Abstract
Cl− channels in the apical membrane of biliary epithelial cells (BECs) provide the driving force for ductular bile formation. Although a cystic fibrosis transmembrane conductance regulator has been identified in BECs and contributes to secretion via secretin binding basolateral receptors and increasing [cAMP]i, an alternate Cl− secretory pathway has been identified that is activated via nucleotides (ATP, UTP) binding apical P2 receptors and increasing [Ca2+]i. The molecular identity of this Ca2+-activated Cl− channel is unknown. The present studies in human, mouse, and rat BECs provide evidence that TMEM16A is the operative channel and contributes to Ca2+-activated Cl− secretion in response to extracellular nucleotides. Furthermore, Cl− currents measured from BECs isolated from distinct areas of intrahepatic bile ducts revealed important functional differences. Large BECs, but not small BECs, exhibit cAMP-stimulated Cl− currents. However, both large and small BECs express TMEM16A and exhibit Ca2+-activated Cl− efflux in response to extracellular nucleotides. Incubation of polarized BEC monolayers with IL-4 increased TMEM16A protein expression, membrane localization, and transepithelial secretion (Isc). These studies represent the first molecular identification of an alternate, noncystic fibrosis transmembrane conductance regulator, Cl− channel in BECs and suggest that TMEM16A may be a potential target to modulate bile formation in the treatment of cholestatic liver disorders.
Keywords: Anion Transport, ATP, Calcium, Chloride Channels, Epithelial Cell, Liver, Purinergic Receptor, Cholangiocyte, TMEM16A, Bile Duct
Introduction
The formation of bile by the liver depends on the transport functions of two complimentary cell types: hepatocytes and intrahepatic bile duct epithelial cells, known as cholangiocytes. Although cholangiocytes account for only ∼2–5% of the nuclear mass of the liver (1), they have a prodigious capacity for secretion (2, 3) and may account for up to 40% of bile volume in humans through regulated ion and water transport (4). Opening of Cl− channels in the apical membrane of cholangiocytes represents the driving force for ductular secretion and at least two Cl− conductances contribute (5). The best characterized pathway involves cAMP-stimulated opening of Cl− channels encoded by the cystic fibrosis transmembrane conductance regulator (CFTR)2 (5, 6). Additionally, there is a separate Ca2+-activated Cl− channel (CaCC) that is involved, but the molecular identity of this channel has not been defined (5, 7).
Increasing evidence suggests that the dominant mechanism for regulation of biliary secretion involves (i) autocrine/paracrine release of ATP into bile, (ii) activation by ATP of purinergic receptors, and (iii) mobilization of intracellular calcium and activation of CaCCs. The magnitude of the Cl− secretory response to ATP is greater than that to cAMP in single human biliary epithelial cells and polarized rat cholangiocyte monolayers (8). Furthermore, many of the effects of cAMP on bile formation appear to be mediated by ATP release into the duct lumen and subsequent activation of apical P2 receptors (9, 10). These general findings are consistent with the clinical observation that only 10–20% of patients with cystic fibrosis (CF) develop clinically significant liver disease despite absent or abnormal CFTR in biliary epithelium (11, 12). Furthermore, in cells with the CF phenotype, activation of P2 receptors still elicits potent secretory responses (13, 14). Together, these findings challenge the conventional model that centers on the concept that cAMP-dependent opening of CFTR-related Cl− channels is the driving force for cholangiocyte secretion. Rather, the operative regulatory pathways appear to take place within the lumen of intrahepatic ducts, where release of ATP into bile is a final common pathway controlling ductular bile formation. Accordingly, molecular definition of the CaCC channel(s) involved has a high priority for future efforts to modulate the volume and composition of bile.
Several candidate proteins have been proposed as CaCCs in other epithelia, including CLC and bestrophin family members (15). However, the biophysical properties of the currents associated with these proteins are not consistent with the ATP-stimulated Cl− currents described in biliary epithelium (8). More recently, TMEM16A, a 114-kDa membrane protein with eight putative transmembrane domains, has been identified as a CaCC in other epithelium (16–18), and the general biophysical properties of the TMEM16A conductance as measured by whole cell patch clamp appear similar to the ATP-stimulated Cl− currents described in biliary cells (8). Although TMEM16A is found in liver (19), its cellular localization and function are unknown.
The aim of the present studies was to identify the molecular basis for Ca2+-activated Cl− currents stimulated by extracellular nucleotides in different biliary cell models. We have utilized single cells (human and mouse), polarized biliary monolayers (rat), and novel models of small and large cholangiocytes isolated from distinct areas of the intrahepatic bile ducts (mouse) to critically assess the potential role of TMEM16A in regulating membrane Cl− permeability and biliary secretion. Lastly, we have explored the potential role of IL-4 on TMEM16A channel expression and function, which may have important implications for Cl− secretion during cholestasis.
EXPERIMENTAL PROCEDURES
Cell Models
Studies in isolated cells were performed using human Mz-Cha-1 cells and mouse cholangiocytes, whereas studies in polarized monolayers were performed utilizing normal rat cholangiocytes (NRCs). Mz-Cha-1, originally isolated from human cholangiocarcinoma cells exhibit phenotypic features of differentiated biliary epithelium (20, 21) and have been utilized as models for purinergic signaling (22) and Ca2+-dependent secretion (23–26). Mouse large cholangiocytes (MLCs) and mouse small cholangiocytes (MSCs) were isolated from normal mice (BALB/c) and immortalized by transfection with the SV40 large T antigen gene as described (27). These cells demonstrate similar properties to those of freshly isolated small and large mouse (28) and rat (29, 30) cholangiocytes. NRC monolayers isolated from intrahepatic bile ducts (31) were cultured on rat tail collagen slabs as described previously (32, 33) and passaged onto collagen-coated semipermeable (24-mm diameter, 0.4-μm pore) transwell supports (Costar Corning, Acton, MA) 7–10 days before electrophysiological and molecular studies. This protocol permits highly polarized cells and the development of a high transepithelial resistance (Rt > 1,000 ohms · cm2) (32, 33). Localization of TMEM16A was performed in whole liver sections from normal and 7-day bile duct-ligated (BDL) rats and mice (Charles River, Wilmington, MA) as described previously (34, 35). All animal experiments were performed in accordance with a protocol approved by the Scott and White Institutional Animal Care and Use Committee and in accordance with the Guide for the Care and Use of Laboratory Animals published by the National Institutes of Health (No. 85-23).
