Abstract
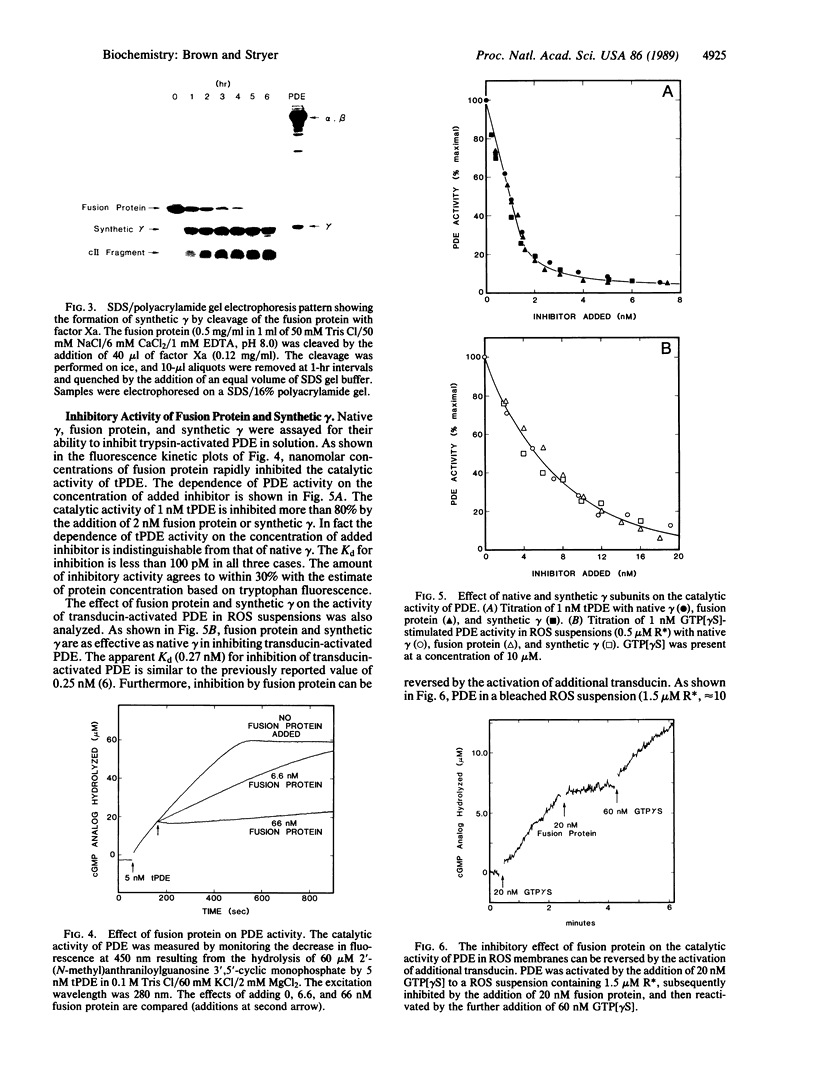
The cGMP phosphodiesterase of vertebrate retinal rod outer segments plays a key role in visual transduction. A functionally active form of the inhibitory gamma subunit of the phosphodiesterase, which keeps the enzyme inactive in the dark, has been obtained in high yield from a synthetic gene expressed in Escherichia coli. A DNA sequence encoding the 87-residue bovine gamma subunit was chemically synthesized and assembled from 10 oligonucleotides. The synthetic gene was cloned into an expression vector that uses the promoter PL of lambda phage. E. coli was transformed with this vector, which encodes a fusion protein consisting of the first 31 residues of the lambda cII protein, a 7-residue joining sequence that is specifically cleaved at its C-terminal end by clotting protease factor Xa, and the 87-residue gamma subunit. The fusion protein was solubilized in 6 M urea and purified by ion-exchange chromatography on a CM-Sephadex column. The typical yield was 1 mg of fusion protein per liter of bacterial culture, which corresponds to the amount of gamma in about 2500 bovine retinas. Proteolytic cleavage of the fusion protein by factor Xa released a synthetic gamma with the same amino acid sequence as that of native gamma. Both fusion protein and synthetic gamma inhibited trypsin-activated phosphodiesterase with high affinity (Kd less than 100 pM). Likewise, both were as effective as native gamma in inhibiting transducin-activated phosphodiesterase in rod outer segment membranes. This inhibition was reversed by the activation of additional transducin. Thus, the N terminus of gamma is not intimately involved in interactions with either the catalytic subunits of the phosphodiesterase or the activated form of transducin. In contrast, a C-terminal deletion mutant terminating at residue 74 of gamma stimulated rather than inhibited the trypsin-activated enzyme. Thus, the C-terminal region of gamma is critical for inhibition of the phosphodiesterase.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Biggin M. D., Gibson T. J., Hong G. F. Buffer gradient gels and 35S label as an aid to rapid DNA sequence determination. Proc Natl Acad Sci U S A. 1983 Jul;80(13):3963–3965. doi: 10.1073/pnas.80.13.3963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deterre P., Bigay J., Forquet F., Robert M., Chabre M. cGMP phosphodiesterase of retinal rods is regulated by two inhibitory subunits. Proc Natl Acad Sci U S A. 1988 Apr;85(8):2424–2428. doi: 10.1073/pnas.85.8.2424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deterre P., Bigay J., Robert M., Pfister C., Kühn H., Chabre M. Activation of retinal rod cyclic GMP-phosphodiesterase by transducin: characterization of the complex formed by phosphodiesterase inhibitor and transducin alpha-subunit. Proteins. 1986 Oct;1(2):188–193. doi: 10.1002/prot.340010210. [DOI] [PubMed] [Google Scholar]
- Ferretti L., Karnik S. S., Khorana H. G., Nassal M., Oprian D. D. Total synthesis of a gene for bovine rhodopsin. Proc Natl Acad Sci U S A. 1986 Feb;83(3):599–603. doi: 10.1073/pnas.83.3.599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fung B. K., Hurley J. B., Stryer L. Flow of information in the light-triggered cyclic nucleotide cascade of vision. Proc Natl Acad Sci U S A. 1981 Jan;78(1):152–156. doi: 10.1073/pnas.78.1.152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hanahan D. Studies on transformation of Escherichia coli with plasmids. J Mol Biol. 1983 Jun 5;166(4):557–580. doi: 10.1016/s0022-2836(83)80284-8. [DOI] [PubMed] [Google Scholar]
