Abstract
PQQ (pyrroloquinoline quinone) improves energy utilization and reproductive performance when added to rodent diets devoid of PQQ. In the present paper we describe changes in gene expression patterns and transcriptional networks that respond to dietary PQQ restriction or pharmacological administration. Rats were fed diets either deficient in PQQ (PQQ−) or supplemented with PQQ (approx. 6 nmol of PQQ/g of food; PQQ+). In addition, groups of rats were either repleted by administering PQQ to PQQ− rats (1.5 mg of PQQ intraperitoneal/kg of body weight at 12 h intervals for 36 h; PQQ−/+) or partially depleted by feeding the PQQ− diet to PQQ+ rats for 48 h (PQQ+/−). RNA extracted from liver and a Codelink® UniSet Rat I Bioarray system were used to assess gene transcript expression. Of the approx. 10000 rat sequences and control probes analysed, 238 were altered at the P<0.01 level by feeding on the PQQ− diet for 10 weeks. Short-term PQQ depletion resulted in changes in 438 transcripts (P<0.01). PQQ repletion reversed the changes in transcript expression caused by PQQ deficiency and resulted in an alteration of 847 of the total transcripts examined (P<0.01). Genes important for cellular stress (e.g. thioredoxin), mitochondriogenesis, cell signalling [JAK (Janus kinase)/STAT (signal transducer and activator of transcription) and MAPK (mitogen-activated protein kinase) pathways] and transport were most affected. qRT-PCR (quantitative real-time PCR) and functional assays aided in validating such processes as principal targets. Collectively, the results provide a mechanistic basis for previous functional observations associated with PQQ deficiency or PQQ administered in pharmacological amounts.
Keywords: gene microarray, Janus kinase/signal transducer and activator of transcription signalling pathway (JAK/STAT signalling pathway), mitogen-activated protein kinase signalling pathway (MAPK signalling pathway), mitochondrion, oxidative metabolism, pyrroloquinoline quinone (PQQ)
Abbreviations: AMPK, AMP-activated protein kinase; ASD, amino-acid-semi-purified-based diet; CF, cystic fibrosis gene; CREB, cAMP-response-element-binding protein; ERK, extracellular-signal-regulated kinase; FDR, false discovery rate; GAPDH, glyceraldehyde-3-phosphate dehydrogenase; GDH, glucose dehydrogenase; HMG-CoA, 3-hydroxy-3-methylglutaryl-CoA; IPA, ingenuity pathways analysis; IPQ, imidazolopyrroloquinoline; JAK, Janus kinase; LC, laboratory chow; MAPK, mitogen-activated protein kinase; MAPKKK, MAPK kinase kinase; mtDNA, mitochondrial DNA; ND-5, nicotinamide adenine dinucleotide dehydrogenase-5; NPY, neuropeptide Y; NRF, nuclear respiratory factor; PGC-1α, peroxisome-proliferator-activated receptor γ co-activator-1α; PQQ, pyrroloquinoline quinone; qRT-PCR, quantitative real-time PCR; SAM, significance analysis of microarrays; STAT, signal transducer and activator of transcription; Tfam, transcription factor A, mitochondrial
INTRODUCTION
A number of physiological properties have been attributed to PQQ (pyrroloquinoline quinone) ranging from classical vitamin/cofactor functions to those important for anti- and pro-oxidant potential, protection of neuronal cells and tissues, and mitochondriogenesis [1–14]. Although a role as a vitamin in animal or human nutrition is unlikely [15–17], similar to other polyphenolic biofactors, PQQ does interact in pathways important for cell signalling [18–21].
When PQQ is omitted from chemically defined diets fed to mice and rats, a wide range of systematic responses are observed including growth impairment, compromised immune responsiveness and abnormal reproductive performance [1–4]. Varying PQQ in highly refined diets also causes modulation in hepatic mitochondrial content and alteration in mitochondrial-related amino acid and lipid metabolism [1,2,20,21]. We have recently provided evidence that changes in mitochondrial amount were associated with modulation in CREB (cAMP-response-element-binding protein) phosphorylation and subsequent PGC-1α (peroxisome-proliferator-activated receptor γ co-activator-1α) directed up-regulation of NRF-1 (nuclear respiratory factor-1), NRF-2 and Tfam (transcription factor A, mitochondrial) mRNA expression [20].
Both PQQ and its principal derivative [the PQQ amino acid adduct IPQ (imidazolopyrroloquinoline)] are widely distributed in animal and plant tissues and fluids ranging from pico- to nano-molar concentrations [21–25]. Given that the systemic effects of PQQ deprivation are influenced at levels of dietary intake in the nanomolar to micromolar range, in the present study purified diets were used to reduce the effects of other bioactive factors and xenobiotics, such as those found in typical rodent chow diets [26]. Furthermore, it is known that Escherichia coli and other organisms commonly found in intestinal microflora do not synthesize PQQ [27–29]. As a consequence, we infer that PQQ in mammals is derived at least in part from dietary or food sources. Accordingly, dietary conditions were also chosen to assess responses to short- and longer-term PQQ deprivation. Gene expression microarray profiling was used as a comprehensive approach to determine whether changes in PQQ dietary protocols or pattern of exposure significantly modulate the expression of specific transcription networks.
EXPERIMENTAL
Reagents
Chemicals and reagents used in diets and assays were obtained from Fisher Chemicals, Sigma–Aldrich, Fluka or Supelco and were of the highest purity available. Amino acids for diet preparations were purchased from Ajinomoto. Reverse transcription and PCR enzymes and reagents were purchased from Applied Biosystems or Ambion. Microarray reagents and kits (Codelink® Rat Whole Genome Bioarray System) and supporting Codelink® statistical software were obtained from Applied Microarrays. RNA extraction and purification kits were purchased from Qiagen. PQQ was a gift from Mitsubishi Gas Chemical Company.
Animal studies, diets and husbandry protocols
Rats were used and housed individually in plastic cages. Recycled fibre, devoid of inks or dyes, was used for bedding (Carefresh Total 1 Clean Bedding). Animals were provided free access to food and water. The water supply was filtered through an activated carbon cartridge (Carbon Capsule 12122; Gellman Sciences), and then filtered through a 0.2 mm bacterial filter (Mini Capsule 12122; Gellman Sciences). Food cups and cages were changed twice weekly. Rats were housed and maintained in an Association for Assessment and Accreditation of Laboratory Animal Care International approved facility with approval from the campus Institutional Animal Care and Use Committee. Rats were fed on an ASD (amino-acid-semi-purified-based diet), which provided all known required nutrients in sufficient quantities to provide maximal growth, reproduction and lactation [1–4]. The ASD contained <5 fmol of PQQ/g.
The effects of PQQ depletion and repletion were examined using male rat pups derived from Sprague–Dawley dams fed on the basal ASD. To produce PQQ deficiency, 10-week-old virgin Sprague–Dawley rats were mated and, during the last 10 days of gestation, two-thirds of the rats were fed the basal ASD (PQQ−), while the remaining one-third were fed a PQQ-supplemented diet (2 mg of PQQ/kg of food or approx. 6 nmol/g of food; PQQ+). Offspring were then assigned to and fed the same diets as their corresponding dams (n=6). At 6.5 weeks post-weaning, half of the PQQ− and PQQ+ rats were divided to generate short-term repleted (PQQ−/+) and depleted (PQQ+/−) groups. The repletion was accomplished by administering PQQ by intraperitoneal injection at 1.5 mg of PQQ/kg of body weight every 12 h for a period of 36 h. Short-term depletion was achieved by switching PQQ+ rats to the PQQ− diet for 48 h. On the basis of previous observations [28,29], the depletion study was conducted for 48 h to allow a washout period for presumed cellular reserves of PQQ. An additional group of rats (n=6) fed a standard LC (laboratory chow) diet was used as a reference.
Microarray analysis and statistics
Total RNA was isolated from the liver of each rat using TRIzol® reagent (Invitrogen) and further purified using Qiagen RNA mini-kits. On-column DNA digestion was performed using an RNase-free DNase kit (Qiagen) to remove DNA residues. To ensure the RNA quality of each sample, integrity and purity were assessed by use of the Agilent Bioanalyser. The biotin-labelled cRNA target was prepared by a linear amplification method using a Codelink® expression array kit (GE Lifesciences). Total RNA (10 μg per sample) was used for cDNA synthesis using DNA oligonucleotides and T7 RNA polymerase. A set of bacterial mRNA controls was also included for the synthesis. The first-strand cDNA synthesis was followed by the second strand synthesis. The resulting DNA served as the template for an in vitro transcription reaction to produce targeted cRNA. The in vitro transcription reaction was performed with the addition of biotinylated nucleotides to label the cRNA. This method produces an approx. 1000-fold linear amplification of the input poly(A)–RNA. RNeasy Mini spin columns (Qiagen) were used for cRNA purification. The quality and yield of cRNA was assessed and subjected to chemical fragmentation.
The fragmented cRNA samples (10 μg each) were applied to bioarrays and hybridized overnight (17 h) in a temperature controlled shaking incubator, washed and followed by streptavidin–phycoerythrin conjugate staining. A subsequent series of less stringent washes were also performed to remove any unbound dye conjugates. Six individual animals per group were used in the analysis; no samples were pooled.
Codelink® expression analysis software provided automatic alignment and data extraction. Positive and negative bacterial controls were added during the target preparation to serve as overall platform performance controls. The Codelink® software also provided background correction. A global Loess function was applied to normalize the microarray data, and to correct for bias and the high-variance data points [30–32]. The probes for each experiment were sorted based on the product of their experimental and reference normalized signal intensities.
