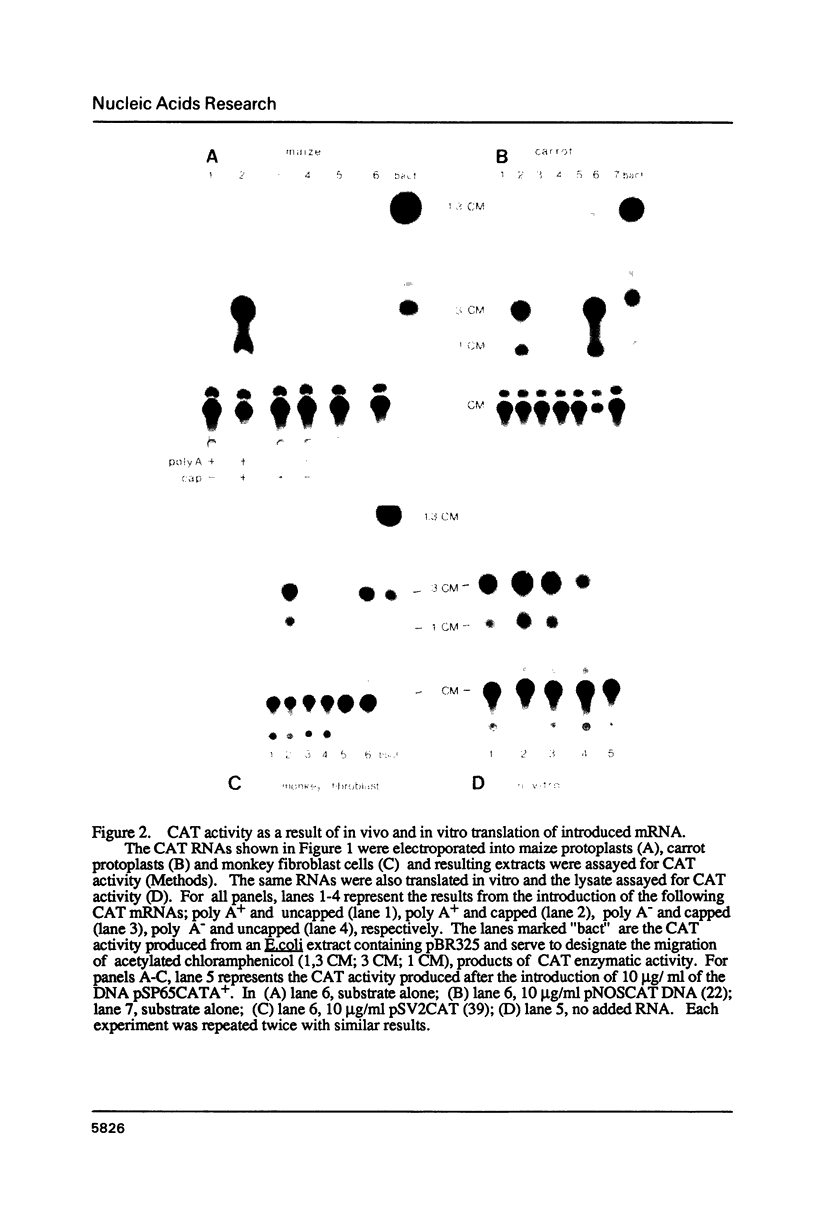
Abstract
A general method to introduce RNA molecules into plant protoplasts and animal cells is described. This technique utilizes the ability of electric pulses of high field strength to form pores in biomembranes. RNA molecules containing the coding region for the bacterial enzyme chloramphenicol acetyltransferase (CAT) were used as a model system. The presence of CAT activity as a result of the in vivo translation of the introduced RNA is entirely dependent on the presence of a 5' cap and greatly increased by the presence of a poly A tail at the 3' end. The introduction of RNA into eukaryotic cells has broad applicability both as an assay for the uptake of nucleic acids into cells independent of transcriptional activity and as a tool to study eukaryotic mRNA translation.
