Abstract
OBJECTIVE
We performed a retrospective analysis of a national cohort of veterans with diabetes to better understand regional, geographic, and racial/ethnic variation in diabetes control as measured by HbA1c.
RESEARCH DESIGN AND METHODS
A retrospective cohort study was conducted in a national cohort of 690,968 veterans with diabetes receiving prescriptions for insulin or oral hypoglycemic agents in 2002 that were followed over a 5-year period. The main outcome measures were HbA1c levels (as continuous and dichotomized at ≥8.0%).
RESULTS
Relative to non-Hispanic whites (NHWs), HbA1c levels remained 0.25% higher in non-Hispanic blacks (NHBs), 0.31% higher in Hispanics, and 0.14% higher in individuals with other/unknown/missing racial/ethnic group after controlling for demographics, type of medication used, medication adherence, and comorbidities. Small but statistically significant geographic differences were also noted with HbA1c being lowest in the South and highest in the Mid-Atlantic. Rural/urban location of residence was not associated with HbA1c levels. For the dichotomous outcome poor control, results were similar with race/ethnic group being strongly associated with poor control (i.e., odds ratios of 1.33 [95% CI 1.31–1.35] and 1.57 [1.54–1.61] for NHBs and Hispanics vs. NHWs, respectively), geographic region being weakly associated with poor control, and rural/urban residence being negligibly associated with poor control.
CONCLUSIONS
In a national longitudinal cohort of veterans with diabetes, we found racial/ethnic disparities in HbA1c levels and HbA1c control; however, these disparities were largely, but not completely, explained by adjustment for demographic characteristics, medication adherence, type of medication used to treat diabetes, and comorbidities.
Diabetes affects more than 23 million Americans, is the seventh leading cause of death in the U.S., and is prevalent worldwide (1,2). However, despite decades of effective treatments for diabetes, control rates for the disease remain below goals set by experts (3,4). A number of studies report higher HbA1c levels in African Americans and Hispanics relative to non-Hispanic whites (NHWs) (5–9).
Current proposed mechanisms for racial/ethnic disparities in HbA1c levels relate to both glucose control and glucose-independent variability in HbA1c levels (5,6,9). Nevertheless, the social determinants of poor adherence need to be elucidated to more adequately understand why Hispanics and non-Hispanic blacks (NHBs) have problems with poor control of their diabetes even when access to care is not a factor. Racial disparities in access to and use of medical care, and provider behavior are lower in the Veterans Health Administration (VHA) than outside the VHA (10). Studies have indicated that quality of care for diabetes is higher in the VHA than outside the VHA (11). In a recent publication by our group based on a cohort of 8,813 NHW and NHB veterans obtaining care at a Veterans Administration (VA) facility in the southeastern U.S., we reported a mean difference in HbA1c levels of 0.43% between NHW and NHB veterans after adjustment for demographic factors and comorbidities, but not type of medication used to treat diabetes or medication adherence (12).
Few studies have examined regional or geographic variations in HbA1c (13–15). Even within the VA, rural residents have disparities in access to care compared with urban residents because of distance to VA facilities (16). Moreover, there are geographic differences in the incidence of diabetes, with levels being higher in the Stroke Belt, composed of the southeastern states, compared with the rest of the U.S. (17). To address this gap in the literature, we performed a retrospective analysis of a national cohort of more than 600,000 diabetic veterans during a 5-year period of observation. We hypothesized that HbA1c levels would be higher in the south, that HbA1c levels would be higher in rural than in urban areas, and that HbA1c levels would be higher in NHBs and Hispanics than in NHWs. In addition, we hypothesized that HbA1c levels would be related to type of medication used to treat diabetes and would vary over time.
