Abstract
The potency and specificity of locked nucleic acid (LNA) antisense oligonucleotides was investigated as a function of length and affinity. The oligonucleotides were designed to target apolipoprotein B (apoB) and were investigated both in vitro and in vivo. The high affinity of LNA enabled the design of short antisense oligonucleotides (12- to 13-mers) that possessed high affinity and increased potency both in vitro and in vivo compared to longer oligonucleotides. The short LNA oligonucleotides were more target specific, and they exhibited the same biodistribution and tissue half-life as longer oligonucleotides. Pharmacology studies in both mice and non-human primates were conducted with a 13-mer LNA oligonucleotide against apoB, and the data showed that repeated dosing of the 13-mer at 1–2 mg/kg/week was sufficient to provide a significant and long lasting lowering of non-high-density lipoprotein (non-HDL) cholesterol without increasing serum liver toxicity markers. The data presented here show that oligonucleotide length as a parameter needs to be considered in the design of antisense oligonucleotide and that potent short oligonucleotides with sufficient target affinity can be generated using the LNA chemistry. Conclusively, we present a 13-mer LNA oligonucleotide with therapeutic potential that produce beneficial cholesterol lowering effect in non-human primates.
INTRODUCTION
Potency and specificity are two key parameters for any antisense oligonucleotide. Other things being equal, potency has until now been considered positively correlated to the affinity of the antisense oligonucleotide for its target sequence (1,2). Specificity however is affected by two somewhat conflicting properties. First, the antisense oligonucleotide needs to be long enough in order to ensure that its target sequence is unique in the transcriptome. Second, as the antisense oligonucleotide is elongated, its ability to discriminate between the matched and mismatched sequences, i.e. its intrinsic specificity, is diminished. Thus, for optimal specificity, antisense oligonucleotides should be as short as possible, but long enough to maintain specificity for the target. In the annotated human transcriptome, this requirement can often be fulfilled with oligonucleotides as short as 12- or 13-mers. Despite this, such short oligonucleotides have so far not been preferred in the antisense field, and oligonucleotides in the size range of 18–22 nt have traditionally been used. This size preference is a consequence of the modest affinity properties of the first and second generation chemical modifications [e.g. phosphorothioate, 2′-O-methyl and 2′-O-methoxyethyl (MOE)] that do not enable sufficient target affinity for very short oligonucleotides to exhibit potent pharmacological activity.
Antisense oligonucleotides and siRNAs have recently been used to target apolipoprotein B-100 (apoB-100; 3–6), and an 20-mer 2′-MOE antisense oligonucleotide is currently in clinical phase 3 trial (7). Thus, in addition to its potential for being a therapeutically relevant target for a new class of therapeutics, apoB is also a convenient reference target for assessing key drug parameters of new classes of antisense oligonucleotides.
The majority of apoB is synthesized in the liver, where its constitutive expression results in the production and secretion of very low-density lipoprotein (VLDL) particles in response to the hepatic lipid content. In plasma, a fraction of VLDL is metabolized to low-density lipoprotein (LDL) (8). Elevated levels of plasma LDL-cholesterol is known to be a major risk factor for atherosclerosis, which in turn is the main underlying cause of coronary heart disease and stroke (9). Given the relationship between the hepatic apoB expression, hepatic VLDL secretion and plasma LDL levels, inhibition of apoB mRNA will lead to a reduction of plasma LDL-cholesterol levels.
In this study, we demonstrate that the high affinity of locked nucleic acid (LNA; 10,11) can be used to construct very short oligonucleotides with high pharmacological activity. We investigate how such size reduction affects key drug parameters such as specificity, potency, biodistribution and tissue half-life.
MATERIALS AND METHODS
Design and synthesis of LNA oligonucleotides
A series of LNA antisense oligonucleotides of different length (10- to 20-mers) were designed with 100% sequence identity to the cynomolgus monkey and human apoB mRNA sequences. The 10- to 16-mers were designed to also have 100% sequence identity to mouse apoB mRNA. (Genbank accession no.: NM_000384 and NM_009693 for human and mouse apoB mRNA respectively; the cynomolgus mRNA was sequenced in-house). All oligonucleotides were designed as gap-mers containing 8–10 DNA nucleotides flanked by 1–5 LNA nucleotides at both ends with all internucleoside linkages phosphorothioated (Table 1).
Table 1.
In vitro properties of oligonucleotides
| Oligonucleotides | |||
|---|---|---|---|
| Oligonucleotidea | Length | Tm versus RNA (°C) | IC50 in Huh-7 cells (nM) |
| 5′-TTCAGcattggtattCAGTG-3′ | 20-mer | 77 | 5.0 ± 1.2 |
| 5′-CAGcattggtatTCAg-3′ | 16-mer | 63 | 2.7 ± 1.3 |
| 5′-CAGcattggtatTCA-3′ | 15-mer | 60 | 0.5 ± 1.3 |
| 5′-AGCattggtatTCA-3′ | 14-mer | 61 | 0.2 ± 1.2 |
| 5′-GCattggtatTCA-3′ | 13-mer | 57 | 0.2 ± 1.3 |
| 5′-GCattggtatTC-3′ | 12-mer | 53 | 0.4 ± 1.4 |
| 5′-CattggtatT-3′ | 10-mer | 44 | ND |
| 5′-gcattggtattc-3′ | 12-mer PS | 34 | ND |
aGap-mer oligonucleotides with LNA (capital) and DNA (plain font). All internucleoside linkages are phosphorothioated. Melting temperatures (Tm) of LNA oligonucleotides were measured against complementary RNA. ApoB mRNA (normalized to GAPDH) IC50 values were determined from three independent experiments ( ± SD). Non-detectable IC50 values, due to low potency, marked ND.
All oligonucleotides were synthesized using standard phosphoramidite protocols on an ÄKTA Oligopilot (GE Healthcare) at 130 µmol to 8 mmol scales employing custom made polystyrene primer supports. The DNA monomers were obtained from Proligo (Sigma-Aldrich) and the LNA monomers and solid support were produced by Santaris Pharma (commercially available from Exiqon, Denmark). After synthesis, the oligonucleotides were cleaved from the support using aqueous ammonia at 65°C overnight. The oligonucleotides were purified by ion exchange and desalted using a Millipore-membrane and were finally characterized by LC-MS (Reverse phase and ESI-MS).
Melting temperature of LNA oligonucleotide/RNA duplexes (Tm)
The binding affinity of the LNA oligonucleotides, determined as the melting temperature (Tm), was measured against complementary RNA sequences as described in Frieden et al. (12). In brief, the LNA oligonucleotide and the complementary RNA was dissolved in RNase free water and mixed with Tm-buffer (200 mM NaCl, 0.2 mM EDTA, 20 mM Na-phosphate, pH 7.0). The solution was heated to 95°C for 3 min and the oligonucleotides were allowed to anneal at room temperature for 30 min. The duplex Tm values were measured on a Lambda 25 UV/VIS Spectrophotometer equipped with a Peltier temperature programmer (PTP6) using PE Templab software (PerkinElmer). The temperature was increased from 20°C to 95°C and then down to 25°C at 1°C/min, and the absorption was recorded at 260 nm every minute. The first derivative and the local maximums of both the melting and annealing were used to assess the duplex melting temperatures.
Cell culturing and transfections
Huh-7 cells (obtained from ECACC) were maintained in complete medium consisting of DMEM (D5671, Sigma-Aldrich), 2 mM GlutaMAX (Invitrogen), 1xNEAA (Invitrogen), 10% FBS (Biochrom AG) and 25 µg/ml Gentamicin (Sigma-Aldrich) at 37°C, 5% CO2. Transfections were performed in 96-well plates (Nunc) using a final oligonucleotide concentration of 0–20 nM. A total of 7500 cells per well were seeded the day before transfection. At the time of transfection, the medium was changed to 80 µl OptiMEM (Invitrogen) containing 2.5 µg/ml Lipofectamine 2000 (Invitrogen) per well. After 7–10 min at room temperature, 20 µl OptiMEM containing oligonucleotide or saline was added. The cells were incubated with the transfection mix for 4 h, whereafter medium was changed to complete medium and cells were incubated for an additional 20 h. Complementary DNA was generated directly from each sample using the TaqMan Gene Expression Cells-to-Ct kit (Applied Biosystems) according to the manufacturer’s instructions.
