Abstract
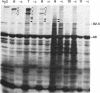
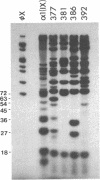
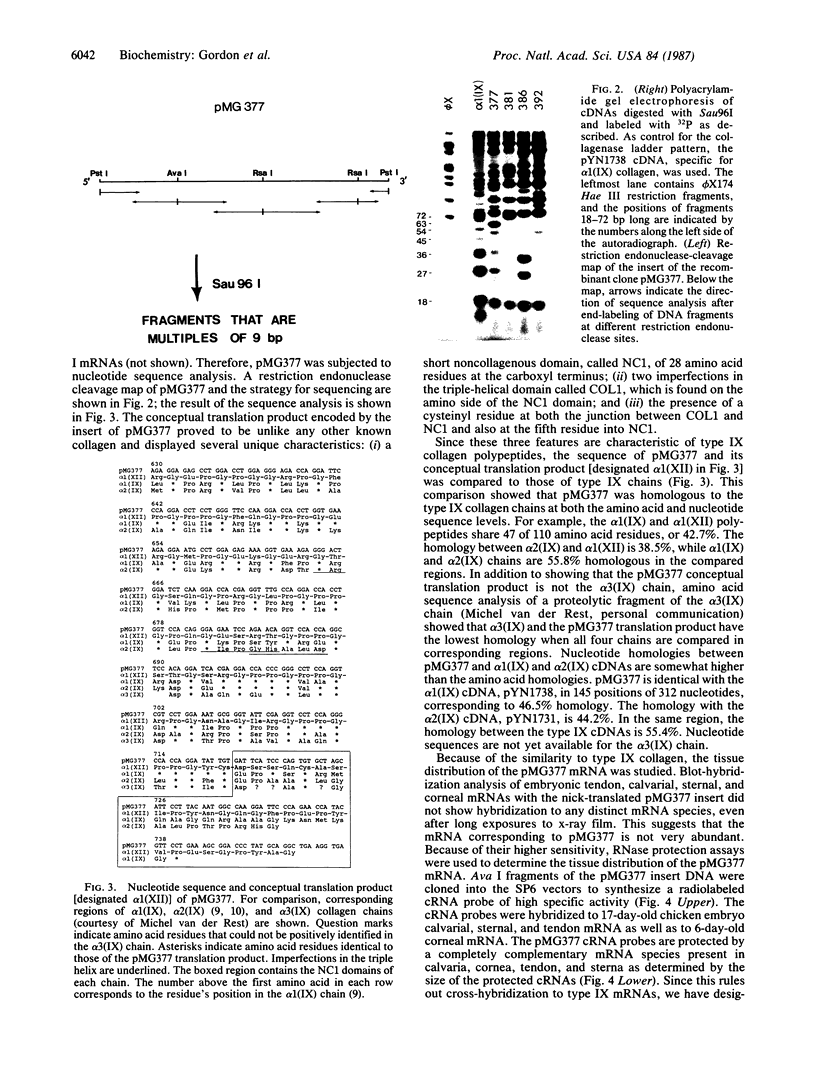
We have screened a cDNA library constructed from tendon fibroblast mRNA for the presence of collagenous coding sequences. Nucleotide sequence analysis of one isolated clone, pMG377, reveals that the clone encodes a polypeptide that is homologous to, yet distinctly different from, type IX short-chain collagen polypeptides. The structure of the conceptual translation product of the cDNA is also different from that of all other collagen types. Therefore, we have given the type IX-like collagen chain encoded by pMG377 the designation alpha 1(XII). Ribonuclease protection assays with single-stranded cRNA probes demonstrate that alpha 1(XII) mRNA is present in several tissues such as calvaria, tendon, and sternal cartilage of 17-day-old chicken embryo and in cornea from 6-day-old embryos. Using pMG377 as the hybridization probe, we isolated a fragment of the corresponding gene from a chicken genomic library. Partial nucleotide sequence analysis of the genomic clone DG12 shows that the exon/intron structure of the alpha 1(XII) collagen gene appears to be homologous to that of the alpha 1(IX) and alpha 2(IX) collagen genes. Our data demonstrate that types IX and XII collagen are two homologous members of a family of unique collagenous proteins that show tissue-specific patterns of expression. Based on their structure and the properties of their genes, we conclude that this family of collagens is distinctly different from that of fibrillar collagens.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aviv H., Leder P. Purification of biologically active globin messenger RNA by chromatography on oligothymidylic acid-cellulose. Proc Natl Acad Sci U S A. 1972 Jun;69(6):1408–1412. doi: 10.1073/pnas.69.6.1408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baron M. H., Maniatis T. Rapid reprogramming of globin gene expression in transient heterokaryons. Cell. 1986 Aug 15;46(4):591–602. doi: 10.1016/0092-8674(86)90885-8. [DOI] [PubMed] [Google Scholar]
- Bruckner P., Vaughan L., Winterhalter K. H. Type IX collagen from sternal cartilage of chicken embryo contains covalently bound glycosaminoglycans. Proc Natl Acad Sci U S A. 1985 May;82(9):2608–2612. doi: 10.1073/pnas.82.9.2608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Butler E. T., Chamberlin M. J. Bacteriophage SP6-specific RNA polymerase. I. Isolation and characterization of the enzyme. J Biol Chem. 1982 May 25;257(10):5772–5778. [PubMed] [Google Scholar]
- Cantatore P., Attardi G. Mapping of nascent light and heavy strand transcripts on the physical map of HeLa cell mitochondrial DNA. Nucleic Acids Res. 1980 Jun 25;8(12):2605–2625. doi: 10.1093/nar/8.12.2605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chirgwin J. M., Przybyla A. E., MacDonald R. J., Rutter W. J. Isolation of biologically active ribonucleic acid from sources enriched in ribonuclease. Biochemistry. 1979 Nov 27;18(24):5294–5299. doi: 10.1021/bi00591a005. [DOI] [PubMed] [Google Scholar]
- Gubler U., Hoffman B. J. A simple and very efficient method for generating cDNA libraries. Gene. 1983 Nov;25(2-3):263–269. doi: 10.1016/0378-1119(83)90230-5. [DOI] [PubMed] [Google Scholar]
- Holmes D. S., Quigley M. A rapid boiling method for the preparation of bacterial plasmids. Anal Biochem. 1981 Jun;114(1):193–197. doi: 10.1016/0003-2697(81)90473-5. [DOI] [PubMed] [Google Scholar]
- King J., Laemmli U. K. Polypeptides of the tail fibres of bacteriophage T4. J Mol Biol. 1971 Dec 28;62(3):465–477. doi: 10.1016/0022-2836(71)90148-3. [DOI] [PubMed] [Google Scholar]