RESEARCH DESIGN AND METHODS
Study population
A national cohort of veterans with type 2 diabetes was created by linking patient and administrative files from the VHA National Patient Care and Pharmacy Benefits Management databases. The Pharmacy Benefits Management database includes use information for every prescription filled in the VA. Veterans were included in the cohort if they had type 2 diabetes defined by two or more International Classification of Diseases, Ninth Revision (ICD-9) codes for diabetes (250, 357.2, 362.0, and 366.41) in the previous 24 months (2000 and 2001) and during 2002 from inpatient stays or outpatient visits on separate days (excluding codes from laboratory tests and other nonclinician visits), and prescriptions for insulin or oral hypoglycemic agents (VA classes HS501 or HS502, respectively) in 2002 (18). Veterans identified as having type 2 diabetes by ICD-9 codes were excluded from the cohort if they did not have prescriptions for diabetic medications (HS501 or HS502) in 2002 (see Supplementary Fig. 1 for cohort definition). The datasets were linked using patient scrambled Social Security Numbers. When the data were merged and limited to include complete adherence data, this resulted in a cohort of 690,968 veterans, of whom 72.86% were NHW, 12.85% were NHB, and 5.11% were Hispanic with type 2 diabetes. There were also 9.18% of veterans with missing or unknown race/ethnicity information. For a comparison of the 690,698 in our study population with those veterans with type 2 diabetes who were not using diabetes medication (n = 201,255), see Supplementary Table 1. The study was approved by our institutional review board and local VA Research and Development committee.
Outcome measure
The primary outcome was HbA1c level. In addition, poor control was defined as HbA1c ≥8.0%.
Primary covariates
The primary covariates of interest were 1) race/ethnicity, classified as NHW, NHB, Hispanic, and other/unknown/missing; 2) location of residence (urban and rural/highly rural) (19); and 3) region, which accounts for the five geographic regions of the country based on the VHA Veteran’s Integrated Service Networks (VISNs): Northeast (VISNs 1, 2, and 3), Mid-Atlantic (VISNs 4, 5, 6, 9, and 10), South (VISNs 7, 8, 16, and 17), Midwest (VISNs 11, 12, 15, 19, and 23), and West (VISNs 18, 20, 21, and 22) (20). The race covariate was retrieved from the 2002 outpatient and inpatient Medical SAS datasets. When missing or unknown, the variable was supplemented using the inpatient 2003 Medical SAS dataset fields for race, the outpatient 2004 Medical SAS dataset fields for race, and the VA Vital Status Centers for Medicare and Medicaid Services field for race.
Demographic variables
Age, sex, marital status (i.e., single or married), and percentage service-connectedness (i.e., degree of disability due to illness or injury that was aggravated by or incurred in military service) were available.
Medication type and medication possession ratio
Veterans were classified as to whether they were taking insulin only, insulin and oral hypoglycemic agents, or oral hypoglycemic agents only. Annual medication possession ratio (MPR) was calculated and defined as the number of days supply divided by 365 days (or if deceased during that year, the number of days until death) over the study period for each veteran for insulin or oral hypoglycemic agents (VA classes HS501 or HS502, respectively). If MPR exceeded one, MPR was set to one. In addition, MPR was dichotomized at >80%.
Comorbidity
Variables included substance abuse, anemia, cancer, cerebrovascular disease, congestive heart failure, cardiovascular disease, depression, hypertension, hypothyroidism, liver disease, lung disease, fluid and electrolyte disorders, obesity, psychoses, peripheral vascular disease, and other (AIDS, rheumatoid arthritis, renal failure, peptic ulcer disease and bleeding, weight loss), and were defined according to ICD-9 codes at entry into the cohort.
Statistical analysis
Models for the relationship between HbA1c and race, urban/rural residence, and VISN were developed in a sequential fashion. We used a generalized linear model approach (lmer function in R) to fit the models for HbA1c as a response variable with race/ethnicity, location of residence (urban/rural), and region as main independent variables of interest with adjustment for potential confounders. The general model had the following regression format, E(Yi |Xi,Zi) = g−1(Xiβ + Ziγi), where g is a monotone link function and Yi is the response for the ith subject with Ti (Ti = 1,…,5) repeated measurements, Xi and Zi represent vectors of fixed and random effect covariates, respectively, and g is considered as the identity link resulting in a linear mixed model for the continuous HbA1c outcome and the logit link for the dichotomous HbA1c outcome. A person-level random effect was included in all models to account for within-individual correlations. This approach accommodates a wide range of distributional assumptions, multilevel data such as longitudinal measurements on subjects, measurement of subjects at different time points, modeling individual level effects, missing data, and time varying or invariant covariates (21). Model fit was assessed using plots of residuals and predicted values. All statistical tests used a two-tailed α = 0.05 level of significance and were performed in R, version 2.11.0.