Mouse studies
Inbred C57BL/6J female mice were obtained from Taconic (Denmark) and fed ad libitum with a commercially pelleted mouse diet (altromin no 1324, Gentofte, Denmark) containing 4 wt% fat. The animal facility was illuminated to give a cycle of 12 h light and 12 h darkness. The temperature was 21 ± 2°C and the relative humidity 55 ± 10%.
LNA oligonucleotides were administered to the mice by tail vein injection (i.v.) based upon body weight. All LNA oligonucleotides were formulated in saline which was also used as the control. The LNA oligonucleotides were administered at 0.5–25 mg/kg/dose. Retro-orbital sinus blood was collected in S-monovette Serum-Gel vials (Sarstedt) for serum preparation. At the time of sacrifice, mice were anesthetized (70% CO2/30% O2) before blood sampling and cervical dislocation was performed after which livers and kidneys were collected in RNA-later (Sigma-Aldrich) or snap frozen in liquid nitrogen. All mouse experiments were performed according to the principles stated in the Danish law on animal experiments, and were approved by the Danish National Committee for Animal Experiments, Ministry of Justice, Denmark.
Oligonucleotide content in tissue
Sample and standard preparation
Tissue samples (100 mg) were collected in 2 ml Eppendorf tubes and kept on dry ice. Extraction buffer 500 µl [0.5% Igepal CA-630 (Sigma-Aldrich), 25 mM Tris pH 8.0, 25 mM EDTA, 100 mM NaCl, pH 8.0] containing proteinase K (1 mg/ml) (Sigma-Aldrich P4850) and two tungsten carbide beads (3 mm) were added. The samples were homogenized mechanically by a Retsch MM300 (8 min at 25 revolutions per seconds) and homogenates were incubated overnight at 37°C. Control tissue from untreated animals were spiked with the relevant oligonucleotides at 5–250 µg/g tissue and treated as described for the samples above.
Extraction of samples, standard and QC-samples
One milliliter phenol–chloroform–isoamyl alcohol [25:24:1(v/v/v)], saturated with 10 M Tris, pH 8.0, and 1 mM EDTA (Sigma P2069) was added to each tissue samples and vortexed for 5 min. Phase separation was achieved by centrifugation at 4000g for 15 min. The aqueous phase (upper-phase) was diluted 100 times. These dilutions were kept at 4°C and were stable for up to 2 weeks.
Oligonucleotide content determination by ELISA (13-mer)
Streptavidin-coated strips (Immobilizer Streptavidin LockWell module plate, Nunc) were washed three times in 300 µl 5× SSCT buffer (750 mM NaCl, 75 mM sodium citrate, 0.05% Tween-20, pH 7.0). Each well was incubated for 30 min at room temperature under gentle agitation with 100 µl of a 0.02 µM solution of biotinylated capture probe (6-mer fully LNA-modified phosphodiester oligonucleotide complementary to the 5′-end of the 13-mer) in 5× SSCT buffer. The wells were aspirated and washed three times with 300 µl of 2×SSCT buffer (300 mM NaCl, 30 mM sodium citrate, 0.05% Tween-20, pH 7.0). One hundred microliters of the extracted and diluted oligonucleotide samples (pmol range) were added to the wells, which were agitated at room temperature for 0.5 h. The wells were aspirated and washed three times with 300 µl of 2× SSCT buffer. One hundred microliters of a 0.025 µM solution of a 5′-digitoxinated conjugated (Dig) detection probe (5× SSCT buffer with 7-mer fully LNA modified phosphorodiester oligonucleotide, complementary to the 3′-end of the 13-mer) was added to each well and incubated for 1 h at room temperature under gentle agitation. The wells were aspirated and washed three times with 300 µl of 2× SSCT buffer. One hundred microliters of anti-Dig-POD Fab fragments (Roche Applied Science) diluted 1:4000 in PBS containing 0.05% Tween-20 (pH 7.2) were added to each well and incubated for 1 h at room temperature under gentle agitation. The wells were aspirated and washed three times with 300 µl of 2× SSCT buffer. One hundred microliters of substrate solution (TMB + Substrate-Chromogen, Dako) was added to each well and incubated for 3–5 min at room temperature under gentle agitation, after which the incubation was stopped by addition of sulphuric acid (100 µl, 0.5 M). The intensity of the color development was measured spectrophotometrically at 450 nm, and the test samples were referenced against the standard samples.
Cholesterol analysis
Serum was analyzed for total cholesterol using ABX Pentra Cholesterol CP (Horiba ABX Diagnostics) according to the manufacturer’s instructions. The cholesterol content in serum lipoprotein classes was determined in fractions separated by ultracentrifugation. The density of the serum samples was adjusted to 1.067 g/ml by addition of NaBr, and the samples were then centrifuged for 4 h at 100 000 r.p.m. in an Optima MAX table top ultracentrifuge using a TLA-100 rotor (Beckman Coulter). The top (non-HDL fraction, mainly VLDL+LDL lipoproteins) and bottom (HDL) fractions were isolated and analyzed for total cholesterol content using the ABX Pentra Cholesterol CP according to the manufacturer’s instructions.
Liver enzyme ALT
The activity of alanine-aminotransferase (ALT) in mouse serum was determined using an enzymatic ALT assay (Horiba ABX Diagnostics) according to the manufacturer’s instructions adjusted to 96-well format. Data were correlated to a 2-fold diluted standard curve generated from an ABX Pentra MultiCal solution. The results were presented as ALT activity in units/liter.
Quantitative whole-body autoradiography
35S was incorporated at the ultimate 5′-phosphate position of the 12- and 16-mer using a slightly modified version of a standard protocol (13) where only carbon disulphide was exchanged with carbon tetrachloride to eliminate thio-oxidation of the ultimate 5′-phosphite with the solvent (carbon disulphide). After chromatographic purification the 35S-labeled oligonucleotides were administered intravenously by tail vein injection to inbred C57BL/6J female mice (Taconic, Denmark) at 9.2 mg/kg for the 12-mer and 14.7 mg/kg for the 16-mer. Individual mice were sacrificed at 5 min, 15 min, 1 h, 4 h, 2 days, 7 days and 21 days after injection of each oligonucleotide. Mice were snap-frozen and sectioned sagittally for whole-body autoradiography, as described by Ullberg (1977 and 1982; 14,15). Twenty micrometer sections were cut at different levels with a cryomicrotome at −20°C and freeze-dried for 24 h. Sections together with calibration sets of whole-blood solutions of the 35S-labeled oligonucleotide were chosen for phosphor imaging analysis to best represent the tissues and organs of interest. Following exposure, the imaging plates were scanned and the radioactive content of the tissues and organs from the radioluminograms were quantified by AIDA software image analysis (Raytest, Germany). Area under the curve (AUC) values (nCi·h/mg tissue) were determined and normalized to the specific radioactivity of each dose (nCi/g animal weight).
Non-human primate study
Cynomolgus monkeys (30–52 months old) had access to an expanded complete commercial primate diet [100 g/animal/day special diet services: OWN (E) short SQC]. The animals (three males and three females per dose level) received a 5 min i.v. infusion of either saline or the 13-mer at 2, 8 or 32 mg/kg. A total of five administrations were given at days 0, 7, 14, 21 and 28. Blood samples were collected from the femoral veins 10 days before the first administration (Day−10) and once a week during the experiment (at Days 7, 14, 21 and 28) prior to the subsequent dose. The animals were fasted for at least 16 h before sampling. Blood samples were analyzed for ALT, serum apoB protein and serum cholesterol (LDL and HDL cholesterol levels) by automated analyzer using commercially available kits (Randox Laboratories). The animals were sacrificed 24 h after the last infusion (Day 29). In the two highest dosing groups, additional animals were included which after the last infusion were left treatment-free for an additional 7 weeks. Blood was collected from these animals at days 35, 42, 56, 70 and 77 after the first administration of oligonucleotide. These animals were sacrificed at the end of the treatment-free period (Day 77). Liver tissue was collected in RNAlater or snap frozen in liquid nitrogen.
