Abstract
Background and objectives: Characterizing relationships of kidney function to healthcare costs in polycystic kidney disease has applications for economic evaluations of standard and emerging therapies.
Design, setting, participants, & measurements: The administrative records (2003 to 2006) of a private health insurer were examined to identify polycystic kidney disease patients (n = 1913) from ICD9 diagnosis codes on billing claims. The first available diagnostic claim was assumed as an index date, and baseline estimated GFR (eGFR) was computed using closest serum creatinine value. The associations of eGFR with annualized charges were modeled by nonlinear and linear regression.
Results: Medical, pharmacy, and total healthcare costs varied significantly by baseline kidney function, such that mean total annualized charges (unadjusted) were approximately 5-fold higher in patients with eGFR < 15 ml/min compared with those with eGFR ≥ 90 ml/min. After adjustment for age and gender, total charges did not differ significantly among patients with eGFR > 30 ml/min, and but rose precipitously with eGFR < 30 ml/min. Each ml/min decline <30 ml/min predicted approximately $5435 higher adjusted annual charges. Results were similar after adjustment for baseline diabetes and cardiovascular disease as identified in claims, while significantly higher adjusted charges were detected with eGFR = 31 to 60 ml/min versus ≥90 ml/min in a subgroup free of diabetes and cardiovascular disease.
Conclusions: Healthcare charges are associated with advanced renal dysfunction in polycystic kidney disease patients. Strategies that prevent loss of renal function below 30 ml/min have the potential to generate substantial reductions in medical charges.
Autosomal dominant polycystic kidney disease (ADPKD) is the most common inherited cystic renal disease as well as the most common single-gene disorder of the kidneys. ADPKD affects approximately 600,000 persons in the United States and 10 million persons worldwide (1). The natural history of ADPKD is one of inevitable pathologic and clinical progression over time, characterized by growth in the number and size of cysts with secondary destruction of functional parenchyma. A recent prospective study of structural and functional progression among 232 ADPKD patients with creatinine clearance >70 ml/min at enrollment documented average increases in total kidney volume of 204 ± 246 ml over 3 years, and decrements in GFR of 4.3 ± 8.1 ml/min per year in participants with baseline kidney volume >1500 ml (2). Incidence estimates for ESRD among ADPKD patients have ranged from 45% to 70% by age 65 years (3–6). As a manifestation of its progressive course, ADPKD ranks as the fourth most common cause of ESRD among adults in the United States (7). At present, no medical therapy has proven to substantially slow the growth of ADPKD-related cysts or the volumetric expansion of polycystic kidneys. However, innovative strategies to blunt renal tubular cell proliferation and fluid secretion, the processes that drive growth of cystic bodies, comprise foci of active research (8,9).
Considered broadly without respect to underlying cause, chronic kidney disease (CKD) is recognized as a source of substantial morbidity and economic burdens. Epidemiologic studies have demonstrated associations between kidney dysfunction and increased risks of adverse outcomes including cardiovascular disease events, hospitalizations, resource utilization, and mortality (10,11). For example, based on data from a large Healthcare Maintenance Organization (HMO), Smith et al. found that patients with CKD stages 2 to 4 had 1.9 to 2.5 times more prescriptions, 1.3 to 1.9 times more outpatient visits, and 1.8 to 4.2 times more inpatient stays than age- and gender-matched controls (11). Notably, however, costs were similar across these functional stages within the CKD sample. Characterizing relationships of kidney function to healthcare costs among patients with CKD due to polycystic kidney disease has practical applications for economic evaluations of standard and emerging therapies in this disease. While ADPKD confers important public health impacts based on its progressive nature within affected patients and relative frequency among heritable disorders, the disease is less common than leading causes of CKD such as diabetes, which by comparison affects approximately 7.2% of the adult population in the United States (15 million Americans) (12). The challenge of assembling large cohorts for outcomes analyses in ADPKD can be addressed by study designs that integrate data across multiple centers or providers.
To advance understanding of the association of kidney function with healthcare costs in polycystic kidney disease, we performed a retrospective study of the administrative records of a large health insurance provider. Administrative insurance data compile dated records of physician encounters, hospitalizations, procedures, and prescriptions that may be examined to classify groups with diagnoses of interest and to study associated resource utilization and healthcare costs. Using linked administrative billing and laboratory data, we sought to identify a cohort of polycystic kidney disease patients receiving care in contemporary practice and to characterize the relationship of kidney function with medical and pharmaceutical charges to the insurer across a broad spectrum of renal function levels.
Materials and Methods
Data Source
Study data were drawn from the administrative records of a large national health insurance provider. The insurance database contains all medical as well as outpatient pharmacy billing claims submitted for beneficiaries during periods of eligibility. Individual claim records contain the date in which a given claim was paid, date in which services were performed, diagnostic and procedure codes, place of service codes, revenue codes, and amount charged. The total population in the database numbers 6 million individuals from across the United States. For the purpose of this analysis, billing claims data were augmented with extracts from outpatient labs for a subsample of beneficiaries, permitting combined examinations of lab results along with expenditures for healthcare services and prescription medications. Data were available for claims and labs submitted in January 2003 to December 2006. Final samples were limited to patients with at least 6 months of insurance eligibility after the diagnosis or capture date of interest. All study participants were simultaneously enrolled in medial and pharmacy benefits with this company exclusively during the study window. This study was approved by the Institutional Review Board of Saint Louis University.
