Abstract
Rapamycin, a potent immunosuppressive drug that disrupts normal signal-transduction processes, inhibited hepatocyte proliferation without evidence of inherent cytotoxicity in rat hepatocytes cultured in conventional medium or in a medium enriched with epidermal growth factor. The antiproliferative effect was dose dependent, uninfluenced by the concentration of epidermal growth factor in the medium and long lasting after a brief exposure. The effect of rapamycin was unaltered by the concomitant presence of FK 506 in the medium, suggesting that different binding affinities of these two drugs or even a separate rapamycin binding site may exist. Hepatocytes harvested 12 and 24 hr after partial hepatectomy were progressively less responsive to the antiproliferative effect of rapamycin. The gene expression of transforming growth factor-β was reduced under in vivo rapamycin treatment, but at the same time the gene expression of albumin and glyceraldehyde-3-phosphate dehydrogenase was unchanged or increased. The experiments confirm that rapamycin has inherent growth-control qualities, and they strengthen the hypothesis that the recently defined immunophilin network is central to many aspects of cellular growth control.
In both rats and dogs, the immunosuppressive drugs cyclosporine (CYA) and FK 506 augment liver regeneration (1–4) and possess other hepatotrophic qualities (5, 6). In contrast, rapamycin (RPM), a powerful immunosuppressant that is chemically related to FK 506 but targeted to a different stage of T-cell activation (7, 8), was recently shown to have antiproliferative properties, including inhibition of regeneration of the liver and of the intestine and kidney (9). We report here studies on the antiproliferative action of RPM on cultured rat hepatocytes in conventional medium, on hepatocytes in epidermal growth factor (EGF)–enriched medium and on hepatocytes harvested at different phases of the regeneration response. We also describe a selective reduction by RPM on the gene expression of transforming growth factor-β (TGF-β). The expression of albumin and glyceraldehyde-3-phosphate dehydrogenase (GAP) genes was unaltered or increased.
MATERIALS AND METHODS
Animals
Hepatocytes for culture were harvested from regenerating or nonregenerating livers of male Fischer F344 rats weighing between 180 and 200 gm (Hilltop Lab Animals, Inc., Scottdale, PA). Seventy-percent hepatectomy was performed as described by Higgins and Anderson (10). The animals were housed in a temperature- and light (6 AM to 6 pM)–controlled room and received food and water ad libitum. All surgical procedures were performed between 8 and 10 AM. Injections were made intraperitoneally.
Materials
Collagenase (type I) (140 to 170 units/mg) was purchased from Worthington Diagnostic Systems (Freehold NJ). Eagles’ MEM and FCS were purchased from GIBCO Laboratories (Grand Island, NY); insulin, HEPES and pyruvic acid were from Sigma Chemical Co. (St. Louis, MO). EGF was from Collaborative Research, Inc. (Bedford, MA); [methyl-3H]thymidine (50 to 80 Ci/mmol) was from Du Pont–New England Nuclear (Boston, MA). Gentamicin was from Elkins-Sinn, Inc. (Cherry Hill, NJ); and scintillant (Aquasol) was from Amersham Corp. (Arlington Heights, IL). RPM was a gift from Dr. Joseph Chang (Wyeth-Ayerst Research, Princeton, NJ). FK 506 was a gift from Fujisawa Pharmaceutical Company Ltd. (Osaka, Japan). CYA was a gift from Sandoz Pharmaceuticals, Inc. (East Hanover, NJ).
Hepatocytes in Primary Culture
Livers were removed from previously unaltered 7-wk-old male rats weighing between 180 and 200 gm or 12 to 24 hr after 70% hepatectomy in some of these animals. Hepatocytes were isolated by a modification (11) of the in situ two-step collagenase perfusion technique of Seglen (12) modified by Jirtle et al. (13). The hepatocytes were dispersed and washed twice with cold Ca+ + -free perfusion buffer and resuspended in basal medium (MEM) supplemented with pyruvate (1 mmol/L), proline (0.26 mmol/L), insulin (10−7 mol/L) and 5% FCS. Viability was determined by trypan blue exclusion, and only preparations with viability greater than 90% at the outset were used. Cell number was determined with a hemocytometer.
The cells were plated at a cell density of 6.5 × 104/well in a Corning 35-mm tissue culture dish (Corning, Inc., Corning, NY) containing 1.5 ml medium and maintained at 37°C in a 5% CO2 atmosphere. After a 3-hr attachment period the medium was aspirated, and 1.5 ml MEM with 5% normal rat serum was added to maintain RPM in solution. EGF and insulin were present in concentrations of 10 ng/ml and 10−7 mol/L, respectively. RPM, FK 506 and CYA were dissolved in alcohol and added in the appropriate concentrations. The amount of alcohol added to the medium was 1 μl/ml; it did not affect hepatocyte proliferation.
In Vitro [3H]thymidine Incorporation
To determine in vitro DNA synthesis, 3 μCi [3H]thymidine was added to each well and maintained from 24 to 48 hr of the culture period. When the cells were harvested, DNA content was determined by the microfluorometric method of Setara and Morley (14), and DNA synthesis was measured by the method of Michalopoulos et al. (15).
