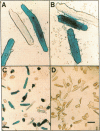
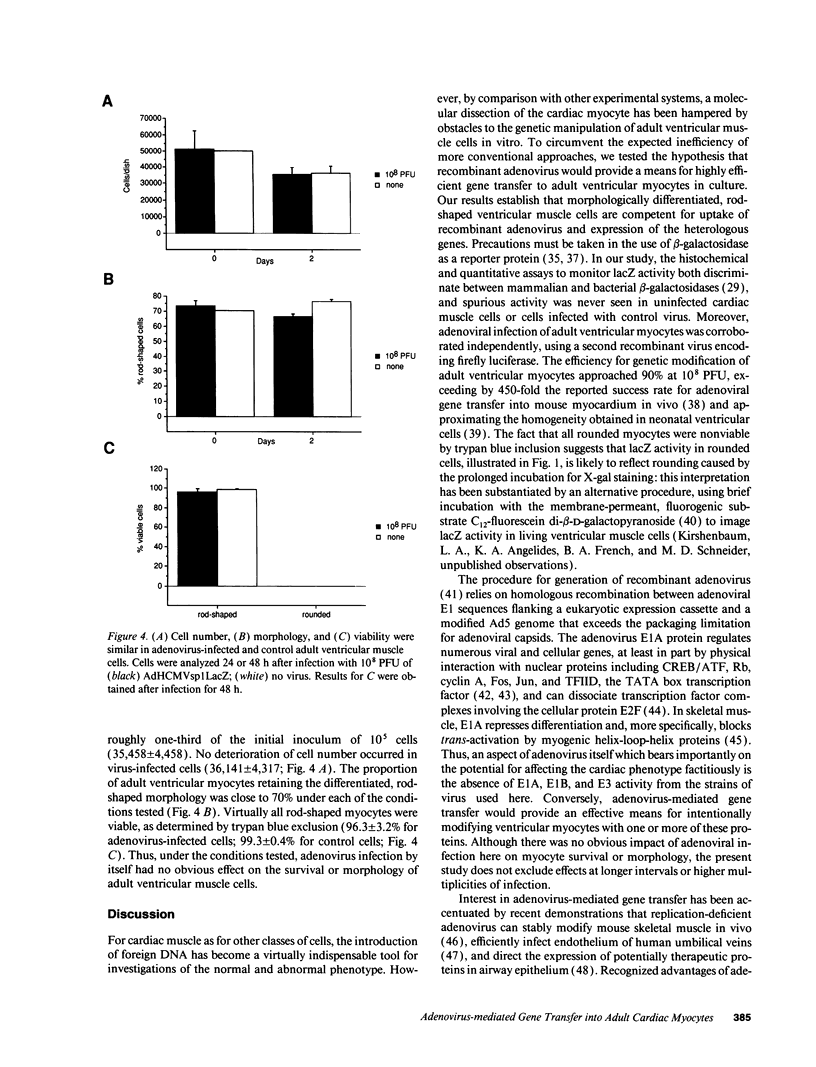
Abstract
Molecular dissection of mechanisms that govern the differentiated cardiac phenotype has, for cogent technical reasons, largely been undertaken to date in neonatal ventricular myocytes. To circumvent expected limitations of other methods, the present study was initiated to determine whether replication-deficient adenovirus would enable efficient gene transfer to adult cardiac cells in culture. Adult rat ventricular myocytes were infected, 24 h after plating, with adenovirus type 5 containing a cytomegalovirus immediate-early promoter-driven lacZ reporter gene and were assayed for the presence of beta-galactosidase 48 h after infection. The frequency of lacZ+ rod-shaped myocytes was half-maximal at 4 x 10(5) plaque-forming units (PFU) and approached 90% at 1 x 10(8) PFU. Uninfected cells and cells infected with lacZ- virus remained colorless. Beta-galactosidase activity concurred with the proportion of lacZ+ cells and was contingent on the exogenous lacZ gene. At 10(8) PFU/dish, cell number, morphology, and viability each were comparable to uninfected cells. Thus, adult ventricular myocytes are amenable to efficient gene transfer with recombinant adenovirus. The relative uniformity for gene transfer by adenovirus should facilitate tests to determine the impact of putative regulators upon the endogenous genes and gene products of virally modified adult ventricular muscle cells.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bihler I., Ho T. K., Sawh P. C. Isolation of Ca2+-tolerant myocytes from adult rat heart. Can J Physiol Pharmacol. 1984 May;62(5):581–588. doi: 10.1139/y84-093. [DOI] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1006/abio.1976.9999. [DOI] [PubMed] [Google Scholar]
- Braun T., Bober E., Arnold H. H. Inhibition of muscle differentiation by the adenovirus E1a protein: repression of the transcriptional activating function of the HLH protein Myf-5. Genes Dev. 1992 May;6(5):888–902. doi: 10.1101/gad.6.5.888. [DOI] [PubMed] [Google Scholar]
- Claycomb W. C., Moses R. L. Growth factors and TPA stimulate DNA synthesis and alter the morphology of cultured terminally differentiated adult rat cardiac muscle cells. Dev Biol. 1988 Jun;127(2):257–265. doi: 10.1016/0012-1606(88)90313-2. [DOI] [PubMed] [Google Scholar]
- Curiel D. T., Agarwal S., Wagner E., Cotten M. Adenovirus enhancement of transferrin-polylysine-mediated gene delivery. Proc Natl Acad Sci U S A. 1991 Oct 1;88(19):8850–8854. doi: 10.1073/pnas.88.19.8850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eppenberger-Eberhardt M., Flamme I., Kurer V., Eppenberger H. M. Reexpression of alpha-smooth muscle actin isoform in cultured adult rat cardiomyocytes. Dev Biol. 1990 Jun;139(2):269–278. doi: 10.1016/0012-1606(90)90296-u. [DOI] [PubMed] [Google Scholar]
- Eustice D. C., Feldman P. A., Colberg-Poley A. M., Buckery R. M., Neubauer R. H. A sensitive method for the detection of beta-galactosidase in transfected mammalian cells. Biotechniques. 1991 Dec;11(6):739-40, 742-3. [PubMed] [Google Scholar]
- Field L. J. Atrial natriuretic factor-SV40 T antigen transgenes produce tumors and cardiac arrhythmias in mice. Science. 1988 Feb 26;239(4843):1029–1033. doi: 10.1126/science.2964082. [DOI] [PubMed] [Google Scholar]
