Abstract
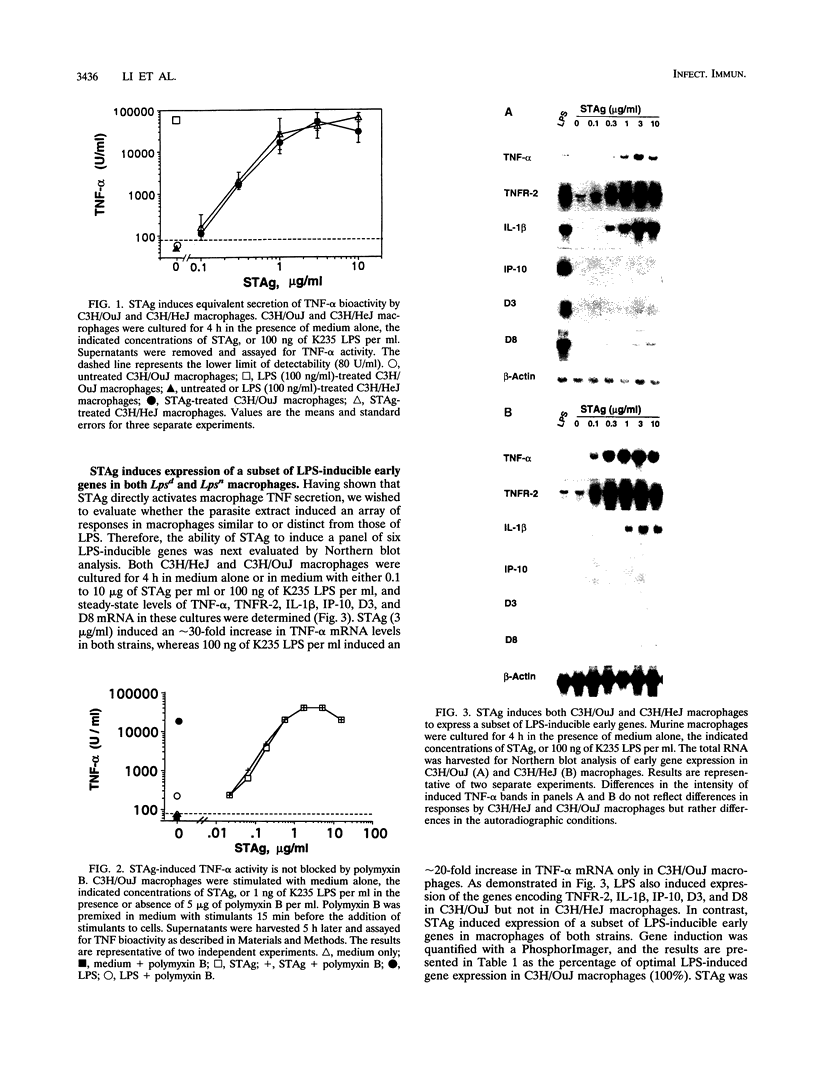
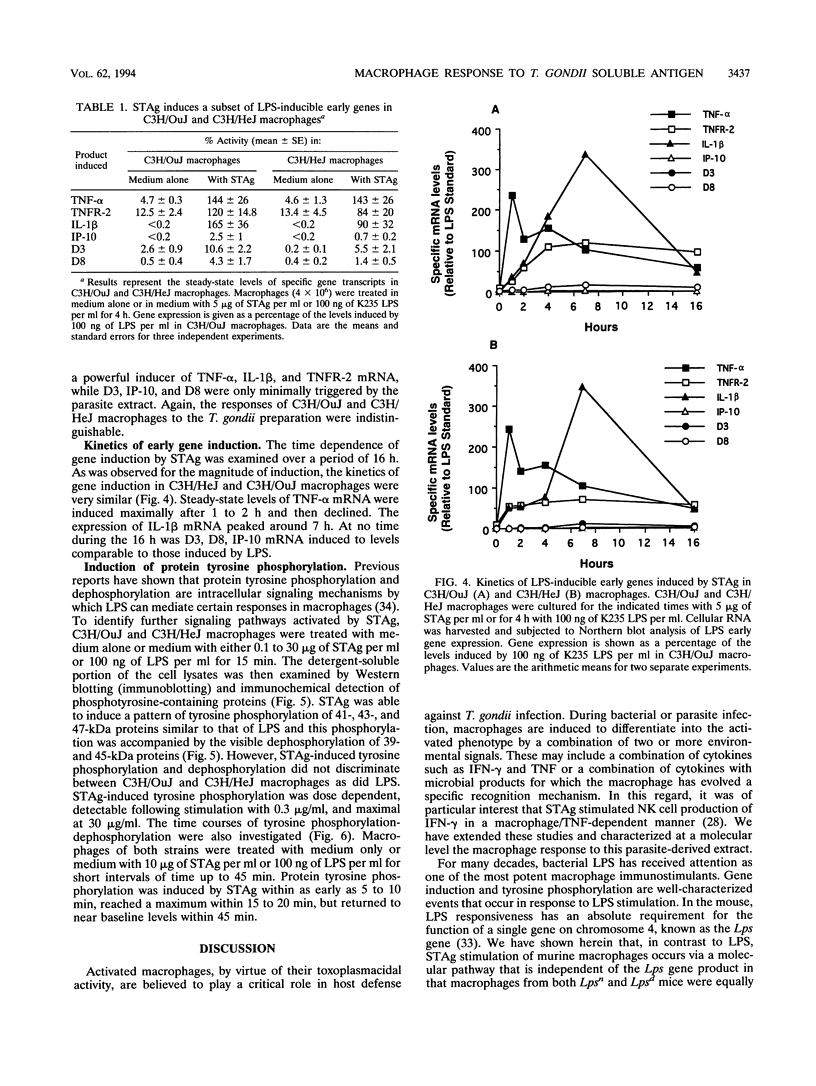
Previous studies have shown that macrophages play an important role in both the initiation of protective responses and the effector mechanism of immunity to Toxoplasma gondii. The purpose of this investigation was to characterize the responses of macrophages to a soluble antigen extract of T. gondii tachyzoites (STAg) in comparison with a prototypic macrophage-activating agent, lipopolysaccharide (LPS), and to determine whether STAg-induced signaling requires a functional Lps gene. Toward this end, tumor necrosis factor (TNF) secretion, a panel of six LPS-inducible genes, and protein tyrosine phosphorylation were examined to gain insights into macrophage responses to STAg. STAg stimulated both C3H/OuJ (Lpsn) and C3H/HeJ (Lpsd) macrophages to secrete bioactive TNF-alpha and to express a subset of LPS-inducible genes (encoding TNF-alpha, TNF receptor 2, and interleukin-1 beta). In contrast to LPS, STAg failed to stimulate Lpsn or Lpsd macrophages to express genes encoding IP-10, D3, or D8. STAg also induced a pattern of tyrosine phosphorylation identical to that induced by LPS; mitogen-activated protein kinase 47-kDa and 43-kDa isoforms and a 41-kDa protein of undetermined identity were inducibly phosphorylated. The ability of STAg to induce TNF-alpha, encoded by a subset of LPS-inducible genes, and tyrosine phosphoproteins was not affected by LPS inhibitors, confirming that the macrophage response to the parasite extract could not be attributed to LPS contamination. We propose that STAg, while differing from LPS in the pattern of macrophage genes induced, may share with LPS two signaling pathways that are intact in Lpsd macrophages.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Adams L. B., Hibbs J. B., Jr, Taintor R. R., Krahenbuhl J. L. Microbiostatic effect of murine-activated macrophages for Toxoplasma gondii. Role for synthesis of inorganic nitrogen oxides from L-arginine. J Immunol. 1990 Apr 1;144(7):2725–2729. [PubMed] [Google Scholar]
- Beaman M. H., Wong S. Y., Remington J. S. Cytokines, Toxoplasma and intracellular parasitism. Immunol Rev. 1992 Jun;127:97–117. doi: 10.1111/j.1600-065x.1992.tb01410.x. [DOI] [PubMed] [Google Scholar]
- Chang H. R., Grau G. E., Pechère J. C. Role of TNF and IL-1 in infections with Toxoplasma gondii. Immunology. 1990 Jan;69(1):33–37. [PMC free article] [PubMed] [Google Scholar]
- Davis R. J. The mitogen-activated protein kinase signal transduction pathway. J Biol Chem. 1993 Jul 15;268(20):14553–14556. [PubMed] [Google Scholar]