Gene expression was assessed by supervised analysis with the SAM (significance analysis of microarrays) algorithm using the Integromics Biomarker Discovery for microarray data analysis software (http://www.integromics.com). SAM is similar to Student's t test, but with permutations to calculate the FDR (false discovery rate) and to pick out genes where the expression level is significantly different between two groups of samples [33–35]. Normalized expression values from Codelink® analysis were used for a two-class unpaired SAM analysis. The SAM software estimated the false discovery rate and generated a q-value for each gene. The q-value for each gene represents the probability that it is falsely called differentially expressed. Similar to a P-value, a smaller q-value indicates a more significant differential expression. This is an advantage over other techniques (e.g. ANOVA followed by a Bonferroni, Tukey, Newman–Keuls or Dunnett's post-test), which assume equal variance and/or independence of given variables. A 5% FDR and an absolute fold-change value of 2.0 were used criteria to screen for differentially expressed genes and filter genes between different groups. Additional statistical data are provided in Supplementary Figures S1 and S2 (at http://www.BiochemJ.org/bj/429/bj4290515add.htm).
IPA (ingenuity pathways analysis)
An additional analysis using IPA software (http://www.ingenuity.com/index.html) was performed to identify principal processes associated with functional categories of genes that responded to changes in PQQ nutritional status. PQQ data sets (PQQ−, PQQ+/− or PQQ−/+) with fold changes of >2.0 (relative to the corresponding control groups) and SAM analysis of q<5.0 were uploaded into the application and each gene was mapped to its corresponding gene object using the Ingenuity pathways knowledge base. A functional and group comparison analysis was then performed to identify functional categories that responded to changes in PQQ status. A Fisher's exact test was performed to calculate whether a given biological function was due to chance. Using the IPA statistical platform, a score of 3 (P<0.01) was used as the cut-off for identifying gene networks that were significantly affected by PQQ.
qRT-PCR (quantitative real-time PCR)
Nine genes were chosen to aid in validating the bioarray results. PCR expression profiles were obtained for carnitine-O-octanoyltransferase, HMG-CoA (3-hydroxy-3-methylglutaryl-CoA) lyase, the cytochrome P450 genes CYP4A3 and CYP7A1, glucokinase, lipocalin 2, monoacylglycerol lipase, Raf1 and STAT3 (signal transducer and activator of transcription 3) as markers for MAPK (mitogen-activated protein kinase)-related signalling, changes in secondary or oxidative metabolism pathways [36].
Two further genes, the c-fos oncogene and NPY (neuropeptide Y), were chosen as additional markers [37–39]. These genes are of relative low abundance and had relatively high FDR values, but were nevertheless chosen for qRT-PCR, because changes in expression can often suggest the involvement of cell signalling associated with MAPK/ERK (extracellular-signal-regulated kinase) or adipocytokine pathways and/or alteration in mtDNA (mitochondrial DNA) expression [40,41]. For c-fos and NPY, total RNA (1 μg from individual samples) containing the first-strand primer [oligo(dT)] was incubated at 65 °C for 5 min and chilled on ice for 2 min. Reverse transcription was performed in a total volume of 20 μl containing 0.2 mM of each dNTP, 200 units of MMLV (Moloney-murine-leukaemia virus) reverse transcriptase and RNAsin at 42 °C for 45 min, followed by 94 °C for 5 min. The cDNA obtained was next diluted to a final concentration of 10 ng/μl and stored at −80 °C.
The qRT-PCR analyses [40,41] were processed with an ABI Prism 7300 sequence detection system (PE Applied Biosystems). GAPDH (glyceraldehyde-3-phosphate dehydrogenase) and β-actin mRNA levels were used as references. Dual-fluorescent non-extendable probes were used and were labelled with 6-carboxyfluorescein at the 5′-end and with 6-carboxytetramethylrhodamine at the 3′-end in all primers and probe combinations (25 μl reaction volumes containing 900 nM primers and 250 nM Taqman® probe) and universal thermal cycling parameters. Amplification involved incubation at 95 °C for 10 min to activate the Taq polymerase, followed by a two-step PCR for 40 cycles: denaturation 15 s at 95 °C, and annealing and extension for 1 min at 60 °C. Following amplification, levels of mRNA expression for the selected gene sequences were normalized relative to either GAPDH or β-actin expression using the comparative cycle time (Ct) method [41,42]. For the relative expression of the c-fos oncogene and NPY, GAPDH was used.
Liver mtDNA levels
The relative amounts of liver mtDNA were measured by qRT-PCR [42–44]. DNA was extracted with phenol/chloroform and precipitated with ethanol. For nuclear DNA quantification, 10 ng of DNA was used as a template. Rat-specific primers were selected using the Primer Express® Software (Applied Biosystems). Primers for nuclear CF (cystic fibrosis) were: forward 5′-AAACTCAGGATAGCTGTCCGTTTAG-3′ and reverse 5′-GCCAAATGATAGCATGGAACTCT-3′. For mtDNA quantification, 0.1 ng of DNA was used as a template and primers for mitochondrial ND-5 (nicotinamide adenine dinucleotide dehydrogenase-5) were: forward 5′-GGATGATGATATGGCCTTGCA-3′ and reverse 5′-CGACTCGGTTGTAGAGGATTGC-3′. PCR was performed using an ABI 7900HT real-time thermocycler (PerkinElmer) coupled with SYBR Green technology (Applied Biosystems) and the following cycling parameters: stage 1, 50 °C for 2 min; stage 2, 95 °C for 10 min; stage 3, 40 cycles for 95 °C for 15 s; 60 °C for 1 min; and stage 4, 95 °C for 15 s; 95 °C for 15 s. The linearity of the dissociation curve was analysed using the ABI 7900HT software. Each sample was analysed in duplicate. The mean cycle time of the linear part of the curve was designated Ct. Relative mitochondrial copy number to nuclear copy number was assessed by a comparative Ct method (ΔCt mitochondria/nuclear = Ct mitochondria−Ct nuclear) to assess for the fold-change for mtDNA/nuclear DNA in liver from PQQ−, PQQ+, PQQ−/+ and PQQ+/− rats. Values are expressed as the means±S.E.M. using a Dunnett'ss test to estimate statistical significance.
Mitochondrial-related genes
In a separate study, groups of PQQ− and PQQ+ rats were used to obtain relative Tfam and PGC-1α mRNA levels and reconfirm relationships related to changes in mtDNA levels. Livers from individual rats were removed intact, weighed, examined and rapidly flash-frozen for RNA and DNA extractions. Blood was also collected in heparinized tubes and immediately centrifuged. Plasma was obtained following centrifugation and frozen at −70 °C until analyses. For the relative expression of Tfam and PGC-1α, β-actin was used for normalization. The targeted PCR Ct value (i.e. the cycle number at which emitted fluorescence exceeds 10× the S.D.) of baseline emissions (measured from cycles 3 to 15) was normalized to the GAPDH or β-actin PCR Ct values by subtracting these values from their respective target PCR Ct value to derive a ΔCt value. From the ΔCt value, the relative mRNA expression level for each targeted transcripts was calculated by relative mRNA expression {[2−(Ct target−Ct control gene)]×100 (to give a percentage)}. A DNA positive control was also run for each set of assays. Results are reported as means±S.E.M. for each sample in triplicate. For the PGC-1α and Tfam primer sets, the sequences used were: PGC-1α, forward 5′-AATGAGCCCGCGAACATATT-3′ and reverse 5′-TGAGGACCGCTAGCAAGTTTG-3′; Tfam, forward direction 5′-AACGCCTAAAGAAGAAAGCACAA-3′ and reverse 5′-CCGAGGTCTTTTTGGTTTTCC-3′.
Plasma sphingosine and triglycerides
Plasma sphingosine and triacylglycerols were measured as indices of functional perturbations in phospholipid and neutral lipid metabolism. Lipids extracted in the presence of authentic internal standards by the Folch method with chloroform/methanol (2:1 v/v). Individual lipid classes were separated by liquid chromatography. Each lipid class was transesterified in 1% (v/v) sulfuric acid in methanol under a nitrogen atmosphere at 100 °C for 45 min. The resulting fatty acid methyl esters were extracted from the mixture with hexane containing 0.05% butylated hydroxytoluene and prepared for gas chromatography under nitrogen. Fatty acid methyl esters were separated and quantified by capillary gas chromatography (Agilent Technologies model 6890) equipped with a 30 m DB-88MS capillary column (Agilent Technologies) and a flame-ionization detector. The basis for these assays evolved from the IPA focusing on canonical lipid pathways that were significantly influenced by changes in PQQ status.
PQQ estimation
A GDH (glucose dehydrogenase)-based assay system was used to estimate dissociable PQQ levels in plasma as described previously [1–4,45]. The recovery of PQQ added to assays as an external spike was >85%.
RESULTS
Animals
PQQ deficiency resulted in a reduction in the relative liver weight when compared with body weight, an effect that was reversed upon PQQ repletion. Changes in body weights were not obvious at week 10 (Table 1, also see the Discussion section). Plasma levels of PQQ for rats fed the basal diet (ASD), as well as changes in the relative levels of mtDNA, largely reflected the levels of dietary PQQ for the rats fed the ASD. Results from rats fed an LC diet are also shown for comparison. The lower values for ‘free’ PQQ in plasma of rats fed LC may reflect: (i) variable intake; (ii) differences due to PQQ adduct formation (e.g. IPQ, see [21]); and/or (iii) a decreased bioavailability of PQQ due to the complexity and extensive processing of LC diets [29]. The PQQ content of LC measured over the last 4 years routinely ranges from 1 to 5 mg of PQQ/kg of diet.
Table 1. PQQ status, body weight, liver/body weight ratio, plasma PQQ levels and liver mtDNA/nuclear DNA ratios.