- Graham F. L. Covalently closed circles of human adenovirus DNA are infectious. EMBO J. 1984 Dec 1;3(12):2917–2922. doi: 10.1002/j.1460-2075.1984.tb02232.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horikoshi N., Maguire K., Kralli A., Maldonado E., Reinberg D., Weinmann R. Direct interaction between adenovirus E1A protein and the TATA box binding transcription factor IID. Proc Natl Acad Sci U S A. 1991 Jun 15;88(12):5124–5128. doi: 10.1073/pnas.88.12.5124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jackson T., Allard M. F., Sreenan C. M., Doss L. K., Bishop S. P., Swain J. L. The c-myc proto-oncogene regulates cardiac development in transgenic mice. Mol Cell Biol. 1990 Jul;10(7):3709–3716. doi: 10.1128/mcb.10.7.3709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson J. E., Wold B. J., Hauschka S. D. Muscle creatine kinase sequence elements regulating skeletal and cardiac muscle expression in transgenic mice. Mol Cell Biol. 1989 Aug;9(8):3393–3399. doi: 10.1128/mcb.9.8.3393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones N., Shenk T. Isolation of deletion and substitution mutants of adenovirus type 5. Cell. 1978 Jan;13(1):181–188. doi: 10.1016/0092-8674(78)90148-4. [DOI] [PubMed] [Google Scholar]
- Kariya K., Karns L. R., Simpson P. C. Expression of a constitutively activated mutant of the beta-isozyme of protein kinase C in cardiac myocytes stimulates the promoter of the beta-myosin heavy chain isogene. J Biol Chem. 1991 Jun 5;266(16):10023–10026. [PubMed] [Google Scholar]
- Kirshenbaum L. A., Thomas T. P., Randhawa A. K., Singal P. K. Time-course of cardiac myocyte injury due to oxidative stress. Mol Cell Biochem. 1992 Apr;111(1-2):25–31. doi: 10.1007/BF00229570. [DOI] [PubMed] [Google Scholar]
- Kitsis R. N., Buttrick P. M., McNally E. M., Kaplan M. L., Leinwand L. A. Hormonal modulation of a gene injected into rat heart in vivo. Proc Natl Acad Sci U S A. 1991 May 15;88(10):4138–4142. doi: 10.1073/pnas.88.10.4138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kovacic-Milivojević B., Gardner D. G. Divergent regulation of the human atrial natriuretic peptide gene by c-jun and c-fos. Mol Cell Biol. 1992 Jan;12(1):292–301. doi: 10.1128/mcb.12.1.292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lemarchand P., Jaffe H. A., Danel C., Cid M. C., Kleinman H. K., Stratford-Perricaudet L. D., Perricaudet M., Pavirani A., Lecocq J. P., Crystal R. G. Adenovirus-mediated transfer of a recombinant human alpha 1-antitrypsin cDNA to human endothelial cells. Proc Natl Acad Sci U S A. 1992 Jul 15;89(14):6482–6486. doi: 10.1073/pnas.89.14.6482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lim C. S., Chapman G. D., Gammon R. S., Muhlestein J. B., Bauman R. P., Stack R. S., Swain J. L. Direct in vivo gene transfer into the coronary and peripheral vasculatures of the intact dog. Circulation. 1991 Jun;83(6):2007–2011. doi: 10.1161/01.cir.83.6.2007. [DOI] [PubMed] [Google Scholar]
- Lin H., Parmacek M. S., Morle G., Bolling S., Leiden J. M. Expression of recombinant genes in myocardium in vivo after direct injection of DNA. Circulation. 1990 Dec;82(6):2217–2221. doi: 10.1161/01.cir.82.6.2217. [DOI] [PubMed] [Google Scholar]
- Luetje C. W., Tietje K. M., Christian J. L., Nathanson N. M. Differential tissue expression and developmental regulation of guanine nucleotide binding regulatory proteins and their messenger RNAs in rat heart. J Biol Chem. 1988 Sep 15;263(26):13357–13365. [PubMed] [Google Scholar]