- Dong Z., Qi X., Fidler I. J. Tyrosine phosphorylation of mitogen-activated protein kinases is necessary for activation of murine macrophages by natural and synthetic bacterial products. J Exp Med. 1993 Apr 1;177(4):1071–1077. doi: 10.1084/jem.177.4.1071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frenkel J. K. Pathophysiology of toxoplasmosis. Parasitol Today. 1988 Oct;4(10):273–278. doi: 10.1016/0169-4758(88)90018-x. [DOI] [PubMed] [Google Scholar]
- Gazzinelli R. T., Hakim F. T., Hieny S., Shearer G. M., Sher A. Synergistic role of CD4+ and CD8+ T lymphocytes in IFN-gamma production and protective immunity induced by an attenuated Toxoplasma gondii vaccine. J Immunol. 1991 Jan 1;146(1):286–292. [PubMed] [Google Scholar]
- Gazzinelli R. T., Hieny S., Wynn T. A., Wolf S., Sher A. Interleukin 12 is required for the T-lymphocyte-independent induction of interferon gamma by an intracellular parasite and induces resistance in T-cell-deficient hosts. Proc Natl Acad Sci U S A. 1993 Jul 1;90(13):6115–6119. doi: 10.1073/pnas.90.13.6115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamilton T. A., Bredon N., Ohmori Y., Tannenbaum C. S. IFN-gamma and IFN-beta independently stimulate the expression of lipopolysaccharide-inducible genes in murine peritoneal macrophages. J Immunol. 1989 Apr 1;142(7):2325–2331. [PubMed] [Google Scholar]
- Hauser W. E., Jr, Tsai V. Acute toxoplasma infection of mice induces spleen NK cells that are cytotoxic for T. gondii in vitro. J Immunol. 1986 Jan;136(1):313–319. [PubMed] [Google Scholar]
- Henricson B. E., Manthey C. L., Perera P. Y., Hamilton T. A., Vogel S. N. Dissociation of lipopolysaccharide (LPS)-inducible gene expression in murine macrophages pretreated with smooth LPS versus monophosphoryl lipid A. Infect Immun. 1993 Jun;61(6):2325–2333. doi: 10.1128/iai.61.6.2325-2333.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joiner K. A., Dubremetz J. F. Toxoplasma gondii: a protozoan for the nineties. Infect Immun. 1993 Apr;61(4):1169–1172. doi: 10.1128/iai.61.4.1169-1172.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luft B. J., Brooks R. G., Conley F. K., McCabe R. E., Remington J. S. Toxoplasmic encephalitis in patients with acquired immune deficiency syndrome. JAMA. 1984 Aug 17;252(7):913–917. [PubMed] [Google Scholar]
- Luft B. J., Remington J. S. Toxoplasmic encephalitis in AIDS. Clin Infect Dis. 1992 Aug;15(2):211–222. doi: 10.1093/clinids/15.2.211. [DOI] [PubMed] [Google Scholar]
