Abstract
The goal of the present investigation was to characterize the development of inhibitory control, an aspect of executive functions, in a sample of prenatally cocaine exposed (CE; n = 165) children compared to an at risk, but prenatally cocaine unexposed (NCE; n = 119) sample across time (i.e. 7.5 to 11.5 years of age). Gender and cumulative risk, a combination of postnatal medical (i.e. low birth weight and APGAR scores) and demographic risk, indexed by maternal educational attainment, were examined as predictors of change in inhibitory control across time and aggression was modeled as an outcome when children reached 14 years of age. Multiple group latent growth models indicated that CE children made more errors at 7.5 years of age during a standard Stroop interference task, however, over time CE children had greater age-related improvements, narrowing the initial gap, with NCE children in the ability to inhibit errors. Gender effects at 7.5 years within the NCE group were identified with NCE boys making initially more errors than NCE girls; both NCE and CE girls improved faster across development compared to NCE and CE boys, respectively. Greater cumulative risk was associated with more errors at 7.5 years in the CE and NCE groups. No differences were observed between CE and NCE children on time to complete the Stroop task at 7.5 years. However, NCE children had greater age-related improvements in their time to complete the Stroop interference task relative to their CE counterparts. NCE girls improved the fastest over time relative to NCE boys; a similar trend emerged (p < .10) with CE girls improving faster over time than CE boys. Although all participants improved across development, higher cumulative risk in both groups was associated with slower age-related improvements (i.e. higher slopes) in the time to complete the Stroop task across development. After accounting for gender and cumulative risk, findings in both groups indicated that those who made more errors at 7.5 years of age and/or who had slower age-related changes (i.e. higher slopes) of time to complete the Stroop task across development were more aggressive as rated by caregivers at 14 years of age. Although qualified by gender and cumulative risk, these findings are consistent with reduced cognitive processing efficiency and executive function difficulties in CE children relative to NCE children. Findings suggest that executive function difficulties in CE children may be subtle as development continues to unfold over time. Furthermore, these findings indicate that development of inhibitory control may be an important mechanism linking prenatal cocaine exposure, gender, and cumulative risk to later adverse outcomes.
Keywords: Prenatal Cocaine Exposure, Executive Functions, Inhibitory Control, Children, Development, Latent Growth Modeling
1. Introduction
The frontal lobes are critical for diverse cognitive functions, such as planning, working memory, prepotent response inhibition, and set shifting as well as other cognitive processes that fall under the rubric of executive functions (1, 2, 3). One aspect of executive functions that is particularly important over the course of development is inhibitory control. Inhibitory control refers to the ability to inhibit a prepotent (i.e. over learned) dominant response in favor of activating a subdominant response (4, 5, 6). While effective inhibitory control has been implicated in better emotion regulation, reasoning, and one’s ability to maintain efforts toward attainment of goals (7, 8, 9, 10), ineffective inhibitory control has been implicated in the development of psychopathology (e.g., ADHD) and externalizing and internalizing difficulties in general (11, 12, 13, 14, 15, 16).
Under typical prenatal conditions, the development of inhibitory processes has been studied in a variety of age groups, from infancy to adulthood, using various tasks. For example, Diamond (17) found improvements during performance on the A not B task in infants from the ages of 7 to 12 months. Kochanska et al. (5) identified improvements in toddler’s ability to delay gratification in two paradigms, “Gift in a Bag” and “Wrapped Gift,” from 22 to 33 months of age, indicating continued improvements during performance of inhibitory control tasks prior to the preschool age period. In older age groups, other’s have observed improvements during performance of inhibitory control tasks from early childhood to late adolescence (e.g., 18) and into adulthood (19).
Imaging (e.g., 20, 21, 22, 23) and electrophysiological (e.g., 24, 25, 26) studies have demonstrated the importance of the frontal lobes during performance of inhibitory control tasks as well as developmental changes in frontal activation during such tasks. In regard to the latter, changes in white matter in the frontal regions appear to underlie changes in performance during tasks that recruit frontal areas for successful completion (for a review, see 27 or 28). In sum, evidence from behavioral inhibitory control tasks, neuroimaging (e.g., fMRI), and electrophysiological (e.g., ERP) studies support the role of the frontal lobes during inhibition of prepotent responses and suggest improvements in inhibitory task performance and changes in brain activation from early in life through adulthood (For a more thorough overview see 4). Furthermore, the studies reviewed above provide evidence of how the development of inhibitory control unfolds across the life span in low-risk, typically developing individuals.
While the studies outlined above are important for understanding typical development of inhibitory control, there are two notable limitations across these studies. First, most studies use cross-sectional designs (e.g., 18, 19, 23) rather than following a single sample of participants over time. While cross-sectional research designs are useful and can identify age group and interindividual differences, information regarding individual differences within intraindividual change and age changes is not obtained when using a cross-sectional design (29, 30). A further limitation of most existing studies of inhibitory control development is the use of low-risk populations (For a recent exception in young children, see 31). While low risk samples define what might be expected under ideal circumstances, such samples do not identify potentially important patterns of development in individuals who have experienced serious challenges to normative developmental trajectories. These challenges encompass prenatal factors such as substance exposure as well as postnatal economic adversity and serious psychopathology among caregivers.
Cocaine exposure in particular has been linked to difficulties with behavior, attention, and executive functions and longitudinal studies of children exposed prenatally to cocaine have identified negative effects of prenatal exposure during performance on various measures of cognitive and emotional functioning, including behavioral measures of executive functions. Early in life, infants exposed to cocaine in utero have been identified as having a “difficult temperament” (i.e. fussy, harder to soothe), which has been linked with later behavioral difficulties, and lower performance during the Bayley Scales of Infant Development motor scales (32). Similarly, Mayes, Cicchetti, Acharyya, and Zhang (33) found that infants exposed to cocaine obtained lower scores on the Bayley Scales of Infant Development, 2nd Ed., compared to non-exposed infants and infants who were exposed in utero to non-cocaine substances (e.g., alcohol). In addition to reduced performance on broad based cognitive measures relative to controls, infants and toddlers exposed to cocaine prenatally have been identified as performing lower on specific measures of cognitive ability, such as visual attention (e.g., 34), spatial working memory (35, 36), and inhibitory control and executive functions (e.g., 37) relative to their non-exposed, same age peers. These findings are consistent with theoretical work, which has drawn on animal and human studies, indicating that prenatal cocaine exposure affects arousal regulatory systems, which then effects prefrontal lobe function (See 38 for additional discussion).
While early work examining the effects of prenatal cocaine exposure has typically covered the developmental periods of infancy through toddlerhood, more recent follow-up studies have been carried out that suggest that individuals exposed to cocaine in utero continue to have difficulties in domains potentially affected by ineffective inhibitory control (e.g., behavioral problems; 15, 16). For example, during the preschool period, children exposed in utero to cocaine were identified as experiencing more frustration and difficulties regulating problem solving behaviors relative to non-exposed children (39). Similarly, Richardson Goldschmidt, and Willford (40) found that preschoolers exposed to cocaine in utero were rated as having more behavioral difficulties and as being more fussy/difficult than children who were not exposed to cocaine in utero. School-aged children prenatally exposed to cocaine have also displayed greater levels of behavioral difficulties relative to their non-exposed peers (41).
In addition to accumulating evidence of inhibitory control difficulties in the prenatally cocaine exposed population, there is growing recognition that some characteristics of prenatally exposed children, such as gender and sociodemographic and medical risk factors, may influence broad cognitive ability, inhibitory control, and/or outcomes associated with inhibitory control difficulties. For example, several studies examining animal models of cocaine exposure have identified greater performance difficulties in male rats relative to female rats (42,43). Gender effects, with males prenatally exposed to cocaine performing more poorly than prenatally exposed females on measures of cognitive function (44) and inhibitory control (45), in human participants have also been reported. Greater behavioral difficulties, potentially linked to poor inhibitory control, have also been reported in males compared to females prenatally exposed to cocaine (46,47). Likewise, in later childhood (i.e. between 10 and 11 years), in utero exposure to cocaine has been associated with increased risky behavior in males (48). Other studies have identified low birth weight and sociodemographic risk as contributing to inhibitory control difficulties in children. For instance, a recent meta-analysis identified early gestational age as a risk factor for executive function difficulties 49); similarly, Li-Girning (50) noted that low birth weight was a particular risk factor for difficulties with inhibition and executive attention. Residing in a low income household (e.g., 31) and/or low maternal education (51) have also been noted as potential sociodemographic characteristics that can contribute to poor executive functions in children.
While the findings reported above are important, there are some noted limitations. For example, most studies reviewed reflect comparisons of cocaine exposed and non-cocaine exposed children at single time points, although all were identified and followed since birth. Only three studies were identified (33,44,52) that have reported on repeated measures data to understand differential development of cognitive ability over time in cocaine exposed compared to non-cocaine exposed participants. However, these studies use either measures of broad cognitive ability or language, typically report on data from the preschool through the school-aged years (44,52), or report on data through the toddler period (33). Studies identified in the current review have not reported on the development of a specific executive function (i.e. inhibitory control) from school-age to early adolescence despite recognition that the effects of prenatal cocaine exposure may be largely subtle, necessitating the need for a more focused cognitive assessment (40). Furthermore, the effects of gender and cumulative risk on inhibitory control development across time in prenatally cocaine exposed children have not been examined despite evidence of gender differences in inhibitory control within this population and the effects of cumulative risk (e.g., low birth weight and sociodemographic risk) on inhibitory control in general.
1.1. The Current Study
Two related issues have been identified: 1) longitudinal work examining inhibitory control development using repeated measures during the time period spanning school-aged to pre-adolescence is limited, and 2) longitudinal studies characterizing the development of specific executive functions in children exposed to cocaine prenatally also have not been reported. The current study seeks to contribute to these areas by examining the development of inhibitory control over a 6 year period spanning the important developmental years from late childhood into pre-adolescence in prenatally cocaine exposed children and in high risk, non-cocaine exposed children using LGM. This time period is particularly important given the ongoing development and further refinement of the ability to inhibit prepotent responses (See 53 for an overview). To evaluate inhibitory control in the current study, a standard Stroop task was administered to children at 7.5, 9.5, and 11.5 years of age. This measure was selected for several reasons: 1) the Stroop task has been commonly used as a clinical measure of executive functions and inhibitory control in neuropsychological evaluations, 2) imaging evidence from prior studies (e.g., 54,55) indicates that the Stroop Color/Word task activates areas in the frontal lobes that have been implicated in theoretical models of the effects of cocaine on executive functions (e.g., 38), and 3) prior work has identified Stroop performance deficits among children with disorders associated with executive dysfunction (e.g., ADHD; 56). Consistent with existing developmental literature, it was anticipated that improvements in inhibitory control would be evident during the transition from late childhood to pre-adolescence in both groups of participants. Nevertheless, consistent with the literature examining the effects of prenatal cocaine exposure on executive functions, it was anticipated that children exposed to cocaine in utero would display a pattern of development consistent with inhibitory control dysfunction (i.e., taking longer to complete the Stroop task and making more errors at 7.5 years and trajectories consistent with slower age-related improvements in task performance) relative to non-cocaine exposed participants.