Detection of TMEM16A
PCR products representing the major isoforms of TMEM16 were detected utilizing specific primers (supplemental Table S1) and oligo(dT) primer and Superscript RNase H-reverse transcriptase (Invitrogen). The reaction mixtures (25 μl) contained 2 μl of cDNA, 1 μl of deoxyribonucleotide triphosphate (10 mmol/liter), 2.5 μl of 10× buffer, 1.5 μl of MgCl2 (25 mmol/liter), 2.5 units Taq Polymerase (Invitrogen), and 50 pmol of each primer. The mixture was subjected to 30 cycles of amplification (94 °C for 60 s; denaturation, 60 °C for 60 s annealing and 72°C for 60 s extension). Quantitative assessment was determined by real-time PCR using the ABI Prism 7900HT Sequence detection system (Applied Biosystems, Invitrogen). Amplification reactions were performed in a final volume of 10 μl containing cDNA from 10 ng of reverse-transcribed total RNA, 150 nm each of forward and reverse primers (supplemental Table S2) and SYBR Green PCR Master Mix (Applied Biosystems). TMEM16A protein was detected by Western blot utilizing goat polyclonal TMEM16A antibody (S-20, catalog no. sc-69343, Santa Cruz Biotechnology, Santa Cruz, CA). Signals were visualized via enhanced chemiluminescence detection (Pierce) and captured with a digital image system (Chemigenius2 photo-documentation system; Syngene, Cambridge, UK), and band intensity was quantified using standard software (GeneTool, Syngene).
Immunofluorescence
Localization of TMEM16A protein was performed in confluent NRC monolayers and in whole liver sections from normal and BDL rats and mice. NRC on collagen-coated tissue culture chamber slides (BD BioCoat) were fixed in 25% acetic acid/75% ethanol (v/v) for 10 min or 4% paraformaldehyde permeabilized with 1% Triton X-100 for 10 min, incubated with 5% normal donkey serum, and then incubated overnight at 4 °C with rabbit anti-TMEM16A antibody (1:100, ab53212, Abcam, Cambridge, MA) and then Dylight 488 conjugated donkey anti-rabbit (Jackson ImmunoResearch Laboratories, 1:600) and counterlabeled with 300 nm DAPI (Molecular Probes, Eugene, OR) or Alexa Fluor 555 phalloidin in separate studies. Control cells were prepared by omitting either primary or secondary antibodies from the incubation solution. The slides were coverslipped with Mowiol 4–88 with 2.5% DABCO and left overnight in the dark at room temperature. The slides were imaged using the Leica TCS SP5 confocal microscope (Leica Micro-systems, CMS GMBH) with custom software (Leica Micro-systems LAS AF). Z-stacks were captured using a step size of 0.35 μm to cover the entire thickness of the sample. Images were acquired using a frame size of 512 × 512 pixels, and four line averaging was used to remove noise from the image. Two channels (Green/Blue) were acquired via the sequential scan option with three photomultiplier tubes. All images were captured using the same electronic settings. Images were then imported in ImageJ (http://rsb.info.nih.gov) using the LOCI Bio-formats plug-in (University of Wisconsin, Madison), and fluorescence intensity was measured in individual cells and confluent monolayers during control and stimulated conditions.
Analysis of TMEM16A expression was performed in frozen liver sections (10 μm) from normal and 7-day BDL rats and mice. Sections were defrosted at room temperature and fixed in 4% paraformaldehyde (1× PBS), permeabilized in PBST (1× PBS with 0.2% Triton), and blocked in 4% BSA (in PBST) for 1 h at room temperature. Primary antibodies for goat anti-cytokeratin (CK)-19 (1:5, rat sections), mouse anti-CK-7 (Santa Cruz Biotechnology, mouse sections) and rabbit anti-TMEM16A antibody (1:100, antibody ab53212, Abcam) were diluted in 1% BSA. Sections were incubated overnight at 4 °C and washed three times for 10 min each with 1× PBST at room temperature. Sections were incubated with Cy3-conjugated anti-goat (1:50) or Cy3-conjugated anti-mouse (1:50), and Dylight 488 conjugated donkey anti-rabbit secondary antibodies (1:600, Jackson ImmunoResearch Laboratories) for 2 h at RT protected from light. In selected experiments, sections were counterlabeled with Alexa Fluor 555 phalloidin. Following incubation, the slides were washed in PBST at room temperature and coverslipped with Antifade gold and DAPI. Images were visualized using an Olympus IX-71 (Tokyo, Japan) confocal microscope.
TMEM16A Silencing
TMEM16A was suppressed by specific anti-TMEM16A siRNA (TMEM16A-HSS123904). 25-nucleotide siRNAs were designed and synthesized by Invitrogen (antisense, AAG UUA GUG AGG UAG GCU GGG AAC C; sense, GGU UCC CAG CCU ACC UCA CUA ACU U) and transfected using FuGENE (5 μg/100 μl). Noncoding StealthTM RNAi (medium guanine cytosine duplex) (Invitrogen) was utilized in control (mock) transfections. Block-itTM Fluorescent Oligo (catalog no. 2013) from Invitrogen was used to optimize transfection conditions and for selection of transfected cells for whole-cell patch clamp current recording. Whole cell patch clamp experiments were done 24–48 h after transfection. NRC monolayers were transfected 2 h after cell plating. Short circuit currents were measured after 5–6 days of transfection. Transfection efficiency and the degree of TMEM16A silencing was evaluated in all models at message level by real-time PCR and at protein level by Western blot analysis.
Measurement of Cl− Currents
Membrane currents were measured via whole-cell patch clamp techniques. Cells on a coverslip were mounted in a chamber (volume ∼ 400 μl), and whole-cell currents were measured with a standard extracellular solution containing the following: 140 mm NaCl, 4 mm KCl, 1 mm CaCl2, 2 mm MgCl2, 1 mm KH2PO4, 10 mm glucose, and 10 mm HEPES/NaOH (pH ∼ 7.40). The standard intracellular (pipette) solution for whole-cell recordings contained the following: 130 mm KCl, 10 mm NaCl, 2 mm MgCl2, 10 mm HEPES/KOH, 0.5 mm CaCl2, 3 mm MgATP2−, and 1 mm EGTA (pH 7.3), corresponding to a free [Ca2+] of ∼100 nm (36). Free [Ca2+]i was calculated using software kindly provided by Guy Droogmans (Katholieke Universiteit Leuven). Patch pipettes were pulled from Corning 7052 glass and had a resistance of 2–5 megohms for the whole cell. Recordings were made with an Axopatch ID amplifier (Axon Instruments, Foster City, CA) and were filtered at 2 kHz and sampled at 4 kHz for storage on a computer and analyzed using pCLAMP (version 10, Axon Instruments, Burlingame, CA) as described previously (37, 38). Two voltage protocols were utilized: 1) a holding potential of −40 mV, with 100-ms steps to 0 and −80 mV at 10-s intervals (for real-time tracings) and 2) a holding potential −40 mV, with 100-ms steps from −100 to +100 mV in 20-mV increments. Current-voltage (I-V) relations were generated from the “step” protocol. Results are compared with control studies measured on the same day to minimize any effects of day-to-day variability and reported as current density (pA/pF) to normalize for differences in cell size (39). Concentration-response data for [Ca2+]i activation of whole-cell currents were fitted to the following equation: Y = Vmax × Xn/(Kn + Xn). Where Y is the current density, X is free [Ca2+]i, Vmax is the maximum current density at −80 mV, K is the half-maximum concentration of free [Ca2+]i, and n is the Hill coefficient.
