Research Program (SARP) and gave written informed consent by signing a consent document approved by the institutional review board at the enrolling center and the SARP Data Safety and Monitoring Board (DSMB). All subjects were screened by history, physical examination, spirometry (before and after two puffs of inhaled albuterol), methacholine provocation, and allergy prick skin testing to a standard panel of aeroallergens. Subjects were nonsmokers, and classified as healthy control subjects if they were free of respiratory symptoms, had normal baseline spirometry, a negative methacholine challenge test, and nitric oxide level less than 50 ppb. Asthma was defined according to the National Asthma Education and Prevention Program guidelines, which include episodic respiratory symptoms, reversible airflow obstruction (documentation of variability of FEV1 and/or FVC by 12% and 200 cm3 either spontaneously or after two puffs of inhaled albuterol), and/or a positive methacholine challenge test (4). Severe asthma was based on the definition used by the proceedings of the American Thoracic Society Workshop on Refractory Asthma (2).
Lung Function
Spirometry was performed with an automated spirometer, consistent with American Thoracic Society (ATS) standards (31). Plethysmographic lung volumes, including total lung capacity (TLC) and residual volume (RV), were measured in 62 subjects with severe asthma and 53 subjects with nonsevere asthma, using methods conforming to ATS guidelines (32), and recorded as the percentage of predicted values obtained with the equations of Stocks and Quanjer (33), with adjustments for African Americans per ATS recommendations (34).
Atopy
Allergy skin testing was done once on each subject during the study. Skin prick testing to 14 common allergens was performed at all SARP sites with the Multi-Test II (Lincoln Diagnostics, Inc., Decatur, IL). Blood was collected for measurement of total serum IgE and a complete blood count.
Exhaled NO (FeNO)
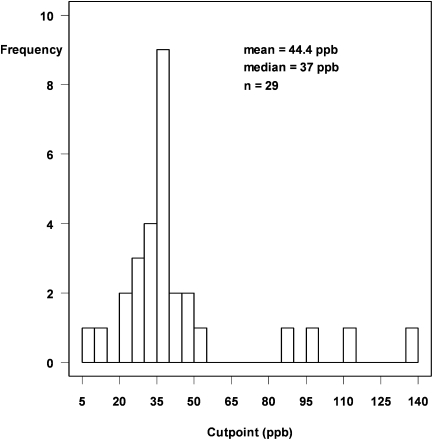
All SARP centers performed online and/or offline NO measurements according to the standards published by the ATS (35). Online FeNO values were used in all data analyses in this article. NO levels were measured online by chemiluminescence at a constant expiratory flow (50 ml/s) in all participating centers. The analyzers were calibrated in accordance with the manufacturer's instructions. Because spirometry can affect the FeNO levels, exhaled gases were collected before spirometry, if completed on the same day. On the basis of data suggesting poor asthma control when FeNO is more than 35 ppb (7), we evaluated clinical characteristics of asthma populations in subgroups of high (>35 ppb) and low (<35 ppb) NO. The rationale for selecting 35 ppb as a cutoff point for high and low NO was based on the published literature (7) and analysis of the data collected in this study. In addition to the published literature, Figure 1 provides the rationale for selecting 35 ppb as a cutoff point for high and low FeNO that is the basis for all data analyses in this study. Relevant variables (as outlined in Table E1 in the online supplement) in the database were analyzed on the basis of receiver operator characteristic (ROC) curves with FeNO as a continuous variable. The cutoff point for each variable was determined on the basis of these ROC curves. Figure 1 represents the frequency distribution of all these cutoff points. The median of all cutoff points for the variables (both categorical and continuous) that showed a significant relationship with FeNO was 37 ppb. This provided support for the validity of our selection of 35 ppb as the cutoff point between high and low FeNO.
Figure 1.
Relevant variables (as outlined in Table E1 in the online supplement) in the database were analyzed on the basis of receiver operator characteristic (ROC) curves with FeNO as a continuous variable. The cutoff point for each variable was determined on the basis of these ROC curves. Shown here is the frequency distribution of all these cutoff points. The median of all cutoff points for the variables (both categorical and continuous) that showed a significant relationship with FeNO was 37 ppb. This provided support for the validity of our selection of 35 ppb as the cutoff point between high and low FeNO.
Total NO Reaction Products
NO reaction products (NOx) in serum samples were measured by an amperometric NO sensor in combination with acidified iodide for the detection of NO derived from total nitrite and nitrate after cadmium/copper-mediated reduction of nitrate to nitrite (ISO-NOP, Nitralyzer II; World Precision Instruments, Sarasota, FL) (36).
Statistical Analyses
Categorical data were summarized as frequencies, and statistical comparisons for categorical variables were performed using Fisher's exact test. Subgroup comparisons within NO level or asthma severity were performed using appropriate contrasts from a logistic regression model including NO level, asthma severity, and their interaction as independent variables. Continuous variables were summarized using the sample size, mean, and standard deviation, and alternatively using the median and interquartile range for variables with skewed distributions. Associations between NO levels and other variables were assessed by linear regression for FeNO as a continuous variable and multiple logistic regression for FeNO (high or low) as categorical variables. Multiple logistic regression modeling is described in more detail in Results. All tests and model fitting were performed with the JMP statistical program, version 5.0 (SAS Institute Inc., Cary, NC) and R version 2.4.1 (www.R-project.org) (37). Models for FeNO as a continuous outcome in a linear regression model and as a dichotomous outcome classified as high or low in a logistic regression model were created. For multivariate analyses and modeling, parsimonious selection of independent variables was performed to avoid confounding that would render the estimated associations with the outcome as noninterpretable or misleading. Similarly, a logistic regression model for which the FeNO outcome would be classified as high or low had to be parsimonious to be mathematically stable.
RESULTS
Characterization of Study Population
Online FeNO levels were measured in 495 individuals enrolled in the Severe Asthma Research Program (SARP). Baseline characteristics are shown in Table 1. On average, healthy control subjects and patients with nonsevere asthma were younger than patients with severe asthma (P < 0.05) (Table 1). As expected, lung functions were lower in patients with severe asthma than in patients with nonsevere asthma or healthy control subjects (Table 1). The detailed clinical description of individuals in the SARP data set was previously published (29). The SARP population included in the current study does not overlap with the SARP subpopulation of children with offline NO values published previously (38).
TABLE 1.
DEMOGRAPHICS, PULMONARY FUNCTION, EXHALED NITRIC OXIDE LEVELS, AND CORTICOSTEROIDS USAGE FOR ALL SUBJECTS
| Control Subjects (n = 49) | n | Nonsevere Asthma (n = 271) | n | Severe Asthma (n = 175) | n | |
|---|---|---|---|---|---|---|
| Mean age, yr* | 32 ± 11 | 49 | 34 ± 12 | 271 | 41 ± 13 | 175 |
| Baseline %FEV1* | 101 ± 15 | 49 | 83 ± 16 | 271 | 58 ± 20 | 175 |
| Maximal %FEV1* | 108 ± 15 | 37 | 93 ± 15 | 256 | 76 ± 20 | 168 |
| % FVC* | 103 ± 11 | 49 | 94 ± 14 | 271 | 80 ± 19 | 176 |
| % FEV1/FVC*, ppb | 97 ± 7 | 49 | 88 ± 12 | 271 | 77 ± 14 | 175 |
| FeNO, ppb | 17 ± 9 | 49 | 43 ± 42 | 271 | 42 ± 41 | 175 |
| Median, IQR* | 14 (11–19) | 49 | 27 (17–55) | 271 | 27 (17–52) | 175 |
| Sex (male), no. | 13 | 49 | 86 | 271 | 65 | 176 |
| Race, C/AA/AI/A/NH/O/U/R/MR | 40/5/0/2/0/0/0/0/2 | 49 | 172/80/0/4/0/8/1/0/6 | 271 | 116/44/0/7/0/2/0/0/6 | 175 |
| Corticosteroids | ||||||
| Inhaled, %* | 0 | 49 | 64% | 271 | 100% | 175 |
| Oral, %* | 0 | 49 | 3% | 271 | 44% | 175 |
| Injected, % | 0 | 49 | 0% | 271 | 3% | 175 |
| Serum IgE levels* | 58 ± 87 | 45 | 267 ± 380 | 239 | 318 ± 730 | 147 |
| Median, IQR* | 32 (10–60) | 45 | 145 (60–330) | 239 | 124 (40–320) | 147 |
| BAL eosinophils, %* | 0.2 ± 0.7 | 21 | 1.1 ± 0.4 | 73 | 1.9 ± 0.5 | 49 |
| Median, IQR* | 0 (0–0.4) | 21 | 0.3 (0–1.2) | 73 | 0.5 (0–1.5) | 49 |
| Blood eosinophils, %* | 2.3 ± 1 | 45 | 4.1 ± 3 | 252 | 4.1 ± 5 | 168 |
| Median, IQR* | 2 (1–2.85) | 45 | 3.7 (2–5) | 252 | 3 (1.5–5) | 168 |
Definition of abbreviations: A = Asian; AA =African American; AI = American Indian or Alaska native; BAL = bronchoalveolar lavage; C = Caucasian; FeNO = exhaled nitric oxide level; IQR = interquartile range; MR = multiple races; n = number of individuals with available data; NH = Native Hawaiian; O = other; R = refused; U = uncertain.
