Abstract
Song in oscine birds is a learned behavior that plays important roles in breeding. Pronounced seasonal differences in song behavior, and in the morphology and physiology of the neural circuit underlying song production are well documented in many songbird species. Androgenic and estrogenic hormones largely mediate these seasonal changes. While much work has focused on the hormonal mechanisms underlying seasonal plasticity in songbird vocal production, relatively less work has investigated seasonal and hormonal effects on songbird auditory processing, particularly at a peripheral level. We addressed this issue in Gambel’s white-crowned sparrow (Zonotrichia leucophrys gambelii), a highly seasonal breeder. Photoperiod and hormone levels were manipulated in the laboratory to simulate natural breeding and non-breeding conditions. Peripheral auditory function was assessed by measuring the auditory brainstem response (ABR) and distortion product otoacoustic emissions (DPOAEs) of males and females in both conditions. Birds exposed to breeding-like conditions demonstrated elevated thresholds and prolonged peak latencies compared with birds housed under non-breeding-like conditions. There were no changes in DPOAEs, however, which indicates that the seasonal differences in ABRs do not arise from changes in hair cell function. These results suggest that seasons and hormones impact auditory processing as well as vocal production in wild songbirds.
Keywords: songbird, hormone, season, auditory, ABR
INTRODUCTION
Seasons and hormones play an important role in coordinating the breeding activity of many animals. In songbirds, song is important in mate attraction and territorial defense. During the breeding season, songs are typically produced more often, are longer, and are more stereotyped in structure than during the rest of the year (Brenowitz 2008). Morphological and physiological changes occur in the underlying song control circuitry (Brenowitz 2008). During the breeding season, some song nuclei are larger (Nottebohm 1981; Brenowitz 1991; Brenowitz et al. 1998) and fire spontaneously at a higher rate (Park et al. 2005; Meitzen et al. 2007b). These seasonal differences in song behavior and neural circuitry are primarily regulated by the sex steroid hormones testosterone and estrogen (Marler et al. 1988; Tramontin et al. 2003; Soma et al. 2004; Meitzen et al. 2007a).
Many studies have used songbirds as model system for examining the effects of seasons and hormones on vocal production (Brenowitz 2008), but relatively few studies have investigated seasonal and hormonal influences on songbird auditory processing. Seasonal changes in auditory processing have been reported for other animals. Seasonal changes in frequency tuning and temporal response properties have been found in the midbrain inferior colliculus of Northern leopard frogs (Goense and Feng 2005). In female green tree frogs (Hyla cinerea), testosterone increases midbrain multiunit thresholds to pure tones that lie within the range of the male advertisement calls (Miranda and Wilczynski 2009b), and females that have mated show frequency-specific, decreased multiunit response strength to noise bursts (Miranda and Wilczynski 2009a). Recordings from auditory nerve afferents in female midshipman fish (Porichthys notatus) demonstrate more precise phase locking during the breeding season, and this auditory phenotype can be induced in non-reproductive fish by administering testosterone or 17β-estradiol (Sisneros and Bass 2003; Sisneros et al. 2004). Both sex-specific and seasonal differences have been found in click-evoked otoacoustic emissions of Rhesus monkeys (McFadden et al. 2006; McFadden 2009).
Behavioral, physiological and morphological observations raise the possibility that seasons and hormones also affect auditory processing in songbirds. Male and female zebra finches housed on a long-day photoperiod learned an operant song discrimination paradigm faster than those housed on a short-day photoperiod (Cynx and Nottebohm 1992), suggesting that day length may influence song perception (though the effect simply may instead reflect seasonal differences in activity or motivation). Similarly, some evidence suggests that estrogen treatment modulates the song-elicited behavioral responses of female birds (Vyas et al. 2009). There are several reports of seasonal and/or hormonal effects on physiological processing in forebrain areas known to respond to auditory stimuli. For instance, the spontaneous electrophysiological profile of neurons in the song nucleus HVC varies as a function of season in male and female canaries (Del Negro and Edeline 2002) and Del Negro and colleagues reported that photoperiod and breeding condition affected song selective neural responses in HVC (Del Negro et al. 2000; Del Negro et al. 2005). Similar effects were found on both passive and active electrophysiological properties of neurons in the white-crowned sparrow robust nucleus of the arcopallium, but the electrical properties of HVC neurons were stable across seasons (Meitzen et al. 2009b). Seasonal effects may also exist in the caudomedial nidopallium (NCM), a forebrain auditory region (Terleph et al. 2008). Estradiol increases evoked activity in NCM (Tremere et al. 2009), and modulates song-induced expression of the immediate early gene egr-1 in several auditory nuclei (Maney et al. 2006; Sanford et al. 2009). Additionally, a recent study using diffusor tensor imaging suggests that the volume of NCM is larger in breeding-condition European starlings (Sturnus vulgaris) (De Groof et al. 2009).
Most of this literature has focused on seasonal changes in auditory function in the forebrain. Seasonal cues, however, may also influence auditory processing at the peripheral level. Seasonal changes at the periphery may be conserved throughout the auditory pathway, giving rise to the seasonal effects observed in higher processing centers. In this respect, it is interesting to note that expression of the alpha subtype of the estrogen receptor (ERα), and aromatase (which catalyzes the synthesis of estrogen from testosterone), were recently reported in the inner ear of zebra finches (Noirot et al. 2009). We have also observed ERα in hair cells and support cells and both ERα and the androgen receptor (AR) in ganglion cells of the inner ear of young chickens and adult white-crowned sparrows (Wang, Brenowitz, Rubel, McCullar, and Oesterle unpublished observations). Lucas and colleagues examined seasonal changes in the amplitude and latencies of evoked responses in six different species of birds (Lucas et al. 2002; Lucas et al. 2007). Their data suggest that the effect of seasonal cues differs between species, but these authors only measured threshold sensitivity in one species (House sparrow, Passer domesticus). Those results were inconclusive, however, due to inadequate sample sizes (Henry and Lucas 2009).
Our study addresses the issue of whether hormonal and photoperiod manipulations that mimic the breeding and non-breeding season affect peripheral auditory processing in Gambel’s white-crowned sparrow (Zonotrichia leucophrys gambelii), a migratory species with highly seasonal breeding. Auditory brainstem response (ABR) and distortion product otoacoustic emission (DPOAE) recordings were used as a measure of peripheral auditory processing.
The ABR is a short-latency neural response typically emitted 10–15 msec after the presentation of an auditory stimulus (Hall 1992). ABR recording has a long history of use as a diagnostic measure of peripheral and brainstem auditory function in humans and animals (Jewett et al. 1970; Achor and Starr 1980; Despland and Galambos 1980; Liberman et al. 2006), and this method has been used to assess avian auditory function (Corwin et al. 1982; Brown-Borg et al. 1987; Burkard et al. 1994; Woolley et al. 2001; Brittan-Powell et al. 2002; Lucas et al. 2002).
Otoacoustic emissions are low-intensity sounds generated by the compressively nonlinear cochlear amplification process of the inner ear (Kemp 1978; Probst et al. 1991; Kemp 2002). In mammals, it is thought that the outer hair cells of the cochlea produce the amplification responsible for emission generation (Dallos 2008; Dallos et al. 2008). Although the exact cellular origin of otoacoustic emission production in non-mammalian vertebrates is currently unknown (Bergevin et al. 2008), DPOAEs can still be effectively used as an indicator of avian inner ear function (Kettembeil et al. 1995; Bergevin et al. 2008).
We report that ABR thresholds were elevated and ABR peak latencies were prolonged in breeding birds, whereas DPOAE amplitudes and thresholds were not affected. Our results show a seasonal effect on auditory thresholds, and suggest that the effect originates post-synaptic to the hair cells.
METHODS
Subjects
Adult male (n=24) and female (n=24) Gambel’s white-crowned sparrows (Zonotrichia leucophrys gambelii) were collected during autumn and spring migrations between 2006 and 2008. Most birds were captured in mist nets in eastern Washington State; a small subset were captured in Davis, California. Birds were housed in outdoor aviaries at the University of Washington for up to 30 weeks before being moved to indoor aviaries. Once inside, all birds were housed in groups on a short-day photoperiod (SD, 8 hr light: 16hr dark) for a minimum of 10 weeks to ensure sensitivity to the stimulating effects of hormones and photoperiod (i.e., photosensitive) (Wingfield et al. 1979). Food and water were available ad libitum. All procedures were approved by the Institutional Animal Care and Use Committee at the University of Washington, Seattle.
Seasonal manipulations
Birds were randomly divided into two groups mimicking breeding and non-breeding conditions. To induce a non-breeding-like condition, birds were housed on a SD photoperiod as above. Birds housed on a SD photoperiod maintain regressed gonads and song nuclei and have basal plasma sex hormone levels typical of the non-breeding season (Wingfield and Farner 1978; Tramontin et al. 2000; Park et al. 2005; Meitzen et al. 2007b). To induce a breeding-like condition, birds were housed on a long day (LD; 20 hr light – 4 hr dark) photoperiod typical of their Alaskan breeding grounds Additionally, these birds were implanted subcutaneously with capsules made from SILASTIC tubing (i.d. 1.0mm; o.d. 2.0mm, length 12mm; VWR, West Chester, PA) filled with crystalline testosterone (males) or estradiol (females) (Tramontin et al. 2003). Implants were rinsed in ethanol and soaked overnight in 0.1M phosphate buffered saline prior to implantation. Supplemental hormone is necessary to raise plasma hormone levels of laboratory-housed birds to physiological levels observed in breeding birds in the wild (Smith et al. 1995). Birds were housed under these conditions for three weeks; this time period is sufficient to induce full breeding-like growth of the song circuits in male white-crowned sparrows (Tramontin et al. 2000; Meitzen et al. 2009a).
Drugs
Birds were anesthetized with 25% urethane (6 μl/g body weight) for all recordings. Body weight (mean +/− S.E.M) was 27.6 +/− 0.64 g (males) and 27.7 +/− 0.75 g (females). The total drug volume was divided evenly into three intramuscular injections separated by 30 minutes. Additional doses (0.67 μl/g) were delivered as necessary to maintain anesthetic state as assessed by toe-pinch.
Experimental set-up
All experiments took place in an acoustically isolated chamber (Acoustic Systems, Austin, TX) between 10:30 and 15:30. We prepared each bird for ABR or DPOAE recording by removing feathers from the top of the head and surrounding the left ear. We swabbed the skin with alcohol and made a small incision at the anterior portion of the dorsal midline of the skull. The skin was retracted and fascia was removed. We cleaned and dried the skull with alcohol and glued a custom-made metal post to the head. The post was securely mounted on a magnetic stand to prevent head movement. We placed the bird on an electric heating pad and maintained the body temperature at 40–42 oC using a cloacal thermal probe and digital controller (TC-1000 Temperature Controller, CWE Inc., Ardmore, PA). For a subset of birds, we placed subcutaneous needle electrodes in the left wing and right leg to monitor electrocardiogram activity throughout the experiment. An electrode in the left leg served as a single-point ground for both the electrocardiogram (when recorded) and the ABR recordings described below. We amplified electrocardiogram signals 1000x (Grass Technologies P15, West Warwick, RI), bandpass filtered them at 100–1000 Hz and displayed them on a digital oscilloscope. The output of a small speaker (Etymotics ER-2B, Elk Grove Village, IL) and microphone (Etymotics ER-10B, Elk Grove Village, IL) were enclosed within a custom-made sound delivery tube affixed to a micromanipulator. We positioned the tube flush against the skull surrounding the left external auditory meatus, and sealed it with petroleum jelly, creating a closed sound delivery system.
At the beginning of each recording session, we calibrated dB SPL values (re: 20 μPa) online using a custom written software program. Click stimuli were presented in dB peak equivalent (pe) SPL. Unless otherwise noted, all pure tones were 10 msec in duration with 2 msec rise-fall times. Clicks were 0.1 msec in duration. We presented all stimuli with alternating polarity. We generated sound stimuli using custom software and delivered them one of two ways. We routed some stimuli through a Delta-44 digital/analog converter (M-Audio, Irwindale, CA), fed them through a PA5 programmable attenuator (Tucker Davis Technologies, Alachua, FL) and delivered them directly to the speaker. We routed the remaining stimuli through an RX6 (Tucker Davis Technologies, Alachua, FL) multifunction processor that performed both digital/analogue conversion and attenuation of the signal before delivery to the speaker.
