Abstract
Diabetes mellitus increases the risk of developing cardiovascular diseases such as coronary artery disease and heart failure. Studies have shown that the heart failure risk is increased in diabetic patients even after adjusting for coronary artery disease and hypertension. Although the cause of this increased heart failure risk is multifactorial, increasing evidence suggests that derangements in cardiac energy metabolism play an important role. In particular, abnormalities in cardiomyocyte mitochondrial energetics appear to contribute substantially to the development of cardiac dysfunction in diabetes. This review will summarize these abnormalities in mitochondrial function and discuss potential underlying mechanisms.
Keywords: Mitochondria, Diabetes, Heart, Metabolism
1. Introduction
It is increasingly recognized that mechanisms independent of coronary artery disease and hypertension contribute to an increased risk for the development of heart failure in diabetic subjects. Already 40 years ago, Rubler et al.1 described four diabetic patients suffering from heart failure, with normal coronary arteries and no other obvious aetiologies for heart failure. This finding has been confirmed in several subsequent studies.2,3 Other studies have shown that the increased risk for developing heart failure persists in diabetic patients after adjusting for age, blood pressure, weight, cholesterol, and coronary artery disease.4,5 These observations led to the introduction of the term ‘diabetic cardiomyopathy’, which describes ventricular dysfunction occurring in diabetic patients that might not be attributable to underlying coronary artery disease and hypertension. It has also been used to describe changes in the heart that amplifies left ventricular dysfunction that occurs in diabetic subjects following pressure overload or ischaemia, relative to similarly affected non-diabetics.6,7 The term now includes diabetic individuals with diastolic dysfunction, the prevalence of which may be as high as 60% in well-controlled Type 2 diabetic patients.6
Although the pathophysiological mechanisms leading to the increased risk for the development of heart failure are certainly multifactorial, emerging evidence suggests that derangements in cardiac energy metabolism play a fundamental role in the pathogenesis of diabetic cardiomyopathy. Recent studies focusing on mitochondrial function point towards an important role of abnormalities in cardiac mitochondria. The current review will describe these mitochondrial abnormalities and discuss potential underlying mechanisms that lead to cardiac mitochondrial dysfunction.
2. Myocardial mitochondrial dysfunction in humans with diabetes
Although only few studies have attempted to examine myocardial mitochondrial function in human diabetes, several studies conducted in recent years suggest that mitochondrial dysfunction likely occurs in the hearts of humans with diabetes or insulin resistance. Peterson et al.8 reported that in young females with obesity and insulin resistance, increased body mass index and impaired glucose tolerance are associated with increased myocardial oxygen consumption, reduced cardiac efficiency (=ratio of cardiac work to O2 consumption), and increased fatty acid utilization. Since both O2 consumption and fatty acid oxidation occur in mitochondria, this study pointed towards alterations in mitochondrial energy metabolism. Support for impairment in cardiac mitochondrial bioenergetics in Type 2 diabetes has come from studies using 31P nuclear magnetic resonance (NMR) spectroscopy, demonstrating that Type 2 diabetic patients had reduced cardiac phosphocreatine (pCr)/ATP ratios, suggesting that mitochondrial high-energy phosphate metabolism may be impaired, although changes in the total creatine pool and increased ATP utilization, as opposed to ATP generation, may also contribute.9,10 These findings are however similar to observations in failing hearts, which are also characterized by decreased pCr/ATP ratios and impairment in mitochondrial respiratory capacity.11–13 In contrast, studies of well-controlled Type 2 diabetics did not reveal significant changes in pCr/ATP ratios.14,15 These studies do not rule out the absence of mitochondrial dysfunction as they could be confounded by changes in the creatine pool or changes in the pCr shuttle. Thus, the possibility exists that 31P nuclear NMR might lack the sensitivity to detect subtle changes in mitochondrial energetics. The most compelling evidence to date comes from a recent study by Anderson et al.16 who directly demonstrated impaired mitochondrial respiratory capacity with palmitoyl-carnitine and glutamate in atrial tissue of Type 2 diabetic patients using the permeabilized cardiac myofibres technique. Mitochondrial dysfunction was present in all diabetics even in those with HbA1c as low as 6.0 and an inverse relationship between HbA1c and mitochondrial respiratory capacity was observed. In addition, they also demonstrated increased mitochondrial H2O2 emission, depletion of glutathione, and increased levels of hydroxynonenal-modified proteins and 3-nitrotyrosine-modified proteins. This study not only provides good evidence for the presence of mitochondrial dysfunction in human diabetic hearts, but also recapitulates data generated in animal models of diabetes mellitus.
3. Mitochondrial dysfunction in animal models of diabetes
Owing to the intrinsic limitations in generating appropriate human samples and performing mechanistic studies in human tissue, most studies addressing the pathogenesis of diabetic cardiomyopathy have been performed in rodent models of Type 1 and Type 2 diabetes (Table 1).17 Early studies in the 1980s by Kuo et al.18,19 demonstrated impaired state 3 respiration of mitochondria isolated from Type 2 diabetic db/db mouse hearts. More recent studies also demonstrated impairment in myocardial mitochondrial state 3 respiration and/or mitochondrial ATP synthesis rates in Type 2 diabetic ob/ob mice, as well as in insulin-resistant pre-diabetic uncoupling protein diphtheria toxin A chain (UCP-DTA) mice.20,21 In addition, reduced expression of respiratory chain complexes was observed in ob/ob and db/db hearts. In some but not all studies, increased myocardial expression of mitochondrial UCPs 2 and 3 was reported for db/db mice or Zucker diabetic fatty (ZDF) rats, and mitochondrial ultrastructural defects and mitochondrial proliferation were demonstrated in ob/ob, db/db, and UCP-DTA mouse hearts.20–22 Similar observations have also been made in hearts of Type 1 diabetic animal models. Impairment in mitochondrial respiratory capacity, pronounced alterations in mitochondrial structure, and altered expression of respiratory chain complexes have been observed in hearts of streptozotocin-induced diabetic rodents, Ins2+/− Akita mice, and OVE26 mice.23–27 Taken together, these studies clearly indicate the presence of myocardial mitochondrial abnormalities in animal models of Type 1 and Type 2 diabetes mellitus. In the next sections, potential molecular mechanisms underlying these abnormalities will be discussed.
Table 1.
Cardiac traits of selected animal models of diabetes mellitus and transgenic animal models mimicking metabolic alterations of diabetic hearts
| Cardiac function | Cardiac efficiency | Mitochondrial function | Mitochondrial content | Fatty acid oxidation | Glucose oxidation | Oxidative stress | Calcium handling | |
|---|---|---|---|---|---|---|---|---|
| Type 1 diabetes | ||||||||
| Streptozotocin | ↓121,122 | ↓37 | ↓25,26 | ↑37 | ↓37 | ↑123,124 | ↓125,126 | |
| OVE26 | ↓67,68 | ↓27,68 | ↑27,68 | ↑67 | ↓67 | |||
| Ins2+/− Akita | ↓23 | =23 | ↓23,24 | ↑23,24 | ↑23 | ↓23 | =23 | |
| Type 2 diabetes | ||||||||
| db/db | ↓22,95 | ↓22 | ↓22 | ↑22 | ↑22,28 | ↓22,28 | ↑22 | ↓97 |
| ob/ob | ↓/↑28,29 | ↓29 | ↓20 | ↑103 | ↑28,29 | ↓28,29 | 65 | ↓65 |
| UCP-DTA | ↓21 | ↑21 | ||||||
| ZDF | ↓127 | ↑30 | ↓127 | |||||
| Goto-Kakizaki | =/↓128,129 | ↑69 | ↓130 | |||||
| Transgenic | ||||||||
| MHC-PPARα | ↓33 | ↑33 | ↓33 | ↑131 | ↓33 | |||
| CIRKO | ↓52,109 | =52 | ↓52 | ↑52 | ↓109 | ↓109 | ↑52 | |
4. Molecular mechanisms of myocardial mitochondrial dysfunction in diabetes
4.1. Fatty acid oxidation, cardiac efficiency, and mitochondrial uncoupling
The heart depends on continuous oxidation of energy metabolic substrates for sufficient generation of ATP to maintain contractile function. Under physiological conditions, the normal heart generates ATP mainly from the mitochondrial oxidation of fatty acids (60–70% of ATP generated) and to a lesser extent from glucose, lactate, and other substrates (30–40%). In contrast, hearts of diabetic animals use relatively more fatty acids to generate ATP, while glucose oxidation rates are decreased.28–30 The increase in myocardial fatty acid oxidative capacity is mediated in part by increased activity of the nuclear hormone receptor peroxisome proliferator-activated receptor α (PPARα). PPARα increases the expression of genes involved in virtually every step of cardiac fatty acid utilization.31,32 Cardiac-specific overexpression of PPARα (MHC-PPARα mice) resulted in increased expression of fatty acid utilization genes and increased fatty acid oxidation.33 Conversely, cardiac expression of genes encoding for proteins of glucose utilization was decreased in parallel with reduced glucose utilization. Thus, mice with cardiac overexpression of PPARα mimicked the metabolic phenotype of the diabetic heart, thereby implicating PPARα in the regulation of cardiac metabolism in the diabetic heart. Indeed, cardiac PPARα expression is increased in ZDF rats, and PPARα target gene expression is increased in 10-week-old db/db mice.28,34 However, increased fatty acid oxidation in 4-week-old ob/ob and db/db mice in the absence of increased PPARα and PPARα target gene expression suggests the presence of additional mechanisms in early Type 2 diabetes.28
Early work by Mjos35,36 in the 1970s demonstrated that increasing cardiac fatty acid uptake by lipid infusion results in higher oxygen extraction but unchanged myocardial contractility in healthy dogs, thus resulting in reduced cardiac efficiency. This phenomenon can similarly be observed in Type 2 diabetes. Myocardial O2 consumption and fatty acid oxidation are increased, and cardiac efficiency is reduced in ob/ob and db/db mice, as well as in obese humans.8,20,22,28,29,37 Thus, reduced cardiac efficiency is a hallmark in obesity and Type 2 diabetes both in rodents and humans. The ZDF rat does not show increased myocardial O2 consumption or reduced cardiac efficiency, but the mechanisms for these differences may be species-related or unique to the ZDF model.30,38
Recent findings suggest increased mitochondrial uncoupling as one underlying mechanism for increased myocardial O2 consumption and reduced cardiac efficiency in Type 2 diabetic hearts. Before presenting the evidence for this, it is important to note that the role of UCPs in the heart is controversial and incompletely understood. Activation of UCPs in the context of increased mitochondrial substrate flux serves a physiological role to decrease the proton gradient and reduce ROS.39,40 We believe, though, that in certain models of diabetes, particularly those that are associated with obesity and insulin resistance, the increase in ROS production is not simply a consequence of an increase in substrate flux, but is also exacerbated by diabetes-induced defects in the electron transport chain (ETC) that promote ROS overproduction. The persistent increase in ROS chronically activates UCPs that might not necessarily reduce flux via the ETC to normalize ROS production (because of ETC defects). However, dissipation of the proton gradient may still reduce ATP generation. Thus, in ob/ob mice, perfused with glucose and palmitate, an increase in oxygen consumption, without a concomitant increase in contractile performance, was observed relative to hearts that were perfused with glucose only.20 This fatty acid-induced increase in myocardial O2 consumption occurred, despite reduced expression of complexes I, III, and V of the respiratory chain of oxidative phosphorylation (OXPHOS). Since ATP/O ratios were decreased and mitochondrial state 4 respiration rates were increased, these data suggest that increased oxygen consumption in ob/ob hearts after addition of fatty acids to the perfusion medium may be the consequence of fatty acid-mediated mitochondrial uncoupling. Mitochondrial uncoupling may increase oxygen consumption without proportionately increasing mitochondrial ATP production. The resulting energy deficit may explain the lack of increase in cardiac contractile function, resulting in reduced cardiac efficiency. Similar results were obtained in a study performed in db/db mice,22 and Duncan et al.21 also reported reduced ATP/O ratios in cardiac mitochondria of insulin-resistant pre-diabetic UCP-DTA mice. Unfortunately, fatty acid oxidation, myocardial O2 consumption, and contractile function in intact hearts were not investigated in UCP-DTA mice.