RESULTS
The study population consisted of 690,968 veterans with diabetes receiving prescriptions for insulin or oral hypoglycemic agents (VA classes HS501 or HS502, respectively) in 2002 that were followed until death, loss to follow-up, or through December 2006. During the follow-up period, 24.65% of individuals in the cohort died. Table 1 shows the characteristics of the sample.
Table 1.
Sample characteristics
| Mean/percent (SD/SE) | |
|---|---|
| Mean HbA1c | 7.48 (1.63) |
| Mean minimum HbA1c | 6.96 (1.49) |
| Mean maximum HbA1c | 8.04 (2.13) |
| Racial/ethnic group | |
| NHW | 72.86 (0.07) |
| NHB | 12.85 (0.11) |
| Hispanic | 5.11 (0.12) |
| Other | 9.18 (0.12) |
| Rural residence | 38.87 (0.09) |
| Geographic region | |
| Northeast | 11.17 (0.11) |
| Mid-Atlantic | 22.57 (0.11) |
| South | 30.00 (0.11) |
| Midwest | 21.12 (0.11) |
| West | 14.88 (0.11) |
| Age | 65.77 (11.28) |
| Male | 97.81 (0.02) |
| Percentage service-connected disability | 12.26 (27.21) |
| Married | 65.58 (0.07) |
| Medication type used to control diabetes | |
| Oral hypoglycemics | 69.37 (0.07) |
| Insulin | 14.28 (0.11) |
| Insulin and oral hypoglycemics | 16.35 (0.11) |
| MPR all (%) | 70.94 (20.53) |
| MPR 501 only (%) | 64.65 (28.26) |
| MPR 502 only (%) | 73.04 (32.34) |
| MPR 501 and 502 (%) | 66.04 (20.36) |
| Comorbidities | |
| Substance abuse | 3.79 (0.12) |
| Anemia | 7.48 (0.12) |
| Cancer | 7.60 (0.12) |
| Cerebrovascular disease | 11.68 (0.11) |
| Congestive heart failure | 11.59 (0.11) |
| Cardiovascular disease | 3.62 (0.12) |
| Depression | 12.42 (0.11) |
| Hypertension | 77.61 (0.06) |
| Hypothyroidism | 6.17 (0.12) |
| Liver disease | 3.14 (0.12) |
| Lung conditions | 13.88 (0.11) |
| Electrolyte disorders | 4.95 (0.12) |
| Obesity | 12.48 (0.11) |
| Psychoses | 4.31 (0.12) |
| Peripheral vascular disease | 12.02 (0.11) |
| Other disease | 3.72 (0.12) |
| Dead | 24.65 (0.11) |
Relative to NHWs, HbA1c was 0.45% higher in NHBs, 0.38% higher in Hispanics, and 0.57% higher in individuals with other/unknown/missing racial/ethnic group after adjusting for trends over time (Table 2). After further adjusting for demographic factors, including location of residence and geographic region, racial/ethnic HbA1c differences were moderately attenuated and changes over time were altered, but the pattern remained similar. Adding medication type, MPR, and comorbidities to the final model altered racial/ethnic group HbA1c differences only slightly. A small, but statistically significant difference in HbA1c with respect to rural/urban residence was found with rural residence being associated with slightly higher HbA1c than urban residence. Small, but statistically significant regional differences were also noted with HbA1c being lowest in the South and highest in the Mid-Atlantic. Adding medication type (i.e., insulin combined with oral hypoglycemic medication, insulin only, and hypoglycemic medication only), MPR, and comorbidities to the final model altered regional differences in HbA1c levels moderately, but HbA1c levels remained lowest in the South and highest in the Mid-Atlantic. Rural/urban differences were attenuated. Medication type was associated with HbA1c levels. Relative to individuals using only oral hypoglycemic medications, HbA1c was 0.53% higher in individuals using insulin only and 0.81% higher in individuals using insulin combined with oral hypoglycemic medication. MPR was not significantly associated with HbA1c levels.
Table 2.