ABR recording
We recorded ABRs using standard subcutaneous needle electrodes. We placed the positive electrode at the vertex of the skull and positioned the reference electrode just dorsal to the (sound stimulated) external auditory meatus. We pre-amplified responses 100x (Grass Technologies P15 amplifier, West Warwick, RI), ran them through a MA3 amplifier with an additional 50 dB post-preamp gain (Tucker Davis, Technologies, Alachua, FL), band pass filtered them from 300–3000 Hz with a 24dB/octave roll-off (Krohn-Hite filter model 3550, Brockton, MA), fed them through a digital oscilloscope and audio monitor, digitized them at a rate of 24.400 kHz, and recorded them using a custom written software program. We sampled responses for a 20 msec window (with a 2 msec stimulus onset delay) or a 30 msec window (with a 10 msec stimulus delay). We used ABR recording in four different paradigms, each of which explored a different aspect of auditory processing. The rationale and methodological details for each paradigm are described below.
Minimum audibility paradigm
We used a total of 17 females (8 non-breeding, 9 breeding) and 24 males (13 non-breeding, 11 breeding) in this paradigm. A subset of these birds also participated in other ABR paradigms and/or DPOAE recording. One non-breeding female was housed in a rooftop aviary up until the day of recording in mid November, and was therefore exposed to day length changes natural to Washington State. Data from this female fell within the range of data from non-breeding females that were housed indoors, so we included them in the subsequent analysis.
The purpose of this experiment was to determine if breeding condition affects basic ABR parameters (i.e. thresholds and response latencies). We tested seven different frequencies (0.5, 1, 2, 3, 4, 6, 8 kHz) within the hearing range of most songbirds, and completely encompassing the spectrum of white-crowned sparrow song (Dooling et al. 2000; Meitzen et al. 2009a). We presented each stimulus at a rate of 19.6/sec starting at 70 dB SPL and decreased the amplitude in 10 dB steps; at or near threshold, we switched to 5 dB intervals. We averaged responses to suprathreshold stimuli across 500 stimulus presentations; for responses at or near threshold, we averaged across 1000 stimulus presentations and recorded the ABR at least twice to determine repeatability. We randomized stimulus presentation order for each subject.
We analyzed all ABR responses offline using custom written software. We defined threshold as the lowest intensity stimulus to elicit a repeatable, visually discernable response of any ABR wave within 10 msec of stimulus onset. To verify threshold estimates, we took two approaches. First, we gave a blind observer trained in audiology a subset (10%) of responses that represented all stimuli and conditions tested. We instructed the observer to estimate the threshold visually, using any ABR wave readily observable. Threshold estimates by one of us (MLC) and the blind observer were highly correlated (Pearson’s Correlation coefficient r = 0.989, p < 0.01, Supplementary Figure 1a). Second, we used a quantitative approach to estimate threshold for the same subset of data. For this approach, a custom written software program automatically detected the largest peak-to-peak voltage difference in a 10 msec window after stimulus onset. Offline, we calculated the maximum peak-to-peak voltage + 2 standard deviations in the pre-stimulus window. Threshold was defined as the lowest stimulus intensity tested that elicited a post-stimulus measurement greater than the pre-stimulus measurement. Quantitative and visually estimated thresholds were significantly correlated (Pearson’s r = 0.941 p <0.0001, Supplementary Figure 1b). We conclude that visually-determined thresholds are valid, representative estimates of auditory sensitivity and therefore present only these data for the remainder of this report.
We determined latency values from the time of stimulus onset for the first two positive peaks numbered sequentially with Arabic numerals as in Brittan-Powell et al. (2005). We generated latency input/output (I/O) functions for each subject and compared latencies across subjects at an iso-intensity level (70 dB SPL). Because many factors can independently affect ABR amplitude measurements (such as head size and electrode placement), we did not compare them in this study.
Forward masking frequency resolution paradigm
Because findings from the minimum audibility paradigm suggested that seasonal and hormonal effects on auditory processing are similar in male and female white-crowned sparrows (see Results section below), we only used males for the additional ABR paradigms described below. We used a total of 11 males (5 non-breeding, 6 breeding) in this experiment. All of these males also participated in the other ABR paradigms.
Breeding condition may affect aspects of auditory processing that are independent of ABR thresholds and wave peak latencies. To determine which other auditory processing parameters were worth closely investigating, we examined the structure of the white-crowned sparrow song. Males produce a single song type, consisting of five syllables: a whistle, a warble, and three buzzes (DeWolfe et al. 1974; Meitzen et al. 2009a, Figure 1). The introductory whistle is a pure tone, the frequency of which does not change seasonally (Meitzen et al. 2009a); many studies suggest that it plays a particularly important role in song identification and learning (Baptista and Morton 1981; Margoliash 1983; Soha and Marler 2000). These findings led us to hypothesize that breeding condition may influence the ability of white-crowned sparrows to resolve the frequency of the introductory whistle.
Fig. 1.
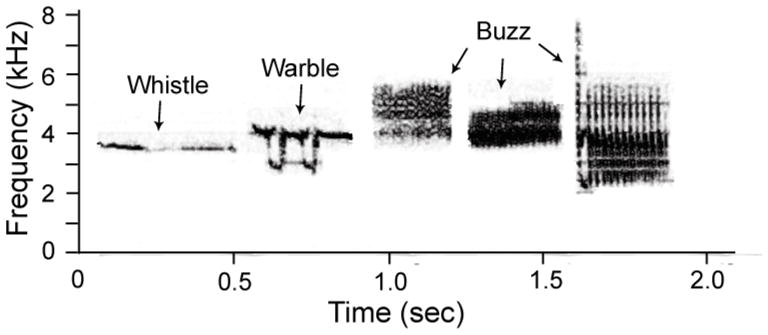
A representative white-crowned sparrow song from a breeding condition male. Songs typically consist of 5 syllables: a whistle, a warble, and 3 buzzes.
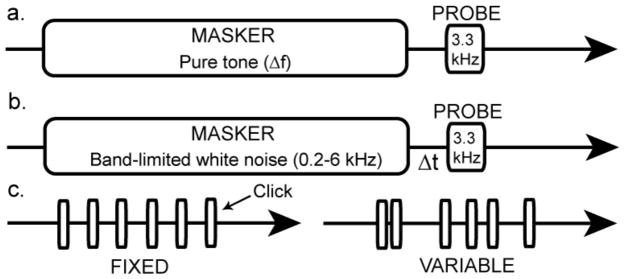
To address this possibility, we used a forward masking paradigm to examine the effect of breeding condition on ABR-derived frequency tuning curves. We set a 10 msec probe tone [3.3 kHz, roughly equivalent to the fundamental frequency of the white-crowned sparrow whistle (Meitzen et al. 2009a)] to a fixed amplitude of 70 dB SPL. Masker stimuli (2.50, 2.70, 2.90, 3.10, 3.20, 3.25, 3.35, 3.40, 3.50, 3.70 and 3.90 kHz) were 100 msec long with 16 msec rise-fall times. We presented the masker after a 10 msec onset delay (to allow for baseline noise capture). The onset of the probe tone occurred 10 msec after the offset of the masker (Figure 2a). We presented each masker-probe combination at a rate of 6.2/sec. We captured the elicited ABR during a 140 msec recording window and averaged it across 500 masker-probe presentations.
Fig. 2.
Stimulus delivery schematics for three of the ABR paradigms. Horizontal arrows indicate passage of time. a) Schematic for the forward masking frequency resolution paradigm. A 100 msec pure tone masker is varied in frequency (Δf). The offset of the masker always occurs 10 msec before the onset of 3.3 kHz probe tone. b) Schematic for the forward masking temporal adaptation paradigm. The offset of a 100 msec band-limited (0.2–6kHz) white noise masker occurs at varying time intervals (Δt) before the onset of the 3.3 kHz probe tone. c) Schematic for the temporal variability paradigm. Clicks were presented at three different rates, with both fixed (left) and variable (right) inter-peak intervals.
At the beginning of each recording, we presented the probe tone alone at 70 dB SPL, which was approximately 20–40 dB above threshold for every subject. We calculated the maximum peak-to-peak voltage difference of the ABR response online for a 10 msec measurement window (starting at the time of probe onset). After obtaining this baseline response amplitude, we began masker-probe trials. In each trial, we presented the masker at an initial amplitude of 40 dB SPL and gradually raised the amplitude by 10 dB steps. We defined threshold as the masker level necessary to reduce the maximum peak-to-peak voltage in the 10 msec measurement window by 50% or more. We verified threshold by repeating the masker-probe presentation. If two out of three repetitions failed to verify the threshold estimation, we raised the masker amplitude by 5 dB until a new threshold was verified. We randomized the order of masker frequency presentations for each subject.
We generated tuning curves offline for each subject and used the quality factor (Q10) as an indicator of sharpness of tuning. Q10 is calculated as the signal (probe) frequency divided by the bandwidth of the tuning curve at 10 dB above the tip. Larger Q values indicate sharper tuning.
Forward masking temporal adaptation paradigm
We used a total of 11 males (5 non-breeding, 6 breeding) in this paradigm. All of these males also participated in the other ABR paradigms.
White-crowned sparrows exhibit seasonal changes in the duration of some song syllables and in the length of the overall song (Meitzen et al. 2009a). These findings raise the possibility that temporal processing also changes seasonally. To address this possibility, we used a forward masking paradigm to examine the effect of breeding condition on temporal adaptation capabilities. We set a 10 msec probe tone (3.3 kHz) to a fixed amplitude of 70 dB SPL as above. The masker stimulus was band-pass filtered white-noise (0.2 – 6 kHz) with a 100 msec duration and 16 msec rise-fall times. We presented the masker after a 10 msec onset delay. The onset of the probe tone occurred 5, 10, 25 or 50 msec after the offset of the masker (Figure 2b). Masker-probe presentations occurred at a rate of 4.9/sec. We captured the elicited ABR during a 161 msec recording window and averaged them across 500 masker-probe presentations.
At the beginning of each recording, we presented the probe tone alone and calculated the maximum peak-to-peak voltage difference online for a 10 msec measurement window as above (starting at the time of probe onset). We then presented the masker at amplitudes of 40, 50, 60 and 70 dB SPL for each masker-probe interval. We randomized the order of masker-probe interval presentations for each subject.
We calculated the decrease of the probe-elicited ABR response amplitude for each masker amplitude and masker-probe interval as follows:
The more the response amplitude decreases, the greater the effect of the masker.
Temporal variability paradigm
We used a total of 10 males (4 non-breeding, 6 breeding) in this paradigm. All of these males also participated in the other ABR paradigms.
In addition to seasonal changes in syllable and song length, white-crowned sparrows also exhibit seasonal fluctuations in song structure variability. During the breeding season, song and syllable duration are less variable than in the non-breeding season (Meitzen et al. 2009a), suggesting that breeding condition may affect other aspects of temporal processing more directly related to temporal variability. To address this issue, we presented clicks (0.1 msec duration) at three presentation rates (19.6/sec, 30.3/sec and 23.2/sec). Rates were either fixed (such that the inter-click interval was constant) or variable (such that the inter-click interval was randomized, but the average rate over the course of all click presentations equaled 19.6/sec, 30.3/sec or 23.2/sec; see Figure 2c).
We initially presented clicks at 70 dB SPL and gradually decreased the amplitude by 10 dB; at or near threshold, we switched to 5 dB intervals. We averaged responses to suprathreshold stimuli across 500 stimulus presentations; at or near threshold we averaged across 1000 stimulus presentations and recorded the ABR at least twice to determine repeatability. We randomized the order of presentation rates for each subject. We determined thresholds visually and measured wave peak latencies offline (as described for the minimum audibility paradigm.)
Distortion Product Otoacoustic Emission (DPOAE) recording
Differences in auditory processing as measured by ABR recording could reflect changes in the VIIIth nerve and/or brainstem, or could reflect changes in sensory processing prior to neuronal activation at the hair cell - ganglion cell synapse (i.e. changes in the external ear canal, middle ear, or inner ear mechanics). To dissociate these possibilities, we recorded DPOAEs from 10 males (5 non-breeding, 5 breeding) and 12 females (6 non-breeding, 6 breeding). Five of the females (2 non-breeding, 3 breeding) and all of the males also participated in the ABR minimum audibility paradigm.
