Table of Contents
| Author List | 1143 |
| Preface | 1145 |
| Executive Summary | 1146 |
| Background to HCT | 1147 |
| Hematopoietic Cell Graft Safety | 1153 |
| Bacterial Infections | 1156 |
| Viral Infections | 1159 |
| Fungal Infections | 1170 |
| Regionally Limited or Rare Infections | 1176 |
| Infection Control and Prevention in Health care Facilities | 1182 |
| Safe Living after Transplantation | 1195 |
| Vaccinations | 1203 |
| References | 1207 |
| Appendices | 1229 |
Executive Committee
Marcie Tomblyn, University of Minnesota, Minneapolis, MN
Tom Chiller, Centers for Disease Control and Prevention, Atlanta, GA
Hermann Einsele, Universitatsklinik Wurzburg Medizinische Klinik und Poliklinik II, Wurzburg, Germany
Ronald Gress, National Institutes of Health, Bethesda, MD
Kent Sepkowitz, Memorial Sloan Kettering Cancer Center, New York, NY
Jan Storek, University of Calgary, Calgary, Alberta, Canada
John R Wingard, University of Florida, Gainesville, FL
Jo-Anne H Young, University of Minnesota, Minneapolis, MN
Michael A Boeckh, University of Washington Fred Hutchinson Cancer Research Center, Seattle, WA
Introduction/Background
Crystal Mackall, National Institutes of Health, Bethesda, MD
Terry Fry, National Institutes of Health, Bethesda, MD
Ronald Gress, National Institutes of Health, Bethesda, MD
Karl Peggs, University College London Hospital, London, United Kingdom
Jan Storek, University of Calgary, Calgary, Alberta, Canada
Antoine Toubert, University Paris Diderot, Hôpital Saint-Louis, Paris, France
Hematopoietic Graft Cell Safety
Dennis Confer, National Marrow Donor Program, Minneapolis, MN
Ronald Gress, National Institutes of Health, Bethesda, MD
Marcie Tomblyn, University of Minnesota, Minneapolis, MN
Gerhard Ehninger, Universitatsklinikum Dresden, Dresden, Germany
BacterialInfection
Dan Engelhard, Hadassah Medical Center, Jerusalem, Israel
Murat Akova, Hacettepe University School of Medicine, Ankara, Turkey
Michael A Boeckh, University of Washington Fred Hutchinson Cancer Research Center, Seattle, WA
Alison Freifeld, Nebraska Medical Center, Omaha, NE
Kent Sepkowitz, Memorial Sloan Kettering Cancer Center, New York, NY
Claudio Viscoli, Ospedale San Martino, Genoa, Italy
James Wade, Medical College of Wisconsin, Milwaukee, WI
Issam Raad, MD Anderson Cancer Center, Houston, TX
Viral Infection
John Zaia, City of Hope, Duarte, CA
Lindsey Baden, Brigham and Women's Hospital, Boston, MA
Michael A Boeckh, University of Washington Fred Hutchinson Cancer Research Center, Seattle, WA
Suparno Chakrabarti, St. George's Hospital, London, United Kingdom
Hermann Einsele, Universitatsklinik Wurzburg Medizinische Klinik und Poliklinik II, Wurzburg, Germany
Per Ljungman, Karolinska University Hospital, Stockholm, Sweden
George McDonald, University of Washington, Seattle, WA
Hans H. Hirsch, University Hospital, Basel, Switzerland
Fungal
Kieren Marr, Johns Hopkins University, Baltimore MD
Eric Bow, University of Manitoba, Winnipeg, Manitoba, Canada
Tom Chiller, Centers for Disease Control and Prevention, Atlanta, GA
Georg Maschmeyer, Center for Hematology, Oncology and Radiotherapy Klinikum Ernst von Bergmann Charlottenstr, Potsdam, Germany
Patricia Ribaud, MD, Hôpital Saint-Louis, Paris, France
Brahm H. Segal, Roswell Park Cancer Institute, Buffalo, NY
William J. Steinbach, Duke University Medical Center, Durham, NC
John R. Wingard, University of Florida, Gainesville, FL
Marcio Nucci, Universidade Federal do Rio de Janeiro, Rio de Janeiro, Brazil
Regionally Limited/Rare Infections
Juan Gea-Banacloche, National Institutes of Health, Bethesda, MD
Henry Masur, National Institutes of Health, Bethesda, MD
Clovis Arns da Cuhna, Universidade Federal do Parana, Curitiba, Brazil
Tom Chiller, Centers for Disease Control and Prevention, Atlanta, GA
Louis Kirchoff, University of Iowa, Iowa City, IA
Peter Shaw, Children's Hospital at Westmead, Sydney, Australia
Marcie Tomblyn, University of Minnesota, Minneapolis, MN
Catherine Cordonnier, Hôpital Henri Mondor, Creteil, France
Infection Prevention and Control
Deborah S. Yokoe, Brigham & Women's Hospital and Dana-Farber Cancer Institute, Boston, MA
Corey Casper, University of Washinton Fred Hutchinson Cancer Research Center, Seattle, WA
Erik R. Dubberke, Washington University School of Medicine, St. Louis, MO
Grace M. Lee, Children's Hospital Boston, Boston, MA
Patricia Muñoz, Hospital General Universitario Gregorio Marañón, University of Madrid, Spain
Tara Palmore, National Institutes of Health, Bethesda, MD
Kent Sepkowitz, Memorial Sloan Kettering Cancer Center, New York, NY
Jo-Anne H Young, University of Minnesota, Minneapolis, MN
J Peter Donnelly, Radboud University Nijmegen Medical Centre, Nijmegen, The Netherlands
Safe Living After HCT
Deborah S. Yokoe, Brigham & Women's Hospital and Dana-Farber Cancer Institute, Boston, MA
Corey Casper, University of Washinton Fred Hutchinson Cancer Research Center, Seattle, WA
Erik R. Dubberke, Washington University School of Medicine, St. Louis, MO
Grace M. Lee, Children's Hospital Boston, Boston, MA
Patricia Muñóz, Hospital General Universitario Gregorio Marañón, University of Madrid, Spain
Tara Palmore, National Institutes of Health, Bethesda, MD
Kent Sepkowitz, Memorial Sloan Kettering Cancer Center, New York, NY
Jo-Anne H Young, University of Minnesota, Minneapolis, MN
J Peter Donnelly, Radboud University Nijmegen Medical Centre, Nijmegen, The Netherlands
Vaccinations
Per Ljungman, Karolinska University Hospital, Stockholm, Sweden
Catherine Cordonnier, Hôpital Henri Mondor, Creteil, France
Hermann Einsele, Universitatsklinik Wurzburg Medizinische Klinik und Poliklinik II, Wurzburg, Germany
Janet Englund, University of Washington/Seattle Children's Hospital and Regional Medical Center, Seattle, WA
Clarisse Martins Machado, Universidade de São Paulo, São Paulo, Brazil
Jan Storek, University of Calgary, Calgary, Alberta
Trudy Small, Memorial Sloan Kettering Cancer Center, New York, NY
Preface
This report, cosponsored by the Center for International Blood and Marrow Transplant Research (CIBMTR), National Marrow Donor Program (NMDP), European Blood and Marrow Transplant Group (EBMT), American Society for Blood and Marrow Transplant (ASBMT), Canadian Blood and Marrow Transplant Group (CBMTG), Infectious Diseases Society of America (IDSA), Society for Healthcare Epidemiology of America (SHEA), Association of Medical Microbiology and Infectious Diseases (AMMI), the Center for Disease Control and Prevention (CDC), and the Health Resources and Services Administration, represents an update of the guidelines published in 2000 for preventing infections among hematopoietic cell transplantion (HCT) recipients [1]. An international group of experts in infectious diseases, HCT, and public health worked together to compile this document with 4 goals in mind: (1) to summarize the current available data in the field, (2) to provide evidence-based recommendations regarding prevention of infectious complications among HCT patients, (3) to serve as a reference for health care providers worldwide who care for HCT recipients, and (4) to serve as a reference for HCT recipients and their nonmedical caregivers. In updating these guidelines, the committee sought to summarize the currently available data and present them as concisely as possible in an evidence-based fashion.
Significant changes in the field of HCT since the publication of the original guidelines necessitated this update. These changes include new antimicrobial agents, broader use of reduced-intensity conditioning (RIC), the increasing age of HCT recipients, and more frequent use of alternative donor stem cell sources such as haploidentical donors and umbilical cord blood. Furthermore, as with any field of medicine, published studies continue to add to the evidence regarding supportive medical care. Despite—or perhaps because of—these changes, infections still occur with increased frequency or severity among HCT recipients as a patient population.
In presenting these guidelines, the committee is not intending to dictate standards of practice. Although considerable effort has gone into ensuring that the guidelines have a global perspective based on the currently available medical evidence, adherence to a particular recommendation may be inconsistent with national or regional guidelines, the availability of specific procedures or medications, or local epidemiological conditions. Individual clinicians may follow practice patterns that, although deviating from these recommendations, are nevertheless effective and sound.
Using These Guidelines
For the purposes of this report, HCT is defined as transplantation of any blood- or marrow-derived hematopoietic stem cells (HSCs), regardless of transplant type (ie, allogeneic or autologous) or cell source (ie, bone marrow [BM], peripheral blood [PB], or umbilical cord blood [UCB]). The definition of immune competence following transplant is loosely defined by the ability of the HCT recipient to receive live vaccine following recovery from transplant. Conventionally, this is thought to occur at approximately 24 months following HCT in patients who are not receiving immunosuppressive therapy and do not have active graft-versus-host disease (GVHD) [1]. For patients with on-going GVHD or continued use of immunosuppressive therapy, it is recommended to consider the patient as immune deficient and still at risk for significant infectious complications.
Unless otherwise noted, the recommendations presented in this report address allogeneic and autologous and pediatric and adult HCT recipients. These recommendations are intended for use by the recipients, their household and other close contacts, transplant and infectious diseases specialists, HCT center personnel, and public health professionals. For most recommendations, prevention strategies are rated by the strength of the recommendation and the quality of the evidence supporting the recommendation (Table 1 ). The principles of this rating system were developed by the IDSA and the U.S. Public Health Service for use in guidelines for preventing opportunistic infections among HIV-infected persons [2]. This rating system allows assessments of the strength of recommendations.
Table 1.
Evidence-Based Rating System Used in the Hematopoietic Cell Transplantation (HCT) Guidelines [2]
| Strength of Recommendation | |
|---|---|
| Category | Definition |
| A | Both strong evidence for efficacy and substantial clinical benefit support recommendation for use. Should always be offered. |
| B | Moderate evidence for efficacy—or strong evidence for efficacy, but only limited clinical benefit—supports recommendation for use. Should generally be offered. |
| C | Evidence for efficacy is insufficient to support a recommendation for or against use, or evidence for efficacy might not outweigh adverse consequences (eg, drug toxicity, drug interactions), or cost of the chemoprophylaxis or alternative approaches. Optional. |
| D | Moderate evidence for lack of efficacy or for adverse outcome supports a recommendation against use. Should generally not be offered. |
| E | Good evidence for lack of efficacy or for adverse outcome supports a recommendation against use. Should never be offered. |
| Quality of Evidence Supporting the Recommendation | |
|---|---|
| Category | Definition |
| I | Evidence from at least one well-executed randomized, controlled trial |
| II | Evidence from at least one well-designed clinical trial without randomization; cohort or case-controlled analytic studies (preferably from more than one center); multiple time-series studies; or dramatic results from uncontrolled experiments |
| III | Evidence from opinions of respected authorities based on clinical experience, descriptive studies, or reports of expert committees |
Executive Summary
In the past decade, modifications in HCT management and supportive care have resulted in changes in recommendations for the prevention of infection in HCT patients. These changes are fueled by new antimicrobial agents, increased knowledge of immune reconstitution, and expanded conditioning regimens and patient populations eligible for HCT. Despite these advances, infection is reported as the primary cause of death in 8% of autologous HCT patients and 17% to 20% of allogeneic HCT recipients [3]. The major changes in this document, including changes in recommendation ratings, are summarized here.
The organization of this document is similar to the previous guidelines. Specifically, the prevention of exposure and disease among pediatric and adult autologous and allogeneic HCT recipients is discussed. The current recommendations consider myeloablative (MA) conditioning and RIC for allogeneic HCT similarly, because data on infectious complications following RIC compared to MA conditioning are sparse 4, 5, 6, 7. However, increased information regarding posttransplant immune recovery highlighting differences between MA and RIC HCT are included.
The sections of the document have been rearranged in an attempt to follow the time course of potential infectious risks for patients receiving HCT. Following the background section, information on hematopoietic cell product safety is provided. The subsequent sections discuss prevention of infection by specific microorganisms. Following organism-specific information, the sections then discuss means of preventing nosocomial infections as well as “dos and don'ts” for patients following discharge posttransplant. Finally, information on vaccinations is provided. This will hopefully allow the reader to follow the prevention practices needed from the time a donor is selected until the patient regains immune competence.
Several topics are new or expanded from the prior document (Table 2 ). These include information on multiple organisms that were previously not discussed, but have seemingly become more clinically relevant in HCT patients over the past decade. Data, and where possible, recommendations, are provided regarding the following organisms that were not included in the previous document: Bordetella pertussis; the polyomaviruses BK and JC; hepatitis A, B, and C viruses (HAV, HBV, HCV); human herpesviruses (HHV) 6, 7, and 8; human metapneumovirus; human immunodeficiency virus (HIV); tuberculosis; nocardiosis; malaria; and leishmaniasis. In recognition of our global society, several organisms are discussed that may be limited to certain regions of the world. Included in that section are also those infections that may be ubiquitous but occur infrequently, such as Pneumocystis jiroveci and Nocardia.
Table 2.
Summary of Changes Compared to the Guidelines Published in 2000 [1]
| Major Changes | Starting Page |
|---|---|
| Updated background on immune recovery following HCT including differences based on conditioning regimen and stem cell sources | 7 |
| Changes to the Bacterial Section | |
| 1) Quinolone prophylaxis is recommended for patients with neutropenia expected to last ≥7 days (BI) | 14 |
| 2) Added recommendations regarding Central Line-Associated Bloodstream Infections (CLABSI) (in addition to the section in Infection Prevetion and Control) | 15 |
|
15 |
| Changes to the Fungal Section | |
| 1) Micafungin is an alternative for prevention of candidiasis during preengraftment (BI) | 32 |
| 2) Voriconazole and Posaconazole may be used for prevention of candidiasis postengraftment (BI) | 33 |
| 3) Itraconazole oral solution as prevention of mold infections (BI—prior, no data) | 34 |
| 4) Posaconazole for prevention of mold infections in patients with GVHD (BI) | 34 |
| PCR screening for Toxoplama gondii can be considered in high-risk patients when unable to tolerate prophylaxis (BII) | 37 |
| Changes in Vaccination Recommendations | |
| 1) Pneumococcal Vaccine: Use PCV vaccine and start 3-6 months post-HCT | 63 |
| 2) Optional to use acellular pertussis vaccine in all patients | 64 |
| 3) Varicella vaccine (Varivax) is optional. Zostavax is contraindicated | 64 |
| 4) Vaccinations with inactivated vaccines may be started as early as 6 months post-HCT (and earlier for PCV and influenza) | 31 |
| 5) Information regarding use of HPV vaccine | 32 |
| Sections added to the Infection Prevention and Control Section | |
| 1) Recommendations regarding multiply drug-resistant Gram-negative bacilli | 49 |
| 2) Recommendations regarding adenovirus | 52 |
| 3) Recommendation regarding viral gastroenteritis | 52 |
|
55 |
| Appendix 1 (Dosing) changes | |
| 1) Alternative CMV prophylaxis/treatment: Foscarnet now AI (prior CIII) and added Valganciclovir and Cidofovir | 89 |
| 2) EBV prophylaxis/treatment with Rituximab | 89 |
| 3) VZV: added alternatives to VZIG for exposure and new information on prophylaxis | 90 |
| 4) Influenza: added dosing information for Oseltamivir and Zanamivir | 91 |
| 5) RSV: Added dosing information | 91 |
| 6) Split the fungal section into data for standard-risk and high-risk patients | 92 |
| 7) Added dosing information for Micafungin, Posaconazole, and Voriconazole | 92 |
| 8) Alternative PCP prophylaxis: Added atovaquone and changed aerosolized Pentamidine to CII (prior CIII) | 92 |
| New Organisms | |
| Bordatella pertussis | 16 |
| Human Metapneumovirus | 23 |
| Polyomaviruses BK and JC | 24 |
| Hepatitis A | 25 |
| Hepatitis B | 25 |
| Hepatitis C | 26 |
| Human Herpes Virus 6 and 7 | 27 |
| Human Herpes Virus 8 | 28 |
| Human Immunodeficiency Virus | 28 |
| Mycobacterium tuberculosis | 34 |
| Nocardia | 38 |
| Leishmania | 39 |
| Malaria | 39 |
GVHD indicates graft-versus-host disease; HCT, hematopoietic cell transplantation; CVM, cytomegalovirus; EBV, Epstein-Barr virus; VZV, varicella zoster virus; PCP, Pneumocystis jiroveci pneumonia; RSV, respiratory syncytial virus; VZIG, varicella-zoster immunoglobulin.
Several other changes should be noted. For bacterial infections, these guidelines now recommend quinolone prophylaxis for patients with neutropenia expected to last as least 7 days (BI). Additionally, the recommendations for contact precautions (AIII), vaccination (BI), and prophylaxis for patients with GVHD (AIII) against Streptococcus pneumoniae have been strengthened. The subsection on central line-associated blood stream infections is now in the bacterial section. The vaccination section has been dramatically expanded. Changes include the recommendations for pneumococcal conjugate vaccine (PCV) rather than polysaccharide vaccine (PPSV-23) for pneumococcal vaccination, starting some vaccinations earlier posttransplant, and the addition of recommendations for Varivax, HPV vaccine, and (the nonuse of) Zostavax vaccine are included. Two additional appendices were added to provide information on desensitization to sulfa drugs and visitor screening questionnaires. Finally, the dosing appendix has merged both adult and pediatric dosing, and provides recommendations for several newer antimicrobial agents that were not previously available.
In summary, the changes and expansion to this document reflect the growing body of literature detailing infectious complications in HCT patients.
Background to HCT, Including Posttransplant Immune Recovery
C. Mackall, T. Fry, R. Gress, K. Peggs, J. Storek, A. Toubert
Hematopoietic cell transplantation (HCT) can be defined as the transfer of hematopoietic stem cells (HSCs) from 1 individual to another (allogeneic HCT) or the return of previously harvested cells to the same individual (autologous HCT) after manipulation of the cells and/or the recipient. The goal of HCT is lifelong engraftment of the administered cells, resulting in some or all of the recipient's lymphohematopoietic system being derived from the HCT graft. Full donor engraftment occurs when the recipient lymphohematopoietic system is fully replaced by progeny of the HCT graft. This is the ultimate goal of many HCT protocols, especially for achieving optimal graft-versus-tumor (GVT) activity in patients with malignant disease 8, 9, 10. In some clinical settings, however, a state of “mixed chimerism,” wherein elements of both the donor and recipient lymphohematopoietic system survive, may be sufficient to cure the underlying condition 11, 12.
Allogeneic HCT can cure or improve outcome in a wide variety of diseases, including leukemia, lymphoma, myeloproliferative disorders, myelodysplastic syndrome (MDS), bone marrow (BM) failure syndromes, congenital immunodeficiencies, enzyme deficiencies, and hemoglobinopathies 13, 14, 15, 16, 17. However, because allogeneic HCT is associated with significant morbidity and mortality because of regimen-related toxicity (RRT) [18], infection [19], and graft versus host disease (GVHD) [20], a recommendation regarding transplantation for the individual patient requires careful risk assessment that takes into account disease status [21], comorbidities, previous therapies, other standard therapies available for the underlying disease [22], donor stem cell source [23], and histoincompatibility [24]. Autologous HCT can improve outcomes in neoplastic diseases [25] and autoimmune conditions [26], and continues to be investigated as a platform for gene therapy [27]. RRT and infections contribute to the morbidity and mortality associated with autologous HCT; however, morbidity because of GVHD generally does not occur after this procedure.
Substantial progress has been made in allogeneic HCT during the past 15 years as a result of improvements in 4 distinct, but interrelated, areas. First, improvements in the supportive care of patients with severe immunosuppression and myelosuppression have diminished morbidity and mortality because of infection 19, 28, 29. Second, the critical contribution of immune-mediated GVT effects toward eradicating malignant disease and facilitating engraftment is now well established, and factors prominently in the design of individual HCT regimens [21]. Third, alternative donor (nonsibling) transplants and new stem cell sources now provide HCT options for a larger percentage of potential candidates 23, 30, 31. Fourth, newer, less toxic preparative regimens have been developed that allow reliable engraftment with a lower risk for treatment-related mortality (TRM) and long-term adverse effects 18, 32.
Classically, transfer of the hematopoietic cell graft was seen as a means to rescue the recipient's lymphohematopoietic system from an otherwise lethal myeloablative (MA) preparative regimen. In this model, the preparative regimen was used as the primary tool to eradicate malignant disease, or to eradicate the recipient's immune system when HCT was used to treat benign diseases. However, careful clinical studies over the last 4 decades have revealed that the effectiveness of allogeneic HCT in eradicating malignant disease is intimately linked to the activity of immunoreactive cells in the graft, most notably T cells and, in some cases, natural killer (NK) cells 24, 33, 34. Indirect demonstration of this graft-versus-leukemia (GVL) effect comes from reports of increased leukemic relapse following syngeneic (identical twin) HCT and increased leukemia relapse following T cell-depleted HCT. Direct demonstration has been provided by the ability of donor lymphocyte infusions (DLIs) to induce remission in substantial numbers of patients with chronic myelogenous leukemia (CML) [35]. Evidence for a GVT effect also exists for other malignancies [36], but the effect appears to be less potent than that observed in CML. Furthermore, even when HCT is performed for benign diseases, the rate and degree of donor engraftment can be enhanced with the use of donor leukocyte infusions, demonstrating that immune cells also contribute to the engraftment process. Thus, HCT has evolved from a therapy designed primarily to provide lymphohematopoietic rescue after MA conditioning to a form of immunotherapy wherein mature immune cells contained within the graft and/or reconstituted from donor progenitors play an active role in eradicating the underlying neoplastic disease and in facilitating donor engraftment.
Expansion of graft sources has contributed to substantial progress in allogeneic HCT. Traditionally, allogeneic HCT involved transfer of marrow grafts harvested from HLA-matched sibling donors, in which case histoincompatibility was limited to minor antigens, for which reliable typing is not routinely available. Approximately 25% to 30% of potential HCT recipients will have a matched sibling donor available. Through the efforts of the NMDP in the United States and donor registries throughout the world, approximately 12 million individuals have undergone preliminary HLA typing over the last 20 years, and as a result, 75% of Caucasian individuals will find a suitably matched unrelated donor (URD). For other racial or ethnic groups, however, the chance of finding a suitable donor using existent registries is substantially less. Recent studies have demonstrated that with proper HLA matching, outcomes following matched URD HCT approach those reported for matched sibling transplants. Umbilical cord blood (UCB) transplants have also shown promise. The benefits of UCB as a stem cell source are ready accessibility and the ability to cross HLA barriers 23, 31, 37, 38. Mismatched related and haploidentical donor grafts also continue to have a role in clinical HCT, especially for patients with congenital immunodeficiency [39] and in specialized centers where this approach continues to be optimized 40, 41. Beyond the multitude of choices regarding donor source, progress during the last 15 years has also demonstrated that granulocyte colony-stimulating factor (G-CSF)-mobilized peripheral blood stem cell (PBSC) allografts provide a reliable source for engraftment, and offer the advantage of improved myelogenous and T cell recovery 30, 42 than with traditional marrow grafts and thus fewer infections. However, G-CSF-mobilized blood grafts also appear to carry a greater risk for chronic GVHD (cGVHD) compared with marrow grafts 23, 43, 44.
New approaches have been developed to minimize the likelihood of graft failure, conditioning toxicity, GVHD, and infections 45, 46, 47, 48. The addition of T cell-depleting agents (eg, antithymocyte globulin [ATG] or alemtuzumab) to conditioning regimens has been associated with a reduced incidence of GVHD and diminished graft rejection, but may delay immune recovery. Nonmyeloablative (NMA) preparative regimens that use cytotoxic drugs or low-dose total body irradiation (TBI) in conditioning regimens have been associated with reduced nonrelapse mortality (NRM), and have provided new options for HCT among the elderly and in patients with substantial comorbidities. The choice of preparative regimen and the decision regarding the relative merit of an MA versus an NMA regimen is complex and should involve a risk-adapted strategy that takes into account the underlying disease, patient age, comorbidities, stem cell source, histoincompatibility, and other relevant factors.
In summary, HCT plays a central role in the treatment of a variety of benign and malignant diseases and the field continues to evolve rapidly, with new options for donor sources and preparative regimens. At the same time, standard treatments for many of the diseases wherein HCT historically provided the mainstay of therapy have also evolved, rendering the decision of whether, when, and how to proceed to HCT highly complex and requiring careful consideration of the individual case in light of evidence-based data. For patients who undergo HCT, the major causes of early morbidity and mortality are disease relapse, acute GVHD (aGVHD), infection, RRT, and graft failure. Long-term survivors of HCT are at risk for a variety of long-term adverse effects, including cGVHD, infections, hypothyroidism, sterility, growth failure and other endocrine disturbances, cataracts, avascular necrosis, disease relapse, and second malignancy. The incidence of each of those adverse effects varies greatly with differing preparative regimens, comorbidities, age at transplantation, and whether the individual experiences cGVHD [49].
Immune System Recovery following HCT
Following MA conditioning, HCT recipients typically experience a period of profound pancytopenia spanning days to weeks depending upon the donor source. The rapidity of neutrophil recovery varies with the type of graft: approximate recovery time is 2 weeks with G-CSF-mobilized PBSC grafts, 3 weeks with marrow grafts, and 4 weeks with UCB grafts. Neutrophil, monocyte, and NK-cell recovery is followed by platelet and red cell recovery, which is followed by B and T cell recovery (Figure 1 ). Simultaneously, MA regimens damage mucosal surfaces and thereby provide a source for bloodstream seeding of commensal pathogens that inhabit the gastrointestinal tract. As a result, infectious complications in the immediate posttransplant period usually present as febrile neutropenia, with the severity of risk related to the depth and duration of neutropenia and the degree of mucosal damage induced.
Figure 1.
Approximate immune cell counts (expressed as percentage of normal counts) peri- and post-MA HCT. Nadirs are higher and occur later after NMA than MA transplantation, as recipient cells persist after NMA transplant for several weeks to months (in the presence of GVHD) or longer (in the absence of GVHD). The orange line represents the innate immune cells (eg, neutrophils, monocytes, and natural killer [NK] cells), the recovery of which is influenced by the graft type (fastest with filgrastim-mobilized blood stem cells, intermediate with marrow, and slowest with UCB). The green line represents the recovery of CD8+ T cells and B cells, the counts of which may transiently become supranormal. B cell recovery is influenced by graft type (fastest after CB transplant), and is delayed by GVHD and/or its treatment. The blue line represents the recovery of relatively radiotherapy/chemotherapy-resistant cells such as plasma cells, tissue dendritic cells (eg, Langerhans cells) and, perhaps, tissue macrophages/microglia. The nadir of these cells may be lower in patients with aGVHD because of graft-versus-host plasma cell/Langerhans cell effect. The red line represents CD4+ T cells, the recovery of which is influenced primarily by T cell content of the graft and patient age (faster in children than adults). From Storek J. Immunological reconstitution after hematopoietic cell transplantation—its relation to the contents of the graft. Expert Opin Biol Ther (Informa). 2008;8:583-597.
Recipients of NMA allogeneic transplants exhibit substantial heterogeneity in the depth and duration of pancytopenia, with some regimens accomplishing reliable engraftment without clinically significant myelosuppression. In regimens with minimal myelosuppression and minimal mucosal toxicity, the risk for infection in the immediate posttransplant period is reduced. In fact, regimens based on low-dose TBI and fludarabine (Flu) can sometimes be performed in the outpatient setting, with a virtual elimination of neutropenic complications.
Although the degree of myelosuppression is milder following NMA regimens, the depth and extent of lymphodepletion tends to be similar, with prolonged periods of immune incompetence observed in recipients of both MA and NMA regimens. This is because engraftment of allogeneic hematopoietic progenitor cells requires significant recipient immunosuppression to prevent graft rejection, even in the context of full HLA matching. With some regimens, essentially complete eradication of recipient lymphocytes is accomplished by the preparative regimen itself. However, with other regimens, depletion of recipient lymphocytes occurs more gradually via the use of donor leukocyte infusions following transplant. In both cases, the vast majority of HCT recipients experience near-total lymphocyte depletion, and thus must undergo lymphoid reconstitution via mature lymphocytes and lymphoid progenitors contained in the graft. Furthermore, except when T cell-depleted HCT grafts are administered, all allogeneic HCT recipients must also receive some form of immunosuppression to prevent GVHD, further limiting immune competence.
Unlike recovery of other hematopoietic lineages, which typically occurs over the course of weeks following HCT, lymphocyte recovery is a prolonged process. Reestablishment of immunocompetence requires at least several months, and some patients continue to demonstrate immune deficits for several years after HCT. In general, NK cells are the first lymphocyte subset to recover, followed by CD8+ T cells, which often reach supranormal levels within 2 to 8 months after HCT. Subsequently, B cells and ultimately CD4+ T cells recover. The pace and extent of recovery of each lymphocyte subset is highly dependent upon several factors, which ultimately determine the degree, extent, and duration of immune incompetence experienced by the individual HCT recipient (Figure 1).
Regeneration of lymphocytes in humans is an inefficient process, which primarily involves 2 distinct pathways. In the first pathway, lymphocytes regenerate from bone marrow (BM) lymphoid progenitors, thus recapitulating ontogeny and regenerating a naïve immune system, similar to that found in a newborn child [50]. NK cell recovery uses this pathway exclusively. Full recovery of NK cell counts is typically achieved within 1 to 2 months following HCT. B cells are also primarily regenerated from lymphoid progenitor cells, as evidenced by the appearance of primitive B cell subsets as the harbinger of B cell immune reconstitution [51]. Unlike NK cell recovery, however, B cell lymphopoiesis is highly dependent upon a specialized marrow microenvironment termed the “bursal equivalent,” which is susceptible to damage by the preparative regimen and is exquisitely sensitive to the toxic effects of GVHD and/or its treatment. Indeed, patients who experience even a limited episode of steroid-responsive GVHD show significantly diminished B cell reconstitution in the long term, compared with patients who do not experience GVHD [52]. Although recent data have demonstrated that mature B cells can also contribute to B cell reconstitution via homeostatic expansion, this pathway appears to be minor compared with the marrow-derived pathway for B cell regeneration.
Restoration of full humoral immune competence following HCT requires reconstitution not only of naïve B cells, but of a memory B cell pool as well. The latter occurs as a result of environmental or vaccine-based exposure to common pathogens and also requires help from CD4+ T cells. Therefore, even HCT recipients who do not experience GVHD and who demonstrate recovery of total B cell numbers within 6 months posttransplant should not be considered to have regained full humoral immunocompetence by this time. For at least 1 year following transplantation, essentially all HCT recipients remain predisposed to infections from encapsulated bacteria and viruses, against which neutralizing antibodies provide a first line of defense. Serum IgG levels provide little insight into B cell reconstitution, because long-lived, radioresistant plasma cells survive many preparative regimens [53] and can produce substantial circulating IgG without providing humoral responses to specific pathogens. The only reliable means by which one can assess humoral immune competence following transplantation is by documenting clinically significant rises in antigen-specific antibodies following vaccination or infection. Indeed, some medical centers use a rise in antibody levels in response to administration of a killed vaccine as a prerequisite for use of live-attenuated vaccines.
T cell regeneration is predominantly driven by a thymic-independent pathway, termed homeostatic peripheral expansion. Here, mature T cells contained within the graft dramatically expand in vivo in response to T cell lymphopenia. This process is driven by a combination of factors, among which are increased availability of homeostatic cytokines, including interleukin (IL)-7 and IL-15, which accumulate during lymphopenia; inflammatory cytokines associated with tissue damage induced by the preparative regimen; and exposure to viral antigens (either environmentally or via reactivation) during the period of profound lymphopenia. Peripheral homoeostatic expansion is much more efficient for CD8+ T cells than for CD4+ T cells [54], resulting in chronically reduced CD4/CD8 ratios in HCT recipients for several months following HCT. Memory T cells are the first to expand after HCT, and may be either of donor origin, in the case of non-T cell-depleted HCT, or originate from host T cells that have survived the conditioning regimen, in the case of T cell-depleted HCT [55]. Memory T cells respond quickly to previously encountered pathogens such as herpesviruses.
Of all factors analyzed thus far, CD4+ counts may provide the most readily available and predictive marker of the restoration of immune competence following HCT. Although the predictive value of low CD4+ counts has not been as extensively studied in recipients of HCT as they have in HIV infection, several studies have demonstrated that CD4+ recovery is associated with diminished infectious risk and improved transplant outcomes 56, 57, 58, 59. When T cell regeneration occurs via the ontogenic or thymic-dependent pathway, there is a substantial rise in CD4+ T cell numbers, with recovery of naïve CD4+ and CD8+ T cells and diversification of the T cell repertoire [60]. However, because the microenvironment of the thymus is highly susceptible to damage from age, therapy, and GVHD, many adult HCT recipients show little or no thymic-dependent T cell regeneration for months to years following HCT 61, 62, 63. A study of adult recipients of autologous HCT for breast cancer revealed that with each advancing decade of patient age between 30 and 60 years, a decreasing percentage of patients achieved effective CD4+ immune reconstitution after 2 years of follow-up [64].
Beyond the general rule that all HCT recipients experience profound immunosuppression at some point, the degree of immunosuppression experienced by individual patients varies greatly and is influenced by several factors. First and foremost is the profound adverse effect of GVHD on the overall process. In essentially every series, GVHD severity correlates with the degree of immunosuppression and infectious complications. This correlation is because of a variety of factors that compound one another, including damage to lymphoid microenvironments, adverse effects of GVHD on homeostatic peripheral expansion, as well as the obvious impact that chronic immunosuppression has on a reconstituting immune system. Second, recipient factors such as age, comorbidities, and infectious exposure prior to transplant contribute substantially to the risk for posttransplant infectious complications. This is illustrated in studies of severe combined immunodeficiency (SCID) patients, wherein outcomes are most successful in children who undergo transplantation before experiencing severe, life-threatening infection [65]. Third, graft-associated factors also play an important role. Recent studies have suggested that PBSC graft recipients show more rapid immune reconstitution, as measured by lymphocyte subsets [42], whereas UCB transplantation in adults 66, 67 and transplantation of profoundly T cell-depleted haploidentical grafts result in poor immune reconstitution and high rates of infectious complications. CD34 dose is crucial and levels of 3 × 106 CD34+ cells/kg or more are associated with an improved hematopoietic recovery, a decreased incidence of fungal infections, and improved overall survival (OS) in recipients of unmanipulated BM transplants from HLA-identical sibling donors [68].
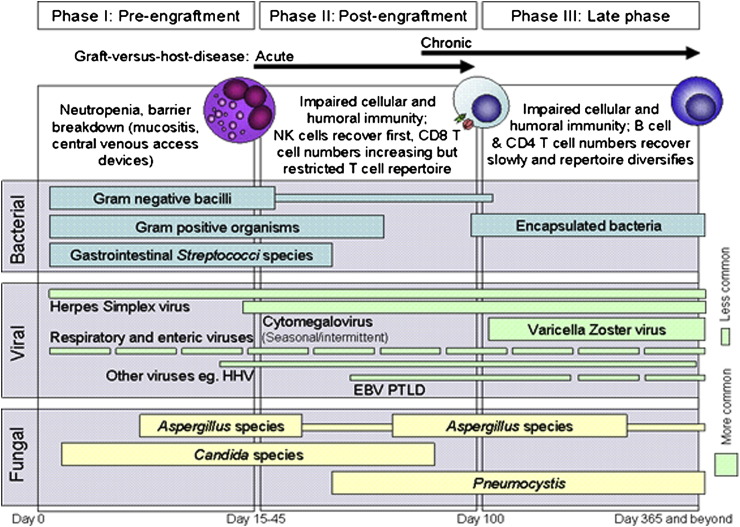
Models that distinguish between infectious complications occurring during different posttransplantation phases have been put forth, based largely upon an MA paradigm. Such a model is shown in Figure 2 , in which phase I is the preengraftment phase (<15-45 days after HCT); phase II, the postengraftment phase (30-100 days after HCT); and phase III, the late phase (>100 days after HCT). During phase I, prolonged neutropenia and breaks in the mucocutaneous barrier result in substantial risk for bacteremia and fungal infections involving Candida species and, as neutropenia continues, Aspergillus species. Additionally, herpes simplex virus (HSV) reactivation occurs during this phase. During phase II, infections relate primarily to impaired cell-mediated immunity. The scope and impact of this defect is determined by the extent of GVHD and immunosuppressive therapy for it. Herpesviruses, particularly cytomegalovirus (CMV), are common infectious agents during this period. Other dominant pathogens during this phase include Pneumocystis jiroveci and Aspergillus species. During phase III, persons with cGVHD and recipients of alternate-donor allogeneic transplants remain most at risk for infection. Common pathogens include CMV, varicella-zoster virus (VZV), and infections with encapsulated bacteria (eg, Streptococcus pneumoniae). The relative risk for these infections is approximately proportional to the severity of the patient's GVHD during phases II and III. For recipients of NMA grafts, substantial differences may be observed during phase I, but the susceptibility to infections during phases II and III are largely similar, and driven primarily by the status of the underlying disease, a history of GVHD, and/or the need for ongoing immunosuppression. The risk of disease from community-acquired respiratory viruses (CRV) is elevated during all 3 phases; in phase III, however, the outpatient status of HCT recipients can complicate efforts to reduce exposure and provide timely intervention.
Figure 2.
Phases of opportunistic infections among allogeneic HCT recipients Abbreviations: EBV, Epstein-Barr virus; HHV6, human herpesvirus 6; PTLD, posttransplant lymphoproliferative disease.
Thus, the risk of infection is primarily determined by the time from transplant and the presence or absence of GVHD. Other factors include donor/host histocompatibility, disease status, graft type, graft contents, conditioning intensity, and neutrophil engraftment (Table 3 ) 6, 7, 52, 69, 70, 71, 72, 73, 74. Unfortunately, there is currently no definitive laboratory marker of immune reconstitution that would predict infectious risk that could be used to tailor infection prophylaxis. It is likely that the degree of immune recovery measured by various assays is associated with clinical outcomes including infection rates, based on retrospective studies that included relatively small numbers of patients (Table 4 ). However, a rigorous proof of the association is lacking. Moreover, most of the published studies have focused on the association of immune assay results with outcomes that can be clearly defined and captured (eg, survival or NRM) rather than with infections. Additionally, information from the published studies is limited because of publication bias (ie, there is a tendency to publish studies that find an association rather than negative studies). Tools are now available to precisely monitor viral-specific (Epstein-Barr virus [EBV], CMV) immune responses (HLA tetramers, interferon-γ production assays), and may help in understanding this issue [75]. Large (ideally, prospective) studies are needed, first to conclusively determine what immune monitoring test has prognostic value and ultimately to determine whether outcomes would improve if such a test were used to tailor infection prophylaxis.
Table 3.
Factors Affecting the Risk of Infection
| Factor | Risk of Infection |
|---|---|
| Type of transplant | Higher risk with allogeneic, lower risk with autologous or syngeneic, depending on graft manipulation and clinical setting, including previous therapies |
| Time from transplant | Lower risk with more time elapsed from transplant |
| Pretransplant factors | Higher risk with extensive pretransplant immunosuppressive therapy (eg, fludarabine, clofaribine), prolonged pretransplant neutropenia, or pretransplant infection |
| GVHD | Higher risk with grade III-IV acute GVHD or extensive chronic GVHD |
| HLA match | Higher risk with HLA-mismatched donors, particularly with haploidentical donors |
| Disease (eg, leukemia) status | Higher risk with more advanced disease at the time of transplant |
| Donor type | Higher risk with marrow unrelated donor than with a fully matching sibling donor |
| Graft type | Highest risk with cord blood, intermediate risk with bone marrow and lowest risk with colony stimulating factor-mobilized blood stem cells. Higher risk with T cell-depleted grafts (depending upon method used) |
| Immunosuppression after transplant | Higher with immunosuppressive drugs, in particular with corticosteroids, antithymocyte globulin, alemtuzumab |
| Conditioning intensity | Lower risk in the first 1-3 months posttransplant with low-dose chemo/radiotherapy |
| Neutrophil engraftment | Higher risk with delayed engraftment/nonengraftment |
GVHD indicates graft-versus-host disease.
Table 4.
Parameters Reported to Correlate with Clinical Outcomes after HCT
| Parameter (Ref.) | Timing | Result | Outcome | Multivariate Analysis |
|---|---|---|---|---|
| Lymphocyte count 813, 814, 815 | Day 15 | <500/μL | Decreased OS and PFS (autologous HCT) | Yes |
| Lymphocyte count 816, 817 | Day 30 | <300/μL | Decreased OS and LFS, Increased NRM (allogeneic HCT) | Yes |
| B cells and monocyte counts [818] | Day 80 | Low (cutoff value not given) | Increased infections | Yes |
| CD4 T cell count [59] | 3 months | <200/μL | Decreased OS, Increased NRM and infections |
|
| CD8 T cell and B cell counts [819] | 6 months | Low (cutoff value not given) | Increased treatment failure (death, relapse or graft failure) | No |
| Every 2 weeks during days 0 to 65 | <7 cells/mL in all samples | Increased risk of recurrent or persistent CMV reactivation | Not specified | |
| CMV-specific lymphoproliferation [248] | 4 months | Undetectable proliferation | Increased late CMV disease | No |
| NK-cell chimerism [817] | First 100 days | Incomplete chimerism | Decreased RFS | Yes |
| NK-cell count [821] | Day 15 | <80/μL | Decreased OS and PFS (autologous HCT) | Yes |
| CD56high NK-cell count [822] | Day 14 | <7/μL† | Decreased OS, increased NRM |
|
| Non-HLA genetics 823, 824, 825, 826 | Pretransplant | At risk allele in donor or recipient | Increased infections, survival | No |
OS indicates overall survival; PFS, progression-free survival; LFS, leukemia-free survival; NRM, nonrelapse mortality; CMV, cytomegalovirus; HCT, hematopoietic cell transplant.
Adapted from Geddes et al. [811] with publisher's permission. Additional studies are needed before any one of the immune tests presented here can be recommended for use in decision making on infection prophylaxis (see text).
Assay measuring the quantity but not quality of CMV-specific T cells.
Patients were split into low, intermediate, and high groups with cutoffs of 4/μL and 9/μL.
In summary, HCT is characterized by a variable period of early infectious complications caused largely by neutropenia and mucosal damage because of the preparative regimen, and such complications are readily predictable based upon clinical findings of mucositis and absolute neutrophil count (ANC). In addition, essentially all HCT recipients experience a prolonged period of immunosuppression characterized by profound defects in cell-mediated and humoral immunity. Unfortunately, there are no readily available surrogate markers to accurately measure the relative risk for individual patients. Consequently, these patients must be monitored carefully and receive early intervention for signs or symptoms of an infectious disease. In most patients, immunocompetence improves progressively with increasing time after transplant, an observation that has led to the current recommendations for revaccination at 6 to 24 months, including with some live vaccines at ∼24 months for patients who are no longer receiving immunosuppression and show no signs of GVHD. However, it is important to realize that many HCT recipients remain immunocompromised far beyond 2 years after transplant—especially individuals with cGVHD, for whom infection remains the most important cause of morbidity and mortality. Future work is needed to augment the degree of immune reconstitution toward pathogens and malignancy and to identify accurate surrogate markers of immunocompetence to guide the long-term management of this high-risk population.
Hematopoietic Cell Graft Safety
D. Confer, R. Gress, M. Tomblyn, G. Ehninger
With allogeneic HCT, the life of the recipient may depend on the timely selection of an acceptable HLA-matched donor. Only a limited number of HLA-matched donors might be identified. Hence, the transplant physician may have to accept a higher risk for transmission of an infectious agent through HCT than would be permitted for routine blood transfusion. This section provides strategies for the HCT physician to minimize transmission of infectious diseases, whenever possible, from donors to recipients. In general, these strategies are dictated by national regulations, and, therefore, ratings are not included.
In addition to recipient safety, clinicians must recognize the donor role and the potential impact upon the donor of the product safety determination 76, 77, 78, 79. Assessment of the donor should include elements related to safety for the donor (eg, uncontrolled hypertension, diabetes mellitus, and coronary artery disease) in addition to determination of product safety. A donor may be cleared to donate from a product safety perspective, but be unsuitable because of medical conditions that render the donation unsafe 76, 77, 78. Because of these potential risks, individuals who donate must provide informed consent.
Assessment of product safety is complicated by the high frequency of international exchange. A survey by the World Marrow Donor Association found that, in 2006, 3269 (39%) adult products and 829 (40%) UCB units were shipped from 1 country to transplant centers in another country. This robust international activity creates further challenges for ensuring product safety related to regional differences in disease risks, regulatory structures, and medical practices.
The desire for HLA tissue matching (which presumably underlies much of the international exchange), results in the potential need to recruit in poorly represented populations. For any given individual, outside of the immediate family, the highest likelihood of an HLA match occurs among persons of the same or similar ethnicity. There is a strong ethical impetus for parity in the likelihood of HLA matching, regardless of the patient's ethnic origins. This may necessitate recruitment of HCT donors from geographic regions where endemic conditions would make the residents unsuitable as routine blood or tissue donors.
Regulations
In the United States, the FDA issues regulations covering product safety and donor eligibility, including screening for the relevant communicable disease agents and disease, as well as laboratory and test kit requirements [80]. FDA Guidance documents on these topics can be found at http://www.fda.gov/cber/index.html. In the European Union, general requirements are defined in the European Commission Directive 2004/23/EC [81], and donor eligibility defined in Commission Directive 2006/17/EC [82]. Requirements similar to those of the FDA and European Union have been or are being developed in Canada (http://www.hc-sc.gc.ca/dhp-mps/brgtherap/reg-init/cell/cto_draft_gd-cto_ebauche_ld-eng.php), Australia, Japan, and elsewhere.
Donor Conditions Affecting Product Safety
Disorders pertinent to HCT can be categorized according to prevalence and severity. Prevalence concerns the likelihood of the disorder within the potential donor population and may be highly dependent upon the donor's geographic location. Severity refers to both the usual consequences of the disorder and the ease of managing those consequences. Highly pertinent disorders 76, 77, 78, 80, 82, 83 include infections caused by HIV, hepatitis B virus (HBV), hepatitis C virus (HCV), human T cell leukemia viruses type I and II (HTLV-I and HTLV-II), and West Nile virus 84, 85, 86, 87, 88, 89, 90. Other pertinent disorders 76, 77, 78, 82, 83, 91 include transmissible spongiform encephalopathy (TSE) 92, 93, 94, 95, CMV infection 96, 97, EBV infection, syphilis, toxoplasmosis 98, 99, and vaccinia [80], as well as viruses used in other live-attenuated vaccines.
Persons acutely infected with CMV or EBV should not donate. Similarly, those with acute toxoplasmosis should not donate until the acute illness has resolved. However, physicians should be aware that in rare cases, persons who are asymptomatically seropositive for Toxoplasma gondii might transmit this infection through HCT [100].
Uncommon disorders include malaria 101, 102, 103, 104, 105, 106, 107, 108 and tick-borne diseases such as Rocky Mountain spotted fever, babesiosis, Q fever (infection caused by Coxiella species), and Colorado tick fever 99, 109, 110, 111, 112, 113, 114. Prospective HCT donors who reside in or have traveled to areas endemic for rickettsia or other tick-borne pathogens and who are suspected of having an acute tick-borne infection should be temporarily deferred as donors until infection with these pathogens is excluded. Blood banking standards strongly recommend deferral for a past history of Q fever or babesiosis because these infections can be chronic and Babesia parasites might persist despite appropriate therapy [115]. Additionally, researchers have recommended deferring persons with acute human ehrlichiosis (eg, active human granulocytic anaplasmosis [116], human monocytic ehrlichiosis, as well as any infections from species within the genus Ehrlichia) from HCT donation.
Chagas disease is transmissible by transfusion 99, 117, 118, 119. Potential donors who reside in or have emigrated from endemic areas should be screened serologically for Trypanosoma cruzi infection. Researchers also recommend deferral of HCT donation if a past history exists because the parasite can persist despite therapy 115, 120, 121, 122.
Prospective donors with symptoms of active tuberculosis (TB) should be evaluated for that disease 123, 124, and those with active TB should not donate until the TB is well controlled (eg, no longer contagious as determined by the donor's primary physician) after appropriate medical therapy. However, transplanting marrow from an untreated, tuberculin skin test-positive donor who has no evidence of active disease poses no known risk. Screening potential donors for TB with tuberculin skin tests is not necessary.
Preventing Transmission of Infections from HCT Donors to Recipients
All prospective HCT donors should be evaluated to determine their general state of health and whether they pose a risk for transmitting infectious diseases to the recipient. Evaluation of donors is achieved through screening and laboratory testing 76, 77, 78, 80. Screening and testing of autologous donors is recommended to ensure the safety of laboratory personnel and to prevent cross contamination. If autologous donors are not screened and tested, their autologous units should be specially labeled and handled as if potentially infected 76, 77, 78, 80. To detect transmissible infections, all HCT donor collection site personnel should follow up-to-date published guidelines and standards for donor screening and testing.
Complete donor screening, which includes history, physical examination, and review of medical records, must be performed within 6 months preceding donation. Prior to each donation procedure, screening must be repeated or updated. Abbreviated screening may be used if complete screening has been performed within the previous 6 months 80, 83. Abbreviated screening is an inquiry about any changes in history, risk factors, or physical findings. This practice is critical because if new risk findings have developed, the potential donor might require further evaluation or deferral. When the donor is 1 month of age or younger, including all UCB donations, the infant's mother should be screened 80, 83.
The donor history includes direct questioning of the donor (which can be done over the telephone), with documentation of all donor responses, review of available medical records, and completion of a risk factor questionnaire (eg, the NMDP questionnaire) [125]. The physical examination of the donor is targeted to detect stigmata associated with transfusion-transmissible disease or high-risk activities.
In addition to donor screening, laboratory testing must be performed to assess for relevant infectious diseases. In the United States, for lymphocyte and UCB donations, a specimen for testing must be obtained within 7 days before or after the donation. For PBSC and BM donations, the specimen may be obtained up to 30 days before donation. In the European Union, the testing specimen must be obtained at the time of donation or within 7 days after donation unless the product can be stored. If storage is possible, the sample may be obtained up to 30 days prior to donation; however, this invokes a further requirement for retesting of the donor 180 days or more after donation. Retesting is not required, however, if nucleic acid testing for HIV, HCV, and HBV is included on the initial specimen. As in the United States, for PBSC or BM that will not be stored, the specimen may be obtained up to 30 days before donation, and there is no requirement for retesting.
In the United States, positive results on screening tests are used for the determination of disease risk regardless of the results obtained with confirmatory tests. The sole exception is testing for syphilis, where a syphilis-specific test is used for determination of eligibility [80].
Use of Potentially Unsafe Products
Oversight by governments and unrelated donor registries generally precludes the use of a volunteer unrelated donor at risk for infectious diseases transmissible by HCT 78, 79, 80, 82, 83. Whether to select a related donor who is at risk for or who has an infectious disease transmissible by HCT should be determined on a case-by-case basis, and is the final responsibility of the HCT physician. If the only possible donor is at risk for, or known to have, infection with a blood-borne pathogen, but the patient is likely to succumb rapidly from his or her disease if an HCT is not received, the physician must carefully weigh the risks and benefits of using potentially infected donor cells. No person should be denied a potentially life-saving HCT procedure solely on the basis of the risk for an infectious disease. However, HCT physicians should avoid transplanting any hematopoietic cell product from an infected or infectious donor unless no other stem cell product can be obtained and the risk for death if transplantation is not performed is deemed to be greater than the risk for morbidity or death from the infection that may be transmitted. If the HCT physician judges that the circumstances of the case justify the use of such a product, the following should be noted in the recipient's chart:
-
•
Knowledge and authorization of the recipient's HCT physician regarding the potential for transmission of an infectious agent during HCT; and
-
•
Informed consent from the recipient or recipient's legal guardian acknowledging the possible transmission of an infectious agent during the transplantation.
Subsequently, the HCT physician should include the infectious agent in the differential diagnosis of any illness that the HCT recipient experiences so that the infection, if transmitted, can be diagnosed early and treated preemptively, if possible.
Infectious products (except those in which CMV seropositivity as the only evidence of infectiousness) should be labeled as being a biohazard or as untested for biohazards, as applicable. A warning label should list all disease agents or diseases for which the donor has shown reactive test results. Tissue intended for autologous use should be labeled “For Autologous Use Only—Use Only for (Patient's Name).”
Safe Handling, Processing, and Storage of Donated Units
Personnel of donation, collection, or transplantation centers, cell-processing laboratories, and courier services should follow current standards for detecting and preventing extrinsic bacterial and fungal contamination of collected stem cell units at the collection site, during processing and transportation, and at the transplant center 126, 127, 128, 129, 130, 131, 132, 133, 134, 135, 136, 137, 138, 139, 140, 141. Current, comprehensive discussions of these issues are detailed in the Food and Drug Administration (FDA) “Good Tissue Practice” regulations [142], in the European Commission regulations [81], and in international standards established by the professional organizations 76, 77, 143.
Bacterial Infection Prevention after HCT
D. Engelhard, M. Akova, M.A. Boeckh, A. Freifeld, K. Sepkowitz, C. Viscoli, J. Wade, I. Raad
In addition to general recommendations regarding bacterial infections, this section provides specific recommendations regarding S. pneumoniae, viridans streptococci (mainly the S. mitis and S. anginosus groups), H. influenzae type b, and central line-associated blood stream infections (CLABSI). Recommendations regarding specific nosocomial infections, including those from Legionella species, methicillin-resistant Staphylococcus aureus (MRSA), Staphylococcus species with reduced susceptibility to vancomycin, vancomycin-resistant Enterococcus (VRE), and Clostridium difficile are found in the Infection Prevention and Control in Health care Facilities section.
General Recommendations
Preventing exposure
Health care workers (HCWs) and others in contact with hematopoietic cell transplantation (HCT) recipients should routinely follow appropriate hand hygiene practices to avoid exposing recipients to bacterial pathogens (AIII) (see Infection Prevention and Control in Health care Facilities). Additional precautions for patients colonized with certain contagious pathogens (eg, MRSA, VRE) and for special situations (eg, diarrhea or respiratory diseases) and instructions with regard to visitors, pets, and plants are described in the Infection Prevention and Control in Health care Facilities section and in the 2007 CDC guidelines [144]. Guidelines for the management of neutropenic fever are published elsewhere [145].
Preventing early disease (0-100 days after HCT)
Antibacterial prophylaxis with a fluoroquinolone (ie, levofloxacin) to prevent bacterial infections should be strongly considered for adult HCT patients with anticipated neutropenic periods of 7 days or more (BI) 146, 147, 148, 149, 150 (Appendix 1). Because of lack of data, there are currently no antimicrobial prophylactic regimens that can be recommended for children. Some experts use levofloxacin for pediatric antibacterial prophylaxis (CIII). Antibacterial prophylaxis is generally started at the time of stem cell infusion and continued until recovery from neutropenia or initiation of empirical antibacterial therapy for fever during neutropenia 148, 149, 150, 151, 152. The prophylaxis should not be continued after recovery from neutropenia. Local epidemiological data should be carefully considered before applying fluoroquinolone prophylaxis, and once it is applied, the emergence of resistance in bacterial pathogens should be monitored closely because of increasing quinolone resistance worldwide among Gram-negative bacteria (eg, Escherichia coli, Klebsiella pneumoniae, and Pseudomonas aeruginosa) [153] and staphylococci 154, 155, 156, 157; plasmid-mediated quinolone resistance in Enterobacteriaceae that is closely related to the beta lactamases that inactivate third-generation cephalosporins 158, 159, 160; and quinolone-related development of a hypervirulent strain of C. difficile (AIII) [161].
The addition of an anti-Gram-positive agent to the prophylaxis regimen is not indicated (DIII) [151]. Specifically, glycopeptides (eg, vancomycin, teicoplanin) should not be used for routine bacterial prophylaxis, either systemically or for prevention of catheter-related infections (DIII). These agents lack benefit for prophylaxis and their use may promote the emergence of resistant microorganisms [151].
Growth factors (eg, granulocyte-macrophage-colony-stimulating factor [GM-CSF] and G-CSF) shorten the duration of neutropenia after HCT 162, 163 and may slightly reduce the risk of infection 164, 165 but have not been shown to reduce mortality 164, 165. Therefore, the routine use of growth factors after HCT is controversial and no recommendation for their use can be made (CI).
Prophylaxis with metronidazole has been shown to reduce anaerobic bacterial growth in the gut, and was associated with a reduction in acute graft-versus-host disease (aGVHD) in recipients of transplants from HLA-identical sibling donors (but not from other donors) in 1 open-labeled randomized trial [166]. However, the evidence is insufficient to recommend routine gut decontamination for HCT candidates, either with metronidazole or with nonabsorbable antibiotics (DIII).
Although intravenous immunoglobulin (IVIG) has been recommended for use in producing immune system modulation for GVHD Prevention, IVIG should not be routinely administered to HCT recipients for prophylaxis of bacterial infection within the first 100 days after transplantation (DI). Some centers check total IgG levels in high-risk HCT recipients (eg, those with unrelated marrow grafts). For patients with severe hypogammaglobulinemia (ie, IgG <400 mg/dL), IVIG prophylaxis may be considered (Appendix 1) (CIII). The IVIG dose and frequency for a hypogammaglobulinemic HCT recipient should be individualized to maintain trough serum IgG concentrations >400 mg/dL, as the half-life of IVIG among HCT recipients (generally, 1-10 days) is much shorter than the half-life among healthy adults (generally, 18-23 days) (BII) 167, 168, 169.
Preventing late disease (>100 days after HCT)
Prolonged antibiotic prophylaxis is recommended only for preventing infection with S. pneumoniae among allogeneic recipients with chronic (cGVHD) for as long as active cGVHD treatment is administered (AIII) [170]. Antibiotic selection should be guided by local antibiotic resistance patterns.
In the absence of severe hypogammaglobulinemia (ie, IgG levels <400 mg/dL, which might be associated with bacteremia or recurrent sinopulmonary infections), routine monthly IVIG administration to HCT recipients >100 days after allogeneic or autologous HCT is not recommended (DI) 171, 172 as a means of preventing bacterial infections.
Recommendations for preventing late bacterial infections are the same among pediatric or adult HCT recipients.
Recommendations Regarding CLABSI
Catheter-associated infections are a leading cause of bloodstream infections in HCT recipients, particularly during the preengraftment phase and in patients with GVHD 173, 174. These infections often result in catheter removal and, much less commonly, in death 175, 176.
In HCT recipients, all central venous catheters (CVCs), whether tunneled or nontunneled, should be inserted using maximal sterile barrier precautions (AI) [177]. The preferred approach is the CLABSI prevention bundle, which consists of hand hygiene, full barrier precautions, cleaning the insertion site with chlorhexidine, avoiding femoral sites for insertion, and removing unnecessary catheters [178]. Although the efficacy of the CLABSI prevention bundle has not been studied in HCT recipients, all 5 elements of the bundle are recommended for this patient population (AII).
Other measures to decrease the risk of CLABSI have been studied. Catheters impregnated with minocycline/rifampin 179, 180, 181 have been shown to decrease CLABSI in patients requiring nontunneled subclavian central venous access, including HCT patients. In 1 retrospective study, the minocycline/rifampin impregnated catheters did not affect the susceptibility of staphylococci to tetracyclines or rifampin [181]. For HCT centers with high CLABSI rates (more than 1 per 1000 catheter days) despite effective implementation of the CLABSI bundle elements, use of additional interventions, such as minocycline/rifampin antimicrobial-impregnated catheters, to prevent CLABSIs should be considered (BIII) [182].
Additional prevention strategies (eg, catheter-site dressing regimens, antimicrobial/antiseptic ointments, and antimicrobial lock prophylaxis) have been evaluated but not extensively assessed among the HCT patient population. A review found a similar risk of infection regardless of whether catheter dressings consisted of a transparent, semipermeable polyurethane dressing, or of sterile gauze and tape [183]. Povidone-iodine ointment, mupirocin ointment, and other antimicrobial ointments applied at the catheter insertion site have failed to show a consistent advantage when compared with no antimicrobial ointment. Recently, data have shown in non-HCT patients that chlorhexadine-impregnated sponges have decreased the rates of catheter related infections 184, 185. Topical antimicrobials should be avoided because of the risk of antimicrobial resistance or increased fungal colonization in immunosuppressed HCT patients (DIII).
Antimicrobial lock prophylaxis using antimicrobial solutions, including those that contain vancomycin, have been studied in patients with long-term venous access devices and who develop neutropenia. A meta-analysis of the prospective, randomized trials testing the vancomycin-lock solution reported a decreased rate of bloodstream infections with vancomycin-susceptible organisms and a delay in the onset of the first bloodstream infection. However, the use of vancomycin-containing locks is not recommended, because of the increased risk of selecting for staphylococci with reduced vancomycin susceptibility [186] (DIII). Other alternatives, including lock solutions containing minocycline/EDTA, taurolidine citrate, or ethanol, have shown success in preventing CLABSI and salvaging vascular access 187, 188, 189, 190. These approaches, though promising, cannot be recommended in routine catheter care until further investigation is completed (DIII)
When adopting prevention practices such as these, HCT programs should institute prospective data collection and reporting that allows for analysis of the success of the practices. HCT teams can also make use of a systemic review that defines benchmark rates of CLABSI for a wide range of catheter types [191]. Although not all of the studies included in the meta-analysis were conducted exclusively among HCT patients, HCT teams can use the results to assess their own center's relative performance, as an aspect of quality improvement efforts.
Recommendations Regarding Streptococcus pneumoniae
Preventing exposure
Standard precautions should be taken with hospitalized patients infected with S. pneumoniae (AIII), including patients with infection caused by drug-resistant strains [192].
Preventing disease
Invasive pneumococcal infection (IPI) is a life-threatening complication that may occur months to years after HCT. The annual incidence of IPI is 8.23/1000 transplants among allogeneic HCT recipients, and higher still among those with cGVHD (20.8/1000 transplants) [193]. Although IPI occurs less frequently in autologous HCT recipients (annual incidence, 3.8/1000 transplants), the risk remains much higher than in an immunocompetent population [193]. Efforts to prevent IPI should include active immunization and prophylactic antibiotics (Table 6 and Appendix 1). Vaccination against S. pneumoniae is recommended for all HCT recipients, preferably with pneumococcal 7-valent conjugate vaccine (BI) (see HCT Recipient Vaccinations).
Table 6.
Vaccinations Recommended for Both Autologous and Allogeneic HCT Recipients
| Vaccine | Recommended for Use after HCT | Time Post-HCT to Initiate Vaccine | Number of Doses∗ | Improved by Donor Vaccination (Practicable Only in Related-Donor Setting) |
|---|---|---|---|---|
|
Yes (BI) | 3-6 months | 3-4† | Yes; may be considered when the recipient is at high risk for chronic GVHD |
|
|
6-12 months | 3§ |
|
|
Yes (BII) | 6-12 months | 3 | Yes |
| Meningococcal conjugate | Follow country recommendations for general population (BII) | 6-12 months | 1 | Unknown |
| Inactivated polio | Yes (BII) | 6-12 months | 3 | Unknown |
|
Follow country recommendations for general population (BII) | 6-12 months | 3 | Likely¶ |
|
Yearly (AII) | 4-6 months | 1 -2⊥ | Unknown |
|
|
24 months | 1 -2‡‡ | Unknown |
|
|
HCT indicates hematopoietic cell transplant; GVHD, graft-versus-host disease.
A uniform specific interval between doses cannot be recommended as various intervals have been used in studies. As a general guideline, a minimum of 1 month between doses may be reasonable.
Following the primary series of 3 PCV doses, a dose of the 23-valent polysaccharide pneumococcal vaccine (PPSV23) to broaden the immune response might be given (BII). For patients with chronic GVHD who are likely to respond poorly to PPSV23, a fourth dose of the PCV should be considered instead of PPSV23 (CIII).
DTaP is preferred, however, if only Tdap is available (eg, because DTaP is not licensed for adults), administer Tdap. Acellular pertussis vaccine is preferred, but the whole-cell pertussis vaccine should be used if it is the only pertussis vaccine available. (See text for more information.)
See text for consideration of an additional dose(s) of Tdap for older children and adults.
Significant improvement of recipient response to hepatitis B vaccine posttransplant can be expected only if the donor receives more than 1 hepatitis vaccine dose prior to donation.
For children <9 years of age, 2 doses are recommended yearly between transplant and 9 years of age [306].
Measles, mumps, and rubella vaccines are usually given together as a combination vaccine. In females with pregnancy potential, vaccination with rubella vaccine either as a single or a combination vaccine is indicated.
In children, 2 doses are favored.
Antibiotic prophylaxis against pneumococcal infection is indicated in patients with cGVHD and those with low IgG levels (AIII). Antibiotic prophylaxis should be administered even to patients who have received pneumococcal vaccine, because not all strains are included in the vaccines, the immunogenicity of vaccines against the vaccine strains in HCT patients is only, at most, about 80% 194, 195, and because of the theoretic concern that strains not included in the vaccine will replace vaccine strains. Oral penicillin remains the preferred choice, but antibiotic selection depends on the local pattern of pneumococcal resistance to penicillin and other antibiotics (ie, second-generation cephalosporins, macrolides, and quinolones) 196, 197, 198, 199. Early empirical antibiotic treatment is required in any HCT patient with suspected IPI, regardless of the time since transplant, the immunization status, and the use of chemoprophylaxis (AIII) [193].