PQQ+, values for rats fed on an ASD containing 2 mg/kg PQQ; PQQ−, values for rats fed on an ASD devoid of PQQ; PQQ+/−, values for rats subjected to a short-term PQQ depletion (48 h); PQQ−/+, values for rats subjected to a PQQ repletion via intraperitoneal injection at 1.5 mg of PQQ/kg of body weight every 12 h for a period of 36 h. The relative amounts of liver mitochondrial DNA (mtDNA) and nuclear DNA measured by real-time PCR. The targeted genes were the nuclear CF and the mitochondrial ND-5 gene. Values within each row labelled with differing superscript letters indicate a difference between the groups of P<0.05 using a Dunnett's test.
| PQQ treatment group | |||||
|---|---|---|---|---|---|
| Parameter | PQQ+ | PQQ− | PQQ+/− | PQQ−/+ | LC |
| Body weight (g) | 220±11a | 225±16a | 221±13a | 231±8a | 250±22a |
| Liver/body weight (% relative to LC values) | 96±5a | 82±4 b | 101±4a | 91±4a | 100±5a |
| Plasma PQQ (nM) | 12±3a | 1.5±2.0b | 8±4a | 17±7a | 4.0±2.0b |
| Liver mtDNA/nuclear DNA | 1.05±0.24a | 0.76±0.13b | 0.82±0.2a,b | 1.3±0.2a | 1.1±0.2a |
Microarray analysis
Of the transcripts analysed by microarray, 10 weeks of PQQ deprivation caused a change in 238 transcripts, and short-term PQQ deficiency (for 48 h) caused a change in 438 transcripts relative to the PQQ+ group (P<0.01). When PQQ-deficient rats were subjected to short-term PQQ repletion, approx. 8.5% or 847 genes (P<0.01) were up- or down-regulated relative to the corresponding PQQ− group. With regard to changes in PQQ status, among the genes altered were genes associated with cellular stress, cell signalling or immune and neural function (Table 2). Changes in relative transcript levels brought about by PQQ deficiency or short-term depletion consistently responded to PQQ repletion (PQQ−/+), which resulted in a reversal in the direction of expression or an amplification in expression. Values for short-term PQQ deficiency were intermediate between the PQQ− and PQQ−/+ groups. Moreover, the results presented in Table 2 are consistent with observations published previously on the effects of PQQ supplementation and deficiency in animal models and cells; e.g. we have reported that PQQ-deficient rats and mice express dyssynchronous changes in development and immune function [3,4].
Table 2. Metabolic stress, cell signalling and immune function-related genes influenced by changes in PQQ status.
An asterisk (*) indicates a significant increase or decrease in gene expression relative to values for the corresponding control group. The analysis was performed by means of SAM analysis using the Integromics Biomarker Discovery microarray data analysis software. Values within each row labelled with differing superscript letters indicate a difference between the groups of P<0.05 using a Dunnett's test. CaM, calmodulin; CDK, cyclin-dependent kinase; CTL, cytotoxic T-cell; IL, interleukin; NF-κβ, nuclear factor κB; NMDA, N-methyl-D-aspartate; PG, prostaglandin; SAPK, stress-activated protein kinase; TGF, transforming growth factor; TNF, tumour necrosis factor.
| PQQ treatment group (fold change relative to PQQ+) | ||||
|---|---|---|---|---|
| Gene | PQQ− | PQQ+/− | PQQ−/+ | Associated functions |
| Metabolic stress and apoptosis-related genes | ||||
| UVB radiation-activated UV96 mRNA | −5.1*a | 1.3b | 7.8*c | UV radiation-induced cellular stress-associated gene |
| Integrin-associated protein | −4.3*a | 1.1b | 6.3*c | Regulation of NF-κβ |
| Receptor-interacting serine-threonine kinase 3 | −2.1*a | 1.0a,b | 1.3b | Apoptosis, component of the TNF receptor-I signalling complex |
| Huntingtin-interacting protein | −2.1*a | −1.6a | 5.6*b | Associated with apoptosis |
| BAX | −2.0a | 1.6b | 8.0*c | Pro-apoptotic regulator that is involved in a wide variety of cellular activities (e.g. mitochondrial membrane porosity) |
| Catechol-O-methyl transferase | −1.5a | 13.6*b | 27.8*c | Cellular stress; involved in IL-1 signalling |
| Smad 5 | −1.2a | −1.4a | 3.2*b | Modulation of TGF-β ligands |
| STAT3 | −1.2a | −1.2a | 7.3*b | Transcription acute-phase response factor that is activated by nuclear co-activator 1 |
| Lyric | −1.2a | 1.1a | 4.1*b | Activates the NF-κβ transcription factor |
| D-site albumin promoter-binding protein | 4.8*a | −2.7*b | −5.9*b | Transcription factor; down-regulation is associated with reduced metabolic stress and apoptosis |
| p75-like apoptosis-inducing death domain protein | 2.7*a | −1.2b | −3.0*c | Apoptosis; is expressed in high amounts in the embryonic tissue and down-regulated in adult tissue |
| 5-Hydroxytryptamine (serotonin) receptor 1F | 1.5a | −1.5a | −3.6*c | Serotonin receptor |
| Heat-shock factor 2 | 1.2a | 1.2a | 2.6*b | Stress-related chaperon transport functions |
| Neuraminidase 2 | 1.1a | −2.9*b | −2.4*b | Sialidase (cleaves neuraminic acid glycosidic linkages), important to the mobility of virus and related particles |
| Prostaglandin D2 synthase | 1.0a | 1.2a | −2.4*b | Glutathione-independent prostaglandin D synthase that catalyses the conversion of PGH2 into PGD2 |
| MAPK and MAPK phosphatases | ||||
| Receptor-interacting serine/threonine kinase 3 | −2.1*a | 1.0b | 1.3b | Component of the TNF receptor-I signaling complex |
| 3-Phosphoinositide-dependent protein kinase-1 | −1.6a | −1.4a | 3.1*b | Signalling processes important for development; activates various MAPK signalling pathways |
| Dual-specificity phosphatase 7 | −1.3a | −1.7a | 2.1*b | Negatively regulate the activity of MAPKs in response to changes in the cellular environment (e.g. heat or metabolic stress) |
| MAPKKK12 | −1.2 a | −1.3a | 3.1*b | An activator of the JNK/SAPK pathway |
| Tyrosine protein kinase pp60 | −1.1a | −1.1a | 2.9*b | A protein kinase C (Ca2+/phospholipid-dependent); known to phosphorylate epidermal growth factor receptor and reduce its affinity for epidermal growth factor |
| Serine/threonine kinase 3 | 2.8*a | 3.2*a | −3.0*b | MAPK that is activated in response to cellular stress |
| MAPK phosphatase | 2.0*a | 1.0a | −2.6*b | Up-regulated during stress; modulates MAPK activities that are sensitive to changes in the cellular environment |
| Protein tyrosine phosphatase 4a1 | 1.5a | −1.6b | −2.8*c | Up-regulated during stress; modulates MAPK activity |
| Cyclin A2 | 1.3a | 1.5a | −3.7*b | Regulators of CDK kinases important to the temporal co-ordination of mitotic events |
| RAS p21 protein activator 1 | 1.2a | −1.2a | −4.0*b | Cytoplasmic GAP1 family of GTPase-activating proteins important to cell proliferation; most abundant in embryonic tissue |
| CaM-kinase II inhibitor | 1.0a | −1.4a | 3.1*b | Serine/threonine protein kinase inhibitor in the Ca2+/CaMdependent protein kinase subfamily |
| MAPK14 | 1.3a | −3.9*b | −2.7*b | A p38 kinase, activated by cellular stress; MAPK 14 activates ERK and kinases in Ras-oncogene related pathways |
| Immune and neural function | ||||
| Cathepsin H | −2.6*a | −4.3*b | 2.4*c | Lysosomal cysteine proteinase |
| Lysozyme | −2.4*a | −1.7*a | 4.0*b | A major component of the innate immune system |
| CTL target antigen | −2.3*a | −1.4b | 1.8*c | Antigen to killer T-cells |
| Complement component 1 | −2.2*a | −1.1b | 1.3c | Important for the complement fixation sites of immunoglobulin |
| Transcription factor 4 | −2.2*a | −1.7*a | 7.0*b | Immune function |
| STAT5B | −2.0*a | −1.2a | 1.6b | T-cell receptor signalling, apoptosis, sexual dimorphism of liver gene expression |
| Basigin | −1.4a | 1.7b | 3.8*b | Member of the immunoglobulin super family with a number of functions (e.g. lymphocyte recognition) related to early development |
| CD14 antigen | −1.7*a | −1.1b | 1.5b,c | Immune response |
| MHC II, DM β class II | −1.7*a | −1.1b | 2.2*c | Immune response |
| JAK3 | 1.0a | 1.6a | 2.3*b | Transduces a signal in response to its activation via tyrosine phosphorylation by interleukin receptors; mutations of JAK3 result in severe immunodeficiency; mice that do not express JAK3 have T-cells and B-cells that fail to respond to many cytokines |
| Presenilin 1 | −3.6*a | −1.6b | 2.4*c | Proteinase associated with amyloidal protein defects |
| RAF-1 | −1.6a | 1.1a | 15.5*b | Receptor important for cellular proliferation and regulation by neuregulins, important for the development of the nervous system |
| Ninjurin | −1.6a | 1.0a | 3.1*b | Nerve injury-induced protein involved in nerve regeneration and in their formation |
| Neuropilin | −1.1a | 1.2a | 5.8*b | Semaphorin binding is especially important in neural system development |
| Cholinergic receptor, nicotinic, α polypeptide 1 | −1.1a | −1.6a,b | −2.0*b | Acetylcholine receptors |
| Neurotrophin-3 | 3.6*a | 2.2*a | 1.1b | Protein growth factors important to peripheral and central nervous system function |
| Benzodiazepine receptor | 2.7*a | −1.4b | −2.0*b | Enhances the effects of the neurotransmitter γ-aminobutyric acid |
| Plexin A3 | 2.0*a | −1.1b | −5.0*c | Interacts with RAC-α serine/threonine protein kinase, a mediator of growth factor-induced neuronal survival |
| Glutamate receptor, ionotropic, NMDA2B | 1.0a | 1.5a | −4.7*b | NMDA receptor important for synaptic transmissions |
| Cholinergic receptor, nicotinic, β polypeptide 1 | 1.4a | 1.1a | −2.6*b | Acetylcholine receptor |
In addition, similar changes in patterns were observed for genes associated with cellular transport, assembly, and cycling, as well as extracellular matrix formation (Table 3), and mitochondrial-related metabolic events and oxidant sensing (Table 4). Table 4 also lists genes that are responsive to changes in PQQ status that are important for lipid-related energy and phospholipid metabolism. These changes complement those observed for genes important for transport, cellular assembly and cell signalling (Tables 2 and 3). In addition, for the approx. 900 total genes in the array that were significantly influenced by either PQQ deficiency or administration, over 10% could be identified in the MitoCarta mouse inventory. The MitoCarta mouse inventory is a collection of 1098 nuclear and mtDNA genes (approx. 4% of the genes in the mammalian genome) that have been judged to be essential to mitochondriogenesis (http://www.broad.mit.edu/pubs/MitoCarta/mouse.mitocarta.html).