Sounds were delivered as described for ABR recording. Two primary tones (F1 and F2) were presented simultaneously. The frequency of F2 varied (0.5, 1.0, 1.5, 2.0, 2.5, 3.0, 3.5, 4.0, 4.5, 5.0, 5.5, 6.0, 6.5, 7.0, 7.5, 8.0, 8.5, 9.0, 9.5 kHz) but we fixed the F2/F1 ratio at 1.15. We determined this ratio value to be optimal for white-crowned sparrows during pilot studies (data not shown). We initially presented the first primary tone (F1) at an amplitude (L1) of 20 dB SPL and systematically increased the amplitude by 5 dB steps to a maximum of 90 dB SPL. The amplitude of the second primary tone (L2) was consistently 10 dB lower than L1. The cubic distortion tone, which corresponds to a frequency of 2F1-F2, is the largest distortion product generated, and was therefore the focus of this study. The presentation order of stimulus frequencies was randomized for each subject.
We measured the amplitude (dB SPL) of the DPOAE for each tone presentation (Figure 3). In addition to absolute amplitude, we estimated DPOAE thresholds for six different F2 frequencies that were also used in the ABR minimum audibility paradigm (1, 2, 3, 4, 6, and 8 kHz). We defined threshold as the lowest L1 amplitude (dB SPL) that met the following three criteria: 1) The amplitude of the DPOAE was at least 3 dB above the immediately surrounding noise floor. 2) The difference between L1 and DPOAE amplitudes did not exceed 85 dB. (This criterion was formed from offline determinations of instrumental and cavity distortions.) 3) The next two DPOAE measurements (elicited by 5 and 10dB increases in L1, respectively) also fit the first two criteria.
Fig. 3.
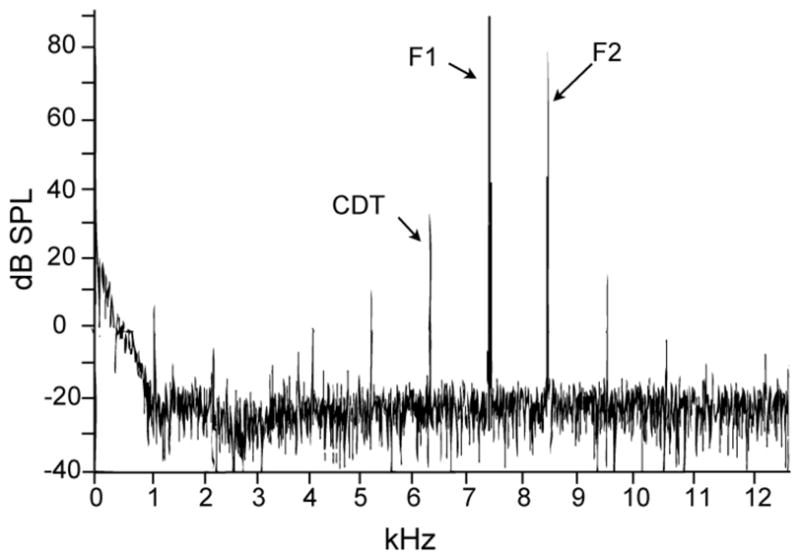
Representative frequency spectrum of a DPOAE recording from a breeding condition female. The primary tones (F1 and F2) were presented at the highest amplitudes (L1 = 90 dB SPL) to enable clear observation of the multiple distortion products. The distortion product with the largest amplitude is the cubic distortion tone (CDT), which corresponds to a frequency of 2F1-F2. F1 and F2 in this example are 7.4 and 8.5 kHz, respectively and the CDT is 6.3 kHz.
Hormone Measurement
At the end of each recording session, we rapidly decapitated subjects and removed basilar papillae for histological processing for a separate study. We collected trunk blood in heparinized tubes and immediately centrifuged it. We separated the plasma and stored it at −80°C until Enzyme-Linked ImmunoSorbent Assay (ELISA). Testosterone and estradiol concentrations were measured using standard kits (Assay Designs, Ann Arbor, MI) and compared to those measured in the wild (Wingfield and Farner 1978).
Assay Validation
We used a testosterone immunoassay kit (Assay Designs catalogue # 900-065) previously validated for the congeneric white-throated sparrow (Zonotrichia albicollis) (Swett and Breuner 2008). No publications reporting the use of the estrogen kit (Assay Designs catalogue # 900-174) in any avian species were found. We therefore validated the use of this kit for white-crowned sparrows. We pooled plasma samples from multiple sparrows and stripped the plasma of steroids with dextran-coated charcoal in assay buffer (Sigma-Aldrich, St. Louis, MO). We spiked stripped plasma with estradiol to ~19.6 ng/ml and assayed a serial dilution of the spiked plasma. The serial dilution paralleled the kit’s standard curve, indicating that endogenous protein elements in white-crowned plasma do not substantially interfere with hormone measurement.
Immunoassay Procedures
We followed the kit instructions to determine testosterone or estradiol levels of experimental subjects. Briefly, we added steroid displacement buffer (1% of raw plasma volume) to each plasma sample, brought the total volume to 200 μl with assay buffer and vortexed. Because LD+ testosterone conditions can generate plasma testosterone levels beyond the highest range of the kit’s detectability (2 ng/ml), a 1:20 dilution of each LD+ testosterone sample was made with assay buffer. We ran 100 μl aliquots of each sample (or LD dilution) in duplicate along with either five testosterone standards (0.008 – 2.000 ng/ml) or 6 estrogen standards (.0293–30.00 ng/ml). We incubated each sample with 50 μl of steroid antibody and alkaline phosphotase-conjugated steroid, emptied and washed all sample wells and added 200 μl of substrate. After adding the stop solution (50μl/well), we read the plate immediately on a Dynex MRX II microplate reader (Chantilly, VA) at 405 nm.
We analyzed samples from subjects tested in the DPOAE paradigm in separate assays from those tested only in the ABR paradigms. The minimum detectable plasma testosterone concentrations were 5.72 x 10−4 ng/ml (ABR tested) and 3.17 x 10−3 ng/ml (DPOAE tested); minimum estradiol concentrations were 1.91 x 10−2 ng/ml (ABR tested) and 5.15 x 10−2 ng/ml (DPOAE tested). Intra-assay variabilities for testosterone measurement were 5.99% (ABR tested) and 5.90% (DPOAE tested); intra-assay variabilities for estradiol measurement were 9.23% (ABR tested) and 7.84% (DPOAE tested). Inter-assay variabilities were 22.4% (testosterone) and 21.7% (estradiol).
One male (ABR) sample fell below the detection limit of the assay. For statistical analysis, we multiplied the detection limit (5.72 x 10−4 ng/ml) by the dilution factor of the sample in question (2.5). We used the resulting value (1.43 x 10−3 ng/ml) for subsequent analysis. One male sample was too concentrated to be detected by the assay, even after a 1:20 dilution. In this case, the concentration of the highest standard (2.0 ng/ml) was multiplied by the dilution factor (20) to give a result of 4.0 ng/ml. All female samples fell within the range of the estradiol assay; however, blood samples were lacking for two females that we used to optimize the DPOAE recording parameters.
Statistics
We made all comparisons with three-way or two-way mixed-model ANOVA (Sex x Breeding Condition x Stimulus Frequency/Stimulus Level) or independent samples t-test. All statistical analyses were made using PASW Statistics 18.0 for Mac (Chicago, IL). Data in all figures are presented as means +/− S.E.M.
RESULTS
Plasma Hormone Levels
Male and female birds housed under breeding (LD + testosterone or LD + estradiol) conditions had significantly higher plasma testosterone or estradiol levels than those housed under non-breeding (SD) conditions. Table 1 shows that testosterone levels from males housed under LD+ testosterone were comparable to those observed in breeding condition males in the wild (Wingfield and Farner, 1978). Estradiol levels from LD+ estradiol females, however, were higher than the physiological range of wild females in breeding condition (4.919 +/− 0.726 vs 0.300–0.400 ng/ml, Wingfield and Farner, 1978).
Table 1.
Plasma Testosterone and Estradiol Levels (mean +/− S.E.M. ng/ml)
| Non-breeding | Breeding | ta | P | |
|---|---|---|---|---|
| Plasma Testosterone | 0.496 +/− 0.146 (n=13) | 13.95 +/− 2.970 (n=11) | 2.07 | <0.0001 |
| Plasma Estradiol | 1.143 +/− 0.243 (n=11) | 4.919 +/− 0.726 (n=11) | 2.09 | <0.0001 |
Independent samples t-test (two-tailed)
ABR
Minimum audibility paradigm
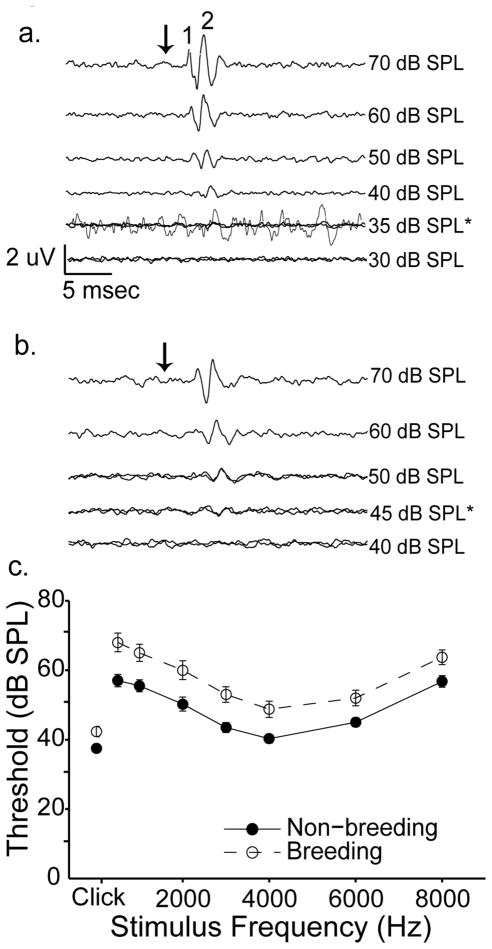
ABR thresholds were significantly affected by breeding condition and stimulus frequency. Males and females had similar overall ABR thresholds (means +/− S.E.M., 51.6 +/− 0.92 vs. 53.5 +/− 1.07 dB SPL, respectively; [F(1,35) = 0.279, p = 0.601]); we therefore pooled their data for analysis. Figure 4 shows representative averaged responses to a 4.0 kHz tone from a non-breeding female (4a) and a breeding female (4b). In these examples, we judged threshold to be 35 dB SPL (4a) and 45 dB SPL (4b). Figure 4c shows group data for thresholds to clicks and tone bursts at 0.5, 1, 2, 3, 4, 6 and 8 kHz. Stimulus frequency significantly affected threshold estimates, with birds showing maximal sensitivity to clicks and 4 kHz pure tones [F(7,245)= 70.54, p<0.001]. Thresholds in white-crowned sparrows housed under breeding-like conditions were higher by 3.90 – 12.3 dB (average 8.23 dB) than in birds housed under non-breeding–like conditions. The effect of breeding condition was significant [F(1,35) = 12.99, p = 0.001]. No significant interactions were found between any of the independent variables (sex, stimulus frequency, or breeding condition; all p > 0.05).
Fig. 4.
Birds housed under breeding-like conditions have higher auditory thresholds than those housed under non-breeding-like conditions. a) Representative ABRs decrease in amplitude and increase in latency as stimulus intensity is decreased. Traces were elicited by a 4000Hz tone from a non-breeding female. The top 4 traces represent averages of 500 stimulus presentations. 35 and 30 dB SPL traces represent averages of 1000 presentations. The black arrow indicates stimulus onset. Scale bars = 2 μV/ 5 msec. Threshold was estimated to be 35 dB SPL and is indicated by the asterisk. One trace elicited by a 35 dB SPL stimulus is enlarged and shown over the original traces to more clearly demonstrate a response. For this trace only, the scale bar = 0.3 uV/5 msec. b) Representative ABR traces from a breeding female demonstrate an elevated threshold. Experimental parameters and figure notations are as in a. The top two traces represent averages from 500 stimulus presentations; the remaining traces were averaged over 1000 presentations. Threshold was estimated at 45 dB SPL. Scale bar is the same for a and b. c) Mean +/− S.E.M. ABR thresholds of birds exposed to breeding-like conditions (open circles) are higher than those housed under non-breeding-like conditions (closed circles) across all stimulus frequencies. Data are presented linearly (rather than logarithmically) for clarity. Thresholds to clicks are shown at the left most portion of each graph and are measured in dB peak equivalent (p.e.) SPL. Each experimental group had an n = 20 (except for clicks, where breeding birds n = 21).