Recommendations Regarding Viridans Streptococci
Preventing exposure
Viridans streptococci are normal commensals, primarily of the oral surfaces. Hence, preventive efforts must focus on preventing systemic infection and disease rather than preventing exposure.
Preventing disease
Chemotherapy-induced oral mucositis is a potential source of viridans streptococcal bacteremia and sepsis. Consequently, before the start of conditioning, dental consults should be considered for all HCT candidates to assess their state of oral health and to perform any needed dental procedures to decrease the risk for oral infections after transplant (AIII) [200]. Many experts recommend that antibiotics active against viridans streptococci be given from the time of transplantation until a minimum of day +21 afterward (CIII). However, this approach has not been systematically studied. Penicillin-, quinolone-, and vancomycin-resistant strains of viridans streptococci have been reported 201, 202. Empiric treatment of any HCT recipient with fever, severe mucositis, and neutropenia should include an agent active against viridans streptococci to prevent complications from this potentially fatal infection [203].
Recommendations Regarding Haemophilus influenzae Type b
Preventing exposure
Vaccination campaigns have markedly reduced the incidence of H. influenzae type b (Hib) disease. However, in the rare event of a patient being hospitalized with Hib, standard precautions are recommended, with droplet precautions added for the first 24 hours after initiation of appropriate antibiotic therapy (BIII) 144, 204.
Preventing disease
Vaccination against Hib is recommended for all HCT recipients, as at least one-third of HCT recipients do not have protective antibody levels of Hib capsular polysaccharide antibodies after HCT, and the Hib conjugate vaccine has excellent efficacy among HCT recipients (BII) 194, 205 (see HCT Recipient Vaccinations).
All HCT recipients who are exposed to persons with Hib disease should receive prophylaxis with 4 days of rifampin [204], or with an alternative antimicrobial agent if rifampin would likely interfere with other prophylactic agents the patient is receiving (eg, extended-spectrum azoles) (BIII) (Appendix 1). Antibiotic prophylaxis is especially indicated for allogeneic HCT recipients with cGVHD, who are at increased risk for developing infections from Hib and other encapsulated organisms (see section on S. pneumoniae) [206].
Recommendations Regarding Bordetella pertussis
Preventing exposure
HCT recipients may be exposed to persons with pertussis, as this disease is increasingly frequent in the general community. Pertussis in an adolescent recipient of an unrelated cord blood transplant (CBT) has been reported [207]. In addition to standard precautions, droplet precautions should be used in patients hospitalized with pertussis; droplet precautions are recommended for 5 days after initiation of effective therapy or, if antibiotic treatment is not given, until 3 weeks after the onset of paroxysmal cough (BIII) [208].
Preventing disease
All HCT recipients who are exposed to persons with pertussis should receive prophylaxis with azithromycin or another macrolide (trimethoprim-sulfamethoxazole [TMP-SMZ] may be an alternative antimicrobial agent) regardless of age and immunization status (BIII) [208]. Following HCT, all HCT recipients should receive vaccination with acellular pertussis (see HCT Recipient Vaccinations).
Viral Disease Prevention after HCT
J. Zaia, L. Baden, M.A. Boeckh, S. Chakrabarti, H. Einsele, P. Ljungman, G.B. McDonald, H. Hirsch
Recommendations Regarding Cytomegalovirus
Preventing exposure
Hematopoietic Cell Transplantation (HCT) candidates should be tested for the presence of serum anti-cytomegalovirus (CMV) IgG antibodies before transplantation to determine their risk for primary CMV infection and reactivation after HCT (AII). CMV is shed intermittently from the oropharynx and the genitourinary tract of both immunocompetent and immunosuppressed subjects. There are no data demonstrating that avoiding these body fluids is feasible or effective in preventing acquisition of CMV in CMV-seronegative HCT recipients. Because CMV-seronegative pregnant health care workers may be at risk for contracting CMV from these and other patients, standard universal precautions should be used in these situations.
With proper management, CMV-seronegative patients have a low risk for contracting CMV infection. To reduce the risk of CMV transmission, blood products from CMV-seronegative donors or leukocyte-depleted blood products should be used in CMV-seronegative allogeneic HCT recipients (AI) 209, 210, 211. The benefit of using either of these products in autologous HCT recipients to prevent CMV transmission is less clear. However, because many autologous HCT recipients have received previous T cell suppressive therapy such as fludarabine (Flu) or alemtuzumab, the use of CMV-safe blood products is recommended (BII). In many centers, and even in entire countries, leukocyte filtration of blood products is mandatory. No controlled study has examined the potential benefit of the combination of seronegative blood products and filtered blood products. Leukocyte filtration should be performed at the blood bank and the established quality standard of <5 × 106 residual leukocytes/unit should be followed (AII) 212, 213.
Preventing disease and disease recurrence
HCT recipients at risk for posttransplant CMV disease (ie, all CMV-seropositive HCT recipients, and all CMV-seronegative recipients with a CMV-seropositive donor) should be placed on a CMV disease prevention program from the time of engraftment until at least 100 days after HCT (ie, phase II) (AI). Physicians should use either prophylaxis or preemptive treatment for allogeneic recipients (AI). In selecting a CMV disease prevention strategy, physicians should assess the risks and benefits of each strategy, the needs and condition of the patient, and the hospital's virology laboratory support capability.
A prophylaxis strategy against early CMV replication (ie, <100 days after HCT) for allogeneic recipients involves administering prophylaxis to all allogeneic recipients at risk throughout the period from engraftment to 100 days after HCT (Appendix 1) (AI). Ganciclovir, high-dose acyclovir, and valacyclovir have all shown efficacy in randomized studies in reducing the risk for CMV infection after HCT 214, 215, 216. If ganciclovir is used, the induction course is usually started at engraftment (AI) 214, 217, 218, although a brief prophylactic course can be added during pretransplant conditioning (CIII). If acyclovir or valacyclovir is used, the patient must also undergo viral monitoring and receive preemptive antiviral therapy if evidence of CMV replication is found (AI) 215, 216. IVIG is not recommended for CMV disease prophylaxis among HCT recipients (EIII).
In patients with CMV disease documented before transplantation, transplantation should be delayed until the disease is adequately treated (BII), and use of secondary anti-CMV prophylaxis during HCT should be considered (BIII) [219]. Such patients should be closely monitored during the HCT procedure, including during the preengraftment phase if the transplant center usually starts monitoring for CMV reactivation following engraftment, and a low threshold for preemptive treatment is used (BIII) [219].
The preemptive strategy targets antiviral treatment to those patients who have evidence of CMV replication after HCT (Appendix 1). It requires the use of sensitive and specific laboratory tests to rapidly diagnose CMV replication after HCT and to enable immediate administration of effective antiviral therapy after CMV replication has been detected. Allogeneic recipients at risk should be screened for the presence of CMV in blood samples ≈1 time/week from 10 days to at least 100 days after HCT (ie, phase II) (AIII). CMV-seropositive cord blood transplant (CBT) recipients are at increased risk of CMV reactivation and disease 220, 221. Thus, some researchers use acyclovir or valacyclovir prophylaxis 215, 216 in combination with preemptive therapy in these patients (CIII). A preemptive strategy against early CMV replication (ie, <100 days after HCT) for allogeneic recipients is preferred over prophylaxis for CMV-seronegative HCT recipients of seropositive donor cells (ie, D-positive and R-negative) because of the low attack rate of CMV replication if screened or filtered blood product support is used (BII).
Diagnostic tests to determine the need for preemptive treatment include the detection of CMV pp65 antigen in leukocytes (antigenemia) 217, 222, CMV DNA by quantitative polymerase chain reaction (PCR) 223, 224, 225, or the detection of CMV RNA [226]. HCT centers performing allogeneic transplants should have the capacity to perform 1 of these tests (AIII). Viral cultures of urine, saliva, blood, or bronchoalveolar washings by rapid shell-vial culture [227] or routine culture 228, 229 are today rarely used because these techniques are less sensitive than CMV-DNA PCR or CMV pp65 antigenemia tests. However, it should be recognized that CMV pp65 antigenemia tests may be falsely negative in patients with neutropenia.
Ganciclovir is often used as a first-line drug for preemptive therapy. Although foscarnet is as effective as ganciclovir [230], it is currently more commonly used as a second-line drug, because of practical reasons (eg, requirement for prehydration and electrolyte monitoring). Allogeneic recipients who are ≤100 days after HCT should begin preemptive treatment if CMV viremia, antigenemia, or DNA is detected (AI). Preemptive therapy should be given for a minimum of 2 weeks (A1) [217]. If CMV is still detected after 2 weeks of therapy, maintenance therapy can be given until CMV is undetectable [230] or it can be continued to day 100 (AI) [217]. After discontinuation of preemptive therapy, routine weekly screening is necessary to at least day 100 because recurrent episodes of CMV viremia commonly occur (BII).
Presently, only the intravenous formulation of ganciclovir has been approved for use in CMV prophylactic or preemptive strategies (AI). Valganciclovir, a prodrug of ganciclovir, has been increasingly used in preemptive therapy 231, 232, 233, 234, and an interim analysis of a randomized, controlled study has shown comparable results in patients treated with i.v. ganciclovir or valganciclovir [235]. Dose adjustment for renal insufficiency is necessary with either drug to avoid hematologic toxicity. Patients who are ganciclovir-intolerant should be treated with foscarnet (AI) [236]. HCT recipients receiving ganciclovir should have absolute neutrophil counts (ANCs) checked ≥2 times/week (BIII). Experts report managing ganciclovir-associated neutropenia by adding granulocyte colony-stimulating factor (G-CSF) [237] or temporarily stopping ganciclovir for ≈2 days if the patient's ANC is <1000 (CIII). Ganciclovir can be restarted when the patient's ANC is ≈1000 for 2 consecutive days. Alternatively, foscarnet can be substituted for ganciclovir if CMV can still be detected in blood. Because neutropenia accompanying ganciclovir administration is usually brief, such patients do not require antifungal or antibacterial prophylaxis (DIII).
True CMV antiviral resistance is quite rare in HCT patients, especially in patients who have never been previously treated with antiviral agents. Increasing antigenemia or CMV DNA load early after initiation of antiviral therapy is usually not a sign of treatment failure in patients who have not been previously treated with antiviral agents, and, therefore, does not necessitate change of therapy 238, 239. However, if the patient develops signs of CMV disease or if the level of antigenemia or the CMV DNA load continues to rise after more than 2 weeks of therapy, resistant CMV should be suspected and a change of therapy considered (BIII). Development of CMV drug resistance early after HCT has been observed in children transplanted for immunodeficiency or those who received T cell-depleted grafts or anti-T cell antibodies [240]. Ganciclovir or foscarnet can be considered as an alternative drug for second-line preemptive therapy (AI) [230]. Cidofovir, a nucleoside analog, can be considered for second-line preemptive therapy, but careful monitoring of renal function is required, and it should be noted that crossresistance with ganciclovir can occur (BII) 241, 242, 243. If possible, samples should be sent to a laboratory capable of documenting antiviral resistance (CIII) [244].
Certain CMV-seropositive autologous recipients are at increased risk for symptomatic CMV replication or disease [245]. These include patients undergoing conditioning regimens including total body irradiation (TBI); patients receiving grafts manipulated to remove T cells; and patients who have recently (eg, within 6 months prior to HCT) received alemtuzumab, Flu, or 2-chlorodeoxyadenosine (CDA). Such patients may benefit from the use of a preemptive strategy that includes monitoring for CMV reactivation until 60 days after HCT (CII). Patients transplanted with CD34+-selected grafts should be treated at any level of antigenemia [245] (BII). Other autologous recipients at high risk who experience moderately high levels of CMV antigenemia (ie, blood levels of ≥5 positive cells/slide) or CMV DNA should receive 2 weeks of preemptive treatment with ganciclovir or foscarnet (CIII) [222]. A prophylactic approach to CMV disease prevention is not recommended for CMV-seropositive autologous recipients (DII) [246].
Because HCT recipients might develop 2 or more reactivations, patients considered to be at increased risk for late CMV disease should be routinely screened for evidence of CMV reactivation as long as substantial immunocompromise persists (BII) [247] (Appendix 1). Risk factors for late CMV disease include allogeneic HCT accompanied by chronic graft-versus-host disease (cGVHD), steroid use, low CD4 counts (<50/mm3), use of grafts from CMV-seronegative donors in CMV-seropositive recipients, and use of unrelated, haploidentical, CB, or T cell-depleted HCTs 248, 249, 250, 251. The indication for antiviral therapy if CMV is detected after day 100 has to be determined on an individual basis depending on the patient's risk factors for developing late CMV disease (BIII). The choice and duration of antiviral therapy are similar to those for CMV infection that occurs during the first 100 days posttransplant.
Strategies for preventing late CMV disease in high-risk patients include the use of continued surveillance and preemptive antiviral therapy [247], as well as prophylaxis with antiviral drugs and cellular immunotherapy for patients with deficient or absent CMV-specific immune system function. Several small phase I/II studies have been published using adoptive transfer of CMV-specific CD4+ and/or CD8+ T cells, especially in patients developing repeated episodes of CMV disease 252, 253, 254, 255. However, none of these adoptive T cell transfer techniques are in routine clinical practice, and, therefore, they cannot be recommended.
Recommendations Regarding Epstein Barr Virus
Preventing exposure
HCT donors and candidates should be tested for the presence of serum anti-EBV IgG antibodies before transplantation to determine risk for primary Epstein-Barr virus (EBV) after HCT. The recommendation is stronger in pediatric patients (AII) than in adults (BII). Although fever and mononucleosis can occur in primary EBV infection, the most significant clinical syndrome associated with EBV replication in HCT recipients, particularly following primary infection, is posttransplant lymphoproliferative disease (PTLD) [256]. This disorder occurs principally in recipients with profound T cell cytopenia (eg, after T cell depletion (TCD), use of anti-T cell antibodies, UCB transplants, and haplo identical transplants) 257, 258, 259. Assessment of blood EBV DNA loads with quantitative PCR testing can identify those at risk for PTLD 260, 261, 262.
EBV disease in HCT recipients typically results from reactivation of endogenous infection or transmission of EBV from the graft [258]. Nevertheless, all transplant candidates, particularly those who are EBV-seronegative, should be advised of behaviors that would decrease the likelihood of EBV exposure (AII) (see Strategies for Safe Living after HCT).
Preventing disease
For prevention of EBV-related PTLD (Appendix 1), it is important to monitor high-risk (eg, after T cell depletion, use of anti-T cell antibodies, UCB transplants, and haplo-identical transplants) patients for EBV DNA load using a blood EBV PCR assay (BII). EBV DNA loads have been shown to rise as early as 3 weeks prior to disease onset. Monitoring for blood EBV DNA loads allows preemptive reduction in immunosuppression, if possible, as the first part of patient management. Because of the variability of PCR techniques and the difference in risk for EBV-related PTLD depending on the degree of T cell lymphopenia, no firm recommendation on the threshold for initiation of preemptive therapy can be made. If there is no response to reduction in immunosuppression, preemptive treatment with rituximab can prevent PTLD (BII) [263]. Infusion of donor-derived, EBV-specific CTL has demonstrated promise in the prophylaxis of EBV lymphoma among recipients of T cell-depleted unrelated or mismatched allogeneic recipients (CII) 264, 265. In addition, expanded donor-derived EBV-specific T cells have been used to control blood EBV DNA loads in this setting, but this procedure remains experimental (CII) 266, 267. Use of B cell depletion to minimize the risk of EBV PTLD has also been proposed (CII) [268]. Finally, prophylaxis or preemptive therapy with currently available antiviral agents is not recommended because of lack of efficacy (DII) 257, 258, 259.
Recommendations Regarding HSV
Preventing exposure
HCT candidates should be tested for serum anti-HSV IgG before transplant (AII); however, type-specific anti-HSV IgG serology testing is not necessary. All HCT candidates, particularly those who are HSV seronegative, should be informed of the importance of avoiding HSV infection while immunocompromised, and should be advised of behaviors that will decrease the likelihood of HSV exposure (AII) (see Strategies for Safe Living after HCT). Any person with disseminated, primary, or severe mucocutaneous HSV disease should be placed under contact precautions for the duration of the illness (AII) [144] to prevent transmission of HSV to HCT recipients.
Preventing disease and disease recurrence
Acyclovir
Acyclovir prophylaxis should be offered to all HSV-seropositive allogeneic recipients to prevent HSV reactivation during the early posttransplant period (AI) 269, 270, 271, 272, 273. The standard approach is to begin acyclovir prophylaxis at the start of the conditioning therapy and continue until engraftment occurs or until mucositis resolves, whichever is longer, or approximately 30 days after HCT (AI) [272] (Appendix 1). Continued use of acyclovir appears to prevent HSV reactivation disease in patients who received it for VZV or CMV prophylaxis (CII) [274]. Routine acyclovir prophylaxis is not indicated for HSV-seronegative HCT recipients, even if the donor is HSV seropositive (DIII). Use of ganciclovir prophylaxis for CMV in HCT recipients is sufficient for prevention of HSV because of this drug's in vitro activity against HSV-1 and HSV-2 (AII) 214, 275, although ganciclovir has not been approved for use against HSV.
Acyclovir-resistant HSV infection occurs mainly in the setting of low-dose prophylaxis, intermittent treatment, or with HSV-seronegative donors 274, 276, 277. Foscarnet is the treatment of choice for resistant disease (BI); cidofovir may serve as an alternative (CIII). If postengraftment acyclovir prophylaxis is given, experts recommend a sufficiently high dose to prevent the emergence of resistance (Appendix 1) [274].
Valacyclovir
Although valacyclovir is not approved for use in preventing HSV disease among HCT recipients, comparative studies have shown that valacyclovir and acyclovir are equally effective in suppression of HSV after autologous HCT 278, 279 for patients who can tolerate oral medications (CIII). Regarding safety, valacyclovir has been used for 1 year in HCT recipients for suppression of VZV without toxicity [280]. Physicians wishing to use valacyclovir among recipients with renal impairment should exercise caution and decrease the dose as needed.
Foscarnet
Because of its substantial renal and infusion-related toxicity, foscarnet is not recommended for routine HSV prophylaxis among HCT recipients (DIII). However, patients who receive foscarnet for other reasons (eg, CMV prophylaxis) do not require additional acyclovir prophylaxis (DIII).
Famciclovir
Presently, data regarding safety and efficacy of famciclovir among HCT recipients are limited; therefore, no recommendations for HSV prophylaxis with famciclovir can be made.
Other recommendations
HSV prophylaxis lasting >30 days after HCT might be considered for persons with frequent recurrences of HSV infection (BIII) (Appendix 1). Acyclovir or valacyclovir can be used during phase I (preengraftment) for administration to HSV-seropositive autologous recipients who are likely to experience substantial mucositis from the conditioning regimen (CIII). Acyclovir prophylaxis doses should be modified for use among children (Appendix 1). Because of limited published data regarding valacyclovir safety and efficacy among children, no recommendations for the pediatric population can be made [281].
Recommendations Regarding VZV
Preventing exposure
HCT candidates should be tested for the presence of serum anti-varicella zoster virus (VZV) IgG antibodies (AII). However, these tests are not 100% reliable, particularly among severely immunosuppressed patients. All HCT candidates and recipients, particularly those who are VZV seronegative, should be informed of the potential seriousness of VZV disease among immunocompromised persons and advised of strategies to decrease their risk for VZV exposure (AII) [282].
Although the majority of VZV disease after HCT is caused by reactivation of endogenous VZV, HCT candidates and recipients who are VZV seronegative, or VZV seropositive and immunocompromised, should avoid exposure to persons with active VZV infections (AII) [283]. Health care workers (HCWs), family members, household contacts, and visitors who are healthy and do not have a reported history of varicella infection or who are VZV-seronegative should receive VZV vaccination before being allowed to visit or have direct contact with an HCT recipient (AIII). Ideally, VZV-susceptible family members, household contacts, and potential visitors of immunocompromised HCT recipients should be vaccinated as soon as the decision is made to perform HCT and the vaccination schedule completed ≈4-6 weeks before the HCT is performed (BIII). To date, no serious disease has been reported among immunocompromised patients from transmission of VZV vaccine virus, and the VZV vaccine strain is susceptible to acyclovir. However, HCT recipients undergoing conditioning therapy should avoid contact with any VZV vaccine recipient who experiences a rash after vaccination (BIII). Rash after vaccination can be because of the wild-type VZV (median: 8 days; range: 1-20 days) or the VZV vaccine strain (median: 21 days; range: 5-42 days) 284, 285.
All HCT recipients with multidermatomal VZV disease should be placed under airborne and contact precautions (AII) [144] to prevent transmission to other HCT recipients. Dermatomal zoster requires contact precautions until all skin lesions are crusted (AII), and some researchers also recommend airborne precautions because in immunocompromised patients there is a high risk for dissemination of the zoster rash (CII). Airborne precautions should be instituted 8 days after exposure to VZV and continued until 21 days after last exposure (AII) or 28 days postexposure if the patient received varicella-zoster immunoglobulin (VZIG) (BII) [144] because a person infected with VZV can be inectious before the rash appears. The VZIG product currently available in the United States is VariZIG [286].
Preventing disease
Antiviral drugs
Long-term acyclovir prophylaxis to prevent recurrent VZV infection is routinely recommended for the first year after HCT for VZV-seropositive allogeneic (BI) 280, 287 and autologous (CII) HCT recipients (Appendix 1). The 1-year regimen of acyclovir is highly effective in reducing the risk of VZV disease during the year of acyclovir administration (BI) 280, 287. Acyclovir prophylaxis may be continued beyond 1 year in allogeneic HCT recipients who have cGVHD or require systemic immunosuppression (BII) 280, 288. The optimal duration of prophylaxis is poorly defined in patients with cGVHD, as there appears to be a persistent risk of VZV reactivation disease even if the acyclovir is continued until all systemic immunosuppressive drugs are discontinued and the CD4+ count exceeds 200 cells/μL [288]. Some clinicians advocate continuing acyclovir prophylaxis until 6 months after discontinuation of all systemic immunosuppressive agents (CIII).
Valacyclovir is a prodrug of acyclovir, and may be used as an alternative to acyclovir at any time that oral medications are used. Valacyclovir may provide higher drug levels in severely immunosuppressed patients than acyclovir (BII). Although valacyclovir is not licensed in the United States for use in HCT recipients, a large randomized trial in HCT recipients found no safety issues with valacyclovir, even when used at very high doses [216]. No data on famciclovir in HCT recipients were found; consequently, no recommendations can be made regarding its use in place of acyclovir or valacyclovir.
Resistance to acyclovir has been rarely documented to date in HCT recipients [289]; however, when clinically suspected or virologically documented acyclovir resistance occurs among patients, HCT physicians should use foscarnet for preemptive treatment of VZV disease (BIII) 289, 290.
Any HCT recipient or candidate undergoing conditioning therapy who experiences a VZV-like rash (whether after exposure to a person with wild-type varicella or shingles or exposure to a VZV vaccinee with a rash) should receive preemptive intravenous acyclovir until ≈2 days after all lesions have crusted (BII). Treatment may be completed with oral valacyclovir if the patient can tolerate oral medication.
Passive immunization and VZV-seronegative HCT recipients
Because of the high morbidity of VZV-associated disease among severely immunocompromised HCT recipients and until further data are published, there are situation-specific indications for the administration of VZIG or VariZIG, where available, for VZV-seronegative HCT recipients. Immunocompromised HCT recipients (ie, an allogeneic patient <24 months after HCT, ≥24 months after HCT and on immunosuppressive therapy, or having cGVHD) should receive VZIG or VariZIG as soon as possible and no later than 96 hours after close or household contact with a person having either chickenpox or shingles (AII). Postexposure acyclovir or valacyclovir (Appendix 1) may be used as an alternative if VZIG or VariZIG is not available (CII) 291, 292. VZIG or VariZIG, acyclovir, or valacyclovir should also be administered to all VZV-seronegative HCT recipients undergoing conditioning therapy who are exposed to a VZV vaccinee having a varicella-like rash (BIII). If VZV-seronegative HCT recipients undergoing conditioning therapy are closely exposed to varicella >3 weeks after receiving VZIG or VariZIG, they should receive another dose of VZIG or VariZIG, or another course of valacyclovir if VZIG or VariZIG is not available (BIII) [282].
Passive immunization and VZV-seropositive HCT recipients
VZIG or VariZIG, acyclovir, or valacyclovir can be used following VZV exposure, including exposure to a VZV vaccinee having a varicella-like rash, for HCT recipients who were VZV-seropositive before HCT and are highly immunosuppressed (ie, because of high-dose steroid therapy, or T CD) (CIII) [292]. These recommendations are made because the vaccinee might be unknowingly incubating wild-type varicella, particularly during the first 14 days after varicella vaccination, and because vaccine-strain VZV has been rarely transmitted by VZV vaccinees with postvaccination vesicular rashes [282]. Furthermore, varicella vaccination is only approximately 85% effective. Thus, vaccine recipients may still become infected with wild-type virus years after vaccination [293] and may thus be a source of transmission to immunocompromised patients.
VZV Vaccines
Use of VZV vaccines (Varivax and Zostavax) is discussed in the HCT Recipient Vaccination section. A vaccine-associated rash occurs in approximately 1% to 5% and 0.5% of recipients of the varicella and zoster vaccine, respectively 294, 295. This rash is a potential source of transmission of vaccine virus strain to HCT recipients. Because the risk of vaccine virus transmission is low, particularly in the absence of a vaccine-associated rash, household members should receive varicella vaccine to protect HCT recipients from potential exposure to wild-type disease (AIII). Individuals who experience a vaccine-associated rash should avoid close contact with HCT recipients in the home setting (BIII). If contact occurs, the HCT recipient should be considered for postexposure prophylaxis with valacyclovir, as outlined earlier (CIII).
An inactivated VZV vaccine has been used investigationally among HCT recipients [296]. Studies are ongoing to further define its utility and no recommendation regarding its use can be made at this time.
Other recommendations
Recommendations for VZV prevention are the same for allogeneic or autologous recipients. Recommendations are also the same for allograft recipients with different-intensity conditioning regimens. Recommendations for preventing VZV disease among pediatric or adult HCT recipients are the same, except that appropriate dose adjustments for acyclovir derivatives and VZIG should be made for pediatric HCT recipients (AIII) (Appendix 1).
Recommendations Regarding CRV Infections: Influenza, Respiratory Syncytial Virus, Human Metapneumovirus, and Parainfluenza Virus
Preventing exposure
Preventing CRV exposure is critical in preventing CRV disease 297, 298. Measures for preventing nosocomial CRV transmission are presented in the Infection Prevention and Control in Health care Facilities: Recommendations Regarding CRV Infections section. Use of PCR testing in donors with respiratory infections remains investigational (CIII). Viral cultures of asymptomatic HCT candidates are unlikely to be useful. Whether multiplex PCR testing can identify asymptomatic shedders before HCT is presently being studied. PCR-based routine surveillance of asymptomatic patients after HCT remains investigational.
HCT recipients with symptoms of an upper respiratory infection (URI) or lower respiratory infection (LRI) should be placed under contact precautions to avoid transmitting infection to other HCT candidates and recipients, HCWs, and visitors until the etiology of the illness is identified (BIII) [144]. Optimal isolation precautions should be modified as needed after the etiology is identified (BIII). HCT recipients and candidates, their family members and visitors, and all HCWs should be informed regarding CRV infection control measures and the potential severity of CRV infections among HCT recipients (BIII) 297, 298, 299.
Preventing disease
HCT physicians should determine the etiology of a URI in an HCT recipient, if possible, because respiratory syncytial virus (RSV), influenza, parainfluenza, and adenovirus URIs can progress to more serious LRI, and certain CRVs can be treated (BIII). Appropriate diagnostic samples include nasopharyngeal washes, swabs or aspirates; throat swabs (in combination with nasal samples); and bronchoalveolar lavage (BAL) fluid. HCT candidates with URI symptoms at the time conditioning therapy is scheduled to start should postpone their conditioning regimen until the URIs resolve, if possible, because certain URIs might progress to LRI during immunosuppression (BIII) 298, 300, 301, 302. The clinical relevance of recently discovered viruses (eg, human bocavirus, non-SARS coronaviruses, human rhinoviruses, human metapneumovirus) that are detectable by molecular methods is currently undefined, and no recommendations can be made for routine screening for these viruses (CIII).
Recommendations regarding influenza
Life-long seasonal influenza vaccination with the trivalent inactivated vaccine is recommended for all HCT candidates and recipients (see Vaccination section) (AII). Additionally, influenza vaccination of family members and close or household contacts is strongly recommended during each influenza season (eg, October-May in the Northern Hemisphere), starting the season before HCT and continuing ≈24 months after HCT (AII) [303] to prevent influenza. All family members and close or household contacts of HCT recipients should continue to be vaccinated annually as long as the HCT recipient's immunocompromise persists, even if beyond 24 months after HCT (AII) [303]. Seasonal influenza vaccination is strongly recommended for all HCWs of HCT recipients (AI) [304].
If HCWs, family members, or other close contacts of HCT recipients receive influenza vaccination during an influenza outbreak, they should receive chemoprophylaxis, if feasible, for 2 weeks after influenza vaccination (BI) while the immunologic response to the vaccine develops. However, if an outbreak occurs with an influenza strain that is not contained in the available influenza vaccine, all healthy family members, close and household contacts, and HCWs of HCT recipients and candidates should receive influenza chemoprophylaxis with an active agent against the current circulating strain of influenza until the end of the outbreak (BIII) [303]. Zanamivir may be given for prevention of influenza A and B, including influenza from strains resistant to oseltamivir. The duration of prophylaxis depends on the type of exposure. Zanamivir can be administered to persons 5 years of age and older for prevention of influenza, and 7 years and older for treatment of influenza. Oseltamivir can be administered to persons ≈1 year of age and older. Patients with influenza should be placed under droplet and standard precautions (AIII) to prevent transmission of influenza to HCT recipients. HCWs with influenza should be excused from patient care until they are no longer infectious (AIII).
HCT recipients <6 months after HCT should receive chemoprophylaxis with neuraminidase inhibitors during community influenza outbreaks that lead to nosocomial outbreaks (AII). During community outbreaks, all HCT recipients who have not yet received a current influenza vaccination should be vaccinated against influenza immediately if it is more than 4 months after HCT (BIII). Additionally, to allow sufficient time for the patient to experience an immunologic response to influenza vaccine, chemoprophylaxis can be used for these HCT recipients for 2 weeks after vaccination during a nosocomial or community influenza outbreak (CIII). Influenza chemoprophylaxis has been recommended for all influenza-exposed HCT recipients who are <24 months after HCT or who are >24 months after HCT and substantially immunocompromised regardless of vaccination history, because of their likely suboptimal immunologic response to influenza vaccine (BII) 305, 306. Drug resistance patterns of circulating influenza strains should guide the choice of prophylactic agent.
Healthy children who receive influenza vaccination for the first time might not generate protective antibodies until 2 weeks after receipt of the second dose of influenza vaccine. Therefore, during an influenza A outbreak, pediatric recipients who are <9 years old, ≤6 months after HCT, and receiving their first influenza vaccination, should be given ≈6 weeks of influenza A chemoprophylaxis after the first dose of influenza vaccine (BIII) (Appendix 1) 307, 308. To prevent severe disease, HCT patients with influenza URI should receive early preemptive therapy with drugs shown to be susceptible to the circulating strain. (AII) 309, 310.
Recommendations regarding RSV
Respiratory secretions of any hospitalized HCT candidate or recipient who experiences signs or symptoms of CRV infection should be tested promptly by viral culture and rapid diagnostic tests for RSV (BIII). If 2 diagnostic samples taken ≈2 days apart do not identify a respiratory pathogen despite persistence of lower respiratory symptoms, BAL and further testing are advised (BIII). This testing is critical because of the high morbidity and case fatality of RSV disease and the frequent presence of significant copathogens among HCT recipients when it occurs during the peritransplant period 311, 312. HCT recipients, particularly those who are preengraftment and lymphopenic or those who have preexisting obstructive airway disease, are at highest risk for severe RSV pneumonia. These patients should have their illness diagnosed early (ie, during RSV URI) and receive aggressive treatment to prevent fatal RSV disease (BIII).
On the basis of retrospective studies as well as a prospective trial with inadequate accrual, some researchers recommend preemptive aerosolized ribavirin for patients with RSV URI, especially those with lymphopenia (during the first 3 months after HCT) and preexisting obstructive lung disease (late after HCT) (CIII) 310, 313. Although a definitive, uniformly effective preemptive therapy for RSV infection among HCT recipients has not been identified, certain other strategies have been proposed, including systemic ribavirin 314, 315, 316, RSV antibodies (ie, passive immunization with high-RSV-titer IVIG, RSV immunoglobulin) in combination with aerosolized ribavirin 301, 317, and RSV monoclonal antibody (mAb) 314, 318. No randomized trial has been completed to test the efficacy of these strategies; therefore, no specific recommendation regarding any of these strategies can be given at this time. To prevent RSV disease, some centers provide monthly palivizumab prophylaxis during RSV season (eg, November-April in the Northern Hemisphere) for pediatric recipients at risk for primary RSV disease (ie, children <4 years old) (CIII). Routine IVIG therapy would still be required if indicated [319] (ie, for those with hypogammaglobulinemia) (Appendix 1).
Recommendations regarding parainfluenza virus
Immunoprophylaxis, withdrawal of immunosuppression to prevent progression, and preemptive treatment for parainfluenza virus infections among HCT recipients have been proposed [320]. However, no recommendation can be made in these guidelines because of insufficient data. No commercially licensed drugs or vaccines against parainfluenza viruses are currently available.
Recommendations regarding human metapneumovirus
Human metapneumovirus can cause pneumonia in HCT recipients [321]. Ribavirin has in vitro and animal model activity against human metapneumovirus 322, 323. However, no recommendations can be made in these guidelines because of lack of treatment data.
Other disease prevention recommendations
The recommendations for preventing CRV infections and their recurrence are the same for allogeneic or autologous recipients. Generally, these recommendations apply to children 308, 319, 324, 325 and adults, but with appropriate adjustments in antiviral drug and influenza vaccine doses for children (Appendix 1). Progression to lower tract disease appears to be less common in HCT recipients with nonmyeloablative (NMA) conditioning, but no specific recommendations for these patients can be made at this time.
Recommendations Regarding Adenoviruses
Preventing exposure
Adenovirus infections after HCT can result from reactivation or de novo acquisition. Because many different serotypes exist and knowledge about crossreactive immunity is limited, pretransplant serologic testing of either the patient or the donor is likely not helpful. Because cellular immune responses are crossreactive across various serotypes and are likely to provide long-term protection against adenovirus reactivation, serious adenovirus infections in adults are uncommon 326, 327. HCT recipients should follow similar preventive measures as for other respiratory or enteric viruses (see recommendations regarding CRV, CMV, and enteric viruses) with regard to contact and nosocomial spread (see Infection Prevention and Control in Health care Facilities and Safe Living after HCT).
Preventing infection and disease
HCT patients can be stratified according to their risk for adenoviral disease:
- •
-
•
Intermediate risk—T cell replete, related-donor allograft recipients without GVHD
-
•
Higher risk—recipients of T cell-depleted (2-3 log10), related- or unrelated-donor transplants 331, 332; HLA mismatched transplant recipients other than allele DRB1 mismatch [333]; patients with GVHD who are receiving systemic steroids 328, 334, 335, and pediatric recipients.
-
•
Highest risk—refractory GVHD, UCB transplantation, haploidentical transplantation, stem cell graft T cell depletion of >2-3 log10, use of anti-T cell antibodies (eg, antithymocyte globulin [ATG], alemtuzumab) 331, 336.
For patients at highest risk, weekly monitoring for active adenovirus infection by PCR for either the first 6 months after HCT or the duration of severe immunosuppression/lymphopenia could be considered (CII) 333, 337. Quantitative PCR testing should be strongly considered for monitoring progression of adenovirus infection and response to treatment (BII). There are no definitive data on a critical value for viral load in peripheral blood to indicate initiation of intervention; thus, no recommendation can be made.
Clearance of adenovirus has been shown to be associated with recovery of adenovirus-specific T cell immunity 338, 339. When possible, rapid tapering or withdrawal of immunosuppression constitutes the best way to prevent progression of adenovirus infection (AII) 331, 332, 340, 341. However, this strategy might not always be feasible in severe GVHD or with severe lymphopenia because of use of anti-T cell antibodies or T CD of the graft. Few antiviral agents have in vivo activity against adenoviruses, and no randomized, placebo-controlled study of antiviral drug therapy for adenoviral infection has been performed. The available data suggest that cidofovir or ribavirin could be used as preemptive antiviral therapy of adenoviral disease in selected high-risk HCT patients (CII) (Appendix 1). A reduction of DNA load has been shown mainly with cidofovir 342, 343 but the evidence of its efficacy in preventing mortality in HCT patients is inconsistent [344]. Differences in responses may be because of strain-specific susceptibilities [345]. The duration of preemptive therapy is subject to tolerance and clearance of viral load.
Current evidence strongly supports the role of adenovirus-specific T cells in controlling the progression of adenoviral disease [346]. However, this approach is at an early stage of development and should not be used outside the context of a clinical trial.
Recommendations Regarding Polyomaviruses BK and JC
Human polyomavirus type I, commonly called BK virus (BKV), and human polyomavirus type II, commonly called JC virus (JCV), infect 50% to 90% of humans worldwide before the age of 10 years, without known symptoms or signs 347, 348. Urinary shedding of BKV and/or JCV occurs in 5% to 20% of healthy immunocompetent blood donors [349]. BKV and JCV are nonenveloped virions found in urban sewage and fairly resistant to environmental inactivation [350].
Polyomavirus disease in HCT patients most often corresponds to secondary BKV replication with impaired polyomavirus-specific cellular immunity. Urinary shedding of BKV occurs in 60% to 80% of HCT recipients 347, 348, 351, 352, 353. The major disease linked to high-level polyomavirus replication is BKV-associated hemorrhagic cystitis (PVHC), which affects 5% to 15% of HCT recipients at 3 ro 6 weeks posttransplant 352, 354. PVHC occurs typically after engraftment, and must be distinguished from hemorrhagic cystitis caused by other pathogens (eg, adenovirus or CMV) and from early-onset hemorrhagic cystitis, which arises preengraftment and has been linked to urotoxic conditioning regimens with cyclophosphamide (Cy), ifosfamide, busulfan (Bu), and/or TBI 352, 354. BKV viruria reaching high viral loads of >107 genome equivalents/mL (geq/mL) is observed in 20% to 80% of HCT patients, but less than one-fifth of HCT recipients develop PVHC [354]. PVHC is diagnosed in HCT patients with postengraftment cystitis who have pain and urinary urgency together with hematuria of grade II (macrohematuria [352]) or higher, high-level BKV replication (ie, ≥107 gEq/mL), and exclusion of other pathogens. There are reports of sporadic cases of JCV-associated PVHC [355], BKV- or JCV-associated polyomavirus nephropathy (PVAN) 347, 356, 357, 358, 359, and JCV- or BKV-mediated polyomavirus multifocal leukoencephalopathy (PVML) 360, 361.
Preventing exposure
There is no evidence to support routine testing of HCT recipients or donors for the presence of BKV-specific or JCV-specific antibodies (DIII). There are no commercially available, standardized, or FDA-approved assays to measure BKV- or JCV-specific antibodies. The role of primary infection, of donor-recipient mismatch, and of BKV-specific antibody titers in HCT recipients is presently unknown.
There is no evidence to support specific infection-control measures for HCT patients with BKV viruria (DIII). In patients with disseminated BKV replication involving the respiratory and the gastrointestinal (GI) tract, separation from other patients with significant immunodeficiency should be considered (CIII).
Preventing disease and disease recurrence
There is no evidence to support the use of quinolones or cidofovir as specific universal prophylaxis for PVHC or other polyomavirus-associated complications (DIII). There is insufficient evidence to support the use of quinolones for preemptive treatment of asymptomatic HCT patients who develop BKV viruria or viremia (DIII). Fluoroquinolones can inhibit BKV replication in tissue culture, and have been reported to reduce BKV loads in HCT patients, but a significant reduction of PVHC has not been demonstrated [362]. Ciprofloxacin and levofloxacin are frequently used, alone or in combination with other antibiotics, in patients undergoing HCT in antibacterial prophylaxis during neutropenia, in empiric or specific antibiotic therapy, and seemingly resistant BKV isolates have been reported 362, 363. There is no evidence to support the use of cidofovir for preemptive treatment of asymptomatic HCT patients who develop BKV viruria or viremia (DIII). Cidofovir has been administered intravenously in a low dose (ie, up to 1 mg/kg 3 times weekly, without probenecid) or a high dose (ie, 5 mg/kg per week with probenecid) to HCT patients with PVHC, but no randomized trials are available proving its clinical efficacy (CIII).
Recommendations Regarding Hepatitis A virus (HAV)
The seroprevalence of HAV varies widely, with higher rates in resource-limited societies. Testing of HCT candidates or donors for HAV IgG antibodies is generally not recommended, as its sole positivity in the absence of IgM indicates remote exposure and has no impact on HCT outcome (DIII). However, testing for IgM is indicated as part of the workup of patients with signs of acute hepatitis (AII). If an HCT candidate tests positive for HAV IgM, transplantation should be delayed because of an increased risk of sinusoidal obstruction syndrome (SOS) following liver-toxic myeloablative (MA) conditioning regimens (DII). If the HCT donor tests positive for HAV IgM, transplantation should be delayed because of a high risk of transmission and increased morbidity and mortality (EII). HAV vaccination recommendations for HCT recipients are provided in Table 7.
Table 7.
Vaccinations Considered Optional or Not Recommended for Both Autologous and Allogeneic HCT Recipients
| Optional | ||
|---|---|---|
| Vaccine | Recommendations for use | Rating |
| Hepatitis A |
|
CIII |
|
Limited data regarding safety and efficacy. |
|
| Human papillomavirus |
|
CIII |
| Yellow fever (live) |
|
|
| Rabies |
|
CIII |
| Tick-borne encephalitis (TBE) |
|
CIII |
| Japanese B encephalitis |
|
CIII |
| Not Recommended | ||
|---|---|---|
| Vaccine | Recommendations for use | Rating |
|
Contraindicated for HCT recipients | EII |
| Oral poliovirus vaccine (live) | Should not be given to HCT recipients since an effective, inactivated alternative exist | EIII |
| Intranasal influenza vaccine (live) |
|
EIII |
| Cholera | No data were found regarding safety and immunogenicity among HCT recipients | DIII |
| Typhoid, oral (live) | No data were found regarding safety and immunogenicity among HCT recipients. | EIII |
| Typhoid (intramuscular) | No data were found regarding safety, immunogenicity, or efficacy among HCT recipients. | DIII |
| Rotavirus | Must be given before 12 weeks of age to be safe. | EIII |
|
No data regarding safety among HCT recipients. | EIII |
HCT indicates hematopoietic cell transplantation; GVHD, graft-versus-host disease.
Current Advisory Committee on Immunization Practices (ACIP) and American Academy of Pediatrics guidelines for postexposure human rabies immunoglobulin and vaccine administration should be followed, which include administering 5 doses of rabies vaccine administered on days 0, 3, 7, 14, and 28 postexposure.
Recommendations Regarding Hepatitis B virus (HBV)
HBV can cause severe hepatitis following HCT. However, rates of HBV-associated cirrhosis and hepatocellular carcinoma do not appear higher in HCT patients than in non-HCT patients [364]. Severe hepatitis B has been observed in HCT recipients in the following situations:
-
•
1. HBV-naïve HCT recipients exposed to HBV via an infected donor, infected blood products, or through sexual contact;
-
•
2. HCT recipients with chronic hepatitis B experiencing prolonged immune suppression;
-
•
3. HCT recipients with serologic evidence of resolved HBV infection who have reverse seroconversion following prolonged immune suppression;
-
•
4. HCT patients—generally in countries with endemic HBV—with latent occult hepatitis B (all serologic markers negative) that activates following prolonged immune suppression [365].
Risk factors for reactivation and exacerbation of HBV replication in HCT recipients include treatment with high-dose steroids 366, 367, Flu/rituximab [368], or alemtuzumab 366, 367, 369. Clinical hepatitis may become further exacerbated during immune recovery and discontinuation of immunosuppression.
Preventing exposure
Testing both recipients and potential donors for evidence of active or past HBV infection is critical to preventing HBV exposure and disease in HCT recipients. The appropriate assays include HBV surface antigen (HBsAg), antibodies to HBV surface antigen (anti-HBs), and antibodies to HBV core antigen (anti-HBc) (AII). All anti-HBc positive and HbsAg-positive donors and recipients should also be tested for HBV DNA (AIII). HBV-naïve HCT candidates should not receive transplants from HBsAg-positive or HBV DNA-positive donors, if another equally suitable donor is available (AII). However, the use of a donor with active HBV replication is not absolutely contraindicated for an HBV-naïve recipient because viral transmission is not universal (BIII). The overriding concern must be HLA matching and other outcome-related issues.
Vaccination of all HBV-naïve HCT candidates should be considered (AIII). An attempt should be made to provide hepatitis B immunization to HBV-seronegative HCT candidates with HBsAg-positive donors, preferably prior to chemotherapy for the initial 2 doses 3 to 4 weeks apart and a third dose 6 months later, ideally prior to HCT (BIII). If this schedule cannot be met, the third dose should be administered a few months after completion of chemotherapy. Of note, the response to vaccination is likely to be poor in patients undergoing chemotherapy. If the postvaccination anti-HBs titer is <10 IU/L or pretransplantation vaccination is impractical, HBIG (0.06 ml/kg) should be administered immediately prior to infusion of stem cells (AIII). HCT recipients who fail to respond to pretransplant vaccination but remain uninfected posttransplant (ie, negative for HBsAg, anti-HBc, anti-HBs, and HBV DNA) should be revaccinated after immune recovery with 1 to 3 doses of hepatitis B vaccine (BII).
If the donor has a detectable HBV DNA load, the following measures are recommended (BIII):
-
•
Administer antiviral treatment to the donor for at least 4 weeks, or until HBV DNA is undetectable (if time permits). Many experts prefer entecavir for this purpose (CIII).
-
•
Reduce harvest volume to the minimum possible without compromising planned CD34+ cell dose and test an aliquot of the cell product for HBV DNA.
-
•
If, at the time of harvest, both the donor and the harvested cells are HBV DNA-negative, monitor alanine aminotransferase (ALT) levels monthly in the recipient for the first 6 months. If the ALT level is increasing, test the recipient for HBV DNA (AIII). If HBV DNA testing is not available, testing for HBsAg is an acceptable alternative. If HBV DNA is detected or a positive HBsAg is identified, antiviral treatment is indicated (AIII).
-
•
If the donor or cell product is positive for HBV DNA at the time of harvest, provide the HCT recipient prophylaxis with lamivudine from day zero through at least 6 months following discontinuation of immunosuppressive drugs. Consider administration of a second dose of HBIG at 4 weeks after transplantation (AIII). Monitor ALT and HBV DNA monthly. If HBV DNA is positive during lamivudine prophylaxis, treatment may be modified as detailed later.
If the donor is anti-HBc positive, but negative for both HBsAg and anti-HBs, the donor should be tested for HBV DNA (BIII). If the HBV DNA assay is positive, treat as described in the bullets earlier for a donor with a detectable HBV viral load (BIII). If HBV DNA testing is negative, repeat testing of the donor at the time of harvest should be considered. If HBV DNA remains negative, HCT is performed without further precaution (DIII). If the repeat test is positive, treat as described before (BIII).
HBV-naïve HCT recipients who are in a monogamous relationship with a known HBsAg carrier, or who are sexually active and not in a long-term monogamous relationship, should always use latex condoms during sexual contact to reduce their risk of primary HBV infection (AIII) [370].
Preventing disease
For HCT candidates with evidence of past exposure to HBV (ie, who are anti-HBc positive), the specific recommendations depend on the pattern of test results. Because the risk of post-HCT hepatitis is reduced via adoptive transfer of a donor's natural immunity, equally suitable donors demonstrating natural immunity (anti-HBs positive, anti-HBc positive) are preferred over donors without natural immunity for recipients with evidence of prior HBV exposure [364].
If the HCT recipient is anti-HBc positive and anti-HBs positive, the risk of HBV reactivation is considered low during chemotherapy/conditioning, but higher following prolonged treatment with prednisone for GVHD. Serum ALT should be followed in such patients, and if the level increases, HBV DNA load should be assessed. Patients with a positive HBV DNA load should receive preemptive antiviral treatment as described later (AIII). Prophylactic antiviral treatment may be considered for anti-HBc positive and anti-HBs positive recipients before, and for 1 to 6 months after, HCT (CIII). Additionally, anti-HBs levels should be monitored every 3 months. Reduction in anti-HBs titer should prompt HBV DNA testing (BIII) [371]. Patients who lose anti-HBs responses, but have no HBV DNA in the serum, should receive active immunization in an attempt to restore protective levels of anti-HBs (BIII). Patients with positive HBV DNA assays should receive antiviral therapy (AIII). The duration of antiviral treatment in this setting has not been studied, but a common practice is to continue therapy for at least 6 months following discontinuation of immune suppressive drugs (BIII) 372, 373, 374. Rebound HBV replication and clinical hepatitis may follow discontinuation of antiviral treatment and should be monitored by regular measurement of ALT and HBV DNA (eg, biweekly) (BIII).
HCT candidates with evidence of active HBV replication (HBsAg positive and/or HBV DNA positive) should have a liver biopsy prior to HCT, because preexisting biopsy-proven cirrhosis and hepatic fibrosis can increase treatment-related morbidity and mortality (TRM) (BIII). Antiviral therapy should be initiated prior to conditioning. If HCT is not urgent, antiviral treatment should be administered for 3 to 6 months prior to conditioning. In patients with persisting HBV DNA while on therapy with lamivudine, treatment may be modified as detailed later. Rebound HBV replication and fulminant hepatitis may occur following discontinuation of antiviral treatment. Therefore, discontinuation of antiviral therapy should be performed judiciously, with frequent monitoring of liver function and HBV DNA (BIII).
HCT candidates who are positive for anti-HBc but negative for HBsAg and anti-Hbs should be tested for HBV DNA. If HBV DNA is undetectable, the patient should receive HBV vaccination as described before and proceed to HCT. Further management regarding monitoring and antiviral treatment should be performed as described for anti-HBc-positive and anti-HBs-positive HCT candidates (BIII). If HBV DNA is positive, proceed to HCT with preemptive antiviral therapy as described above.
Antiviral treatment
Therapy should be coordinated between the HCT physician and infectious disease and/or hepatology specialist(s) with expertise in chronic viral hepatitis. Lamivudine (100 mg/day) is the first choice for antiviral therapy (AI). Antiviral therapy should be continued for at least 6 months posttransplant in autologous HCT patients, for 6 months following discontinuation of immune suppressive drugs in allogeneic HCT patients, and longer in patients receiving immunosuppression for cGVHD (BIII). Extended use of lamivudine not only results in reduction of HBV reactivation and the resultant complications, but also lowers HBV-associated mortality 375, 376.
Recommendations Regarding Hepatitis C Virus (HCV)
HCT from donors who are HCV-RNA-positive invariably transmits HCV to uninfected recipients, with development of viremia in the immediate posttransplantation period [377]. Conversely, the risk of transmission is decreased if HCV-RNA is undetectable at the time of hematopoietic cell donation [377]. There is no evidence of adverse short-term effects of HCV infection in HCT recipients, and HCV-infected HCT patients have similar morbidity up to 10 years after transplant [378]. Subsequently, however, these patients are at risk for progression to cirrhosis, which may occur more rapidly than in non-HCT patients. The cumulative incidence of biopsy-proven cirrhosis is 11% and 24% at 15 and 20 years posttransplant, respectively, with a median of 18 years compared with 40 years for non-HCT HCV-infected patients [379]. Extrahepatic manifestations and infection with HCV genotype 3 increase the risk for progression.
Preventing exposure
All HCT candidates should be assessed for the risk of HCV infection with a careful history, physical examination, and serum ALT testing (BIII), as well as measurement of anti-HCV antibody titers (AII). Even if anti-HCV titers are negative, nucleic acid testing for HCV RNA should be undertaken in patients whose history indicates increased risk for HCV infection (eg, transfusion with blood not tested for HCV-reliable testing of the blood supply began in 1992 in developed countries; i.v. or inhaled drug abuse; or tattoos) or who have an unexplained elevation of serum ALT (AII) [380].
HCV-infected patients requiring HCT and for whom there is no alternative donor can proceed with HCT from an HCV-positive donor, provided they have full understanding of the long-term side effects (BIII) [378]. The donor should be assessed for chronic liver disease and other extrahepatic manifestations of HCV, which might contraindicate donation (EIII).
Similar to recipients, all donors should be screened for anti-HCV antibodies, and those found to be anti-HCV-positive or at high risk for HCV infection should be tested for HCV-RNA (AII). If feasible, viral clearance with standard combination antiviral therapy prior to stem cell harvest may be attempted in donors with detectable HCV RNA. However, there are few reports on success of this method 381, 382. Both the donor and recipient should be counseled on individual risks (BIII).
Preventing disease progression
All HCT candidates with HCV infection must be assessed for evidence of chronic liver disease. To assess the risk of conditioning and HCT, liver biopsy is warranted in the following clinical situations (AIII):
-
•
Associated iron overload
-
•
History of excessive alcohol intake
-
•
History of hepatitis C for >10 years
-
•
Clinical evidence of chronic liver disease.
Patients with evidence of cirrhosis or hepatic fibrosis should not be considered for conventional MA conditioning therapy that contains either Cy or TBI ≥12 Gy (DIII), because those regimens are associated with a 9.6-fold increased risk of fatal SOS in these patients [378]. Instead, regimens that do not contain either Cy or TBI, which pose a lower risk for fatal SOS, should be used [383]. For patients with cirrhosis, however, even an RIC regimen poses a mortality risk [384].
Treatment for chronic HCV should be considered in all HCV-infected HCT recipients, because limited data suggest improved outcome in those who respond to combination therapy (BIII) [385]. To qualify for antiviral treatment, the patient must be in complete remission from the original disease, be ≥2 years posttransplant without evidence of either protracted aGVHD or cGVHD, have been off immunosuppression for 6 months, and have normal blood counts and serum creatinine (BIII). Treatment should consist of full-dose peginterferon and ribavirin (BIII). Dose modifications should be made if intolerance develops (eg, development of cytopenias). In survivors whose neutrophil and platelet counts are below normal at baseline, daily interferon alpha can be substituted for peginterferon to assess hematologic toxicity before moving to peginterferon. Treatment should be continued for 24 to 48 weeks, depending on response.
Human Herpesviruses 6 and 7
Preventing exposure
Human herpesvirus 6 (HHV-6) is the cause of the classic childhood illness roseola, which is also known as exanthema subitum or sixth disease [386]. Clinical disease associated with human herpesvirus 7 (HHV-7) infection remains to be defined. Nearly all children are infected with HHV-6 by 2 to 3 years of age 387, 388, 389 and HHV-7 by the age of 5 years [390].
Preventing disease and disease recurrence
The spectrum of HHV-6-associated complications after HCT has not been completely described. HHV-6 reactivation is common during the early allogeneic HCT transplant period, with viremia occurring in approximately 40% to 60% of patients 387, 391, 392, 393, 394. The clinical significance of detection of HHV-6 viremia is unknown, although it has been associated in the posttransplant setting with hepatitis, fever, rash, idiopathic pulmonary syndrome, and delayed platelet and monocyte engraftment. HHV-6 can also be chromosomally integrated, potentially resulting in a false-positive PCR assay result [395]. A posttransplantation acute limbic encephalitis syndrome associated with HHV-6 reactivation in the cerebral spinal fluid (CSF) has been reported [396]. This syndrome is uncommon, occurring in approximately 1% to 2% of HCT patients in some series. It typically occurs 1 to 2 months posttransplantation and appears to be more common after receipt of a UCB or HLA-mismatched graft. Manifestations include profound memory loss, seizures, hyponatremia, mild CSF pleocytosis, and significant mesial temporal lobe abnormalities on magnetic resonance imaging (MRI) 396, 397. In addition, HHV-6 may interfere with MHC class I antigen presentation and augment local immunosuppression [398]; however, the implications of this viral property are unknown. The role of HHV-7 in posttransplant complications remains to be defined.
At this time, there are no data to guide preemptive monitoring or a prophylactic antiviral strategy to prevent potential HHV-6-associated disease (DIII). Ganciclovir, cidofovir, and foscarnet have variable in vitro activity against HHV-6, and may have a role in treating HHV-6-associated disease 399, 400. There are no data to support recommendations for monitoring of potential HHV-7-associated disease (DIII).
Human Herpesvirus 8
Preventing exposure
Human herpesvirus type 8 (HHV-8) is the cause of Kaposi's sarcoma, and is also known as KSHV. Unlike other herpesvirus infections, HHV-8 infection is not ubiquitous. There is significant geographic variability in prevalence of HHV-8 infection, with high infection rates reported in sub-Saharan Africa (50%), modest rates in the United States (about 5%), and low rates in Japan (<1%). In the United States, higher rates of HHV-8 infection have been identified in men who have sex with men, perhaps indicating sexual transmission or enhanced transmission through saliva.
Preventing disease and disease recurrence
HHV-8-associated disease (eg, Kaposi's sarcoma) occurs only rarely after HCT 401, 402, 403, 404. At this time, there are no data to guide monitoring or preemptive antiviral treatment for posttransplant HHV-8-associated disease (DIII).
Human Immunodeficiency Virus
In patients with HIV and receiving HAART therapy, cancer is now the leading cause of death [405]. Consequently, HCT may need to be considered for these patients.
Preventing exposure
As described in the Hematopoietic Cell Graft Safety section, regulations are in place in both the United State and the European Union for evaluation of donors to minimize risk of transmissible diseases 78, 79, 80, 82, 83. Using a related donor with known HIV harbors significant risks that likely outweigh any benefits of transplantation to a seronegative recipient, and should not be considered (DIII).
Preventing disease and disease recurrence
Patients with HIV and a malignancy treated by transplantation should not automatically be excluded from this potentially life-saving therapy. Reports of autologous transplantation suggest that this is a feasible approach in patients with controlled HIV disease (BIII) 406, 407, 408, 409, 410. Outcomes after allogeneic transplantation are quite limited, although 1 report suggests that in the current era of HAART therapy, this may be considered (CIII) [411]. Because of significant complexity in the management of HIV-positive patients, it is recommended that any HIV-positive patient considered for HCT be enrolled on a clinical trial and the patient be comanaged with an HIV specialist.
Fungal Infection Prevention after HCT
K.A. Marr, E. Bow, T. Chiller, G. Maschmeyer, P. Ribaud, B. Segal, W. Steinbach, J.R. Wingard, M. Nucci
General Recommendations
Preventing exposure
Hematopoietic cell transplantation (HCT) recipients and candidates undergoing conditioning therapy should avoid substances, including certain foods (Table 5 ), which increase the risk of exposure to fungi (DIII).
Table 5.
Foods That Hematopoietic Cell Transplant (HCT) Recipients May Eat (BIII) or Should Avoid (DIII) Until Having Reached the Milestone of Live Virus Vaccination following HCT
| Food Groups | May Eat (BIII) | Do Not Eat (DIII) |
|---|---|---|
| Dairy |
|
|
| Meat and meat substitutes |
|
|
| Fruits and nuts |
|
|
| Entrees, soups | ·All cooked entrees and soups | ·All miso products (such as miso soup and miso paste) |
| Vegetables |
|
|
| Bread, grain, and cereal products |
|
· Raw (not baked or cooked) grain products (such as raw oats) |
| Beverages |
|
|
| Desserts |
|
·Unrefrigerated cream-filled pastry products (not shelf-stable‡) |
| Fats |
|
·Fresh salad dressings (stored in the grocer's refrigerated case) containing raw eggs or cheeses listed as “Do Not Eat” under “Dairy.” |
| Other |
|
|
Concern arising from the detection of potential pathogens in food has not been supported by documented evidence of such organisms as the source of opportunistic infections in immunocompromised persons. The potential benefit of food safety recommendations directed specifically toward HCT recipients must be weighed against the uncertain value of such recommendations [767] and their potential to adversely affect patients` nutritional intake and/or quality of life.
Shelf-stable refers to unopened canned, bottled, or packaged food products that can be stored before opening at room temperature; container may require refrigeration after opening.
Aseptically packaged, shelf-stable tofu and pasteurized tofu do not need to be boiled. Nonpasteurized tofu must be cut into 1-inch cubes or smaller, and boiled a minimum of 5 minutes in water or broth before eating or using in recipes.
Rinse under clean, running water before use, including produce that is to be cooked or peeled (such as bananas, oranges, and melon).
Bring tap water to a rolling boil and boil for 15-20 minutes. Store boiled water in the refrigerator. Discard water not used within 48 hours (2 days).
Recommend using boiled or bottled water if using a water service other than city water service. Please see Water Safety Guidelines in “Food Safety Guidelines.”
See Water Safety Guidelines in “Food Safety Guidelines” for approved bottled water treatments.
Honey products are not allowed for any child less than 1 year of age and not allowed for children with SCIDS until 9 months posttransplant.
Preventing disease
Growth factors (eg, granulocyte-macrophage colony-stimulating factor [GM-CSF] and G-CSF) shorten the duration of neutropenia after HCT [412]. However, a meta-analysis showed that use of growth factors did not reduce the attack rate of invasive fungal disease [165], and therefore, no recommendation can be made for the use of growth factors for prophylaxis against invasive fungal disease (CI).
Topical antifungal drugs applied to the skin or mucosa (eg, nystatin or clotrimazole) might reduce colonization by yeasts and molds in the area of application. However, these agents have not been proved to prevent locally invasive or disseminated yeast or mold infections and their use for prophylaxis is unclear (CIII). Performing fungal surveillance cultures is not indicated for asymptomatic HCT recipients (DII) 413, 414.
Other recommendations
Patients receiving antifungal prophylaxis who develop clinical signs or symptoms of infection should be evaluated for breakthrough bloodstream or pulmonary fungal infections (AIII). Such infections may occur because the prophylactic drug has no activity against the organism; because the organism has developed resistance to the drug; or because of other factors, such as severe immunosuppression or low serum levels of the prophylactic agent [415].
Recommendations Regarding Yeast Infections
Preventing exposure
The risk for invasive candidiasis is significantly higher during the early posttransplant period (phase I) because of neutropenia, severe mucositis, and the presence of a central venous catheter (CVC) [416]. During phase II, the risk factors for invasive candidiasis are the presence of a CVC and severe gastrointestinal (GI) graft-versus-host disease (GVHD) [416]. Among autologous HCT recipients, the risk of invasive candidiasis is minimal once neutropenia and mucositis have resolved.
Invasive candidiasis is usually caused by dissemination of endogenous Candida species that have colonized a patient's GI tract [417]. Consequently, methods of preventing exogenous yeast exposure usually do not prevent invasive yeast infections after HCT. However, because Candida species can be carried on the hands, HCWs and others in contact with HCT recipients should follow appropriate hand-washing practices to avoid exposing patients to fungal pathogens (AII).
Preventing disease
Fluconazole is the drug of choice for the prophylaxis of invasive candidiasis before engraftment in allogeneic HCT recipients, and may be started from the beginning or just after the end of the conditioning regimen (AI) 418, 419. Low-dose fluconazole has variable efficacy; therefore, doses lower than 200 mg are not recommended (DII). The optimal duration of fluconazole prophylaxis—specifically, whether prophylaxis confers significant benefits when continued postengraftment—is not defined. A post hoc analysis of a randomized clinical trial has shown that fluconazole given until day 75 posttransplant was associated with prolonged protection against invasive candidiasis, even beyond the period of prophylaxis [420].
Fluconazole is not effective against Candida krusei, and has variable activity against C. glabrata, which can become resistant [421]; moreover, the use of fluconazole for prophylaxis may result in the selection of azole-resistant Candida species (principally C. glabrata and C. krusei) [422]. Therefore, fluconazole is not recommended for prevention of such infections (DI). This may be a consideration in patients who are known to be colonized with fluconazole-resistant Candida species.
Cros-sresistance to azoles (ie, fluconazole, voriconazole, posaconazole) may occur among Candida species, particularly C. glabrata 423, 424. Therefore, clinicians should be alert for the possibility of breakthrough infection with resistant organisms in patients receiving prophylaxis with any azole drug (CIII).
Micafungin is an alternative prophylactic agent, as 1 study has shown it to be comparable to fluconazole for preventing possible or documented fungal infection (BI) [425]. Use of micafungin as a prophylactic agent is limited by the necessity of i.v. infusion and cost. There have been case reports of sporadic resistance to echinocandin antifungal agents [426].
Itraconazole oral solution has been shown to prevent invasive fungal infections, but use of this drug is limited by poor tolerability and toxicities (CI) 427, 428. If antimold activity is warranted in antifungal prophylaxis, posaconazole and voriconazole are options 429, 430. Posaconazole has not been studied in the preengraftment phase of HCT. Few HCT recipients require prophylaxis against Candida in the postengraftment period (phases II and III), but if needed (eg, because of the presence of GVHD), options include voriconazole (BI) [430] and posaconazole (BI) [429].
Oral, nonabsorbable antifungal drugs, including oral amphotericin B (500 mg suspension every 6 hours), nystatin, and clotrimazole troches, might reduce superficial colonization and control local mucosal candidiasis, but have not been demonstrated to reduce invasive candidiasis (CIII).
Other recommendations
HCT candidates with candidemia or invasive candidiasis can safely receive transplants [431] if (1) their infection was diagnosed early and treated immediately and aggressively with effective anti-Candida therapy; and (2) there is evidence of infection control before the transplant (BIII). Such patients should continue receiving therapeutic doses of an appropriate antifungal drug throughout phase I (BII) and until a careful review of clinical, laboratory, and serial computed tomography scans verifies resolution of candidiasis (BII). As radiographic abnormalities may persist for a long period of time among patients with hepatosplenic candidiasis, complete resolution is not necessary before transplantation, provided that the patient has received appropriate therapy and shows clinical improvement.