Autoradiography
For autoradiographic studies, hepatocytes were plated in 35-mm dishes as previously described (11, 16). [3H]thymidine, 7.5 μCi/dish, was present for 24 hr before fixation. Fixed cells in the dishes were covered with emulsion (Kodak NTB3), and the dishes were developed after 10 days. The labeling index was determined by counting a total of 1,000 cells/dish.
Gene-expression Studies
Total cellular RNA from rat livers or from primary cultures of rat hepatocytes was extracted by RNAzol using the manufacturer’s procedure (Biotecx, Houston, TX). Total RNA (20 μg) was subjected to electrophoresis in a 1% agarose gel in 10 mmol/L Na3PO4 with constant recirculation. The fractionated RNA was transferred to Zitabind nylon membrane (Whatman, Maidstone, UK) overnight in 20 × standard saline citrate. After transfer, the blot was fixed by UV light (short wave, 254 nm). cDNA probes were labeled with 32p with a random-primed labeling kit (Boehringer, Indianapolis, IN). Prehybridization was performed at 70°C for 30 min to 3 hr with Church buffer (1 % BSA, 7% SDS, 0.5 mol/L sodium phosphate, 1 mmol/L EDTA); then 32P-labeled, denatured probes in Church buffer were added at 70°C for hybridization (17). After hybridization, the membranes were washed twice in buffer A (1% BSA, 5% SDS, 40 mmol/L sodium phosphate and 1 mmol/L EDTA), at 70°C for 20 min and then washed in buffer B (1% SDS, 40 mmol/L sodium phosphate and 1 mmol/L EDT A) at 70°C for 20 min (four times). The washed, air-dried membranes were autoradiographed at −70° C. To check for equivalent transfer of RNAs from agarose gel to membrane, the gel was stained with ethidium bromide after capillary transfer; little RNA was found to remain in any lane. Before probe hybridization, the membranes were checked in UV light; similar amounts of RNA were found to have been transferred to each lane. The probes used were TGF-β1 (gift from R. Derynk; Genentech, San Francisco, CA), rat albumin (gift from T. Sargent, NIH) and glyceraldehyde-3-phosphate dehydrogenase (GAP) (gift of Dr. S. Thorgeirsson, NIH). For FK binding protein/RPM binding protein gene transcripts, RNA was reverse-transcribed by oligonucleotide primers (primers from R. F. Standaert and S. L. Schreiber, Department of Chemistry, Harvard University, [18]), and the resulting complementary DNA was amplified by PCR. The products were subjected to electrophoresis on a 2% agarose gel with molecular weight markers.
Other Methods and Statistical Analysis
Proteins were determined by the method of Lowry et al. (19). An unpaired, one-tailed Student’s t test was used for statistical analysis of the data.
RESULTS
Hepatocytes From Resting Liver
Hepatocyte Viability in Resting Liver
More than 95% of the control hepatocytes were alive 24 and 48 hr after the 3-hr attachment period, a viability that was not affected by any dose of RPM. Nor were there changes in the hepatocyte DNA content or ALT concentration in the culture medium (Table 1).
Table 1.
Viability, DNA concentration and transaminase levels of hepatocytes cultured in serum-free medium containing RPM
| RPM dose | 24 hr |
48 hr |
|||
|---|---|---|---|---|---|
| Viability percentage | DNA(μg/dish) | Viability percentage | DNA(μg/dish) | ALT (U/ml) | |
| Control | 95 ± 4a | 3.2 ± 0.2 | 96 ± 3 | 3.3 ± 0.2 | 10 ± 2 |
| RPM | |||||
| 0.4 ng/ml | 94 ± 3 | 3.3 ± 0.3 | 96 ± 4 | 3.3 ± 0.2 | 11 ± 4 |
| 4 ng/ml | 96 ± 4 | 3.2 ± 0.3 | 95 ± 5 | 3.2 ± 0.3 | 10 ± 3 |
| 10 ng/ml | 94 ± 3 | 3.3 ± 0.3 | 97 ± 3 | 3.3 ± 0.2 | 11 ± 2 |
Data expressed as the average from nine dishes and three different animals (mean ± S.D.).
Hepatocyte Proliferation
DNA synthesis was depressed by RPM in a dose-related manner, an effect not seen with FK 506 or CYA (Fig. 1, bottom).
Fig. 1.