The cynomolgus monkey study was a part of our clinical program and was conducted by a certified contract organization (AAALAC accredited and approved by the National Ministry of Agriculture) in accordance with the testing facility’s standard operating procedure.
Quantitative PCR
Total RNA was extracted from tissue homogenates using the RNeasy mini kit (Qiagen) according to the manufacturer’s instructions. First strand cDNA was generated from total RNA by a reverse transcription reaction using random decamers, 0.5 µg total RNA, and the M-MLV RT enzyme (Ambion), according to manufacturer’s instructions. mRNA quantification of selected genes was carried out using commercially available TaqMan assays (Applied Biosystems). An applied Biosystems 7500Fast real-time PCR instrument was used for amplification. Data were analyzed and quantified using 7500Fast SDS software. ApoB mRNA levels were normalized to GAPDH or β-actin mRNA and presented relative to saline control.
Histopathology
Mouse liver samples were cryosectioned for Oil Red O staining and formalin-fixed followed by cryosectioning before hematoxylin and eosin staining in accordance with previously published method by Moewis (16). The Oil Red O staining was quantified using an image analysis system (Leica Q500, Cambridge, UK) and the percentage of Oil Red O staining calculated as area of Oil Red O staining relative to total analyzed area.
Statistical analysis
Statistical analysis was performed using one-way ANOVA with Bonferroni’s Multiple Comparison Test as post test to determine the exact nature of the differences if the data followed a Gaussian distribution. Otherwise, the non-parametric Kruskal Wallis test with Dunn’s Multiple Comparison test was used as a post test. P < 0.05 was considered to be of statistical significance. The GraphPad Prism version 4.03 was used for the statistical analysis of data from the mouse experiments and the Provantis data acquisition system was used to analyze the primate data. The primate pharmacodynamic data (serum apoB mRNA and protein versus oligonucleotide concentration in liver) were analyzed using an inhibitory effect Emax model (model 103) in the WinNonlin program (version 5.2.1; Pharsight Corporation).
RESULTS
In vitro properties of LNA oligonucleotides
The target affinity of the LNA oligonucleotides was measured Tm determined against the complementary RNA sequences. The highest Tm was found for the 20-mer (77°C), with Tm decreasing as length and LNA content was reduced (Table 1).
The Huh-7 human hepatoma cell line, which expresses high levels of apoB mRNA, was used to evaluate the RNA silencing properties of the oligonucleotides employing standard transfection procedures. The 15-, 14-, 13- and 12-mer LNA oligonucleotides all potently reduced apoB mRNA expression in a dose-dependent manner and with essentially identical IC50 values (∼0.5 nM). Molecules below the 12-mer size, i.e. the 10-mer LNA oligonucleotide, did not induce any detectable degree of target inhibition. Above the 15-mer size, i.e. the 16- and 20-mer, target inhibition decreased with increasing length (IC50 values of the 16-mer and 20-mer at 2.7 nM and 5 nM, respectively; Table 1), despite the fact that target affinity increased. The 12-mer phosphorothioate without LNA nucleotides did not reduce the expression of apoB mRNA at the concentrations used in this experiment.
Potency assessment in mice
The 12- to 16-mer LNA oligonucleotides were formulated in saline and dosed at 5.0 mg/kg i.v. on three successive days in mice that were subsequently sacrificed 24 h after the last dose. All oligonucleotides reduced the expression of apoB mRNA in the liver, and this correlated with a decrease in the levels of total serum cholesterol at the time of sacrifice (Table 2). The two shortest, the 12- and 13-mer, were found to be the most potent, resulting in ∼80–90% reduction in both apoB mRNA and total serum cholesterol relative to the saline control. In comparison, the 16-mer produced only a 16% reduction in apoB mRNA and a 35% reduction in total serum cholesterol (Table 2). The reductions in total cholesterol were primarily due to a reduction in non-HDL cholesterol resulting in an improved HDL/non-HDL ratio with all tested oligonucleotides. No significant changes were observed in serum ALT with any of the oligonucleotides (Table 2).
Table 2.
In vivo potency of LNA oligonucleotides
| Parameter | LNA oligonucleotide |
|||||
|---|---|---|---|---|---|---|
| Saline | 16-mer | 15-mer | 14-mer | 13-mer | 12-mer | |
| ApoB mRNA in liver (% of saline group) | 100 ± 15a | 84 ± 9a,b | 78 ± 18b | 18 ± 6c | 7 ± 3c | 8 ± 2c |
| Total cholesterol in serum (mg/dl) | 84 ± 13a | 55 ± 10b | 54 ± 7b | 22 ± 5c | 10 ± 2c | 14 ± 3c |
| HDL/(non-HDL) ratio* | 2 ± 0a | 4 ± 1a,c | 5 ± 1a,c | 11 ± 4b | 10 ± 6b,c | 8 ± 3b,c |
| ALT in serum (U/L) | 52 ± 17 | 42 ± 8 | 25 ± 5 | 68 ± 14 | 55 ± 8 | 75 ± 15 |
Mice were administered 5 mg/kg LNA oligonucleotide i.v. on three successive days and sacrificed 24 h after last dose. ApoB mRNA expression in liver was normalized to GAPDH levels (in percent of saline control group). Absolute total serum cholesterol levels, lipoprotein fractions and ALT were measured in serum. a,b,cValues in the same row but with different superscript letters are significantly different (P < 0.05). *Values for non-HDL (mainly VLDL+LDL) and HDL represent percent of total serum cholesterol recovered in the respective fraction after ultracentrifugation. Data represents mean values ± SD, n = 6.
Mis-match specificity in mice
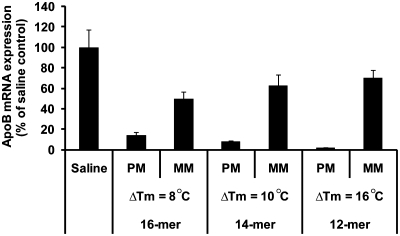
The expected advantage of shorter oligonucleotides to discriminate between the correct and mismatched targets was examined by comparing the potency of the 12-, 14- and 16-mers to that of single-base mismatched versions of these oligonucleotides which created an A–A mis-match to the apoB target in the oligonucleotide gap. This single mismatch results in an 8.33%, 7.14 % and 6.25% theoretical mis-pairing for the 12-, 14- and 16-mer, respectively and caused a decrease in the Tm values compared to the fully matched LNA oligonucleotides. The relative change in target affinity (ΔTm) increased with the increased percentage mis-pairing; 16°C (12-mer), 10°C (14-mer) and 8°C (16-mer) confirming the improved intrinsic specificity for shorter oligonucleotides. The potency of the three sets of matched/mis-matched oligonucleotides was evaluated in vivo in mouse livers and kidneys. As the fully matched LNA oligonucleotides was found to have different potencies (vide supra), the animals were dosed at 2.5, 5.0 and 25 mg/kg for the 12-, 14- and 16-mer, respectively, to establish a similar level of hepatic apoB mRNA reduction. The mice were dosed on three successive days, and sacrificed 24 h after the last dose. In the liver, the perfect matched LNA oligonucleotides exhibited an equal 75% reduction of apoB mRNA, whereas none of the mis-matched LNA oligonucleotides produced any effect (data not shown) thus preventing any conclusion on specificity versus length. In kidney, however, the mis-matched oligonucleotides showed to be less potent as size decreased an observation which was even further augmented by an increased potency of the matched oligonucleotides (Figure 1).
Figure 1.
