Abstract
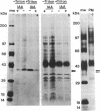
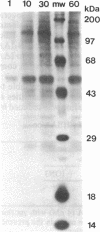
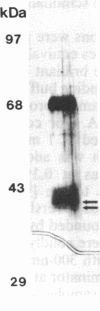
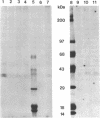
Plasma membrane vesicles were isolated from zucchini (Cucurbita pepo) hypocotyl tissue by aqueous phase partitioning and assessed for homogeneity by the use of membrane-specific enzyme assays. The highly pure (ca. 95%) plasma membrane vesicles maintained a pH differential across the membrane and accumulated a tritiated azido analogue of 3-indoleacetic acid (IAA), 5-azido-[7-3H]IAA ([3H]N3IAA), in a manner similar to the accumulation of [3H]IAA. The association of the [3H]N3IAA with membrane vesicles was saturable and subject to competition by IAA and auxin analogues. Auxin-binding proteins were photoaffinity labeled by addition of [3H]N3IAA to plasma membrane vesicles prior to exposure to UV light (15 sec; 300 nm) and detected by subsequent NaDodSO4/PAGE and fluorography. When the reaction temperature was lowered to - 196°C, high-specific-activity labeling of a 40-kDa and a 42-kDa polypeptide was observed. Triton X-100 (0.1%) increased the specific activity of labeling and reduced the background, which suggests that the labeled polypeptides are intrinsic membrane proteins. The labeled polypeptides are of low abundance, as expected for auxin receptors. Further, the addition of IAA and auxin analogues to the photoaffinity reaction mixture resulted in reduced labeling that was qualitatively similar to their effects on the accumulation of radiolabeled IAA in membrane vesicles. Collectively, these results suggest that the radiolabeled polypeptides are auxin receptors. The covalent nature of the label should facilitate purification and further characterization of the receptors.
Keywords: auxin receptors, aqueous phase partitioning, 3-indoleacetic acid, Cucurbita pepo
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Chowdhry V., Westheimer F. H. Photoaffinity labeling of biological systems. Annu Rev Biochem. 1979;48:293–325. doi: 10.1146/annurev.bi.48.070179.001453. [DOI] [PubMed] [Google Scholar]
- Cross J. W., Briggs W. R. Properties of a Solubilized Microsomal Auxin-binding Protein from Coleoptiles and Primary Leaves of Zea mays. Plant Physiol. 1978 Jul;62(1):152–157. doi: 10.1104/pp.62.1.152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacobs M., Gilbert S. F. Basal localization of the presumptive auxin transport carrier in pea stem cells. Science. 1983 Jun 17;220(4603):1297–1300. doi: 10.1126/science.220.4603.1297. [DOI] [PubMed] [Google Scholar]
- Jones A. M., Melhado L. L., Ho T. H., Leonard N. J. Azido auxins: quantitative binding data in maize. Plant Physiol. 1984 Feb;74(2):295–301. doi: 10.1104/pp.74.2.295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones A. M., Melhado L. L., Ho T. H., Pearce C. J., Leonard N. J. Azido auxins : photoaffinity labeling of auxin-binding proteins in maize coleoptile with tritiated 5-azidoindole-3-acetic Acid. Plant Physiol. 1984 Aug;75(4):1111–1116. doi: 10.1104/pp.75.4.1111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lomax T. L., Conley P. B., Schilling J., Grossman A. R. Isolation and characterization of light-regulated phycobilisome linker polypeptide genes and their transcription as a polycistronic mRNA. J Bacteriol. 1987 Jun;169(6):2675–2684. doi: 10.1128/jb.169.6.2675-2684.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lomax T. L., Mehlhorn R. J., Briggs W. R. Active auxin uptake by zucchini membrane vesicles: quantitation using ESR volume and delta pH determinations. Proc Natl Acad Sci U S A. 1985 Oct;82(19):6541–6545. doi: 10.1073/pnas.82.19.6541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Löbler M., Klämbt D. Auxin-binding protein from coleoptile membranes of corn (Zea mays L.). I. Purification by immunological methods and characterization. J Biol Chem. 1985 Aug 15;260(17):9848–9853. [PubMed] [Google Scholar]
- Melhado L. L., Jones A. M., Leonard N. J., Vanderhoef L. N. Azido auxins: synthesis and biological activity of fluorescent photoaffinity labeling agents. Plant Physiol. 1981 Aug;68(2):469–475. doi: 10.1104/pp.68.2.469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ripp K. G., Viitanen P. V., Hitz W. D., Franceschi V. R. Identification of membrane protein associated with sucrose transport into cells of developing soybean cotyledons. Plant Physiol. 1988 Dec;88(4):1435–1445. doi: 10.1104/pp.88.4.1435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sandstrom R. P., Deboer A. H., Lomax T. L., Cleland R. E. Latency of Plasma Membrane H-ATPase in Vesicles Isolated by Aqueous Phase Partitioning : Increased substrate Accessibility or Enzyme Activation. Plant Physiol. 1987 Nov;85(3):693–698. doi: 10.1104/pp.85.3.693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimomura S., Sotobayashi T., Futai M., Fukui T. Purification and properties of an auxin-binding protein from maize shoot membranes. J Biochem. 1986 May;99(5):1513–1524. doi: 10.1093/oxfordjournals.jbchem.a135621. [DOI] [PubMed] [Google Scholar]
- Spector T. Refinement of the coomassie blue method of protein quantitation. A simple and linear spectrophotometric assay for less than or equal to 0.5 to 50 microgram of protein. Anal Biochem. 1978 May;86(1):142–146. doi: 10.1016/0003-2697(78)90327-5. [DOI] [PubMed] [Google Scholar]
- Tolbert N. E. Isolation of subcellular organelles of metabolism on isopycnic sucrose gradients. Methods Enzymol. 1974;31:734–746. doi: 10.1016/0076-6879(74)31077-4. [DOI] [PubMed] [Google Scholar]