Although the primary goal of this investigation is to add to the growing literature examining change over time in inhibitory control in high risk groups, such as children who experienced prenatal cocaine exposure, several secondary goals were also examined. First, the effects of gender on inhibitory control development in both groups were examined in the current investigation. Based on prior findings (e.g., 44,45), we anticipated that interindividual differences in Stroop performance across time would be accounted for by gender, with males exhibiting greater difficulties while performing the Stroop task than females. Given the nature of the analyses performed in the current study, examining potential gender effects allows for the examination of possible Gender by Exposure Group by Time interactions. Furthermore, given work that suggests that medical (e.g., low birth weight) and socidemographic (e.g. low maternal education) risk can contribute to executive function outcomes (31,49,50,51), we also examined the contribution of the combination of these risk factors to the development of inhibitory control.
Finally, we examined the effects of inhibitory control development on aggressive behavior during adolescence. Given prior work linking poor inhibitory control with behavioral difficulties (11, 12, 13, 14, 15, 16), we anticipated that, after controlling for gender and cumulative risk, those children who performed more poorly during the initial Stroop evaluation as well as children who had slower, age-related change (i.e. higher slopes) in inhibitory control, both in terms of errors and length of time to complete the task, would have more difficulties with aggressive behavior as rated by parents. Group differences are examined between prenatally cocaine exposed and prenatally cocaine unexposed participants on associations between Stroop performance over time and aggressive behavior outcomes, but specific expectations were not formulated given mixed findings in the literature regarding behavioral difficulties in prenatally cocaine exposed compared to unexposed participants (e.g., 15,16,40,41,57,58).
2. Method
2.1 Participants
Participants in the current study consisted of 293 high-risk families (i.e. families from a low-income, urban environment) with new born infants exposed to cocaine (and other drugs; n = 172 or 58.7%), exposed to non-cocaine substances (e.g., alcohol, tobacco, and/or marijuana; n = 46 or 15.7%), or non-drug exposed (n = 75 or 25.6%) that were recruited from a moderately sized northeastern urban area to participate in a longitudinal study examining normal and abnormal development of executive and regulatory functions. Children in the present sample were drawn from a larger sample of 369 children who have been participating in a fifteen year longitudinal study of the effects of fetal cocaine exposure on physical, cognitive, social and emotional development. Children (and their mothers) who were exposed to drugs other than cocaine (primarily alcohol, tobacco, and/or marijuana) were also enrolled in the study, as were non-drug using mothers and their non-drug exposed children. The sample was recruited at birth over a five-year period and children and their parents have been seen biannually. Families were originally recruited when they presented for prenatal care at the Women’s Center of Yale-New Haven Hospital or, in the case of no prenatal care, when they were admitted to the postpartum ward. The larger sample from which the children for this study were drawn consisted of 81 % African American, 6.5% Hispanic and 12.5% Caucasian children, all of who come from the greater New Haven area. Out of the 76 participants not included in the current analyses, 67 did not have any relevant data available for the visits in question (i.e. were missing IQ data or all Stroop assessments) and 9 participants (3 non-drug exposed participants, 1 non-cocaine, but other drug exposed participant, and 5 cocaine exposed participants) were excluded from participation due to an IQ lower than 70. With regard to missing data (i.e. only those that were either missing IQ data or all Stroop assessments), there were no differences between these participants and participants with data present in terms of maternal age and birth weight (p’s > .05); however, there was a difference between these groups of participants on head circumference (M = 33.29 cm’s for those without missing data vs. M = 32.72 cm’s for those with missing data, p < .05).
Prenatal drug-exposure status was ascertained at the time of recruitment into the longitudinal follow-up study (either prenatally or at the time of delivery). For those women interviewed prenatally, the majority were interviewed in their late second or third trimester. After obtaining verbal consent for an interview, all women were questioned about substance use in a detailed interview that covered lifetime use (prior to the current pregnancy) of cocaine, tobacco, alcohol, marijuana, and other drugs (e.g., sedatives, opiates), and frequency and amount of use of these agents during the preceding 30 days.
Women interviewed prenatally were also interviewed again at the time of delivery either to confirm their continued use or to ascertain new use. For all women regardless of drug use history, a urine sample was obtained for toxicology. Standard urine screening for drug level or metabolites of cocaine (e.g., benzoylecognine), opioids, benzodiazepines, and tetrahydrocannabinol (THC) was performed using the Abbott TDx system and the recommended cutoff levels (59). Urine samples were rated as positive if the quantity of drug or metabolite was > 300 gms/ml. The TDx system is highly sensitive and specific for the detection of illicit drug use, and benzoylecognine is detectable for three days after use (60).
Prenatal cocaine-exposure status was determined by a combination of maternal report and urine toxicology from the prenatal or immediate postpartum period. Infants were considered cocaine-exposed prenatally if maternal self-reports were positive, even if urine toxicological results were negative. Conversely, if mothers reported that they did not use cocaine, but clinic or hospital urine toxicological results were positive, infants were also considered exposed. Every mother with a positive history and/or positive urine toxicology for cocaine-use was invited to join the study. Non-cocaine-exposed status was ascertained by negative urine toxicology and a negative maternal history of cocaine during pregnancy and at the time of delivery. Non-cocaine/non-substance exposed participants were eligible to participate as a non-substance exposed comparison group.
Of the 293 participants, as stated above, nine were eliminated from analyses due to IQ’s lower than 70. Differences between non-exposed and non-cocaine, but other drug exposed participants were not observed on head circumference, first or second APGAR scores, IQ scores (measured at 48 months of age), or maternal age; however, non-exposed infants attained significantly higher birth weights (M = 6.64 lbs, SD = 1.12) than infants prenatally exposed to non-cocaine substances (M = 6.16 lbs, SD = 1.27; t [114] = 2.14, p = .03). Differences between non-exposed and non-cocaine, but other drug exposed children also were not observed on the 168 month CBCL aggression outcome. Given the aims of the current study and the general lack of evidence of developmental differences between non-drug exposed and non-cocaine exposed participants, these participants were collapsed into one group herein called the NCE comparison group (NCE). This resulted in 119 NCE participants and 165 cocaine-exposed (CE) participants.
Most participants reported an African-American racial background (68.1% of NCE participants and 87.3% cocaine-exposed participants) with the cocaine-exposed group consisting of significantly greater numbers of minority participants than the NCE group, χ2 (4) = 20.90, p < .05. Consistent with the at risk nature of both participant groups, 14.30% of primary caregivers of NCE participants reported attaining less than a high school education level while 26.70% of primary caregivers of CE participants reported attaining less than a high school education. Similarly, 2.5% of primary caregivers of NCE participants and none of the cocaine-using primary caregivers reported attaining a bachelor’s degree. Despite the relatively low educational attainment of both groups, between group educational differences were observed, χ2 (3) = 14.89, p < .05, with primary caregivers of CE participants attaining significantly less education than primary caregivers of NCE participants. Despite ethnic and education differences between groups, interactions between groups, examined with ANOVAs, were not significant for any of the stroop assessments (i.e. time to complete the stroop color/word task or errors during the stroop color/word task; all ps > .05)1.
Approximately equal numbers of males (43.7% vs. 50.3%) and females (56.3% vs. 49.7%) were in the NCE and cocaine-exposed groups, respectively. Two (Exposure Group: NCE vs. cocaine + other drug exposed) by two (Gender: males vs. females) ANOVA’s were conducted to examine group differences on birth weight, head circumference, first and second APGAR scores, and 48 month IQ assessments. Consistent with the higher-risk nature of the CE group, main effects of exposure group were obtained on measures of birth weight, head circumference, second APGAR scores, and IQ. An exposure group main effect was not identified on first APGAR scores. No main effects for gender were identified for the variables examined. Exposure Group by Gender interaction effects were also not significant, indicating that gender effects unique to a given group were not observed in terms of the variables examined above (See Table 1 for sample sizes and means by group and gender).
Table 1.
Means by Exposure Group and Gender for Birth Measures and IQ
| Measure | Cocaine Exposed Males (SD; n) | Non-Exposed Males (SD; n) | Cocaine Exposed Females (SD; n) | Non-Exposed Females (SD; n) |
|---|---|---|---|---|
| Birth Weight (lbs)a | 5.71 (1.16; 81) | 6.56 (1.33; 50) | 5.36 (1.23; 79) | 6.38 (1.09; 66) |
| Head Circumference (cm)a | 32.75 (2.17; 80) | 34.09 (2.39; 49) | 32.43 (2.36; 78) | 33.48 (1.84; 63) |
| APGAR 1 | 7.96 (1.78; 81) | 8.06 (1.59; 48) | 8.07 (1.44; 75) | 8.46 (1.08; 66) |
| APGAR 2a | 8.68 (0.96; 81) | 9.00 (0.29; 48) | 8.78 (0.64; 76) | 8.91 (0.45; 66) |
| Toddler IQa | 87.46 (10.54; 57) | 91.07 (11.42; 46) | 88.04 (10.56; 51) | 95.04 (14.18; 55) |
= main effect for exposure group
All effects noted are p < .05
The 165 mothers for the present study sample who were identified as cocaine-users had used cocaine at least since the beginning of their pregnancy (and the majority had used for months and years before their pregnancy) and the cocaine-using women did not stop their use before delivery. None had used cocaine only before but not during their pregnancy. No mother in the sample had used opiates based on their report or on toxicology findings. The 165 cocaine-using mothers in this sample reported 5.34 mean years of lifetime cocaine use (SD = 3.29), and a mean of 5.58 days of cocaine use during pregnancy in the 30 days prior to the interview (SD = 6.96).
There were significant differences between the CE and NCE groups in the reported amount of other drugs used during pregnancy. The CE group more often reported using alcohol, tobacco, or marijuana during their pregnancy in addition to their cocaine use. Seventy-four percent (74.54%) of cocaine-using women compared to 35.29% of non-cocaine-using women reported alcohol use during their pregnancy (χ2 [2] = 43.76, p < .01). Similarly, 79.39% and 52.72% of cocaine-using women compared to 18.48% and 11.76% of non-cocaine-users reported tobacco (χ2 [2] = 103.21, p < .01) or marijuana (χ2 [2] = 50.62, p < .01) use, respectively, during their pregnancy.