Many ABR peak latency values were not measurable at 70 dB SPL, either because of high thresholds at the extremes of the stimulus frequencies tested (500 and 8000 Hz) or because of elevated thresholds in breeding condition birds. As a result, the missing values (11.5% of the total number of data points) were not randomly distributed among the data set (see Supplementary Table 1 for more detailed information). A biased sample of missing data can confound statistical analyses and obscure interpretation of the results. Therefore, we calculated average peak latency values for each stimulus and experimental group (breeding males, non-breeding males, breeding females, and non-breeding females). Missing data points were filled in with the appropriate mean values and the completed dataset was analyzed by ANOVA, as above.
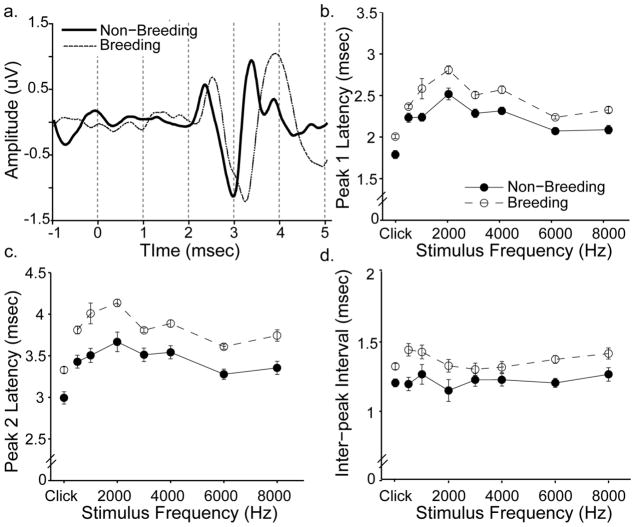
ABR peak latencies were significantly affected by breeding condition and stimulus frequency. Males and females had similar latency values for peak 1 (mean +/− S.E.M., 2.28 +/− 0.02 vs. 2.34 +/− 0.03 msec, respectively) and peak 2 (3.55 +/− 0.031 vs. 3.65 +/− 0.04 msec). The effect of sex was not significant (peak 1 [F (1,35) = 0.694, p = 0.411]; peak 2 [F(1,37) = 0.887, p = 0.353] we therefore pooled their data for analysis. Figure 5a shows representative ABR traces from a breeding and non-breeding female overlaid. Peak latencies from the breeding bird were delayed relative to the non-breeding bird. Group data for peak latencies generated by 70 dB SPL clicks and pure tone bursts are shown in Figure 5b-c. Latency values depended on stimulus frequency, with clicks evoking the lowest values. The effect of frequency was highly significant for both peak 1 [F(7,259) = 63.40, p <0.001] and peak 2 [F(7,259) = 31.52, p<0.001]. The differences between breeding and non-breeding peak 1 latencies ranged from 0.135–0.349 msec (average 0.233 msec); peak 2 latencies differed by more (range 0.286–0.513 msec, average 0.378 msec). The effect of breeding condition was statistically significant across all stimuli tested (peak 1; [F(1,37) = 18.94, p<0.001]; peak 2 [F(1,37) = 26.06, p <0.001]). A significant interaction between stimulus frequency and sex was observed for peak 1 latency values [F(7,259) = 2.846, p = 0.007]; no other interactions were found (all p>0.05).
Fig. 5.
Birds housed under breeding-like conditions have longer ABR peak latencies and inter-peak intervals than those housed under non-breeding-like conditions. a) Representative ABR traces from a breeding (thin line) and non-breeding (thick line) female in response to a 4 kHz tone. Traces are aligned in time and stimulus onset occurs at time zero. The breeding bird has a delayed response compared to the non-breeding bird; note that this temporal disparity increases between peak 1 and peak 2. b) Peak 1 latencies of birds exposed to breeding-like conditions (open circles) are longer than those housed under non-breeding-like conditions (closed circles). The same pattern was observed for peak 2 latencies (c) and inter-peak intervals (d). Data are means +/− S.E.M. generated in response to iso-intensity tones (70 dB SPL) and clicks (70 dB p.e. SPL). Missing data points were filled in with appropriate group averages before data were plotted and analyzed (see main text). Breeding birds n = 19; non-breeding birds n = 20.
Breeding condition also affected inter-peak intervals (Figure 5d). Males and females had similar inter-peak intervals (mean +/− S.E.M., 1.28 +/− 0.02 msec vs. 1.31 +/− 0.02 msec, respectively). The effect of sex was not significant [F(1,37) = 0.557, p = 0.460]; we again pooled their data for analysis. Birds in breeding condition had longer inter-peak intervals than birds in non-breeding condition. These differences ranged from 0.067–0.256 msec (average 0.146 msec) and were statistically significant [F(1,37) = 17.09, p < 0.001]. While stimulus frequency did not affect inter-peak intervals independently [F(7,259) = 1.681, p =0.114], a significant interaction was found between all three independent variables (stimulus frequency, breeding condition and sex), [F(7,259) = 2.747, p = 0.009].
These data suggest that seasons and hormones affect peripheral auditory sensitivity. One possible concern was that these findings reflected group differences in baseline noise levels, rather than differences in sensory processing, per se. High baseline noise levels could cause difficulty in the extraction of responses at low stimulus amplitudes, leading to higher threshold estimates. To examine this possibility, we calculated the root mean square deviation (RMSD) of the voltage values during the first two msec of the pre-stimulus window for each response. A large RMSD value indicates greater variability in the pre-stimulus window, reflecting a higher baseline noise level. We averaged RMSD values across all responses to obtain a single value for each bird. We then compared these values as a function of breeding condition.
We only used responses averaged over 1000 stimulus presentations for this analysis. We chose this selection criterion because the effect of baseline noise on threshold detection was of primary interest, and all traces at or near threshold were averaged over 1000 stimulus presentations. Birds in breeding and non-breeding condition had similar RMSD values (mean +/− S.E.M., 0.077 +/− 0.025 μV vs. 0.073 +/− 0.022 μV, respectively) that did not differ statistically [F(1,37) = 0.093, p =0.762]). The results of this analysis suggest that seasonal and hormonal differences in ABR thresholds and latencies and reflect differences in auditory sensitivity at the level of the inner ear and/or central nervous system.
Forward masking frequency resolution paradigm
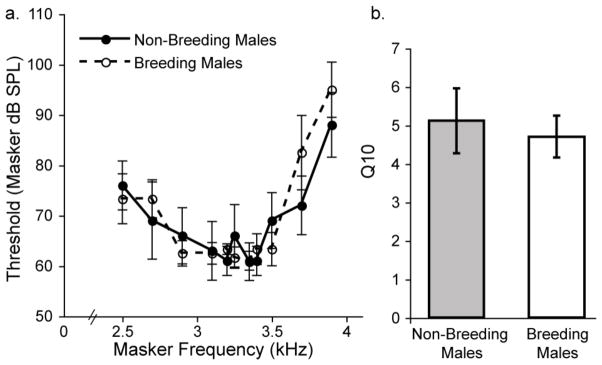
Breeding condition did not affect frequency tuning. Figure 6a shows averaged frequency tuning curves from breeding males and non-breeding males. The curves show substantial overlap at all masker frequencies tested and did not differ statistically [F (1,9) = 0.057, p = 0.817]. We calculated and compared Q10 values for each subject to determine whether tuning sharpness varied as a function of breeding condition. Average Q10 values are shown in Figure 6b. Breeding males and non-breeding males had similar Q10 values (means +/− S.E.M.; breeding 4.70 +/− 0.54; non-breeding 5.21 +/− 0.84). A two-sample t-test revealed no significant difference between the groups [t = 2.447, p = 0.651].
Fig. 6.
Breeding condition does not affect frequency tuning. a) Thresholds for a 3.3 kHz probe tone in a forward masking paradigm are similar for breeding males (open circles; n = 5) and non-breeding males (closed circles; n = 5) across all masker frequencies. b) Average Q10 values (indicative of tuning sharpness) did not differ between breeding males (open bar) and non-breeding males (shaded bar). One subject in each group had tuning curves too broad to accurately measure Q10. Thus, n = 4 for each group in b.
Forward masking temporal processing paradigm
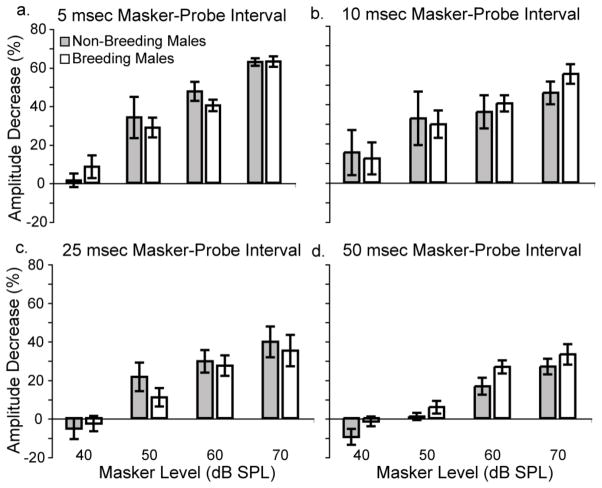
Breeding condition did not affect temporal adaptation. Figures 7a–d show the percent decrease of the probe response as a function of masker level (40, 50, 60 and 70 dB SPL) for four different masker-probe intervals (5, 10, 25 and 50 msec). Response amplitudes of breeding males and non-breeding males decrease by similar amounts across all masker levels for 5, 10, and 25 msec masker-probe intervals (Figures 7a–c). Separate two-way ANOVAs revealed no effect of breeding condition for these three intervals (all p>0.4). At the longest masker-probe interval, however, breeding condition did affect the amount by which response amplitudes decreased (Figure 7d). Response amplitudes of males in breeding condition decreased an average of 5.84% more than males in non-breeding condition when the masker and probe were separated by 50 msec. This difference was significant [F(1,8) = 6.867, p=0.031].
Fig. 7.
Breeding condition only affects temporal adaptation at the longest masker-probe interval tested. a) The probe-elicited ABR response amplitude decreases by a similar amount for males in breeding (open bars) and non-breeding (shaded bars) conditions when the masker and probe are separated by 5 msec. Similar results were found for (b) 10 msec and (c) 25 msec masker-probe intervals. d) When the masker and probe are separated by 50 msec, breeding males show a significantly greater decrease in response amplitude than non-breeding males. Data are means +/− S.E.M. Breeding males n = 6; non-breeding males n = 5 (except n = 4 at 40 and 50 dB SPL masker levels for 10, 25 and 50 msec intervals, and 40 dB SPL masker level for 5 msec interval).
Temporal variability paradigm
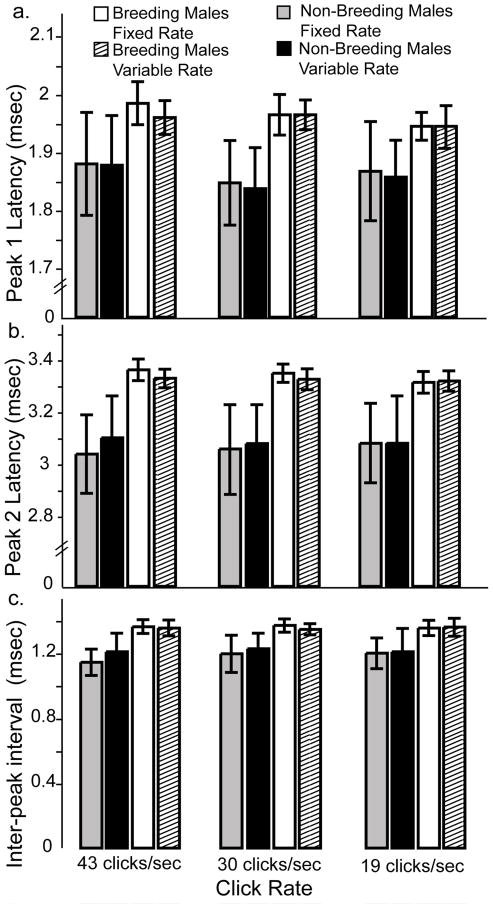
Breeding condition did not affect processing of temporally variable stimuli. Figure 8a–c shows the peak 1, peak 2, and inter-peak latencies, respectively, as a function of click rate. Fixed and variable rates elicited similar latencies from breeding and non-breeding males. Separate two-way ANOVAs revealed no effect of temporal variability on peak 1, peak 2 or inter-peak latencies (all p>0.2).