Transepithelial Cl− Secretion
NRC monolayers were utilized to study integrated secretory responses. Cells were grown to confluence on collagen-treated polycarbonate filters with a pore size of 0.4 μm (Costar, Cambridge, MA) until resistance was >1,000 ohms·cm2 (EVOHM; World Precision Instruments, Sarasota, FL) (32). Cells were mounted in a Trans-24 mini-perfusion system for tissue culture cups (Jim's Instrument Manufacturing, Inc., Iowa City, Iowa). All experiments were carried out at 37 °C, and basolateral and apical (luminal) sides were bubbled with O2 through air-lift circulators. The standard extracellular buffer solution containing the following: 140 mm NaCl, 4 mm KCl, 1 mm KH2PO4, 2 mm MgCl2, 1 mm CaCl2, 5 mm glucose, and 10 mm HEPES/NaOH (pH 7.3). Transepithelial voltage (Vt) was clamped to 0 mV, and the short circuit current Isc was recorded through agar bridges (3% agar in 1 m KCl) connected to Ag-AgCl electrodes (cartridge electrodes, World Precision Instruments). The Isc represents the net sum of the transepithelial anion and cation fluxes and reflects the level of ion and fluid secretion (33). Studies included paired, same-day monolayers to minimize any potential effects of day-to-day variability.
Reagents
IL-4 was purchased from Cell Sciences (Canton, MA), and forskolin was obtained from Enzo Life Sciences (formerly BIOMOL). The CFTR inhibitor, CFTRinh172, was a kind gift from Dr. Nitin Sonawane and Dr. Alan Verkman (University of California, San Francisco, CA). All other reagents, including ATP and UTP, were obtained from Sigma-Aldrich.
Statistics
Results are presented as the means ± S.E., with n representing the number of culture plates or repetitions for each assay as indicated. Student's paired or unpaired t test or analysis of variance for multiple comparisons was used to assess statistical significance as indicated, and p values < 0.05 were considered to be statistically significant.
RESULTS
Expression of TMEM16A mRNA and Protein in Biliary Epithelia
Utilizing selective primers for the specific TMEM16 isoforms, PCR products of the predicted size for TMEM16A were detected in human, mouse, and rat biliary cells (Fig. 1). TMEM16F, TMEM16J, and TMEM16K were also identified in all biliary cells used in this study, whereas none of the models expressed TMEM16B. Additionally, Western blot analysis, utilizing a specific anti-TMEM16A antibody, detected a predicted protein band ∼ 114 kDa in all biliary cells (Fig. 1B). Immunostaining of both rat and mouse whole liver sections revealed only a weak signal in hepatocytes, but a strong signal in cholangiocytes (Fig. 1D and supplemental Fig. S1). To confirm the predominant cholangiocyte location, liver sections from BDL mice and rats, which results in cholangiocyte proliferation, were likewise labeled. These sections revealed strong TMEM16A signal in cholangiocytes but undetectable signal in hepatocytes. In these sections, as well as in polarized NRC monolayers, the TMEM16A signal was most prominent in apical compartments (Fig. 1, C and D, and supplemental Fig. S1). Collectively, these results demonstrate the presence of TMEM16A in biliary epithelia with a predominant apical membrane location.
FIGURE 1.
Expression and localization of TMEM16A in human Mz-Cha-1 cells, MSCs and MLCs, NRC monolayers, and mouse and rat whole liver. A, RT-PCR. Species-specific TMEM16A primers were used to detect TMEM16A expression in all models represented by a band size at ∼305 bp for human and ∼300 bp for mouse and rat (as described under “Experimental Procedures”). TMEM16 b, j, f, and k isoforms were also evaluated. A λ DNA-HindIII digest ladder was used to delineate the size of respective amplicons. B, Western blot utilizing polyclonal anti-TMEM16A antibody (as described under “Experimental Procedures”). TMEM16A protein (band at ∼114 kDa) is present in whole-cell lysates of Mz-Cha-1, MLC, MSC, and NRC cells; the loading control was β-actin. C, membrane localization of TMEM16A protein in polarized NRC monolayers. Staining with anti-TMEM16A antibody (green) demonstrates TMEM16A protein in both apical and basolateral membranes with predominance in apical compartments. Cell nuclei were counterlabeled with DAPI (blue). The lower panel represents the Z-axis view; arrows: A, apical; BL, basolateral membrane (scale bar, 10 μm). D, localization of TMEM16A in whole liver sections of BDL mice (top) and rat (bottom). TMEM16A (green) co-localizes with cholangiocyte markers CK19 (rat) and CK7 (mouse). Cell nuclei stained with DAPI (blue). The rat bile duct is outlined by a dotted line (top right panel). Scale bar, 10 μm.
TMEM16A Is a Ca2+-activated Cl− Channel in Human Biliary Cells
In human Mz-Cha-1 biliary cells under whole-cell patch clamp recording conditions, exposure to the Ca2+ ionophore, ionomycin, resulted in simultaneous increases in current density recorded at 0 and −80 mV, representing K+ and Cl− currents, respectively (Fig. 2 and supplemental Fig. S2) as described previously (5, 7, 25, 26). The sustained Cl− currents exhibited time-dependent activation at positive membrane potentials, a reversal potential of −15.2 mV (ECl− = 0 mV), and outward rectification. Exposure to the potent Ca2+-mobilizing agonists, ATP or UTP, resulted in currents with similar properties (Fig. 2B). Transfection of cells with anti-TMEM16A siRNA significantly decreased expression of TMEM16A mRNA by 70 ± 7% and protein by 77 ± 2% (Fig. 2C) and significantly inhibited Cl− currents in response to ionomycin (by 84 ± 5%), ATP (by 81 ± 5%), or UTP (by 57 ± 8%) compared with cells transfected with nontargeting siRNA (Fig. 2, D–F). Ca2+-activated K+ currents, measured at 0 mV, were not significantly affected by TMEM16A silencing. Together, these studies demonstrate that TMEM16A contributes to Ca2+-activated Cl− currents in human biliary cells.
