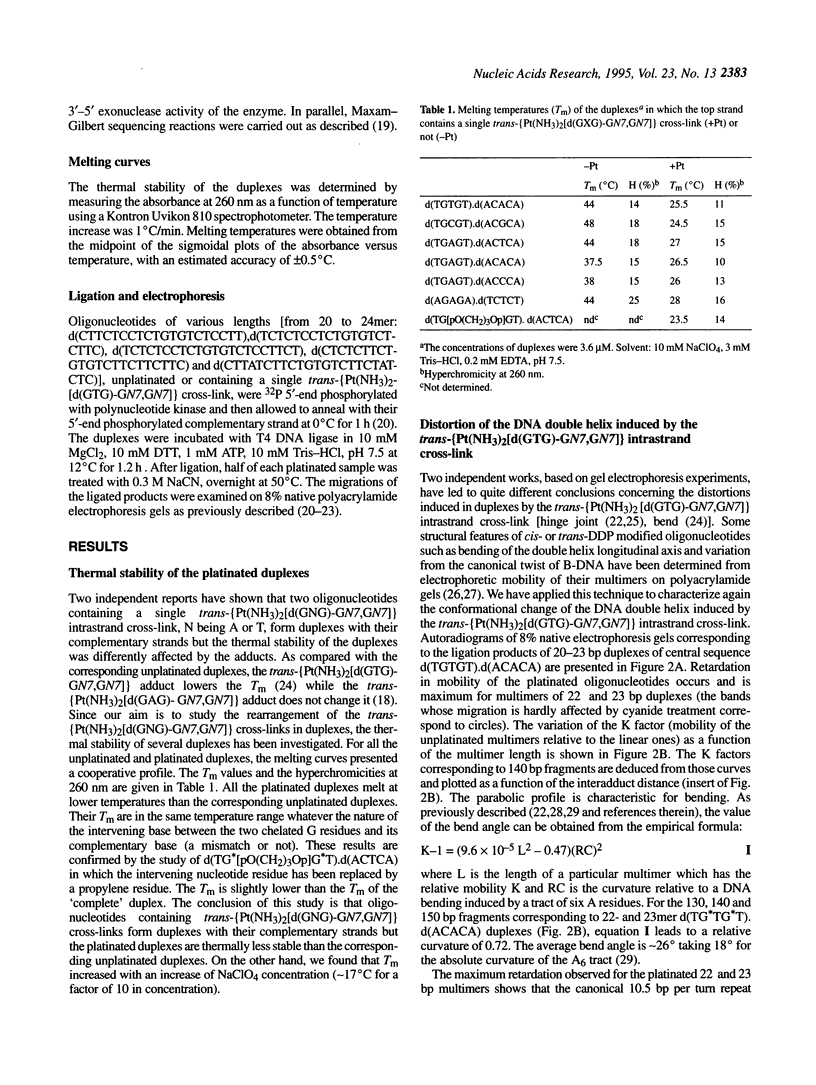
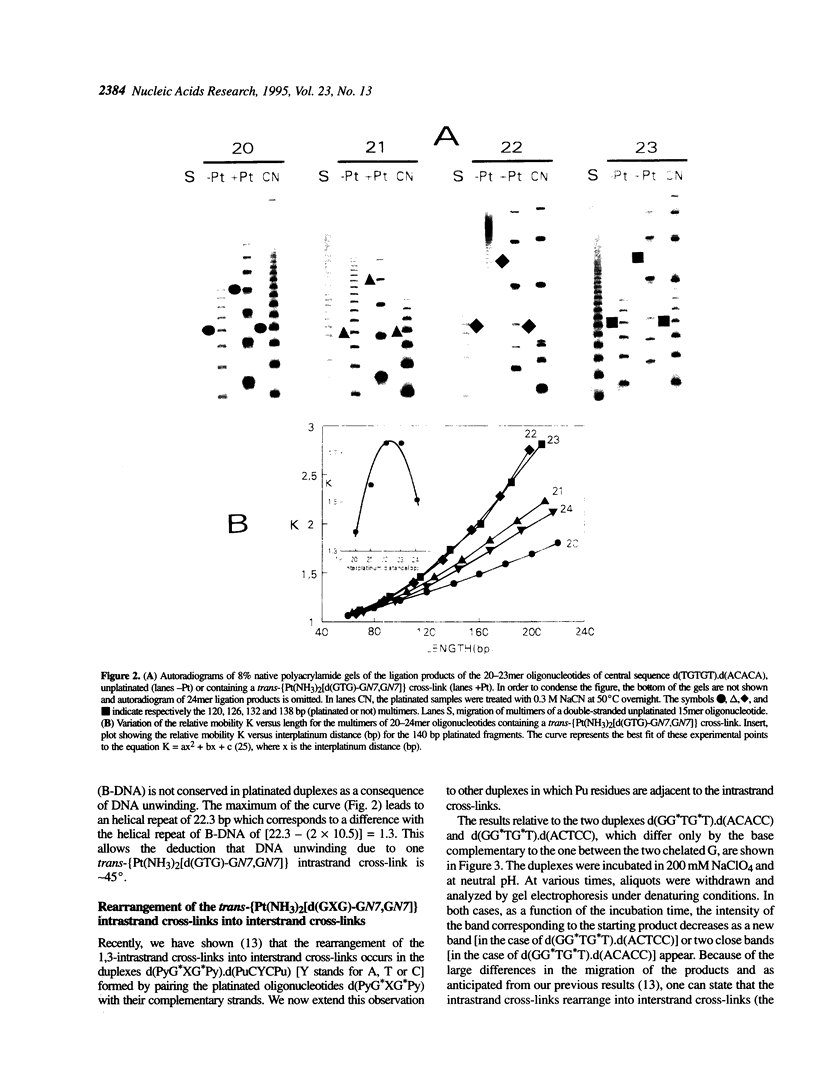
Abstract
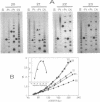
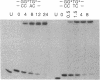
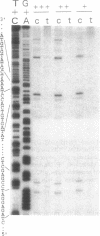
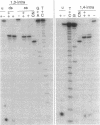
The reaction between trans-diamminedichloroplatinum(II) and single-stranded oligonucleotides containing the sequence d(GXG) (X being an adenine, cytosine or thymine residue) yields trans-[Pt(NH3)2[(GXG)-GN7,GN7]] intrastrand cross-links. These cross-links do not prevent the pairing of the platinated oligonucleotides with their complementary strands but they decrease the thermal stability of the duplexes. The thermal stability is not much affected by the chemical nature of the X residue and its complementary base. By gel electrophoresis, it is shown that the trans- [Pt(NH3)2[d(GTG)-GN7,GN7]] cross-link bends the DNA double helix (26 degrees) and unwinds it (45 degrees). The pairing of the platinated oligonucleotides with their complementary strands promotes the rearrangement of the 1,3-intrastrand cross-links into interstrand cross-links. At a given temperature, the nature of the X residue, its complementary base and of the base pairs adjacent to the adducts do not dramatically affect the rate of the reaction. To know whether trans-[Pt(NH3)2[d(GXG)-GN7,GN7]] cross-links do not rearrange in some sequences, the location of these adducts was searched in double-stranded DNA after reaction with trans-diamminedichloroplatinum(II) by means of the 3'-5' exonuclease activity of T4 DNA polymerase. At low level of platination, trans-[Pt(NH3)2[d(GXG)-GN7,GN7]] cross-links were not detected. Monofunctional adducts and interstrand cross-links were mainly formed. These results are discussed in relation with the clinical inefficiency of trans-diamminedichloroplatinum(II).