Mismatch specificity in mice. Mice were administered i.v. injections of saline, the 12-, 14- and 16-mer and their respective one mismatch versions at 2.5, 5 and 25 mg/kg, respectively on three successive days and sacrificed 24 h after last dose. Kidneys were analyzed for apoB mRNA expression normalized to GAPDH (in percent of saline control group). Data represent mean values ± SD, n = 5. MM indicates mismatch and PM perfect match. ΔTm values represent the difference in Tm between PM and MM oligonucleotides measured against RNA complementary to the perfect match oligonucleotides.
Quantitative whole-body autoradiography
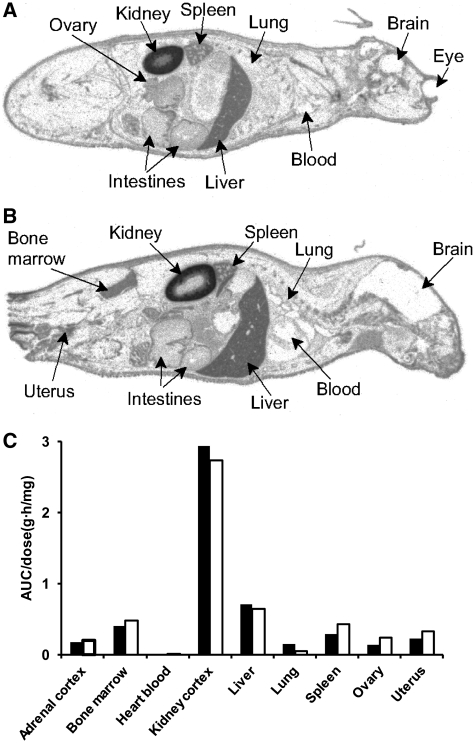
To understand whether the different potencies of the 12- to 16-mer LNA oligonucleotides were the consequence of differences in biodistribution or tissue half-life, the 12-mer and the 16-mer LNA oligonucleotides were covalently labeled at the 5′-internucleoside bond with 35S, and injected i.v. in mice at identical molar concentrations [labeled LNA oligonucleotides are highly stable in tissues, allowing a correlation between the presence of radioactivity and intact oligonucleotide (11,17)]. Mice were sacrificed at different time points after dosing, ranging from 5 min to 21 days, snap-frozen, sectioned, subjected to whole-body autoradiography, and quantified by using whole-blood solutions of the 35S-labeled oligonucleotides as reference standard. The two LNA oligonucleotides exhibited a very similar distribution pattern across different tissues (Figure 2A and B). The highest levels of radioactivity/oligonucleotide accumulation were found in kidney cortex, liver, bone marrow, spleen, ovary, uterus and adrenal cortex (Figure 2C). Furthermore, in the liver, the distribution rate and peak value time were similar for the two oligonucleotides, 75 nCi·h/mg and 4 h, respectively. The elimination half-lives in liver were found to be 9 days for the 12-mer and 14 days for the 16-mer.
Figure 2.
Quantitative whole-body autoradiography. Tissue distribution in mice of two 35S-labelled oligonucleotides, the 12- and 16-mer (A and B, respectively), administered i.v. Individual mice were sacrificed at 24 h post-administration, snap-frozen and sectioned for whole-body autoradiography. Following exposure the imaging plates were scanned, tissues and organs intensity were quantified by image analyses. (C) Area under the curve values normalized to the specific radioactivity of the dose levels of 16-mer (dark bars) and 12-mer (white bars).
Single dose studies in mice
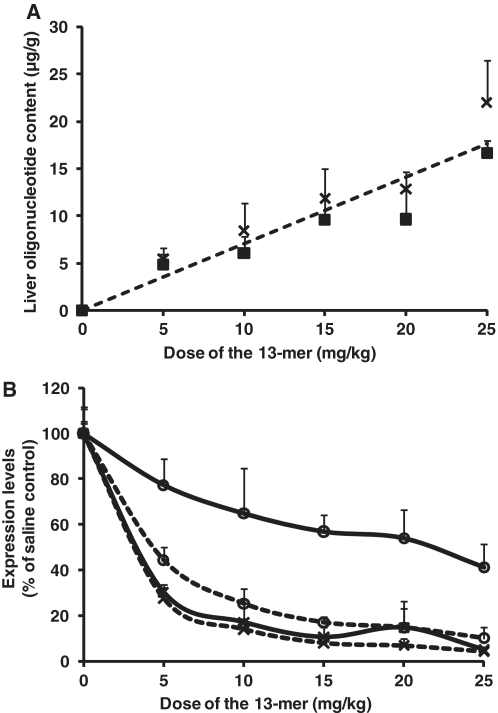
To examine the pharmacological properties of a short LNA oligonucleotide, the 13-mer was selected for further in vivo studies. Mice were administered single i.v. injections at 5, 10, 15, 20 or 25 mg/kg and sacrificed 24 and 48 h after dosing. The liver content of the 13-mer was measured and found to correlate linearly with dose level (R2 = 0.88), at ∼0.7 µg oligonucleotide/g liver for each 1.0 mg/kg dosed (Figure 3A). The apoB mRNA expression reached maximum reduction at the dose levels 10–15 mg/kg at 24 and 48 h after dosing (>83%, Figure 3B). Higher doses of the oligonucleotide (resulting in higher liver content), did not produce further significant reduction in either the expression of the apoB mRNA or total serum cholesterol 48 h after dosing. The reduction in apoB mRNA was rapid and preceded the effect on total serum cholesterol, which increased from 24 to 48 h after dosing. Thus, the maximum effect in mice in this study was reached at a dose of 10–15 mg/kg, which corresponds to an accumulation of 7–11 µg of the 13-mer per gram of liver.
Figure 3.
Single dose in mice. Mice were administered a single i.v. injection of 5, 10, 15, 20 and 25 mg/kg of the 13-mer and sacrificed 24 and 48 h after dosing. (A) The 13-mer oligonucleotide content was measured in liver and presented as function of dose level at 24 h (crosses) and 48 h (boxes) after dosing. The linear correlation (R2 = 0.88) showed oligonucleotide content of ∼0.7 µg oligonucleotide/g liver for each 1 mg/kg administered. (B) Liver apoB mRNA expression (crosses) and serum total cholesterol (circles) were measured for all dose levels 24 h (solid line) and 48 h (dashed line) after dosing. Data are expressed as percentage of respectively, apoB mRNA and serum total cholesterol in saline control and presented as mean values ± SD, n = 5/dose level and time point. The apoB mRNA expression was normalized to the expression of GAPDH.
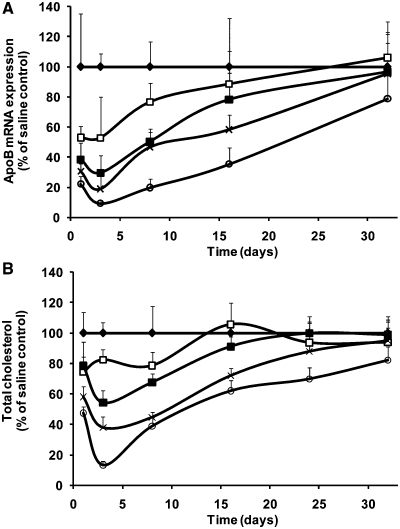
The duration of effect in mice was studied after single i.v. injections of 0.5, 1.0, 2.5 and 5.0 mg/kg of the 13-mer. The mice were sacrificed 1, 3, 8, 16 and 32 days after dosing. A single dose resulted in a rapid and dose-dependent reduction of liver apoB mRNA expression and total serum cholesterol (Figure 4). The lowest and highest dose (0.5 mg/kg and 5 mg/kg), respectively, resulted in a reduction in apoB mRNA expression of 47% (P < 0.01) and 78% (P < 0.001) and total serum cholesterol of 25% (P < 0.05) and 52% (P < 0.001) already 1 day after dosing.
Figure 4.
Duration of effect in mice. Mice were administered a single i.v. injection of saline (diamonds) or 13-mer at 0.5 (white boxes), 1 (dark boxes), 2.5 (crosses) and 5 mg/kg (circles). Livers were analyzed for (A) apoB mRNA expression normalized to GAPDH and (B) serum total cholesterol levels (both in percent of control groups) at days 1, 3, 8, 16 and 32 after administration. (Data represent mean values ± SD, n = 5).