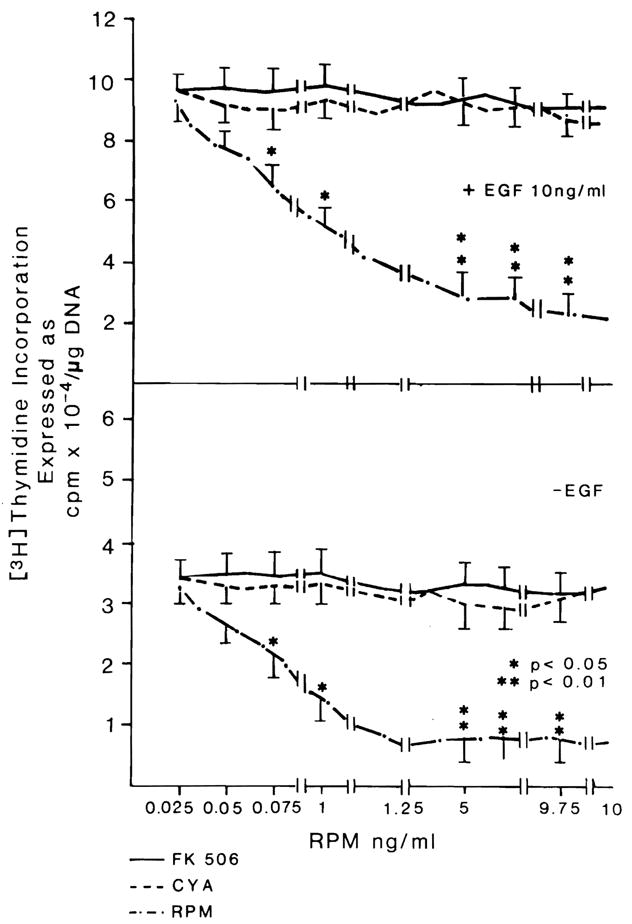
Effect of different doses of FK 506, CYA and RPM on DNA synthesis in hepatocytes cultured in the presence (top) or absence (bottom) of EGF. FK506 and CYA were previously shown to be inactive in the system at 10 times the maximum doses used for these experiments (24). n = 9 for all data points, which represent three experiments with triplicate determinations. Data expressed as mean ± S.D.
Comparable results were obtained with higher-baseline DNA synthesis, which occurs after enrichment of the medium with EGF (Fig. 1, top). The results with RPM were confirmed by autoradiography (Table 2). For any given dose of RPM, the degree of DNA synthesis depression was similar no matter what the amount of EGF added to the medium between 10 and 40 ng/ml (Fig. 2). In experiments in which washing and medium change terminated the briefer exposures to RPM, the inhibitory effect of RPM was the same whether contact with RPM was for the entire 48 hr of culture or for intervals as short as the first 3 hr (Fig. 3).
Table 2.
DNA concentration and labeling index of hepatocytes cultured in the presence of different concentrations of RPM
| RPM dose | DNA (μg/dish) | Labeled nuclei (%) |
|---|---|---|
| Control | 6.1 + 1.3a | 10 + 1.5 |
| RPM | ||
| 10 ng/ml | 3.3 + 0.2b | 5.8 + 0.9c |
| 2.5 ng/ml | 3.3 + 0.4b | 6.1 + 0.8b |
| 1 ng/ml | 3.7 + 0.3b | 6.5 + 0.6b |
| 0.25 ng/ml | 4.2 + 0.35 | 7.1 + 0.8 |
Data are average from six dishes for each RPM concentration. Medium was enriched with 5% normal rat serum.
p < 0.05 vs. control.
p < 0.01 vs. control.
Fig. 2.
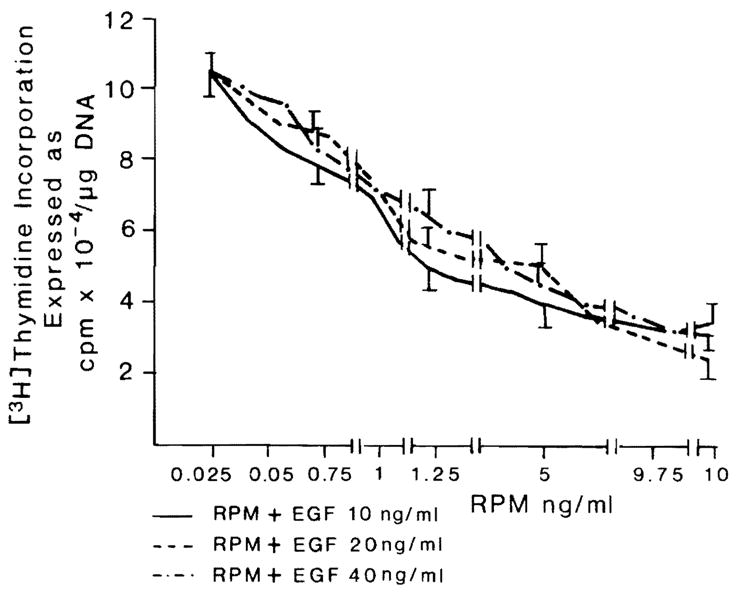
Effect of different doses of EGF on hepatocyte proliferation in the presence of different concentrations of RPM. n = 9 for all data points. Data expressed as mean ± S.D.
Fig. 3.