Fisher's, analysis of variance, or Kruskal-Wallis P < 0.05 among three groups.
NO in Asthma
NO levels were higher in patients with asthma as compared with control subjects, but there was no significant difference in average FeNO between severe and nonsevere asthma (FeNO [ppb]: control, 17 ± 9; nonsevere, 43 ± 42; severe, 42 ± 41; P = 0.01) (Table 1). The proportion of individuals with high FeNO was the same in severe and nonsevere asthma (nonsevere, 109/271 [40%]; severe, 70/175 [40%]).
The High-NO Phenotype in Asthma
There were equal proportions of patients with severe and nonsevere asthma in the low- and high-FeNO groups. In general, patients with asthma and high FeNO demonstrated several distinct characteristics when compared with patients with asthma and low FeNO. Demographically, patients with asthma and high FeNO were younger (age, yr [mean ± SD]: low FeNO, 38 ± 12; high FeNO, 36 ± 13; P = 0.03) and diagnosed with asthma at a younger age (age, yr [mean ± SD]: low FeNO, 16 ± 13; high FeNO, 14 ± 14; P = 0.05) and less likely to be female (female [% of population]: low FeNO, 70%; high FeNO, 60%; P = 0.02).
On pulmonary function testing, the high- and low-FeNO groups had similar baseline FEV1 and FVC, but the FEV1/FVC ratio (% predicted) was lower in high FeNO, indicating increased airflow limitation in this group. The high-FeNO group also had more airway reactivity as shown by greater FEV1 reversibility after maximal bronchodilation and lower PC20 (provocative concentration of methacholine causing a 20% fall in FEV1). They had more hyperinflation with a higher total lung capacity (TLC), a higher residual lung volume (RV), and a higher RV/TLC ratio (Table 2).
TABLE 2.
PULMONARY FUNCTION BY EXHALED NITRIC OXIDE
| Characteristic | Low FeNO (≤35 ppb) | n | High FeNO (>35 ppb) | n | P Value |
|---|---|---|---|---|---|
| Baseline FVC, % predicted | 85 ± 18 | 267 | 87 ± 19 | 179 | 0.20 |
| Maximal FVC, % of predicted | 93 ± 16 | 253 | 100 ± 15 | 170 | <0.001 |
| Baseline FEV1, % predicted | 74 ± 20 | 267 | 73 ± 23 | 179 | 0.80 |
| Maximal FEV1, % of predicted | 85 ± 20 | 253 | 90 ± 18 | 170 | 0.005 |
| FEV1/FVC ratio, % of predicted | 86 ± 14 | 267 | 81 ± 14 | 179 | <0.001 |
| Maximal FEV1 reversal, % | 14 ± 16 | 253 | 20 ± 17 | 170 | |
| Median (IQR)* | 10 (5–18) | 253 | 16 (8–26) | 170 | <0.001 |
| PC20 | 4.3 ± 6 | 203 | 1.7 ± 3 | 123 | |
| Median (IQR)* | 1.8 (0.5–4.9) | 203 | 0.7 (0.3–1.6) | 123 | <0.001 |
| TLC, % predicted | 106 ± 12 | 88 | 115 ± 14 | 43 | 0.002 |
| FRC, % predicted | 101 ± 24 | 84 | 119 ± 30 | 40 | <0.001 |
| FRC/TLC, % predicted | 95 ± 18 | 84 | 103 ± 16 | 40 | 0.008 |
| RV, % predicted | 124 ± 42 | 88 | 153 ± 57 | 43 | 0.003 |
| RV/TLC, % predicted | 111 ± 30 | 88 | 126 ± 40 | 42 | 0.03 |
Definition of abbreviations: IQR = interquartile range; PC20 = provocative concentration of methacholine causing a 20% fall in FEV1; RV = reserve volume; TLC = total lung capacity.
Wilcoxon rank sum P values reported rather than t test.
High-FeNO patients with asthma, whether severe or nonsevere, were more likely to be atopic as shown by more positive skin tests (number of positive skin tests [mean ± SD]: low FeNO, 3.4 ± 3; high FeNO, 4.2 ± 3; P = 0.004), higher serum IgE level (serum IgE [mean ± SD]: low FeNO, 219 ± 366; high FeNO, 340 ± 402; P = 0.0001), and higher blood eosinophils (% blood eosinophils [mean ± SD]: low FeNO, 3.4 ± 3.7; high FeNO, 5.1 ± 3.9; P = 0.0001). They also had more eosinophils in sputum (% sputum eosinophils [mean ± SD]: low FeNO, 3 ± 7; high FeNO, 13 ± 23; P = 0.0001), suggesting more evidence of allergic airway inflammation. Interestingly, patients with asthma and high FeNO levels were less likely to have seen a physician in the last 12 months (%: low FeNO, 72%; high FeNO, 63%; P = 0.04), but more likely to have been in the emergency room (%: low FeNO, 66%; high FeNO, 73%; P = 0.05) over the same time period, or admitted to the intensive care unit in the past (%: low FeNO, 16%; high FeNO, 25%; P = 0.02).
Patients with asthma and low NO levels were more likely to be overweight (body mass index, kg/m2 [mean ± SD]: low FeNO, 31 ± 9; high FeNO, 28 ± 8; P = 0.002), have systemic hypertension (%: low FeNO, 16%; high FeNO, 8%; P = 0.05), and be on treatment for diabetes (%: low FeNO, 40%; high FeNO, 11%; P = 0.01).
Characterizing the High-FeNO Phenotype in Severe Asthma
In patients with severe asthma, high FeNO levels identified a phenotype that appeared to be the most severe of all groups, including low-FeNO severe asthma, and high- or low-FeNO nonsevere asthma groups. Individuals with severe asthma and high FeNO levels tended to share several characteristics. They had the greatest airway reactivity of any group defined as the magnitude of FEV1 reversal after maximal bronchodilation and by PC20. They had the greatest degree of airflow limitation and the most hyperinflation (Table 3). They also had high numbers of eosinophils in the sputum (Table 4). Emergency room use and intensive care unit admissions were greatest in this group (Table 3). In contrast to FeNO, NO metabolites (NOx) in serum were higher in all patients with severe asthma as a group in comparison with patients with nonsevere asthma (NOx, μM: nonsevere, 36 ± 23; severe, 42 ± 24; P = 0.0009) and were unrelated to FeNO levels (R = 0.002; P = 0.5). Serum NOx was not related to clinical characteristics such as lung function or atopy (all P > 0.2).
TABLE 3.