Given the differences between CE and NCE mothers in terms of their use of other substances, associations between use/no use of tobacco, alcohol, and marijuana and birth, IQ, and Stroop interference (i.e. time and errors) variables were examined. In many cases, within the NCE group there was a high degree of overlap among substances used. That is, many mothers, who, for example, used tobacco, also used alcohol (72.72%) and/or marijuana (31.81%). Similarly, significant inter-correlations between types of substances used were also observed. Alcohol use during pregnancy was significantly related to tobacco use (r = .37, p < .01) and to marijuana use (r = .39, p < .01) during pregnancy. Likewise, use of tobacco during pregnancy was significantly related to use of marijuana during pregnancy (r = .30, p < .01). Among the birth variables and IQ, head circumference was associated with marijuana use prenatally (rpb = −.21, p < .05). All other associations between use of each substance and birth variables and IQ were not significant. Associations between use/no use of each substance and time to complete the Stroop task and errors during completion of the Stroop task are presented in Table 2. Although findings largely support no deleterious effects of substances on Stroop performance, unexpectedly, those exposed prenatally to alcohol made fewer errors during the Stroop task at 90 months of age.
Table 2.
Correlations1,2 between Use/No Use of Alcohol, Tobacco, and Marijuana and Stroop Variables for Cocaine Exposed3 and Non-Cocaine Exposed Participants
| Variable | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1. Prenatal Alcohol Exp. | --- | --- | --- | −.13 | −.09 | −.08 | −.09 | −.14 | −.03 | .08 | .02 | −.09 | .06 | −.22 |
| 2. Prenatal Tobacco Exp. | --- | --- | --- | −.06 | −.02 | .05 | −.11 | −.13 | −.14 | .06 | −.06 | −.13 | −.04 | −.14 |
| 3. Prenatal Marijuana Exp. | --- | --- | --- | .09 | .04 | .04 | .07 | −.08 | −.01 | −.11 | −.05 | −.01 | −.09 | −.06 |
| 4. Stroop Time 90 Months | −.13 | .04 | .03 | --- | .27* | .29* | .14 | .12 | .03 | −.08 | −.18* | −.13 | −.19* | .05 |
| 5. Stroop Time 114 Months | −.14 | .08 | −.16 | .47* | --- | .62* | .02 | .39* | .24* | .06 | .02 | .01 | .04 | .07 |
| 6. Stroop Time 138 Months | −.15 | −.05 | −.04 | .52* | .73* | --- | .12 | .27* | .26* | .10 | .07 | −.07 | .06 | .16 |
| 7. Stroop Errors 90 Months | −.27* | −.02 | .02 | .40* | .17 | .31* | --- | .24* | .15 | −.04 | .12 | .06 | .12 | .05 |
| 8. Stroop Errors 114 Months | −.03 | −.01 | −.15 | .08 | .36* | .35* | .14 | --- | .40* | .01 | .09 | −.02 | .09 | .20 |
| 9. Stroop Errors 138 Months | .07 | −.08 | .07 | .21 | .30* | .52* | .21 | .45* | --- | −.05 | .14 | −.01 | .07 | .04 |
| 10. Maternal Less than HS Ed. | −.10 | −.02 | .01 | −.08 | .14 | .02 | .05 | .12 | .09 | --- | .13 | .10 | .78* | −.01 |
| 11. Birth Weight Less than 2500 Gs | −.04 | .12 | .12 | .06 | −.05 | .08 | .10 | −.10 | −.09 | −.11 | --- | −.02 | .69* | −.05 |
| 12. Second APGAR Less than 7 | .13 | −.04 | .25* | −.04 | na4 | −.12 | .01 | na | −.16 | −.05 | .26* | --- | .22* | −.18 |
| 13. Risk | −.11 | .08 | .10 | −.06 | .10 | .08 | .12 | .06 | −.01 | .75* | .55* | .20* | --- | −.12 |
| 14. CBCL Aggression 168 Months | −.09 | −.05 | .15 | −.13 | .04 | .27 | .15 | −.02 | .29 | .02 | .09 | na | .08 | --- |
Associations between variables 1, 2, 3, 10, 11, and 12 and between these variables and other variables in the table are Point-Biserial correlations. When appropriate, variables in this table reflect log transformations (See Table 3 and 4) to account for substantial distributional non-normality (e.g. skew).
Samples sizes for pairs of variables are different throughout due to missing data across time.
Associations between variables within the CE group are presented to the left and above the diagonal; associations between variables within the NCE group are presented to the right and below the diagonal.
Due to the low occurrence of APGAR scores below 7 in the NCE sample, associations could not be computed for selected pairs of variables when no variation in APGAR scores was present.
p < .05
Within the CE group, all mothers who used cocaine prenatally also reported using alcohol, tobacco, or marijuana or a combination of these three substances (75.8% used two or more of these substances in addition to cocaine). As was observed within the NCE group, significant inter-correlations between types of substances used were also observed within the CE group. Alcohol use during pregnancy was significant related to tobacco use (r = .43, p < .01) and to marijuana use (r = .37, p < .01) during pregnancy and use of tobacco was significantly related to use of marijuana during pregnancy (r = .24, p < .01). All associations between use of marijuana, tobacco, or alcohol and birth variables and IQ were not significant. Likewise, no associations between use/no use of alcohol, tobacco, or marijuana and Stroop time or errors were statistically significant (See Table 2).
2.2. Measures
2.2.1. Cumulative Risk
To account for postnatal medical and demographic risk, a risk score was calculated for all participants and incorporated as a predictor into the LGM models present below. Each participant received up to three points, with one point for 1) maternal education less than a high school degree or equivalent, 2) birth weight less than 2,500 grams (i.e. was considered to have a low birth weight), or 3) for a second APGAR score of less than 7. Higher scores reflected higher risk.
2.2.2. Stroop
Based on a standard Stroop paradigm (61), two different sets of stimuli were presented. Set 1 consisted of three words representing color names (red, green, blue) printed in lower-case letters in a congruent color (e.g., “red” in red type). Set 2 included the same three color names printed in lower-case letters in incongruent color (e.g., the word ‘red’ printed in blue). All stimuli were presented on a white background. When presented with the second set of stimuli, subjects were asked to name the color of the type instead of naming or reading the word. For the purposes of the present investigation, only completion time and errors for the incongruent condition is summarized. We focused on the incongruent condition because as opposed to congruent task, the incongruent task is widely considered to reflect prepotent response inhibition (i.e. inhibitory control) and has also been extensively used as a measure of inhibitory control in the clinical literature (e.g., 62, 63, 64, 65).
2.2.3. KABC
The Kaufman Assessment Battery for Children (K-ABC; 66) is a norm-referenced, individually administered test of cognitive ability based on research and theory in cognition and neuropsychology regarding sequential and simultaneous information processing. The K-ABC is for use with children 2.5 through 12.5 years old.
2.2.4. Child Behavior Checklist
To evaluate behavioral outcomes that could be attributed to prenatal cocaine exposure, difficulties with inhibitory control as measured using the Stroop task, or both, caregivers completed the Child Behavior Checklist (CBCL; 67). For the purposes of this study, the aggression scale, which assesses aggressive, acting out behaviors, was used.
2.3 Procedure
All participants were followed from birth at predetermined intervals. IQ was assessed when toddlers reached 48 months of age and the Stroop task was administered at 90, 114, and 138 months of age. Finally, primary caregivers completed the CBCL when children reached 168 months of age. Each visit occurred in a standardized, laboratory environment with minimal distractions. All participants were accompanied by their parents or an adult caregiver. Families were compensated for the visit and children were provided age appropriate toys.
2.4 Analytic Strategy
Prior to all structural equation modeling (SEM) analyses, variables were examined for normality by group. Utilizing the recommendations outlined by Tabachnick and Fidell (68), a z-test (Skew/Std. Error of Skew) was conducted to determine if the degree of skew for each variable was significantly different from zero. All variables were transformed utilizing a log transformation if the results of the z-test were greater than or equal to 2 or less than or equal to −2. One of the assumptions underlying LGM is that measurements contained in the model are all on the same scale. Thus, if stroop information (i.e. time and/or errors) at 90 months was severely skewed, but stroop information at 114 months was not skewed, no transformations were performed. Similarly, if stroop information at 90 months was negatively skewed, but stroop information at 114 months was positively skewed, no transformation occurred because of differences in the transformation procedure for positively and negatively skewed data. Descriptive statistics for non-transformed and, if necessary, transformed data are displayed in Table 3 for NCE participants and in Table 4 for CE participants.
Table 3.
Descriptive Statistics for Non-Cocaine Exposed Participants for all Variables used in Multiple Group LGM Analyses
| Variable | Mean (SD) | Skew | S.E. of Skew | z | T. Mean (SD) | T. Skew | T. S.E. of Skew | z |
|---|---|---|---|---|---|---|---|---|
| Risk | 0.33 (0.51) | 1.12 | 0.23 | 4.87** | 0.51 (0.06) | 0.99 | 0.23 | 4.30* |
| 90 Month Stroop Errors | 12.43 (14.52) | 3.74 | 0.25 | 14.96** | 1.08 (0.28) | 0.72 | 0.25 | 2.88* |
| 90 Month Stroop Time | 256.15 (52.31) | 0.87 | 0.25 | 3.48** | 2.40 (0.09) | 0.34 | 0.25 | 1.36 |
| 114 Month Stroop Errors | 9.51 (6.89) | 1.63 | 0.26 | 6.27** | 1.04 (0.23) | −0.12 | 0.26 | −0.46 |
| 114 Month Stroop Time | 217.93 (42.80) | 1.17 | 0.26 | 4.50** | 2.33 (0.08) | 0.59 | 0.26 | 2.27* |
| 138 Month Stroop Errors | 8.03 (6.15) | 2.43 | 0.28 | 8.69** | 0.99 (0.22) | −0.06 | 0.28 | −0.21 |
| 138 Month Stroop Time | 187.74 (37.24) | 1.01 | 0.28 | 3.61** | 2.27 (0.08) | 0.45 | 0.28 | 1.61 |
| 168 Month Aggression | 6.46 (6.15) | 1.46 | 0.38 | 3.82** | 0.90 (0.26) | 0.19 | 0.38 | 0.50 |
p < .05;
p < .01
T = Transformed
Table 4.