Regarding the mechanism leading to mitochondrial uncoupling in diabetic mice, a current concept is the activation of mitochondrial UCPs. In the brown adipose tissue, mitochondrial uncoupling is mediated by uncoupling protein 1 (UCP1) and serves non-shivering thermogenesis.41 UCP1 is an H+ translocase, which channels protons from the intermembrane space back into the matrix space, thereby bypassing the F0F1-ATPase, reducing ATP production with the generation of heat (i.e. uncoupling of oxygen consumption from ATP production). About 20 years after the discovery of UCP1, four more UCP homologues have been identified (UCP2, UCP3, UCP4, and UCP5/BCMP1), two of which (UCP2 and UCP3) are expressed in myocardial tissue.42–44 Several studies have demonstrated uncoupling activity for UCP3 in muscle tissue.45,46 The group of Brand47,48 also demonstrated that both fatty acids and superoxide can activate UCP3 protein activity in skeletal muscle. Superoxides can activate UCP3 activity either by direct interaction with the protein or indirectly by generating lipid peroxidation products, which then activate UCP3.
In a recent study, Boudina et al.22 directly demonstrated that mitochondrial uncoupling occurs in hearts of diabetic db/db mice and that this uncoupling is mainly mediated by increased UCP activity. Mitochondria from Type 2 diabetic db/db mouse hearts pre-perfused with glucose and palmitate exhibited increased respiration in the presence of oligomycin, reduced ATP synthesis, and reduced ATP/O ratios, compared with similarly perfused wild-type mice. In addition, pre-perfusion of db/db hearts with glucose and palmitate increased mitochondrial ADP-stimulated oxygen consumption compared with hearts that were perfused with glucose only. These data suggest the presence of fatty acid-induced mitochondrial uncoupling in db/db mouse hearts. Finally, Boudina et al. demonstrated that mitochondrial proton leak was increased in db/db mitochondria and that the addition of GDP, an inhibitor of UCPs, almost completely restored proton leak to wild-type levels. The remaining proton leak could be attributed to activation of the adenine nucleotide translocase (ANT), another protein capable of uncoupling mitochondrial respiration.49 Although UCP3 protein levels were not increased in db/db mice in this study, this does not preclude the fact that increased cardiac UCP expression, as observed under conditions of increased fatty acid delivery and utilization, may also additionally increase mitochondrial uncoupling activity.
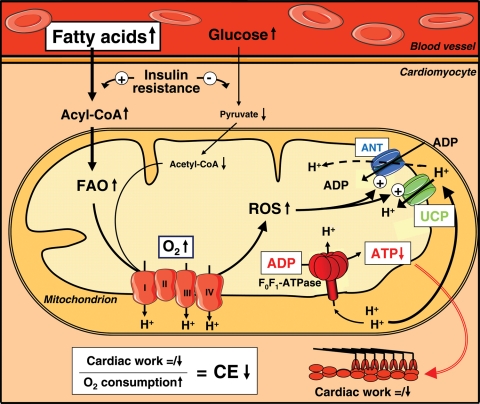
Importantly, db/db hearts exhibited increased mitochondrial H2O2 generation, and overproduction of mitochondrial ROS occurring in conjunction with augmented electron delivery from increased fatty acid oxidation.50 On the basis of the capability of superoxide to activate UCP activity, we propose a model in which ROS-induced mitochondrial uncoupling may reduce cardiac efficiency in Type 2 diabetic hearts (illustrated in Figure 1). The increased cardiac fatty acid uptake and β-oxidation that characterizes many models of Type 2 diabetes increase the delivery of reducing equivalents to the ETC. However, defects in the ETC increase the likelihood that excessive ROS generation leading to increased lipid peroxidation will occur as a result of increased fatty acid utilization, which in turn increases UCP and ANT activity. Increased mitochondrial uncoupling will increase cardiac oxygen consumption and may thereby increase fatty acid oxidation rates further. However, the increase in oxygen consumption would not be accompanied by an appropriate increase in ATP synthesis. Therefore, cardiac work would not increase and cardiac efficiency would be reduced. Ultimately, mitochondrial uncoupling may result in reduced myocardial high-energy reserves and thereby contribute to the development of cardiac contractile dysfunction in Type 2 diabetes.
Figure 1.
Fatty acid-induced mitochondrial uncoupling and impaired cardiac efficiency. In Type 2 diabetes, increased delivery of fatty acids to the heart may result in increased fatty acid uptake and oxidation. The resulting increase in reducing equivalent delivery to the respiratory chain may increase ROS production, leading to activation of UCPs and proton leak via ANT. The resulting increase in mitochondrial uncoupling leads to increased mitochondrial O2 consumption, thereby increasing fatty acid oxidation even further. Since the increase in O2 consumption results from mitochondrial uncoupling, ATP synthesis would not increase proportionately, resulting in reduced cardiac efficiency, subsequent cardiac energy deficit, and ultimately contractile dysfunction. FAO, fatty acid oxidation; GO, glucose oxidation; ROS, reactive oxygen species, UCP, uncoupling protein; ANT, adenine nucleotide translocator.
In contrast to Type 2 diabetic models, impaired cardiac efficiency and mitochondrial uncoupling were not observed in some models of Type 1 diabetes. Thus, in Ins2+/− Akita mice, cardiac efficiency was not reduced, despite increased cardiac fatty acid utilization.23 In addition, pre-perfusion of Akita hearts with fatty acids did not increase oxygen consumption rates in the presence of oligomycin, did not decrease ATP/O ratios, and did not increase mitochondrial proton leak, despite increased expression of UCP3. Similarly, Herlein et al.51 reported decreased mitochondrial state 4 respiration and unchanged ADP/O ratios, associated with increased UCP3 expression in rats rendered diabetic by streptozotocin injections for 2 months. Thus, it is tempting to speculate that, despite increased fatty acid oxidation and UCP3 expression, Type 1 diabetic hearts may not develop fatty acid-induced mitochondrial uncoupling leading to impaired cardiac efficiency. Of note, How et al.37 reported that cardiac efficiency was reduced in streptozotocin-diabetic mice, but mitochondrial uncoupling was also not observed. More studies are needed to evaluate whether fatty acid-induced uncoupling is indeed unique to models of Type 2 diabetes, and if so, what differences may underlie a predisposition of Type 2 diabetic models to develop fatty acid-induced mitochondrial uncoupling. Studies in mice with cardiomyocyte-restricted deletion of insulin receptors, which develop fatty acid-induced mitochondrial uncoupling in the absence of hyperglycaemia, suggest that a potential mechanism for the difference between models of Type 1 and Type 2 diabetes could be insulin resistance.52
4.2. Oxidative stress
The mitochondrial respiratory chain is the principal source of cellular oxygen radicals (ROS), such as superoxide and hydroxyl anions. The primary factor governing mitochondrial ROS generation is the redox state of the respiratory chain.53 If the membrane potential across the inner mitochondrial membrane rises above a certain threshold value, a large stimulation of ROS generation occurs.54 Electrons mainly leak from complexes I and III of the ETC and thereby generate incompletely reduced forms of oxygen.39,55 A rise in membrane potential can occur as a consequence of augmented delivery of electrons to the respiratory chain, which can result either from increased glucose or fatty acid oxidation, or also occur as a result of changes in ETC stoichiometry that could, for example, lead to an increase in reverse electron flow.39,54,56 There is also evidence that increased cytosolic generation of ROS might precipitate increased mitochondrial ROS overproduction, which underscores the pathogenic importance of ROS generation from mitochondrial and non-mitochondrial sources.57,58 ROS can induce cellular damage by direct oxidation of proteins, by conversion of lipids to reactive lipid peroxidation products, by increasing protein tyrosine nitration by generation of reactive nitrogen species, and by interaction with DNA. Mitochondrial DNA has been proposed to be particularly susceptible to oxidative damage.59 Increased levels of ROS not only result from overproduction of ROS, but may also be the consequence of decreased efficiency of inhibitory scavenger systems, such as superoxide dismutases (Cu/Zn-SOD and Mn-SOD), catalase, and the glutathione peroxidase system.
There is convincing experimental and clinical evidence that the production of ROS is increased in multiple tissues in models of Type 1 and Type 2 diabetes. Numerous studies demonstrated an association between increased free radical production and the progression of diabetic complications in various tissues including the heart.60–62 Several pathways have been identified by which increased serum glucose levels can increase ROS production, including glucose auto-oxidation, formation of advanced glycation end products, increased polyol pathway flux, increased hexosamine pathway flux, and activation of protein kinase C.63 Brownlee's64 group provided strong evidence that ROS from mitochondria activates many of these pathological pathways that induce diabetic complications. Although most of the ROS may be predominantly derived from mitochondria, evidence for increased activity of cytosolic ROS generation via NADPH oxidase also exists.65 Since ROS have a very short half-life, they are believed to cause damage close to their origin. Thus, mitochondria would not only be the origin but also the target of oxidative stress.