Full text
PDF







Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alton N. K., Vapnek D. Nucleotide sequence analysis of the chloramphenicol resistance transposon Tn9. Nature. 1979 Dec 20;282(5741):864–869. doi: 10.1038/282864a0. [DOI] [PubMed] [Google Scholar]
- Asselbergs F. A. Post-synthetic fate of the translation products of messenger RNA microinjected into Xenopus oocytes. Mol Biol Rep. 1979 Dec 31;5(4):199–208. doi: 10.1007/BF00782889. [DOI] [PubMed] [Google Scholar]
- Banerjee A. K. 5'-terminal cap structure in eucaryotic messenger ribonucleic acids. Microbiol Rev. 1980 Jun;44(2):175–205. doi: 10.1128/mr.44.2.175-205.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bergmann J. E., Lodish H. F. Translation of capped and uncapped vesicular stomatitis virus and reovirus mRNA'S. Sensitivity to m7GpppAm and ionic conditions. J Biol Chem. 1979 Jan 25;254(2):459–468. [PubMed] [Google Scholar]
- Birnboim H. C., Doly J. A rapid alkaline extraction procedure for screening recombinant plasmid DNA. Nucleic Acids Res. 1979 Nov 24;7(6):1513–1523. doi: 10.1093/nar/7.6.1513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brawerman G. The Role of the poly(A) sequence in mammalian messenger RNA. CRC Crit Rev Biochem. 1981;10(1):1–38. doi: 10.3109/10409238109114634. [DOI] [PubMed] [Google Scholar]
- Diacumakos E. G. Methods for micromanipulation of human somatic cells in culture. Methods Cell Biol. 1973;7:287–311. doi: 10.1016/s0091-679x(08)61783-5. [DOI] [PubMed] [Google Scholar]
- Dimitriadis G. J. Translation of rabbit globin mRNA introduced by liposomes into mouse lymphocytes. Nature. 1978 Aug 31;274(5674):923–924. doi: 10.1038/274923a0. [DOI] [PubMed] [Google Scholar]
- Ecker J. R., Davis R. W. Inhibition of gene expression in plant cells by expression of antisense RNA. Proc Natl Acad Sci U S A. 1986 Aug;83(15):5372–5376. doi: 10.1073/pnas.83.15.5372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fraley R. T., Dellaporta S. L., Papahadjopoulos D. Liposome-mediated delivery of tobacco mosaic virus RNA into tobacco protoplasts: A sensitive assay for monitoring liposome-protoplast interactions. Proc Natl Acad Sci U S A. 1982 Mar;79(6):1859–1863. doi: 10.1073/pnas.79.6.1859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Furuichi Y., LaFiandra A., Shatkin A. J. 5'-Terminal structure and mRNA stability. Nature. 1977 Mar 17;266(5599):235–239. doi: 10.1038/266235a0. [DOI] [PubMed] [Google Scholar]
- Gluzman Y. SV40-transformed simian cells support the replication of early SV40 mutants. Cell. 1981 Jan;23(1):175–182. doi: 10.1016/0092-8674(81)90282-8. [DOI] [PubMed] [Google Scholar]
- Gorman C. M., Moffat L. F., Howard B. H. Recombinant genomes which express chloramphenicol acetyltransferase in mammalian cells. Mol Cell Biol. 1982 Sep;2(9):1044–1051. doi: 10.1128/mcb.2.9.1044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Green M. R., Maniatis T., Melton D. A. Human beta-globin pre-mRNA synthesized in vitro is accurately spliced in Xenopus oocyte nuclei. Cell. 1983 Mar;32(3):681–694. doi: 10.1016/0092-8674(83)90054-5. [DOI] [PubMed] [Google Scholar]
- Greene J. J., Dieffenbach C. W., Ts'o P. O. Inactivation of interferon mRNA in the shutoff of human interferon synthesis. Nature. 1978 Jan 5;271(5640):81–83. doi: 10.1038/271081a0. [DOI] [PubMed] [Google Scholar]
- Grässman A. Mikrochirurgische Zellkerntransplantation bei Säugetierzellsn. Exp Cell Res. 1970 Jun;60(3):373–382. doi: 10.1016/0014-4827(70)90530-6. [DOI] [PubMed] [Google Scholar]
- Huez G., Bruck C., Cleuter Y. Translational stability of native and deadenylylated rabbit globin mRNA injected into HeLa cells. Proc Natl Acad Sci U S A. 1981 Feb;78(2):908–911. doi: 10.1073/pnas.78.2.908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krieg P. A., Melton D. A. Functional messenger RNAs are produced by SP6 in vitro transcription of cloned cDNAs. Nucleic Acids Res. 1984 Sep 25;12(18):7057–7070. doi: 10.1093/nar/12.18.7057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Littauer U. Z., Soreq H. The regulatory function of poly(A) and adjacent 3' sequences in translated RNA. Prog Nucleic Acid Res Mol Biol. 1982;27:53–83. doi: 10.1016/s0079-6603(08)60597-8. [DOI] [PubMed] [Google Scholar]
- Lockard R. E., Lane C. Requirement for 7-methylguanosine in translation of globin mRNA in vivo. Nucleic Acids Res. 1978 Sep;5(9):3237–3247. doi: 10.1093/nar/5.9.3237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melton D. A., Krieg P. A., Rebagliati M. R., Maniatis T., Zinn K., Green M. R. Efficient in vitro synthesis of biologically active RNA and RNA hybridization probes from plasmids containing a bacteriophage SP6 promoter. Nucleic Acids Res. 1984 Sep 25;12(18):7035–7056. doi: 10.1093/nar/12.18.7035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Merrick W. C. Translation of exogenous mRNAs in reticulocyte lysates. Methods Enzymol. 1983;101:606–615. doi: 10.1016/0076-6879(83)01041-1. [DOI] [PubMed] [Google Scholar]
