Abstract
Previous studies from our laboratory demonstrated that melatonin inhibits nitric oxide (NO)-induced relaxation in porcine coronary arteries. The present study was designed to further characterize the mechanisms underlying this inhibitory effect of melatonin. Western immunoblot studies identified the presence of melatonin type 2 (MT2) receptors, but not MT1 or MT3 receptors, in porcine coronary arteries. Immunohistochemical analysis revealed that MT2 receptors colocalized with α-actin in the smooth muscle cell layer. In coronary arterial rings suspended in organ chambers for isometric tension recording, melatonin (10−7 M) inhibited relaxations induced by the exogenous NO donor sodium nitroprusside (SNP; 10−9 to 10−5 M) and by the α2-adrenoceptor agonist 5-bromo-6-[2-imidazolin-2-yl-amino]-quinoxaline (UK14,304; 10−9 to 10−5 M), an endothelium-dependent vasodilator. The inhibitory effect of melatonin on SNP- and UK14,304-induced relaxations was abolished in the presence of the selective MT2 receptor antagonists 4-phenyl-2-propionamidotetralin (4P-PDOT; 10−7 M) and luzindole (10−7 M). In contrast to melatonin, the selective MT3 receptor agonist 5-methoxycarbonylamino-N-acetyltryptamine (5-MCA-NAT; 10−7 M) had no effect on the concentration-response curves to either SNP or UK14,304. Melatonin (10−7 M) had no effect on coronary artery relaxation induced by 8-bromoguanosine 3′,5′-cyclic monophosphate, but it significantly attenuated the increase in intracellular cyclic GMP levels in response to SNP (10−5 M). This effect of melatonin was abolished in the presence of 4P-PDOT (10−7 M). Taken together, these data support the view that melatonin acts on MT2 receptors in coronary vascular smooth muscle cells to inhibit NO-induced increases in cyclic GMP and coronary arterial relaxation, thus demonstrating a novel function for MT2 receptors in the vasculature.
Introduction
Melatonin is the primary hormone secreted by the pineal gland and is thought to play a central role in the biological regulation of circadian rhythms. Several extrapineal sources of melatonin have also been identified, including mast cells, leukocytes, platelets, and endothelial cells (Finocchiaro et al., 1991; Kvetnoy, 1999). Increasing evidence indicates that melatonin plays a role in cardiovascular homeostasis by regulating blood vessel diameter, thereby affecting arterial blood pressure and local blood flow to organs and tissues (Krause et al., 2000; Pandi-Perumal et al., 2008). Moreover, alterations in circulating melatonin levels are associated with several cardiovascular disorders, including hypertension (Jonas et al., 2003), ischemic heart disease (Brugger et al., 1995), myocardial infarction (Domínguez-Rodríguez et al., 2002), and heart failure (Girotti et al., 2003).
Melatonin regulates vascular tone by interacting with specific receptors that are present in mammalian arteries (Viswanathan et al., 1990; Stankov et al., 1993). Three distinct melatonin receptor subtypes, termed MT1, MT2, and MT3 receptors, have been identified and shown to mediate the physiological effects of melatonin (Nosjean et al., 2000; Dubocovich et al., 2003). The effects of melatonin on vascular function are complex inasmuch as melatonin receptor activation causes vasoconstriction in certain arteries (Evans et al., 1992; Geary et al., 1997; Ting et al., 1997; Viswanathan et al., 1997) and vasodilation in others (Satake et al., 1991; Weekley, 1991; Doolen et al., 1998). Furthermore, melatonin receptors are not expressed in all blood vessels (Mahle et al., 1997), thus limiting the actions of the hormone to specific regions of the circulation. It is also clear that melatonin receptor expression in the cardiovascular system is altered in a variety of conditions, such as Alzheimer's disease (Savaskan et al., 2001), aging (Sánchez-Hidalgo et al., 2009), myocardial infarction (Sallinen et al., 2007), and coronary artery disease (Ekmekcioglu et al., 2003).
In addition to directly causing vasoconstriction or vasodilation, we have reported that melatonin activates receptors in coronary arteries to exert a powerful indirect effect on vascular tone by inhibiting nitric oxide (NO)-induced smooth muscle relaxation (Yang et al., 2001). Because NO is a key regulator of coronary arterial smooth muscle tone, inhibition of the NO signaling pathway is likely to contribute to the mechanism(s) by which melatonin influences coronary arterial function under normal and pathophysiological conditions. Thus, the primary goal of the present study was to identify the melatonin receptor subtype that mediates the inhibitory effects of melatonin on NO-induced relaxation in porcine coronary arteries.
Materials and Methods
Tissue Preparation.
Fresh porcine hearts were obtained from a local abattoir and were immediately immersed in ice-cold physiological salt solution. After transfer to the laboratory, the left anterior descending coronary artery was dissected from surrounding myocardium, cleaned of adherent fat and connective tissue, and cut into rings 4 to 5 mm in length. Four to eight coronary arterial rings were prepared from each heart. In some rings, the endothelium was removed by gently rubbing the intimal surface with a fine forceps.
Western Immunoblot Analysis.
After collection, coronary arteries from six hearts were immediately frozen in liquid nitrogen. Tissues were homogenized in lysis buffer and supplemented with a protease and phosphatase inhibitor cocktail (Santa Cruz Biotechnology Inc., Santa Cruz, CA) at 4°C using an IKA Ultra-Turrax T8 homogenizer (IKA Works Inc., Wilmington, NC). The tissue homogenates were kept on ice for 10 min and then centrifuged at 10,000g for 10 min. Supernatant was collected, and protein determination was performed using a Bio-Rad Dc Protein assay kit (Bio-Rad Laboratories, Hercules, CA). Aliquots of supernatant containing equal amounts of protein (100 μg) were separated on 12% polyacrylamide gel by SDS-polyacrylamide gel electrophoresis, and proteins were electroblotted onto a polyvinylidene difluoride membrane (Bio-Rad Laboratories). Blots were blocked with 5% nonfat dry milk in phosphate-buffered saline (PBS, pH 7.4) and incubated overnight at 4°C with a primary antibody specific for MT1 receptors, MT2 receptors, or MT3 receptors using a dilution of 1:200 (Santa Cruz Biotechnology Inc.). Membranes were washed two times for 15 min using PBS Tween 20 and incubated with a horseradish peroxidase-linked secondary antibody (Santa Cruz Biotechnology Inc.). To ensure equal loading, the blots were analyzed for β-tubulin expression using an anti-β-tubulin antibody (Santa Cruz Biotechnology Inc.). Immunodetection was performed using an enhanced chemiluminescence light detection kit (Thermo Fisher Scientific, Waltham, MA).
Immunohistochemistry.