Most studies investigating the contribution of oxidative stress to diabetic cardiomyopathy have been performed in Type 1 diabetic models. Lashin et al.26 showed that 4-hydroxy-2-nonenal is produced within cardiac mitochondria and forms an adduct with complex II of the respiratory chain, and that this modification was associated with decreased succinate-supported respiration and complex II enzymatic activity in streptozotocin-induced Type 1 diabetic rats. A proteomic approach by Turko et al.66 revealed tyrosine nitration of several cardiac mitochondrial proteins in alloxan-induced Type 1 diabetic rats, including proteins involved in energy metabolism (succinyl-CoA:3-oxoacid CoA transferase, creatine kinase). Ye et al.67 demonstrated that catalase overexpression restores impaired mitochondrial morphology and cardiomyocyte contractility of Type 1 diabetic OVE26 mice. It was further shown that incubation of cardiomyocytes from OVE26 mice in a high-glucose medium results in increased ROS production and that this increase could be prevented either by catalase overexpression or by inhibition of electron transport at complex I or II, indicating a mitochondrial source for ROS production in OVE26 mouse hearts. Another recent report from this group demonstrated that reductions in OVE26 cardiomyocyte contractility could be completely reversed and that impaired mitochondrial state 3 respiration could, at least in part, be restored, by the overexpression of the mitochondrial SOD isoform (Mn-SOD).68 The accompanying changes in mitochondrial morphology (swelling, mottled matrix, broken mitochondrial membrane) and the increase in mitochondrial biogenesis could be reversed as well. Taken together, these studies suggest that in Type 1 diabetic hearts, ROS may be produced within mitochondria and that these ROS may have varied effects on mitochondrial function. Many of these studies have examined downstream consequences of ROS overproduction on mitochondrial function and protein modifications. However, they do not exclude the possibility that an increase in mitochondrial ROS production in vivo in these models of Type 1 diabetes could result from an initial increase in cytosol ROS generation. Indeed, results from recent reports in which mitochondrial ROS levels were directly measured suggest that there is no intrinsic increase in mitochondrial ROS generation in Type 1 diabetes. Thus, studies in the Type 1 diabetes Akita mouse model and streptozotocin-diabetic rats revealed no evidence for increased mitochondrial ROS generation when directly measured using a variety of approaches.23,51 Mitochondrial respiratory dysfunction and severe mitochondrial morphological abnormalities were nonetheless present in those hearts. Although some of these differences may be related to unique characteristics of the animal models used, they also suggest that additional (ROS-independent) mechanisms might impair mitochondrial function in these models.17
Reports of increased mitochondrial ROS production in models of Type 2 diabetes are limited. Increased lipid peroxidation and reduced mitochondrial levels of glutathione and coenzyme Q have been reported in hearts of Goto-Kakizaki rats.69 Ye et al.67 showed that overexpression of mitochondrial catalase in insulin-resistant and obese Ay mice restores impaired cardiomyocyte contractility. Conti et al.70 reported induction of the mitochondrial antioxidant defence system in obese Zucker rats. Most compelling evidence to date comes from Boudina et al.22 who recently reported that db/db mice show increased mitochondrial H2O2 production, increased levels of malondialdehyde and 4-hydroxy-2-nonenal protein adducts, and increased mitochondrial Mn-SOD expression in the heart. Thus, it is likely that increased mitochondrial ROS production and subsequent oxidative stress occur in hearts of Type 2 diabetic animal models. Interestingly, similar changes were observed in mitochondria obtained from mice with cardiomyocyte-restricted deletion of insulin receptors, suggesting a potential role for insulin resistance in this process.52
Taken together, these studies suggest that oxidative stress may contribute to myocardial mitochondrial dysfunction in diabetes. However, the underlying mechanisms of ROS-induced mitochondrial dysfunction may only partially overlap between models of Type 1 and Type 2 diabetes. ROS-mediated damage leading to mitochondrial dysfunction may ultimately result in reduced ATP production and thereby impair contractility. In Type 2 diabetic hearts, mitochondrial ROS may play an even more central role in impairing mitochondrial energy metabolism due to additional existence of fatty acid-induced mitochondrial uncoupling (Figure 1).
4.3. Mitochondrial calcium handling
Cardiomyocyte contraction relies upon intricate interactions between Ca2+ and ATP, both of which must be present in adequate amounts. Excitation of the cardiomyocyte results in increased Ca2+ influx via L-type Ca2+ channels, which in turn triggers the release of significant amounts of Ca2+ from the sarcoplasmic reticulum, resulting in binding of Ca2+ to troponin C on actin filaments and subsequent conformational changes in contractile-regulatory proteins which allow interaction between actin and myosin cross-bridges. ATP is required for the dissociation of the actomyosin interaction to allow the start of a new contractile cycle and for Ca2+-re-uptake into the sarcoplasmic reticulum by the sarcoendoplasmic reticulum Ca2+-ATPase 2a (SERCA2a).
In addition to its role in E–C coupling, Ca2+ also regulates energy metabolism, i.e. ATP production. After a cytosolic increase in Ca2+ concentration, intramitochondrial Ca2+ concentration rises as well. There is growing evidence that the mitochondrial Ca2+ concentration can track cytosolic Ca2+ concentrations during the cardiac cycle, although the rapid influx and efflux mechanisms participating in Ca2+ exchange between the cytosol and mitochondria are still unclear.71–73 Ca2+ exchange between these subcellular compartments is believed to provide a mechanism for matching energy production to energy demand under physiological conditions or increased workload and is termed the ‘parallel activation model’.71 Ca2+ can up-regulate the activity of several Ca2+-sensitive metabolic enzymes, including the mitochondrial dehydrogenases pyruvate dehydrogenase, isocitrate dehydrogenase, and α-ketoglutarate dehydrogenase.74,75 Activation of intramitochondrial Ca2+-sensitive dehydrogenases by Ca2+ has been directly documented in isolated cardiac mitochondria, and mitochondrial membrane potential, ATP generation, and tricarboxylic acid (TCA) cycle activity (measured by mitochondrial NADH content) all increase following an elevation in mitochondrial Ca2+.76–79 In addition, Territo et al.80 demonstrated in isolated cardiac mitochondria that Ca2+ is required for ATP production via the F0F1-ATPase and that the Ca2+ Km for activation of the F0F1-ATPase falls within physiological levels. This study also estimated that the activation of F0F1-ATPase may have been responsible for >60% of the Ca2+-induced activation of OXPHOS, whereas the contribution of Ca2+-sensitive dehydrogenases accounted for ∼40%. Thus, Ca2+-mediated activation of oxidative metabolism likely results in increased energy substrate oxidation and ATP production, and Ca2+ exchange between the cytosol and the mitochondria has therefore been suggested to provide a simple but elegant way to coordinate the rate of ATP production with its use for cardiac contraction.81
Given the importance of the regulatory interactions between mitochondrial energy metabolism and Ca2+ handling, it seems plausible that impaired mitochondrial Ca2+ handling may contribute to the development of contractile dysfunction in diabetes. In streptozotocin-induced diabetes, the rate of Ca2+ uptake into rat heart mitochondria is low compared with normal rats and is accompanied by a reduction of α-ketoglutarate-supported mitochondrial state 3 respiration.25,77,82,83 In contrast, succinate-supported respiration, which is initiated by the Ca2+-insensitive enzyme succinate dehydrogenase, was unaffected.25 Importantly, mitochondrial Ca2+ concentrations in diabetic hearts were in the range where modulation of dehydrogenase activation occurs, suggesting that decreased flux through Ca2+-sensitive dehydrogenases may indeed contribute to impaired ATP generation in these hearts.
The lower mitochondrial concentrations of Ca2+ may be the consequence of the smaller systolic transients in cytosolic Ca2+ concentration which have been observed in several studies in myocytes from Type 1 diabetic hearts.84,85 This may be the consequence of reduced Ca2+ loading of the sarcoplasmic reticulum and decreased activity of SERCA2a in these hearts.86,87 Several studies observed reduced expression of SERCA2a in streptozotocin-induced diabetic animals.88,89 In addition, altered phospholamban–SERCA2a interaction, free radical-induced damage, and reduced glycolytic ATP supply to SERCA2a have been observed in diabetic hearts.90–93 Another possibility may be that mitochondria from Type 1 diabetic hearts have depressed capacity to accumulate Ca2+, because of an enhanced sensitivity to induction of MPTP opening. Oliveira et al.94 observed that in response to a Ca2+ stimulus, mitochondria from both wild-type and streptozotocin-induced diabetic rats accumulated the same amount of calcium, but mitochondria from the diabetic hearts were not able to retain the accumulated Ca2+, unless the MPTP inhibitor cyclosporin was present.
Less conclusive data are available for Type 2 diabetic models. Recent studies demonstrated reduced intracellular Ca2+ release upon electrical stimulation, a slowed intracellular Ca2+ decay rate, and impaired mitochondrial Ca2+ handling in Type 2 diabetic ob/ob mice.95,96 In db/db mice, Ca2+ transients from isolated myocytes showed lower systolic and diastolic Ca2+ levels, and the decay rate of Ca2+ transients was also reduced.97 In the same study, increased Ca2+ leakage from the sarcoplasmic reticulum was observed, and the impairment in sarcoplasmic reticulum Ca2+ handling was attributed to a decline in sarcoplasmic reticulum activity, likely due to a mild decrease in SERCA2a expression and a large increase in phospholamban expression.97 Data on mitochondrial Ca2+-handling were not provided in this study.
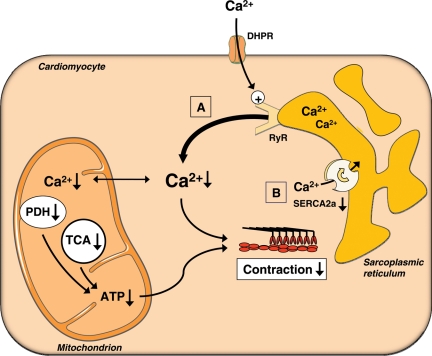
Taken together, these data support the hypothesis that impaired mitochondrial Ca2+ handling may compromise cardiac energy metabolism and thereby contribute to the development of contractile dysfunction in diabetic hearts. It is tempting to speculate that lower cytosolic Ca2+ levels may reduce mitochondrial Ca2+ transients, which may result in reduced activation of mitochondrial metabolic enzymes and therefore impaired ATP production and contractility (Figure 2).
Figure 2.
Impaired mitochondrial Ca2+ handling. Impaired sarcoplasmic reticulum Ca2+ release (A) or reduced re-uptake (B) may limit peak cytosolic Ca2+ levels, which may limit intramitochondrial Ca2+ accumulation during contraction. This may reduce the attendant increase in dehydrogenase activation and ATP synthesis. Reduced ATP synthesis and lower cytosolic Ca2+ transients may both contribute to contractile dysfunction or impaired relaxation in diabetes. PDH, pyruvate dehydrogenase; TCA, tricarboxylic acid; DHPR, dihydropyridine receptor; RyR, ryanodine receptor.
4.4. Mitochondrial biogenesis
Mitochondrial biogenesis is a complex and dynamic process that regulates and determines the content and composition of mitochondria in tissues. Stimuli that increase mitochondrial biogenesis increase mitochondrial DNA replication as well as increased incorporation of nuclear-encoded proteins into mitochondrial membranes and matrix. There is also a coordinate increase in the biogenesis of mitochondrial membranes, which involves increased synthesis of mitochondrial phospholipids. The ways in which diabetes may influence these complex and highly regulated steps in mitochondrial biogenesis are incompletely understood. A major regulator of mitochondrial biogenesis is the highly inducible transcriptional co-activator peroxisome proliferator-activated receptor γ co-activator 1α (PGC-1α), which docks to a variety of transcription factors to amplify their activity and their target gene expression, including oestrogen-related receptor α, nuclear respiratory factors 1 and 2, and also PPARα.98 When ectopically expressed in fat or muscle cells, PGC-1α robustly increased mitochondrial DNA content, increased the expression of a large set of nuclear and mitochondrial-encoded mitochondrial genes, and increased mitochondrial respiration.99 In the heart, high-level cardiomyocyte-specific overexpression of PGC-1α dramatically increased cardiomyocyte mitochondrial volume density, but precipitated contractile dysfunction.100,101 Interestingly, knockout of PGC-1α in the heart resulted in reduced expression of fatty acid oxidation, TCA cycle, and OXPHOS genes, but did not alter mitochondrial volume density.102
In hearts of insulin-resistant and/or diabetic animals, mitochondrial content is increased.21,22 UCP-DTA mice showed increased mitochondrial volume density, increased mitochondrial DNA content, and increased OXPHOS expression. In contrast, ADP-stimulated mitochondrial respiration was not increased, and ATP synthesis was reduced.21 Similarly, mitochondrial volume density and mitochondrial DNA content were increased in ob/ob and db/db mice, but these alterations were also accompanied by impairment in ADP-stimulated respiration and ATP synthesis.20,22,103 Thus, cardiac mitochondrial biogenesis occurs in insulin-resistant and/or diabetic mice but is not accompanied by a coordinate increase in mitochondrial function. These observations suggest that the diabetes-associated changes in mitochondrial biogenesis might not be associated with a coordinate increase in all of the processes that are required to generate competent mitochondria and raise important questions about the underlying mechanisms that may drive mitochondrial biogenesis in these hearts, and if the mitochondrial biogenesis observed is adaptive or maladaptive.