Table 3. Cellular transport, assembly, growth, cell cycling and extracellular matrix formation genes influenced by changes in PQQ status.
An asterisk (*) indicates a significant increase or decrease in gene expression relative to values for the corresponding control group. The analysis was performed by means of SAM analysis using the Integromics Biomarker Discovery microarray data analysis software. Values within each row labelled with differing superscript letters indicate a difference between the groups of P<0.05 using a Dunnett's test. ABC, ATP-binding-cassette; CDK, cyclin-dependent kinase; DMT, divalent metal transporter; ER, endoplasmic reticulum; MARCKS, myristoylated alanine-rich protein C-kinase substrate; SNAP, soluble N-ethylmaleimide-sensitive fusion protein-attachment protein; VAMP, vesicle-associated membrane protein.
| PQQ treatment group (fold change relative to PQQ+) | ||||
|---|---|---|---|---|
| Gene | PQQ− | PQQ+/− | PQQ−/+ | Associated functions |
| Transport | ||||
| Solute carrier family 28, member 2 | −2.1*a | 1.0b | 1.0b | Sodium-coupled nucleoside transporter |
| Solute carrier family 34, member 2 | −2.0*a | 2.2*b | 4.2*c | Sodium-dependent phosphate transport |
| Transporter 1, ATP-binding cassette, subfamily B | −2.8*a | 1.3b | 2.3*c | Member of the ABC transport family; cholesterol efflux regulatory protein |
| Embigin 5 | −2.0*a | 1.2a | 1.5b | Monocarboxylate transporter |
| DMT1-associated protein | −1.2a | −1.1a | 2.1*b | Divalent metal ion (e.g. Fe2+) transporter |
| Aquaporin 8 | −2.2*a | 1.0b | 3.6*c | Water transport; localized in the inner mitochondrial membrane |
| Solute carrier family 2, member 13 | 1.3a | 1.1a | −3.3*b | Member of the facilitated glucose transporter family |
| Solute carrier family 4, member 4 | 1.0a | −1.1a | 3.4*b | Electrogenic sodium bicarbonate co-transporter |
| Solute carrier family 4, member 1 | 1.6a | −2.2*b | −4.0*c | Chloride-bicarbonate cotransporter |
| Solute carrier family 6 | 1.3a | 1.0a | −2.7*b | Co-transport of Na+ and Cl− and neurotransmitter molecules (family includes carriers for -aminobutyric acid, noradrenaline/adrenaline, dopamine, serotonin, proline, glycine, choline, betaine and taurine) |
| Solute carrier family 14, member 2 | 1.2a | −5.6*b | −1.2a | Urea transporter |
| Solute carrier family 17 | 1.2a | 1.0a | 6.5*b | Phosphate transporter |
| Solute carrier family 18 | 2.3*a | −1.6b | −6.8*c | Vesicular monoamine transporter |
| Solute carrier family 20, member 1 | 1.7*a | −1.1a | −3.0*c | Phosphate transporter |
| Similar to solute carrier family 2, member 5 | 2.4*a | 2.5*a | −1.4b | Integral membrane transporter, similar to those in the facilitated glucose transporter family |
| Similar to solute carrier 37 | 1.2a | −1.3a | −2.0*b | Glycerol 3-phosphate transporter |
| Cellular assembly | ||||
| Epimorphin | −2.5*a | 1.1b | 2.3b | Intracellular vesicle docking |
| MARCKS-like protein | −1.9*a | 1.0b | 1.0b | Interacts with dynactin, ER-to-Golgi transport |
| Signal transducer and activator of transcription 5B | −2.0*a | −1.2a | 1.6b | Activation of tethering signalling components, apoptosis |
| Epimorphin | −1.2a | 1.3a | 2.3*b | Intracellular vesicle docking protein |
| Golgi SNAP protein, member 2 | −1.2a | −1.9a | 5.4*b | Trafficking of membrane proteins that travel among the medial- and trans-Golgi compartment |
| GTPase Rab8b | −2.3*a | −2.0*a | 2.9*b | Rab GTPases regulate many steps of membrane trafficking: vesicle formation, vesicle movement along actin and tubulin networks, and membrane fusion |
| Transgelin | −1.6a | −1.0a | 3.4*b | Transformation and shape-change; actin cross-linking/gelling |
| Karyopherin α2 | −1.0a | 1.0a | 3.0*b | Involved in transporting molecules through the pores of the nuclear envelope: a part of the importin-β superfamily |
| Trans-Golgi network protein 1 | 4.7*a | 1.1b | 1.3b | Trans-golgi assembly and recycling of trans-Golgi components |
| Chimerin 1 (Chimaerin) | 2.9*a | 1.4b | −6.3*c | Regulation of Rac, a subfamily of the Rho family GTPases involved in cell organization and regulation |
| Chimerin 2 | 1.6a | −1.4a | −4.9*c | Induces translocation of the protein from the cytosol to the Golgi apparatus |
| Heat-shock 27 kDa protein family, member 3 | 1.4a | −1.1a | −3.5*b | Hsp27 functions as a molecular chaperone and is involved in the regulation of cell growth and differentiation |
| ADP-ribosylation factor guanine-nucleotide exchange factor 2 (brefeldin A-inhibited) | 1.4a | 1.0a | −1.9*b | Intracellular vesicular trafficking |
| Dynactin 4 | 1.1a | 1.1a | −2.3*b | Aids in bidirectional intracellular organelle transport by binding to dynein and kinesin II and linking them to the organelles to be transported |
| GRB2-associated binding protein 2 | 1.6a | 1.5a | −2.6*b | Principal activator of phosphatidylinositol 3-kinase involved in cellular growth, proliferation, motility and intracellular trafficking |
| Vesicle-associated membrane protein 8 | −1.2 a | 1.2 a | 2.3*b | Synaptobrevins/VAMPs, syntaxins and the 25-kDa synaptosomalassociated proteins (SNAP25) are components involved in the docking and/or fusion of synaptic vesicles with the presynaptic membranes |
| Growth, cell cycling and extracellular matrix formation | ||||
| Connective tissue growth factor | −3.1*a | −1.2b | 4.3*c | Cellular matrix induction |
| Collagen type XXVII | −2.9*a | −1.2b | 3.8*c | In the ‘fibrillar’ class of collagens |
| Prolyl 4-hydroxylase α subunit | −3.1*a | −3.1*a | 3.4*b | Catalyses the formation of 4-hydroxyproline |
| Cyclin D2 | −1.2 a | −1.1 a | 2.5*b | Regulation of transcription and mRNA processing |
| Cyclin-dependent kinase inhibitor 1C, p57 | −1.2 a | 1.4 a | 2.7*b | A negative regulator of cell proliferation |
| Septin-like protein | −1.1 a | 1.2 a | 1.9*b | Apparent cell division cycle regulatory protein |
| Similar to methionyl-tRNA synthetase | 3.9*a | −1.7 | −14.7 | Apparent relationship with protein translation |
| p21 (CDKN1A)-activated kinase 2 | 1.0a | −1.2a | −2.3*b | Kinase in the CDK important for cell cycling |
| Cyclin A2 | 1.3a | 1.5a | −3.7*b | Cyclins function as regulators of CDKs; CDKs are involved in the regulation of transcription and mRNA processing |
| p21 (CDKN1A)-activated kinase 3 | 2.1*a | −1.2b | −8.0*c | Tightly controlled by p53; important for cell cycling and G1-phase arrest in response to a variety of stress stimuli |
| Prominin | 1.3 a | −9.0*b | −29.8*c | Localizes to cellular protrusions in haemopoietic and neuronal stem cells |
| Integrin α E1 | 1.1 a | 1.0a | 2.4*b | One of many integrin receptors that mediate attachment between a cell and the tissues |
| Lamin B receptor | 2.3*a | −1.8*b | −6.5*c | Aids in anchoring the lamina and heterochromatin to cellular membranes |
| Chondroadherin | 1.3 a | −1.9*b | −5.1*c | Binds cells via the integrin α-family of receptors and controls cell spreading |
| Chondroitin sulfate proteoglycan 3 | 1.3 a | −1.6a | −2.1*a,b | Cell matrix component |
Table 4. Mitochondrial, secondary metabolism and lipid-related genes influenced by changes in PQQ status.