Because autologous recipients generally have an overall lower risk for invasive fungal infection than allogeneic recipients, most autologous recipients do not require routine antiyeast prophylaxis (CIII). However, experts recommend administering antiyeast prophylaxis to a subpopulation of autologous recipients who have underlying hematologic malignancies (eg, lymphoma, leukemia, or myeloma) and who have or will have prolonged neutropenia and mucosal damage from intense conditioning regimens or graft manipulation, or have received fludarabine (Flu) or 2-CDA within the 6 months prior to HCT (BIII). Recommendations regarding the prevention of invasive yeast infections are the same among pediatric and adult HCT recipients, except that appropriate dose adjustments for prophylactic drugs should be made for pediatric recipients (Appendix 1).
When considering continuation of antifungal therapy in patients with prior infection, HCT clinicians should be mindful of drug interactions, especially with calcineurin inhibitors, particularly with drugs in the azole class, which affect cytochrome P450 metabolism [432]. Results of recent studies suggest differential toxicities and effects on cyclophosphamide (Cy) metabolites with coadministration of different azole drugs 432, 433. Specifically, coadministration of fluconazole was associated with fewer early toxicities and different metabolites compared with itraconazole [433]. This was hypothesized to be because of fluconazole inhibition of cytochrome P450 2C9 providing a “protective” effect compared to itraconazole, which inhibits only P450 3A4 [432]. In general, clinicians should be careful about coadministering drugs that have an impact on any cytochrome subunits active in conditioning metabolism.
Recommendations Regarding Mold Infections
Preventing exposure
Nosocomial mold infections among HCT recipients result primarily from respiratory exposure to and direct contact with fungal spores [434]. Measures for minimizing exposure to mold in HCT candidates and recipients are discussed in the Infection Prevention and Control in Health care Facilities section.
In addition to air, water may also be a source of pathogenic fungi. Opportunistic molds (eg, Aspergillus and Fusarium species) are present in the water and on water-related surfaces of hospitals caring for cancer patients, and molecular studies have suggested potential relatedness of environmental and clinical strains among patients with aspergillosis and fusariosis 435, 436, 437. Therefore, hospital water should be considered a potential source of nosocomial invasive mold infections. Although some investigators believe it is necessary to decrease patient exposure during periods of severe immunosuppression [438], current data are insufficient to support the formulation of definitive recommendations. Cleaning of water-related structures in patients' bathrooms may be attempted 435, 436.
Preventing disease
Invasive mold infections have a trimodal incidence distribution among allogeneic HCT recipients 439, 440, 441. Before engraftment (ie, during phase I), the main risk factor is prolonged neutropenia, and therefore, the risk is higher with bone marrow (BM) and umbilical cord blood (UCB) transplants and lower with peripheral blood (PB) and nonmyeloablative (NMA) transplants. In addition, the risk is higher among patients with prolonged low-level neutropenia prior to transplant, such as aplastic anemia (AA) patients. In phases II and III, the main risk factor is severe cell-mediated immunodeficiency caused by GVHD and its treatment. Therefore, recipients of transplants with higher risks for severe GVHD (unrelated donor, mismatched transplant, haploidentical) are at greater risk of mold infections. Patients at high risk for mold infections should be considered for prophylaxis with mold-active drugs during periods of risk (BI).
Trials assessing the efficacy of cyclodextrin oral solution and intravenous formulations of itraconazole have shown efficacy in preventing mold infections (BI), but the benefit was offset by poor tolerance and toxicity of the oral formulation, with ∼25% discontinuation for GI side effects 427, 433.One study has shown micafungin to be effective in preventing invasive fungal infections (including fever) when administered during neutropenia [425], but the incidence of invasive aspergillosis is low during the preengraftment phase, so antimold efficacy could only show a display of activity rather than efficacy (BI). Experience with other echinocandins (eg, caspofungin) demonstrates some efficacy (CII), but breakthrough mold infections during echinocandin prophylaxis have been reported [442]. Fluconazole has no activity against molds [443]. Based on the results of a study presented to date only in abstract form [430], voriconazole appears to be an alternative to fluconazole prophylaxis. Because the results of this study have not been presented in detail, the committee is not assigning a recommendation. Specific molds are resistant to certain drugs (eg, voriconazole does not have activity against Zygomycetes, and Scedosporium prolificans is resistant to all available antifungal agents).
In patients with GVHD, posaconazole has been reported to prevent invasive mold infections (BI) [429]. In 1 study, patients with GVHD were randomized to receive either fluconazole (400 mg) or posaconazole (200 mg 3 times daily). Results showed a trend toward a lower incidence of invasive fungal infection, with particular differences in mold infections. The optimal duration of prophylaxis in the setting of GVHD is not defined.
Aerosolized liposomal amphotericin B has been found to be effective in reducing invasive pulmonary aspergillosis in 1 randomized trial that included leukemia and HCT patients and can be considered as an alternative to mold-active azoles (BII) [444]. However, administration of the drug was interrupted by cough in many patients, and the optimal aerosol dose and delivery device have not yet been determined. Thus, further study is needed before its role can be determined.
Other recommendations
Patients with prior invasive aspergillosis should receive secondary prophylaxis with a mold-active drug (AII). The optimal drug has not been determined, but voriconazole has been shown to have benefit for this indication (AII) [445].
Recommendations Regarding Serum Level Monitoring
Absorption of itraconazole and posaconazole is poor in patients who are not eating. Meta-analyses suggest that the efficacy of itraconazole is associated with bioavailability; blood levels of >0.5 μg/mL are associated with effective prevention of proven invasive fungal infection [446]. Posaconazole levels for therapeutic effectiveness have not been established. Median levels of posaconazole are relatively lower in patients who have aGVHD and symptomatic diarrhea [447]. Voriconazole blood levels of at least 1 μg/mL are thought to be required for efficacy. Low voriconazole levels have been reported in patients with documented breakthrough infections [415].
There is relative controversy over the utility of routine monitoring of azole drug levels, but most clinicians agree that measurement of levels should be considered in patients who have documented breakthrough infection. A change to, or addition of, an antifungal agent of a different class is advisable until the blood level has been determined. If the level is found to be low, then consideration can be given to resuming the earlier drug at a higher dose (CIII) 415, 448.
Regionally Limited or Rare Infections—Prevention after HCT
J. Gea-Banacloche, H. Masur, C. Arns da Cuhna, T. Chiller, L. Kirchoff, P. Shaw, M. Tomblyn, C. Cordonnier
This section covers infections that are prevalent only in certain geographic areas, or that occur uncommonly among hematopoietic cell transplantation (HCT) recipients. Diseases covered are tuberculosis, Pneumocystis jiroveci Pneumonia (PCP), toxoplasmosis, strongyloidiasis, Chagas disease, leishmaniasis, malaria, and Nocardia infection. Other infections, such as babesiosis, can be transmitted in the course of transplant via blood products, in addition to the hematopoietic cell graft [449]. Guidelines for prevention of these graft-transmissible infections are found in the Hematopoietic Cell Safety Section.
Recommendations Regarding Mycobacterium tuberculosis
Tuberculosis (TB) is uncommon among HCT recipients. The main risk factor is undergoing transplant in a country with a high endemic rate of TB 450, 451. In countries where TB is uncommon, TB is more prevalent among patients who have come for HCT from countries or were born in countries with a higher rate of endemic TB 452, 453. Patients with household members at risk (eg, recent immigrants, correctional facility discharges) also deserve particular attention. It must always be remembered that there is no definitive test to exclude TB and a high level of clinical awareness must be maintained in all immunosuppressed patients.
The rate of TB reported after HCT is 10 times less than after solid organ transplantation [454], as HCT patients do not receive life-long immunosuppression. However, patients with prolonged immunosuppression, such as those with chronic graft-versus-host disease (cGVHD), remain at risk for TB. Because control of TB is T cell mediated, restoration of T cell function over time and with cessation of immunosuppression reduces the patient's risk of TB.
Most patients who develop TB after HCT have not had clearly identified risk factors 455, 456. Most had normal pretransplant chest radiographs and no direct history of contact with TB. Most had been transplanted for leukemia, had received total body irradiation (TBI)-based conditioning, and were allograft recipients. Most were receiving treatment for GVHD and were >100 days posttransplant.
Although most cases of TB have occurred in allogeneic HCT recipients, 20% have occurred in autologous recipients. Despite this low rate 454, 457, diagnostic vigilance must be maintained. For example, mycobacterial infection may occur more frequently in patients who have undergone autologous HCT for chronic lymphocytic leukemia (CLL), in whom prior therapy with fludarabine (Flu) and alemtuzumab induces profound T cell deficiency, and susceptibility to a variety of opportunistic infections [458].
Preventing exposure
HCT candidates and recipients should avoid exposure to persons or environments where there is a substantial risk of respiratory contact with individuals with active TB. It is prudent to advise HCT candidates and recipients that certain occupations (eg, volunteer work or employment in health care facilities, correctional institutions, or shelters for the homeless) can increase their risk for TB exposure (BIII) [459].
Preventing disease
Evaluation of patients before HCT
Experts recommend evaluation for latent or active TB in patients who are candidates for HCT (BII). Assessment should include a history of:
-
•
Prior active TB
-
•
Prior exposure—evaluate as high-priority contacts as per CDC guidelines [460].
-
•
Results of previous tuberculin skin tests (TSTs) or interferon-gamma release assays (IGRA).
Experts disagree about the convenience or benefit of routinely obtaining a TST or IGRA in every transplant candidate. Interpretation of the TST may also be complicated by a history of prior Bacillus Calmette-Guérin (BCG) vaccination, although tuberculin reactivity following BCG tends to wane over time [461]. The American Thoracic Society (ATS) states that a positive TST may be considered diagnostic of latent M. tuberculosis infection in BCG recipients who are at risk for recent M. tuberculosis infection or who have a medical condition that increases the risk for progression to active TB (CIII) [462]. Because of prior chemotherapy-induced immunosuppression, the TST is not as sensitive in HCT candidates as it is in the healthy population. IGRAs are specific for TB, but a negative test does not exclude latent TB infection, particularly in the immunocompromised patient. In a meta-analysis, IGRAs were found to be more sensitive than TST in immunocompromised patients suspected of having TB [463]. This was particularly the case among patients with autoimmune disease and receiving antitumor necrosis factor therapy (eg, infliximab), who are at particular risk of TB [464]. Studies specifically in HCT patients are awaited. However, Canada and the United States have made recommendations for use of IGRAs in immunocompromised patients 465, 466.
Any patient with a recently positive TST or IGRA or a history of a positive test and no prior preventive therapy, should be evaluated for active TB. At a minimum, the patient should be asked about symptoms of systemic disease and respiratory symptoms such as cough and shortness of breath, and a chest radiograph should be assessed (AII) [462]. If active TB is detected, therapy and appropriate isolation should be initiated. HCT should be delayed until the active infection is deemed controlled based on clinical judgment, because no objective definition of adequate control has been formulated. If the TST or IGRA is positive, but no active TB identified, treatment for latent TB infection (Appendix 1) should be initiated but the HCT need not be delayed.
Indications for treatment of latent TB infection or prophylaxis
Because of high risk of reactivation or new infection, prophylaxis should be administered to immunocompromised HCT recipients or candidates who:
-
•
Have been exposed to someone with active, infectious (ie, sputum-smear positive) pulmonary or laryngeal TB, regardless of the HCT recipient's or candidate's TST or IGRA status (BIII)
-
•
Have a positive TST result—regardless of prior BCG vaccination—without previous treatment and no evidence of active TB disease (BII). A positive TST with a history of BCG vaccination is still considered by the ATS as an indication for prophylaxis in patients who “have medical conditions that increase the risk for disease” [462], which presumably include HCT (CIII)
-
•
Have a positive IGRA result, without previous treatment and no evidence of active TB (BII).
A report of high frequency of reactivation of previously treated TB following transplantation, especially in some parts of the world where the endemic prevalence of TB is high, suggests such patients may be at high risk, and therefore, isoniazid (INH) prophylaxis should be considered (CIII) [467]. The value of prophylaxis in countries with a high rate of TB, or in HCT patients from such countries, should be considered at an institutional level. Because of the high prevalence of multidrug-resistant TB in some of these countries, single-agent prophylaxis may be ineffective. Consequently, maintaining a very high index of suspicion and providing early intervention may be preferable to universal prophylaxis. An HCT candidate or recipient who has been exposed to an active case of extrapulmonary, and therefore noninfectious, TB does not require preventive therapy (DIII).
Prophylactic regimens
INH is well tolerated post-HCT even with concurrent fluconazole use 450, 467, 468. Concurrent use with itraconazole is not recommended, and the impact of voriconazole or posaconazole is not known. INH with pyridoxine should be continued for at least 9 months and until immunosuppression dosages are substantially reduced (ie, prednisolone <0.5 mg/kg in children or <20 mg daily in adults) (Appendix 1). INH can be started at the completion of conditioning therapy, or prior to conditioning if feasible or if the clinical situation puts the patient at higher risk of infection. Rifampin given for 4 months is effective but the potential for substantial drug-drug interactions between rifampin and immunosuppressive and other agents generally makes this option impractical. The combination of pyrazinamide and rifampin (PZA/RIF) has known significant liver toxicity and its use post-HCT is not recommended (EII) [469].
Vaccination
BCG vaccination is contraindicated for HCT candidates (EII). Disseminated BCG infection has been reported among immunocompromised persons receiving BCG [470].
Donor evaluation
Donors who live in or originate from countries where TB is endemic have an increased risk of TB or latent TB similar to the rest of the population. There is no known risk from transplanting hematopoietic progenitor cells from an untreated donor with latent or active TB. However, it would be prudent for prospective donors with active TB to defer donation until the TB is well controlled, as determined by the donor's primary physician (EIII).
Recommendations Regarding PCP
Preventing exposure
Most cases of PCP were traditionally thought to occur from reactivation of latent infection. However, suggestive animal data and well-documented human cases indicate that clinical disease can result from recent infection. Moreover, previously infected patients can be reinfected with new strains of Pneumocystis 471, 472, 473, 474, 475, 476. These observations and some intriguing clusters of PCP cases have suggested that person-to-person transmission is possible 477, 478, 479, 480.
Neither the CDC nor most hospitals mandate respiratory precautions regarding patients with PCP. However, many clinicians consider it logical that HCT recipients should avoid exposure to patients with PCP (CIII). There are no data-based guidelines for how long such PCP patients should be isolated.
Preventing disease and disease recurrence
Allogeneic stem cell transplant (SCT) recipients
PCP is a generally preventable complication of HCT. Programs must have vigorous systems to assure that PCP prophylaxis is prescribed by providers and adhered to by patients (AII). PCP prophylaxis is usually prescribed for allogeneic recipients from engraftment until at least 6 months after HCT (AII) [481]. Some health care providers initiate PCP prophylaxis prior to transplantation, depending on the underlying disease and the pretransplant conditioning regimens or prior chemotherapy (CII). Prophylaxis should be continued for longer than 6 months in patients who continue to receive immunosuppressive drugs (AII) 481, 482, 483, 484.
The preferred regimen for PCP prophylaxis is trimethoprim-sulfamethoxazole (TMP-SMX), because of its superior efficacy compared with aerosolized pentamidine, oral dapsone, or oral atovaquone (AII) 485, 486. TMP-SMX also provides some protection against other pathogens, including Toxoplasma, Nocardia, enteric pathogens, Plasmodium species, urinary pathogens, and some respiratory pathogens. Although dapsone and atovaquone probably provide some protection against Toxoplasma, the broader spectrum of activity of TMP-SMX is among the reasons this drug is preferred.
TMP-SMX can delay engraftment, and thus is not usually administered before engraftment occurs. TMP-SMX is variably tolerated, and toxicities include myelosuppression, hypersensitivity, hyperkalemia, nephritis, hepatitis, and pancreatitis. The optimal dosage has not been defined in HCT patients. Many HCT programs use once- or twice-daily regimens (single-strength or double-strength tablets) 2 to 7 days per week (CIII), and all appear efficacious.
There are no properly powered trials on which to base a recommendation for alternative regimens. Oral dapsone 487, 488, aerosolized pentamidine [489], and oral atovaquone [490] have all been used (CII). However, none is as effective as TMP-SMX for PCP prophylaxis [491]; therefore, every effort should be made to administer TMP-SMX. Oral desensitization regimens have been used successfully for HIV-infected patients and others with rash [492], and similar protocols have been used in HCT recipients with a success rate of approximately 80% 491, 493. Desensitization should be attempted when feasible (CII) (Appendix 3).
Oral dapsone is convenient, but a substantial number of patients allergic to TMP-SMX will also be intolerant of dapsone. Therefore, this drug is not recommended in patients who have had life-threatening reactions to TMP-SMX. Adverse reactions to dapsone include hypersensitivity, hemolysis (especially in patients with glucose-6-phosphate dehydrogenase [G6PD] deficiency), hepatitis, and methemoglobinemia. Patients with G6PD deficiency should not receive this drug [488]. Aerosolized pentamidine is well tolerated, with bronchospasm as the only common adverse effect, but may be less effective than other regimens [494]. Atovaquone is expensive but well tolerated; however, it is not well absorbed unless ingested with fatty foods. Thus, atovaquone might not be an optimal choice in patients with gut GVHD.
Recurrence of PCP in HCT recipients is rare. Nevertheless, patients recovering from PCP should continue on chemoprophylaxis until their immunosuppression is resolved (AII), although there are no clear parameters for defining this endpoint.
Autologous SCT recipients
PCP develops in some autologous HCT recipients, although the rate is much lower than among allogeneic recipients. PCP prophylaxis should be considered for autologous HCT recipients whose immunosuppression is substantial, either from an underlying condition or its treatment. Such patients include those with underlying lymphoma, leukemia, or myeloma, especially when intensive treatment or conditioning regimens have included purine analogues (Flu, cladribine [2CdA]) or high-dose corticosteroids (BIII) 480, 488. Duration of PCP prophylaxis in this setting has not been evaluated, but common practice is to extend prophylaxis 3 to 6 months posttransplantation (CIII). For patients with ongoing immunomodulatory therapy posttransplantation, a longer duration of prophylaxis may be necessary.
Recommendations Regarding Toxoplasma gondii
Preventing exposure
In immunocompetent hosts, initial acquisition of Toxoplasma most often occurs through exposure to cat feces or undercooked meat. All HCT recipients should be provided information regarding strategies to reduce their risk for exposure to Toxoplasma species (see Strategies for Safe Living). Although an unusual source of toxoplasmosis after HCT, potential donors for allogeneic HCT could be tested for IgG antibodies to T. gondii antibodies [495] because T. gondii has been reported to be transmitted by leukocyte transfusion [496] and HCT [497] (CIII).
Preventing disease and disease recurrence
Because most toxoplasmosis among HCT recipients is caused by disease reactivation, candidates for allogeneic HCT should be tested for IgG antibody to determine whether they are at risk for disease reactivation after HCT (BIII) [495]. However, toxoplasmosis has occurred in a limited number of HCT patients who were seronegative for T. gondii pretransplant [498]. Most cases observed after allogeneic HCT occurred between 3 and 26 weeks posttransplant 499, 500, 501, but later occurrence has also been reported in patients receiving long-term immunosuppression. The incidence of T. gondii disease varies according to the seroprevalence in the geographic area. Clinical reactivation occurs among 2% to 6% of those who are seropositive prior to HCT 495, 500, 501, 502. CB transplant recipients may be at higher risk than other HCT recipients [500].
In allogeneic recipients who are seropositive for toxoplasmosis, the risk of reactivation varies with the kind of transplant and the degree of immunosuppression. In a prospective study from Spain, weekly screening with quantitative PCR testing of blood in allogeneic seropositive HCT recipients over the first 6 months after transplant has shown that reactivation occurred in 16% (8%-21%) of seropositive allogeneic HCT recipients [500]. Nucleic-acid based reactivation typically precedes clinical symptoms by 4 to 16 days 98, 500.
To facilitate early identification of reactivated infection, PCR screening can be considered in patients at high risk (ie, patients who were seropositive before transplant and who received a CB graft, have developed GVHD and require immunosuppressive treatment, or who are not receiving TMP-SMX) (BII). PCR should preferably be performed in a reference laboratory by a quantitative method, using strict measures to reduce the risk of false positive results (because of contamination) and false negative results (because of the presence of inhibitors) (BIII) [503]. Brain MRI or CT scans in HCT patients with toxoplasmosis may not demonstrate the characteristic ring-enhancing lesions of this disease [504]; therefore, a diagnostic workup (eg, PCR testing of cerebrospinal fluid (CSF) or bronchoalveolar lavage (BAL) samples and/or blood) should be initiated in patients with brain or pulmonary lesions who are seropositive for Toxoplasma (CIII).
T. gondii reactivation and disease are both rare, but have been reported, in patients receiving TMP-SMX for P. jiroveci prophylaxis 500, 505. In high-risk patients who are intolerant to TMP-SMX, a specific strategy for T. gondii should be implemented, because other P. jiroveci prophylactic drugs either do not penetrate the body site of reactivation (ie, aerosolized pentamidine) or their prophylactic efficacy after HCT is unknown (ie, dapsone, atovaquone). Such patients can either be treated prophylactically with 1 or more drugs effective against T. gondii (ie, clindamycin, pyrimethamine plus leucovorin, pyrimethamine plus sulfadiazine, or pyrimethamine and sulfadoxine plus leucovorin [506]), or be monitored with PCR testing and given preemptive treatment (Appendix 1) (CIII) [506]. There are no published prospective studies on the benefit/risk ratio of such strategies.
After therapy for toxoplasmosis, HCT recipients should continue receiving suppressive doses of TMP-SMZ or an alternate regimen for the duration of their immunosuppression (BIII) (Appendix 1).
Other recommendations
Except for the rare patients with pretransplant toxoplasmic chorioretinitis, who may benefit from secondary prophylaxis [507], recipients of autologous transplants are at negligible risk for toxoplasmosis reactivation. No prophylaxis or screening for toxoplasmosis infection is recommended for autologous transplant recipients (CIII). Indications for toxoplasmosis prophylaxis are the same in children and adults, but children should receive pediatric doses (Appendix 1).
Recommendations Regarding Nocardia Infection
Preventing exposure
All HCT recipients should be provided information on ways to reduce risks of exposure to Nocardia spp. Specific information should include the ubiquitous nature of this Gram-positive, weakly acid-fast, aerobic actinomycete. Nocardia spp. are found primarily in dirt and stagnant water. Patients should be advised to avoid gardening, soil, and plants while receiving immunosuppressive therapy (CIII). Because infection occurs via inhalation, patients who wish to continue gardening may minimize exposure by wearing protective gear such as an N-95 mask and gloves (CIII).
Preventing disease and disease recurrence
The incidence of Nocardia infections is increased in patients with HCT compared to the general population (128 cases/105 person/years versus 0.3-0.4 cases/105 person/years) 508, 509, 510. The median time to onset is approximately 200 days following transplant 508, 510. Risk factors for disease include immunosuppression, corticosteroid use, neutropenia, active GVHD, and concomitant opportunistic infections, especially cytomegalovirus (CMV) 510, 511, 512.
One study suggested that there may be decreased risk of infection in patients receiving TMP/SMX prophylaxis (CIII) [508]. Notably, between 40% and 60% of cases in HCT patients occurred while the patient was receiving TMP/SMX for PCP prophylaxis 510, 512. Interestingly, most of these breakthrough cases were successfully treated with high-dose TMP/SMX. However, therapy for Nocardia infection should be chosen on the basis of appropriate identification of the Nocardia isolate and, if available, susceptibility testing, becvause other treatment options are possible.
Recommendations Regarding Strongyloides stercoralis
Preventing exposure
Allogeneic HCT recipients should avoid contact with outhouses and cutaneous exposure to soil or other surfaces that might be contaminated with human feces (AIII) [513]. Allogeneic HCT recipients who work in settings where they could be exposed to fecal matter (eg, hospitals or institutions) should wear gloves when working with patients or in areas with potential fecal contamination (AIII).
Preventing disease and disease recurrence
Histories should be obtained for all patients before HCT to identify potential exposure to Strongyloides through travel or residence in high-risk areas (ie, moist temperate areas such as the tropics, subtropics, or the southeastern United States and Europe) (BIII) [513]. HCT candidates who have unexplained peripheral eosinophilia or who have resided in or traveled to areas endemic for strongyloidiasis, even during the distant past, should be screened for asymptomatic strongyloidiasis before HCT (BIII). Options available for screening include stool examination and serologic testing with an enzyme-linked immunosorbent assay (ELISA) [514] or an indirect immunofluorescence antibody test (IFAT) [515]. Generally, ELISA is the preferred screening method, but its sensitivity may be relatively low (<70%) in some immunocompromised patients (BIII) [516]. Although stool examinations for strongyloidiasis are specific, the sensitivity achieved with ≥3 stool examinations is 60% to 70%; the sensitivity achieved with concentrated stool exams is, at best, 80% [513]. A total of ≥3 stool examinations should be performed if serologic tests are unavailable or if strongyloidiasis is clinically suspected in a seronegative patient (BIII).
HCT candidates with pretransplant screening tests positive for Strongyloides species or those with an unexplained eosinophilia and a travel or residence history indicating exposure to Strongyloides stercoralis, should receive empiric treatment before transplantation, preferably with ivermectin (Appendix 1) (BIII) [517]. There is no evidence supporting universal preemptive treatment, but given ivermectin's favorable safety profile and low cost, some clinicians advocate preemptive treatment for patients from endemic areas who have no symptoms, no eosinophilia, and negative screening test results (CIII).
To prevent recurrence among HCT candidates, parasite clearance after therapy should be verified with ≥3 consecutive negative stool examinations before proceeding with HCT (AIII). Data are insufficient to recommend a drug prophylaxis regimen after HCT to prevent recurrence of strongyloidiasis. HCT recipients who had strongyloidiasis before or after HCT should be monitored carefully for signs and symptoms of recurrent infection for at least 6 months after treatment (BIII).
Other recommendations
Unlike with allogeneic HCT, hyperinfection strongyloidiasis has been reported only rarely after autologous HCT; however, the same screening precautions should be used among autologous recipients (BIII). Indications for empiric treatment for strongyloidiasis before HCT are the same among children and adults except for children weighing <15 kg, for whom the preferred drug is albendazole (BIII) (Appendix 1).
Recommendations Regarding Trypanosoma cruzi and Leishmania
Preventing exposure
Trypanosoma cruzi, the etiologic agent of Chagas disease, can be transmitted through blood transfusion [518]. Transfusion-associated transmission of T. cruzi is a well-known phenomenon in endemic countries, and 7 instances have been reported in the United States and Canada, all in immunocompromised patients who had fulminant courses of acute Chagas disease. Serologic screening of donated blood for T. cruzi in the United States began in January 2007, and although the FDA has not mandated screening, more than 70% of donated units are currently being screened. Sleeping sickness (T. brucei rhodesiense and T.b. gambiense) and leishmaniasis (various species belonging to the genus Leishmania) could also be transmitted via contaminated transfusions, although the possibility of leishmaniae or African trypanosomes causing problems in an HCT patient is extremely remote if the donor and recipient are from nonendemic areas.
Reactivation of T. cruzi has been reported in HCT patients, but there are few reports of visceral leishmaniasis following HCT 519, 520, 521, and this case seemed to be acquired more than a year after transplantation, and not preexistent or transmitted by the stem cell product.
Donors or recipients who were born, received a blood transfusion, or ever lived at least 6 months in a Chagas disease endemic area (eg, parts of South and Central America and Mexico) should be screened serologically for anti-T. cruzi serum IgG antibody (BIII) [522]. Similarly, a donor or recipient whose mother was born in a Chagas disease endemic area or who has an extensive maternal family history of cardiac disease (eg, cardiomegaly and dysrhythmias) should be screened because of the risk of congenital transmission (CIII) 518, 523. Persons who lived <6 months in a Chagas-endemic area but who had high-risk living conditions (eg, having had extensive exposure to the Chagas disease vector—the reduviid bug—or having lived in dwellings with mud walls, unmilled logs and sticks, or a thatched roof) should also be screened for evidence of T. cruzi infection (BIII).
In the absence of a gold standard test for Chagas disease, screening for infection should be performed with at least 2 serologic tests (eg, enzyme immunoassay, indirect hemagglutination, indirect fluorescent antibody, or radioimmunoprecipitation assay) (BIII) [524]. Persons with active Chagas disease (DIII) or a past history of Chagas disease (CIII) should not serve as HCT donors.
Preventing disease
HCT candidates at risk for infection with T. cruzi should be screened for serum IgG anti-T. cruzi antibody (BIII) [522]. T. cruzi seropositivity is not a contraindication to HCT 522, 525. However, if an acute illness occurs in a T. cruzi-seropositive HCT recipient, particularly during neutropenia, T. cruzi reactivation should be included in the differential diagnosis (BIII) [525]. Experts have proposed use of benznidazole or nifurtimox for preemptive therapy or prophylaxis of recurrent T. cruzi among seropositive HCT recipients [526], but insufficient data were found to make a recommendation. For additional information regarding the epidemiology of Chagas disease and for questions regarding evaluation and treatment, contact CDC/National Center for Zoonotic, Vector-borne and Enteric Diseases/Division of Parasitic Diseases (http://www.cdc.gov/chagas, (770) 488-7775, chagas@cdc.gov).
Other recommendations
Recommendations are the same for autologous or allogeneic recipients. However, recurrence of Chagas disease is probably less likely to occur among autologous recipients because of the shorter duration of immunosuppression. Recommendations are the same among children or adults.
Recommendations Regarding Malaria
In 2006, there were an estimated 247 million cases of malaria with more than 880,000 deaths [527]. Malaria is not an opportunistic disease, but it is increasingly relevant in SCT for several reasons:
-
•
More patients with either a history of malaria or residence in an endemic malaria region are being transplanted;
-
•
More residents from endemic regions are being considered as donors; and
-
•
More recipients are being exposed to malaria after transplant.
Preventing exposure
Preventing transmission through the stem cell product
Malaria, including falciparum malaria, has been transmitted by HCT even when the donor smears pretransplant are reportedly negative 106, 107, 108, 528. All donors from endemic regions should be asked about a malaria history. Donors who have traveled to an area where malaria transmission occurs should be deferred from donating for 1 year after their return. Former or current residents of an area where malaria transmission occurs should be deferred for 3 years. If those deferral times are not feasible, the donor should receive empiric treatment for malaria prior to donating [108]. Blood smears, immunochromatogenic tests, and PCR are all inappropriate tests for evaluating asymptomatic potential donors.
If the donor is diagnosed with active infection, collection of the stem cell product should be delayed if possible until treatment has been completed and negative confirmatory testing obtained (AIII). If this is not feasible, preemptive treatment of the recipient seems reasonable [106], but there is no evidence to make a recommendation.
Preventing mosquito bites
HCT recipients who cannot avoid areas with malaria should try to avoid bites by Plasmodium-carrying mosquitoes. Anopheles mosquitoes are most active at night. It is recommended HCT recipients traveling to endemic malaria areas use insecticide-treated bed nets (ITNs) if they are sleeping in an open-air setting (AII). This practice has shown to reduce severe malaria in children [529] and is recommended by the CDC and the World Health Organization for malaria control. Individual infection may be decreased by the use of long sleeves, long pants, and mosquito repellents (AI) (eg, DEET [30%-50%]). The use of mosquito repellent seems to add to the protective effect of ITNs [530].
Preventing disease
HCT recipients from endemic areas or living in endemic areas
HCT recipients from endemic areas should be screened by blood smears, rapid tests, and/or PCR for active disease before transplantation (AIII). Following HCT, patients should be screened if they develop fever, pancytopenia, hemophagocytosis syndrome, hepatosplenomegaly, or other symptoms suggestive of malaria (AIII). However, reported occurrences of malaria in HCT recipients are rare, and evidence is insufficient to make specific recommendations for prophylaxis even in patients living in endemic areas. Involvement of a malarial specialist should be considered for additional management.
HCT recipients in nonendemic areas who are traveling to malaria endemic areas
HCT recipients traveling to areas endemic for malaria should consult their physicians to choose an antimalarial regimen that is effective against the prevalent Plasmodium and that can be tolerated and is free of interactions with the patient's current medications (AIII). The CDC has a Web site with comprehensive information regarding travel (http://wwwn.cdc.gov/travel/default.aspx).
Infection Prevention and Control in Health Care Facilities Where Hematopoietic Cell Transplantation Recipients are Treated
D. Yokoe, C. Casper, E. Dubberke, G. Lee, P. Muñoz, T. Palmore, K. Sepkowitz, J.-A.H. Young, J.P. Donnelly
Room Ventilation
Hematopoietic Cell Transplantation (HCT) center personnel should follow published guidelines for hospital room design and ventilation (BIII) 531, 532. HCT recipients should ideally be placed in Protective Environment rooms that incorporate the following features:
-
•
≥12 air exchanges/hour
-
•
Central or point-of-use HEPA filters with 99.97% efficiency for removing particles ≥0.3 μm in diameter (AIII) 532, 533, 534. Correct filtration is particularly critical in HCT centers with ongoing construction and renovation 535, 536. Filters should be replaced regularly based on manufacturers' recommendations, and, when there is ongoing construction, filtration efficiency should be monitored frequently to best determine appropriate time for replacement (AIII).
-
•
Directed airflow so that air intake occurs at 1 side of the room and air exhaust occurs at the opposite side (BIII) [531].
-
•
Consistent positive air pressure differential between the patient's room and the hallway ≥2.5 Pa (ie, 0.01 inches by water gauge) (BIII) 531, 532.
-
•
Well-sealed rooms (eg, filling the gaps between walls and windows, outlets, floor, and ceiling) should always be used for HCT patients to prevent infiltration of air from outside the room that could allow entry of spores and hinder maintenance of proper pressure differential (BIII) 532, 534, 537.
-
•
Continuous pressure monitoring, especially while rooms are occupied 531, 532 (BIII). Consideration should be given to using monitoring systems that will set off an alarm when the pressure differential between any Protective Environment room and adjacent hallway or anteroom falls to less than 2.5 Pa, to alert staff to possible engineering failures (CIII).
-
•
Self-closing doors to maintain constant pressure differentials (BIII) [532]. To enable the nursing staff to observe the HCT recipient even when the doors are closed, windows can be installed in either the door or the wall of the HCT recipient's room (CIII) [532].
When a shortage of Protective Environment rooms to accommodate all HCT patients exists, the allocation of these rooms should be prioritized for those at highest risk of invasive mold infection (eg, expected prolonged neutropenia, receiving treatment for GVHD) (BIII). In that situation, portable, industrial-grade HEPA filters may be used in non-Protective Environment rooms to accommodate other vulnerable patients, and should be placed centrally in patient rooms so that space is available around all surfaces to allow free air circulation (BIII). Portable HEPA filters have been shown to remove airborne fungal spores [538] and mycobacteria [539], and have been used as part of a successful strategy to prevent fungal infections during construction [540]. However, whether they are as effective as central, or point of use, HEPA filters at preventing infection has not been studied.
A laminar air flow (LAF) room contains HEPA-filtered air that moves in parallel, unidirectional flow (the air enters the room from 1 wall and exits the room on the opposite wall) [541]. Some studies have shown that LAF may protect patients from infection during Aspergillosis outbreaks related to hospital construction [542]. However, routine LAF room use for all HCT recipients has not been shown to provide substantial overall survival (OS) benefit, and therefore, LAF systems are not recommended for newly constructed rooms in HCT centers (DII) 531, 532.
There are no recommendations regarding the pressure differential between a HCT center and adjoining buildings, but at least 1 study demonstrated that negative pressure with respect to an adjoining building contributed to an Aspergillus outbreak [543]. HCT center maintenance personnel should prevent birds from nesting near hospital air-intake ducts (AII) 531, 534, 544, 545. HCT centers should have provisions for backup emergency power and redundant air-handling and pressurization systems to maintain a constant number of air exchanges and room pressurization when the central ventilation system is shut off for maintenance and repair (BIII) [546]. Additionally, an Infection Control Risk Assessment (ICRA) should be created by infection control personnel and maintenance personnel to develop protocols to protect HCT patients at all times from bursts of mold spores that might occur when air-handling systems are restarted after routine maintenance shut-downs (BIII) [546]. Because of this risk, air-handling systems should never be shut off in HCT units for energy conservation purposes.
Anterooms for HCT center rooms are optional, except in the case of HCT recipients requiring airborne precautions for certain infections (BIII). Anterooms should be used to ensure appropriate air balance relative to the Protective Environment and the hallway; independent exhaust of contaminated air to the outside should be provided, or a HEPA filter placed in the exhaust duct if return air must be recirculated (BIII) 531, 532. If a Protective Environment airborne infection isolation room with an anteroom is not available, the patient should be placed in a standard airborne infection isolation room and a portable, industrial-grade HEPA filter used to enhance removal of spores in the room (BIII) [531].
Construction, Renovation, and Building Cleaning
Construction and renovation
Hospital construction and renovation may result in an increased risk for health care-associated invasive mold infection, particularly aspergillosis, among HCT recipients 531, 547, 548. Therefore, persons responsible for HCT center construction or renovation should consult published recommendations regarding environmental controls during construction (AIII) 531, 549.
When construction or renovation is undertaken, plans should include intensified mold-control measures. An ICRA should be planned prior to construction on a facility or design of a new facility (AIII) 531, 532, 550. Construction and renovation infection control planning committees should include engineers, architects, housekeeping staff, infection control personnel, the director of the HCT center, the administration, construction project managers, and safety officers. Close coordination among these participants will facilitate compliance with infection control measures during construction, renovation, and demolition (BIII) 550, 551. Whenever possible, HCT recipients should avoid construction or renovation areas (AIII) 531, 543, 552, 553. Also, HCT center staff should avoid transporting equipment and supplies used by HCT recipients through construction or renovation areas.
During construction or renovation, HCT center staff should take steps to minimize fungal spore counts in patient rooms by adhering to room ventilation recommendations [549]. During outdoor construction and demolition, the intake air should be sealed (BIII), if possible; if not, filters should be checked frequently to verify that they are well-seated and replaced when necessary. Additionally, to protect HCT patient care areas during fire drills and emergencies, weather stripping should be placed around stairwell doors, or alternatively, the stairwell air should be filtered to the level of safety of the adjacent hospital air (BIII). False ceilings should be avoided whenever possible (BII) [547]. If use of false ceilings cannot be avoided, the area above false ceilings should be vacuumed routinely to minimize dust and, therefore, fungal exposure to patients (BIII) [547].
A number of measures have been used to minimize the impact of construction on HCT recipients [554]. During hospital construction or renovation, hospitals should construct rigid, dust-proof barriers with airtight seals [531] between patient care and construction or renovation areas to prevent dust from entering patient care areas (BIII) [go to www.ashe.org/ashe/codes/cdc/pdfs/assessment_icra.pdf for more information]. These barriers (eg, sealed drywall) should be impermeable to Aspergillus spores 531, 534, 549, 552, 555. Construction and renovation areas should have negative air pressure-relative HCT patient care areas to ensure that air flows from patient care areas toward construction areas (AII) 543, 549. If impervious barriers cannot be created around the construction area, patients should be moved from the area until renovation is complete and the area has been cleaned appropriately (BIII). HCT centers should direct pedestrian traffic occurring near construction or renovation areas away from patient care areas to limit the opening and closing of doors or other barriers that might cause dust dispersion, entry of contaminated air, or tracking of dust into patient areas 531, 556, particularly HCT center patient care areas (BIII). If possible, specific access routes, such as corridors, elevators, entrances, exits, even bathrooms should be dedicated to construction use only [531]. Tacky floor mats should be placed at the threshold of construction areas to minimize tracking of dust. Visible dust and debris tracked out of the construction zone should be vacuum cleaned with HEPA filters in the vacuum exhaust [554]. Construction workers, whose clothing might be contaminated with mold spores, should use the construction elevator and avoid contact with patients, patient care areas, other elevators, and nonconstruction areas (BIII). A portable, industrial-grade HEPA filter should be used between a construction zone and the HCT unit if a large area is under construction and negative pressure differential cannot be guaranteed (AII) 543, 550. Monitoring of air quality during construction may include, daily particle counts, environmental air sampling, and more frequent measurements of ventilation pressure differentials (CIII) 543, 557.
Monitoring for clinical cases of aspergillosis and other invasive mold infections should be performed, with enhanced surveillance of microbiologic, pathologic, and radiologic data to identify trends suggesting an environmental mold source (BIII) (see Surveillance section). Routine microbiologic air sampling for fungal spores in HCT units is not recommended, as its role has not been established (DIII) 558, 559, 560. However, during a suspected outbreak, there may be a role for microbiologic air sampling in patient care areas (CIII). If microbiologic air sampling is performed as part of an outbreak investigation, sample volumes of at least 1000 L may achieve a higher degree of sensitivity than smaller samples 543, 561.
Allogeneic HCT recipients should avoid construction areas inside and outside the health care facility. HCT recipients may benefit from wearing N95 respirators while outside of HEPA-filtered areas, especially during periods of health care facility construction and renovation (CIII) 543, 552, 553. Although they have been utilized for patient use, there are no commercially available masks, including N95 respirators, which have been tested specifically for efficacy in reducing patient exposure to Aspergillus or other mold in hospital construction or renovation areas. Standard surgical masks provide negligible protection against mold spores. Whether or not the addition of mold-active prophylaxis would provide additional protection during periods of heavy construction has not been specifically studied, and, therefore, no recommendation can be made.
Newly constructed or renovated areas should be cleaned and disinfected before patients are allowed to enter them (AIII) [534]. Fungal-contaminated materials that cannot be removed and replaced should be decontaminated with copper-8-quinolinolate after cleaning (BIII) 540, 562. Also, areas above dropped ceilings in rooms located under or adjacent to construction areas should be vacuumed (BIII). Additionally, the ventilation, direction of airflow, and room pressurization should be tested and correctly adjusted before patients are allowed to enter (BIII) [549].
Cleaning
HCT centers should be cleaned at least daily with special attention to dust control (BIII). Wet-dusting should be performed; dusting techniques that aerosolize dust should be avoided (BIII) 531, 563, 564. Many studies show that surface contamination is associated with nosocomial infections, but there is a paucity of data showing that routine surface disinfection reduces the rate of infections [565]. Nonetheless, exhaust vents, window sills, and all horizontal surfaces should be cleaned with cloths and mop heads that have been premoistened with an approved hospital disinfectant (BIII) 531, 566. Thorough cleaning during and after any construction activity, including minor renovation projects, is critical (BIII) [549].
Floor surfaces and finishes should be smooth, nonporous, and scrubbable (eg, vinyl, linoleum, tile) to minimize dust levels (BIII) [144]. Carpeting should not be installed in HCT center hallways outside of patient rooms or inside the rooms (DIII) because contaminated carpeting has been associated with outbreaks of aspergillosis among HCT recipients 567, 568. HCT recipients should not be exposed to vacuuming that could cause aerosolization of fungal spores (eg, Aspergillus species) (EIII). All vacuum cleaners used in the HCT center should be fitted with HEPA filters 531, 549. An approved disinfectant [539] should be used daily for environmental disinfection and when wet vacuuming is performed in the HCT center (BIII). If an HCT center provides care for infants, phenolic disinfectants can be used to clean the floors, provided they are diluted according to the product label. Phenolic compounds, however, should not be used to clean basinets or incubators (DIII) [539].
Water leaks should be cleaned up and repaired as soon as possible but within 72 hours to prevent mold proliferation in floor and wall coverings, ceiling tiles, and cabinetry in and around all HCT patients care areas (BIII) 531, 564, 569. If cleanup and repair are delayed ≥72 hours after the water leak, the involved materials should be assumed to contain fungi and handled accordingly (eg, discarded preferably or cleaned) [531]. A moisture meter to detect water penetration of walls should be used whenever possible to guide decision making (BIII). For example, if the wall does not have <20% moisture content ≥72 hours after water penetration, the affected portions of the wall should be removed (BIII) [531].
Design and selection of furnishings should focus on creating and maintaining a dust-free environment. Upholstery should be smooth, nonporous, and easily disinfected to minimize contamination with potential nosocomial pathogens [570]. Finishes (ie, wall coverings, window shades, and countertops) used in HCT centers should also be scrubbable, nonporous, and easily disinfected to minimize dust accumulation (BIII).
Isolation and Barrier Precautions
HCT centers should follow published guidelines for hospital isolation practices, including CDC guidelines for preventing healthcare-associated infections (AIII) 144, 534, 571. However, the efficacy of specific isolation precautions in preventing health care-associated infections among HCT recipients has not been evaluated. HCT recipients should be placed in single-patient rooms (BIII), if possible. If the availability of single-patient rooms is limited, their use should be prioritized for the most severely immunosuppressed patients (eg, HCT recipients during their initial transplant admission, particularly allogeneic HCT recipients, or patients who are receiving immunosuppressive therapy for GVHD during readmissions) (BIII).
In general, allogeneic HCT recipients are likely to benefit from protective isolation measures (ie, a Protective Environment), including HEPA filtration and/or laminar airflow, to reduce exposure to environmental fungi (eg, Aspergillus sp.) and reduce treatment-related mortality (TRM; see Ventilation section) (BIII) 144, 572. The efficacy of protective isolation measures for autologous HCT recipients is not well established (BIII) [573], and individual circumstances should guide the prioritization of protective environment rooms in settings where the number of these rooms is limited (eg, some patients who receive tandem autologous HCT may be more immunosuppressed than those who receive nonmyeloablative (NMA) allogeneic HCT) (BIII).
At a minimum, standard precautions should be followed for all patient contacts (AIII). Standard precautions include hand hygiene and wearing of appropriate personal protective equipment (ie, gloves, surgical masks or eye and face protection, and gowns) during procedures and activities that are likely to generate splashes or sprays of blood, body fluids, secretions or excretions, or cause soiling of clothing [144]. Some centers use additional protective precautions (eg, gloves and mask, either during respiratory season or year round) in an attempt to further reduce the risk of transmission of respiratory viruses from health care workers and visitors to patients. However, there are insufficient data to provide recommendations regarding the use of additional protective precautions.
When indicated on the basis of coexisting conditions, HCT recipients should also be placed on airborne, droplet, or contact precautions in addition to standard precautions (AIII) [144]. Adherence to isolation precautions is critical in preventing transmission of infectious agents among HCT recipients, HCWs, and visitors (AIII). HCT recipients with illnesses because of respiratory or gastrointestinal viruses can have prolonged or episodic excretion of organisms (eg, RSV, adenovirus, rotavirus). Guidance regarding the duration of isolation precautions for specific pathogens is provided in the Control of Specific Healthcare-Associated Infections section.
HCT recipients may benefit from wearing masks or N95 respirators during the preengraftment period when they are outside of their hospital rooms, especially during periods of hospital construction and renovation (CIII). All HCT recipients who are immunocompromised (phases I-III of immune system recovery) and candidates undergoing conditioning therapy should minimize the time spent in crowded areas to avoid exposure to persons with CRV infections (BIII).
Hand Hygiene
Hand hygiene is the mainstay of infection prevention in the hospital, and is an essential element of Standard Precautions for all patients (AII) [144]. Hand hygiene includes both use of alcohol-based hand rubs and handwashing with soap (plain or antimicrobial) and water. HCT center HCWs whose duties include direct patient contact should not wear artificial fingernails or extenders because these have been associated with outbreaks of Gram-negative bacillus and candidal infections (EIII) 574, 575, 576. HCT center policies should encourage visitors to perform hand hygiene before and after each patient visit (BIII). HCT recipients and candidates and their household contacts should be educated about the importance of hand hygiene during the HCT hospitalization and after hospital discharge (BIII).
In the absence of visible soiling of hands or contact with spore-forming organisms (see Recommendations regarding C. difficile), the preferred method of hand hygiene is with an alcohol-based hand rub because of its superior microbicidal activity, reduced drying of the skin, and convenience (AII) 577, 578, 579, 580, 581. When hands are visibly dirty or soiled with blood or body fluids, hands should be washed using soap and water (AI) [579]. Hand hygiene should be performed under the following circumstances [579]:
-
•
Before direct contact with patients;
-
•
After contact with blood, body fluids or excretions, mucous membranes, nonintact skin, or wound dressing;
-
•
After contact with a patient's skin;
-
•
When hands will be moving from a contaminated to a clean body site during patient care;
-
•
After contact with medical equipment or the hospital environment in the immediate vicinity of the patient; and
-
•
If gloves are worn, before donning and after removing gloves.
Equipment
Equipment and devices should be cleaned, disinfected or sterilized, and maintained as directed by established guidelines (AIII) 532, 534, 539, 582, 583, 584, 585, 586. Opened and unopened wound-dressing supplies (eg, adhesive bandages 587, 588 and surgical and elastic adhesive tape [589]) should be examined for factors that, if left unchecked, could result in mold contamination and possible subsequent cutaneous transmission to patients (BII). This should consist of discarding all bandages and wound dressings that are out of date, have damaged packaging, or are visually contaminated by construction debris or moisture (BIII). When arm boards are used to provide support for intravenous lines, only sterile dressing materials should be used [590], and arm boards should be changed frequently (eg, daily) (BIII). Additionally, nonsterile tongue depressors inserted into a piece of foam tubing should not be used as splints for intravenous and arterial catheter sites because these have been associated with an outbreak of fatal invasive nosocomial Rhizopus microsporus among preterm (ie, very low birthweight) infants (DII) [591].
Plants, Play Areas, and Toys
Although exposure to plants and flowers has not conclusively caused invasive mold infections among HCT recipients, most experts recommend that plants and dried or fresh flowers should not be allowed in hospital rooms during conditioning or after HCT (phases I-III of immune system recovery) because Aspergillus species have been isolated from the soil of potted ornamental plants (eg, cacti), the surface of dried flower arrangements, and fresh flowers (DIII) 533, 534, 547, 592. In addition, high counts of Gram-negative bacteria have been found in vase water of cut flowers, with Pseudomonas most frequently isolated [531]. To minimize the risk of mold infection, HCT recipients and candidates should avoid contact with soil-based materials (eg, clay or potting soil) (DIII).
Toys are commonly colonized with bacteria (eg, S. aureus, Enterococcus) and viruses associated with respiratory and gastrointestinal (GI) illnesses 593, 594, 595, 596. There is no evidence to date that toys can serve as a source of mold infection. Water-retaining bath toys should not be used by immunocompromised HCT recipients and candidates as they have been associated with an outbreak of Pseudomonas aeruginosa in a pediatric oncology ward (DII) [597].
Published recommendations for washing and disinfecting toys should be followed (BIII) [598]. Cloth or plush toys should be washed in a hot cycle of a washing machine at least once weekly and more often as needed (BIII). Alternatively, machine washing in a cold cycle is acceptable if laundry chemicals for cold water washing are used in proper concentration [598]. Hard plastic toys should be scrubbed with warm soapy water using a brush to clean crevices, rinsed in clean water, immersed in a mild bleach solution that should be made fresh daily (see section on bleach disinfection) for 10 to 20 minutes, rinsed again, and allowed to air dry [539]. Alternatively, hard plastic toys can be washed in a dishwasher or hot cycle of a washing machine (BIII). Broviac dolls (which are used to demonstrate medical procedures [eg, insertion of catheters] to children to lessen their fears) should be disassembled upon completion of play and washed and disinfected before other children are allowed to play with them (BIII) 539, 582. Infants, toddlers, and children who put toys in their mouths should not share toys (BIII) [598]. Toys that cannot be washed or disinfected after use should be avoided (DIII). Occupational and physical therapy items should be cleaned and disinfected as previously described (BIII).
Play areas for pediatric HCT recipients and candidates undergoing conditioning therapy should be cleaned and disinfected daily, or more frequently if needed (BIII). Toys, games, and videos should be allowed in playrooms in HCT centers only if they can be kept clean and disinfected (BIII). Toys, games, and videos that are provided in an individual patient room should be thoroughly washed before they are brought into the room and thereafter at least once weekly or more frequently as needed (BIII) 539, 582, 598.
Disposable play items should be offered whenever possible to children on contact precautions (BIII). If a child on contact precautions has used a toy, game, or video, the item should be thoroughly cleaned and disinfected before being used by other children (BIII). When a child no longer requires contact precautions, any toys, games, and videos used during the period of isolation should be thoroughly cleaned and disinfected (BIII) 539, 582, 598. All cloth or plush toys used by a child on contact precautions should be washed in a washing machine before the toy is given to another child or placed in a play area (BIII). Toys that have been used in an isolation room and that cannot be thoroughly disinfected should be discarded (BIII).
Health care Personnel
HCT centers should provide a written comprehensive policy regarding immunizations and vaccinations for HCT center employees that meets current CDC, Advisory Committee on Immunization Practices, and Healthcare Infection Control Practices Advisory Committee recommendations (BIII) [304]. Immunization of all HCWs with all recommended vaccines is critical to prevent transmission of vaccine-preventable diseases to HCT recipients and candidates undergoing conditioning therapy. Where an equivalently effective option exists, HCWs caring for HCT recipients should preferentially receive inactivated vaccines (eg, trivalent inactivated influenza vaccine, inactivated polio vaccine) rather than live vaccines to minimize the theoretical risks of transmission of vaccine virus to HCT recipients (see Family Members Who Receive Live Vaccines section) (AIII).
Every effort should be made to restrict from direct patient care activities all HCWs with infections that are potentially transmissible to HCT recipients or candidates (AII). The extent of work restrictions (eg, leave from work versus temporary reassignment to nonpatient care duties) will depend on the specific infection. HCWs with draining skin and soft tissue infections or other skin or mucous membrane lesions (eg, HSV lip lesions) that cannot be completely covered should also be restricted from patient contact (BIII). Published recommendations regarding the duration of work restrictions for HCWs with infectious diseases should be followed (BIII) 599, 600. HCT center HCWs with blood-borne viral infections (ie, HIV or hepatitis B or C viruses [HBV, HCV]) should not be restricted from patient contact (DIII) 601, 602. Work exclusion policies should be designed to encourage HCWs to report their illnesses or exposures (AII).
HCT Center Visitors
HCT centers should have written policies regarding the screening of all visitors for communicable infections (BIII). In some institutions, trained personnel (eg, administrative or nursing personnel) perform active screening at key entry points to HCT units, particularly during the respiratory virus season. At a minimum, signs should be posted to inform the public about visitation restrictions. Ideally, staff should actively screen visitors daily (Appendix 2). Visitors with signs or symptoms suggestive of communicable infections (eg, fever, upper respiratory infection (URI), or flu-like symptoms, diarrhea, vomiting) or recent known exposure to communicable infections (eg, chickenpox, mumps, measles, pertussis) should be excluded from direct contact with HCT recipients or candidates undergoing conditioning therapy (BII) [603]. This is particularly important during periods of widespread CRV activity in the surrounding community or suspected nosocomial CRV outbreaks. Staff should request that affected visitors leave the HCT center until signs and symptoms of infection have resolved or, for recent exposures to communicable infections, until the incubation period for that infection has passed without the appearance of signs or symptoms suggestive of active infection (AII). Visitors should also be screened for recent receipt of live vaccines and excluded as appropriate (see Family Members Who Receive Live Vaccines section). The screening process should include all visitors who stay overnight in the rooms of HCT recipients or candidates.
No absolute minimum age requirement for HCT center visitors exists; however, all visitors must be able to follow appropriate hand hygiene and isolation precautions (AIII). The number of HCT center visitors at any 1 time should be limited to a number that permits the nursing staff to perform appropriate infection screening and adequate instruction and supervision of hand hygiene and glove and mask use, as appropriate (BIII).
Hospitals with nearby family lodging facilities (eg, Ronald McDonald houses), should share information with these facilities regarding practices to prevent spread of communicable infections within these types of facilities and to health care facilities (BIII).
Patient Skin and Oral Care
To optimize skin care, HCT recipients should take daily showers or baths using a mild soap during and after transplantation (BIII). For patients with GVHD, regular lubrication of dry, intact skin with emollients may decrease pruritus and maintain skin integrity [604]. Ointments and creams are more effective than lotions and less likely to sting when applied to sensitive skin [604].
Routine inspection of skin sites likely to be portals of infection (eg, perineum, intravascular access sites) is recommended during neutropenia (BIII). HCT recipients and candidates undergoing conditioning therapy should maintain good perineal hygiene to minimize loss of skin integrity and risk for infection (BIII). To facilitate this, HCT centers should develop protocols for patient perineal care, including recommendations for gentle but thorough perineal cleaning after each bowel movement and thorough drying of the perineum after each episode of urination (BIII). After using the toilet, females should always wipe the perineum from front to back to prevent fecal contamination of the urethra and urinary tract infections (AIII). To prevent vaginal irritation and to avoid the risk for cervical and vaginal abrasions that can serve as portals of entry for infection, menstruating HCT recipients should not use tampons (DIII). The use of rectal thermometers, enemas, or suppositories; internal rectal exams; and sexual practices involving anal penetration are contraindicated among HCT recipients to avoid skin or mucosal breakdown, which can introduce pathogens (DIII).
To reduce the risk of oral and dental infections, all HCT candidates and their caregivers should be educated regarding the importance of maintaining good oral and dental hygiene for at least the first year after HCT (AIII). HCT candidates should be informed that establishment of the best possible periodontal health before HCT is of substantial benefit in avoiding short- and long-term oral infections and that maintenance of oral hygiene after HCT can minimize the severity of infections and facilitate healing of mucositis, particularly before engraftment (BIII) 200, 605. If time permits, all HCT candidates should undergo a dental evaluation and relevant treatment before the start of conditioning therapy (AIII) 606, 607. Patients with prior exposure to bisphosphonates should be treated according to established guidelines 608, 609, 610. Prophylaxis against oral mucositis with keratinocyte growth factor-1 (palifermin) can be considered for patients undergoing myeloablative transplantation [611]. Palifermin reduces the incidence, severity, and duration of mucositis, and the incidence of febrile neutropenia, and may diminish the risk of bacteremia following autologous HCT [611]. Elective dentistry should be postponed until the patient has demonstrated substantial immune recovery [607]. Patients' dentists should eliminate likely sources of dental infection, for example, by restoring teeth with moderate to severe caries and repairing ill-fitting dental prostheses (BIII) [607]. Dentists should extract teeth compromised by moderate to severe periodontal disease, nonrestorable carious teeth, and partially impacted teeth 200, 607. Ideally, at least 10 to 14 days should elapse between the completion of tissue-invasive oral procedures and onset of conditioning therapy, to enable adequate healing and monitoring for postsurgical complications (AIII) [607].
HCT recipients with mucositis and HCT candidates undergoing conditioning therapy should maintain oral hygiene by performing oral rinses 4 to 6 times/day with sterile water, normal saline, or sodium bicarbonate solutions (AIII) [605]. HCT recipients and candidates should brush their teeth 2 to 3 times/day with a soft regular toothbrush that is replaced regularly 605, 607. If the recipient cannot tolerate this, a foam tooth swab can be used (CIII), although these control plaque and prevent caries less effectively than toothbrushes and must be used gently as they can exacerbate mucositis 607, 612. Using toothpaste is optional, depending on the recipient's tolerance (CIII) [607]. HCT recipients and candidates undergoing conditioning therapy who are skilled at dental flossing should floss daily if this can be done without trauma (BIII). Routine dental supervision to monitor and guide the patient's maintenance of oral and dental hygiene should be provided or encouraged (BIII).
To decrease the risk for mechanical trauma and infection of oral mucosa, HCT patients should not wear fixed orthodontic appliances or space maintainers from the start of conditioning therapy until preengraftment mucositis resolves or during any subsequent periods of mucositis (EIII) [607]. Dental and transplant teams and the patient's community dentist should coordinate removal of these appliances and long-term rehabilitation of any oral lesions (BIII). Patients should minimize the use of removable dentures during conditioning and the early posttransplantation period to reduce the potential for mucosal injury. It is recommended that HCT recipients at risk for mucositis wear dentures only while eating, clean them twice daily with a soft toothbrush, and, when not wearing them, soak dentures in antimicrobial denture soaking solution that is changed daily (BIII) [607].
Patients with GVHD of the oral cavity should undergo frequent dental evaluation because of the accelerated pace of dental caries in those patients (BIII) [613]. Dental caries, gingivitis, and periodontal disease must be managed promptly to avoid infectious sequelae, including periodontitis and dental abscesses. Regular brushing, flossing, rinsing, fluoride treatments, and management of xerostomia are basic elements of oral care of patients with GVHD (BIII) [613].
Preventing Intravascular Catheter-Associated Infections
HCT personnel should follow established practices for preventing infections related to intravascular devices (see Bacterial Infections: Recommendations Regarding Catheter-Associated Infections). For central venous access among children, nonimplanted devices are preferred because implantable devices (defined as central venous catheters [CVCs] that are tunneled beneath skin with a subcutaneous port that can be accessed with a needle) have an insufficient number of lumens for optimal patient management immediately after HCT (AIII). In addition, especially among children <4 years old, the skin is often too fragile to tolerate repeated punctures over a port site.
Patients or parents and caretakers should be trained in the care of intravascular devices (AIII) [182]. For devices that are not totally implantable, contact with tap water at the central venous catheter skin site should be avoided (BIII), and patients should cover and protect the catheter tip or end cap during bathing or showering to avoid contamination from tap water. Intravenous infusions given outside of clinic should be started by a caregiver, not the patient (BII) [614].
Prevention and Control of Specific Healthcare-Associated Infections
Recommendations Regarding Legionella Species
HCT physicians should always include Legionnaires' disease (LD) in the differential diagnosis of pneumonia among HCT recipients (AIII) 531, 534. Legionella outbreaks have occurred on transplant wards 615, 616, 617. Appropriate tests to confirm LD include culture of sputum, BAL, and tissue specimens; testing BAL specimens for Legionella by direct fluorescent antibody; and testing for Legionella pneumophila serogroup 1 antigen in urine. However, several of the Legionella outbreaks in immunocompromised patients described in the literature were because of species other than L. pneumophila serogroup 1 species, and therefore would not have been detected by the urine antigen assay 615, 616, 617. Currently, sufficient evidence is lacking to make recommendations regarding Legionella PCR testing.
The incubation period for LD is usually 2 to 10 days. Thus, laboratory-confirmed legionellosis that occurs in a patient who has been hospitalized continuously for ≥10 days before the onset of illness is regarded as a definite case of nosocomial LD, and a laboratory-confirmed infection that occurs 2 to 9 days after hospital admission is a possible case of nosocomial LD 531, 534. When a definite or possible case of laboratory-confirmed nosocomial LD 615, 618 is identified in a person who was in the inpatient HCT center during all or part of the 2 to 10 days before illness onset, or if 2 or more cases of laboratory-confirmed LD occur among patients who had visited an outpatient HCT center, the case(s) should be reported to the local or state health department if the disease is reportable in that state or country or if assistance is needed (AIII) 531, 534. Additionally, in consultation with the hospital infection control team, a thorough epidemiologic and environmental investigation should be conducted to determine the likely environmental source(s) of Legionella species (eg, showers, tap water faucets, cooling towers, and hot water tanks) (AI) 531, 534. The source of Legionella infection should be identified and decontaminated (AIII). Extensive hospital investigation of an isolated case of possible nosocomial LD might not be indicated if the patient has had limited contact with the inpatient HCT center during most of the incubation period (CIII).
HCT centers should follow published recommendations regarding prevention of nosocomial Legionellosis (BIII) 531, 534. HCT center personnel should use only sterile water (ie, not distilled nonsterile water) to fill reservoirs of nebulization devices [534] and to rinse nebulization devices and other semicritical respiratory-care equipment after cleaning or disinfection (BII). HCT centers should not use large-volume room air humidifiers that create aerosols (eg, by Venturi principle, ultrasound, or spinning disk), and thus, are actually nebulizers (DI) [534] unless these humidifiers are sterilized or subjected to daily high-level disinfection and filled with sterile water only (CIII) [534].
When a new hospital with an HCT center is constructed, the cooling towers should be placed so that the tower drift is directed away from the hospital's air-intake system, and the cooling towers should be designed so that the volume of aerosol drift is minimized (BII) [534]. For operational hospital cooling towers, hospitals should install drift eliminators, regularly use an effective biocide, maintain cooling towers according to the manufacturer's recommendations, and keep adequate maintenance records (BII) [534].
Decorative fountains should not be installed in HCT units or areas in a healthcare facility frequented by HCT recipients (BIII). A clonal outbreak of LD linked to a decorative fountain despite efforts to prevent Legionella contamination was recently described [619]. Available data do not suggest that drinking tap water poses a risk for Legionella exposure among HCT recipients in the absence of an outbreak.
Because HCT recipients are at higher risk for disease and death from legionellosis compared with other hospitalized persons [615], periodic routine culturing for Legionella species in water samples from the HCT center's potable water supply could be included as part of an overall strategy for preventing LD (CIII) 531, 534, 620, 621. However, the optimal methodology (ie, frequency or number of sites) for environmental surveillance cultures in HCT centers has not been determined, and the cost-effectiveness of this strategy has not been evaluated 531, 534. Because a “safe” concentration of Legionella organisms in potable water has not been established, the goal should be to maintain water systems with no detectable organisms (AIII) 531, 534. If Legionella species are detected in the water supplying an HCT center, the following measures should be performed until Legionella species are no longer detected by culture 531, 534:
-
•
Decontaminate the water supply (AII).
-
•
Prevent patients from showering with LD-contaminated water. Instead, provide HCT recipients with sponge baths using water that is not contaminated with Legionella species (eg, use water that is not from the HCT center's potable water system) (BIII).
-
•
Do not use faucets containing LD-contaminated water in patient rooms or the HCT center or ambulatory HCT clinics to avoid creating infectious aerosols (DIII).
-
•
Provide HCT recipients with sterile water instead of tap water for drinking, brushing teeth, or flushing nasogastric tubes during proven or suspected Legionellosis outbreaks (BIII).
Recommendations regarding methicillin-resistant Staphylococcus aureus (MRSA)
HCT centers should follow published recommendations to prevent healthcare-associated transmission of MRSA 144, 622, 623, 624, 625. To prevent MRSA transmission, adherence to the following infection control practices is recommended 144, 579, 623, 624:
-
•
Perform hand hygeine with either an alcohol-based hand rub or wash hands with soap and water if soiled before and after all patient contact or contact with the patients' potentially contaminated equipment or environment (AII);
-
•
Use contact precautions for patients colonized or infected with MRSA, including the use of gloves and gowns (AII);
-
•
Ensure adherence to standard environmental cleaning with an effective disinfectant (BIII) 531, 539.