In UCP-DTA mice, mitochondrial biogenesis was accompanied by a significant increase in PGC-1α and OXPHOS gene expression, suggesting that PGC-1α signalling may drive the mitochondrial biogenesis in these mice.21 However, PGC-1α expression was not increased and even decreased at later ages in ob/ob mice that developed decreased OXPHOS protein content, suggesting that mitochondrial proliferation may have occurred via a PGC-1α-independent pathway.20,28 Moreover, in db/db mice, although mitochondrial biogenesis was shown to be associated with increased expression of PGC-1α expression, there was no coordinate up-regulation in OXPHOS subunit gene expression.22 These discrepancies may argue against an increase in PGC-1α signalling as a unifying underlying hypothesis for increased mitochondrial biogenesis in diabetic hearts. However, model-specific differences between UCP-DTA mice and models of Type 2 diabetes with impaired leptin action cannot be ruled out. The question remains if increased mitochondrial biogenesis per se is beneficial or detrimental. The mitochondrial biogenic response could represent a compensatory mechanism since mitochondrial function is impaired. If true, then this mechanism represents only a partial adaptation because overall respiratory capacity was not increased and even impaired in mitochondria from UCP-DTA, ob/ob, and db/db mice.
Cellular mitochondrial content reflects the balance between degradation of existing mitochondria and generation of new ones. Cellular processes such as autophagy or increased degradation or turnover of mitochondrial proteins could contribute to imbalance between mitochondrial mass and function as observed in diabetes. There is little known about the contributions of these processes to altered mitochondrial mass or changes in mitochondrial structure in the context of diabetes. On the basis of studies performed in other cells and tissues, it can be hypothesized that diabetes-associated changes in metabolism, such as reduced insulin signalling or increased oxidative stress, could lead to increased autophagy.104,105 Elucidation of these questions remains an important area for future research.
4.5. Remodelling and post-translational modifications of the mitochondrial proteome
Several comparative proteomic analyses demonstrated that remodelling of the mitochondrial proteome occurs in Type 1 diabetic hearts.24,27,106 Combining stable isotope labelling and two-dimensional gel electrophoresis, Turko and Murad106 reported increased abundance of several proteins involved in mitochondrial fatty acid oxidation in mitochondria of streptozotocin-diabetic rats, whereas a selection of OXPHOS subunits was down-regulated. Similarly, increased cardiac levels of mitochondrial fatty acid oxidation proteins were observed in two other independent studies using whole-cell proteomic approaches in Type 1 diabetic OVE26 mice and streptozotocin-diabetic rats, respectively.27,107 Using gel-independent label-free mass spectrometry-based expression analysis of the mitochondrial proteome, we recently reported the detection of 123 mitochondrial proteins in hearts of wild-type and Type 1 diabetic Akita mice, 23% of which were significantly different in abundance between the groups.24 As in previous studies, mitochondrial content of fatty acid oxidation proteins was increased in Akita hearts, although to a lesser extent than in OVE26 mice or streptozotocin-diabetic rats. In addition, several OXPHOS subunits and TCA cycle enzymes were down-regulated in diabetic mitochondria, associated with decreased expression of OXPHOS genes, regulators of mitochondrial biogenesis (including PGC-1α and β), and impairment in mitochondrial respiratory function. Thus, the consistently observed increase in cardiac fatty acid oxidation rates and impairment in mitochondrial respiration rates may be based on increased levels of fatty acid oxidation proteins and decreased abundance of OXPHOS subunits in diabetic mitochondria, respectively. In Akita mice, reduced mitochondrial OXPHOS subunit content may be related to reduced transcriptional activity of the PGC-1 signalling cascade, although other mechanisms such as increased protein turnover or reduced mitochondrial protein import cannot be ruled out. Similar studies investigating the mitochondrial proteome in models of Type 2 diabetes remain to be undertaken.
Post-translational modifications of mitochondrial proteins have been described in diabetes and include nitrosylation and O-linked glycosylation.66,108 Reversal of O-linked glycosylation by overexpression of N-acetyl-β-d-glucosaminidase restored the activity of ATP synthase and increased oxygen consumption in cultured cardiomyocytes that were exposed to high glucose. Thus, proteomic analyses have shed important new insights into the pathogenesis of mitochondrial dysfunction in diabetes. Future studies combining these approaches with analyses of metabolic intermediates (metabolomics) are likely to yield additional new insights.
4.6. Impaired cardiac insulin signalling
Most rodent models mimicking the metabolic syndrome and Type 2 diabetes exhibit hyperinsulinaemia and insulin resistance in multiple tissues. These models also exhibit insulin resistance in the heart and myocardial contractile dysfunction.28,29 Thus, the question arises if impaired cardiac insulin signalling per se may contribute to the development of contractile dysfunction. Since all of these models are characterized by at least one or several systemic metabolic alterations, such as hyperlipidaemia, hyperglycaemia, or hormonal alterations, the evaluation of the contribution of impaired cardiac insulin signalling to contractile dysfunction is challenging. Thus, we generated mice with a cardiomyocyte-restricted deletion of the insulin receptor (CIRKO mice).109 These mice allow the investigation of impaired myocardial insulin signalling without confounding effects of systemic metabolic alterations and obesity. CIRKO mice show a modest and age-dependent decrease in contractile function.52,109 This contractile dysfunction is associated with reduced insulin-stimulated glucose uptake and a decrease in the oxidation of glucose and fatty acids as they age and as contractile dysfunction occurs.109 In addition, Boudina et al.52 recently demonstrated that CIRKO hearts have impaired mitochondrial respiration and ATP synthesis rates, reduced expression of genes encoding for OXPHOS subunits, UCPs, and fatty acid oxidation proteins, as well as decreased mitochondrial levels of fatty acid oxidation proteins, TCA cycle proteins, and remodelling of the OXPHOS subunit composition. Importantly, CIRKO mice also exhibit increased mitochondrial H2O2 generation and mitochondrial uncoupling in the presence of long chain fatty acids, which could be prevented by pre-treatment of CIRKO mice with the mitochondrial superoxide dismutase mimetic manganese[III]tetrakis(4-benzoic acid)porphyrin. Since Type 2 diabetic mice exhibit cardiac insulin resistance, this study suggests that fatty acid-induced mitochondrial uncoupling in Type 2 diabetic mice may be related to impaired cardiac insulin signalling. Thus, impaired cardiomyocyte insulin signalling has profound effects on cardiac mitochondria and may play a fundamental role in the pathogenesis of diabetes-induced abnormalities in cardiac mitochondrial function.
With respect to the pathogenesis of cardiac insulin resistance and concomitant mitochondrial dysfunction in diabetic hearts, the role of adipocytokines deserves mention. Obesity is associated with altered circulating concentrations of various adipokines such as leptin and resistin, which might have independent effects on cardiac metabolism. For example, resistin has been shown to impair cardiomyocyte glucose uptake.110 The most abundant adipocytokine secreted by adipose tissue is adiponectin, which may exert diverse cardiovascular effects. Adiponectin administration may attenuate cardiac hypertrophy following pressure overload both in adiponectin-knockout mice and Type 2 diabetic db/db mice,111 and reduces myocardial infarct area following ischaemia–reperfusion.112 Hypo-adiponectinaemia is a consistent feature of Type 2 diabetes and insulin-resistant states, and given the physiological effects of adiponectin to increase FA oxidation in skeletal muscle and liver, the reduced concentrations might be might be mechanistically linked to the pathogenesis of insulin resistance.113–115 Importantly, adiponectin also regulates mitochondrial function. Adiponectin can increase mitochondrial biogenesis, palmitate oxidation, and citrate synthase activity in primary human myotubes, and adiponectin-knockout mice have decreased cytochrome c oxidase activity, decreased citrate synthase activity, reduced mitochondrial DNA content, and reduced PGC-1α expression in skeletal muscle.116 In addition, a recent report by Iwabu et al.117 demonstrated that adiponectin and adiponectin receptor 1 regulate PGC-1α expression and acetylation, and thereby mitochondrial content via activation of the AMPK/SIRT1 pathway and activation of CaMKKβ. Thus, these studies suggest a link between adiponectin, mitochondrial function, and the pathogenesis of insulin resistance and Type 2 diabetes. Owing to overlapping metabolic signalling mechanisms in oxidative skeletal muscle and cardiac muscle, it is tempting to speculate that reduced adiponectin levels in diabetes may also negatively impact cardiac mitochondrial function and insulin sensitivity.
5. Conclusions
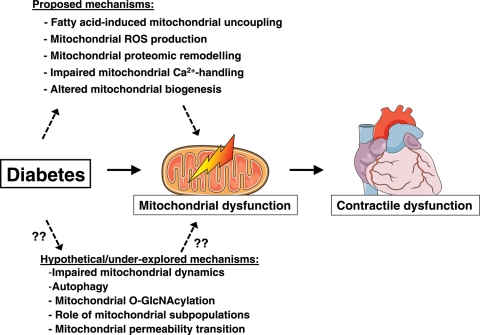
We have discussed several potential mechanisms that may contribute to changes in myocardial mitochondrial energetics in Type 1 and Type 2 diabetic hearts. It is likely that no one single mechanism, but rather the combination of several mechanisms, may lead to mitochondrial dysfunction (summarized in Figure 3). We propose that the resultant mitochondrial dysfunction will compromise cardiac ATP generation, ultimately leading to contractile dysfunction. We have presented evidence that suggests that important differences may exist between models of Type 1 and Type 2 diabetes in the pathogenesis of mitochondrial dysfunction. Additional studies will be required to completely elucidate the molecular basis for these differences. Novel but relatively under-explored hypotheses may include impairment in mitochondrial dynamics, increased O-linked β-N-acetylglucosamine glycosylation of mitochondrial proteins, differential roles of myocardial mitochondrial subpopulations, and contributions of increased mitochondrial permeability transition108,118–120 (Figure 3). Given the dramatic increase in the prevalence of the metabolic syndrome and subsequent Type 2 diabetes, it might be predicted that our societies might face an explosive increase in heart disease. Thus, a compelling case can be made for additional mechanistic studies both in animal models and in particular also in humans that will address fundamental underlying pathophysiological mechanisms with the goal to reveal useful targets for the development of therapeutic strategies that may ameliorate or prevent cardiac disease in diabetic subjects.
Figure 3.
Proposed and hypothetical/under-explored mechanisms leading to cardiac mitochondrial dysfunction in diabetes.
Conflict of interest: none declared.
Funding
H.B. was supported by a research fellowship grant from the German Research Foundation (DFG). Research in the Abel laboratory is supported by grants [UO1HL70525, UO1 HL087947 (Animal Models of Diabetes Complications Consortium—AMDCC), and RO1 HL70070, RO1 HL73167] from the National Institutes of Health and grants from the Juvenile Diabetes Research Foundation, the American Diabetes Association, and the American Heart Association.
