Abstract
Compensatory endocytosis (CE) ensures recycling of membrane components and maintenance of plasma membrane size; however, the mechanisms, regulation, and physiological functions of clathrin-independent modes of CE are poorly understood. CE was studied in umbrella cells, which undergo regulated exocytosis of subapical discoidal/fusiform vesicles (DFV) during bladder filling, and may then replenish the pool of DFV by internalizing apical membrane during voiding. We found that voiding-stimulated CE, which depended on β1 integrin-associated signalling pathways, occurred by a dynamin-, actin-, and RhoA-regulated mechanism and was independent of caveolins, clathrin, and flotillin. Internalized apical membrane and fluid were initially found in ZO-1-positive vesicles, which were distinct from DFV, classical early endosomes, or the Golgi, and subsequently in lysosomes. We conclude that clathrin-independent CE in umbrella cells functions to recover membrane during voiding, is integrin regulated, occurs by a RhoA- and dynamin-dependent pathway, and terminates in degradation and not recapture of membrane in DFV.
Keywords: clathrin-independent endocytosis, compensatory endocytosis, integrin, RhoA, uroepithelium
Introduction
Compensatory endocytosis (CE) is a crucial process whereby regulated secretory cells such as pancreatic acinar cells, neurons, and neuroendocrine cells recover plasma membrane constituents added during an earlier bout of exocytosis (Gundelfinger et al, 2003; Sokac and Bement, 2006; Wu et al, 2007; Barg and Machado, 2008). In cells such as neurons, CE ensures the rapid cycling and reuse of a limited pool of proteins and lipids that are required for neurotransmission (Gundelfinger et al, 2003; Wu et al, 2007), and in all regulated secretory cells, CE serves to maintain cell surface area by balancing exocytosis with endocytosis (Gundelfinger et al, 2003; Sokac and Bement, 2006; Wu et al, 2007; Barg and Machado, 2008). The best-understood type of CE is mediated by the classical clathrin-dependent pathway, which is prevalent in neuronal and neuroendocrine cells and requires clathrin, accessory, and adaptor proteins such as AP-2 and the GTPase dynamin (Gundelfinger et al, 2003; Traub, 2009).
In contrast, other forms of CE may be mediated by clathrin-independent pathways (Mayor and Pagano, 2007; Gong et al, 2008; Sandvig et al, 2008); however, the mechanisms and physiological significance of these alternative modes of internalization are poorly understood. In adrenal chromaffin cells, rapid CE is dependent on dynamin-1, but is otherwise independent of clathrin (Artalejo et al, 2002). In oocytes, acinar cells, and type II alveolar cells, relatively large exocytic granules fuse, become coated with actin, and then remain associated with the plasma membrane for an extended period of time (from seconds to several minutes) before internalization by CE (Valentijn et al, 2000; Sokac et al, 2003; van Weeren et al, 2004; Yu and Bement, 2007). The mechanism of scission remains unclear, but studies in oocytes indicate that actin may work in conjunction with multiple myosin motor proteins and Cdc42 to promote internalization (Sokac et al, 2003; Yu and Bement, 2007). Other forms of clathrin-independent CE may include kiss-and-run fusion events (Gundelfinger et al, 2003; He and Wu, 2007; Barg and Machado, 2008), bulk endocytosis in neuronal cells (Holt et al, 2003; Clayton et al, 2007; Wu et al, 2007), and a pathway in neuronal cells that is clathrin and dynamin independent, but is otherwise uncharacterized (Xu et al, 2008). In cells that lack regulated secretory pathways, several forms of clathrin-independent pinocytosis exist including macropinocytosis, the circular dorsal ruffle pathway, caveolin-mediated endocytosis, flotillin-mediated endocytosis, the Cdc42-regulated glycosylphosphatidylinositol-enriched endosomal compartments (GEEC) pathway, possibly an Arf6-regulated pathway, and the RhoA pathway (McNiven, 2006; Mayor and Pagano, 2007; Gong et al, 2008; Sandvig et al, 2008; Hansen and Nichols, 2009). However, information about the function of these mechanisms in CE is scant.
The bladder umbrella cell, which forms the outermost layer of the stratified uroepithelium (Khandelwal et al, 2009), may provide important insight into clathrin-independent forms of CE. Bladder filling increases umbrella cell apical membrane tension, which stimulates the Ca2+-regulated exocytosis of a subapical pool of discoidal- or fusiform-shaped vesicles (DFV) (Khandelwal et al, 2009; Yu et al, 2009). This exocytic response is modulated by a filling-induced CE, which is triggered by increased basolateral tension as the bladder fills, and possibly by a voiding-induced CE when the bladder empties and basolateral tension increases as the tissue refolds (Yu et al, 2009). How tension is sensed at the basolateral membrane and then transduced into changes in apical endocytosis remains a mystery. Intriguingly, adult umbrella cells are reported neither to express caveolins (Fong et al, 2003; Barresi et al, 2006), nor do they express clathrin under their apical surface (Born et al, 2003), indicating that apical endocytosis in these cells is likely through a clathrin- and caveolar-independent pathway. However, the specific mechanism(s) is unknown. The text-book model proposes that on voiding excess membrane recovered by CE replenishes the DFV population (Khandelwal et al, 2009). However, earlier demonstrations of voiding-associated endocytosis are vague, in part, because the act of filling the bladder with tracers can induce internalization (Amano et al, 1991; Truschel et al, 2002; Yu et al, 2009). Furthermore, endocytosed membrane and fluid show little co-localization with DFV, and instead these markers appear in multivesicular bodies and lysosomes, indicating that degradation may be their final fate (Amano et al, 1991; Truschel et al, 2002; Alenghat et al, 2004; Khandelwal et al, 2008). Other studies indicate that endocytosis does not occur in differentiated cultures of umbrella cells (Kreft et al, 2009), or that after voiding, excess membrane may be pinched off the apical surface in a fission process (Born et al, 2003).
Our studies provide insight into the regulation, possible mechanisms, and function of clathrin-independent CE. We observed that voiding stimulates recovery of umbrella cell apical membrane through a dynamin- and RhoA-dependent form of CE that is dependent on basolateral integrins. The latter may modulate basolateral membrane mechanotransduction and regulate RhoA activity downstream of focal adhesion kinase (FAK) and phosphoinositide-3 kinase (PI-3K). Furthermore, the majority of endocytosed cargo was initially found in peripheral junction-associated apical endosomes (PJAEs), not DFV, and a large fraction of endocytosed cargo was degraded in lysosomes and not recaptured in DFV.
Results
Bladder voiding stimulates CE at the apical surface of umbrella cells
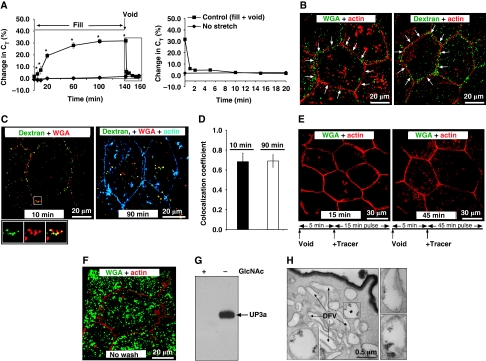
Bladder filling was simulated by mounting isolated rabbit uroepithelium in an Ussing stretch chamber and bowing the tissue outwards by slowly filling the mucosal hemichamber (Supplementary Figure S1). This led to an increase in transepithelial capacitance (CT, where 1 μF≈1 cm2 of apical surface area) (Figure 1A, left panel), which was earlier shown to result from the regulated apical exocytosis of DFV (Lewis and de Moura, 1982; Truschel et al, 2002; Wang et al, 2003; Khandelwal et al, 2008; Yu et al, 2009). The exocytic response can be kinetically separated into an early stage, which in these studies occurs during the first 20–30 min, and a subsequent late stage (Balestreire and Apodaca, 2007; Yu et al, 2009). These two stages are differentially regulated (Balestreire and Apodaca, 2007; Yu et al, 2009). The early stage response is modulated by filling-induced CE (Yu et al, 2009); however, these events were not apparent in Figure 1A because we sampled the initial changes in CT less frequently than our earlier studies (Yu et al, 2009). Tissue incubated in the absence of stretch served as a control, and no change in CT was observed (Figure 1A). The rise in filling-induced CT was abruptly reversed when experimental voiding was induced by quickly lowering the level of fluid in the mucosal hemichamber until the tissue bowed inwards towards the mucosal hemichamber (Figure 1A, right panel; Supplementary Figure S1). This treatment increases basolateral membrane tension and mimics the rapid release of urine and refolding of the bladder mucosa that occurs during voiding (Yu et al, 2009). The decrease in CT occurred rapidly (within 1 min) and similar to other forms of CE resulted in the near complete recovery of membrane, regardless of the preceding period of exocytosis (30–300 min; data not shown; Balestreire and Apodaca, 2007).
Figure 1.
