Abstract
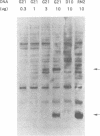
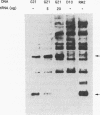
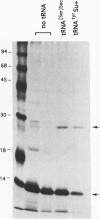
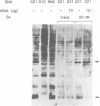
Translation of UGA as selenocysteine instead of termination occurs in numerous proteins, and the process of recording UGA requires specific signals in the corresponding mRNAs. In eukaryotes, stem-loops in the 3' untranslated region of the mRNAs confer this function. Despite the presence of these signals, selenocysteine incorporation is inefficient. To investigate the reason for this, we examined the effects of the amount of deiodinase cDNA on UGA readthrough in transfected cells, quantitating the full-length and UGA terminated products by Western blotting. The gene for the selenocysteine-specific tRNA was also cotransfected to determine if it was limiting. We find that the concentrations of both the selenoprotein DNA and the tRNA affect the ratio of selenocysteine incorporation to termination. Selenium depletion was also found to decrease readthrough. The fact that the truncated peptide is synthesized intracellularly demonstrates unequivocally that UGA can serve as both a stop and a selenocysteine codon in a single mRNA. Mutation of UGA to UAA (stop) or UUA (leucine) in the deiodinase mRNA abolishes deiodinase activity; but activity is partially restored when selenocysteine tRNAs containing complementary mutations are contransfected. Thus, UGA is not essential for selenocysteine incorporation in mammalian cells, provided that codon:anticodon complementarity is maintained.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Baker R. D., Baker S. S., LaRosa K., Whitney C., Newburger P. E. Selenium regulation of glutathione peroxidase in human hepatoma cell line Hep3B. Arch Biochem Biophys. 1993 Jul;304(1):53–57. doi: 10.1006/abbi.1993.1320. [DOI] [PubMed] [Google Scholar]
- Baron C., Heider J., Böck A. Interaction of translation factor SELB with the formate dehydrogenase H selenopolypeptide mRNA. Proc Natl Acad Sci U S A. 1993 May 1;90(9):4181–4185. doi: 10.1073/pnas.90.9.4181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berry M. J., Banu L., Chen Y. Y., Mandel S. J., Kieffer J. D., Harney J. W., Larsen P. R. Recognition of UGA as a selenocysteine codon in type I deiodinase requires sequences in the 3' untranslated region. Nature. 1991 Sep 19;353(6341):273–276. doi: 10.1038/353273a0. [DOI] [PubMed] [Google Scholar]
- Berry M. J., Banu L., Harney J. W., Larsen P. R. Functional characterization of the eukaryotic SECIS elements which direct selenocysteine insertion at UGA codons. EMBO J. 1993 Aug;12(8):3315–3322. doi: 10.1002/j.1460-2075.1993.tb06001.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berry M. J., Banu L., Larsen P. R. Type I iodothyronine deiodinase is a selenocysteine-containing enzyme. Nature. 1991 Jan 31;349(6308):438–440. doi: 10.1038/349438a0. [DOI] [PubMed] [Google Scholar]
- Berry M. J., Maia A. L., Kieffer J. D., Harney J. W., Larsen P. R. Substitution of cysteine for selenocysteine in type I iodothyronine deiodinase reduces the catalytic efficiency of the protein but enhances its translation. Endocrinology. 1992 Oct;131(4):1848–1852. doi: 10.1210/endo.131.4.1396330. [DOI] [PubMed] [Google Scholar]
- Burk R. F., Hill K. E. Regulation of selenoproteins. Annu Rev Nutr. 1993;13:65–81. doi: 10.1146/annurev.nu.13.070193.000433. [DOI] [PubMed] [Google Scholar]
- Böck A., Forchhammer K., Heider J., Baron C. Selenoprotein synthesis: an expansion of the genetic code. Trends Biochem Sci. 1991 Dec;16(12):463–467. doi: 10.1016/0968-0004(91)90180-4. [DOI] [PubMed] [Google Scholar]
- Chu F. F., Doroshow J. H., Esworthy R. S. Expression, characterization, and tissue distribution of a new cellular selenium-dependent glutathione peroxidase, GSHPx-GI. J Biol Chem. 1993 Feb 5;268(4):2571–2576. [PubMed] [Google Scholar]