References
- 1.Rubler S, Dlugash J, Yuceoglu YZ, Kumral T, Branwood AW, Grishman A. New type of cardiomyopathy associated with diabetic glomerulosclerosis. Am J Cardiol. 1972;30:595–602. doi: 10.1016/0002-9149(72)90595-4. doi:10.1016/0002-9149(72)90595-4. [DOI] [PubMed] [Google Scholar]
- 2.Hamby RI, Zoneraich S, Sherman L. Diabetic cardiomyopathy. JAMA. 1974;229:1749–1754. doi:10.1001/jama.229.13.1749. [PubMed] [Google Scholar]
- 3.Regan TJ, Lyons MM, Ahmed SS, Levinson GE, Oldewurtel HA, Ahmad MR, et al. Evidence for cardiomyopathy in familial diabetes mellitus. J Clin Invest. 1977;60:884–899. doi: 10.1172/JCI108843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ho KK, Pinsky JL, Kannel WB, Levy D. The epidemiology of heart failure: the Framingham study. J Am Coll Cardiol. 1993;22:6A–13A. doi: 10.1016/0735-1097(93)90455-a. doi:10.1016/0735-1097(93)90455-A. [DOI] [PubMed] [Google Scholar]
- 5.Kannel WB, McGee DL. Diabetes and cardiovascular disease. The Framingham study. JAMA. 1979;241:2035–2038. doi: 10.1001/jama.241.19.2035. doi:10.1001/jama.241.19.2035. [DOI] [PubMed] [Google Scholar]
- 6.Bell DS. Diabetic cardiomyopathy. Diabetes Care. 2003;26:2949–2951. doi: 10.2337/diacare.26.10.2949. doi:10.2337/diacare.26.10.2949. [DOI] [PubMed] [Google Scholar]
- 7.Fein FS. Diabetic cardiomyopathy. Diabetes Care. 1990;13:1169–1179. doi: 10.2337/diacare.13.11.1169. doi:10.2337/diacare.13.11.1169. [DOI] [PubMed] [Google Scholar]
- 8.Peterson LR, Herrero P, Schechtman KB, Racette SB, Waggoner AD, Kisrieva-Ware Z, et al. Effect of obesity and insulin resistance on myocardial substrate metabolism and efficiency in young women. Circulation. 2004;109:2191–2196. doi: 10.1161/01.CIR.0000127959.28627.F8. doi:10.1161/01.CIR.0000127959.28627.F8. [DOI] [PubMed] [Google Scholar]
- 9.Diamant M, Lamb HJ, Groeneveld Y, Endert EL, Smit JW, Bax JJ, et al. Diastolic dysfunction is associated with altered myocardial metabolism in asymptomatic normotensive patients with well-controlled type 2 diabetes mellitus. J Am Coll Cardiol. 2003;42:328–335. doi: 10.1016/s0735-1097(03)00625-9. doi:10.1016/S0735-1097(03)00625-9. [DOI] [PubMed] [Google Scholar]
- 10.Scheuermann-Freestone M, Madsen PL, Manners D, Blamire AM, Buckingham RE, Styles P, et al. Abnormal cardiac and skeletal muscle energy metabolism in patients with type 2 diabetes. Circulation. 2003;107:3040–3046. doi: 10.1161/01.CIR.0000072789.89096.10. doi:10.1161/01.CIR.0000072789.89096.10. [DOI] [PubMed] [Google Scholar]
- 11.Casademont J, Miro O. Electron transport chain defects in heart failure. Heart Fail Rev. 2002;7:131–139. doi: 10.1023/a:1015372407647. [DOI] [PubMed] [Google Scholar]
- 12.Neubauer S, Horn M, Cramer M, Harre K, Newell JB, Peters W, et al. Myocardial phosphocreatine-to-ATP ratio is a predictor of mortality in patients with dilated cardiomyopathy. Circulation. 1997;96:2190–2196. doi: 10.1161/01.cir.96.7.2190. [DOI] [PubMed] [Google Scholar]
- 13.Neubauer S, Krahe T, Schindler R, Horn M, Hillenbrand H, Entzeroth C, et al. 31P magnetic resonance spectroscopy in dilated cardiomyopathy and coronary artery disease. Altered cardiac high-energy phosphate metabolism in heart failure. Circulation. 1992;86:1810–1818. doi: 10.1161/01.cir.86.6.1810. [DOI] [PubMed] [Google Scholar]
- 14.Rijzewijk LJ, van der Meer RW, Lamb HJ, de Jong HW, Lubberink M, Romijn JA, et al. Altered myocardial substrate metabolism and decreased diastolic function in nonischemic human diabetic cardiomyopathy: studies with cardiac positron emission tomography and magnetic resonance imaging. J Am Coll Cardiol. 2009;54:1524–1532. doi: 10.1016/j.jacc.2009.04.074. doi:10.1016/j.jacc.2009.04.074. [DOI] [PubMed] [Google Scholar]
- 15.van der Meer RW, Rijzewijk LJ, de Jong HW, Lamb HJ, Lubberink M, Romijn JA, et al. Pioglitazone improves cardiac function and alters myocardial substrate metabolism without affecting cardiac triglyceride accumulation and high-energy phosphate metabolism in patients with well-controlled type 2 diabetes mellitus. Circulation. 2009;119:2069–2077. doi: 10.1161/CIRCULATIONAHA.108.803916. doi:10.1161/CIRCULATIONAHA.108.803916. [DOI] [PubMed] [Google Scholar]
- 16.Anderson EJ, Kypson AP, Rodriguez E, Anderson CA, Lehr EJ, Neufer PD. Substrate-specific derangements in mitochondrial metabolism and redox balance in the atrium of the type 2 diabetic human heart. J Am Coll Cardiol. 2009;54:1891–1898. doi: 10.1016/j.jacc.2009.07.031. doi:10.1016/j.jacc.2009.07.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bugger H, Abel ED. Rodent models of diabetic cardiomyopathy. Dis Model Mech. 2009;2:454–466. doi: 10.1242/dmm.001941. doi:10.1242/dmm.001941. [DOI] [PubMed] [Google Scholar]
- 18.Kuo TH, Giacomelli F, Wiener J. Oxidative metabolism of Polytron versus Nagarse mitochondria in hearts of genetically diabetic mice. Biochim Biophys Acta. 1985;806:9–15. doi: 10.1016/0005-2728(85)90076-3. [DOI] [PubMed] [Google Scholar]
- 19.Kuo TH, Moore KH, Giacomelli F, Wiener J. Defective oxidative metabolism of heart mitochondria from genetically diabetic mice. Diabetes. 1983;32:781–787. doi: 10.2337/diab.32.9.781. doi:10.2337/diabetes.32.9.781. [DOI] [PubMed] [Google Scholar]
- 20.Boudina S, Sena S, O'Neill BT, Tathireddy P, Young ME, Abel ED. Reduced mitochondrial oxidative capacity and increased mitochondrial uncoupling impair myocardial energetics in obesity. Circulation. 2005;112:2686–2695. doi: 10.1161/CIRCULATIONAHA.105.554360. doi:10.1161/CIRCULATIONAHA.105.554360. [DOI] [PubMed] [Google Scholar]
- 21.Duncan JG, Fong JL, Medeiros DM, Finck BN, Kelly DP. Insulin-resistant heart exhibits a mitochondrial biogenic response driven by the peroxisome proliferator-activated receptor-alpha/PGC-1alpha gene regulatory pathway. Circulation. 2007;115:909–917. doi: 10.1161/CIRCULATIONAHA.106.662296. doi:10.1161/CIRCULATIONAHA.106.662296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Boudina S, Sena S, Theobald H, Sheng X, Wright JJ, Hu XX, et al. Mitochondrial energetics in the heart in obesity-related diabetes: direct evidence for increased uncoupled respiration and activation of uncoupling proteins. Diabetes. 2007;56:2457–2466. doi: 10.2337/db07-0481. doi:10.2337/db07-0481. [DOI] [PubMed] [Google Scholar]
- 23.Bugger H, Boudina S, Hu XX, Tuinei J, Zaha VG, Theobald HA, et al. Type 1 diabetic akita mouse hearts are insulin sensitive but manifest structurally abnormal mitochondria that remain coupled despite increased uncoupling protein 3. Diabetes. 2008;57:2924–2932. doi: 10.2337/db08-0079. doi:10.2337/db08-0079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Bugger H, Chen D, Riehle C, Soto J, Theobald HA, Hu XX, et al. Tissue-specific remodeling of the mitochondrial proteome in type 1 diabetic akita mice. Diabetes. 2009;58:1986–1997. doi: 10.2337/db09-0259. doi:10.2337/db09-0259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Flarsheim CE, Grupp IL, Matlib MA. Mitochondrial dysfunction accompanies diastolic dysfunction in diabetic rat heart. Am J Physiol. 1996;271:H192–H202. doi: 10.1152/ajpheart.1996.271.1.H192. [DOI] [PubMed] [Google Scholar]
- 26.Lashin OM, Szweda PA, Szweda LI, Romani AM. Decreased complex II respiration and HNE-modified SDH subunit in diabetic heart. Free Radic Biol Med. 2006;40:886–896. doi: 10.1016/j.freeradbiomed.2005.10.040. doi:10.1016/j.freeradbiomed.2005.10.040. [DOI] [PubMed] [Google Scholar]
- 27.Shen X, Zheng S, Thongboonkerd V, Xu M, Pierce WM, Jr, Klein JB, et al. Cardiac mitochondrial damage and biogenesis in a chronic model of type 1 diabetes. Am J Physiol Endocrinol Metab. 2004;287:E896–E905. doi: 10.1152/ajpendo.00047.2004. doi:10.1152/ajpendo.00047.2004. [DOI] [PubMed] [Google Scholar]
- 28.Buchanan J, Mazumder PK, Hu P, Chakrabarti G, Roberts MW, Yun U, et al. Reduced cardiac efficiency and altered substrate metabolism precedes the onset of hyperglycemia and contractile dysfunction in two mouse models of insulin resistance and obesity. Endocrinology. 2005;146:5341–5349. doi: 10.1210/en.2005-0938. doi:10.1210/en.2005-0938. [DOI] [PubMed] [Google Scholar]
- 29.Mazumder PK, O'Neill BT, Roberts MW, Buchanan J, Yun UJ, Cooksey RC, et al. Impaired cardiac efficiency and increased fatty acid oxidation in insulin-resistant ob/ob mouse hearts. Diabetes. 2004;53:2366–2374. doi: 10.2337/diabetes.53.9.2366. doi:10.2337/diabetes.53.9.2366. [DOI] [PubMed] [Google Scholar]
- 30.Wang P, Lloyd SG, Zeng H, Bonen A, Chatham JC. Impact of altered substrate utilization on cardiac function in isolated hearts from Zucker diabetic fatty rats. Am J Physiol Heart Circ Physiol. 2005;288:H2102–H2110. doi: 10.1152/ajpheart.00935.2004. doi:10.1152/ajpheart.00935.2004. [DOI] [PubMed] [Google Scholar]
- 31.Aoyama T, Peters JM, Iritani N, Nakajima T, Furihata K, Hashimoto T, et al. Altered constitutive expression of fatty acid-metabolizing enzymes in mice lacking the peroxisome proliferator-activated receptor alpha (PPARalpha) J Biol Chem. 1998;273:5678–5684. doi: 10.1074/jbc.273.10.5678. doi:10.1074/jbc.273.10.5678. [DOI] [PubMed] [Google Scholar]
- 32.Barger PM, Kelly DP. PPAR signaling in the control of cardiac energy metabolism. Trends Cardiovasc Med. 2000;10:238–245. doi: 10.1016/s1050-1738(00)00077-3. doi:10.1016/S1050-1738(00)00077-3. [DOI] [PubMed] [Google Scholar]