Voiding stimulates CE. (A) Left panel: To simulate bladder filling, uroepithelial tissue was mounted in an Ussing stretch chamber and the mucosal hemichamber was slowly filled until the tissue bowed outwards. At the indicated time, experimental voiding was induced by removing fluid in the mucosal hemichamber until the tissue bowed inwards. The change in CT was measured in quiescent tissue (no stretch) or tissue that was bowed outwards and then inwards (control). The boxed region is expanded in the panel on the right. Right panel: Changes in CT in response to experimental voiding. The time scale is renumbered so that the initiation of voiding occurs at t=0. Values are mean±s.e.m. (n⩾7), and those values significantly different than no stretch samples (P<0.05) are indicated with an asterisk. (B) Uptake of FITC-labelled WGA or dextran added to the mucosal hemichamber 10 min before voiding and then processed for immunofluorescence 5 min after experimental voiding. A 3D reconstruction is shown. The junctional complex surrounding individual umbrella cells is indicated by arrows. F-actin associated with the apical plasma membrane is difficult to discern in these images, but is apparent along the lateral surfaces of the umbrella cells. The scattered patches of actin observed at the base of the cells are the cortical actin associated with the plasma membrane of the underlying intermediate cell layer. The distribution of markers near the junctions is more apparent in Supplementary movies S1 and S2. (C) Distribution of FITC-dextran and Alexa647-WGA co-internalized for 10 or 90 min after experimental voiding. The boxed region in the left-hand panel is magnified in images below. (D) Co-localization coefficient for the fraction of FITC-dextran that co-localized with Alexa647-WGA. Values are mean±s.e.m. (n⩾7). (E) Uptake of FITC-WGA added 5 min after voiding and then incubated for 15 or 45 min. (F) Distribution of FITC-WGA added to the apical surface of the umbrella cells and then fixed and visualized. (G) Membrane fractions of uroepithelium were incubated with agarose-WGA in the presence (+) or absence (−) of N-acetyl glucosamine (GlcNAc). The bound proteins were resolved by SDS–PAGE and UP3a detected by western blot analysis. (H) TEM of rat bladder filled with cationized ferritin and induced to void. Left panel: DFV are indicated with arrows and a cationized ferritin-positive PJAE is indicated with an asterisk. The boxed PJAE is magnified in the inset. Right panels: examples of cationized ferritin-positive PJAEs.
To confirm that endocytosis occurred during experimental voiding, the membrane marker FITC-conjugated wheat germ agglutinin (WGA) or the fluid-phase marker FITC-conjugated dextran was added to the mucosal hemichamber 10 min before release of the mucosal pressure head. On voiding, both FITC-WGA and FITC-dextran appeared in apically localized vesicular structures that clustered near the junctional complexes that we term as PJAEs (Figure 1B; Supplementary movies S1 and S2). When co-internalized, FITC-dextran and Alexa647-WGA were often found in identical structures (Figure 1C and D; co-localization coefficient of ∼0.7), indicating that PJAEs receive both fluid and membrane markers. A small amount of tracer was internalized during the 10-min incubation before voiding, but was <3% of the amount that was taken up during experimental voiding (mean intensity of 1.2±0.7 before voiding versus 43.7±9.5 afterwards; mean±s.e.m., n=4). Estimates of the fraction of apical membrane that was endocytosed during experimental voiding were ∼25% (25.5±4.14% using FITC-WGA or 23.06±5.15% using dextran; mean±s.e.m., n⩾15), which is in reasonable concordance with the ∼30% decrease in apical surface area we measured by capacitance. We also observed that internalization of tracer was restricted to a short time window (⩽5 min) just after pressure release, and no PJAEs were labelled when tracer was added 5 min after voiding, and then chased for 15–45 min (Figure 1E). The appearance of FITC-WGA in PJAEs was not due to preferential binding of proteins/lipids near the junctional complex, as surface-bound FITC-WGA was distributed across the apical pole of the umbrella cells (Figure 1F). Furthermore, FITC-WGA likely served as a bulk membrane marker: when membrane fractions of uroepithelium were incubated with agarose-WGA, they bound to uroplakin 3A (UP3a; Figure 1G), a protein constituent of DFV and the cell surface plaques that comprise ∼90% of the umbrella cell apical plasma membrane (Wu et al, 2009).
CE was also observed in rat bladders that were filled in situ and then allowed to void in the presence of FITC-dextran or WGA (e.g. Figure 4E). However, in rat umbrella cells, there was a tendency for the endocytic structures to appear somewhat more randomly dispersed in the apical cytoplasm. The ultrastructure of PJAEs was examined by instilling cationized ferritin into rat bladders just before voiding. Cationized ferritin bound avidly to all regions of the umbrella cell apical membrane and appeared as a thick coat on the extracellular surface of the cells (Figure 1H). Whereas the majority of subapical DFV lacked ferritin, this marker was observed in vesicular structures that were ∼0.5 μm in diameter (examples of which are shown in Figure 1H). Taken together, these results indicate that voiding stimulates a rapid CE that retrieves bulk apical membrane into vesicular PJAEs.
Internalized membrane and fluid are targeted for degradation
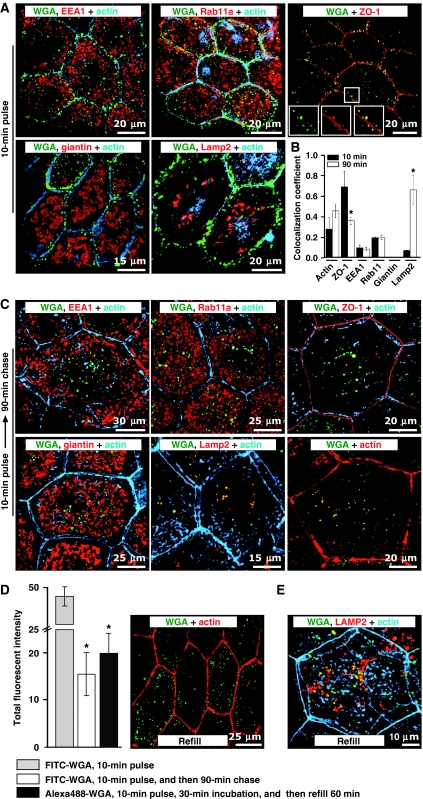
Next, we examined the fate of apically internalized membrane after voiding-induced CE. Within 10 min of internalization, FITC-WGA-labelled PJAEs showed minimal co-localization with the classical marker of early endosomes EEA1 (Supplementary movie S3), the Golgi marker giantin, or the late endosome- and lysosome-associated protein LAMP2 (Figure 2A and B; Supplementary Figure S2A). There was a small degree of co-localization of endocytic tracer with Rab11a (Figure 2A and B; Supplementary Figure S2A and Supplementary movie S4), which in umbrella cells label the DFV (Khandelwal et al, 2008). This prompted us to examine whether endocytosed WGA colocalized with exogenously expressed human growth hormone, which is efficiently packaged into DFV (Kerr et al, 1998; Khandelwal et al, 2008). In these experiments, we observed minor co-localization of these two markers (Supplementary Figure S3; co-localization coefficient of 0.08±0.03, n=7), which is consistent with our electron micrographs that show endocytic tracer is excluded from the majority of DFV (Figure 1H). In contrast, there was a high degree of co-localization between endocytosed tracers and actin or the tight junction-associated protein ZO-1 (∼70% Figure 2A and B; Supplementary Figure S2A and Supplementary movie S5).
Figure 2.
Delivery of endocytosed membrane to late endosomes/lysosomes. (A) Co-localization of EEA1, Rab11a, ZO-1, giantin, or LAMP2 with FITC-WGA 10 min after experimental voiding. (B) Co-localization coefficients for FITC-WGA and the indicated marker 10 or 90 min after experimental voiding. Values are mean±s.e.m. (n⩾7). Those values significantly different from the 10-min time point (P<0.05) are indicated with an asterisk. (C) Distribution of FITC-WGA and indicated markers 90 min after voiding. (D) Left panel: Intensity of labelled WGA pulsed for 10 min after experimental voiding ± an 80-min chase in the absence of additional marker. Alternatively, WGA was pulsed for 10 min after voiding, chased for 30 min in the absence of filling, and then chased for an additional 60 min during which time the chamber was re-filled. Values are mean±s.e.m. (n⩾7) and those values significantly different than samples fixed after the pulse (P<0.05) are indicated with an asterisk. Right panel: Distribution of Alexa488-WGA that was internalized for 10 min after voiding, chased for 30 min, and then chased for 60 min during which time the mucosal hemichamber was re-filled. (E) Co-localization of Alexa488-WGA and LAMP2 in tissue that was refilled.
Forty-five minutes after voiding, there was little change in the distribution of the PJAEs or their co-localization with the above markers (data not shown), indicating that PJAEs may be long lived. However, by 90 min, there was a decrease in the intensity of the FITC-WGA signal, which could be prevented to some extent by treating the tissue with leupeptin, an inhibitor of some lysosomal proteinases. In these leupeptin-treated tissues, FITC-WGA-labelled endosomes were no longer concentrated near the cell junctions; instead, they appeared deeper in the cell cytoplasm. These endosomes showed only a small degree of co-localization with EEA1, Rab11a, or giantin and the amount of ZO-1 co-localization decreased (Figure 2B and C; Supplementary Figure S2B). However, they showed a striking increase in co-localization with LAMP2 (Figure 2B and C; Supplementary Figure S2B), consistent with delivery of endocytosed cargo to LAMP2-positive late endosomes/lysosomes and subsequent degradation.
In the preceding experiments, the uroepithelium underwent one cycle of filling and voiding, but afterwards, the tissue was left in a quiescent state. To rule out the possibility that subsequent re-filling may induce recycling or otherwise alter the fate of the endocytosed markers, we internalized WGA, conjugated to the pH-insensitive fluorophore Alexa488, during voiding. The tissue was then incubated for 30 min with leupeptin and N-acetyl glucosamine (to prevent external WGA from rebinding), and then re-filled for 60 min. Relative to the original pool of endocytosed marker, there was a significant loss of WGA signal after a 90-min chase in both quiescent and re-stretched tissue (Figure 2D). However, there was no significant difference in the distribution or the total amount of fluorophore-labelled WGA that remained in quiescent or re-filled tissue after 90 min (Figure 2D; P<0.05), indicating that re-filling did not alter the fate of the endocytosed marker. The loss in WGA signal may reflect degradation by a leupeptin-insensitive pathway or recycling of marker. Furthermore, a large fraction of Alexa488-WGA co-localized with Lamp2-positive structures (Figure 2E, co-localization coefficient 0.69±0.06, n=6), consistent with delivery of cargo to late endosomes/lysosomes even in filled tissue. Taken together, these data indicate that the majority of internalized membrane and fluid are rapidly delivered to PJAEs and not DFV, and the fate of internalized cargo seems to be relatively slow degradation in late endosomes and lysosomes through a non-canonical delivery route.