- Konomi H., Seyer J. M., Ninomiya Y., Olsen B. R. Peptide-specific antibodies identify the alpha 2 chain as the proteoglycan subunit of type IX collagen. J Biol Chem. 1986 May 25;261(15):6742–6746. [PubMed] [Google Scholar]
- Lozano G., Ninomiya Y., Thompson H., Olsen B. R. A distinct class of vertebrate collagen genes encodes chicken type IX collagen polypeptides. Proc Natl Acad Sci U S A. 1985 Jun;82(12):4050–4054. doi: 10.1073/pnas.82.12.4050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melton D. A., Krieg P. A., Rebagliati M. R., Maniatis T., Zinn K., Green M. R. Efficient in vitro synthesis of biologically active RNA and RNA hybridization probes from plasmids containing a bacteriophage SP6 promoter. Nucleic Acids Res. 1984 Sep 25;12(18):7035–7056. doi: 10.1093/nar/12.18.7035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller E. J. The structure of fibril-forming collagens. Ann N Y Acad Sci. 1985;460:1–13. doi: 10.1111/j.1749-6632.1985.tb51152.x. [DOI] [PubMed] [Google Scholar]
- Ninomiya Y., Gordon M., van der Rest M., Schmid T., Linsenmayer T., Olsen B. R. The developmentally regulated type X collagen gene contains a long open reading frame without introns. J Biol Chem. 1986 Apr 15;261(11):5041–5050. [PubMed] [Google Scholar]
- Ninomiya Y., Olsen B. R. Synthesis and characterization of cDNA encoding a cartilage-specific short collagen. Proc Natl Acad Sci U S A. 1984 May;81(10):3014–3018. doi: 10.1073/pnas.81.10.3014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ninomiya Y., van der Rest M., Mayne R., Lozano G., Olsen B. R. Construction and characterization of cDNA encoding the alpha 2 chain of chicken type IX collagen. Biochemistry. 1985 Jul 16;24(15):4223–4229. doi: 10.1021/bi00336a061. [DOI] [PubMed] [Google Scholar]
- Noro A., Kimata K., Oike Y., Shinomura T., Maeda N., Yano S., Takahashi N., Suzuki S. Isolation and characterization of a third proteoglycan (PG-Lt) from chick embryo cartilage which contains disulfide-bonded collagenous polypeptide. J Biol Chem. 1983 Aug 10;258(15):9323–9331. [PubMed] [Google Scholar]
- Overbeek P. A., Merlino G. T., Peters N. K., Cohn V. H., Moore G. P., Kleinsmith L. J. Characterization of five members of the actin gene family in the sea urchin. Biochim Biophys Acta. 1981 Dec 28;656(2):195–205. doi: 10.1016/0005-2787(81)90087-3. [DOI] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Southern E. M. Detection of specific sequences among DNA fragments separated by gel electrophoresis. J Mol Biol. 1975 Nov 5;98(3):503–517. doi: 10.1016/s0022-2836(75)80083-0. [DOI] [PubMed] [Google Scholar]
- Takanami M. RNA polymerase nascent product analysis. Methods Enzymol. 1980;65(1):497–499. doi: 10.1016/s0076-6879(80)65058-7. [DOI] [PubMed] [Google Scholar]
- Vaughan L., Winterhalter K. H., Bruckner P. Proteoglycan Lt from chicken embryo sternum identified as type IX collagen. J Biol Chem. 1985 Apr 25;260(8):4758–4763. [PubMed] [Google Scholar]
- Yamauchi M., Kuboki Y., Sasaki S., Mechanic G. L. New pepsin-solubilized low molecular weight collagenous component possibly unique to periodontal ligament. Biochemistry. 1986 Apr 22;25(8):1997–2002. doi: 10.1021/bi00356a024. [DOI] [PubMed] [Google Scholar]
- Zinn K., DiMaio D., Maniatis T. Identification of two distinct regulatory regions adjacent to the human beta-interferon gene. Cell. 1983 Oct;34(3):865–879. doi: 10.1016/0092-8674(83)90544-5. [DOI] [PubMed] [Google Scholar]
- van der Rest M., Mayne R., Ninomiya Y., Seidah N. G., Chretien M., Olsen B. R. The structure of type IX collagen. J Biol Chem. 1985 Jan 10;260(1):220–225. [PubMed] [Google Scholar]