FIGURE 2.
Ca2+-activated Cl− currents in human Mz-Cha-1 cells are mediated via TMEM16A. A and B, representative whole-cell recordings. Currents were measured during basal conditions and during exposure to ionomycin (2 μm; A) or ATP (100 μm; B). Currents measured at −80 mV (open circles), representing ICl−, and at 0 mV (closed circles), representing IK+, are shown. Voltage protocols shown in the inset. Maximum current for this study are indicated by black dots immediately below the trace. A voltage-step protocol (from a holding potential of −40 mV, 100-ms steps from −100 to +100 mV in 20-mV increments) was obtained at a(star) (basal), b(star) (maximal outward current response), and c(star) (maximal inward current response) as indicated. The I-V plots were generated from these protocols and shows the current-voltage relation during basal (●), ionomycin-, or ATP-stimulated conditions (maximal outward currents (K+) are represented by ▿, and maximal inward currents (Cl−) are represented by ○. C, representative Western blot and cumulative data demonstrating change in TMEM16A protein and mRNA levels in control cells, cells transfected with nontargeting siRNA (mock), and cells transfected with anti-TMEM16A siRNA (*, p < 0.05 versus control or mock levels). D and E, representative whole-cell recordings from cells transfected with anti-TMEM16A siRNA in response to ionomycin (2 μm; D) or ATP (100 μm; E). Voltage-step protocols were obtained at indicated points (a(star) and b(star)), shown below the trace, and plotted as an I-V relation (inset). F, cumulative data demonstrating maximal current density (−pA/pF) measured at −80 mV in response to ionomycin, ATP, and UTP (100 μm) in cells transfected with nontargeting siRNA (mock) or cells transfected with anti-TMEM16A siRNA (bars represent mean ± S.E.; n = 6 for ionomycin; n = 14 for ATP; n = 7 for UTP). *, p < 0.05 versus mock-transfected.
Heterogeneity in Mouse Cholangiocyte Cl− Channels
Cholangiocytes isolated from distinct areas of the intrahepatic biliary tree are functionally heterogeneous. In mouse and rat, the large cholangiocytes (∼14 μm) forming the large intrahepatic ducts express CFTR and AE2, whereas the small cholangiocytes (∼8 μm), forming the small upstream ducts, do not (29, 35, 40). To study the functional consequences of these expression studies, the biophysical properties of MSCs and MLCs were studied under whole-cell patch clamp conditions. Exposure of MLC to a cAMP mixture (forskolin, isobutylmethylxanthine, and cpt-cAMP) to activate CFTR resulted in large Cl− currents at −80 mV. Currents were not voltage-dependent, reversed at −13 mV (ECl− = 0), and demonstrated a linear, or only mildly outward rectifying, IV relation and were significantly inhibited by the CFTR channel blocker CFTRinh172 (Fig. 3, A and C). Subsequent exposure of cells to ATP resulted in instantaneous activation of Cl− currents with time-dependent activation and outward rectification. In some studies, the ATP-stimulated currents demonstrated an oscillatory pattern (∼30%). The magnitude of ATP-stimulated currents was 3-fold greater than those stimulated by the cAMP mixture (Fig. 3C). In contrast, exposure of MSC to the cAMP mixture failed to activate Cl− currents (Fig. 3B). However, subsequent exposure to ATP resulted in large currents as measured at −80 mV with identical biophysical properties to those in MLC including time-dependent activation, reversal near ECl−, and outward rectification. Thus, both small and large mouse cholangiocytes express TMEM16A (Fig. 1) and exhibit Ca2+-activated Cl− currents in response to ATP, but only large cholangiocytes exhibit cAMP-stimulated Cl− currents.
FIGURE 3.
Representative whole cell recordings from single MLC (A) and MSC (B) measured at −80 mV representing ICl−. Exposure to cAMP mixture (10 μm forskolin, 100 μm isobutylmethylxanthine, and 500 μm cpt-cAMP), ATP (100 μm), and CFTRinh172 (5 μm) is indicated by the bar. The voltage-step protocol (100-ms steps from −100 to +100 mV in 20-mV increments) was obtained at a(star), b(star), and c(star) as indicated and used to generate the I-V relation during cAMP mixture (●) and ATP-stimulated (○) conditions. C, left panel, maximum current density (−pA/pF) measured at −80 mV in response to cAMP mixture or ATP in MLC (n = 6) or MSC (n = 5). *, current density in response to ATP was significantly greater than cAMP mixture (p < 0.05). **, current density in response to cAMP mixture was significantly greater in MLC versus MSC (p < 0.05). #, current density in response to cAMP mixture was significantly inhibited by CFTRinh172 (p < 0.05). The right panel shows quantitative levels of TMEM16A mRNA by real-time PCR (p = not significant between MLC and MSC).
TMEM16A Represents Ca2+-activated Cl− Channel in Mouse Cholangiocytes
To characterize and identify the molecular basis of the ATP-stimulated Cl− currents in mouse cholangiocytes, whole-cell currents were measured in cells transfected with anti-TMEM16A siRNA and compared with nontransfected cells (control) and cells transfected with nontargeting siRNA (mock) (Fig. 4). First, in control or mock-transfected MSC, exposure to ATP resulted in characteristic Ca2+-activated Cl− currents, which were inhibited by the nonspecific Cl− channel blocker NPPB, and the Ca2+-activated Cl− channel blocker niflumic acid (Fig. 4A, 4B). In separate studies, transfection of MSC with anti-TMEM16A siRNA abolished the ATP-, UTP-, and ionomycin-stimulated currents compared with control or mock-transfected cells (Fig. 4, C and D, and supplemental Fig. S3). The efficiency of anti-TMEM16A siRNA on protein levels was assessed by Western blot and demonstrated a decrease of 79 ± 3% from mock-transfected cells, which correlated with a significant decrease in whole-cell Ca2+-activated Cl− currents of 72 ± 7% in response to ionomycin, of 71 ± 10% in response to ATP, and of 77 ± 10% in response to UTP.
FIGURE 4.