CE occurs through a caveolar-, clathrin-, and flotillin-independent pathway
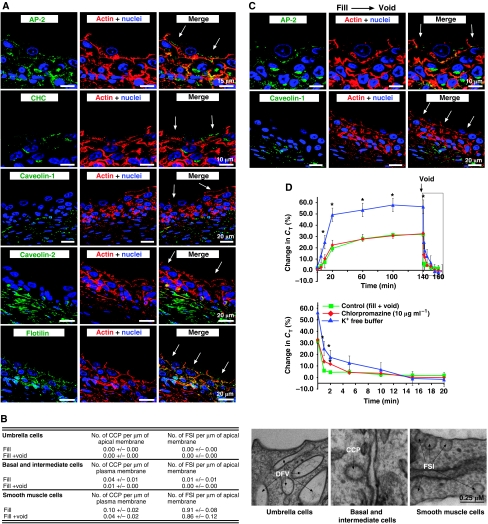
We next explored possible mechanisms for voiding-induced CE. Similar to earlier studies (Born et al, 2003), we found that there was no expression of clathrin at the apical surface of umbrella cells (Figure 3A). We also found no AP-2 adaptor complex at this domain (Figure 3A), although some AP-2 staining was detected at the basolateral surface of the umbrella cells as well as the plasma membrane of the underlying intermediate and basal cell layers (Figure 3A). Furthermore, we observed no morphologically identifiable clathrin-coated pits at the apical surface of umbrella cells examined by transmission electron microscopy (Figure 3B). However, a small number of clathrin-coated pits were observed on the plasma membranes of the intermediate and basal cells as well as in scattered smooth muscle cells, which lie just below the uroepithelium (Figure 3B). We also examined whether AP-2 was redistributed to the apical plasma membrane immediately after experimental voiding; however, there was no apparent change in the distribution of AP-2 under these conditions (Figure 3C), and there was no appearance of clathrin-coated pits at the apical plasma membrane immediately after voiding (Figure 3B). Treatment with inhibitors of clathrin-dependent endocytosis, including chlorpromazine or incubation in K+-free medium (Ivanov, 2008), marginally slowed the rate of CE, but did not prevent complete return of CT to baseline (Figure 3D). Chlorpromazine had no effect on exocytosis, but K+-free treatment significantly stimulated release, presumably a result of altering the umbrella cell membrane potential. As a positive control, we showed that chlorpromazine inhibited the basolateral uptake of transferrin (Supplementary Figure S4A and B), which is internalized by a clathrin-dependent mechanism (Sabharanjak et al, 2002).
Figure 3.
CE is through a caveolin-, clathrin-, and flotillin-independent mechanism. (A) Distribution of the α adaptin of AP-2, clathrin heavy chain (CHC), caveolin-1, -2, or flotillin-1 in cryosections of uroepithelial tissue. The location of an umbrella cell apical membrane is indicated by arrows. (B) Analysis of the number of clathrin-coated pits (CCP) and flask-shaped invaginations (FSI) per micron of the indicated membrane in samples fixed before voiding or fixed immediately after voiding. Values are mean±s.e.m. (n=25). Panels to the right are electron micrographs of the sampled membranes. CCPs or FSIs are indicated with arrows. (C) Distribution of the α adaptin of AP-2 and caveolin-1 in tissue that was experimentally filled and then fixed 5-min after voiding. (D) Effect of chlorpromazine (10 μg/ml) or K+-free buffer on CT responses during experimental filling and voiding. The boxed region in the upper panel is expanded in the lower panel. Induction of voiding is indicated by the arrow. Control data are reproduced from Figure 1A. Values are mean±s.e.m. (n⩾7), and those values significantly different than control samples (P<0.05) are indicated with an asterisk.
Caveolae and flotillin carriers were also unlikely to be responsible for apical endocytosis. First, multiple antibodies failed to detect the caveolar-associated proteins caveolin-1 and -2 in cryosections of the uroepithelium (Figure 3A and data not shown); however, both proteins were expressed in the underlying capillary endothelium and scattered rests of smooth muscle cells (Figure 3A), as well as the detrusor muscle (data not shown). Second, RT–PCR confirmed that message for caveolin-1 or -2 was absent in the uroepithelium, but was present in the bladder proper (Supplementary Figure S5). Third, ultrastructural analysis showed that there were neither flask-shaped 50–100 nm invaginations, characteristic of caveolae and flotillin-associated carriers (Frick et al, 2007), at the apical surface of the umbrella cells (Figure 3B), nor was flotillin-1 staining observed at this surface (Figure 3A). There were few flask-shaped invaginations at the plasma membranes of the basal and intermediate cells (Figure 3B), and an antibody to flotillin-1 showed weak staining along the basolateral surface of the umbrella cells and the plasma membranes of the underlying cell layers (Figure 3A). In contrast, flask-shaped invaginations were relatively abundant along the surface of the smooth muscle cells (Figure 3B). Experimental voiding did not stimulate the expression of flask-shaped invaginations or cause redistribution of caveolins or flotillin-1 to the apical surface of the umbrella cells (Figure 3B and C). In summary, these data indicate that CE at the apical membrane of umbrella cells is likely to be independent of caveolin, clathrin, or flotillin.
CE is actin, dynamin, and RhoA dependent
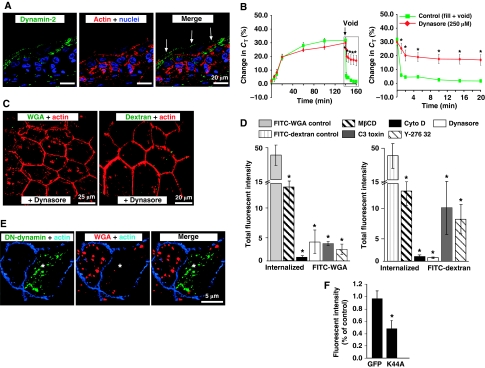
The GTPase dynamin is known to be important for several clathrin-independent pathways of endocytosis (Mayor and Pagano, 2007), and we observed that dynamin-2 was found localized underneath the entire plasma membrane of the umbrella cells, as well as that of the first intermediate cell layer (Figure 4A). This is consistent with a recent report that dynamin-2 is expressed in umbrella cells and associated with DFV (Terada et al, 2009). However, it is unknown if voiding-induced CE is dependent on dynamin. In tissue culture cells, the dynamin-1 and -2-specific inhibitor dynasore is often used at ∼80 μM (Macia et al, 2006); however, the results in bladder tissue were variable at this concentration, possibly because of problems with drug access or turnover. When used at 250 μM, dynasore had no effect on exocytosis, but it inhibited the voiding-induced decrease in CT by ∼60% (Figure 4B), and markedly impaired the uptake of FITC-WGA and FITC-dextran (Figure 4C and D) as well as the number of labelled endocytic structures (Supplementary Figure S7). As further evidence of the function of dynamin-2 in CE, we used adenovirus-mediated in situ transduction of umbrella cells to express green fluorescent protein (GFP) or a GFP-labelled dominant-negative mutant of dynamin-2 (DN-dynaminK44A). FITC-WGA uptake was observed in non-transduced umbrella cells, but was significantly inhibited by ∼50% in cells expressing DN-dynaminK44A (Figure 4E and F). Internalization was not perturbed in cells expressing GFP alone (Figure 4F; Supplementary Figure S6).
Figure 4.
Dependence of CE on dynamin. (A) Localization of dynamin-2 in uroepithelial tissue. The apical surface of an umbrella cell is indicated by arrows. (B) Effect of dynasore on CT during filling and voiding. Voiding was initiated at the arrow. The boxed region in the left panel is expanded in the right-hand panel. Control data are reproduced from Figure 1A. Values are mean±s.e.m. (n⩾5), and those values significantly different than control samples (P<0.5) are indicated with an asterisk. (C) Uptake of FITC-WGA and FITC-dextran in tissue treated with dynasore (250 μM). (D) Quantitation of FITC-WGA and FITC-dextran uptake in tissue treated with MβCD (10 mM), cyto-D (25 μg/ml), C3 toxin (1 μg/ml), dynasore (250 μM), or Y-27632 (25 μM). Values are mean±s.e.m. (n⩾10), and those values significantly different than control samples (P<0.05) are indicated with an asterisk. (E, F) Rat bladders were transduced in situ with adenovirus expressing GFP alone or GFP-labelled DN-dynaminK44A (DN-dynamin). The bladder was filled with Alexa647-WGA, stimulated to undergo voiding, excised, and then fixed and processed for immunofluorescence. (E) A transduced cell expressing DN-dynaminK44A (K44A) is indicated with an asterisk. (F) The uptake of Alex647-WGA was quantified in cells expressing GFP or DN-dynaminK44A (K44A) and normalized to uptake in adjacent cells that did not express exogenous protein. Data are mean±s.e.m. (n⩾70).