PULMONARY FUNCTION AND EXHALED NITRIC OXIDE LEVEL BY SEVERITY
| Characteristic | Severe Asthma, Low FeNO | n | Nonsevere Asthma, Low FeNO | n | Severe Asthma, High FeNO | n | Nonsevere Asthma, High FeNO | n | Low FeNO: Severe vs. Nonsevere Asthma | High FeNO: Severe vs. Nonsevere Asthma | Severe Asthma: Low vs. High FeNO | Nonsevere Asthma: Low vs. High FeNO |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Baseline FVC, % predicted | 75 ± 18 | 105 | 92 ± 15 | 162 | 75 ± 21 | 70 | 95 ± 14 | 109 | <0.001 | <0.001 | 0.97 | 0.13 |
| Maximal FVC, % predicted | 88 ± 17 | 101 | 97 ± 14 | 153 | 95 ± 18 | 67 | 103 ± 12 | 103 | <0.001 | <0.001 | 0.004 | <0.001 |
| Baseline FEV1, % predicted | 60 ± 19 | 105 | 83 ± 16 | 162 | 56 ± 22 | 70 | 83 ± 17 | 109 | <0.001 | <0.001 | 0.24 | 0.76 |
| Maximal FEV1, % predicted | 74 ± 20 | 101 | 91 ± 15 | 153 | 80 ± 19 | 67 | 96 ± 15 | 103 | <0.001 | <0.001 | 0.009 | 0.04 |
| FEV1/FVC ratio, % predicted | 79 ± 15 | 105 | 90 ± 11 | 162 | 74 ± 14 | 70 | 86 ± 12 | 109 | <0.001 | <0.001 | 0.011 | 0.03 |
| Maximal FEV1 reversal, % | 18 ± 23 | 101 | 11 ± 9 | 153 | 23 ± 19 | 67 | 17 ± 15 | 103 | ||||
| Median (IQR)* | 14 (6–22) | 101 | 8 (5–14) | 153 | 21 (9–29) | 67 | 13 (7–22) | 103 | 0.002 | 0.01 | 0.005 | <0.001 |
| PC20 | 3.9 ± 6 | 53 | 4.4 ± 6 | 149 | 1.5 ± 3 | 27 | 1.7 ± 3 | 96 | ||||
| Median (IQR)* | 1 (0.2–4.5) | 53 | 2 (0.6–5) | 149 | 0.6 (0.2–1.7) | 27 | 0.7 (0.3–1.6) | 96 | 0.10 | 0.40 | 0.01 | <0.001 |
| TLC, % predicted | 107 ± 13 | 41 | 104 ± 12 | 31 | 117 ± 17 | 21 | 112 ± 10 | 22 | 0.55 | 0.28 | 0.006 | 0.05 |
| FRC, % predicted | 103 ± 27 | 38 | 96 ± 21 | 31 | 124 ± 34 | 18 | 115 ± 26 | 22 | 0.40 | 0.36 | 0.005 | 0.01 |
| FRC/TLC, % predicted | 96 ± 21 | 38 | 92 ± 14 | 31 | 104 ± 16 | 18 | 102 ± 17 | 22 | 0.43 | 0.84 | 0.10 | 0.03 |
| RV, % predicted | 143 ± 44 | 41 | 109 ± 34 | 31 | 176 ± 58 | 21 | 131 ± 47 | 22 | 0.004 | 0.001 | 0.005 | 0.11 |
| RV/TLC, % predicted | 128 ± 30 | 41 | 99 ± 21 | 31 | 141 ± 41 | 20 | 111 ± 33 | 22 | <0.001 | 0.002 | 0.12 | 0.21 |
| ER in past 12 mo | 38% | 105 | 12% | 161 | 53% | 70 | 18% | 109 | <0.001 | <0.001 | 0.05 | 0.14 |
| Ever had an ICU admission due to asthma | 34% | 105 | 5% | 162 | 44% | 70 | 12% | 108 | <0.001 | <0.001 | 0.26 | 0.04 |
| BMI | 32 ± 8 | 103 | 30 ± 9 | 162 | 30 ± 8 | 66 | 28 ± 7 | 109 | 0.08 | 0.13 | 0.08 | 0.016 |
Definition of abbreviations: BMI = body mass index; ER = emergency room; ICU = intensive care unit; IQR = interquartile range; RV = reserve volume; TLC = total lung capacity.
High FeNO defined as >35 ppb, and low FeNO as ≤35 ppb.
Wilcoxon rank sum P values reported rather than those based on contrasts from analysis of variance.
TABLE 4.
INFLAMMATORY CELLS IN BLOOD, BRONCHOALVEOLAR LAVAGE, AND SPUTUM BY SEVERITY AND EXHALED NITRIC OXIDE LEVELS
| Characteristic | Severe Asthma, Low FeNO | n | Nonsevere Asthma, Low FeNO | n | Severe Asthma, High FeNO | n | Nonsevere Asthma, High FeNO | n | Low FeNO: Severe vs. Nonsevere Asthma | High FeNO: Severe vs. Nonsevere Asthma | Severe Asthma: Low vs. High FeNO | Nonsevere Asthma: Low vs. High FeNO |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Inflammatory cells in blood | ||||||||||||
| Total WBC | 7.8 ± 3 | 103 | 6.8 ± 2 | 153 | 8 ± 2 | 64 | 6.5 ± 2 | 104 | 0.001 | <0.001 | 0.76 | 0.19 |
| Monocytes, % | 5.7 ± 2 | 103 | 6.4 ± 2 | 152 | 6.0 ± 3 | 63 | 6.8 ± 2 | 104 | 0.23 | 0.83 | 0.06 | 0.16 |
| Neutrophils, % | 62 ± 12 | 103 | 58 ± 10 | 152 | 63 ± 15 | 64 | 55 ± 10 | 104 | 0.27 | 0.46 | 0.81 | 0.63 |
| Lymphocytes, % | 28 ± 11 | 103 | 32 ± 9 | 152 | 25 ± 10 | 64 | 33 ± 9 | 104 | 0.16 | <0.001 | 0.27 | 0.11 |
| Eosinophils, % | 3.8 ± 5.2 | 103 | 3.1 ± 2.1 | 152 | 4.7 ± 3.9 | 64 | 5.3 ± 3.9 | 104 | 0.20 | 0.27 | 0.13 | <0.001 |
| Basophils, % | 0.4 ± 0.5 | 103 | 0.6 ± 0.5 | 144 | 0.5 ± 0.5 | 64 | 0.5 ± 0.6 | 104 | 0.005 | 0.36 | 0.34 | 0.60 |
| Inflammatory cells in BAL | ||||||||||||
| BAL total cells | 8 ± 6 | 27 | 9 ± 8 | 48 | 4.8 ± 3 | 22 | 8.9 ± 6 | 27 | 0.55 | 0.02 | 0.09 | 0.95 |
| BAL macrophages, % | 91 ± 10 | 27 | 90 ± 16 | 47 | 82 ± 24 | 22 | 81 ± 26 | 26 | 0.74 | 0.85 | 0.10 | 0.07 |
| BAL neutrophils, % | 3.0 ± 4 | 27 | 2.1 ± 4 | 47 | 2.6 ± 4 | 22 | 3.2 ± 7 | 26 | 0.42 | 0.62 | 0.72 | 0.34 |
| BAL lymphocytes, % | 3.8 ± 3 | 27 | 5.3 ± 6 | 47 | 8.9 ± 13 | 22 | 6.4 ± 6 | 26 | 0.40 | 0.23 | 0.02 | 0.56 |
| BAL eosinophils, % | 1.9 ± 6 | 27 | 0.8 ± 2.2 | 47 | 1.8 ± 3 | 22 | 1.5 ± 2.4 | 26 | 0.18 | 0.77 | 0.93 | 0.39 |
| Inflammatory cells in sputum | ||||||||||||
| Total cells, millions | 4.1 ± 7 | 61 | 3.0 ± 4 | 117 | 3.3 ± 5 | 34 | 2.4 ± 3 | 80 | 0.24 | 0.98 | 0.52 | 0.76 |
| Total WBCs, millions | 3.1 ± 6 | 61 | 2.1 ± 4 | 117 | 2.5 ± 5 | 34 | 2.4 ± 9 | 79 | 0.27 | 0.92 | 0.61 | 0.76 |
| Viability of WBCs, % | 61 ± 24 | 62 | 63 ± 21 | 117 | 58 ± 23 | 34 | 62 ± 22 | 79 | 0.48 | 0.30 | 0.53 | 0.83 |
| Bronchial epithelial cells, % | 4.8 ± 6 | 62 | 2.9 ± 5 | 117 | 2.4 ± 3 | 34 | 3.7 ± 4 | 79 | 0.02 | 0.21 | 0.03 | 0.30 |
| Sputum macrophages, % | 52 ± 25 | 56 | 56 ± 26 | 99 | 39 ± 32 | 28 | 63 ± 26 | 66 | 0.38 | <0.001 | 0.05 | 0.11 |
| Sputum lymphocytes, % | 4.2 ± 5 | 56 | 2.5 ± 3 | 99 | 3.1 ± 6 | 28 | 2.7 ± 2 | 66 | 0.008 | 0.64 | 0.20 | 0.82 |
| Sputum neutrophils, % | 40 ± 25 | 56 | 39 ± 26 | 99 | 32 ± 30 | 28 | 27 ± 23 | 66 | 0.79 | 0.36 | 0.23 | 0.006 |
| Sputum eosinophils, % | 3 ± 5 | 56 | 2.2 ± 4 | 99 | 25 ± 33 | 28 | 7 ± 13 | 66 | 0.38 | <0.001 | <0.001 | 0.02 |
Definition of abbreviation: BAL = bronchoalveolar lavage; WBCs = white blood cells.