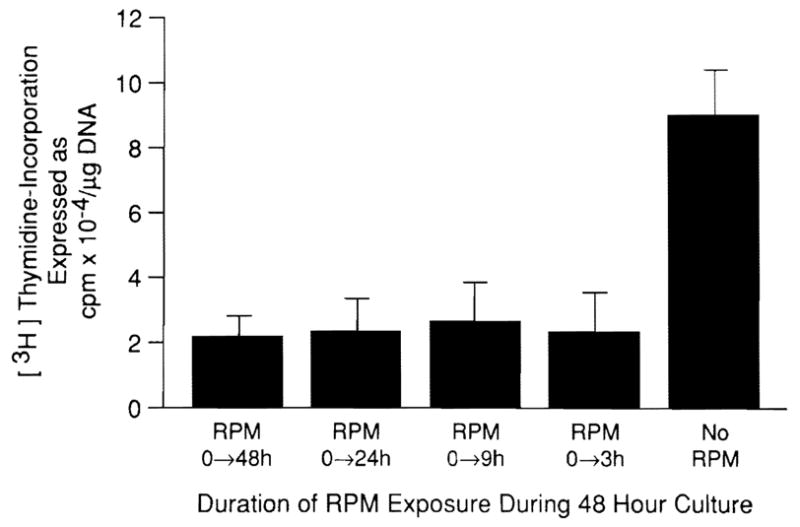
Effect of the time of RPM exposure on hepatocyte proliferation in medium enriched with 10 ng/ml EGF. The dish contained 5 ng/ml RPM at zero time; at the end of RPM exposure, the medium was removed and after three washes was replaced with RPM-free medium. n = 9 for all data points. Data expressed as mean ± S.D.
The inhibitory effect of RPM was not changed by adding FK 506, even with FK 506 doses approximately 4,000 times greater than those of RPM (Fig. 4).
Fig. 4.
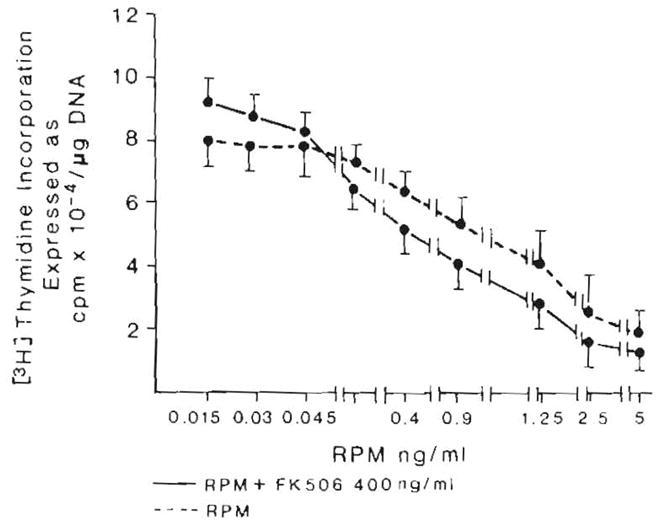
Effect of FK 506 on the inhibition induced by RPM. n = 9 for all data points. Data expressed as mean ± S.D.
Hepatocytes From Regenerating Liver
Hepatocytes harvested 12 and 24 hr after 70% hepatectomy and cultured in EGF-enriched medium were responsive to RPM inhibition but far less so than hepatocytes from normal rats (Fig. 5). RPM resistance was more pronounced in hepatocytes from rats 24 hr after partial hepatectomy than in those harvested 12 hr after hepatectomy. Inhibition of DNA synthesis could be seen only with the highest RMP dose of 5 ng/ml.
Fig 5.
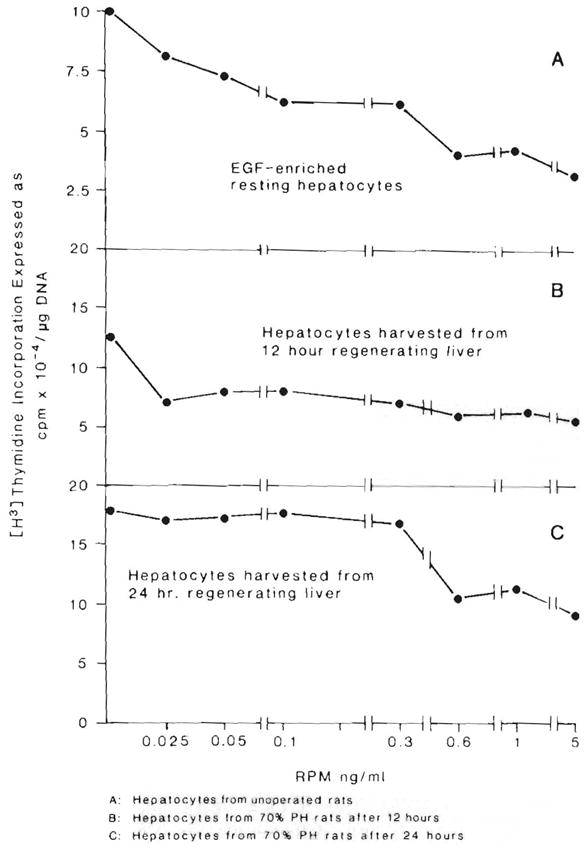
RPM inhibition of DNA synthesis in EGF-enriched control hepatocytes (A) compared with that in 12-hour (B) and 24-hour (C) regenerating hepatocytes cultured in EGF-enriched medium. The control animal data is the same shown in Figure 1 (top).