Regression coefficient estimates and 95% CIs for HbA1c in veterans with type 2 diabetes
| Model 1* | Model 2* | Final model* | |
|---|---|---|---|
| Intercept |
7.424 (7.411–7.437) |
7.534 (7.518–7.551) |
7.4205 (7.4027–7.4383) |
| Time |
−0.136 (−0.144 to −0.127) |
−0.160 (−0.168 to −0.151) |
−0.1603 (−0.1689 to −0.1517) |
| Time-squared |
0.027 (0.026–0.029) |
0.029 (0.028–0.030) |
0.0279 (0.0265–0.0292) |
| Racial/ethnic group |
|||
| NHB |
0.454 (0.444–0.465) |
0.265 (0.254–0.276) |
0.2529 (0.2424–0.2634) |
| Hispanic |
0.375 (0.360–0.391) |
0.317 (0.301–0.333) |
0.3139 (0.2987–0.3290) |
| Other |
0.570 (0.557–0.582) |
0.142 (0.129–0.155) |
0.1799 (0.1674–0.1924) |
| Residence (rural vs. urban) |
0.010 (0.002–0.017) |
0.0046 (−0.0025 to 0.0117) |
|
| Geographic region (reference: Northeast) |
|||
| South |
−0.054 (−0.066 to −0.041) |
−0.0797 (−0.0915 to −0.0678) |
|
| Mid-Atlantic |
0.040 (0.027–0.053) |
0.0131 (0.0007–0.0255) |
|
| Midwest |
−0.026 (−0.039 to −0.013) |
−0.0703 (−0.0829 to −0.0578) |
|
| West |
−0.027 (−0.041 to −0.014) |
−0.0605 (−0.0737 to −0.0472) |
|
| Age (1-year increase)† |
−0.031 (−0.031 to −0.031) |
−0.0261 (−0.0265 to −0.0258) |
|
| Sex (female vs. male) |
−0.029 (−0.052 to −0.005) |
−0.0118 (−0.0346 to 0.0110) |
|
| SCD (1% increase) |
−0.001 (−0.002 to −0.001) |
−0.0014 (−0.0015 to −0.0113) |
|
| Marital status (single vs. married) |
0.096 (0.088–0.103) |
0.1007 (0.0934–0.1079) |
|
| Medication type used to control diabetes (reference: oral hypoglycemics) |
|||
| Insulin |
0.5326 (0.5239–0.05414) |
||
| Insulin and oral hypoglycemics |
0.8146 (0.8070–0.8221) |
||
| Nonadherence (MPR ≤80% vs. >80%) |
0.0031 (−0.0021 to 0.0084) |
||
| Comorbidities (disease vs. no disease) |
|||
| Substance abuse |
0.0215 (0.0033–0.0397) |
||
| Anemia |
−0.1592 (−0.1725 to −0.1460) |
||
| Cancer |
−0.0577 (−0.0708 to −0.0447) |
||
| Cerebrovascular disease |
−0.0685 (−0.0792 to −0.0577) |
||
| Congestive heart failure |
0.0794 (0.0682 to 0.0907) |
||
| Cardiovascular disease |
0.0334 (0.0151–0.0516) |
||
| Depression |
0.0464 (0.0360–0.0568) |
||
| Hypertension |
−0.0810 (−0.0893 to −0.0727) |
||
| Hypothyroidism |
−0.0331 (−0.0470 to −0.0192) |
||
| Liver disease |
−0.2818 (−0.3013 to −0.2624) |
||
| Lung conditions |
−0.0171 (−0.0270 to −0.0072) |
||
| Electrolyte disorders |
0.0230 (0.0070–0.0390) |
||
| Obesity |
−0.0465 (−0.0565 to −0.0365) |
||
| Psychoses |
−0.0572 (−0.0742 to −0.0402) |
||
| Peripheral vascular disease |
0.0255 (0.0149–0.0361) |
||
| Other disease | −0.1578 (−0.1759 to −0.1397) | ||
SCD, service-connected disability.
*Includes a random subject term.
†Age centered.