- 33.Finck BN, Lehman JJ, Leone TC, Welch MJ, Bennett MJ, Kovacs A, et al. The cardiac phenotype induced by PPARalpha overexpression mimics that caused by diabetes mellitus. J Clin Invest. 2002;109:121–130. doi: 10.1172/JCI14080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Young ME, Patil S, Ying J, Depre C, Ahuja HS, Shipley GL, et al. Uncoupling protein 3 transcription is regulated by peroxisome proliferator-activated receptor (alpha) in the adult rodent heart. FASEB J. 2001;15:833–845. doi: 10.1096/fj.00-0351com. doi:10.1096/fj.00-0351com. [DOI] [PubMed] [Google Scholar]
- 35.Kjekshus JK, Mjos OD. Effect of free fatty acids on myocardial function and metabolism in the ischemic dog heart. J Clin Invest. 1972;51:1767–1776. doi: 10.1172/JCI106978. doi:10.1172/JCI106978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Mjos OD. Effect of free fatty acids on myocardial function and oxygen consumption in intact dogs. J Clin Invest. 1971;50:1386–1389. doi: 10.1172/JCI106621. doi:10.1172/JCI106621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.How OJ, Aasum E, Severson DL, Chan WY, Essop MF, Larsen TS. Increased myocardial oxygen consumption reduces cardiac efficiency in diabetic mice. Diabetes. 2006;55:466–473. doi: 10.2337/diabetes.55.02.06.db05-1164. doi:10.2337/diabetes.55.02.06.db05-1164. [DOI] [PubMed] [Google Scholar]
- 38.Young ME, Guthrie PH, Razeghi P, Leighton B, Abbasi S, Patil S, et al. Impaired long-chain fatty acid oxidation and contractile dysfunction in the obese Zucker rat heart. Diabetes. 2002;51:2587–2595. doi: 10.2337/diabetes.51.8.2587. doi:10.2337/diabetes.51.8.2587. [DOI] [PubMed] [Google Scholar]
- 39.Brand MD, Affourtit C, Esteves TC, Green K, Lambert AJ, Miwa S, et al. Mitochondrial superoxide: production, biological effects, and activation of uncoupling proteins. Free Radic Biol Med. 2004;37:755–767. doi: 10.1016/j.freeradbiomed.2004.05.034. doi:10.1016/j.freeradbiomed.2004.05.034. [DOI] [PubMed] [Google Scholar]
- 40.Brand MD, Esteves TC. Physiological functions of the mitochondrial uncoupling proteins UCP2 and UCP3. Cell Metab. 2005;2:85–93. doi: 10.1016/j.cmet.2005.06.002. doi:10.1016/j.cmet.2005.06.002. [DOI] [PubMed] [Google Scholar]
- 41.Nicholls DG, Locke RM. Thermogenic mechanisms in brown fat. Physiol Rev. 1984;64:1–64. doi: 10.1152/physrev.1984.64.1.1. [DOI] [PubMed] [Google Scholar]
- 42.Ledesma A, de Lacoba MG, Rial E. The mitochondrial uncoupling proteins. Genome Biol. 2002;3:Reviews3015. doi: 10.1186/gb-2002-3-12-reviews3015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Murray AJ, Anderson RE, Watson GC, Radda GK, Clarke K. Uncoupling proteins in human heart. Lancet. 2004;364:1786–1788. doi: 10.1016/S0140-6736(04)17402-3. doi:10.1016/S0140-6736(04)17402-3. [DOI] [PubMed] [Google Scholar]
- 44.Murray AJ, Panagia M, Hauton D, Gibbons GF, Clarke K. Plasma free fatty acids and peroxisome proliferator-activated receptor alpha in the control of myocardial uncoupling protein levels. Diabetes. 2005;54:3496–3502. doi: 10.2337/diabetes.54.12.3496. doi:10.2337/diabetes.54.12.3496. [DOI] [PubMed] [Google Scholar]
- 45.Clapham JC, Arch JR, Chapman H, Haynes A, Lister C, Moore GB, et al. Mice overexpressing human uncoupling protein-3 in skeletal muscle are hyperphagic and lean. Nature. 2000;406:415–418. doi: 10.1038/35019082. doi:10.1038/35019082. [DOI] [PubMed] [Google Scholar]
- 46.Vidal-Puig AJ, Grujic D, Zhang CY, Hagen T, Boss O, Ido Y, et al. Energy metabolism in uncoupling protein 3 gene knockout mice. J Biol Chem. 2000;275:16258–16266. doi: 10.1074/jbc.M910179199. doi:10.1074/jbc.M910179199. [DOI] [PubMed] [Google Scholar]
- 47.Echtay KS, Esteves TC, Pakay JL, Jekabsons MB, Lambert AJ, Portero-Otin M, et al. A signalling role for 4-hydroxy-2-nonenal in regulation of mitochondrial uncoupling. EMBO J. 2003;22:4103–4110. doi: 10.1093/emboj/cdg412. doi:10.1093/emboj/cdg412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Echtay KS, Roussel D, St-Pierre J, Jekabsons MB, Cadenas S, Stuart JA, et al. Superoxide activates mitochondrial uncoupling proteins. Nature. 2002;415:96–99. doi: 10.1038/415096a. doi:10.1038/415096a. [DOI] [PubMed] [Google Scholar]
- 49.Brand MD, Pakay JL, Ocloo A, Kokoszka J, Wallace DC, Brookes PS, et al. The basal proton conductance of mitochondria depends on adenine nucleotide translocase content. Biochem J. 2005;392:353–362. doi: 10.1042/BJ20050890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Yamagishi SI, Edelstein D, Du XL, Kaneda Y, Guzman M, Brownlee M. Leptin induces mitochondrial superoxide production and monocyte chemoattractant protein-1 expression in aortic endothelial cells by increasing fatty acid oxidation via protein kinase A. J Biol Chem. 2001;276:25096–25100. doi: 10.1074/jbc.M007383200. doi:10.1074/jbc.M007383200. [DOI] [PubMed] [Google Scholar]
- 51.Herlein JA, Fink BD, O'Malley Y, Sivitz WI. Superoxide and respiratory coupling in mitochondria of insulin-deficient diabetic rats. Endocrinology. 2009;150:46–55. doi: 10.1210/en.2008-0404. doi:10.1210/en.2008-0404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Boudina S, Bugger H, Sena S, O'Neill BT, Zaha VG, Ilkun O, et al. Contribution of impaired myocardial insulin signaling to mitochondrial dysfunction and oxidative stress in the heart. Circulation. 2009;119:1272–1283. doi: 10.1161/CIRCULATIONAHA.108.792101. doi:10.1161/CIRCULATIONAHA.108.792101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Skulachev VP. Anion carriers in fatty acid-mediated physiological uncoupling. J Bioenerg Biomembr. 1999;31:431–445. doi: 10.1023/a:1005492205984. doi:10.1023/A:1005492205984. [DOI] [PubMed] [Google Scholar]
- 54.Korshunov SS, Skulachev VP, Starkov AA. High protonic potential actuates a mechanism of production of reactive oxygen species in mitochondria. FEBS Lett. 1997;416:15–18. doi: 10.1016/s0014-5793(97)01159-9. doi:10.1016/S0014-5793(97)01159-9. [DOI] [PubMed] [Google Scholar]
- 55.St-Pierre J, Buckingham JA, Roebuck SJ, Brand MD. Topology of superoxide production from different sites in the mitochondrial electron transport chain. J Biol Chem. 2002;277:44784–44790. doi: 10.1074/jbc.M207217200. doi:10.1074/jbc.M207217200. [DOI] [PubMed] [Google Scholar]
- 56.Skulachev VP. Role of uncoupled and non-coupled oxidations in maintenance of safely low levels of oxygen and its one-electron reductants. Q Rev Biophys. 1996;29:169–202. doi: 10.1017/s0033583500005795. doi:10.1017/S0033583500005795. [DOI] [PubMed] [Google Scholar]
- 57.Garciarena CD, Caldiz CI, Correa MV, Schinella GR, Mosca SM, Chiappe de Cingolani GE, et al. Na+/H+ exchanger-1 inhibitors decrease myocardial superoxide production via direct mitochondrial action. J Appl Physiol. 2008;105:1706–1713. doi: 10.1152/japplphysiol.90616.2008. doi:10.1152/japplphysiol.90616.2008. [DOI] [PubMed] [Google Scholar]
- 58.Zhou L, Aon MA, Almas T, Cortassa S, Winslow RL, O'Rourke B. A reaction-diffusion model of ROS-induced ROS release in a mitochondrial network. PLoS Comput Biol. 6:e1000657. doi: 10.1371/journal.pcbi.1000657. doi:10.1371/journal.pcbi.1000657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Wallace DC. Mitochondrial genetics: a paradigm for aging and degenerative diseases? Science. 1992;256:628–632. doi: 10.1126/science.1533953. doi:10.1126/science.1533953. [DOI] [PubMed] [Google Scholar]
- 60.Marra G, Cotroneo P, Pitocco D, Manto A, Di Leo MA, Ruotolo V, et al. Early increase of oxidative stress and reduced antioxidant defenses in patients with uncomplicated type 1 diabetes: a case for gender difference. Diabetes Care. 2002;25:370–375. doi: 10.2337/diacare.25.2.370. doi:10.2337/diacare.25.2.370. [DOI] [PubMed] [Google Scholar]
- 61.Rosen P, Nawroth PP, King G, Moller W, Tritschler HJ, Packer L. The role of oxidative stress in the onset and progression of diabetes and its complications: a summary of a Congress Series sponsored by UNESCO-MCBN, the American Diabetes Association and the German Diabetes Society. Diabetes Metab Res Rev. 2001;17:189–212. doi: 10.1002/dmrr.196. doi:10.1002/dmrr.196. [DOI] [PubMed] [Google Scholar]
- 62.Van Dam PS, Van Asbeck BS, Erkelens DW, Marx JJ, Gispen WH, Bravenboer B. The role of oxidative stress in neuropathy and other diabetic complications. Diabetes Metab Rev. 1995;11:181–192. doi: 10.1002/dmr.5610110303. doi:10.1002/dmr.5610110303. [DOI] [PubMed] [Google Scholar]
- 63.Jay D, Hitomi H, Griendling KK. Oxidative stress and diabetic cardiovascular complications. Free Radic Biol Med. 2006;40:183–192. doi: 10.1016/j.freeradbiomed.2005.06.018. doi:10.1016/j.freeradbiomed.2005.06.018. [DOI] [PubMed] [Google Scholar]
- 64.Nishikawa T, Edelstein D, Du XL, Yamagishi S, Matsumura T, Kaneda Y, et al. Normalizing mitochondrial superoxide production blocks three pathways of hyperglycaemic damage. Nature. 2000;404:787–790. doi: 10.1038/35008121. [DOI] [PubMed] [Google Scholar]
- 65.Li SY, Yang X, Ceylan-Isik AF, Du M, Sreejayan N, Ren J. Cardiac contractile dysfunction in Lep/Lep obesity is accompanied by NADPH oxidase activation, oxidative modification of sarco(endo)plasmic reticulum Ca2+-ATPase and myosin heavy chain isozyme switch. Diabetologia. 2006;49:1434–1446. doi: 10.1007/s00125-006-0229-0. doi:10.1007/s00125-006-0229-0. [DOI] [PubMed] [Google Scholar]
