Abstract
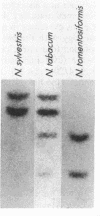
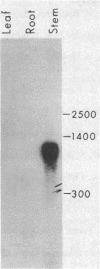
Plant peroxidases play a major role in lignin formation and wound healing and are believed to be involved in auxin catabolism and defense to pathogen attack. The function of the anionic peroxidase isozymes is best understood in tobacco. These isozymes catalyze the formation of the lignin polymer and form rigid cross-links between lignin, cellulose, and extensin in the secondary plant cell wall. We report the purification of the anionic peroxidase isozymes from tobacco and their partial amino acid sequence. An oligonucleotide probe deduced from the amino acid sequence was used to screen a tobacco leaf cDNA library and a 1200-base-pair cDNA clone was isolated and sequenced in its entirety. The predicted amino acid sequence revealed a 22-amino acid signal peptide and a 302-amino acid mature protein (Mr, 32,311). The amino acid sequence was compared to that of the cationic peroxidases from horseradish and turnip and was found to be 52% and 46% homologous, respectively. By RNA blot analysis, the messenger for the tobacco isozyme was found to be abundant in stem tissue while expressed at very low levels in leaf and root tissue. Four distinguishable copies of the gene were found on genomic DNA blots. The gene copy number may reflect the allotetraploid nature of Nicotiana tabacum.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Benton W. D., Davis R. W. Screening lambdagt recombinant clones by hybridization to single plaques in situ. Science. 1977 Apr 8;196(4286):180–182. doi: 10.1126/science.322279. [DOI] [PubMed] [Google Scholar]
- Birecka H., Miller A. Cell wall and protoplast isoperoxidases in relation to injury, indoleacetic Acid, and ethylene effects. Plant Physiol. 1974 Apr;53(4):569–574. doi: 10.1104/pp.53.4.569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Espelie K. E., Franceschi V. R., Kolattukudy P. E. Immunocytochemical localization and time course of appearance of an anionic peroxidase associated with suberization in wound-healing potato tuber tissue. Plant Physiol. 1986 Jun;81(2):487–492. doi: 10.1104/pp.81.2.487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fitzgerald M., Shenk T. The sequence 5'-AAUAAA-3'forms parts of the recognition site for polyadenylation of late SV40 mRNAs. Cell. 1981 Apr;24(1):251–260. doi: 10.1016/0092-8674(81)90521-3. [DOI] [PubMed] [Google Scholar]
- Hattori M., Sakaki Y. Dideoxy sequencing method using denatured plasmid templates. Anal Biochem. 1986 Feb 1;152(2):232–238. doi: 10.1016/0003-2697(86)90403-3. [DOI] [PubMed] [Google Scholar]
- Lapeyre B., Amalric F. A powerful method for the preparation of cDNA libraries: isolation of cDNA encoding a 100-kDal nucleolar protein. Gene. 1985;37(1-3):215–220. doi: 10.1016/0378-1119(85)90275-6. [DOI] [PubMed] [Google Scholar]
- Lehrach H., Diamond D., Wozney J. M., Boedtker H. RNA molecular weight determinations by gel electrophoresis under denaturing conditions, a critical reexamination. Biochemistry. 1977 Oct 18;16(21):4743–4751. doi: 10.1021/bi00640a033. [DOI] [PubMed] [Google Scholar]
- Mazza G., Welinder K. G. Covalent structure of turnip peroxidase 7. Cyanogen bromide fragments, complete structure and comparison to horseradish peroxidase C. Eur J Biochem. 1980 Jul;108(2):481–489. doi: 10.1111/j.1432-1033.1980.tb04745.x. [DOI] [PubMed] [Google Scholar]
- Ohtsuka E., Matsuki S., Ikehara M., Takahashi Y., Matsubara K. An alternative approach to deoxyoligonucleotides as hybridization probes by insertion of deoxyinosine at ambiguous codon positions. J Biol Chem. 1985 Mar 10;260(5):2605–2608. [PubMed] [Google Scholar]
- Rigby P. W., Dieckmann M., Rhodes C., Berg P. Labeling deoxyribonucleic acid to high specific activity in vitro by nick translation with DNA polymerase I. J Mol Biol. 1977 Jun 15;113(1):237–251. doi: 10.1016/0022-2836(77)90052-3. [DOI] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Southern E. M. Detection of specific sequences among DNA fragments separated by gel electrophoresis. J Mol Biol. 1975 Nov 5;98(3):503–517. doi: 10.1016/s0022-2836(75)80083-0. [DOI] [PubMed] [Google Scholar]
- Thomas P. S. Hybridization of denatured RNA and small DNA fragments transferred to nitrocellulose. Proc Natl Acad Sci U S A. 1980 Sep;77(9):5201–5205. doi: 10.1073/pnas.77.9.5201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Welinder K. G. Amino acid sequence studies of horseradish peroxidase. Amino and carboxyl termini, cyanogen bromide and tryptic fragments, the complete sequence, and some structural characteristics of horseradish peroxidase C. Eur J Biochem. 1979 Jun 1;96(3):483–502. doi: 10.1111/j.1432-1033.1979.tb13061.x. [DOI] [PubMed] [Google Scholar]
- Wood W. I., Gitschier J., Lasky L. A., Lawn R. M. Base composition-independent hybridization in tetramethylammonium chloride: a method for oligonucleotide screening of highly complex gene libraries. Proc Natl Acad Sci U S A. 1985 Mar;82(6):1585–1588. doi: 10.1073/pnas.82.6.1585. [DOI] [PMC free article] [PubMed] [Google Scholar]