There is insufficient evidence at present to recommend routine screening of all HCT recipients for MRSA or the use of topical or systemic antimicrobial therapy for patients with asymptomatic MRSA colonization, although these are areas of active research 626, 627. If high rates of MRSA persist despite implementation of basic infection control practices, HCT centers should consider adjunctive strategies, such as implementing a program to obtain MRSA surveillance cultures on admission and serially (eg, weekly) (BII) [628] with or without decolonization therapy (BIII) [627]; routine bathing of patients with chlorhexidine (BIII) [629]; cohorting of MRSA patients in designated areas (eg, bays, patient care areas); or by assigning care to dedicated staff (CIII) [624]. The potential impact of rapid molecular techniques to identify S. aureus nasal carriage to prevent MRSA transmission is another area of active research, and recommendations regarding use of rapid techniques for MRSA cannot be provided at this time. The emergence of community-associated MRSA infection has further complicated the prevention and control of MRSA. Optimal strategies for prevention of transmission and infection with community-associated MRSA are currently unknown.
The optimal duration of contact precautions for patients with MRSA is unknown, although studies have shown that patients can remain persistently colonized for extended periods of time. Some HCT centers may opt to implement MRSA discontinuation criteria (eg, continue contact precautions until all antimicrobials active against the MRSA isolate are discontinued and 3 consecutive screening cultures taken on separate days are negative) (CIII) [624].
For patients with recurrent MRSA infection, eradication of the carrier state can be attempted by applying a 2% mupirocin calcium ointment to the nares, use of topical antiseptics such as chlorhexidine for bathing, or administration of systemic antimicrobials, although these strategies have not been shown to be consistently effective (Appendix 1) (CII) 630, 631. High-level mupirocin-resistant MRSA has been reported in Europe, the Middle East, and South America 632, 633, 634, 635, 636, but is less common in the United States. As with any antimicrobial agent, incorrect use or overuse of mupirocin can result in the emergence of mupirocin-resistant staphylococci; therefore, mupirocin use should be coordinated with the local hospital infection control team 632, 633.
Selection of systemic antimicrobials used to treat MRSA infection should be guided by susceptibility patterns. Recommendations for treatment of MRSA infections are beyond the scope of this guideline; refer to published treatment guidelines.
Recommendations regarding Staphylococcus species with reduced susceptibility to vancomycin
All HCT centers should have sufficient laboratory capability to identify all Staphylococcus isolates and their antimicrobial susceptibility patterns, including vancomycin susceptibility (AII) 637, 638, 639. Additionally, HCT centers should conduct routine surveillance for the emergence of Staphylococcus species strains with reduced susceptibility to vancomycin (AIII) 640, 641. Reduced susceptibility to vancomycin is defined as those S. aureus strains that have a vancomycin minimum inhibitory concentration (MIC) of ≥2 μg/mL and those coagulase-negative staphylococci that have a vancomycin MIC of ≥4 μg/mL.
If a patient in an HCT center is colonized or infected with a staphylococcal strain that has reduced susceptibility to vancomycin, infection control personnel should follow published guidelines for the control of such species (BIII) 638, 642, 643. Avoiding overuse and misuse of antimicrobials will decrease the emergence of Staphylococcus species with reduced susceptibility to vancomycin 641, 642. Therefore, medical and ancillary staff members who are responsible for monitoring antimicrobial use patterns in the facility should routinely review vancomycin use patterns (AII) 638, 642, 643. Additionally, HCT center personnel should institute prudent use of all antimicrobials, including vancomycin, to prevent the emergence of Staphylococcus with reduced susceptibility to vancomycin (AII) 638, 640, 642, 643, 644, 645.
Recommendations regarding VRE
HCT recipients frequently have multiple risk factors for VRE colonization and infection, and VRE infection is associated with poor outcomes among HCT recipients 646, 647. A history of resolved VRE bacteremia or known VRE colonization, however, is not a contraindication for HCT (BIII).
To reduce the risk of VRE infection, HCT clinicians should minimize the use and duration of treatment with vancomycin and antimicrobial agents with anti-anaerobic coverage (eg, metronidazole and third-generation cephalosporins) (AII) 638, 648, 649, 650. Although both oral vancomycin and metronidazole promote overgrowth of VRE in stool, the risk of acquiring VRE or promoting VRE overgrowth should not be a defining consideration when selecting oral vancomycin for treatment of severe or recurrent C. difficile infection (BIII) 650, 651.
Patients colonized with VRE typically remain colonized for long periods extending beyond the hospitalization during which the initial VRE-positive culture result was obtained 652, 653. VRE may “reemerge” after prior negative cultures when the patient is reexposed to antimicrobials 624, 654. Contact precautions for HCT recipients with past VRE colonization or infection should be continued during hospital readmissions (AIII). Although there are insufficient data to provide recommendations regarding the optimal duration of precautions for HCT recipients with a history of VRE, individual HCT centers can consider implementing criteria for discontinuation of contact precautions (eg, 3 consecutive sets of screening cultures negative for VRE obtained on separate days for a patient who is not receiving antimicrobial therapy active against the VRE isolate).
To prevent VRE transmission, the following infection control measures are recommended 579, 655:
-
•
Perform hand hygiene with an alcohol-based hand rub or by washing hands with soap and water if soiled before and after all patient contacts or contact with the patients' potentially contaminated equipment or environment (AII);
-
•
Use contact precautions for patients colonized or infected with VRE, including the use of gloves and gowns (AII).
-
•
Ensure adherence to standard environmental cleaning with an appropriate disinfectant (BIII) [539].
Antimicrobial treatment of VRE carriers has not been adequately studied, and carries potential risks, including toxicity and the development of drug-resistant organisms; therefore, it should be discouraged (DIII). The effectiveness of active surveillance cultures for VRE to prevent health care-associated transmission of VRE is unknown. VRE rectal or stool-active surveillance cultures to identify colonized patients can be considered if there is evidence for ongoing transmission of VRE on a HCT unit (CIII) [622].
Recommendations regarding multidrug-resistant Gram-negative bacilli (MDR-GNB)
MDR-GNB are defined as GNB that are resistant to 1 or more classes of antimicrobial agents, including those producing extended-spectrum beta-lactamases (ESBLs) and carbapenemases. In addition to Escherichia coli and Klebsiella pneumoniae, MDR-GNB includes highly resistant strains of Acinetobacter baumannii and organisms such as Stenotrophomonas maltophilia, Burkholderia cepacia, and Ralstonia pickettii with intrinsically broad antimicrobial resistance. A detailed discussion of MDR-GNB and recommendations for prevention was published in 2006 [624]. Judicious use of antimicrobial agents is important for control of MDR-GNB (BII). Several studies have demonstrated a temporal association between formulary changes and decreased incidence of targeted MDR-GNB [656].
As with other multiply drug-resistant organisms, adherence to the following infection control measures is important to prevent health care-associated transmission 144, 579:
-
•
Perform hand hygiene with an alcohol-based hand rub or by washing hands with soap and water if soiled before and after all patient contacts or contact with the patients' potentially contaminated equipment or environment (AII);
-
•
Use contact precautions, including gowns and gloves, for patients colonized or infected with MDR-GNB that are of epidemiologic importance for each HCT center (AII);
-
•
Ensure adherence to standard environmental cleaning with an effective disinfectant (BIII) 531, 539.
Based on current evidence, it is unclear whether the addition of an active surveillance-culturing (ASC) program for MDR-GNB to basic infection control practices is useful. Some studies have reported successful reduction of extended-spectrum beta-lactamase-producing Enterobacteriaceae over a 6-year period using a multifaceted control program that included use of ASC 657, 658. Other reports suggest that the use of ASC is not necessary to control endemic MDR-GNBs [659]. However, the CDC and the Health care Infection Control Practices Advisory Committee now recommends that a single round of surveillence be conducted in high risk settings (eg, units where patients are exposed to broad spectrum antimicrobials) if previously unidentified cases of carbapenem-resistant or carbapenemase-producing Klebsiella spp. or E. coli are identified on review of mircobiology records [660]. Units experiencing high rates of MDR-GNB infection can consider the use of ASC as a component of their control and prevention program (CIII), although there is insufficient information to provide a recommendation regarding the use of ASC to prevent transmission of MDR-GNB.
Recommendations regarding C. difficile
HCT centers should follow published recommendations for preventing, controlling, and treating C. difficile infection (CDI) (AI) 661, 662. Patients with CDI should be placed on contact precautions for the duration of illness (BIII) 144, 662. All HCWs who have contact with a C. difficile-infected patient or the patient's environment should don gloves (AI) and gowns (BIII) before entering the patient's room 144, 661, 662, 663, 664. Contact precautions should be continued at least until the patient is asymptomatic (BIII) [661]. A “test of cure” to determine whether contact precautions can be discontinued is discouraged (DII) [661].
If there is evidence of ongoing transmission of C. difficile despite implementation of the basic prevention practices described above, HCT centers should consider maintaining contact precautions even after diarrhea has resolved and until hospital discharge (CIII) [665]. During nosocomial CDI outbreaks, HCT centers should work closely with their Infection Prevention and Control staff to ensure implementation of appropriate control measures (AIII). The following practices are not recommended for prevention of C. difficile transmission:
-
•
Performing routine stool surveillance cultures or toxin assays for C. difficile among asymptomatic patients or HCWs, even during outbreaks (DIII);
-
•
Culturing HCWs' hands for C. difficile (EIII);
-
•
Treating asymptomatic C. difficile carriers to prevent clinical infection (EII) [666].
Hand hygiene compliance may decrease when soap and water is the preferred method. Alcohol-based hand rubs are associated with increased hand hygiene compliance and are more effective at removing nonspore-forming organisms than soap and water 579, 667. However, the use of alcohol-based hand hygiene products after caring for a patient with CDI remains controversial, because alcohol is not sporicidal. Hand washing with soap and water has been shown to be effective at removing Bacillus atrophaeus spores [668]. Presumably, if gloves are worn when caring for a patient with C. difficile infection, spores are physically removed during glove removal. In the setting of an outbreak or ongoing C. difficile transmission in a HCT center, the center should consider instructing visitors and HCWs to wash hands with soap and water after contact with patients with CDI or their equipment or environment (BIII). If soap and water are used, proper technique (ie, a minimum 15 to 30 seconds of hand washing) should be ensured (BI) [579].
C. difficile is primarily transmitted from the hands of HCWs. However, C. difficile can also be acquired from the hospital environment. Currently there are no approved sporicidal hospital disinfectants. Sodium hypochlorite-based cleaning agents (ie, household bleach in water at a 1:10 dilution or at least 5000 ppm available chlorine) are sporicidal within 10 minutes [669]. One study demonstrated a significant decline in CDI rates on an HCT unit with the use of a bleach-based cleaning agent [670]. The use of bleach-containing cleaning agents for environmental disinfection should be considered when there is evidence of ongoing C. difficile transmission (BII). The optimal frequency (eg, daily cleaning or cleaning after discharge of the patient from the room) and extent (eg, only rooms of patients with CDI, all patient rooms on the unit, or the addition of areas outside of patient rooms) of cleaning with bleach-containing agents are unresolved issues.
Recommendations regarding community respiratory virus infections
HCT centers should institute appropriate precautions and infection control measures for preventing transmission of CRV among hospitalized HCT recipients and candidates undergoing conditioning therapy (AIII) 310, 534, 563, 671. HCT recipients or candidates with URI or lower respiratory infection (LRI) symptoms because of suspected CRV infection should empirically be placed on contact plus droplet precautions until a specific pathogen has been identified. After identification, precautions should be adjusted based on published guidelines to avoid transmitting infection to other HCT candidates and recipients, HCWs, and visitors (BIII) [144]. Pathogen-specific CRV isolation precautions include:
-
•
Contact precautions for RSV and parainfluenza;
-
•
Droplet precautions for influenza;
-
•
Droplet plus contact precautions for adenovirus; and
-
•
Airborne plus contact precautions for primary or disseminated varicella infection.
Personal protective equipment (eg, gown, gloves, surgical mask, and eye protection) should be donned prior to entering and discarded upon exiting a patient's room to ensure that personal protective equipment is always changed between patients (AIII) [144].
When caring for an HCT recipient or candidate undergoing conditioning therapy with URI or LRI, HCWs and visitors should disinfect hands with an alcohol-based gel or, if hands are visibly soiled, with soap and water (AII):
-
•
Before and after each contact with a patient;
-
•
After handling respiratory secretions or fomites potentially contaminated with patients' secretions; and,
- •
Adherence to hand hygiene is critical because most respiratory infections are transmitted by direct contact, particularly by hand to nose and eye. Use of a mask without appropriate hand disinfection, glove-wearing, or facial protection is insufficient to prevent transmission of CRV infections. While performing aerosol-generating procedures (eg, bronchoscopy, open suctioning of the respiratory tract [ie, unless in-line suctioning catheters are used], endotracheal intubation), HCWs should wear a face shield that fully covers the front and side of the face, a mask with an attached shield, or a mask and goggles, along with gloves and gowns to avoid contamination from the patient's respiratory secretions regardless of the presence or absence of CRV symptoms (AIII).
HCT centers should consider daily screening of all persons who enter the center, including HCWs and visitors, for URI symptoms, especially during nosocomial or community outbreaks of CRV infection (Appendix 2) (BIII). HCT center HCWs with URI symptoms should be restricted from patient contact and reassigned to nonpatient care duties until symptoms resolve (BII). Visitors with URI symptoms should be asked to defer their visit to the HCT center until their URI symptoms resolve (AII) [298]. HCWs and visitors with infectious conjunctivitis should be restricted from direct patient contact until the drainage resolves (AII) [599].
Respiratory secretions of any hospitalized HCT candidate or recipient with signs or symptoms of CRV infection should be tested promptly by viral culture or rapid diagnostic tests for CRV (BIII) (see Viral Infections section). Appropriate samples include nasopharyngeal washes, swabs, aspirates (with or without throat swabs), and BAL fluid. This practice permits timely initiation of isolation precautions to prevent transmission to other patients and HCWs as well as preemptive treatment of certain CRVs (eg, influenza) [672] that might prevent severe disease and death among HCT recipients [534].
HCT centers should obtain nasopharyngeal swabs, throat swabs, or aspirates for culture, PCR, or rapid antigen testing to help determine whether patients have stopped shedding virus (BIII). To prevent nosocomial transmission of CRV [673], HCT centers should recognize that prolonged CRV shedding may occur. Viral shedding has been reported to last ≥4 months for influenza [305], ≥2 years for adenovirus 674, 675, and ≥22 days for RSV [673], mainly in patients treated with steroid doses of >1 mg/kg [309]. RSV viral shedding has been reported to last 112 days in a child with severe combined immunodeficiency [676]. HCT recipients with CRV infection should be placed on the appropriate precautions for at least the duration of illness (AII) and precautions should be continued for the duration of hospitalization or viral shedding to prevent transmission within the unit (CIII).
Some HCT centers conduct routine CRV surveillance among asymptomatic HCT recipients to detect outbreaks and implement infection control measures as early as possible [677]. To date, however, data are insufficient to provide recommendations regarding routine surveillance testing of asymptomatic patients for CRV infections.
During periods of widespread RSV or influenza activity in the surrounding community or suspected health care-associated CRV outbreaks, all HCT recipients and candidates with signs or symptoms of respiratory infection should be tested for RSV and influenza infection (ie, the presence of RSV and/or influenza antigen in respiratory secretions tested by enzyme-linked immunosorbent assay and viral culture). During an outbreak of health care-associated RSV infection, managers should cohort health care personnel as much as is practical (ie, restrict personnel who care for RSV-infected patients from giving care to uninfected patients) (CIII) 297, 298, 678. No recommendation can be made for cohorting of personnel during an outbreak of other health care-associated CRV infections.
HCWs and close contacts of HCT recipients should receive yearly influenza vaccine at the start of the influenza season, preferably with trivalent inactivated influenza vaccine rather than live attenuated influenza vaccine to avoid concerns about transmission of vaccine virus (Table 7) (AI). Use of influenza vaccine along with prophylaxis and early antiviral therapy among at-risk healthcare workers and high-risk patients reduces the spread of influenza within healthcare facilities [534]. During an outbreak of health care-associated influenza infection, in addition to use of droplet precautions for patients with suspected or confirmed influenza and rapid influenza virus testing for symptomatic patients, HCT centers should consider rapid influenza virus testing of symptomatic HCT staff; administration of inactivated influenza vaccine to unvaccinated HCT staff and appropriate patients (see HCT Recipient Vaccinations section); and administration of influenza antiviral chemoprophylaxis to HCT staff and patients according to current recommendations (BIII) [603]. Healthcare personnel with influenza should be excluded from work for 5 days following the onset of symptoms (AIII) 303, 679.
Preventing CRV exposure among HCT recipients after hospital discharge is more challenging because of high CRV prevalence. Preventive measures should be individualized in accordance with the immunologic status and tolerance of the patient. In outpatient waiting rooms, patients with CRV infections should be separated to the extent possible from other patients and should be instructed to use respiratory hygiene/cough etiquette (BIII).
Recommendations Regarding Adenovirus
Adenovirus can cause large outbreaks in hospital settings and chronic care facilities. Outbreaks of diarrheal illness because of adenovirus have been previously described among adult HCT recipients [680]. Transmission can occur via inhalation of aerosolized droplets; direct and indirect contact through contaminated surfaces; fecal-oral spread; exposure to infected tissue or blood; and rarely, contaminated water [330]. Sputum or oral secretions of infected adults contain 106 to 107 particles/mL. Nonimmune adults can be infected by inhaling as few as 5 viral particles [681]. Prolonged fecal shedding has been demonstrated in HIV-infected patients with and without diarrhea. Immunocompromised hosts are known to have asymptomatic shedding of adenovirus from the GI and respiratory tracts for months after initial infection. Furthermore, immunosuppressed individuals may either reactivate endogenous infection or acquire new infection. Adenovirus survives on nonporous surfaces up to 35 days 682, 683, 684.
Recommendations for isolation precautions in the hospital setting depend on the type of syndrome caused by adenovirus. HCT recipients with adenoviral gastroenteritis should be placed on contact precautions for at least the duration of illness (AIII), and precautions should be continued for the duration of hospitalization or viral shedding (CIII) to prevent transmission within the unit. For cases of respiratory illness or disseminated infection associated with adenovirus, droplet and contact precautions should be maintained for at least the duration of illness (AIII) [144]. For cases of adenoviral conjunctivitis in immunosuppressed patients, contact precautions (AIII) and droplet precautions (CIII) should be instituted for at least the duration of illness (usually 5-7 days) (CIII) [144].
Hand hygiene with either an alcohol-based hand rub or soap and water has been shown to be effective against adenovirus (BIII) [685]. Environmental disinfection of surfaces with hospital-approved disinfectants (eg, chlorine-based products, ethyl alcohol, ethanol mixed with quaternary ammonium compounds) is important to limit the spread of adenoviral infection [686].
Recommendations Regarding Viral Gastroenteritis
Viral gastroenteritis is most commonly spread by the fecal-oral route. Common pathogens include rotavirus, norovirus, astrovirus, and adenovirus (see the Adenovirus section for adenovirus-specific recommendations). To prevent the acquisition and spread of viral gastroenteritis among HCT recipients, HCT centers should ensure adherence to hand hygiene, appropriate isolation precautions, and environmental disinfection (AII) [531]. Appropriate precautions should be maintained for at least the duration of illness (AII). Because HCT recipients may continue to shed virus after symptoms resolve, HCT centers may choose to continue precautions for the duration of hospitalization, or the duration of viral shedding if diagnostic laboratory testing is available (CIII).
Rotavirus
Rotavirus is the most common cause of severe gastroenteritis in infants and young children worldwide. Virus is shed in high concentrations in the stool of infected children and is transmitted primarily by the fecal-oral route, through person-to-person contact and fomites [687]. Environmental contamination is common because rotavirus can survive on nonporous surfaces for more than 10 days 688, 689. Health care-associated transmission because of rotavirus infection has been linked to toys [594] and contaminated hands [690]. Contact precautions should be implemented for HCT recipients with suspected or confirmed rotavirus gastroenteritis to prevent transmission in the health care setting (AIII). Alcohol-based hand gel has some virucidal activity against rotavirus and is sufficient for routine hand hygiene unless hands are visibly soiled [685]. Because prolonged shedding can occur in immunocompromised patients, HCT staff should ensure consistent environmental cleaning and disinfection and removal of soiled diapers (AIII) [144]. If soiled diapers need to be weighed outside of the patient room, it is important to ensure environmental disinfection of items in contact with soiled diapers (eg, cover the scale with paper, appropriately discard soiled diapers and paper, and disinfect the scale after each use) (BIII).
Norovirus
Noroviruses are the most common cause of outbreaks of nonbacterial gastroenteritis. Fecal-oral transmission is most common, although environmental and fomite contaminations are also important sources of infection. Aerosolization of vomitus resulting in droplets that contaminate surfaces or are swallowed also contribute to transmission. Infected individuals are contagious up to 72 hours after recovery. The low infectious dose (<100 particles), multiple modes of transmission, prolonged duration of viral shedding even among asymptomatic people, and the ability to survive high levels of chlorine and varying temps (freezing and heating to 60 °C) contribute to ease of norovirus transmission. Detection of viral RNA in stools by use of RT-PCR assays shows that virus can be isolated as long as 7 days after symptom onset. To reduce transmission of this highly infectious organism, several strategies need to be employed, including hand washing with soap and water, contact precautions, wearing masks while cleaning areas contaminated by feces or vomitus, and minimal handling of soiled linens and clothes (BIII). Use of alcohol-based hand rubs may be inadequate for preventing norovirus transmission 691, 692, 693.
Noroviruses are resistant to usual cleaning and disinfection agents [144]. A hypochlorite-based cleaning agent is recommended for use on hard, nonporous environmental surfaces at a concentration of 1000 ppm (1/3 cup bleach in 1 gallon water for 1:50 dilution) to 5,000 ppm (1 and 2/3 cup bleach in 1 gallon water for 1:10 dilution), depending on the level of soiling and types of environmental surfaces (BII) [694]. In HCT centers without access to prediluted bleach products, staff should prepare a fresh dilution of bleach daily and discard unused portions. Environmental cleaning and disinfection with a hypochlorite-based cleaning agent should be performed at minimum on patient discharge from the room (ie, terminal cleaning) and more frequently as needed (BIII). Quaternary ammonium compounds do not have significant activity because noroviruses are nonenveloped virus particles. Phenolic-based disinfectants are active against noroviruses, but may require concentrations 2 to 4 times higher than manufacturers' recommendations for routine use. Heat disinfection (pasteurization to 60 °C) has been suggested for items that cannot be cleaned with bleach-based disinfectants [695].
Astroviruses
Outbreaks of astrovirus gastroenteritis have occurred in hospitals, nursing homes, and daycare centers 696, 697, 698. Infection can also occur in HCT recipients [699]. Astroviruses can be transmitted via the fecal-oral route, direct and indirect contact, and possibly via contaminated water [700]. Furthermore, virus can persist on fecally contaminated surfaces for several months at relatively low humidity 531, 689. Contact precautions and environmental disinfection should be used to control the spread of astrovirus infection among HCT recipients during known outbreaks (AII). Quaternary ammonium compounds and chlorine solutions can be effective in inactivating enteric viruses provided a cleaning step to remove organic matter precedes terminal disinfection [531].
Infection Control Surveillance
Institutions should follow standard guidelines for surveillance of epidemiologically significant nosocomial pathogens (eg, MRSA, VRE, MDR-GNB, C. difficile, invasive mold infections) and their susceptibility patterns (BIII) 624, 701. In the absence of epidemiologic clusters of infections, there is no need to perform routine bacterial surveillance cultures of the HCT center environment, equipment, or devices used for respiratory therapy, pulmonary-function testing, or delivery of inhalation anesthesia (DIII) [534]. Furthermore, in the absence of a nosocomial fungal outbreak, it is not necessary to perform routine fungal cultures of devices or dust (eg, settle plates) in the rooms of HCT recipients and candidates undergoing conditioning therapy (DIII). However, HCT center personnel should perform routine surveillance for cases of invasive mold disease, including aspergillosis, occurring among HCT recipients (BIII). The optimal surveillance definition for nosocomial invasive mold disease is unclear. Because HCT recipients have frequent contact with ambulatory healthcare facilities and the incubation period for invasive mold disease is unknown, all cases of invasive mold infection should be tracked regardless of time to onset after admission (BIII). Cases of invasive mold disease with onset of symptoms ≥7 days after hospital admission are more likely to be hospital acquired. Surveillance definitions can include definitions for proven, probable, and possible cases and criteria can include culture, histology, host factors, indirect tests for detection of fungal antigen or cell-wall constituents, and clinical (including radiology) data (BIII) [702]. An increase in the number of cases or in the incidence of invasive mold disease among HCT recipients should trigger careful evaluation of the HCT center environment for sources of mold exposure. In addition, the ventilation system should be evaluated to ensure adequate filtration, air flow, and air pressure differentials (see Ventilation section) (BIII) [547].
Safe Living after HCT
D. Yokoe, C. Casper, E. Dubberke, G. Lee, P. Muñoz, T. Palmore, K. Sepkowitz, J.-A.H. Young, J.P. Donnelly
Avoiding Environmental Exposures
Hematopoietic cell transplantation (HCT) recipients and their caregivers should be educated regarding strategies to avoid environmental exposures to opportunistic pathogens (AIII).
Preventing infections primarily transmitted by direct contact
HCT recipients and candidates should perform frequent and consistent hand hygiene (AII), especially related to the following;
-
•
before eating or preparing food;
-
•
after changing diapers;
-
•
after touching plants or dirt;
-
•
after urinating or defecating;
-
•
after touching animals;
-
•
after touching body fluids or excretions or items that might have had contact with human or animal feces (eg, clothing, bedding, toilets, or bedpans);
-
•
after going outdoors or to a public place;
-
•
after removing gloves;
-
•
after collecting or depositing garbage; and
-
•
before and after touching catheters or wounds [579].
Conscientious hand hygiene using an alcohol-based hand gel or soap and water is especially critical during the first 6 months after HCT and during other periods of substantial immunosuppression (eg, treatment of GVHD, systemic steroid use, or relapse of the underlying disease for which the transplant was performed). Pediatric HCT recipients and candidates should be supervised by adults during hand disinfection to ensure thorough cleaning. Hand hygiene should be performed with an alcohol-based hand gel or with soap and water (AII) 703, 704. HCT recipients who visit or live on farms should follow published recommendations for preventing cryptosporidiosis 705, 706.
HCT recipients should avoid changing diapers whenever possible (BIII). Even if gloves are worn, hands should always be washed after gloves are removed, as gloves may have defects that are not easily visible (AIII) 707, 708.
Preventing infections primarily transmitted through respiratory exposures
CRV infections can cause severe disease and mortality in HCT recipients [709]. To prevent respiratory infections after hospital discharge, HCT recipients should observe the following precautions:
-
•
Perform frequent and thorough hand hygiene (BIII) and avoid touching their eyes, nose, and mouth unless they have disinfected their hands to avoid inoculating themselves with CRV;
-
•
Avoid close contact with persons with respiratory illnesses (BIII). When close contact is unavoidable, both the HCT recipient and the symptomatic contact should be encouraged to disinfect their hands frequently, as this has been shown to reduce the transmission of respiratory and other illnesses 710, 711, 712, 713. Symptomatic contacts should also consider wearing surgical masks [714] or, at a minimum, cover their mouth and nose with a disposable tissue when sneezing or coughing [715] or, if tissues are not available, using their upper sleeves [603]. Although HCT recipients can wear masks themselves when in contact with symptomatic persons, the degree of protection from wearing masks has not been determined.
-
•
Exercise particularly careful hand hygiene when in crowded areas (eg, shopping malls or public elevators) where close contact with persons with respiratory illnesses is likely, especially during the autumn and winter months (AII).
Visitors to HCT centers can introduce communicable infections through environmental contamination or direct spread. All visitors should perform hand hygiene before and after visits. All visitors should be screened for symptoms of infection: fever, rash, vomiting, diarrhea, upper respiratory symptoms, or conjunctivitis, and should not be allowed to visit a HCT recipient while they are symptomatic (BIII).
HCT recipients and candidates who are undergoing conditioning therapy are also at risk for infections caused by fungal, parasitic, and bacterial pathogens associated with exposure to soil, plants, or their aerosols (eg, Toxoplasma gondii, Histoplasma capsulatum, Cryptococcus neoformans, Nocardia species, and Aspergillus species). To prevent such infections, HCT recipients and candidates should follow the following precautions:
-
•
During the first 6 months after HCT and periods of substantial immunosuppression (eg, treatment for GVHD, systemic steroid use, or relapse of the underlying disease for which the transplant was performed), HCT recipients and candidates who are undergoing conditioning therapy should avoid gardening or direct contact with soil, plants, or their aerosols (BIII). If such activity is unavoidable, these individuals should wear protective gloves to prevent abrasions or lacerations and surgical or N95 masks while touching plants or soil, although the effectiveness of gloves and masks to protect against infections associated with exposure to soil is unknown 716, 717, 718, 719, 720, 721. HCT recipients should avoid creating plant or soil aerosols through activities such as spreading mulch 722, 723. HCT recipients should always thoroughly wash their hands with soap and water after soil or plant contact.
-
•
HCT recipients should avoid construction or excavation sites or other dust-laden environments for the first 6 months after HCT and during other periods of substantial immunosuppression to avoid exposures to molds [724]. Outpatient HCT recipients should be advised of travel routes to the HCT center that will avoid or minimize exposure to construction sites (CIII).
Coccidioidomycosis is uncommon after allogeneic HCT, but can present as severe disease 725, 726. HCT recipients traveling to or residing in areas where coccidioidomycosis is endemic (eg, the American Southwest, Mexico, and Central and South America) should avoid activities that cause exposure to dust [727], and should minimize exposure to dust or disturbed soil, including construction or excavation sites, areas with recent earthquakes, farms, or other rural areas (CIII). Histoplasmosis (Histoplasma capsulatum infection) after allogeneic HCT is also rare. However, HCT recipients in histoplasmosis-endemic areas should avoid exposure to bird-roosting sites and caves [728] and should avoid exposure to bird or bat droppings (CIII) 729, 730.
HCT candidates or recipients should be advised that certain activities and occupations (eg, work in healthcare settings, prisons, jails, or homeless shelters) can increase their risk for tuberculosis (TB) exposure 731, 732. In deciding whether a HCT recipient should continue activities in these settings, physicians should evaluate the patient's specific duties, the precautions used to prevent TB exposure in the workplace, and the prevalence of TB in the community. The decision to continue or terminate such activities should be made jointly between patient and physician. HCT recipients should avoid exposure to persons with active TB, particularly during the first 6 months after HCT and during other periods of substantial immunosuppression (BIII).
Persons whose occupations involve animal contact (eg, veterinarians, pet store employees, farmers, or slaughterhouse workers) may be at increased risk for toxoplasmosis, Q fever, brucellosis, and other zoonotic diseases. Although data are insufficient to justify a general recommendation against HCT recipients working in such settings, these exposures should be avoided during the first 6 months after HCT and during other periods of substantial immunosuppression (BIII).
HCT recipients should exercise caution when handling household trash. Air surrounding solid waste has been shown to contain culturable, aerosolized bacteria, and fungi [733]. Although there have been no cases of infection definitively linked to routine handling of solid waste, it is recommended that HCT recipients wear masks and use gloves when handling substantial volumes of waste (CIII).
Recommendations Regarding Household Members who Receive Live-Attenuated Vaccines
Household members should receive recommended vaccines to prevent illness in the household and reduce the risk of transmission of infections to the HCT recipient (Table 7). When a choice between a live-attenuated vaccine and an inactivated vaccine exists (eg, influenza, polio), household members should receive inactivated vaccine to reduce the potential risk for transmission of the vaccine virus. To ensure that HCT patients are optimally protected from vaccine-preventable diseases through vaccination of household contacts while minimizing the risk of transmission of vaccine virus, clinicians should consider the following data and current recommendations:
-
•
Polio vaccine (AII): household members of HCT recipients should receive inactivated polio vaccine (IPV), because oral polio vaccine (OPV) is a live-attenuated vaccine that has been rarely associated with paralysis in healthy recipients and their contacts. If OPV vaccine is inadvertently given to a household contact, close contact should be avoided for approximately 4 to 6 weeks after vaccination (AII). If this is not feasible, HCT recipients should perform careful hand hygiene after any contact with feces from the vaccinated household member (eg, after changing the diaper of a vaccinated child) and avoid contact with saliva (eg, shared food or utensils) to reduce the risk of transmission of vaccine-associated polio virus. Infants and children who have recently received the OPV vaccine should be excluded from visiting the HCT unit for 4 to 6 weeks after receipt of vaccine to minimize the risk of transmission and vaccine-associated paralytic poliomyelitis among HCT recipients (AII) [734].
-
•
Rotavirus vaccine (CIII): 2 rotavirus vaccines are currently available. RV5 is a human-bovine reassortant vaccine available as a 3-dose series for use in infants at 2, 4, and 6 months of age to prevent rotavirus gastroenteritis [735]. RV1 is a human strain that is available as a 2-dose series for use in children at 2 and 4 months of age 736, 737, 738. Shedding has been shown to occur up to 15 days after a dose of RV5 and 30 days after RV1, although there have been no documented cases of transmission of vaccine virus 735, 739. Thus, routine standard precautions are currently recommended for recently immunized infants. To minimize the risk of potential vaccine virus transmission, HCT recipients should avoid handling diapers if possible. If this is not feasible, HCT recipients should perform careful hand hygiene after any contact with feces from the vaccinated household member (AIII). Until further data on the risks of transmission of vaccine virus become available, HCT units may wish to exclude infants who have recently received rotavirus vaccine from visiting for 2 to 4 weeks after receiving a vaccine dose (CIII).
-
•
Live-attenuated influenza vaccine (LAIV): studies have demonstrated that 80% of vaccine recipients 8 to 36 months of age shed 1 or more vaccine virus strains for a mean duration of 7.6 days. Thus far, there have not been any documented cases of transmission to an immunocompromised host, and only 1 case of documented transmission has occurred in an immunocompetent host, with an estimated transmission rate of 0.6% to 2.9% in a population of young children enrolled in a childcare center [740]. In adults, shedding mostly occurs within the first 3 days, but can continue for as long as 7 days after vaccination 740, 741. In HIV-infected individuals who had a CDC class of A1-2 and a plasma HIV RNA count <10,000 and CD4 count >200, none shed LAIV virus longer than 10 days. Furthermore, clinical trial data suggest that when vaccine virus is shed by vaccine recipients, none have reverted to wild type 742, 743. Because an alternative trivalent inactivated influenza vaccine (TIV) exists, household members of HCT recipients should receive TIV to avoid potential concerns for transmission of vaccine virus (AI). Health care center personnel and HCT center visitors who receive LAIV instead of TIV should avoid contact with severely immunosuppressed persons for 7 days after vaccination (CIII) [307].
-
•
Zoster vaccine: herpes zoster vaccine is currently recommended for adults 60 years of age and older in the United States [744]. There is no evidence to date that transmission of vaccine-associated virus has occurred. However, HCT centers should exclude visitors who develop a varicella- or zoster-like rash after vaccination (AIII). If a household member develops a varicella or zoster-like rash after vaccination, close contact with the HCT recipient should be avoided and affected areas should be covered (AIII).
-
•
Varicella vaccine (AIII): In 1 small study of 37 healthy children receiving vaccine and their 30 immunocompromised siblings, there was no evidence of vaccine virus transmission [745]. In the postlicensure era, transmission from immunocompetent persons after vaccination has been documented by PCR from only 5 persons resulting in 6 secondary infections, with over 55 million doses estimated to have been given [282]. Because the risk of vaccine virus transmission is low, particularly in the absence of a vaccine-associated rash, household members should receive varicella vaccine to protect HCT recipients from potential exposure to wild-type disease (AIII). Individuals who experience a vaccine-associated rash within 1 month after varicella vaccination should be excluded from visiting the HCT center and should avoid close contact with HCT recipients in the home setting (BIII). Measures such as removal of the vaccinee or transplant recipient from the domicile have not been studied.
-
•
Measles, mumps, rubella (MMR) vaccine (AIII): Household members should receive age-appropriate MMR vaccination as recommended. However, vaccine recipients who develop a fever and/or rash postvaccination should be excluded from visiting the HCT center while symptomatic and should avoid close contact with HCT recipients in the home setting (BIII).
Safe Sex
Sexually active patients who are not in long-term monogamous relationships should always use latex condoms during sexual contact to reduce their risk for exposure to cytomegalovirus (CMV), herpes simplex virus (HSV), HIV, human papilloma virus (HPV), hepatitis b virus (HBV), hepatitis c virus (HCV), and other sexually transmitted infections (STIs) (AII). However, even long-time monogamous partners can be discordant for these infections. Reinfection with some STIs has been reported in seropositive recipients with long-term partners years after transplant [370]. Therefore, sexually active HCT recipients in long-term monogamous relationships should consider using latex condoms during sexual contact to reduce the risk for exposure to these STIs (CIII). Additionally, contact with oral or genital secretions has been associated with acquisition of many STIs, including those from HSV, CMV, Epstein-Barr virus (EBV), HHV-8, and HPV. Severely immunosuppressed HCT recipients should consider completely avoiding unprotected sexual activity (ie, activities that involve contact of the HCT recipient's mucous membranes with saliva, semen, or vaginal secretions) for as long as they remain severely immunodeficient (CIII). Sexually active HCT recipients should avoid sexual practices that could result in oral exposure to feces (AIII) 2, 746.
Animal Safety
Preventing pet-transmitted zoonotic infections
HCT physicians should advise recipients and candidates undergoing conditioning therapy of the potential infection risks posed by pet ownership; however, they should not routinely advise HCT recipients to part with their pets, with limited exceptions. Immunocompromised HCT recipients and candidates should avoid adopting ill or juvenile pets (eg, cats <6 months old) [747] and any stray animals (BIII) 2, 746. Children are more likely to be bitten or scratched by pets, and thus the acquisition of new pets by children who are recent HCT recipients or candidates for HCT is not recommended (DIII). Generally, immunocompromised HCT recipients and candidates undergoing conditioning therapy should minimize direct contact with animals 99, 748, 749, particularly animals that are ill (eg, with diarrhea) (BIII) [747]. Immunocompromised persons who choose to own pets should be aware that the risk of infection varies with the type of animal and where the pet was acquired, with larger, mature animals from established vendors posing a lower risk than other types of animals [747]. Immunocompromised HCT recipients and candidates should avoid ownership of, or contact with, reptiles (eg, lizards, snakes, turtles, and iguanas), because of the risk for salmonellosis (BII) 747, 750, 751, 752. Additionally, patients should be informed that salmonellosis can occur from fomite contact alone [752]. Therefore, HCT recipients and candidates should avoid contact with a reptile, its food, or anything that it has touched. If such contact occurs, recipients and candidates should disinfect their hands thoroughly afterward (AIII). Immunocompromised HCT recipients and candidates should avoid ownership of, or contact with, ducklings and chicks because of the risk for acquiring Salmonella or Campylobacter species infections (BIII) 753, 754. Immunocompromised HCT recipients and candidates should avoid ownership of, or contact with, exotic pets (eg, chinchillas or monkeys or other nonhuman primates) (BII).
HCT recipients and candidates need to be vigilant regarding maintenance of their pet's health (BIII). If the pet becomes ill, the owner should seek veterinary care promptly, to minimize the possible transmission of the pet's illness to the owner [747]. Any pet that experiences diarrhea should be tested for infection (BIII) with Cryptosporidium 2, 746, Giardia species [747], Salmonella, and Campylobacter 2, 747. Owners should ensure that pets are up to date on recommended vaccinations (with the exception of live vaccines) (BIII) [755]. Routine screening of healthy animals for zoonotic diseases is not recommended (DIII) [747].
Feeding pets only high-quality commercial pet foods reduces the possibility of illness caused by spoiled or contaminated foods, thus reducing the possibility of transmitting illness from the pet to the HCT recipient. If eggs, poultry, or meat products are given to the pet as supplements, they should be well cooked. Any dairy products given to pets should be pasteurized (BIII) [747]. Pets should be prevented from drinking toilet bowl water, scavenging, hunting, eating other animals' feces, and from having access to garbage (BIII) [747].
If HCT recipients handle pets or clean cages, they should perform hand hygiene with an alcohol-based hand gel or soap and water (preferred if hands are visibly soiled or after contact with feces). Adults should supervise hand disinfection of pediatric HCT recipients (BIII). HCT candidates and recipients should avoid contact with animal feces (BIII) to reduce the risk for toxoplasmosis, cryptosporidiosis, salmonellosis, and campylobacteriosis [747], and should not clean pet litter boxes or cages or dispose of animal waste (DIII) [1]. If patients are unable to find assistance with these tasks, they should wear disposable gloves during such activities and wash their hands thoroughly afterward (BIII). Bird cage linings and cat litter should be cleaned regularly (eg, daily) [747]. All persons, but particularly immunocompromised HCT candidates and recipients, should wear gloves whenever handling items contaminated with bird droppings (BIII) because droppings can be a source of Cryptococcus neoformans, Mycobacterium avium, or H. capsulatum [747]. To minimize potential exposure to Mycobacterium marinum, immunocompromised HCT recipients and candidates should not clean fish tanks (DIII). If this task cannot be avoided, patients should wear disposable gloves during such activities and disinfect their hands thoroughly afterward (BIII) 747, 754.
HCT candidates or recipients may also come into contact with animals in the medical setting. These animals are typically divided into 3 categories: service, therapy, and visitation [756]. In most settings, access to service animals can not legally be restricted. Animals are increasingly used as adjunctive therapy for patients with cancer and other chronic illnesses. Although many studies attest to the overall benefits of such “therapy” or “visitation” animals in various clinical settings, the risk of acquiring a zoonotic infection for immunosuppressed HCT candidates or recipients needs to be carefully considered, and thus, no recommendation can be given.
Preventing toxoplasmosis
Many cases of primary toxoplasmosis are acquired through eating undercooked meat 747, 749. However, all HCT recipients and candidates, particularly those who are T. gondii seronegative, should be informed of the risks for contracting toxoplasmosis from ingestion of oocytes after contact with cat feces (BIII), although they need not be required to give away their cats (DII). In households with cats, litter boxes should not be placed in kitchens, dining rooms, or other areas where food preparation and eating occur [747]. Additionally, litter boxes should be cleaned daily by someone other than the HCT recipient during the first 6 months after HCT and during periods of substantial immunosuppression to reduce the risk for transmitting toxoplasmosis to the HCT recipient (BIII). Daily litter box changes will minimize the risk for fecal transmission of T. gondii oocysts, because fecal oocysts require >1 days of incubation to become infectious. If HCT recipients perform this task during the first 6 months after HCT and during subsequent periods of substantial immunocompromise (eg, during GVHD, systemic steroid use, or relapse of the underlying disease for which the transplant was performed), they should wear disposable gloves [747]. Gloves should be discarded after a single use (BIII). Soiled, dried litter should be disposed of carefully to prevent aerosolizing the T. gondii oocysts (BIII). Cat feces (but not litter) can be flushed down the toilet, although this is not recommended because T. gondii oocysts are not consistently inactivated by sewage systems (DIII). Also, persons who clean cat litter, particularly HCT recipients, should wash their hands thoroughly with soap and water afterward to reduce their risk for acquiring toxoplasmosis (BIII).
HCT recipients and candidates with cats should keep their cats inside (BIII) and should not adopt or handle stray cats (DIII). Cats should be fed only canned or dried commercial food or well-cooked table food, not raw or undercooked meats, to eliminate the possibility of causing an illness that could be transmitted from the cat to the HCT recipient (BIII). Pet cats of HCT recipients do not need to be tested for toxoplasmosis (EII). Playground sandboxes should be kept covered when not in use to prevent cats from soiling them (BIII). HCT recipients and candidates undergoing conditioning therapy should avoid drinking raw goat's milk, to decrease the risk for acquiring toxoplasmosis (DIII). Toxoplasmosis may also be acquired after contact with cat feces encountered while gardening.
Water and Other Beverage Safety
HCT recipients should avoid walking, wading, swimming, or playing in recreational water (eg, ponds or lakes) that is likely to be contaminated with Cryptosporidium, E. coli O157:H7 749, 757, 758, 759, sewage, or animal or human waste (DII). HCT recipients should also avoid swallowing such water (eg, while swimming) 2, 757, 759, as well as any water taken directly from rivers and lakes (EIII) 2, 746.
Water and other beverages may pose a risk to immunocompromised HCT candidates and recipients because of bacterial, viral, and parasitic pathogens. HCT recipients should not use well water from private wells or from public wells in communities with limited populations (DIII) because tests for microbial contamination are performed too infrequently (eg, in certain locations, tests are performed ≤1 times/month) to detect sporadic bacterial contamination. However, drinking well water from municipal wells serving highly populated areas is regarded as safe from bacterial contamination because the water is tested ≥2 times/day for bacterial contamination. If HCT recipients consume tap water, they should routinely monitor mass media (eg, radio, television, or newspapers) in their area and immediately implement any boil-water advisories that might be issued for immunocompromised persons by state or local governments (BIII). A boil-water advisory means that all tap water should be boiled for >1 minute before it is consumed [760].
Although municipal tap water is generally safe, it may not be completely free of Cryptosporidium. Although limited data exist regarding the risks for and epidemiology of Cryptosporidium disease among HCT recipients [761], HCT recipients should avoid possible exposures to Cryptosporidium (DIII) because this pathogen has been reported to cause severe, chronic diarrhea, malnutrition, and death among other immunocompromised persons 746, 762, 763. To eliminate the risk for Cryptosporidium exposure from tap water, HCT recipients can boil tap water for ≥1 minute before consuming it (CIII) [746]. Alternatively, they can use certain types of water filters or a home distiller to reduce their risk for exposure to Cryptosporidium [746] and other waterborne pathogens (CIII). If a home water filter is used, it should be capable of removing particles ≥1 μm in diameter, or should filter by reverse osmosis (for a list of filters certified under National Sanitation Foundation [NSF] Standard 053 for cyst (ie, Cryptosporidium) removal, contact the NSF International consumer line or http://www.nsf.org). However, the majority of these filters are not capable of removing smaller microbes (eg, bacteria or viruses), and therefore, should be used only on properly treated municipal water. The majority of these devices are inappropriate for use on water from an unchlorinated private well to control viral or bacterial pathogens.
Bottled water can be consumed if it conforms to regional standards (eg, the FDA for the United States and Directive 80/777/EEC for the EU) and has been processed to remove Cryptosporidium by 1 of 3 processes: reverse osmosis, distillation, or 1-μm particulate absolute filtration. HCT recipients should contact the bottler directly to confirm that a specific bottled water has undergone 1 of these processes. The International Bottled Water Association can be contacted in the United States at (703) 683-5213 from 9 a.m. to 5 p.m. EST or anytime at their Internet site (http://www.bottledwater.org) to obtain contact information regarding water bottlers.
Patients can take other precautions in the absence of boil-water advisories to further reduce their risk for cryptosporidiosis. These extra precautions include avoiding fountain beverages and ice made from tap water at restaurants, bars, and theaters; fruit drinks made from frozen concentrate mixed with tap water; and iced tea or coffee made with tap water [746]. Drinks that are likely to be Cryptosporidium-safe for HCT recipients include nationally distributed brands of bottled or canned carbonated soft drinks; commercially packaged noncarbonated drinks that contain fruit juice; fruit juices that do not require refrigeration until after opening (eg, those that are stored unrefrigerated on grocery shelves); canned or bottled soda, seltzer, or fruit drinks; steaming hot (>175° F [80 °C]) tea or coffee; juices labeled as pasteurized; and nationally distributed brands of frozen fruit juice concentrate that are reconstituted with water from a safe source 705, 746. HCT recipients should not drink nonpasteurized milk or fruit or vegetable juices (eg, apple cider or orange juice), to avoid infection with Brucella species, E. coli O157:H7, Salmonella species, Cryptosporidium, and other pathogens (DII) 763, 764, 765, 766.
Food Safety
Recommendations on food safety are based largely on observations in the general population. Concern arising from the detection of potential pathogens in food has not been supported by documented evidence of such organisms as the source of opportunistic infections in immunocompromised persons. The potential benefit of food safety recommendations directed specifically toward HCT recipients must be weighed against the uncertain value of such recommendations [767] and their potential to adversely affect patients` nutritional intake and/or quality of life.
HCT candidates and persons who will prepare food for them after HCT should review general food safety practices (AIII) [768] and should be educated regarding additional food safety practices appropriate for HCT recipients. This review and education should be done before the conditioning regimen (ie, chemotherapy and radiation) begins (BIII). Adherence to these guidelines will decrease the risk for food-borne disease among HCT recipients.
Food safety practices appropriate for all persons
Raw poultry, meats, fish, and seafood should be handled on separate surfaces (eg, cutting board or counter top) from other food items. Persons preparing food should always use separate cutting boards or wash the board(s) with warm water and soap between cutting different food items (AIII). To prevent foodborne illnesses caused by Campylobacter jejuni and Salmonella enteritidis, which can cause severe and invasive infections among immunocompromised persons [769], uncooked meats should not come in contact with other foods (DIII). After preparing raw poultry, meats, fish, and seafood and before preparing other foods, food handlers should wash their hands thoroughly in warm, soapy water. Any cutting boards, counters, knives, and other utensils used should also be washed thoroughly in warm, soapy water (AIII). Food preparers should keep shelves, countertops, refrigerators, freezers, utensils, sponges, towels, and other kitchen items clean (AIII). All fresh produce should be washed thoroughly under running water before serving (AIII) [768]. Persons preparing food should follow published recommendations regarding safe food thawing (BIII) [770].
Persons cooking food for HCT recipients should follow established guidelines for monitoring internal cooking temperatures for meats, which is the only method for determining if meat has been adequately cooked (AII). Different kinds of meat should be cooked to varying internal temperatures, all >150°F (66 °C) (AII). Specifically, food oversight agencies such as the U.S. Department of Agriculture recommend that poultry be cooked to an internal temperature of 180°F (82 °C). Other meats and egg-containing casseroles and soufflés should be cooked to an internal temperature of >160°F (71 °C). Cold foods should be stored at <40°F (4 °C), and hot foods should be kept at >140°F (60 °C) (BIII). Food preparers should wash their hands before and after handling leftovers (AIII); use clean utensils and food-preparation surfaces (AIII); divide leftovers into small units and store in shallow containers for quick cooling (AII); refrigerate leftovers within 2 hours of cooking (AII) or discard leftovers that were kept at room temperature for >2 hours (AIII); reheat leftovers or heat partially cooked foods to >165°F (74 °C) throughout before serving (AII); bring leftover soups, sauces, and gravies to a rolling boil before serving (AIII); and follow published guidelines for cold storage of food (AII). Leftover foods placed in the refrigerator should be dated and discarded after 72 hours.
Additional food safety practices appropriate for HCT recipients
HCT recipients' diets should be restricted prior to engraftment to decrease the risk for exposure to food-borne infections from bacteria, yeasts, molds, viruses, and parasites. A low-microbial diet is recommended for HCT recipients prior to engraftment 771, 772, although evidence for its efficacy to prevent infection is lacking (CIII) [773].
Once HCT recipients have engrafted, they should follow a diet that balances the risk for acquiring food-borne illnesses with the importance of proper nutritional supplementation, as recommended below and outlined in Table 5. This diet should be continued for 3 months after autologous HCT, and allogeneic HCT recipients should remain on this diet until all immunosuppressive drugs are discontinued and the patient has reached the milestone of receiving live virus vaccines. However, the HCT physician should have final responsibility for determining when the diet can be discontinued safely.
HCT recipients should not eat raw or undercooked meat, including beef, poultry, pork, lamb, venison or other wild game, or combination dishes containing raw or undercooked meats or sweetbreads from these animals (eg, sausages or casseroles) (EII). HCT recipients and candidates should only consume meat that is well done when they or their caretakers do not have direct control over food preparation (eg, when eating in a restaurant) (AI). Hot dogs and deli-style ready-to-eat meats should be avoided unless heated until steaming (AII).
HCT recipients should not consume raw or undercooked eggs or foods that might contain them (eg, certain preparations of hollandaise sauce, Caesar and other salad dressings, homemade mayonnaise, and homemade eggnog) because of the risk for infection with Salmonella enteritidis (EII) [774]. HCT recipients should not consume raw or undercooked seafood (eg, oysters or clams), to prevent exposure to Vibrio species, viral gastroenteritis, and Cryptosporidium parvum (EII) [769].
Fruits and vegetables provide essential nutritional elements to HCT patients, but certain precautions should be taken to prevent acquisition of infection. In general, it may be helpful to remind patients of the adage “If you can't peel or wash it, don't eat it.” Most infections from agricultural products are acquired either through contamination of the item while in the field or subsequent to harvesting during processing. Washing of fruits and vegetables in tap water is advisable even for those with skin or rinds, organic foods, and prepackaged items labeled as “prewashed” (BIII). Such washing may prevent many contamination-related infections, but not all. HCT recipients should avoid fruits and vegetables that may confer higher risk of infection, including raw vegetable sprouts (E. coli and Salmonella), fresh salsa, and berries (EII).
Other food items that have been associated with food-borne outbreaks in the past and, therefore, which HCT patients should avoid, include unroasted raw nuts or nuts in the shell, miso products, raw grain products, nonpasteurized milk products (milk, cheese, yogurt), cheeses containing uncooked vegetables, cheeses with molds (ie, blue, Stilton, Roquefort, and gorgonzola), soft cheeses (eg, brie and feta), smoked or pickled seafood, raw honey, and tempe products (EII).
To date, no evidence exists that there is a greater risk for acquiring infection from eating at a fast food restaurant than at a conventional sit-down restaurant. Several steps can be taken to ensure the safety of food consumed at such establishments (AIII): (1) ask that food be freshly prepared (ie, avoid food that has been sitting under heat lamps); (2) ask if fruit juices are pasteurized; (3) avoid any raw fruits and vegetables when dining out; (4) ask for single-serving condiment packages (avoid use of public self-serve condiment containers); (5) avoid salad bars, delicatessens, buffets, smorgasbords, etc.; (6) set utensils on a napkin or clean tablecloth or placemat (rather than directly on the table); and (7) if planning to take the leftovers home, transfer the food directly to a box at the table. Similarly, although consumption of prepared food purchased from street vendors in industrialized countries has not been associated with infection, food preparation under stringent sanitary conditions cannot be guaranteed and therefore such food should be avoided (DIII); the same is true of food brought by others to gatherings such as potlucks or picnics.
The consumption of foods with “active” or “live” yeast cultures (“probiotics”) has been promoted as an effective means of preventing infections. Studies to date have not been definitive, but there is some support in the literature for the possibility that consumption of probiotics reduces antibiotic-associated diarrhea [775] or genitourinary infections [776]. The use of probiotics has not been examined in the setting of HCT. Disseminated infection from probiotic administration has been reported in HCT patients, however [777], and HCT recipients should avoid the use of probiotics (DIII). HCT candidates undergoing conditioning therapy and HCT recipients with neutropenia (ie, ANC <1000/mL3) or during periods of substantial immunocompromise should also avoid exposure to naturopathic medicines that might contain molds (DIII) [778]. HCT recipients wishing to take naturopathic medications are advised to use them only as prescribed by a licensed naturopathic physician working in consultation with the recipient's transplant and infectious disease physicians (CIII).
Travel Safety
For HCT recipients, particularly allogeneic recipients who are chronically immunosuppressed, travel to developing countries can pose substantial risks for exposure to opportunistic pathogens. HCT recipients should not plan travel to developing countries without consulting their physicians (EIII), and should not undertake such travel until the period of severe immunosuppression has resolved. Generally, allogeneic recipients should not plan travel to developing countries for 6 to 12 months after HCT, and should refrain from travel in the setting of immunosuppressive treatment for GVHD. Autologous recipients can travel to developing countries 3 to 6 months after HCT if their HCT providers agree that this is safe and appropriate.
HCT recipients should be educated about regarding strategies to minimize the risk for acquiring infections while traveling. Selected strategies include the following:
HCT recipients should obtain updated, detailed health information for international travelers from health organizations (AIII) [779];
-
•
“Adventure” travel (eg, kayaking, participating in triathlons), has been associated with epidemic infections (such as leptospirosis), and should be avoided (DIII) 780, 781, 782;
-
•
Air travel is generally safe, although rarely may be associated with acquisition of respiratory illnesses (eg, influenza, tuberculosis, measles) [783]. HCT recipients should use techniques such as social distancing and hand hygiene to avoid infections from other passengers in close proximity who exhibit respiratory symptoms (BIII)
-
•
Cruise ships are generally considered to be safe, but may be associated with acquisition of GI (eg, norovirus) and Legionella infections. HCT recipients should be fastidious about washing hands with soap and water frequently while on cruises and should promptly report any symptoms that may arise to their transplant team (AII).
-
•
Food safety is of paramount importance while traveling to developing countries. HCT recipients should avoid consuming (DIII) raw fruits and vegetables; tap water or any potentially untreated or contaminated water; ice made from tap water or any potentially contaminated water; nonpasteurized milk or any nonpasteurized dairy products; fresh fruit juices, food, and drinks from street vendors; and raw or undercooked eggs. Steaming hot foods, fruits peeled by oneself, bottled and canned processed drinks, and hot coffee or tea are probably safe [784]. Travelers should plan for treating their drinking water while in developing countries. If bottled water is not available, boiling is the best method of making water safe. However, if boiling of water is not feasible, the traveler should carry supplies for disinfecting water (eg, commercially available iodine disinfection tablets or a portable water filter) (BIII) 784, 785. As referenced in the food safety section, water disinfection does not mitigate the risk for cryptosporidiosis and viral infections.
Antimicrobial prophylaxis for traveler's diarrhea is not recommended routinely for HCT recipients traveling to developing countries (DIII). However, HCT physicians who wish to provide prophylaxis to HCT recipients who are traveling can prescribe a fluoroquinolone (eg, ciprofloxacin) (Appendix 1) (CIII). Some clinicians recommend using bismuth subsalicylate to prevent traveler's diarrhea in adults [784]. However, no data were found regarding safety and efficacy among HCT recipients, and salicylates are not recommended for use among persons younger than 18 years of age, because salicylates are associated with Reye's syndrome [784].
HCT recipients' immunization status should be assessed and their vaccinations updated as needed before travel (Table 6) [784]. Influenza chemoprophylaxis with oseltamivir or zanamivir (depending on the circulating strain and susceptibilities) can be used for immunocompromised HCT recipients who could be exposed to influenza while traveling (CIII), but travelers should be vigilant about the risk of pandemic influenza or severe acute respiratory syndrome (SARS).
Vaccination of HCT Recipients
P. Ljungman, C. Cordonnier, H. Einsele, J. Englund, C.M. Machado, J. Storek, T. Small
Vaccination recommendations have previously been developed and published separately by the EBMT and the CDC, the IDSA, and the ASBMT 1, 786, 787. The purpose of the vaccination schedule in these joint updated guidelines is to provide guidance for HCT centers around the globe.
Antibody titers to vaccine-preventable diseases (eg, tetanus, polio, measles, mumps, rubella) decline during the 1 to 10 years after allogeneic or autologous hematopoietic cell transplantation (HCT) 788, 789, 790, 791, 792 if the recipient is not revaccinated. The clinical relevance of decreased antibodies to vaccine-preventable diseases among HCT recipients is not immediately apparent because a limited number of cases of vaccine-preventable diseases are reported among HCT recipients. However, vaccine-preventable diseases still pose risks to the population. Additionally, evidence exists that certain vaccine-preventable diseases such as pneumococcal infection, Haemophilus influenzae type b (Hib) infection, measles, varicella, and influenza can pose increased risk for HCT recipients 193, 309, 793, 794, 795, 796, 797, 798. Therefore, HCT recipients should be routinely revaccinated after HCT so that they can experience immunity to the same vaccine-preventable diseases as others (Table 6). Although several studies have evaluated safety and immunogenicity (eg, serologic studies of antibody titers after vaccination) of different vaccines in allogeneic HCT recipients, limited and only indirect data were found regarding vaccine efficacy among HCT recipients (ie, whether vaccinated HCT recipients have decreased attack rates of infections compared with unvaccinated HCT recipients) [798].
For a vaccine to mount a response thought to be clinically relevant (eg, a 4-fold rise in specific antibody levels or a rise to a level considered protective), adaptive (T and B cell) immunity posttransplant must have been at least partially reconstituted. B cell counts, which are typically zero or near-zero in the first 1 to 3 months after HCT, return to normal by 3 to 12 months posttransplant 799, 800. In patients treated with rituximab posttransplant, B cell recovery is generally delayed for 6 months following the last dose [801]. Regardless of the time to recovery, the newly generated B cells often demonstrate impaired antigen-specific responses because of a limited capability of naïve B cells to undergo somatic mutation and isotype switching during the first year after transplant. T cell counts are low in the first 1 to 3 months posttransplant (CD4+ counts are typically <200/μL). Thereafter, the recovery of T cells, particularly CD4+ T cells, is influenced by patient age at HCT, substantial T cell depletion (TCD) of the graft, and the presence or absence of cGVHD. Most patients <18 years at HCT without cGVHD will recover CD4+ cell counts of >200/μL by 6 to 9 months after transplant, whereas adults, particularly those with cGVHD, may require >2 years.
Most of the circulating T cells in the first year after transplantation, particularly in adults, are memory/effector T cells, likely derived from the T cells infused with the graft and capable of responding to antigens encountered by the donor prior to the transplant. Naïve T cells capable of responding to new antigens start to be generated only at 6 to 12 months posttransplant, earlier in young children and later in older adults. Because HCT recipients have varying immune system recovery after HCT, it has been proposed that different vaccination schedules be recommended for recipients of different types of HCT, with the rationale that, for example, autologous HCT patients do not lose immunity as frequently or rapidly as patients after allogeneic HCT. The existing evidence suggests, however, that loss of immunity is also common after autologous HCT (particularly in patients who have received multiple courses of chemotherapy before HCT), and that responses to vaccination are similar to those that occur after allogeneic HCT 194, 790, 791, 802, 803, 804, 805. It should also be recognized that limited information regarding vaccine immunogenicity exists for patients transplanted with umbilical cord blood (UCB) or haploidentical grafts, or after reduced-intensity conditioning (RIC). For the sake of simplicity, therefore, the committee has chosen to recommend the same vaccination schedule for all HCT recipients until additional data are published.
T cell response to vaccines for pathogens encountered pretransplant (eg, varicella zoster virus [VZV]) can be observed as soon as 1 to 6 months posttransplant. Antibody response to vaccines for pathogens encountered pretransplant (eg, tetanus toxoid) can be observed at 6 to 12 months after HCT. T cell or antibody response to vaccines for pathogens not encountered pretransplant (eg, hepatitis B virus [HBV] in most European and American adults) can usually be observed later (1 year or more posttransplant). HCT recipients are similar to young children in that they respond poorly to pure polysaccharide antigens such as those included in the 23-valent polysaccharide pneumococcal vaccine. Pure polysaccharide antigens elicit antibody responses later after HCT than protein antigens (eg, diphtheria toxoid) or polysaccharide-protein conjugates (eg, Hib capsular polysaccharide conjugated to a carrier protein). Graft-versus-host disease (GVHD) and/or its treatment hamper T cell and antibody responses to vaccines. Because studies have shown that patients with chronic GVHD (cGVHD) can mount responses to vaccines and clearly need protection against pneumococci, these guidelines do not recommend postponing vaccination in patients with GVHD with the exception of withholding live vaccines. However, when vaccinating patients with active GVHD, it may be prudent to measure specific antibody levels before and after vaccination, to determine their level of protection and need for booster immunizations.
The committee has split the recommendations into different categories. In Table 6, the vaccines are listed for which evidence exists regarding safety and immunogenicity and that are generally recommended to be used in HCT recipients. Since the previous version of the vaccination guidelines was published, new vaccines have been introduced. In addition, clinicians at HCT centers get questions from patients, family members, and HCWs regarding vaccinations in special situations, such as after disease exposure or before travel to areas endemic for infections not previously considered in these recommendations. Therefore, comments are made regarding these vaccines and situations (Table 7), although very limited or no data exist. Finally, there are situations when vaccination of family members, household contacts, and HCWs is recommended to minimize exposure of vaccine-preventable diseases among HCT recipients (Table 8 ).
Table 8.
Vaccinations for Family, Close Contacts, and Healthcare Workers (HCWs) of HCT Recipients
| Vaccine | Recommendations for Use | Rating |
|---|---|---|
| Hepatitis A [829] |
|
BIII |
|
||
| AII | ||
| AI | ||
| Polio‡ | Not routinely recommended for adults but inactivated§ polio vaccine should be administered when polio vaccination is indicated | AII |
| Rotavirus |
|
CIII |
| Measles-mumps-rubella (MMR) (live) |
|
AIII |
| Pertussis | Vaccination with DTaP is recommended for children < 7 years and with Tdap for adolescents and adults. | BIII |
|
|
AIII |
HCT indicates hematopoietic cell transplant.
Children aged 6 months to <9 years receiving influenza vaccination for the first time require 2 doses and those who only receive 1 dose in the first year should receive 2 doses the following year 304, 307.
If HCWs, family members, or other close contacts of HCT recipients receive influenza vaccination during an influenza outbreak, they should also receive chemoprophylaxis, based on strain susceptibility (BI), while the vaccinee develops an immunologic response to the vaccine. However, if a nosocomial outbreak occurs with an influenza strain that is not contained in the available influenza vaccine, HCWs, family members, and other close contacts of HCT recipients and candidates should be administered influenza chemoprophylaxis based on strain susceptibility, until the end of the outbreak (BIII) 307, 830, 831.
Vaccine-strain polio virus in oral polio vaccine can be transmitted person to person; therefore, oral polio vaccine administration is contraindicated among household contacts of immunocompromised persons. If oral polio vaccine is inadvertently administered to a household contact of an HCT recipient, ACIP's and the American Academy of Pediatrics' recommendations should be followed to minimize close contact with the immunocompromised person for 4-6 weeks after vaccination 304, 832, 833.