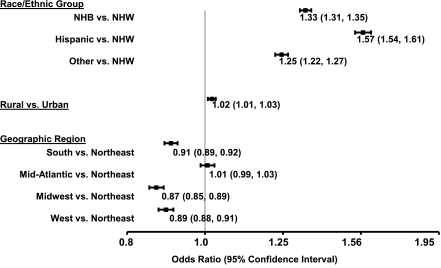
Relative to NHWs, the odds of having uncontrolled versus controlled HbA1c (defined as HbA1c >8.0%) was 1.71-fold (95% CI 1.68–1.73) higher in NHBs, 1.63-fold (1.60–1.66) higher in Hispanics, and 1.91-fold (1.87–1.94) higher in individuals with other/unknown/missing racial/ethnic group after adjusting for trends over time (Table 3). The odds of having uncontrolled versus controlled HbA1c initially decreased, but then increased over time. After further adjusting for demographic factors (including location of residence and geographic region), changes over time were altered, but the pattern remained similar. Odds ratios relative to NHWs for uncontrolled HbA1c were 1.35 (1.33–1.37), 1.53 (1.50–1.57), and 1.14 (1.11–1.16) for NHBs, Hispanics, and individuals with other/unknown/missing racial/ethnic group over time, respectively (Fig. 1). The odds of having uncontrolled HbA1c levels decreased with increasing age, were lower in women than men, and were higher in single versus married individuals, but were not associated with service-connected disability. Small, but statistically significant differences in the odds of having uncontrolled HbA1c with respect to rural/urban residence were found; rural residence was associated with a slightly higher odds of having uncontrolled HbA1c compared with urban residence (Fig. 1). Small, but statistically significant regional differences were also noted with the odds of having uncontrolled HbA1c lowest in the Midwest and highest in the Mid-Atlantic (Fig. 1). Adding medication type, MPR, and comorbidities to a final model altered changes in HbA1c control over time, racial/ethnic group differences in HbA1c control, and regional differences in HbA1c control only slightly. Medication type was associated with HbA1c control relative to individuals using only oral hypoglycemic medications. The odds of having uncontrolled HbA1c were 2.35-fold (2.32–2.38) higher in individuals using insulin only and 3.53-fold (3.49–3.57) higher in individuals using insulin combined with oral hypoglycemic medication. In addition, having an MPR less than 80% was associated with a 5% increase in the odds of having uncontrolled HbA1c (odds ratio 1.05 [1.04–1.06]).
Table 3.
Odds ratios and 95% CIs for poor control (HbA1c ≥8) in veterans with type 2 diabetes
| Model 1* | Model 2* | Final model* | |
|---|---|---|---|
| Racial/ethnic group (reference: NHW) |
|||
| NHB |
1.707 (1.683–1.732) |
1.353 (1.333–1.374) |
1.333 (1.311–1.354) |
| Hispanic |
1.629 (1.595–1.664) |
1.534 (1.499–1.569) |
1.572 (1.536–1.608) |
| Other |
1.905 (1.874–1.937) |
1.135 (1.114–1.155) |
1.245 (1.222–1.268) |
| Residence (rural vs. urban) |
1.026 (1.014–1.037) |
1.020 (1.009–1.032) |
|
| Geographic region (reference: Northeast) |
|||
| South |
0.952 (0.934–0.969) |
0.907 (0.890–0.924) |
|
| Mid-Atlantic |
1.053 (1.033–1.074) |
1.007 (0.987–1.027) |
|
| Midwest |
0.941 (0.922–0.959) |
0.870 (0.852–0.888) |
|
| West |
0.951 (0.931–0.971) |
0.894 (0.875–0.913) |
|
| Age (1-year increase)† |
0.959 (0.959–0.960) |
0.965 (0.965–0.966) |
|
| Sex (female vs. male) |
0.920 (0.888–0.953) |
0.943 (0.910–0.977) |
|
| SCD (1% increase) |
0.999 (0.999–1.000) |
0.998 (0.998–0.999) |
|
| Marital status (single vs. married) |
1.149 (1.136–1.161) |
1.162 (1.149–1.175) |
|
| Medication type used to control diabetes (reference: oral hypoglycemics) |
|||
| Insulin |
2.349 (2.316–2.383) |
||
| Insulin and oral hypoglycemics |
3.530 (3.487–3.573) |
||
| Nonadherence (MPR ≤80% vs. >80%) |
1.048 (1.038–1.058) |
||
| Comorbidities (disease vs. no disease) |
|||
| Substance abuse |
1.002 (0.975–1.029) |
||
| Anemia |
0.837 (0.819–0.856) |
||
| Cancer |
0.924 (0.903–0.944) |
||
| Cerebrovascular disease |
0.932 (0.915–0.948) |
||
| Congestive heart failure |
1.136 (1.116–1.156) |
||
| Cardiovascular disease |
1.065 (1.034–1.096) |
||
| Depression |
1.071 (1.054–1.088) |
||
| Hypertension |
0.907 (0.896–0.919) |
||
| Liver disease |
0.766 (0.742–0.790) |
||
| Lung conditions |
0.998 (0.973–1.004) |
||
| Electrolyte disorders |
1.022 (0.997–1.048) |
||
| Obesity |
0.964 (0.949–0.979) |
||
| Psychoses |
0.929 (0.905–0.954) |
||
| Peripheral vascular disease |
1.033 (1.016–1.051) |
||
| Other disease | 0.847 (0.822–0.872) | ||
SCD, service-connected disability.