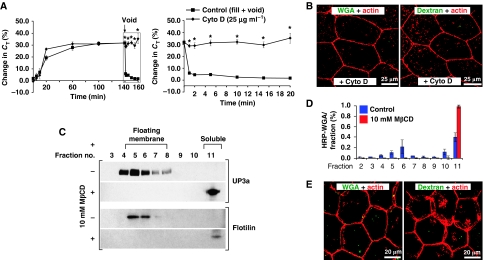
Many clathrin-independent endocytic pathways require actin and are dependent on cholesterol (Mayor and Pagano, 2007; Sandvig et al, 2008). To test whether CE is actin dependent, we treated tissue with the actin-disrupting agent cytochalasin D (cytoD). As cytoD impairs exocytosis, and can negatively impact tissue integrity, we limited pretreatment to 15 min before voiding. CytoD completely inhibited the recovery of membrane (Figure 5A), and also impaired the uptake of FITC-WGA and FITC-dextran (Figures 4D and 5B; Supplementary Figure S7). The apical plasma membrane of umbrella cells is especially rich in cholesterol and sphingolipids and is highly detergent resistant (Hicks et al, 1974; Wu et al, 1990). To examine the function of cholesterol and associated detergent-resistant membranes in CE, we treated tissue with 10 mM methyl-β-cyclodextrin (MβCD), a cholesterol-sequestering agent. When isolated membranes were analysed in sucrose flotation gradients, MβCD treatment resulted in a complete shift of UP3a-positive apical membranes from a lighter ‘floating' fraction to a denser ‘soluble' fraction (Figure 5C). HRP-WGA, prebound to the apical membrane at 4°C, was found in both the floating and soluble regions of the gradient (Figure 5D). As the whole of the apical membrane of the umbrella cell is reported to be detergent resistant (Wu et al, 1990), the soluble fraction of HRP-WGA may reflect dissociation of this lectin during the processing of the tissue. Similar to UP3a, all of the HRP-WGA shifted to the soluble pool on treatment of tissue with MβCD (Figure 5D). In MβCD-treated tissue, CE was markedly inhibited and there was little uptake of FITC-WGA (Figure 5E).
Figure 5.
CE is actin and cholesterol dependent. (A) Changes in CT in control tissue or that treated with cytoD (25 μg/ml). Voiding was induced at the arrow. The boxed region is expanded in the right-hand panel. Control data are reproduced from Figure 1A. Values are mean±s.e.m. (n⩾5), and those values significantly different than control samples (P<0.05) are indicated with an asterisk. (B) Uptake of FITC-WGA or FITC-dextran in tissue treated with cytoD. (C) Effect of MβCD treatment on umbrella cell detergent-resistant membranes. The apical surface of umbrella cells was incubated in the presence or absence of MβCD, and Triton X-100-resistant (floating membrane) and -soluble membranes resolved by sucrose flotation gradients. The localization of UP3a or flotillin in the gradients was determined by western blotting. (D) Association of HRP-WGA with floating and soluble membrane fractions (±MβCD treatment). Data are mean±s.e.m. (n=3). (E) Uptake of FITC-WGA or FITC-dextran in tissue treated with MβCD.
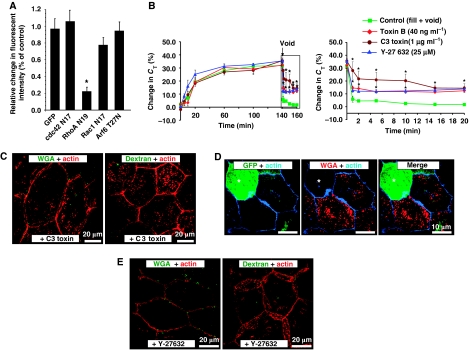
In addition to excluding clathrin-, caveolar, and flotillin-dependent pathways, our data also seem to rule out a function for the dynamin-independent macropinocytotic pathways and the dynamin-independent CDC42-regulated GEEC and Arf6 pathways (Naslavsky et al, 2004; Chadda et al, 2007; Mayor and Pagano, 2007; Hansen and Nichols, 2009). In fact, expression of dominant-negative mutants of Cdc42 or Arf6 (which impairs endocytic recycling) had no significant effects on voiding-induced CE (Figure 6A). This prompted us to explore the possible involvement of the RhoA pathway in CE. Treatment with the Clostridium difficile toxin B, a broad-spectrum inhibitor of Rho-family GTPases that is cell permeable, had little effect on exocytosis, but inhibited membrane recovery by ∼40% (Figure 6B). Similarly, a membrane-permeant variant of Clostridium botulinum C3 transferase, an inhibitor of the RhoA subfamily, but not Cdc42 or Rac1, also had no effect on exocytosis, but significantly reduced CE (Figure 6B), and impaired endocytosis of FITC-WGA and FITC-dextran (Figures 4D and 6C; Supplementary Figure S7). To further confirm the involvement of RhoA, we expressed dominant-negative RhoA (DN-RhoAN19) in conjunction with GFP, or GFP alone, in rat bladder umbrella cells. Uptake of Alexa647-WGA was inhibited by ∼80% in cells expressing DN-RhoAN19 (Figure 6A and D), but as described above, expression of GFP alone had no impact on uptake of Alexa647-WGA (Figure 6A; Supplementary Figure S6). As Rho-associated coiled coil-containing protein kinases (ROCKs) are major downstream effectors of RhoA, we treated the uroepithelium with the ROCK inhibitor Y-27632. This drug had no impact on exocytosis, but significantly inhibited membrane recovery by ∼40% (Figure 6B) and uptake of FITC-labelled WGA or dextran by ⩾70% (Figures 4D and 6E; Supplementary Figure S7). Finally, the RhoA endocytic pathway is also reported to be regulated by Rac1 (Grassart et al, 2008); however, we observed no significant effect of expressing DN-Rac1 in umbrella cell CE (Figure 6A). Taken together, the above data indicate that umbrella cell CE is related to the clathrin independent, but dynamin-dependent RhoA pathway for endocytosis (Lamaze et al, 2001). Furthermore, ROCKs may act downstream of RhoA to modulate CE, possibly by altering the actin cytoskeleton.
Figure 6.
RhoA dependence of CE in umbrella cells. (A) Uptake of Alexa647-WGA was quantified in cells expressing GFP or the indicated ras family GTPase and normalized to uptake in adjacent cells that did not express exogenous protein. GFP data is reproduced from Figure 4F. Data are mean±s.e.m. (n⩾70). (B) Effect of C3 toxin (1 μg/ml), toxin B (40 ng/ml), and Y-27632 (25 μM) on changes in CT. Voiding was induced at the arrow. The boxed region in the left-hand panel is expanded in the right-hand panel. Control data are reproduced from Figure 1A. Values are mean±s.e.m. (n⩾5), and those values significantly different than controls (P<0.05) are indicated with an asterisk. (C) Internalization of FITC-WGA or FITC-dextran in C3 toxin-treated cells. (D) Uptake of Alexa647-WGA in rat umbrella cells transduced in situ with GFP and dominant-negative RhoA (transduced cell is marked with an asterisk). (E) Effect of Y-27632 on uptake of FITC-WGA or FITC-dextran after experimental voiding.
Integrin signalling is involved in modulating CE
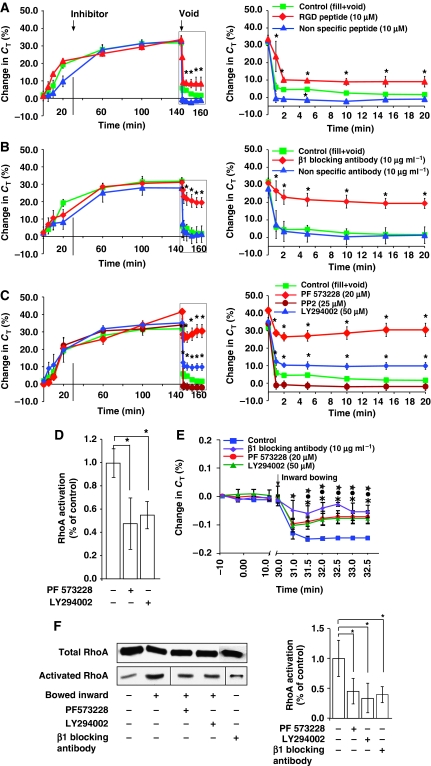
Both filling-induced and voiding-induced CE are coupled to increased tension (i.e. stretch) of the umbrella cell basolateral membrane, but how is unknown (Yu et al, 2009). In other cell types, membrane stretch is known to stimulate integrin activation, which in turn can lead to RhoA activation and changes in cell function (Tzima et al, 2001; Torsoni et al, 2005; Zhang et al, 2007; Lim et al, 2008). As the β1 integrin is prominently localized to the basolateral surface of the umbrella cell and plasma membrane of underlying cell layers (Supplementary Figure S8) (Southgate et al, 1995), we earlier hypothesized that integrins may have a function in basolateral membrane mechanotransduction and thus may modulate filling- and voiding-induced CE (Yu et al, 2009). To explore this possibility, we treated the serosal surface of tissue with RGD peptide (GRGDNP), an integrin receptor substrate mimetic. To selectively examine the effects on voiding-induced CE, peptide was added after the tissue was bowed outwards, which as described above triggers filling-induced CE (Yu et al, 2009). RGD peptide treatment blocked voiding-induced membrane recovery by ∼30% (Figure 7A); whereas control peptide (GRADSP) did not affect voiding-induced CE (Figure 7A). We also treated the serosal surface of the tissue with a β1 integrin-specific function-blocking antibody, which also significantly slowed CE and inhibited the overall drop in CT by ∼55% (Figure 7B). Incubation with control antibody did not significantly alter CE (Figure 7B), and treatment with RGD peptide or neutralizing antibody did not impact exocytosis.
Figure 7.