Characterizing the High-FeNO Phenotype in Nonsevere Asthma
In patients with nonsevere asthma, high FeNO similarly identified a more severe subgroup. In fact, the patients with nonsevere asthma and high FeNO shared more similarities with patients with severe asthma and high FeNO than with patients with nonsevere asthma and low FeNO. For instance, the nonsevere group with high FeNO had more airway reactivity defined by the magnitude of FEV1 reversal after maximal bronchodilation and by PC20, and significantly more airflow limitation and hyperinflation than patients with nonsevere asthma and low FeNO levels. They also had more eosinophilic inflammation (Table 4) and more intensive care unit (although not emergency room) admissions. These individuals were the thinnest among all groups (Table 3).
FeNO and Lung Volumes
TLC increased linearly with increased air trapping as measured by elevated ratio of RV to TLC. In addition, there was an independent additive increase in TLC in subjects with higher FeNO (P = 0.0005 for FeNO effect, P < 0.0001 for RV/TLC effect; analysis of covariance). There was no effect of the designated severe or nonsevere asthma grouping (P > 0.9) on TLC independent of air trapping and FeNO effects within each of the severity groups. This indicates that air trapping and FeNO are independent determinants for lung hyperinflation in asthma.
FeNO and Use of Corticosteroids and Other Medications
The greater reactivity in the high-FeNO asthma subgroups suggested that these patients had greater airway inflammation and/or less antiinflammatory therapy. All patients with severe asthma in this study were by definition receiving some form of corticosteroids (2). There was no difference in the use of inhaled corticosteroid or leukotriene modifiers among patients with asthma and high or low FeNO, but more patients in the high-FeNO group were taking oral corticosteroids (Table 5). Individuals with high FeNO were more likely to be taking theophylline (Table 5). When corticosteroid use was further analyzed by asthma severity in addition to FeNO levels, again there was no significant difference in inhaled corticosteroid use between the high- and low-FeNO groups regardless of severity. The group with high FeNO and severe asthma had the highest proportion of oral corticosteroid use (percent oral corticosteroid use: severe asthma–low FeNO, 37%; nonsevere asthma–low FeNO, 1%; severe asthma–high FeNO, 56%; nonsevere asthma–high FeNO, 5%; P = 0.01). Whereas only a small number of individuals were taking theophylline, patients with severe asthma and high FeNO levels were much more likely to be taking daily theophylline than members of any of the other groups (percent theophylline use: severe asthma–low FeNO, 13%; nonsevere asthma–low FeNO, 1%; severe asthma–high FeNO, 29%; nonsevere asthma–high FeNO, 3%; P = 0.01). Thus, the finding of high FeNO in the severe and nonsevere asthma subgroups was likely not due to less corticosteroid therapy than in the low-FeNO subgroups. Multivariate analyses and modeling for determinants of FeNO did not indicate an influence of corticosteroid use on FeNO levels (Table 6).
TABLE 5.
MEDICATION USE BY EXHALED NITRIC OXIDE LEVELS
| Characteristic | Low FeNO (≤35 ppb) | n | High FeNO (>35 ppb) | n | P Value* |
|---|---|---|---|---|---|
| Inhaled corticosteroids | 28% | 267 | 28% | 179 | 0.9 |
| Oral corticosteroids | 15% | 267 | 25% | 179 | 0.01 |
| Injectable corticosteroids | 2.2% | 267 | 3.9% | 179 | 0.3 |
| Inhaled corticosteroids and β-agonist | 57% | 267 | 50% | 179 | 0.1 |
| Total β-agonists | 91% | 267 | 91% | 179 | 0.9 |
| Total long-acting β-agonist | 65% | 267 | 59% | 179 | 0.2 |
| Total inhaled corticosteroids | 73% | 267 | 70% | 179 | 0.5 |
| Total other corticosteroids | 16% | 267 | 25% | 179 | 0.02 |
| Leukotriene modifiers | 29% | 267 | 31% | 179 | 0.6 |
| Theophylline | 6% | 267 | 13% | 179 | 0.01 |
Fisher's exact test P values.
TABLE 6.
RESULTS OF MULTIVARIATE LOGISTIC REGRESSION ANALYSIS WITH HIGH EXHALED NITRIC OXIDE (>35 ppb) AS THE OUTCOME
| All Asthma (n = 335) |
Nonsevere Asthma (n = 210) |
Severe Asthma (n = 125) |
||||
|---|---|---|---|---|---|---|
| Characteristic | Odds Ratio (95% CI) | P Value | Odds Ratio (95% CI) | P Value | Odds Ratio (95% CI) | P Value |
| Sex | 0.99 (0.55–1.61) | 0.96 | 1.04 (0.50–2.05) | 0.90 | 1.11 (0.40–2.70) | 0.82 |
| Age | 0.99 (0.97–1.01) | 0.57 | 0.97 (0.95–1.00) | 0.058 | 1.02 (0.99–1.05) | 0.22 |
| BMI | 0.99 (0.95–1.02) | 0.40 | 1.02 (0.98–1.07) | 0.36 | 0.94 (0.89–1.00) | 0.06 |
| Activity score | 1.42 (1.10–1.80) | 0.005 | 1.47 (1.08–2.03) | 0.02 | 1.52 (0.89–2.31) | 0.08 |
| Wheezing | 1.14 (0.92–1.38) | 0.17 | 1.17 (0.89–1.50) | 0.24 | 1.19 (0.80–1.68) | 0.33 |
| Log maximal FEV1 reversal | 1.19 (1.01–1.41) | 0.03 | 1.22 (0.99–1.51) | 0.06 | 1.29 (0.91–1.73) | 0.1 |
| Log IgE | 1.88 (1.19–3.01) | 0.007 | 2.71 (1.38–5.36) | 0.004 | 1.52 (0.74–3.20) | 0.25 |
| Log percent eosinophils in the blood | 1.35 (1.11–1.62) | 0.002 | 1.15 (1.14–1.93) | 0.003 | 1.31 (0.97–1.78) | 0.07 |
| Total ICS | 0.74 (0.40–1.39) | 0.34 | 0.68 (0.34–1.40) | 0.30 | 3.14 (0.07–95.54) | 0.53 |
| Total other CS | 1.34 (0.66–3.20) | 0.41 | 2.05 (0.18–22.45) | 0.55 | 1.76 (0.68–5.15) | 0.23 |
| Theophylline use | 2.92 (1.23–7.54) | 0.02 | 9.0 (0.71–102.62) | 0.08 | 2.9 (1.13–9.30) | 0.04 |
| Seen a doctor in the last 12 mo | 0.58 (0.31–1.09) | 0.09 | 0.75 (0.35–1.61) | 0.46 | 0.5 (0.12–2.89) | 0.38 |
| Visited ER in the last 12 mo | 2.33 (1.20–4.69) | 0.01 | 2.65 (0.87–7.90) | 0.09 | 2.5 (1.08–7.69) | 0.05 |
Definition of abbreviations: BMI = body mass index; CI = confidence interval; CS = corticosteroids; ER = emergency room; ICS = inhaled corticosteroids.
n = number of individuals who had a complete set of all variables to run the model.