The effect of a single dose was long lasting, and correlated with the estimated elimination half-life of 9–14 days determined for the 12- and 16-mer (vide supra). At the highest dose (5.0 mg/kg), hepatic apoB mRNA expression and total serum cholesterol were reduced by ∼90%, 3 days after dosing (P < 0.001), and the effect was still significant after 3 weeks (Figure 4).
Multiple dose study in mice
The data from the single dose studies indicated that dosing once weekly or biweekly would be sufficient to achieve sustained and significant reductions in serum cholesterol. Accordingly, a multiple dose study was conducted in which mice were dosed with the 13-mer either once weekly (Group 1) or biweekly (Group 2) for a total of 7 weeks. In both groups, mice received the 13-mer i.v. at 1.0, 2.5 or 5.0 mg/kg. The mice in Group 1 received a total of seven doses, and the mice in Group 2 received a total of four doses of each concentration. In both groups, serum sampling for cholesterol analysis was conducted weekly and immediately before administration of the next dose. Livers were collected for apoB mRNA analysis at the end of the study (Day 49), 1 week after the last dose.
Reductions in hepatic apoB mRNA levels at the end of the study were ∼70%, 80% and 90% for dose levels 1.0, 2.5 and 5.0 mg/kg, and were similar both in mice dosed weekly and biweekly (data not shown).
These reductions in apoB mRNA expression were paralleled by a significant decrease in apoB containing lipoproteins (non-HDL) throughout the study. At 1 mg/kg weekly dosing, non-HDL cholesterol levels were gradually reduced throughout the dosing period, reaching ∼80% reduction compared to saline control level at the end of the experimental period (Figure 5A). The 2.5 mg/kg weekly dose further improved the effect after the first 2 weeks, and led to a ∼90% reduction in non-HDL levels relative to saline control at the end of the study. Consistent with the substantial activity of the 2.5 mg/kg dose, no further reduction in non-HDL cholesterol was observed with the 5 mg/kg dose. All three dose levels also affected HDL cholesterol levels, albeit significantly less (P < 0.001) than the effects on non-HDL cholesterol, reaching 40–60% relative to saline levels at the end of the study (Figure 5B).
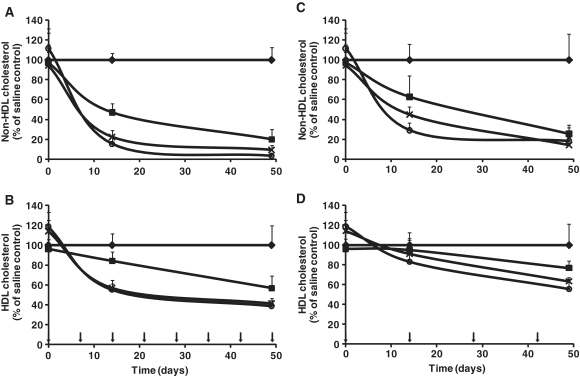
Figure 5.
Multiple dosing in mice. Mice were administered i.v. injections weekly (group 1, left panel) or biweekly (group 2, right panel) for 49 days with either saline (diamonds) or the 13-mer at 1 (dark boxes), 2.5 (crosses) or 5 mg/kg (circles) (arrows on bottom figure indicates dosing days). Serum was analyzed for non-HDL cholesterol (mainly VLDL+LDL) (A and C) and HDL cholesterol (B and D) at 0, 14 and 49 days after first administration. (Data represent mean values ± SD, n = 7).
Biweekly dosing produced a rather similar picture between the three dose groups as weekly dosing, although both the rate of reduction and maximum reduction at the end of the study was smaller on both measured parameters, non-HDL (Figure 5C) and HDL cholesterol (Figure 5D). Similar to what was observed in the weekly dosing group the effect on HDL in biweekly dosing was also significantly less than the effect on non-HDL (P < 0.001). Overall, the HDL/non-HDL ratios at the end of the study were identical for the two different dose regimens, ∼4-fold increase compared to saline treated animals.
Multiple dose study in non-human primates
Wild-type mice have at least 70% of their serum cholesterol carried as part of HDL particles, whereas cynomolgus monkeys have almost equal distribution between HDL and non-HDL cholesterol (18). Thus, cynomolgus monkeys more closely resemble humans that have ∼70% of serum cholesterol in LDL particles (19). Male and female cynomolgus monkeys were therefore used in a multiple dose study, where they were dosed i.v. with the 13-mer once a week for 4 weeks (a total of 5 doses). The dose levels were 2, 8 and 32 mg/kg/week. A recovery group from the saline and the two highest dose level groups were kept treatment-free for an additional period of 7 weeks.
Following treatment with the 13-mer, LDL cholesterol levels were reduced in a dose-dependent manner. Close to maximal effect was reached already after the second administration where the dose levels 2, 8 and 32mg/kg/week resulted in ∼45%, 70% and 90% reduction in LDL cholesterol, respectively, and the LDL cholesterol remained hereafter at similar levels for the rest of the treatment period in all dose groups, relative to saline (Figure 6A). In the mid- and high-dose groups, LDL cholesterol was also significantly reduced throughout the recovery phase, with significant reductions still evident 7 weeks after last dose (Figure 6A). The LDL cholesterol levels correlated linearly with serum apoB protein concentration with a correlation coefficient R2 = 0.94 (data not shown).
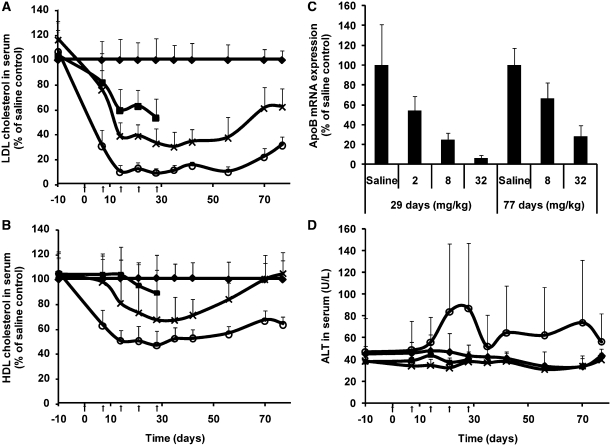
Figure 6.
Multiple dosing in non-human primates. Cynomolgus monkeys were administered i.v. injections of the either saline (diamonds) or the 13-mer at 2 (dark boxes), 8 (crosses) or 32 (circles) mg/kg once a week for 28 days (arrows on figures indicates dosing days). Serum was sampled 10 days prior to first administration (Day−10) and every week from the first administration. LDL cholesterol (A), HDL cholesterol (B), ALT activity (D) in serum relative to saline control group was measured (±SD, n = 10). A subset of animals at the 8 and 32 mg/kg dose levels were kept treatment-free for an additional seven week period (n = 4). ApoB mRNA expression in liver normalized to β-actin and relative to saline control group (C) at the end of the treatment period (Day 29, ±SD, n = 6) and at the end of the treatment-free period (Day 77, ±SD, n = 4).
During the treatment period, HDL cholesterol was also affected in a dose-dependent manner, albeit significantly less than non-HDL, and only marginally—if at all—at the lowest dose (Figure 6B). HDL cholesterol remained reduced in the high dose group during the 7 weeks of recovery whereas it returned to the saline control level in the mid-dose group (Figure 6B).
Liver apoB mRNA expression was determined for all three dose groups at the end of the treatment period (Day 29), and the end of the recovery period (Day 77) for the mid- and high-dose groups. ApoB mRNA was reduced in a dose-dependent manner, reaching a 90% reduction in the high-dose group (Figure 6C) at the end of dosing, where also LDL cholesterol was reduced by ∼90% (vide supra). ApoB mRNA levels increased during the recovery period but were still reduced by 35% and 70% in the mid- and high-dose groups, respectively at Day 77.