Although vaccine-associated paralytic poliomyelitis has not been reported among HCT recipients after exposure to household contacts inadvertently vaccinated with oral polio vaccine, inactivated polio vaccine should be used among family members, close contacts, and HCWs to avoid person-to-person transmission of vaccine-strain polio virus [832].
HCWs, family members, close contacts, and visitors who do not have a documented history of varicella-zoster infection or who are seronegative should receive varicella vaccine before being allowed to visit or have direct contact with an HCT recipient (AIII). Ideally, varicella-zoster- susceptible HCWs, family members, household contacts, and potential visitors of immunocompromised HCT recipients should be vaccinated as soon as the decision to perform an HCT is made. The vaccination dose or doses should be completed >4 weeks before the conditioning regimen begins or >6 weeks (42 days) before contact with the HCT recipient is planned (BIII). If a varicella vaccinee develops a postvaccination rash within 42 days of vaccination, the vaccinee should avoid contact with HCT recipients until all rash lesions are crusted or the rash has resolved 282, 304
Children 12 months to 12 years should receive two doses 3 months apart; adolescents ≥13 years and adults should receive 2 doses 4 weeks apart.
Donor vaccination
Vaccination of the donor has been shown to improve the posttransplant immunity of the patient in the case of tetanus toxoid, 7-valent PCV, and Hib-conjugate vaccines. No recommendations are made regarding donor vaccination, because of practical and ethical difficulties surrounding this issue.
Serological testing
HCT patients are immunosuppressed to varying degrees, and it is therefore prudent to test immunity to some infections before or after vaccination.
Testing before vaccination
Testing for antibodies to measles is recommended in adults, with vaccination performed only if the patient is seronegative (CIII) (Table 6). If vaccination against varicella is contemplated, testing of immunity should be performed and vaccination given to seronegative patients only (CIII) (Table 6).
Testing after vaccination
Testing after vaccination can be indicated either to assess the response to vaccination and the need for additional doses or to check durability of response during long-term follow-up. Testing to assess the response to vaccination against pneumococcal disease is recommended at 1 month or later after the third or fourth dose of pneumococcal vaccine (BIII) (Table 6). There are different methods for assessment of pneumococcal antibody levels, each having advantages and disadvantages; thus, no specific method can be recommended. Likewise, as a widely accepted definition of adequate response to pneumococcal vaccine is lacking, guidelines for revaccination of nonresponders are not given. Testing to assess the response to HBV vaccination is also recommended. Testing should be done 1 month or later after the third vaccine dose (BIII). A second 3-dose vaccination schedule is recommended in nonresponders (CIII). The interval between the first and second series has to be determined individually because nonresponsiveness to HBV vaccine can have different causes (eg, cGVHD, in which case it may be prudent to revaccinate only after GVHD has abated).
Regular testing of long-term HCT survivors for maintenance of antibody levels is recommended (BIII). Testing should be conducted approximately every 4 to 5 years to assess for immunity to HBV, measles, tetanus, diphtheria, and polio (BIII). Testing for immunity to pneumococcus might reasonably be repeated every 2 years for the first 4 years (BIII). The need for revaccination has to be assessed on an individual basis.
Comments Regarding Specific Vaccines
Pneumococcal vaccine
There are 2 types of pneumococcal vaccine: a conjugate vaccine (PCV) and a polysaccharide vaccine (PPSV23). As with most conjugate vaccines compared with polysaccharide vaccines, PCV is more immunogenic than PPSV23. However, the spectrum of protection is narrower, as PCV7 covers only 7 strains of pneumococci whereas PPSV23 covers 23 strains. When given during the first year after transplantation, PPSV23 elicits inadequate responses. Four prospective trials demonstrate better responses with PCV in HCT recipients 195, 797, 806, 807; thus, PCV is the preferred vaccine. It is likely beneficial to use PPSV23 for the fourth dose (after 3 doses of PCV) (BII), to broaden the immune response [808]. A fourth dose of PCV might increase the response rate in patients with cGVHD, who are less likely to respond to PPSV23 (CIII).
If a microbiologically documented pneumococcal infection occurs after pneumococcal vaccination, documenting the serotype of the strain is recommended (BIII), to know whether the serotype is among those included in the vaccine, which can be indicative of a nonresponse to the vaccine(s). Such patients should receive additional doses of pneumococcal vaccine, with the choice of vaccine (ie, PCV or PPSV23) depending on the documented strain (BIII).
The time posttransplant to initiate routine vaccination with PCV is controversial. One trial showed similar antibody responses with vaccination started at 3 months (early) and 9 months (late) posttransplant [808]. Thus, early vaccination may be preferred as it may protect against not only late but also early pneumococcal disease. It should be noted that early vaccination may not prime for a PPSV23 boost as efficiently as the late vaccination. Also, early vaccination may result in a shorter lasting antibody response. Therefore, if vaccination is started early, it may be particularly important to determine pneumococcal antibody levels and, if these are “low,” revaccinate (BIII).
Diphtheria-tetanus vaccine
There are 2 general types of diphtheria and tetanus vaccines: those containing “full”-dose diphtheria toxoid in combination with tetanus toxoid (DT) and those containing reduced dose diphtheria toxoid (such as Td; the lower case “d” indicates reduced diphtheria toxoid, while the tetanus content is essentially the same in TD and Td). Use of reduced diphtheria toxoid vaccines following transplant can be associated with lack of response. Therefore, posttransplant patients should be viewed as “never vaccinated” and full toxoid vaccines should be used if possible. The DT vaccine is not approved in individuals >7 years of age in the United States because of side effects, although experience with adult HCT recipients receiving DT vaccine indicates a lower risk for side effects than in previously vaccinated healthy adults. Adult transplant recipients might have an adequate response to the diphtheria portion of Td. However, whether the response is equal to TD has not been studied. Checking diphtheria antibody level after vaccination with Td might therefore be warranted in situations where an increased risk for diphtheria might be envisaged.
Pertussis vaccine
For the general population, the Advisory Committee on Immunization Practices and the CDC have recommended use of acellular (rather than whole cell) pertussis vaccine in pediatric vaccination regimens since 1997. Because of the steady increase in pertussis over the last decade and the licensing of 2 vaccines containing tetanus, reduced dose diphtheria, and reduced dose pertussis (Tdap), the CDC now recommends that adolescents (10-18 years of age) and adults (19-64 years of age) receive a single dose of Tdap to replace their routine adult tetanus and diphtheria toxoids (Td) booster. For adolescents, the preferred age range is 11 to 12 years. For adults (11-64 years), a single Tdap is recommended to replace their routine Td booster if given ≥10 years earlier. The recommended interval of 10 years between Td and Tdap was because of concerns of local site reactions. However, for adults who have contact with infants <12 months, HCWs, and those in community outbreaks of pertussis, intervals as short as 2 years after the last Td is now recommended. The aim is to decrease the reservoir of pertussis that affects infants and immunocompromised individuals regardless of age.
HCT recipients may be particularly vulnerable to complications from pertussis because of pulmonary damage from chemotherapy and/or TBI, even in the absence of cGVHD. Because posttransplant patients should be viewed as “never vaccinated” and receive full doses of toxoids, their “DT” vaccine should include full-dose (not reduced dose) acellular pertussis toxoid (DTaP) if available. In the United States, DTaP is available for children <7 years. The adult pertussis vaccine used frequently by community physicians, Tdap, contains tetanus toxoid, reduced-dose diphtheria toxoid, and reduced-dose acellular pertussis toxoid. Tdap is different from the DTaP vaccine currently given to babies and young children, as it contains lesser quantities of diphtheria and pertussis proteins and is much less likely than DTaP to cause side effects such as pain, redness, and tenderness.
Preliminary data in autologous and allogeneic transplant recipients demonstrate that the response to reduced dose pertussis in Tdap is poor, regardless of when the vaccine is given 809, 810. Use of Tdap as the initial tetanus-containing vaccine in adult autologous HCT recipients was associated with lack of response to the tetanus as well as the pertussis portion, suggesting that this vaccine should be used as a booster vaccine rather than as part of the primary series [810]. Vaccines with higher tetanus and pertussis content may be more immunogenic in HCT recipients and thus should be considered for the initial vaccination.
Influenza vaccine
Life-long seasonal influenza vaccination with inactivated influenza vaccine is recommended for all HCT candidates and recipients (AII). When vaccination should be initiated after HCT depends on the epidemiologic situation, but it is likely that the effectiveness is better later after HCT. Whether influenza vaccination is beneficial during the first months following nonmyeloablative conditioning is unknown. If the vaccine is given earlier than 6 months after HCT regardless of conditioning intensity, a second dose could be contemplated (CIII). The live intranasal influenza vaccine should not be used because an inactivated alternative exists (EIII).
During community outbreaks, HCT recipients who have not yet received a current influenza vaccination should be vaccinated against influenza immediately if they are more than 4 months after HCT (BIII). A second dose can also be given, especially if the first dose is given <6 months after HCT (CIII). For pediatric HCT recipients and candidates >6 months old, annual seasonal influenza vaccination is recommended (BIII). Children <9 years old who are receiving influenza vaccination for the first time require 2 doses administered ≈1 month apart (AI).
Varicella vaccines
There are 2 main varicella vaccines, directed against preventing chickenpox (Varivax) or shingles (Zostavax). The difference between these vaccines is the number of plaque forming units of attenuated virus. The chickenpox vaccine has lower viral titers and can be used for all HCT recipients who have met the criteria for live virus vaccination. The new shingles vaccine should not be used because of the much higher viral titers.
HBV vaccine
Vaccination is recommended for HBsAg or HBcAb-positive patients, because vaccination can reduce the risk for reverse seroconversion (BII). For HBsAg or HBcAb-negative HCT patients, the recommendations for the general population in their country of residence should be followed.
Meningococcal vaccine
Both polysaccharide-based and conjugate vaccines exist. It is reasonable to assume that, as is true of vaccines against pneumococci and Hib, conjugated meningococcal vaccine will give more stable immune responses than polysaccharide-based vaccines, although no comparative study of the 2 vaccine types has been performed 811, 812.
Footnotes
Recommendations of the Center for International Blood and Marrow Transplant Research (CIBMTR®), the National Marrow Donor Program (NMDP), the European Blood and Marrow Transplant Group (EBMT), the American Society of Blood and Marrow Transplantation (ASBMT), the Canadian Blood and Marrow Transplant Group (CBMTG), the Infectious Diseases Society of America (IDSA), the Society for Healthcare Epidemiology of America (SHEA), the Association of Medical Microbiology and Infectious Diseases Canada (AMMI), and the Centers for Disease Control and Prevention (CDC)
Appendix 1: Dosing Charts for Prevention or Preemptive Treatment of Specific Infections in HCT Patients
I. Bacterial Infections
Pathogen: Bacterial infections during the first 100 days after HCT
| Indication | First Choice | Alternatives |
|---|---|---|
| Prevention of bacterial infections for adult HCT patients with anticipated neutropenic periods of 7 days or more |
|
Azithromycin 250 mg once daily (CIII) |
| Prevention of bacterial infections among adult or adolescent HCT recipients with severe hypogammaglobulinemia (ie, serum IgG level <400 mg/dL) | IVIG, 500 mg/kg/week (CIII)∗, † | None |
| Prevention of bacterial infections among allogeneic pediatric HCT recipients with severe hypogammaglobulinemia (ie, serum IgG level <400 mg/dL) | IVIG, 400 mg/kg/month (CIII)∗, † | None |
IVIG indicates intravenous immunoglobulin; HCT, hematopoietic cell transplantation.
Persons with IgA deficiency should not receive standard immunoglobulin products (DIII). Researchers have reported that use of IgA-depleted immunoglobulin preparations can be used with caution among these persons 834, 835, 836.
Researchers propose checking serum IgG levels every 2 weeks among patients receiving IVIG replacement therapy.
Pathogen: Bacterial infections beyond 100 days post-HCT
| Indication | First Choice | Alternatives |
|---|---|---|
| Prevention of late bacterial infections with antibiotic prophylaxis | Prolonged antibiotic prophylaxis is recommended only for preventing infection with S. pneumoniae among allogeneic recipients with cGVHD (see below). | |
| Prevention of late bacterial infections with vaccinations | Immunizations (Table 6) are recommended for preventing infection with S. pneumoniae and H. influenzae type b | |
| Prevention of bacterial infections among HCT recipients with severe hypogammaglobulinemia (ie, serum IgG level <400 mg/dL) | IVIG, 500 g/kg every 3-4 weeks (CIII) | None |
IVIG indicates intravenous immunoglobulin; HCT, hematopoietic cell transplantation; cGVHD, chronic graft-versus-host disease.
Notes: See the note in the previous table for IVIG administration for IgA-deficient patients.
Pathogen: Streptococcus pneumoniae
| Indication | First Choice | Alternatives |
|---|---|---|
| Immunization against pneumococcal disease for adult or pediatric HCT recipients |
|
|
|
|
Macrolides or fluoroquinolones∗ or second-generation cephalosporins (CIII) |
HCT indicates hematopoietic cell transplantation; GVHD, graft-versus-host disease.
Fluoroquinolones generally should not be used in children. However, if a fluoroquinolone is considered, suggested dosing is as follows: levofloxacin 10 mg/kg/dose daily or ciprofloxacin 10-15 mg/kg/dose twice daily.
Pathogen: Haemophilus influenzae type b (Hib)
| Indication | First Choice | Alternatives |
|---|---|---|
| Immunization against Hib for adult or pediatric HCT recipients |
|
None |
|
None |
HCT indicates hematopoietic cell transplantation.
There is potential for substantial drug-drug interactions between rifampin and immunosuppressive agents and other drugs
Pathogen: Methicillin-resistant Staphylococcus aureus (MRSA)
| Indication | First Choice | Alternatives |
|---|---|---|
| Elimination of methicillin- resistant Staphylococcus aureus (MRSA) carrier state among HCT patients with recurrent MRSA infection (CIII) |
|
Systemic antimicrobials (eg, rifampin, doxycycline) |
HCT indicates hematopoietic cell transplantation.
Note: No regimen for MRSA decolonization has proved consistently effective, but a decrease in MRSA burden may be accomplished.
Pathogen: Bordetella pertussis
| Indication | First Choice | Alternatives |
|---|---|---|
| Immunization against pertussis for adult or pediatric HCT recipients |
|
|
| Postexposure prophylaxis for adult or pediatric HCT recipients, regardless of vaccination status |
|
II. Viral Infections
Pathogen: Cytomegalovirus
| Indication | First Choice | Alternatives |
|---|---|---|
|
|
|
|
|
|
|
|
|
GVHD indicates graft-versus-host disease; HCT, hematopoietic cell transplantation; PCR, polymerase chain reaction; CMV, cytomegalovirus; TBI, total body irradiation.
Notes: Patients who do not tolerate standard doses of ganciclovir should be treated with foscarnet.
Ganciclovir and foscarnet doses should be reduced in patients with renal impairment. Prehydration is required for foscarnet and cidofovir administration.
Pathogen: Epstein-Barr virus
| Indication | First Choice | Alternatives |
|---|---|---|
| Prevention of Epstein-Barr virus-related posttransplant lymphoproliferative disease in high-risk patients (BII) |
|
None |
Pathogen: Herpes simplex virus
| Indication | First Choice | Alternatives |
|---|---|---|
|
|
|
| Prevention of late reactivation among seropositive HCT recipients |
|
|
HCT indicates hematopoietic cell transplantation; HSV, herpes simplex virus.
Note: For patients requiring prophylaxis for cytomegalovirus and herpes simplex virus after engraftment, ganciclovir alone provides effective prophylaxis for both pathogens.
For long-term prophylaxis, the higher dose of acyclovir is recommended for maximal viral suppression and minimization of resistance.
Pathogen: Varicella-zoster virus
| Indication | First Choice | Alternatives |
|---|---|---|
|
|
|
|
|
|
GVHD indicates graft-versus-host disease; HCT, hematopoietic cell transplantation; VZV, varicella zoster virus.
Lower doses of acyclovir, as low as 200 mg twice daily (BII) (at this dose there is no prevention of HSV reactivation disease).
Pathogen: Adenovirus
| Indication | First Choice | Alternatives |
|---|---|---|
| Preemptive therapy among high-risk seropositive HCT recipients (CII) |
|
|
HCT indicates hematopoietic cell transplant.
∗Data on optimal dosing of oral ribavirin for adenovirus infection are very limited and based on maximum tolerated doses 837, 838; dose reduction may be required when hemolytic anemia occurs.
The 1 mg/kg 3 times a week dose may cause less renal toxicity, but it is insufficient to treat concomitant CMV infection; if concomitant CMV infection is present, the 5 mg/kg/week is recommended.
Dose may be rounded to nearest dose that can be divided by 200 (capsule size); an oral suspension is available.
Pathogen: Influenza
| Indication | First Choice | Alternatives |
|---|---|---|
| Prevention of influenza A or B |
|
None |
|
|
|
HCT indicates hematopoietic cell transplantation; cGVHD, chronic graft-versus-host disease.
Notes: Rimantadine dose should be reduced for patients with impaired renal function or for severely impaired hepatic function.
Amantadine dose should be reduced for renal impairment.
Neither rimantadine nor amantadine are Federal Drug Administration-approved for children <1 year old.
Children <9 years old receiving influenza vaccination for the first time require 2 doses of vaccine spaced ≥1 month apart.
Pathogen: Respiratory syncytial virus
| Indication | First Choice | Alternatives |
|---|---|---|
|
|
None |
|
|
IVIG indicates intravenous immunoglobulin; HCT, hematopoietic cell transplant.
III. Fungal Infections
Patients at standard risk for fungal infections
| Indication | First Choice | Alternatives |
|---|---|---|
|
|
|
HCT indicates hematopoietic cell transplantation.
Patients at high risk for infections from molds or fluconazole-resistant Candida species
| Indication | First Choice | Alternatives |
|---|---|---|
| Prophylaxis in the setting of prolonged neutropenia | Micafungin, 50 mg i.v. once daily (BI) | |
| Prophylaxis in the setting of GVHD | Posaconazole, 200 mg orally 3 times daily (BI) |
|
GVHD indicates graft-versus-host disease.
IV. Regionally Limited or Rare Infections
Pathogen: Pneumocystis jiroveci
| Indication | First Choice | Alternatives |
|---|---|---|
|
|
|
HCT indicates hematopoietic cell transplant; cGVHD, chronic graft-versus-host disease.
Notes: Patients who are receiving sulfadiazine-pyrimethamine for toxoplasmosis therapy are protected against Pneumocystis jiroveci and do not need additional prophylaxis.Trimethoprim-sulfamethoxazole is not recommended for patients <2 months old, because of risk for kernicterus.
Pathogen: Toxoplasma gondii
| Indication | First Choice | Alternatives |
|---|---|---|
|
|
|
HCT indicates hematopoietic cell transplantation.
Note: Among allogeneic HCT recipients, clinical toxoplasmosis has occurred despite the use of trimethoprim-sulfamethoxazole for Pneumocystis jiroveci prophylaxis [100].
Trimethoprim-sulfamethoxazole is not recommended for patients <2 months old, because of risk for kernicterus.
Pathogen: Strongyloides species
| Indication | First choice | Alternatives |
|---|---|---|
|
|
HCT indicates hematopoietic cell transplant.
Notes: Among immunocompromised patients, multiple courses at 2-week intervals might be required; however, cure might not be achievable.
Safety and efficacy of ivermectin has not been established during pregnancy.
Albendazole and thiabendazole are contraindicated during pregnancy.
Pathogen: Traveler's diarrhea
| Indication | First Choice | Alternatives |
|---|---|---|
|
|
|
HCT indciates hematopoietic cell transplantation.
Notes: Use of aspirin-containing products, including bismuth subsalicylate, is contraindicated in persons <18 years old unless prescribed by a physician, because these products have been associated with Reye's syndrome [840].
Trimethoprim-sulfamethoxazole is not recommended for patients <2 months old, because of risk for kernicterus.
Resistance to trimethoprim-sulfamethoxazole is common in tropical areas.
Usual doses of trimethoprim-sulfamethoxazole for Pneumocystis jiroveci pneumonia prophylaxis should provide limited protection against traveler's diarrhea
Fluoroquinolones are not approved for use among children <18 years old.
Pathogen: Mycobacterium tuberculosis
| Indication | First Choice | Alternatives |
|---|---|---|
|
|
Rifampin, 600 mg orally once daily for 4 months ∗ (CIII) |
HCT indicates hematopoietic cell transplant; TST, tuberculin skin tests; IGRA, interferon-gamma release assays.
Notes: A twice-weekly schedule of isoniazid and pyridoxine can be administered (CIII). The twice-weekly isoniazid dose is 15 mg/kg orally or intramuscularly (maximum dose, 900 mg). The twice-weekly pyridoxine dose is 50-100 mg orally.
A 2-month pyrazinamide/rifampin preventive therapy regimen should not be used because of the risk of fatal hepatitis (EIII) [469].
Persons who are intolerant to isoniazid or have been exposed to isoniazid-resistant tuberculosis should be referred to a tuberculosis specialist (AIII).
All intermittent dosing strategies should be administered as directly observed therapy (AIII).
There is potential for substantial drug-drug interactions between rifampin and immunosuppressive agents and other drugs.
Appendix 2: Sample Screening Tool for Visitors to HCT Centers
Visitor Screening Questionnaire
(adapted from a questionnaire developed by Children's Hospital Boston)
At our institution, it is a top priority to ensure a safe environment for all patients, families, visitors and staff. Part of this commitment includes daily wellness screening of parents/guardians, other family members, and visitors here to see hospitalized patients. This screening process helps us to prevent the spread of infection. To assist us in this effort, please read and answer the questions on this form carefully.
The name of the patient you are visiting: ___________________________________
The nursing unit that the patient is on: _____________________________________
Number of Adults visiting (age 18 and older) __________
Number of Adolescents visiting (age 13 to 17) __________
Number of Children visiting (12 and younger) __________
Note: on certain units, children 12 and younger must be siblings of the patient
If you answer Yes to either of the following questions, please let the front desk staff member know that you need to speak with the charge nurse or the patient's bedside nurse before proceeding into the unit.
-
1.In the past 3 weeks have any of the visitors indicated above had contact with a person who is ill with chickenpox, measles, mumps, or whooping cough (also known as pertussis), such as in daycare, school, at home or work?
- Yes □ No □
-
2.Have any of the visitors been sick with fever, diarrhea or vomiting in the past 3 days or had any cold symptoms including fever, cough, sore throat, or runny nose in the past 3 days?
- Yes □ No □
If yes, please explain: __________________________________________________________________________________________________________________________________________
__________________________________________________________
Signature of Adult Relationship to patient Date
Screening reviewer _________________________________________________________
Hospital personnel action: ___________________________________________________
________________________________________________________________________
_________________________________________________________________________
Appendix 3: Desensitization Protocol for HCT Patients with Sulfa Allergies∗
All HCT patients with sulfa allergies should undergo desensitization at the earliest possible time. If desensitization cannot be performed before transplantation, the desensitization process should be started posttransplantation when an absolute neutrophil count >500/mm3 is sustained for 72 hours or by day +30.
A stock solution (Standard Pediatric Oral Suspension, trimethoprim [TMP] 40 mg/sulfamethoxazole [SMX] 200 mg per 5 mL is used, followed by single-strength tablets (80 mg TMP/400 mg SMX).
Adult Sulfa Desensitization Schedule
On days 1 through 5: the stock suspension is diluted: One (1) mL of stock +
9 mL saline in a 10-mL syringe = 4 mg/mL SMX
Day 1: Take 0.25 mL = 1 mg SMX
Day 2: Take 0.50 mL = 2 mg SMX
Day 3: Take 1 mL = 4 mg SMX
Day 4: Take 2 mL = 8 mg SMX
Day 5: Take 4 mL = 16 mg SMX
On days 6 through 9, the stock solution is used full strength:
Day 6: Take 0.5 mL of stock = 20 mg SMX
Day 7: Take 1 mL of stock = 40 mg SMX
Day 8: Take 2 mL of stock = 80 mg SMX
Day 9: Take 4 mL of stock = 160 mg SMX
Day 10: Take 1 single-strength tablet (400 mg SMX).
If no rash appears, continue dosing at 1 single-strength tablet twice daily for 30 days. If the desensitization process is interrupted at this point for reasons other than TMP/SMX allergy, then give a test dose of half a single-strength tablet. If the patient tolerates this test dose, then restart dosing at 1 single-strength tablet twice daily. After 30 days of continuous therapy, if no reaction has occurred, full therapy can be given.
Pediatric Adult Sulfa Desensitization Schedule
On days 1 through 5, the stock suspension is diluted: One (1) mL of stock +
9 mL saline in a 10 mL syringe = 0.8 mg/mL TMP (4 mg/mL SMX)
Day 1: Take 0.25 mL = 0.2 mg TMP
Day 2: Take 0.50 mL = 0.4 mg TMP
Day 3: Take 1 mL = 0.8 mg TMP
Day 4: Take 2 mL = 1.6 mg TMP
Day 5: Take 4 mL = 3.2 mg TMP
On days 6 through 9, the stock solution is used full strength:
Day 6: Take 0.5 mL of stock = 4 mg TMP
Day 7: Take 1 mL of stock = 8 mg TMP
Day 8: Take 2 mL of stock = 16 mg TMP
Day 9: Take 4 mL of stock = 32 mg TMP
Day 10: If <12 years old, take 1/2 single-strength tablet (40 mg TMP);
if >12 years old: take 1 single-strength tablet (80 mg TMP)
Day 11: If <12 years old: Take 1/2 single-strength tablet (40 mg TMP) twice daily;
if >12 years old: Take 1 single-strength tablet (80 mg TMP) twice daily
If no rash appears, continue dosing as above twice daily for 30 days. If the desensitization process is interrupted at this point for reasons other than TMP/SMX allergy, then give a test dose of half a single-strength tablet if the patient is >12 years old, and 1/4 single-strength tablet if the patient is <12 years old. If the patient tolerates this test dose, then restart dosing at 1 single-strength tablet twice daily for children >12 years old and 1/2 single strength-table twice daily for children <12 years old. After 30 days of continuous therapy, if no reaction has occurred, full therapy can be given.
∗Modified from Purdy et al.,45 Ann Intern Med. 1984:100;512-514.
References
- 1.Dykewicz C.A., Jaffe H.W., Kaplan J.E. Guidelines for preventing opportunistic infections among hematopoietic stem cell transplant recipients. MMWR Recomm Rep. 2000;49:1–125. CE1-7. [PubMed] [Google Scholar]
- 2.Centers for Disease Control and Prevention (CDC) 1999 USPHS/IDSA guidelines for the prevention of opportunistic infections in persons infected with human immunodeficiency virus. MMWR Morb Mortal Wkly Rep. 1999;48:1–66. [Google Scholar]
- 3.CIBMTR Summary Slides. http://www.cibmtr.org/SERVICES/Observational_Research/Summary_Slides/index.html
- 4.Bachanova V., Brunstein C.G., Burns L.J. Fewer infections and lower infection-related mortality following non-myeloablative versus myeloablative conditioning for allotransplantation of patients with lymphoma. Bone Marrow Transplant. 2009;43:237–244. doi: 10.1038/bmt.2008.313. [DOI] [PubMed] [Google Scholar]
- 5.Meijer E., Dekker A.W., Lokhorst H.M., Petersen E.J., Nieuwenhuis H.K., Verdonck L.F. Low incidence of infectious complications after nonmyeloablative compared with myeloablative allogeneic stem cell transplantation. Transpl Infect Dis. 2004;6:171–178. doi: 10.1111/j.1399-3062.2004.00075.x. [DOI] [PubMed] [Google Scholar]
- 6.Junghanss C., Boeckh M., Carter R.A. Incidence and outcome of cytomegalovirus infections following nonmyeloablative compared with myeloablative allogeneic stem cell transplantation, a matched control study. Blood. 2002;99:1978–1985. doi: 10.1182/blood.v99.6.1978. [DOI] [PubMed] [Google Scholar]
- 7.Junghanss C., Marr K.A., Carter R.A. Incidence and outcome of bacterial and fungal infections following nonmyeloablative compared with myeloablative allogeneic hematopoietic stem cell transplantation: a matched control study. Biol Blood Marrow Transplant. 2002;8:512–520. doi: 10.1053/bbmt.2002.v8.pm12374456. [DOI] [PubMed] [Google Scholar]
- 8.Baron F., Sandmaier B.M. Chimerism and outcomes after allogeneic hematopoietic cell transplantation following nonmyeloablative conditioning. Leukemia. 2006;20:1690–1700. doi: 10.1038/sj.leu.2404335. [DOI] [PubMed] [Google Scholar]
- 9.Huss R., Deeg H.J., Gooley T. Effect of mixed chimerism on graft-versus-host disease, disease recurrence and survival after HLA-identical marrow transplantation for aplastic anemia or chronic myelogenous leukemia. Bone Marrow Transplant. 1996;18:767–776. [PubMed] [Google Scholar]
- 10.Welniak L.A., Blazar B.R., Murphy W.J. Immunobiology of allogeneic hematopoietic stem cell transplantation. Annu Rev Immunol. 2007;25:139–170. doi: 10.1146/annurev.immunol.25.022106.141606. [DOI] [PubMed] [Google Scholar]
- 11.Gaziev J., Lucarelli G. Stem cell transplantation and gene therapy for hemoglobinopathies. Curr Hematol Rep. 2005;4:126–131. [PubMed] [Google Scholar]
- 12.Horwitz M.E., Barrett A.J., Brown M.R. Treatment of chronic granulomatous disease with nonmyeloablative conditioning and a T-cell-depleted hematopoietic allograft. N Engl J Med. 2001;344:881–888. doi: 10.1056/NEJM200103223441203. [DOI] [PubMed] [Google Scholar]
- 13.Giralt S. Allogeneic hematopoietic progenitor cell transplantation for the treatment of chronic myelogenous leukemia in the era of tyrosine kinase inhibitors: lessons learned to date. Clin Lymphoma Myeloma. 2007;7(Suppl 3):S102–S104. doi: 10.3816/clm.2007.s.009. [DOI] [PubMed] [Google Scholar]
- 14.Oliansky D.M., Rizzo J.D., Aplan P.D. The role of cytotoxic therapy with hematopoietic stem cell transplantation in the therapy of acute myeloid leukemia in children: an evidence-based review. Biol Blood Marrow Transplant. 2007;13:1–25. doi: 10.1016/j.bbmt.2006.10.024. [DOI] [PubMed] [Google Scholar]
- 15.Dreger P., Corradini P., Kimby E. Indications for allogeneic stem cell transplantation in chronic lymphocytic leukemia: the EBMT transplant consensus. Leukemia. 2007;21:12–17. doi: 10.1038/sj.leu.2404441. [DOI] [PubMed] [Google Scholar]
- 16.de Witte T., Suciu S., Brand R., Muus P., Kroger N. Autologous stem cell transplantation in myelodysplastic syndromes. Semin Hematol. 2007;44:274–277. doi: 10.1053/j.seminhematol.2007.08.006. [DOI] [PubMed] [Google Scholar]
- 17.Davies J.K., Guinan E.C. An update on the management of severe idiopathic aplastic anaemia in children. Br J Haematol. 2007;136:549–564. doi: 10.1111/j.1365-2141.2006.06461.x. [DOI] [PubMed] [Google Scholar]
- 18.Aschan J. Risk assessment in haematopoietic stem cell transplantation: conditioning. Best Pract Res Clin Haematol. 2007;20:295–310. doi: 10.1016/j.beha.2006.09.004. [DOI] [PubMed] [Google Scholar]
- 19.Leather H.L., Wingard J.R. Infections following hematopoietic stem cell transplantation. Infect Dis Clin North Am. 2001;15:483–520. doi: 10.1016/s0891-5520(05)70157-4. [DOI] [PubMed] [Google Scholar]
- 20.Fraser C.J., Scott Baker K. The management and outcome of chronic graft-versus-host disease. Br J Haematol. 2007;138:131–145. doi: 10.1111/j.1365-2141.2007.06652.x. [DOI] [PubMed] [Google Scholar]
- 21.Chaidos A., Kanfer E., Apperley J.F. Risk assessment in haemotopoietic stem cell transplantation: disease and disease stage. Best Pract Res Clin Haematol. 2007;20:125–154. doi: 10.1016/j.beha.2006.10.003. [DOI] [PubMed] [Google Scholar]
- 22.Baccarani M., Saglio G., Goldman J. Evolving concepts in the management of chronic myeloid leukemia: recommendations from an expert panel on behalf of the European LeukemiaNet. Blood. 2006;108:1809–1820. doi: 10.1182/blood-2006-02-005686. [DOI] [PubMed] [Google Scholar]
- 23.Urbano-Ispizua A. Risk assessment in haematopoietic stem cell transplantation: stem cell source. Best Pract Res Clin Haematol. 2007;20:265–280. doi: 10.1016/j.beha.2006.09.006. [DOI] [PubMed] [Google Scholar]
- 24.Petersdorf E.W. Risk assessment in haematopoietic stem cell transplantation: histocompatibility. Best Pract Res Clin Haematol. 2007;20:155–170. doi: 10.1016/j.beha.2006.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Bjorkstrand B., Gahrton G. High-dose treatment with autologous stem cell transplantation in multiple myeloma: past, present, and future. Semin Hematol. 2007;44:227–233. doi: 10.1053/j.seminhematol.2007.08.010. [DOI] [PubMed] [Google Scholar]
- 26.Passweg J., Tyndall A. Autologous stem cell transplantation in autoimmune diseases. Semin Hematol. 2007;44:278–285. doi: 10.1053/j.seminhematol.2007.08.001. [DOI] [PubMed] [Google Scholar]
- 27.Sadelain M., Rivella S., Lisowski L., Samakoglu S., Riviere I. Globin gene transfer for treatment of the beta-thalassemias and sickle cell disease. Best Pract Res Clin Haematol. 2004;17:517–534. doi: 10.1016/j.beha.2004.08.002. [DOI] [PubMed] [Google Scholar]
- 28.Leather H.L., Wingard J.R. New strategies of antifungal therapy in hematopoietic stem cell transplant recipients and patients with hematological malignancies. Blood Rev. 2006;20:267–287. doi: 10.1016/j.blre.2006.03.001. [DOI] [PubMed] [Google Scholar]
- 29.Collin B.A., Leather H.L., Wingard J.R., Ramphal R. Evolution, incidence, and susceptibility of bacterial bloodstream isolates from 519 bone marrow transplant patients. Clin Infect Dis. 2001;33:947–953. doi: 10.1086/322604. [DOI] [PubMed] [Google Scholar]
- 30.Powles R., Mehta J., Kulkarni S. Allogeneic blood and bone-marrow stem-cell transplantation in haematological malignant diseases: a randomised trial. Lancet. 2000;355:1231–1237. doi: 10.1016/S0140-6736(00)02090-0. [DOI] [PubMed] [Google Scholar]
- 31.Wagner J.E., Barker J.N., DeFor T.E. Transplantation of unrelated donor umbilical cord blood in 102 patients with malignant and nonmalignant diseases: influence of CD34 cell dose and HLA disparity on treatment-related mortality and survival. Blood. 2002;100:1611–1618. doi: 10.1182/blood-2002-01-0294. [DOI] [PubMed] [Google Scholar]
- 32.Baron F., Storb R. Hematopoietic cell transplantation after reduced-intensity conditioning for older adults with acute myeloid leukemia in complete remission. Curr Opin Hematol. 2007;14:145–151. doi: 10.1097/MOH.0b013e3280168462. [DOI] [PubMed] [Google Scholar]
- 33.Kolb H.J., Schmid C., Barrett A.J., Schendel D.J. Graft-versus-leukemia reactions in allogeneic chimeras. Blood. 2004;103:767–776. doi: 10.1182/blood-2003-02-0342. [DOI] [PubMed] [Google Scholar]
- 34.Ruggeri L., Mancusi A., Burchielli E. NK cell alloreactivity and allogeneic hematopoietic stem cell transplantation. Blood Cells Mol Dis. 2008;40:84–90. doi: 10.1016/j.bcmd.2007.06.029. [DOI] [PubMed] [Google Scholar]
- 35.Dazzi F., Fozza C. Disease relapse after haematopoietic stem cell transplantation: risk factors and treatment. Best Pract Res Clin Haematol. 2007;20:311–327. doi: 10.1016/j.beha.2006.10.002. [DOI] [PubMed] [Google Scholar]
- 36.Barkholt L., Bregni M., Remberger M. Allogeneic haematopoietic stem cell transplantation for metastatic renal carcinoma in Europe. Ann Oncol. 2006;17:1134–1140. doi: 10.1093/annonc/mdl086. [DOI] [PubMed] [Google Scholar]
- 37.Eapen M., Rubinstein P., Zhang M.J. Outcomes of transplantation of unrelated donor umbilical cord blood and bone marrow in children with acute leukaemia: a comparison study. Lancet. 2007;369:1947–1954. doi: 10.1016/S0140-6736(07)60915-5. [DOI] [PubMed] [Google Scholar]
- 38.Gluckman E., Rocha V. Donor selection for unrelated cord blood transplants. Curr Opin Immunol. 2006;18:565–570. doi: 10.1016/j.coi.2006.07.014. [DOI] [PubMed] [Google Scholar]
- 39.Buckley R.H., Schiff S.E., Schiff R.I. Haploidentical bone marrow stem cell transplantation in human severe combined immunodeficiency. Semin Hematol. 1993;30:92–101. discussion 102–104. [PubMed] [Google Scholar]
- 40.Aversa F., Tabilio A., Velardi A. Treatment of high-risk acute leukemia with T-cell-depleted stem cells from related donors with one fully mismatched HLA haplotype. N Engl J Med. 1998;339:1186–1193. doi: 10.1056/NEJM199810223391702. [DOI] [PubMed] [Google Scholar]
- 41.Aversa F. Haploidentical haematopoietic stem cell transplantation for acute leukaemia in adults: experience in Europe and the United States. Bone Marrow Transplant. 2008;41:473–481. doi: 10.1038/sj.bmt.1705966. [DOI] [PubMed] [Google Scholar]
- 42.Storek J., Dawson M.A., Storer B. Immune reconstitution after allogeneic marrow transplantation compared with blood stem cell transplantation. Blood. 2001;97:3380–3389. doi: 10.1182/blood.v97.11.3380. [DOI] [PubMed] [Google Scholar]
- 43.Schmitz N., Eapen M., Horowitz M.M. Long-term outcome of patients given transplants of mobilized blood or bone marrow: a report from the International Bone Marrow Transplant Registry and the European Group for Blood and Marrow Transplantation. Blood. 2006;108:4288–4290. doi: 10.1182/blood-2006-05-024042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Dey B.R., Shaffer J., Yee A.J. Comparison of outcomes after transplantation of peripheral blood stem cells versus bone marrow following an identical nonmyeloablative conditioning regimen. Bone Marrow Transplant. 2007;40:19–27. doi: 10.1038/sj.bmt.1705688. [DOI] [PubMed] [Google Scholar]
- 45.Champlin R., Khouri I., Anderlini P. Nonmyeloablative preparative regimens for allogeneic hematopoietic transplantation. Biology and current indications. Oncology (Williston Park) 2003;17:94–100. discussion 103–107. [PubMed] [Google Scholar]
- 46.Georges G.E., Storb R. Review of “minitransplantation”: nonmyeloablative allogeneic hematopoietic stem cell transplantation. Int J Hematol. 2003;77:3–14. doi: 10.1007/BF02982597. [DOI] [PubMed] [Google Scholar]
- 47.Feinstein L., Storb R. Nonmyeloablative hematopoietic cell transplantation. Curr Opin Oncol. 2001;13:95–100. doi: 10.1097/00001622-200103000-00003. [DOI] [PubMed] [Google Scholar]
- 48.Giralt S., Thall P.F., Khouri I. Melphalan and purine analog-containing preparative regimens: reduced-intensity conditioning for patients with hematologic malignancies undergoing allogeneic progenitor cell transplantation. Blood. 2001;97:631–637. doi: 10.1182/blood.v97.3.631. [DOI] [PubMed] [Google Scholar]
- 49.Hows J.M., Passweg J.R., Tichelli A. Comparison of long-term outcomes after allogeneic hematopoietic stem cell transplantation from matched sibling and unrelated donors. Bone Marrow Transplant. 2006;38:799–805. doi: 10.1038/sj.bmt.1705531. [DOI] [PubMed] [Google Scholar]
- 50.Lewin S.R., Heller G., Zhang L. Direct evidence for new T-cell generation by patients after either T-cell-depleted or unmodified allogeneic hematopoietic stem cell transplantations. Blood. 2002;100:2235–2242. [PubMed] [Google Scholar]
- 51.Small T.N., Keever C.A., Weiner-Fedus S., Heller G., O'Reilly R.J., Flomenberg N. B-cell differentiation following autologous, conventional, or T-cell depleted bone marrow transplantation: a recapitulation of normal B-cell ontogeny. Blood. 1990;76:1647–1656. [PubMed] [Google Scholar]
- 52.Storek J., Wells D., Dawson M.A., Storer B., Maloney D.G. Factors influencing B lymphopoiesis after allogeneic hematopoietic cell transplantation. Blood. 2001;98:489–491. doi: 10.1182/blood.v98.2.489. [DOI] [PubMed] [Google Scholar]
- 53.Manz R.A., Thiel A., Radbruch A. Lifetime of plasma cells in the bone marrow. Nature. 1997;388:133–134. doi: 10.1038/40540. [DOI] [PubMed] [Google Scholar]
- 54.Mackall C.L., Fleisher T.A., Brown M.R. Distinctions between CD8+ and CD4 + T-cell regenerative pathways result in prolonged T-cell subset imbalance after intensive chemotherapy. Blood. 1997;89:3700–3707. [PubMed] [Google Scholar]
- 55.Roux E., Dumont-Girard F., Starobinski M. Recovery of immune reactivity after T-cell-depleted bone marrow transplantation depends on thymic activity. Blood. 2000;96:2299–2303. [PubMed] [Google Scholar]
- 56.Storek J., Gooley T., Witherspoon R.P., Sullivan K.M., Storb R. Infectious morbidity in long-term survivors of allogeneic marrow transplantation is associated with low CD4 T cell counts. Am J Hematol. 1997;54:131–138. doi: 10.1002/(sici)1096-8652(199702)54:2<131::aid-ajh6>3.0.co;2-y. [DOI] [PubMed] [Google Scholar]
- 57.Berger M., Figari O., Bruno B. Lymphocyte subsets recovery following allogeneic bone marrow transplantation (BMT): CD4+ cell count and transplant-related mortality. Bone Marrow Transplant. 2008;41:55–62. doi: 10.1038/sj.bmt.1705870. [DOI] [PubMed] [Google Scholar]
- 58.Talmadge J.E. Lymphocyte subset recovery following allogeneic bone marrow transplantation: CD4(+)-cell count and transplant-related mortality. Bone Marrow Transplant. 2008;41:19–21. doi: 10.1038/sj.bmt.1705871. [DOI] [PubMed] [Google Scholar]
- 59.Kim D.H., Sohn S.K., Won D.I., Lee N.Y., Suh J.S., Lee K.B. Rapid helper T-cell recovery above 200 × 10 6/l at 3 months correlates to successful transplant outcomes after allogeneic stem cell transplantation. Bone Marrow Transplant. 2006;37:1119–1128. doi: 10.1038/sj.bmt.1705381. [DOI] [PubMed] [Google Scholar]
- 60.Dumont-Girard F., Roux E., van Lier R.A. Reconstitution of the T-cell compartment after bone marrow transplantation: restoration of the repertoire by thymic emigrants. Blood. 1998;92:4464–4471. [PubMed] [Google Scholar]
- 61.Mackall C.L., Fleisher T.A., Brown M.R. Age, thymopoiesis, and CD4 + T-lymphocyte regeneration after intensive chemotherapy. N Engl J Med. 1995;332:143–149. doi: 10.1056/NEJM199501193320303. [DOI] [PubMed] [Google Scholar]
- 62.Weinberg K., Annett G., Kashyap A., Lenarsky C., Forman S.J., Parkman R. The effect of thymic function on immunocompetence following bone marrow transplantation. Biol Blood Marrow Transplant. 1995;1:18–23. [PubMed] [Google Scholar]
- 63.Storek J., Joseph A., Dawson M.A., Douek D.C., Storer B., Maloney D.G. Factors influencing T-lymphopoiesis after allogeneic hematopoietic cell transplantation. Transplantation. 2002;73:1154–1158. doi: 10.1097/00007890-200204150-00026. [DOI] [PubMed] [Google Scholar]
- 64.Hakim F.T., Memon S.A., Cepeda R. Age-dependent incidence, time course, and consequences of thymic renewal in adults. J Clin Invest. 2005;115:930–939. doi: 10.1172/JCI22492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Myers L.A., Patel D.D., Puck J.M., Buckley R.H. Hematopoietic stem cell transplantation for severe combined immunodeficiency in the neonatal period leads to superior thymic output and improved survival. Blood. 2002;99:872–878. doi: 10.1182/blood.v99.3.872. [DOI] [PubMed] [Google Scholar]
- 66.Komanduri K.V., St John L.S., de Lima M. Delayed immune reconstitution after cord blood transplantation is characterized by impaired thymopoiesis and late memory T cell skewing. Blood. 2007;110:4543–4551. doi: 10.1182/blood-2007-05-092130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Thomson B.G., Robertson K.A., Gowan D. Analysis of engraftment, graft-versus-host disease, and immune recovery following unrelated donor cord blood transplantation. Blood. 2000;96:2703–2711. [PubMed] [Google Scholar]
- 68.Bittencourt H., Rocha V., Chevret S. Association of CD34 cell dose with hematopoietic recovery, infections, and other outcomes after HLA-identical sibling bone marrow transplantation. Blood. 2002;99:2726–2733. doi: 10.1182/blood.v99.8.2726. [DOI] [PubMed] [Google Scholar]
- 69.Ochs L., Shu X.O., Miller J. Late infections after allogeneic bone marrow transplantation: comparison of incidence in related and unrelated donor transplant recipients. Blood. 1995;86:3979–3986. [PubMed] [Google Scholar]
- 70.Sullivan K.M., Mori M., Sanders J. Late complications of allogeneic and autologous marrow transplantation. Bone Marrow Transplant. 1992;10(Suppl 1):127–134. [PubMed] [Google Scholar]
- 71.Atkinson K., Farewell V., Storb R. Analysis of late infections after human bone marrow transplantation: role of genotypic nonidentity between marrow donor and recipient and of nonspecific suppressor cells in patients with chronic graft-versus-host disease. Blood. 1982;60:714–720. [PubMed] [Google Scholar]
- 72.Hakim F.T., Mackall C.L. The immune system: effector and target of GVHD. In: Ferrara J.L.M., Deeg H.J., Burakoff S.J., editors. Graft-vs.-Host Disease. Marcel Dekker; New York: 1997. pp. 257–289. [Google Scholar]
- 73.Maris M., Boeckh M., Storer B. Immunologic recovery after hematopoietic cell transplantation with nonmyeloablative conditioning. Exp Hematol. 2003;31:941–952. doi: 10.1016/s0301-472x(03)00201-7. [DOI] [PubMed] [Google Scholar]
- 74.van Burik J.A., Brunstein C.G. Infectious complications following unrelated cord blood transplantation. Vox Sang. 2007;92:289–296. doi: 10.1111/j.1423-0410.2007.00900.x. [DOI] [PubMed] [Google Scholar]
- 75.Cwynarski K., Ainsworth J., Cobbold M. Direct visualization of cytomegalovirus-specific T-cell reconstitution after allogeneic stem cell transplantation. Blood. 2001;97:1232–1240. doi: 10.1182/blood.v97.5.1232. [DOI] [PubMed] [Google Scholar]
- 76.AABB . AABB; Bethesda, MD: 2007. Standards for Cellular Therapy Product Services. [Google Scholar]
- 77.Fact-Jacie . Foundation for the Accreditation of Cellular Therapy; Omaha, NE: 2006. International Standards for Cellular Therapy Product Collection, Processing, and Administration. [Google Scholar]
- 78.National Marrow Donor Program . 19th ed. National Marrow Donor Program; Minneapolis, MN: 2004. National Marrow Donor Program Standards. [Google Scholar]
- 79.World Marrow Donor Association . World Marrow Donor Association; Leiden, The Netherlands: 2007. International Standards for Unrelated Hemtatopoietic Stem Cell Donor Registries. [DOI] [PubMed] [Google Scholar]
- 80.Food and Drug Administration . U.S. Department of Health and Human Services; Washington, DC: 2007. Guidance for Industry: Eligibility Determination for Donors of Human Cells, Tissues, and Cellular- and Tissue-Based Products (HCT/Ps) [Google Scholar]
- 81.European Commission Commission Directive 2004/23/EC. Official J Eur Union. 2004;L 102:48–58. [Google Scholar]
- 82.European Commission Commission Directive 2006/17/EC. Official J Eur Union. 2006;L 38:40–52. [Google Scholar]
- 83.US National Archives and Records Administration . GPO; Washington, DC: 2004. 21 CFR, Part 1271, Subpart C—Donor Eligibility. [Google Scholar]
- 84.Centers for Disease Control and Prevention West Nile virus transmission through blood transfusion—South Dakota, 2006. MMWR Morb Mortal Wkly Rep. 2007;56:76–79. [PubMed] [Google Scholar]
- 85.Centers for Disease Control and Prevention West Nile virus update—United States, January 1-November 13, 2007. MMWR Morb Mortal Wkly Rep. 2007;56:1191–1192. [PubMed] [Google Scholar]
- 86.Centers for Disease Control and Prevention West Nile virus infections in organ transplant recipients—New York and Pennsylvania, August-September, 2005. MMWR Morb Mortal Wkly Rep. 2005;54:1021–1023. [PubMed] [Google Scholar]
- 87.Centers for Disease Control and Prevention Transfusion-associated transmission of West Nile virus—Arizona, 2004. MMWR Morb Mortal Wkly Rep. 2004;53:842–844. [PubMed] [Google Scholar]
- 88.Centers for Disease Control and Prevention Detection of West Nile virus in blood donations—United States, 2003. MMWR Morb Mortal Wkly Rep. 2003;52:769–772. [PubMed] [Google Scholar]
- 89.Centers for Disease Control and Prevention Update: detection of West Nile virus in blood donations—United States, 2003. MMWR Morb Mortal Wkly Rep. 2003;52:916–919. [PubMed] [Google Scholar]
- 90.Centers for Disease Control and Prevention Investigation of blood transfusion recipients with West Nile virus infections. MMWR Morb Mortal Wkly Rep. 2002;51:823. [PubMed] [Google Scholar]
- 91.Food and Drug Administration Suitability determination for donors of human cellular and tissue-based products: proposed rule. Fed. Reg. 1999;64:52696–52723. [Google Scholar]
- 92.Hewitt P.E., Llewelyn C.A., Mackenzie J., Will R.G. Creutzfeldt-Jakob disease and blood transfusion: results of the UK Transfusion Medicine Epidemiological Review study. Vox Sang. 2006;91:221–230. doi: 10.1111/j.1423-0410.2006.00833.x. [DOI] [PubMed] [Google Scholar]
- 93.Ludlam C.A., Turner M.L. Managing the risk of transmission of variant Creutzfeldt Jakob disease by blood products. Br J Haematol. 2006;132:13–24. doi: 10.1111/j.1365-2141.2005.05796.x. [DOI] [PubMed] [Google Scholar]
- 94.Hart J., Leier B., Nahirniak S. Informed consent for blood transfusion: should the possibility of prion risk be included? Transfus Med Rev. 2004;18:177–183. doi: 10.1016/j.tmrv.2004.03.001. [DOI] [PubMed] [Google Scholar]
- 95.Llewelyn C.A., Hewitt P.E., Knight R.S. Possible transmission of variant Creutzfeldt-Jakob disease by blood transfusion. Lancet. 2004;363:417–421. doi: 10.1016/S0140-6736(04)15486-X. [DOI] [PubMed] [Google Scholar]
- 96.Ljungman P., Perez-Bercoff L., Jonsson J. Risk factors for the development of cytomegalovirus disease after allogeneic stem cell transplantation. Haematologica. 2006;91:78–83. [PubMed] [Google Scholar]
- 97.Nichols W.G., Corey L., Gooley T., Davis C., Boeckh M. High risk of death due to bacterial and fungal infection among cytomegalovirus (CMV)-seronegative recipients of stem cell transplants from seropositive donors: evidence for indirect effects of primary CMV infection. J Infect Dis. 2002;185:273–282. doi: 10.1086/338624. [DOI] [PubMed] [Google Scholar]
- 98.Matsuo Y., Takeishi S., Miyamoto T. Toxoplasmosis encephalitis following severe graft-vs-host disease after allogeneic hematopoietic stem cell transplantation: 17 yr experience in Fukuoka BMT group. Eur J Haematol. 2007;79:317–321. doi: 10.1111/j.1600-0609.2007.00919.x. [DOI] [PubMed] [Google Scholar]
- 99.Kotton C.N. Zoonoses in solid-organ and hematopoietic stem cell transplant recipients. Clin Infect Dis. 2007;44:857–866. doi: 10.1086/511859. [DOI] [PubMed] [Google Scholar]
- 100.Slavin M.A., Meyers J.D., Remington J.S., Hackman R.C. Toxoplasma gondii infection in marrow transplant recipients: a 20 year experience. Bone Marrow Transplant. 1994;13:549–557. [PubMed] [Google Scholar]
- 101.Kitchen A., Mijovic A., Hewitt P. Transfusion-transmitted malaria: current donor selection guidelines are not sufficient. Vox Sang. 2005;88:200–201. doi: 10.1111/j.1423-0410.2005.00610.x. [DOI] [PubMed] [Google Scholar]
- 102.Kitchen A.D., Barbara J.A., Hewitt P.E. Documented cases of post-transfusion malaria occurring in England: a review in relation to current and proposed donor-selection guidelines. Vox Sang. 2005;89:77–80. doi: 10.1111/j.1423-0410.2005.00661.x. [DOI] [PubMed] [Google Scholar]
- 103.Centers for Disease Control and Prevention Probable transfusion-transmitted malaria—Houston, Texas, 2003. MMWR Morb Mortal Wkly Rep. 2003;52:1075–1076. [PubMed] [Google Scholar]
- 104.Mungai M., Tegtmeier G., Chamberland M., Parise M. Transfusion-transmitted malaria in the United States from 1963 through 1999. New Engl J Med. 2001;344:1973–1978. doi: 10.1056/NEJM200106283442603. [DOI] [PubMed] [Google Scholar]
- 105.Centers for Disease Control and Prevention Transfusion-transmitted malaria—Missouri and Pennsylvania, 1996-1998. MMWR Morb Mortal Wkly Rep. 1999;48:253–256. [PubMed] [Google Scholar]
- 106.Villeneuve L., Cassaing S., Magnaval J.F. Plasmodium falciparum infection following allogeneic bone-marrow transplantation. Ann Trop Med Parasitol. 1999;93:533–535. [PubMed] [Google Scholar]
- 107.O'Donnell J., Goldman J.M., Wagner K. Donor-derived Plasmodium vivax infection following volunteer unrelated bone marrow transplantation. Bone Marrow Transplant. 1998;21:313–314. doi: 10.1038/sj.bmt.1701073. [DOI] [PubMed] [Google Scholar]
- 108.Lefrere F., Besson C., Datry A. Transmission of Plasmodium falciparum by allogeneic bone marrow transplantation. Bone Marrow Transplant. 1996;18:473–474. [PubMed] [Google Scholar]
- 109.Herwaldt B.L., Neitzel D.F., Gorlin J.B. Transmission of Babesia microti in Minnesota through four blood donations from the same donor over a 6-month period. Transfusion. 2002;42:1154–1158. doi: 10.1046/j.1537-2995.2002.00189.x. [DOI] [PubMed] [Google Scholar]
- 110.Kain K.C., Jassoum S.B., Fong I.W., Hannach B. Transfusion-transmitted babesiosis in Ontario: first reported case in Canada. CMAJ. 2001;164:1721–1723. [PMC free article] [PubMed] [Google Scholar]
- 111.Kjemtrup A.M., Lee B., Fritz C.L., Evans C., Chervenak M., Conrad P.A. Investigation of transfusion transmission of a WA1-type babesial parasite to a premature infant in California. Transfusion. 2002;42:1482–1487. doi: 10.1046/j.1537-2995.2002.00245.x. [DOI] [PubMed] [Google Scholar]
- 112.Linden J.V., Wong S.J., Chu F.K., Schmidt G.B., Bianco C. Transfusion-associated transmission of babesiosis in New York state. Transfusion. 2000;40:285–289. doi: 10.1046/j.1537-2995.2000.40030285.x. [DOI] [PubMed] [Google Scholar]
- 113.Pantanowitz L., Telford S.R., Cannon M.E. Tick-borne diseases in transfusion medicine. Transfus Med Rev. 2002;12:85–106. doi: 10.1046/j.1365-3148.2002.00358.x. [DOI] [PubMed] [Google Scholar]
- 114.Pantanowitz L., Telford S.R., Cannon M.E. The impact of babesiosis on transfusion medicine. Transfus Med Rev. 2002;16:131–143. doi: 10.1053/tmrv.2002.31462. [DOI] [PubMed] [Google Scholar]
- 115.AABB . AABB; Bethesda, MD: 2008. Standards for Blood Banks and Transfusion Services. [Google Scholar]
- 116.Klein M.B., Miller J.S., Nelson C.M., Goodman J.L. Primary bone marrow progenitors of both granulocytic and monocytic lineages are susceptible to infection with the agent of human granulocytic ehrlichiosis. J Infect Dis. 1997;176:1405–1409. doi: 10.1086/517332. [DOI] [PubMed] [Google Scholar]
- 117.Centers for Disease Control and Prevention Blood donor screening for chagas disease—United States, 2006-2007. MMWR Morb Mortal Wkly Rep. 2007;56:141–143. [PubMed] [Google Scholar]
- 118.Centers for Disease Control and Prevention Chagas disease after organ transplantation—Los Angeles, California, 2006. MMWR Morb Mortal Wkly Rep. 2006;55:798–800. [PubMed] [Google Scholar]
- 119.Centers for Disease Control and Prevention Chagas disease after organ transplantation—United States, 2001. MMWR Morb Mortal Wkly Rep. 2002;51:210–212. [PubMed] [Google Scholar]
- 120.Leiby D.A., Lenes B.A., Tibbals M.A., Tames-Olmedo M.T. Prospective evaluation of a patient with Trypanosoma cruzi infection transmitted by transfusion. N Engl J Med. 1999;341:1237–1239. doi: 10.1056/NEJM199910143411615. [DOI] [PubMed] [Google Scholar]
- 121.Leiby D.A., Read E.J., Lenes B.A. Seroepidemiology of Trypanosoma cruzi, etiologic agent of Chagas' disease, in US blood donors. J Infect Dis. 1997;176:1047–1052. doi: 10.1086/516534. [DOI] [PubMed] [Google Scholar]
- 122.Dodd R.Y. Transmission of parasites by blood transfusion. Vox Sang. 1998;74(Suppl 2):161–163. doi: 10.1111/j.1423-0410.1998.tb05415.x. [DOI] [PubMed] [Google Scholar]
- 123.Aljurf M., Gyger M., Alrajhi A. Mycobacterium tuberculosis infection in allogeneic bone marrow transplantation patients. Bone Marrow Transplantation. 1999;24:551–554. doi: 10.1038/sj.bmt.1701930. [DOI] [PubMed] [Google Scholar]
- 124.Tavil B., Gulhan B., Ozcelik U. Tuberculin skin test positivity in pediatric allogeneic BMT recipients and donors in Turkey. Pediatr Transplant. 2007;11:414–418. doi: 10.1111/j.1399-3046.2007.00679.x. [DOI] [PubMed] [Google Scholar]
- 125.National Marrow Donor Program . National Marrow Donor Program; Minneapolis, MN: 2007. Donor Health History Screening Questionnaire. [Google Scholar]
- 126.Husebekk A., Skaug K., Kolstad A., Dahl I.M., Gutteberg T., Skogen B. Hepatitis B virus-infected peripheral blood progenitor cell harvests in liquid nitrogen freezer containing non-infectious products. Transfusion. 2004;44:942–943. doi: 10.1111/j.1537-2995.2004.00379.x. [DOI] [PubMed] [Google Scholar]
- 127.Reich-Slotky R., Semidei-Pomales M., Schwartz J. Microbial pathogens of hematopoietic stem cells—screening and testing for infectious diseases. Rev Med Microbiol. 2007;18:17–27. [Google Scholar]
- 128.Padley D.J., Dietz A.B., Gastineau D.A. Sterility testing of hematopoietic progenitor cell products: a single-institution series of culture-positive rates and successful infusion of culture-positive products. Transfusion. 2007;47:636–643. doi: 10.1111/j.1537-2995.2007.01165.x. [DOI] [PubMed] [Google Scholar]
- 129.Majado M.J., Garcia-Hernandez A., Morales A. Influence of harvest bacterial contamination on autologous peripheral blood progenitor cells post-transplant. Xenotransplantation. 2007;39:121–125. doi: 10.1038/sj.bmt.1705549. [DOI] [PubMed] [Google Scholar]
- 130.Khuu H.M., Patel N., Carter C.S., Murray P.R., Read E.J. Sterility testing of cell therapy products: parallel comparison of automated methods with a CFR-compliant method. Transfusion. 2006;46:2071–2082. doi: 10.1111/j.1537-2995.2006.01041.x. [DOI] [PubMed] [Google Scholar]
- 131.Klein M.A., Kadidlo D., McCullough J., McKenna D.H., Burns L.J. Microbial contamination of hematopoietic stem cell products: incidence and clinical sequelae. Biol Blood Marrow Transplant. 2006;12:1142–1149. doi: 10.1016/j.bbmt.2006.06.011. [DOI] [PubMed] [Google Scholar]
- 132.Kelly M., Roy D.C., Labbe A.C., Laverdiere M. What is the clinical significance of infusing hematopoietic cell grafts contaminated with bacteria? Rev Med Microbiol. 2006;38:183–188. doi: 10.1038/sj.bmt.1705421. [DOI] [PubMed] [Google Scholar]
- 133.Kamble R., Pant S., Selby G.B. Microbial contamination of hematopoietic progenitor cell grafts-incidence, clinical outcome, and cost-effectiveness: an analysis of 735 grafts. Transfusion. 2005;45:874–878. doi: 10.1111/j.1537-2995.2005.04178.x. [DOI] [PubMed] [Google Scholar]
- 134.Larrea L., de la Rubia J., Soler M.A. Quality control of bacterial contamination in autologous peripheral blood stem cells for transplantation. Haematologica. 2004;89:1232–1237. [PubMed] [Google Scholar]
- 135.Khuu H.M., Stock F., McGann M. Comparison of automated culture systems with a CFR/USP-compliant method for sterility testing of cell-therapy products. Cytotherapy. 2004;6:183–195. doi: 10.1080/14653240410005997. [DOI] [PubMed] [Google Scholar]
- 136.Lowder J.N., Whelton P. Microbial contamination of cellular products for hematolymphoid transplantation therapy: assessment of the problem and strategies to minimize the clinical impact. Cytotherapy. 2003;5:377–390. doi: 10.1080/14653240310003044. [DOI] [PubMed] [Google Scholar]
- 137.Padley D.J., Greiner C.W., Heddlesten-Rediskel T.L., Hopkins M.K., Maasi M.L., Gastineau D.A. Endogenous microbial contamination of cultured autologous preparations in trials of cancer immunotherapy. Cytotherapy. 2003;5:147–152. doi: 10.1080/14653240310001046. [DOI] [PubMed] [Google Scholar]
- 138.Honohan A., Olthuis H., Bernards A.T., van Beckhoven J.M., Brand A. Microbial contamination of cord blood stem cells. Vox Sang. 2002;82:32–38. doi: 10.1046/j.1423-0410.2002.00133.x. [DOI] [PubMed] [Google Scholar]
- 139.Gee A.P. Product release assays. Cytotherapy. 1999;1:485–491. doi: 10.1080/0032472031000141309. [DOI] [PubMed] [Google Scholar]
- 140.Gagnon J.A., Porter K., Guerin D., Gluck S. Bacterial decontamination of blood stem cell apheresis products. J Clin Apher. 1998;13:103–107. doi: 10.1002/(sici)1098-1101(1998)13:3<103::aid-jca2>3.0.co;2-g. [DOI] [PubMed] [Google Scholar]
- 141.Espinosa M.T.F., Fox R., Creger R.J., Lazarus H.M. Microbiologic contamination of peripheral blood progenitor cells collected for hematopoietic cell transplantation. Transfusion. 1996;36:789–793. doi: 10.1046/j.1537-2995.1996.36996420754.x. [DOI] [PubMed] [Google Scholar]
- 142.US National Archives and Records Administration . GPO; Washington, DC: 2004. 21 CFR, Part 1271, Subpart D—Good Tissue Practice. [Google Scholar]
- 143.NetCord-FACT . Foundation for the Accreditation of Cellular Therapy; Omaha, NE: 2006. International Standards for Cord Blood Collection, Processing, Testing, Banking, Selection, and Release. [Google Scholar]
- 144.Siegel J.D., Rhinehart E., Jackson M., Chiarello L. 2007 Guideline for isolation precautions: preventing transmission of infectious agents in health care settings. Am J Infect Control. 2007;35:S65–S164. doi: 10.1016/j.ajic.2007.10.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Hughes W.T., Armstrong D., Bodey G.P. 2002 Guidelines for the use of antimicrobial agents in neutropenic patients with cancer. Clin Infect Dis. 2002;34:730. doi: 10.1086/339215. [DOI] [PubMed] [Google Scholar]
- 146.Gafter-Gvili A., Fraser A., Paul M., Leibovici L. Meta-analysis: antibiotic prophylaxis reduces mortality in neutropenic patients. Ann Intern Med. 2005;142:979–995. doi: 10.7326/0003-4819-142-12_part_1-200506210-00008. [DOI] [PubMed] [Google Scholar]
- 147.van de Wetering M.D., de Witte M.A., Kremer L.C., Offringa M., Scholten R.J., Caron H.N. Efficacy of oral prophylactic antibiotics in neutropenic afebrile oncology patients: a systematic review of randomised controlled trials. Eur J Cancer. 2005;41:1372–1382. doi: 10.1016/j.ejca.2005.03.006. [DOI] [PubMed] [Google Scholar]
- 148.Bucaneve G., Castagnola E., Viscoli C., Leibovici L., Menichetti F. Quinolone prophylaxis for bacterial infections in afebrile high risk neutropenic patients. Eur J Cancer. 2007;(Suppl 5):5–12. [Google Scholar]
- 149.Bucaneve G., Micozzi A., Menichetti F. Levofloxacin to prevent bacterial infection in patients with cancer and neutropenia. N Engl J Med. 2005;353:977–987. doi: 10.1056/NEJMoa044097. [DOI] [PubMed] [Google Scholar]
- 150.Cruciani M., Rampazzo R., Malena M. Prophylaxis with fluoroquinolones for bacterial infections in neutropenic patients: a meta-analysis. Clin Infect Dis. 1996;23:795–805. doi: 10.1093/clinids/23.4.795. [DOI] [PubMed] [Google Scholar]
- 151.Cruciani M., Malena M., Bosco O., Nardi S., Serpelloni G., Mengoli C. Reappraisal with meta-analysis of the addition of Gram-positive prophylaxis to fluoroquinolone in neutropenic patients. J Clin Oncol. 2003;21:4127–4137. doi: 10.1200/JCO.2003.01.234. [DOI] [PubMed] [Google Scholar]
- 152.Engels E.A., Lau J., Barza M. Efficacy of quinolone prophylaxis in neutropenic cancer patients: a meta-analysis. J Clin Oncol. 1998;16:1179–1187. doi: 10.1200/JCO.1998.16.3.1179. [DOI] [PubMed] [Google Scholar]
- 153.The European Antimicrobial Resistance Surveillance System (EARSS) Annual Report, 2006.