The relative resistance to the inhibitory effects of RPM by hepatocytes from regenerating liver compared with hepatocytes from normal liver could be due to differences in the ability to bind RPM because both FK 506 and RPM have been reported to bind to FK/RPM binding protein (FKBP). This possibility was weakened by the absence of any substantial decrease in the messenger RNA amounts for FKBP in hepatocytes from regenerating liver compared with resting liver (Fig. 6). A small increase in FKBP mRNA is shown in the gel in hepatocytes from regenerating liver. We think that this is not significant, since various dilutions of resting and regenerating hepatocyte RNA’s (data not shown) revealed essentially no differences in FKBP mRNA amounts.
Fig 6.
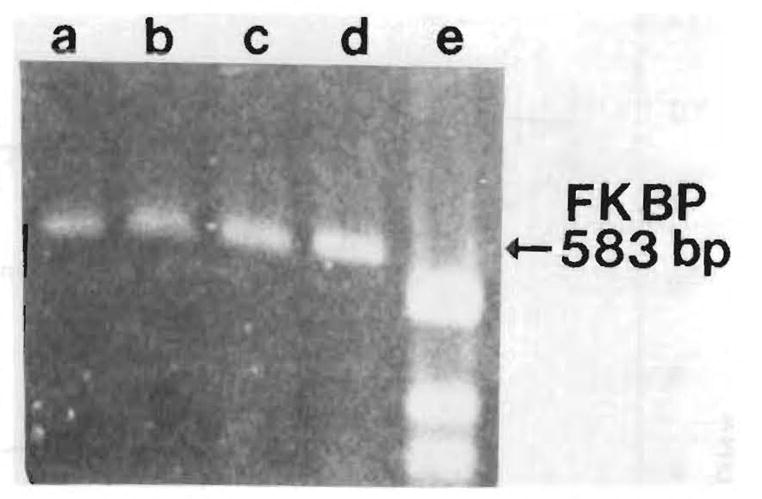
Expression of FKBP mRNA by reverse-transcription polymerase chain reaction. Lanes a and b are hepatocytes from normal liver; lanes c and d are hepatocytes from regenerating liver; lane e shows molecular-weight markers.
Changes in Gene Expression In Vivo
Two rats each were treated for 5, 4, 3, 2, and 1 day with 0.1 mg/kg/day rapamycin intramuscularly and killed for RNA extraction. The TGF-β mRNA, which normally is found in low levels in resting liver, was suppressed completely after 4 days of therapy (Fig. 7) in resting rats. The mRNA levels of the two nongrowth genes, albumin (Fig. 8) and GAP (Fig. 9), had different changes. Albumin mRNA initially showed a slight decrease but then increased again. GAP mRNA actually increased at the time of minimum TGF-β mRNA expression.
Fig 7.

Northern blot of TGF-β1 mRNA levels from livers of resting rats treated with RPM for 0, 1, 2, 3, 4 or 5 days.
Fig 8.

Northern blot of rat albumin mRNA levels. RNA samples are identical to those of Figure 7.
Fig 9.
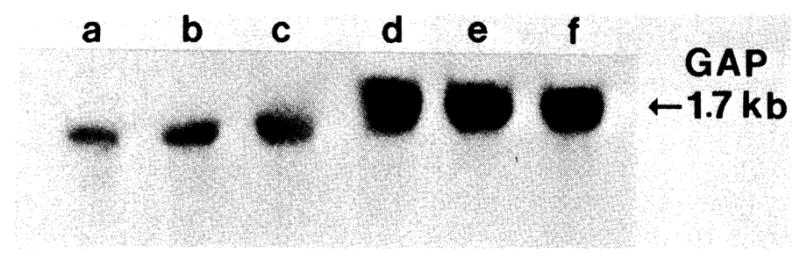
Northern blot of GAP mRNA levels. RNA samples are identical to those of Figure 7.
Two additional rats underwent 70% hepatectomy after 5 days of RPM treatment. The TGF-β mRNA transcripts failed to increase after the partial hepatectomy (lanes a and b, Fig. 10), in contrast to the sharp increase in two saline solution–treated hepatectomy controls.
Fig 10.
