Abstract
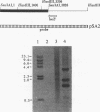
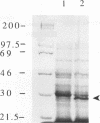
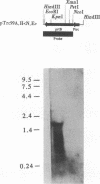
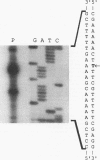
A clone expressing a Treponema denticola chymotrypsinlike protease from recombinant plasmid pSA2 was identified in a genomic library of T. denticola ATCC 35405. Nucleotide sequencing of the insert identified an open reading frame, designated the prtB gene, which codes for the protease. Two potential inverted repeat sequences are present both upstream and downstream from the prtB gene. The prtB gene would code for a putative protein of 273 amino acids with a calculated molecular mass of 30.4 kDa and an estimated pI of 7.0. The G+C content of the gene is 40.3%. The results of maxicell analysis are consistent with the expression of a 30-kDa protease from the prtB gene. Preliminary characterization of the protease indicated that it was inhibited by the protease inhibitors phenylmethylsulfonyl fluoride, diisopropylfluorophosphate, and N-tosyl-L-phenylalanine chloromethyl ketone but not by N alpha-p-tosyl-L-lysine chloromethyl ketone. Purification of the protease was accomplished with the PinPoint protein purification system following construction of site-directed mutagenized plasmid pXa-3:2. The purified protease degraded human and bovine serum albumins as well as casein. Furthermore, hemolysis of sheep erythrocytes by the protease was observed. Northern (RNA) blot analysis of mRNA extracted from strain 35405 indicated a single 1.9-kb mRNA species containing the prtB transcript. In addition, the results of primer extension analysis indicated that transcription was initiated primarily at a T residue. However, no corresponding -10 and -35 sequences related to Escherichia coli promoter sequences were identified. The availability of the purified protein and its gene will aid in evaluating the potential role of the protease in the physiology and virulence of T. denticola since proteases may play a key role in oral treponemal pathogenicity.
Full text
PDF

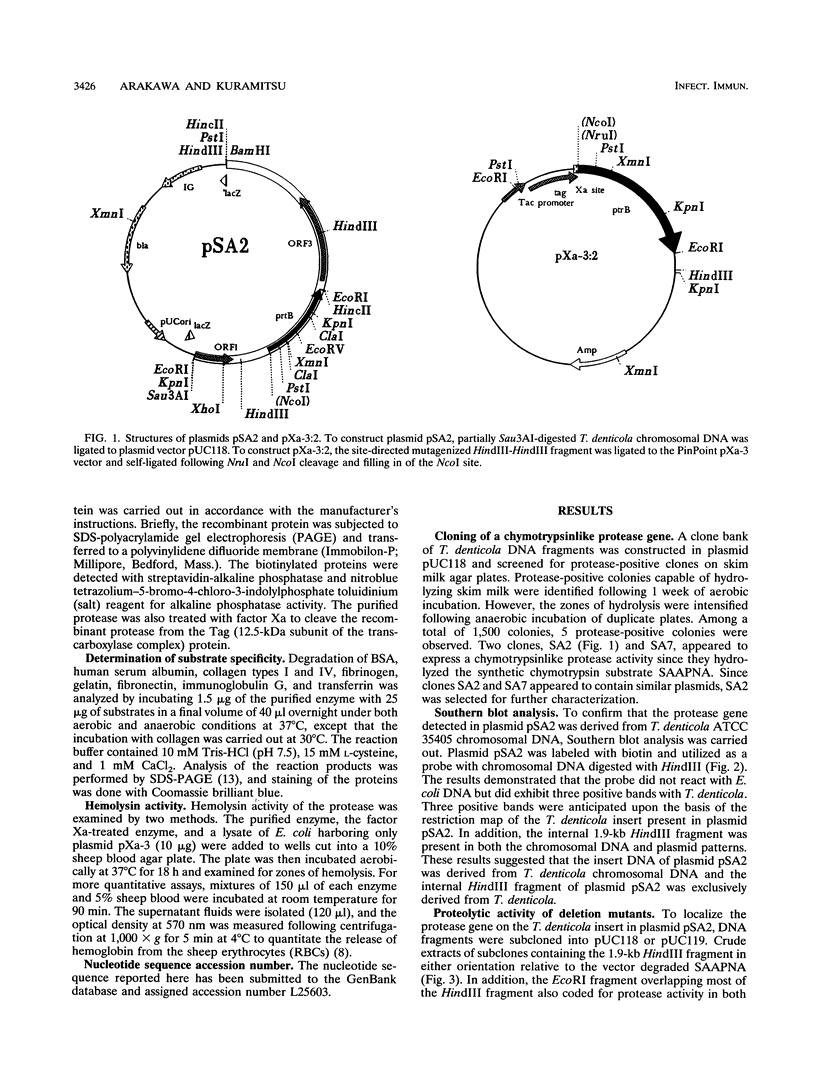


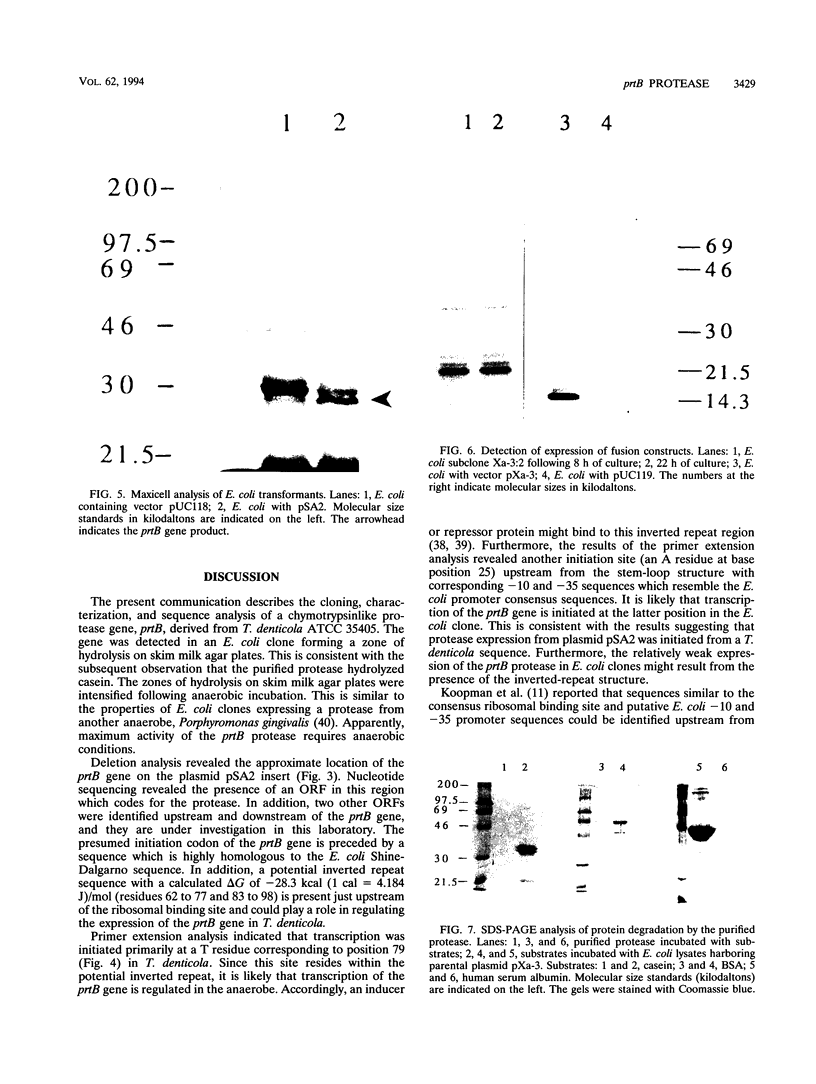




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Baehni P. C., Song M., McCulloch C. A., Ellen R. P. Treponema denticola induces actin rearrangement and detachment of human gingival fibroblasts. Infect Immun. 1992 Aug;60(8):3360–3368. doi: 10.1128/iai.60.8.3360-3368.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boehringer H., Berthold P. H., Taichman N. S. Studies on the interaction of human neutrophils with plaque spirochetes. J Periodontal Res. 1986 May;21(3):195–209. doi: 10.1111/j.1600-0765.1986.tb01452.x. [DOI] [PubMed] [Google Scholar]