*Sequentially built models that each include time and time squared.
†Age centered.
Figure 1.
Odds ratios and 95% CIs for poor control (HbA1c ≥8) in veterans with type 2 diabetes, final model.
In the final models, HbA1c levels and control (i.e., defined as HbA1c ≤8.0%) were also associated with comorbidities; however, the strength and direction of the association depended on the comorbidity (Tables 2 and 3).
CONCLUSIONS
The findings of this study are important for the following reasons. First, few prior studies have examined geographic or regional differences in either HbA1c testing or HbA1c levels, and prior national studies have not examined geographic or regional differences in HbA1c levels. Moreover, prior national studies on racial/ethnic differences in HbA1c levels at the VA have not focused on HbA1c levels in Hispanics relative to NHWs or adjusted for type of diabetes medication, medication adherence, or comorbidities. These unique features of our study combined with the unique setting of the VA, where racial/ethnic disparities in access to and use of medical care, and provider behavior are limited, are novel aspects of our study that contribute to the literature and to our understanding of factors that influence HbA1c levels. Finally, understanding geographic and regional differences in HbA1c levels and control may have important policy implications if we are able to identify factors within each setting that contribute to differences.
Despite equal access in the VHA, we report longitudinal racial/ethnic differences in HbA1c. A major contribution of this study is the ability to control for demographic factors, type of medication used (as a surrogate for disease severity), medication adherence, and a variety of comorbidities. In addition, the study included a large sample of Hispanics. Previous VA studies have not examined HbA1c levels in Hispanics relative to NHWs or NHBs. Notably, our adjusted reported mean difference in HbA1c levels between NHBs and NHWs is smaller (0.25%) than that reported in a recent cross-sectional study (0.47%), which adjusted for plasma glucose levels, demographics, and a few other variables, not including type of medication used (9). The magnitude of the racial/ethnic disparity was similar in our study as in a smaller VA study (n = 4,080) that reported glucose-independent mean differences in HbA1c levels between NHBs and NHWs of ∼0.2% among individuals receiving consistent primary care at the VA (22).
Studies outside the VHA have consistently reported racial/ethnic disparities (7). A meta-analysis of disparities in HbA1c levels between NHWs and NHBs with diabetes, which included 11 articles published between 1993 and 2005, reported crude unadjusted HbA1c differences between NHWs and NHBs of ∼0.65% (7). Our adjusted estimate of mean difference in HbA1c between NHBs and NHWs (0.25%) is substantially lower (7). Studies conducted outside a managed care setting generally report wider disparities. One smaller (n = 1,034) study conducted in a nationally representative sample reported mean differences in HbA1c levels of 0.73% between NHBs and NHWs and 0.74% between Latinos and NHWs after adjusting for demographics, clinical characteristics, access and quality of care, health behaviors, medication adherence, and self-management attitudes (23).
HbA1c levels were highly correlated with average glucose levels over the proceeding 3-month period (24); however, glucose is not the only factor that affects HbA1c levels. Current proposed mechanisms for racial/ethnic disparities in HbA1c levels are physiologic, genetic, socioeconomic, and cultural in nature. The social determinants of poor adherence need to be better elucidated to adequately understand why Hispanics and NHBs have more problems with poor control of their diabetes even when access to care is not a factor. However, in considering mechanisms in racial/ethnic disparities in HbA1c levels, it is important to remember that glucose-adjusted racial and ethnic disparities in HbA1c are estimated to be less than 0.5% in individuals with diabetes (5,6,9).
Few studies have examined geographic or regional differences in HbA1c testing or control (13–15). Results from these studies indicate that HbA1c testing is lower in rural than urban areas, especially in the rural south (13–15). In one study conducted in Oregon, HbA1c testing rates in areas with rural health clinics were higher than in areas without rural health clinics and similar to levels in urban areas (15). Thus, there may be important policy implications given the geographic differences that we found.