Descriptive Statistics for Cocaine Exposed Participants for all Variables used in Multiple Group LGM Analyses
| Variable | Mean (SD) | Skew | S.E. of Skew | z | T. Mean (SD) | T. Skew | T. S.E. of Skew | z |
|---|---|---|---|---|---|---|---|---|
| Risk | 0.77 (.74) | 0.48 | 0.20 | 2.45* | 0.57 (0.08) | 0.23 | 0.20 | 1.15 |
| 90 Month Stroop Errors | 15.84 (17.28) | 2.90 | 0.23 | 12.61** | 1.16 (0.30) | 0.39 | 0.23 | 1.70 |
| 90 Month Stroop Time | 255.79 (52.30) | 0.55 | 0.23 | 2.39* | 2.40 (0.09) | 0.16 | 0.23 | 0.70 |
| 114 Month Stroop Errors | 12.16 (13.50) | 3.80 | 0.21 | 18.10** | 1.08 (0.28) | 0.41 | 0.21 | 1.95 |
| 114 Month Stroop Time | 227.82 (44.68) | 1.06 | 0.21 | 5.05** | 2.35 (0.08) | 0.51 | 0.21 | 2.43* |
| 138 Month Stroop Errors | 9.02 (7.86) | 3.18 | 0.21 | 15.14** | 1.01 (0.23) | 0.26 | 0.21 | 1.24 |
| 138 Month Stroop Time | 196.98 (34.20) | 0.82 | 0.21 | 3.91** | 2.29 (0.07) | 0.31 | 0.21 | 1.48 |
| 168 Month Aggression | 6.93 (6.31) | 1.37 | 0.29 | 4.72** | 0.91 (0.28) | −0.07 | 0.29 | 0.24 |
p < .05;
p < .01
T = Transformed
Latent growth modeling (LGM) was conducted with EQS 6.1 (69). Because LGM is carried out within the broader framework of SEM, LGM retains all the advantages of the SEM framework (e.g., estimation of measurement error and the ability to incorporate observed [i.e. manifest] and unobserved [i.e. latent] variables). However, several conditions must be met when performing LGM. For example, measurements for each individual must be taken a minimum of three times. It also is also ideal for the spacing of measurements to be the same for all individuals (see 70, 71). Given these two requirements, in the present investigation, three measurements were obtained and all participants were assessed at approximately the same ages across time.
When using an LGM approach to model change across time, an initial growth model is fit to repeated measures for each participant, resulting in intercept and slope factors with corresponding factor residuals (70, 72). Intercept and slope residuals (i.e. error terms) in the LGM framework represent potential deviations from the average intercepts and slopes (i.e. individual differences). In the present study, consistent with the hypotheses outlined above, two multiple-group LGM’s were conducted, with one examining developmental trajectories of time to complete the stroop interference task and the other examining number of errors on the stroop interference task. To examine group differences in the absence of predictors (i.e. gender and risk) and outcome (i.e. aggression), an initial model for time and errors was run. Pathways representing the mean intercept (i.e. mean initial levels) for time and errors and mean slope (i.e. mean change over time) for time and errors were constrained to be equal across the NCE and CE groups2. Subsequent models with gender and cumulative risk entered as predictors of interindividual differences in the slope and intercept of Stroop time and errors and the aggressive behavior outcomes were run to evaluate remaining hypotheses (i.e. the effects of gender and risk on inhibitory control development and the effects of inhibitory control development on aggressive behavior). Importantly, the effects of inhibitory control on subsequent aggression accounted for gender and risk. Predictor/outcome models were also initially run with all pathways constrained to be equal across groups.
For all multiple group LGM models, Lagrange Multiplier tests (LM) were used to determine if pathways constrained to be equal should be unconstrained (i.e. the constraints should be relaxed) in subsequent analyses. Additionally, if relaxed constraints and a subsequent analysis were warranted based on LM test results, the improvement in model fit was examined using a chi-square difference test. This approach allows one to determine whether participants that are NCE or CE reflect membership in the same underlying population in terms of inhibitory control development across time and whether the potential influence of gender and cumulative risk on inhibitory control and associations between gender, cumulative risk, and inhibitory control variables with the aggressive behavior outcome were similar or different in each group. All models were evaluated using standard fit indices widely used in SEM applications (e.g., CFI, GFI, AIC, and RMSEA). The interested reader can find additional information regarding the LM test, change in chi-square, and fit indices as well as the use of EQS in Byrne (70) and LGM more specifically in Duncan, Duncan, and Strycker (71).
2.5 Missing Data
Rates of missing data for the CE and NCE groups for each variable utilized in the LGM analyses are presented in Table 5. For each group (CE and NCE) a series of independent t-tests were conducted to examine potential systematic patterns to missing data. Beginning at the 48 month IQ assessment, maternal age and infant birth variables (i.e. weight, head circumference, and first and second APGAR scores) were evaluated. At the 90 month evaluation, maternal age and birth variables, as well as 48 month IQ was examined. This process was repeated at each of the evaluation time points with data collected at the previous time point entered into subsequent missing data analyses. For the CE group of participants, one of the 62 comparisons was significant; maternal caregivers of children who completed the 48 month IQ evaluation were significantly older when the index child was born (M = 29.99, SD = 4.58) than mothers whose children did not complete the 48 month IQ evaluation (M = 28.30, SD = 4.39), t (163) = 2.28, p < .05. Two comparisons out of 62 were significant in the NCE group. Children who completed the 138 month stroop evaluation performed slightly better on the 90 month stroop errors (M = 1.03, SD = .23) than children who did not complete the 138 month stroop evaluation (M = 1.15, SD = .32), t (91) = −2.15, p < .05. At the 168 month evaluation of aggression, those participating in the visit made fewer errors during the 90 month Stroop evaluation (M = 0.99, SD = .22) than those who did not participate in the 168 month visit (M = 1.10, SD = .24), t (95) = −2.27, p < .05. In sum, across both groups, three out of 124 missing data comparisons were statistically significant. These findings do not suggest a systematic pattern to the missing data in this data set.
Table 5.
Rates of Missing Data by Group
| Variable | Cocaine Exposed | Non-Cocaine Exposed |
|---|---|---|
| Cumulative Risk | 6.1% | 4.2% |
| KABC | 34.5% | 15.1% |
| 90 Month Stroop | 35.2% | 21.8% |
| 114 Month Stroop | 18.8% | 26.1% |
| 138 Month Stroop | 17.6% | 37.8% |
| 168 Month Aggression | 55.2% | 67.2% |
Based on these findings, missing data was handled with Maximum Likelihood Estimation (MLE). MLE incorporates all available data from each participant at each time point by utilizing an expectation maximization algorithm to obtain estimates of missing data (69, 73). It has been recommended that the amount of missing data be no greater than 90% for any single variable when utilizing the MLE method for missing data (74). In the current study, the largest amount of missing data for any one variable in the CE group is 55.2% and for the NCE group is 67.2%. These rates of missing data are well within the recommended limits for use of the MLE missing data procedure.
3. Results
3.1 Multiple Group LGM’s
3.1.1. Stroop errors initial model
The initial multiple group model examining change in stroop interference task errors fit the data well, χ2 (10) = 12.05, p > .05, CFI = 1.00, AGFI = .98, AIC = −7.95, RMSEA = .00 (90% Confidence: .00 to .08). LM test results indicated that removing the constraint associated with the mean intercept would result in a significant decrease in the overall model Chi-square. Consistent with the LM test results, the multiple group LGM with the intercept constraint released resulted in a significant improvement in overall model fit, χ2 (9) = 7.22, p > .05, CFI = 1.00, AGFI = .98, AIC = −8.87, RMSEA = .00 (90% Confidence: .00 to .08). Further examination of LM test results of the model with the intercept constraint released indicated that releasing the constraint associated with slope of stroop errors would result in an additional improvement in model fit. A subsequent analysis of the multiple group model with both the mean slope and mean intercept constraints released resulted in an improved fit to the data, χ2 (8) = 5.24, p > .05, CFI = 1.00, AGFI = .98, AIC = −8.94, RMSEA = .00 (90% Confidence: .00 to .07). LM test results on the final model did not indicate releasing additional constraints and the final model was a significant improvement in fit relative to the model with all parameters constrained to be equal, Δ χ2 (2) = 6.81, p < .05. Findings (See Table 6) indicated that cocaine exposed participants initially committed significantly more errors on the stroop task than non-cocaine exposed participants. While both groups of participants improved their performance on the stroop task over time, as indexed by the decreasing slope trajectories of stroop errors, findings indicate that cocaine exposed participants improved faster over time than non-exposed participants in terms of errors committed during performance of the stroop task.
Table 6.
Parameter Estimates for Stroop Task Errors in the Initial Multiple Group LGM
| Parameter | Coefficient (S.E.)1 | z value |
|---|---|---|
| Mean Intercept | ||
| Non-Cocaine Exposed | 1.07 (.02) | 46.58** |
| Cocaine Exposed | 1.15 (.02) | 58.85** |
| Mean Slope | ||
| Non-Cocaine Exposed | −0.04 (.01) | −2.52** |
| Cocaine Exposed | −0.07 (.01) | −5.57** |
| Intercept Variance | 0.02 (.006) | 2.66** |
| Slope Variance | 0.007 (.003) | 2.58** |
| Covariance | −0.003 (.003) | −0.77 |
| Error Variances | ||
| E1 | 0.06 (.009) | 6.36** |
| E2 | 0.04 (.005) | 8.79** |
| E3 | 0.01 (.006) | 2.38** |
Coefficients presented are not standardized
p < .05;
p < .01
3.1.2. Stroop errors with predictors of intercept and slope and aggression outcome
The multiple group model with gender and risk entered as predictors of intercept and slope and the 168 month aggressive behavior outcome with all parameters constrained was a inadequate fit the data, χ2 (32) = 86.39, p < .05, CFI = .95, GFI = .92, AIC = 22.39, RMSEA = .11 (90% Confidence: .08 to .14). However, in this initial run with all between groups constraints imposed on the model, LM test results indicated that the following nine constraints should be released: mean intercept, mean slope, mean risk, gender to intercept, risk to intercept, slope to aggression, intercept residual, gender residual, and risk residual. Consistent with the LM test results, the multiple group LGM with the above constraints released resulted in a significant improvement in overall model fit, Δ χ2 (9) = 65.20, p < .05; χ2 (23) = 21.19, p > .05, CFI = 1.00, GFI = .95, AIC = −24.80, RMSEA = .00 (90% Confidence: .00 to .04). LM test results on this final model did not indicate the need to release additional model constraints.
Findings indicated that initial differences between groups in errors during stroop task performance are qualified by the effects of gender and risk. For both the NCE group and the CE group, higher risk status was associated with more errors during the stroop task at 7.5 years of age (z = 2.12, p < .05, both groups). Furthermore, while there were no gender differences between initial number of errors during performance on the stroop task for CE participants, within the NCE group, girls made significantly fewer errors at 7.5 years of age than boys (z = −3.80, p < .05). While gender accounted for interindividual differences associated with the intercept in the NCE group, interindividual differences remained (i.e. were not accounted for by gender; z = 3.46, p < .05) associated with the intercept of the CE participants. With regard to the slope of Stroop errors, both groups of participants improved their performance on the stroop task over time, as indexed by decreasing slope trajectories. Gender, but not risk, was a significant predictor of interindividual differences in the slope of stroop errors with girls in both groups improving faster over development than boys (z = −2.29, p < .05, both groups).
With regard to aggressive behavior at 168 months of age, gender and risk were not significant predictors. However, after accounting for gender and risk, higher intercept in both groups was a significant predictor of increased aggressive behavior (z = 2.27, p < .05). That is, children who made more errors on the Stroop at 7.5 years of age were rated by their caregivers as displaying more aggressive behavior at 14 years of age, regardless or prenatal cocaine exposure status. Slope of errors was not a significant predictor of aggressive behavior in either group (See Figure 1 and 2 for trajectories by CE/NCE group and by gender and by risk; See Figure 3 for Standardized Coefficients for significant findings).