An asterisk (*) indicates a significant increase or decrease in gene expression relative to values for the corresponding control group. The analysis was performed by means of SAM analysis using the Integromics Biomarker Discovery microarray data analysis software. Values within each row labelled with differing superscript letters indicate a difference between the groups of P<0.05 using a Dunnett's test. HO, haem oxygenase; IL, interleukin.
| PQQ treatment group (fold change relative to PQQ+) | ||||
|---|---|---|---|---|
| Gene | PQQ− | PQQ+/− | PQQ−/+ | Associated functions |
| Mitochondrial and metabolism-related | ||||
| Mitochondrial tumour suppressor gene 1 | 1.7*a | 1.3a | −1.7*b | Mitochondrial-related apoptosis |
| STAT3 | −1.4a | 1.1a | 7.0*b | Interacts with IL-6; required for optimal mitochondrial function |
| ATP synthase, H+ transporting, mitochondrial F1 complex, γ polypeptide 1 | −1.4a | −1.1a | 2.0*b | Subunit of mitochondrial ATP synthase |
| ATP synthase, H+ transporting, mitochondrial F1 complex, O subunit | −1.3a | 1.2a,b | 1.8b* | Subunit of mitochondrial ATP synthase |
| ATP synthase, H+ transporting, mitochondrial F1 complex, β polypeptide | 1.0a | −1.2a | 2.5*b | Subunit of mitochondrial ATP synthase |
| ATP synthase, H+ transporting, mitochondrial Fo complex, subunit c, isoform 1 (5 other subunitsdetected) | −1.1a | 1.2a | 2.3*b | Subunit of mitochondrial ATP synthase |
| ATP synthase, H+ transporting, mitochondrial F0 complex, subunit c (subunit 9) | 1.0 a | 1.1a | 2.3*b | Subunit of mitochondrial ATP synthase |
| Cytochrome c oxidase subunit 4A | −1.1a | 1.2a | 2.6*b | Subunit of cytochrome c oxidase |
| Cytochrome c oxidase subunit VIIb | −1.1a | −1.1a | 2.3*b | Subunit of cytochrome c oxidase |
| HO-1 | −1.7 *a | −1.1a | 1.9*b | HO-1 is a protective antioxidant enzyme that prevents apoptosis; inducible |
| HO-2 | 1.1a | −1.1 a | −1.2a | Constitutive HO involved in haem catabolism used a control for HO-1 and HO-3 |
| HO-3 | −3.2*a | −1.4b | 22.9*c | HO-3 is not catalytically active, but is thought to function as an oxygen sensor |
| Hypoxia-inducible factor 1 | 1.0a | −1.2 a | −2.3*b | Functions as an oxygen sensor |
| Phosphoserine aminotransferase 1 | −6.7*a | −1.0a | 3.4*b | Formation of phosphoserine from 3-phosphohydroxypyruvate |
| Glycerol kinase | −2.5*a | 1.0b | 2.3*c | Triacylglycerol formation |
| Succinyl-CoA ligase (ATP-forming), β chain | −1.4a | 1.0a | 2.3*b | Energy production and conversion |
| Glucokinase regulatory protein | −1.7a | 1.3b | 4.7*c | Glucokinase/glucose regulation |
| Glucokinase | −1.9*a | −2.9*b | 2.1*c | Glucose regulation |
| Fructose-2,6-biphosphatase 4 | −1.9*a | 1.6b | 3.2*c | Important for glycolysis and gluconeogenesis |
| Glutamate oxaloacetate transaminase 2 | 1.3a | 1.6a | 1.3a | Amino acid metabolism |
| D-site albumin promoter-binding protein | 4.5*a | −2.8*b | −5.7*c | A member of the proline and acidic amino acid-rich basic leucine zipper transcription factor family; binds to an upstream promoter in the insulin gene |
| Glycogen synthase 2 | 1.7a | −1.1b | −2.6*c | Regulation of glycogen synthesis |
| Pyruvate dehydrogenase phosphatase isoenzyme 1 | 1.0a | −1.1a | −2.7*b | Energy production and conversion |
| Ornithine decarboxylase | 1.4a | 1.0a | −2.3*b | Urea cycle regulation |
| Serine dehydratase | 5.5*a | −1.4b | −3.7*c | Converts serine into pyruvate; threonine into propionyl-CoA |
| Secondary metabolism-related | ||||
| Thioredoxin 2 | −1.1a | 1.1a | 2.2*b | Contributes to antioxidant activity by facilitating the reduction of proteins by cysteine thiol–disulfide exchange |
| Aryl hydrocarbon receptor nuclear translocator | −8.7*a | −1.1b | 9.6*b | Induction of enzymes important to xenobiotic metabolism; forms a complex with ligand-bound aryl hydrocarbon receptors required for receptor function |
| Cytochrome P4508B1 hydrolase | −2.8*a | 1.2b | 2.5*b | Phase 1 xenobiotic metabolism |
| Cytochrome P4504A3 hydrolase | −1.4a | 2.1*b | 3.6*b | Phase 1 xenobiotic metabolism |
| Cytochrome P4507A1 hydrolase | −2.1*a | −3.0*b | −3.0*b | Phase 1 xenobiotic metabolism |
| Cysteine–sulfinate decarboxylase | −4.5*a | −1.0b | 1.0b | Hypotaurine metabolism |
| Glutathione transferase, mu 5 | −1.2a | −1.2a | 2.4*b | Phase II reaction functions |
| Lipid and phospholipid-related | ||||
| Fatty acid elongase 2 | −1.2a | 2.5*b | 12.5*c | Fatty acid chain elongation |
| Fatty acid desaturase | −2.0*a | −1.2b | 1.9*c | Formation of unsaturated acyl chains in fatty acids |
| Carnitine-O-octanoyl transferase 1 | −2.0*a | 1.6b | 1.9*b | Mitochondrial transport fatty acid transport enzyme |
| Adiponectin receptor 2 | −1.4a | 1.1a | 2.8*b | Receptor for adiponectin binding; levels of adiponectin are inversely correlated with body fat percentage in adults |
| Choline kinase | −2.1*a | 1.3*b | 1.6b | Participates in glycine, serine and threonine and glycerophospholipid metabolism |
| Mevalonate pyrophosphate decarboxylase | −1.9*a | −1.1a | 2.0*b | Enzyme in the mevalonate pathway |
| HMG-CoA lyase | −1.4a | 1.0a | 2.4*b | Cholesterol biosynthesis |
| Farnesyl diphosphate synthase | −1.4a | 1.7b | 2.0*b | Conversion of geranyl diphosphate and isopentenyl diphosphate into farnesyl diphosphate |
| 2,3-Oxidosqualene:lanosterol cyclase | −1.4a | −2.1*a | 1.4b | Cholesterol synthesis |
| Isopentenyl-diphosphate Δ isomerase | −1.9*a | −1.6a | 1.8*b | Steroid synthesis regulation |
| 2,3-Oxidosqualene:lanosterol cyclase | −1.7*a | −1.4a | 1.4a | Steroid synthesis |
| Cytochrome P4508B1, sterol 12α hydrolase | −2.7*a | 1.2b | 3.0*c | Steroid synthesis |
| Lipocalcin 2 | 1.4a | −1.6b | 5.4c | Transport small hydrophobic molecules such as steroids, bilins, retinoids and lipids |
| Leptin receptor | 2.0*a | 1.3a | −2.0*b | Regulates adipose-tissue mass through hypothalamic effects on satiety and energy expenditure |
| Sphingomyelin phosphodiesterase | 2.0*a | 1.0a,b | −1.4b | Sphingomyelin regulation; sphingomyelin is converted into phosphocholine and ceramide |
| Ng22 protein | 1.8*a | −1.4b | −2.1*b | Choline transporter-like protein 4, phospholipid metabolism |
| CDP-diacylglycerol synthase (phosphatidate cytidylyltransferase) 1 | 1.3a | 1.3a | −1.8*b | A CTP-diacylglycerol or diacylglycerol synthetase that participates in glycerophospholipid metabolism and phosphatidylinositol signaling systems |
| Sphingolipid G-protein-coupled-receptor 5 | 1.5a | −2.9*b | −3.3*b | Lipid signaling molecule that binds sphingosine 1-phosphate |
| Lysophospholipase | 1.9*a | 1.4a | 1.3b | Glycerophospholipid metabolism; converts 2-lysophosphatidylcholine into glycerophosphocholine |
| Monoacylglycerol lipase | 1.0a | −1.4a | 3.6*b | Functions together with hormone-sensitive lipase to hydrolyse intracellular triacylglycerols |
| Fatty acid CoA ligase, long chain 5 | 1.3a | 2.5*b | 4.0*c | Conversion of long-chain fatty acids into acyl-CoA esters |
| Stearoyl-CoA desaturase 2 | 2.1*a | 4.0*b | −1.2c | A Δ9-fatty acid desaturase |
| Phosphatidylinositol-binding clathrin assembly protein | 1.0a | −1.2a | 1.9*b | Clathin assembly and phospholipid metabolism |
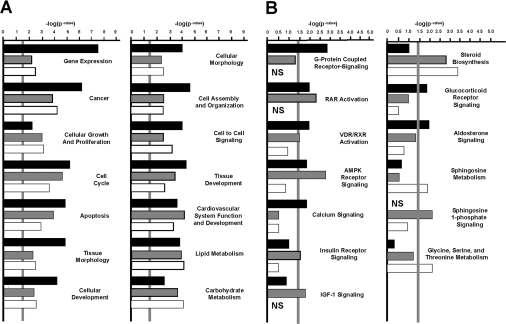
To verify the various categories that are presented in Tables 2–4, an IPA tool was run using results from all the four ASD groups (PQQ+, PQQ−, PQQ+/− and PQQ−/+). In keeping with the categories and partial list of genes presented in Tables 2–4, IPA resulted in similar categories that included canonical relationships important for cellular growth and proliferation, apoptosis, cell cycling and organization, cell–cell communication and cellular defence (Figure 1A). More direct functional or specific categories included a number of specific cell signalling pathways (e.g. G-protein-coupled receptor cycling, AMPK (AMP-activated protein kinase) signalling, IGF (insulin growth factor)-1 and insulin receptor signalling), in addition to the genes related to steroid synthesis, energy regulation, sphingosine metabolism, cellular differentiation, and glycine, serine and threonine metabolism (Figure 1B).
Figure 1. Network-assisted transcription profiling.
PQQ-influenced genes belong to multiple activity categories. The −log of the P-values relative to the PQQ+ group are indicated, with P<0.05 denoted by the vertical grey line. Dietary groups correspond to: PQQ-deficient (PQQ−), black bars; short-term PQQ-deficient group (PQQ+/−), grey bars; and PQQ-repleted (PQQ−/+), white bars. (A) Canonical clusters and relationships and (B) potential pathways and processes affected by PQQ. NS (not significant) indicates P>0.4.