Frozen tissue blocks of freshly isolated porcine coronary arteries (n = 4) were sectioned at 8 μm and mounted onto ProbeOn Plus microscopic slides (Thermo Fisher Scientific). Sections were fixed with ice-cold acetone and air-dried for 30 min at room temperature. Nonspecific antibody binding was blocked with normal donkey serum [1.5% (v/v); Santa Cruz Biotechnology Inc.] in PBS for 1 h at room temperature. Sections were incubated overnight at 4°C in PBS containing a primary antibody against MT2 receptors (Santa Cruz Biotechnology Inc.) and/or smooth muscle α-actin (Santa Cruz Biotechnology Inc.). For colocalization of the MT2 receptors with smooth muscle α-actin, double immunofluorescent staining was performed by incubating the tissue sections with more than one primary antibody at the same time. Detection of the primary antibodies against the MT2 receptors and smooth muscle α-actin was accomplished using Alexa Fluor 488-conjugated (i.e., rabbit anti-goat IgG; Invitrogen, Carlsbad, CA) and Texas red-conjugated (i.e., goat anti-mouse IgG; Santa Cruz Biotechnology Inc.) secondary antibodies, respectively. Controls were incubated with either nonimmune goat IgG or primary antibody immunoneutralized with a specific blocking peptide (Santa Cruz Biotechnology Inc.) instead of primary antibody. All dilutions and thorough washes between stages were performed using PBS containing Triton X-100 [0.3% (v/v)] unless otherwise stated. Sections were drained by blotting with filter paper, and a drop of mounting medium containing an anti-fade reagent (Vector Laboratories, Burlingame, CA) was added to the slides. The images of the sections were obtained using an Olympus confocal laser-scanning microscope (Olympus, Tokyo, Japan). The images were generated using Olympus FluoView FV300 (v. 4.3) confocal software and Adobe Photoshop 5.5 (Adobe Systems, Mountain View, CA).
Organ Chamber Studies.
Coronary arterial rings were suspended in water-jacketed organ chambers filled with 25 ml of physiological salt solution, as described previously (Yang et al., 2001). The organ chamber solution was aerated with a mixture of 95% O2/5% CO2, and the temperature was maintained at 37°C throughout the experiment. Each ring was suspended by means of two fine stainless steel wire clips passed through the lumen; one clip was anchored inside the organ chamber, and the other was connected to a force transducer (model FT03; Grass Instruments, Quincy, MA). Isometric tension was measured and recorded on a Grass polygraph. The tissues were stretched progressively to the optimal point of their length–tension relationship using 20 mM KCl to generate a standard contractile response. After this procedure, the rings were allowed to equilibrate at their optimal length for at least 30 min before exposure to any vasoactive substances. The absence or presence of intact endothelium was confirmed in each preparation by the absence or presence of relaxation to the endothelium-dependent vasodilator bradykinin (10−7 M).
Relaxation of coronary arteries was studied in rings contracted with the thromboxane A2-mimetic, 9,11-dideoxy-11α,9α-epoxymethano-prostaglandin F2α (U46619, 1–3 × 10−9 M), which produces a contraction that is approximately 25 to 50% of the maximal response to U46619. After the U46619-induced contraction had reached a stable plateau, relaxation responses to increasing concentrations of sodium nitroprusside (SNP; 10−9 to 10−5 M), the α2-adrenoceptor agonist UK14,304 (10−9 to 10−5 M), or 8-Br-cGMP (10−7 to 10−4 M) were obtained in the absence and presence of melatonin (10−7 M), which was added to the organ chambers immediately before the addition of U46619. This concentration of melatonin and incubation time were previously determined to have the greatest inhibitory effect on responses to NO (Yang et al., 2001). In some experiments, the preparations were incubated with the MT2 receptor antagonists 4P-PDOT (10−7 M) or luzindole (10−7 M) for 30 min before exposure to melatonin, and these inhibitors were present in the organ chambers throughout the remainder of the experiment. In a separate series of experiments, concentration-response curves to SNP and UK14,304 were also obtained in the absence and presence of the MT3 receptor-selective agonist 5-MCA-NAT (10−7 M), which was added to the organ chambers in place of melatonin.
Cyclic GMP Measurements.
An enzyme linked immunosorbent assay was used to measure cGMP levels in porcine coronary arteries. Coronary arterial rings were suspended in water-jacketed organ chambers filled with 25 ml of physiological salt solution and were allowed to equilibrate for at least 1 h at 37°C. After equilibration, coronary artery rings were treated with SNP (10−5 M) for 5 min in the presence and absence of melatonin (10−7 M). In some experiments, the rings were pretreated with 4P-PDOT (20 min; 10−7 M) before exposure to melatonin and SNP. After drug treatments, rings were frozen in liquid nitrogen and homogenized with an IKA Ultra-Turrax T8 homogenizer (IKA Works Inc.) at 4°C in 0.1 N hydrochloric acid. The tissue homogenates were centrifuged at least 600g for 10 min. The cyclic GMP and total protein content was determined in the supernatant using a direct cyclic GMP enzyme immunoassay kit (Assay Designs, Ann Arbor, MI) and a Bio-Rad Dc Protein assay kit (Bio-Rad Laboratories), respectively. Cyclic GMP levels were expressed as picomole per microgram of protein.
Data Analysis.
Relaxation responses are expressed as a percentage of the initial tension induced by U46619. For each vasodilator, both Emax and EC50 values were determined. The EC50 values were converted to negative logarithms and expressed as log molar EC50 (pD2). Results are expressed as mean ± S.E., and n refers to the number of animals from which blood vessels were taken. Values were compared by a Student's t test for paired or unpaired observations, or by analysis of variance with a post hoc Bonferroni's multiple-comparison analysis to determine significance between groups, as appropriate. Values were considered to be significantly different when P < 0.05.
Drugs and Solutions.
Bradykinin, melatonin, and sodium nitroprusside were obtained from Sigma-Aldrich (St. Louis, MO); luzindole, 5-MCA-NAT, 4P-PDOT, and UK14,304 were obtained from Tocris Bioscience (Ellisville, MO); and U46619 was obtained from Cayman Chemical (Ann Arbor, MI). Drug solutions were prepared daily, kept on ice, and protected from light until used. All drugs were dissolved initially in distilled water with the exception of melatonin, 5-MCA-NAT, 4P-PDOT, and luzindole, which were dissolved in ethanol before further dilution in distilled water. Drugs were added to the organ chambers in volumes no greater than 0.2 ml. Drug concentrations are reported as final molar concentration in the organ chamber. The composition of the physiological salt solution was as follows: 118.3 mM NaCl, 4.7 mM KCl, 2.5 mM CaCl2, 1.2 mM MgSO4, 1.2 mM KH2PO4, 25.0 mM NaHCO3, and 11.1 mM glucose.
Results
Receptor Expression.