Identification of Patients with Polycystic Kidney Disease
Patients with ADPKD were identified by billing claims with a corresponding International Classification of Diseases 9th Edition (ICD-9) code (753.13, “polycystic kidney disease, autosomal dominant”) within the available claims data. As cysts may be recognized in ADPKD patients before the diagnosis is formalized, we also considered claims for “polycystic kidney, unspecified type” (753.12). In categorizing patients as 753.13 versus 753.12, a mutually exclusive diagnostic hierarchy was used such that individuals with any appearance of 753.13 codes during observation were considered to have ADPKD, whereas patients without 753.13 claims but any 753.12 claims were considered to have polycystic kidney disease/unspecified. We defined the first diagnosis claim in the data set as the analysis “index date.”
Classification of Renal Function and Baseline Characteristics
Age and gender of the study samples were ascertained from insurance eligibility files. Renal function was computed as estimated GFR (eGFR) from closest serum creatinine values reported in the laboratory results database within ±365 days of index polycystic kidney disease claim. eGFR was computed according to the abbreviated Modification of Diet in Renal Disease (aMDRD) equation (13) without racial adjustment as: eGFR (ml/min per 1.73 m2) = 186 × (serum creatinine) − 1.154 × (Age) − 0.203 × (0.742 if female). As race information is not tracked in the administrative data, we assumed all subjects to be nonblack in accord with “best practice in laboratory medicine” recommendations (14) and the approach of previous investigations with administrative data (15,16). The computed eGFR was assumed to apply to the individual at the time of the index claim. Level of kidney function was classified according to the GFR-based schema of the National Kidney Foundation, Kidney Disease Outcomes Quality Initiative (NKF/KDOQI) (17) as: ≥90, 60 to 89, 30 to 59, 15 to 29, or <15 ml/min. To restrict the sample to patients with pre-ESRD polycystic diseases, we excluded individuals with billing claims before the index date for dialysis, transplantation, and/or associated complications based on ICD-9 diagnosis codes (V45.11, V45.12, 585.6, V56.0, V56.31, V56.32, V56.8,, E870.2, E871.2, E872.2, E874.2, E879.1, 996.56, 996.68, 996.73, 996.62, 996.81), ICD-9 procedure codes (39.95, 54.98, 55.69, 00.91, 00.92, 00.93), Common Procedural Terminology/Healthcare Common Procedure Coding System codes (90918–90937, 90945, 90947, 90951 to 90970, 90999, 99512, 99559, G0308 to G0327, 0505F, 0507F, 4052F, 4053F, 4054F, 4055F, 50360, 50365), or the Diagnosis Related Group for kidney transplantation (302).
The presence of comorbid diabetes mellitus and of cardiovascular disease at baseline sampling were defined by billing claims with corresponding ICD-9 diagnosis codes within ±30 days of the index date, using coding algorithms similar to previous reports (18–22) as: Diabetes mellitus (250, 357.2, 362.0, 366.41, 648.0); cardiovascular disease composite including coronary artery disease (414), myocardial infarction (410.x), angina pectoris (411,413), heart failure (398.91, 422, 425, 428, 402.x1, 404.x1, 404.x3, V42.1), or stroke/transient ischemic attack (433 to 435).
Ascertainment of Healthcare Charges
Costs for all medical services and outpatient pharmaceutical prescriptions were derived from charges reported on claims paid by the insurer. Monetary figures were adjusted to the prices in the year 2008 Medical Care Component of the Consumer Price Index (23). Annualized charges were computed among patients with at least 6 months of insurance eligibility after the index date to avoid magnification of outlier effects in patients captured with short eligibility windows. Annualization was performed by aggregating total charges per patient over 6 months after index date and multiplying by 2 to represent estimated costs for 1 year of care. Medical charges from inpatient sources were also examined separately. The charge contributions of renal replacement services were examined among those who required sustained dialysis (defined as at least two dialysis claims over ≥30 days) or kidney transplant during observation. Dialysis charges were annualized among dialysis-dependent patients, whereas transplant charges were defined as those associated with the transplant hospitalization.
Statistical Analyses
Data sets were merged and analyzed with SAS for Windows software, version 9.2 (SAS Institute Inc., Cary, NC). Distributions of age, gender, and baseline diabetes and cardiovascular disease diagnoses in patients with specified ADPKD/753.13 versus unspecified/753.12 polycystic kidney disease, as well as trait distributions by eGFR level within a given group, were compared by the two-sample t test (or ANOVA) for age and the Chi-square test for categorical factors. The Kruskal-Wallis test, a nonparametric equivalent to one-way ANOVA, was used to compare insurance charges in patients with specified ADPKD versus unspecified polycystic kidney disease, as well as to compare unadjusted charges across eGFR levels. Pairwise comparisons of unadjusted charges in a given eGFR level versus eGFR > 90 ml/min were performed by the Wilcoxon test, with statistical significance for pairwise comparisons defined as P < 0.01 based on a Bonferroni correction for multiple comparisons. To assess the shape of the relationship of eGFR with annualized healthcare charges, a general linear model incorporating splines was constructed using a mid-range eGFR (60 ml/min) as a reference and including form break points (knots) at 15, 30, 45, and 90 ml/min. The resulting shape informed construction of a linear regression model including adjustment for gender and age. Models were constructed for total charges as well as components of total medical (excluding outpatient pharmacy), inpatient medical, and pharmacy charges. Additional linear regressions on charges were fit adding comorbidities of diabetes, cardiovascular disease and their interaction, as well as in a sample free of baseline diabetes and cardiovascular disease.