- Boehringer H., Taichman N. S., Shenker B. J. Suppression of fibroblast proliferation by oral spirochetes. Infect Immun. 1984 Jul;45(1):155–159. doi: 10.1128/iai.45.1.155-159.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng S. L., Chan E. C. The routine isolation, growth, and maintenance of the intermediate-size anaerobic oral spirochetes from periodontal pockets. J Periodontal Res. 1983 Jul;18(4):362–368. doi: 10.1111/j.1600-0765.1983.tb00371.x. [DOI] [PubMed] [Google Scholar]
- Dzink J. L., Socransky S. S., Haffajee A. D. The predominant cultivable microbiota of active and inactive lesions of destructive periodontal diseases. J Clin Periodontol. 1988 May;15(5):316–323. doi: 10.1111/j.1600-051x.1988.tb01590.x. [DOI] [PubMed] [Google Scholar]
- Frank R. M. Bacterial penetration in the apical pocket wall of advanced human periodontitis. J Periodontal Res. 1980 Nov;15(6):563–573. doi: 10.1111/j.1600-0765.1980.tb00315.x. [DOI] [PubMed] [Google Scholar]
- Grenier D. Characteristics of hemolytic and hemagglutinating activities of Treponema denticola. Oral Microbiol Immunol. 1991 Aug;6(4):246–249. doi: 10.1111/j.1399-302x.1991.tb00485.x. [DOI] [PubMed] [Google Scholar]
- Grenier D., Uitto V. J., McBride B. C. Cellular location of a Treponema denticola chymotrypsinlike protease and importance of the protease in migration through the basement membrane. Infect Immun. 1990 Feb;58(2):347–351. doi: 10.1128/iai.58.2.347-351.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haapasalo M., Müller K. H., Uitto V. J., Leung W. K., McBride B. C. Characterization, cloning, and binding properties of the major 53-kilodalton Treponema denticola surface antigen. Infect Immun. 1992 May;60(5):2058–2065. doi: 10.1128/iai.60.5.2058-2065.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koopman M. B., de Leeuw O. S., van der Zeijst B. M., Kusters J. G. Cloning and DNA sequence analysis of a Serpulina (Treponema) hyodysenteriae gene encoding a periplasmic flagellar sheath protein. Infect Immun. 1992 Jul;60(7):2920–2925. doi: 10.1128/iai.60.7.2920-2925.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kunkel T. A., Roberts J. D., Zakour R. A. Rapid and efficient site-specific mutagenesis without phenotypic selection. Methods Enzymol. 1987;154:367–382. doi: 10.1016/0076-6879(87)54085-x. [DOI] [PubMed] [Google Scholar]
- LISTGARTEN M. A. ELECTRON MICROSCOPIC OBSERVATIONS ON THE BACTERIAL FLORA OF ACUTE NECROTIZING ULCERATIVE GINGIVITIS. J Periodontol. 1965 Jul-Aug;36:328–339. doi: 10.1902/jop.1965.36.4.328. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Listgarten M. A. Direct microscopy of periodontal pathogens. Oral Microbiol Immunol. 1986 Nov;1(1):31–38. doi: 10.1111/j.1399-302x.1986.tb00314.x. [DOI] [PubMed] [Google Scholar]
- Listgarten M. A., Helldén L. Relative distribution of bacteria at clinically healthy and periodontally diseased sites in humans. J Clin Periodontol. 1978 May;5(2):115–132. doi: 10.1111/j.1600-051x.1978.tb01913.x. [DOI] [PubMed] [Google Scholar]