Western immunoblot analysis revealed the presence of melatonin receptor protein in porcine coronary arteries. A strong immunoreactive band at 36 kDa corresponding to MT2 receptors was detected in immunoblots of porcine coronary artery homogenates (Fig. 1, lanes 1–3) and in rat heart used as a positive control (Fig. 1, lane 5; Sánchez-Hidalgo et al., 2009). The MT2 receptor protein immunoreactivity was abolished when the primary antibody was neutralized with a specific blocking peptide. By contrast, neither MT1 nor MT3 receptor expression was detected in porcine coronary arteries (data not shown).
Fig. 1.

Western immunoblot analysis of MT2 receptor protein in porcine coronary artery homogenates. Immunoreactive bands at 36 kDa are for porcine coronary artery samples from three animals (lanes 1–3) and rat heart used as a positive control (lane 5). No immunoreactivity was detected in loading buffer (negative control, lane 4). β-Tubulin is shown at 55 kDa.
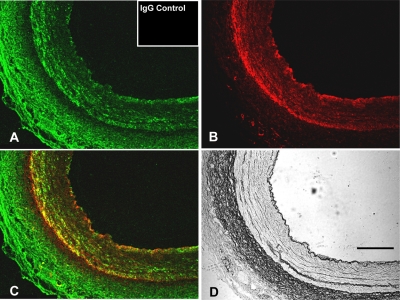
In immunohistochemical studies, MT2 receptor protein was immunodetected in porcine coronary arteries (Fig. 2A) and localized to the smooth muscle cell layer and adventitia (Fig. 2, A and C). Incubation of arteries with nonimmune IgG followed by the secondary antibody resulted in a complete lack of staining (Fig. 2A, inset). Smooth muscle cell actin (SMCA) was detected in the smooth muscle cell layer (Fig. 2B), and MT2 receptors were colocalized with SMCA in the smooth muscle cell layer of the coronary artery (Fig. 2C).
Fig. 2.
Representative images of immunofluorescent colocalization of MT2 receptors and SMCA in porcine coronary artery. A, MT2 receptor (green fluorescence). B, SMCA (red fluorescence). C, colocalized areas (yellow fluorescence). D, image taken using light microscopy. Inset in A demonstrates a lack of staining after incubation of arteries with nonimmune IgG followed by the secondary antibody. Scale bar, 50 μm.
Pharmacologic Studies.
SNP (10−9 to 10−5 M), an exogenous NO donor (Kowaluk et al., 1992), caused concentration-dependent relaxations in isolated porcine coronary artery rings (without endothelium) contracted by the thromboxane A2 mimetic U46619 (1–3 × 10−9 M) (Fig. 3, top). In the presence of melatonin (10−7 M), the concentration-response curve to SNP was shifted to the right in a parallel manner (Fig. 3, top). The pD2 values for SNP in the absence and presence of melatonin were 7.49 ± 0.1 and 6.79 ± 0.2, respectively (P < 0.05). Melatonin had no effect on the maximal response to SNP, inasmuch as SNP caused complete (i.e., 100%) relaxation in both untreated and melatonin-treated rings. Melatonin itself had no direct effect on resting tension or on the level of U46619-induced contraction in coronary artery rings, as reported previously (Yang et al., 2001).
Fig. 3.
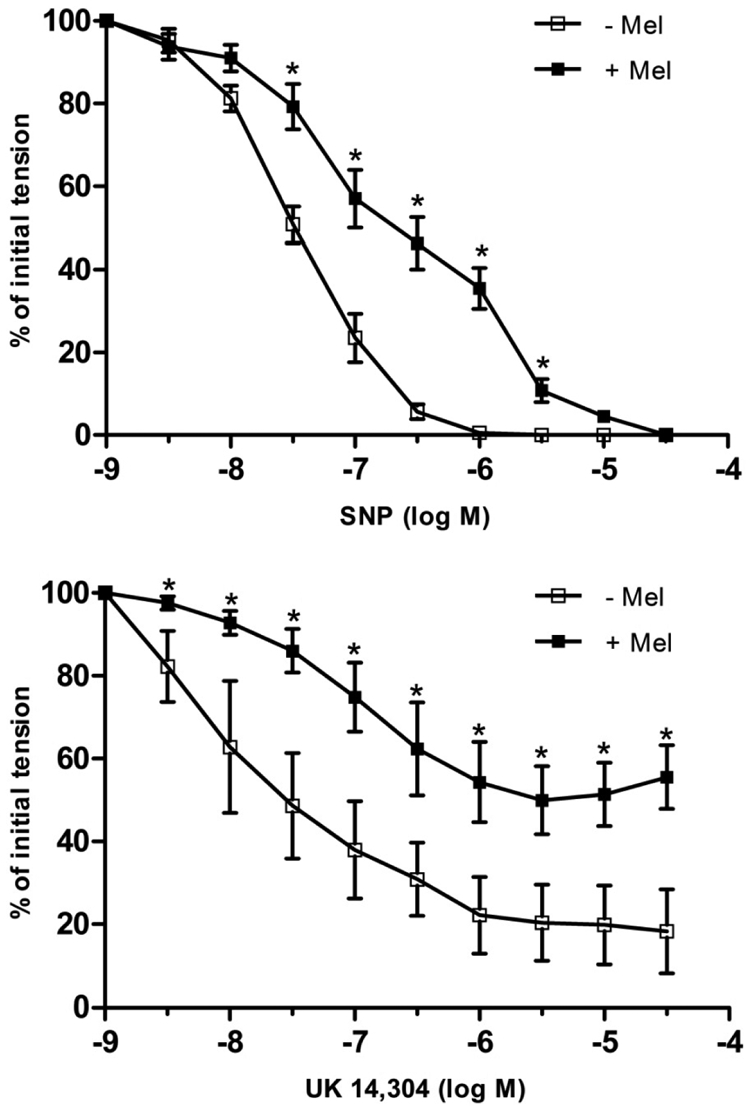
Log concentration-response curves for SNP (top) or UK14,304 (bottom) in producing relaxation of isolated porcine coronary arteries in the absence and presence of melatonin (Mel; 10−7 M). Data are expressed as a percentage of the initial increase in tension induced by U46619 (1–3 × 10−9 M), which averaged 2.48 ± 0.2 and 2.31 ± 0.3 g in the absence and presence of melatonin, respectively (P > 0.05; Student's t test). Each point represents the mean ± S.E.M. (n = 4–5). Statistically significant difference (*, P < 0.05; ANOVA) in the presence of melatonin is indicated.
