Abstract
Background: Autoantibodies may be present in a variety of underlying cancers several years before tumours can be detected and testing for their presence may allow earlier diagnosis. We report the clinical validation of an autoantibody panel in newly diagnosed patients with lung cancer (LC).
Patients and methods: Three cohorts of patients with newly diagnosed LC were identified: group 1 (n = 145), group 2 (n = 241) and group 3 (n = 269). Patients were individually matched by gender, age and smoking history to a control individual with no history of malignant disease. Serum samples were obtained after diagnosis but before any anticancer treatment. Autoantibody levels were measured against a panel of six tumour-related antigens (p53, NY-ESO-1, CAGE, GBU4-5, Annexin 1 and SOX2). Assay sensitivity was tested in relation to demographic variables and cancer type/stage.
Results: The autoantibody panel demonstrated a sensitivity/specificity of 36%/91%, 39%/89% and 37%/90% in groups 1, 2 and 3, respectively, with good reproducibility. There was no significant difference between different LC stages, indicating that the antigens included covered the different types of LC well.
Conclusion: This assay confirms the value of an autoantibody panel as a diagnostic tool and offers a potential system for monitoring patients at high risk of LC.
Keywords: autoantibodies, clinical validation, lung cancer, newly diagnosed patients
introduction
Lung cancer (LC) is the worldwide leading cause of cancer-related mortality [1]. Tobacco smoking is estimated to cause upwards of 90% of cases, and other recognised risk factors include passive smoking, occupational exposure, especially to asbestos and radon exposure [1]. Outcomes are substantially better with early localised disease compared with locally advanced and metastatic disease, with 5-year survival rates of 53%, 23.7% and 3.5%, respectively [2].
Although the latent period of LC in smokers is reported to be at least 20 years [1], ∼85% of patients with LC remain undiagnosed until the disease is symptomatic and has reached an advanced stage [2]. At present, there is nothing to offer for early diagnosis, although ongoing clinical trials are investigating the use of spiral computed tomography (CT) in ‘at-risk’ individuals [3–12]. However, the radiation dose delivered and the substantial costs limit its widespread application as a screening procedure [13]. Furthermore, the high rate of false positives (as high as 50% in a prevalence round) [5] dictates that many individuals require follow-up examinations and a substantial proportion of individuals undergo unnecessary thoracotomy [14]. Application of a filter such as a blood-based marker to identify smokers at the highest risk for LC may improve the positive predictive value (PPV) of these screening tools [11, 15].
There is a considerable body of evidence documenting the presence of circulating antibodies to autologous cellular antigens [referred to as tumour-associated antigens (TAA)] in serum samples from patients with a variety of cancers, including LC [16–24]. Monitoring persons at increased risk of cancer for the presence of serum autoantibodies may allow earlier detection of the disease.
The panel of proteins selected for investigation comprised a number of well-recognised TAA, four of which (p53, NY-ESO-1, CAGE and GBU4-5) have been described by ourselves in a previous publication to induce the production of autoantibodies or immune biomarkers in LC [24]. In brief, p53 is a tumour suppressor gene, which is often mutated in cancer and to which autoantibodies were first described [25], autoantibodies to this protein have also been detected in some cases, even before the cancer diagnosis [26, 27]. NY-ESO-1 and CAGE are both cancer testis antigens whose expression has been described in a number of solid tumours [28, 29] and with GBU4-5, a protein of unknown function that encodes a DEAD box domain, have also been described as inducing autoantibodies in LC [24, 30, 31].
The remaining antigens SOX2, a member of the SOXB1 family of proteins that is described as inducing an autoantibody responses in small-cell lung cancer (SCLC) [32, 33], and Annexin I, a phospholipid-binding protein to which autoantibodies, have also been described [18].
The selection of these antigens was confirmed following screening of a panel of >20 potential antigens as being of greatest diagnostic utility for the diagnosis of all non-small-cell lung cancer (NSCLC) and SCLC cancer (C. Chapman, unpublished observations).
This manuscript reports the clinical validation set for these autoantibodies in the serum of patients with newly diagnosed LC (before any treatment) and matched controls.
patients and methods
patients
Findings from three separate groups of patients with newly diagnosed LC are reported. The third group is the final validation set where the data were run in a blinded manner. All patients with LC were as far as possible individually matched by gender, age and smoking history to a control individual with no previous history of malignant disease. In patients with LC, blood samples were obtained after diagnosis but before receiving any anticancer treatment. Demographic characteristics of the control versus the study population are given in the Appendix 1.
Group 1 comprised 145 patients with stage I/II LC (including NSCLC and SCLC) and 146 controls treated in centres in the United States and Russia. All subjects in this group were smokers; baseline patient characteristics are shown in Table 1. Group 2 comprised 241 patients with LC treated at a single centre in Germany as part of a collaborative study (Table 1). Tumour pathological information was available for the patients with LC, including tumour, node, metastasis staging and NSCLC histology (Table 2). In group 2, an additional 88 sera from unmatched individuals (25 normal and 63 with benign lung conditions) supplied by the single centre were analysed (Appendix 1).
Table 1.