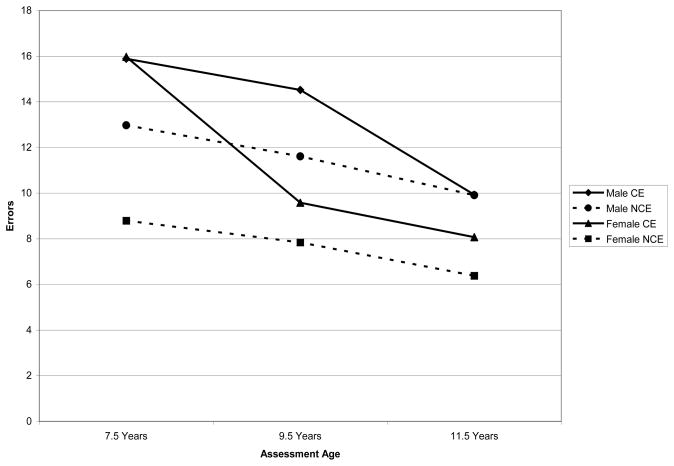
Figure 1.
Trajectory of Errors During the Stroop Color-Word Task Performance by Gender and Prenatal Cocaine Exposure Status
Figure 2.
Trajectory of Errors During the Stroop Color-Word Task by Risk and Prenatal Cocaine Exposure Status
Figure 3.
Final Latent Growth Model of Errors During the Stroop Color-Word Task with Gender, Cumulative Risk, and Aggressive Behavior
3.1.2. Stroop time initial model
Similar to the findings regarding Stroop errors, the initial multiple group LGM examining time to complete the stroop task was a good fit to the data, χ2 (10) = 11.36, p > .05, CFI = 1.00, AGFI = .99, AIC = −8.64, RMSEA = .00 (90% Confidence: .00 to .05). LM test results indicated that removing the constraint associated with the slope would result in a significant improvement in the overall model Chi-square, χ2 (1) = 13.62, p < .05. The multiple group LGM with the slope constraint released resulted in a significant improvement in overall model fit, χ2 (9) = 3.60, p > .05, CFI = 1.00, AGFI = .99, AIC = −14.40, RMSEA = .00 (90% Confidence: .00 to .03). LM test results did not indicate further constraints should be released. The final model resulted in a significant change in chi-square relative to the initial model, Δ χ2 (1) = 7.76. These findings indicated that although the intercept’s (i.e. initial starting point) for the groups are similar, how the groups change over development (i.e. slope) in terms of time to complete the stroop task is different. That is to say, although both groups are able to complete the task faster, as indicated by decreasing slope trajectories, however, non-cocaine exposed participants had steeper age-related improvements (i.e. are faster in task performance over development) than cocaine exposed participants (See Table 7).
Table 7.
Parameter Estimates for Stroop Task Completion Time in the Initial Multiple Group LGM
| Parameter | Coefficient (S.E.)1 | z value |
|---|---|---|
| Mean Intercept | 2.40 (.005) | 496.34** |
| Mean Slope | ||
| Non-Cocaine Exposed | −0.07 (.004) | −18.95** |
| Cocaine Exposed | −0.05 (.003) | −17.19** |
| Intercept Variance | 0.004 (.0007) | 5.64** |
| Slope Variance | 0.001 (.0002) | 4.24** |
| Covariance | −0.001 (.0005) | −2.04* |
| Error Variances | ||
| E1 | 0.004 (.001) | 5.38** |
| E2 | 0.003 (.0004) | 8.22** |
| E3 | 0.001 (.001) | 1.04 |
Coefficients presented are not standardized
p < .05;
p < .01
3.1.3. Stroop time with predictors of intercept and slope and aggression outcome
The initial multiple group LGM examining time to complete the stroop task with predictors and the aggressive behavior outcome was a poor fit to the data, χ2 (32) = 884.28, p < .05, CFI = .10, GFI = .87, AIC = 820.28, RMSEA = .43 (90% Confidence: .40 to .45). LM test results indicated that releasing twelve of the twenty-two constraints (mean slope, mean risk, gender to slope, mean aggression, intercept to aggression, slope to aggression, gender to aggression, risk to aggression, gender residual, mean gender, risk residual, and aggression residual3) was appropriate and would result in a substantial improvement in model fit. The multiple group LGM with the restraints noted above released resulted in a significant improvement in overall model fit, Δ χ2 (12) = 808.31, p < .05; χ2 (20) = 11.96, p > .05, CFI = 1.00, GFI = .98, AIC = −28.03, RMSEA = .00 (90% Confidence: .00 to .03). LM test results did not indicate further constraints should be released.
No differences on intercept of time to complete the Stroop task were observed. Furthermore, gender and risk did not account for variance in the intercept of time to complete the stroop (i.e. the time to complete the stroop at 7.5 years). However, while participants in the CE group generally improved faster over development in their ability to complete the Stroop task in a timely manner, this effect is qualified by gender differences in the NCE group. Girls in the NCE group demonstrated significantly greater improvements in the amount of time to complete the Stroop task over development relative to boys in the NCE group (z = −4.05, p < .05). Similarly, there was a trend (z = −1.42, p < .10) such that girls in the CE group improved faster over development compared to CE boys. In both groups, higher risk was associated with higher slopes (z = 3.10, p < .05, both groups). That is, those in both the CE and NCE groups who received higher risk scores improved at a slower rate over development than those at lower risk.
The 14 year CBCL aggression outcome, after controlling for gender and risk, was predicted by the slope of time to complete the Stroop task, with higher slopes predicting increased aggression. However, this effect is qualified, somewhat, by differences between the CE and NCE groups. The effect of slope on subsequent aggressive behavior was stronger in the NCE group (z = 2.51, p < .05) whereas in the CE group, the effect of slope on subsequent aggression was marginal (z = 1.47, p < .10; See Figure 4 and 5 for trajectories by CE/NCE group and by gender and by risk; See Figure 6 for Standardized Coefficients for significant findings).
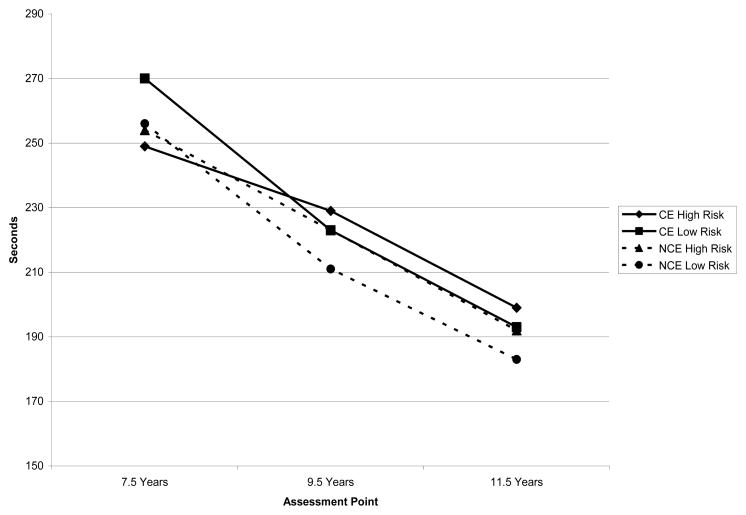
Figure 4.
Trajectory of Time to Complete the Stroop Color-Word Task by Gender and Prenatal Cocaine Exposure Status
Figure 5.
Trajectory of Time to Complete the Stroop Color-Word Task by Risk and Prenatal Cocaine Exposure Status
Figure 6.
Final Latent Growth Model of Time to Complete the Stroop Color-Word Task with Gender, Cumulative Risk, and Aggressive Behavior
3.1.3. Other substances
Although preliminary analyses suggested that the effects, if any, of prenatal exposure to non-cocaine substances (i.e. alcohol, marijuana, and tobacco) on performance during the Stroop task (Time and Error variables) would be negligible, a total of six additional LGM models (i.e. one model for Stroop Time and Errors for each of the three substances) were fit to the data to examine potential exposure effects. Prenatal exposure to non-cocaine substances was coded dichotomously (exposed/not exposed prenatally) for each substance for all participants. Each of the six multiple-group LGM models was a poor fit to the data, which provided additional evidence that prenatal exposure to non-cocaine substances in this sample did not appear to influence performance during the Stroop task that was used to measure inhibitory control.
4. Discussion
The findings in the current study largely conform to hypotheses and make several important contributions. This study adds to the literature by examining how inhibitory control development unfolds across development within two high risk groups of children. Furthermore, this study demonstrates that the influence of cocaine exposure on development of inhibitory control between 7.5 and 11.5 years of age is moderated by gender and that development of inhibitory control in both groups of high risk children is also influenced by demographic and early postnatal medical risk. The role of developing inhibitory control on aggressive behavior outcomes in both groups was also evaluated and hypotheses were examined using a methodology (LGM) that is scarcely employed in these literatures (See 52 for a notable exception). In terms of broad developmental expectations, although both samples were high risk, with one exposed to cocaine, as well as other substances, and the other comprised of a mixed group of non-exposed and non-cocaine, but other substance exposed participants, improvements in inhibitory control were evident from 7.5 to 11.5 years of age in both groups. This finding conforms to expectations based on findings in cross-sectional studies (e.g., 18, 19, 23) that have identified improvements in performance during tasks measuring inhibitory control from childhood through adulthood. This finding also suggests a degree of resilience within the inhibitory control system given that both groups in the current study come from backgrounds that are characterized by varying degrees of risk.
Evidence was obtained which largely conformed to expectations of group differences in developmental trajectories of inhibitory control. Initially, NCE participants obtained fewer errors on a standard Stroop Color/Word task compared to participants who were exposed prenatally to cocaine. Although both groups experienced decreases in errors over time, the CE group improved faster (i.e. had a steeper declining slope) than the NCE group. These findings were qualified by gender effects. In the NCE group, boys made significantly more errors initially than girls whereas there were no differences in errors between boys and girls in the CE group. Girls in the CE and NCE groups improved faster across time than boys; however, it was evident that while boys in the CE group were performing similarly to their counterparts in the NCE group by 11.5 years of age, CE girls had not caught up to their NCE counterparts in terms of number of errors committed during the Stroop task. The pattern of findings related to errors during the Stroop task is broadly consistent with existing literature (46,47,48) indicating greater effects in boys exposed prenatally to cocaine relative to girls prenatally exposed to cocaine and adds to existing sex differences literature in this population by demonstrating that sex effects may become evident, particularly within the CE exposed population, as development progresses. This finding, as it pertains to errors committed during the Stroop task, also suggests that CE exposed girls may continue to display greater difficulties relative to NCE girls across development whereas CE boys appear to perform similarly to their NCE counterparts by 11.5 years of age.