The endothelium-dependent vasodilator UK14,304 (10−9 to 10−5 M) (Flavahan et al., 1989; Bockman et al., 1996) caused concentration-dependent relaxations in endothelium-intact coronary arterial rings (Fig. 3, bottom). The response to UK14,304 was markedly inhibited in the presence of melatonin (10−7 M) (Fig. 3, bottom). Melatonin caused a rightward shift in the concentration-response curve to UK14,304 (pD2 = 7.65 ± 0.2 versus 6.73 ± 0.2 in the absence and presence of melatonin, respectively; P < 0.05), as well as a reduction in the maximal level of relaxation (Emax = 80 ± 9% relaxation versus 53 ± 8% relaxation in the absence and presence of melatonin, respectively; P < 0.05).
In contrast to melatonin, the MT3 receptor-selective agonist 5-MCA-NAT (10−7 M) (Pintor et al., 2001) had no effect on the concentration-response curves to either SNP or UK14,304 (Fig. 4; P > 0.05). Pretreatment of coronary arterial rings with either of the selective MT2 receptor antagonists, 4P-PDOT (10−7 M) or luzindole (10−7 M) (Dubocovich, 1988; Dubocovich et al., 1997; Browning et al., 2000), abolished the inhibitory effect of melatonin on relaxation induced by either SNP (Fig. 5) or UK14,304 (Fig. 6). The concentration-response curves to SNP and UK14,304 were unaffected by the presence of either antagonist alone (data not shown).
Fig. 4.
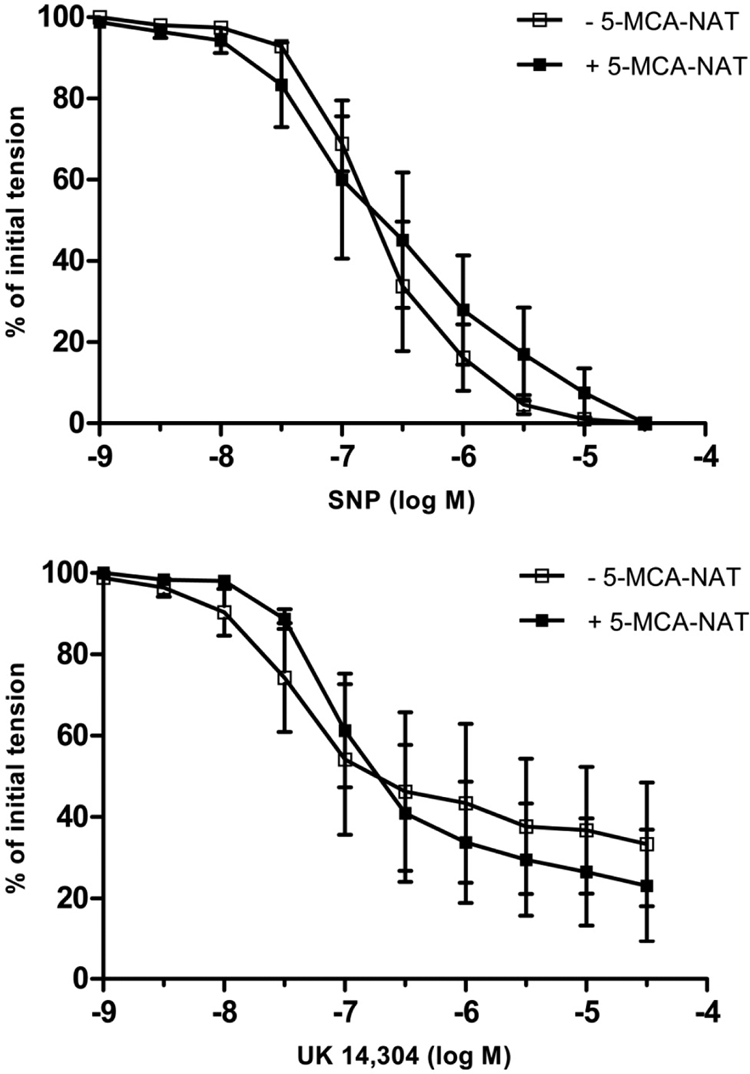
Log concentration-response curves for SNP (top) or UK14,304 (bottom) in producing relaxation of isolated porcine coronary arteries in the absence and presence of 5-MCA-NAT (10−7 M). Data are expressed as a percentage of the initial increase in tension induced by U46619 (1–3 × 10−9 M), which averaged 5.09 ± 0.9 and 5.15 ± 0.7 g in the absence and presence of melatonin, respectively (P > 0.05; Student's t test). Each point represents the mean ± S.E.M. (n = 4).
Fig. 5.
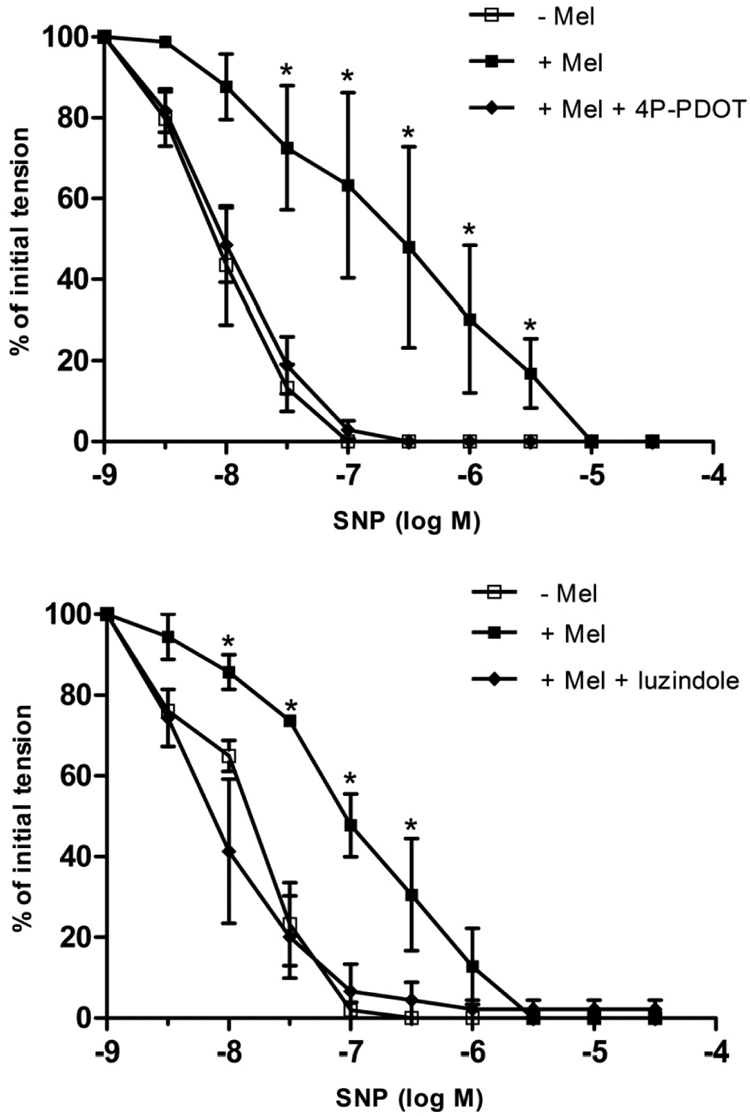
Effect of the selective MT2 receptor antagonists 4P-PDOT (10−7 M) (top) or luzindole (10−7 M) (bottom) on SNP-induced relaxation of isolated porcine coronary artery arteries (without endothelium) in the absence and presence of melatonin (Mel; 10−7 M). Data are expressed as a percentage of the initial increase in tension induced by U46619 (1–3 × 10−9 M), which averaged 2.66 ± 0.2 g in control rings and did not differ significantly in rings treated with melatonin, 4P-PDOT, or luzindole (P > 0.05; ANOVA). Each point represents the mean ± S.E.M. (n = 4–5). Statistically significant difference (*, P < 0.05; ANOVA) from untreated control is indicated.