- 66.Turko IV, Li L, Aulak KS, Stuehr DJ, Chang JY, Murad F. Protein tyrosine nitration in the mitochondria from diabetic mouse heart. Implications to dysfunctional mitochondria in diabetes. J Biol Chem. 2003;278:33972–33977. doi: 10.1074/jbc.M303734200. doi:10.1074/jbc.M303734200. [DOI] [PubMed] [Google Scholar]
- 67.Ye G, Metreveli NS, Donthi RV, Xia S, Xu M, Carlson EC, et al. Catalase protects cardiomyocyte function in models of type 1 and type 2 diabetes. Diabetes. 2004;53:1336–1343. doi: 10.2337/diabetes.53.5.1336. doi:10.2337/diabetes.53.5.1336. [DOI] [PubMed] [Google Scholar]
- 68.Shen X, Zheng S, Metreveli NS, Epstein PN. Protection of cardiac mitochondria by overexpression of MnSOD reduces diabetic cardiomyopathy. Diabetes. 2006;55:798–805. doi: 10.2337/diabetes.55.03.06.db05-1039. doi:10.2337/diabetes.55.03.06.db05-1039. [DOI] [PubMed] [Google Scholar]
- 69.Santos DL, Palmeira CM, Seica R, Dias J, Mesquita J, Moreno AJ, et al. Diabetes and mitochondrial oxidative stress: a study using heart mitochondria from the diabetic Goto-Kakizaki rat. Mol Cell Biochem. 2003;246:163–170. doi:10.1023/A:1023475022025. [PubMed] [Google Scholar]
- 70.Conti M, Renaud IM, Poirier B, Michel O, Belair MF, Mandet C, et al. High levels of myocardial antioxidant defense in aging nondiabetic normotensive Zucker obese rats. Am J Physiol Regul Integr Comp Physiol. 2004;286:R793–R800. doi: 10.1152/ajpregu.00521.2002. [DOI] [PubMed] [Google Scholar]
- 71.Balaban RS. Cardiac energy metabolism homeostasis: role of cytosolic calcium. J Mol Cell Cardiol. 2002;34:1259–1271. doi: 10.1006/jmcc.2002.2082. doi:10.1006/jmcc.2002.2082. [DOI] [PubMed] [Google Scholar]
- 72.Isenberg G, Han S, Schiefer A, Wendt-Gallitelli MF. Changes in mitochondrial calcium concentration during the cardiac contraction cycle. Cardiovasc Res. 1993;27:1800–1809. doi: 10.1093/cvr/27.10.1800. doi:10.1093/cvr/27.10.1800. [DOI] [PubMed] [Google Scholar]
- 73.Lukacs GL, Kapus A. Measurement of the matrix free Ca2+ concentration in heart mitochondria by entrapped fura-2 and quin2. Biochem J. 1987;248:609–613. doi: 10.1042/bj2480609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Denton RM, Randle PJ, Martin BR. Stimulation by calcium ions of pyruvate dehydrogenase phosphate phosphatase. Biochem J. 1972;128:161–163. doi: 10.1042/bj1280161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Nichols BJ, Denton RM. Towards the molecular basis for the regulation of mitochondrial dehydrogenases by calcium ions. Mol Cell Biochem. 1995;149–150:203–212. doi: 10.1007/BF01076578. doi:10.1007/BF01076578. [DOI] [PubMed] [Google Scholar]
- 76.Duchen MR. Ca(2+)-dependent changes in the mitochondrial energetics in single dissociated mouse sensory neurons. Biochem J. 1992;283:41–50. doi: 10.1042/bj2830041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Hansford RG, Zorov D. Role of mitochondrial calcium transport in the control of substrate oxidation. Mol Cell Biochem. 1998;184:359–369. doi:10.1023/A:1006893903113. [PubMed] [Google Scholar]
- 78.Jouaville LS, Pinton P, Bastianutto C, Rutter GA, Rizzuto R. Regulation of mitochondrial ATP synthesis by calcium: evidence for a long-term metabolic priming. Proc Natl Acad Sci USA. 1999;96:13807–13812. doi: 10.1073/pnas.96.24.13807. doi:10.1073/pnas.96.24.13807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Robb-Gaspers LD, Burnett P, Rutter GA, Denton RM, Rizzuto R, Thomas AP. Integrating cytosolic calcium signals into mitochondrial metabolic responses. EMBO J. 1998;17:4987–5000. doi: 10.1093/emboj/17.17.4987. doi:10.1093/emboj/17.17.4987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Territo PR, Mootha VK, French SA, Balaban RS. Ca(2+) activation of heart mitochondrial oxidative phosphorylation: role of the F(0)/F(1)-ATPase. Am J Physiol Cell Physiol. 2000;278:C423–C435. doi: 10.1152/ajpcell.2000.278.2.C423. [DOI] [PubMed] [Google Scholar]
- 81.Duchen MR. Roles of mitochondria in health and disease. Diabetes. 2004;53((Suppl. 1)):S96–S102. doi: 10.2337/diabetes.53.2007.s96. [DOI] [PubMed] [Google Scholar]
- 82.Pierce GN, Dhalla NS. Heart mitochondrial function in chronic experimental diabetes in rats. Can J Cardiol. 1985;1:48–54. [PubMed] [Google Scholar]
- 83.Tanaka Y, Konno N, Kako KJ. Mitochondrial dysfunction observed in situ in cardiomyocytes of rats in experimental diabetes. Cardiovasc Res. 1992;26:409–414. doi: 10.1093/cvr/26.4.409. doi:10.1093/cvr/26.4.409. [DOI] [PubMed] [Google Scholar]
- 84.Lagadic-Gossmann D, Buckler KJ, Le Prigent K, Feuvray D. Altered Ca2+ handling in ventricular myocytes isolated from diabetic rats. Am J Physiol. 1996;270:H1529–H1537. doi: 10.1152/ajpheart.1996.270.5.H1529. [DOI] [PubMed] [Google Scholar]
- 85.Yu Z, Quamme GA, McNeill JH. Depressed [Ca2+]i responses to isoproterenol and cAMP in isolated cardiomyocytes from experimental diabetic rats. Am J Physiol. 1994;266:H2334–H2342. doi: 10.1152/ajpheart.1994.266.6.H2334. [DOI] [PubMed] [Google Scholar]
- 86.Bouchard RA, Bose D. Influence of experimental diabetes on sarcoplasmic reticulum function in rat ventricular muscle. Am J Physiol. 1991;260:H341–H354. doi: 10.1152/ajpheart.1991.260.2.H341. [DOI] [PubMed] [Google Scholar]
- 87.Ganguly PK, Pierce GN, Dhalla KS, Dhalla NS. Defective sarcoplasmic reticular calcium transport in diabetic cardiomyopathy. Am J Physiol. 1983;244:E528–E535. doi: 10.1152/ajpendo.1983.244.6.E528. [DOI] [PubMed] [Google Scholar]
- 88.Netticadan T, Temsah RM, Kent A, Elimban V, Dhalla NS. Depressed levels of Ca2+-cycling proteins may underlie sarcoplasmic reticulum dysfunction in the diabetic heart. Diabetes. 2001;50:2133–2138. doi: 10.2337/diabetes.50.9.2133. doi:10.2337/diabetes.50.9.2133. [DOI] [PubMed] [Google Scholar]
- 89.Zhong Y, Ahmed S, Grupp IL, Matlib MA. Altered SR protein expression associated with contractile dysfunction in diabetic rat hearts. Am J Physiol Heart Circ Physiol. 2001;281:H1137–H1147. doi: 10.1152/ajpheart.2001.281.3.H1137. [DOI] [PubMed] [Google Scholar]
- 90.Belke DD, Dillmann WH. Altered cardiac calcium handling in diabetes. Curr Hypertens Rep. 2004;6:424–429. doi: 10.1007/s11906-004-0035-3. doi:10.1007/s11906-004-0035-3. [DOI] [PubMed] [Google Scholar]
- 91.Dhalla NS, Liu X, Panagia V, Takeda N. Subcellular remodeling and heart dysfunction in chronic diabetes. Cardiovasc Res. 1998;40:239–247. doi: 10.1016/s0008-6363(98)00186-2. doi:10.1016/S0008-6363(98)00186-2. [DOI] [PubMed] [Google Scholar]
- 92.Xu KY, Zweier JL, Becker LC. Functional coupling between glycolysis and sarcoplasmic reticulum Ca2+ transport. Circulation research. 1995;77:88–97. doi: 10.1161/01.res.77.1.88. [DOI] [PubMed] [Google Scholar]
- 93.Zima AV, Blatter LA. Redox regulation of cardiac calcium channels and transporters. Cardiovasc Res. 2006;71:310–321. doi: 10.1016/j.cardiores.2006.02.019. doi:10.1016/j.cardiores.2006.02.019. [DOI] [PubMed] [Google Scholar]
- 94.Oliveira PJ, Seica R, Coxito PM, Rolo AP, Palmeira CM, Santos MS, et al. Enhanced permeability transition explains the reduced calcium uptake in cardiac mitochondria from streptozotocin-induced diabetic rats. FEBS Lett. 2003;554:511–514. doi: 10.1016/s0014-5793(03)01233-x. doi:10.1016/S0014-5793(03)01233-X. [DOI] [PubMed] [Google Scholar]
- 95.Dong F, Zhang X, Yang X, Esberg LB, Yang H, Zhang Z, et al. Impaired cardiac contractile function in ventricular myocytes from leptin-deficient ob/ob obese mice. J Endocrinol. 2006;188:25–36. doi: 10.1677/joe.1.06241. doi:10.1677/joe.1.06241. [DOI] [PubMed] [Google Scholar]
- 96.Fauconnier J, Lanner JT, Zhang SJ, Tavi P, Bruton JD, Katz A, et al. Insulin and inositol 1,4,5-trisphosphate trigger abnormal cytosolic Ca2+ transients and reveal mitochondrial Ca2+ handling defects in cardiomyocytes of ob/ob mice. Diabetes. 2005;54:2375–2381. doi: 10.2337/diabetes.54.8.2375. doi:10.2337/diabetes.54.8.2375. [DOI] [PubMed] [Google Scholar]
- 97.Belke DD, Swanson EA, Dillmann WH. Decreased sarcoplasmic reticulum activity and contractility in diabetic db/db mouse heart. Diabetes. 2004;53:3201–3208. doi: 10.2337/diabetes.53.12.3201. doi:10.2337/diabetes.53.12.3201. [DOI] [PubMed] [Google Scholar]
- 98.Kelly DP, Scarpulla RC. Transcriptional regulatory circuits controlling mitochondrial biogenesis and function. Genes Dev. 2004;18:357–368. doi: 10.1101/gad.1177604. doi:10.1101/gad.1177604. [DOI] [PubMed] [Google Scholar]
- 99.Wu Z, Puigserver P, Andersson U, Zhang C, Adelmant G, Mootha V, et al. Mechanisms controlling mitochondrial biogenesis and respiration through the thermogenic coactivator PGC-1. Cell. 1999;98:115–124. doi: 10.1016/S0092-8674(00)80611-X. doi:10.1016/S0092-8674(00)80611-X. [DOI] [PubMed] [Google Scholar]
- 100.Lehman JJ, Barger PM, Kovacs A, Saffitz JE, Medeiros DM, Kelly DP. Peroxisome proliferator-activated receptor gamma coactivator-1 promotes cardiac mitochondrial biogenesis. J Clin Invest. 2000;106:847–856. doi: 10.1172/JCI10268. doi:10.1172/JCI10268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Russell LK, Mansfield CM, Lehman JJ, Kovacs A, Courtois M, Saffitz JE, et al. Cardiac-specific induction of the transcriptional coactivator peroxisome proliferator-activated receptor gamma coactivator-1alpha promotes mitochondrial biogenesis and reversible cardiomyopathy in a developmental stage-dependent manner. Circ Res. 2004;94:525–533. doi: 10.1161/01.RES.0000117088.36577.EB. doi:10.1161/01.RES.0000117088.36577.EB. [DOI] [PubMed] [Google Scholar]