DISCUSSION
This study provides evidence that subclassification by FeNO defines asthma phenotypes independent of current definitions for asthma severity. Patients with asthma who have high FeNO levels share several characteristics regardless of their asthma severity as it is currently defined. Patients with asthma and high FeNO are younger and diagnosed with asthma at a younger age. They are atopic and have more eosinophilic airway inflammation, more airway reactivity, more airflow limitation, and more hyperinflation. The fact that patients with high FeNO were more likely to have gone to an emergency room or admitted to an ICU over the past 12 months also suggests that they may be less aware of early symptoms of their disease. Within the severe asthma group of subjects, high FeNO identifies a severe asthma phenotype that has the greatest eosinophilic airway inflammation, the most severe airflow limitation, and uses emergent care most often.
NO is produced by nitric oxide synthases (NOS), including constitutive (neuronal, or type 1, and endothelial, or type 3) and inducible (type 2) enzymes, all isoforms of which are present in the lung (39, 40). Abnormalities in NOS1 and NOS2 genotype and expression are associated with asthma and increased NOS activity is associated with increased FeNO levels (41–43). FeNO represents a balance between NO production and consumption (10). In particular, NO is rapidly consumed by reaction with superoxide. There is direct evidence that more severe obstruction in asthma is associated with increased spontaneous and stimulus-induced generation of superoxide by inflammatory cells in the airway (44).
The independent association of elevated FeNO with increased TLC is a novel finding, and suggests that there is an inflammatory component affecting lung mechanics that is separate from the air-trapping mechanism. Increased TLC has been associated with acute asthma exacerbation and with poorly controlled chronic asthma, and many of these subjects exhibit a decrease in TLC after therapy with bronchodilators and corticosteroids (45–47). Further studies are needed to determine the nature of the interaction between NO and TLC in asthma, but the current study shows that FeNO is associated with hyperinflation in asthma. Furthermore, FeNO in severe asthma might reflect airway-remodeling processes (48, 49). Because many of the variables that are related to NO are also related to severity, relationships between variables and FeNO were also evaluated within severity group by multivariate analyses. Multivariate analyses and modeling confirmed most of the associations suggested by the univariate analyses and revealed new findings as well. For instance, the relationship between FeNO and markers of atopy and eosinophilic inflammation was confirmed in nonsevere asthma, which suggests a strong dependence of FeNO on these variables. However, the multivariate significance of factors that influence FeNO in all asthma was driven primarily by the nonsevere asthma group. Most features did not significantly influence FeNO in severe asthma. This suggests that features other than those evaluated in this study may be determinants of high FeNO levels in severe asthma.
There are several possible explanations for the presence of high FeNO in patients with severe asthma. Because the FeNO levels of patients with nonsevere asthma decrease in response to corticosteroid therapy (22–27), the greater corticosteroid use in the severe asthma group would be expected to result in low levels of FeNO. In this context, one possible explanation for the high FeNO may be inadequate corticosteroid therapy. However, high FeNO levels in patients with severe asthma on high-dose oral or injectable corticosteroids are difficult to explain on this basis. Noncompliance with therapy is possible, but this explanation needs to invoke that patients with severe asthma with high FeNO are less compliant than patients with severe asthma with low FeNO, even though they report similar corticosteroid use. Importantly, high FeNO was not related to corticosteroid therapy in any analysis. Furthermore, the current ATS workshop criteria are meant to distinguish patients with severe asthma, but not to define those with nonsevere asthma. This may partially explain why the patients with high FeNO have similar characteristics in both patient groups. Taken together with previous studies (1, 3, 23, 28, 50–54), the high-FeNO severe asthma group may have relative resistance to corticosteroid therapy. In support of this, individuals with severe asthma and high FeNO are more likely to be taking theophylline. Given the current clinical practice of reserving theophylline use in asthma for individuals not responding to traditional therapies, greater use of daily theophylline in the high-FeNO group is another indirect marker of more difficult to treat, less corticosteroid-responsive asthma.
Analyses of the characteristics of individuals with low FeNO and severe asthma in this study also provide new information about asthma phenotypes. FeNO levels reflect a balance between the rates of NO production and its consumption, which is largely related to oxidant–NO reactions (10, 55, 56). Thus, low levels of FeNO in asthma may be related to less NO synthesis or greater oxidative consumption. Mechanisms that affect NO production include factors that modify NOS enzyme activity or expression, alter nonenzymatic release of NO from storage pools, or change the denitrifying organisms that colonize the upper airway (57–61). However, the end products of NO consumption are greater in severe asthma than in nonsevere asthma, which suggests that total NO production is greater in severe asthma but may not be reflected by levels in the exhaled breath because of greater oxidative consumption. In support of this concept, features of metabolic syndrome, which is characterized by oxidative stress and abnormalities of NO metabolism, were observed in the low-FeNO asthma group, that is, higher body mass index, hypertension, and diabetes.
Strengths of this study include the large cohort, the well-characterized population, and the prospective and standardized method of data collection. The main imitation of this study is also the fact that it is a cohort and not a randomized controlled trial. FeNO levels were analyzed in a cross-sectional fashion and not based on or before and after an intervention. Certain variables such as compliance with therapy could not be completely accounted for and verified.
Clinical asthma phenotypes have been recognized for some time (51, 62), but quantitative biomarkers have not been previously identified in severe asthma (51). This has limited the clear discrimination and understanding of severe asthma. Detailed and quantitative phenotypes will further our understanding of the pathobiology and genetics that contribute to severe asthma genesis (51). Although the current definition of asthma severity is useful for clinical research, it is cumbersome to use and impractical for the busy office setting. The availability of an easy-to-measure, noninvasive marker would greatly simplify and improve severe asthma management (63). The current findings suggest that evaluation of multiple quantitative biologic markers, such as FeNO, circulating NO reaction products, and sputum eosinophils, may provide a cumulative index for definition of asthma severity in the future. Here, FeNO is identified as a biomarker that distinguishes a group of patients with severe airflow obstruction, hyperreactivity, hyperinflation, and persistent airway inflammation. Although the retrospective nature of our analysis has precluded us from determining whether FeNO could predict future risk of exacerbations in asthma, its correlation with ER visits and hospital and ICU admission suggests a great potential for FeNO in identifying those patients with the most severe disease in clinical practice. Prospective studies would be helpful in confirming this fact that is suggested by our findings and to ascertain the determinants of the high-FeNO phenotype in patients with severe asthma, who are refractory to therapy.
Supplementary Material
Acknowledgments
The authors thank A. Janocha for the performance of NOx analyses.
Supported by HL69170, AI70649, HL04265, HL68863, HL69116, HL69174, HL69167, HL069155, HL69130, HL69149, HL69170, HL069349, ACRN U10HL74225, P01/U01HL67663, and P01/U01 HL67663; by a Third Frontier grant (TECH09-003) from the Ohio Department of Development; and by M01 RR018390 from the National Center for Research Resources.
This article has an online supplement, which is accessible from this issue's table of contents at www.atsjournals.org
Originally Published in Press as DOI: 10.1164/rccm.200905-0695OC on February 4, 2010
The Severe Asthma Research Program (SARP) is a multicenter asthma research group funded by the NHLBI and consisting of the following contributors (Steering Committee members are marked with an asterisk): Brigham and Women's Hospital, Boston, MA—Elliot Israel,* Bruce D. Levy, Gautham Marigowda; Cleveland Clinic, Cleveland, OH—Serpil C. Erzurum,* Raed A. Dweik, Suzy A. A. Comhair, Marcelle Baaklini, Daniel Laskowski, Jacqueline Pyle; Emory University, Atlanta, GA—W. Gerald Teague,* Anne M. Fitzpatrick, Eric Hunter; Imperial College School of Medicine, London, UK—Kian F. Chung,* Mark Hew, Alfonso Torrego, Sally Meah, Mun Lim; National Jewish Medical and Research Center, Denver, CO—Sally E. Wenzel,* Diane Rhodes; University of Pittsburgh, Pittsburgh, PA—William J. Calhoun,* Melissa P. Clark, Renee Folger, Rebecca Z. Wade; Bill T. Ameredes, Dori Smith; University of Virginia, Charlottesville, VA—Benjamin Gaston,* Peter Urban; University of Wisconsin, Madison, WI—William W. Busse,* Nizar Jarjour, Erin Billmeyer, Ronald L. Sorkness, Cheri Swenson, Gina Crisafi; Wake Forest University, Winston-Salem, NC—Eugene R. Bleecker,* Deborah Meyers, Wendy Moore, Stephen Peters, Annette Hastie, Gregory Hawkins, Jeffrey Krings, Regina Smith; Washington University in St. Louis, St. Louis, MO—Mario Castro,* Leonard Bacharier, Iftikhar Hussain, Jaime Tarsi; Data Coordinating Center, Denver, CO—James R. Murphy,* Douglas Curran-Everett; NHLBI, Bethesda, MD—Patricia Noel.