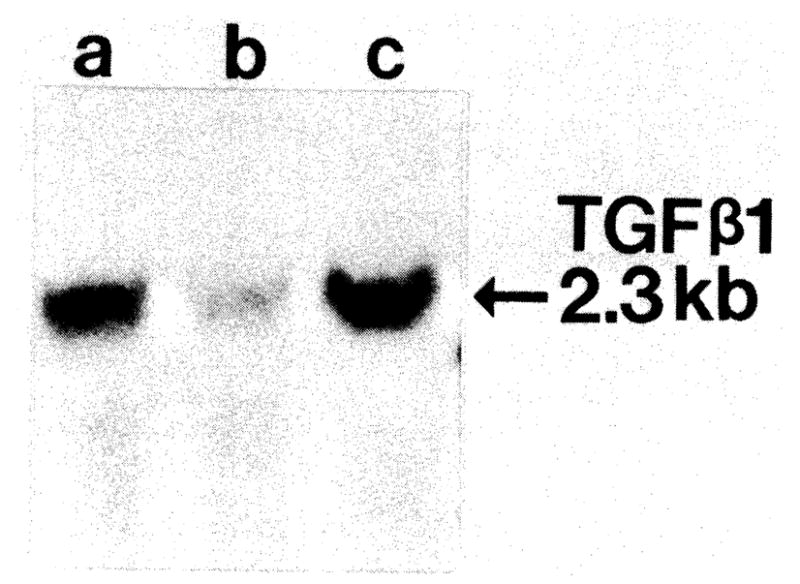
Northern blot of TGF-β1 mRNA levels from livers after partial hepatectomy (48 hr) treated without (lane a) or with (lane b) RPM. Lane c is RNA from nonparenchymal liver cells (positive control).
DISCUSSION
The immunosuppression of CYA, FK 506 and RPM is caused by variable modification of signal transduction in immune cells (7, 8, 20). CYA and FK 506 inhibit the transcription of early T-cell activation genes, including interleukin-2 (21, 22), whereas RPM blocks downstream events resulting from T-cell activation (7, 8). The reason for this difference in drug action is not known. The binding sites of CYA (cyclophilin) and FK 506 (FKBP) are different low-molecular-weight proteins (23, 24).
Although RPM was initially thought to bind to FKBP, recent genetic evidence in yeast experiments has suggested that a separate binding protein (RBP) closely related to FKBP may also exist (25). The demonstration herein reported of an absence of competitive action in our in vitro growth-control system is compatible with this possibility but does not prove it. Other unpublished observations are consistent with the existence of a binding protein with a different molecular weight and binding affinity than FKBP. In the Harvard laboratories (Albers MW et al., Unpublished observation, 1991), a 30-kD protein called FKBP30, which has a sequence related to that of the original FKBP (called FKBP12) has been shown to bind RPM more than 100 times more selectively than FK 506.
Collectively, these ubiquitous binding proteins–which have in common enzyme peptidyl-prolyl isomerase (PPIase) activity (23, 24)–have been conceptualized as an immunophilin network with potential pleiotropic activities not limited to those affecting the immune system (6,18,26). Whether PPIase is central to the function of the immunophilin system or merely a marker of it is unresolved.
The possibility that CYA, FK 506 or both alter immunologically independent signaling pathways that affect growth was raised by in vivo experiments in rats and dogs showing that both drugs augment liver regeneration and have other hepatotrophic actions (6, 24). Further experiments showed that this augmentation was not related to T-cell or natural killer cell deficiency (27). The demonstration in intact rats of an opposite (antiproliferative) effect of RPM with reduction or abolition of liver, kidney and intestinal regeneration was of unusual interest because it was the first example of down-regulation by a drug of this physiological manifestation of the immunophilin system (9).
The in vitro observations reported herein support our previous conclusion from in vivo experiments that RPM is a growth inhibitor (9). The ease with which growth regulation by RPM could be detected in hepatocyte cultures was in contrast to the previous difficulty of demonstrating the stimulation of isolated hepatocytes either by CYA or FK 506 (28), which had been anticipated from the results of in vivo experiments with these drugs. Immune modulation as the primary explanation of the RPM effect was greatly weakened in our current experiments because more than 99% of the cells in our cultures have been shown to be hepatocytes with liver-specific enzymes (11, 15). However, because the cells were plated in 5% FCS and grown in 5% normal serum, it can not be excluded that RPM prevented the trophic action of a factor(s) released into the sera by immunocytes. The RPM inhibition of proliferation of either resting hepatocytes or EGF-driven hepatocytes was unambiguously demonstrated over a wide dose range.
Brief exposure to RPM had as profound an effect on nonregenerating hepatocytes during the succeeding 2 days as continuous exposure for the same period. Proliferation was harder to suppress in cultured hepatocytes committed to regeneration by prior partial hepatectomy, but even 24 hr after hepatectomy these hepatocytes (which were at the peak of the regeneration response) could be partially inhibited with high RPM doses. Growth inhibition was not due to inherent hepatotoxicity because cell viability was not jeopardized in the conditions of our culture model over the full 48-hr testing period. In addition, the gene expression of albumin and GAP were unaltered or actually enhanced.
Further investigations will be required to establish the molecular basis for the growth-regulatory qualities of these immunosuppressive drugs and their interactions with other putative growth factors. Our finding of a profound decrease in the expression of TGF-β, but not in that of non-growth-associated genes such as albumin or GAP, suggests that the inhibitory actions of RPM on liver growth are intricately associated with liver-growth gene expression, although the mechanism of this effect is not clear.