- Lynn W. A., Golenbock D. T. Lipopolysaccharide antagonists. Immunol Today. 1992 Jul;13(7):271–276. doi: 10.1016/0167-5699(92)90009-V. [DOI] [PubMed] [Google Scholar]
- Manthey C. L., Brandes M. E., Perera P. Y., Vogel S. N. Taxol increases steady-state levels of lipopolysaccharide-inducible genes and protein-tyrosine phosphorylation in murine macrophages. J Immunol. 1992 Oct 1;149(7):2459–2465. [PubMed] [Google Scholar]
- Manthey C. L., Perera P. Y., Qureshi N., Stütz P. L., Hamilton T. A., Vogel S. N. Modulation of lipopolysaccharide-induced macrophage gene expression by Rhodobacter sphaeroides lipid A and SDZ 880.431. Infect Immun. 1993 Aug;61(8):3518–3526. doi: 10.1128/iai.61.8.3518-3526.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McIntire F. C., Sievert H. W., Barlow G. H., Finley R. A., Lee A. Y. Chemical, physical, biological properties of a lipopolysaccharide from Escherichia coli K-235. Biochemistry. 1967 Aug;6(8):2363–2372. doi: 10.1021/bi00860a011. [DOI] [PubMed] [Google Scholar]
- Murray H. W., Cohn Z. A. Macrophage oxygen-dependent antimicrobial activity. I. Susceptibility of Toxoplasma gondii to oxygen intermediates. J Exp Med. 1979 Oct 1;150(4):938–949. doi: 10.1084/jem.150.4.938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murray H. W. Interferon-gamma, the activated macrophage, and host defense against microbial challenge. Ann Intern Med. 1988 Apr;108(4):595–608. doi: 10.7326/0003-4819-108-4-595. [DOI] [PubMed] [Google Scholar]
- Murray H. W., Szuro-Sudol A., Wellner D., Oca M. J., Granger A. M., Libby D. M., Rothermel C. D., Rubin B. Y. Role of tryptophan degradation in respiratory burst-independent antimicrobial activity of gamma interferon-stimulated human macrophages. Infect Immun. 1989 Mar;57(3):845–849. doi: 10.1128/iai.57.3.845-849.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohmori Y., Hamilton T. A. A macrophage LPS-inducible early gene encodes the murine homologue of IP-10. Biochem Biophys Res Commun. 1990 May 16;168(3):1261–1267. doi: 10.1016/0006-291x(90)91164-n. [DOI] [PubMed] [Google Scholar]
- Ohmori Y., Strassman G., Hamilton T. A. cAMP differentially regulates expression of mRNA encoding IL-1 alpha and IL-1 beta in murine peritoneal macrophages. J Immunol. 1990 Nov 15;145(10):3333–3339. [PubMed] [Google Scholar]