- Morikawa H., Iida A., Matsui C., Ikegami M., Yamada Y. Gene transfer into intact plant cells by electroinjection through cell walls and membranes. Gene. 1986;41(1):121–124. doi: 10.1016/0378-1119(86)90274-x. [DOI] [PubMed] [Google Scholar]
- Neumann E., Schaefer-Ridder M., Wang Y., Hofschneider P. H. Gene transfer into mouse lyoma cells by electroporation in high electric fields. EMBO J. 1982;1(7):841–845. doi: 10.1002/j.1460-2075.1982.tb01257.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nudel U., Soreq H., Littauer U. Z. Globin mRNA species containing poly(A) segments of different lengths. Their functional stability in Xenopus oocytes. Eur J Biochem. 1976 Apr 15;64(1):115–121. doi: 10.1111/j.1432-1033.1976.tb10279.x. [DOI] [PubMed] [Google Scholar]
- Ostro M. J., Giacomoni D., Lavelle D., Paxton W., Dray S. Evidence for translation of rabbit globin mRNA after liposome-mediated insertion into a human cell line. Nature. 1978 Aug 31;274(5674):921–923. doi: 10.1038/274921a0. [DOI] [PubMed] [Google Scholar]
- Ou-Lee T. M., Turgeon R., Wu R. Expression of a foreign gene linked to either a plant-virus or a Drosophila promoter, after electroporation of protoplasts of rice, wheat, and sorghum. Proc Natl Acad Sci U S A. 1986 Sep;83(18):6815–6819. doi: 10.1073/pnas.83.18.6815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pelham H. R., Jackson R. J. An efficient mRNA-dependent translation system from reticulocyte lysates. Eur J Biochem. 1976 Aug 1;67(1):247–256. doi: 10.1111/j.1432-1033.1976.tb10656.x. [DOI] [PubMed] [Google Scholar]
- Potter H., Weir L., Leder P. Enhancer-dependent expression of human kappa immunoglobulin genes introduced into mouse pre-B lymphocytes by electroporation. Proc Natl Acad Sci U S A. 1984 Nov;81(22):7161–7165. doi: 10.1073/pnas.81.22.7161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riggs C. D., Bates G. W. Stable transformation of tobacco by electroporation: evidence for plasmid concatenation. Proc Natl Acad Sci U S A. 1986 Aug;83(15):5602–5606. doi: 10.1073/pnas.83.15.5602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roberts B. E., Paterson B. M. Efficient translation of tobacco mosaic virus RNA and rabbit globin 9S RNA in a cell-free system from commercial wheat germ. Proc Natl Acad Sci U S A. 1973 Aug;70(8):2330–2334. doi: 10.1073/pnas.70.8.2330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sippel A. E., Stavrianopoulos J. G., Schutz G., Feigelson P. Translational properties of rabbit globin mRNA after specific removal of poly(A) with ribonuclease H. Proc Natl Acad Sci U S A. 1974 Nov;71(11):4635–4639. doi: 10.1073/pnas.71.11.4635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soreq H., Sagar A. D., Sehgal P. B. Translational activity and functional stability of human fibroblast beta 1 and beta 2 interferon mRNAs lacking 3'-terminal RNA sequences. Proc Natl Acad Sci U S A. 1981 Mar;78(3):1741–1745. doi: 10.1073/pnas.78.3.1741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soreq H. The biosynthesis of biologically active proteins in mRNA-microinjected Xenopus oocytes. CRC Crit Rev Biochem. 1985;18(3):199–238. doi: 10.3109/10409238509085134. [DOI] [PubMed] [Google Scholar]
- Stacey D. W., Allfrey V. G. Microinjection studies of duck globin messenger RNA translation in human and avian cells. Cell. 1976 Dec;9(4 Pt 2):725–732. doi: 10.1016/0092-8674(76)90136-7. [DOI] [PubMed] [Google Scholar]
- Williamson R., Crossley J., Humphries S. Translation of mouse globin messenger ribonucleic acid from which the poly(adenylic acid) sequence has been removed. Biochemistry. 1974 Feb 12;13(4):703–707. doi: 10.1021/bi00701a011. [DOI] [PubMed] [Google Scholar]
- Winkler M. M., Bruening G., Hershey J. W. An absolute requirement for the 5' cap structure for mRNA translation in sea urchin eggs. Eur J Biochem. 1983 Dec 1;137(1-2):227–232. doi: 10.1111/j.1432-1033.1983.tb07819.x. [DOI] [PubMed] [Google Scholar]
- Wodnar-Filipowicz A., Szczesna E., Zan-Kowalczewska M., Muthukrishnan S., Szybiak U., Legocki A. B., Filipowicz W. 5'-Terminal 7-methylguanosine and mRNA function. The effect of enzymatic decapping and of cap analogs on translation of tobacco-mosaic-virus RNA and globin mRNA in vitro. Eur J Biochem. 1978 Dec 1;92(1):69–80. doi: 10.1111/j.1432-1033.1978.tb12724.x. [DOI] [PubMed] [Google Scholar]