- 102.Arany Z, He H, Lin J, Hoyer K, Handschin C, Toka O, et al. Transcriptional coactivator PGC-1 alpha controls the energy state and contractile function of cardiac muscle. Cell Metab. 2005;1:259–271. doi: 10.1016/j.cmet.2005.03.002. doi:10.1016/j.cmet.2005.03.002. [DOI] [PubMed] [Google Scholar]
- 103.Boudina S, Abel ED. Mitochondrial uncoupling: a key contributor to reduced cardiac efficiency in diabetes. Physiology (Bethesda) 2006;21:250–258. doi: 10.1152/physiol.00008.2006. [DOI] [PubMed] [Google Scholar]
- 104.Levine B, Kroemer G. Autophagy in the pathogenesis of disease. Cell. 2008;132:27–42. doi: 10.1016/j.cell.2007.12.018. doi:10.1016/j.cell.2007.12.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Nishida K, Yamaguchi O, Otsu K. Crosstalk between autophagy and apoptosis in heart disease. Circ Res. 2008;103:343–351. doi: 10.1161/CIRCRESAHA.108.175448. doi:10.1161/CIRCRESAHA.108.175448. [DOI] [PubMed] [Google Scholar]
- 106.Turko IV, Murad F. Quantitative protein profiling in heart mitochondria from diabetic rats. J Biol Chem. 2003;278:35844–35849. doi: 10.1074/jbc.M303139200. doi:10.1074/jbc.M303139200. [DOI] [PubMed] [Google Scholar]
- 107.Hamblin M, Friedman DB, Hill S, Caprioli RM, Smith HM, Hill MF. Alterations in the diabetic myocardial proteome coupled with increased myocardial oxidative stress underlies diabetic cardiomyopathy. J Mol Cell Cardiol. 2007;42:884–895. doi: 10.1016/j.yjmcc.2006.12.018. doi:10.1016/j.yjmcc.2006.12.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Hu Y, Suarez J, Fricovsky E, Wang H, Scott BT, Trauger SA, et al. Increased enzymatic O-GlcNAcylation of mitochondrial proteins impairs mitochondrial function in cardiac myocytes exposed to high glucose. J Biol Chem. 2009;284:547–555. doi: 10.1074/jbc.M808518200. doi:10.1074/jbc.M808518200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Belke DD, Betuing S, Tuttle MJ, Graveleau C, Young ME, Pham M, et al. Insulin signaling coordinately regulates cardiac size, metabolism, and contractile protein isoform expression. J Clin Invest. 2002;109:629–639. doi: 10.1172/JCI13946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Graveleau C, Zaha VG, Mohajer A, Banerjee RR, Dudley-Rucker N, Steppan CM, et al. Mouse and human resistins impair glucose transport in primary mouse cardiomyocytes, and oligomerization is required for this biological action. J Biol Chem. 2005;280:31679–31685. doi: 10.1074/jbc.M504008200. doi:10.1074/jbc.M504008200. [DOI] [PubMed] [Google Scholar]
- 111.Shibata R, Ouchi N, Ito M, Kihara S, Shiojima I, Pimentel DR, et al. Adiponectin-mediated modulation of hypertrophic signals in the heart. Nat Med. 2004;10:1384–1389. doi: 10.1038/nm1137. doi:10.1038/nm1137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Shibata R, Sato K, Pimentel DR, Takemura Y, Kihara S, Ohashi K, et al. Adiponectin protects against myocardial ischemia-reperfusion injury through AMPK- and COX-2-dependent mechanisms. Nat Med. 2005;11:1096–1103. doi: 10.1038/nm1295. doi:10.1038/nm1295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Arita Y, Kihara S, Ouchi N, Takahashi M, Maeda K, Miyagawa J, et al. Paradoxical decrease of an adipose-specific protein, adiponectin, in obesity. Biochem Biophys Res Commun. 1999;257:79–83. doi: 10.1006/bbrc.1999.0255. doi:10.1006/bbrc.1999.0255. [DOI] [PubMed] [Google Scholar]
- 114.Berg AH, Combs TP, Du X, Brownlee M, Scherer PE. The adipocyte-secreted protein Acrp30 enhances hepatic insulin action. Nat Med. 2001;7:947–953. doi: 10.1038/90992. doi:10.1038/90992. [DOI] [PubMed] [Google Scholar]
- 115.Yamauchi T, Kamon J, Waki H, Terauchi Y, Kubota N, Hara K, et al. The fat-derived hormone adiponectin reverses insulin resistance associated with both lipoatrophy and obesity. Nat Med. 2001;7:941–946. doi: 10.1038/90984. doi:10.1038/90984. [DOI] [PubMed] [Google Scholar]
- 116.Civitarese AE, Ukropcova B, Carling S, Hulver M, DeFronzo RA, Mandarino L, et al. Role of adiponectin in human skeletal muscle bioenergetics. Cell Metab. 2006;4:75–87. doi: 10.1016/j.cmet.2006.05.002. doi:10.1016/j.cmet.2006.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Iwabu M, Yamauchi T, Okada-Iwabu M, Sato K, Nakagawa T, Funata M, et al. Adiponectin and AdipoR1 regulate PGC-1alpha and mitochondria by Ca(2+) and AMPK/SIRT1. Nature. 2010;464:1313–1319. doi: 10.1038/nature08991. doi:10.1038/nature08991. [DOI] [PubMed] [Google Scholar]
- 118.Dabkowski ER, Williamson CL, Bukowski VC, Chapman RS, Leonard SS, Peer CJ, et al. Diabetic cardiomyopathy-associated dysfunction in spatially distinct mitochondrial subpopulations. Am J Physiol Heart Circ Physiol. 2009;296:H359–H369. doi: 10.1152/ajpheart.00467.2008. doi:10.1152/ajpheart.00467.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Miki T, Miura T, Hotta H, Tanno M, Yano T, Sato T, et al. Endoplasmic reticulum stress in diabetic hearts abolishes erythropoietin-induced myocardial protection by impairment of phospho-glycogen synthase kinase-3beta-mediated suppression of mitochondrial permeability transition. Diabetes. 2009;58:2863–2872. doi: 10.2337/db09-0158. doi:10.2337/db09-0158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Yu T, Sheu SS, Robotham JL, Yoon Y. Mitochondrial fission mediates high glucose-induced cell death through elevated production of reactive oxygen species. Cardiovasc Res. 2008;79:341–351. doi: 10.1093/cvr/cvn104. doi:10.1093/cvr/cvn104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Kajstura J, Fiordaliso F, Andreoli AM, Li B, Chimenti S, Medow MS, et al. IGF-1 overexpression inhibits the development of diabetic cardiomyopathy and angiotensin II-mediated oxidative stress. Diabetes. 2001;50:1414–1424. doi: 10.2337/diabetes.50.6.1414. doi:10.2337/diabetes.50.6.1414. [DOI] [PubMed] [Google Scholar]
- 122.Nielsen LB, Bartels ED, Bollano E. Overexpression of apolipoprotein B in the heart impedes cardiac triglyceride accumulation and development of cardiac dysfunction in diabetic mice. J Biol Chem. 2002;277:27014–27020. doi: 10.1074/jbc.M203458200. doi:10.1074/jbc.M203458200. [DOI] [PubMed] [Google Scholar]
- 123.Singh VP, Le B, Khode R, Baker KM, Kumar R. Intracellular angiotensin II production in diabetic rats is correlated with cardiomyocyte apoptosis, oxidative stress, and cardiac fibrosis. Diabetes. 2008;57:3297–3306. doi: 10.2337/db08-0805. doi:10.2337/db08-0805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Wold LE, Ren J. Streptozotocin directly impairs cardiac contractile function in isolated ventricular myocytes via a p38 map kinase-dependent oxidative stress mechanism. Biochem Biophys Res Commun. 2004;318:1066–1071. doi: 10.1016/j.bbrc.2004.04.138. doi:10.1016/j.bbrc.2004.04.138. [DOI] [PubMed] [Google Scholar]
- 125.Suarez J, Scott B, Dillmann WH. Conditional increase in SERCA2a protein is able to reverse contractile dysfunction and abnormal calcium flux in established diabetic cardiomyopathy. Am J Physiol Regul Integr Comp Physiol. 2008;295:R1439–R1445. doi: 10.1152/ajpregu.00736.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Zhao XY, Hu SJ, Li J, Mou Y, Chen BP, Xia Q. Decreased cardiac sarcoplasmic reticulum Ca2+-ATPase activity contributes to cardiac dysfunction in streptozotocin-induced diabetic rats. J Physiol Biochem. 2006;62:1–8. doi: 10.1007/BF03165800. doi:10.1007/BF03165800. [DOI] [PubMed] [Google Scholar]
- 127.Golfman LS, Wilson CR, Sharma S, Burgmaier M, Young ME, Guthrie PH, et al. Activation of PPARgamma enhances myocardial glucose oxidation and improves contractile function in isolated working hearts of ZDF rats. Am J Physiol Endocrinol Metab. 2005;289:E328–E336. doi: 10.1152/ajpendo.00055.2005. doi:10.1152/ajpendo.00055.2005. [DOI] [PubMed] [Google Scholar]
- 128.Chandler MP, Morgan EE, McElfresh TA, Kung TA, Rennison JH, Hoit BD, et al. Heart failure progression is accelerated following myocardial infarction in type 2 diabetic rats. Am J Physiol Heart Circ Physiol. 2007;293:H1609–H1616. doi: 10.1152/ajpheart.01338.2006. doi:10.1152/ajpheart.01338.2006. [DOI] [PubMed] [Google Scholar]
- 129.Iltis I, Kober F, Desrois M, Dalmasso C, Lan C, Portha B, et al. Defective myocardial blood flow and altered function of the left ventricle in type 2 diabetic rats: a noninvasive in vivo study using perfusion and cine magnetic resonance imaging. Invest Radiol. 2005;40:19–26. [PubMed] [Google Scholar]
- 130.Darmellah A, Baetz D, Prunier F, Tamareille S, Rucker-Martin C, Feuvray D. Enhanced activity of the myocardial Na+/H+ exchanger contributes to left ventricular hypertrophy in the Goto-Kakizaki rat model of type 2 diabetes: critical role of Akt. Diabetologia. 2007;50:1335–1344. doi: 10.1007/s00125-007-0628-x. doi:10.1007/s00125-007-0628-x. [DOI] [PubMed] [Google Scholar]
- 131.Finck BN, Han X, Courtois M, Aimond F, Nerbonne JM, Kovacs A, et al. A critical role for PPARalpha-mediated lipotoxicity in the pathogenesis of diabetic cardiomyopathy: modulation by dietary fat content. Proc Natl Acad Sci USA. 2003;100:1226–1231. doi: 10.1073/pnas.0336724100. doi:10.1073/pnas.0336724100. [DOI] [PMC free article] [PubMed] [Google Scholar]