Results
Sample Characteristics
Among 2,041,973 patients in the administrative database with at least one reported serum creatinine result within their period of observed insurance benefits eligibility and at least 6 months of benefits, 1913 (0.09%) had billing claims with diagnosis codes for polycystic kidney disease (496 with 753.13; 1417 with 753.12 alone). Among patients categorized as ADPKD/753.13, 245 (49.4%) also had claims with unspecified/753.12 polycystic kidney disease diagnosis codes in the available claims data. There were no significant differences in the distributions of age, gender, or baseline diabetes or cardiovascular disease in those with diagnosis codes for ADPKD, specifically compared with unspecified polycystic kidney disease (Table 1). The eGFR distributions in patients with 753.13 diagnoses compared with 753.12 alone also did not significantly differ (P = 0.31): >90 ml/min: 13.7% versus 16.0%; 60 to 89 ml/min: 33.7% versus 37.3%; 30 to 59 ml/min: 31.9% versus 29.9%; 15 to 29 ml/min: 12.1% versus 10.0%; <15 ml/min: 8.7% versus 6.5%. The mean age of the combined study sample of polycystic kidney patients was 46.7 years, and 43% were men. Diabetes and cardiovascular disease were present in 5.3% and 2.2% of the full cohort, respectively; 1781 subjects were free of both these diagnoses. Compared with patients in the combined cohort with eGFR > 90 ml/min, those with lower levels of renal function were older, more commonly male, and had a higher baseline prevalence of cardiovascular disease.
Table 1.
Demographic traits of the study sample by polycystic kidney disease diagnosis and kidney function level
| Combined Cohort, n (%)a | ADPKD, ICD9 753.13, n (%)a | Polycystic Kidney Unspecified, ICD9 753.12, n (%)a | |
|---|---|---|---|
| Full Cohort | n = 1913 | n = 496 | n = 1417 |
| Age in years, mean ± SD | 46.7 ± 12.5b | 46.8 ± 12.4b | 46.7 ± 12.5b |
| Male gender | 828 (43.3)b | 221 (44.6)b | 607 (42.8)b |
| Diabetes | 102 (5.3) | 31 (6.3) | 71 (5.0) |
| Cardiovascular disease | 43 (2.2)b | 13 (2.6)b | 30 (2.1) |
| eGFR ≥90 | n = 295 | n = 68 | n = 227 |
| Age in years, mean ± SD | 37.4 ± 12.1 | 36.9 ± 11.9 | 37.6 ± 12.2 |
| Male gender | 94 (31.9) | 23 (33.8) | 71 (31.3) |
| Diabetes | 14 (4.8) | 3 (4.4) | 11 (4.9) |
| Cardiovascular disease | 3 (1.0) | 0 | 3 (1.3) |
| eGFR 60 to 89 | n = 696 | n = 167 | n = 529 |
| Age in years, mean ± SD | 44.1 ± 11.5 | 43.5 ± 11.3 | 44.2 ± 11.6 |
| Male gender | 272 (39.1) | 67 (40.1) | 205 (38.8) |
| Diabetes | 31 (4.5) | 8 (4.8) | 23 (4.4) |
| Cardiovascular disease | 5 (0.7) | 3 (1.8) | 2 (0.4) |
| eGFR 30 to 59 | n = 582 | n = 158 | n = 424 |
| Age in years, mean ± SD | 51.2 ± 11.0 | 50.3 ± 11.0 | 51.5 ± 11.0 |
| Male gender | 299 (51.4) | 79 (50.0) | 220 (51.9) |
| Diabetes | 33 (5.7) | 15 (9.5) | 18 (4.3) |
| Cardiovascular disease | 18 (3.1) | 7 (4.4) | 11 (2.6) |
| eGFR 15 to 29 | n = 205 | n = 60 | n = 145 |
| Age in years, mean ± SD | 53.1 ± 10.1 | 53.4 ± 9.1 | 53.0 ± 10.5 |
| Male gender | 98 (47.8) | 34 (56.7) | 64 (44.1) |
| Diabetes | 13 (6.3) | 3 (5.0) | 10 (6.9) |
| Cardiovascular disease | 8 (3.9) | 1 (1.7) | 7 (4.8) |
| eGFR <15 | n = 135 | n = 43 | n = 92 |
| Age in years, mean ± SD | 52.1 ± 11.2 | 52.7 ± 12.5 | 51.8 ± 10.6 |
| Male gender | 65 (48.2) | 18 (41.9) | 47 (51.1) |
| Diabetes | 11 (8.2) | 2 (4.6) | 9 (9.8) |
| Cardiovascular disease | 9 (6.7) | 2 (4.6) | 7 (7.6) |
Percentages reflect proportions within the polycystic kidney diagnosis code category with the indicated demographic or clinical traits (“column percentages”).
There were no statistically significant differences in distributions of listed clinical traits in 753.13 and 743.12 subgroups at the P < 0.05 level.
P < 0.05 for variation in the distribution of clinical traits according to eGFR level within a polycystic kidney diagnosis code category.
Unadjusted Annualized Charges by eGFR Level
There were no significant differences in annualized charges among patients with diagnosis codes for ADPKD versus unspecified polycystic kidney disease (P values for charge comparisons: Total medical, 0.69; inpatient, 0.54; outpatient pharmacy, 0.31; total charges, 0.76). A combined study sample of patients with 753.12 and 753.13 diagnosis codes was thus considered in subsequent analyses. Annualized medical and pharmaceutical charges differed significantly across levels of kidney function in the study sample (Table 2). Specifically, mean annual medical charges (unadjusted) increased from $24,427 in polycystic kidney disease patients with baseline eGFR >90 ml/min to $134,784 in those with baseline eGFR < 15 ml/min (P < 0.0001). Considering inpatient medical charges separately, mean annual inpatient expenses were $10,721 in polycystic kidney disease patients with baseline eGFR ≥ 90 ml/min but were $37,304 in those patients with eGFR < 15 ml/min (P < 0.0001). Mean annual outpatient pharmacy charges increased from $2093 to $5355 across baseline eGFR levels of >90 ml/min to <15 ml/min (P < 0.0001), and mean total healthcare charges per year rose from $26,521 to $140,139 across these levels of renal function (P < 0.0001).