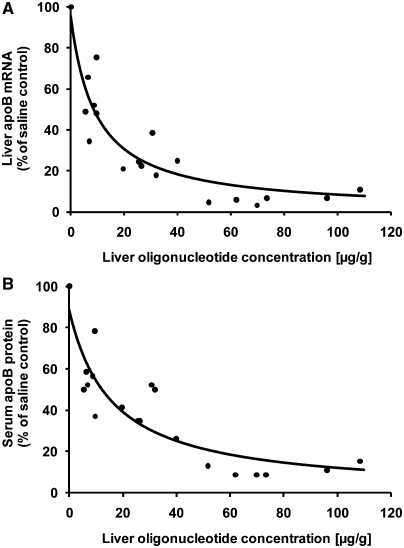
The amount of LNA oligonucleotide in liver was measured in all animals at the end of the dosing period, and was found to correlate closely with the reduction in both apoB mRNA and serum protein (Figure 7). The best estimated non-linear regression fit appeared to follow the inhibitory effect Emax model (R2 = 0.90 for mRNA and R2 = 0.88 for protein). According to this fit, a 50% reduction in apoB mRNA and protein was obtained at a liver concentration of the LNA oligonucleotide of ∼10 and 15 µg/g liver, respectively.
Figure 7.
Oligonucleotide content in liver relative to apoB levels in cynomolgus monkeys. The liver tissue content of the 13-mer was measured in all samples from the multiple dosing study in cynomolgus monkeys after the end of the treatment period (Day 29). Data are presented relative to apoB mRNA expression in liver (A) and serum apoB protein levels (B). Best estimated Emax fit curves are plotted with R2 = 0.88 and R2 = 0.90 for mRNA and protein levels, respectively.
Although small fluctuations were observed in serum ALT levels for the monkeys in the 32 mg/kg/week group, no statistically significant changes were recorded for any of the groups throughout the entire experimental period (Figure 6D).
DISCUSSION
LNA has been used extensively to inhibit RNA expression. In addition, LNA gapmers have been described as a highly potent class of oligonucleotides for mRNA inhibition (11,20). These molecules are usually designed with a central DNA segment (6–10 nucleotides) that is flanked at each molecular terminus by 2–4 LNA nucleotides. This design is particularly potent for three synergistic reasons: the LNA nucleotides provide a high binding affinity for the target; the central DNA region triggers RNase H cleavage activity; and since all the phosphate groups are thioated, the gapmer is highly nuclease resistant. All these basic properties of LNA oligonucleotides have been extensively commented on in the literature (17,21–31).
We demonstrate here that LNA oligonucleotides, in length as short as 12 nt, exhibit high antisense activity in both liver and kidney of mice (32) and non-human primates. The data further show that both affinity and size are major determinants for the potency of LNA oligonucleotides. The importance of affinity is illustrated by the fact that the 12-mer LNA oligonucleotide (Tm = 53°C), exhibited an IC50 value of 0.4 nM, whereas the iso-sequential, non-LNA containing, phosphorothioate oligonucleotide (Tm = 34°C) had no effect. The fact that potency correlates positively with affinity for iso-sequential oligonucleotides has also been described for other targets (1,2). The 10-mer LNA oligonucleotide had a much reduced affinity (Tm = 44°C), compared to the 12-mer, which is likely the reason that it did not exhibit any activity.
The importance of size is illustrated by the fact that the longer oligonucleotides (>14-mer) did not exhibit higher potencies despite their inherent higher affinities. This pattern was augmented in vivo where potency decreased significantly with increasing size, illustrated by the fact that the 12- and 13-mers were approximately 11 times more potent than the 16-mer.
Both the 12-mer and the 16-mer LNA oligonucleotides accumulated identically in the target organs (liver and kidney) over time in mice, so the differences in potencies between the differently sized oligonucleotides cannot be explained by distribution differences. Neither can the potency differences be explained by differences in tissue half-lives which appeared to be longer for the 16-mer compared to the 12-mer.
Taken together, these differences might be explained by the presence of a ‘threshold affinity’ that an oligonucleotide must achieve in order to exhibit high potency, and beyond which further increase in affinity will not necessarily correlate with additional potency. At the same time, the data also indicate that there is a ‘length penalty’ that reduces potency and which not necessarily is compensated by increased affinity.
Although only scarcely experimentally supported at this time, this ‘length penalty’ could be linked to protein binding. Phosphorothioate oligonucleotides are known to bind several plasma proteins in a length-dependent manner (33) so the increased potency of shorter LNA oligonucleotides could be the result of faster and more efficient intracellular availability for target hybridization—provided by reduced protein binding. Contrary to this, longer oligonucleotides would bind proteins more strongly rendering them less efficient for target hybridization. Finally, oligonucleotides with more than 15 phosphorothioate linkages have been shown to inhibit the activity of human RNase H (34) suggesting that this could be another contributing factor to the observed ‘length penalty’.
Recently Stein et al. (35) demonstrated that LNA oligonucleotides can be taken up by cells in culture by simply adding the LNA oligonucleotides without transfection reagents to the medium, a process termed gymnotic delivery. The data indicated that in cell cultures under gymnotic delivery shorter LNA oligonucleotides appeared to act faster and be most potent. Furthermore, it was reported that the potency/length relation found in vitro correlated with the in vivo outcome (35). These data indicated that in vitro results obtained under gymnotic delivery may be more predictive, compared to classical transfection assays, for the in vivo outcome (35). As an extension of this work with apoB targeting oligonucleotides (35), a study in cell cultures under gymnotic delivery using Bcl-2 targeting oligonucleotides was conducted (Supplementary Data). In this study, eight LNA gapmers were tested, of which seven were truncated from a 22-mer base sequence. It appeared that all three 12-mers were significantly more potent inhibitors of Bcl-2 compared to the longer ones (>14-mers), and in parallel to the data described above, the onset of action of the shortmers was faster. Work to resolve this mechanism is currently ongoing in our laboratories.
Our data also indicate that short LNA oligonucleotides are better at discriminating between matched and mis-matched target sequences. The ΔTm (Tm matched-Tm mis-matched) increased as the LNA oligonucleotide size decreased, and that correlated in vivo with higher target specificity (Figure 1). This discrimination was observed in kidney which accumulates the highest amount of oligonucleotide per gram tissue, allowing the mis-matched LNA oligonucleotides to reach concentrations that could exhibit some activity. In the liver, none of the mismatched LNA oligonucleotides showed any activity at the doses employed in the study thus preventing any conclusion on specificity versus length.
The present discussion is based on data obtained with a single target (apoB) and a relatively small number of oligonucleotides. Thus, it obviously does not allow for any generalization as to how affinity and size will affect the potency and specificity of LNA oligonucleotides against other targets. It merely points to the fact that oligonucleotide size as a parameter is important for potency, and furthermore, the data illustrate that in order to exploit that parameter for making better antisense oligonucleotides—with higher potency and specificity—a high affinity chemical component like LNA has to be used.
Inhibiting apoB mRNA serves as a new and potentially powerful strategy for cholesterol lowering therapies (7,36–38). With this in mind, the present study was also directed at understanding what sort of potency could be achieved in mice, and more importantly, in non-human primates with appropriately designed LNA oligonucleotides. The most intensively studied LNA oligonucleotide was the 13-mer, which in mice provided a very potent and long lasting reduction in all the parameters studied. In fact, in mice, a single dose of 0.5 mg/kg of the 13-mer administered i.v. without any formulation, reduced apoB mRNA by 47% and total serum cholesterol by 25% one day after dosing, and with activity still being significant one week after. Consistent with these characteristics, dosing the 13-mer at 1 mg/kg biweekly produced a reduction in non-HDL cholesterol of ∼75% at the end of a 7-week study. In comparison, Crooke et al. (3) reported that an 20-mer 2′-MOE oligonucleotide, dosed twice weekly for 4 weeks at 50 mg/kg, was able to achieve a 60% reduction in total serum cholesterol in mice at the end of the study.