Lung cancer patient characteristics
| Group 1 (n = 145) | Group 2 (n = 241) | Group 3 (n = 269) | |
| Median age, years (range) | 66 (41–87) | 63 (28–87) | 65 (38–87) |
| Patients >60 years, n (%) | 96 (66.2) | 140 (58.1) | 171 (63.6) |
| Gender, n (%) | |||
| Male | 81 (55.9) | 172 (71.4) | 199 (74.0) |
| Female | 64 (44.1) | 69 (28.6) | 70 (26.0) |
| Smoking history, n (%) | |||
| Current | 145 | 0 | 132 (49.1) |
| Previous | 0 | 0 | 76 (28.3) |
| Never | 0 | 0 | 24 (8.9) |
| Not determined | 0 (0.0) | 241 (100.0) | 37 (13.8) |
Table 2.
Tumour stage and histology according to gender
| Group 1 (n = 145) |
Group 2 (n = 241) |
Group 3 (n = 255a) |
||||
| Male (n = 81) | Female (n = 64) | Male (n = 172) | Female (n = 69) | Male (n = 188) | Female (n = 67) | |
| Tumour type, n (%) | ||||||
| NSCLC | 71 (87.7) | 52 (81.3) | 125 (72.7) | 46 (66.7) | 141 (75.0) | 41 (61.2) |
| SCLC | 10 (12.3) | 12 (18.8) | 47 (27.3) | 23 (33.3) | 47 (25.0) | 26 (38.8) |
| NSCLC stage, n (%) | ||||||
| I | 41 (57.7) | 40 (76.9) | 0 (0.0) | 0 (0.0) | 71 (50.4) | 15 (36.6) |
| II | 30 (42.3) | 12 (23.1) | 0 (0.0) | 1 (2.2) | 42 (29.8) | 11 (26.8) |
| III | 0 (0.0) | 0 (0.0) | 38 (30.4) | 11 (23.9) | 13 (9.2) | 1 (2.4) |
| IV | 0 (0.0) | 0 (0.0) | 63 (50.4) | 25 (54.3) | 1 (0.7) | 2 (4.9) |
| Unknown | 0 (0.0) | 0 (0.0) | 24 (19.2) | 9 (19.6) | 14 (9.9) | 12 (29.3) |
| NSCLC histology, n (%) | ||||||
| Squamous | 16 (22.5) | 5 (9.6) | 38 (30.4) | 4 (8.7) | 78 (55.3) | 10 (24.4) |
| Adenocarcinoma | 16 (22.5) | 13 (25.0) | 37 (29.6) | 19 (41.3) | 44 (31.2) | 23 (56.1) |
| Large cell | 2 (2.8) | 0 | 4 (3.2) | 2 (4.3) | 5 (3.5) | 0 (0.0) |
| Bronchoalveolar | 1 (1.4) | 18 (34.6) | 0 (0.0) | 0 (0.0) | 1 (0.7) | 5 (12.2) |
| Tubular adenocarcinoma | 0 | 0 | 0 | 0 | 2 (1.4) | 0 |
| Not determined | 4 (5.6) | 12 (23.1) | 46 (36.8) | 21 (45.6) | 9 (6.4) | 1 (2.4) |
| Other | 32 (45.1) | 4 (7.7) | 0 | 0 | 2 (1.4) | 2 (4.9) |
| SCLC stage, n (%) | ||||||
| Limited SCLC | 0 | 0 | 21 (44.7) | 6 (26.1) | 14 (29.8) | 15 (57.7) |
| Extensive SCLC | 0 | 0 | 19 (40.4) | 14 (60.9) | 8 (17.0) | 3 (11.5) |
| Not determined | 10 (100.0) | 12 (100.0) | 7 (14.9) | 3 (13.0) | 25 (53.2) | 8 (30.8) |
Tumour histology and stage data available for 255 of the 269 patients comprising group 3.
NSCLC, non-small-cell lung cancer; SCLC, small-cell lung cancer.
Group 3 comprised 269 patients with LC treated at centres in the United States, UK and Ukraine (Table 1). This group was assembled to validate the calibration and control scheme for the autoantibody assay. Tumour pathological information was available for the patients with LC (Table 2). The timeline for collection of samples from patients is shown in supplemental Table S1 (available at Annals of Oncology online).
Serum samples in group 1 were evaluated for autoantibodies against p53, NY-ESO-1, CAGE and GBU4-5. Serum samples in groups 2 and 3 were evaluated for autoantibodies against the same four antigens plus Annexin 1 and SOX2. In groups 2 and 3, samples from patients with cancers, matched normals, benign lung disease and control sera for the assay were interspersed in the order samples were assayed so that any batch effects would be spread over all sample types. The laboratory staff running the assay was blinded to the disease state of individual samples. Group 2, therefore, was a validation set for the results seen in group 1 for four of the antigens (i.e. p53, NY-ESO1, CAGE and GBU4-5) with the added value of Annexin 1 and SOX2. Group 3 validated a calibrated and controlled assay on the whole panel of six antigens.
autoantibody assay
Autoantibodies were determined by a quality-controlled, semi-automated indirect enzyme-linked immunosorbent assay in which samples were allowed to react with a titration series of antigen concentrations. All liquid handling steps were carried out using an automated liquid handling system. Briefly, purified recombinant antigens were diluted to provide a semi-log titration series for each antigen from 160 to 1.6 nM [34]. Control antigens consisting of the purified BirA or NusA tags were also included to allow subtraction of the signal due to nonspecific binding to bacterial contaminants. Antigen dilutions were adsorbed to the surface of microtitre plate wells in phosphate buffer at room temperature. After washing in phosphate-buffered saline containing 0.1% Tween 20 (pH 7.6), microtitre plates were blocked with a gelatine-based blocking buffer. Serum samples (diluted 1 in 110 in a blocking buffer) were then added to the plates and allowed to incubate at room temperature with shaking for 90 min. Following incubation, plates were washed and horseradish peroxidase-conjugated rabbit anti-human IgG (Dako, Glostrup, Denmark) was added. After a 60-min incubation, the plates were washed and 3,3′,5,5′-tetramethylbenzidine was added. Colour formation was allowed to proceed for 15 min before the optical density (OD) of each well was determined spectrophotometrically at 650 nm [35].