Strengths of our study include the study population, all veterans with type 2 diabetes receiving care at the VHA had prescriptions for diabetes medication in 2002, the longitudinal design with 5 years of follow-up data, the extensive data available on comorbidities, the ability to identify racial/ethnic group in >90% of the cohort, the longitudinal information on the type of medication used to treat diabetes, and the information on medication nonadherence as measured by an MPR <80%. There are also limitations of our study. First, we do not have information on plasma glucose levels and are therefore unable to examine glucose-independent variability in HbA1c levels. Second, MPR is really only a proxy for medication adherence, and because of wastage the reliability of MPR for insulin needs further validation; however, prior studies of insulin MPR have been conducted using VA data (25). Third, the VA medical record does not include information on socioeconomic status, which is likely to be an important factor. Fourth, women make up a small proportion, and women veterans may not be representative of women in the general population.
In summary, in a national longitudinal cohort of veterans with diabetes, we found racial/ethnic disparities in HbA1c levels and HbA1c control after adjustment for demographic characteristics, medication adherence, type of medication used to treat diabetes, and comorbidities. As efforts are made to better elucidate the mechanisms of racial/ethnic disparities in HbA1c levels, it will be important to determine whether similar HbA1c levels across racial/ethnic groups have the same impact on macrovascular outcomes and mortality risk. Moreover, understanding geographic differences in HbA1c levels and control may have important policy implications if the findings are replicated in non-VA samples and we are able to identify factors within each setting that contribute to differences.
Supplementary Material
Acknowledgments
This study was supported by Grant IIR-06-219 funded by the VHA Health Services Research and Development program. The funding agency did not participate in the design and conduct of the study; collection, management, analysis, and interpretation of the data; and preparation, review, or approval of the manuscript. The article represents the views of the authors and not those of the VA or Health Services Research and Development.
No potential conflicts of interest relevant to this article were reported.
L.E.E. conceived and designed the study, researched data, analyzed and interpreted data, reviewed and edited the manuscript, and supervised the study. M.G. and K.J.H. analyzed and interpreted data, wrote the manuscript, and reviewed and edited the manuscript. R.N.A., C.E., and G.E.G. analyzed and interpreted data and wrote the manuscript. P.D.M. conceived and designed the study, researched data, analyzed and interpreted data, wrote the manuscript, reviewed and edited the manuscript, and supervised the study.
Footnotes
This article contains Supplementary Data online at http://care.diabetesjournals.org/lookup/suppl/doi:10.2337/dc10-1504/-/DC1.
References
- 1.Centers for Disease Control and Prevention National Diabetes Fact Sheet: General Information and Estimates on Diabetes in the United States, 2007. Atlanta, GA, U.S. Department of Health and Human Services, 2007 [Google Scholar]
- 2.Wild S, Roglic G, Green A, Sicree R, King H. Global prevalence of diabetes: estimates for the year 2000 and projections for 2030. Diabetes Care 2004;27:1047–1053 [DOI] [PubMed] [Google Scholar]
- 3.Saaddine JB, Cadwell B, Gregg EW, et al. Improvements in diabetes processes of care and intermediate outcomes: United States, 1988-2002. Ann Intern Med 2006;144:465–474 [DOI] [PubMed] [Google Scholar]
- 4.Healthy People 2010 [Internet]. Available from http://www.healthypeople.gov/ Accessed 26 May 2010
- 5.Herman WH, Dungan KM, Wolffenbuttel BH, et al. Racial and ethnic differences in mean plasma glucose, hemoglobin A1c, and 1,5-anhydroglucitol in over 2000 patients with type 2 diabetes. J Clin Endocrinol Metab 2009;94:1689–1694 [DOI] [PubMed] [Google Scholar]