Reducing the expression of hepatic apoB prevents liver secretion of VLDL particles. This gives rise to lipid accumulation in the liver (8,39) and elevations of liver enzymes, which has been observed after apoB inhibition employing siRNA (4). Consistent with these reports, we observed mild accumulation (up to 6–7% of total liver weight) of neutral lipids in our multiple dose study in mice, and this was associated with mild increases in ALT (up to 3-fold; data not shown). Dosing three times 5 mg/kg did not cause any significant increase in ALT (Table 1).
In monkeys where the HDL/LDL ratio is closer to the ratio in human, the 13-mer also demonstrated strong potency and long lasting effect. The low-dose group (2.0 mg/kg, administered weekly for 4 weeks) achieved an oligonucleotide concentration in liver of ∼10 µg oligonucleotide/g liver, and produced a reduction in non-HDL cholesterol of 45%. Notably, this dose level did not significantly affect HDL which only was affected at the higher doses, indicating that dose regimens can be found that will potently and exclusively affect the non-HDL components. At this dose level, there were no elevations in liver enzymes and no accumulation of liver lipids (data not shown).
Reduction of apoB mRNA expression in monkeys with a 20-mer 2′-MOE oligonucleotide has previously been reported (38). In this study, the 20-mer was dosed to high-fat fed cynomolgus monkeys, at 35 mg/kg twice a week for 5 weeks (total of ∼385 mg/kg), resulting in a 70% reduction in LDL-cholesterol. The EC50 for both serum apoB protein and liver apoB mRNA was reported to be reached at 300 µg oligonucleotide/g liver compared to the estimated EC50 of the 13-mer LNA oligonucleotide of ∼10 and 15 µg/g for apoB mRNA and apoB serum protein, respectively (Figure 7).
In conclusion, we have shown that oligonucleotide length as a parameter can be exploited to improve the design of antisense oligonucleotides. We have illustrated that in order to design potent oligonucleotides it is important to balance target affinity against oligonucleotide length, and that the most potent oligonucleotides are likely among the shortest exhibiting the sequence dependent threshold affinity. The use of LNA provides the target affinity needed for the design of shorter oligonucleotides with high in vivo potency and therapeutic potential. Such molecules delivered systemically without delivery vehicles were here demonstrated to potently down-regulate apoB mRNA in liver and lower serum non-HDL cholesterol in mice and non-human primates.
SUPPLEMENTARY DATA
Supplementary data are available at NAR Online.
FUNDING
Funding for open access charge: Santaris Pharma A/S Kogle Allé 6 DK-2970 Hoersholm Denmark.
Conflict of interest statement. The work presented in this manuscript is performed at Santaris Pharma, the company that produces LNA oligonucleotides for therapeutic purpose.
Supplementary Material
ACKNOWLEDGEMENTS
The authors wish to thank Prof. Cy A. Stein, Albert Einstein College of Medicine, NY, for fruitful discussions and for reviewing the article. We would like to thank Heidi W. Hovring, Lisbeth Bang, Rikke Solberg, Louise Degn Nielsen, Bettina Nordbo, Lene Sodborg Jorgensen, Gitte Baerentzen, Andreas Rassov and Helle Knudsen for their excellent technical support.
REFERENCES
- 1.Seth PP, Siwkowski A, Allerson CR, Vasquez G, Lee S, Prakash TP, Wancewicz EV, Witchell D, Swayze EE. Short antisense oligonucleotides with novel 2′-4′ conformationaly restricted nucleoside analogues show improved potency without increased toxicity in animals. J. Med. Chem. 2009;52:10–13. doi: 10.1021/jm801294h. [DOI] [PubMed] [Google Scholar]
- 2.Swayze EE, Siwkowski AM, Wancewicz EV, Migawa MT, Wyrzykiewicz TK, Hung G, Monia BP, Bennett CF. Antisense oligonucleotides containing locked nucleic acid improve potency but cause significant hepatotoxicity in animals. Nucleic Acids Res. 2007;35:687–700. doi: 10.1093/nar/gkl1071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Crooke RM, Graham MJ, Lemonidis KM, Whipple CP, Koo S, Perera RJ. An apolipoprotein B antisense oligonucleotide lowers LDL cholesterol in hyperlipidemic mice without causing hepatic steatosis. J. Lipid Res. 2005;46:872–878. doi: 10.1194/jlr.M400492-JLR200. [DOI] [PubMed] [Google Scholar]
- 4.Nishina K, Unno T, Uno Y, Kubodera T, Kanouchi T, Mizusawa H, Yokota T. Efficient in vivo delivery of siRNA to the liver by conjugation of alpha-tocopherol. Mol. Ther. 2008;16:734–740. doi: 10.1038/mt.2008.14. [DOI] [PubMed] [Google Scholar]
- 5.Soutschek J, Akinc A, Bramlage B, Charisse K, Constien R, Donoghue M, Elbashir S, Geick A, Hadwiger P, Harborth J, et al. Therapeutic silencing of an endogenous gene by systemic administration of modified siRNAS. Nature. 2004;432:173–178. doi: 10.1038/nature03121. [DOI] [PubMed] [Google Scholar]
- 6.Zimmermann TS, Lee AC, Akinc A, Bramlage B, Bumcrot D, Fedoruk MN, Harborth J, Heyes JA, Jeffs LB, John M, et al. RNAi-mediated gene silencing in non-human primates. Nature. 2006;441:111–114. doi: 10.1038/nature04688. [DOI] [PubMed] [Google Scholar]
- 7.Yu RZ, Geary RS, Flaim JD, Riley GC, Tribble DL, Vanvliet AA, Wedel MK. Lack of pharmacokinetic interaction of mipomersen sodium (ISIS 301012), a 2′-O-methoxyethyl modified antisense oligonucleotide targeting apolipoprotein B-100 messenger RNA, with simvastatin and ezetimibe. Clin. Pharmacokinet. 2009;48:39–50. doi: 10.2165/0003088-200948010-00003. [DOI] [PubMed] [Google Scholar]
- 8.Olofsson SO, Wiklund O, Boren J. Apolipoproteins A-I and B: biosynthesis, role in the development of atherosclerosis and targets for intervention against cardiovascular disease. Vasc. Health Risk Manag. 2007;3:491–502. [PMC free article] [PubMed] [Google Scholar]
- 9.Hansson GK, Libby P. The immune response in atherosclerosis: a double-edged sword. Nat. Rev. Immunol. 2006;6:508–519. doi: 10.1038/nri1882. [DOI] [PubMed] [Google Scholar]
- 10.Grünweller A, Hartmann RK. Locked nucleic acid oligonucleotides: the next generation of antisense agents? Biodrugs. 2007;21:235–243. doi: 10.2165/00063030-200721040-00004. [DOI] [PubMed] [Google Scholar]
- 11.Koch T, Ørum H. Locked nucleic acid. In: Crooke ST, editor. Antisense Drug Technology. Boca Raton: CRC Press; 2008. pp. 519–564. [Google Scholar]
- 12.Frieden M, Christensen SM, Mikkelsen ND, Rosenbohm C, Thrue CA, Westergaard M, Hansen HF, Orum H, Koch T. Expanding the design horizon of antisense oligonucleotides with alpha-L-LNA. Nucleic Acids Res. 2003;31:6365–6372. doi: 10.1093/nar/gkg820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zon G, Stec W. Phosphorothioate oligonucleotides. In: Eckstein F, editor. Oligonucleotide Analogues, Chapter 4. Oxford University Press; 1991. pp. 102–103. [Google Scholar]
- 14.Ullberg S. The technique of whole body autoradiography. Cryosectioning of large specimens. Science tools, LKB Inst. J., Special Issue on Whole-body Autoradiography. 1977:2–28. [Google Scholar]