Function of β1 integrin and associated signalling pathways in voiding-induced CE. (A) Changes in CT for tissue treated with RGD peptide (10 μM) or non-specific peptide (10 μM). (B) Effect of β1-blocking antibody (10 μg/ml) or non-specific antibody (10 μg/ml) on changes in CT. (C) Changes in CT for tissue treated with PF573228 (20 μM), PP2 (25 μM), or LY294002 (50 μM). (A–C) Inhibitor was added at the indicated time. Voiding was induced at the arrow and the boxed region in the left-hand panel is expanded in the right-hand panel. Control data are reproduced from Figure 1A. Values are mean±s.e.m. (n⩾5), and those values significantly different than controls (P<0.05) are indicated with an asterisk. (D) Effect of PF573228 or LY294002 on RhoA activation in intact bladders. Values are mean±s.e.m. (n=3) and those values significantly different than control samples (P<0.05) are indicated with an asterisk. (E) Changes in CT in response to inwards bowing. Tissue was left untreated (control) or treated with blocking antibody, PF573228, or LY294002. Values are mean±s.e.m. (n⩾5), and those values significantly different than control samples (P<0.05) are indicated with an asterisk (PF573228) or a closed circle (LY294002). (F) Left panel: Total and activated RhoA in control tissue or that treated with blocking antibody, PF573228, or LY294002. Right panel: RhoA activation expressed as a per cent of control. Values are mean±s.e.m. (n=6), and those values significantly different than control samples (P<0.05) are indicated with an asterisk.
Integrin activation is transmitted to other cellular organelles by various molecules including FAK, PI-3K, and Src, all of which can alter GTPase function (Schaller, 2001; Mitra et al, 2005; Lim et al, 2008; Tomar et al, 2009). To explore the possible involvement of these kinases in CE, we treated tissue with selective inhibitors including the FAK inhibitor PF573228, the Src inhibitor PP2, or the PI3-K inhibitor LY294002. Treatment with PF573228 or LY294002 significantly inhibited CE by ∼70 and ∼30%, respectively, whereas PP2 did not alter the change in CT (Figure 7C). These treatments did not affect exocytosis. Furthermore, when excised, but otherwise intact bladders were filled, and then treated with PF573228 or LY294002 before voiding, they suppressed RhoA activation (Figure 7D), indicating that they may act upstream of RhoA. However, these experiments are complex to interpret because it is difficult to tease out any effects on filling-induced changes in RhoA activation, and those that occur in response to voiding. To examine the latter response in isolation, we measured CT in tissue that was bowed inwards without exposure to a preceding period of outwards bowing. We earlier showed that inward bowing stimulates apical endocytosis (Yu et al, 2009). In these tissues, β1 integrin-blocking antibody, PF573228, and LY294002 all significantly inhibited endocytosis (Figure 7E). Furthermore, we observed that inwards bowing stimulated activation of RhoA (∼5-fold) and that treatment with blocking antibody, PF573228, or LY294002 caused a significant decrease in RhoA activation (Figure 7F). Taken together, our results indicate that during voiding, integrins may initiate basolateral membrane mechanotransduction and then effect changes in CE by modulating RhoA activity downstream of FAK- and PI3-K.
Discussion
To date, there is little information available about clathrin-independent mechanisms of CE, even though they may have important functions in diverse functions including neurotransmission, secretion, and fertilization (Sokac and Bement, 2006; Clayton et al, 2007). In part, this reflects the fact that these forms of endocytosis can lack specific cargoes, carriers, inhibitors, or machineries (Mayor and Pagano, 2007; Sandvig et al, 2008; Hansen and Nichols, 2009). Furthermore, their physiological relevance is unclear, in part, because many have been defined in cultured cells (Mayor and Pagano, 2007), some are only revealed when other forms of endocytosis are inhibited (Damke et al, 1994), and in the case of CE, they are often observed only after massive stimulation of exocytosis (Holt et al, 2003; Clayton et al, 2008). The umbrella cell model is ideal for these studies as the apical membrane of differentiated umbrella cells lacks coated pits, caveolae, or flotillin-positive carriers, and CE in these cells is apparently a pathway for bulk retrieval of apical membrane and is unlikely to require multiple mechanisms for cargo-selective endocytosis.
Our data further show that voiding stimulates the rapid uptake of apical membrane into apical endosomes, which we term as PJAEs, that deliver their contents to late endosomes and lysosomes without apparent transit through an EEA1-positive early endosome compartment. Furthermore, we find little evidence of recycling of endocytosed apical membrane components into DFV for reuse on subsequent bladder filling. Instead, the internalized membrane components seem fated for degradation, although some recycling from PJAEs cannot be ruled out. PJAEs are associated with ZO-1, a protein that can interact with the actin cytoskeleton, which promotes delivery of endosomal cargo to lysosomes (Durrbach et al, 1996). In the upper urinary tract, kidney proximal tubule cells also deliver apically internalized membrane and fluid components to lysosomes (Christensen and Gburek, 2004); however, endocytosis in these cells is by a constitutive clathrin-dependent process, is not directly coupled to exocytosis, and is required for the selective retrieval of molecules from the glomerular filtrate. In contrast, our data indicate that CE in umbrella cells is a clathrin-independent mechanism for apical membrane recovery that is triggered by signal propagation downstream of basolateral membrane stretch.
In kidney epithelial cells, endothelial cells, and cardiac myocytes, integrins are thought to be important players in sensing membrane stretch and can modulate RhoA activation (Tzima et al, 2001; Torsoni et al, 2005; Zhang et al, 2007; Lim et al, 2008). Furthermore, integrins may impact endocytosis in other cell systems (del Pozo et al, 2004, 2005; Singh et al, 2010). However, a function for integrins in CE and RhoA-regulated internalization pathways has not been explored. Intriguingly, we observed that RGD peptide and neutralizing antibodies to β1 integrin impaired CE in umbrella cells. Integrin-dependent activation of RhoA can be mediated by Src, FAK, or PI-3K, which functions by inhibiting p190RhoGAP or stimulating p190RhoGEF (Mitra et al, 2005; Lim et al, 2008; Tomar et al, 2009). In our studies, we find that FAK and PI-3K, but not Src, are important for CE and that inhibition of FAK or PI-3K impairs activation of RhoA. Intriguingly, bulk endocytosis in bipolar neurons is also dependent on PI-3K (Holt et al, 2003); however a function for integrins or RhoA in this pathway is unknown. Voltage-gated Ca2+ channels (Smith et al, 2000; Rosa et al, 2007; Wu et al, 2007; Yao et al, 2009), granule membrane proteins and lipids (Cremona et al, 1999; Gundelfinger et al, 2003; Poskanzer et al, 2003; Deak et al, 2004), post-translational modifications of proteins (Engisch and Nowycky, 1998; Henkel et al, 2001, 2004; Graham et al, 2002; Clayton et al, 2007), and actin (Valentijn et al, 2000; Sokac et al, 2003; Nemoto et al, 2004; Yu and Bement, 2007) are known to be important regulators of CE. Our data indicate that integrin signalling also has a function in initiating or promoting CE, possibly by stimulating RhoA activation in response to voiding.
CE in umbrella cell is dependent on actin, cholesterol, dynamin, RhoA, and possibly ROCK. Although a similar pathway has been described for the endocytosis of the IL2 receptor β chain (Lamaze et al, 2001), the pathways may not be identical as the IL2 receptor pathway is constitutive (Lamaze et al, 2001), dependent on Rac1 (and associated Pak1/Pak2 kinases) (Grassart et al, 2008), and is not known to be integrin regulated. Although a RhoA-regulated CE pathway is unlikely to be the sole mechanism of clathrin-independent endocytosis in cells with a regulated secretory pathway, a requirement for the cytoskeleton, molecules that promote fission, and Rho-family GTPases may be common to multiple forms of clathrin-independent CE. For example, in oocytes, an actin coat surrounds the cortical granules and myosin-dependent and Cdc42-regulated compression of the coat may promote vesicle scission (Bement et al, 2000; Sokac et al, 2003; Yu and Bement, 2007). Similar mechanisms could be important in other cell types such as pancreatic acinar cells and alveolar cells, which also form actin coats around their secretory granules (Nemoto et al, 2004; van Weeren et al, 2004).
Dynamin is a common requirement for other forms of clathrin-independent CE (Artalejo et al, 2002; Jockusch et al, 2005) and may have an important function in vesicle scission. However, dynamin could also modulate CE by altering actin dynamics, as dynamin interacts with cortactin as well as other scaffolding proteins (e.g. syndapin and intersectin) that link dynamin to the Arp2/3-regulatory protein N-WASP (Schafer, 2004). Even though DFV are apparently not endocytic in nature (Khandelwal et al, 2008, 2009), dynamin is associated with their cytoplasmic surface (Terada et al, 2009). This interaction may occur before DFV fusion, likely as a prelude to dynamin-dependent CE. This would be similar to the reported association of actin, clathrin, and dynamin with post-Golgi exocytic carriers before kiss-and-run fusion (Jaiswal et al, 2009). Interestingly, we observed that the sites of PJAEs were at the cell periphery, but dynamin was distributed under the whole of the apical surface. One possible explanation is that endocytic carriers, possibly PJAEs, form homogenously under the apical membrane, and subsequently accumulate near the junctions.
In summary, we show that clathrin-independent CE is critically important in the recovery of umbrella cell apical membrane after voiding. The physiological stimulus for this event is increased basolateral tension, which through or in conjunction with β1 integrin stimulates PI-3K- and FAK-dependent activation of RhoA. In turn, RhoA recruits ROCK, which may act by promoting alterations in the actin cytoskeleton that in conjunction with myosin motors could help to pull apical membranes inwards as well as generate tension in these membranes as a preamble to scission. In other forms of endocytosis BAR-domain containing proteins, which may act by sensing or inducing membrane curvature, are thought to be important in the formation endocytic pits (Yarar et al, 2007; Lundmark et al, 2008). A particularly intriguing example is the protein GRAF1, which has a BAR domain, recruits dynamin, and is a GTPase-activating protein for RhoA (Lundmark et al, 2008). Thus, GRAF1, or a protein similar to it, may help to promote CE in umbrella cells by forming/sculpting apical membrane into endocytic pits, recruiting and/or modulating dynamin-dependent scission, and promoting termination of the endocytic reaction by stimulating RhoA GTPase hydrolysis. The internalized apical membrane would then be captured within or delivered to PJAEs, and then may be recycled or transferred to lysosomes in which it is degraded.