Calibration standards of known potency are not available for assays to measure autoantibodies against TAAs. Therefore, a calibration system was devised which utilised fluids drained from pleural or ascitic cavities of patients with LC [36]. The calibration system was only evaluated for group 3 samples. A reportable dilution range for each antigen, giving acceptable calibration precision, was determined at 7.5%–92.5% of the upper asymptote of the average calibration curve, equivalent to ∼5.0 natural log reference units (RU). These data were used to construct a calibration curve of OD versus log dilution to which a four-parameter model plot was fitted [37]. The background-corrected OD value for each unknown sample was then converted to a calibrated log RU.
Samples were judged to be positive if they fulfilled two criteria—i.e. they showed a dose response to the antigen titration series and the measured autoantibody signal to one or more of the antigens was above the accepted cut-off set for that antigen assay. The autoantibody signal for a sample was defined as above the cut-off when the result was greater than the calculated cut-off for the control population at either of the two highest points on the titration curve. All assays were carried out as two replicates and the mean value taken as the overall assay measurement.
optimisation of assay cut-offs
A specificity of 90% was selected in order to produce a test which could be used for early detection in a high-risk population and which would be health economically viable. For all groups, cut-offs based on mean + 3 standard deviations (SDs) of the normal population were used. In addition, for groups 2 and 3, the cut-offs were optimised using a Monte Carlo direct search method [38] to find a set of antigen-specific cut-offs yielding the maximum sensitivity for the fixed specificity of 90%.
For a set of possible cut-offs for the six panel antigens chosen by Monte Carlo sampling over the feasible range, the specificity/sensitivity was first estimated from the data. This was carried out 100 000 times. All combinations with a specificity of ∼90% were then extracted and the combination yielding the maximum sensitivity used. This is a process dependent on assay conditions and when new batches of proteins, or new types of protein, are introduced to the panel, new cut-offs will have to be calculated.
To support the quoted specificity/sensitivity panel results, the area under the curve (AUC) and standard error (SE) for the respective receiver operating characteristic (ROC) curve was calculated for each group. The ROC curve was constructed by calculating the specificity and sensitivity of the test for a succession of deviations from the original cut-offs, with the same deviation for each antigen in the panel.
adjustment for LCs in the control populations
The cut-offs are best set by comparing the results in a group of patients with known LC and a group of high-risk individuals (e.g. smokers and ex-smokers) who are known to not have the disease. However, the latter population is difficult to identify since the CT screening studies have clearly shown there are a percentage of smokers/ex-smokers who at any one time are ‘harbouring’ an asymptomatic LC. In the prevalence round, the percentage of undiagnosed occult cancers has been reported to be between 0.5% and 2.7% in heavy smokers, while in incidence rounds, it has been reported to be up to 2.3% [3–12]. For this reason, adjusted specificity and sensitivity values assuming some degree of occult LCs in the control populations were also calculated.
The method used to calculate and adjust for the presence of undiagnosed cancers in the controls used LC prediction models for which the most important predictors are age, current smoking status and smoking history, and family history of smoking-related cancers [39].
results
autoantibody expression
In group 1, autoantibodies to four antigens (p53, NY-ESO-1, CAGE and GBU4-5) were measured as raw OD values. Using cut-offs based on mean + 3 SDs gave a sensitivity of 36% with a specificity of 91% (50 of the 137, 8 unassessable). The sensitivities and specificities for each of these four antigens and the reproducibility of these assays have been reported elsewhere [35]. The sensitivity and specificity of the panel was similar for males and females. The ROC curve AUC was 0.71 (SE = 0.03).
In group 2, autoantibodies to six antigens (p53, NY-ESO-1, CAGE, GBU4-5 plus Annexin 1 and SOX2) were measured as raw OD values, with cut-offs based on mean + 3 SDs producing sensitivity and specificity values of 34% (80 of the 234, 7 unassessable) and 91%, respectively. Again, individual sensitivities and specificities for these six antigens have been reported elsewhere [35]. Using individually optimised cut-offs for each antigen, the overall sensitivity was 39% (33%–45%) (91 of the 234), with a specificity of 89%. In an at-risk population of 20 LCs per 1000 population, this would result in a PPV of 7.2% (i.e. 1 in 13.9 persons with a positive test would have a LC) and a negative predictive value (NPV) of 98.6%. The ROC curve AUC was 0.63 (SE = 0.03).
Of the 88 unmatched sera received from the group 2 centre, 8 of the 88 (9%) were positive, of which none of the 25 (0%) normal sera had raised autoantibodies, while 8 of the 63 (13%) of individuals with benign lung disease had raised autoantibodies. Follow-up data could only be obtained for one of these eight individuals who was found to have developed a gastric cancer, giving a specificity of at least 89% (55 of the 62).