- Hurley J. B., Stryer L. Purification and characterization of the gamma regulatory subunit of the cyclic GMP phosphodiesterase from retinal rod outer segments. J Biol Chem. 1982 Sep 25;257(18):11094–11099. [PubMed] [Google Scholar]
- Johnson J. D., Walters J. D., Mills J. S. A continuous fluorescence assay for cyclic nucleotide phosphodiesterase hydrolysis of cyclic GMP. Anal Biochem. 1987 Apr;162(1):291–295. doi: 10.1016/0003-2697(87)90039-x. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Liebman P. A., Evanczuk A. T. Real time assay of rod disk membrane cGMP phosphodiesterase and its controller enzymes. Methods Enzymol. 1982;81:532–542. doi: 10.1016/s0076-6879(82)81074-4. [DOI] [PubMed] [Google Scholar]
- Liebman P. A., Parker K. R., Dratz E. A. The molecular mechanism of visual excitation and its relation to the structure and composition of the rod outer segment. Annu Rev Physiol. 1987;49:765–791. doi: 10.1146/annurev.ph.49.030187.004001. [DOI] [PubMed] [Google Scholar]
- Lipkin V. M., Dumler I. L., Muradov K. G., Artemyev N. O., Etingof R. N. Active sites of the cyclic GMP phosphodiesterase gamma-subunit of retinal rod outer segments. FEBS Lett. 1988 Jul 18;234(2):287–290. doi: 10.1016/0014-5793(88)80100-5. [DOI] [PubMed] [Google Scholar]
- Miki N., Baraban J. M., Keirns J. J., Boyce J. J., Bitensky M. W. Purification and properties of the light-activated cyclic nucleotide phosphodiesterase of rod outer segments. J Biol Chem. 1975 Aug 25;250(16):6320–6327. [PubMed] [Google Scholar]
- Nagai K., Thøgersen H. C. Synthesis and sequence-specific proteolysis of hybrid proteins produced in Escherichia coli. Methods Enzymol. 1987;153:461–481. doi: 10.1016/0076-6879(87)53072-5. [DOI] [PubMed] [Google Scholar]
- Ovchinnikov YuA, Lipkin V. M., Kumarev V. P., Gubanov V. V., Khramtsov N. V., Akhmedov N. B., Zagranichny V. E., Muradov K. G. Cyclic GMP phosphodiesterase from cattle retina. Amino acid sequence of the gamma-subunit and nucleotide sequence of the corresponding cDNA. FEBS Lett. 1986 Aug 18;204(2):288–292. doi: 10.1016/0014-5793(86)80830-4. [DOI] [PubMed] [Google Scholar]
- Stryer L. Cyclic GMP cascade of vision. Annu Rev Neurosci. 1986;9:87–119. doi: 10.1146/annurev.ne.09.030186.000511. [DOI] [PubMed] [Google Scholar]
- Varadarajan R., Szabo A., Boxer S. G. Cloning, expression in Escherichia coli, and reconstitution of human myoglobin. Proc Natl Acad Sci U S A. 1985 Sep;82(17):5681–5684. doi: 10.1073/pnas.82.17.5681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wensel T. G., Stryer L. Reciprocal control of retinal rod cyclic GMP phosphodiesterase by its gamma subunit and transducin. Proteins. 1986 Sep;1(1):90–99. doi: 10.1002/prot.340010114. [DOI] [PubMed] [Google Scholar]
- Zweig M., Showalter S. D., Du Bois G. C., Sisk W. P., Court D. L. Detection of heterologous fusion proteins in Escherichia coli with a monoclonal antibody. Gene. 1987;55(1):47–53. doi: 10.1016/0378-1119(87)90247-2. [DOI] [PubMed] [Google Scholar]