- Listgarten M. A., Levin S. Positive correlation between the proportions of subgingival spirochetes and motile bacteria and susceptibility of human subjects to periodontal deterioration. J Clin Periodontol. 1981 Apr;8(2):122–138. doi: 10.1111/j.1600-051x.1981.tb02352.x. [DOI] [PubMed] [Google Scholar]
- Loesche W. J., Giordano J., Hujoel P. P. The utility of the BANA test for monitoring anaerobic infections due to spirochetes (Treponema denticola) in periodontal disease. J Dent Res. 1990 Oct;69(10):1696–1702. doi: 10.1177/00220345900690101301. [DOI] [PubMed] [Google Scholar]
- Loesche W. J., Syed S. A., Stoll J. Trypsin-like activity in subgingival plaque. A diagnostic marker for spirochetes and periodontal disease? J Periodontol. 1987 Apr;58(4):266–273. doi: 10.1902/jop.1987.58.4.266. [DOI] [PubMed] [Google Scholar]
- Loesche W. J. The role of spirochetes in periodontal disease. Adv Dent Res. 1988 Nov;2(2):275–283. doi: 10.1177/08959374880020021201. [DOI] [PubMed] [Google Scholar]
- MacDougall J. H., Beighton D., Russell R. R. Cloning and expression of protease genes from Treponema denticola in Escherichia coli. Oral Microbiol Immunol. 1991 Oct;6(5):270–274. doi: 10.1111/j.1399-302x.1991.tb00490.x. [DOI] [PubMed] [Google Scholar]
- Mayrand D., Holt S. C. Biology of asaccharolytic black-pigmented Bacteroides species. Microbiol Rev. 1988 Mar;52(1):134–152. doi: 10.1128/mr.52.1.134-152.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mikx F. H., Jacobs F., Satumalay C. Cell-bound peptidase activities of Treponema denticola ATCC 33520 in continuous culture. J Gen Microbiol. 1992 Sep;138(9):1837–1842. doi: 10.1099/00221287-138-9-1837. [DOI] [PubMed] [Google Scholar]
- Mikx F. H., Maltha J. C., van Campen G. J. Spirochetes in early lesions of necrotizing ulcerative gingivitis experimentally induced in beagles. Oral Microbiol Immunol. 1990 Apr;5(2):86–89. doi: 10.1111/j.1399-302x.1990.tb00233.x. [DOI] [PubMed] [Google Scholar]
- Mikx F. H., Ngassapa D. N., Reijntjens F. M., Maltha J. C. Effect of splint placement on black-pigmented Bacteroides and spirochetes in the dental plaque of beagle dogs. J Dent Res. 1984 Nov;63(11):1284–1288. doi: 10.1177/00220345840630110601. [DOI] [PubMed] [Google Scholar]
- Mikx F. H., de Jong M. H. Keratinolytic activity of cutaneous and oral bacteria. Infect Immun. 1987 Mar;55(3):621–625. doi: 10.1128/iai.55.3.621-625.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miyamoto M., Noji S., Kokeguchi S., Kato K., Kurihara H., Murayama Y., Taniguchi S. Molecular cloning and sequence analysis of antigen gene tdpA of Treponema denticola. Infect Immun. 1991 Jun;59(6):1941–1947. doi: 10.1128/iai.59.6.1941-1947.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moore L. V., Moore W. E., Cato E. P., Smibert R. M., Burmeister J. A., Best A. M., Ranney R. R. Bacteriology of human gingivitis. J Dent Res. 1987 May;66(5):989–995. doi: 10.1177/00220345870660052401. [DOI] [PubMed] [Google Scholar]