Characterization of ATP-stimulated Cl− currents in MSC. A, whole-cell currents were measured at −80 mV during basal conditions and during exposure to ATP (100 μm) (upper trace). A voltage-step protocol was obtained at a(star) (basal) and b(star) (maximal current response) as indicated (shown below the trace) and plotted as the I-V relation during basal (●) and ATP-stimulated (○) conditions. Middle plots show ATP-stimulated currents in the presence (bars below trace) or absence of the Cl− channel inhibitors NPPB (100 μm) or niflumic acid (100 μm). B, cumulative data demonstrating maximum current density (−pA/pF) measured at −80 mV (n = 4–9 each). *, NPPB and niflumic acid significantly blocked ATP-stimulated currents (p < 0.05 versus control for each). C, representative whole-cell current recording in response to ATP (100 μm) from MSC cells transfected with nontargeting siRNA (control-mock, top panel) or siRNA against TMEM16A (bottom panel). Voltage-step protocols obtained at a(star) (basal) and b(star) (maximal current response) as indicated. The I-V plots shown were generated from these protocols and demonstrate basal (●) and ATP-stimulated (○) conditions. Right panel, effect of anti-TMEM16A siRNA on protein expression, representative Western blot (top), and cumulative data (bottom). D, cumulative data demonstrating maximum current density (−pA/pF) measured at −80 mV in response to ATP (100 μm, n = 4), or UTP (100 μm, n = 4) in MSCs transfected with nontargeting siRNA (mock) or anti-TMEM16A siRNA. *, p < 0.05 versus mock transfection.
To determine Ca2+-sensitivity of Cl− current activation, whole-cell studies were performed with varying the Ca2+ concentration in the pipette. As shown in Fig. 5, current density increased with increasing the free [Ca2+] in the pipette. Maximal current activation occurred at a Ca2+ concentration of 5 μm, and the calculated K½-maximum was 668 ± 54 nm free [Ca2+] (Fig. 5B). As shown, the current-voltage relationship demonstrated outward rectification at submaximal calcium levels but became linear at high pipette Ca2+ levels, a characteristic of endogenous Ca2+-activated Cl− channels in other models (15). Transfection of cells with anti-TMEM16A siRNA significantly decreased expression of TMEM16A mRNA and protein (supplemental Fig. S4) and abolished the Ca2+-activated currents (Fig. 5, C and D). Together, the molecular analysis, biophysical properties, and pharmacological profile of these studies in single human and mouse cholangiocytes are consistent with previous reports of TMEM16A in other epithelia (16–18) and provide evidence that TMEM16A contributes to membrane Cl− permeability in response to Ca2+-mobilizing agonists in biliary epithelial cells.
FIGURE 5.
Biophysical properties of whole-cell Ca2+-activated Cl− currents in mouse cholangiocytes. A, representative whole-cell currents (utilizing voltage-step protocol; see “Experimental Procedures”) recorded from MLC cells in response to increasing [Ca2+]i in pipette solution and corresponding I-V relation plotted on the right. B, [Ca2+]i dose-response curve for Cl− currents. Data were plotted from maximum current density (pA/pF) measured at −80 mV in response to different intracellular (pipette) Ca2+ concentrations. Each point represents mean ± S.E. (n = 3–5 each) fit to the Hill equation (see “Experimental Procedures”). C, anti-TMEM16A siRNA inhibits Ca2+-activated Cl− currents. Representative spontaneous whole-cell currents, measured at −80 mV, in response to 1 μm free Ca2+ in pipette solution from MLC cells transfected with nontargeting siRNA (mock, ○) or anti-TMEM16A siRNA (●). D, (cumulative data) values represent maximum current density measured at −80 mV in response to 1 μm free Ca2+ in pipette (n = 6–7 each). *, p < 0.05 versus mock.
TMEM16A Contributes to Transepithelial Secretion in Polarized Biliary Monolayers
To determine the potential contribution of TMEM16A to integrated secretory responses, studies were performed in polarized NRC monolayers. First, NRC monolayers express TMEM16A with enrichment in the apical membrane (Fig. 1, A and C). Next, confluent NRC monolayers were placed in an Ussing chamber, and Isc was measured in response to ATP/UTP. In control monolayers, addition of ATP to the apical membrane resulted in a large increase in Isc, which was unaffected by the CFTR inhibitor, CFTRinh172 but was inhibited by the nonspecific Cl− channel inhibitor NPPB or the Ca2+-activated Cl− channel inhibitor niflumic acid (Fig. 6, A and B). To confirm that the transepithelial secretory response is mediated by TMEM16A, NRC monolayers were transfected with anti-TMEM16A siRNA or nontargeted siRNA (mock), and Isc was again recorded in response to extracellular nucleotides. The efficiency of gene silencing by anti-TMEM16A siRNA was assessed by real-time PCR and demonstrated a 76 ± 19% decrease in TMEM16A levels, which correlated with a 54 ± 3% decrease in the ATP-stimulated Isc and a 57 ± 3% decrease in UTP-stimulated Isc (Fig. 6, C and D). Together, these studies in polarized cholangiocyte monolayers demonstrate that TMEM16A contributes to ATP- and UTP-stimulated transepithelial secretion.
FIGURE 6.
TMEM16A contributes to transepithelial secretion in polarized cholangiocyte monolayers. Short circuit current (Isc) across NRC monolayers was measured under voltage clamp conditions in an Ussing chamber. In these studies, agonists were added to the apical chamber. A, representative recording. Addition of ATP (200 μm) increased Isc, which was unaffected by CFTRinh172 (5 μm) but inhibited by NPPB (100 μm). B, cumulative data representing change in Isc in response to the addition of ATP in the presence or absence of the Cl− channel inhibitors. The y axis values are reported as ΔIsc (maximum Isc − basal Isc). *, NPPB and niflumic acid (n. acid) significantly inhibited the ATP-stimulated Isc (p < 0.05, n = 3 each). C, representative recordings of short circuit current Isc stimulated by UTP (100 μm) in control NRC (black) or NRC-transfected with anti-TMEM16A siRNA (blue). Anti-TMEM16A siRNA significantly decreased TMEM16A mRNA expression (inset). D, average change in Isc after addition of UTP or ATP in control NRC (mock) or NRC transfected with anti-TMEM16A siRNA. The y axis values are reported as ΔIsc (maximum Isc − basal Isc). *, p < 0.05 versus mock transfected, n = 5–6 each.