- Lundgren E., Terracio L., Mårdh S., Borg T. K. Extracellular matrix components influence the survival of adult cardiac myocytes in vitro. Exp Cell Res. 1985 Jun;158(2):371–381. doi: 10.1016/0014-4827(85)90462-8. [DOI] [PubMed] [Google Scholar]
- MacGregor G. R., Mogg A. E., Burke J. F., Caskey C. T. Histochemical staining of clonal mammalian cell lines expressing E. coli beta galactosidase indicates heterogeneous expression of the bacterial gene. Somat Cell Mol Genet. 1987 May;13(3):253–265. doi: 10.1007/BF01535207. [DOI] [PubMed] [Google Scholar]
- Mann D. L., Kent R. L., Parsons B., Cooper G., 4th Adrenergic effects on the biology of the adult mammalian cardiocyte. Circulation. 1992 Feb;85(2):790–804. doi: 10.1161/01.cir.85.2.790. [DOI] [PubMed] [Google Scholar]
- McGrory W. J., Bautista D. S., Graham F. L. A simple technique for the rescue of early region I mutations into infectious human adenovirus type 5. Virology. 1988 Apr;163(2):614–617. doi: 10.1016/0042-6822(88)90302-9. [DOI] [PubMed] [Google Scholar]
- Nadal-Ginard B., Mahdavi V. Molecular basis of cardiac performance. Plasticity of the myocardium generated through protein isoform switches. J Clin Invest. 1989 Dec;84(6):1693–1700. doi: 10.1172/JCI114351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parker T. G., Chow K. L., Schwartz R. J., Schneider M. D. Differential regulation of skeletal alpha-actin transcription in cardiac muscle by two fibroblast growth factors. Proc Natl Acad Sci U S A. 1990 Sep;87(18):7066–7070. doi: 10.1073/pnas.87.18.7066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parker T. G., Chow K. L., Schwartz R. J., Schneider M. D. Positive and negative control of the skeletal alpha-actin promoter in cardiac muscle. A proximal serum response element is sufficient for induction by basic fibroblast growth factor (FGF) but not for inhibition by acidic FGF. J Biol Chem. 1992 Feb 15;267(5):3343–3350. [PubMed] [Google Scholar]
- Parker T. G., Packer S. E., Schneider M. D. Peptide growth factors can provoke "fetal" contractile protein gene expression in rat cardiac myocytes. J Clin Invest. 1990 Feb;85(2):507–514. doi: 10.1172/JCI114466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parmacek M. S., Vora A. J., Shen T., Barr E., Jung F., Leiden J. M. Identification and characterization of a cardiac-specific transcriptional regulatory element in the slow/cardiac troponin C gene. Mol Cell Biol. 1992 May;12(5):1967–1976. doi: 10.1128/mcb.12.5.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Piper H. M., Jacobson S. L., Schwartz P. Determinants of cardiomyocyte development in long-term primary culture. J Mol Cell Cardiol. 1988 Sep;20(9):825–835. doi: 10.1016/s0022-2828(88)80007-5. [DOI] [PubMed] [Google Scholar]
- Quantin B., Perricaudet L. D., Tajbakhsh S., Mandel J. L. Adenovirus as an expression vector in muscle cells in vivo. Proc Natl Acad Sci U S A. 1992 Apr 1;89(7):2581–2584. doi: 10.1073/pnas.89.7.2581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raychaudhuri P., Bagchi S., Devoto S. H., Kraus V. B., Moran E., Nevins J. R. Domains of the adenovirus E1A protein required for oncogenic activity are also required for dissociation of E2F transcription factor complexes. Genes Dev. 1991 Jul;5(7):1200–1211. doi: 10.1101/gad.5.7.1200. [DOI] [PubMed] [Google Scholar]