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Albert A. C., Roman A. M., Bouche G., Leng M., Rahmouni A. R. Gradual and oriented B-Z transition in 5'-untranscribed region of mouse ribosomal DNA. J Biol Chem. 1994 Jul 29;269(30):19238–19244. [PubMed] [Google Scholar]
- Anin M. F., Leng M. Distortions induced in double-stranded oligonucleotides by the binding of cis- or trans-diammine-dichloroplatinum(II) to the d(GTG) sequence. Nucleic Acids Res. 1990 Aug 11;18(15):4395–4400. doi: 10.1093/nar/18.15.4395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bellon S. F., Coleman J. H., Lippard S. J. DNA unwinding produced by site-specific intrastrand cross-links of the antitumor drug cis-diamminedichloroplatinum(II). Biochemistry. 1991 Aug 13;30(32):8026–8035. doi: 10.1021/bi00246a021. [DOI] [PubMed] [Google Scholar]
- Bellon S. F., Lippard S. J. Bending studies of DNA site-specifically modified by cisplatin, trans-diamminedichloroplatinum(II) and cis-[Pt(NH3)2(N3-cytosine)Cl]+. Biophys Chem. 1990 Apr;35(2-3):179–188. doi: 10.1016/0301-4622(90)80007-t. [DOI] [PubMed] [Google Scholar]
- Brabec V., Leng M. DNA interstrand cross-links of trans-diamminedichloroplatinum(II) are preferentially formed between guanine and complementary cytosine residues. Proc Natl Acad Sci U S A. 1993 Jun 1;90(11):5345–5349. doi: 10.1073/pnas.90.11.5345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brabec V., Síp M., Leng M. DNA conformational change produced by the site-specific interstrand cross-link of trans-diamminedichloroplatinum(II). Biochemistry. 1993 Nov 2;32(43):11676–11681. doi: 10.1021/bi00094a025. [DOI] [PubMed] [Google Scholar]
- Comess K. M., Costello C. E., Lippard S. J. Identification and characterization of a novel linkage isomerization in the reaction of trans-diamminedichloroplatinum(II) with 5'-d(TCTACGCGTTCT). Biochemistry. 1990 Feb 27;29(8):2102–2110. doi: 10.1021/bi00460a020. [DOI] [PubMed] [Google Scholar]
- Dalbiès R., Boudvillain M., Leng M. Linkage isomerization reaction of intrastrand cross-links in trans-diamminedichloroplatinum(II)-modified single-stranded oligonucleotides. Nucleic Acids Res. 1995 Mar 25;23(6):949–953. doi: 10.1093/nar/23.6.949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dalbiès R., Payet D., Leng M. DNA double helix promotes a linkage isomerization reaction in trans-diamminedichloroplatinum(II)-modified DNA. Proc Natl Acad Sci U S A. 1994 Aug 16;91(17):8147–8151. doi: 10.1073/pnas.91.17.8147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Decoville M., Schwartz A., Locker D., Leng M. Detection of minor adducts in cisplatin-modified DNA by transcription footprinting. FEBS Lett. 1993 May 24;323(1-2):55–58. doi: 10.1016/0014-5793(93)81447-8. [DOI] [PubMed] [Google Scholar]
- Eastman A., Barry M. A. Interaction of trans-diamminedichloroplatinum(II) with DNA: formation of monofunctional adducts and their reaction with glutathione. Biochemistry. 1987 Jun 16;26(12):3303–3307. doi: 10.1021/bi00386a009. [DOI] [PubMed] [Google Scholar]
- Eastman A., Jennerwein M. M., Nagel D. L. Characterization of bifunctional adducts produced in DNA by trans-diamminedichloroplatinum(II). Chem Biol Interact. 1988;67(1-2):71–80. doi: 10.1016/0009-2797(88)90087-7. [DOI] [PubMed] [Google Scholar]
- Eastman A. The formation, isolation and characterization of DNA adducts produced by anticancer platinum complexes. Pharmacol Ther. 1987;34(2):155–166. doi: 10.1016/0163-7258(87)90009-x. [DOI] [PubMed] [Google Scholar]