- 154.MacDougall C., Powell J.P., Johnson C.K., Edmond M.B., Polk R.E. Hospital and community fluoroquinolone use and resistance in Staphylococcus aureus and Escherichia coli in 17 US hospitals. Clin Infect Dis. 2005;41:435–440. doi: 10.1086/432056. [DOI] [PubMed] [Google Scholar]
- 155.Bonadio M., Morelli G., Mori S., Riccioni R., Papineschi F., Petrini M. Fluoroquinolone resistance in hematopoietic stem cell transplant recipients with infectious complications. Biomed Pharmacother. 2005;59:511–516. doi: 10.1016/j.biopha.2005.06.008. [DOI] [PubMed] [Google Scholar]
- 156.Kirby J.T., Fritsche T.R., Jones R.N. Influence of patient age on the frequency of occurrence and antimicrobial resistance patterns of isolates from hematology/oncology patients: report from the Chemotherapy Alliance for Neutropenics and the Control of Emerging Resistance Program (North America) Diagn Microbiol Infect Dis. 2006;56:75–82. doi: 10.1016/j.diagmicrobio.2006.06.004. [DOI] [PubMed] [Google Scholar]
- 157.Blahova J., Kralikova K., Krcmery V.S. Four years of monitoring antibiotic resistance in microorganisms from bacteremic patients. J Chemother. 2007;19:665–669. doi: 10.1179/joc.2007.19.6.665. [DOI] [PubMed] [Google Scholar]
- 158.Robicsek A., Jacoby G.A., Hooper D.C. The worldwide emergence of plasmid-mediated quinolone resistance. Lancet Infect Dis. 2006;6:629–640. doi: 10.1016/S1473-3099(06)70599-0. [DOI] [PubMed] [Google Scholar]
- 159.Oktem I.M., Gulay Z., Bicmen M., Gur D. HITIT Project Study Group. qnrA prevalence in extended-spectrum beta-lactamase-positive Enterobacteriaceae isolates from Turkey. Jpn J Infect Dis. 2008;61:13–17. [PubMed] [Google Scholar]
- 160.Poirel L., Cattoir V., Nordmann P. Is plasmid-mediated quinolone resistance a clinically significant problem? Clin Microbiol Infect. 2008;14:295–297. doi: 10.1111/j.1469-0691.2007.01930.x. [DOI] [PubMed] [Google Scholar]
- 161.Loo V.G., Poirier L., Miller M.A. A predominantly clonal multi-institutional outbreak of Clostridium difficile-associated diarrhea with high morbidity and mortality. N Engl J Med. 2005;353:2442–2449. doi: 10.1056/NEJMoa051639. [DOI] [PubMed] [Google Scholar]
- 162.Vose J.M., Armitage J.O. Clinical applications of hematopoietic growth factors. J Clin Oncol. 1995;13:1023–1035. doi: 10.1200/JCO.1995.13.4.1023. [DOI] [PubMed] [Google Scholar]
- 163.Moreau P., Fiere D., Bezwoda W.R. Prospective randomized placebo-controlled study of granulocyte-macrophage colony-stimulating factor without stem-cell transplantation after high-dose melphalan in patients with multiple myeloma. J Clin Oncol. 1997;15:660–666. doi: 10.1200/JCO.1997.15.2.660. [DOI] [PubMed] [Google Scholar]
- 164.Dekker A., Bulley S., Beyene J., Dupuis L.L., Doyle J.J., Sung L. Meta-analysis of randomized controlled trials of prophylactic granulocyte colony-stimulating factor and granulocyte-macrophage colony-stimulating factor after autologous and allogeneic stem cell transplantation. J Clin Oncol. 2006;24:5207–5215. doi: 10.1200/JCO.2006.06.1663. [DOI] [PubMed] [Google Scholar]
- 165.Sung L., Nathan P.C., Alibhai S.M., Tomlinson G.A., Beyene J. Meta-analysis: effect of prophylactic hematopoietic colony-stimulating factors on mortality and outcomes of infection. Ann Intern Med. 2007;147:400–411. doi: 10.7326/0003-4819-147-6-200709180-00010. [DOI] [PubMed] [Google Scholar]
- 166.Beelen D.W., Elmaagacli A., Muller K.D., Hirche H., Schaefer U.W. Influence of intestinal bacterial decontamination using metronidazole and ciprofloxacin or ciprofloxacin alone on the development of acute graft-versus-host disease after marrow transplantation in patients with hematologic malignancies: final results and long-term follow-up of an open-label prospective randomized trial. Blood. 1999;93:3267–3275. [PubMed] [Google Scholar]
- 167.Rand K.J., Houck H., Ganju A., Babington R.G., Elfenbein G.J. Pharmacokinetics of cytomegalovirus specific IgG antibody following intravenous immunoglobulin in bone marrow transplant patients. Bone Marrow Transplant. 1989;4:679–683. [PubMed] [Google Scholar]
- 168.Bosi A., De Majo E., Guidi S. Kinetics of anti-CMV antibodies after administration of intravenous immunoglobulins to bone marrow transplant recipients. Haematologica. 1990;75:109–112. [PubMed] [Google Scholar]
- 169.Buckley R.H., Schiff R.I. Use of intravenous immune globulin in immunodeficiency diseases. N Engl J Med. 1991;325:110–117. doi: 10.1056/NEJM199107113250207. [DOI] [PubMed] [Google Scholar]
- 170.Bowden R.A., Myers J.D. Infection complicating bone marrow transplantation. In: Rubin R.H., Young L.S., editors. Clinical Approach to Infection in the Compromised Host. Plenum Medical Book Co; New York: 1994. pp. 601–628. [Google Scholar]
- 171.Sullivan K.M., Storek J., Kopecky K.J. A controlled trial of long-term administration of intravenous immunoglobulin to prevent late infection and chronic graft-vs.-host disease after marrow transplantation: clinical outcome and effect on subsequent immune recovery. Biol. Blood Marrow Transplant. 1996;2:44–53. [PubMed] [Google Scholar]
- 172.Wolff S.N., Fay J.W., Herzig R.H. High-dose weekly intravenous immunoglobulin to prevent infections in patients undergoing autologous bone marrow transplantation or severe myelosuppressive therapy. A study of the American Bone Marrow Transplant Group. Ann Intern Med. 1993;118:937–942. doi: 10.7326/0003-4819-118-12-199306150-00004. [DOI] [PubMed] [Google Scholar]
- 173.Yamasaki S., Heike Y., Mori S. Infectious complications in chronic graft-versus-host disease: a retrospective study of 145 recipients of allogeneic hematopoietic stem cell transplantation with reduced- and conventional-intensity conditioning regimens. Transpl Infect Dis. 2008;10:252–259. doi: 10.1111/j.1399-3062.2007.00291.x. [DOI] [PubMed] [Google Scholar]
- 174.Aksu G., Ruhi M.Z., Akan H. Aerobic bacterial and fungal infections in peripheral blood stem cell transplants. Bone Marrow Transplant. 2001;27:201–205. doi: 10.1038/sj.bmt.1702739. [DOI] [PubMed] [Google Scholar]
- 175.Kim D.H., Bae N.Y., Sung W.J. Hickman catheter site infections after allogeneic stem cell transplantation: single-center experience. Transplant Proc. 2004;36:3203–3207. doi: 10.1016/j.transproceed.2004.10.089. [DOI] [PubMed] [Google Scholar]
- 176.Adler A., Yaniv I., Solter E. Catheter-associated bloodstream infections in pediatric hematology-oncology patients: factors associated with catheter removal and recurrence. J Pediatr Hematol Oncol. 2006;28:23–28. [PubMed] [Google Scholar]
- 177.Raad I., Hohn D.C., Gilbreath B.J. Prevention of central venous catheter-related infections by using maximal sterile barrier precautions during insertion. Infect Control Hosp Epidemiol. 1994;15:231–238. [PubMed] [Google Scholar]
- 178.Pronovost P., Needham D., Berenholtz S. An intervention to decrease catheter-related bloodstream infections in the ICU. N Engl J Med. 2006;355:2725–2732. doi: 10.1056/NEJMoa061115. [DOI] [PubMed] [Google Scholar]
- 179.Hanna H., Benjamin R., Chatzinikolaou I. Long-term silicone central venous catheters impregnated with minocycline and rifampin decrease rates of catheter-related bloodstream infection in cancer patients: a prospective randomized clinical trial. J Clin Oncol. 2004;22:3163–3171. doi: 10.1200/JCO.2004.04.124. [DOI] [PubMed] [Google Scholar]
- 180.Darouiche R.O., Berger D.H., Khardori N. Comparison of antimicrobial impregnation with tunneling of long-term central venous catheters: a randomized controlled trial. Ann Surg. 2005;242:193–200. doi: 10.1097/01.sla.0000171874.29934.61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Chatzinikolaou I., Hanna H., Graviss L. Clinical experience with minocycline and rifampin-impregnated central venous catheters in bone marrow transplantation recipients: efficacy and low risk of developing staphylococcal resistance. Infect Control Hosp Epidemiol. 2003;24:961–963. doi: 10.1086/502167. [DOI] [PubMed] [Google Scholar]
- 182.O'Grady N.P., Alexander M., Dellinger E.P. Guidelines for the prevention of intravascular catheter-related infections. Centers for Disease Control and Prevention. MMWR Recomm Rep. 2002;51:1–29. [PubMed] [Google Scholar]
- 183.Gillies D., O'Riordan L., Carr D., Frost J., Gunning R., O'Brien I. Gauze and tape and transparent polyurethane dressings for central venous catheters. Cochrane Database Syst Rev. 2003 doi: 10.1002/14651858.CD003827. CD003827. [DOI] [PubMed] [Google Scholar]
- 184.Ruschulte H., Franke M., Gastmeier P. Prevention of central venous catheter related infections with chlorhexidine gluconate impregnated wound dressings: a randomized controlled trial. Ann Hematol. 2009;88:267–272. doi: 10.1007/s00277-008-0568-7. [DOI] [PubMed] [Google Scholar]
- 185.Timsit J.F., Schwebel C., Bouadma L. Chlorhexidine-impregnated sponges and less frequent dressing changes for prevention of catheter-related infections in critically ill adults: a randomized controlled trial. JAMA. 2009;301:1231–1241. doi: 10.1001/jama.2009.376. [DOI] [PubMed] [Google Scholar]
- 186.Safdar N., Maki D.G. Use of vancomycin-containing lock or flush solutions for prevention of bloodstream infection associated with central venous access devices: a meta-analysis of prospective, randomized trials. Clin Infect Dis. 2006;43:474–484. doi: 10.1086/505976. [DOI] [PubMed] [Google Scholar]
- 187.Raad I., Buzaid A., Rhyne J. Minocycline and ethylenediaminetetraacetate for the prevention of recurrent vascular catheter infections. Clin Infect Dis. 1997;25:149–151. doi: 10.1086/514518. [DOI] [PubMed] [Google Scholar]
- 188.Chatzinikolaou I., Zipf T.F., Hanna H. Minocycline-ethylenediaminetetraacetate lock solution for the prevention of implantable port infections in children with cancer. Clin Infect Dis. 2003;36:116–119. doi: 10.1086/344952. [DOI] [PubMed] [Google Scholar]
- 189.Betjes M.G., van Agteren M. Prevention of dialysis catheter-related sepsis with a citrate-taurolidine-containing lock solution. Nephrol Dial Transplant. 2004;19:1546–1551. doi: 10.1093/ndt/gfh014. [DOI] [PubMed] [Google Scholar]
- 190.Broom J., Woods M., Allworth A. Ethanol lock therapy to treat tunnelled central venous catheter-associated blood stream infections: results from a prospective trial. Scand J Infect Dis. 2008;40:399–406. doi: 10.1080/00365540701756953. [DOI] [PubMed] [Google Scholar]
- 191.Maki D.G., Kluger D.M., Crnich C.J. The risk of bloodstream infection in adults with different intravascular devices: a systematic review of 200 published prospective studies. Mayo Clin Proc. 2006;81:1159–1171. doi: 10.4065/81.9.1159. [DOI] [PubMed] [Google Scholar]
- 192.American Academy of Pediatrics/Committee on Infectious Diseases . Pneumococcal infections. In: Pickering LK, editor. 2006 Red Book: Report of the Committee on Infectious Diseases. American Academy of Pediatrics; Elk Grove Village, IL: 2006. pp. 525–537. [Google Scholar]
- 193.Engelhard D., Cordonnier C., Shaw P.J. Early and late invasive pneumococcal infection following stem cell transplantation: a European Bone Marrow Transplantation survey. Br J Haematol. 2002;117:444–450. doi: 10.1046/j.1365-2141.2002.03457.x. [DOI] [PubMed] [Google Scholar]
- 194.Patel S.R., Ortin M., Cohen B.J. Revaccination with measles, tetanus, poliovirus, Haemophilus influenzae type B, meningococcus C, and pneumococcus vaccines in children after hematopoietic stem cell transplantation. Clin Infect Dis. 2007;44:625–634. doi: 10.1086/511641. [DOI] [PubMed] [Google Scholar]
- 195.Cordonnier C, Labopin M, V C, et al. Influence of immunisation timing on the response to conjugate-pneumococcal vaccine after allogeneic stem cell transplant: final results of the EBMT IDWP01 Trial. Bone Marrow Transplant. 2008;45.
- 196.Tauro S., Dobie D., Richardson G., Hastings M., Mahendra P. Recurrent penicillin-resistant pneumococcal sepsis after matched unrelated donor (MUD) transplantation for refractory T cell lymphoma. Bone Marrow Transplant. 2000;26:1017–1019. doi: 10.1038/sj.bmt.1702647. [DOI] [PubMed] [Google Scholar]
- 197.Perez Retortillo J.A., Marco F., Richard C. Pneumococcal pericarditis with cardiac tamponade in a patient with chronic graft-versus-host disease. Bone Marrow Transplant. 1998;21:299–300. doi: 10.1038/sj.bmt.1701072. [DOI] [PubMed] [Google Scholar]
- 198.Haddad P.A., Repka T.L., Weisdorf D.J. Penicillin-resistant Streptococcus pneumoniae septic shock and meningitis complicating chronic graft versus host disease: a case report and review of the literature. Am J Med. 2002;113:152–155. doi: 10.1016/s0002-9343(02)01154-3. [DOI] [PubMed] [Google Scholar]
- 199.Jenkins S.G., Brown S.D., Farrell D.J. Trends in antibacterial resistance among Streptococcus pneumoniae isolated in the USA: update from PROTEKT US Years 1-4. Ann Clin Microbiol Antimicrob. 2008;7:1. doi: 10.1186/1476-0711-7-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Goldman K.E. Dental management of patients with bone marrow and solid organ transplantation. Dent Clin North Am. 2006;50:659–676. doi: 10.1016/j.cden.2006.06.009. viii. [DOI] [PubMed] [Google Scholar]
- 201.Prabhu R.M., Piper K.E., Litzow M.R., Steckelberg J.M., Patel R. Emergence of quinolone resistance among viridans group streptococci isolated from the oropharynx of neutropenic peripheral blood stem cell transplant patients receiving quinolone antimicrobial prophylaxis. Eur J Clin Microbiol Infect Dis. 2005;24:832–838. doi: 10.1007/s10096-005-0037-3. [DOI] [PubMed] [Google Scholar]
- 202.Alcaide F., Linares J., Pallares R. In vitro activities of 22 beta-lactam antibiotics against penicillin-resistant and penicillin-susceptible viridans group streptococci isolated from blood. Antimicrob Agents Chemother. 1995;39:2243–2247. doi: 10.1128/aac.39.10.2243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Steiner M., Villablanca J., Kersey J. Viridans streptococcal shock in bone marrow transplantation patients. Am J Hematol. 1993;42:354–358. doi: 10.1002/ajh.2830420405. [DOI] [PubMed] [Google Scholar]
- 204.American Academy of Pediatrics. Haemophilus influenzae infections. In: LK Pickering, ed. 2006 Red Book: Report of the Committee on Infectious Diseases. Elk Grove Village, IL: American Academy of Pediatrics, 2006:310–318.
- 205.Barra A., Cordonnier C., Preziosi M.P. Immunogenicity of Haemophilus influenzae type b conjugate vaccine in allogeneic bone marrow recipients. J Infect Dis. 1992;166:1021–1028. doi: 10.1093/infdis/166.5.1021. [DOI] [PubMed] [Google Scholar]
- 206.Sable C.A., Donowitz G.A. Infections in bone marrow transplant recipients. Clin Infect Dis. 1994;18:273–284. doi: 10.1093/clinids/18.3.273. [DOI] [PubMed] [Google Scholar]
- 207.Suzuki N., Mizue N., Hori T., Hatakeyama N., Kudoh T., Tsutsumi H. Pertussis in adolescence after unrelated cord blood transplantation. Bone Marrow Transplant. 2003;32:967. doi: 10.1038/sj.bmt.1704250. [DOI] [PubMed] [Google Scholar]
- 208.American Academy of Pediatrics/Committee on Infectious Diseases . Pertussis. In: Pickering L.K., editor. 2006 Red Book: Report of the Committee on Infectious Diseases. American Academy of Pediatrics; Elk Grove Village, IL: 2006. p. 502. [Google Scholar]
- 209.Nichols W.G., Price T.H., Gooley T., Corey L., Boeckh M. Transfusion-transmitted cytomegalovirus infection after receipt of leukoreduced blood products. Blood. 2003;101:4195–4200. doi: 10.1182/blood-2002-10-3143. [DOI] [PubMed] [Google Scholar]
- 210.Ljungman P., Larsson K., Kumlien G. Leukocyte depleted, unscreened blood products give a low risk for CMV infection and disease in CMV seronegative allogeneic stem cell transplant recipients with seronegative stem cell donors. Scand J Infect Dis. 2002;34:347–350. doi: 10.1080/00365540110080412. [DOI] [PubMed] [Google Scholar]
- 211.Bowden R., Cays M., Schoch G. Comparison of filtered blood (FB) to seronegative blood products (SB) for prevention of cytomegalovirus (CMV) infection after marrow transplant. Blood. 1995;86:3598–3603. [PubMed] [Google Scholar]
- 212.Blajchman M.A., Goldman M., Freedman J.J., Sher G.D. Proceedings of a consensus conference: prevention of post-transfusion CMV in the era of universal leukoreduction. Transfus Med Rev. 2001;15:1–20. doi: 10.1053/tmrv.2001.19946. [DOI] [PubMed] [Google Scholar]
- 213.Ratko T.A., Cummings J.P., Oberman H.A. Evidence-based recommendations for the use of WBC-reduced cellular blood components. Transfusion. 2001;41:1310–1319. doi: 10.1046/j.1537-2995.2001.41101310.x. [DOI] [PubMed] [Google Scholar]
- 214.Goodrich J.M., Bowden R.A., Fisher L., Keller C., Schoch G., Meyers J.D. Ganciclovir prophylaxis to prevent cytomegalovirus disease after allogeneic marrow transplant. Ann Intern Med. 1993;118:173–178. doi: 10.7326/0003-4819-118-3-199302010-00003. [DOI] [PubMed] [Google Scholar]
- 215.Prentice H.G., Gluckman E., Powles R.L. Impact of long-term acyclovir on cytomegalovirus infection and survival after allogeneic bone marrow transplantation. European Acyclovir for CMV Prophylaxis Study Group. Lancet. 1994;343:749–753. doi: 10.1016/s0140-6736(94)91835-x. [DOI] [PubMed] [Google Scholar]
- 216.Ljungman P., de La Camara R., Milpied N. Randomized study of valacyclovir as prophylaxis against cytomegalovirus reactivation in recipients of allogeneic bone marrow transplants. Blood. 2002;99:3050–3056. doi: 10.1182/blood.v99.8.3050. [DOI] [PubMed] [Google Scholar]
- 217.Boeckh M., Gooley T.A., Myerson D., Cunningham T., Schoch G., Bowden R.A. Cytomegalovirus pp65 antigenemia-guided early treatment with ganciclovir versus ganciclovir at engraftment after allogeneic marrow transplantation: a randomized double-blind study. Blood. 1996;88:4063–4071. [PubMed] [Google Scholar]
- 218.Winston D.J., Ho W.G., Bartoni K. Ganciclovir prophylaxis of cytomegalovirus infection and disease in allogeneic bone marrow transplant recipients. Results of a placebo-controlled, double-blind trial. Ann Intern Med. 1993;118:179–184. doi: 10.7326/0003-4819-118-3-199302010-00004. [DOI] [PubMed] [Google Scholar]
- 219.Fries B.C., Riddell S.R., Kim H.W. Cytomegalovirus disease before hematopoietic cell transplantation as a risk for complications after transplantation. Biol Blood Marrow Transplant. 2005;11:136–148. doi: 10.1016/j.bbmt.2004.11.016. [DOI] [PubMed] [Google Scholar]
- 220.Takami A., Mochizuki K., Asakura H., Yamazaki H., Okumura H., Nakao S. High incidence of cytomegalovirus reactivation in adult recipients of an unrelated cord blood transplant. Haematologica. 2005;90:1290–1292. [PubMed] [Google Scholar]
- 221.Matsumura T., Narimatsu H., Kami M. Cytomegalovirus infections following umbilical cord blood transplantation using reduced intensity conditioning regimens for adult patients. Biol Blood Marrow Transplant. 2007;13:577–583. doi: 10.1016/j.bbmt.2006.12.454. [DOI] [PubMed] [Google Scholar]
- 222.Boeckh M., Stevens-Ayers T., Bowden R.A. Cytomegalovirus pp65 antigenemia after autologous marrow and peripheral blood stem cell transplantation. J Infect Dis. 1996;174:907–912. doi: 10.1093/infdis/174.5.907. [DOI] [PubMed] [Google Scholar]
- 223.Boeckh M., Boivin G. Quantitation of cytomegalovirus: methodologic aspects and clinical applications. Clin Microbiol Rev. 1998;11:533–554. doi: 10.1128/cmr.11.3.533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Drew W.L. Laboratory diagnosis of cytomegalovirus infection and disease in immunocompromised patients. Curr Opin Infect Dis. 2007;20:408–411. doi: 10.1097/QCO.0b013e32821f6010. [DOI] [PubMed] [Google Scholar]
- 225.Lilleri D., Gerna G., Furione M. Use of a DNAemia cut-off for monitoring human cytomegalovirus infection reduces the number of pre-emptively treated children and young adults receiving haematopoietic stem cell transplantation as compared to qualitative pp65-antigenemia. Blood. 2007;110:2757–2760. doi: 10.1182/blood-2007-03-080820. [DOI] [PubMed] [Google Scholar]
- 226.Gerna G., Lilleri D., Baldanti F. Human cytomegalovirus immediate-early mRNAemia versus pp65 antigenemia for guiding pre-emptive therapy in children and young adults undergoing hematopoietic stem cell transplantation: a prospective, randomized, open-label trial. Blood. 2003;101:5053–5060. doi: 10.1182/blood-2002-12-3636. [DOI] [PubMed] [Google Scholar]
- 227.JC Mendez, IG Sia, CV Paya. Human cytomegalovirus. In: Lennette EH, ed. Laboratory Diagnosis of Viral Infections. New York: Marcel Decker, Inc., 1999:361–372.
- 228.Goodrich J.M., Mori M., Gleaves C.A. Early treatment with ganciclovir to prevent cytomegalovirus disease after allogeneic bone marrow transplantation. N Engl J Med. 1991;325:1601–1607. doi: 10.1056/NEJM199112053252303. [DOI] [PubMed] [Google Scholar]
- 229.Schmidt G.M., Horak D.A., Niland J.C., Duncan S.R., Forman S.J., Zaia J.A. A randomized, controlled trial of prophylactic ganciclovir for cytomegalovirus pulmonary infection in recipients of allogeneic bone marrow transplants; The City of Hope-Stanford-Syntex CMV Study Group. N Engl J Med. 1991;324:1005–1011. doi: 10.1056/NEJM199104113241501. [DOI] [PubMed] [Google Scholar]
- 230.Reusser P., Einsele H., Lee J. Randomized multicenter trial of foscarnet versus ganciclovir for preemptive therapy of cytomegalovirus infection after allogeneic stem cell transplantation. Blood. 2002;99:1159–1164. doi: 10.1182/blood.v99.4.1159. [DOI] [PubMed] [Google Scholar]
- 231.Ayala E., Greene J., Sandin R. Valganciclovir is safe and effective as pre-emptive therapy for CMV infection in allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant. 2006;37:851–856. doi: 10.1038/sj.bmt.1705341. [DOI] [PubMed] [Google Scholar]
- 232.Einsele H., Reusser P., Bornhauser M. Oral valganciclovir leads to higher exposure to ganciclovir than intravenous ganciclovir in patients following allogeneic stem cell transplantation. Blood. 2006;107:3002–3008. doi: 10.1182/blood-2005-09-3786. [DOI] [PubMed] [Google Scholar]
- 233.van der Heiden P.L., Kalpoe J.S., Barge R.M., Willemze R., Kroes A.C., Schippers E.F. Oral valganciclovir as pre-emptive therapy has similar efficacy on cytomegalovirus DNA load reduction as intravenous ganciclovir in allogeneic stem cell transplantation recipients. Bone Marrow Transplant. 2006;37:693–698. doi: 10.1038/sj.bmt.1705311. [DOI] [PubMed] [Google Scholar]
- 234.Busca A., de Fabritiis P., Ghisetti V. Oral valganciclovir as preemptive therapy for cytomegalovirus infection post allogeneic stem cell transplantation. Transpl Infect Dis. 2007;9:102–107. doi: 10.1111/j.1399-3062.2006.00183.x. [DOI] [PubMed] [Google Scholar]
- 235.Volin L., Barkholt L., Nihtinen A. An open-label randomised study of oral valganciclovir versus intravenous ganciclovir for pre-emptive therapy of cytomegalovirus infection after allogeneic stem cell transplantation [abstract] Bone Marrow Transplant. 2008;41(Suppl 1):47. [Google Scholar]
- 236.Moretti S., Zikos P., Van Lint M.T. Forscarnet vs ganciclovir for cytomegalovirus (CMV) antigenemia after allogeneic hemopoietic stem cell transplantation (HSCT): a randomised study. Bone Marrow Transplant. 1998;22:175–180. doi: 10.1038/sj.bmt.1701302. [DOI] [PubMed] [Google Scholar]
- 237.Boeckh M., Hoy C., Torok-Storb B. Occult cytomegalovirus infection of marrow stroma. Clin Infect Dis. 1998;26:209–210. doi: 10.1086/517022. [DOI] [PubMed] [Google Scholar]
- 238.Nichols W.G., Corey L., Gooley T. Rising pp65 antigenemia during preemptive anticytomegalovirus therapy after allogeneic hematopoietic stem cell transplantation: risk factors, correlation with DNA load, and outcomes. Blood. 2001;97:867–874. doi: 10.1182/blood.v97.4.867. [DOI] [PubMed] [Google Scholar]
- 239.Gerna G., Lilleri D., Zecca M. Rising antigenemia levels may be misleading in pre-emptive therapy of human cytomegalovirus infection in allogeneic hematopoietic stem cell transplant recipients. Haematologica. 2005;90:526–533. [PubMed] [Google Scholar]
- 240.Eckle T., Prix L., Jahn G. Drug-resistant human cytomegalovirus infection in children after allogeneic stem cell transplantation may have different clinical outcomes. Blood. 2000;96:3286–3289. [PubMed] [Google Scholar]
- 241.Cesaro S., Zhou X., Manzardo C. Cidofovir for cytomegalovirus reactivation in pediatric patients after hematopoietic stem cell transplantation. J Clin Virol. 2005;34:129–132. doi: 10.1016/j.jcv.2005.02.009. [DOI] [PubMed] [Google Scholar]
- 242.Platzbecker U., Bandt D., Thiede C. Successful preemptive cidofovir treatment for CMV antigenemia after dose-reduced conditioning and allogeneic blood stem cell transplantation. Transplantation. 2001;71:880–885. doi: 10.1097/00007890-200104150-00010. [DOI] [PubMed] [Google Scholar]
- 243.Ljungman P., Deliliers G.L., Platzbecker U. Cidofovir for cytomegalovirus infection and disease in allogeneic stem cell transplant recipients. The Infectious Diseases Working Party of the European Group for Blood and Marrow Transplantation. Blood. 2001;97:388–392. doi: 10.1182/blood.v97.2.388. [DOI] [PubMed] [Google Scholar]
- 244.Chou S. Cytomegalovirus UL97 mutations in the era of ganciclovir and maribavir. Rev Med Virol. 2008;18:233–246. doi: 10.1002/rmv.574. [DOI] [PubMed] [Google Scholar]
- 245.Holmberg L.A., Boeckh M., Hooper H. Increased incidence of cytomegalovirus disease after autologous CD34-selected peripheral blood stem cell transplantation. Blood. 1999;94:4029–4035. [PubMed] [Google Scholar]
- 246.Boeckh M., Gooley T.A., Reusser P., Buckner C.D., Bowden R.A. Failure of high-dose acyclovir to prevent cytomegalovirus disease after autologous marrow transplantation. J Infect Dis. 1995;172:939–943. doi: 10.1093/infdis/172.4.939. [DOI] [PubMed] [Google Scholar]
- 247.Peggs K.S., Preiser W., Kottaridis P.D. Extended routine polymerase chain reaction surveillance and pre-emptive antiviral therapy for cytomegalovirus after allogeneic transplantation. Br J Haematol. 2000;111:782–790. [PubMed] [Google Scholar]
- 248.Krause H., Hebart H., Jahn G., Muller C.A., Einsele H. Screening for CMV-specific T cell proliferation to identify patients at risk of developing late onset CMV disease. Bone Marrow Transplant. 1997;19:1111–1116. doi: 10.1038/sj.bmt.1700801. [DOI] [PubMed] [Google Scholar]
- 249.Gor D., Sabin C., Prentice H.G. Longitudinal fluctuations in cytomegalovirus load in bone marrow transplant patients: relationship between peak virus load, donor/recipient serostatus, acute GVHD and CMV disease. Bone Marrow Transplant. 1998;21:597–605. doi: 10.1038/sj.bmt.1701139. [DOI] [PubMed] [Google Scholar]
- 250.Zaia J.A., Gallez-Hawkins G.M., Tegtmeier B.R. Late cytomegalovirus disease in marrow transplantation is predicted by virus load in plasma. J Infect Dis. 1997;176 doi: 10.1086/517301. 782–725. [DOI] [PubMed] [Google Scholar]
- 251.Ljungman P., Aschan J., Azinge J.N. Cytomegalovirus viraemia and specific T-helper cell responses as predictors of disease after allogeneic marrow transplantation. Br J Haematol. 1993;83:118–124. doi: 10.1111/j.1365-2141.1993.tb04641.x. [DOI] [PubMed] [Google Scholar]
- 252.Walter E.A., Greenberg P.D., Gilbert M.J. Reconstitution of cellular immunity against cytomegalovirus in recipients of allogeneic bone marrow by transfer of T-cell clones from the donor. N Engl J Med. 1995;333:1038–1044. doi: 10.1056/NEJM199510193331603. [DOI] [PubMed] [Google Scholar]
- 253.Cobbold M., Khan N., Pourgheysari B. Adoptive transfer of cytomegalovirus-specific CTL to stem cell transplant patients after selection by HLA-peptide tetramers. J Exp Med. 2005;202:379–386. doi: 10.1084/jem.20040613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 254.Peggs K.S., Verfuerth S., Pizzey A. Adoptive cellular therapy for early cytomegalovirus infection after allogeneic stem-cell transplantation with virus-specific T-cell lines. Lancet. 2003;362:1375–1377. doi: 10.1016/S0140-6736(03)14634-X. [DOI] [PubMed] [Google Scholar]
- 255.Einsele H., Roosnek E., Rufer N. Infusion of cytomegalovirus (CMV)-specific T cells for the treatment of CMV infection not responding to antiviral chemotherapy. Blood. 2002;99:3916–3922. doi: 10.1182/blood.v99.11.3916. [DOI] [PubMed] [Google Scholar]
- 256.Buchsbaum R.J., Fabry J.A., Lieberman J. EBV-specific cytotoxic T lymphocytes protect against human EBV-associated lymphoma in scid mice. Immunol Lett. 1996;52:145–152. doi: 10.1016/0165-2478(96)02602-8. [DOI] [PubMed] [Google Scholar]
- 257.Shapiro R.S., McClain K., Frizzera G. Epstein-Barr virus associated B cell lymphoproliferative disorders following bone marrow transplantation. Blood. 1988;71:1234–1243. [PubMed] [Google Scholar]
- 258.Zutter M.M., Martin P.J., Sale G.E. Epstein-Barr virus lymphoproliferation after bone marrow transplantation. Blood. 1988;72:520–529. [PubMed] [Google Scholar]
- 259.Gerritsen E.J., Stam E.D., Hermans J. Risk factors for developing EBV-related B cell lymphoproliferative disorders (BLPD) after non-HLA-identical BMT in children. Bone Marrow Transplant. 1996;18:377–382. [PubMed] [Google Scholar]
- 260.Lucas K.G., Burton R.L., Zimmerman S.E. Semiquantitative Epstein-Barr virus (EBV) polymerase chain reaction for the determination of patients at risk for EBV-induced lymphoproliferative disease after stem cell transplantation. Blood. 1998;91:3654–3661. [PubMed] [Google Scholar]
- 261.Gustafsson A., Levitsky V., Zou J.Z. Epstein-Barr virus (EBV) load in bone marrow transplant recipients at risk to develop posttransplant lymphoproliferative disease: prophylactic infusion of EBV-specific cytotoxic T cells. Blood. 2000;95:807–814. [PubMed] [Google Scholar]
- 262.van Esser J.W., van der Holt B., Meijer E. Epstein-Barr virus (EBV) reactivation is a frequent event after allogeneic stem cell transplantation (SCT) and quantitatively predicts EBV-lymphoproliferative disease following T-cell-depleted SCT. Blood. 2001;98:972–978. doi: 10.1182/blood.v98.4.972. [DOI] [PubMed] [Google Scholar]
- 263.Meerbach A., Wutzler P., Hafer R., Zintl F., Gruhn B. Monitoring of Epstein-Barr virus load after hematopoietic stem cell transplantation for early intervention in post-transplant lymphoproliferative disease. J Med Virol. 2008;80:441–454. doi: 10.1002/jmv.21096. [DOI] [PubMed] [Google Scholar]
- 264.Papadopoulos E.B., Ladanyi M., Emanuel D. Infusions of donor leukocytes to treat Epstein-Barr virus-associated lymphoproliferative disorders after allogeneic bone marrow transplantation. N Engl J Med. 1994;330:1185–1191. doi: 10.1056/NEJM199404283301703. [DOI] [PubMed] [Google Scholar]
- 265.Rooney C.M., Smith C.A., Ng C.Y. Infusion of cytotoxic T cells for the prevention and treatment of Epstein-Barr virus-induced lymphoma in allogeneic transplant recipients. Blood. 1998;92:1549–1555. [PubMed] [Google Scholar]
- 266.Heslop H.E., Perez M., Benaim E., Rochester R., Brenner M.K., Rooney C.M. Transfer of EBV-specific CTL to prevent EBV lymphoma post bone marrow transplant. J Clin Apher. 1999;14:154–156. doi: 10.1002/(sici)1098-1101(1999)14:3<154::aid-jca9>3.0.co;2-k. [DOI] [PubMed] [Google Scholar]
- 267.Lucas K.G., Sun Q., Burton R.L. A phase I-II trial to examine the toxicity of CMV- and EBV-specific cytotoxic T lymphocytes when used for prophylaxis against EBV and CMV disease in recipients of CD34-selected/T cell-depleted stem cell transplants. Hum Gene Ther. 2000;11:1453–1463. doi: 10.1089/10430340050057521. [DOI] [PubMed] [Google Scholar]
- 268.Cavazzana-Calvo M., Bensoussan D., Jabado N. Prevention of EBV-induced B-lymphoproliferative disorder by ex vivo marrow B-cell depletion in HLA-phenoidentical or non-identical T-depleted bone marrow transplantation. Br J Haematol. 1998;103:543–551. doi: 10.1046/j.1365-2141.1998.00972.x. [DOI] [PubMed] [Google Scholar]
- 269.Saral R., Burns W.H., Laskin O.L., Santos G.W., Lietman P.S. Acyclovir prophylaxis of herpes-simplex-virus infections. N Engl J Med. 1981;305:63–67. doi: 10.1056/NEJM198107093050202. [DOI] [PubMed] [Google Scholar]
- 270.Gluckman E., Lotsberg J., Devergie A. Prophylaxis of herpes infections after bone-marrow transplantation by oral acyclovir. Lancet. 1983;2:706–708. doi: 10.1016/s0140-6736(83)92248-1. [DOI] [PubMed] [Google Scholar]
- 271.Wade J.C., Newton B., McLaren C., Flournoy N., Keeney R.E., Meyers J.D. Intravenous acyclovir to treat mucocutaneous herpes simplex virus infection after marrow transplantation: a double-blind trial. Ann Intern Med. 1982;96:265–269. doi: 10.7326/0003-4819-96-3-265. [DOI] [PubMed] [Google Scholar]
- 272.Wade J.C., Newton B., Flournoy N., Meyers J.D. Oral acyclovir for prevention of herpes simplex virus reactivation after marrow transplantation. Ann Intern Med. 1984;100:823–828. doi: 10.7326/0003-4819-100-6-823. [DOI] [PubMed] [Google Scholar]
- 273.Johnson J.R., Egaas S., Gleaves C.A., Hackman R., Bowden R.A. Hepatitis due to herpes simplex virus in marrow-transplant ecipients. Clin Infect Dis. 1992;14:38–45. doi: 10.1093/clinids/14.1.38. [DOI] [PubMed] [Google Scholar]
- 274.Erard V., Wald A., Corey L., Leisenring W.M., Boeckh M. Use of long-term suppressive acyclovir after hematopoietic stem-cell transplantation: impact on herpes simplex virus (HSV) disease and drug-resistant HSV disease. J Infect Dis. 2007;196:266–270. doi: 10.1086/518938. [DOI] [PubMed] [Google Scholar]
- 275.Smith K.O., Galloway K.S., Kennell W.L., Ogilvie K.K., Radatus B.K. A new nucleoside analog, 9-[[2-hydroxy-1-(hydroxymethyl)ethoxyl]methyl]guanine, highly active in vitro against herpes simplex virus types 1 and 2. Antimicrob Agents Chemother. 1982;22:55–61. doi: 10.1128/aac.22.1.55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 276.Langston A.A., Redei I., Caliendo A.M. Development of drug-resistant herpes simplex virus infection after haploidentical hematopoietic progenitor cell transplantation. Blood. 2002;99:1085–1088. doi: 10.1182/blood.v99.3.1085. [DOI] [PubMed] [Google Scholar]
- 277.Nichols W.G., Boeckh M., Carter R.A., Wald A., Corey L. Transferred herpes simplex virus immunity after stem-cell transplantation: clinical implications. J Infect Dis. 2003;187:801–808. doi: 10.1086/367894. [DOI] [PubMed] [Google Scholar]
- 278.Dignani M.C., Mykietiuk A., Michelet M. Valacyclovir prophylaxis for the prevention of Herpes simplex virus reactivation in recipients of progenitor cells transplantation. Bone Marrow Transplant. 2002;29:263–267. doi: 10.1038/sj.bmt.1703354. [DOI] [PubMed] [Google Scholar]
- 279.Eisen D., Essell J., Broun E.R., Sigmund D., DeVoe M. Clinical utility of oral valacyclovir compared with oral acyclovir for the prevention of herpes simplex virus mucositis following autologous bone marrow transplantation or stem cell rescue therapy. Bone Marrow Transplant. 2003;31:51–55. doi: 10.1038/sj.bmt.1703817. [DOI] [PubMed] [Google Scholar]
- 280.Erard V., Guthrie K.A., Varley C. One-year acyclovir prophylaxis for preventing varicella-zoster virus disease after hematopoietic cell transplantation: no evidence of rebound varicella-zoster virus disease after drug discontinuation. Blood. 2007;110:3071–3077. doi: 10.1182/blood-2007-03-077644. [DOI] [PubMed] [Google Scholar]
- 281.Dekker C.L., Prober C.G. Pediatric uses of valacyclovir, penciclovir and famciclovir. Pediatr Infect Dis J. 2001;20:1079–1081. doi: 10.1097/00006454-200111000-00014. [DOI] [PubMed] [Google Scholar]
- 282.Marin M., Guris D., Chaves S.S., Schmid S., Seward J.F. Prevention of varicella: recommendations of the Advisory Committee on Immunization Practices (ACIP) MMWR Recomm Rep. 2007;56:1–40. [PubMed] [Google Scholar]
- 283.Josephson A., Gombert M.E. Airborne transmission of nosocomial varicella from localized zoster. J Infect Dis. 1988;158:238–241. doi: 10.1093/infdis/158.1.238. [DOI] [PubMed] [Google Scholar]
- 284.Galea S.A., Sweet A., Beninger P. The safety profile of varicella vaccine: a 10-year review. J Infect Dis. 2008;(197 Suppl. 2):S165–S169. doi: 10.1086/522125. [DOI] [PubMed] [Google Scholar]
- 285.Oxman M.N., Levin M.J., Johnson G.R. A vaccine to prevent herpes zoster and postherpetic neuralgia in older adults. N Engl J Med. 2005;352:2271–2284. doi: 10.1056/NEJMoa051016. [DOI] [PubMed] [Google Scholar]
- 286.Centers for Disease Control and Prevention (CDC) A new product (VariZIG) for postexposure prophylaxis of varicella available under an investigational new drug application expanded access protocol. MMWR Morb Mortal Wkly Rep. 2006;55:209–210. [PubMed] [Google Scholar]
- 287.Boeckh M., Kim H.W., Flowers M.E., Meyers J.D., Bowden R.A. Long-term acyclovir for prevention of varicella zoster virus disease after allogeneic hematopoietic cell transplantation—a randomized double-blind placebo-controlled study. Blood. 2006;107:1800–1805. doi: 10.1182/blood-2005-09-3624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 288.Thomson K.J., Hart D.P., Banerjee L., Ward K.N., Peggs K.S., Mackinnon S. The effect of low-dose aciclovir on reactivation of varicella zoster virus after allogeneic haemopoietic stem cell transplantation. Bone Marrow Transplant. 2005;35:1065–1069. doi: 10.1038/sj.bmt.1704959. [DOI] [PubMed] [Google Scholar]
- 289.Hatchette T., Tipples G.A., Peters G., Alsuwaidi A., Zhou J., Mailman T.L. Foscarnet salvage therapy for acyclovir-resistant varicella zoster: report of a novel thymidine kinase mutation and review of the literature. Pediatr Infect Dis J. 2008;27:75–77. doi: 10.1097/INF.0b013e3181598315. [DOI] [PubMed] [Google Scholar]
- 290.Safrin S., Berger T.G., Gilson I. Foscarnet therapy in five patients with AIDS and acyclovir-resistant varicella-zoster virus infection. Ann Intern Med. 1991;115:19–21. doi: 10.7326/0003-4819-115-1-19. [DOI] [PubMed] [Google Scholar]
- 291.Weinstock D.M., Boeckh M., Sepkowitz K.A. Postexposure prophylaxis against varicella zoster virus infection among hematopoietic stem cell transplant recipients. Biol Blood Marrow Transplant. 2006;12:1096–1097. doi: 10.1016/j.bbmt.2006.06.005. [DOI] [PubMed] [Google Scholar]
- 292.Boeckh M. Prevention of VZV infection in immunosuppressed patients using antiviral agents. Herpes. 2006;13:60–65. [PubMed] [Google Scholar]
- 293.Adler A.L., Casper C., Boeckh M., Heath J., Zerr D.M. An outbreak of varicella with likely breakthrough disease in a population of pediatric cancer patients. Infect Control Hosp Epidemiol. 2008;29:866–870. doi: 10.1086/590358. [DOI] [PubMed] [Google Scholar]
- 294.Merck. Varivax: Varicella Vaccine Live, 2008.
- 295.Merck. Zostavax: Zoster Vaccine Live, 2007.
- 296.Hata A., Asanuma H., Rinki M. Use of an inactivated varicella vaccine in recipients of hematopoietic-cell transplants. N Engl J Med. 2002;347:26–34. doi: 10.1056/NEJMoa013441. [DOI] [PubMed] [Google Scholar]
- 297.Garcia R., Raad I., Abi-Said D. Nosocomial respiratory syncytial virus infections: prevention and control in bone marrow transplant patients. Infect Control Hosp Epidemiol. 1997;18:412–416. doi: 10.1086/647640. [DOI] [PubMed] [Google Scholar]
- 298.Raad I., Abbas J., Whimbey E. Infection control of nosocomial respiratory viral disease in the immunocompromised host. Am J Med. 1997;102:48–52. doi: 10.1016/s0002-9343(97)00011-9. discussion 53–54. [DOI] [PubMed] [Google Scholar]
- 299.Sable C.A., Hayden F.G. Orthomyxoviral and paramyxoviral infections in transplant patients. Infect Dis Clin North Am. 1995;9:987–1003. [PubMed] [Google Scholar]
- 300.Whimbey E., Champlin R.E., Couch R.B. Community respiratory virus infections among hospitalized adult bone marrow transplant recipients. Clin Infect Dis. 1996;22:778–782. doi: 10.1093/clinids/22.5.778. [DOI] [PubMed] [Google Scholar]
- 301.Whimbey E., Champlin R.E., Englund J.A. Combination therapy with aerosolized ribavirin and intravenous immunoglobulin for respiratory syncytial virus disease in adult bone marrow transplant recipients. Bone Marrow Transplant. 1995;16:393–399. [PubMed] [Google Scholar]
- 302.Folz R.J., Elkordy M.A. Coronavirus pneumonia following autologous bone marrow transplantation for breast cancer. Chest. 1999;115:901–905. doi: 10.1378/chest.115.3.901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 303.Fiore A.E., Shay D.K., Haber P. Prevention and control of influenza. Recommendations of the Advisory Committee on Immunization Practices (ACIP), 2007. MMWR Recomm Rep. 2007;56:1–54. [PubMed] [Google Scholar]
- 304.Centers for Disease Control and Prevention (CDC) Immunization of health care workers: recommendations of the Advisory Committee on Immunization Practices (ACIP) and the Hospital Infection Control Practices Advisory Committee. MMWR Morb Mortal Wkly Rep. 1997;46:1–42. [Google Scholar]
- 305.Hayden F.G. Prevention and treatment of influenza in immunocompromised patients. Am J Med. 1997;102:55–60. doi: 10.1016/s0002-9343(97)80013-7. discussion 75–76. [DOI] [PubMed] [Google Scholar]
- 306.Engelhard D., Nagler A., Hardan I. Antibody response to a two-dose regimen of influenza vaccine in allogeneic T cell-depleted and autologous BMT recipients. Bone Marrow Transplant. 1993;11:1–5. [PubMed] [Google Scholar]
- 307.Fiore A.E., Shay D.K., Broder K. Prevention and control of influenza. Recommendations of the Advisory Committee on Immunization Practices (ACIP), 2008. MMWR Recomm Rep. 2008;57:1–60. [PubMed] [Google Scholar]
- 308.American Academy of Pediatrics. Influenza. In: LK Pickering, ed. Red Book: 2006 Report of the Committee on Infectious Diseases. Elk Grove Village, IL: American Academy of Pediatrics, 2006:401–411.
- 309.Nichols W.G., Guthrie K.A., Corey L., Boeckh M. Influenza infections after hematopoietic stem cell transplantation: risk factors, mortality, and the effect of antiviral therapy. Clin Infect Dis. 2004;39:1300–1306. doi: 10.1086/425004. [DOI] [PubMed] [Google Scholar]
- 310.Chemaly R.F., Ghosh S., Bodey G.P. Respiratory viral infections in adults with hematologic malignancies and human stem cell transplantation recipients: a retrospective study at a major cancer center. Medicine (Baltimore) 2006;85:278–287. doi: 10.1097/01.md.0000232560.22098.4e. [DOI] [PubMed] [Google Scholar]
- 311.Martin M.A., Bock M.J., Pfaller M.A., Wenzel R.P. Respiratory syncytial virus infections in adult bone marrow transplant recipients. Lancet. 1988;1:1396–1397. doi: 10.1016/s0140-6736(88)92208-8. [DOI] [PubMed] [Google Scholar]
- 312.Hertz M.I., Englund J.A., Snover D., Bitterman P.B., McGlave P.B. Respiratory syncytial virus-induced acute lung injury in adult patients with bone marrow transplants: a clinical approach and review of the literature. Medicine (Baltimore) 1989;68:269–281. doi: 10.1097/00005792-198909000-00002. [DOI] [PubMed] [Google Scholar]
- 313.Boeckh M., Englund J., Li Y. Randomized controlled multicenter trial of aerosolized ribavirin for respiratory syncytial virus upper respiratory tract infection in hematopoietic cell transplant recipients. Clin Infect Dis. 2007;44:245–249. doi: 10.1086/509930. [DOI] [PubMed] [Google Scholar]
- 314.Khanna N., Widmer A.F., Decker M. Respiratory syncytial virus infection in patients with hematological diseases: single-center study and review of the literature. Clin Infect Dis. 2008;46:402–412. doi: 10.1086/525263. [DOI] [PubMed] [Google Scholar]
- 315.Sparrelid E., Ljungman P., Ekelof-Andstrom E. Ribavirin therapy in bone marrow transplant recipients with viral respiratory tract infections. Bone Marrow Transplant. 1997;19:905–908. doi: 10.1038/sj.bmt.1700752. [DOI] [PubMed] [Google Scholar]
- 316.Ljungman P., Ward K.N., Crooks B.N. Respiratory virus infections after stem cell transplantation: a prospective study from the Infectious Diseases Working Party of the European Group for Blood and Marrow Transplantation. Bone Marrow Transplant. 2001;28:479–484. doi: 10.1038/sj.bmt.1703139. [DOI] [PubMed] [Google Scholar]
- 317.De Vincenzo J.P., Leombruno D., Soiffer R.J., Siber G.R. Immunotherapy of respiratory syncytial virus pneumonia following bone marrow transplantation. Bone Marrow Transplant. 1996;17:1051–1056. [PubMed] [Google Scholar]
- 318.de Fontbrune F.S., Robin M., Porcher R. Palivizumab treatment of respiratory syncytial virus infection after allogeneic hematopoietic stem cell transplantation. Clin Infect Dis. 2007;45:1019–1024. doi: 10.1086/521912. [DOI] [PubMed] [Google Scholar]
- 319.American Academy of Pediatrics. Respiratory syncytial virus. In: LK Pickering, ed. Red Book: 2006 Report of the Committee on Infectious Diseases. Elk Grove Village, IL: American Academy of Pediatrics, 2006:560–566.
- 320.Englund J.A., Piedra P.A., Whimbey E. Prevention and treatment of respiratory syncytial virus and parainfluenza viruses in immunocompromised patients. Am J Med. 1997;102:61–70. doi: 10.1016/s0002-9343(97)00014-4. discussion 75–76. [DOI] [PubMed] [Google Scholar]
- 321.Englund J.A., Boeckh M., Kuypers J. Brief communication: fatal human metapneumovirus infection in stem-cell transplant recipients. Ann Intern Med. 2006;144:344–349. doi: 10.7326/0003-4819-144-5-200603070-00010. [DOI] [PubMed] [Google Scholar]
- 322.Wyde P.R., Chetty S.N., Jewell A.M., Boivin G., Piedra P.A. Comparison of the inhibition of human metapneumovirus and respiratory syncytial virus by ribavirin and immune serum globulin in vitro. Antiviral Res. 2003;60:51–59. doi: 10.1016/s0166-3542(03)00153-0. [DOI] [PubMed] [Google Scholar]
- 323.Hamelin M.E., Prince G.A., Boivin G. Effect of ribavirin and glucocorticoid treatment in a mouse model of human metapneumovirus infection. Antimicrob Agents Chemother. 2006;50:774–777. doi: 10.1128/AAC.50.2.774-777.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 324.American Academy of Pediatrics. Parainfluenza viral infections. In: LK Pickering, ed. Red Book: 2006 Report of the Committee on Infectious Diseases. Elk Grove Village, IL: American Academy of Pediatrics, 2006:479–481.
- 325.American Academy of Pediatrics. Adenovirus infections. In: LK Pickering, ed. Red Book: 2006 Report of the Committee on Infectious Diseases. Elk Grove Village, IL: American Academy of Pediatrics. 2006:202–204.
- 326.MS Horwitz. Adenoviruses. In: Knipe DM, Howley PM, ed. Fields Virology. Philadelphia, PA: Lippincott, Williams and Wilkins, 2001:2301–2326.