Risk status, defined as a combination of low birth weight, APGAR scores, and low maternal education attainment, emerged as an important contributor to the initial number of errors committed during the Stroop task in both groups at 7.5 years of age. High risk CE and NCE participants made more errors at 7.5 years than low risk CE and NCE participants, respectively. This finding is inconsistent with Bendersky et al.’s (45) finding that prenatal exposure to cocaine was unrelated to performance during an inhibitory control task only for participants coming from a high risk background. In the current study, cocaine exposure status was related to performance (i.e. number of errors) during an inhibitory control task at 7.5 years of age (i.e. intercept in the growth model) even for high risk CE participants. However, across development in both groups, the effects of risk on inhibitory control, as indexed by errors during the Stroop task, diminished. This suggests that risk may be more salient for the inhibitory control system in younger, school aged children, with those exposed to cocaine prenatally even more vulnerable during this time period than non-cocaine exposed, but nonetheless high risk children. It should be noted, however, that differences between findings reported in this study and findings reported by Bendersky et al. could also be a function of differences in the manner in which risk was determined. In the current study, postnatal medical risk variables (i.e. low birth weight and APGAR) were combined with one demographic risk characteristic (i.e. low maternal education) whereas Bendersky et al. focused exclusively on multiple sociodemographic risk characteristics. Future work should consider incorporating variables associated with early postnatal medical risk (e.g., birth weight) as well as multiple sociodemographic characteristics.
Both the CE and NCE groups improved across the study time frame in terms of how quickly the Stroop Color/Word task was completed, which is similar to findings related to errors during the Stroop task outlined above and consistent with continued frontal lobe development as children age. However, relative to the developmental pattern observed with respect to errors during the Stroop Color/Word task, findings indicated that the time to complete the task decreased faster in NCE participants than in CE participants between 7.5 and 11.5 years of age. That is, differences in time to complete the Stroop Color/Word task in CE exposed children may only become evident over time. When sex is considered, however, it appears that NCE boys and CE boys performed similarly across development. Nevertheless, girls in the NCE group improved over time faster than boys in the NCE group with a trend for a similar pattern of findings within the CE group. These findings are also consistent with gender differences identified by other investigators (e.g., 46,47,48), suggesting greater resilience among girls prenatally exposed to cocaine relative to prenatally CE boys, and girls in general that are at elevated risk of difficulties due to demographic risk. However, it is also noteworthy that CE girls appear to have greater inhibitory control difficulties than their same sex, NCE counterparts. In addition to important gender differences in the developmental trajectory of inhibitory control as indexed by the length of time to complete the Stroop task, risk was also an important predictor of change in time to complete the Stroop task between 7.5 and 11.5 years of age. In both groups, higher risk was associated with higher slopes. That is, while both groups (i.e. CE and NCE) improved over development, CE and NCE participants that were higher in risk took longer to complete the task as development progressed, which is indicative of greater inhibitory control difficulties.
Finally, models incorporated a parent report measure of aggressive behavior that was obtained when children reached 14 years of age. Consistent with expectations, after accounting for gender and risk, participants with greater inhibitory control difficulties as indicated by commission of more errors on the Stroop task at 7.5 years of age showed higher levels of aggressive behavior at 14 years of age. Likewise, higher slopes of time to complete the Stroop task in the NCE group, with a similar trend (p < .10) in the CE group, also predicted increased difficulties with aggressive behavior at 14 years of age. These findings are consistent with other studies that have noted associations between low inhibitory control and greater externalizing behavior difficulties (11,12,13,14,15,16). The lack of differences between CE and NCE participants in terms of predictors of aggressive behavior outcomes and in mean levels of aggression at 14 years of age is not entirely consistent with some prior work insomuch as prior work has linked more externalizing problems with prenatal cocaine exposure (15,16,40,41). However, this is not necessarily unexpected as difficulties with inhibitory control, regardless of etiology, would be anticipated to result in greater behavioral difficulties on the basis of prior work. Similarly, consistent with existing work, this study identifies risk as well as prenatal cocaine exposure, with the noted moderation by gender, as potential contributors to inhibitory control difficulties. This collective set of findings is consistent with studies that find that higher cumulative risk, prenatal cocaine exposure, and gender, places individuals, typically boys, at greater risk for externalizing problems, and suggests that inhibitory control may be a critical mechanism linking earlier risks including prenatal exposures to later behavioral problems.
When considered within the broader context of prior work in the area of performance on measures of inhibitory control, collectively the findings outlined above suggest that over time, the CE participants in this study made gains in the ability to inhibit responses (i.e. make fewer errors), especially for boys, but at the greater cost of decreased efficiency, particularly for girls. Gender differences notwithstanding, this finding is consistent with a previous study that examined ERP’s during the Stroop task in a smaller sample of CE vs. non-substance exposed children, which identified CE participants as having slower task processing as a result of the recruitment of more diverse regions as opposed to non-substance exposed participants that had greater specialization and efficiency of regions that in turn enhanced cognitive processing during the Stroop task (26). Future studies, using ERP or other physiological methods, may want to consider gender differences and/or changes over time/development in the study of underlying physiological differences between CE and NCE children that may reflect behavioral observations of task performance during specific measures of cognitive function, such as inhibitory control. This study is also in line with and adds to previous studies indicating that cocaine-exposure results in cognitive impairment in domains such as visual attention (e.g., 34), executive functions (e.g., 37), and poor regulation of emotion (e.g., 40). The findings in this study also are consistent with what might be expected based on theoretical formulations of the effects of prenatal cocaine exposure on the developing central nervous system and specifically executive functions (e.g., 38). Importantly, the current study adds to the nascent literature on the effects of prenatal cocaine exposure on cognitive functions in older children and early adolescents (e.g., 48, 36, 75) and extends this growing literature by examining inhibitory control, a more specific cognitive function, as has been previously suggested (40). Additionally, the current investigation is one of only a few studies that currently employs a longitudinal approach in the study of executive functions in an at risk population as opposed to the more common cross-sectional approach. By using a longitudinal, repeated measures design, this study moves beyond the examination of age differences and examines age/developmental changes over time (See 29 or 30 for discussion).
Clinical implications of the findings in this study also are present. For example, repeated assessment across development of specific cognitive functions in CE children may be necessary to identify specific cognitive difficulties, given that differentiation, especially for girls, of the time it took to complete the Stroop Color/Word task was the most evident at 11.5 years of age and was less so earlier in life for participants in the present study. Similarly, at earlier ages, findings in this study suggest that there may be greater inhibitory deficits (e.g., Stroop Errors, as well as potential deficits on other measures of prefrontal function) during performance of cognitive measures with improvements evident when these children are followed over time. However, it also is plausible that on speeded measures, performance may progressively decline relative to age expected norms. This means that on particularly complex, challenging timed measures, it will be important to be able to dissociate speed of task completion, which may in part reflect aspects of cognitive efficiency, from other functions believed to underlie task completion. Insomuch as difficulties with inhibitory control have been linked to behavioral problems in prior research (15,16) as well as in the current investigation, children prenatally exposed to cocaine and those who experience greater levels of early medical and demographic risk, regardless of prenatal cocaine exposure status, may remain at increased risk for behavioral difficulties (e.g., impulsivity and aggression) and problems in school as they approach early adolescence. This being said, it should be emphasized that just as with most other vulnerabilities, individual children that have been prenatally exposed to cocaine and those with varying levels of other risk factors, will likely vary widely in the degree and range of effects on their cognitive development, with such effects potentially moderated by gender, as was observed in the current study. Thus, while the findings in the current study suggest a general pattern of inhibitory control development, cognitive assessments and subsequent recommendations/interventions will need to be tailored to individual children based on their referral questions and needs.
While the findings in the current study have made several unique contributions to the literature on inhibitory control development in general, and such development in a high risk sample, specifically, the present study is not without limitations. For example, children prenatally exposed to cocaine also were exposed, collectively, to a number of other substances, such as alcohol, marijuana, and/or tobacco. This raises two possibilities: 1) that there is some synergistic effect of these substances on the developing nervous system and specifically on frontal systems associated with inhibitory control or 2) that cocaine as well as other substances independently and uniquely affect inhibitory control, with both of these serving as possible explanations for the effects observed in this study. Nevertheless, to the extent possible, we attempted to account for this by comparing the CE participants to a mixed group of participants, some of whom had not experienced any substance expose and others that had been exposed to non-cocaine substances. The process by which the non-cocaine comparison group was formed was supported by no differences between non-cocaine, but other drug exposed children, and non-substance exposed children on nearly all outcome measures at birth, IQ in the preschool period, and on all three of the Stroop assessments. Thus, this increases the likelihood that the effects obtained are indeed due to either prenatal cocaine exposure alone or to synergistic interaction of cocaine with other substances. Similarly, although all cocaine using mothers also used other substances, there was not evidence to suggest systematic differences between children of cocaine using mothers who used one, two, or three other substances based on analyses conducted within the CE group. Consistent with these findings, exploratory LGM’s failed to identify effects of other substances on inhibitory control development in this study. Nevertheless, while attempts were made to examine the possible influence of other substances on inhibitory control development, it remains possible that a dose-response effect of specific substances or combinations of substances on Stroop performance remained undetected as a function of the use of dichotomous measures that were used to assess the potential effect of other substances.
From a statistical and methodological standpoint, this study identified that in both groups of participants, residuals associated with the slope and intercept factors were significant. When using LGM, this is a strong indicator of the presence of interindividual differences (70, 76) in the initial starting point and in the trajectory of inhibitory control over time that may be accounted for from systematic sources (e.g., may be accounted for by aspects of the environment, parent-child relationship, and other aspects of the individual). Although gender and cumulative risk accounted for some variability (i.e. interindividual differences), with the intercept residual associated with errors during the Stroop task in the NCE group reduced to non-significant levels once gender and cumulative risk was considered, remaining variability in performance time in the CE and NCE groups and errors in the CE group suggests that even within the prenatally cocaine/polysubstance exposed population, there may be individuals that are more or less resilient to the effects of in utero substance exposure on cognitive development, such as inhibitory control. Future studies will need to take into account and model such possibilities within a developmentally sensitive framework.
Two other limitations should also be acknowledged. First, there were relatively high rates of missing data for the 14 year aggression outcome. Although analyses did not suggest systematic differences between those who completed the 14 year assessment and those who did not, it remains a possibility that effects went undetected. For example, high rates of missing data may have impeded the ability for statistical analyses to differentiate the effects of slopes and intercepts of Stroop time and errors, gender, and risk between the CE and NCE groups on aggressive behavior outcomes. Finally, a low risk comparison group was not included in the current investigation. This means that while a typical developmental pattern was observed in both groups, differences between groups in this study notwithstanding, inclusion of a low risk “normative” control group may have revealed differences, such as delays, in inhibitory control development that could be better attributed to characteristics of both the CE and NCE groups. Nevertheless, while this limitation should be addressed in future work, in some respects, findings obtained in the current investigation comparing a CE group of children to another high risk group are all the more notable, and indicative of the potentially long lasting effects of prenatal cocaine exposure against the backdrop of a high risk, but NCE comparison group of children. In spite of the noted limitations, which should be accounted for or examined in future research, the current study demonstrates the potential for using developmentally sensitive approaches that model age-related changes in aspects of cognition and how early influences, such as prenatal substance exposure, as well as the effects of risk and moderation by gender of prenatal substance exposure, can affect long term cognitive development, and adds to the growing literature examining specific cognitive effects associated with prenatal cocaine exposure through late school-age/early adolescent children.