- Moore W. E., Holdeman L. V., Cato E. P., Smibert R. M., Burmeister J. A., Palcanis K. G., Ranney R. R. Comparative bacteriology of juvenile periodontitis. Infect Immun. 1985 May;48(2):507–519. doi: 10.1128/iai.48.2.507-519.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nitzan D., Sperry J. F., Wilkins T. D. Fibrinolytic activity of oral anaerobic bacteria. Arch Oral Biol. 1978;23(6):465–470. doi: 10.1016/0003-9969(78)90078-x. [DOI] [PubMed] [Google Scholar]
- Ohta K., Makinen K. K., Loesche W. J. Purification and characterization of an enzyme produced by Treponema denticola capable of hydrolyzing synthetic trypsin substrates. Infect Immun. 1986 Jul;53(1):213–220. doi: 10.1128/iai.53.1.213-220.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olsen I. Attachment of Treponema denticola to cultured human epithelial cells. Scand J Dent Res. 1984 Feb;92(1):55–63. doi: 10.1111/j.1600-0722.1984.tb00860.x. [DOI] [PubMed] [Google Scholar]
- Oosterwaal P. J., Matee M. I., Mikx F. H., van 't Hof M. A., Renggli H. H. The effect of subgingival debridement with hand and ultrasonic instruments on the subgingival microflora. J Clin Periodontol. 1987 Oct;14(9):528–533. doi: 10.1111/j.1600-051x.1987.tb00995.x. [DOI] [PubMed] [Google Scholar]
- Pabo C. O., Lewis M. The operator-binding domain of lambda repressor: structure and DNA recognition. Nature. 1982 Jul 29;298(5873):443–447. doi: 10.1038/298443a0. [DOI] [PubMed] [Google Scholar]
- Pabo C. O., Sauer R. T. Protein-DNA recognition. Annu Rev Biochem. 1984;53:293–321. doi: 10.1146/annurev.bi.53.070184.001453. [DOI] [PubMed] [Google Scholar]
- Park Y., McBride B. C. Cloning of a Porphyromonas (Bacteroides) gingivalis protease gene and characterization of its product. FEMS Microbiol Lett. 1992 May 1;71(3):273–278. doi: 10.1016/0378-1097(92)90721-y. [DOI] [PubMed] [Google Scholar]
- Que X. C., Kuramitsu H. K. Isolation and characterization of the Treponema denticola prtA gene coding for chymotrypsinlike protease activity and detection of a closely linked gene encoding PZ-PLGPA-hydrolyzing activity. Infect Immun. 1990 Dec;58(12):4099–4105. doi: 10.1128/iai.58.12.4099-4105.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reijntjens F. M., Mikx F. H., Wolters-Lutgerhorst J. M., Maltha J. C. Adherence of oral treponemes and their effect on morphological damage and detachment of epithelial cells in vitro. Infect Immun. 1986 Feb;51(2):642–647. doi: 10.1128/iai.51.2.642-647.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saglie R., Newman M. G., Carranza F. A., Jr, Pattison G. L. Bacterial invasion of gingiva in advanced periodontitis in humans. J Periodontol. 1982 Apr;53(4):217–222. doi: 10.1902/jop.1982.53.4.217. [DOI] [PubMed] [Google Scholar]