In group 3, autoantibodies to the same six antigens as group 2 were measured as raw OD values and converted into calibrated RU. Using the ODs and applying cut-offs based on mean + 3 SDs gave a sensitivity of 32% (85 of the 269) with a specificity of 91%. Using RU values with individually optimised cut-offs for each antigen, the sensitivity was 37% (31%–43%) (100 of the 269), with a specificity of 90%. In an at-risk population of 20 LCs per 1000 population, this would result in a PPV of 7.0% (i.e. 1 in 14.3) and an NPV of 98.6%. The ROC curve AUC was 0.64 (SE = 0.02).
Individual antigen sensitivity and specificity are shown in supplemental Table S2 (available at Annals of Oncology online).
adjustment for occult LCs within the control population
Adjustment generated specific cut-offs for each antigen for the different methods. The sensitivities for each antigen for a fixed specificity of 90% are shown for the unadjusted and adjusted method in Table 3. The most conservative estimate for adjusted sensitivity is 40%, which in an at-risk group of 20 LCs per 1000 population would give a PPV of 7.5% (i.e. 1 in 13.3) and an NPV of 98.7%.
Table 3.
Comparison of performancea before and after adjustment for the presence of undiagnosed occult cancers in the control population
| Adjustment method | Group 2 | Group 3 | |
| Unadjusted | Sensitivity | 39% (91/234) | 37% (100/269) |
| Specificity | 89% (207/232) | 90% (242/269) | |
| Occult cancer rate (5%) | Sensitivity | 42% (99/234) | 40% (108/269) |
| Specificity | 90% (197/220) | 90% (230/255) | |
| Occult cancer rate (11%) | Sensitivity | 46% (108/234) | 43% (115/269) |
| Specificity | 89% (184/206) | 90% (214/238) |
Sensitivity for specificity of 90% ± 1%, based on optimised cut-offs for individual antigens.
effect of patient and disease characteristics on autoantibody assay sensitivity and specificity
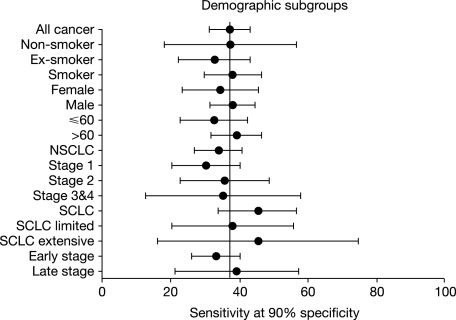
The calibrated group 3 dataset with an unadjusted sensitivity and specificity of 37% and 90%, respectively, was used to assess whether patient characteristics, tumour type or stage gave rise to significant variation in the specificity/sensitivity (Figure 1). Statistical comparison of subgroups with remaining controls demonstrated no significant difference in sensitivity according to patient gender, smoking status and age or tumour type or stage (P > 0.10). There was also no significant difference in sensitivity between those NSCLC tumours where the subtype was known and those where it was unknown.
Figure 1.
Forest plot showing the sensitivity at a fixed 90% specificity by patient demographics, tumour characteristics and lung cancer stage. Line shows unadjusted sensitivity of 37% (all stages of cancers) in group 3 (n = 269). NSCLC, non-small-cell lung cancer; SCLC, small-cell lung cancer.
discussion
This report confirms a validated assay for the detection of autoantibodies to selected cancer-associated antigens in the peripheral blood. The value of a test for early cancer detection is usually defined via a number of related parameters, including sensitivity, specificity, PPV and NPV. A percentage of smokers/ex-smokers are ‘harbouring’ an asymptomatic LC at any one time. Even with the most conservative estimation of occult LCs, the panel of autoantibodies can identify 40% of primary LCs, including early stage of disease, with a specificity of 90% against age-matched, gender-matched and smoking history-matched controls. The specificity was similar (at least 89%) for patients with benign disease.
Autoantibodies to p53 [26, 27, 40, 41], NY-ESO-1 [30, 31], CAGE [29, 42], GBU4-5 [31], Annexin 1 [16, 18, 43] and SOX2 [44] have all been shown to be capable of inducing autoantibodies in patients with LC. The data in this manuscript further confirm the value of a panel of autoantibodies over a single autoantibody assay [19, 23, 24, 35]. Recent publications have reported autoantibodies to a natural form of Annexin 1 [43] and other antigens (e.g. 14-3-3 theta [43, 45] and LAMR1 [43]), which are elevated in LC and up to 1 year before clinical diagnosis. The combination of 14-3-3 theta, Annexin 1 and LAMR1 gave an AUC on a combined ROC curve of 0.73. While these results were based on a research assay, it is possible that adding 14-3-3 theta and/or LAMR1 to the current panel might increase the sensitivity.
Group 3 data confirm that there was no significant difference between different stages of LC, although due to sample size the confidence intervals were sometimes wide. Further evaluation of the data was, therefore, carried out by comparing early-stage (stage I/II NSCLC plus limited SCLC) with late-stage (stage III/IV NSCLC plus extensive SCLC) disease, which again showed no difference. The presence of such a signal in early-stage disease is precisely what would be expected of an in vivo amplification signal such as the humoural immune response. This is in contrast to cancer-associated antigens, which are markers of tumour burden and not useful for the early detection or screening of breast [46, 47] or colorectal cancer [48, 49].