Acknowledgments
This work is supported by NIDA grants RO1-DA-06025 (LCM), DA-017863 (LCM) and KO5 (LCM). The authors gratefully acknowledge the loyal participation of the families over many years as well as the research staff who have so respectfully worked with the families over time and to whom so many of the families turn for help and support.
The authors thank Vince Smeriglio for his ever present support, good counsel, and generous friendship. His attentiveness in his NIH program role made possible the careers of many young investigators, including one of us (LM) and through his support of our generation, he continues to impact the next generation of scholars as evidenced by the lead author of this manuscript (DB). Vince’s understanding of the complexity of research in the real world made him an effective and indefatigable champion of the difficult but necessary longitudinal studies of prenatal cocaine exposure and much of the present science in this area is built on the foundation of his support and enthusiasm. Vince, there are countless families with their now adolescent children and many young investigators who owe you a considerable debt of gratitude.
Footnotes
Tables with means, standard deviations, and test statistics are not presented for given the non-significant findings in this set of analyses.
Although in the current study slope and intercept means and, in subsequent models, the influence of gender and risk on interindividual differences within and between groups were of primary interest, we choose to constrain all parameters to be equal to examine other possible differences between populations, such as the covariance between intercept and slope residuals.
Due to different data (i.e. time to complete the Stroop task) being considered in the LGM model and used in the estimation of missing data, it is possible for constraints that did not need to be released in the prior LGM modeling errors during the Stroop task to contribute to poor model fit in this model.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Contributor Information
David J. Bridgett, Northern Illinois University
Linda C. Mayes, Yale University, School of Medicine
5. References
- 1.Kimberg DY, D’Esposito M, Farah MJ. Cognitive functions in the prefrontal cortex-working memory and executive control. Current Directions in Psychological Science. 1997;6(6):185–192. [Google Scholar]
- 2.Mesulam M. The human frontal lobes: Transcending the default mode through contingent encoding. In: Stuss DT, Knight RT, editors. Principles of frontal lobe function. New York, NY: Oxford University Press; 2002. pp. 8–30. [Google Scholar]
- 3.Wagner G, Koch K, Reichenbach JR, Sauer H, Schlosser RGM. The special involvement of the rostrolateral prefrontal cortex in planning abilities: An event-related fMRI study with the Tower of London paradigm. Neuropsychologia. 2006;44:2337–2347. doi: 10.1016/j.neuropsychologia.2006.05.014. [DOI] [PubMed] [Google Scholar]
- 4.Diamond A. Normal development of the prefrontal cortex from birth to young adulthood: Cognitive functions, anatomy, and biochemistry. In: Stuss DT, Knight RT, editors. Principles of frontal lobe function. New York, NY: Oxford University Press; 2002. pp. 127–180. [Google Scholar]
- 5.Kochanska G, Murray KT, Harlan ET. Effortful control in early childhood: Continuity and change, antecedents, and implications for social development. Developmental Psychology. 2000;36:220–232. [PubMed] [Google Scholar]
- 6.Rothbart MK, Posner MI. Temperament and the development of self-regulation. In: Hartlage L, Telzrow CF, editors. The neuropsychology of individual differences: A developmental perspective. New York, NY: Plenum; 1985. pp. 93–123. [Google Scholar]
- 7.Carlson SM, Wang TS. Inhibitory control and emotion regulation in preschool children. Cognitive Development. 2007;22:489–510. [Google Scholar]
- 8.Fox NA, Calkins SD. The development of self-control of emotion: Intrinsic and extrinsic influences. Motivation and Emotion. 2003;27(1):7–26. [Google Scholar]
- 9.Hadley SJ, Capon A, Beveridge M, Evans J, Dennis I. Working memory, inhibitory control and the development of children’s reasoning. Thinking & Reasoning. 2004;10(2):175–195. [Google Scholar]
- 10.Silverman IW, Ippolito MF. Goal-directedness and its relation to inhibitory control among toddlers. Infant Behavior & Development. 1997;20(2):271–273. [Google Scholar]
- 11.Barkley RA. Behavioral inhibition, sustained attention, and executive functions: Constructing a unifying theory of ADHD. Psychological Bulletin. 1997;121:65–94. doi: 10.1037/0033-2909.121.1.65. [DOI] [PubMed] [Google Scholar]
- 12.Barkley RA. Attention deficit hyperactivity disorder: A handbook for diagnosis and treatment. New York, NY: Guilford Press; 1998. [Google Scholar]
- 13.Eisenberg N, Fabes RA, Guthrie IK, Murphy BC, Maszk P, Holmgen R, et al. The relations of regulation and emotionality to problem behavior in elementary school children. Development and Psychopathology. 1996;8:141–162. [Google Scholar]
- 14.Huijbregts SCJ, Warren AJ, de Sonneville LMJ, Swaab-Barneveld H. Hot and cool forms of inhibitory control and externalizing behavior in children of mothers who smoked during pregnancy: An exploratory study. Journal of Abnormal Child Psychology. 2008;36(3):323–333. doi: 10.1007/s10802-007-9180-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zhou Q, Lengua LJ, Wang Y. The relations of temperament reactivity and effortful control to children’s adjustment problems in China and the United States. Developmental Psychology. 2009;45(3):724–739. doi: 10.1037/a0013776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Muris P, Meesters C, Blijlevens P. Self-reported reactive and regulative temperament in early adolescence: Relations to internalizing and externalizing problem behavior and ‘Big Three’ personality factors. Journal of Adolescence. 2007;30(6):1035–1049. doi: 10.1016/j.adolescence.2007.03.003. [DOI] [PubMed] [Google Scholar]
- 17.Diamond A. Development of the ability to use recall to guide action, as indicated by infant’s performance on AB. Child Development. 1985;56:868–883. [PubMed] [Google Scholar]
- 18.Leon-Carrion J, Garcia-Orza J, Perez-Santamaria FJ. Development of the inhibitory component of the executive functions in children and adolescents. International Journal of Neuroscience. 2004;114:1291–1311. doi: 10.1080/00207450490476066. [DOI] [PubMed] [Google Scholar]
- 19.Williams BR, Ponesse JS, Schachar RJ, Logan GD, Tannock R. Development of inhibitory control across the life span. Developmental Psychology. 1999;35(1):205–213. doi: 10.1037//0012-1649.35.1.205. [DOI] [PubMed] [Google Scholar]
- 20.Bunge SA, Dudukovic NM, Thomason ME, Vaidya CJ, Gabrieli JDE. Immature frontal lobe contributions to cognitive control in children: Evidence from fMRI. Neuron. 2002;33:301–311. doi: 10.1016/s0896-6273(01)00583-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Rubia K, Overmeyer S, Taylor E, Brammer M, Williams S, Simmons A, Andrew C, Bullmore E. Functional frontalisation with age: Mapping neurodevelopmental trajectories with fMRI. Neuroscience and Biobehavioral Reviews. 2000;24:13–19. doi: 10.1016/s0149-7634(99)00055-x. [DOI] [PubMed] [Google Scholar]
- 22.Rubia K, Smith AB, Taylor E, Brammer M. Linear age-correlated functional development of right inferior fronto-striato-cerebellar networks during response inhibition and anterior cingulate during error-related processes. Human Brain Mapping. 2007;28:1163–1177. doi: 10.1002/hbm.20347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Velanova K, Wheeler ME, Luna B. Maturational changes in anterior cingulate an frontoparietal recruitment support the development of error processing and inhibitory control. Cerebral Cortex. 2008;18:2505–2522. doi: 10.1093/cercor/bhn012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Jonkman LM, Lansbergen M, Stauder JEA. Developmental differences in behavioral and event-related responses associated with response preparation and inhibition in a go/nogo task. Psychophysiology. 2003;40:752–761. doi: 10.1111/1469-8986.00075. [DOI] [PubMed] [Google Scholar]
- 25.West R. Neural correlates of cognitive control and conflict detection in the Stroop and digit-location tasks. Neuropsychologia. 2003;41:1122–1135. doi: 10.1016/s0028-3932(02)00297-x. [DOI] [PubMed] [Google Scholar]
- 26.Mayes LC, Molfese DL, Key APF, Hunter NC. Event-related potentials in cocaine-exposed children during a Stroop task. Neurotoxicology and Teratology. 2005;27:797–813. doi: 10.1016/j.ntt.2005.05.011. [DOI] [PubMed] [Google Scholar]
- 27.Casey BJ, Giedd JN, Thomas KM. Structural and functional brain development and its relation to cognitive development. Biological Psychology. 2000;54:241–257. doi: 10.1016/s0301-0511(00)00058-2. [DOI] [PubMed] [Google Scholar]
- 28.Lenroot RK, Giedd JN. Brain development in children and adolescents: Insights from anatomical magnetic resonance imaging. Neuroscience and Biobehavioral Reviews. 2006;30:718–729. doi: 10.1016/j.neubiorev.2006.06.001. [DOI] [PubMed] [Google Scholar]
- 29.Miller SA. Developmental research methods. 2. Upper Saddle River, NJ: Prentice-Hall; 1998. [Google Scholar]
- 30.Robinson K, Schmidt T, Teti DM. Issues in the use of longitudinal and cross-sectional designs. In: Teti DM, editor. Handbook of research methods in developmental science. Oxford, UK: Blackwell Publishing; 2005. pp. 3–20. [Google Scholar]
- 31.Moilanen KL, Shaw DS, Dishion TJ, Gardner F, Wilson M. Predictors of longitudinal growth in inhibitory control in early childhood. Social Development. 2010;19(2):326–347. doi: 10.1111/j.1467-9507.2009.00536.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Richardson GA, Goldschmidt L, Willford J. The effects of prenatal cocaine use on infant development. Neurotoxicology and Teratology. 2008;30:96–106. doi: 10.1016/j.ntt.2007.12.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Mayes LC, Cicchetti D, Acharyya S, Zhang H. Developmental trajectories of cocaine-and-other-drug-exposed and non-cocaine-exposed children. Developmental and Behavioral Pediatrics. 2003;24(5):323–335. doi: 10.1097/00004703-200310000-00003. [DOI] [PubMed] [Google Scholar]