Conflict of Interest Statement: R.A.D. received more than $100,001 from Actelion, more than $100,001 from Gilead, and more than $100,001 from Novartis in industry-sponsored grants. R.L.S. received up to $1,000 from Synairgen in consultancy fees, and more than $100,001 from Modus Biological Membranes in industry-sponsored grants. The spouse/life partner of R.L.S. received $5,001–$10,000 from GlaxoSmithKline in advisory board fees, and $5,001–$10,000 from GlaxoSmithKline and $1,001–$5,000 from AstraZeneca in lecture fees. S.W. received $1,001–$5,000 from Amgen, $10,001–$50,000 from GlaxoSmithKline, $1,001–$5,000 from Wyeth, and $1,001–$5,000 from Genentech in consultancy fees; $1,001–$5,000 from GlaxoSmithKline, $10,001–$50,000 from Amira, $10,001–$50,000 from Epigenesis, and $5,001–$10,000 from Novartis in advisory board fees; $1,001–$5,000 from Critical Therapeutics and $1,001–$5,000 from Genentech in lecture fees; $50,001–$100,000 from GlaxoSmithKline, $5,001–$10,000 from Amgen, $10,001–$50,000 from MedImmune, $50,001–$100,000 from Ception, and $10,001–$50,000 from Aerovance in industry-sponsored grants; and holds stock ownership or options in Amira and Epigenesis. J.H. does not have a financial relationship with a commercial entity that has an interest in the subject of this manuscript. D.C.-E. does not have a financial relationship with a commercial entity that has an interest in the subject of this manuscript. S.A.A.C. does not have a financial relationship with a commercial entity that has an interest in the subject of this manuscript. E.R.B. does not have a financial relationship with a commercial entity that has an interest in the subject of this manuscript. W.B. received up to $1,000 from Alexion, $1,001–$5,000 from AstraZeneca, $1,001–$5,000 from Boehringer Ingelheim, up to $1,000 from Dainippon–Sumitomo, up to $1,000 from Funxional Therapeutics, Ltd., $1,001–$5,000 from Novartis, and $1,001–$5,000 from TEVA in consultancy fees; $1,001–$5,000 from Altair, $1,001–$5,000 from Amgen, $1,001–$5,000 from Abbott Laboratories, $1,001–$5,000 from Asthmatx, Inc., $1,001–$5,000 from Bristol–Meyers Squib, $1,001–$5,000 from Centocor, up to $1,000 from Genentech, up to $1,000 from GlaxoSmithKline, $1,001–$5,000 from Merck, $1,001–$5,000 from Schering Plough, $1,001–$5,000 from Pfizer, and $1,001–$5,000 from Wyeth in advisory board fees; $1,001–$5,000 from Merck in lecture fees; and $10,001–$50,000 from Novartis, $5,001–$10,000 from Centocor, more than $100,001 from GlaxoSmithKline, more than $100,001 from MedImmune, and $50,001–$100,000 from Ception in industry–sponsored grants. W.J.C. received $10,001–$50,000 from AstraZeneca and $5,001–$10,000 from Sepracor in consultancy fees, and $5,001–$10,000 from Merck in advisory board fees. M.C. received $10,001–$50,000 from Asthmatx, $1,001–$5,000 from Schering, $1,001–$5,000 from Electrocore, and $1,001–$5,000 from BMS in consultancy fees; $5,001–$10,000 Genentech in advisory board fees; $50,001–$100,000 from AstraZeneca, $10,001–$50,000 from Boehringer Ingelheim, $10,001–$50,000 from Pfizer, $5,001–$10,000 from Genentech, and $5,001–$10,000 from Merck in lecture fees; more than $100,001 from Asthmatx, more than $100,001 from Amgen, more than $100,001 from Centocor, more than $100,001 from Ception, and more than $100,001 from GlaxoSmithKline in industry-sponsored grants; $1,001–$5,000 from Elsevier in royalties; and $10,001–$50,000 from Pfizer, more than $100,001 from Genentech, more than $100,001 from MedImmune, more than $100,001 from Merck, and more than $100,001 from Novartis in contracted research. K.F.C. received $1,001–$5,000 from Gilead for ad hoc consulting; $1,001–$5,000 from GlaxoSmithKline, $1,001–$5,000 from Chiesi, $1,001–$5,000 from Merck, and $1,001–$5,000 from Boehringer Ingelheim for serving on an advisory board; $1,001–$5,000 from AstraZeneca, $1,001–$5,000 from GlaxoSmithKline, and $1,001–$5,000 from Chiesi in nonpromotional lecture fees; $10,001–$50,000 from Novartis for a phase 3 clinical trial, $1,001–$5,000 from Schering Plough for a phase 3 clinical trial, $1,001–$5,000 from AstraZeneca for phase 3 research, and $50,001–$100,000 from GlaxoSmithKline (collaborative) in industry-sponsored grants; and holds a patent from Imperial Innovations on use of p38 MAPK inhibitors to reverse corticosteroid resistance in airway disease. E.I. does not have a financial relationship with a commercial entity that has an interest in the subject of this manuscript. N.J. received $10,001–$50,000 from Asthmatx for review of CT scans, concept discussions, and advisory group meetings; $1,001–$5,000 from GlaxoSmithKline and $1,001–$5,000 from Genentech in advisory board fees; $1,001–$5,000 from Merck and Co. in lecture fees; and more than $100,001 from GlaxoSmithKline and more than $100,001 from Genentech in industry-sponsored grants. W.M. does not have a financial relationship with a commercial entity that has an interest in the subject of this manuscript. S.P. does not have a financial relationship with a commercial entity that has an interest in the subject of this manuscript. G.T. received $10,001–$50,000 from Merck and Co. and up to $1,000 from Aerocrine in lecture fees. The B.G. laboratory received up to $1,000 from Galleon in consultancy fees, more than $100,001 from Galleon in industry-sponsored grants, and more than $100,001 from the NIH in sponsored grants; $1,001–$5,000 from Williams and Connelly as an expert witness; holds patents from Galleon (for control of breathing: therapy), Respiratory Research (for breath condensate analysis), Airbase (for asthma treatment), and N30 (for asthma and CF treatment); and has received $5,001–$10,000 from Respiratory Research in royalties. S.C.E. received more than $100,001 from Asthmatx as an investigator in industry-sponsored grants.
References
- 1.Busse WW, Banks-Schlegel S, Wenzel SE. Pathophysiology of severe asthma. J Allergy Clin Immunol 2000;106:1033–1042. [DOI] [PubMed] [Google Scholar]
- 2.American Thoracic Society. Proceedings of the ATS workshop on refractory asthma: current understanding, recommendations, and unanswered questions. Am J Respir Crit Care Med 2000;162:2341–2351. [DOI] [PubMed] [Google Scholar]
- 3.Wenzel SE, Schwartz LB, Langmack EL, Halliday JL, Trudeau JB, Gibbs RL, Chu HW. Evidence that severe asthma can be divided pathologically into two inflammatory subtypes with distinct physiologic and clinical characteristics. Am J Respir Crit Care Med 1999;160:1001–1008. [DOI] [PubMed] [Google Scholar]
- 4.National Asthma Education and Prevention Program, National Heart, Lung, and Blood Institute. Guidelines for the diagnosis and the management of asthma: expert panel report II. Bethesda, MD: National Institutes of Health; 1997.