- 15.Ullberg S, Larsson B, Tjälve H. Autoradiography. In: Glenn J, editor. Biological Application of Radiotracers. Boca Raton, FL, CRC Press: CRC Press; 1982. pp. 55–108. [Google Scholar]
- 16.Moewis G. Histopatologisk Teknik. Stockholm: Almqvist & Wiksell Förlag; 1978. [Google Scholar]
- 17.Fluiter K, Frieden M, Vreijling J, Rosenbohm C, De Wissel MB, Christensen SM, Koch T, Orum H, Baas F. On the in vitro and in vivo properties of four locked nucleic acid nucleotides incorporated into an anti-H-Ras antisense oligonucleotide. Chembiochem. 2005;6:1104–1109. doi: 10.1002/cbic.200400419. [DOI] [PubMed] [Google Scholar]
- 18.Leblanc M, Belanger MC, Julien P, Tchernof A, Labrie C, Belanger A, Labrie F. Plasma lipoprotein profile in the male cynomolgus monkey under normal, hypogonadal, and combined androgen blockade conditions. J. Clin. Endocrinol. Metab. 2004;89:1849–1857. doi: 10.1210/jc.2003-031233. [DOI] [PubMed] [Google Scholar]
- 19.Greeve J, Altkemper I, Dieterich JH, Greten H, Windler E. Apolipoprotein B mRNA editing in 12 different mammalian species: hepatic expression is reflected in low concentrations of apob-containing plasma lipoproteins. J. Lipid Res. 1993;34:1367–1383. [PubMed] [Google Scholar]
- 20.Koch T, Rosenbohm C, Frydenlund HF, Hansen B, Straarup EM, Kauppinen S. Locked nucleic acid: properties and therapeutic aspects. In: KURRECK J, editor. Therapeutic Oligonucleotides. Cambridge: The Royal Society of Chemistry; 2008. pp. 103–141. [Google Scholar]
- 21.Braasch DA, Liu Y, Corey DR. Antisense inhibition of gene expression in cells by oligonucleotides incorporating locked nucleic acids: effect of mRNA target sequence and chimera design. Nucleic Acids Res. 2002;30:5160–5167. doi: 10.1093/nar/gkf651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Elayadi AN, Braasch DA, Corey DR. Implications of high-affinity hybridization by locked nucleic acid oligomers for inhibition of human telomerase. Biochemistry. 2002;41:9973–9981. doi: 10.1021/bi025907j. [DOI] [PubMed] [Google Scholar]
- 23.Elmen J, Zhang HY, Zuber B, Ljungberg K, Wahren B, Wahlestedt C, Liang Z. Locked nucleic acid containing antisense oligonucleotides enhance inhibition of HIV-1 genome dimerization and inhibit virus replication. Febs Lett. 2004;578:285–290. doi: 10.1016/j.febslet.2004.11.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Frieden M, Christensen SM, Mikkelsen ND, Rosenbohm C, Thrue CA, Westergaard M, Hansen HF, Orum H, Koch T. Expanding the design horizon of antisense oligonucleotides with alpha-L-LNA. Nucleic Acids Res. 2003;31:6365–6372. doi: 10.1093/nar/gkg820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Frieden M, Hansen HF, Koch T. Nuclease stability of LNA oligonucleotides and LNA-DNA chimeras. Nucleosides Nucleotides Nucleic Acids. 2003;22:1041–1043. doi: 10.1081/NCN-120022731. [DOI] [PubMed] [Google Scholar]
- 26.Grünweller A, Wyszko E, Bieber B, Jahnel R, Erdmann VA, Kurreck J. Comparison of differnt antisense strategies in mammalian cells using locked nucleic acids, 2′-OME RNA, phosphorothioates and small intering RNA. Nucleic Acids Res. 2003;31:3185–3193. doi: 10.1093/nar/gkg409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Jepsen JS, Pfundheller HM, Lykkesfeldt AE. Downregulation of P21(WAF1/CIP1) and estrogen receptor alpha in MCF-7 cells by antisense oligonucleotides containing locked nucleic acid (LNA) Oligonucleotides. 2004;14:147–156. doi: 10.1089/1545457041526281. [DOI] [PubMed] [Google Scholar]
- 28.Kurreck J, Wyszko E, Gillen C, Erdmann VA. Design of antisense oligonucleotides stabilized by locked nucleic acids. Nucleic Acids Res. 2002;30:1911–1918. doi: 10.1093/nar/30.9.1911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lennox KA, Sabel JL, Johnson MJ, Moreira BG, Fletcher CA, Rose SD, Behlke MA, Laikhter AL, Walder JA, Dagle JM. Characterization of modified antisense oligonucleotides in Xenopus laevis embryos. Oligonucleotides. 2006;16:26–42. doi: 10.1089/oli.2006.16.26. [DOI] [PubMed] [Google Scholar]
- 30.Mong JA, Devidze N, Goodwillie A, Pfaff DW. Reduction of lipocalin-type prostaglandin D synthase in the preoptic area of female mice mimics estradiol effects on arousal and sex behavior. Proc. Natl Acad. Sci. USA. 2003;100:15206–15211. doi: 10.1073/pnas.2436540100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Simões-Wüst AP, Hopkins-Donaldson S, Sigrist B, Belyanskaya L, Stahel RA, Zangemeister-Wittke U. A Functionally improved locked nucleic acid antisense oligonucleotide inhibits BCL-2 and BCL-XL Expression and facilitates tumor cell apoptosis. Oligonucleotides. 2004;14:1–11. doi: 10.1089/oli.2004.14.199. [DOI] [PubMed] [Google Scholar]
- 32.Krzystanek M, Pedersen TX, Bartels ED, Kjaehr J, Straarup EM, Nielsen LB. Expression of apolipoprotein B in the kidney attenuates renal lipid accumulation. J. Biol. Chem. 2010;285:10583–10590. doi: 10.1074/jbc.M109.078006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Watanabe TA, Geary RS, Levin AA. Plasma protein binding of an antisense oligonucleotide targeting human ICAM-1 (ISIS 2302) Oligonucleotides. 2006;16:169–180. doi: 10.1089/oli.2006.16.169. [DOI] [PubMed] [Google Scholar]
- 34.Gao WY, Han FS, Storm C, Egan W, Cheng YC. Phosphorothioate oligonucleotides are inhibitors of human DNA polymerases and RNAse H: implications for antisense technology. Mol. Pharmacol. 1992;41:223–229. [PubMed] [Google Scholar]
- 35.Stein CA, Hansen JB, Lai J, Wu S, Voskresenskiy A, Hog A, Worm J, Hedtjarn M, Souleimanian N, Miller P, et al. Efficient gene silencing by delivery of locked nucleic acid antisense oligonucleotides, unassisted by transfection reagents. Nucleic Acids Res. 2010;38:e3. doi: 10.1093/nar/gkp841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Akdim F, Stroes ES, Kastelein JJ. Antisense apolipoprotein B therapy: where do we stand? Curr. Opin. Lipidol. 2007;18:397–400. doi: 10.1097/MOL.0b013e328248b4ad. [DOI] [PubMed] [Google Scholar]
- 37.Kastelein JJ, Wedel MK, Baker BF, Su J, Bradley JD, Yu RZ, Chuang E, Graham MJ, Crooke RM. Potent reduction of apolipoprotein B and low-density lipoprotein cholesterol by short-term administration of an antisense inhibitor of apolipoprotein B. Circulation. 2006;114:1729–1735. doi: 10.1161/CIRCULATIONAHA.105.606442. [DOI] [PubMed] [Google Scholar]
- 38.Yu RZ, Lemonidis KM, Graham MJ, Matson JE, Crooke RM, Tribble DL, Wedel MK, Levin AA, Geary RS. Cross-species comparison of in vivo PK/PD relationships for second-generation antisense oligonucleotides targeting apolipoprotein B-100. Biochem. Pharmacol. 2009;77:910–919. doi: 10.1016/j.bcp.2008.11.005. [DOI] [PubMed] [Google Scholar]
- 39.Schonfeld G, Patterson BW, Yablonskiy DA, Tanoli TS, Averna M, Elias N, Yue P, Ackerman J. Fatty liver in familial hypobetalipoproteinemia: triglyceride assembly into VLDL particles is affected by the extent of hepatic steatosis. J. Lipid Res. 2003;44:470–478. doi: 10.1194/jlr.M200342-JLR200. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.