Previous publications [16–24, 50] have highlighted the potential value of a panel of autoantibodies for the early detection of cancer. Using a panel of antigens, autoantibodies have been reported up to 5 years before screening CT scans [22] in LC and up to 4 years before screening mammography in young women at increased risk [21, 23]. Other authors have highlighted individual autoantibodies such as p53 autoantibodies detected before diagnosis of cancer in smokers with chronic obstructive pulmonary disease [27] or in patients with asbestosis [41]. In the latter publication, the average lead time (time from first positive sample to diagnosis) was 3.5 years (range 1–12 years). There are similar publications on other single autoantibodies [45, 51, 52]. These findings all indicate the induction of autoantibodies happening relatively early in the process of carcinogenesis.
This panel assay is the first to show reproducible results with a calibration and control system and offers a potential system for monitoring a population at high risk of LC, either alone or in conjunction with imaging modalities (e.g. CT). The similar sensitivities and specificities measured for these three datasets and with different batches of proteins utilised emphasise the robustness of these autoantibody assays and also confirm the value of a panel of autoantibodies over a single autoantibody assay.
At a fixed 90% specificity, the sensitivity of 40% is a conservative estimate of the performance of the assay both in terms of estimating the level of clinically occult LCs (supplemental Table S2, available at Annals of Oncology online) and also the sensitivity reported for SCLC (n = 73) in group 3. The latter is lower than the 55% sensitivity and 90% specificity, which the authors will report in a larger consecutive series (n = 242) from a single centre (C. J. Chapman, A. J. Thorpe, A. Murray et al., unpublished data).
The sensitivity of 40% with a specificity of 90% are similar to mammography in high-risk young women [53], while the incidence of LCs in heavy smokers is at least three times the incidence of breast cancer in a typical cohort of high-risk young women [5, 7]. Therefore, in terms of absolute number of cancers, this test should detect more LCs for every 1000 high-risk persons tested than screening mammography would detect breast cancers in a high-risk group of young women, even if mammography were 100% sensitive rather than its current 40% [53]. This has to be seen in the context of a disease (i.e. LC), which has a mortality rate between 85% and 95%. By way of contrast, annual CT in the Mayo CT screening trial had a specificity of 49% (with a sensitivity of 67%) in the prevalence round. In an at-risk group of 20 of the 1000, CT gave a PPV of 2.5% (i.e. 1:40) and an NPV of 98.7%. The autoantibody test with a sensitivity of 40% and a specificity of 90% would have a PPV of 7.5% (i.e. 1 in 13.3) and an NPV of 98.7% in a similar-risk group.
While such comparisons serve to highlight the potential value of an autoantibody test for LC that has a specificity of 90%, the authors envisage the autoantibody technology and imaging as being complementary.
funding
Oncimmune Ltd and the University of Nottingham.
disclosure
CJC and JFRR are consultants to Oncimmune Ltd, a University of Nottingham spinout company and JFRR holds stock. AM and GH are full-time employees of Oncimmune Ltd. CR holds stock option and is also a consultant to Oncimmune Ltd. WCW is the scientific advisor for Oncimmune Ltd. GH is an employee of Oncimmune Ltd. GHF is the Chairman of Oncimmune Ltd and holds stock. ACB holds stock and options in Oncimmune Ltd and has a significant conflict.
Supplementary Material
Acknowledgments
The authors would like to thank Petra Stieber and Joachim von Pawel for their role in the collection of samples and data included in this study. They also acknowledge Céline Parsy-Kowalska’s role in protein production and Jared Allen’s contribution as the data manager. The manuscript was drafted by the authors. However, the authors acknowledge and thank Sandra Cuscó, PhD, from Complete Medical Communications, who provided final editorial assistance funded by Oncimmune Ltd and the University of Nottingham.
appendix 1
demographic characteristics of the control versus the study population
A total of 655 lung cancer (LC) sera (476 were from patients with non-small-cell lung cancer, 165 with small-cell lung cancer, 1 lung sarcoma and 13 of unknown histology) were compared directly with 655 normal sera, which were analysed as controls. In addition, sera from 88 unmatched individuals (25 normal and 63 with benign lung conditions) supplied by the group 2 centre were analysed as controls to check the positivity rate in known benign lung disease. Samples were obtained, with full informed consent, at different sites.
Controls for patients in group 1 were matched on the basis of gender and age (±4 years). As all subjects in this group were smokers, pack-year matching was attempted, but a tight match was prohibited by lack of information. There were 81 males and 64 females in the LC group and 83 males and 62 females in the control group. The median age (range) of the LC patients and controls were 66 (41–87) and 66 (41–87) years, respectively.
In group 2, there were 172 males and 69 females in the LC group and 171 males and 69 females in the control group. The median age (range) of the LC patients and controls were 63 (28–87) and 63 (28–87) years, respectively. Controls for group 2 were selected from a prospective collection of blood samples taken from a larger sample set of a normal population in the Midlands of England. Patients with LC were initially matched to controls on the basis of gender, age (±3 years) and smoking history. In <5% of cases, these criteria could not be met, so a choice had to be made to either extend the age-match criteria or ignore the gender-match stipulation. Since the authors have never observed a significant gender difference, age and smoking history were given priority over gender. In 37 LC patients, the exact smoking history was unknown, and in a further four patients, age matching was >3 years.