- Sancar A., Hack A. M., Rupp W. D. Simple method for identification of plasmid-coded proteins. J Bacteriol. 1979 Jan;137(1):692–693. doi: 10.1128/jb.137.1.692-693.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sancar A., Rupert C. S. Determination of plasmid molecular weights from ultraviolet sensitivities. Nature. 1978 Mar 30;272(5652):471–472. doi: 10.1038/272471a0. [DOI] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Savitt E. D., Socransky S. S. Distribution of certain subgingival microbial species in selected periodontal conditions. J Periodontal Res. 1984 Mar;19(2):111–123. doi: 10.1111/j.1600-0765.1984.tb00800.x. [DOI] [PubMed] [Google Scholar]
- Schmidt E. F., Bretz W. A., Hutchinson R. A., Loesche W. J. Correlation of the hydrolysis of benzoyl-arginine naphthylamide (BANA) by plaque with clinical parameters and subgingival levels of spirochetes in periodontal patients. J Dent Res. 1988 Dec;67(12):1505–1509. doi: 10.1177/00220345880670121201. [DOI] [PubMed] [Google Scholar]
- Slots J., Genco R. J. Black-pigmented Bacteroides species, Capnocytophaga species, and Actinobacillus actinomycetemcomitans in human periodontal disease: virulence factors in colonization, survival, and tissue destruction. J Dent Res. 1984 Mar;63(3):412–421. doi: 10.1177/00220345840630031101. [DOI] [PubMed] [Google Scholar]
- Socransky S. S., Haffajee A. D. Effect of therapy on periodontal infections. J Periodontol. 1993 Aug;64(8 Suppl):754–759. doi: 10.1902/jop.1993.64.8s.754. [DOI] [PubMed] [Google Scholar]
- Uitto V. J., Chan E. C., Quee T. C. Initial characterization of neutral proteinases from oral spirochetes. J Periodontal Res. 1986 Mar;21(2):95–100. doi: 10.1111/j.1600-0765.1986.tb01442.x. [DOI] [PubMed] [Google Scholar]
- Uitto V. J., Grenier D., Chan E. C., McBride B. C. Isolation of a chymotrypsinlike enzyme from Treponema denticola. Infect Immun. 1988 Oct;56(10):2717–2722. doi: 10.1128/iai.56.10.2717-2722.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wallich R., Moter S. E., Simon M. M., Ebnet K., Heiberger A., Kramer M. D. The Borrelia burgdorferi flagellum-associated 41-kilodalton antigen (flagellin): molecular cloning, expression, and amplification of the gene. Infect Immun. 1990 Jun;58(6):1711–1719. doi: 10.1128/iai.58.6.1711-1719.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yanisch-Perron C., Vieira J., Messing J. Improved M13 phage cloning vectors and host strains: nucleotide sequences of the M13mp18 and pUC19 vectors. Gene. 1985;33(1):103–119. doi: 10.1016/0378-1119(85)90120-9. [DOI] [PubMed] [Google Scholar]