Fig. 8.
Breeding condition does not affect processing of temporally variable stimuli. Clicks were presented at three rates with both fixed and variable inter-click intervals. a) Non-breeding males show similar peak 1 latencies to fixed (grey bars) and variable (black bars) inter-click intervals for all presentation rates tested. Though breeding males showed a trend towards longer latencies in general, their responses to fixed (open bars) and variable (striped bars) stimuli were also similar. Similar results were found for peak 2 latencies (b) and inter-peak intervals (c). Data are mean +/− S.E.M. Breeding males n = 6, non-breeding males n = 4.
As expected from the latency analyses presented above, breeding males showed a trend of longer peak latencies and inter-peak intervals than non-breeding males. These differences did not reach significance, however, probably due to small sample sizes (n = 4–6).
DPOAE
The results from the ABR study indicated that breeding condition affects auditory sensitivity. To determine whether the effect could be explained by processing changes at levels prior to synaptic responses, DPOAEs were elicited by a range of frequencies (1 – 9.5 kHz) from males and females in breeding and non-breeding conditions.
Breeding condition and sex did not affect iso-intensity DPOAE amplitudes. DPOAE amplitudes of males and females were similar overall, and did not differ significantly [F(1,15) = 1.181, p> 0.20]; we therefore pooled their data for analysis. Figure 9a shows DPOAE amplitudes as a function of the second primary (F2) frequency. These DPOAEs were elicited while L1 was held constant at 70 dB SPL. DPOAE amplitudes depended on primary tone frequency, with mid to high frequencies eliciting the largest DPOAEs. The effect of frequency was significant [F(17,255) = 17.95, p <0.001]. DPOAE amplitudes were similar for birds in breeding and non-breeding conditions across all frequencies (mean +/− S.E.M., −9.12 +/− 0.82 vs. −8.17 +/− 0.83 dB SPL) and did not differ statistically [F(1,15) = 0.009, p = 0.927]. No significant interactions were observed (all p>0.05).
Fig. 9.
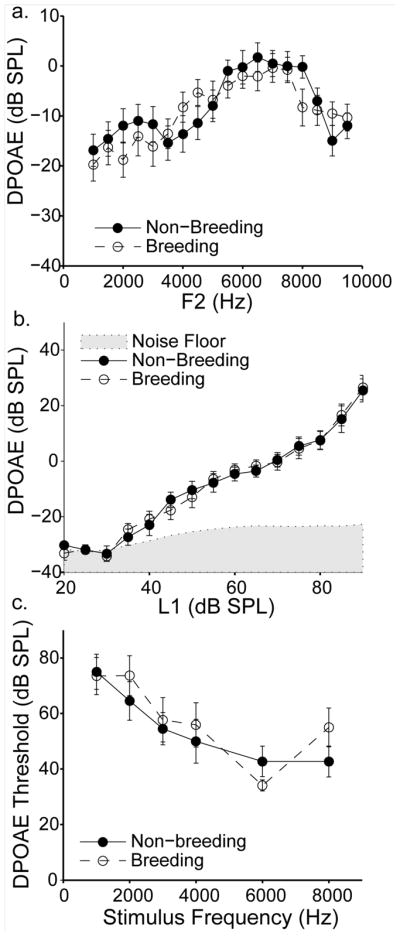
Breeding condition does not affect DPOAE amplitudes or thresholds. a) DPOAE amplitude changes systematically with F2 frequency, but no amplitude differences are observed between breeding (open circles) and non-breeding (closed circles) birds. DPOAEs were elicited by iso-intensity primary tones (L1 = 70 dB SPL) for all frequencies tested. b) DPOAE amplitude increases with increasing stimulus level, but no difference is observed between breeding (open circles) and non-breeding (closed circles) birds. The amplitude of the noise floor immediately surrounding the DPOAE frequency is indicated by the shaded grey area. DPOAEs were elicited by iso-frequency primary tones (F2 = 7 kHz) for all levels tested. c) DPOAE threshold decreases with increasing stimulus frequency, but no difference is observed between breeding (open circles) and non-breeding (closed circles) birds. Data are means +/− S.E.M. Breeding birds n = 11 (except n = 8 at 20 and 25 dB SPL in b); non-breeding birds n = 11 (except n = 7 at 1000Hz in c).
While breeding condition did not affect iso-intensity DPOAE amplitudes, it was possible that seasons/hormones affected amplitudes across the dynamic range of stimulus levels. Figure 9b shows DPOAE amplitudes as a function of the level of the first primary tone (L1). These DPOAEs were elicited while F2 was held constant at 7 kHz. This F2 value was chosen because it elicited relatively strong DPOAE amplitudes at 70 dB (see Figure 9a). DPOAE amplitudes were similar for males and females across stimulus levels [F(1,15) = 2.35, p = 0.146); we again pooled their data for analysis. DPOAE amplitudes increased with higher stimulus levels; this effect was significant [F(14,210) = 141.8, p < 0.001]). DPOAE amplitudes were similar, however, for birds in breeding and non-breeding conditions across all stimulus levels [F(1,15) = 2.62, p = 0.127). No significant interactions were observed (all p >0.2).
While DPOAE thresholds depend on DPOAE amplitudes, other factors can affect threshold independently, such as the level of the noise floor. This fact raised the possibility that breeding condition affected DPOAE thresholds without directly affecting DPOAE absolute amplitude measurements. To address this issue, DPOAE thresholds were measured for six F2 frequencies that were also used in the ABR minimum audibility paradigm (1, 2, 3, 4, 6 and 8kHz). Figure 9c shows DPOAE threshold as a function of F2 frequency. Again, males and females had similar threshold values (mean +/− S.E.M., 51.9 +/− 2.89 vs. 60.2+/− 3.07 dB SPL) that did not differ statistically [p> 0.20] and we pooled their data for analysis. In general, the frequencies that elicited that highest amplitude DPOAEs (6–8 kHz) also elicited the lowest thresholds. The effect of stimulus frequency was significant [F(5, 85) = 15.29, p <0.001]. Threshold values of birds in breeding condition and non-breeding condition were similar (mean +/− S.E.M., 58.1 +/− 3.18 vs. 54.9 +/− 2.92 dB SPL) and did not differ statistically, [F(1,17) = 0.092, p = 0.77).
SUMMARY
To summarize, we found that birds housed under breeding-like laboratory conditions had higher ABR thresholds, longer peak latencies, and increased inter-peak intervals. As measured by ABR methods, temporal processing and frequency tuning were unaffected by breeding state. In addition, otoacoustic emissions appeared to be unaffected by breeding state.
DISCUSSION
The main purpose of this study was to determine whether breeding condition affects auditory processing at the level of the inner ear and brainstem pathways in a highly seasonal songbird. A robust effect of breeding condition was found. Birds exposed to breeding-like conditions had higher ABR thresholds, longer peak latencies, and longer inter-peak intervals than birds housed under non-breeding-like conditions. No measurable effects were found for ABR analyses of frequency resolution or temporal adaptation or for more peripheral measures of auditory function (otoacoustic emissions).
One important note is that while males in this study demonstrated testosterone levels comparable to male white-crowned sparrows free living in the wild, females had much higher estradiol levels than free living birds, even when housed under non-breeding conditions. While the exogenous treatment can account for the high estradiol levels in the breeding condition females, the underlying cause of elevated estradiol in non-breeding females remains unclear. It is possible that the ELISA kit used to measure estradiol levels detected endogenous estrogen-like compounds in the white-crowned sparrow plasma, giving artificially high measurements, though this explanation seems unlikely given that stripped and spiked white-crowned sparrow plasma dilutions paralleled the kit’s standard curves. Alternatively, the social environment of the non-breeding females may have been a contributing factor. It is known that experimentally elevating hormone levels in female white-crowned sparrows can increase the hormone levels of their mates (Moore 1982). Many of the non-breeding females in this study were housed in single sex aviaries before experimentation, and unidentified social or endocrine cues may have elevated estradiol levels in these birds. (It should be noted, however, that birds in single-sex aviaries always had full visual and auditory contact with members of the opposite sex.) Though the effect of social interactions between females on their hormone levels has not been addressed in birds, effects of this type have been documented in other taxa (McClintock 1971). These reports, however, are controversial (see Schank 2001). It is important to recognize, however, that even though we cannot explain the high estradiol levels measured in this study, our experimental groups did show large relative differences in estradiol levels (~4X higher in breeding vs. non-breeding conditions).
Breeding condition affects ABR thresholds
Both male and female white-crowned sparrows showed significantly higher ABR thresholds when housed under breeding-like conditions than under non-breeding like conditions. On average, this difference amounted to about 8 – 10 dB, a substantial amount given that a 10 dB amplitude increase is perceived as twice as loud by humans (Stevens and Poulton 1956). We were careful to verify ABR threshold estimates in several ways. First, visually estimated thresholds showed strong correlations with both blind observer and quantitative estimates. Second, though ABR threshold estimates are approximately 10– 30 dB less sensitive than behavioral estimates (Borg and Engstrom 1983; Brittan-Powell et al. 2002), the audiograms presented here are similar in shape to behavioral audiograms from song sparrows (Melospiza melodia) and swamp sparrows (Melospiza Georgiana) (Okanoya and Dooling 1988). Third, RMSD analysis demonstrated that group differences in threshold estimates could not be attributed solely to differences in baseline noise levels. Last, given the fact that there are numerous reports in the literature of absolute peak latency delays as a function of hearing loss (Coats and Martin 1977; Coats 1978; Jerger and Mauldin 1978; Rosenhamer et al. 1981), the fact that breeding condition birds demonstrated both elevated thresholds and prolonged peak latencies for all stimuli tested supports the validity of the threshold differences observed.
The results reported here differ from those of a previous study that showed no effect of season on ABR thresholds or latencies (Henry and Lucas 2009). The discrepancy between the studies may result from a number of different factors. First, Henry and Lucas used house sparrows (Passer domesticus), a species that shows much less pronounced seasonal breeding (Nehls 1981) than does the Gambel’s subspecies of white-crowned sparrow used in our study. Second, the birds in the Lucas study were divided into three different groups for seasonal comparisons (those caught in March–May, June–July and Sep–Nov). It is well known, however, that the seasonal fluctuations in hormone levels are not synchronous within the individuals in a population. Therefore, such comparisons are more robust when subjects are selected on the basis of their hormonal and breeding state, rather than by calendar date (Wingfield and Farner 1978). It is possible that if Henry and Lucas had measured plasma hormone levels and examined their data on the basis of that comparison, they might have observed effects similar to those reported here.
The results of our study also differ from the published literature in another important way. Previous work has demonstrated seasonal differences in auditory processing in other taxa, including both frogs and fish (Goense and Feng 2005; Miranda and Wilczynski 2009a; Sisneros 2009). In all of these cases, sensitivity or frequency tuning has shifted in a direction that enhances reception of mating calls or vocalizations during breeding conditions. These findings led to the a priori hypothesis that white-crowned sparrows in breeding condition would have greater auditory sensitivity than birds in non-breeding condition. Surprisingly, the results here demonstrate the opposite of what was expected; sparrows in non-breeding condition have greater auditory sensitivity than birds in breeding condition. The possible behavioral significance and adaptive value of these findings are discussed below.
Breeding condition affects ABR latencies
Both absolute and inter-peak latencies were prolonged in breeding condition white-crowned sparrows. Studies of humans and other mammals suggest that the first peak of the ABR reflects activity of the auditory nerve (Sohmer et al. 1974; Buchwald and Huang 1975; Starr and Hamilton 1976; Achor and Starr 1980; Moller et al. 1981; Moller and Jannetta 1983). Similar wave 1 latencies have been reported for both mammalian and avian species (Buchwald and Huang 1975; Achor and Starr 1980; Katayama 1985, present data; Burkard et al. 1996; Brittan-Powell et al. 2002; Brittan-Powell et al. 2005), suggesting that the wave 1 generator is the same for both animal classes. Additionally, Brittan-Powell and Dooling (2002) demonstrated that wave 1 of the ABR corresponds to the first deflection of the compound action potential in budgerigars (Melopsittacus undulatus); this finding supports the notion that wave 1 of the avian ABR reflects activity of the auditory nerve. The breeding condition increase in white-crowned sparrow wave 1 latencies therefore suggests a hormonal effect that originates early in the auditory pathway.