- Pennica D., Hayflick J. S., Bringman T. S., Palladino M. A., Goeddel D. V. Cloning and expression in Escherichia coli of the cDNA for murine tumor necrosis factor. Proc Natl Acad Sci U S A. 1985 Sep;82(18):6060–6064. doi: 10.1073/pnas.82.18.6060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rossomando A. J., Payne D. M., Weber M. J., Sturgill T. W. Evidence that pp42, a major tyrosine kinase target protein, is a mitogen-activated serine/threonine protein kinase. Proc Natl Acad Sci U S A. 1989 Sep;86(18):6940–6943. doi: 10.1073/pnas.86.18.6940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sher A., Oswald I. P., Hieny S., Gazzinelli R. T. Toxoplasma gondii induces a T-independent IFN-gamma response in natural killer cells that requires both adherent accessory cells and tumor necrosis factor-alpha. J Immunol. 1993 May 1;150(9):3982–3989. [PubMed] [Google Scholar]
- Suzuki Y., Orellana M. A., Schreiber R. D., Remington J. S. Interferon-gamma: the major mediator of resistance against Toxoplasma gondii. Science. 1988 Apr 22;240(4851):516–518. doi: 10.1126/science.3128869. [DOI] [PubMed] [Google Scholar]
- Tannenbaum C. S., Koerner T. J., Jansen M. M., Hamilton T. A. Characterization of lipopolysaccharide-induced macrophage gene expression. J Immunol. 1988 May 15;140(10):3640–3645. [PubMed] [Google Scholar]
- Thomas S. M., DeMarco M., D'Arcangelo G., Halegoua S., Brugge J. S. Ras is essential for nerve growth factor- and phorbol ester-induced tyrosine phosphorylation of MAP kinases. Cell. 1992 Mar 20;68(6):1031–1040. doi: 10.1016/0092-8674(92)90075-n. [DOI] [PubMed] [Google Scholar]
- Tokunaga K., Taniguchi H., Yoda K., Shimizu M., Sakiyama S. Nucleotide sequence of a full-length cDNA for mouse cytoskeletal beta-actin mRNA. Nucleic Acids Res. 1986 Mar 25;14(6):2829–2829. doi: 10.1093/nar/14.6.2829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weinstein S. L., Gold M. R., DeFranco A. L. Bacterial lipopolysaccharide stimulates protein tyrosine phosphorylation in macrophages. Proc Natl Acad Sci U S A. 1991 May 15;88(10):4148–4152. doi: 10.1073/pnas.88.10.4148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weinstein S. L., June C. H., DeFranco A. L. Lipopolysaccharide-induced protein tyrosine phosphorylation in human macrophages is mediated by CD14. J Immunol. 1993 Oct 1;151(7):3829–3838. [PubMed] [Google Scholar]
- Weinstein S. L., Sanghera J. S., Lemke K., DeFranco A. L., Pelech S. L. Bacterial lipopolysaccharide induces tyrosine phosphorylation and activation of mitogen-activated protein kinases in macrophages. J Biol Chem. 1992 Jul 25;267(21):14955–14962. [PubMed] [Google Scholar]
- Wong S. Y., Remington J. S. Biology of Toxoplasma gondii. AIDS. 1993 Mar;7(3):299–316. doi: 10.1097/00002030-199303000-00001. [DOI] [PubMed] [Google Scholar]