Materials and methods
Reagents and antibodies
Unless specified otherwise, all chemicals, toxins, and proteins were obtained from Sigma-Aldrich (St Louis, MO). Mouse monoclonal antibodies against LAMP2 (AC17), Rab11a (8H10), UP3a (K8B12), and rat anti-ZO-1 (R40.76) were described earlier (Anderson et al, 1988; Nabi et al, 1991; Truschel et al, 1999; Lapierre et al, 2003). Other antibodies used in these studies include mouse monoclonal antibodies specific for caveolin-1 and -2, dynamin-2, EEA1, and flotillin-1 (all from BD Biosciences; San Jose, CA), mouse monoclonal anti-giantin (provided by Dr Adam Linstedt, Carnegie Mellon University), and mouse monoclonal anti-AP2 α subunit antibody AP.6 and anti-clathrin heavy chain (provided by Dr Linton Traub, University of Pittsburgh). Similar results were obtained with monoclonal antibodies to caveolin-2 obtained from Novus Biologicals (Littleton, CO) and Cell Signaling Technology (Danvers, MA). Fluorophore- or HRP-conjugated, affinity-purified, and minimal cross-reacting goat anti-mouse or -rat antibodies were purchased from Jackson ImmunoResearch Laboratories (West Grove, PA). FITC-, HRP-, and Agarose-bound WGA were obtained from Vector laboratories (Burlingame, CA) and Alexa488- and Alexa647-WGA were obtained from Invitrogen (Carlsbad, CA). Rhodamine phalloidin and TO-PRO3 were obtained from Molecular Probes-Invitrogen. Cell permeable C3 transferase was obtained from Cytoskeleton Inc. (Denver, CO).
Animals
Urinary bladders were obtained from female New Zealand White Rabbits (3–4 kg), or female Sprague–Dawley rats (250–300 g). Rabbits were euthanized by injecting 300 mg of Nembutal into their ear vein, and rats were euthanized by inhalation of 100% CO2. All animal studies were approved by the University of Pittsburgh Institutional Animal Care and Use Committee.
Mounting of uroepithelium, simulation of filling and voiding, and measurement of CT
Rabbit bladders were excised and the uroepithelium dissected away from the underlying musculature and mounted in Ussing stretch chambers as described earlier (Wang et al, 2003; Yu et al, 2009). Each bladder yielded 3–4 preparations. To simulate bladder filling, Krebs buffer was added to the mucosal hemichamber at 100 μl/min using a NE-1600 pump (New Era Pump Systems, NY), filling the chamber to its capacity and bowing the tissue outwards. The chamber was then sealed, and an additional 0.5 ml of Krebs solution was infused at the same filling rate. To experimentally mimic bladder voiding, the sealed mucosal hemichamber luer ports were opened, and excess Krebs buffer was removed with a 20-ml syringe fitted with an 18-gauge needle until the tissue bowed inwards. The change in CT was measured as described earlier (Yu et al, 2009). The experiments were repeated several times (n⩾4), using separate rabbit bladders for each treatment group. Data are derived from multiple tissue preparations, which were obtained from multiple animals.
Internalization of endocytic markers in rabbit uroepithelial tissue and rat bladders
To internalize markers in rabbit uroepithelial tissue, the mucosal hemichamber was filled for 140 min, and then FITC- or Alexa488/647-WGA (25 μg/ml) or FITC-dextran (2.5 mg/ml) was isovolumetrically added to the mucosal chamber for 10 min. Voiding was subsequently induced as described above. In some experiments, tracer was added after voiding. When indicated, 40 μM leupeptin was added to the mucosal and serosal hemichambers just after voiding. For experiments that measured uptake of FITC-WGA, the labelled tissue was placed in ice-cold buffer and then washed three times 30 min on a shaker at 4°C with 50 mM N-acetyl glucosamine to remove cell surface-bound lectin, and then fixed with 4% paraformaldehyde in 100 mM cacodylate buffer. On FITC-dextran internalization, the tissue was washed three times 5 min with Krebs buffer and then fixed in periodate-lysine-paraformaldehyde fixative (Brown and Farquhar, 1989). Fixed tissues were immunostained, mounted, and imaged as described below. Experiments exploring uptake of cationized ferritin are described in Supplementary data.
Uptake of endocytic markers in adenovirus-transduced rat bladders
Rat bladders were transduced in situ with adenoviruses expressing GFP-DN-dynaminK44A (kindly provided by Dr Ora Weisz, University of Pittsburgh), or GFP, cdc42N17, Rac1N17, Arf6T27N, or RhoAN19 (kindly provided by Dr James Bamburg, Colorodo State University, and Dr James Casanova, University of Virginia) as described earlier (Khandelwal et al, 2008). Before filling the bladders, concentrated endocytic tracer was taken up in a syringe, which was then carefully filled with Krebs buffer to limit mixing with the tracer. This protocol insured that endocytic tracer was not delivered until the majority of buffer was expelled from the syringe. Bladders were catheterized with a Jelco 22-gauge IV catheter trimmed to ∼1 cm in length, the buffer and tracer were then instilled into the bladder over a period of 3–4 min using a syringe pump, and the bladder was left filled for 30 min. Subsequently, the fluid was allowed to void. The bladder was rapidly excised and then washed, fixed, immunostained, and images were acquired and data analysed as described below.
Acquisition of images and image analyses
Tissue was immunostained and confocal images of random regions of tissue were acquired by confocal microscopy as described earlier (Khandelwal et al, 2008). The collection parameters were set so that all pixel values fell within the 8-bit range of the image files. 3D reconstructions of the individual Z-series were generated using Volocity software (Perkin Elmers; Waltham, MA). Before quantitation, images were processed in Volocity using a ‘fine' (3 × 3 median) noise reduction filter. Methods for estimation of the amount of apical membrane internalized and for the number of endocytic objects in tissue samples are provided in Supplementary data.
Mander correlation coefficients were obtained using Volocity as described earlier (Khandelwal et al, 2008). The analysis was performed multiple times (n⩾3), using separate bladders for each treatment group. Multiple image stacks (⩾7), derived from multiple tissue preparations and animals, were used in the analysis. The total mean intensity of endocytic objects in rabbit tissue was determined as follows. Image stacks were projected into a single image using the ‘brightest point' option in Volocity and then exported TIFF files opened in Image J (http://rsbweb.nih.gov/ij). Objects with fixed thresholds >45 were chosen using the ‘analyse parameter' function. The thresholded objects in control tissue had intensity values in the range of ∼120–130 (signal-to-noise ratio of ∼3.0). The ‘object area' and ‘mean intensity for each object' were multiplied, and the resulting values (mean intensity x μm2) exported to Excel (Microsoft Inc.; Redmond, WA) and then summed to yield values for total mean intensity x μm2. As assigning PJAES to individual umbrella cells was difficult, the latter value was divided by the calibrated area of the projected image (∼7000–10 000 μm2), which yielded the total mean intensity for the sampled image stack. These experiments were performed at least three times, and at least 10 image stacks, obtained from multiple preparations and animals, were analysed per condition.
To measure mean fluorescent intensity of endocytosed tracer in rat umbrella cells expressing virally expressed proteins (or adjacent cells not expressing these proteins), individual umbrella cell boundary areas were selected using the freehand tool in Volocity. The ‘crop selection' command was executed, the resulting image was selected, fluorescent objects with fixed threshold values >45 were chosen, and the measure object command was used to measure the mean fluorescent intensity of the thresholded objects in the selected cell (control values of ∼160–190; signal-to-noise ratio of ∼3.5–4.2). The total mean fluorescent intensity for all objects was determined by summing the individual values in Excel. Data are reported as a ratio of total mean fluorescent intensity of endocytic tracer in cells expressing dominant-negative protein versus cells in the field that did not. For each condition, the experiments were repeated multiple times (n⩾6), each time using a different rat bladder, and data obtained for at least 70 transduced and non-transduced cells were reported as mean±s.e.m.
Binding of WGA to UP3a, RT–PCR, analysis of plasma membrane-associated clathrin-coated pits and flask-shaped invaginations, isolation of detergent-resistant membranes, and RhoA activation assays
Methods for these techniques can be found in Supplementary data.
Statistical analysis
Data are reported as mean±s.e.m. Statistically significant differences between means were determined using a two-tailed Student's t-test; P<0.05 was considered statistically significant. One-way ANOVA, with Bonferonni's correction, was used when making multiple comparisons.
Supplementary Material
Acknowledgments
We thank Dr Adam Linstedt for his invaluable assistance in quantifying the endocytosis data and the thoughtful comments and suggestions of Dr Linstedt, Dr Shalini Mitra, and Dr Linton Traub. We also thank Drs Adam Linstedt, Mark McNiven, Linton Traub, Ora Weisz, and James Bamburg for their kind gifts of reagents. This work was supported by an AHA Postdoctoral Fellowship (to PK), NIH grants R37-DK54425 and RO1-DK077777 (to GA), and the Urinary Tract Epithelial Imaging Core of the Pittsburgh Center for Kidney Research (P30DK079307).
Footnotes
The authors declare that they have no conflict of interest.