The generator of wave 2 is less clear. Intracranial recordings, clinical evidence and estimates of conduction times and synaptic delays suggest that wave 2 of the human ABR reflects processing in the proximal portion of the auditory nerve (Moller and Jannetta 1981, 1983; Hall 2007). The lack of such studies in birds and the dramatic difference in length of the auditory nerve axons in humans (25 mm, Hall 2007) and songbirds (1–3 mm, personal communication- E.W. Rubel), however, suggests that this conclusion does not necessarily apply to avian species. Additionally confounding is the fact that previous studies have suggested that components of wave 2 of the avian ABR actually correspond to wave 3 of the human ABR (Katayama 1985; Brittan-Powell et al. 2002; Hall 2007), which is thought to have multiple brainstem generators (Hall 2007).
Though no conclusive statement can be made about the location of the wave 2 generator(s) in this study, much can still be learned from the effect of breeding condition on the wave 2 latency. In particular, wave 2 latency values increased more than wave 1 latencies in breeding condition birds, leading to a significant difference in inter-peak intervals. Inter-wave latencies reflect signal conduction times along the auditory pathway (Ponton et al. 1996). Longer inter-wave latencies therefore suggest that axonal conduction velocity and/or synaptic transmission is slower in breeding condition white-crowned sparrows. Possible mechanisms to explain these findings are discussed below.
Effect of acoustic stimulation on breeding condition differences
Breeding condition male birds, including white-crowned sparrows, sing at higher rates than non-breeding males (Catchpole and Slater 1995; Meitzen et al. 2009a). Birds in different breeding conditions in our study were held in separate rooms, leading to the possibility that breeding birds were more acoustically stimulated than non-breeding birds and that this extra stimulation contributed to their lower auditory sensitivity. While we did not formally measure the differences in singing and calling rates between breeding and non-breeding birds during housing, breeding females were often housed in a room with no males. Wild female white-crowned sparrows rarely sing without testosterone stimulation, and almost never do so in captivity (Baptista and Petrinovich 1986). (Indeed, the two authors who work with white-crowned sparrows routinely (EB and MLC) have never observed a captive female sing under either breeding or non-breeding conditions). Therefore, breeding condition females that were housed without males would be less acoustically stimulated than their non-breeding counterparts that were housed in rooms with many males. Conversely, we would expect breeding condition males to be more stimulated than their non-breeding counterparts. As similar ABR findings were observed for males and females, it is unlikely that differences in acoustic stimulation can explain the differences in auditory sensitivity reported in this study.
Cellular origins of breeding condition differences
Increased ABR thresholds and prolonged wave 1 latencies in breeding condition white-crowned sparrows initially raised the possibility that seasons and hormones act on the hair cells or other auditory processing components presynaptic to the auditory nerve afferents. Others have suggested similar models (Sisneros et al. 2004; Henry and Lucas 2009), and hair cells presented themselves as interesting candidates because of their known expression of hormone receptors in numerous species (Stenberg et al. 1999; Stenberg et al. 2001; Sisneros et al. 2004; Hultcrantz et al. 2006; Noirot et al. 2009). The distortion product otoacoustic emission (DPOAE) analyses, however, do not support the idea of a functional change at the hair cell level. DPOAEs, like other types of otoacoustic emissions, are indices of cochlear function and are now widely used in clinical diagnostic settings (Harris 1990; Pak et al. 2000; Akdogan and Ozkan 2006). Changes in DPOAE thresholds and input-output functions are reliable and valid indicators of changes in auditory sensitivity (Lonsbury-Martin et al. 1991; Kemp 2002). If preneural changes in auditory function were responsible for the elevated ABR thresholds and increased peak 1 latencies found in breeding condition birds, one would expect to find decreased DPOAE amplitudes and/or increased DPOAE thresholds. No measurable effect of breeding condition was found for DPOAEs recorded from male or female birds, suggesting that the functional change does not occur peripheral to the hair cell-auditory nerve synapse. It is important to note, however, that the site of hormone action and the site of the functional change are not necessarily the same. For example, steroid hormones have been implicated in the regulation of synaptic signaling (Mitsushima et al. 2009), leading to the possibility that hormones act on hair cells to modulate neurotransmitter release at the hair cell-auditory nerve synapse. Similarly, elevated aromatase expression or activity in the auditory hair cells of breeding condition birds may contribute to functional changes at the level of the auditory nerve, an idea discussed more fully below.
Steroid hormones are well known regulators of ionic currents and neurotransmitter receptor expression (McEwen 1991; Zakon 1998). Hormone binding in cochlear ganglion cells or auditory nerve fibers could therefore directly affect axon conduction time and intrinsic excitability, leading to the latency and threshold differences observed here. Anatomical evidence supports this possibility. Positive staining for both ERα and ERβ has been documented in type I and II spiral ganglion cells of mice, rats and humans (Stenberg et al. 1999; Stenberg et al. 2001). Similarly, Forlano and colleagues found ERα mRNA and aromatase expression in the auditory nerve fibers of the midshipman fish (Porichthys notatus); aromatase was also found in the ganglion cell somata (Forlano et al. 2005). Additionally, both ERα and AR are expressed in the cochlear ganglion cells of white-crowned sparrows (Wang, Brenowitz and Rubel, unpublished observations). The contribution of seasonal and hormonal effects on descending efferent pathways cannot be ruled out conclusively at this time, however.
Male and female white-crowned sparrows showed similar changes in auditory processing even though they were treated with two different hormones (testosterone and estradiol). Testosterone can be aromatized to estradiol in vivo, however, suggesting that estradiol may mediate the changes observed in both sexes. Previous research has shown that aromatase expression is elevated in breeding condition birds (Fusani et al. 2000), and aromatase activity is highest during the breeding season (Riters et al. 2001; Soma et al. 2003). Additionally, other investigators have demonstrated that testosterone treatment increases aromatase activity in the central nervous system of quails (Schumacher and Balthazart 1986), doves (Steimer and Hutchison 1981), canaries (Fusani et al. 2001) and possibly white-crowned sparrows (Park et al. 2005). Aromatase is expressed in the auditory hair cells of zebra finches (Noirot et al. 2009). Notably, aromatase is also expressed in the auditory nerve of two species of fish (Gelinas and Callard 1997; Forlano et al. 2005), though it is not yet known whether this is also true for birds. These findings support the idea that estradiol production is elevated in breeding condition male white-crowned sparrows and available to bind to ERs in the cochlear ganglion cells.
It should be noted that the data presented here may partly result from steroid independent effects of photoperiod, such as seasonal regulation of aromatase or steroid receptor expression (Smith et al. 1997; Soma et al. 1999; Riters et al. 2001; Park et al. 2005). To investigate this possibility further, we looked for correlations between hormone level and ABR thresholds and latencies (Supplementary Figures 2 and 3). Though some of the latency measures significantly correlated with hormone levels in males, we observed no significant correlations for females, nor for ABR thresholds in either sex. These findings suggest that steroid independent effects of photoperiod may play a role in the regulation of auditory processing. One cannot necessarily rule out hormones as a causal factor, however; an alternative possibility is that after a threshold hormone concentration is reached, a physiological response occurs which then levels off when the concentration reaches a ceiling level. This model seems to explain seasonal changes in the morphology and electrical activity of neurons in the telencephalic song control nuclei (Brenowitz 2008).
Behavioral Significance
We predicted that auditory thresholds would be lower in breeding condition birds than in non-breeding birds, but observed the opposite pattern. The thresholds observed in white-crowned sparrows exposed to breeding-like conditions (ranging from 25 – 90 dB SPL) are not outside the range of ‘normal’ thresholds of other songbirds (Dooling et al. 2000), suggesting that these findings may best be interpreted as enhanced sensitivity during the non-breeding season rather than impaired hearing during the breeding season.
One possible explanation for this finding relates to seasonal changes in vocal production. White-crowned sparrow song is known to be shorter, more variable, and less frequently produced outside the breeding season (Meitzen et al. 2009a). In addition, non-breeding song is produced at lower amplitudes (Supplemental Figure 4). Previous work in other avian species has indicated that song may serve as a flocking and/or roosting signal in birds that form social groups outside the breeding season (Brenowitz 1981). If non-breeding song plays a similar role in white-crowned sparrows, then increased auditory sensitivity may facilitate group cohesion in the non-breeding period. Future work should address this issue and other perceptual implications of seasonal/hormonal effects on auditory processing.
Supplementary Material
Acknowledgments
We thank Brandon Warren, Karin Lent, and Mike Famulare for technical assistance, Kristen Richards Preble for animal care, Jason Sanchez for threshold verification, and members of the Brenowitz and Rubel labs for constructive discussion and support. This work was supported by the NIH/NIDCD grants DC000033, DC003829, DC004661, the Seattle Chapter of Achievement Rewards for College Scientists Foundation (MLC), and the Washington Research Foundation (MLC). All procedures were approved by the Institutional Animal Care and Use Committee at the University of Washington, Seattle.
Abbreviations
- ABR
Auditory brainstem response
- AR
Androgen receptor
- DPOAE
Distortion product otoacoustic emission
- ER
Estrogen receptor
- F1
First primary tone
- F2
Second primary tone
- LD
Long-day
- L1
Level of the first primary tone
- L2
Level of the second primary tone
- NCM
Caudomedial nidopallium
- Q
Quality factor
- SD
Short-day
Footnotes
CONFLICT OF INTEREST
The authors declare that they have no conflict of interest.