- Rockman H. A., Ross R. S., Harris A. N., Knowlton K. U., Steinhelper M. E., Field L. J., Ross J., Jr, Chien K. R. Segregation of atrial-specific and inducible expression of an atrial natriuretic factor transgene in an in vivo murine model of cardiac hypertrophy. Proc Natl Acad Sci U S A. 1991 Sep 15;88(18):8277–8281. doi: 10.1073/pnas.88.18.8277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosenfeld M. A., Yoshimura K., Trapnell B. C., Yoneyama K., Rosenthal E. R., Dalemans W., Fukayama M., Bargon J., Stier L. E., Stratford-Perricaudet L. In vivo transfer of the human cystic fibrosis transmembrane conductance regulator gene to the airway epithelium. Cell. 1992 Jan 10;68(1):143–155. doi: 10.1016/0092-8674(92)90213-v. [DOI] [PubMed] [Google Scholar]
- Schneider M. D., Parker T. G. Cardiac growth factors. Prog Growth Factor Res. 1991;3(1):1–26. doi: 10.1016/0955-2235(91)90010-2. [DOI] [PubMed] [Google Scholar]
- Sen A., Dunnmon P., Henderson S. A., Gerard R. D., Chien K. R. Terminally differentiated neonatal rat myocardial cells proliferate and maintain specific differentiated functions following expression of SV40 large T antigen. J Biol Chem. 1988 Dec 15;263(35):19132–19136. [PubMed] [Google Scholar]
- Shenk T., Flint J. Transcriptional and transforming activities of the adenovirus E1A proteins. Adv Cancer Res. 1991;57:47–85. doi: 10.1016/s0065-230x(08)60995-1. [DOI] [PubMed] [Google Scholar]
- Shimohama S., Rosenberg M. B., Fagan A. M., Wolff J. A., Short M. P., Breakefield X. O., Friedmann T., Gage F. H. Grafting genetically modified cells into the rat brain: characteristics of E. coli beta-galactosidase as a reporter gene. Brain Res Mol Brain Res. 1989 Jun;5(4):271–278. doi: 10.1016/0169-328x(89)90061-2. [DOI] [PubMed] [Google Scholar]
- Springhorn J. P., Ellingsen O., Berger H. J., Kelly R. A., Smith T. W. Transcriptional regulation in cardiac muscle. Coordinate expression of Id with a neonatal phenotype during development and following a hypertrophic stimulus in adult rat ventricular myocytes in vitro. J Biol Chem. 1992 Jul 15;267(20):14360–14365. [PubMed] [Google Scholar]
- Thompson W. R., Nadal-Ginard B., Mahdavi V. A MyoD1-independent muscle-specific enhancer controls the expression of the beta-myosin heavy chain gene in skeletal and cardiac muscle cells. J Biol Chem. 1991 Nov 25;266(33):22678–22688. [PubMed] [Google Scholar]
- Tsika R. W., Bahl J. J., Leinwand L. A., Morkin E. Thyroid hormone regulates expression of a transfected human alpha-myosin heavy-chain fusion gene in fetal rat heart cells. Proc Natl Acad Sci U S A. 1990 Jan;87(1):379–383. doi: 10.1073/pnas.87.1.379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Volz A., Piper H. M., Siegmund B., Schwartz P. Longevity of adult ventricular rat heart muscle cells in serum-free primary culture. J Mol Cell Cardiol. 1991 Feb;23(2):161–173. doi: 10.1016/0022-2828(91)90103-s. [DOI] [PubMed] [Google Scholar]
- Zhang Y. Z., Naleway J. J., Larison K. D., Huang Z. J., Haugland R. P. Detecting lacZ gene expression in living cells with new lipophilic, fluorogenic beta-galactosidase substrates. FASEB J. 1991 Dec;5(15):3108–3113. doi: 10.1096/fasebj.5.15.1720751. [DOI] [PubMed] [Google Scholar]
- Zimmerman K. A., Yancopoulos G. D., Collum R. G., Smith R. K., Kohl N. E., Denis K. A., Nau M. M., Witte O. N., Toran-Allerand D., Gee C. E. Differential expression of myc family genes during murine development. 1986 Feb 27-Mar 5Nature. 319(6056):780–783. doi: 10.1038/319780a0. [DOI] [PubMed] [Google Scholar]