Table 2.
Annualized medical, pharmacy and total charges according to kidney function level in the sample of polycystic kidney disease patientsa
| eGFR Level | n | Medical Charges ($) |
||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Total Medical |
Inpatient Medical |
Outpatient Pharmacy Charges ($) |
Total Charges ($) |
|||||||||||
| Mean | Median | Mean | Median | Mean | Median | Mean | Median | |||||||
| ≥90 | 295 | 24,427 | 5758 | 10,721 | 0 | 2093 | 714 | 26,521 | 7669 | |||||
| 60 to 89 | 696 | 19,064 | 4532 | 8300 | 0 | 2296 | c | 1054 | 21,360 | 6660 | ||||
| 30 to 59 | 582 | 27,093 | 6381 | 13,185 | c | 0 | 4154 | c | 1744 | 31,247 | c | 9423 | ||
| 15 to 29 | 205 | 38,549 | 7513 | 19,374 | c | 0 | 3257 | c | 1818 | 41,806 | c | 10,499 | ||
| <15 | 135 | 134,784 | c | 42,910 | 37,304 | c | 0 | 5355 | c | 3317 | 140,139 | c | 46,779 | |
| Pb | <0.0001 | <0.0001 | <0.0001 | <0.0001 | ||||||||||
Charges were adjusted to the $2008 medical component of the Consumer Price Index.
Variation in annualized costs by eGFR level was compared by the Kruskal-Wallis test, a nonparametric equivalent to one-way ANOVA. Pairwise comparisons of unadjusted charges in a given eGFR level versus eGFR >90 ml/min were performed by the Wilcoxon test, with statistical significance for pairwise comparisons defined as P < 0.01 (c) based on a Bonferroni correction for multiple comparisons.
There were 187 patients who progressed to dialysis-dependence during observation (proportions per each baseline eGFR level: ≥90 ml/min: 2.0%; 60 to 89 ml/min: 1.4%; 30 to 59 ml/min: 5.4%; 15 to 29 ml/min: 22.4%; <15 ml/min: 68.8%). The mean annualized charges for dialysis services were $131,890. Thirty-five patients received kidney transplants during observation (proportions per baseline eGFR level: ≥60 ml/min: 0%; 30 to 59 ml/min: 0.9%; 15 to 29 ml/min: 5.9%; <15 ml/min: 16.3%). The mean charges per transplant hospitalization were $119,931. Among patients with eGFR < 15ml/min at baseline, 71% of total medical charges were related to dialysis or transplant services.
Nonlinear and Linear Regression
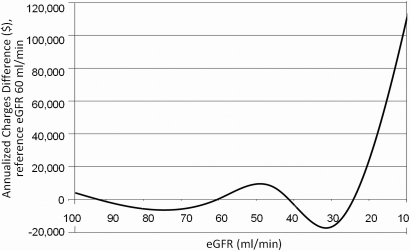
Exploration of the relationship of eGFR with total annualized healthcare charges by spline regression in the study sample is shown in Figure 1. This method compares additional charges associated with each ml/min eGFR difference compared with a mid-range reference of 60 ml/min using a flexible functional form. The resulting plot demonstrates mild fluctuation in charges around a baseline of $0 (negligible difference from reference) until eGFR falls below 30 ml/min, at which point annual costs rise precipitously with further decrements in eGFR.
Figure 1.
Associations of eGFR (referenced to 60 ml/min) with annualized total healthcare charges in polycystic kidney disease patients by spline regression. Plot illustrates annual charge differences compared with reference eGFR of 60 ml/min. Charges were adjusted to the $2008 medical component of the Consumer Price Index.
The exploration in Figure 1 was used to inform construction of a multivariate linear regression model. Here annual charges associated with kidney function >30 ml/min were considered according to KDOQI levels rather than by ml/min changes. Patients with eGFR >90 ml/min were chosen as the reference on clinical grounds, and models were adjusted for age and gender. While eGFR levels 31 to 60 ml/min were associated with a trend toward higher annual charges compared with eGFR > 90 ml/min (P = 0.21), the effect estimates for function levels >30 ml/min were nonsignificant. Given the sharp rise in charges with eGFR < 30 ml/min seen using the spline form, we modeled the charges associated with each ml/min decline below 30 ml/min. By the resulting linear fit, each ml/min decline in eGFR < 30 was associated with an increase in adjusted annual charges of approximately $5435 compared with eGFR > 90 ml/min (P < 0.0001). Patterns were similar for total medical (excluding pharmacy) charges, such that each ml/min decrement in eGFR was associated with $5332 higher adjusted annual medical charges. Outpatient pharmacy charges, which comprised a smaller fraction of total charges, showed an increase at earlier stages of kidney dysfunction. Adjusted annual pharmacy charges were approximately $1500 higher in patients with eGFR = 31 to 60 ml/min compared with >90 ml/min (P = 0.003), and then rose an estimated $199 per ml/min decline <30 (P = 0.001). By contrast, inpatient medical charges were not significantly associated with renal function after covariate adjustment.