References
- Achor LJ, Starr A. Auditory brain stem responses in the cat. I. Intracranial and extracranial recordings. Electroencephalography and clinical neurophysiology. 1980;48:154–173. doi: 10.1016/0013-4694(80)90301-6. [DOI] [PubMed] [Google Scholar]
- Akdogan O, Ozkan S. Otoacoustic emissions in children with otitis media with effusion. International journal of pediatric otorhinolaryngology. 2006;70:1941–1944. doi: 10.1016/j.ijporl.2006.07.004. [DOI] [PubMed] [Google Scholar]
- Baptista LF, Morton ML. Interspecific song acquisition by a white-crowned sparrow. Auk. 1981;98:383–385. [Google Scholar]
- Baptista LF, Petrinovich L. Song development in the white-crowned sparrow: Social factors and sex differences. Animal Behaviour Anim Behav. 1986;34:1359–1371. [Google Scholar]
- Bergevin C, Freeman DM, Saunders JC, Shera CA. Otoacoustic emissions in humans, birds, lizards, and frogs: evidence for multiple generation mechanisms. Journal of comparative physiology. 2008;194:665–683. doi: 10.1007/s00359-008-0338-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borg E, Engstrom B. Hearing thresholds in the rabbit. A behavioral and electrophysiological study. Acta oto-laryngologica. 1983;95:19–26. doi: 10.3109/00016488309130911. [DOI] [PubMed] [Google Scholar]
- Brenowitz EA. ‘Territorial song’ as a flocking signal in red-winged blackbirds. Anim Behav. 1981;29:641–642. [Google Scholar]
- Brenowitz EA. Altered perception of species-specific song by female birds after lesions of a forebrain nucleus. Science (New York, NY. 1991;251:303–305. doi: 10.1126/science.1987645. [DOI] [PubMed] [Google Scholar]
- Brenowitz EA. Plasticity of the song control system in adult birds. In: Zeigler P, Marler P, editors. Neuroscience of birdsong. Cambridge University Press; New York: 2008. p. 332. [Google Scholar]
- Brenowitz EA, Baptista LF, Lent K, Wingfield JC. Seasonal plasticity of the song control system in wild Nuttall’s white-crowned sparrows. Journal of neurobiology. 1998;34:69–82. doi: 10.1002/(sici)1097-4695(199801)34:1<69::aid-neu6>3.0.co;2-a. [DOI] [PubMed] [Google Scholar]
- Brittan-Powell EF, Dooling RJ, Gleich O. Auditory brainstem responses in adult budgerigars (Melopsittacus undulatus) The Journal of the Acoustical Society of America. 2002;112:999–1008. doi: 10.1121/1.1494807. [DOI] [PubMed] [Google Scholar]
- Brittan-Powell EF, Lohr B, Hahn DC, Dooling RJ. Auditory brainstem responses in the Eastern Screech Owl: an estimate of auditory thresholds. The Journal of the Acoustical Society of America. 2005;118:314–321. doi: 10.1121/1.1928767. [DOI] [PubMed] [Google Scholar]
- Brown-Borg HM, Beck MM, Jones TA. Origins of peripheral and brainstem auditory responses in the White Leghorn chick. Comparative biochemistry and physiology. 1987;88:391–396. doi: 10.1016/0300-9629(87)90052-1. [DOI] [PubMed] [Google Scholar]
- Buchwald JS, Huang C. Far-field acoustic response: origins in the cat. Science (New York, NY. 1975;189:382–384. doi: 10.1126/science.1145206. [DOI] [PubMed] [Google Scholar]
- Burkard R, Jones S, Jones T. Conventional and cross-correlation brain-stem auditory evoked responses in the white leghorn chick: rate manipulations. The Journal of the Acoustical Society of America. 1994;95:2136–2144. doi: 10.1121/1.408675. [DOI] [PubMed] [Google Scholar]
- Burkard R, McGee J, Walsh EJ. Effects of stimulus rate on the feline brain-stem auditory evoked response during development. I. Peak latencies. The Journal of the Acoustical Society of America. 1996;100:978–990. doi: 10.1121/1.416209. [DOI] [PubMed] [Google Scholar]
- Catchpole CK, Slater PJB. Bird Song: Biological themes and variations. University Press; Cambridge, Great Britian: 1995. [Google Scholar]
- Coats AC. Human auditory nerve action potentials and brain stem evoked responses. Arch Otolaryngol. 1978;104:709–717. [PubMed] [Google Scholar]
- Coats AC, Martin JL. Human auditory nerve action potentials and brain stem evoked responses: effects of audiogram shape and lesion location. Arch Otolaryngol. 1977;103:605–622. doi: 10.1001/archotol.1977.00780270073012. [DOI] [PubMed] [Google Scholar]
- Corwin JT, Bullock TH, Schweitzer J. The auditory brain stem response in five vertebrate classes. Electroencephalography and clinical neurophysiology. 1982;54:629–641. doi: 10.1016/0013-4694(82)90117-1. [DOI] [PubMed] [Google Scholar]
- Cynx J, Nottebohm F. Role of gender, season, and familiarity in discrimination of conspecific song by zebra finches (Taeniopygia guttata) Proceedings of the National Academy of Sciences of the United States of America. 1992;89:1368–1371. doi: 10.1073/pnas.89.4.1368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dallos P. Cochlear amplification, outer hair cells and prestin. Current opinion in neurobiology. 2008;18:370–376. doi: 10.1016/j.conb.2008.08.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dallos P, Wu X, Cheatham MA, Gao J, Zheng J, Anderson CT, Jia S, Wang X, Cheng WH, Sengupta S, He DZ, Zuo J. Prestin-based outer hair cell motility is necessary for mammalian cochlear amplification. Neuron. 2008;58:333–339. doi: 10.1016/j.neuron.2008.02.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Groof G, Verhoye M, Poirier C, Leemans A, Eens M, Darras VM, Van der Linden A. Structural changes between seasons in the songbird auditory forebrain. J Neurosci. 2009;29:13557–13565. doi: 10.1523/JNEUROSCI.1788-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Del Negro C, Edeline JM. Sex and season influence the proportion of thin spike cells in the canary HVc. Neuroreport. 2002;13:2005–2009. doi: 10.1097/00001756-200211150-00003. [DOI] [PubMed] [Google Scholar]
- Del Negro C, Kreutzer M, Gahr M. Sexually stimulating signals of canary (Serinus canaria) songs: evidence for a female-specific auditory representation in the HVc nucleus during the breeding season. Behavioral neuroscience. 2000;114:526–542. doi: 10.1037//0735-7044.114.3.526. [DOI] [PubMed] [Google Scholar]
- Del Negro C, Lehongre K, Edeline JM. Selectivity of canary HVC neurons for the bird’s own song: modulation by photoperiodic conditions. J Neurosci. 2005;25:4952–4963. doi: 10.1523/JNEUROSCI.4847-04.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Despland PA, Galambos R. The auditory brainstem response (ABR) is a useful diagnostic tool in the intensive care nursery. Pediatric research. 1980;14:154–158. doi: 10.1203/00006450-198002000-00018. [DOI] [PubMed] [Google Scholar]
- DeWolfe BB, Kaska DD, Peyton LJ. Prominent variations in the songs of gambel’s white-crowned sparrows. Bird-Banding. 1974;45:224–252. [Google Scholar]
- Dooling RJ, Lohr B, Dent ML. Hearing in Birds and Reptiles. In: Dooling RJ, Popper AN, Fay RR, editors. Comparative Hearing: Birds and Reptiles. Springer-Verlag; New York: 2000. pp. 308–359. [Google Scholar]
- Forlano PM, Deitcher DL, Bass AH. Distribution of estrogen receptor alpha mRNA in the brain and inner ear of a vocal fish with comparisons to sites of aromatase expression. The Journal of comparative neurology. 2005;483:91–113. doi: 10.1002/cne.20397. [DOI] [PubMed] [Google Scholar]
- Fusani L, Hutchison JB, Gahr M. Testosterone regulates the activity and expression of aromatase in the canary neostriatum. Journal of neurobiology. 2001;49:1–8. doi: 10.1002/neu.1061. [DOI] [PubMed] [Google Scholar]
- Fusani L, Van’t Hof T, Hutchison JB, Gahr M. Seasonal expression of androgen receptors, estrogen receptors, and aromatase in the canary brain in relation to circulating androgens and estrogens. Journal of neurobiology. 2000;43:254–268. [PubMed] [Google Scholar]
- Gelinas D, Callard GV. Immunolocalization of aromatase- and androgen receptor-positive neurons in the goldfish brain. General and comparative endocrinology. 1997;106:155–168. doi: 10.1006/gcen.1997.6891. [DOI] [PubMed] [Google Scholar]
- Goense JB, Feng AS. Seasonal changes in frequency tuning and temporal processing in single neurons in the frog auditory midbrain. Journal of neurobiology. 2005;65:22–36. doi: 10.1002/neu.20172. [DOI] [PubMed] [Google Scholar]
- Hall JW. Handbook of Auditory Evoked Responses. Allyn and Bacon; Needham Heights, MA: 1992. [Google Scholar]
- Hall JW. New Handbook of Auditory Evoked Responses. Allyn and Bacon; Boston, MA: 2007. [Google Scholar]
- Harris FP. Distortion-product otoacoustic emissions in humans with high frequency sensorineural hearing loss. Journal of speech and hearing research. 1990;33:594–600. doi: 10.1044/jshr.3303.594. [DOI] [PubMed] [Google Scholar]
- Henry KS, Lucas JR. Vocally correlated seasonal auditory variation in the house sparrow (Passer domesticus) The Journal of experimental biology. 2009;212:3817–3822. doi: 10.1242/jeb.033035. [DOI] [PubMed] [Google Scholar]
- Hultcrantz M, Simonoska R, Stenberg AE. Estrogen and hearing: a summary of recent investigations. Acta oto-laryngologica. 2006;126:10–14. doi: 10.1080/00016480510038617. [DOI] [PubMed] [Google Scholar]
- Jerger J, Mauldin L. Prediction of sensorineural hearing level from the brain stem evoked response. Arch Otolaryngol. 1978;104:456–461. doi: 10.1001/archotol.1978.00790080038010. [DOI] [PubMed] [Google Scholar]
- Jewett DL, Romano MN, Williston JS. Human auditory evoked potentials: possible brain stem components detected on the scalp. Science (New York, NY. 1970;167:1517–1518. doi: 10.1126/science.167.3924.1517. [DOI] [PubMed] [Google Scholar]
- Katayama A. Postnatal development of auditory function in the chicken revealed by auditory brain-stem responses (ABRs) Electroencephalography and clinical neurophysiology. 1985;62:388–398. doi: 10.1016/0168-5597(85)90048-6. [DOI] [PubMed] [Google Scholar]
- Kemp DT. Stimulated acoustic emissions from within the human auditory system. The Journal of the Acoustical Society of America. 1978;64:1386–1391. doi: 10.1121/1.382104. [DOI] [PubMed] [Google Scholar]
- Kemp DT. Otoacoustic emissions, their origin in cochlear function, and use. British medical bulletin. 2002;63:223–241. doi: 10.1093/bmb/63.1.223. [DOI] [PubMed] [Google Scholar]
- Kettembeil S, Manley GA, Siegl E. Distortion-product otoacoustic emissions and their anaesthesia sensitivity in the European starling and the chicken. Hearing research. 1995;86:47–62. doi: 10.1016/0378-5955(95)00053-7. [DOI] [PubMed] [Google Scholar]
- Liberman MC, Tartaglini E, Fleming JC, Neufeld EJ. Deletion of SLC19A2, the high affinity thiamine transporter, causes selective inner hair cell loss and an auditory neuropathy phenotype. J Assoc Res Otolaryngol. 2006;7:211–217. doi: 10.1007/s10162-006-0035-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lonsbury-Martin BL, Cutler WM, Martin GK. Evidence for the influence of aging on distortion-product otoacoustic emissions in humans. The Journal of the Acoustical Society of America. 1991;89:1749–1759. doi: 10.1121/1.401009. [DOI] [PubMed] [Google Scholar]
- Lucas JR, Freeberg TM, Krishnan A, Long GR. A comparative study of avian auditory brainstem responses: correlations with phylogeny and vocal complexity, and seasonal effects. Journal of comparative physiology. 2002;188:981–992. doi: 10.1007/s00359-002-0359-x. [DOI] [PubMed] [Google Scholar]
- Lucas JR, Freeberg TM, Long GR, Krishnan A. Seasonal variation in avian auditory evoked responses to tones: a comparative analysis of Carolina chickadees, tufted titmice, and white-breasted nuthatches. Journal of comparative physiology. 2007;193:201–215. doi: 10.1007/s00359-006-0180-z. [DOI] [PubMed] [Google Scholar]
- Maney DL, Cho E, Goode CT. Estrogen-dependent selectivity of genomic responses to birdsong. The European journal of neuroscience. 2006;23:1523–1529. doi: 10.1111/j.1460-9568.2006.04673.x. [DOI] [PubMed] [Google Scholar]
- Margoliash D. Acoustic parameters underlying the responses of song-specific neurons in the white-crowned sparrow. J Neurosci. 1983;3:1039–1057. doi: 10.1523/JNEUROSCI.03-05-01039.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marler P, Peters S, Ball GF, Dufty AM, Jr, Wingfield JC. The role of sex steroids in the acquisition and production of birdsong. Nature. 1988;336:770–772. doi: 10.1038/336770a0. [DOI] [PubMed] [Google Scholar]
- McClintock M. Menstrual Synchrony and Suppression. Nature. 1971;229:244. doi: 10.1038/229244a0. [DOI] [PubMed] [Google Scholar]
- McEwen BS. Non-genomic and genomic effects of steroids on neural activity. Trends in pharmacological sciences. 1991;12:141–147. doi: 10.1016/0165-6147(91)90531-v. [DOI] [PubMed] [Google Scholar]