- 5.Green RH, Brightling CE, McKenna S, Hargadon B, Parker D, Bradding P, Wardlaw AJ, Pavord ID. Asthma exacerbations and sputum eosinophil counts: a randomised controlled trial. Lancet 2002;360:1715–1721. [DOI] [PubMed] [Google Scholar]
- 6.Jayaram L, Pizzichini MM, Cook RJ, Boulet LP, Lemiere C, Pizzichini E, Cartier A, Hussack P, Goldsmith CH, Laviolette M, et al. Determining asthma treatment by monitoring sputum cell counts: effect on exacerbations. Eur Respir J 2006;27:483–494. [DOI] [PubMed] [Google Scholar]
- 7.Smith AD, Cowan JO, Brassett KP, Herbison GP, Taylor DR. Use of exhaled nitric oxide measurements to guide treatment in chronic asthma. N Engl J Med 2005;352:2163–2173. [DOI] [PubMed] [Google Scholar]
- 8.Olin AC, Bake B, Toren K. Fraction of exhaled nitric oxide at 50 mL/s: reference values for adult lifelong never-smokers. Chest 2007;131:1852–1856. [DOI] [PubMed] [Google Scholar]
- 9.Smith AD, Cowan JO, Filsell S, McLachlan C, Monti-Sheehan G, Jackson P, Taylor DR. Diagnosing asthma: comparisons between exhaled nitric oxide measurements and conventional tests. Am J Respir Crit Care Med 2004;169:473–478. [DOI] [PubMed] [Google Scholar]
- 10.Dweik RA, Comhair SA, Gaston B, Thunnissen FB, Farver C, Thomassen MJ, Kavuru M, Hammel J, Abu-Soud HM, Erzurum SC. NO chemical events in the human airway during the immediate and late antigen-induced asthmatic response. Proc Natl Acad Sci USA 2001;98:2622–2627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Guo FH, Comhair SA, Zheng S, Dweik RA, Eissa NT, Thomassen MJ, Calhoun W, Erzurum SC. Molecular mechanisms of increased nitric oxide (NO) in asthma: evidence for transcriptional and post-translational regulation of NO synthesis. J Immunol 2000;164:5970–5980. [DOI] [PubMed] [Google Scholar]
- 12.Kharitonov SA, Yates D, Robbins RA, Logan-Sinclair R, Shinebourne EA, Barnes PJ. Increased nitric oxide in exhaled air of asthmatic patients. Lancet 1994;343:133–135. [DOI] [PubMed] [Google Scholar]
- 13.Massaro AF, Mehta S, Lilly CM, Kobzik L, Reilly JJ, Drazen JM. Elevated nitric oxide concentrations in isolated lower airway gas of asthmatic subjects. Am J Respir Crit Care Med 1996;153:1510–1514. [DOI] [PubMed] [Google Scholar]
- 14.Persson MG, Zetterstrom O, Agrenius V, Ihre E, Gustafsson LE. Single-breath nitric oxide measurements in asthmatic patients and smokers. Lancet 1994;343:146–147. [DOI] [PubMed] [Google Scholar]
- 15.Thomassen MJ, Raychaudhuri B, Dweik RA, Farver C, Buhrow L, Malur A, Connors MJ, Drazba J, Hammel J, Erzurum SC, et al. Nitric oxide regulation of asthmatic airway inflammation with segmental allergen challenge. J Allergy Clin Immunol 1999;104:1174–1182. [DOI] [PubMed] [Google Scholar]
- 16.Khatri SB, Hammel J, Kavuru MS, Erzurum SC, Dweik RA. Temporal association of nitric oxide levels and airflow in asthma after whole lung allergen challenge. J Appl Physiol 2003;95:436–440, discussion 435. [DOI] [PubMed] [Google Scholar]
- 17.Deykin A, Halpern O, Massaro AF, Drazen JM, Israel E. Expired nitric oxide after bronchoprovocation and repeated spirometry in patients with asthma. Am J Respir Crit Care Med 1998;157:769–775. [DOI] [PubMed] [Google Scholar]
- 18.Massaro AF, Gaston B, Kita D, Fanta C, Stamler JS, Drazen JM. Expired nitric oxide levels during treatment of acute asthma. Am J Respir Crit Care Med 1995;152:800–803. [DOI] [PubMed] [Google Scholar]
- 19.Delgado-Corcoran C, Kissoon N, Murphy SP, Duckworth LJ. Exhaled nitric oxide reflects asthma severity and asthma control. Pediatr Crit Care Med 2004;5:48–52. [DOI] [PubMed] [Google Scholar]
- 20.Payne DN, Adcock IM, Wilson NM, Oates T, Scallan M, Bush A. Relationship between exhaled nitric oxide and mucosal eosinophilic inflammation in children with difficult asthma, after treatment with oral prednisolone. Am J Respir Crit Care Med 2001;164:1376–1381. [DOI] [PubMed] [Google Scholar]
- 21.Silkoff PE, Sylvester JT, Zamel N, Permutt S. Airway nitric oxide diffusion in asthma: role in pulmonary function and bronchial responsiveness. Am J Respir Crit Care Med 2000;161:1218–1228. [DOI] [PubMed] [Google Scholar]
- 22.Silkoff PE, McClean P, Spino M, Erlich L, Slutsky AS, Zamel N. Dose–response relationship and reproducibility of the fall in exhaled nitric oxide after inhaled beclomethasone dipropionate therapy in asthma patients. Chest 2001;119:1322–1328. [DOI] [PubMed] [Google Scholar]
- 23.ten Brinke A, Zwinderman AH, Sterk PJ, Rabe KF, Bel EH. “Refractory” eosinophilic airway inflammation in severe asthma: effect of parenteral corticosteroids. Am J Respir Crit Care Med 2004;170:601–605. [DOI] [PubMed] [Google Scholar]
- 24.Lim S, Jatakanon A, John M, Gilbey T, O'Connor BJ, Chung KF, Barnes PJ. Effect of inhaled budesonide on lung function and airway inflammation: assessment by various inflammatory markers in mild asthma. Am J Respir Crit Care Med 1999;159:22–30. [DOI] [PubMed] [Google Scholar]
- 25.Dupont LJ, Rochette F, Demedts MG, Verleden GM. Exhaled nitric oxide correlates with airway hyperresponsiveness in steroid-naive patients with mild asthma. Am J Respir Crit Care Med 1998;157:894–898. [DOI] [PubMed] [Google Scholar]
- 26.Mattes J, Storm van's Gravesande K, Reining U, Alving K, Ihorst G, Henschen M, Kuehr J. NO in exhaled air is correlated with markers of eosinophilic airway inflammation in corticosteroid-dependent childhood asthma. Eur Respir J 1999;13:1391–1395. [PubMed] [Google Scholar]
- 27.Kharitonov SA, Yates DH, Barnes PJ. Inhaled glucocorticoids decrease nitric oxide in exhaled air of asthmatic patients. Am J Respir Crit Care Med 1996;153:454–457. [DOI] [PubMed] [Google Scholar]
- 28.European Network for Understanding Mechanisms of Severe Asthma. The ENFUMOSA cross-sectional European multicentre study of the clinical phenotype of chronic severe asthma. Eur Respir J 2003;22:470–477. [DOI] [PubMed] [Google Scholar]
- 29.Moore WC, Bleecker ER, Curran-Everett D, Erzurum SC, Ameredes BT, Bacharier L, Calhoun WJ, Castro M, Chung KF, Clark MP, et al. Characterization of the severe asthma phenotype by the National Heart, Lung, and Blood Institute's Severe Asthma Research Program. J Allergy Clin Immunol 2007;119:405–413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Dweik RA, Erzurum SC; Severe Asthma Research Program. Low levels of exhaled nitric oxide in severe asthma [abstract]. Proc Am Thorac Soc 2005;2:A244. [Google Scholar]
- 31.American Thoracic Society. Standardization of spirometry, 1994 update. Am J Respir Crit Care Med 1995;152:1107–1136. [DOI] [PubMed] [Google Scholar]
- 32.Wanger J, Clausen JL, Coates A, Pedersen OF, Brusasco V, Burgos F, Casaburi R, Crapo R, Enright P, van der Grinten CP, et al. Standardisation of the measurement of lung volumes. Eur Respir J 2005;26:511–522. [DOI] [PubMed] [Google Scholar]
- 33.Stocks J, Quanjer PH. Reference values for residual volume, functional residual capacity and total lung capacity: ATS workshop on lung volume measurements [official statement of the European Respiratory Society]. Eur Respir J 1995;8:492–506. [DOI] [PubMed] [Google Scholar]
- 34.American Thoracic Society. Lung function testing: selection of reference values and interpretative strategies. Am Rev Respir Dis 1991;144:1202–1218. [DOI] [PubMed] [Google Scholar]
- 35.American Thoracic Society, European Respiratory Society. ATS/ERS recommendations for standardized procedures for the online and offline measurement of exhaled lower respiratory nitric oxide and nasal nitric oxide, 2005. Am J Respir Crit Care Med 2005;171:912–930. [DOI] [PubMed] [Google Scholar]
- 36.Xu W, Kaneko FT, Zheng S, Comhair SA, Janocha AJ, Goggans T, Thunnissen FB, Farver C, Hazen SL, Jennings C, et al. Increased arginase II and decreased NO synthesis in endothelial cells of patients with pulmonary arterial hypertension. FASEB J 2004;18:1746–1748. [DOI] [PubMed] [Google Scholar]
- 37.R Development Core Team. R: a language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing; 2006.