Although not common in the study sample of polycystic kidney patients, both comorbid diabetes mellitus and cardiovascular disease were associated with increased healthcare charges (Table 3). In the absence of cardiovascular disease, diabetes at baseline predicted approximately $27,847 higher annual charges independent of age, gender, and eGFR. In the absence of diabetes, cardiovascular disease was associated with approximately $188,220 higher annual charges independent of age, gender, and eGFR. There was a significant interaction between these conditions in relation to costs, such that comorbid diabetes and cardiovascular disease had similar cost implications as diabetes alone in this sample. The age and gender-adjusted association of eGFR with annual charges in the full sample was similar after additional adjustment for diabetes and cardiovascular disease. Specifically, each ml/min decline in eGFR <30 was associated with $5316 higher adjusted annual charges.
Table 3.
Relationships of eGFR with annualized healthcare charges by multivariate linear regression in the full study sample (n = 1913)a
| Variables | Parameter Estimate ($ per year [SEM])b | P | Parameter Estimate ($ per year [SEM])c | P |
|---|---|---|---|---|
| Total Charges | ||||
| Intercept | −5886 (8537) | 0.49 | 3197 (8266) | 0.70 |
| Age (per year) | 579 (174) | 0.0009 | 301 (170) | 0.08 |
| Male gender | −107 (4134) | 0.98 | −2271 (3988) | 0.57 |
| eGFR | ||||
| >90 ml/min level | reference | reference | ||
| 61 to 90 ml/min level | 1866 (5656) | 0.74 | 3556 (5456) | 0.51 |
| 31 to 60 ml/min level | 7590 (6087) | 0.21 | 7738 (5869) | 0.19 |
| Per ml/min ≤30 | 5435 (449) | <0.0001 | 5316 (433) | <0.0001 |
| Diabetes mellitus | — | — | 27,847 (9292) | 0.0028 |
| Cardiovascular disease | — | — | 188,220 (15847) | <0.0001 |
| Diabetes–cardiovascular disease interaction | — | — | −185,935 (29767) | <0.0001 |
| Total Medical Charges | ||||
| Intercept | −6228 (8551) | 0.47 | 2791 (8281) | 0.74 |
| Age (per year) | 547 (174) | 0.0017 | 273 (170) | 0.11 |
| Male gender | −865 (4162) | 0.84 | −2981 (4015) | 0.46 |
| eGFR | ||||
| >90 ml/min level | reference | reference | ||
| 61 to 90 ml/min level | 2067 (5676) | 0.72 | 3702 (5476) | 0.50 |
| 31 to 60 ml/min level | 6292 (6096) | 0.30 | 6346 (5878) | 0.28 |
| Per ml/min ≤30 | 5332 (448) | <0.0001 | 5215 (433) | <0.0001 |
| Diabetes mellitus | — | — | 25,104 (9265) | 0.007 |
| Cardiovascular disease | — | — | 186,664 (15797) | <0.0001 |
| Diabetes–cardiovascular disease interaction | — | — | −184,495 (29667) | <0.0001 |
| Inpatient Charges | ||||
| Intercept | −3465 (27799) | 0.90 | 17,240 (26967) | 0.52 |
| Age (per year) | 1437 (520) | 0.006 | 998 (517) | 0.05 |
| Male gender | −4782 (14202) | 0.74 | −10,690 (13628) | 0.43 |
| eGFR | ||||
| >90 ml/min level | reference | reference | ||
| 61 to 90 ml/min level | 1262 (20653) | 0.95 | 680 (19776) | 0.97 |
| 31 to 60 ml/min level | −5328 (20169) | 0.79 | −11,895 (19331) | 0.54 |
| Per ml/min <30 | 783 (1164) | 0.50 | 563 (1114) | 0.61 |
| Diabetes mellitus | — | — | −18,965 (24284) | 0.44 |
| Cardiovascular disease | — | — | 176,913 (30285) | <0.0001 |
| Diabetes–cardiovascular disease interaction | — | — | −193,981 (53821) | 0.0004 |
| Outpatient Pharmacy Charges | ||||
| Intercept | 542 (712) | 0.45 | 674 (714) | 0.35 |
| Age (per year) | 41 (15) | 0.0045 | 35 (15) | 0.02 |
| Male gender | 581 (343) | 0.09 | 566 (343) | 0.10 |
| eGFR | ||||
| >90 ml/min level | reference | reference | ||
| 61 to 90 ml/min level | 18 (472) | 0.97 | 37 (471) | 0.94 |
| 31 to 60 ml/min level | 1491 (502) | 0.003 | 1514 (501) | 0.003 |
| Per ml/min ≤30 | 119 (37) | 0.001 | 118 (36) | 0.001 |
| Diabetes mellitus | — | — | 2484 (789) | 0.002 |
| Cardiovascular disease | — | — | 1066 (1341) | 0.43 |
| Diabetes–cardiovascular disease interaction | — | — | −1514 (2464) | 0.54 |
Charges were adjusted to the $2008 medical component of the Consumer Price Index.
Regression model among the full sample, excluding parameters for diabetes and cardiovascular disease.
Regression model among the full sample, including parameters for diabetes and cardiovascular disease.
Considering the sample of 1781 polycystic kidney disease patients who were free of diabetes and cardiovascular disease diagnoses at baseline, eGFR = 31 to 60 ml/min was associated with approximately $10,788 higher total annual charges compared with eGFR >90 ml/min that now reached statistical significance (P = 0.045) (Table 4). Subsequent decrements in eGFR < 30 min/ml were associated with estimated increases in total charges of $4914 per ml/min. Associations of eGFR with total medical only, inpatient and outpatient pharmacy charges in the sample without diabetes and cardiovascular disease were similar to results in the full sample.
Table 4.