TGF-β is a potent bifunctional growth-regulating peptide that can stimulate fibroblast growth, but inhibits the growth of most epithelial cells, including hepatocytes in vitro and in vivo (17, 29, 30). We postulate that the action of an externally administered inhibitor of growth, such as RPM, may alter the requirement for an endogenous putative inhibitor, such as TGF-β, and might explain our findings and constitute an example of intracellular homeostasis at the level of gene expression.
It now seems possible that the alterations in signal transduction that have been caused in immune cells by FK 506 and other drugs may play an analogous role in nonimmunologic functions subserved by the immunophilin network. The possibility that biologically active complexes are formed by ligands and the cytosolic binding proteins has been suggested by the recent demonstration that recombinant FKBP has physiologic growth control activity in vivo (31), presumably after binding with endogenous ligands in these drug-free dogs. Other evidence comes from genetic studies in Saccharomyces cerevisiae that demonstrate that, for example, the complexes of yeast FKBP and RPM or human FKBP and RPM are responsible for mediating the antibiotic actions of RPM in yeast (25).
Acknowledgments
We gratefully acknowledge the influence on this work of S. L. Schreiber and M. W. Albers (Department of Chemistry, Harvard University), particularly in the interpretation of the results.
Aided by Research Grants from the Department of Veterans Affairs and project grants DK 29961 and CA 35373 from the National Institutes of Health.
References
- 1.Makowka L, Svanas G, Esquivel C, Venkataramanan R, Todo S, Iwatsuki S, Van Thiel DH, et al. The effect of cyclosporine on hepatic regeneration. Surg Forum. 1986;37:352–354. [PMC free article] [PubMed] [Google Scholar]
- 2.Kahn D, Lai HS, Romovacek H, Makowka L, Van Thiel DH, Starzl TE. Cyclosporin A augments the regeneration response after partial hepatectomy in the rat. Transplant Proc. 1988;20(suppl 3):850–852. [PMC free article] [PubMed] [Google Scholar]
- 3.Kim YI, Calne RY, Nagasue N. Cyclosporin A stimulates proliferation of the liver cells after partial hepatectomy in rats. Surg Gynecol Obstet. 1988;166:317–311. [PubMed] [Google Scholar]
- 4.Francavilla A, Barone M, Todo S, Zeng Q, Porter KA, Starzl TE. Augmentation of rat liver regeneration by FK 506 compared with cyclosporine. Lancet. 1989;2:1248–1249. doi: 10.1016/s0140-6736(89)91853-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Mazzaferro V, Porter KA, Scotti-Foglieni CL, Venkataramanan R, Makowka L, Rossaro L, Francavilla A, et al. The hepatotrophic influence of cyclosporine. Surgery. 1990;107:533–539. [PMC free article] [PubMed] [Google Scholar]
- 6.Starzl TE, Porter KA, Mazzaferro V, Todo S, Fung J, Francavilla A. Hepatotrophic effects of FK 506 in dogs. Transplantation. 1991;51:67–70. doi: 10.1097/00007890-199101000-00010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Dumont FJ, Staruch MJ, Koprak SL, Melino MR, Sigal NH. Distinct mechanisms of suppression of murine T cell activation by the related macrolides FK 506 and rapamycin. J Immunol. 1990;144:251–258. [PubMed] [Google Scholar]
- 8.Bierer BE, Mattila PS, Standaert RF, Herzenberg LA, Burakoff SJ, Crabtree G, Schreiber SL. Two distinct signal transmission pathways in T lymphocytes are inhibited by complexes formed between an immunophilin and either FK 506 or rapamycin. Proc Natl Acad Sci USA. 1990;87:9231–9235. doi: 10.1073/pnas.87.23.9231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Francavilla A, Starzl TE, Scotti C, Carrieri C, Azzarone A, Zeng DH, Porter KA, et al. Rapamycin inhibits liver, kidney, and intestine regeneration. Transplantation. 1992;53:496–498. doi: 10.1097/00007890-199202010-00045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Higgins GM, Anderson RM. Experimental pathology of liver. I. Restoration of liver of the white rat following partial surgical removal. Arch Pathol. 1931;12:186–202. [Google Scholar]
- 11.Francavilla A, Ove P, Polimeno L, Sciascia C, Coetzee ML, Starzl TE. Epidermal growth factor and proliferation in rat hepatocytes in primary cultures isolated at different times after partial hepatectomy. Cancer Res. 1986;46:1318–1323. [PMC free article] [PubMed] [Google Scholar]
- 12.Seglen PO. Preparation of isolated rat liver cell. Methods Cell Biol. 1976;13:29–83. doi: 10.1016/s0091-679x(08)61797-5. [DOI] [PubMed] [Google Scholar]