- 327.Flomenberg P., Piaskowski V., Truitt R.L., Casper J.T. Characterization of human proliferative T cell responses to adenovirus. J Infect Dis. 1995;171:1090–1096. doi: 10.1093/infdis/171.5.1090. [DOI] [PubMed] [Google Scholar]
- 328.La Rosa A.M., Champlin R.E., Mirza N. Adenovirus infections in adult recipients of blood and marrow transplants. Clin Infect Dis. 2001;32:871–876. doi: 10.1086/319352. [DOI] [PubMed] [Google Scholar]
- 329.Howard D.S., Phillips I.G., Reece D.E. Adenovirus infections in hematopoietic stem cell transplant recipients. Clin Infect Dis. 1999;29:1494–1501. doi: 10.1086/313514. [DOI] [PubMed] [Google Scholar]
- 330.Ison M.G. Adenovirus infections in transplant recipients. Clin Infect Dis. 2006;43:331–339. doi: 10.1086/505498. [DOI] [PubMed] [Google Scholar]
- 331.Chakrabarti S., Mautner V., Osman H. Adenovirus infections following allogeneic stem cell transplantation: incidence and outcome in relation to graft manipulation, immunosuppression, and immune recovery. Blood. 2002;100:1619–1627. doi: 10.1182/blood-2002-02-0377. [DOI] [PubMed] [Google Scholar]
- 332.Kampmann B., Cubitt D., Walls T. Improved outcome for children with disseminated adenoviral infection following allogeneic stem cell transplantation. Br J Haematol. 2005;130:595–603. doi: 10.1111/j.1365-2141.2005.05649.x. [DOI] [PubMed] [Google Scholar]
- 333.Lion T., Baumgartinger R., Watzinger F. Molecular monitoring of adenovirus in peripheral blood after allogeneic bone marrow transplantation permits early diagnosis of disseminated disease. Blood. 2003;102:1114–1120. doi: 10.1182/blood-2002-07-2152. [DOI] [PubMed] [Google Scholar]
- 334.Flomenberg P., Babbitt J., Drobyski W.R. Increasing incidence of adenovirus disease in bone marrow transplant recipients. J Infect Dis. 1994;169:775–781. doi: 10.1093/infdis/169.4.775. [DOI] [PubMed] [Google Scholar]
- 335.Shields A.F., Hackman R.C., Fife K.H., Corey L., Meyers J.D. Adenovirus infections in patients undergoing bone-marrow transplantation. N Engl J Med. 1985;312:529–533. doi: 10.1056/NEJM198502283120901. [DOI] [PubMed] [Google Scholar]
- 336.Myers G.D., Krance R.A., Weiss H. Adenovirus infection rates in pediatric recipients of alternate donor allogeneic bone marrow transplants receiving either antithymocyte globulin (ATG) or alemtuzumab (Campath) Bone Marrow Transplant. 2005;36:1001–1008. doi: 10.1038/sj.bmt.1705164. [DOI] [PubMed] [Google Scholar]
- 337.Erard V., Huang M.L., Ferrenberg J. Quantitative real-time polymerase chain reaction for detection of adenovirus after T cell-replete hematopoietic cell transplantation: viral load as a marker for invasive disease. Clin Infect Dis. 2007;45:958–965. doi: 10.1086/521851. [DOI] [PubMed] [Google Scholar]
- 338.Feuchtinger T., Lucke J., Hamprecht K. Detection of adenovirus-specific T cells in children with adenovirus infection after allogeneic stem cell transplantation. Br J Haematol. 2005;128:503–509. doi: 10.1111/j.1365-2141.2004.05331.x. [DOI] [PubMed] [Google Scholar]
- 339.Myers G.D., Bollard C.M., Wu M.F. Reconstitution of adenovirus-specific cell-mediated immunity in pediatric patients after hematopoietic stem cell transplantation. Bone Marrow Transplant. 2007;39:677–686. doi: 10.1038/sj.bmt.1705645. [DOI] [PubMed] [Google Scholar]
- 340.van Tol M.J., Kroes A.C., Schinkel J. Adenovirus infection in paediatric stem cell transplant recipients: increased risk in young children with a delayed immune recovery. Bone Marrow Transplant. 2005;36:39–50. doi: 10.1038/sj.bmt.1705003. [DOI] [PubMed] [Google Scholar]
- 341.Avivi I., Chakrabarti S., Milligan D.W. Incidence and outcome of adenovirus disease in transplant recipients after reduced-intensity conditioning with alemtuzumab. Biol Blood Marrow Transplant. 2004;10:186–194. doi: 10.1016/j.bbmt.2003.11.001. [DOI] [PubMed] [Google Scholar]
- 342.Neofytos D., Ojha A., Mookerjee B. Treatment of adenovirus disease in stem cell transplant recipients with cidofovir. Biol Blood Marrow Transplant. 2007;13:74–81. doi: 10.1016/j.bbmt.2006.08.040. [DOI] [PubMed] [Google Scholar]
- 343.Yusuf U., Hale G.A., Carr J. Cidofovir for the treatment of adenoviral infection in pediatric hematopoietic stem cell transplant patients. Transplantation. 2006;81:1398–1404. doi: 10.1097/01.tp.0000209195.95115.8e. [DOI] [PubMed] [Google Scholar]
- 344.Symeonidis N., Jakubowski A., Pierre-Louis S. Invasive adenoviral infections in T-cell-depleted allogeneic hematopoietic stem cell transplantation: high mortality in the era of cidofovir. Transpl Infect Dis. 2007;9:108–113. doi: 10.1111/j.1399-3062.2006.00184.x. [DOI] [PubMed] [Google Scholar]
- 345.Morfin F., Dupuis-Girod S., Mundweiler S. In vitro susceptibility of adenovirus to antiviral drugs is species-dependent. Antivir Ther. 2005;10:225–229. [PubMed] [Google Scholar]
- 346.Feuchtinger T., Matthes-Martin S., Richard C. Safe adoptive transfer of virus-specific T-cell immunity for the treatment of systemic adenovirus infection after allogeneic stem cell transplantation. Br J Haematol. 2006;134:64–76. doi: 10.1111/j.1365-2141.2006.06108.x. [DOI] [PubMed] [Google Scholar]
- 347.Hirsch H.H., Steiger J., Polyomavirus B.K. Lancet Infect Dis. 2003;3:611–623. doi: 10.1016/s1473-3099(03)00770-9. [DOI] [PubMed] [Google Scholar]
- 348.Knowles W.A. Discovery and epidemiology of the human polyomaviruses BK virus (BKV) and JC virus (JCV) Adv Exp Med Biol. 2006;577:19–45. doi: 10.1007/0-387-32957-9_2. [DOI] [PubMed] [Google Scholar]
- 349.Egli A., Infanti L., Stebler C., Bodaghi S., Gosert R., Hirsch H.H., Polyomavirus B.K. (BKV) and JC (JCV) replication in plasma and urine in healthy blood donors (Abstract #1058) Am J Transplant. 2008;8(S2):460. [Google Scholar]
- 350.Bofill-Mas S., Formiga-Cruz M., Clemente-Casares P., Calafell F., Girones R. Potential transmission of human polyomaviruses through the gastrointestinal tract after exposure to virions or viral DNA. J Virol. 2001;75:10290–10299. doi: 10.1128/JVI.75.21.10290-10299.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 351.Knowles W.A., Pillay D., Johnson M.A., Hand J.F., Brown D.W. Prevalence of long-term BK and JC excretion in HIV-infected adults and lack of correlation with serological markers. J Med Virol. 1999;59:474–479. [PubMed] [Google Scholar]
- 352.Arthur R.R., Shah K.V., Baust S.J., Santos G.W., Saral R. Association of BK viruria with hemorrhagic cystitis in recipients of bone marrow transplants. N Engl J Med. 1986;315:230–234. doi: 10.1056/NEJM198607243150405. [DOI] [PubMed] [Google Scholar]
- 353.Arthur R.R., Shah K.V., Charache P., Saral R. BK and JC virus infections in recipients of bone marrow transplants. J Infect Dis. 1988;158:563–569. doi: 10.1093/infdis/158.3.563. [DOI] [PubMed] [Google Scholar]
- 354.Bedi A., Miller C.B., Hanson J.L. Association of BK virus with failure of prophylaxis against hemorrhagic cystitis following bone marrow transplantation. J Clin Oncol. 1995;13:1103–1109. doi: 10.1200/JCO.1995.13.5.1103. [DOI] [PubMed] [Google Scholar]
- 355.Gorczynska E., Turkiewicz D., Rybka K. Incidence, clinical outcome, and management of virus-induced hemorrhagic cystitis in children and adolescents after allogeneic hematopoietic cell transplantation. Biol Blood Marrow Transplant. 2005;11:797–804. doi: 10.1016/j.bbmt.2005.06.007. [DOI] [PubMed] [Google Scholar]
- 356.Hirsch H.H., Knowles W., Dickenmann M. Prospective study of polyomavirus type BK replication and nephropathy in renal-transplant recipients. N Engl J Med. 2002;347:488–496. doi: 10.1056/NEJMoa020439. [DOI] [PubMed] [Google Scholar]
- 357.Stracke S., Helmchen U., von Muller L., Bunjes D., Keller F. Polyoma virus-associated interstitial nephritis in a patient with acute myeloic leukaemia and peripheral blood stem cell transplantation. Nephrol Dial Transplant. 2003;18:2431–2433. doi: 10.1093/ndt/gfg361. [DOI] [PubMed] [Google Scholar]
- 358.Limaye A.P., Smith K.D., Cook L. Polyomavirus nephropathy in native kidneys of non-renal transplant recipients. Am J Transplant. 2005;5:614–620. doi: 10.1046/j.1600-6143.2003.00209.x. [DOI] [PubMed] [Google Scholar]
- 359.Drachenberg C.B., Hirsch H.H., Papadimitriou J.C. Polyomavirus BK versus JC replication and nephropathy in renal transplant recipients: a prospective evaluation. Transplantation. 2007;84:323–330. doi: 10.1097/01.tp.0000269706.59977.a5. [DOI] [PubMed] [Google Scholar]
- 360.Kharfan-Dabaja M.A., Ayala E., Greene J., Rojiani A., Murtagh F.R., Anasetti C. Two cases of progressive multifocal leukoencephalopathy after allogeneic hematopoietic cell transplantation and a review of the literature. Bone Marrow Transplant. 2007;39:101–107. doi: 10.1038/sj.bmt.1705548. [DOI] [PubMed] [Google Scholar]
- 361.Hix J.K., Braun W.E., Isada C.M. Delirium in a renal transplant recipient associated with BK virus in the cerebrospinal fluid. Transplantation. 2004;78:1407–1408. doi: 10.1097/01.tp.0000137106.09925.8b. [DOI] [PubMed] [Google Scholar]
- 362.Rinaldo C.H., Hirsch H.H. Antivirals for the treatment of polyomavirus BK replication. Expert Rev Antiinfect Ther. 2007;5:105–115. doi: 10.1586/14787210.5.1.105. [DOI] [PubMed] [Google Scholar]
- 363.Leung A.Y., Chan M.T., Yuen K.Y. Ciprofloxacin decreased polyoma BK virus load in patients who underwent allogeneic hematopoietic stem cell transplantation. Clin Infect Dis. 2005;40:528–537. doi: 10.1086/427291. [DOI] [PubMed] [Google Scholar]
- 364.Hui C.K., Lie A., Au W.Y. A long-term follow-up study on hepatitis B surface antigen-positive patients undergoing allogeneic hematopoietic stem cell transplantation. Blood. 2005;106:464–469. doi: 10.1182/blood-2005-02-0698. [DOI] [PubMed] [Google Scholar]
- 365.Carpenter P.A., Huang M.L., McDonald G.B. Activation of occult hepatitis B from a seronegative patient after hematopoietic cell transplant: a cautionary tale. Blood. 2002;99:4245–4246. doi: 10.1182/blood-2001-12-0239. [DOI] [PubMed] [Google Scholar]
- 366.Tur-Kaspa R., Shaul Y., Moore D.D. The glucocorticoid receptor recognizes a specific nucleotide sequence in hepatitis B virus DNA causing increased activity of the HBV enhancer. Virology. 1988;167:630–633. [PubMed] [Google Scholar]
- 367.Tur-Kaspa R., Burk R.D., Shaul Y., Shafritz D.A. Hepatitis B virus DNA contains a glucocorticoid-responsive element. Proc Natl Acad Sci USA. 1986;83:1627–1631. doi: 10.1073/pnas.83.6.1627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 368.Aksoy S., Harputluoglu H., Kilickap S. Rituximab-related viral infections in lymphoma patients. Leuk Lymphoma. 2007;48:1307–1312. doi: 10.1080/10428190701411441. [DOI] [PubMed] [Google Scholar]
- 369.Moses S.E., Lim Z.Y., Sudhanva M. Lamivudine prophylaxis and treatment of hepatitis B Virus-exposed recipients receiving reduced intensity conditioning hematopoietic stem cell transplants with alemtuzumab. J Med Virol. 2006;78:1560–1563. doi: 10.1002/jmv.20705. [DOI] [PubMed] [Google Scholar]
- 370.Hui C.K., Yu J., Au W.Y. Sexual transmission of hepatitis B infection despite the presence of hepatitis B virus immunity in recipients of allogeneic bone marrow transplantation. J Clin Virol. 2005;32:173–178. doi: 10.1016/j.jcv.2004.08.003. [DOI] [PubMed] [Google Scholar]
- 371.Goyama S., Kanda Y., Nannya Y. Reverse seroconversion of hepatitis B virus after hematopoietic stem cell transplantation. Leuk Lymphoma. 2002;43:2159–2163. doi: 10.1080/1042819021000033042. [DOI] [PubMed] [Google Scholar]
- 372.Lau G.K., Lie A., Liang R. Prophylactic lamivudine therapy for hepatitis B patients undergoing immunosuppressive therapy. Blood. 2002;100:3054. doi: 10.1182/blood-2002-07-1976. [DOI] [PubMed] [Google Scholar]
- 373.Lau G.K., Leung Y.H., Fong D.Y. High hepatitis B virus (HBV) DNA viral load as the most important risk factor for HBV reactivation in patients positive for HBV surface antigen undergoing autologous hematopoietic cell transplantation. Blood. 2002;99:2324–2330. doi: 10.1182/blood.v99.7.2324. [DOI] [PubMed] [Google Scholar]
- 374.Lau G.K., He M.L., Fong D.Y. Preemptive use of lamivudine reduces hepatitis B exacerbation after allogeneic hematopoietic cell transplantation. Hepatology. 2002;36:702–709. doi: 10.1053/jhep.2002.35068. [DOI] [PubMed] [Google Scholar]
- 375.Hsiao L.T., Chiou T.J., Liu J.H. Extended lamivudine therapy against hepatitis B virus infection in hematopoietic stem cell transplant recipients. Biol Blood Marrow Transplant. 2006;12:84–94. doi: 10.1016/j.bbmt.2005.09.001. [DOI] [PubMed] [Google Scholar]
- 376.Lin P.C., Poh S.B., Lee M.Y., Hsiao L.T., Chen P.M., Chiou T.J. Fatal fulminant hepatitis B after withdrawal of prophylactic lamivudine in hematopoietic stem cell transplantation patients. Int J Hematol. 2005;81:349–351. doi: 10.1532/IJH97.A10411. [DOI] [PubMed] [Google Scholar]
- 377.Shuhart M.C., Myerson D., Childs B.H. Marrow transplantation from hepatitis C virus seropositive donors: transmission rate and clinical course. Blood. 1994;84:3229–3235. [PubMed] [Google Scholar]
- 378.Strasser S.I., Myerson D., Spurgeon C.L. Hepatitis C virus infection and bone marrow transplantation: a cohort study with 10-year follow-up. Hepatology. 1999;29:1893–1899. doi: 10.1002/hep.510290609. [DOI] [PubMed] [Google Scholar]
- 379.Peffault dL, Levy V., Asselah T. Long-term outcome of hepatitis C infection after bone marrow transplantation. Blood. 2004;103:1618–1624. doi: 10.1182/blood-2003-06-2145. [DOI] [PubMed] [Google Scholar]
- 380.Locasciulli A., Alberti A. Hepatitis C virus serum markers and liver disease in children with leukemia. Leuk Lymphoma. 1995;17:245–249. doi: 10.3109/10428199509056828. [DOI] [PubMed] [Google Scholar]
- 381.Vance E.A., Soiffer R.J., McDonald G.B., Myerson D., Fingeroth J., Ritz J. Prevention of transmission of hepatitis C virus in bone marrow transplantation by treating the donor with alpha-interferon. Transplantation. 1996;62:1358–1360. doi: 10.1097/00007890-199611150-00032. [DOI] [PubMed] [Google Scholar]
- 382.Surapaneni S.N., Hari P., Knox J., Daniel J., Saeian K. Suppressive anti-HCV therapy for prevention of donor to recipient transmission in stem cell transplantation. Am J Gastroenterol. 2007;102:449–451. doi: 10.1111/j.1572-0241.2006.00937.x. [DOI] [PubMed] [Google Scholar]
- 383.McDonald G.B. Review article: management of hepatic disease following haematopoietic cell transplant. Aliment Pharmacol Ther. 2006;24:441–452. doi: 10.1111/j.1365-2036.2006.03001.x. [DOI] [PubMed] [Google Scholar]
- 384.Hogan W.J., Maris M., Storer B. Hepatic injury after nonmyeloablative conditioning followed by allogeneic hematopoietic cell transplantation: a study of 193 patients. Blood. 2004;103:78–84. doi: 10.1182/blood-2003-04-1311. [DOI] [PubMed] [Google Scholar]
- 385.Peffault dL, Asselah T., Levy V. Treatment of chronic hepatitis C virus in allogeneic bone marrow transplant recipients. Bone Marrow Transplant. 2005;36:709–713. doi: 10.1038/sj.bmt.1705120. [DOI] [PubMed] [Google Scholar]
- 386.Yamanishi K., Okuno T., Shiraki K. Identification of human herpesvirus-6 as a causal agent for exanthem subitum. Lancet. 1988;1:1065–1067. doi: 10.1016/s0140-6736(88)91893-4. [DOI] [PubMed] [Google Scholar]
- 387.Zerr D.M., Corey L., Kim H.W., Huang M.L., Nguy L., Boeckh M. Clinical outcomes of human herpesvirus 6 reactivation after hematopoietic stem cell transplantation. Clin Infect Dis. 2005;40:932–940. doi: 10.1086/428060. [DOI] [PubMed] [Google Scholar]
- 388.Zerr D.M., Meier A.S., Selke S.S. A population-based study of primary human herpesvirus 6 infection. N Engl J Med. 2005;352:768–776. doi: 10.1056/NEJMoa042207. [DOI] [PubMed] [Google Scholar]
- 389.Cone R.W., Huang M.L., Ashley R., Corey L. Human herpesvirus 6 DNA in peripheral blood cells and saliva from immunocompetent individuals. J Clin Microbiol. 1993;31:1262–1267. doi: 10.1128/jcm.31.5.1262-1267.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 390.Yoshikawa T., Asano Y., Kobayashi I. Seroepidemiology of human herpesvirus 7 in healthy children and adults in Japan. J Med Virol. 1993;41:319–323. doi: 10.1002/jmv.1890410412. [DOI] [PubMed] [Google Scholar]
- 391.Imbert-Marcille B.M., Tang X.W., Lepelletier D. Human herpesvirus 6 infection after autologous or allogeneic stem cell transplantation: a single-center prospective longitudinal study of 92 patients. Clin Infect Dis. 2000;31:881–886. doi: 10.1086/318142. [DOI] [PubMed] [Google Scholar]
- 392.Ogata M., Kikuchi H., Satou T. Human herpesvirus 6 DNA in plasma after allogeneic stem cell transplantation: incidence and clinical significance. J Infect Dis. 2006;193:68–79. doi: 10.1086/498531. [DOI] [PubMed] [Google Scholar]
- 393.Sashihara J., Tanaka-Taya K., Tanaka S. High incidence of human herpesvirus 6 infection with a high viral load in cord blood stem cell transplant recipients. Blood. 2002;100:2005–2011. [PubMed] [Google Scholar]
- 394.Wang L.R., Dong L.J., Lu D.P. Surveillance of active human herpesvirus 6 infection in Chinese patients after hematopoietic stem cell transplantation with 3 different methods. Int J Hematol. 2006;84:262–267. doi: 10.1532/IJH97.A10607. [DOI] [PubMed] [Google Scholar]
- 395.Ward K.N., Leong H.N., Nacheva E.P. Human herpesvirus 6 chromosomal integration in immunocompetent patients results in high levels of viral DNA in blood, sera, and hair follicles. J Clin Microbiol. 2006;44:1571–1574. doi: 10.1128/JCM.44.4.1571-1574.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 396.Seeley W.W., Marty F.M., Holmes T.M. Post-transplant acute limbic encephalitis: clinical features and relationship to HHV6. Neurology. 2007;69:156–165. doi: 10.1212/01.wnl.0000265591.10200.d7. [DOI] [PubMed] [Google Scholar]
- 397.Gorniak R.J., Young G.S., Wiese D.E., Marty F.M., Schwartz R.B. MR imaging of human herpesvirus-6-associated encephalitis in 4 patients with anterograde amnesia after allogeneic hematopoietic stem-cell transplantation. AJNR. 2006;27:887–891. [PMC free article] [PubMed] [Google Scholar]
- 398.Glosson N.L., Hudson A.W. Human herpesvirus-6A and -6B encode viral immunoevasins that downregulate class I MHC molecules. Virology. 2007;365:125–135. doi: 10.1016/j.virol.2007.03.048. [DOI] [PubMed] [Google Scholar]
- 399.Zerr D.M., Gupta D., Huang M.L., Carter R., Corey L. Effect of antivirals on human herpesvirus 6 replication in hematopoietic stem cell transplant recipients. Clin Infect Dis. 2002;34:309–317. doi: 10.1086/338044. [DOI] [PubMed] [Google Scholar]
- 400.De Bolle L., Naesens L., De Clercq E. Update on human herpesvirus 6 biology, clinical features, and therapy. Clin Microbiol Rev. 2005;18:217–245. doi: 10.1128/CMR.18.1.217-245.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 401.de Medeiros B.C., Rezuke W.N., Ricci A., Jr. Kaposi's sarcoma following allogeneic hematopoietic stem cell transplantation for chronic myelogenous leukemia. Acta Haematol. 2000;104:115–118. doi: 10.1159/000039743. [DOI] [PubMed] [Google Scholar]
- 402.Bruno B., Sorasio R., Barozzi P. Kaposi's sarcoma triggered by endogenous HHV-8 reactivation after non-myeloablative allogeneic haematopoietic transplantation. Eur J Haematol. 2006;76 doi: 10.1111/j.1600-0609.2005.00601.x. 342–327. [DOI] [PubMed] [Google Scholar]
- 403.Gentile G., Capobianchi A., Volpi A. Human herpesvirus 8 DNA in serum during seroconversion in allogeneic bone marrow transplant recipients. J Natl Cancer Inst. 2005;97:1008–1011. doi: 10.1093/jnci/dji177. [DOI] [PubMed] [Google Scholar]
- 404.Cuzzola M., Irrera G., Iacopino O. Bone marrow failure associated with herpesvirus 8 infection in a patient undergoing autologous peripheral blood stem cell transplantation. Clin Infect Dis. 2003;37:e102–e106. doi: 10.1086/377268. [DOI] [PubMed] [Google Scholar]
- 405.Bonnet F., Lewden C., May T. Malignancy-related causes of death in human immunodeficiency virus-infected patients in the era of highly active antiretroviral therapy. Cancer. 2004;101:317–324. doi: 10.1002/cncr.20354. [DOI] [PubMed] [Google Scholar]
- 406.Spitzer T.R., Ambinder R.F., Lee J.Y. Dose-reduced busulfan, cyclophosphamide, and autologous stem cell transplantation for human immunodeficiency virus-associated lymphoma: AIDS Malignancy Consortium study 020. Biol Blood Marrow Transplant. 2008;14:59–66. doi: 10.1016/j.bbmt.2007.03.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 407.Gabarre J., Azar N., Autran B., Katlama C., Leblond V. High-dose therapy and autologous haematopoietic stem-cell transplantation for HIV-1-associated lymphoma. Lancet. 2000;355:1071–1072. doi: 10.1016/S0140-6736(00)02041-9. [DOI] [PubMed] [Google Scholar]
- 408.Molina A., Krishnan A.Y., Nademanee A. High dose therapy and autologous stem cell transplantation for human immunodeficiency virus-associated non-Hodgkin lymphoma in the era of highly active antiretroviral therapy. Cancer. 2000;89:680–689. [PubMed] [Google Scholar]
- 409.Krishnan A., Molina A., Zaia J. Autologous stem cell transplantation for HIV-associated lymphoma. Blood. 2001;98:3857–3859. doi: 10.1182/blood.v98.13.3857. [DOI] [PubMed] [Google Scholar]
- 410.Re A., Cattaneo C., Michieli M. High-dose therapy and autologous peripheral-blood stem-cell transplantation as salvage treatment for HIV-associated lymphoma in patients receiving highly active antiretroviral therapy. J Clin Oncol. 2003;21:4423–4427. doi: 10.1200/JCO.2003.06.039. [DOI] [PubMed] [Google Scholar]
- 411.Gupta V., Tomblyn M., Pederson T. Allogeneic hematopoietic stem cell transplantation in HIV-positive patients with hematologic disorders: a report from the center for international blood and marrow transplant research (CIBMTR) Biol Blood Marrow Transplant. 2009;15(7):864–871. doi: 10.1016/j.bbmt.2009.03.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 412.American Society of Clinical Oncology Recommendations for the use of hematopoietic colony-stimulating factors: evidence-based, clinical practice guidelines. J Clin Oncol. 1994;12:2471–2508. doi: 10.1200/JCO.1994.12.11.2471. [DOI] [PubMed] [Google Scholar]
- 413.Riley D.K., Pavia A.T., Beatty P.G., Denton D., Carroll K.C. Surveillance cultures in bone marrow transplant recipients: worthwhile or wasteful? Bone Marrow Transplant. 1995;15:469–473. [PubMed] [Google Scholar]
- 414.Walsh T.J. Role of surveillance cultures in prevention and treatment of fungal infections. NCI Monogr. 1990:43–45. [PubMed] [Google Scholar]
- 415.Trifilio S., Singhal S., Williams S. Breakthrough fungal infections after allogeneic hematopoietic stem cell transplantation in patients on prophylactic voriconazole. Bone Marrow Transplant. 2007;40:451–456. doi: 10.1038/sj.bmt.1705754. [DOI] [PubMed] [Google Scholar]
- 416.Barnes P.D., Marr K.A. Risks, diagnosis and outcomes of invasive fungal infections in haematopoietic stem cell transplant recipients. Br J Haematol. 2007;139:519–531. doi: 10.1111/j.1365-2141.2007.06812.x. [DOI] [PubMed] [Google Scholar]
- 417.Nucci M., Anaissie E. Revisiting the source of candidemia: skin or gut? Clin Infect Dis. 2001;33:1959–1967. doi: 10.1086/323759. [DOI] [PubMed] [Google Scholar]
- 418.Goodman J.L., Winston D.J., Greenfield R.A. A controlled trial of fluconazole to prevent fungal infections in patients undergoing bone marrow transplantation. N Engl J Med. 1992;326:845–851. doi: 10.1056/NEJM199203263261301. [DOI] [PubMed] [Google Scholar]
- 419.Slavin M.A., Osborne B., Adams R. Efficacy and safety of fluconazole prophylaxis for fungal infections after marrow transplantation—a prospective, randomized, double-blind study. J Infect Dis. 1995;171:1545–1552. doi: 10.1093/infdis/171.6.1545. [DOI] [PubMed] [Google Scholar]
- 420.Marr K.A., Seidel K., Slavin M.A. Prolonged fluconazole prophylaxis is associated with persistent protection against candidiasis-related death in allogeneic marrow transplant recipients: long-term follow-up of a randomized, placebo-controlled trial. Blood. 2000;96:2055–2061. [PubMed] [Google Scholar]
- 421.Pfaller M.A., Diekema D.J., Gibbs D.L. Results from the ARTEMIS DISK Global Antifungal Surveillance study, 1997 to 2005: an 8.5-year analysis of susceptibilities of Candida species and other yeast species to fluconazole and voriconazole determined by CLSI standardized disk diffusion testing. J Clin Microbiol. 2007;45:1735–1745. doi: 10.1128/JCM.00409-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 422.Marr K.A., Seidel K., White T.C., Bowden R.A. Candidemia in allogeneic blood and marrow transplant recipients: evolution of risk factors after the adoption of prophylactic fluconazole. J Infect Dis. 2000;181:309–316. doi: 10.1086/315193. [DOI] [PubMed] [Google Scholar]
- 423.Pfaller M.A., Messer S.A., Boyken L., Tendolkar S., Hollis R.J., Diekema D.J. Geographic variation in the susceptibilities of invasive isolates of Candida glabrata to 7 systemically active antifungal agents: a global assessment from the ARTEMIS Antifungal Surveillance Program conducted in 2001 and 2002. J Clin Microbiol. 2004;42:3142–3146. doi: 10.1128/JCM.42.7.3142-3146.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 424.Tsai H.F., Bard M., Izumikawa K. Candida glabrata erg1 mutant with increased sensitivity to azoles and to low oxygen tension. Antimicrob Agents Chemother. 2004;48:2483–2489. doi: 10.1128/AAC.48.7.2483-2489.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 425.van Burik J.A., Ratanatharathorn V., Stepan D.E. Micafungin versus fluconazole for prophylaxis against invasive fungal infections during neutropenia in patients undergoing hematopoietic stem cell transplantation. Clin Infect Dis. 2004;39:1407–1416. doi: 10.1086/422312. [DOI] [PubMed] [Google Scholar]
- 426.Perlin D.S. Resistance to echinocandin-class antifungal drugs. Drug Resist Update. 2007;10:121–130. doi: 10.1016/j.drup.2007.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 427.Winston D.J., Maziarz R.T., Chandrasekar P.H. Intravenous and oral itraconazole versus intravenous and oral fluconazole for long-term antifungal prophylaxis in allogeneic hematopoietic stem-cell transplant recipients. A multicenter, randomized trial. Ann Intern Med. 2003;138:705–713. doi: 10.7326/0003-4819-138-9-200305060-00006. [DOI] [PubMed] [Google Scholar]
- 428.Oren I., Rowe J.M., Sprecher H. A prospective randomized trial of itraconazole vs fluconazole for the prevention of fungal infections in patients with acute leukemia and hematopoietic stem cell transplant recipients. Bone Marrow Transplant. 2006;38:127–134. doi: 10.1038/sj.bmt.1705418. [DOI] [PubMed] [Google Scholar]
- 429.Ullmann A.J., Lipton J.H., Vesole D.H. Posaconazole or fluconazole for prophylaxis in severe graft-versus-host disease. N Engl J Med. 2007;356:335–347. doi: 10.1056/NEJMoa061098. [DOI] [PubMed] [Google Scholar]
- 430.Wingard J.R., Carter S.L., Walsh T.J. Results of a randomized, double-blind trial of fluconazole vs. voriconazole for the prevention of invasive fungal infections in 600 allogeneic blood and marrow transplant patients. Blood. 2007;110:55a. [Google Scholar]
- 431.Bjerke J.W., Meyers J.D., Bowden R.A. Hepatosplenic candidiasis—a contraindication to marrow transplantation? Blood. 1994;84:2811–2814. [PubMed] [Google Scholar]
- 432.Upton A., McCune J.S., Kirby K.A. Fluconazole coadministration concurrent with cyclophosphamide conditioning may reduce regimen-related toxicity postmyeloablative hematopoietic cell transplantation. Biol Blood Marrow Transplant. 2007;13:760–764. doi: 10.1016/j.bbmt.2007.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 433.Marr K.A., Crippa F., Leisenring W. Itraconazole versus fluconazole for prevention of fungal infections in patients receiving allogeneic stem cell transplants. Blood. 2004;103:1527–1533. doi: 10.1182/blood-2003-08-2644. [DOI] [PubMed] [Google Scholar]
- 434.Denning D.W. Invasive aspergillosis. Clin Infect Dis. 1998;26:781–803. doi: 10.1086/513943. quiz 804–805. [DOI] [PubMed] [Google Scholar]
- 435.Anaissie E.J., Kuchar R.T., Rex J.H. Fusariosis associated with pathogenic fusarium species colonization of a hospital water system: a new paradigm for the epidemiology of opportunistic mold infections. Clin Infect Dis. 2001;33:1871–1878. doi: 10.1086/324501. [DOI] [PubMed] [Google Scholar]
- 436.Anaissie E.J., Stratton S.L., Dignani M.C. Cleaning patient shower facilities: a novel approach to reducing patient exposure to aerosolized Aspergillus species and other opportunistic molds. Clin Infect Dis. 2002;35:E86–E88. doi: 10.1086/342305. [DOI] [PubMed] [Google Scholar]
- 437.Anaissie E.J., Stratton S.L., Dignani M.C. Pathogenic molds (including Aspergillus species) in hospital water distribution systems: a 3-year prospective study and clinical implications for patients with hematologic malignancies. Blood. 2003;101:2542–2546. doi: 10.1182/blood-2002-02-0530. [DOI] [PubMed] [Google Scholar]
- 438.Anaissie E.J., Stratton S.L., Dignani M.C. Pathogenic Aspergillus species recovered from a hospital water system: a 3-year prospective study. Clin Infect Dis. 2002;34:780–789. doi: 10.1086/338958. [DOI] [PubMed] [Google Scholar]
- 439.Baddley J.W., Stroud T.P., Salzman D., Pappas P.G. Invasive mold infections in allogeneic bone marrow transplant recipients. Clin Infect Dis. 2001;32:1319–1324. doi: 10.1086/319985. [DOI] [PubMed] [Google Scholar]
- 440.Marr K.A., Carter R.A., Crippa F., Wald A., Corey L. Epidemiology and outcome of mould infections in hematopoietic stem cell transplant recipients. Clin Infect Dis. 2002;34:909–917. doi: 10.1086/339202. [DOI] [PubMed] [Google Scholar]
- 441.Nucci M., Marr K.A., Queiroz-Telles F. Fusarium infection in hematopoietic stem cell transplant recipients. Clin Infect Dis. 2004;38:1237–1242. doi: 10.1086/383319. [DOI] [PubMed] [Google Scholar]
- 442.Madureira A., Bergeron A., Lacroix C. Breakthrough invasive aspergillosis in allogeneic haematopoietic stem cell transplant recipients treated with caspofungin. Int J Antimicrob Agents. 2007;30:551–554. doi: 10.1016/j.ijantimicag.2007.07.026. [DOI] [PubMed] [Google Scholar]
- 443.Kanafani Z.A., Perfect J.R. Antimicrobial resistance: resistance to antifungal agents: mechanisms and clinical impact. Clin Infect Dis. 2008;46:120–128. doi: 10.1086/524071. [DOI] [PubMed] [Google Scholar]
- 444.Rijnders B.J., Cornelissen J.J., Slobbe L. Aerosolized liposomal amphotericin B for the prevention of invasive pulmonary aspergillosis during prolonged neutropenia: a randomized, placebo-controlled trial. Clin Infect Dis. 2008;46:1401–1408. doi: 10.1086/586739. [DOI] [PubMed] [Google Scholar]
- 445.Cordonnier C., Maury S., Pautas C. Secondary antifungal prophylaxis with voriconazole to adhere to scheduled treatment in leukemic patients and stem cell transplant recipients. Bone Marrow Transplant. 2004;33:943–948. doi: 10.1038/sj.bmt.1704469. [DOI] [PubMed] [Google Scholar]
- 446.Glasmacher A., Prentice A., Gorschluter M. Itraconazole prevents invasive fungal infections in neutropenic patients treated for hematologic malignancies: evidence from a meta-analysis of 3,597 patients. J Clin Oncol. 2003;21:4615–4626. doi: 10.1200/JCO.2003.04.052. [DOI] [PubMed] [Google Scholar]
- 447.Krishna G., Martinho M., Chandrasekar P., Ullmann A.J., Patino H. Pharmacokinetics of oral posaconazole in allogeneic hematopoietic stem cell transplant recipients with graft-versus-host disease. Pharmacotherapy. 2007;27:1627–1636. doi: 10.1592/phco.27.12.1627. [DOI] [PubMed] [Google Scholar]
- 448.Pascual A., Calandra T., Bolay S., Buclin T., Bille J., Marchetti O. Voriconazole therapeutic drug monitoring in patients with invasive mycoses improves efficacy and safety outcomes. Clin Infect Dis. 2008;46:201–211. doi: 10.1086/524669. [DOI] [PubMed] [Google Scholar]
- 449.Cirino C.M., Leitman S.F., Williams E. Transfusion-associated babesiosis with an atypical time course after nonmyeloablative transplantation for sickle cell disease. Ann Intern Med. 2008;148:794–795. doi: 10.7326/0003-4819-148-10-200805200-00018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 450.Budak-Alpdogan T., Tangun Y., Kalayoglu-Besisik S. The frequency of tuberculosis in adult allogeneic stem cell transplant recipients in Turkey. Biol Blood Marrow Transplant. 2000;6:370–374. doi: 10.1016/s1083-8791(00)70013-9. [DOI] [PubMed] [Google Scholar]
- 451.Ip M.S., Yuen K.Y., Woo P.C. Risk factors for pulmonary tuberculosis in bone marrow transplant recipients. Am J Respir Crit Care Med. 1998;158:1173–1177. doi: 10.1164/ajrccm.158.4.9712072. [DOI] [PubMed] [Google Scholar]
- 452.Erdstein A.A., Daas P., Bradstock K.F., Robinson T., Hertzberg M.S. Tuberculosis in allogeneic stem cell transplant recipients: still a problem in the 21st century. Transpl Infect Dis. 2004;6:142–146. doi: 10.1111/j.1399-3062.2004.00068.x. [DOI] [PubMed] [Google Scholar]
- 453.Garces Ambrossi G., Jakubowski A., Feinstein M.B., Weinstock D.M. Active tuberculosis limited to foreign-born patients after allogeneic hematopoietic stem cell transplant. Bone Marrow Transplant. 2005;36:741–743. doi: 10.1038/sj.bmt.1705129. [DOI] [PubMed] [Google Scholar]
- 454.de la Camara R., Martino R., Granados E. Tuberculosis after hematopoietic stem cell transplantation: incidence, clinical characteristics and outcome. Spanish Group on Infectious Complications in Hematopoietic Transplantation. Bone Marrow Transplant. 2000;26:291–298. doi: 10.1038/sj.bmt.1702506. [DOI] [PubMed] [Google Scholar]
- 455.Akan H., Arslan O., Akan O.A. Tuberculosis in stem cell transplant patients. J Hosp Infect. 2006;62:421–426. doi: 10.1016/j.jhin.2005.09.020. [DOI] [PubMed] [Google Scholar]
- 456.Yuen K.Y., Woo P.C. Tuberculosis in blood and marrow transplant recipients. Hematol Oncol. 2002;20:51–62. doi: 10.1002/hon.681. [DOI] [PubMed] [Google Scholar]
- 457.Cordonnier C., Martino R., Trabasso P. Mycobacterial infection: a difficult and late diagnosis in stem cell transplant recipients. Clin Infect Dis. 2004;38:1229–1236. doi: 10.1086/383307. [DOI] [PubMed] [Google Scholar]
- 458.Saadeh C.E., Srkalovic G. Mycobacterium avium complex infection after alemtuzumab therapy for chronic lymphocytic leukemia. Pharmacotherapy. 2008;28:281–284. doi: 10.1592/phco.28.2.281. [DOI] [PubMed] [Google Scholar]
- 459.Kaplan J.E., Masur H., Holmes K.K. Guidelines for preventing opportunistic infections among HIV-infected persons—2002. Recommendations of the U.S. Public Health Service and the Infectious Diseases Society of America. MMWR Recomm Rep. 2002;51:1–52. [PubMed] [Google Scholar]
- 460.Centers for Disease Control and Prevention (CDC) Guidelines for the investigation of contacts of persons with infectious tuberculosis. Recommendations from the National Tuberculosis Controllers Association and CDC. MMWR Recomm Rep. 2005;54:1–47. [PubMed] [Google Scholar]
- 461.Centers for Disease Control and Prevention (CDC) The role of BCG vaccine in the prevention and control of tuberculosis in the United States. A joint statement by the Advisory Council for the Elimination of Tuberculosis and the Advisory Committee on Immunization Practices. MMWR Recomm Rep. 1996;45:1–18. [PubMed] [Google Scholar]
- 462.Targeted tuberculin testing and treatment of latent tuberculosis infection American Thoracic Society. MMWR Recomm Rep. 2000;49:1–51. [PubMed] [Google Scholar]
- 463.Menzies D., Pai M., Comstock G. Meta-analysis: new tests for the diagnosis of latent tuberculosis infection: areas of uncertainty and recommendations for research. Ann Intern Med. 2007;146:340–354. doi: 10.7326/0003-4819-146-5-200703060-00006. [DOI] [PubMed] [Google Scholar]
- 464.Matulis G., Juni P., Villiger P.M., Gadola S.D. Detection of latent tuberculosis in immunosuppressed patients with autoimmune diseases: performance of a Mycobacterium tuberculosis antigen-specific interferon gamma assay. Ann Rheum Dis. 2008;67:84–90. doi: 10.1136/ard.2007.070789. [DOI] [PubMed] [Google Scholar]
- 465.Interferon gamma release assays for latent tuberculosis infection An Advisory Committee Statement (ACS) Can Commun Dis Rep. 2007;33:1–18. [PubMed] [Google Scholar]
- 466.Kobashi Y., Mouri K., Obase Y., Fukuda M., Miyashita N., Oka M. Clinical evaluation of QuantiFERON TB-2G test for immunocompromised patients. Eur Respir J. 2007;30:945–950. doi: 10.1183/09031936.00040007. [DOI] [PubMed] [Google Scholar]
- 467.Lee J., Lee M.H., Kim W.S. Tuberculosis in hematopoietic stem cell transplant recipients in Korea. Int J Hematol. 2004;79:185–188. doi: 10.1532/ijh97.a10219. [DOI] [PubMed] [Google Scholar]
- 468.Ahmed P., Anwar M., Khan B. Role of isoniazid prophylaxis for prevention of tuberculosis in haemopoietic stem cell transplant recipients. J Pak Med Assoc. 2005;55:378–381. [PubMed] [Google Scholar]
- 469.Update: adverse event data and revised American Thoracic Society/CDC recommendations against the use of rifampin and pyrazinamide for treatment of latent tuberculosis infection—United States, 2003. MMWR Morb Mortal Wkly Rep. 2003;52:735–739. [PubMed] [Google Scholar]
- 470.Kroger A.T., Atkinson W.L., Marcuse E.K., Pickering L.K. General recommendations on immunization: recommendations of the Advisory Committee on Immunization Practices (ACIP) MMWR Recomm Rep. 2006;55:1–48. [PubMed] [Google Scholar]
- 471.Brazinsky J.H., Phillips J.E. Pneumocystis pneumonia transmission between patients with lymphoma. JAMA. 1969;209:1527. doi: 10.1001/jama.209.10.1527. [DOI] [PubMed] [Google Scholar]
- 472.Chave J.P., David S., Wauters J.P., Van Melle G., Francioli P. Transmission of Pneumocystis carinii from AIDS patients to other immunosuppressed patients: a cluster of Pneumocystis carinii pneumonia in renal transplant recipients. AIDS. 1991;5:927–932. doi: 10.1097/00002030-199108000-00002. [DOI] [PubMed] [Google Scholar]
- 473.Goesch T.R., Gotz G., Stellbrinck K.H., Albrecht H., Weh H.J., Hossfeld D.K. Possible transfer of Pneumocystis carinii between immunodeficient patients. Lancet. 1990;336:627. doi: 10.1016/0140-6736(90)93420-t. [DOI] [PubMed] [Google Scholar]
- 474.Bensousan T., Garo B., Islam S., Bourbigot B., Cledes J., Garre M. Possible transfer of Pneumocystis carinii between kidney transplant recipients. Lancet. 1990;336:1066–1067. doi: 10.1016/0140-6736(90)92534-o. [DOI] [PubMed] [Google Scholar]
- 475.Ruskin J., Remington J.S. The compromised host and infection. I. Pneumocystis carinii pneumonia. JAMA. 1967;202:1070–1074. [PubMed] [Google Scholar]
- 476.Watanabe J.M., Chinchinian H., Weitz C., McIvanie S.K. Pneumocystis carinii pneumonia in a family. JAMA. 1965;193:685–686. doi: 10.1001/jama.1965.03090080047017. [DOI] [PubMed] [Google Scholar]
- 477.Vargas S.L., Ponce C.A., Gigliotti F. Transmission of Pneumocystis carinii DNA from a patient with P. carinii pneumonia to immunocompetent contact health care workers. J Clin Microbiol. 2000;38:1536–1538. doi: 10.1128/jcm.38.4.1536-1538.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 478.Wohl A.R., Simon P., Hu Y.W., Duchin J.S. The role of person-to-person transmission in an epidemiologic study of Pneumocystis carinii pneumonia. AIDS. 2002;16:1821–1825. doi: 10.1097/00002030-200209060-00016. [DOI] [PubMed] [Google Scholar]
- 479.Miller R.F., Ambrose H.E., Novelli V., Wakefield A.E. Probable mother-to-infant transmission of Pneumocystis carinii f. sp. hominis infection. J Clin Microbiol. 2002;40:1555–1557. doi: 10.1128/JCM.40.4.1555-1557.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 480.Manoloff E.S., Francioli P., Taffe P., Van Melle G., Bille J., Hauser P.M. Risk for Pneumocystis carinii transmission among patients with pneumonia: a molecular epidemiology study. Emerg Infect Dis. 2003;9:132–134. doi: 10.3201/eid0901.020141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 481.Tuan I.Z., Dennison D., Weisdorf D.J. Pneumocystis carinii pneumonitis following bone marrow transplantation. Bone Marrow Transplant. 1992;10:267–272. [PubMed] [Google Scholar]
- 482.De Castro N., Neuville S., Sarfati C. Occurrence of Pneumocystis jiroveci pneumonia after allogeneic stem cell transplantation: a 6-year retrospective study. Bone Marrow Transplant. 2005;36:879–883. doi: 10.1038/sj.bmt.1705149. [DOI] [PubMed] [Google Scholar]
- 483.Chen C.S., Boeckh M., Seidel K. Incidence, risk factors, and mortality from pneumonia developing late after hematopoietic stem cell transplantation. Bone Marrow Transplant. 2003;32:515–522. doi: 10.1038/sj.bmt.1704162. [DOI] [PubMed] [Google Scholar]
- 484.Lyytikainen O., Ruutu T., Volin L. Late onset Pneumocystis carinii pneumonia following allogeneic bone marrow transplantation. Bone Marrow Transplant. 1996;17:1057–1059. [PubMed] [Google Scholar]
- 485.Green H., Paul M., Vidal L., Leibovici L. Prophylaxis for Pneumocystis pneumonia (PCP) in non-HIV immunocompromised patients. Cochrane Database Syst Rev. 2007 doi: 10.1002/14651858.CD005590.pub2. CD005590. [DOI] [PubMed] [Google Scholar]
- 486.Green H., Paul M., Vidal L., Leibovici L. Prophylaxis of Pneumocystis pneumonia in immunocompromised non-HIV-infected patients: systematic review and meta-analysis of randomized controlled trials. Mayo Clin Proc. 2007;82:1052–1059. doi: 10.4065/82.9.1052. [DOI] [PubMed] [Google Scholar]
- 487.Maltezou H.C., Petropoulos D., Choroszy M. Dapsone for Pneumocystis carinii prophylaxis in children undergoing bone marrow transplantation. Bone Marrow Transplant. 1997;20:879–881. doi: 10.1038/sj.bmt.1700978. [DOI] [PubMed] [Google Scholar]
- 488.Sangiolo D., Storer B., Nash R. Toxicity and efficacy of daily dapsone as Pneumocystis jiroveci prophylaxis after hematopoietic stem cell transplantation: a case-control study. Biol Blood Marrow Transplant. 2005;11:521–529. doi: 10.1016/j.bbmt.2005.04.011. [DOI] [PubMed] [Google Scholar]
- 489.Link H., Vohringer H.F., Wingen F., Bragas B., Schwardt A., Ehninger G. Pentamidine aerosol for prophylaxis of Pneumocystis carinii pneumonia after BMT. Bone Marrow Transplant. 1993;11:403–406. [PubMed] [Google Scholar]
- 490.Chan C., Montaner J., Lefebvre E.A. Atovaquone suspension compared with aerosolized pentamidine for prevention of Pneumocystis carinii pneumonia in human immunodeficiency virus-infected subjects intolerant of trimethoprim or sulfonamides. J Infect Dis. 1999;180:369–376. doi: 10.1086/314893. [DOI] [PubMed] [Google Scholar]
- 491.Souza J.P., Boeckh M., Gooley T.A., Flowers M.E., Crawford S.W. High rates of Pneumocystis carinii pneumonia in allogeneic blood and marrow transplant recipients receiving dapsone prophylaxis. Clin Infect Dis. 1999;29:1467–1471. doi: 10.1086/313509. [DOI] [PubMed] [Google Scholar]
- 492.Purdy B.H., Philips D.M., Summers R.W. Desensitization for sulfasalazine skin rash. Ann Intern Med. 1984;100:512–514. doi: 10.7326/0003-4819-100-4-512. [DOI] [PubMed] [Google Scholar]
- 493.Walter E, Chauncey T, Boeckh M, Hackman B, Kennedy M, Bowden R. PCP prophylaxis after marrow transplantation: efficacy of trimethoprim-sulfamethoxazole (trim-sulfa), role of trim-sulfa desensitization and iv pentamidine [abstract 32]. In: Program and abstracts of the 33d Interscience Conference on Antimicrobial Agents and Chemotherapy (New Orleans). Washington, DC: American Society for Microbiology, 1993:125.
- 494.Vasconcelles M.J., Bernardo M.V., King C., Weller E.A., Antin J.H. Aerosolized pentamidine as pneumocystis prophylaxis after bone marrow transplantation is inferior to other regimens and is associated with decreased survival and an increased risk of other infections. Biol Blood Marrow Transplant. 2000;6:35–43. doi: 10.1016/s1083-8791(00)70050-4. [DOI] [PubMed] [Google Scholar]
- 495.Derouin F., Devergie A., Auber P. Toxoplasmosis in bone marrow-transplant recipients: report of seven cases and review. Clin Infect Dis. 1992;15:267–270. doi: 10.1093/clinids/15.2.267. [DOI] [PubMed] [Google Scholar]
- 496.Siegel S.E., Lunde M.N., Gelderman A.H. Transmission of toxoplasmosis by leukocyte transfusion. Blood. 1971;37:388–394. [PubMed] [Google Scholar]
- 497.Jurges E., Young Y., Eltumi M. Transmission of toxoplasmosis by bone marrow transplant associated with Campath-1G. Bone Marrow Transplant. 1992;9:65–66. [PubMed] [Google Scholar]
- 498.Chandrasekar P.H., Momin F. Disseminated toxoplasmosis in marrow recipients: a report of three cases and a review of the literature. Bone Marrow Transplant Team. Bone Marrow Transplant. 1997;19:685–689. doi: 10.1038/sj.bmt.1700736. [DOI] [PubMed] [Google Scholar]
- 499.Bretagne S., Costa J.M.F.F., Jabot-Lestang L., aud-Camus F., Cordonnier C. Prospective study of toxoplasma reactivation by polymerase chain reaction in allogeneic stem cell transplant recipients. Transpl Infect Dis. 2000;2:127–132. doi: 10.1034/j.1399-3062.2000.020305.x. [DOI] [PubMed] [Google Scholar]
- 500.Martino R., Bretagne S., Einsele H. Early detection of Toxoplasma infection by molecular monitoring of Toxoplasma gondii in peripheral blood samples after allogeneic stem cell transplantation. Clin Infect Dis. 2005;40:67–78. doi: 10.1086/426447. [DOI] [PubMed] [Google Scholar]
- 501.Lim Z., Baker B., Zuckerman M. Toxoplasmosis following alemtuzumab based allogeneic haematopoietic stem cell transplantation. J Infect. 2007;54:e83–e86. doi: 10.1016/j.jinf.2006.04.013. [DOI] [PubMed] [Google Scholar]
- 502.Small T.N., Leung L., Stiles J. Disseminated toxoplasmosis following T cell-depleted related and unrelated bone marrow transplantation. Bone Marrow Transplant. 2000;25:969–973. doi: 10.1038/sj.bmt.1702370. [DOI] [PubMed] [Google Scholar]
- 503.Costa J.M., Munoz C., Kruger D. Quality control for the diagnosis of Toxoplasma gondii reactivation in SCT patients using PCR assays. Bone Marrow Transplant. 2001;28:527–528. doi: 10.1038/sj.bmt.1703175. [DOI] [PubMed] [Google Scholar]
- 504.Ionita C., Wasay M., Balos L., Bakshi R. MR imaging in toxoplasmosis encephalitis after bone marrow transplantation: paucity of enhancement despite fulminant disease. AJNR Am J Neuroradiol. 2004;25:270–273. [PMC free article] [PubMed] [Google Scholar]
- 505.Roemer E., Blau I.W., Basara N. Toxoplasmosis, a severe complication in allogeneic hematopoietic stem cell transplantation: successful treatment strategies during a 5-year single-center experience. Clin Infect Dis. 2001;32:E1–E8. doi: 10.1086/317548. [DOI] [PubMed] [Google Scholar]
- 506.Foot A.B., Garin Y.J., Ribaud P., Devergie A., Derouin F., Gluckman E. Prophylaxis of toxoplasmosis infection with pyrimethamine/sulfadoxine (Fansidar) in bone marrow transplant recipients. Bone Marrow Transplant. 1994;14:241–245. [PubMed] [Google Scholar]
- 507.Peacock J.J.E., Greven C.M., Cruz J.M., Hurd D.D. Reactivation toxoplasmic retinochoroiditis in patients undergoing bone marrow transplantation: is there a role for chemoprophylaxis? Bone Marrow Transplant. 1995;15:983–987. [PubMed] [Google Scholar]
- 508.Daly A.S., McGeer A., Lipton J.H. Systemic nocardiosis following allogeneic bone marrow transplantation. Transpl Infect Dis. 2003;5:16–20. doi: 10.1034/j.1399-3062.2003.00007.x. [DOI] [PubMed] [Google Scholar]
- 509.Filice G.A. Nocardiosis in persons with human immunodeficiency virus infection, transplant recipients, and large, geographically defined populations. J Lab Clin Med. 2005;145:156–162. doi: 10.1016/j.lab.2005.01.002. [DOI] [PubMed] [Google Scholar]
- 510.van Burik J., Hackman R.C., Nadeem S.Q. Nocardiosis after bone marrow transplantation: A retrospective study. Clin Infect Dis. 1997;24:1154–1160. doi: 10.1086/513654. [DOI] [PubMed] [Google Scholar]
- 511.Martinez Tomas R., Menendez Villanueva R., Reyes Calzada S. Pulmonary nocardiosis: risk factors and outcomes. Respirology. 2007;12:394–400. doi: 10.1111/j.1440-1843.2007.01078.x. [DOI] [PubMed] [Google Scholar]
- 512.Peleg A.Y., Husain S., Qureshi Z.A. Risk factors, clinical characteristics, and outcome of nocardia infection in organ transplant recipients: a matched case-control study. Clin Infect Dis. 2007;44:1307–1314. doi: 10.1086/514340. [DOI] [PubMed] [Google Scholar]
- 513.Liu L.X., Weller P.F. Strongyloidiasis and other intestinal nematode infections. Infect Dis Clin North Am. 1993;7:655–682. [PubMed] [Google Scholar]
- 514.Conway D.J., Atkins N.S., Lillywhite J.E. Immunodiagnosis of Strongyloides stercoralis infection: a method for increasing the specificity of the indirect ELISA. Trans R Soc Trop Med Hyg. 1993;87:173–176. doi: 10.1016/0035-9203(93)90477-8. [DOI] [PubMed] [Google Scholar]
- 515.Boscolo M., Gobbo M., Mantovani W. Evaluation of an indirect immunofluorescence assay for strongyloidiasis as a tool for diagnosis and follow-up. Clin Vaccine Immunol. 2007;14:129–133. doi: 10.1128/CVI.00278-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 516.Schaffel R., Nucci M., Carvalho E. The value of an immunoenzymatic test (enzyme-linked immunosorbent assay) for the diagnosis of strongyloidiasis in patients immunosuppressed by hematologic malignancies. Am J Trop Med Hyg. 2001;65:346–350. doi: 10.4269/ajtmh.2001.65.346. [DOI] [PubMed] [Google Scholar]
- 517.Keiser P.B., Nutman T.B. Strongyloides stercoralis in the immunocompromised population. Clin Microbiol Rev. 2004;17:208–217. doi: 10.1128/CMR.17.1.208-217.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 518.Leiby D.A., Fucci M.H., Stumpf R.J. Trypanosoma cruzi in a low- to moderate-risk blood donor population: seroprevalence and possible congenital transmission. Transfusion. 1999;39:310–315. doi: 10.1046/j.1537-2995.1999.39399219290.x. [DOI] [PubMed] [Google Scholar]
- 519.Sirvent-von Bueltzingsloewen A., Marty P., Rosenthal E. Visceral leishmaniasis: a new opportunistic infection in hematopoietic stem-cell-transplanted patients. Bone Marrow Transplant. 2004;33:667–668. doi: 10.1038/sj.bmt.1704396. [DOI] [PubMed] [Google Scholar]
- 520.Agteresch H.J., van 't Veer M.B., Cornelissen J.J., Sluiters J.F. Visceral leishmaniasis after allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant. 2007;40:391–393. doi: 10.1038/sj.bmt.1705728. [DOI] [PubMed] [Google Scholar]
- 521.Rodriguez-Wilhelmi P., Panizo C., Ruza E., Rocha E. Treatment of visceral leishmaniasis with liposomal amphotericin B in three immunocompromised patients. Med. Clin (Barc) 2001;116:37–38. doi: 10.1016/s0025-7753(01)71711-3. [DOI] [PubMed] [Google Scholar]
- 522.Dictar M., Sinagra A., Veron M.T. Recipients and donors of bone marrow transplants suffering from Chagas' disease: management and preemptive therapy of parasitemia. Bone Marrow Transplant. 1998;21:391–393. doi: 10.1038/sj.bmt.1701107. [DOI] [PubMed] [Google Scholar]
- 523.Congenital infection with Trypanosoma cruzi: from mechanisms of transmission to strategies for diagnosis and control. Rev Soc Bras Med Trop. 2003;36:767–771. doi: 10.1590/s0037-86822003000600024. [DOI] [PubMed] [Google Scholar]
- 524.Moraes-Souza H., Bordin J.O. Strategies for prevention of transfusion-associated Chagas' disease. Transfus Med Rev. 1996;10:161–170. doi: 10.1016/s0887-7963(96)80057-5. [DOI] [PubMed] [Google Scholar]
- 525.Altclas J., Sinagra A., Jaimovich G. Reactivation of chronic Chagas' disease following allogeneic bone marrow transplantation and successful pre-emptive therapy with benznidazole. Transpl Infect Dis. 1999;1:135–137. doi: 10.1034/j.1399-3062.1999.010207.x. [DOI] [PubMed] [Google Scholar]
- 526.Altclas J., Sinagra A., Dictar M. Chagas disease in bone marrow transplantation: an approach to preemptive therapy. Bone Marrow Transplant. 2005;36:123–129. doi: 10.1038/sj.bmt.1705006. [DOI] [PubMed] [Google Scholar]
- 527.World Health Organization . World Health Organization; Geneva, Switzerland: 2008. World Malaria Report 2008. [Google Scholar]
- 528.Shamsi T., Hashmi K., Adil S. The stem cell transplant program in Pakistan—the first decade. Bone Marrow Transplant. 2008;42(Suppl 1):S114–S117. doi: 10.1038/bmt.2008.137. [DOI] [PubMed] [Google Scholar]
- 529.D'Alessandro U., Olaleye B.O., McGuire W. A comparison of the efficacy of insecticide-treated and untreated bed nets in preventing malaria in Gambian children. Trans R Soc Trop Med Hyg. 1995;89:596–598. doi: 10.1016/0035-9203(95)90401-8. [DOI] [PubMed] [Google Scholar]
- 530.Hill N., Lenglet A., Arnez A.M., Carneiro I. Plant based insect repellent and insecticide treated bed nets to protect against malaria in areas of early evening biting vectors: double blind randomised placebo controlled clinical trial in the Bolivian Amazon. BMJ. 2007;335:1023. doi: 10.1136/bmj.39356.574641.55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 531.Sehulster L., Chinn R.Y. Guidelines for environmental infection control in health-care facilities. Recommendations of CDC and the Healthcare Infection Control Practices Advisory Committee (HICPAC) MMWR Recomm Rep. 2003;52:1–42. [PubMed] [Google Scholar]
- 532.Guidelines for Design and Construction of Health Care Facilities. American Institute of Architects Press; Washington DC: 2006. American Institute of Architects Facility Guidelines Institute, AIA Academy of Architecture for Health, US Dept.of Health & Human Services. [Google Scholar]
- 533.Rhame F.S., Streifel A.J., Kersey J.H., Jr., McGlave P.B. Extrinsic risk factors for pneumonia in the patient at high risk of infection. Am J Med. 1984;76:42–52. doi: 10.1016/0002-9343(84)90243-2. [DOI] [PubMed] [Google Scholar]
- 534.Tablan O.C., Anderson L.J., Besser R., Bridges C., Hajjeh R. Guidelines for preventing health-care-associated pneumonia, 2003: recommendations of CDC and the Healthcare Infection Control Practices Advisory Committee. MMWR Recomm Rep. 2004;53:1–36. [PubMed] [Google Scholar]
- 535.Benet T., Nicolle M.C., Thiebaut A. Reduction of invasive aspergillosis incidence among immunocompromised patients after control of environmental exposure. Clin Infect Dis. 2007;45:682–686. doi: 10.1086/521378. [DOI] [PubMed] [Google Scholar]
- 536.Hahn T., Cummings K.M., Michalek A.M., Lipman B.J., Segal B.H., McCarthy P.L., Jr. Efficacy of high-efficiency particulate air filtration in preventing aspergillosis in immunocompromised patients with hematologic malignancies. Infect Control Hosp Epidemiol. 2002;23:525–531. doi: 10.1086/502101. [DOI] [PubMed] [Google Scholar]
- 537.Rice N., Streifel A., Vesley D. An evaluation of hospital special-ventilation-room pressures. Infect Control Hosp Epidemiol. 2001;22:19–23. doi: 10.1086/501819. [DOI] [PubMed] [Google Scholar]
- 538.Engelhart S., Hanfland J., Glasmacher A., Krizek L., Schmidt-Wolf I.G., Exner M. Impact of portable air filtration units on exposure of haematology-oncology patients to airborne A spergillus fumigatus spores under field conditions. J Hosp Infect. 2003;54:300–304. doi: 10.1016/s0195-6701(03)00176-2. [DOI] [PubMed] [Google Scholar]
- 539.Rutala W.A. APIC guideline for selection and use of disinfectants. 1994, 1995, and 1996 APIC Guidelines Committee. Association for Professionals in Infection Control and Epidemiology, Inc. Am J Infect Control. 1996;24:313–342. doi: 10.1016/s0196-6553(96)90066-8. [DOI] [PubMed] [Google Scholar]
- 540.Loo V.G., Bertrand C., Dixon C. Control of construction-associated nosocomial aspergillosis in an antiquated hematology unit. Infect Control Hosp Epidemiol. 1996;17:360–364. doi: 10.1086/647317. [DOI] [PubMed] [Google Scholar]
- 541.Barnes R.A., Rogers T.R. Control of an outbreak of nosocomial aspergillosis by laminar air-flow isolation. J Hosp Infect. 1989;14:89–94. doi: 10.1016/0195-6701(89)90110-2. [DOI] [PubMed] [Google Scholar]
- 542.Cornet M., Levy V., Fleury L. Efficacy of prevention by high-efficiency particulate air filtration or laminar airflow against Aspergillus airborne contamination during hospital renovation. Infect Control Hosp Epidemiol. 1999;20:508–513. doi: 10.1086/501661. [DOI] [PubMed] [Google Scholar]
- 543.Thio C.L., Smith D., Merz W.G. Refinements of environmental assessment during an outbreak investigation of invasive aspergillosis in a leukemia and bone marrow transplant unit. Infect Control Hosp Epidemiol. 2000;21:18–23. doi: 10.1086/501691. [DOI] [PubMed] [Google Scholar]
- 544.Kistemann T., Huneburg H., Exner M., Vacata V., Engelhart S. Role of increased environmental Aspergillus exposure for patients with chronic obstructive pulmonary disease (COPD) treated with corticosteroids in an intensive care unit. Int J Hyg Environ Health. 2002;204:347–351. doi: 10.1078/1438-4639-00106. [DOI] [PubMed] [Google Scholar]
- 545.Vilchez R.A., Fung J., Kusne S. Cryptococcosis in organ transplant recipients: an overview. Am J Transplant. 2002;2:575–580. doi: 10.1034/j.1600-6143.2002.20701.x. [DOI] [PubMed] [Google Scholar]
- 546.Streifel AJ. Design and maintenance of hospital ventilation systems and the prevention of airborne infections. In: Mayhall C, ed. Hospital Epidemiology and Infection Control. Philadelphia, PA: Lippincott, Williams & Wilkins, 2004.
- 547.Walsh T.J., Dixon D.M. Nosocomial aspergillosis: environmental microbiology, hospital epidemiology, diagnosis and treatment. Eur J Epidemiol. 1989;5:131–142. doi: 10.1007/BF00156818. [DOI] [PubMed] [Google Scholar]
- 548.Vonberg R.P., Gastmeier P. Nosocomial aspergillosis in outbreak settings. J Hosp Infect. 2006;63:246–254. doi: 10.1016/j.jhin.2006.02.014. [DOI] [PubMed] [Google Scholar]
- 549.Bartley J. Prevention of infections related to construction, renovation, and demolition systems. In: Mayhall C ed. Hospital Epidemiology and Infection Control. Philadelphia, PA: Lippincott, Williams & Wilkins, 2004.
- 550.Kidd F., Buttner C., Kressel A.B. Construction: a model program for infection control compliance. Am J Infect Control. 2007;35:347–350. doi: 10.1016/j.ajic.2006.07.011. [DOI] [PubMed] [Google Scholar]
- 551.Srinivasan A., Beck C., Buckley T. The ability of hospital ventilation systems to filter Aspergillus and other fungi following a building implosion. Infect Control Hosp Epidemiol. 2002;23:520–524. doi: 10.1086/502100. [DOI] [PubMed] [Google Scholar]
- 552.Berthelot P., Loulergue P., Raberin H. Efficacy of environmental measures to decrease the risk of hospital-acquired aspergillosis in patients hospitalised in haematology wards. Clin Microbiol Infect. 2006;12:738–744. doi: 10.1111/j.1469-0691.2006.01499.x. [DOI] [PubMed] [Google Scholar]
- 553.Raad I., Hanna H., Osting C. Masking of neutropenic patients on transport from hospital rooms is associated with a decrease in nosocomial aspergillosis during construction. Infect Control Hosp Epidemiol. 2002;23:41–43. doi: 10.1086/501967. [DOI] [PubMed] [Google Scholar]
- 554.Cooper E.E., O'Reilly M.A., Guest D.I., Dharmage S.C. Influence of building construction work on Aspergillus infection in a hospital setting. Infect Control Hosp Epidemiol. 2003;24:472–476. doi: 10.1086/502239. [DOI] [PubMed] [Google Scholar]
- 555.Mahieu L.M., De Dooy J.J., Van Laer F.A., Jansens H., Ieven M.M. A prospective study on factors influencing Aspergillus spore load in the air during renovation works in a neonatal intensive care unit. J Hosp Infect. 2000;45:191–197. doi: 10.1053/jhin.2000.0773. [DOI] [PubMed] [Google Scholar]
- 556.Weems J.J., Jr., Davis B.J., Tablan O.C., Kaufman L., Martone W.J. Construction activity: an independent risk factor for invasive aspergillosis and zygomycosis in patients with hematologic malignancy. Infect Control. 1987;8:71–75. doi: 10.1017/s0195941700067114. [DOI] [PubMed] [Google Scholar]
- 557.Nihtinen A., Anttila V.J., Richardson M., Meri T., Volin L., Ruutu T. The utility of intensified environmental surveillance for pathogenic moulds in a stem cell transplantation ward during construction work to monitor the efficacy of HEPA filtration. Bone Marrow Transplant. 2007;40:457–460. doi: 10.1038/sj.bmt.1705749. [DOI] [PubMed] [Google Scholar]
- 558.Falvey D.G., Streifel A.J. Ten-year air sample analysis of Aspergillus prevalence in a university hospital. J Hosp Infect. 2007;67:35–41. doi: 10.1016/j.jhin.2007.06.008. [DOI] [PubMed] [Google Scholar]
- 559.Lai K.K. A cluster of invasive aspergillosis in a bone marrow transplant unit related to construction and the utility of air sampling. Am J Infect Control. 2001;29:333–337. doi: 10.1067/mic.2001.117038. [DOI] [PubMed] [Google Scholar]
- 560.Morris G., Kokki M.H., Anderson K., Richardson M.D. Sampling of Aspergillus spores in air. J Hosp Infect. 2000;44:81–92. doi: 10.1053/jhin.1999.0688. [DOI] [PubMed] [Google Scholar]
- 561.Streifel A.J. Aspergillus and construction. In: Kundsin R.B., editor. Architectural Design and Indoor Microbial Pollution. Oxford University Press; New York: 1988. pp. 198–216. [Google Scholar]
- 562.Opal S.M., Asp A.A., Cannady P.B., Jr., Morse P.L., Burton L.J., Hammer P.G. Efficacy of infection control measures during a nosocomial outbreak of disseminated aspergillosis associated with hospital construction. J Infect Dis. 1986;153:634–637. doi: 10.1093/infdis/153.3.634. [DOI] [PubMed] [Google Scholar]
- 563.Lee I., Barton T.D. Viral respiratory tract infections in transplant patients: epidemiology, recognition and management. Drugs. 2007;67:1411–1427. doi: 10.2165/00003495-200767100-00004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 564.Curtis L., Cali S., Conroy L. Aspergillus surveillance project at a large tertiary-care hospital. J Hosp Infect. 2005;59:188–196. doi: 10.1016/j.jhin.2004.05.017. [DOI] [PubMed] [Google Scholar]
- 565.Dettenkofer M., Wenzler S., Amthor S., Antes G., Motschall E., Daschner F.D. Does disinfection of environmental surfaces influence nosocomial infection rates? A systematic review. Am J Infect Control. 2004;32:84–89. doi: 10.1016/j.ajic.2003.07.006. [DOI] [PubMed] [Google Scholar]
- 566.Rutala W.A., Weber D.J. The benefits of surface disinfection. Am J Infect Control. 2004;32:226–231. doi: 10.1016/j.ajic.2004.04.197. [DOI] [PubMed] [Google Scholar]
- 567.Gerson S.L., Parker P., Jacobs M.R., Creger R., Lazarus H.M. Aspergillosis due to carpet contamination. Infect Control Hosp Epidemiol. 1994;15:221–223. [PubMed] [Google Scholar]
- 568.Richet H.M., McNeil M.M., Davis B.J. Aspergillus fumigatus sternal wound infections in patients undergoing open heart surgery. Am J Epidemiol. 1992;135:48–58. doi: 10.1093/oxfordjournals.aje.a116201. [DOI] [PubMed] [Google Scholar]
- 569.Pegues C.F., Daar E.S., Murthy A.R. The epidemiology of invasive pulmonary aspergillosis at a large teaching hospital. Infect Control Hosp Epidemiol. 2001;22:370–374. doi: 10.1086/501915. [DOI] [PubMed] [Google Scholar]
- 570.Noskin G.A., Bednarz P., Suriano T., Reiner S., Peterson L.R. Persistent contamination of fabric-covered furniture by vancomycin-resistant enterococci: implications for upholstery selection in hospitals. Am J Infect Control. 2000;28:311–313. doi: 10.1067/mic.2000.108129. [DOI] [PubMed] [Google Scholar]
- 571.Mangram A.J., Horan T.C., Pearson M.L., Silver L.C., Jarvis W.R. Guideline for prevention of surgical site infection, 1999. Hospital Infection Control Practices Advisory Committee. Infect Control Hosp Epidemiol. 1999;20:250–278. doi: 10.1086/501620. quiz 279–280. [DOI] [PubMed] [Google Scholar]
- 572.Passweg J.R., Rowlings P.A., Atkinson K.A. Influence of protective isolation on outcome of allogeneic bone marrow transplantation for leukemia. Bone Marrow Transplant. 1998;21:1231–1238. doi: 10.1038/sj.bmt.1701238. [DOI] [PubMed] [Google Scholar]
- 573.Dadd G., McMinn P., Monterosso L. Protective isolation in hemopoietic stem cell transplants: a review of the literature and single institution experience. J Pediatr Oncol Nurs. 2003;20:293–300. doi: 10.1177/1043454203254985. [DOI] [PubMed] [Google Scholar]
- 574.Gupta A., Della-Latta P., Todd B. Outbreak of extended-spectrum beta-lactamase-producing Klebsiella pneumoniae in a neonatal intensive care unit linked to artificial nails. Infect Control Hosp Epidemiol. 2004;25:210–215. doi: 10.1086/502380. [DOI] [PubMed] [Google Scholar]
- 575.Moolenaar R.L., Crutcher J.M., San Joaquin V.H. A prolonged outbreak of Pseudomonas aeruginosa in a neonatal intensive care unit: did staff fingernails play a role in disease transmission? Infect Control Hosp Epidemiol. 2000;21:80–85. doi: 10.1086/501739. [DOI] [PubMed] [Google Scholar]
- 576.Boszczowski I., Nicoletti C., Puccini D.M. Outbreak of extended spectrum beta-lactamase-producing Klebsiella pneumoniae infection in a neonatal intensive care unit related to onychomycosis in a health care worker. Pediatr Infect Dis J. 2005;24:648–650. doi: 10.1097/01.inf.0000168844.55898.8f. [DOI] [PubMed] [Google Scholar]
- 577.Pittet D., Hugonnet S., Harbarth S. Effectiveness of a hospital-wide programme to improve compliance with hand hygiene. Infection Control Programme. Lancet. 2000;356:1307–1312. doi: 10.1016/s0140-6736(00)02814-2. [DOI] [PubMed] [Google Scholar]
- 578.Widmer A.F. Replace hand washing with use of a waterless alcohol hand rub? Clin Infect Dis. 2000;31:136–143. doi: 10.1086/313888. [DOI] [PubMed] [Google Scholar]
- 579.Boyce J.M., Pittet D. Guideline for hand hygiene in health-care settings. Recommendations of the Healthcare Infection Control Practices Advisory Committee and the HICPAC/SHEA/APIC/IDSA Hand Hygiene Task Force. Society for Healthcare Epidemiology of America/Association for Professionals in Infection Control/Infectious Diseases Society of America. MMWR Recomm Rep. 2002;51:1–45. quiz CE1-4. [PubMed] [Google Scholar]
- 580.Bischoff W.E., Reynolds T.M., Sessler C.N., Edmond M.B., Wenzel R.P. Handwashing compliance by health care workers: the impact of introducing an accessible, alcohol-based hand antiseptic. Arch Intern Med. 2000;160:1017–1021. doi: 10.1001/archinte.160.7.1017. [DOI] [PubMed] [Google Scholar]
- 581.Hugonnet S., Perneger T.V., Pittet D. Alcohol-based hand rub improves compliance with hand hygiene in intensive care units. Arch Intern Med. 2002;162:1037–1043. doi: 10.1001/archinte.162.9.1037. [DOI] [PubMed] [Google Scholar]
- 582.National Antimicrobial Information Network. List of EPA registered products.