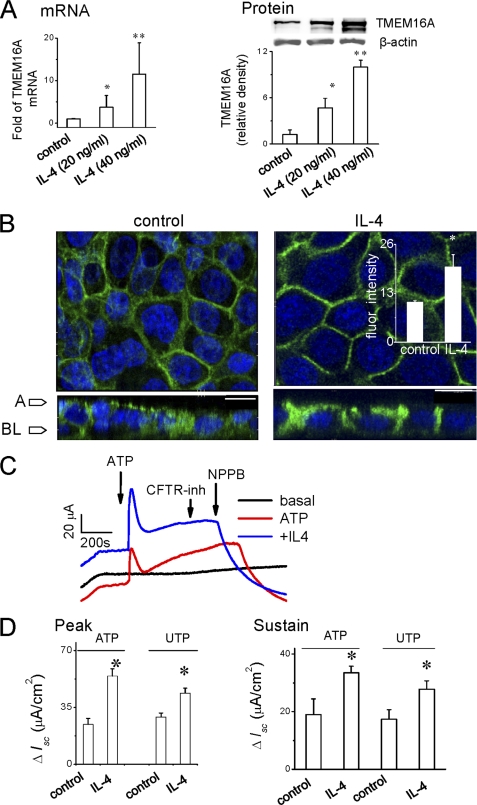
TMEM16A Is Regulated by IL-4
It has previously been demonstrated that exposure of respiratory epithelial cells to IL-4 increases TMEM16A expression and Ca2+-activated Cl− secretion (16). If similar effects are observed in biliary epithelium, it may have implications during cholestasis where inflammatory cytokines exert potent effects on hepatobiliary channels and transporters (41). To determine the effects of IL-4 on TMEM16A and transepithelial secretion Isc in biliary epithelium, polarized NRC monolayers were cultured on transwell filters with or without overnight treatment with IL-4, mounted in an Ussing chamber, and exposed to ATP or UTP, and Isc was measured. The results (Fig. 7) reveal that IL-4 exposure increased (i) relative TMEM16A mRNA and protein in a dose-dependent manner, (ii) membrane localization of TMEM16A, and (iii) the magnitude of both peak and sustained nucleotide-stimulated Isc compared with control (untreated) monolayers. The ATP- and UTP-stimulated increase in Isc in IL-4-treated monolayers was abolished by NPPB or niflumic acid but was unaffected by CFTRinh172 (supplemental Fig. S5). Thus, IL-4 increases cholangiocyte plasma membrane expression of TMEM16A and increases transepithelial secretion in response to extracellular nucleotides.
FIGURE 7.
IL-4 increases TMEM16A expression and transepithelial secretion in polarized NRC monolayers. A, TMEM16A mRNA and protein levels assessed by real-time PCR (left) and Western blot (right), respectively, with or without overnight treatment with IL-4 (20 or 40 ng/ml). Representative Western blot is shown in the inset; β-actin was used as the loading control. *, p < 0.05 versus control monolayers (without IL-4, n = 3 each). **, p < 0.05 versus control monolayers (n = 3–4 each). B, localization of TMEM16A protein in NRC monolayers. Labeling with anti-TMEM16A antibody (green) demonstrates presence of TMEM16A protein on cell membrane with predominant apical location (left panel). Nuclei were counterlabeled with DAPI (blue). Overnight treatment with IL-4 (40 ng/ml) increased membrane expression of TMEM16A. The inset shows the relative increase in green (TMEM16A) fluorescence intensity in IL-4 treated cells versus control (n = 31–36, p < 0.01). The Z-axis views are shown below panels; arrows: A, apical; BL, basolateral membrane (scale bar, 10 μm). C, representative short circuit current Isc measurements under basal (nonstimulated) conditions (black) and in response to ATP (200 μm) in control NRC (red) and after overnight treatment with IL-4 (10 ng/ml) (blue). In these representative recordings, agonists were added to the apical chamber at the indicated time points. D, average change in Isc in response to ATP or UTP in control (nontreated) and IL-4-treated monolayers. The y axis values are reported as ΔIsc (maximum Isc − basal Isc). The left panel shows peak currents, and the right panel shows sustained currents. *, IL-4 significantly increased Isc both at peak and sustained level versus control (without IL-4) monolayers (n = 6–12 each, p < 0.05).
DISCUSSION
In these studies of human, rat, and mouse biliary epithelial cells, we have identified TMEM16A as the molecular basis for Ca2+-activated Cl− channels stimulated by extracellular nucleotides. Furthermore, we have identified the mechanism by which small cholangiocytes, which do not express CFTR (35), contribute to ductular secretion. Lastly, the finding that IL-4 up-regulates biliary TMEM16A expression and nucleotide-stimulated secretion may have important implications for cholestatic liver diseases associated with proinflammatory mediators. Accordingly, these studies represent the first molecular identification of a non-CFTR Cl− channel in biliary epithelium and therefore compliment and extend the original biophysical characterization of Cl− currents in biliary epithelial cells first published almost 20 years ago (5). As these are the first studies of TMEM16A in biliary epithelium, several points are in order.
First, although these studies demonstrate a predominant role of TMEM16A in ATP-stimulated Cl− secretion, we cannot exclude contributions from other Cl− channels or TMEM16 isoforms. TMEM16A/ANO-1 is a 114-kDa membrane protein with 960 amino acids and eight transmembrane domains and is one of the 10 members of TMEM16/ANO family of proteins (16–18). Only TMEM16A/ANO1 (16–18) and TMEM16B/ANO2 (42, 43) are associated with Ca2+-activated Cl− currents in other epithelia. In the current studies, TMEM16A was identified in all biliary models, whereas TMEM16B was not present in any models. Furthermore, TMEM16A gene silencing decreased protein expression by 75% which corresponded to a 75% decrease in the magnitude of Ca2+-activated Cl− current density as assessed by whole-cell patch clamp. The residual Cl− currents may be secondary to residual TMEM16A protein (due to the less than 100% efficiency of gene silencing), and the correlation between the decrease in TMEM16A protein expression and Cl− currents provides strong evidence that this is the predominant operative channel. However, the possibility that the other TMEM16 isoforms (f, j, and k) identified in biliary epithelium form novel Cl−-permeable heteromultimers cannot be excluded.
Second, the studies in polarized NRC monolayers demonstrate that TMEM16A is activated through nucleotides binding P2 receptors on the apical membrane. This is in contrast to CFTR, which is activated by binding of secretin to basolateral receptors (6). Thus, TMEM16A represents a critical member of the purinergic signaling pathway involved in hepatobiliary coupling, by which ATP released into bile by hepatocytes can stimulate P2 receptors on downstream cholangiocytes to increase ductular Cl− transport. Together with the recent findings demonstrating that fluid flow, or shear, at the cholangiocyte apical membrane is a potent stimulus for ATP release (44), a model emerges in which increases in bile flow rate or viscosity is transmitted to mechanosensitive ATP release, autocrine/paracrine P2 receptor binding, and TMEM16A-mediated increases in membrane Cl− permeability, ultimately resulting in bile dilution.