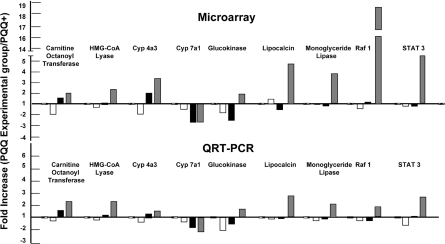
qRT-PCR analysis for selected array genes
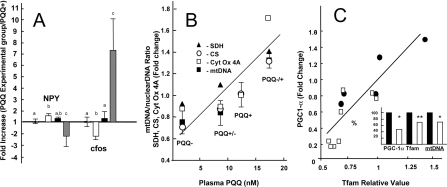
Overall, an excellent correspondence was observed for the genes chosen for microarray and the qRT-PCR data comparisons (Figure 2). Likewise, changes in c-fos and NPY (Figure 3A) were responsive to the changes in PQQ status, in keeping with what may be inferred from the IPA for functional and canonical clusters. In Figure 3(B), the relationships between plasma PQQ levels and the fold changes for citrate synthase, succinic acid dehydrogenase and cytochrome oxidase taken from the microarray data (see Supplementary Figures S1 and S2) and changes for the mtDNA/nuclearDNA (see also Table 1) are shown. These results are in keeping with previous experiments that have used both morphological analysis, and estimates of the mtDNA/nuclear DNA ratio or cytochrome oxidase activity, as indices of the qRT-PCR mitochondrial levels [1–4]. As additional validation, Tfam and PGC-1α transcripts were examined in a separate experiment (Figure 3C). Tfam encodes a transcription factor that is an important activator of mitochondrial transcription. PGC-1α is a transcriptional co-activator that regulates the genes involved in energy metabolism. Both PGC-1α and Tfam were influenced by changes in dietary PQQ status in a manner consistent with the gene array data presented.
Figure 2. qRT-PCR validation of selected genes.
Expression profiles were obtained for carnitine-O-octanoyltransferase, HMG-CoA lyase, CYP4a3, CYP7a1, glucokinase, lipocalin 2, monoacylglycerol lipase, Raf1, and STAT3 and closely match the profiles observed in the microarray analysis (Tables 1–4). Values represent fold changes in expression relative to a corresponding control group. Ratios: Control, PQQ+/PQQ+ (=1.0), PQQ deficient, PQQ−/PQQ+ (white bars); PQQ partially depleted PQQ(+/−)/PQQ+ (black bars); PQQ-repleted PQQ(−/+)/PQQ− (grey bars).
Figure 3. NPY, c-fos, mtDNA/nuclear DNA ratio, and relative changes in succinic acid dehydrogenase, citrate synthetase, cytochrome oxidase-related transcripts, PGC-1α and Tfam in response to dietary PQQ status.
(A) Changes in NPY and c-fos levels in response to changes in PQQ exposure. Values represent fold changes in expression relative to a corresponding control group. Ratios: Control, PQQ+/PQQ+ (=1.0), PQQ deficient, PQQ−/PQQ+ (white bars); PQQ partially depleted PQQ(+/−)/PQQ+ (black bars); PQQ-repleted PQQ(−/+)/PQQ− (grey bars). The superscript letter values indicate that there was a difference between groups (P<0.05 using a Dunnett's post-test). (B) Plasma PQQ and the relative content or fold changes for liver mtDNA and succinic acid dehydrogenase (SDH), citrate synthetase (CS) and cytochrome oxidase-related transcripts (Cyt OX). Values for r2 (plasma PQQ compared with relative fold-changes) ranged from approx. 0.72 to 70.95. (C) Relationship of Tfam to PGC-1α (relative changes in transcript levels) in PQQ− rats (□) or PQQ+ rats (●). Changes in PGC-1α and Tfam transcripts levels (estimated by qRT-PCR) and the mtDNA/nuclear DNA ratio are expressed as a percentage of control values (insert). *P<0.05; **P<0.2.
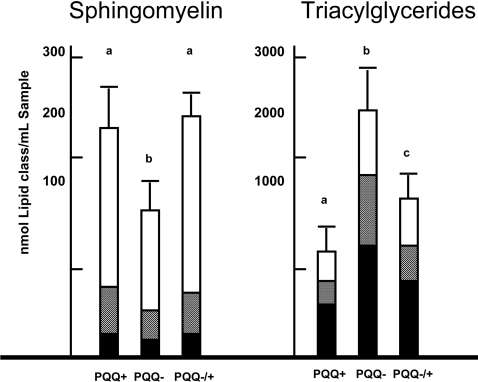
Plasma lipids
Figure 4 shows the levels of triacylglycerols and sphingosine and the relative composition of fatty acid classes contained in each group of rats (e.g. the percentage of saturated, mono-unsaturated and polyunsaturated fatty acids). On the basis of the results from the gene array assessment, it was predicted that triacylglycerols levels would be elevated and sphingosine levels decreased in response to PQQ dietary deficiency.
Figure 4. Plasma sphingomyelin and triacylglycerol levels.
PQQ exposure had a significant effect on plasma sphingomyelin and triacylglycerol levels. The superscript letters indicate that there was a difference between groups (P<0.05 using a Dunnett's post-test). The major fatty acid classes contained in the sphingomyelin and triacylglycerol fractions are: polyunsaturated fatty acids (black), mono-unsaturated fatty acids (grey) and saturated fatty acids (white bars). Results are means+S.E.M (n=6).
DISCUSSION
A wide range of functions has been attributed to PQQ (e.g. improved growth, immune responsiveness and reproductive performance). We have emphasized previously the role of PQQ in mitochondrial function and biogenesis [1–4,20,21]; however, the biochemical and cellular mechanisms responsible for such effects remain obscure. Accordingly, in order to better understand the effects of PQQ and its mechanism of action, we performed expression microarray analysis on rats subject to differing PQQ exposure protocols. An ASD was chosen because it could be formulated to be nutritionally complete with all known essential nutrients, yet free of PQQ [1–4]. It was noteworthy that merely supplementing the ASD with micromolar amounts of PQQ improves the global expression of numerous genes and supports growth and reproduction similar to mice or rats fed LC diets [1–4].
In the present study we terminated the experiments at a time when rats are sexually mature. We have observed previously in mice that, although differences in body weight may occur at weaning, differences in body weight (PQQ− compared with PQQ+) are not apparent by 6–8 weeks post-weaning [4]. Thus the inability to gain weight or growth was in part excluded as a variable. Regarding plasma PQQ levels, the values for ASD-fed rats reflected the dietary exposure to PQQ with the caveat that the lower values for plasma PQQ for rats fed the LC diets may reflect variable intake, PQQ adduct formation because of the complexity of the LC diet or decreased bioavailability of PQQ as a result of LC processing and preparation. Another caveat is that the GDH assay used for PQQ measurements only measures ‘free’ PQQ and not PQQ bound to protein or in the form of IPQ, the principal PQQ–amino acid adduct. The extent to which IPQ may contribute to the total functional body pool of PQQ remains to be fully assessed.
When defined in functional categories or clusters, genes that were influenced or changed significantly in response to PQQ deficiency were rapidly reversed or normalized upon repletion with PQQ. This was particularly apparent for genes associated with cell signalling, stress and apoptosis. Components of the JAK/STAT signalling pathway seemed to be a major target, which is important for the regulation of cellular responses to a large number of cytokines and growth factors. The apparent changes observed in c-fos (up-regulation) and NPY (down-regulation) mRNA levels are consistent with alterations in JAK/STAT-related pathways [36–39].
The responsiveness of BAX and STAT3 to changes in PQQ status was also viewed as important. STAT3 is activated in response to various cytokines and growth factors and is involved in the control of respiration by mitochondria [46]. STAT3-null mouse embryos do not develop beyond embryonic day 7 [47]. STATs in combination with JAKs are cell signalling alternatives to the second messenger systems [46]. STAT3 is also present in mitochondria and when down-regulated can affect the activities of complexes I and II in the electron transport chain [46]. The BAX gene was the first identified as the pro-apoptotic member of the Bcl-2 protein family [48,49]. The majority of BAX is found in the cytosol, but, upon initiation of apoptotic signalling, BAX inserts into the outer mitochondrial membrane and aids in opening of the mitochondrial voltage-dependent anion channels and plays a role in mitochondrial outer membrane permeabilization. Such observations are consistent with the relative changes associated with mitochondriogenesis (e.g. a lower ratio of mtDNA to nuclear DNA, lower levels of liver PGC-1α and Tfam mRNA, and the disproportionately high number of mitochondrial-related genes that respond significantly to changes in PQQ status). The results are also consistent with previous observations showing that cytochrome oxidase and succinic acid dehydrogenase enzymatic activity is relatively lower in PQQ-deprived rats, a good indirect measure of mitochondrial numbers [1,20]. Moreover, the elevation in triacylglycerol and decrease in sphingosine levels provide additional validation and a functional consequence in the altering transcriptional networks by PQQ.