References
- Alenghat FJ, Nauli SM, Kolb R, Zhou J, Ingber DE (2004) Global cytoskeletal control of mechanotransduction in kidney epithelial cells. Exp Cell Res 301: 23–30 [DOI] [PubMed] [Google Scholar]
- Amano O, Kataoka S, Yamamoto T (1991) Turnover of asymmetric unit membranes in the transitional epithelial superficial cells of the rat urinary bladder. Anatom Record 229: 9–15 [DOI] [PubMed] [Google Scholar]
- Anderson JM, Stevenson BR, Jesaitis LA, Goodenough DA, Mooseker MS (1988) Characterization of ZO-1, a protein component of the tight junction from mouse liver and Madin-Darby canine kidney cells. J Cell Biol 106: 1141–1149 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Artalejo CR, Elhamdani A, Palfrey HC (2002) Sustained stimulation shifts the mechanism of endocytosis from dynamin-1-dependent rapid endocytosis to clathrin- and dynamin-2-mediated slow endocytosis in chromaffin cells. Proc Natl Acad Sci USA 99: 6358–6363 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Balestreire EM, Apodaca G (2007) Apical EGF receptor signaling: regulation of stretch-dependent exocytosis in bladder umbrella cells. Mol Biol Cell 18: 1312–1323 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barg S, Machado JD (2008) Compensatory endocytosis in chromaffin cells. Acta Physiol (Oxf) 192: 195–201 [DOI] [PubMed] [Google Scholar]
- Barresi V, Grosso M, Bulfamante G, Vitarelli E, Ghioni MC, Barresi G (2006) Caveolin-1 immuno-expression in human fetal tissues during mid and late gestation. Eur J Histochem 50: 183–190 [PubMed] [Google Scholar]
- Bement WM, Benink H, Mandato CA, Swelstad BB (2000) Evidence for direct membrane retrieval following cortical granule exocytosis in Xenopus oocytes and eggs. J Exp Zool 286: 767–775 [DOI] [PubMed] [Google Scholar]
- Born M, Pahner I, Ahnert-Hilger G, Jons T (2003) The maintenance of the permeability barrier of bladder facet cells requires a continuous fusion of discoid vesicles with the apical plasma membrane. Eur J Cell Biol 82: 343–350 [DOI] [PubMed] [Google Scholar]
- Brown WJ, Farquhar MG (1989) Immunoperoxidase methods for the localization of antigens in cultured cells and tissue sections by electron microscopy. In Methods in Cell Biology, Alan M Tartakoff (ed), Vol. 31, pp 553–569. Palo Alto, CA: Academic Press [DOI] [PubMed] [Google Scholar]
- Chadda R, Howes MT, Plowman SJ, Hancock JF, Parton RG, Mayor S (2007) Cholesterol-sensitive Cdc42 activation regulates actin polymerization for endocytosis via the GEEC pathway. Traffic 8: 702–717 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Christensen EI, Gburek J (2004) Protein reabsorption in renal proximal tubule-function and dysfunction in kidney pathophysiology. Pediatr Nephrol 19: 714–721 [DOI] [PubMed] [Google Scholar]
- Clayton EL, Evans GJ, Cousin MA (2007) Activity-dependent control of bulk endocytosis by protein dephosphorylation in central nerve terminals. J Physiol 585: 687–691 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clayton EL, Evans GJ, Cousin MA (2008) Bulk synaptic vesicle endocytosis is rapidly triggered during strong stimulation. J Neurosci 28: 6627–6632 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cremona O, Di Paolo G, Wenk MR, Luthi A, Kim WT, Takei K, Daniell L, Nemoto Y, Shears SB, Flavell RA, McCormick DA, De Camilli P (1999) Essential role of phosphoinositide metabolism in synaptic vesicle recycling. Cell 99: 179–188 [DOI] [PubMed] [Google Scholar]
- Damke H, Baba T, Warnock DE, Schmid SL (1994) Induction of mutant dynamin specifically blocks endocytic coated vesicle formation. J Cell Biol 127: 915–934 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deak F, Schoch S, Liu X, Sudhof TC, Kavalali ET (2004) Synaptobrevin is essential for fast synaptic-vesicle endocytosis. Nat Cell Biol 6: 1102–1108 [DOI] [PubMed] [Google Scholar]
- del Pozo MA, Alderson NB, Kiosses WB, Chiang HH, Anderson RG, Schwartz MA (2004) Integrins regulate Rac targeting by internalization of membrane domains. Science 303: 839–842 [DOI] [PubMed] [Google Scholar]
- del Pozo MA, Balasubramanian N, Alderson NB, Kiosses WB, Grande-Garcia A, Anderson RG, Schwartz MA (2005) Phospho-caveolin-1 mediates integrin-regulated membrane domain internalization. Nat Cell Biol 7: 901–908 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Durrbach A, Louvard D, Coudrier E (1996) Actin filaments facilitate two steps of endocytosis. J Cell Sci 109: 457–465 [DOI] [PubMed] [Google Scholar]
- Engisch KL, Nowycky MC (1998) Compensatory and excess retrieval: two types of endocytosis following single step depolarizations in bovine adrenal chromaffin cells. J Physiol 506 (Pt 3): 591–608 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fong A, Garcia E, Gwynn L, Lisanti MP, Fazzari MJ, Li M (2003) Expression of caveolin-1 and caveolin-2 in urothelial carcinoma of the urinary bladder correlates with tumor grade and squamous differentiation. Am J Clin Pathol 120: 93–100 [DOI] [PubMed] [Google Scholar]
- Frick M, Bright NA, Riento K, Bray A, Merrified C, Nichols BJ (2007) Coassembly of flotillins induces formation of membrane microdomains, membrane curvature, and vesicle budding. Curr Biol 17: 1151–1156 [DOI] [PubMed] [Google Scholar]
- Gong Q, Huntsman C, Ma D (2008) Clathrin-independent internalization and recycling. J Cell Mol Med 12: 126–144 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Graham ME, O'Callaghan DW, McMahon HT, Burgoyne RD (2002) Dynamin-dependent and dynamin-independent processes contribute to the regulation of single vesicle release kinetics and quantal size. Proc Natl Acad Sci USA 99: 7124–7129 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grassart A, Dujeancourt A, Lazarow PB, Dautry-Varsat A, Sauvonnet N (2008) Clathrin-independent endocytosis used by the IL-2 receptor is regulated by Rac1, Pak1 and Pak2. EMBO Rep 9: 356–362 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gundelfinger ED, Kessels MM, Qualmann B (2003) Temporal and spatial coordination of exocytosis and endocytosis. Nat Rev Mol Cell Biol 4: 127–139 [DOI] [PubMed] [Google Scholar]
- Hansen CG, Nichols BJ (2009) Molecular mechanisms of clathrin-independent endocytosis. J Cell Sci 122: 1713–1721 [DOI] [PMC free article] [PubMed] [Google Scholar]
- He L, Wu LG (2007) The debate on the kiss-and-run fusion at synapses. Trends Neurosci 30: 447–455 [DOI] [PubMed] [Google Scholar]
- Henkel AW, Kang G, Kornhuber J (2001) A common molecular machinery for exocytosis and the ‘kiss-and-run' mechanism in chromaffin cells is controlled by phosphorylation. J Cell Sci 114: 4613–4620 [DOI] [PubMed] [Google Scholar]
- Henkel AW, Vogt TK, Henkel MK (2004) Staurosporine restores GTPgammaS induced block of rapid endocytosis in chromaffin cells. FEBS Lett 571: 103–106 [DOI] [PubMed] [Google Scholar]
- Hicks M, Ketterer B, Warren R (1974) The ultrastructure and chemistry of the luminal plasma membrane of the mammalian urinary bladder: a structure with low permeability to water and ions. Phil Trans Royal Soc London—Series B: Biol Sci 268: 23–38 [DOI] [PubMed] [Google Scholar]
- Holt M, Cooke A, Wu MM, Lagnado L (2003) Bulk membrane retrieval in the synaptic terminal of retinal bipolar cells. J Neurosci 23: 1329–1339 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ivanov AI (2008) Pharmacological inhibition of endocytic pathways: is it specific enough to be useful? Methods Mol Biol 440: 15–33 [DOI] [PubMed] [Google Scholar]
- Jaiswal JK, Rivera VM, Simon SM (2009) Exocytosis of post-Golgi vesicles is regulated by components of the endocytic machinery. Cell 137: 1308–1319 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jockusch WJ, Praefcke GJ, McMahon HT, Lagnado L (2005) Clathrin-dependent and clathrin-independent retrieval of synaptic vesicles in retinal bipolar cells. Neuron 46: 869–878 [DOI] [PubMed] [Google Scholar]
- Kerr DE, Liang F, Bondioli KR, Zhao H, Kreibich G, Wall RJ, Sun TT (1998) The bladder as a bioreactor: urothelium production and secretion of growth hormone into urine. Nat Biotechnol 16: 75–79 [DOI] [PubMed] [Google Scholar]
- Khandelwal P, Abraham SN, Apodaca G (2009) Cell biology and physiology of the uroepithelium. Am J Physiol Renal Physiol 297: F1477–F1501 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khandelwal P, Ruiz G, Balestreire-Hawryluk E, Weisz OA, Goldenring JA, Apodaca G (2008) Rab11a-dependent exocytosis of discoidal/fusiform vesicles in bladder umbrella cells. Proc Natl Acad Sci USA 105: 15773–15778 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kreft ME, Romih R, Kreft M, Jezernik K (2009) Endocytotic activity of bladder superficial urothelial cells is inversely related to their differentiation stage. Differentiation 77: 48–59 [DOI] [PubMed] [Google Scholar]
- Lamaze C, Dujeancourt A, Baba T, Lo CG, Benmerah A, Dautry-Varsat A (2001) Interleukin 2 receptors and detergent-resistant membrane domains define a clathrin-independent endocytic pathway. Mol Cell 7: 661–671 [DOI] [PubMed] [Google Scholar]