- 6.Herman WH, Ma Y, Uwaifo G, et al. Diabetes Prevention Program Research Group Differences in A1C by race and ethnicity among patients with impaired glucose tolerance in the Diabetes Prevention Program. Diabetes Care 2007;30:2453–2457 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kirk JK, D’Agostino RB, Jr, Bell RA, et al. Disparities in HbA1c levels between African-American and non-Hispanic white adults with diabetes: a meta-analysis. Diabetes Care 2006;29:2130–2136 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Weatherspoon LJ, Kumanyika SK, Ludlow R, Schatz D. Glycemic control in a sample of black and white clinic patients with NIDDM. Diabetes Care 1994;17:1148–1153 [DOI] [PubMed] [Google Scholar]
- 9.Ziemer DC, Kolm P, Weintraub WS, et al. Glucose-independent, black-white differences in hemoglobin A1c levels: a cross-sectional analysis of 2 studies. Ann Intern Med 2010;152:770–777 [DOI] [PubMed] [Google Scholar]
- 10.Heisler M, Smith DM, Hayward RA, Krein SL, Kerr EA. Racial disparities in diabetes care processes, outcomes, and treatment intensity. Med Care 2003;41:1221–1232 [DOI] [PubMed] [Google Scholar]
- 11.Kerr EA, Gerzoff RB, Krein SL, et al. Diabetes care quality in the Veterans Affairs Health Care System and commercial managed care: the TRIAD study. Ann Intern Med 2004;141:272–281 [DOI] [PubMed] [Google Scholar]
- 12.Egede LE, Mueller M, Echols CL, Gebregziabher M. Longitudinal differences in glycemic control by race/ethnicity among veterans with type 2 diabetes. Med Care 2010;48:527–533 [DOI] [PubMed] [Google Scholar]
- 13.Andrus MR, Kelley KW, Murphey LM, Herndon KC. A comparison of diabetes care in rural and urban medical clinics in Alabama. J Community Health 2004;29:29–44 [DOI] [PubMed] [Google Scholar]
- 14.Weingarten JP, Jr, Brittman S, Hu W, Przybyszewski C, Hammond JM, FitzGerald D. The state of diabetes care provided to Medicare beneficiaries living in rural America. J Rural Health 2006;22:351–358 [DOI] [PubMed] [Google Scholar]
- 15.Kirkbride K, Wallace N. Rural health clinics and diabetes-related primary care for Medicaid beneficiaries in Oregon. J Rural Health 2009;25:247–252 [DOI] [PubMed] [Google Scholar]
- 16.Weeks WB, Bott DM, Lamkin RP, Wright SM. Veterans Health Administration and Medicare outpatient health care utilization by older rural and urban New England veterans. J Rural Health 2005;21:167–171 [DOI] [PubMed] [Google Scholar]
- 17.Voeks JH, McClure LA, Go RC, et al. Regional differences in diabetes as a possible contributor to the geographic disparity in stroke mortality: the REasons for Geographic And Racial Differences in Stroke Study. Stroke 2008;39:1675–1680 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Miller DR, Safford MM, Pogach LM. Who has diabetes? Best estimates of diabetes prevalence in the Department of Veterans Affairs based on computerized patient data. Diabetes Care 2004;27(Suppl. 2):B10–B21 [DOI] [PubMed] [Google Scholar]
- 19.West AN, Lee RE, Shambaugh-Miller MD, et al. Defining “rural” for veterans’ health care planning. J Rural Health 2010;26:301–309 [DOI] [PubMed] [Google Scholar]
- 20.Department of Veterans Affairs Field Research Advisory Committee. Department of Veterans Affairs, Veterans Health Administration, Office of Research and Development, Field Research Advisory Committee Operating Procedures [Internet], 2004. Available from http://www.research.va.gov/about/frac/FRAC-ops.pdf Accessed 19 February 2011
- 21.Diggle PJ, Heagerty P, Liang K-Y, Zeger SL. Analysis of Longitudinal Data. 2nd ed. Oxford, England, Oxford University Press, 2002 [Google Scholar]
- 22.Twombly JG, Long Q, Zhu M, et al. Diabetes care in black and white veterans in the southeastern U.S. Diabetes Care 2010;33:958–963 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Heisler M, Faul JD, Hayward RA, Langa KM, Blaum C, Weir D. Mechanisms for racial and ethnic disparities in glycemic control in middle-aged and older Americans in the health and retirement study. Arch Intern Med 2007;167:1853–1860 [DOI] [PubMed] [Google Scholar]
- 24.Nathan DM, Kuenen J, Borg R, Zheng H, Schoenfeld D, Heine RJ, A1c-Derived Average Glucose Study Group Translating the A1C assay into estimated average glucose values. Diabetes Care 2008;31:1473–1478 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Cramer JA, Pugh MJ. The influence of insulin use on glycemic control: how well do adults follow prescriptions for insulin? Diabetes Care 2005;28:78–83 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.