Fig. 6.
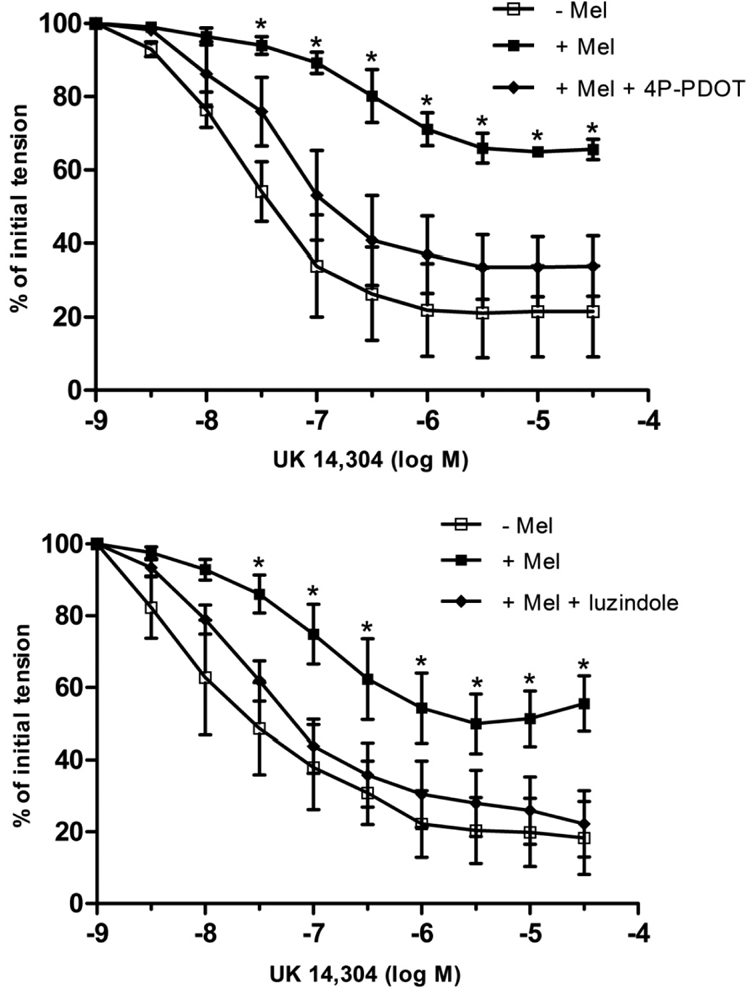
Effect of the selective MT2 receptor antagonists 4P-PDOT (10−7 M) (top) or luzindole (10−7 M) (bottom) on UK14,304-induced relaxation of isolated porcine coronary artery arteries (with endothelium) in the absence and presence of melatonin (10−7 M). Data are expressed as a percentage of the initial increase in tension induced by U46619 (1–3 × 10−9 M), which averaged 2.28 ± 0.3 g in control rings and did not differ significantly in rings treated with melatonin, 4P-PDOT, or luzindole (P > 0.05; ANOVA). Each point represents the mean ± S.E.M. (n = 4–7). Statistically significant difference (*, P < 0.05; ANOVA) from untreated control is indicated.
Cyclic GMP Studies.
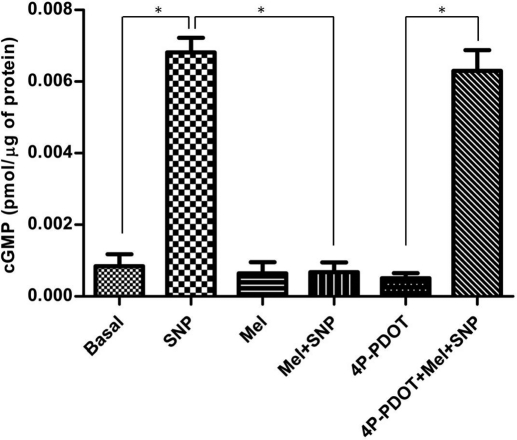
SNP (10−5 M) significantly increased cyclic GMP levels in coronary arteries (Fig. 7). Incubation of coronary arteries with melatonin (10−7 M) had no effect on basal cyclic GMP levels but markedly attenuated the SNP-induced increase in cyclic GMP. In the presence of 4P-PDOT (10−7 M), the inhibitory effect of melatonin on the SNP-induced increase in cyclic GMP was abolished. 4P-PDOT itself had no effect on cyclic GMP levels.
Fig. 7.
Effect of melatonin (Mel; 10−7 M) on SNP (10−5 M)-induced increases in coronary artery cyclic GMP levels in the absence and presence of the MT2-selective antagonist 4P-PDOT (10−7 M). Data are expressed as mean ± S.E.M. (n = 6). Statistically significant difference (*, P < 0.05; ANOVA) between treatment groups is indicated.
In isolated coronary artery rings contracted with U46619, the cell membrane-permeable cyclic GMP analog 8-Br-cGMP (10−7 to 10−4 M) caused concentration-dependent relaxations (pD2 = 4.75 ± 0.2, Emax = 99 ± 1%; n = 3) that were unaffected by the presence of melatonin (10−7 M) (pD2 = 4.55 ± 0.3, Emax = 99 ± 1%; P > 0.05).
Discussion
The results of this study demonstrate the presence of structural and functional MT2 receptors in porcine coronary arterial smooth muscle cells and show that activation of these receptors by melatonin inhibits NO-induced increases in cyclic GMP as well as the ability of the smooth muscle to relax in response to NO. These conclusions are supported by the observations that 1) MT2 receptor protein was expressed in porcine coronary arteries; 2) MT2 receptor proteins colocalized with α-actin, thus indicating that MT2 receptors are present in the smooth muscle cell layer; and 3) the inhibitory effect of melatonin on NO-induced cyclic GMP accumulation and smooth muscle relaxation was abolished in the presence of selective MT2 receptor antagonists. These findings suggest a novel role for MT2 receptors in regulating vascular tone; before the present study, the only previously known vasomotor response to MT2 receptor activation was vasodilation (Doolen et al., 1998; Masana et al., 2002).