Relationships of eGFR with annualized healthcare charges by multivariate linear regression in the polycystic kidney disease sample free of baseline diabetes and cardiovascular disease (n = 1781)a
| Variable | Parameter Estimate ($ per year [SEM]) | P |
|---|---|---|
| Total Charges | ||
| Intercept | 3403 (7540) | 0.65 |
| Age (per year) | 318 (156) | 0.04 |
| Male gender | −4664 (3641) | 0.20 |
| eGFR | ||
| >90 ml/min level | reference | |
| 61 to 90 ml/min level | 3344 (4966) | 0.50 |
| 31 to 60 ml/min level | 10,788 (5386) | 0.045 |
| Per ml/min <30 | 4914 (403) | <0.0001 |
| Total Medical Charges | ||
| Intercept | 3042 (7541) | 0.69 |
| Age (per year) | 291 (156) | 0.06 |
| Male gender | −5581 (3660) | 0.13 |
| eGFR | ||
| >90 ml/min level | reference | |
| 61 to 90 ml/min level | 3460 (4975) | 0.49 |
| 31 to 60 ml/min level | 9604 (5384) | 0.07 |
| Per ml/min ≤30 | 4805 (402) | <0.0001 |
| Inpatient Charges | ||
| Intercept | −5199 (26,584) | 0.85 |
| Age (per year) | 1339 (520) | 0.01 |
| Male gender | −27,079 (13,632) | 0.048 |
| eGFR | ||
| >90 ml/min level | reference | |
| 61 to 90 ml/min level | 5722 (19,262) | 0.77 |
| 31 to 60 ml/min level | 10,428 (19,194) | 0.59 |
| Per ml/min <30 | 1713 (1106) | 0.12 |
| Pharmacy Charges | ||
| Intercept | 595 (735) | 0.42 |
| Age (per year) | 35 (15) | 0.02 |
| Male gender | 681 (353) | 0.05 |
| eGFR | ||
| >90 ml/min level | reference | |
| 61 to 90 ml/min level | 108 (484) | 0.82 |
| 31 to 60 ml/min level | 1428 (518) | 0.006 |
| Per ml/min <30 | 128 (38) | 0.0009 |
Charges were adjusted to the $2008 medical component of the Consumer Price Index.
Discussion
ADPKD is a progressive genetic disorder that leads to advanced kidney failure in approximately half of affected patients by mid-adulthood. We examined administrative records of a large health insurance provider to assemble a cohort of patients with polycystic kidney disease and investigate the relationship of renal function with healthcare charges to the insurer. The study yielded several key findings: (1) medical, pharmacy, and total healthcare costs varied significantly by baseline kidney function level, such that mean total annual charges (unadjusted) were approximately fivefold higher in polycystic kidney patients with eGFR < 15 ml/min compared with those with eGFR ≥ 90 ml/min; (2) after adjustment for age and gender, total charges did not differ significantly in the full sample among those with eGFR > 30 ml/min, but rose precipitously in patients with eGFR <30 ml/min compared with higher levels of function; (3) comorbid diabetes and cardiovascular disease were significant correlates of charges in patients with polycystic kidney disease, but the association of eGFR and charges persisted after adjustment for these comorbidities; and (4) in a subgroup free of diabetes and/or cardiovascular disease at baseline, a significant rise in adjusted charges was detected at eGFR = 31 to 60 compared with >90 ml/min.
Quantification of the costs associated with health states offer valuable inputs for economic evaluations including cost-benefit analyses of standard and emerging disease management strategies. The dramatic increase in healthcare charges for polycystic kidney disease patients with eGFR < 30 ml/min is important but not surprising. While the study sample in this analysis was selected to be free of indications of dialysis or transplantation at baseline, GFR <30 ml/min is the functional level at which many patients begin preparing for dialysis or transplant and face increased burdens of kidney failure-related complications such as anemia, hypertension, and bone and mineral disorders (24). Renal replacement therapy often begins after GFR falls below 10 to 15 ml/min. Notably, outpatient medical charges appeared to be the main driver of the association of eGFR with total charges in this analysis, with a lesser but consistent contribution from outpatient pharmacy charges, whereas inpatient charges were not significantly associated with renal function in this sample.
Other investigators have shown positive associations between healthcare costs and proximity to dialysis in CKD patients. For example, St. Peter et al. found that costs in a Medicare/Medicaid population increased from $993 per-patient per-month 2 years before dialysis to $6300 per-patient per-month 6 months after dialysis initiation, with a positive gradient in between these periods (25). Similar observations have been made in managed care populations (26). Consistent with our observation of nonsignificant changes in adjusted charges in polycystic kidney disease patients with eGFR > 90 to 30 ml/min, Smith et al. found that while care of CKD patients was more expensive than that of controls, provider-side costs were stable across pre-ESRD CKD stages 2 to 4 in an HMO setting (11). This study extends understanding of relationships of renal function and healthcare costs to patients with polycystic kidney disease as the cause of CKD, demonstrating sharp accrual of economic impact at advanced stages of renal insufficiency. Assuming novel interventions to slow progression of ADPKD prove clinically effective in the future, the economic benefits of such interventions are likely to be greatest when function loss below approximately 30 ml/min can be prevented.