- 38.Fitzpatrick AM, Gaston BM, Erzurum SC, Teague WG. Features of severe asthma in school-age children: atopy and increased exhaled nitric oxide. J Allergy Clin Immunol 2006;118:1218–1225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kobzik L, Bredt DS, Lowenstein CJ, Drazen J, Gaston B, Sugarbaker D, Stamler JS. Nitric oxide synthase in human and rat lung: immunocytochemical and histochemical localization. Am J Respir Cell Mol Biol 1993;9:371–377. [DOI] [PubMed] [Google Scholar]
- 40.Dweik RA, Laskowski D, Abu-Soud HM, Kaneko F, Hutte R, Stuehr DJ, Erzurum SC. Nitric oxide synthesis in the lung: regulation by oxygen through a kinetic mechanism. J Clin Invest 1998;101:660–666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.De Sanctis GT, MacLean JA, Hamada K, Mehta S, Scott JA, Jiao A, Yandava CN, Kobzik L, Wolyniec WW, Fabian AJ, et al. Contribution of nitric oxide synthases 1, 2, and 3 to airway hyperresponsiveness and inflammation in a murine model of asthma. J Exp Med 1999;189:1621–1630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Wechsler ME, Grasemann H, Deykin A, Silverman EK, Yandava CN, Israel E, Wand M, Drazen JM. Exhaled nitric oxide in patients with asthma: association with NOS1 genotype. Am J Respir Crit Care Med 2000;162:2043–2047. [DOI] [PubMed] [Google Scholar]
- 43.Grasemann H, Yandava CN, Storm van's Gravesande K, Deykin A, Pillari A, Ma J, Sonna LA, Lilly C, Stampfer MJ, Israel E, et al. A neuronal NO synthase (NOS1) gene polymorphism is associated with asthma. Biochem Biophys Res Commun 2000;272:391–394. [DOI] [PubMed] [Google Scholar]
- 44.Calhoun WJ, Reed HE, Moest DR, Stevens CA. Enhanced superoxide production by alveolar macrophages and air-space cells, airway inflammation, and alveolar macrophage density changes after segmental antigen bronchoprovocation in allergic subjects. Am Rev Respir Dis 1992;145:317–325. [DOI] [PubMed] [Google Scholar]
- 45.Blackie SP, al-Majed S, Staples CA, Hilliam C, Pare PD. Changes in total lung capacity during acute spontaneous asthma. Am Rev Respir Dis 1990;142:79–83. [DOI] [PubMed] [Google Scholar]
- 46.Gold WM, Kaufman HS, Nadel JA. Elastic recoil of the lungs in chronic asthmatic patients before and after therapy. J Appl Physiol 1967;23:433–438. [DOI] [PubMed] [Google Scholar]
- 47.Woolcock AJ, Read J. The static elastic properties of the lungs in asthma. Am Rev Respir Dis 1968;98:788–794. [DOI] [PubMed] [Google Scholar]
- 48.van Veen IH, Ten Brinke A, Sterk PJ, Sont JK, Gauw SA, Rabe KF, Bel EH. Exhaled nitric oxide predicts lung function decline in difficult-to-treat asthma. Eur Respir J 2008;32:344–349. [DOI] [PubMed] [Google Scholar]
- 49.Sugiura H, Komaki Y, Koarai A, Ichinose M. Nitrative stress in refractory asthma. J Allergy Clin Immunol 2008;121:355–360. [DOI] [PubMed] [Google Scholar]
- 50.Wenzel SE. Phenotypes in asthma: useful guides for therapy, distinct biological processes, or both? Am J Respir Crit Care Med 2004;170:579–580. [DOI] [PubMed] [Google Scholar]
- 51.Wenzel SE. Asthma: defining of the persistent adult phenotypes. Lancet 2006;368:804–813. [DOI] [PubMed] [Google Scholar]
- 52.Chanez P, Wenzel SE, Anderson GP, Anto JM, Bel EH, Boulet LP, Brightling CE, Busse WW, Castro M, Dahlen B, et al. Severe asthma in adults: what are the important questions? J Allergy Clin Immunol 2007;119:1337–1348. [DOI] [PubMed] [Google Scholar]
- 53.in't Veen JC, Smits HH, Hiemstra PS, Zwinderman AE, Sterk PJ, Bel EH. Lung function and sputum characteristics of patients with severe asthma during an induced exacerbation by double-blind steroid withdrawal. Am J Respir Crit Care Med 1999;160:93–99. [DOI] [PubMed] [Google Scholar]
- 54.ten Brinke A, Zwinderman AH, Sterk PJ, Rabe KF, Bel EH. Factors associated with persistent airflow limitation in severe asthma. Am J Respir Crit Care Med 2001;164:744–748. [DOI] [PubMed] [Google Scholar]
- 55.Comhair SA, Ricci KS, Arroliga M, Lara AR, Dweik RA, Song W, Hazen SL, Bleecker ER, Busse WW, Chung KF, et al. Correlation of systemic superoxide dismutase deficiency to airflow obstruction in asthma. Am J Respir Crit Care Med 2005;172:306–313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Comhair SA, Bhathena PR, Dweik RA, Kavuru M, Erzurum SC. Rapid loss of superoxide dismutase activity during antigen-induced asthmatic response. Lancet 2000;355:624. [DOI] [PubMed] [Google Scholar]
- 57.Ricciardolo FL, Gaston B, Hunt J. Acid stress in the pathology of asthma. J Allergy Clin Immunol 2004;113:610–619. [DOI] [PubMed] [Google Scholar]
- 58.Ricciardolo FL, Sterk PJ, Gaston B, Folkerts G. Nitric oxide in health and disease of the respiratory system. Physiol Rev 2004;84:731–765. [DOI] [PubMed] [Google Scholar]
- 59.Que LG, Liu L, Yan Y, Whitehead GS, Gavett SH, Schwartz DA, Stamler JS. Protection from experimental asthma by an endogenous bronchodilator. Science 2005;308:1618–1621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Gaston B, Sears S, Woods J, Hunt J, Ponaman M, McMahon T, Stamler JS. Bronchodilator S-nitrosothiol deficiency in asthmatic respiratory failure. Lancet 1998;351:1317–1319. [DOI] [PubMed] [Google Scholar]
- 61.Wu H, Romieu I, Sienra-Monge JJ, Estela Del Rio-Navarro B, Anderson DM, Jenchura CA, Li H, Ramirez-Aguilar M, Del Carmen Lara-Sanchez I, London SJ. Genetic variation in S-nitrosoglutathione reductase (GSNOR) and childhood asthma. J Allergy Clin Immunol 2007;120:322–328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Kiley J, Smith R, Noel P. Asthma phenotypes. Curr Opin Pulm Med 2007;13:19–23. [DOI] [PubMed] [Google Scholar]
- 63.Reddel HK, Taylor DR, Bateman ED, Boulet LP, Boushey HA, Busse WW, Casale TB, Chanez P, Enright PL, Gibson PG, et al. An official American Thoracic Society/European Respiratory Society statement: asthma control and exacerbations: standardizing endpoints for clinical asthma trials and clinical practice. Am J Respir Crit Care Med 2009;180:59–99. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.