- Koo H. S., Crothers D. M. Calibration of DNA curvature and a unified description of sequence-directed bending. Proc Natl Acad Sci U S A. 1988 Mar;85(6):1763–1767. doi: 10.1073/pnas.85.6.1763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koo H. S., Drak J., Rice J. A., Crothers D. M. Determination of the extent of DNA bending by an adenine-thymine tract. Biochemistry. 1990 May 1;29(17):4227–4234. doi: 10.1021/bi00469a027. [DOI] [PubMed] [Google Scholar]
- Koo H. S., Wu H. M., Crothers D. M. DNA bending at adenine . thymine tracts. Nature. 1986 Apr 10;320(6062):501–506. doi: 10.1038/320501a0. [DOI] [PubMed] [Google Scholar]
- Lemaire M. A., Schwartz A., Rahmouni A. R., Leng M. Interstrand cross-links are preferentially formed at the d(GC) sites in the reaction between cis-diamminedichloroplatinum (II) and DNA. Proc Natl Acad Sci U S A. 1991 Mar 1;88(5):1982–1985. doi: 10.1073/pnas.88.5.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lepre C. A., Chassot L., Costello C. E., Lippard S. J. Synthesis and characterization of trans-[Pt(NH3)2Cl2] adducts of d(CCTCGAGTCTCC).d(GGAGACTCGAGG). Biochemistry. 1990 Jan 23;29(3):811–823. doi: 10.1021/bi00455a031. [DOI] [PubMed] [Google Scholar]
- Malinge J. M., Schwartz A., Leng M. Characterization of the ternary complexes formed in the reaction of cis-diamminedichloroplatinum (II), ethidium bromide and nucleic acids. Nucleic Acids Res. 1987 Feb 25;15(4):1779–1797. doi: 10.1093/nar/15.4.1779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marshall W. S., Caruthers M. H. Phosphorodithioate DNA as a potential therapeutic drug. Science. 1993 Mar 12;259(5101):1564–1570. doi: 10.1126/science.7681216. [DOI] [PubMed] [Google Scholar]
- Maxam A. M., Gilbert W. A new method for sequencing DNA. Proc Natl Acad Sci U S A. 1977 Feb;74(2):560–564. doi: 10.1073/pnas.74.2.560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Payet D., Gaucheron F., Sip M., Leng M. Instability of the monofunctional adducts in cis-[Pt(NH3)2(N7-N-methyl-2-diazapyrenium)Cl](2+)-modified DNA: rates of cross-linking reactions in cis-platinum-modified DNA. Nucleic Acids Res. 1993 Dec 25;21(25):5846–5851. doi: 10.1093/nar/21.25.5846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pinto A. L., Lippard S. J. Sequence-dependent termination of in vitro DNA synthesis by cis- and trans-diamminedichloroplatinum (II). Proc Natl Acad Sci U S A. 1985 Jul;82(14):4616–4619. doi: 10.1073/pnas.82.14.4616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rampino N. J., Bohr V. A. Rapid gene-specific repair of cisplatin lesions at the human DUG/DHFR locus comprising the divergent upstream gene and dihydrofolate reductase gene during early G1 phase of the cell cycle assayed by using the exonucleolytic activity of T4 DNA polymerase. Proc Natl Acad Sci U S A. 1994 Nov 8;91(23):10977–10981. doi: 10.1073/pnas.91.23.10977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rice J. A., Crothers D. M., Pinto A. L., Lippard S. J. The major adduct of the antitumor drug cis-diamminedichloroplatinum(II) with DNA bends the duplex by approximately equal to 40 degrees toward the major groove. Proc Natl Acad Sci U S A. 1988 Jun;85(12):4158–4161. doi: 10.1073/pnas.85.12.4158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zou Y., Van Houten B., Farrell N. Sequence specificity of DNA-DNA interstrand cross-link formation by cisplatin and dinuclear platinum complexes. Biochemistry. 1994 May 10;33(18):5404–5410. doi: 10.1021/bi00184a007. [DOI] [PubMed] [Google Scholar]