- Lapierre LA, Dorn MC, Zimmerman CF, Navarre J, Burnette JO, Goldenring JR (2003) Rab11b resides in a vesicular compartment distinct from Rab11a in parietal cells and other epithelial cells. Exp Cell Res 290: 322–331 [DOI] [PubMed] [Google Scholar]
- Lewis S, de Moura J (1982) Incorporation of cytoplasmic vesicles into apical membrane of mammalian urinary bladder epithelium. Nature 297: 685–688 [DOI] [PubMed] [Google Scholar]
- Lim Y, Lim ST, Tomar A, Gardel M, Bernard-Trifilo JA, Chen XL, Uryu SA, Canete-Soler R, Zhai J, Lin H, Schlaepfer WW, Nalbant P, Bokoch G, Ilic D, Waterman-Storer C, Schlaepfer DD (2008) PyK2 and FAK connections to p190Rho guanine nucleotide exchange factor regulate RhoA activity, focal adhesion formation, and cell motility. J Cell Biol 180: 187–203 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lundmark R, Doherty GJ, Howes MT, Cortese K, Vallis Y, Parton RG, McMahon HT (2008) The GTPase-activating protein GRAF1 regulates the CLIC/GEEC endocytic pathway. Curr Biol 18: 1802–1808 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Macia E, Ehrlich M, Massol R, Boucrot E, Brunner C, Kirchhausen T (2006) Dynasore, a cell-permeable inhibitor of dynamin. Dev Cell 10: 839–850 [DOI] [PubMed] [Google Scholar]
- Mayor S, Pagano RE (2007) Pathways of clathrin-independent endocytosis. Nat Rev Mol Cell Biol 8: 603–612 [DOI] [PMC free article] [PubMed] [Google Scholar]
- McNiven MA (2006) Big gulps: specialized membrane domains for rapid receptor-mediated endocytosis. Trends Cell Biol 16: 487–492 [DOI] [PubMed] [Google Scholar]
- Mitra SK, Hanson DA, Schlaepfer DD (2005) Focal adhesion kinase: in command and control of cell motility. Nat Rev Mol Cell Biol 6: 56–68 [DOI] [PubMed] [Google Scholar]
- Nabi IR, Le Bivic A, Fambrough D, Rodriguez-Boulan E (1991) An endogenous MDCK lysosomal membrane glycoprotein is targeted basolaterally before delivery to lysosomes. J Cell Biol 115: 1573–1584 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Naslavsky N, Weigert R, Donaldson JG (2004) Characterization of a nonclathrin endocytic pathway: membrane cargo and lipid requirements. Mol Biol Cell 15: 3542–3552 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nemoto T, Kojima T, Oshima A, Bito H, Kasai H (2004) Stabilization of exocytosis by dynamic F-actin coating of zymogen granules in pancreatic acini. J Biol Chem 279: 37544–37550 [DOI] [PubMed] [Google Scholar]
- Poskanzer KE, Marek KW, Sweeney ST, Davis GW (2003) Synaptotagmin I is necessary for compensatory synaptic vesicle endocytosis in vivo. Nature 426: 559–563 [DOI] [PubMed] [Google Scholar]
- Rosa JM, de Diego AM, Gandia L, Garcia AG (2007) L-type calcium channels are preferentially coupled to endocytosis in bovine chromaffin cells. Biochem Biophys Res Commun 357: 834–839 [DOI] [PubMed] [Google Scholar]
- Sabharanjak S, Sharma P, Parton RG, Mayor S (2002) GPI-anchored proteins are delivered to recycling endosomes via a distinct cdc42-regulated, clathrin-independent pinocytic pathway. Dev Cell 2: 411–423 [DOI] [PubMed] [Google Scholar]
- Sandvig K, Torgersen ML, Raa HA, van Deurs B (2008) Clathrin-independent endocytosis: from nonexisting to an extreme degree of complexity. Histochem Cell Biol 129: 267–276 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schafer DA (2004) Regulating actin dynamics at membranes: a focus on dynamin. Traffic 5: 463–469 [DOI] [PubMed] [Google Scholar]
- Schaller MD (2001) Biochemical signals and biological responses elicited by the focal adhesion kinase. Biochim Biophys Acta 1540: 1–21 [DOI] [PubMed] [Google Scholar]
- Singh RD, Marks DL, Holicky EL, Wheatley CL, Kaptzan T, Sato SB, Kobayashi T, Ling K, Pagano RE (2010) Gangliosides and beta1-integrin are required for caveolae and membrane domains. Traffic 11: 348–360 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith RM, Bailbakov B, Ikebuchi Y, White BH, Lambert NA (2000) Exocytotic insertion of calcium channels constrains compensatory endocytosis to sites of exocytosis. J Cell Bio 148: 755–767 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sokac AM, Bement WM (2006) Kiss-and-coat and compartment mixing: coupling exocytosis to signal generation and local actin assembly. Mol Biol Cell 17: 1495–1502 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sokac AM, Co C, Taunton J, Bement W (2003) Cdc42-dependent actin polymerization during compensatory endocytosis in Xenopus eggs. Nat Cell Biol 5: 727–732 [DOI] [PubMed] [Google Scholar]
- Southgate J, Kennedy W, Hutton KA, Trejdosiewicz LK (1995) Expression and in vitro regulation of integrins by normal human urothelial cells. Cell Adhesion Commun 3: 231–242 [DOI] [PubMed] [Google Scholar]
- Terada N, Ohno N, Saitoh S, Saitoh Y, Fujii Y, Kondo T, Katoh R, Chan C, Abraham SN, Ohno S (2009) Involvement of dynamin-2 in formation of discoid vesicles in urinary bladder umbrella cells. Cell Tissue Res 337: 91–102 [DOI] [PubMed] [Google Scholar]
- Tomar A, Lim ST, Lim Y, Schlaepfer DD (2009) A FAK-p120RasGAP-p190RhoGAP complex regulates polarity in migrating cells. J Cell Sci 122: 1852–1862 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torsoni AS, Marin TM, Velloso LA, Franchini KG (2005) RhoA/ROCK signaling is critical to FAK activation by cyclic stretch in cardiac myocytes. Am J Physiol Heart Circ Physiol 289: H1488–H1496 [DOI] [PubMed] [Google Scholar]
- Traub LM (2009) Tickets to ride: selecting cargo for clathrin-regulated internalization. Nat Rev Mol Cell Biol 10: 583–596 [DOI] [PubMed] [Google Scholar]
- Truschel ST, Ruiz WG, Shulman T, Pilewski J, Sun T-T, Zeidel ML, Apodaca G (1999) Primary uroepithelial cultures: a model system to analyze umbrella cell barrier function. J Biol Chem 274: 15020–15029 [DOI] [PubMed] [Google Scholar]
- Truschel ST, Wang E, Ruiz WG, Leung SM, Rojas R, Lavelle J, Zeidel M, Stoffer D, Apodaca G (2002) Stretch-regulated exocytosis/endocytosis in bladder umbrella cells. Mol Biol Cell 13: 830–846 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tzima E, del Pozo MA, Shattil SJ, Chien S, Schwartz MA (2001) Activation of integrins in endothelial cells by fluid shear stress mediates Rho-dependent cytoskeletal alignment. EMBO J 20: 4639–4647 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valentijn JA, Valentijn K, Pastore LM, Jamieson JD (2000) Actin coating of secretory granules during regulated exocytosis correlates with the release of rab3D. Proc Natl Acad Sci USA 97: 1091–1095 [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Weeren L, de Graaff AM, Jamieson JD, Batenburg JJ, Valentijn JA (2004) Rab3D and actin reveal distinct lamellar body subpopulations in alveolar epithelial type II cells. Am J Respir Cell Mol Biol 30: 288–295 [DOI] [PubMed] [Google Scholar]
- Wang E, Truschel ST, Apodaca G (2003) Analysis of hydrostatic pressure-induced changes in umbrella cell surface area. Methods 30: 207–217 [DOI] [PubMed] [Google Scholar]
- Wu E-R, Manabe M, Yu J, Sun T-T (1990) Large scale purification and immunolocalization of bovine uroplakins I, II and III. J Biol Chem 265: 19170–19179 [PubMed] [Google Scholar]
- Wu LG, Ryan TA, Lagnado L (2007) Modes of vesicle retrieval at ribbon synapses, calyx-type synapses, and small central synapses. J Neurosci 27: 11793–11802 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu XR, Kong XP, Pellicer A, Kreibich G, Sun TT (2009) Uroplakins in urothelial biology, function, and disease. Kidney Int 75: 1153–1165 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu J, McNeil B, Wu W, Nees D, Bai L, Wu LG (2008) GTP-independent rapid and slow endocytosis at a central synapse. Nat Neurosci 11: 45–53 [DOI] [PubMed] [Google Scholar]
- Yao CK, Lin YQ, Ly CV, Ohyama T, Haueter CM, Moiseenkova-Bell VY, Wensel TG, Bellen HJ (2009) A synaptic vesicle-associated Ca2+ channel promotes endocytosis and couples exocytosis to endocytosis. Cell 138: 947–960 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yarar D, Waterman-Storer CM, Schmid SL (2007) SNX9 couples actin assembly to phosphoinositide signals and is required for membrane remodeling during endocytosis. Dev Cell 13: 43–56 [DOI] [PubMed] [Google Scholar]
- Yu HY, Bement WM (2007) Multiple myosins are required to coordinate actin assembly with coat compression during compensatory endocytosis. Mol Biol Cell 18: 4096–4105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu W, Khandelwal P, Apodaca G (2009) Distinct apical and basolateral membrane requirements for stretch-induced membrane traffic at the apical surface of bladder umbrella cells. Mol Biol Cell 20: 282–295 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang SJ, Truskey GA, Kraus WE (2007) Effect of cyclic stretch on beta1D-integrin expression and activation of FAK and RhoA. Am J Physiol Cell Physiol 292: C2057–C2069 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.