Third, these studies suggest that TMEM16A represents a potential target for regulation of secretion by cAMP-independent pathways and for pharmacological therapy of cystic fibrosis and other cholestatic liver diseases. In fact, in other secretory cells, the level of expression of Cl− channels other than CFTR is an important determinant of organ level disease in CF (45). In the CF mouse model for example, increased Ca2+-activated Cl− channel expression in respiratory epithelium is associated with mild pulmonary disease (45). Additionally, gallbladder epithelium from CF mice exhibits brisk secretory responses in response to UTP (14), suggesting that nucleotide-stimulated secretion, via Ca2+-activated pathways, represents a viable strategy to increase secretion and bile flow in CF biliary epithelium.
Fourth, these studies are the first to identify the molecular basis of secretion in small mouse cholangiocytes isolated from small “upstream” intrahepatic ducts (<15 μm in diameter) (27). As far as we know, these are the first patch clamp studies to compare the biophysical properties of cholangiocytes isolated from distinct segments of the intrahepatic bile duct. Small cholangiocytes do not express CFTR and do not exhibit cAMP-stimulated Cl− secretion (35). It seemed unusual that these small, cholangiocytes forming the smallest ducts, and the first to be exposed to canalicular bile rich in bile acids, did not possess the necessary secretory apparatus to increase fluid secretion and dilute bile. The identification of robust ATP-stimulated Ca2+-activated Cl− efflux via TMEM16A provides an answer.
Lastly, cholestasis may have profound effects on cholangiocyte Cl− transport through down-regulation of IP3 receptors (46) and altered Ca2+ and cAMP signaling (46). These effects may be mediated in part by proinflammatory cytokines that are elevated during cholestasis. For example, IL-5 may have direct effects on CFTR expression and cAMP-stimulated Cl− transport (41). In contrast, IL-4 appears to mediate an adaptive response through up-regulation of TMEM16A expression resulting in an increase in transepithelial secretion. Thus, interleukin-mediated effects on channel expression and/or function may be complex within the inflammatory milieu of the cholestatic liver with some interleukins increasing, and others decreasing, cellular transport. Defining the roles of interleukins in channel expression and regulation may have important implications during cholestasis.
In summary, these studies have identified the molecular identity of the Ca2+-activated Cl− channel responsible for nucleotide-mediated secretion in biliary epithelium. Modulation of TMEM16A, or other components of the ATP-stimulated secretory apparatus, might provide novel strategies for the management of liver diseases characterized by impaired bile flow.
Supplementary Material
Acknowledgments
Confocal imaging was carried out in the University of Texas Southwestern Live Cell Imaging Facility.
This work was supported, in whole or in part, by NIDDK, National Institutes of Health Grants DK054811 (to G. A.), DK 50574 (to D. C. R.), and DK078587 (to A. P. F.). This work was also supported by the American Association for the Study of Liver Diseases American Liver Foundation Liver Scholar award (to A. K. D.), the Children's Medical Center Foundation (to A. K. D.), Cystic Fibrosis Foundation Grant FERANC08G0 (to A. P. F.), and the Veterans Affairs Merit award (to G. A.).

The on-line version of this article (available at http://www.jbc.org) contains supplemental Tables S1 and S2 and Figs. S1–S5.
- CFTR
- cystic fibrosis transmembrane conductance regulator
- NRC
- normal rat cholangiocyte(s)
- MLC
- mouse large cholangiocytes
- MSC
- mouse small cholangiocytes
- BDL
- bile duct-ligated.
REFERENCES
- 1. Kumar U., Jordan T. W. (1986) Liver 6, 369–378 [DOI] [PubMed] [Google Scholar]
- 2. Preisig R., Cooper H. L., Wheeler H. O. (1962) J. Clin. Invest. 41, 1152–1162 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Wheeler H. O., Ramos O. L. (1960) J. Clin. Invest. 39, 161–170 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Fitz J. G. (2002) Semin. Liver Dis. 22, 241–249 [DOI] [PubMed] [Google Scholar]
- 5. Fitz J. G., Basavappa S., McGill J., Melhus O., Cohn J. A. (1993) J. Clin. Invest. 91, 319–328 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. McGill J. M., Basavappa S., Gettys T. W., Fitz J. G. (1994) Am. J. Physiol. 266, G731–736 [DOI] [PubMed] [Google Scholar]
- 7. Schlenker T., Fitz J. G. (1996) Am. J. Physiol. 271, G304–310 [DOI] [PubMed] [Google Scholar]
- 8. Dutta A. K., Woo K., Doctor R. B., Fitz J. G., Feranchak A. P. (2008) Am. J. Physiol. Gastrointest. Liver Physiol. 295, G1004–1015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Minagawa N., Nagata J., Shibao K., Masyuk A. I., Gomes D. A., Rodrigues M. A., Lesage G., Akiba Y., Kaunitz J. D., Ehrlich B. E., Larusso N. F., Nathanson M. H. (2007) Gastroenterology 133, 1592–1602 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Fiorotto R., Spirlì C., Fabris L., Cadamuro M., Okolicsanyi L., Strazzabosco M. (2007) Gastroenterology 133, 1603–1613 [DOI] [PubMed] [Google Scholar]
- 11. Colombo C., Battezzati P. M., Crosignani A., Morabito A., Costantini D., Padoan R., Giunta A. (2002) Hepatology 36, 1374–1382 [DOI] [PubMed] [Google Scholar]
- 12. Feranchak A. P., Sokol R. J. (2001) Sem. Liv. Disease 21, 471–488 [DOI] [PubMed] [Google Scholar]