Cardiovascular disease has been regarded as the most costly disease in the United States, as the combination of high prevalence and case management that may entail expensive procedures was estimated to account for $400 billion direct and indirect costs for the nation in 2006 (27). Diabetes mellitus was associated with approximately $174 billion in total costs in the United States in 2007 (28), although some of the costs of diabetes are mediated by end organ complications such as cardiovascular disease and kidney failure. We therefore estimated the associations of baseline cardiovascular disease and diabetes with healthcare charges in our sample of polycystic kidney disease patients. While uncommon in this sample, both comorbidities were significantly associated with annual total charges. Importantly, however, the relationship of eGFR with costs was not significantly affected by adjustment for these conditions. In a sample selected to be free of baseline diabetes and cardiovascular disease, the significant association between eGFR and total charges expanded to the patients with eGFR = 31 to 60 ml/min.
Our study has limitations. As with any observational study, our findings represent associations and do not prove causal links between the studied variables. A cross-sectional design was used based on the nature of the available data. Although prospective longitudinal studies may demonstrate a somewhat different form of the relationship of GFR with healthcare costs, based on the nature of the variables of interest, it is unlikely that reverse causation (i.e., that costs caused changes in eGFR) explain the findings. Billing claims have been demonstrated to provide sensitive measures of diagnoses such as diabetes and cardiovascular disease (18,22), but administrative coding algorithms for identifying polycystic kidney disease have not been validated with external data. Analogous to the approach of prior studies of ADPKD in administrative data (29,30), we combined patients with 753.13 and 753.12 claims in the main analysis based on the similarity of these groups. Lack of information on participant race may have produced some misclassification of renal function. As the aMDRD equation includes a scaling factor of 1.21 for black race, eGFR was underestimated for African American participants by approximately 21%. However, as African Americans account for only 12.5% of the general United States population and are underrepresented among the privately insured in this county (31), potential misclassification of CKD based on race likely affected only a small number of participants. We used charges as a surrogate measure for costs based on the billing information available in the source data. While charges are frequently examined as proxy measures in studies of healthcare economics, charge data may vary based on inclusion of different cost items and modification of charges over time and between centers. Moreover, charges can be approximately 1.6 to 1.8 times greater than costs (32). However, the relative relationship between cost and charges is unlikely to vary across levels of renal function and, therefore, should not invalidate the key findings of this investigation. Findings may differ under other reimbursement systems such as public insurance. We were also unable to study indirect costs to the patient. Despite these limitations, the data source used enabled study of insurance data in nearly 2000 patients with polycystic kidney disease, a sample much larger than could be assembled at a single-center center or even multi-institutional level.
In conclusion, we observed a strong association of healthcare charges with advanced renal dysfunction in privately insured polycystic kidney disease patients, independent of age and comorbid diseases such as diabetes and cardiovascular disease. This evidence is relevant to economic evaluations of new interventions that could prevent or delay progression of polycystic kidney disease. Strategies that prevent loss of renal function below 30 ml/min have the potential to generate substantial reductions in medical charges.
Disclosures
None.
Acknowledgments
Support for data use fees and research staff time was provided by Novartis Pharmaceuticals, Corp. The analyses, interpretation, medical writing, and reporting of these data are the sole responsibility of the authors.
Institution at which work was performed: Saint Louis University, St. Louis, Missouri.
Footnotes
Published online ahead of print. Publication date available at www.cjasn.org.
References
- 1.Grantham J, Cowley BJ, Torres VE: Progression of autosomal dominant polycystic kidney disease (ADPKD) to renal failure. In: The Kidney: Physiology and Pathophysiology, edited by Seldin DW, Geibisch G, Philadelphia, Lippincott Williams and Wilkins, 2000, pp 2513–2536 [Google Scholar]
- 2.Grantham JJ, Torres VE, Chapman AB, Guay-Woodford LM, Bae KT, King BF, Jr., Wetzel LH, Baumgarten DA, Kenney PJ, Harris PC, Klahr S, Bennett WM, Hirschman GN, Meyers CM, Zhang X, Zhu F, Miller JP: Volume progression in polycystic kidney disease. N Engl J Med 354: 2122–2130, 2006 [DOI] [PubMed] [Google Scholar]
- 3.Mitcheson H, Williams G, Castro JE: Clinical aspects of polycystic disease of the kidneys. BMJ 1: 1196–1199, 1977 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bear JC, McManamon P, Morgan J, Payne RH, Lewis H, Gault MH, Churchill DN: Age at clinical onset and at ultrasonographic detection of adult polycystic kidney disease: Data for genetic counselling. Am J Med Genet 18: 45–53, 1984 [DOI] [PubMed] [Google Scholar]