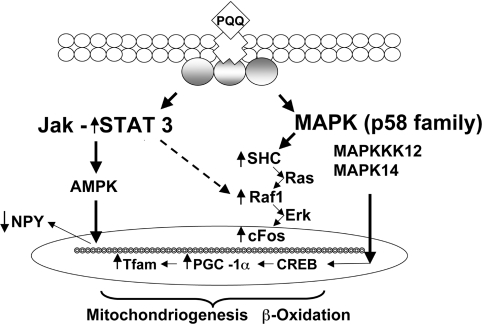
As a summary, the changes discussed above are highlighted in Figure 5, which integrates how varying PQQ exposure alters cell signalling. JAK/STAT and MAPK-related pathways seem particularly influenced by PQQ. As examples, MAPK14, a kinase in the p38 kinase family, is required for BAX translocation to mitochondria, cytochrome c release and apoptosis [49,50]. MAPK14 was up-regulated by PQQ deprivation (Table 2). In contrast, MAPKKK (MAPK kinase kinase) 12 was up-regulated by PQQ repletion (Table 2). The up-regulation of MAPKKK12 can be linked to CREB activation and increased mitochondriogenesis [50], in keeping with our previous observations that PQQ can influence CREB phosphorylation and PGC-1α and Tfam mRNA expression [20], as well as from the results presented in Figure 3; these results also indicate that JAK/STAT signalling is activated (e.g. based in part on changes in STAT3, NPY and BAX). PQQ seems to modulate cell signalling pathways important for both apoptosis [49–51] and mitochondrial assembly [52]. As a final point, it is also of importance that among the antioxidant enzymes influenced by PQQ, significant changes in thioredoxin-related transcripts were observed (Table 4). Thioredoxin not only plays a role in anti-oxidative defence, but also catalyses the inter- and intra-molecular disulfide bonds in proteins [53]. An important activity is S-glutathionylation and deglutathionylation to protect thiol groups in proteins from oxidation, which is particularly important for the regulation of transcription factor activity [53].
Figure 5. Proposed pathways regulated by dietary PQQ.
Several major signalling pathways are modulated by changes in PQQ dietary status. Given the broad range of responses, PQQ is proposed to bind to a cell surface receptor that triggers responses from one or more transducers. The JAK/STAT3 pathway is highlighted, because STAT3 is up-regulated and influences the expression of a variety of genes that play key roles in cellular processes such as cell proliferation, differentiation and apoptosis. The link to AMPK is based in part on the down-regulation of NPY. Cross-talk between Raf-1 and STAT3 is inferred (broken arrow), because of the known inter-relationships between MAPK/ERK signalling and STAT3, and the up-regulation of c-Fos, Raf-1 and Ras following exposure to either dietary or pharmacological levels of PQQ. The MAPK p58 family (e.g., MAPKKK12 and MAPK14) is also involved. The combination of the two cell signalling pathways can lead to the up-regulation or activation of PGC-1α, PPARα (peroxisome-proliferator-activated receptor α) and CREB as end products.
Taken together, the observations suggest that a closer examination of PQQ and its related derivatives is justified, given that compromised mitochondrial function is easily linked to diseases and metabolic disorders [54]. For example, in contrast with other compounds that provoke changes in mitochondrial number and function (quercetin, hydroxytyrosol and resveratrol [36]), the effects of PQQ occur at low levels of intake (at nano- to micro-molar levels in diets in contrast with near millimolar levels) and are dynamic, i.e. appear to be modulated by not only the amount but also the duration of PQQ exposure.
Online data
AUTHOR CONTRIBUTION
Eskouhie Tchaparian and Robert Rucker organized and wrote the manuscript. Eskouhie Tchaparian and Lisa Marshal generated the gene microarray data. Kathryn Bauerly, Winyoo Chowanadisai, Michael Satre and Calliandra Harris independently contributed to efforts related to validation of the microarray data or analysis of PQQ and/or animal husbandry and maintenance. Gene Cutler assisted with data analysis and informatics.
FUNDING
This work was supported in part by the Mitsubishi Gas Chemical Company and the Center for Health Related Research, University of California Davis.
References
- 1.Bauerly K. A., Storms D., Harris C. B., Hajizadeh S., Sun M. Y., Cheung C. P., Satre M. A., Fascetti A. J., Tchaparian E., Rucker R. B. Pyrroloquinoline quinone nutritional status alters lysine metabolism and modulates mitochondrial DNA content in the mouse and rat. Biochim. Biophys. Acta. 2006;1760:1741–1748. doi: 10.1016/j.bbagen.2006.07.009. [DOI] [PubMed] [Google Scholar]
- 2.Stites T., Storms D., Bauerly K., Mah J., Harris C., Fascetti A., Rogers Q., Tchaparian E., Satre M., Rucker R. B. Pyrroloquinoline quinone modulates mitochondrial quantity and function in mice. J. Nutr. 2006;136:390–396. doi: 10.1093/jn/136.2.390. [DOI] [PubMed] [Google Scholar]
- 3.Steinberg F., Stites T. E., Anderson P., Storms D., Chan I., Eghbali S., Rucker R. B. Pyrroloquinoline quinone improves growth and reproductive performance in mice fed chemically defined diets. Exp. Biol. Med. 2003;228:160–166. doi: 10.1177/153537020322800205. [DOI] [PubMed] [Google Scholar]
- 4.Steinberg F. M., Gershwin M. E., Rucker R. B. Dietary pyrroloquinoline quinone: growth and immune response in BALB/c mice. J. Nutr. 1994;124:744–753. doi: 10.1093/jn/124.5.744. [DOI] [PubMed] [Google Scholar]
- 5.Ishii T., Akagawa M., Naito Y., Handa O., Takagi T., Mori T., Kumazawa S., Yoshikawa T., Nakayama T. Pro-oxidant action of pyrroloquinoline quinone: characterization of protein oxidative modifications. Biosci. Biotechnol. Biochem. 2010;74:663–666. doi: 10.1271/bbb.90764. [DOI] [PubMed] [Google Scholar]
- 6.Hamagishi Y., Murata S., Kamei H., Oki T., Adachi O., Ameyama M. New biological properties of pyrroloquinoline quinone and its related compounds: inhibition of chemiluminescence, lipid peroxidation and rat paw edema. J. Pharmacol. Exp. Ther. 1990;255:980–985. [PubMed] [Google Scholar]
- 7.Hirakawa A., Shimizu K., Fukumitsu H., Furukawa S. Pyrroloquinoline quinone attenuates iNOS gene expression in the injured spinal cord. Biochem. Biophys. Res. Commun. 2009;378:308–312. doi: 10.1016/j.bbrc.2008.11.045. [DOI] [PubMed] [Google Scholar]
- 8.Li H., Liu S. Q., Peng H., Zhang N. Pyrroloquinoline quinone enhances regeneration of transected sciatic nerve in rats. Chin. J. Traumatol. 2005;8:225–229. [PubMed] [Google Scholar]
- 9.Zhang J. J., Zhang R. F., Meng X. K. Protective effect of pyrroloquinoline quinone against Aβ-induced neurotoxicity in human neuroblastoma SH-SY5Y cells. Neurosci. Lett. 2009;464:165–169. doi: 10.1016/j.neulet.2009.08.037. [DOI] [PubMed] [Google Scholar]
- 10.Zhang P., Xu Y., Sun J., Li X., Wang L., Jin L. Protection of pyrroloquinoline quinone against methylmercury-induced neurotoxicity via reducing oxidative stress. Free Radical Res. 2009;43:224–233. doi: 10.1080/10715760802677348. [DOI] [PubMed] [Google Scholar]
- 11.Zhu B. Q., Simonis U., Cecchini G., Zhou H. Z., Li L., Teerlink J. R., Karliner J. S. Comparison of pyrroloquinoline quinone and/or metoprolol on myocardial infarct size and mitochondrial damage in a rat model of ischemia/reperfusion injury. J. Cardiovasc. Pharmacol. Ther. 2006;11:119–128. doi: 10.1177/1074248406288757. [DOI] [PubMed] [Google Scholar]
- 12.Zhu B. Q., Zhou H. Z., Teerlink J., Karliner J. S. Pyrroloquinoline quinone (PQQ) decreases myocardial infarct size and improves cardiac function in rat models of ischemia and ischemia/reperfusion. Cardiovasc. Drugs Ther. 2004;18:421–431. doi: 10.1007/s10557-004-6219-x. [DOI] [PubMed] [Google Scholar]
- 13.Nunome K., Miyazaki S., Nakano M., Iguchi-Ariga S., Ariga H. Pyrroloquinoline quinone prevents oxidative stress-induced neuronal death probably through changes in oxidative status of DJ-1. Biol. Pharm. Bull. 2008;31:1321–1326. doi: 10.1248/bpb.31.1321. [DOI] [PubMed] [Google Scholar]
- 14.Liu S., Li H., Ou Yang J., Peng H., Wu K., Liu Y., Yang J. Enhanced rat sciatic nerve regeneration through silicon tubes filled with pyrroloquinoline quinone. Microsurgery. 2005;25:329–337. doi: 10.1002/micr.20126. [DOI] [PubMed] [Google Scholar]
- 15.Kasahara T., Kato T. Nutritional biochemistry: a new redox-cofactor vitamin for mammals. Nature. 2003;422:832. doi: 10.1038/422832a. [DOI] [PubMed] [Google Scholar]
- 16.Felton L. M., Anthony C. Biochemistry: role of PQQ as a mammalian enzyme cofactor? Nature. 2005;433:E10. doi: 10.1038/nature03322. [DOI] [PubMed] [Google Scholar]
- 17.Rucker R.B., Storms D., Sheets A., Tchaparian E., Fascetti A. Biochemistry: is pyrroloquinoline quinone a vitamin? Nature. 2005;433:E10–E11. doi: 10.1038/nature03323. [DOI] [PubMed] [Google Scholar]
- 18.Kumazawa T., Hiwasa T., Takiguchi M., Suzuki O., Sato K. Activation of Ras signaling pathways by pyrroloquinoline quinone in NIH3T3 mouse fibroblasts. Int. J. Mol. Med. 2007;19:765–770. [PubMed] [Google Scholar]