- 13. Paradiso A. M., Ribeiro C. M., Boucher R. C. (2001) J. Gen. Physiol. 117, 53–67 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Clarke L. L., Harline M. C., Gawenis L. R., Walker N. M., Turner J. T., Weisman G. A. (2000) Am. J. Physiol. Gastrointest. Liver Physiol. 279, G132–138 [DOI] [PubMed] [Google Scholar]
- 15. Hartzell C., Putzier I., Arreola J. (2005) Annu. Rev. Physiol. 67, 719–758 [DOI] [PubMed] [Google Scholar]
- 16. Caputo A., Caci E., Ferrera L., Pedemonte N., Barsanti C., Sondo E., Pfeffer U., Ravazzolo R., Zegarra-Moran O., Galietta L. J. (2008) Science 322, 590–594 [DOI] [PubMed] [Google Scholar]
- 17. Yang Y. D., Cho H., Koo J. Y., Tak M. H., Cho Y., Shim W. S., Park S. P., Lee J., Lee B., Kim B. M., Raouf R., Shin Y. K., Oh U. (2008) Nature 455, 1210–1215 [DOI] [PubMed] [Google Scholar]
- 18. Schroeder B. C., Cheng T., Jan Y. N., Jan L. Y. (2008) Cell 134, 1019–1029 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Ferrera L., Caputo A., Ubby I., Bussani E., Zegarra-Moran O., Ravazzolo R., Pagani F., Galietta L. J. (2009) J. Biol. Chem. 284, 33360–33368 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Knuth A., Gabbert H., Dippold W., Klein O., Sachsse W., Bitter-Suermann D., Prellwitz W., Meyer zum Büschenfelde K. H. (1985) J. Hepatol. 1, 579–596 [DOI] [PubMed] [Google Scholar]
- 21. Basavappa S., Middleton J., Mangel A. W., McGill J. M., Cohn J. A., Fitz J. G. (1993) Gastroenterology 104, 1796–1805 [DOI] [PubMed] [Google Scholar]
- 22. Feranchak A. P., Roman R. M., Doctor R. B., Salter K. D., Toker A., Fitz J. G. (1999) J. Biol. Chem. 274, 30979–30986 [DOI] [PubMed] [Google Scholar]
- 23. Roman R. M., Wang Y., Fitz J. G. (1996) Am. J. Physiol. 271, G239–248 [DOI] [PubMed] [Google Scholar]
- 24. Wang Y., Roman R., Schlenker T., Hannun Y. A., Raymond J., Fitz J. G. (1997) J. Clin. Invest. 99, 2890–2897 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Feranchak A. P., Doctor R. B., Troetsch M., Brookman K., Johnson S. M., Fitz J. G. (2004) Gastroenterology 127, 903–913 [DOI] [PubMed] [Google Scholar]
- 26. Dutta A. K., Khimji A. K., Sathe M., Kresge C., Parameswara V., Esser V., Rockey D. C., Feranchak A. P. (2009) Am. J. Physiol. Gastrointest. Liver Physiol. 297, G1009–G1018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Ueno Y., Alpini G., Yahagi K., Kanno N., Moritoki Y., Fukushima K., Glaser S., LeSage G., Shimosegawa T. (2003) Liver Int. 23, 449–459 [DOI] [PubMed] [Google Scholar]
- 28. Francis H., Glaser S., Demorrow S., Gaudio E., Ueno Y., Venter J., Dostal D., Onori P., Franchitto A., Marzioni M., Vaculin S., Vaculin B., Katki K., Stutes M., Savage J., Alpini G. (2008) Am. J. Physiol. Cell Physiol. 295, C499–513 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Alpini G., Roberts S., Kuntz S. M., Ueno Y., Gubba S., Podila P. V., LeSage G., LaRusso N. F. (1996) Gastroenterology 110, 1636–1643 [DOI] [PubMed] [Google Scholar]
- 30. Alpini G., Ulrich C., Roberts S., Phillips J. O., Ueno Y., Podila P. V., Colegio O., LeSage G. D., Miller L. J., LaRusso N. F. (1997) Am. J. Physiol. Gastrointest. Liver Physiol. 272, G289–297 [DOI] [PubMed] [Google Scholar]
- 31. Vroman B., LaRusso N. F. (1996) Lab. Invest. 74, 303–313 [PubMed] [Google Scholar]
- 32. Salter K. D., Roman R. M., LaRusso N. R., Fitz J. G., Doctor R. B. (2000) Lab. Invest. 80, 1775–1778 [DOI] [PubMed] [Google Scholar]
- 33. Schlenker T., Romac J. M., Sharara A. I., Roman R. M., Kim S. J., LaRusso N., Liddle R. A., Fitz J. G. (1997) Am. J. Physiol. 273, G1108–1117 [DOI] [PubMed] [Google Scholar]
- 34. Ishii M., Vroman B., LaRusso N. F. (1989) Gastroenterology 97, 1236–1247 [DOI] [PubMed] [Google Scholar]
- 35. Glaser S. S., Gaudio E., Rao A., Pierce L. M., Onori P., Franchitto A., Francis H. L., Dostal D. E., Venter J. K., DeMorrow S., Mancinelli R., Carpino G., Alvaro D., Kopriva S. E., Savage J. M., Alpini G. D. (2009) Lab. Invest. 89, 456–469 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Chang D., Hsieh P. S., Dawson D. C. (1988) Comput. Biol. Med. 18, 351–366 [DOI] [PubMed] [Google Scholar]
- 37. Lidofsky S. D., Xie M. H., Sostman A., Scharschmidt B. F., Fitz J. G. (1993) J. Biol. Chem. 268, 14632–14636 [PubMed] [Google Scholar]
- 38. Fitz J. G., Sostman A. H. (1994) Am. J. Physiol. 266, G544–553 [DOI] [PubMed] [Google Scholar]
- 39. Feranchak A. P., Berl T., Capasso J., Wojtaszek P. A., Han J., Fitz J. G. (2001) J. Clin. Invest. 108, 1495–1504 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Alpini G., Glaser S., Robertson W., Rodgers R. E., Phinizy J. L., Lasater J., LeSage G. D. (1997) Am. J. Physiol. 272, G1064–1074 [DOI] [PubMed] [Google Scholar]
- 41. McGill J. M., Yen M. S., Cummings O. W., Alpini G., LeSage G., Pollok K. E., Miller B., Engle S. K., Stansfield A. P. (2001) Am. J. Physiol. Gastrointest. Liver Physiol. 280, G738–745 [DOI] [PubMed] [Google Scholar]
- 42. Stephan A. B., Shum E. Y., Hirsh S., Cygnar K. D., Reisert J., Zhao H. (2009) Proc. Natl. Acad. Sci. U.S.A. 106, 11776–11781 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Stöhr H., Heisig J. B., Benz P. M., Schöberl S., Milenkovic V. M., Strauss O., Aartsen W. M., Wijnholds J., Weber B. H., Schulz H. L. (2009) J. Neurosci. 29, 6809–6818 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Woo K., Dutta A. K., Patel V., Kresge C., Feranchak A. P. (2008) J. Physiol. 586, 2779–2798 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Clarke L. L., Grubb B. R., Yankaskas J. R., Cotton C. U., McKenzie A., Boucher R. C. (1994) Proc. Natl. Acad. Sci. U.S.A. 91, 479–483 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Shibao K., Hirata K., Robert M. E., Nathanson M. H. (2003) Gastroenterology 125, 1175–1187 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.