The group 2 centre also supplied 88 unmatched samples from individuals who were either thought to be normal (n = 25) or have a range of benign lung diseases (n = 63), including mass/nodule (n = 3), autoimmune lung disease (n = 10), chronic obstructive pulmonary disease/emphysema (n = 2), benign pleural effusion (n = 2), allergic/inflammatory/infective conditions (n = 25) (e.g. allergic alveolitis, Wegner’s granulomatosis, asthma, sarcoid, vasculitis, Dessler’s syndrome, mycoplasmosis, tuberculosis) and nonspecified lung disorders (n = 21). A set of individually matched controls for this group of LC patients was selected from a prospective collection of blood samples taken from a normal population in the UK. Controls were matched on the basis of gender and age. With the exception of one patient who was matched to ±4 years, controls were matched to patient age ±2 years. Smoking history was not known for the patients with LC, so controls were simply selected from a population of smokers and ex-smokers.
In group 3, there were 199 males and 70 females in the LC group and 187 males and 82 females in the control group. The median age (range) in the LC and control groups was 65 (38–87) and 65 (38–86) years, respectively.
The matched controls in group 3 were collected as part of a larger sample set of the normal population (n = 766) in the Midwest United States and demographic data included ethnicity. Evaluation of calibrated reference unit (RU) for autoantibody expression demonstrated that when controlled for age, there was no significant difference between ethnic groups [Caucasians (n = 614), African Americans (n = 108), Hispanics (n = 27) and Native Americans (n = 17)] in terms of calibrated RUs (data not shown). There was a further set of samples from 125 normal individuals who were located in Florida and age matched, gender matched and smoking history matched to a similar number of the controls in the Midwest United States (n = 125). The Florida samples were part of another larger prospective collection of sera from the normal population. Comparison of the 125 samples from each of these two normal populations from different geographic and ethnic backgrounds showed no significant difference in the calibrated RU values for any of the six antigens (data not shown).
References
- 1.Boyle P, Levin B, editors. World Cancer Report 2008. Lyon, France: IARC; 2008. [Google Scholar]
- 2.Horner MJ, Ries LAG, Krapcho M, et al., editors. SEER Cancer Statistics Review, 1975–2006. Bethesda, MD: National Cancer Institute; 2009. http://seer.cancer.gov/csr/1975_2006/, based on November 2008 SEER data submission, posted to the SEER website (February 2010, date last accessed) [Google Scholar]
- 3.Sone S, Takashima S, Li F, et al. Mass screening for lung cancer with mobile spiral computed tomography scanner. Lancet. 1998;351:1242–1245. doi: 10.1016/S0140-6736(97)08229-9. [DOI] [PubMed] [Google Scholar]
- 4.Henschke CI, McCauley DI, Yankelevitz DF, Altorki NK. Early lung cancer action project: overall design and findings from baseline screening. Lancet. 1999;354:99–105. doi: 10.1016/S0140-6736(99)06093-6. [DOI] [PubMed] [Google Scholar]
- 5.Swensen SJ, Jett JR, Hartman TE, et al. CT screening for lung cancer: five-year prospective experience. Radiology. 2005;235:259–265. doi: 10.1148/radiol.2351041662. [DOI] [PubMed] [Google Scholar]
- 6.Sobue T, Moriyama N, Kaneko M, et al. Screening for lung cancer with low-dose helical computed tomography: anti-lung cancer association project. J Clin Oncol. 2002;20:911–920. doi: 10.1200/JCO.2002.20.4.911. [DOI] [PubMed] [Google Scholar]
- 7.Henschke CI, Naidich DP, Yankelevitz DF, et al. Early lung cancer action project: initial findings on repeat scanning. Cancer. 2001;92:153–159. doi: 10.1002/1097-0142(20010701)92:1<153::aid-cncr1303>3.0.co;2-s. [DOI] [PubMed] [Google Scholar]
- 8.Diederich S, Wormanns D, Semik M, et al. Screening for early lung cancer with low-dose spiral CT: prevalence in 817 asymptomatic smokers. Radiology. 2002;222:773–781. doi: 10.1148/radiol.2223010490. [DOI] [PubMed] [Google Scholar]
- 9.Nawa T, Nakagawa T, Suzushi S, et al. Lung cancer screening using low-dose spiral CT: results of baseline and 1-year follow-up studies. Chest. 2002;122:15–20. doi: 10.1378/chest.122.1.15. [DOI] [PubMed] [Google Scholar]
- 10.Gohagan J, Marcus P, Fagerstrom R, et al. Writing Committee, Lung Screening Study Research Group. Baseline findings of a randomized feasibility trial of lung cancer screening with spiral CT scan vs chest radiograph: the Lung Screening Study of the National Cancer Institute. Chest. 2004;126:114–121. doi: 10.1378/chest.126.1.114. [DOI] [PubMed] [Google Scholar]
