Abstract
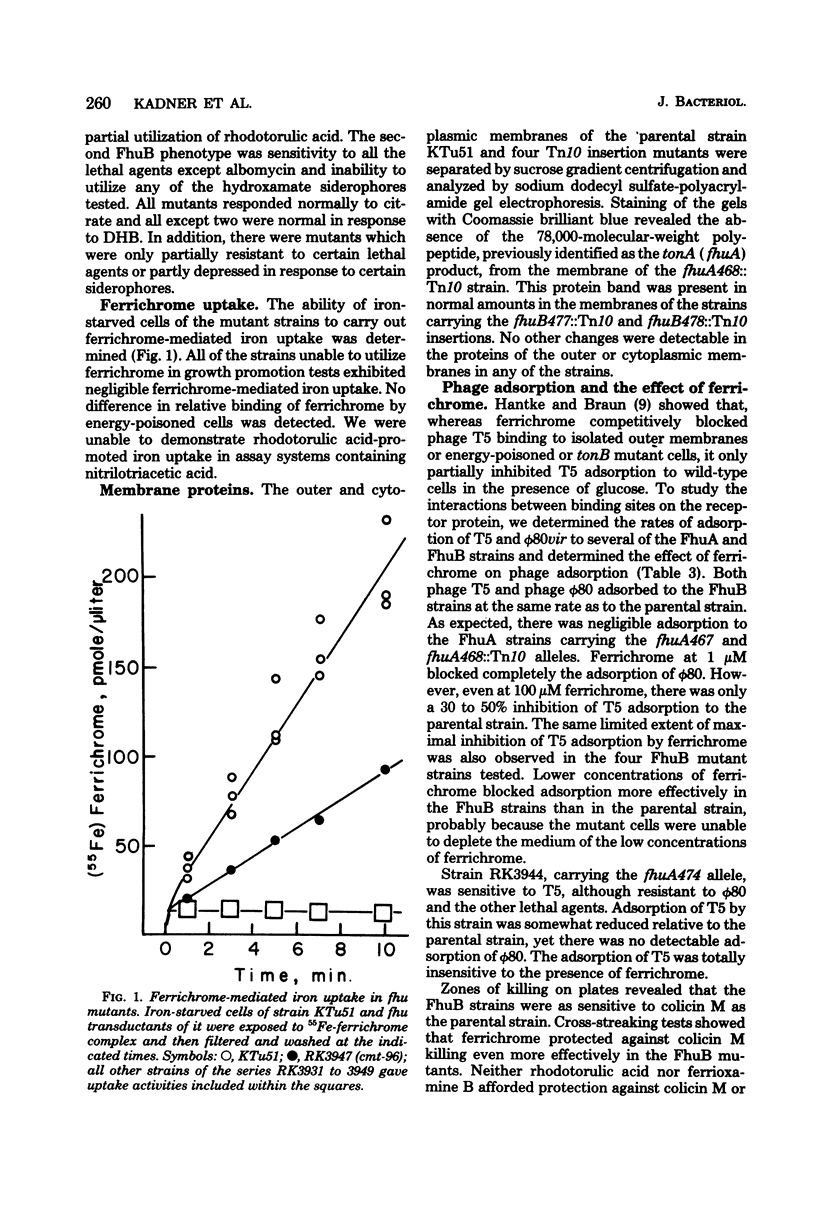
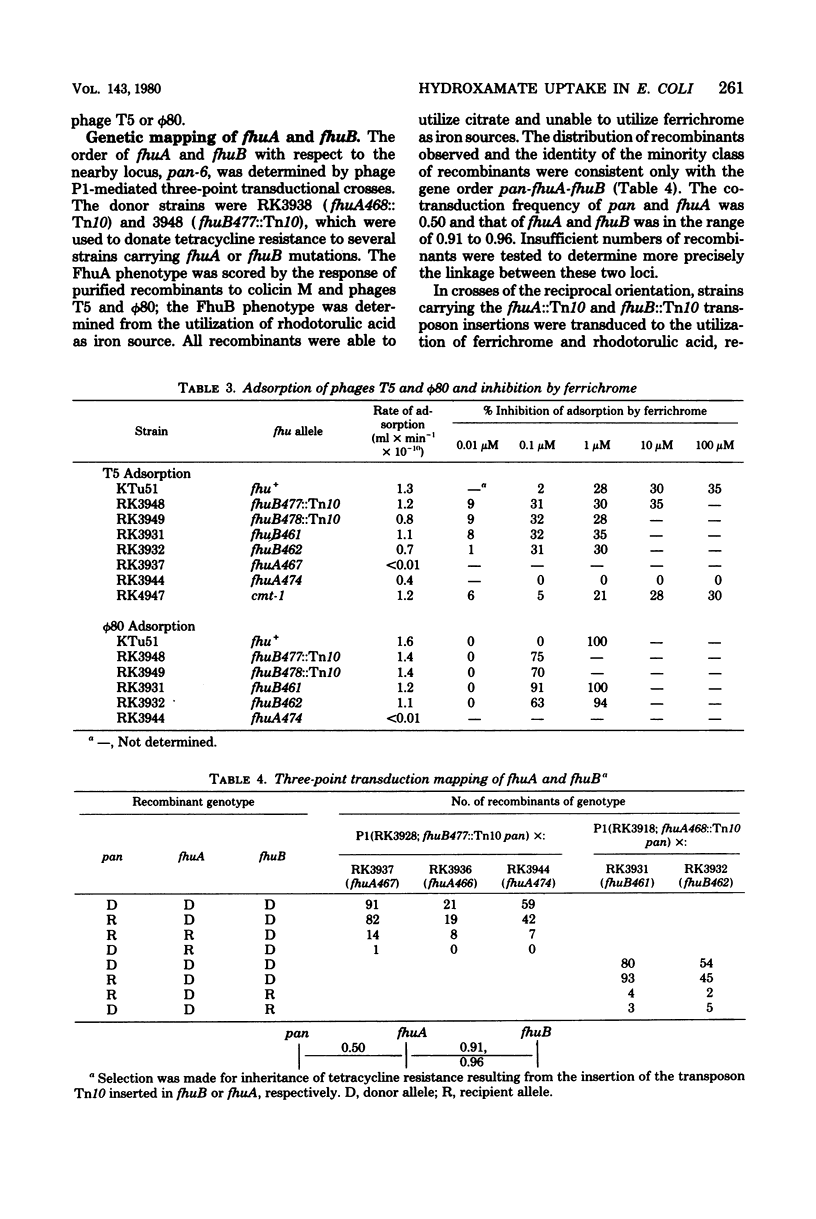
Cells of Escherichia coli can derive iron from a variety of chelators (siderophores) in addition to enterochelin, the catechol derivative excreted by many enteric bacteria. The genetic control of hydroxamate siderophore utilization was investigated in mutants of E. coli K-12 selected for resistance to lethal agents which adsorb to the tonA protein of the outer membrane (albomycin, colicin M, and phages T5 and φ80). Many of the mutants were unable to utilize hydroxamate siderophores as an iron source. This phenotype was termed Fhu, for ferric hydroxamate uptake. Mutants carrying lesions in the tonA region of the chromosome were studied and fell into several types. Members of one class had lost some or all of the tonA receptor protein's functions in that they were resistant to the lethal agents and unable to utilize ferrichrome and its analogs, although able to respond to the hydroxamate rhodotorulic acid (FhuA phenotype). Other mutants were unable to utilize any of the hydroxamate siderophores tested and were resistant to albomycin, althogh many were sensitive to the other lethal agents (FhuB phenotype). Members of these classes lacked ferrichrome-mediated iron uptake. Strains carrying transposon Tn10 insertions in fhuA (previously termed tonA) lacked the 78,000-molecular-weight outer membrane protein previously described; insertions in fhuB retained this protein and had no detectable change in outer membrane composition. Three-point transduction crosses revealed the gene order to be pan-fhuA-fhuB-metD in the min 3.5 region of the chromosome map. Complementation analyses with F′ merodiploid strains showed that fhuA and fhuB comprise separate transcription units which are both required for utilization of ferrichrome. Response of diploid strains to rhodotorulic acid suggested the existence of a third gene, fhuC, required for utilization of this siderophore, but not ferrichrome. It is suggested that fhuB encodes a transport component in the cytoplasmic membrane that is necessary for the uptake of all hydroxamate siderophores following their receptor-mediated passage across the outer membrane.