- Feng Y. X., Copeland T. D., Oroszlan S., Rein A., Levin J. G. Identification of amino acids inserted during suppression of UAA and UGA termination codons at the gag-pol junction of Moloney murine leukemia virus. Proc Natl Acad Sci U S A. 1990 Nov;87(22):8860–8863. doi: 10.1073/pnas.87.22.8860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Forchhammer K., Leinfelder W., Böck A. Identification of a novel translation factor necessary for the incorporation of selenocysteine into protein. Nature. 1989 Nov 23;342(6248):453–456. doi: 10.1038/342453a0. [DOI] [PubMed] [Google Scholar]
- Gelpi C., Sontheimer E. J., Rodriguez-Sanchez J. L. Autoantibodies against a serine tRNA-protein complex implicated in cotranslational selenocysteine insertion. Proc Natl Acad Sci U S A. 1992 Oct 15;89(20):9739–9743. doi: 10.1073/pnas.89.20.9739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gossen M., Bujard H. Tight control of gene expression in mammalian cells by tetracycline-responsive promoters. Proc Natl Acad Sci U S A. 1992 Jun 15;89(12):5547–5551. doi: 10.1073/pnas.89.12.5547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hatfield D. L., Lee B. J., Price N. M., Stadtman T. C. Selenocysteyl-tRNA occurs in the diatom Thalassiosira and in the ciliate Tetrahymena. Mol Microbiol. 1991 May;5(5):1183–1186. doi: 10.1111/j.1365-2958.1991.tb01891.x. [DOI] [PubMed] [Google Scholar]
- Hatfield D., Choi I. S., Mischke S., Owens L. D. Selenocysteyl-tRNAs recognize UGA in Beta vulgaris, a higher plant, and in Gliocladium virens, a filamentous fungus. Biochem Biophys Res Commun. 1992 Apr 15;184(1):254–259. doi: 10.1016/0006-291x(92)91186-t. [DOI] [PubMed] [Google Scholar]
- Hatfield D., Diamond A. UGA: a split personality in the universal genetic code. Trends Genet. 1993 Mar;9(3):69–70. doi: 10.1016/0168-9525(93)90215-4. [DOI] [PubMed] [Google Scholar]
- Heider J., Baron C., Böck A. Coding from a distance: dissection of the mRNA determinants required for the incorporation of selenocysteine into protein. EMBO J. 1992 Oct;11(10):3759–3766. doi: 10.1002/j.1460-2075.1992.tb05461.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heider J., Leinfelder W., Böck A. Occurrence and functional compatibility within Enterobacteriaceae of a tRNA species which inserts selenocysteine into protein. Nucleic Acids Res. 1989 Apr 11;17(7):2529–2540. doi: 10.1093/nar/17.7.2529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karimpour I., Cutler M., Shih D., Smith J., Kleene K. C. Sequence of the gene encoding the mitochondrial capsule selenoprotein of mouse sperm: identification of three in-phase TGA selenocysteine codons. DNA Cell Biol. 1992 Nov;11(9):693–699. doi: 10.1089/dna.1992.11.693. [DOI] [PubMed] [Google Scholar]
- Konecki D. S., Aune K. C., Tate W., Caskey C. T. Characterization of reticulocyte release factor. J Biol Chem. 1977 Jul 10;252(13):4514–4520. [PubMed] [Google Scholar]
- Lee B. J., Kang S. G., Hatfield D. Transcription of Xenopus selenocysteine tRNA Ser (formerly designated opal suppressor phosphoserine tRNA) gene is directed by multiple 5'-extragenic regulatory elements. J Biol Chem. 1989 Jun 5;264(16):9696–9702. [PubMed] [Google Scholar]
- Lee B. J., Rajagopalan M., Kim Y. S., You K. H., Jacobson K. B., Hatfield D. Selenocysteine tRNA[Ser]Sec gene is ubiquitous within the animal kingdom. Mol Cell Biol. 1990 May;10(5):1940–1949. doi: 10.1128/mcb.10.5.1940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stadtman T. C. Biosynthesis and function of selenocysteine-containing enzymes. J Biol Chem. 1991 Sep 5;266(25):16257–16260. [PubMed] [Google Scholar]
- Vallette F., Mege E., Reiss A., Adesnik M. Construction of mutant and chimeric genes using the polymerase chain reaction. Nucleic Acids Res. 1989 Jan 25;17(2):723–733. doi: 10.1093/nar/17.2.723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vendeland S. C., Beilstein M. A., Chen C. L., Jensen O. N., Barofsky E., Whanger P. D. Purification and properties of selenoprotein W from rat muscle. J Biol Chem. 1993 Aug 15;268(23):17103–17107. [PubMed] [Google Scholar]
- Zinoni F., Heider J., Böck A. Features of the formate dehydrogenase mRNA necessary for decoding of the UGA codon as selenocysteine. Proc Natl Acad Sci U S A. 1990 Jun;87(12):4660–4664. doi: 10.1073/pnas.87.12.4660. [DOI] [PMC free article] [PubMed] [Google Scholar]