- 5.Churchill DN, Bear JC, Morgan J, Payne RH, McManamon PJ, Gault MH: Prognosis of adult onset polycystic kidney disease re-evaluated. Kidney Int 26: 190–193, 1984 [DOI] [PubMed] [Google Scholar]
- 6.Fick GM, Gabow PA: Natural history of autosomal dominant polycystic kidney disease. Annu Rev Med 45: 23–29, 1994 [DOI] [PubMed] [Google Scholar]
- 7.Incidence and prevalence of ESRD. United States Renal Data System. Am J Kidney Dis 32: S38–S49, 1998 [DOI] [PubMed] [Google Scholar]
- 8.Calvert JP: Strategies to inhibit cyst formation in ADPKD. Clin J Am Soc Nephrol 3: 1205–1211, 2008 [DOI] [PubMed] [Google Scholar]
- 9.Leuenroth SJ, Crews CM: Targeting cyst initiation in ADPKD. J Am Soc Nephrol 20: 1–3, 2009 [DOI] [PubMed] [Google Scholar]
- 10.Go AS, Chertow GM, Fan D, McCulloch CE, Hsu CY: Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N Engl J Med 351: 1296–1305, 2004 [DOI] [PubMed] [Google Scholar]
- 11.Smith DH, Gullion CM, Nichols G, Keith DS, Brown JB: Cost of medical care for chronic kidney disease and comorbidity among enrollees in a large HMO population. J Am Soc Nephrol 15: 1300–1306, 2004 [DOI] [PubMed] [Google Scholar]
- 12.Cheung BM, Ong KL, Cherny SS, Sham PC, Tso AW, Lam KS: Diabetes prevalence and therapeutic target achievement in the United States, 1999 to 2006. Am J Med 122: 443–453, 2009 [DOI] [PubMed] [Google Scholar]
- 13.Levey AS, Greene T, Kusek JW, Beck G: A simplified equation to prediction glomerular filtration rate from serum creatinine. J Am Soc Nephrol 11: 155A, 2000 [Google Scholar]
- 14.Smellie WS, Shaw N, Bowley R, Stewart MF, Kelly AM, Twomey PJ, Chadwick PR, Houghton JB, Ng JP, McCulloch AJ: Best practice in primary care pathology: Review 10. J Clin Pathol 60: 1195–1204, 2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Middleton RJ, Foley RN, Hegarty J, Cheung CM, McElduff P, Gibson JM, Kalra PA, O'Donoghue DJ, New JP: The unrecognized prevalence of chronic kidney disease in diabetes. Nephrol Dial Transplant 21: 88–92, 2006 [DOI] [PubMed] [Google Scholar]
- 16.Philipneri MD, Rocca Rey LA, Schnitzler MA, Abbott KC, Brennan DC, Takemoto SK, Buchanan PM, Burroughs TE, Willoughby LM, Lentine KL: Delivery patterns of recommended chronic kidney disease care in clinical practice: Administrative claims-based analysis and systematic literature review. Clin Exp Nephrol 12: 41–52, 2008 [DOI] [PubMed] [Google Scholar]
- 17.National Kidney Foundation: K/DOQI clinical practice guidelines for chronic kidney disease: Evaluation, classification, and stratification. Am J Kidney Dis 39: S1–S266, 2002 [PubMed] [Google Scholar]
- 18.Hebert PL, Geiss LS, Tierney EF, Engelgau MM, Yawn BP, McBean AM: Identifying persons with diabetes using Medicare claims data. Am J Med Qual 14: 270–277, 1999 [DOI] [PubMed] [Google Scholar]
- 19.Kasiske BL, Snyder JJ, Gilbertson D, Matas AJ: Diabetes mellitus after kidney transplantation in the United States. Am J Transplant 3: 178–185, 2003 [DOI] [PubMed] [Google Scholar]
- 20.Stevens LA, Fares G, Fleming J, Martin D, Murthy K, Qiu J, Stark PC, Uhlig K, Van Lente F, Levey AS: Low rates of testing and diagnostic codes usage in a commercial clinical laboratory: Evidence for lack of physician awareness of chronic kidney disease. J Am Soc Nephrol 16: 2439–2448, 2005 [DOI] [PubMed] [Google Scholar]
- 21.U.S. Renal Data System: USRDS 2008 Annual Data Report, Methods Appendix. Bethesda, National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases, 2008. Available at http://www.usrds.org/2008/pdf/V2_Appendices_2008.pdf Accessed July 6, 2009 [Google Scholar]
- 22.Lentine KL, Schnitzler MA, Abbott KC, Bramesfeld K, Buchanan PM, Brennan DC: Sensitivity of billing claims for cardiovascular disease events among kidney transplant recipients. Clin J Am Soc Nephrol 4: 1213–1221, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bureau of Labor Statistics Consumer Price Index, U.S. Average Medical Care. Available at http://data.bls.gov/PDQ/servlet/SurveyOutputServlet Accessed August 7, 2009
- 24.London R, Solis A, Goldberg GA, Wade S, Ryu S: Health care resource utilization and the impact of anemia management in patients with chronic kidney disease. Am J Kidney Dis 40: 539–548, 2002 [DOI] [PubMed] [Google Scholar]
- 25.St Peter WL, Khan SS, Ebben JP, Pereira BJ, Collins AJ: Chronic kidney disease: The distribution of health care dollars. Kidney Int 66: 313–321, 2004 [DOI] [PubMed] [Google Scholar]
- 26.Robbins JD, Kim JJ, Zdon G, Chan WW, Jones J: Resource use and patient care associated with chronic kidney disease in a managed care setting. J Manag Care Pharm 9: 238–247, 2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Mensah GA, Brown DW: An overview of cardiovascular disease burden in the United States. Health Aff (Millwood) 26: 38–48, 2007 [DOI] [PubMed] [Google Scholar]
- 28.National Diabetes Statistics, 2007. Estimated Diabetes Costs in the United States in 2007. Available at http://diabetes.niddk.nih.gov/DM/PUBS/statistics/#costs Accessed August 7, 2009
- 29.Freedman BI, Soucie JM, Chapman A, Krisher J, McClellan WM: Racial variation in autosomal dominant polycystic kidney disease. Am J Kidney Dis 35: 35–39, 2000 [DOI] [PubMed] [Google Scholar]
- 30.Perrone RD, Kausz TA, Chavers BM, Weinhandl ED, Collins AJ: Incidence of autosomal dominant polycystic kidney disease (ADPKD) among ESRD patients in the United States. J Am Soc Nephrol 16 (Suppl): 358A, 2005 [Google Scholar]
- 31.DeNavas-Walt C, Proctor BD, Smith JC: Income, poverty, and health insurance coverage in the United States: 2007. Current Population Reports, US Census Bureau Issued2008
- 32.Health Care Financing Administration Prospective Payment System: Standardizing File. Washington DC, Department of Health and Human Services, 1998 [Google Scholar]