High-affinity melatonin binding sites have been identified, characterized, and labeled as MT1, MT2, and MT3 (Dubocovich and Markowska, 2005). Whereas MT1 and MT2 receptors belong to the G-protein-coupled family of membrane receptors, the MT3 receptor is identified as the enzyme quinone reductase 2, an intracellular melatonin binding site (Nosjean et al., 2000). Specific membrane melatonin receptors have been detected in many tissues (e.g., brain, retina, heart, adipocytes) from several different species (Stankov et al., 1993; Mahle et al., 1997; Brydon et al., 2001; Masana et al., 2002; Dubocovich and Markowska, 2005). Within the cardiovascular system, high-affinity melatonin binding sites were identified in several vascular beds (Viswanathan et al., 1990; Stankov and Fraschini, 1993; Stankov et al., 1993), suggesting a role for melatonin in local control of the blood vessel diameter. Indeed, a functional role for melatonin binding sites and receptors was established by studies demonstrating that melatonin causes vasoconstriction in certain vascular beds and vasodilation in others. For example, melatonin causes direct vasoconstriction of cerebral arteries (Capsoni et al., 1995; Geary et al., 1997; Viswanathan et al., 1997) and vasodilation in caudal arteries (Doolen et al., 1998; Masana et al., 2002) in rats.
The coronary circulation is also a site of action for the receptor-dependent effects of melatonin. Previous studies from our laboratory demonstrated that, in isolated porcine coronary arteries, physiologically relevant concentrations of melatonin have no direct effect on vasomotor tone but cause inhibition of NO-induced smooth muscle relaxation. This effect of melatonin is abolished by the specific MT receptor antagonist N-[2-napth-1-yl-ethyl]-cyclobutyl carboxamide (S20928) (Yang et al., 2001); however, S20928 is nonselective with regard to individual MT receptor subtypes (Petit et al., 1999; Audinot et al., 2003). Thus, in the present study, we used a combination of molecular and pharmacological techniques to identify the specific MT receptor subtype mediating the inhibitory effect of melatonin on NO-induced relaxation.
At present, a limited number of potent and selective drugs are available for pharmacologically characterizing MT receptor subtypes. The most useful agents include luzindole and 4P-PDOT, which at low concentrations (≤0.1 μM) are selective antagonists of MT2 receptors (Boutin et al., 2005), and 5-MCA-NAT, which is a selective MT3 receptor agonist (Pintor et al., 2001). No current commercially available antagonists are selective for MT1 receptors. Using these pharmacologic probes in functional studies, we found that in contrast to melatonin 5-MCA-NAT had no effect on NO-induced relaxation of isolated coronary arteries, suggesting that the inhibitory effect of melatonin does not involve activation of MT3 receptors. However, the inhibitory effect of melatonin on NO-induced relaxation was abolished in the presence of either 4P-PDOT or luzindole, two chemically unrelated antagonists (Boutin et al., 2005), strongly suggesting a role for MT2 receptors in the response to melatonin in coronary arteries. The results of these functional studies are consistent with our immunoblot data indicating that neither MT1 nor MT3 receptors are expressed in these arteries. Moreover, the expression of MT2 receptors in the coronary vascular smooth muscle cells, which is the site of action for NO, was confirmed by immunoblot and immunohistochemical studies. Taken together, these data strongly support the view that melatonin acts on MT2 receptors in vascular smooth muscle to inhibit the actions of NO in porcine coronary arteries.
The physiological role of MT2 receptors in the vasculature is not yet fully understood. In the rat caudal artery, MT2 receptors are expressed in the vascular smooth muscle layer, where they mediate vasodilation in response to melatonin (Doolen et al., 1998; Masana et al., 2002). MT2 receptors are also expressed in human coronary arteries (Ekmekcioglu et al., 2003), but the functional effects of melatonin in the human coronary circulation are unknown. The present findings demonstrate that in addition to mediating vasodilation in some arteries (Doolen et al., 1998; Masana et al., 2002), MT2 receptors may be functionally coupled to impaired NO-induced vasodilation in other arteries (e.g., coronary arteries) and may provide new strategies for assessing melatonin receptor function in human coronary arteries.
In the present study, melatonin inhibited porcine coronary arterial relaxation in response to both the α2-adrenoceptor agonist UK14,304, an endothelium-dependent vasodilator that acts solely by releasing NO from endothelial cells in this tissue (Flavahan et al., 1989; Bockman et al., 1996), and SNP, an exogenous NO donor (Kowaluk et al., 1992). One potential site of action for melatonin could be endothelial NO synthase (eNOS). Indeed, melatonin inhibits NO production in rat microvascular endothelial cells (Silva et al., 2007), although this effect is not observed in larger arteries of the rat (Monroe and Watts, 1998). Although an effect of melatonin on eNOS in porcine coronary arteries cannot be ruled out, the observation that melatonin also inhibits relaxation induced by SNP, which is independent of eNOS (Kowaluk et al., 1992), suggests a site of action for melatonin other than, or in addition to, eNOS. A likely possibility is that melatonin acts directly on the vascular smooth muscle cells, which express MT2 receptors (Figs. 1 and 2) and are the primary site of action for the vasorelaxing effect of NO. The primary mechanism by which NO relaxes vascular smooth muscle is by increasing intracellular cyclic GMP levels, followed by activation of protein kinase G and the subsequent phosphorylation of several regulatory proteins (Ignarro et al., 1981; Murad, 1986). Thus, our observation that melatonin attenuates the NO-induced increase in intracellular cyclic GMP levels in coronary arteries provides a plausible mechanism that could account for the inhibitory action of melatonin on relaxation to either endogenous or exogenous NO. Moreover, the effects of melatonin on both NO-induced increases in cyclic GMP and arterial relaxation were attenuated by pharmacologic blockade of MT2 receptors, further suggesting that MT2 receptors mediate both effects of melatonin and that these effects are functionally linked to one another. These results are consistent with previous reports that found that activation of melatonin receptors is linked to decreased cyclic GMP accumulation in some other cell types (Petit et al., 1999; Stumpf et al., 2008). Judging from the lack of effect of melatonin on 8-Br-cGMP-induced relaxation, we find it unlikely that melatonin inhibits signaling events downstream from the NO-induced increase in intracellular cyclic GMP levels.
In summary, the results of the present study support the view that melatonin acts on MT2 receptors in vascular smooth muscle cells to inhibit NO-induced increases in intracellular cyclic GMP and coronary arterial relaxation, thus demonstrating a novel function for MT2 receptors in the vasculature. Because MT2 receptors are expressed in human coronary arteries, and their expression is altered in cardiovascular disease (Ekmekcioglu et al., 2003), these findings may provide clues as to the physiological and pathophysiological roles of melatonin in human coronary circulation.