- 34.Heffelfinger A, Craft S, Shyken J. Visual attention in children with prenatal cocaine exposure. Journal of the International Neuropsychological Society. 1997;3:237–245. [PubMed] [Google Scholar]
- 35.Schroder M, Snyder P, Sielski I, Mayes LC. Impaired performance of children exposed in-utero to cocaine on a novel test of visuospatial working memory. Brain and Cognition. 2004;55(2):409–12. doi: 10.1016/j.bandc.2004.02.062. [DOI] [PubMed] [Google Scholar]
- 36.Mayes LC, Snyder PJ, Langlois E, Hunter N. Visuospatial working memory in school-aged children exposed in Utero to cocaine. Child Neuropsychology. 2007;13:205–218. doi: 10.1080/09297040600888753. [DOI] [PubMed] [Google Scholar]
- 37.Espy KA, Kaufman PM, Glisky ML. Neuropsychologic function in toddlers exposed to cocaine in utero: A preliminary study. Developmental Neuropsychology. 1999;15(3):447–460. [Google Scholar]
- 38.Mayes LC. A behavioral teratogenic model of the impact of prenatal cocaine exposure on arousal regulatory systems. Neurotoxicology and Teratology. 2002;24(3):385–395. doi: 10.1016/s0892-0362(02)00200-3. [DOI] [PubMed] [Google Scholar]
- 39.Dennis T, Bendersky M, Ramsay D, Lewis M. Reactivity and regulation in children prenatally exposed to cocaine. Developmental Psychology. 2006;42(4):688–697. doi: 10.1037/0012-1649.42.4.688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Richardson GA, Goldschmidt L, Willford J. Continued effects of prenatal cocaine use: Preschool development. Neurotoxicology and Teratology. 2009;31:325–333. doi: 10.1016/j.ntt.2009.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Bada H, Das A, Bauer C, Shankaran S, Lester B, LaGasse L, Hammond J, Wright L, Higgins R. Impact of prenatal cocaine exposure on child behavior problems through school age. Pediatrics. 2007;119:348–359. doi: 10.1542/peds.2006-1404. [DOI] [PubMed] [Google Scholar]
- 42.Markowski VP, Cox C, Weiss B. Prenatal cocaine exposure produces gender specific motor effects in aged rats. Neurotoxicology and Teratology. 1998;20:43–53. doi: 10.1016/s0892-0362(97)00076-7. [DOI] [PubMed] [Google Scholar]
- 43.Spear LP, Silveri MM, Casale M, Katovic NM, Campbell JO, Douglas LA. Cocaine development: A retrospective perspective. Neurotoxicology and Teratology. 2002;24:321–327. doi: 10.1016/s0892-0362(02)00194-0. [DOI] [PubMed] [Google Scholar]
- 44.Bennett DS, Bendersky M, Lewis M. Children’s cognitive ability from 4 to 9 years old as a function of prenatal cocaine exposure, environmental risk, and maternal verbal intelligence. Developmental Psychology. 2008;44(4):919–928. doi: 10.1037/0012-1649.44.4.919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Bendersky M, Gambini G, Lastella A, Bennett DS, Lewis M. Inhibitory motor control at five years as a function of prenatal cocaine exposure. Journal of Developmental and Behavioral Pediatrics. 2003;24(5):345–351. doi: 10.1097/00004703-200310000-00005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Delaney-Black V, Covington CY, Nordstrom B, Ager J, Janisse J, Hannigan JH, Chiodo L, Sokol RJ. Prenatal cocaine: Quantity of exposure and gender modification. Journal of Developmental and Behavioral Pediatrics. 2004;25(4):254–263. doi: 10.1097/00004703-200408000-00005. [DOI] [PubMed] [Google Scholar]
- 47.Bailey BN, Sood BG, Sokol RJ, Ager J, Janisse J, Hannigan JH, Covington C, Delaney-Black V. Gender and alcohol moderate prenatal cocaine effects on teacher-report of child behavior. Neurotoxicology and Teratology. 2005;27:181–189. doi: 10.1016/j.ntt.2004.10.004. [DOI] [PubMed] [Google Scholar]
- 48.Bennett D, Bendersky M, Lewis M. Preadolescent health risk behavior as a function of prenatal cocaine exposure and gender. Journal of Developmental Behavioral Pediatrics. 2007;28:467–472. doi: 10.1097/DBP.0b013e31811320d8. [DOI] [PubMed] [Google Scholar]
- 49.Mulder H, Pitchford NJ, Hagger MS, Neil M. Development of executive function and attention in preterm children: A systematic review. Developmental Neuropsychology. 2009;34(4):393–421. doi: 10.1080/87565640902964524. [DOI] [PubMed] [Google Scholar]
- 50.Li-Grining CP. Effortful control among low-income preschoolers in three cities: Stability, change, and individual differences. Developmental Psychology. 2007;43(1):208–221. doi: 10.1037/0012-1649.43.1.208. [DOI] [PubMed] [Google Scholar]
- 51.Hughes C, Ensor R. Independence and interplay between maternal and child risk factors for preschool problem behaviors? International Journal of Behavioral Development. 2009;33:312–322. [Google Scholar]
- 52.Bandstra ES, Vogel AC, Morrow CE, Xue L, Anthony JC. Severity of prenatal cocaine exposure and child language functioning through age seven years: A longitudinal latent growth curve analysis. Substance Use & Misuse. 2004;39(1):25–59. doi: 10.1081/JA-120027765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Best JR, Miller PH, Jones LL. Executive functions after age 5: Changes are correlates. Developmental Review. 2009;29:180–200. doi: 10.1016/j.dr.2009.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Bench CJ, Frith CD, Grasby PM, Friston KJ, Paulesu E, Frackowiak RSJ, Dolan RJ. Investigations of the functional anatomy of attention using the Stroop test. Neuropsychologia. 1993;31(9):907–922. doi: 10.1016/0028-3932(93)90147-r. [DOI] [PubMed] [Google Scholar]
- 55.Alvarex JA, Emory E. Executive function and the frontal lobes: A meta-analytic review. Neuropsychology Review. 2006;16(1):17–42. doi: 10.1007/s11065-006-9002-x. [DOI] [PubMed] [Google Scholar]
- 56.Homack S, Riccio CA. A meta-analysis of the sensitivity and specificity of the Stroop color and word test with children. Archives of Clinical Neuropsychology. 2004;19:725–743. doi: 10.1016/j.acn.2003.09.003. [DOI] [PubMed] [Google Scholar]
- 57.Warner TD, Behnke M, Hou W, Garvan CW, Wobie K, Eyler FD. Predicting caregiver-reported behavior problems in cocaine-exposed children at 3 years. Developmental and Behavioral Pediatrics. 2006;27(2):83–92. doi: 10.1097/00004703-200604000-00001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Accornero VH, Morrow CE, Bandstra ES, Johnson AL, Anthony JC. Behavioral outcome of preschoolers exposed prenatally to cocaine: Role of maternal behavioral health. Journal of Pediatric Psychology. 2002;27(3):259–269. doi: 10.1093/jpepsy/27.3.259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Poklis A. Evaluation of TDx cocaine metabolite. Journal of Analytical Toxicology. 1987;11:228–230. doi: 10.1093/jat/11.5.228. [DOI] [PubMed] [Google Scholar]
- 60.Walters R. An abused drug assay system. No. 2331 1987. Mar, [Google Scholar]
- 61.Stroop JR. Studies of interference in serial verbal reactions. Journal of Experimental Psychology: Human Perception and Performance. 1935;18:643–662. [Google Scholar]
- 62.Brocki KC, Nyberg L, Thorell LB, Bohlin G. Early and concurrent and longitudinal symptoms of ADHD and ODD: Relations to different types of inhibitory control and working memory. Journal of Child Psychology and Psychiatry. 2007;48(10):1033–1041. doi: 10.1111/j.1469-7610.2007.01811.x. [DOI] [PubMed] [Google Scholar]
- 63.Cothran DL, Larsen R. Comparison of inhibition in two timed reaction tasks: The color and emotion stroop tasks. The Journal of Psychology. 2008;142(4):373–385. doi: 10.3200/JRLP.142.4.373-385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Ciairano S, Visu-Petra L, Settanni M. Executive inhibitory control and cooperative behavior during early school years: A follow-up study. Journal of Abnormal Child Psychology. 2007;35:335–345. doi: 10.1007/s10802-006-9094-z. [DOI] [PubMed] [Google Scholar]
- 65.Polderman TJC, de Geus EJC, Hoekstra RA, Bartles M, van Leeuwen M, Verhulst FC, Posthuma D, Boomsma DI. Attention problems, inhibitory control, and intelligence index overlapping genetic factors: A study in 9-, 12-, and 18-year-old twins. Neuropsychology. 2009;23(3):381–391. doi: 10.1037/a0014915. [DOI] [PubMed] [Google Scholar]
- 66.Kaufman A, Kaufman N. Kaufman Assessment Battery for Children: Administration and Scoring Manual. Circle Pines, MN: American Guidance Service; 1984. [Google Scholar]
- 67.Achenbach TM, Rescorla LA. Manual for the ASEBA school-age forms & profiles. Burlington, VT: University of Vermont; 2001. [Google Scholar]
- 68.Tabachnick BG, Fidell LS. Using multivariate statistics. 5. New York, NY: Pearson; 2007. [Google Scholar]
- 69.Bentler PM. EQS 6.1 structural equations program manual. Encino, CA: Multivariate Software; 2004. [Google Scholar]
- 70.Byrne BM. Structural equation modeling with EQS: Basic concepts, applications, and programming. 2. Mahwah, NJ: Lawrence Erlbaum Associates, Inc; 2006. [Google Scholar]
- 71.Duncan TE, Duncan SC, Strycker LA. An introduction to latent variable growth curve modeling: Concepts, Issues, and Applications. 2. Manwah, NJ: Lawrence Erlbaum Associations, Inc; 2006. [Google Scholar]
- 72.Curran PJ, Hussong AM. The use of latent trajectory models in psychopathology research. Journal of Abnormal Psychology. 2003;112:526–544. doi: 10.1037/0021-843X.112.4.526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Pettit GS, Keiley MK, Laird RD, Bates JE, Dodge KA. Predicting the developmental course of mother reported monitoring across childhood and adolescence from early proactive parenting, child temperament, and parent’s worries. Journal of Family Psychology. 2007;21(2):206–217. doi: 10.1037/0893-3200.21.2.206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Muthen LK, Muthen BO. Mplus user’s guide: The comprehensive modeling program for applied researchers. Los Angeles: Muthen & Muthen; 1998. [Google Scholar]
- 75.Savage J, Brodsky NL, Malmud E, Giannetta JM, Hurt H. Attentional functioning and impulse control in cocaine-exposed and control children at age ten years. Developmental and Behavioral Pediatrics. 2005;26(1):42. [PubMed] [Google Scholar]
- 76.Byrne BM, Crombie G. Modeling and testing change: An introduction tothe latent growth curve model. Understanding Statistics. 2003;2:177–203. [Google Scholar]