- 13.Jirtle RL, Michalopoulos G, McLaine JR, Crowley J. Transplantation system for determining the clonogenic survival of parenchymal hepatocytes exposed to ionizing radiation. Cancer Res. 1981;41:3512–3518. [PubMed] [Google Scholar]
- 14.Setaro F, Morley CGD. A modified fluorimetric method for the determination of microgram quantities of DNA from cell tissue cultures. Anal Biochem. 1976;71:313–317. doi: 10.1016/0003-2697(76)90043-9. [DOI] [PubMed] [Google Scholar]
- 15.Michalopoulos G, Cianciulli HD, Novotny R, Kligerman AD, Strom SC, Jirtle RL. Liver regeneration studies with rat hepatocytes in primary culture. Cancer Res. 1982;42:4673–4680. [PubMed] [Google Scholar]
- 16.Thaler TJ, Michalopoulos GH. Hepatoprotein A: partial characterization and trypsin activation of hepatocyte growth factor. Cancer Res. 1985;45:2545–2549. [PubMed] [Google Scholar]
- 17.Carr BI, Huang TH, Itakura K, Neel M, Marceau N. TGFB gene transcription in normal and neoplastic liver growth. J Cell Biochem. 1989;39:477–487. doi: 10.1002/jcb.240390413. [DOI] [PubMed] [Google Scholar]
- 18.Standaert RF, Galat A, Verdine GL, Schreiber SL. Molecular cloning and overexpression of the human FK506-binding protein FKBP. Nature. 1990;346:671–674. doi: 10.1038/346671a0. [DOI] [PubMed] [Google Scholar]
- 19.Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. Protein measurement with the Folin phenol reagent. J Biol Chern. 1951;193:265–275. [PubMed] [Google Scholar]
- 20.Schreiber SL. Chemistry and biology of the immunophilins and their immunosuppressive ligands. Science. 1991;251:283–287. doi: 10.1126/science.1702904. [DOI] [PubMed] [Google Scholar]
- 21.Tocci MJ, Matkovich DA, Collier KA, Kwok P, Dumont F, Lin S, Degudicibus S, et al. The immunosuppressant FK 506 selectively inhibits expression of early T cell activation genes. J Immunol. 1989;143:718–726. [PubMed] [Google Scholar]
- 22.Sawada S, Suzuki G, Kawase Y, Takaku F. Novel immunosuppressive agent, FK 506: in vitro effects on the cloned T cell activation. J Immunol. 1987;139:1797–1803. [PubMed] [Google Scholar]
- 23.Siekierka JJ, Hung SHY, Poe M, Lin CS, Sigal NH. A cytosolic binding protein for the immunosuppressant FK 506 has peptidyl-prolyl isomerase activity but is distinct from cyclophilin. Nature. 1989;341:755–757. doi: 10.1038/341755a0. [DOI] [PubMed] [Google Scholar]
- 24.Harding MW, Galat A, Uehling DE, Schreiber SL. A receptor for the immunosuppressant FK506 is a cis-trans peptidyl-prolyl isomerase. Nature. 1989;341:758–760. doi: 10.1038/341758a0. [DOI] [PubMed] [Google Scholar]
- 25.Koltin Y, Faucette L, Bergsma DJ, Levy MA, Cafferkey R, Koser PL, Johnson RK, et al. Rapamycin sensitivity in saccharomyces cerevisiae is mediated by a peptidyl-prolyl cis-trans isomerase related to human FK 506-binding protein. Mol Cell Biol. 1991;11:1718–1723. doi: 10.1128/mcb.11.3.1718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Starzl TE, Fung JJ. Contempo 90: transplantation. JAMA. 1990;263:2686–2687. [PMC free article] [PubMed] [Google Scholar]
- 27.Francavilla A, Starzl TE, Barone M, Zeng Q, Porter KA, Zeevi A, Markus P, et al. Studies on mechanisms of augmentation of liver regeneration by cyclosporine and FK 506. Hepatology. 1991;14:140–143. doi: 10.1002/hep.1840140123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Francavilla A, Barone M, Starzl TE, Zeevi A, Scotti C, Carrieri G, Mazzaferro V, et al. FK 506 as a growth control factor. Transplant Proc. 1990;22:90–92. [PMC free article] [PubMed] [Google Scholar]
- 29.Braun L, Mead JE, Panzica M, Mikumo R, Bell GI, Fausto N. Transforming growth factor B mRNA increases during liver regeneration: a possible paracrine mechanism of growth regulation. Proc Natl Acad Sci USA. 1988;85:1539–1543. doi: 10.1073/pnas.85.5.1539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lawrence DA, Pirchen R, Kryceve-Martinerie C, Jullien P. Normal embryo fibroblasts release transforming growth factors in a latent form. J Cell Physiol. 1984;121:184–188. doi: 10.1002/jcp.1041210123. [DOI] [PubMed] [Google Scholar]
- 31.Starzl TE, Schrieber SL, Albers MW, Porter KA, Foglieni CS, Francavilla A. Hepatotrophic properties in dogs of human FKBP, the binding protein for FK 506 and rapamycin. Transplantation. 1991;52:751–753. doi: 10.1097/00007890-199110000-00038. [DOI] [PMC free article] [PubMed] [Google Scholar]