This work was supported by the National Institutes of Health National Heart, Lung, and Blood Institute [Grant HL077204] (to S.T.O.).
Article, publication date, and citation information can be found at http://jpet.aspetjournals.org.
doi:10.1124/jpet.110.174482.
- MT
- melatonin
- NO
- nitric oxide
- PBS
- phosphate-buffered saline
- U44619
- 9,11-dideoxy-11α,9α-epoxymethano-prostaglandin F2α
- SNP
- sodium nitroprusside
- UK14,304
- 5-bromo-6-[2-imidazolin-2-yl-amino]-quinoxaline
- 8-Br-cGMP
- 8-bromoguanosine 3′,5′-cyclic monophosphate
- 4P-PDOT
- 4-phenyl-2-propionamidotetralin
- 5-MCA-NAT
- 5-methoxycarbonylamino-N-acetyltryptamine
- SMCA
- smooth muscle cell actin
- S20928
- N-[2-napth-1-yl-ethyl]-cyclobutyl carboxamide
- eNOS
- endothelial NO synthase
- ANOVA
- analysis of variance.
Authorship Contributions
Participated in research design: Tunstall, Shukla, Grazul-Bilska, Sun, and O'Rourke.
Conducted experiments: Tunstall and Shukla.
Performed data analysis: Tunstall, Shukla, Grazul-Bilska, Sun, and O'Rourke.
Wrote or contributed to the writing of the manuscript: Tunstall, Shukla, Grazul-Bilska, and O'Rourke.
Other: O'Rourke obtained funding for the research.
References
- Audinot V, Mailliet F, Lahaye-Brasseur C, Bonnaud A, Le Gall A, Amossé C, Dromaint S, Rodriguez M, Nagel N, Galizzi JP, et al. (2003) New selective ligands of human cloned melatonin MT1 and MT2 receptors. Naunyn Schmiedebergs Arch Pharmacol 367:553–561 [DOI] [PubMed] [Google Scholar]
- Bockman CS, Gonzalez-Cabrera I, Abel PW. (1996) Alpha-2 adrenoceptor subtype causing nitric oxide-mediated vascular relaxation in rats. J Pharmacol Exp Ther 278:1235–1243 [PubMed] [Google Scholar]
- Boutin JA, Audinot V, Ferry G, Delagrange P. (2005) Molecular tools to study melatonin pathways and actions. Trends Pharmacol Sci 26:412–419 [DOI] [PubMed] [Google Scholar]
- Browning C, Beresford I, Fraser N, Giles H. (2000) Pharmacological characterization of human recombinant melatonin mt(1) and MT(2) receptors. Br J Pharmacol 129:877–886 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brugger P, Marktl W, Herold M. (1995) Impaired nocturnal secretion of melatonin in coronary heart disease. Lancet 345:1408. [DOI] [PubMed] [Google Scholar]
- Brydon L, Petit L, Delagrange P, Strosberg AD, Jockers R. (2001) Functional expression of MT2 (Mel1b) melatonin receptors in human PAZ6 adipocytes. Endocrinology 142:4264–4271 [DOI] [PubMed] [Google Scholar]
- Capsoni S, Stankov BM, Fraschini F. (1995) Reduction of regional cerebral blood flow by melatonin in young rats. Neuroreport 6:1346–1348 [DOI] [PubMed] [Google Scholar]
- Domínguez-Rodríguez A, Abreu-González P, García MJ, Sanchez J, Marrero F, de Armas-Trujillo D. (2002) Decreased nocturnal melatonin levels during acute myocardial infarction. J Pineal Res 33:248–252 [DOI] [PubMed] [Google Scholar]
- Doolen S, Krause DN, Dubocovich ML, Duckles SP. (1998) Melatonin mediates two distinct responses in vascular smooth muscle. Eur J Pharmacol 345:67–69 [DOI] [PubMed] [Google Scholar]
- Dubocovich ML. (1988) Luzindole (N-0774): a novel melatonin receptor antagonist. J Pharmacol Exp Ther 246:902–910 [PubMed] [Google Scholar]
- Dubocovich ML, Markowska M. (2005) Functional MT1 and MT2 melatonin receptors in mammals. Endocrine 27:101–110 [DOI] [PubMed] [Google Scholar]
- Dubocovich ML, Masana MI, Iacob S, Sauri DM. (1997) Melatonin receptor antagonists that differentiate between the human Mel1a and Mel1b recombinant subtypes are used to assess the pharmacological profile of the rabbit retina ML1 presynaptic heteroreceptor. Naunyn Schmiedebergs Arch Pharmacol 355:365–375 [DOI] [PubMed] [Google Scholar]
- Dubocovich ML, Rivera-Bermudez MA, Gerdin MJ, Masana MI. (2003) Molecular pharmacology, regulation and function of mammalian melatonin receptors. Front Biosci 8:d1093–d1108 [DOI] [PubMed] [Google Scholar]
- Ekmekcioglu C, Thalhammer T, Humpeler S, Mehrabi MR, Glogar HD, Hölzenbein T, Markovic O, Leibetseder VJ, Strauss-Blasche G, Marktl W. (2003) The melatonin receptor subtype MT2 is present in the human cardiovascular system. J Pineal Res 35:40–44 [DOI] [PubMed] [Google Scholar]
- Evans BK, Mason R, Wilson VG. (1992) Evidence for direct vasoconstrictor activity of melatonin in “pressurized” segments of isolated caudal artery from juvenile rats. Naunyn Schmiedebergs Arch Pharmacol 346:362–365 [DOI] [PubMed] [Google Scholar]
- Finocchiaro LM, Nahmod VE, Launay JM. (1991) Melatonin biosynthesis and metabolism in peripheral blood mononuclear leucocytes. Biochem J 280:727–731 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flavahan NA, Shimokawa H, Vanhoutte PM. (1989) Pertussis toxin inhibits endothelium-dependent relaxations to certain agonists in porcine coronary arteries. J Physiol 408:549–560 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geary GG, Krause DN, Duckles SP. (1997) Melatonin directly constricts rat cerebral arteries through modulation of potassium channels. Am J Physiol 273:H1530–H1536 [DOI] [PubMed] [Google Scholar]
- Girotti L, Lago M, Ianovsky O, Elizari MV, Dini A, Lloret SP, Albornoz LE, Cardinali DP. (2003) Low urinary 6-sulfatoxymelatonin levels in patients with severe congestive heart failure. Endocrine 22:245–248 [DOI] [PubMed] [Google Scholar]
- Ignarro LJ, Lippton H, Edwards JC, Baricos WH, Hyman AL, Kadowitz PJ, Gruetter CA. (1981) Mechanism of vascular smooth muscle relaxation by organic nitrates, nitrites, nitroprusside and nitric oxide: evidence for the involvement of S-nitrosothiols as active intermediates. J Pharmacol Exp Ther 218:739–749 [PubMed] [Google Scholar]