Full text
PDF






Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Braun V., Hancock R. E., Hantke K., Hartmann A. Functional organization of the outer membrane of escherichia coli: phage and colicin receptors as components of iron uptake systems. J Supramol Struct. 1976;5(1):37–58. doi: 10.1002/jss.400050105. [DOI] [PubMed] [Google Scholar]
- Braun V., Hantke K., Stauder W. Identification of the sid outer membrane receptor protein in Salmonella typhimurium SL1027. Mol Gen Genet. 1977 Oct 20;155(2):227–229. doi: 10.1007/BF00393164. [DOI] [PubMed] [Google Scholar]
- Braun V., Schaller K., Wolff H. A common receptor protein for phage T5 and colicin M in the outer membrane of Escherichia coli B. Biochim Biophys Acta. 1973 Sep 27;323(1):87–97. doi: 10.1016/0005-2736(73)90433-1. [DOI] [PubMed] [Google Scholar]
- Coulton J. W., Naegeli H. U., Braun V. Iron supply of Escherichia coli with polymer-bound ferricrocin. Eur J Biochem. 1979 Aug 15;99(1):39–47. doi: 10.1111/j.1432-1033.1979.tb13228.x. [DOI] [PubMed] [Google Scholar]
- Cox G. B., Gibson F., Luke R. K., Newton N. A., O'Brien I. G., Rosenberg H. Mutations affecting iron transport in Escherichia coli. J Bacteriol. 1970 Oct;104(1):219–226. doi: 10.1128/jb.104.1.219-226.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DAVIS B. D., MINGIOLI E. S. Mutants of Escherichia coli requiring methionine or vitamin B12. J Bacteriol. 1950 Jul;60(1):17–28. doi: 10.1128/jb.60.1.17-28.1950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davies J. K., Reeves P. Genetics of resistance to colicins in Escherichia coli K-12: cross-resistance among colicins of group B. J Bacteriol. 1975 Jul;123(1):96–101. doi: 10.1128/jb.123.1.96-101.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frost G. E., Rosenberg H. Relationship between the tonB locus and iron transport in Escherichia coli. J Bacteriol. 1975 Nov;124(2):704–712. doi: 10.1128/jb.124.2.704-712.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hantke K., Braun V. Functional interaction of the tonA/tonB receptor system in Escherichia coli. J Bacteriol. 1978 Jul;135(1):190–197. doi: 10.1128/jb.135.1.190-197.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heller K., Braun V. Accelerated adsorption of bacteriophage T5 to Escherichia coli F, resulting from reversible tail fiber-lipopolysaccharide binding. J Bacteriol. 1979 Jul;139(1):32–38. doi: 10.1128/jb.139.1.32-38.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kadner R. J. Transport and utilization of D-methionine and other methionine sources in Escherichia coli. J Bacteriol. 1977 Jan;129(1):207–216. doi: 10.1128/jb.129.1.207-216.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luckey M., Neilands J. B. Iron transport in Salmonella typhimurium LT-2: prevention, by ferrichrome, of adsorption of bacteriophages ES18 and ES18.h1 to a common cell envelope receptor. J Bacteriol. 1976 Aug;127(2):1036–1037. doi: 10.1128/jb.127.2.1036-1037.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luckey M., Pollack J. R., Wayne R., Ames B. N., Neilands J. B. Iron uptake in Salmonella typhimurium: utilization of exogenous siderochromes as iron carriers. J Bacteriol. 1972 Sep;111(3):731–738. doi: 10.1128/jb.111.3.731-738.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pugsley A. P., Reeves P. Characterization of group B colicin-resistant mutants of Escherichia coli K-12: colicin resistance and the role of enterochelin. J Bacteriol. 1976 Jul;127(1):218–228. doi: 10.1128/jb.127.1.218-228.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pugsley A. P., Reeves P. Iron uptake in colicin B-resistant mutants of Escherichia coli K-12. J Bacteriol. 1976 Jun;126(3):1052–1062. doi: 10.1128/jb.126.3.1052-1062.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wayne R., Neilands J. B. Evidence for common binding sites for ferrichrome compounds and bacteriophage phi 80 in the cell envelope of Escherichia coli. J Bacteriol. 1975 Feb;121(2):497–503. doi: 10.1128/jb.121.2.497-503.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woodrow G. C., Langman L., Young I. G., Gibson F. Mutations affecting the citrate-dependent iron uptake system in Escherichia coli. J Bacteriol. 1978 Mar;133(3):1524–1526. doi: 10.1128/jb.133.3.1524-1526.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wookey P., Rosenberg H. Involvement of inner and outer membrane components in the transport of iron and in colicin B action in Escherichia coli. J Bacteriol. 1978 Feb;133(2):661–666. doi: 10.1128/jb.133.2.661-666.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]