- 19.Naito Y., Kumazawa T., Kino I., Suzuki O. Effects of pyrroloquinoline quinone (PQQ) and PQQ-oxazole on DNA synthesis of cultured human fibroblasts. Life Sci. 1993;52:1909–1915. doi: 10.1016/0024-3205(93)90631-c. [DOI] [PubMed] [Google Scholar]
- 20.Chowanadisai W., Bauerly K. A., Tchaparian E., Wong A., Cortopassi G. A., Rucker R. B. Pyrroloquinoline quinone stimulates mitochondrial biogenesis through cAMP response element-binding protein phosphorylation and increased PGC-1α expression. J. Biol. Chem. 2010;285:142–152. doi: 10.1074/jbc.M109.030130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Rucker R. B., Chowanadisai W., Nakano M. Potential physiological importance of pyrroloquinoline quinone. Altern. Med. Rev. 2009;14:268–277. [PubMed] [Google Scholar]
- 22.Mitchell A. E., Jones A. D., Mercer R. S., Rucker R. B. Characterization of pyrroloquinoline quinone amino acid derivatives by electrospray ionization mass spectrometry and detection in human milk. Anal. Biochem. 1999;269:317–325. doi: 10.1006/abio.1999.4039. [DOI] [PubMed] [Google Scholar]
- 23.Kumazawa T., Sato K., Seno H., Ishii A., Suzuki O. Levels of pyrroloquinoline quinone in various foods. Biochem. J. 1995;307:331–333. doi: 10.1042/bj3070331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kumazawa T., Seno H., Suzuki O. Failure to verify high levels of pyrroloquinoline quinone in eggs and skim milk. Biochem. Biophys. Res. Commun. 1993;193:1–5. doi: 10.1006/bbrc.1993.1581. [DOI] [PubMed] [Google Scholar]
- 25.Kumazawa T., Seno H., Urakami T., Matsumoto T., Suzuki O. Trace levels of pyrroloquinoline quinone in human and rat samples detected by gas chromatography/mass spectrometry. Biochim. Biophys. Acta. 1992;1156:62–66. doi: 10.1016/0304-4165(92)90096-d. [DOI] [PubMed] [Google Scholar]
- 26.Rudolf J. L., Bauerly K. A., Tchaparian E., Rucker R. B., Mitchell A. E. The influence of diet composition on phase I and II biotransformation enzyme induction. Arch, Toxicol. 2008;82:893–901. doi: 10.1007/s00204-008-0310-1. [DOI] [PubMed] [Google Scholar]
- 27.Matsushita K., Arents J. C., Bader R., Yamada M., Adachi O., Postma P. W. Escherichia coli is unable to produce pyrroloquinoline quinone (PQQ) Microbiology. 1997;143:3149–3156. doi: 10.1099/00221287-143-10-3149. [DOI] [PubMed] [Google Scholar]
- 28.Smidt C. R., Bean-Knudsen D., Kirsch D. G., Rucker R. B. Does the intestinal microflora synthesize pyrroloquinoline quinone? Biofactors. 1991;3:53–59. [PubMed] [Google Scholar]
- 29.Smidt C. R., Unkefer C. J., Houck D. R., Rucker R. B. Intestinal absorption and tissue distribution of [14C]pyrroloquinoline quinone in mice. Proc. Soc. Biol. Med. 1991;197:27–31. doi: 10.3181/00379727-197-43219. [DOI] [PubMed] [Google Scholar]
- 30.Ramakrishnan R., Dorris D., Lublinsky A., Nguyen A., Domanus M., Prokhorova A., Gieser L., Touma E., Lockner R., Tata M., et al. An assessment of Motorola CodeLink microarray performance for gene expression profiling applications. Nucleic Acids Res. 2002;30:e30. doi: 10.1093/nar/30.7.e30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Diez D., Alvarez R., Dopazo A. Codelink: an R package for analysis of GE Healthcare gene expression bioarrays. Bioinformatics. 2007;23:1168–1169. doi: 10.1093/bioinformatics/btm072. [DOI] [PubMed] [Google Scholar]
- 32.Wu W., Dave N., Tseng G. C., Richards T., Xing E. P., Kaminski N. Comparison of normalization methods for CodeLink Bioarray data. BMC Bioinform. 2005;6:309. doi: 10.1186/1471-2105-6-309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Tusher V. G., Tibshirani R., Chu G. Significance analysis of microarrays applied to the ionizing radiation response. Proc. Natl. Acad. Sci. U.S.A. 2001;98:5116–5121. doi: 10.1073/pnas.091062498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Zang S., Guo R., Zhang L., Lu Y. Integration of statistical inference methods and a novel control measure to improve sensitivity and specificity of data analysis in expression profiling studies. J. Biomed. Inform. 2007;40:552–560. doi: 10.1016/j.jbi.2007.01.002. [DOI] [PubMed] [Google Scholar]
- 35.Zhang S. A comprehensive evaluation of SAM, the SAM R-package and a simple modification to improve its performance. BMC Bioinformatics. 2007;8:230. doi: 10.1186/1471-2105-8-230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Cadenas E., Packer L., Surh Y-J., Dong Z. Dietary Modulation of Cell Signaling Pathways. Boca Raton: CRC Press, Taylor and Francis Group; 2008. pp. 1–465. [Google Scholar]
- 37.Hagmeyer B. M., Angel P., van Dam H. Modulation of AP-1/ATF transcription factor activity by the adenovirus-E1A oncogene products. BioEssays. 1999;17:621–629. doi: 10.1002/bies.950170708. [DOI] [PubMed] [Google Scholar]
- 38.Kaipio K., Kallio J., Pesonen U. Mitochondrial targeting signal in human neuropeptide Y gene. Biochem. Biophys. Res. Commun. 2005;337:633–640. doi: 10.1016/j.bbrc.2005.09.093. [DOI] [PubMed] [Google Scholar]
- 39.Rebagliati I., Ricci C., Zaninovich A., Tang F. Neuropeptide Y suppressed the T3-induced rise in brown fat mitochondrial respiration in hypothyroid rats. Endocr. Res. 2001;27:367–375. doi: 10.1081/erc-100106014. [DOI] [PubMed] [Google Scholar]
- 40.Asin-Cayuela J., Gustafsson C. M. Mitochondrial transcription and its regulation in mammalian cells. Trends Biochem. Sci. 2007;32:111–117. doi: 10.1016/j.tibs.2007.01.003. [DOI] [PubMed] [Google Scholar]
- 41.Cochem H. M., Kelso G. F., James A. M., Ross M. F., Trnka J., Mahendiran T., Asin-Cayuela J., Blaikie F. H., Manas A. R., Porteous C. M., et al. Mitochondrial targeting of quinones: therapeutic implications. Mitochondrion. 2007;7(Suppl. 1):S94–S102. doi: 10.1016/j.mito.2007.02.007. [DOI] [PubMed] [Google Scholar]
- 42.Bookout A. L., Cummins C. L., Mangelsdorf D. J., Pesola J. M., Kramer M. F. High-throughput real-time quantitative reverse transcription PCR. Curr. Protoc. Mol. Biol. 2006;15 doi: 10.1002/0471142727.mb1508s73. Unit 15.8. [DOI] [PubMed] [Google Scholar]
- 43.Bookout A. L., Mangelsdorf D. J. Quantitative real-time PCR protocol for analysis of nuclear receptor signaling pathways. Nucl. Recept. Signaling. 2003;1:e012. doi: 10.1621/nrs.01012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Wong A., Cortopassi G. Reproducible quantitative PCR of mitochondrial and nuclear DNA copy number using the LightCycler. Methods Mol. Biol. 2002;197:129–137. doi: 10.1385/1-59259-284-8:129. [DOI] [PubMed] [Google Scholar]
- 45.Ameyama M., Nonobe M., Shinagawa E., Matsushita K., Adachi O. Method of enzymatic determination of pyrroloquinoline quinone. Anal. Biochem. 1985;151:263–267. doi: 10.1016/0003-2697(85)90174-5. [DOI] [PubMed] [Google Scholar]
- 46.Wegrzyn J., Potla R., Chwae Y. J., Sepuri N. B., Zhang Q., Koeck T., Derecka M., Szczepanek K., Szelag M., Gornicka A., et al. Function of mitochondrial Stat3 in cellular respiration. Science. 2009;323:793–797. doi: 10.1126/science.1164551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Takeda K., Noguchi K., Shi W., Tanaka T., Matsumoto M., Yoshida N., Kishimoto T., Akira S. Targeted disruption of the mouse Stat3 gene leads to early embryonic lethality. Proc. Natl. Acad. Sci. U.S.A. 1997;94:3801–3804. doi: 10.1073/pnas.94.8.3801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Wang H. G., Rapp U. R., Reed J. C. Bcl-2 targets the protein kinase Raf-1 to mitochondria. Cell. 1996;87:629–638. doi: 10.1016/s0092-8674(00)81383-5. [DOI] [PubMed] [Google Scholar]
- 49.Van Laethem A., Van Kelst S., Lippens S., Declercq W., Vandenabeele P., Janssens S., Vandenheede J. R., Garmyn M., Agostinis P. Activation of p38 MAPK is required for Bax translocation to mitochondria, cytochrome c release and apoptosis induced by UVB irradiation in human keratinocytes. FASEB J. 2004;18:1946–1948. doi: 10.1096/fj.04-2285fje. [DOI] [PubMed] [Google Scholar]
- 50.Wang H. G., Rapp U. R., Reed J. C. Bcl-2 targets the protein kinase Raf-1 to mitochondria. Cell. 1996;87:629–638. doi: 10.1016/s0092-8674(00)81383-5. [DOI] [PubMed] [Google Scholar]
- 51.Vermeulen L., Berghe W. V., Beck I. M., De Bosscher K., Haegeman G. The versatile role of MSKs in transcriptional regulation. Trends Biochem. Sci. 2009;34:311–318. doi: 10.1016/j.tibs.2009.02.007. [DOI] [PubMed] [Google Scholar]
- 52.Spiegelman B. M. Transcriptional control of mitochondrial energy metabolism through the PGC1 coactivators. Novartis Found. Symp. 2007;287:60–63. [PubMed] [Google Scholar]
- 53.Kalinina E. V., Chernov N. N., Saprin A. N. Involvement of thio-, peroxi-, and glutaredoxins in cellular redox-dependent processes. Biochemistry (Moscow) 2008;73:1493–1510. doi: 10.1134/s0006297908130099. [DOI] [PubMed] [Google Scholar]
- 54.Johannsen D. L., Ravussin E. The role of mitochondria in health and disease. Curr. Opin. Pharmacol. 2009;9:780–786. doi: 10.1016/j.coph.2009.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.