- Jonas M, Garfinkel D, Zisapel N, Laudon M, Grossman E. (2003) Impaired nocturnal melatonin secretion in non-dipper hypertensive patients. Blood Press 12:19–24 [PubMed] [Google Scholar]
- Kowaluk EA, Seth P, Fung HL. (1992) Metabolic activation of sodium nitroprusside to nitric oxide in vascular smooth muscle. J Pharmacol Exp Ther 262:916–922 [PubMed] [Google Scholar]
- Krause ND, Geary GG, Doolen S, Duckles PS. (2000) Melatonin and cardiovascular function, in Melatonin after Four Decades: An Assessment of Its Potential (Olcese J. ed) pp 299–310, Kluwer Academic/Plenum Publishers, New York [Google Scholar]
- Kvetnoy IM. (1999) Extrapineal melatonin: location and role within diffuse neuroendocrine system. Histochem J 31:1–12 [DOI] [PubMed] [Google Scholar]
- Mahle CD, Goggins GD, Agarwal P, Ryan E, Watson AJ. (1997) Melatonin modulates vascular smooth muscle tone. J Biol Rhythms 12:690–696 [DOI] [PubMed] [Google Scholar]
- Masana MI, Doolen S, Ersahin C, Al-Ghoul WM, Duckles SP, Dubocovich ML, Krause DN. (2002) MT(2) melatonin receptors are present and functional in rat caudal artery. J Pharmacol Exp Ther 302:1295–1302 [DOI] [PubMed] [Google Scholar]
- Monroe KK, Watts SW. (1998) The vascular reactivity of melatonin. Gen Pharmacol 30:31–35 [DOI] [PubMed] [Google Scholar]
- Murad F. (1986) Cyclic guanosine monophosphate as a mediator of vasodilation. J Clin Invest 78:1–5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nosjean O, Ferro M, Coge F, Beauverger P, Henlin JM, Lefoulon F, Fauchere JL, Delagrange P, Canet E, Boutin JA. (2000) Identification of the melatonin-binding site MT3 as the quinone reductase 2. J Biol Chem 275:31311–31317 [DOI] [PubMed] [Google Scholar]
- Pandi-Perumal SR, Trakht I, Srinivasan V, Spence DW, Maestroni GJ, Zisapel N, Cardinali DP. (2008) Physiological effects of melatonin: role of melatonin receptors and signal transduction pathways. Prog Neurobiol 85:335–353 [DOI] [PubMed] [Google Scholar]
- Petit L, Lacroix I, de Coppet P, Strosberg AD, Jockers R. (1999) Differential signaling of human Mel1a and Mel1b melatonin receptors through the cyclic guanosine 3′-5′-monophosphate pathway. Biochem Pharmacol 58:633–639 [DOI] [PubMed] [Google Scholar]
- Pintor J, Martin L, Pelaez T, Hoyle CH, Peral A. (2001) Involvement of melatonin MT(3) receptors in the regulation of intraocular pressure in rabbits. Eur J Pharmacol 416:251–254 [DOI] [PubMed] [Google Scholar]
- Sánchez-Hidalgo M, Guerrero Montávez JM, Carrascosa-Salmoral Mdel P, Naranjo Gutierrez Mdel C, Lardone PJ, de la Lastra Romero CA. (2009) Decreased MT1 and MT2 melatonin receptor expression in extrapineal tissues of the rat during physiological aging. J Pineal Res 46:29–35 [DOI] [PubMed] [Google Scholar]
- Sallinen P, Mänttäri S, Leskinen H, Ilves M, Vakkuri O, Ruskoaho H, Saarela S. (2007) The effect of myocardial infarction on the synthesis, concentration and receptor expression of endogenous melatonin. J Pineal Res 42:254–260 [DOI] [PubMed] [Google Scholar]
- Satake N, Oe H, Shibata S. (1991) Vasorelaxing action of melatonin in rat isolated aorta; possible endothelium dependent relaxation. Gen Pharmacol 22:1127–1133 [DOI] [PubMed] [Google Scholar]
- Savaskan E, Olivieri G, Brydon L, Jockers R, Kräuchi K, Wirz-Justice A, Müller-Spahn F. (2001) Cerebrovascular melatonin MT1-receptor alterations in patients with Alzheimer's disease. Neurosci Lett 308:9–12 [DOI] [PubMed] [Google Scholar]
- Silva CL, Tamura EK, Macedo SM, Cecon E, Bueno-Alves L, Farsky SH, Ferreira ZS, Markus RP. (2007) Melatonin inhibits nitric oxide production by microvascular endothelial cells in vivo and in vitro. Br J Pharmacol 151:195–205 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stankov B, Fraschini F. (1993) High affinity melatonin binding sites in the vertebrate brain. Neuro Endocrinol Lett 15:149–164 [Google Scholar]
- Stankov B, Capsoni S, Lucini V, Fauteck J, Gatti S, Gridelli B, Biella G, Cozzi B, Fraschini F. (1993) Autoradiographic localization of putative melatonin receptors in the brains of two Old World primates: Cercopithecus aethiops and Papio ursinus. Neuroscience 52:459–468 [DOI] [PubMed] [Google Scholar]
- Stumpf I, Mühlbauer E, Peschke E. (2008) Involvement of the cGMP pathway in mediating the insulin-inhibitory effect of melatonin in pancreatic beta-cells. J Pineal Res 45:318–327 [DOI] [PubMed] [Google Scholar]
- Ting KN, Dunn WR, Davies DJ, Sugden D, Delagrange P, Guardiola-Lemaître B, Scalbert E, Wilson VG. (1997) Studies on the vasoconstrictor action of melatonin and putative melatonin receptor ligands in the tail artery of juvenile Wistar rats. Br J Pharmacol 122:1299–1306 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Viswanathan M, Laitinen JT, Saavedra JM. (1990) Expression of melatonin receptors in arteries involved in thermoregulation. Proc Natl Acad Sci USA 87:6200–6203 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Viswanathan M, Scalbert E, Delagrange P, Guardiola-Lemaître B, Saavedra JM. (1997) Melatonin receptors mediate contraction of a rat cerebral artery. Neuroreport 8:3847–3849 [DOI] [PubMed] [Google Scholar]
- Weekley LB. (1991) Melatonin-induced relaxation of rat aorta: interaction with adrenergic agonists. J Pineal Res 11:28–34 [DOI] [PubMed] [Google Scholar]
- Yang Q, Scalbert E, Delagrange P, Vanhoutte PM, O'Rourke ST. (2001) Melatonin potentiates contractile responses to serotonin in isolated porcine coronary arteries. Am J Physiol 280:H76–H82 [DOI] [PubMed] [Google Scholar]