- McFadden D. Masculinization of the mammalian cochlea. Hearing research. 2009;252:37–48. doi: 10.1016/j.heares.2009.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McFadden D, Pasanen EG, Raper J, Lange HS, Wallen K. Sex differences in otoacoustic emissions measured in rhesus monkeys (Macaca mulatta) Hormones and behavior. 2006;50:274–284. doi: 10.1016/j.yhbeh.2006.03.012. [DOI] [PubMed] [Google Scholar]
- Meitzen J, Moore IT, Lent K, Brenowitz EA, Perkel DJ. Steroid hormones act transsynaptically within the forebrain to regulate neuronal phenotype and song stereotypy. J Neurosci. 2007a;27:12045–12057. doi: 10.1523/JNEUROSCI.3289-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meitzen J, Perkel DJ, Brenowitz EA. Seasonal changes in intrinsic electrophysiological activity of song control neurons in wild song sparrows. Journal of comparative physiology. 2007b;193:677–683. doi: 10.1007/s00359-007-0222-1. [DOI] [PubMed] [Google Scholar]
- Meitzen J, Thompson CK, Choi H, Perkel DJ, Brenowitz EA. Time course of changes in Gambel’s white-crowned sparrow song behavior following transitions in breeding condition. Hormones and behavior. 2009a;55:217–227. doi: 10.1016/j.yhbeh.2008.10.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meitzen J, Weaver AL, Brenowitz EA, Perkel DJ. Plastic and stable electrophysiological properties of adult avian forebrain song-control neurons across changing breeding conditions. J Neurosci. 2009b;29:6558–6567. doi: 10.1523/JNEUROSCI.5571-08.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miranda JA, Wilczynski W. Female reproductive state influences the auditory midbrain response. Journal of comparative physiology. 2009a;195:341–349. doi: 10.1007/s00359-008-0410-7. [DOI] [PubMed] [Google Scholar]
- Miranda JA, Wilczynski W. Sex differences and androgen influences on midbrain auditory thresholds in the green tree-frog, Hyla cinera. Hearing research. 2009b doi: 10.1016/j.heares.2009.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mitsushima D, Takase K, Funabashi T, Kimura F. Gonadal steroids maintain 24 h acetylcholine release in the hippocampus: organizational and activational effects in behaving rats. J Neurosci. 2009;29:3808–3815. doi: 10.1523/JNEUROSCI.5301-08.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moller AR, Jannetta PJ. Compound action potentials recorded intracranially from the auditory nerve in man. Experimental neurology. 1981;74:862–874. doi: 10.1016/0014-4886(81)90259-4. [DOI] [PubMed] [Google Scholar]
- Moller AR, Jannetta PJ. Interpretation of brainstem auditory evoked potentials: results from intracranial recordings in humans. Scand Audiol. 1983;12:125–133. doi: 10.3109/01050398309076235. [DOI] [PubMed] [Google Scholar]
- Moller AR, Jannetta PJ, Moller MB. Neural generators of brainstem evoked potentials. Results from human intracranial recordings. The Annals of otology, rhinology, and laryngology. 1981;90:591–596. doi: 10.1177/000348948109000616. [DOI] [PubMed] [Google Scholar]
- Moore MC. Hormonal response of free-living male white-crowned sparrows to experimental manipulation of female sexual behavior. Hormones and behavior. 1982;16:323–329. doi: 10.1016/0018-506x(82)90030-7. [DOI] [PubMed] [Google Scholar]
- Nehls HB. Familiar Birds of the Northwest. Portland Audubon Society; Portland: 1981. [Google Scholar]
- Noirot IC, Adler HJ, Cornil CA, Harada N, Dooling RJ, Balthazart J, Ball GF. Presence of aromatase and estrogen receptor alpha in the inner ear of zebra finches. Hearing research. 2009;252:49–55. doi: 10.1016/j.heares.2009.04.012. [DOI] [PubMed] [Google Scholar]
- Nottebohm F. A brain for all seasons: cyclical anatomical changes in song control nuclei of the canary brain. Science (New York, NY. 1981;214:1368–1370. doi: 10.1126/science.7313697. [DOI] [PubMed] [Google Scholar]
- Okanoya K, Dooling RJ. Hearing in the swamp sparrow (Melospiza georgiana) and the song sparrow (Melospiza melodia) Anim Behav. 1988;36:726–732. [Google Scholar]
- Pak MW, Ng MH, Leung CB, van Hasselt CA. Cochlear deafness in a Chinese family with Fechtner’s syndrome. The American journal of otology. 2000;21:345–350. doi: 10.1016/s0196-0709(00)80043-5. [DOI] [PubMed] [Google Scholar]
- Park KH, Meitzen J, Moore IT, Brenowitz EA, Perkel DJ. Seasonal-like plasticity of spontaneous firing rate in a songbird pre-motor nucleus. Journal of neurobiology. 2005;64:181–191. doi: 10.1002/neu.20145. [DOI] [PubMed] [Google Scholar]
- Ponton CW, Moore JK, Eggermont JJ. Auditory brain stem response generation by parallel pathways: differential maturation of axonal conduction time and synaptic transmission. Ear and hearing. 1996;17:402–410. doi: 10.1097/00003446-199610000-00006. [DOI] [PubMed] [Google Scholar]
- Probst R, Lonsbury-Martin BL, Martin GK. A review of otoacoustic emissions. The Journal of the Acoustical Society of America. 1991;89:2027–2067. doi: 10.1121/1.400897. [DOI] [PubMed] [Google Scholar]
- Riters LV, Baillien M, Eens M, Pinxten R, Foidart A, Ball GF, Balthazart J. Seasonal variation in androgen-metabolizing enzymes in the diencephalon and telencephalon of the male European starling (Sturnus vulgaris) Journal of neuroendocrinology. 2001;13:985–997. doi: 10.1046/j.1365-2826.2001.00723.x. [DOI] [PubMed] [Google Scholar]
- Rosenhamer HJ, Lindstrom B, Lundborg T. On the use of click-evoked electric brainstem responses in audiological diagnosis. IV. Interaural latency differences (wave V) in cochlear hearing loss. Scand Audiol. 1981;10:67–73. doi: 10.3109/01050398109076164. [DOI] [PubMed] [Google Scholar]
- Sanford SE, Lange HS, Maney DL. Topography of estradiol-modulated genomic responses in the songbird auditory forebrain. Developmental neurobiology. 2009;70:73–86. doi: 10.1002/dneu.20757. [DOI] [PubMed] [Google Scholar]
- Schank JC. Measurement and cycle variability: reexamining the case for ovarian-cycle synchrony in primates. Behavioural processes. 2001;56:131–146. doi: 10.1016/s0376-6357(01)00194-2. [DOI] [PubMed] [Google Scholar]
- Schumacher M, Balthazart J. Testosterone-induced brain aromatase is sexually dimorphic. Brain research. 1986;370:285–293. doi: 10.1016/0006-8993(86)90483-x. [DOI] [PubMed] [Google Scholar]
- Sisneros JA. Seasonal plasticity of auditory saccular sensitivity in the vocal plainfin midshipman fish, Porichthys notatus. Journal of neurophysiology. 2009;102:1121–1131. doi: 10.1152/jn.00236.2009. [DOI] [PubMed] [Google Scholar]
- Sisneros JA, Bass AH. Seasonal plasticity of peripheral auditory frequency sensitivity. J Neurosci. 2003;23:1049–1058. doi: 10.1523/JNEUROSCI.23-03-01049.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sisneros JA, Forlano PM, Deitcher DL, Bass AH. Steroid-dependent auditory plasticity leads to adaptive coupling of sender and receiver. Science (New York, NY. 2004;305:404–407. doi: 10.1126/science.1097218. [DOI] [PubMed] [Google Scholar]
- Smith GT, Brenowitz EA, Beecher MD, Wingfield JC. Seasonal changes in testosterone, neural attributes of song control nuclei, and song structure in wild songbirds. J Neurosci. 1997;17:6001–6010. doi: 10.1523/JNEUROSCI.17-15-06001.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith GT, Brenowitz EA, Wingfield JC, Baptista LF. Seasonal changes in song nuclei and song behavior in Gambel’s white-crowned sparrows. Journal of neurobiology. 1995;28:114–125. doi: 10.1002/neu.480280110. [DOI] [PubMed] [Google Scholar]
- Soha JA, Marler P. A species-specific acoustic cue for selective song learning in the white-crowned sparrow. Anim Behav. 2000;60:297–306. doi: 10.1006/anbe.2000.1499. [DOI] [PubMed] [Google Scholar]
- Sohmer H, Feinmesser M, Szabo G. Sources of electrocochleographic responses as studied in patients with brain damage. Electroencephalography and clinical neurophysiology. 1974;37:663–669. doi: 10.1016/0013-4694(74)90081-9. [DOI] [PubMed] [Google Scholar]
- Soma KK, Hartman VN, Wingfield JC, Brenowitz EA. Seasonal changes in androgen receptor immunoreactivity in the song nucleus HVc of a wild bird. The Journal of comparative neurology. 1999;409:224–236. [PubMed] [Google Scholar]
- Soma KK, Schlinger BA, Wingfield JC, Saldanha CJ. Brain aromatase, 5 alpha-reductase, and 5 beta-reductase change seasonally in wild male song sparrows: relationship to aggressive and sexual behavior. Journal of neurobiology. 2003;56:209–221. doi: 10.1002/neu.10225. [DOI] [PubMed] [Google Scholar]
- Soma KK, Tramontin AD, Featherstone J, Brenowitz EA. Estrogen contributes to seasonal plasticity of the adult avian song control system. Journal of neurobiology. 2004;58:413–422. doi: 10.1002/neu.10288. [DOI] [PubMed] [Google Scholar]
- Starr A, Hamilton AE. Correlation between confirmed sites of neurological lesions and abnormalities of far-field auditory brainstem responses. Electroencephalography and clinical neurophysiology. 1976;41:595–608. doi: 10.1016/0013-4694(76)90005-5. [DOI] [PubMed] [Google Scholar]
- Steimer T, Hutchison JB. Androgen increases formation of behaviourally effective oestrogen in dove brain. Nature. 1981;292:345–347. doi: 10.1038/292345a0. [DOI] [PubMed] [Google Scholar]
- Stenberg AE, Wang H, Fish J, 3rd, Schrott-Fischer A, Sahlin L, Hultcrantz M. Estrogen receptors in the normal adult and developing human inner ear and in Turner’s syndrome. Hearing research. 2001;157:87–92. doi: 10.1016/s0378-5955(01)00280-5. [DOI] [PubMed] [Google Scholar]
- Stenberg AE, Wang H, Sahlin L, Hultcrantz M. Mapping of estrogen receptors alpha and beta in the inner ear of mouse and rat. Hearing research. 1999;136:29–34. doi: 10.1016/s0378-5955(99)00098-2. [DOI] [PubMed] [Google Scholar]
- Stevens SS, Poulton EC. The estimation of loudness by unpracticed observers. Journal of Experimental Psychology. 1956;51:71–78. doi: 10.1037/h0041089. [DOI] [PubMed] [Google Scholar]
- Swett MB, Breuner CW. Interaction of testosterone, corticosterone and corticosterone binding globulin in the white-throated sparrow (Zonotrichia albicollis) Comp Biochem Physiol A Mol Integr Physiol. 2008;151:226–231. doi: 10.1016/j.cbpa.2008.06.031. [DOI] [PubMed] [Google Scholar]
- Terleph TA, Lu K, Vicario DS. Response properties of the auditory telencephalon in songbirds change with recent experience and season. PLoS ONE. 2008;3:e2854. doi: 10.1371/journal.pone.0002854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tramontin AD, Hartman VN, Brenowitz EA. Breeding conditions induce rapid and sequential growth in adult avian song control circuits: a model of seasonal plasticity in the brain. J Neurosci. 2000;20:854–861. doi: 10.1523/JNEUROSCI.20-02-00854.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tramontin AD, Wingfield JC, Brenowitz EA. Androgens and estrogens induce seasonal-like growth of song nuclei in the adult songbird brain. Journal of neurobiology. 2003;57:130–140. doi: 10.1002/neu.10263. [DOI] [PubMed] [Google Scholar]
- Tremere LA, Jeong JK, Pinaud R. Estradiol shapes auditory processing in the adult brain by regulating inhibitory transmission and plasticity-associated gene expression. J Neurosci. 2009;29:5949–5963. doi: 10.1523/JNEUROSCI.0774-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vyas A, Harding C, Borg L, Bogdan D. Acoustic characteristics, early experience, and endocrine status interact to modulate female zebra finches’ behavioral responses to songs. Hormones and behavior. 2009;55:50–59. doi: 10.1016/j.yhbeh.2008.08.005. [DOI] [PubMed] [Google Scholar]
- Wingfield JC, Crim JW, Mattocks PW, Jr, Farner DS. Responses of photosensitive and photorefractory male white-crowned sparrows (Zonotrichia leucophrys gambelii) to synthetic mammalian luteinizing hormone releasing hormone (syn-LHRH) Biology of reproduction. 1979;21:801–806. doi: 10.1095/biolreprod21.4.801. [DOI] [PubMed] [Google Scholar]
- Wingfield JC, Farner DS. The annual cycle of plasma irLH and steroid hormones in feral populations of the white-crowned sparrow, Zonotrichia leucophrys gambelii. Biology of reproduction. 1978;19:1046–1056. doi: 10.1095/biolreprod19.5.1046. [DOI] [PubMed] [Google Scholar]
- Woolley SM, Wissman AM, Rubel EW. Hair cell regeneration and recovery of auditory thresholds following aminoglycoside ototoxicity in Bengalese finches. Hearing research. 2001;153:181–195. doi: 10.1016/s0378-5955(00)00217-3. [DOI] [PubMed] [Google Scholar]
- Zakon HH. The effects of steroid hormones on electrical activity of excitable cells. Trends in neurosciences. 1998;21:202–207. doi: 10.1016/s0166-2236(97)01209-5. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.