- 11.McWilliams A, Mayo J, MacDonald S, et al. Lung cancer screening: a different paradigm. Am J Respir Crit Care Med. 2003;168:1167–1173. doi: 10.1164/rccm.200301-144OC. [DOI] [PubMed] [Google Scholar]
- 12.Kaneko M, Eguchi K, Ohmatsu H, et al. Peripheral lung cancer: screening and detection with low-dose spiral CT versus radiography. Radiology. 1996;201:798–802. doi: 10.1148/radiology.201.3.8939234. [DOI] [PubMed] [Google Scholar]
- 13.Berrington de Gonzalez A, Mahesh M, Kim K, et al. Projected cancer risks from computed tomography scans performed in the United States in 2007. Arch Intern Med. 2009;169:2071–2077. doi: 10.1001/archinternmed.2009.440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sone S, Li F, Yang ZG, et al. Results of three-year mass screening programme for lung cancer using mobile low-dose spiral computed tomography scanner. Br J Cancer. 2001;84:25–32. doi: 10.1054/bjoc.2000.1531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.McWilliams AM, MacAulay C, Gazdar AF, Lam S. Innovative molecular and imaging approaches for the detection of lung cancer and its precursor lesions. Oncogene. 2002;21:6949–6959. doi: 10.1038/sj.onc.1205831. [DOI] [PubMed] [Google Scholar]
- 16.Tan EM. Autoantibodies as reporters identifying aberrant cellular mechanisms in tumorigenesis. J Clin Invest. 2001;108:1411–1415. doi: 10.1172/JCI14451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Houghton AN. Cancer antigens: immune recognition of self and altered self. J Exp Med. 1994;180:1–4. doi: 10.1084/jem.180.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Brichory FM, Misek DE, Yim AM, et al. An immune response manifested by the common occurrence of annexins I and II autoantibodies and high circulating levels of IL-6 in lung cancer. Proc Natl Acad Sci U S A. 2001;98:9824–9829. doi: 10.1073/pnas.171320598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zhang J, Casiano CA, Peng X, et al. Enhancement of antibody detection in cancer using panel of recombinant tumor-associated antigens. Cancer Epidemiol Bio and Prev. 2003;12:136–143. [PubMed] [Google Scholar]
- 20.Nesterova M, Johnson N, Cheadle C, et al. Autoantibody biomarker opens a new gateway for cancer diagnosis. Biochimica et Biophysica Acta. 2006;1762:398–403. doi: 10.1016/j.bbadis.2005.12.010. [DOI] [PubMed] [Google Scholar]
- 21.Robertson JFR, Chapman CJ, Cheung KL, et al. Auto-antibodies in early breast cancer. J Clin Oncol. 2005;23:549. [Google Scholar]
- 22.Zhong L, Coe SP, Stromberg AJ, et al. Profiling tumor-associated antibodies for early detection of non-small cell lung cancer. J Thor Oncol. 2006;1:513–519. [PubMed] [Google Scholar]
- 23.Chapman C, Murray A, Chakrabarti J, et al. Autoantibodies in breast cancer: their use as an aid to early diagnosis. Ann Oncol. 2007;18:868–873. doi: 10.1093/annonc/mdm007. [DOI] [PubMed] [Google Scholar]
- 24.Chapman CJ, Murray A, McElveen JE, et al. Autoantibodies in lung cancer—possibilities for early detection and subsequent cure. Thorax. 2008;63:228–233. doi: 10.1136/thx.2007.083592. [DOI] [PubMed] [Google Scholar]
- 25.Crawford LV, Pim DC, Bulbrook RD. The detection of antibodies against the cellular protein p53 in sera from patients with breast-cancer. Int J Cancer. 1982;30:403–408. doi: 10.1002/ijc.2910300404. [DOI] [PubMed] [Google Scholar]
- 26.Li Y, Brandt-Rauf PW, Carney WP, et al. Circulating anti-p53 antibodies in lung cancer and relationship to histology and smoking. Biomarkers. 1999;4:381–390. doi: 10.1080/135475099230769. [DOI] [PubMed] [Google Scholar]
- 27.Trivers GE, DeBenedetti VM, Cawley HL, et al. Anti p53 antibodies in sera from patients with chronic obstructive pulmonary disease can predate a diagnosis of cancer. Clin Cancer Res. 1996;2:1767–1775. [PubMed] [Google Scholar]
- 28.Gnjatic S, Nishikawa H, Jungbluth AA, et al. NY-ESO-1: review of an immunogenic tumor antigen. Adv Cancer Res. 2006;95:1–30. doi: 10.1016/S0065-230X(06)95001-5. [DOI] [PubMed] [Google Scholar]
- 29.Cho B, Lim Y, Lee DY, et al. Identification and characterization of a novel cancer/testis antigen gene CAGE. Biochem Biophys Res Commun. 2002;292:715–726. doi: 10.1006/bbrc.2002.6701. [DOI] [PubMed] [Google Scholar]
- 30.Krause P, Tureci O, Micke P, et al. SeroGRID: an improved method for the rapid selection of antigens with disease related immunogenicity. J Immunol Methods. 2003;283:261–267. doi: 10.1016/j.jim.2003.09.014. [DOI] [PubMed] [Google Scholar]
- 31.Türeci O, Mack U, Luxemberger U, et al. Humoral responses of lung cancer patients against tumor antigen NY-ESO-1. Cancer Lett. 2006;236:64–71. doi: 10.1016/j.canlet.2005.05.008. [DOI] [PubMed] [Google Scholar]
- 32.Vural B, Chen L-C, Saip P, et al. Frequency of SOX Group B (SOX1, 2, 3) and ZIC2 antibodies in Turkish patients with small cell lung carcinoma and their correlation with clinical parameters. Cancer. 2005;103:2575–2583. doi: 10.1002/cncr.21088. [DOI] [PubMed] [Google Scholar]
- 33.Maddison P, Thorpe A, Silcocks P, et al. Autoimmunity to SOX2, clinical phenotype and survival in patients with small-cell lung cancer. Lung Cancer. 2010 doi: 10.1016/j.lungcan.2010.03.002. April 3 [Epub ahead of print] [DOI] [PubMed] [Google Scholar]
- 34.Robertson JFR, Barnes T, Murray A, Chapman C. Improved Immunoassay Methods WO2006/126/008. Nottingham, GB: Oncimmune Ltd; 2006. [Google Scholar]
- 35.Murray A, Chapman CJ, Healey G, et al. Technical validation of an autoantibody test for lung cancer. Ann Oncol. 2010;21:1687–1693. doi: 10.1093/annonc/mdp606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Robertson JFR, Murray A, Chapman C, Barnes A. Calibrator for Immunoassays WO2009/081165. Nottingham, GB: Oncimmune Ltd; 2007. [Google Scholar]
- 37.Motulsky H, Christopoulos A. Fitting Models to Biological Data Using Linear and Nonlinear Regression (Chapter 41), Madison Avenue, NY: Oxford University Press; 2004. [Google Scholar]
- 38.Geyer C. Estimation and optimization of functions. In: Gilks WR, Richardson ST, Speigelhalter DJ, editors. Markov Chain Monte Carlo In Practice. London: Chapman and Hall; 1999. pp. 241–258. [Google Scholar]
- 39.Spitz MR, Hong WK, Amos CI, et al. A risk model for prediction of lung cancer. J Natl Cancer Inst. 2007;99:715–726. doi: 10.1093/jnci/djk153. [DOI] [PubMed] [Google Scholar]
- 40.Sangrajrang S, Sornprom A, Chernrungroj G, Soussi T. Serum p53 antibodies in patients with lung cancer: correlation with clinicopathologic features and smoking. Lung Cancer. 2003;39:297–301. doi: 10.1016/s0169-5002(02)00509-3. [DOI] [PubMed] [Google Scholar]
- 41.Li Y, Karjalainen A, Koskinen H, et al. p53 Autoantibodies predict subsequent development of cancer. Int J Cancer. 2005;114:157–160. doi: 10.1002/ijc.20715. [DOI] [PubMed] [Google Scholar]
- 42.Cho B, Lee H, Jeong S, et al. Promoter hypomethylation of a novel cancer/testis antigen gene CAGE is correlated with its aberrant expression and is seen in premalignant stage of gastric cancer. Biochem Biophys Res Commun. 2002;307:52–63. doi: 10.1016/s0006-291x(03)01121-5. [DOI] [PubMed] [Google Scholar]
- 43.Qiu J, Choi G, Li L, et al. Occurrence of autoantibodies to annexin I, 14-3-3 theta and LAMR1 in prediagnostic lung cancer sera. J Clin Oncol. 2008;26:5060–5066. doi: 10.1200/JCO.2008.16.2388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Gure AO, Stockert E, Scanlan M, et al. Serological identification of embryonic neural proteins as highly immunogenic tumor antigens in small cell lung cancer. Proc Natl Acad Sci U S A. 2000;97:4198–4203. doi: 10.1073/pnas.97.8.4198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Pereira-Faca SR, Kuick R, Puravs E, et al. Identification of 14-3-3u as an antigen that induces a humoral response in lung cancer. Cancer Res. 2007;67:12000–12006. doi: 10.1158/0008-5472.CAN-07-2913. [DOI] [PubMed] [Google Scholar]
- 46.Robertson JFR, Pearson D, Price MR, et al. Assessment of four monoclonal antibodies as serum markers in breast cancer. Eur J Cancer. 1990;26:1127–1132. doi: 10.1016/0277-5379(90)90268-x. [DOI] [PubMed] [Google Scholar]
- 47.Robertson JFR, Pearson D, Price MR, et al. Prospective assessment of the role of five serum markers in breast cancer. Cancer Immunol Immunother. 1991;33:403–410. doi: 10.1007/BF01741602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Laurence DJR, Stevens U, Bettelheim R, et al. Role of plasma carcinoembryonic antigen in the diagnosis of gastro-intestinal, mammary and bronchial carcinoma. Br Med J. 1972;3:605–609. doi: 10.1136/bmj.3.5827.605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Thomas MW, Robertson JFR, Price MR, Hardcastle JD. Failure of CA 19-9 to detect asymptomatic colorectal carcinoma. Br J Cancer. 1991;63:975–976. doi: 10.1038/bjc.1991.213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Robertson JFR, Graves CRL, Price MR. Tumour Markers US 7,402,403, B1. Nottingham, GB: Oncimmune Ltd; 1999. [Google Scholar]
- 51.Rohayem J, Diestelkoetter P, Weigle B, et al. Antibody response to the tumor-associated inhibitor of apoptosis protein surviving in cancer patients. Cancer Res. 2000;60:1815–1817. [PubMed] [Google Scholar]
- 52.Suzuki H, Graziano DF, McKolanis J, Finn OJ. T cell-dependent antibody responses against aberrantly expressed cyclin B1 protein in patients with cancer and premalignant disease. Clin Cancer Res. 2005;11:1521–1526. doi: 10.1158/1078-0432.CCR-04-0538. [DOI] [PubMed] [Google Scholar]
- 53.Pisano ED, Gatsonis C, Hendrick E, et al. Diagnostic performance of digital versus film mammography for breast cancer screening. N Engl J Med. 2005;353:1773–1783. doi: 10.1056/NEJMoa052911. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.