- 583.CDC/National Center for Infectious Diseases/Hospital Infections Program . US Department of Health and Human Services, CDC; Atlanta, GA: 2000. Sterilization or Disinfection of Medical Devices: General Purposes. [Google Scholar]
- 584.Favero M., Bond W. Sterilization, disinfection, and antispsis int he hospital. In: Hauser W.J. Jr., Herrman J.L., Isenberg H.D., Shadomy H.J., editors. Manual of Clincial Microbiology. American Society for Microbiology; Washington DC: 1991. pp. 183–200. [Google Scholar]
- 585.Johnson S., Gerding D. Clostridium difficile. In: Mayhall C.G., editor. Hospital Epidemiology and Infection Control. Lippincott Williams & Wilkins; Philadelphia, PA: 1999. pp. 467–476. [Google Scholar]
- 586.Rutala W.A., Weber D.J. Disinfection of endoscopes: review of new chemical sterilants used for high-level disinfection. Infect Control Hosp Epidemiol. 1999;20:69–76. doi: 10.1086/501544. [DOI] [PubMed] [Google Scholar]
- 587.Centers for Disease Control and Prevention (CDC) Nosocomial outbreak of Rhizopus infections associated with Elastoplast wound dressings—Minnesota. MMWR Morb Mortal Wkly Rep. 1978;27:33–34. [Google Scholar]
- 588.Centers for Disease Control and Prevention (CDC) Follow-up on Rhizopus infections associated with Elastoplast bandages—United States. MMWR Morb Mortal Wkly Rep. 1978;27:243–244. [Google Scholar]
- 589.Bryce E.A., Walker M., Scharf S. An outbreak of cutaneous aspergillosis in a tertiary-care hospital. Infect Control Hosp Epidemiol. 1996;17:170–172. doi: 10.1086/647266. [DOI] [PubMed] [Google Scholar]
- 590.McCarty J.M., Flam M.S., Pullen G., Jones R., Kassel S.H. Outbreak of primary cutaneous aspergillosis related to intravenous arm boards. J Pediatr. 1986;108:721–724. doi: 10.1016/s0022-3476(86)81051-4. [DOI] [PubMed] [Google Scholar]
- 591.Mitchell S.J., Gray J., Morgan M.E., Hocking M.D., Durbin G.M. Nosocomial infection with Rhizopus microsporus in preterm infants: association with wooden tongue depressors. Lancet. 1996;348:441–443. doi: 10.1016/s0140-6736(96)05059-3. [DOI] [PubMed] [Google Scholar]
- 592.Staib F. Ecological and epidemiological aspects of aspergilli pathogenic for man and animal in Berlin (West) Zentralbl Bakteriol Mikrobiol Hyg [A] 1984;257:240–245. [PubMed] [Google Scholar]
- 593.Hutto C., Little E.A., Ricks R., Lee J.D., Pass R.F. Isolation of cytomegalovirus from toys and hands in a day care center. J Infect Dis. 1986;154:527–530. doi: 10.1093/infdis/154.3.527. [DOI] [PubMed] [Google Scholar]
- 594.Rogers M., Weinstock D.M., Eagan J., Kiehn T., Armstrong D., Sepkowitz K.A. Rotavirus outbreak on a pediatric oncology floor: possible association with toys. Am J Infect Control. 2000;28:378–380. doi: 10.1067/mic.2000.109908. [DOI] [PubMed] [Google Scholar]
- 595.Fleming K., Randle J. Toys—friend or foe? A study of infection risk in a paediatric intensive care unit. Paediatr Nurs. 2006;18:14–18. [PubMed] [Google Scholar]
- 596.Davies M.W., Mehr S., Garland S.T., Morley C.J. Bacterial colonization of toys in neonatal intensive care cots. Pediatrics. 2000;106:E18. doi: 10.1542/peds.106.2.e18. [DOI] [PubMed] [Google Scholar]
- 597.Buttery J.P., Alabaster S.J., Heine R.G. Multiresistant Pseudomonas aeruginosa outbreak in a pediatric oncology ward related to bath toys. Pediatr Infect Dis J. 1998;17:509–513. doi: 10.1097/00006454-199806000-00015. [DOI] [PubMed] [Google Scholar]
- 598.Centers for Disease Control and Prevention (CDC) US Department of Health and Human Services, CDC; Atlanta, GA: 2000. ABC's of Safe and Healthy Child Care: An On-line Handbook for Child Care Providers. [Google Scholar]
- 599.Bolyard E.A., Tablan O.C., Williams W.W., Pearson M.L., Shapiro C.N., Deitchmann S.D. Guideline for infection control in healthcare personnel, 1998. Hospital Infection Control Practices Advisory Committee. Infect Control Hosp Epidemiol. 1998;19:407–463. doi: 10.1086/647840. [DOI] [PubMed] [Google Scholar]
- 600.Stover B.H., Bratcher D.F. Varicella-zoster virus: infection, control, and prevention. Am J Infect Control. 1998;26:369–381. doi: 10.1016/s0196-6553(98)80025-4. quiz 382–384. [DOI] [PubMed] [Google Scholar]
- 601.Centers for Disease Control and Prevention (CDC) Recommendations for preventing transmission of human immunodeficiency virus and hepatitis B virus to patients during exposure-prone invasive procedures. MMWR Recomm Rep. 1991;40:1–9. [PubMed] [Google Scholar]
- 602.Management of healthcare workers infected with hepatitis B virus, hepatitis C virus, human immunodeficiency virus, or other bloodborne pathogens. AIDS/TB Committee of the Society for Healthcare Epidemiology of America. Infect Control Hosp Epidemiol. 1997;18:349–363. [PubMed] [Google Scholar]
- 603.Centers for Disease Control and Prevention (CDC) Centers for Disease Control and Prevention; Atlanta, GA: 2007. Infection Control Guidance for the Prevention and Control of Influenza in Acute-Care Facilities. [Google Scholar]
- 604.Couriel D., Carpenter P.A., Cutler C. Ancillary therapy and supportive care of chronic graft-versus-host disease: national institutes of health consensus development project on criteria for clinical trials in chronic Graft-versus-host disease: V. Ancillary Therapy and Supportive Care Working Group Report. Biol Blood Marrow Transplant. 2006;12:375–396. doi: 10.1016/j.bbmt.2006.02.003. [DOI] [PubMed] [Google Scholar]
- 605.Keefe D.M., Schubert M.M., Elting L.S. Updated clinical practice guidelines for the prevention and treatment of mucositis. Cancer. 2007;109:820–831. doi: 10.1002/cncr.22484. [DOI] [PubMed] [Google Scholar]
- 606.Barker G.J. Current practices in the oral management of the patient undergoing chemotherapy or bone marrow transplantation. Support Care Cancer. 1999;7:17–20. doi: 10.1007/s005200050217. [DOI] [PubMed] [Google Scholar]
- 607.Schubert M.M., Peterson D.E., Lloid M.E. Oral complications. In: Thomas E.D., Blume K.G., Forman S.J., editors. Hematopoietic Cell Transplantation. Blackwell Science, Inc.; Oxford, England: 1999. pp. 751–763. [Google Scholar]
- 608.Kyle R.A., Yee G.C., Somerfield M.R. American Society of Clinical Oncology 2007 clinical practice guideline update on the role of bisphosphonates in multiple myeloma. J Clin Oncol. 2007;25:2464–2472. doi: 10.1200/JCO.2007.12.1269. [DOI] [PubMed] [Google Scholar]
- 609.Lacy M.Q., Dispenzieri A., Gertz M.A. Mayo clinic consensus statement for the use of bisphosphonates in multiple myeloma. Mayo Clin Proc. 2006;81:1047–1053. doi: 10.4065/81.8.1047. [DOI] [PubMed] [Google Scholar]
- 610.Migliorati C.A., Siegel M.A., Elting L.S. Bisphosphonate-associated osteonecrosis: a long-term complication of bisphosphonate treatment. Lancet Oncol. 2006;7:508–514. doi: 10.1016/S1470-2045(06)70726-4. [DOI] [PubMed] [Google Scholar]
- 611.Spielberger R., Stiff P., Bensinger W. Palifermin for oral mucositis after intensive therapy for hematologic cancers. N Engl J Med. 2004;351:2590–2598. doi: 10.1056/NEJMoa040125. [DOI] [PubMed] [Google Scholar]
- 612.Rubenstein E.B., Peterson D.E., Schubert M. Clinical practice guidelines for the prevention and treatment of cancer therapy-induced oral and gastrointestinal mucositis. Cancer. 2004;100:2026–2046. doi: 10.1002/cncr.20163. [DOI] [PubMed] [Google Scholar]
- 613.Schubert M.M., Correa M.E. Oral graft-versus-host disease. Dent Clin North Am. 2008;52:79–109. doi: 10.1016/j.cden.2007.10.004. viii-ix. [DOI] [PubMed] [Google Scholar]
- 614.Do A.N., Ray B.J., Banerjee S.N. Bloodstream infection associated with needleless device use and the importance of infection-control practices in the home health care setting. J Infect Dis. 1999;179:442–448. doi: 10.1086/314592. [DOI] [PubMed] [Google Scholar]
- 615.Kool J.L., Fiore A.E., Kioski C.M. More than 10 years of unrecognized nosocomial transmission of Legionnaires' disease among transplant patients. Infect Control Hosp Epidemiol. 1998;19:898–904. doi: 10.1086/647760. [DOI] [PubMed] [Google Scholar]
- 616.Oren I., Zuckerman T., Avivi I., Finkelstein R., Yigla M., Rowe J.M. Nosocomial outbreak of Legionella pneumophila serogroup 3 pneumonia in a new bone marrow transplant unit: evaluation, treatment and control. Bone Marrow Transplant. 2002;30:175–179. doi: 10.1038/sj.bmt.1703628. [DOI] [PubMed] [Google Scholar]
- 617.Knirsch C.A., Jakob K., Schoonmaker D. An outbreak of Legionella micdadei pneumonia in transplant patients: evaluation, molecular epidemiology, and control. Am J Med. 2000;108:290–295. doi: 10.1016/s0002-9343(99)00459-3. [DOI] [PubMed] [Google Scholar]
- 618.Centers for Disease Control and Prevention (CDC) Sustained transmission of nosocomial Legionnaires disease—Arizona and Ohio. MMWR Morb Mortal Wkly Rep. 1997;46:416–421. [PubMed] [Google Scholar]
- 619.Palmore TN, Stock F, White M, et al. “A cluster of cases of nosocomial legionnaires disease linked to contaminated hospital decorative water fountain” Infect Control Hosp Epidemiol. 2009 Aug;30(8):764-8. [DOI] [PMC free article] [PubMed]
- 620.Squier C.L., Stout J.E., Krsytofiak S. A proactive approach to prevention of health care-acquired Legionnaires' disease: the Allegheny County (Pittsburgh) experience. Am J Infect Control. 2005;33:360–367. doi: 10.1016/j.ajic.2005.01.012. [DOI] [PubMed] [Google Scholar]
- 621.Stout J.E., Muder R.R., Mietzner S. Role of environmental surveillance in determining the risk of hospital-acquired legionellosis: a national surveillance study with clinical correlations. Infect Control Hosp Epidemiol. 2007;28:818–824. doi: 10.1086/518754. [DOI] [PubMed] [Google Scholar]
- 622.Muto C.A., Jernigan J.A., Ostrowsky B.E. SHEA guideline for preventing nosocomial transmission of multidrug-resistant strains of Staphylococcus aureus and enterococcus. Infect Control Hosp Epidemiol. 2003;24:362–386. doi: 10.1086/502213. [DOI] [PubMed] [Google Scholar]
- 623.Dutch Workingparty Infection Prevention. Policy for Methicillin-resistant Staphylococcus aureus;The Lancet Infectious Diseases, 2005;5(10): 653-663. [DOI] [PMC free article] [PubMed]
- 624.Siegel J.D., Rhinehart E., Jackson M., Chiarello L. Management of Multidrug-Resistant Organisms In Healthcare Settings, 2006. CDC; Atlanta, GA: 2006. Healthcare Infection Control Practices Advisory Committee. [DOI] [PubMed] [Google Scholar]
- 625.Coia J.E., Duckworth G.J., Edwards D.I. Guidelines for the control and prevention of meticillin-resistant Staphylococcus aureus (MRSA) in healthcare facilities. J Hosp Infect. 2006;63:S1–S44. doi: 10.1016/j.jhin.2006.01.001. [DOI] [PubMed] [Google Scholar]
- 626.Harbarth S., Fankhauser C., Schrenzel J. Universal screening for methicillin-resistant Staphylococcus aureus at hospital admission and nosocomial infection in surgical patients. JAMA. 2008;299:1149–1157. doi: 10.1001/jama.299.10.1149. [DOI] [PubMed] [Google Scholar]
- 627.Robicsek A., Beaumont J.L., Paule S.M. Universal surveillance for methicillin-resistant Staphylococcus aureus in 3 affiliated hospitals. Ann Intern Med. 2008;148:409–418. doi: 10.7326/0003-4819-148-6-200803180-00003. [DOI] [PubMed] [Google Scholar]
- 628.Huang S.S., Yokoe D.S., Hinrichsen V.L. Impact of routine intensive care unit surveillance cultures and resultant barrier precautions on hospital-wide methicillin-resistant Staphylococcus aureus bacteremia. Clin Infect Dis. 2006;43:971–978. doi: 10.1086/507636. [DOI] [PubMed] [Google Scholar]
- 629.Ridenour G., Lampen R., Federspiel J., Kritchevsky S., Wong E., Climo M. Selective use of intranasal mupirocin and chlorhexidine bathing and the incidence of methicillin-resistant Staphylococcus aureus colonization and infection among intensive care unit patients. Infect Control Hosp Epidemiol. 2007;28:1155–1161. doi: 10.1086/520102. [DOI] [PubMed] [Google Scholar]
- 630.Harbarth S., Dharan S., Liassine N., Herrault P., Auckenthaler R., Pittet D. Randomized, placebo-controlled, double-blind trial to evaluate the efficacy of mupirocin for eradicating carriage of methicillin-resistant. Staphylococcus aureus. Antimicrob Agents Chemother. 1999;43:1412–1416. doi: 10.1128/aac.43.6.1412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 631.Simor A.E., Loeb M. The management of infection and colonization due to methicillin-resistant Staphylococcus aureus: a CIDS/CAMM position paper. Can J Infect Dis. 2004;15:39–48. doi: 10.1155/2004/531434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 632.Leski T.A., Gniadkowski M., Skoczynska A., Stefaniuk E., Trzcinski K., Hryniewicz W. Outbreak of mupirocin-resistant Staphylococci in a hospital in Warsaw, Poland, due to plasmid transmission and clonal spread of several strains. J Clin Microbiol. 1999;37:2781–2788. doi: 10.1128/jcm.37.9.2781-2788.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 633.Schmitz F., Lindenlauf E., Hofmann B. The prevalence of low- and high-level mupirocin resistance in staphylococci from 19 European hospitals. J Antimicrob Chemother. 1998;42:489–495. doi: 10.1093/jac/42.4.489. [DOI] [PubMed] [Google Scholar]
- 634.Irish D., Eltringham I., Teall A. Control of an outbreak of an epidemic methicillin-resistant Staphylococcus aureus also resistant to mupirocin. J Hosp Infect. 1998;39:19–26. doi: 10.1016/s0195-6701(98)90239-0. [DOI] [PubMed] [Google Scholar]
- 635.Udo E.E., Jacob L.E., Mathew B. The spread of a mupirocin-resistant/methicillin-resistant Staphylococcus aureus clone in Kuwait hospitals. Acta Tropica. 2001;80:155–161. doi: 10.1016/s0001-706x(01)00171-1. [DOI] [PubMed] [Google Scholar]
- 636.Bastos M.D., Mondino P.J., Azevedo M.L., Santos K.R., Giambiaqui-deMarval M.M. Molecular characterization and transfer among Staphylococcus strains of a plasmid conferring high-level resistance to mupirocin. Eur J Clin Microbiol Infect Dis. 1999;18:393–398. doi: 10.1007/s100960050306. [DOI] [PubMed] [Google Scholar]
- 637.Centers for Disease Control. Vancomycin-Intermediate/Resistant Staphylococcus aureus Laboratory Testing Algorithm. Atlanta, GA: CDC, 2009:1.
- 638.Centers for Disease Control and Prevention (CDC) Recommendations for preventing the spread of vancomycin resistance. Recommendations of the Hospital Infection Control Practices Advisory Committee (HICPAC) MMWR Recomm Rep. 1995;22:1–13. [PubMed] [Google Scholar]
- 639.NCCLS Performance standards for antimicrobial disk susceptibility testing. Fourteenth informational supplement. NCCLS document. 2004:M100–M514. [Google Scholar]
- 640.Centers for Disease Control and Prevention (CDC) Update: Staphylococcus aureus with reduced susceptibility to vancomycin—United States, 1997. MMWR Morb Mortal Wkly Rep. 1997;46:813–815. [PubMed] [Google Scholar]
- 641.Smith T.L., Pearson M.L., Wilcox K.R. emergence of vancomycin resistance in. Staphylococcus aureus. N Engl J Med. 1999;340:493–501. doi: 10.1056/NEJM199902183400701. [DOI] [PubMed] [Google Scholar]
- 642.Centers for Disease Control and Prevention Interim guidelines for prevention and control of Staphylococcal infection associated with reduced susceptibility to vancomycin. MMWR Morb Mortal Wkly Rep. 1997;46:626–628. [PubMed] [Google Scholar]
- 643.Centers for Disease Control and Prevention Staphylococcus aureus resistant to vancomycin—United States, 2002. MMWR Morb Mortal Wkly Rep. 2002;51:565–567. [PubMed] [Google Scholar]
- 644.Waldvogel F.A. New resistance in Staphylococcus aureus. N Engl J Med. 1999;340:556–557. doi: 10.1056/NEJM199902183400709. [DOI] [PubMed] [Google Scholar]
- 645.Dellit T.H., Owens R.C., McGowan J.E., Jr. Infectious Diseases Society of America and the Society for Healthcare Epidemiology of America guidelines for developing an institutional program to enhance antimicrobial stewardship. Clin Infect Dis. 2007;44:159–177. doi: 10.1086/510393. [DOI] [PubMed] [Google Scholar]
- 646.Avery R., Kalaycio M., Pohlman B. Early vancomycin-resistant enterococcus (VRE) bacteremia after allogeneic bone marrow transplantation is associated with a rapidly deteriorating clinical course. Bone Marrow Transplant. 2005;35:497–499. doi: 10.1038/sj.bmt.1704821. [DOI] [PubMed] [Google Scholar]
- 647.Dubberke E.R., Hollands J.M., Georgantopoulos P. Vancomycin-resistant enterococcal bloodstream infections on a hematopoietic stem cell transplant unit: are the sick getting sicker? Bone Marrow Transplant. 2006;38:813–819. doi: 10.1038/sj.bmt.1705530. [DOI] [PubMed] [Google Scholar]
- 648.Montecalvo M.A., Shay D.K., Patel P. Bloodstream infections with vancomycin-resistant enterococci. Arch Intern Med. 1996;156:1458–1462. [PubMed] [Google Scholar]
- 649.Kirkpatrick B.D., Harrington S.M., Smith D. An outbreak of vancomycin-dependent Enterococcus faecium in a bone marrow transplant unit. Clin Infect Dis. 1999;29:1268–1273. doi: 10.1086/313456. [DOI] [PubMed] [Google Scholar]
- 650.Donskey C.J., Chowdhry T.K., Hecker M.T. Effect of antibiotic therapy on the density of vancomycin-resistant enterococci in the stool of colonized patients. N Engl J Med. 2000;343:1925–1932. doi: 10.1056/NEJM200012283432604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 651.Zar F.A., Bakkanagari S.R., Moorthi K.M., Davis M.B. A comparison of vancomycin and metronidazole for the treatment of Clostridium difficile-associated diarrhea, stratified by disease severity. Clin Infect Dis. 2007;45:302–307. doi: 10.1086/519265. [DOI] [PubMed] [Google Scholar]
- 652.Montecalvo M.A., de Lencastre H., Carraher M. Natural history of colonization with vancomycin-resistant. Enterococcus faecium. Infect Control Hosp Epidemiol. 1995;16:680–685. doi: 10.1086/647041. [DOI] [PubMed] [Google Scholar]
- 653.Baden L.R., Thiemke W., Skolnik A. Prolonged colonization with vancomycin-resistant Enterococcus faecium in long-term care patients and the significance of “clearance”. Clin Infect Dis. 2001;33:1654–1660. doi: 10.1086/323762. [DOI] [PubMed] [Google Scholar]
- 654.Donskey C.J., Hoyen C.K., Das S.M., Helfand M.S., Hecker M.T. Recurrence of vancomycin-resistant Enterococcus stool colonization during antibiotic therapy. Infect Control Hosp Epidemiol. 2002;23:436–440. doi: 10.1086/502081. [DOI] [PubMed] [Google Scholar]
- 655.Boyce J.M., Mermel L.A., Zervos M.J. Controlling vancomycin-resistant enterococci. Infect Control Hosp Epidemiol. 1995;16:634–637. doi: 10.1086/647028. [DOI] [PubMed] [Google Scholar]
- 656.Pena C., Pujol M., Ardanuy C. Epidemiology and successful control of a large outbreak due to Klebsiella pneumoniae producing extended-spectrum beta-lactamases. Antimicrob Agents Chemother. 1998;42:53–58. doi: 10.1128/aac.42.1.53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 657.Troche G., Joly L.M., Guibert M., Zazzo J.F. Detection and treatment of antibiotic-resistant bacterial carriage in a surgical intensive care unit: a 6-year prospective survey. Infect Control Hosp Epidemiol. 2005;26:161–165. doi: 10.1086/502521. [DOI] [PubMed] [Google Scholar]
- 658.Reddy P., Malczynski M., Obias A. Screening for extended-spectrum beta-lactamase-producing Enterobacteriaceae among high-risk patients and rates of subsequent bacteremia. Clin Infect Dis. 2007;45:846–852. doi: 10.1086/521260. [DOI] [PubMed] [Google Scholar]
- 659.Gardam M.A., Burrows L.L., Kus J.V. Is surveillance for multidrug-resistant enterobacteriaceae an effective infection control strategy in the absence of an outbreak? J Infect Dis. 2002;186:1754–1760. doi: 10.1086/345921. [DOI] [PubMed] [Google Scholar]
- 660.Centers for Disease Control and Prevention (CDC) Guidance for control of infections with carbapenem-resistant or carbapenemase-producing Enterobacteriaceae in acute care facilities. MMWR Morb Mortal Wkly Rep. 2009;58:256–260. [PubMed] [Google Scholar]
- 661.Gerding D.N., Johnson S., Peterson L.R., Mulligan M.E., Silva J., Jr. Clostridium difficile-associated diarrhea and colitis. Infect Control Hosp Epidemiol. 1995;16:459–477. doi: 10.1086/648363. [DOI] [PubMed] [Google Scholar]
- 662.Johnson S., Gerding D.N. Clostridium difficile-associated diarrhea. Clin Infect Dis. 1998;26:1027–1034. doi: 10.1086/520276. [DOI] [PubMed] [Google Scholar]
- 663.McFarland L.V., Mulligan M.E., Kwok R.Y., Stamm W.E. Nosocomial acquisition of Clostridium difficile infection. N Engl J Med. 1989;320:204–210. doi: 10.1056/NEJM198901263200402. [DOI] [PubMed] [Google Scholar]
- 664.Johnson S., Gerding D.N., Olson M.M. Prospective, controlled study of vinyl glove use to interrupt Clostridium difficile nosocomial transmission. Am J Med. 1990;88:137–140. doi: 10.1016/0002-9343(90)90462-m. [DOI] [PubMed] [Google Scholar]
- 665.Bobulsky G.S., Al-Nassir W.N., Riggs M.M., Sethi A.K., Donskey C.J. Clostridium difficile skin contamination in patients with C. difficile-associated disease. Clin Infect Dis. 2008;46:447–450. doi: 10.1086/525267. [DOI] [PubMed] [Google Scholar]
- 666.Johnson S., Homann S.R., Bettin K.M. Treatment of asymptomatic Clostridium difficile carriers (fecal excretors) with vancomycin or metronidazole. A randomized, placebo-controlled trial. Ann Intern Med. 1992;117:297–302. doi: 10.7326/0003-4819-117-4-297. [DOI] [PubMed] [Google Scholar]
- 667.Boyce J.M., Ligi C., Kohan C., Dumigan D., Havill N.L. Lack of association between the increased incidence of Clostridium difficile-associated disease and the increasing use of alcohol-based hand rubs. Infect Control Hosp Epidemiol. 2006;27:479–483. doi: 10.1086/504362. [DOI] [PubMed] [Google Scholar]
- 668.Weber D.J., Sickbert-Bennett E., Gergen M.F., Rutala W.A. Efficacy of selected hand hygiene agents used to remove Bacillus atrophaeus (a surrogate of Bacillus anthracis) from contaminated hands. JAMA. 2003;289:1274–1277. doi: 10.1001/jama.289.10.1274. [DOI] [PubMed] [Google Scholar]
- 669.Perez J., Springthorpe V.S., Sattar S.A. Activity of selected oxidizing microbicides against the spores of Clostridium difficile: relevance to environmental control. Am J Infect Control. 2005;33:320–325. doi: 10.1016/j.ajic.2005.04.240. [DOI] [PubMed] [Google Scholar]
- 670.Mayfield J.L., Leet T., Miller J., Mundy L.M. Environmental control to reduce transmission of. Clostridium difficile. Clin Infect Dis. 2000;31:995–1000. doi: 10.1086/318149. [DOI] [PubMed] [Google Scholar]
- 671.Dykewicz C.A. Guidelines for preventing opportunistic infections among hematopoietic stem cell transplant recipients: focus on community respiratory virus infections. Biol Blood Marrow Transplant. 2001;7(Suppl):19S–22S. doi: 10.1053/bbmt.2001.v7.pm11777100. [DOI] [PubMed] [Google Scholar]
- 672.Whimbey E., Englund J.A., Couch R.B. Community respiratory virus infections in immunocompromised patients with cancer. Am J Med. 1997;102:10–18. doi: 10.1016/S0002-9343(97)80004-6. discussion 25–26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 673.Harrington R.D., Hooton T.M., Hackman R.C. An outbreak of respiratory syncytial virus in a bone marrow transplant center. J Infect Dis. 1992;165:987–993. doi: 10.1093/infdis/165.6.987. [DOI] [PubMed] [Google Scholar]
- 674.Fox J.P., Brandt C.D., Wassermann F.E. The virus watch program: a continuing surveillance of viral infections in metropolitan New York families. VI. Observations of adenovirus infections: virus excretion patterns, antibody response, efficiency of surveillance, patterns of infections, and relation to illness. Am J Epidemiol. 1969;89:25–50. doi: 10.1093/oxfordjournals.aje.a120913. [DOI] [PubMed] [Google Scholar]
- 675.Hillis W.O., Cooper M.R., Bang F.B. Adenovirus infection in West Bengal. I: persistence of viruses in infants and young children. Indian J Med Res. 1973;61:980–988. [PubMed] [Google Scholar]
- 676.Hall C.B., Powell K.R., MacDonald N.E. Respiratory syncytial viral infection in children with compromised immune function. N Engl J Med. 1986;315:77–81. doi: 10.1056/NEJM198607103150201. [DOI] [PubMed] [Google Scholar]
- 677.Roghmann M., Ball K., Erdman D., Lovchik J., Anderson L.J., Edelman R. Active surveillance for respiratory virus infections in adults who have undergone bone marrow and peripheral blood stem cell transplantation. Bone Marrow Transplant. 2003;32:1085–1088. doi: 10.1038/sj.bmt.1704257. [DOI] [PubMed] [Google Scholar]
- 678.Madge P., Paton J.Y., McColl J.H., Mackie P.L. Prospective controlled study of four infection-control procedures to prevent nosocomial infection with respiratory syncytial virus. Lancet. 1992;340:1079–1083. doi: 10.1016/0140-6736(92)93088-5. [DOI] [PubMed] [Google Scholar]
- 679.Vu D., Peck A.J., Nichols W.G. Safety and tolerability of oseltamivir prophylaxis in hematopoietic stem cell transplant recipients: a retrospective case-control study. Clin Infect Dis. 2007;45:187–193. doi: 10.1086/518985. [DOI] [PubMed] [Google Scholar]
- 680.Jalal H., Bibby D.F., Tang J.W. First reported outbreak of diarrhea due to adenovirus infection in a hematology unit for adults. J Clin Microbiol. 2005;43:2575–2580. doi: 10.1128/JCM.43.6.2575-2580.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 681.Musher D.M. How contagious are common respiratory tract infections? N Engl J Med. 2003;348:1256–1266. doi: 10.1056/NEJMra021771. [DOI] [PubMed] [Google Scholar]
- 682.Nauheim R.C., Romanowski E.G., Araullo-Cruz T. Prolonged recoverability of desiccated adenovirus type 19 from various surfaces. Ophthalmology. 1990;97:1450–1453. doi: 10.1016/s0161-6420(90)32389-8. [DOI] [PubMed] [Google Scholar]
- 683.Gordon Y.J., Gordon R.Y., Romanowski E., Araullo-Cruz T.P. Prolonged recovery of desiccated adenoviral serotypes 5, 8, and 19 from plastic and metal surfaces in vitro. Ophthalmology. 1993;100:1835–1839. doi: 10.1016/s0161-6420(93)31389-8. discussion 1839–18340. [DOI] [PubMed] [Google Scholar]
- 684.Mahl M.C., Sadler C. Virus survival on inanimate surfaces. Can J Microbiol. 1975;21:819–823. doi: 10.1139/m75-121. [DOI] [PubMed] [Google Scholar]
- 685.Sattar S.A., Abebe M., Bueti A.J., Jampani H., Newman J., Hua S. Activity of an alcohol-based hand gel against human adeno-, rhino-, and rotaviruses using the fingerpad method. Infect Control Hosp Epidemiol. 2000;21:516–519. doi: 10.1086/501796. [DOI] [PubMed] [Google Scholar]
- 686.Rutala W.A., Peacock J.E., Gergen M.F., Sobsey M.D., Weber D.J. Efficacy of hospital germicides against adenovirus 8, a common cause of epidemic keratoconjunctivitis in health care facilities. Antimicrob Agents Chemother. 2006;50:1419–1424. doi: 10.1128/AAC.50.4.1419-1424.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 687.Butz A.M., Fosarelli P., Dick J., Cusack T., Yolken R. Prevalence of rotavirus on high-risk fomites in day-care facilities. Pediatrics. 1993;92:202–205. [PubMed] [Google Scholar]
- 688.Sattar S.A., Lloyd-Evans N., Springthorpe V.S., Nair R.C. Institutional outbreaks of rotavirus diarrhoea: potential role of fomites and environmental surfaces as vehicles for virus transmission. J Hyg (Lond) 1986;96:277–289. doi: 10.1017/s0022172400066055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 689.Gallimore C.I., Taylor C., Gennery A.R. Environmental monitoring for gastroenteric viruses in a pediatric primary immunodeficiency unit. J Clin Microbiol. 2006;44:395–399. doi: 10.1128/JCM.44.2.395-399.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 690.Ansari S.A., Sattar S.A., Springthorpe V.S., Wells G.A., Tostowaryk W. Rotavirus survival on human hands and transfer of infectious virus to animate and nonporous inanimate surfaces. J Clin Microbiol. 1988;26:1513–1518. doi: 10.1128/jcm.26.8.1513-1518.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 691.Lages S.L., Ramakrishnan M.A., Goyal S.M. In-vivo efficacy of hand sanitisers against feline calicivirus: a surrogate for norovirus. J Hosp Infect. 2008;62:159–163. doi: 10.1016/j.jhin.2007.11.018. [DOI] [PubMed] [Google Scholar]
- 692.Kampf G., Grotheer D., Steinmann J. Efficacy of three ethanol-based hand rubs against feline calicivirus, a surrogate virus for norovirus. J Hosp Infect. 2005;60:144–149. doi: 10.1016/j.jhin.2004.12.005. [DOI] [PubMed] [Google Scholar]
- 693.Gehrke C., Steinmann J., Goroncy-Bermes P. Inactivation of feline calicivirus, a surrogate of norovirus (formerly Norwalk-like viruses), by different types of alcohol in vitro and in vivo. J Hosp Infect. 2004;56:49–55. doi: 10.1016/j.jhin.2003.08.019. [DOI] [PubMed] [Google Scholar]
- 694.Duizer E., Bijkerk P., Rockx B., De Groot A., Twisk F., Koopmans M. Inactivation of caliciviruses. Appl Environ Microbiol. 2004;70:4538–4543. doi: 10.1128/AEM.70.8.4538-4543.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 695.Centers for Disease Control and Prevention (CDC) National Center for Preparedness, Detection, and Control of Infectious Diseases; Atlanta, GA: 2006. Norovirus in Healthcare Facilities Fact Sheet. [Google Scholar]
- 696.Gallimore C.I., Taylor C., Gennery A.R. Use of a heminested reverse transcriptase PCR assay for detection of astrovirus in environmental swabs from an outbreak of gastroenteritis in a pediatric primary immunodeficiency unit. J Clin Microbiol. 2005;43:3890–3894. doi: 10.1128/JCM.43.8.3890-3894.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 697.Marshall J.A., Bruggink L.D., Sturge K., Subasinghe N., Tan A., Hogg G.G. Molecular features of astrovirus associated with a gastroenteritis outbreak in an aged-care centre. Eur J Clin Microbiol Infect Dis. 2007;26:67–71. doi: 10.1007/s10096-006-0250-8. [DOI] [PubMed] [Google Scholar]
- 698.Glass R.I., Noel J., Mitchell D. The changing epidemiology of astrovirus-associated gastroenteritis: a review. Arch Virol Suppl. 1996;12:287–300. doi: 10.1007/978-3-7091-6553-9_31. [DOI] [PubMed] [Google Scholar]
- 699.Sebire N.J., Malone M., Shah N., Anderson G., Gaspar H.B., Cubitt W.D. Pathology of astrovirus associated diarrhoea in a paediatric bone marrow transplant recipient. J Clin Pathol. 2004;57:1001–1013. doi: 10.1136/jcp.2004.017178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 700.LeBaron C.W., Furutan N.P., Lew J.F. Viral agents of gastroenteritis. Public health importance and outbreak management. MMWR Recomm Rep. 1990;39:1–24. [PubMed] [Google Scholar]
- 701.RP Gaynes Horan TC. Surveillance of nosocomial infections. In: CG Mayhall, ed. Hospital Epidemiology and Infection Control. Philadelphia, PA: Lippincott Williams & Wilkins, 1999:1285–1317.
- 702.De Pauw B., Walsh T.J., Donnelly J.P. Revised definitions of invasive fungal disease from the European Organization for Research and Treatment of Cancer/Invasive Fungal Infections Cooperative Group and the National Institute of Allergy and Infectious Diseases Mycoses Study Group (EORTC/MSG) Consensus Group. Clin Infect Dis. 2008;25:1813–1821. doi: 10.1086/588660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 703.Lee G.M., Salomon J.A., Friedman J.F. Illness transmission in the home: a possible role for alcohol-based hand gels. Pediatrics. 2005;115:852–860. doi: 10.1542/peds.2004-0856. [DOI] [PubMed] [Google Scholar]
- 704.Sandora T.J., Taveras E.M., Shih M.C. A randomized, controlled trial of a multifaceted intervention including alcohol-based hand sanitizer and hand-hygiene education to reduce illness transmission in the home. Pediatrics. 2005;116:587–594. doi: 10.1542/peds.2005-0199. [DOI] [PubMed] [Google Scholar]
- 705.Centers for Disease Control and Prevention . CDC; Atlanta, GA: 2005. Preventing Cryptosporidiosis: A Guide for People with Compromised Immune System. [Google Scholar]
- 706.Masur H., Kaplan J.E., Holmes K.K. Guidelines for preventing opportunistic infections among HIV-infected persons—2002. Recommendations of the U.S. Public Health Service and the Infectious Diseases Society of America. Ann Intern Med. 2002;137:435–478. doi: 10.7326/0003-4819-137-5_part_2-200209031-00002. [DOI] [PubMed] [Google Scholar]
- 707.Kerr L.N., Chaput M.P., Cash L.D. Assessment of the durability of medical examination gloves. J Occup Environ Hyg. 2004;1 doi: 10.1080/15459620490491803. 607–512. [DOI] [PubMed] [Google Scholar]
- 708.Korniewicz D.M., El-Masri M., Broyles J.M., Martin C.D., O'Connell K.P. Performance of latex and nonlatex medical examination gloves during simulated use. Am J Infect Control. 2002;30:133–138. doi: 10.1067/mic.2002.119512. [DOI] [PubMed] [Google Scholar]
- 709.Kim Y.J., Boeckh M., Englund J.A. Community respiratory virus infections in immunocompromised patients: hematopoietic stem cell and solid organ transplant recipients, and individuals with human immunodeficiency virus infection. Semin Respir Crit Care Med. 2007;28:222–242. doi: 10.1055/s-2007-976494. [DOI] [PubMed] [Google Scholar]
- 710.Luby S.P., Agboatwalla M., Feikin D.R. Effect of handwashing on child health: a randomised controlled trial. Lancet. 2005;366:225–233. doi: 10.1016/S0140-6736(05)66912-7. [DOI] [PubMed] [Google Scholar]
- 711.Roberts L., Smith W., Jorm L., Patel M., Douglas R.M., McGilchrist C. Effect of infection control measures on the frequency of upper respiratory infection in child care: a randomized, controlled trial. Pediatrics. 2000;105:738–742. doi: 10.1542/peds.105.4.738. [DOI] [PubMed] [Google Scholar]
- 712.Ryan M.A., Christian R.S., Wohlrabe J. Handwashing and respiratory illness among young adults in military training. Am J Prev Med. 2001;21:79–83. doi: 10.1016/s0749-3797(01)00323-3. [DOI] [PubMed] [Google Scholar]
- 713.White C., Kolble R., Carlson R. The effect of hand hygiene on illness rate among students in university residence halls. Am J Infect Control. 2003;31:364–370. doi: 10.1016/s0196-6553(03)00041-5. [DOI] [PubMed] [Google Scholar]
- 714.Lau J.T., Tsui H., Lau M., Yang X. SARS transmission, risk factors, and prevention in Hong Kong. Emerg Infect Dis. 2004;10:587–592. doi: 10.3201/eid1004.030628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 715.Chatterjee A., Plummer S., Heybrock B. A modified “cover your cough” campaign prevents exposures of employees to pertussis at a children's hospital. Am J Infect Control. 2007;35:489–491. doi: 10.1016/j.ajic.2007.02.008. [DOI] [PubMed] [Google Scholar]
- 716.Centers for Disease Control and Prevention Legionnaires' Disease associated with potting soil—California, Oregon, and Washington, May-June 2000. MMWR Morb Mortal Wkly Rep. 2000;49:777–778. [PubMed] [Google Scholar]
- 717.Lopez A., Dietz V.J., Wilson M., Navin T.R., Jones J.L. Preventing congenital toxoplasmosis. MMWR Recomm Rep. 2000;49:59–68. [PubMed] [Google Scholar]
- 718.O'Connor B.A., Carman J., Eckert K., Tucker G., Givney R., Cameron S. Does using potting mix make you sick? Results from a Legionella longbeachae case-control study in South Australia. Epidemiol Infect. 2007;135:34–39. doi: 10.1017/S095026880600656X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 719.De Groote M.A., Pace N.R., Fulton K., Falkinham J.O., 3rd Relationships between Mycobacterium isolates from patients with pulmonary mycobacterial infection and potting soils. Appl Environ Microbiol. 2006;72:7602–7606. doi: 10.1128/AEM.00930-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 720.Wilson M., Robson J., Pyke C.M., McCormack J.G. Saksenaea vasiformis breast abscess related to gardening injury. Aust N Z J Med. 1998;28:845–846. doi: 10.1111/j.1445-5994.1998.tb01573.x. [DOI] [PubMed] [Google Scholar]
- 721.Golledge C. An infection from a penetrating plant wound. Aust Fam Phys. 1995;24:2099. [PubMed] [Google Scholar]
- 722.Siddiqui S., Anderson V.L., Hilligoss D.M. Fulminant mulch pneumonitis: an emergency presentation of chronic granulomatous disease. Clin Infect Dis. 2007;45:673–681. doi: 10.1086/520985. [DOI] [PubMed] [Google Scholar]
- 723.Stark D., Milliken S., Marriott D., Harkness J. Rhizopus microsporus var. rhizopodiformis sinus-orbital zygomycosis in an immunosuppressed patient: successful treatment with posaconazole after a complicated clinical course. J Med Microbiol. 2007;56:699–701. doi: 10.1099/jmm.0.47069-0. [DOI] [PubMed] [Google Scholar]
- 724.Praz-Christinaz S.M., Lazor-Blanchet C., Binet I., Boillat M.A., Danuser B. Occupational risk assessment of aspergillosis after renal transplantation. Transpl Infect Dis. 2007;9:175–181. doi: 10.1111/j.1399-3062.2007.00223.x. [DOI] [PubMed] [Google Scholar]
- 725.Glenn T.J., Blair J.E., Adams R.H. Coccidioidomycosis in hematopoietic stem cell transplant recipients. Med Mycol. 2005;43:705–710. doi: 10.1080/13693780500147840. [DOI] [PubMed] [Google Scholar]
- 726.Blair J.E. Coccidioidomycosis in patients who have undergone transplantation. Ann N Y Acad Sci. 2007;1111:365–376. doi: 10.1196/annals.1406.009. [DOI] [PubMed] [Google Scholar]
- 727.Centers for Disease Control and Prevention Increase in coccidioidomycosis—Arizona, 1998-2001. MMWR Morb Mortal Wkly Rep. 2003;52:109–112. [PubMed] [Google Scholar]
- 728.Lyon G.M., Bravo A.V., Espino A. Histoplasmosis associated with exploring a bat-inhabited cave in Costa Rica, 1998-1999. Am J Trop Med Hyg. 2004;70:438–442. [PubMed] [Google Scholar]
- 729.Huhn G.D., Austin C., Carr M. Two outbreaks of occupationally acquired histoplasmosis: more than workers at risk. Environ Health Perspect. 2005;113:585–589. doi: 10.1289/ehp.7484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 730.Centers for Disease Control and Prevention Outbreak of histoplasmosis among industrial plant workers—Nebraska, 2004. MMWR Morb Mortal Wkly Rep. 2004;53:1020–1022. [PubMed] [Google Scholar]
- 731.Jie T., Matas A.J., Gillingham K.J., Sutherland D.E., Dunn D.L., Humar A. Mycobacterial infections after kidney transplant. Transplant Proc. 2005;37 doi: 10.1016/j.transproceed.2004.12.167. 937–739. [DOI] [PubMed] [Google Scholar]
- 732.Centers for Disease Control and Prevention Prevention and control of tuberculosis in correctional and detention facilities: recommendations from CDC. Endorsed by the Advisory Council for the Elimination of Tuberculosis, the National Commission on Correctional Health Care, and the American Correctional Association. MMWR Recomm Rep. 2006;55:1–44. [PubMed] [Google Scholar]
- 733.Lavoie J., Dunkerley C.J., Kosatsky T., Dufresne A. Exposure to aerosolized bacteria and fungi among collectors of commercial, mixed residential, recyclable and compostable waste. Sci Total Environ. 2006;370:23–28. doi: 10.1016/j.scitotenv.2006.05.016. [DOI] [PubMed] [Google Scholar]
- 734.Prevots D.R., Burr R.K., Sutter R.W., Murphy T.V. Poliomyelitis prevention in the United States. Updated recommendations of the Advisory Committee on Immunization Practices (ACIP) MMWR Recomm Rep. 2000;49:1–22. quiz CE1-7. [PubMed] [Google Scholar]
- 735.Parashar U.D., Alexander J.P., Glass R.I. Prevention of rotavirus gastroenteritis among infants and children. Recommendations of the Advisory Committee on Immunization Practices (ACIP) MMWR Recomm Rep. 2006;55:1–13. [PubMed] [Google Scholar]
- 736.Vesikari T., Karvonen A., Korhonen T. A randomized, double-blind study of the safety, transmissibility and phenotypic and genotypic stability of cold-adapted influenza virus vaccine. Pediatr Infect Dis J. 2006;25:590–595. doi: 10.1097/01.inf.0000220229.51531.47. [DOI] [PubMed] [Google Scholar]
- 737.Araujo E.C., Clemens S.A., Oliveira C.S. Safety, immunogenicity, and protective efficacy of two doses of RIX4414 live attenuated human rotavirus vaccine in healthy infants. J Pediatr (Rio J) 2007;83:217–224. doi: 10.2223/JPED.1600. [DOI] [PubMed] [Google Scholar]
- 738.Ruiz-Palacios G.M., Perez-Schael I., Velazquez F.R. Safety and efficacy of an attenuated vaccine against severe rotavirus gastroenteritis. N Engl J Med. 2006;354:11–22. doi: 10.1056/NEJMoa052434. [DOI] [PubMed] [Google Scholar]
- 739.Clinical Review for STN 125265/0 Rotarix; 2008.
- 740.Ali T., Scott N., Kallas W. Detection of influenza antigen with rapid antibody-based tests after intranasal influenza vaccination (FluMist) Clin Infect Dis. 2004;38:760–762. doi: 10.1086/382887. [DOI] [PubMed] [Google Scholar]
- 741.Talbot T.R., Crocker D.D., Peters J. Duration of virus shedding after trivalent intranasal live attenuated influenza vaccination in adults. Infect Control Hosp Epidemiol. 2005;26:494–500. doi: 10.1086/502574. [DOI] [PubMed] [Google Scholar]
- 742.Cha T.A., Kao K., Zhao J., Fast P.E., Mendelman P.M., Arvin A. Genotypic stability of cold-adapted influenza virus vaccine in an efficacy clinical trial. J Clin Microbiol. 2000;38:839–845. doi: 10.1128/jcm.38.2.839-845.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 743.Buonagurio D.A., O'Neill R.E., Shutyak L. Genetic and phenotypic stability of cold-adapted influenza viruses in a trivalent vaccine administered to children in a day care setting. Virology. 2006;347:296–306. doi: 10.1016/j.virol.2005.11.006. [DOI] [PubMed] [Google Scholar]
- 744.ACIP Provisional Recommendations for use of Zoster Vaccine. 2008.
- 745.Diaz P.S., Au D., Smith S. Lack of transmission of the live attenuated varicella vaccine virus to immunocompromised children after immunization of their siblings. Pediatrics. 1991;87:166–170. [PubMed] [Google Scholar]
- 746.Centers for Disease Control and Prevention (CDC). “Crypto”—Cryptosporidiosis: prevention—immunocompromised persons. 2008.
- 747.Angulo F.J., Glaser C.A., Juranek D.D., Lappin M.R., Regnery R.L. Caring for pets of immunocompromised persons. J Am Vet Med Assoc. 1994;205:1711–1718. [PubMed] [Google Scholar]
- 748.Elliot D.L., Tolle S.W., Goldberg L., Miller J.B. Pet-associated illness. N Engl J Med. 1985;313:985–995. doi: 10.1056/NEJM198510173131605. [DOI] [PubMed] [Google Scholar]
- 749.Glaser C.A., Angulo F.J., Rooney J.A. Animal-associated opportunistic infections among persons infected with the human immunodeficiency virus. Clin Infect Dis. 1994;18:14–24. doi: 10.1093/clinids/18.1.14. [DOI] [PubMed] [Google Scholar]
- 750.Dalton C., Hoffman R., Pape J. Iguana-associated salmonellosis in children. Pediatr Infect Dis J. 1995;14:319–320. doi: 10.1097/00006454-199504000-00014. [DOI] [PubMed] [Google Scholar]
- 751.Centers for Disease Control and Prevention (CDC) Reptile-associated salmonellosis—selected states, 1998-2002. MMWR Morb Mortal Wkly Rep. 2003;52:1206–1209. [PubMed] [Google Scholar]
- 752.Mermin J., Hoar B., Angulo F.J. Iguanas and Salmonella marina infection in children: a reflection of the increasing incidence of reptile-associated salmonellosis in the United States. Pediatrics. 1997;99:399–402. doi: 10.1542/peds.99.3.399. [DOI] [PubMed] [Google Scholar]
- 753.Centers for Disease Control and Prevention (CDC) Salmonellosis associated with chicks and ducklings—Michigan and Missouri, Spring 1999. MMWR Morb Mortal Wkly Rep. 2000;49:297–299. [PubMed] [Google Scholar]
- 754.Adams R.M. Animals in schools: a zoonosis threat? Pediatr Infect Dis J. 1998;17:174–176. doi: 10.1097/00006454-199802000-00025. [DOI] [PubMed] [Google Scholar]
- 755.Berkelman R.L. Human illness associated with use of veterinary vaccines. Clin Infect Dis. 2003;37:407–414. doi: 10.1086/375595. [DOI] [PubMed] [Google Scholar]
- 756.DiSalvo H., Haiduven D., Johnson N. Who let the dogs out? Infection control did: utility of dogs in health care settings and infection control aspects. Am J Infect Control. 2006;34:301–307. doi: 10.1016/j.ajic.2005.06.005. [DOI] [PubMed] [Google Scholar]
- 757.Centers for Disease Control and Prevention (CDC) Lake-associated outbreak of Escherichia coli O157:H7—Illinois, 1995. MMWR Morb Mortal Wkly Rep. 1996;45:437–439. [PubMed] [Google Scholar]
- 758.Centers for Disease Control and Prevention (CDC) Outbreak of cryptosporidiosis associated with a water sprinkler fountain—Minnesota, 1997. MMWR Morb Mortal Wkly Rep. 1998;47:856–860. [PubMed] [Google Scholar]
- 759.Kramer M.H., Sorhage F.E., Goldstein S.T., Dalley E., Wahlquist S.P., Herwaldt B.L. First reported outbreak in the United States of cryptosporidiosis associated with a recreational lake. Clin Infect Dis. 1998;26:27–33. doi: 10.1086/516271. [DOI] [PubMed] [Google Scholar]
- 760.Centers for Disease Control and Prevention (CDC) Centers for Disease Control and Prevention; Atlanta, GA: 2008. Risks from Food and Water (Drinking and Recreational) [Google Scholar]
- 761.van Kraaij M.G., Dekker A.W., Verdonck L.F. Infectious gastro-enteritis: an uncommon cause of diarrhoea in adult allogeneic and autologous stem cell transplant recipients. Bone Marrow Transplant. 2000;26:299–303. doi: 10.1038/sj.bmt.1702484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 762.American Academy of Pediatrics . Cryptosporidiosis. In: Pickering L.K., editor. Red Book: 2003 Report of the Committee on Infectious Diseases. American Academy of Pediatrics; Elk Grove Village, IL: 2003. pp. 123–127. [Google Scholar]
- 763.American Public Health Association . American Public Health Association; Washington, DC: 2004. Cryptosporidiosis. Control of Communicable Disease Manual. 138-141. [Google Scholar]
- 764.Keene W.E., Hedberg K., Herriott D.E. A prolonged outbreak of Escherichia coli O157:H7 infections caused by commercially distributed raw milk. J Infect Dis. 1997;176:815–818. doi: 10.1086/517310. [DOI] [PubMed] [Google Scholar]
- 765.Shen M.W. Diagnostic and therapeutic challenges of childhood brucellosis in a nonendemic country. Pediatrics. 2008;121:e1178–e1183. doi: 10.1542/peds.2007-1874. [DOI] [PubMed] [Google Scholar]
- 766.Vojdani J.D., Beuchat L.R., Tauxe R.V. Juice-associated outbreaks of human illness in the United States, 1995 through 2005. J Food Protein. 2008;71:356–364. doi: 10.4315/0362-028x-71.2.356. [DOI] [PubMed] [Google Scholar]
- 767.Gardner A., Mattiuzzi G., Faderl S. Randomized comparison of cooked and noncooked diets in patients undergoing remission induction therapy for acute myeloid leukemia. J Clin Oncol. 2008;26:5684–5688. doi: 10.1200/JCO.2008.16.4681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 768.Handle and Prepare Food Safely, 2008.
- 769.Todd E.C. Epidemiology of foodborne diseases: a worldwide review. World Health Stat Q. 1997;50:30–50. [PubMed] [Google Scholar]
- 770.The Big Thaw—Safe Defrosting Methods for Consumers, 2008.
- 771.Moe G.L. Low-microbial diets for patients with granulocytopenia. In: Bloch A.S., editor. Nutrition Management of the Cancer Patient. Aspen Publishing, Inc.; Rockville, MD: 1990. pp. 125–134. [Google Scholar]
- 772.Aker S.N., Lenssen P. Nutritional support of patients with hematologic malignancies. In: Benz E.J. Jr., Cohen J.J., Burie B., editors. Hematology: Basic Principles and Practice. Churchill Livingstone; New York: 2000. pp. 1501–1514. [Google Scholar]
- 773.Moody K., Charlson M.E., Finlay J. The neutropenic diet: what's the evidence? J Pediatr Hematol Oncol. 2002;24:717–721. doi: 10.1097/00043426-200212000-00007. [DOI] [PubMed] [Google Scholar]
- 774.Centers for Disease Control and Prevention (CDC) Outbreaks of Salmonella serotype enteritidis infection associated with eating shell eggs—United States, 1999-2001. MMWR Morb Mortal Wkly Rep. 2003;51:1149–1152. [PubMed] [Google Scholar]
- 775.Sazawal S., Hiremath G., Dhingra U., Malik P., Deb S., Black R.E. Efficacy of probiotics in prevention of acute diarrhoea: a meta-analysis of masked, randomised, placebo-controlled trials. Lancet Infect Dis. 2006;6:374–382. doi: 10.1016/S1473-3099(06)70495-9. [DOI] [PubMed] [Google Scholar]
- 776.Falagas M.E., Betsi G.I., Athanasiou S. Probiotics for the treatment of women with bacterial vaginosis. Clin Microbiol Infect. 2007;13:657–664. doi: 10.1111/j.1469-0691.2007.01688.x. [DOI] [PubMed] [Google Scholar]
- 777.Antony S.J., Stratton C.W., Dummer J.S. Lactobacillus bacteremia: description of the clinical course in adult patients without endocarditis. Clin Infect Dis. 1996;23:773–778. doi: 10.1093/clinids/23.4.773. [DOI] [PubMed] [Google Scholar]
- 778.Oliver M.R., Van Voorhis W.C., Boeckh M., Mattson D., Bowden R.A. Hepatic mucormycosis in a bone marrow transplant recipient who ingested naturopathic medicine. Clin Infect Dis. 1996;22:521–524. doi: 10.1093/clinids/22.3.521. [DOI] [PubMed] [Google Scholar]
- 779.Centers for Disease Control and Prevention (CDC) Centers for Disease Control and Prevention; Atlanta, GA: 2007. Travelers's Health: Destinations. [Google Scholar]
- 780.Centers for Disease Control and Prevention (CDC) Outbreak of acute febrile illness among athletes participating in triathlons—Wisconsin and Illinois, 1998. MMWR Morb Mortal Wkly Rep. 1998;47:585–588. [PubMed] [Google Scholar]
- 781.Centers for Disease Control and Prevention (CDC) Outbreak of leptospirosis among white-water rafters—Costa Rica, 1996. MMWR Morb Mortal Wkly Rep. 1997;46:577–579. [PubMed] [Google Scholar]
- 782.Sejvar J., Bancroft E., Winthrop K. Leptospirosis in “Eco-Challenge” athletes, Malaysian Borneo, 2000. Emerg Infect Dis. 2003;9:702–707. doi: 10.3201/eid0906.020751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 783.Leder K., Newman D. Respiratory infections during air travel. Intern Med J. 2005;35:50–55. doi: 10.1111/j.1445-5994.2004.00696.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 784.Centers for Disease Control and Prevention (CDC) Centers for Disease Control and Prevention; Atlanta, GA: 2008. Travelers' Health: Yellow Book. [Google Scholar]
- 785.Centers for Disease Control and Prevention (CDC). Water Treatment Methods. Atlanta, GA: Centers for Disease Control and Prevention.
- 786.Ljungman P., Cordonnier C., de Bock R. Immunisations after bone marrow transplantation: results of a European survey and recommendations from the infectious diseases working party of the European Group for Blood and Marrow Transplantation. Bone Marrow Transplant. 1995;15:455–460. [PubMed] [Google Scholar]
- 787.Ljungman P., Engelhard D., de la Camara R. Vaccination of stem cell transplant recipients: recommendations of the Infectious Diseases Working Party of the EBMT. Bone Marrow Transplant. 2005;35:737–746. doi: 10.1038/sj.bmt.1704870. [DOI] [PubMed] [Google Scholar]
- 788.Ljungman P., Fridell E., Lönnqvist B. Efficacy and safety of vaccination of marrow transplant recipients with a live attenuated measles, mumps, and rubella vaccine. J Infect Dis. 1989;159:610–615. doi: 10.1093/infdis/159.4.610. [DOI] [PubMed] [Google Scholar]
- 789.Ljungman P., Wiklund H.M., Duraj V. Response to tetanus toxoid immunization after allogeneic bone marrow transplantation. J Infect Dis. 1990;162:496–500. doi: 10.1093/infdis/162.2.496. [DOI] [PubMed] [Google Scholar]
- 790.Pauksen K., Duraj V., Ljungman P. Immunity to and immunization against measles, rubella and mumps in patients after autologous bone marrow transplantation. Bone Marrow Transplant. 1992;9:427–432. [PubMed] [Google Scholar]
- 791.Pauksen K, V H, Ljungman P, et al. Immunity to poliovirus and immunization with inactivated poliovaccine after autologous bone marrow transplantation. Clin Infect Dis. 1994;18:547–552. [DOI] [PubMed]
- 792.Guinan E.C., Molrine D.C., Antin J.H. Polysaccharide conjugate vaccine responses in bone marrow transplant patients. Transplantation. 1994;57:677–684. doi: 10.1097/00007890-199403150-00009. [DOI] [PubMed] [Google Scholar]
- 793.Youssef S., Rodriguez G., Rolston K.V., Champlin R.E., Raad, Safdar A. Streptococcus pneumoniae infections in 47 hematopoietic stem cell transplantation recipients: clinical characteristics of infections and vaccine-breakthrough infections, 1989-2005. Medicine. 2007;86:69–77. doi: 10.1097/md.0b013e31803eb176. [DOI] [PubMed] [Google Scholar]
- 794.Locksley R.M., Flournoy N., Sullivan K.M., Meyers J.D. Infection with varicella-zoster virus after marrow transplantation. J Infect Dis. 1985;152:1172–1181. doi: 10.1093/infdis/152.6.1172. [DOI] [PubMed] [Google Scholar]
- 795.Nakano T., Shimono Y., Sugiyama K. Clinical features of measles in immunocompromised children. Acta Paediatr Jpn. 1996;38:212–217. doi: 10.1111/j.1442-200x.1996.tb03472.x. [DOI] [PubMed] [Google Scholar]
- 796.Machado C.M., Goncalves F.B., Pannuti C.S., Dulley F.L., de Souza V.A. Measles in bone marrow transplant recipients during an outbreak in Sao Paulo, Brazil. Blood. 2002;99:83–87. doi: 10.1182/blood.v99.1.83. [DOI] [PubMed] [Google Scholar]
- 797.Kumar D., Chen M.H., Welsh B. A randomized, double-blind trial of pneumococcal vaccination in adult allogeneic stem cell transplant donors and recipients. Clin Infect Dis. 2007;45:1576–1582. doi: 10.1086/523583. [DOI] [PubMed] [Google Scholar]
- 798.Machado C.M. Influenza infections after hematopoietic stem cell transplantation. Clin Infect Dis. 2005;41:273–274. doi: 10.1086/431304. [DOI] [PubMed] [Google Scholar]
- 799.Parkman R., Weinberg K. Immunological reconstitution following hematopoietic stem cell transplantation. In: Blume K., Forman S., Appelbaum F., editors. Thomas's Hematopoietic Cell Transplantation. Blackwell Science; Malden, MA: 2003. pp. 853–861. [Google Scholar]
- 800.Storek J., Witherspoon R. Immunological reconstitution after hemopoietic stem cell transplantation. In: Atkinson K., Champlin R., Ritz J., Fibbe W., Ljungman P., Brenner M., editors. Clinical Bone Marrow and Blood Stem Cell Transplantation. Cambridge University Press; Cambridge: 2004. pp. 194–226. [Google Scholar]
- 801.Horwitz S.M., Negrin R.S., Blume K.G. Rituximab as adjuvant to high-dose therapy and autologous hematopoietic cell transplantation for aggressive non-Hodgkin lymphoma. Blood. 2004;103:777–783. doi: 10.1182/blood-2003-04-1257. [DOI] [PubMed] [Google Scholar]
- 802.Nordoy T., Husebekk A., Aaberge I.S. Humoral immunity to viral and bacterial antigens in lymphoma patients 4-10 years after high-dose therapy with ABMT. Serological responses to revaccinations according to EBMT guidelines. Bone Marrow Transplant. 2001;28:681–687. doi: 10.1038/sj.bmt.1703228. [DOI] [PubMed] [Google Scholar]
- 803.Pauksen K., Linde A., Hammarstrom V. Granulocyte-macrophage colony-stimulating factor as immunomodulating factor together with influenza vaccination in stem cell transplant patients. Clin Infect Dis. 2000;30:342–348. doi: 10.1086/313663. [DOI] [PubMed] [Google Scholar]
- 804.Gandhi M.K., Egner W., Sizer L. Antibody responses to vaccinations given within the first two years after transplant are similar between autologous peripheral blood stem cell and bone marrow transplant recipients. Bone Marrow Transplant. 2001;28:775–781. doi: 10.1038/sj.bmt.1703239. [DOI] [PubMed] [Google Scholar]
- 805.van der Velden A.M., Claessen A.M., van Velzen-Blad H., Biesma D.H., Rijker G.T. Development of functional Haemophilus influenzae type b antibodies after vaccination. Haematologica. 2005;90:1582–1584. [PubMed] [Google Scholar]
- 806.Meisel R., Kuypers L., Dirksen U. Pneumococcal conjugate vaccine provides early protective antibody responses in children after related and unrelated allogeneic hematopoietic stem cell transplantation. Blood. 2007;109:2322–2326. doi: 10.1182/blood-2006-06-032284. [DOI] [PubMed] [Google Scholar]
- 807.Molrine D.C., Antin J.H., Guinan E.C. Donor immunization with pneumococcal conjugate vaccine and early protective antibody responses following allogeneic hematopoietic cell transplantation. Blood. 2003;101:831–836. doi: 10.1182/blood-2002-03-0832. [DOI] [PubMed] [Google Scholar]
- 808.Cordonnier C., Labopin M., Chesnel V. Immune response to the 23-valent polysaccharide pneumococcal vaccine (PPV23) after the 7-valent conjugate vaccine (PCV7) in allogeneic stem cell transplant (SCT) recipients: results of the EBMT IDWP01 trial. Blood. 2008;112:350. doi: 10.1016/j.vaccine.2010.01.025. [DOI] [PubMed] [Google Scholar]
- 809.Papadopoulos E.B., Young J.W., Kernan N.A. Use of the tetanus toxoid, reduced dose diphtheria and pertussis vaccine (Tdap) in allogeneic transplant (alloHCT) recipients [Abstract] Blood. 2008;112:2214. [Google Scholar]
- 810.Small TN, Zelenetz AD, Noy A, et al. Pertussis Immunity and Response to tetanus toxoid, reduced diphtheria toxoid and acellular pertussis vaccine (Tdap) following autologous peripheral blood stem cell transplantation. Biol Blood Marrow Transplant (in press) [DOI] [PubMed]
- 811.Parkkali T., Kayhty H., Lehtonen H. Tetravalent meningococcal polysaccharide vaccine is immunogenic in adult allogeneic BMT recipients. Bone Marrow Transplant. 2001;27:79–84. doi: 10.1038/sj.bmt.1702742. [DOI] [PubMed] [Google Scholar]
- 812.Mahler M., Kernan N.A., Boulad F. Immunogenicity of the tetravalent protein-conjugated meningococcal vaccine in recipients of related and unrelated allogeneic hematopoietic cell transplants (HCT) [Abstract] Blood. 2008;112:1163. doi: 10.1016/j.bbmt.2011.07.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 813.Geddes M., Storek J. Immune reconstitution following hematopoietic stem-cell transplantation. Best Pract Res Clin Haematol. 2007;20:329–348. doi: 10.1016/j.beha.2006.09.009. [DOI] [PubMed] [Google Scholar]
- 814.Joao C., Porrata L.F., Inwards D.J. Early lymphocyte recovery after autologous stem cell transplantation predicts superior survival in mantle-cell lymphoma. Bone Marrow Transplant. 2006;37:865–871. doi: 10.1038/sj.bmt.1705342. [DOI] [PubMed] [Google Scholar]
- 815.Savani B.N., Mielke S., Rezvani K. Absolute lymphocyte count on day 30 is a surrogate for robust hematopoietic recovery and strongly predicts outcome after T cell-depleted allogeneic stem cell transplantation. Biol Blood Marrow Transplant. 2007;13:1216–1223. doi: 10.1016/j.bbmt.2007.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 816.Savani B.N., Rezvani K., Mielke S. Factors associated with early molecular remission after T cell-depleted allogeneic stem cell transplantation for chronic myelogenous leukemia. Blood. 2006;107:1688–1695. doi: 10.1182/blood-2005-05-1897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 817.Baron F., Baker J.E., Storb R. Kinetics of engraftment in patients with hematologic malignancies given allogeneic hematopoietic cell transplantation after nonmyeloablative conditioning. Blood. 2004;104:2254–2262. doi: 10.1182/blood-2004-04-1506. [DOI] [PubMed] [Google Scholar]
- 818.Storek J., Espino G., Dawson M.A., Storer B., Flowers M.E.D., Maloney D.G. Low B cell and monocyte counts on day 80 are associated with high infection rates between day 100 and 365 after allogeneic marrow transplantation. Blood. 2000;96:3290–3293. [PubMed] [Google Scholar]
- 819.Novitzky N., Davison G.M., Hale G., Waldmann H. Immune reconstitution at 6 months following T-cell depleted hematopoietic stem cell transplantation is predictive for treatment outcome. Transplantation. 2002;74:1551–1559. doi: 10.1097/00007890-200212150-00012. [DOI] [PubMed] [Google Scholar]
- 820.Boeckh M., Nakamura R., Cornelissen J. Immune monitoring with iTAg (TM) MHC tetramers for prediction of recurrent or persistent CMV infection in allogeneic stem cell transplant recipients: a prospective multicenter clinical trial. Biol Blood Marrow Transplant. 2006;12:79. doi: 10.1182/blood-2010-03-273508. [DOI] [PubMed] [Google Scholar]
- 821.Porrata L.F., Inwards D.J., Ansell S.M. Early lymphocyte recovery predicts superior survival after autologous stem cell transplantation in non-Hodgkin lymphoma: a prospective study. Biol Blood Marrow Transplant. 2008;14:807–816. doi: 10.1016/j.bbmt.2008.04.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 822.Chang Y.J., Zhao X.Y., Huang X.J. Effects of the NK cell recovery on outcomes of unmanipulated haploidentical blood and marrow transplantation for patients with hematologic malignancies. Biol Blood Marrow Transplant. 2008;14:323–334. doi: 10.1016/j.bbmt.2007.12.497. [DOI] [PubMed] [Google Scholar]
- 823.Rocha V., Franco R.F., Porcher R. Host defense and inflammatory gene polymorphisms are associated with outcomes after HLA-identical sibling bone marrow transplantation. Blood. 2002;100:3908–3918. doi: 10.1182/blood-2002-04-1033. [DOI] [PubMed] [Google Scholar]
- 824.Holler E., Rogler G., Brenmoehl J. Prognostic significance of NOD2/CARD15 variants in HLA-identical sibling hematopoietic stem cell transplantation: effect on long-term outcome is confirmed in 2 independent cohorts and may be modulated by the type of gastrointestinal decontamination. Blood. 2006;107:4189–4193. doi: 10.1182/blood-2005-09-3741. [DOI] [PubMed] [Google Scholar]
- 825.Dickinson A.M. Risk assessment in haematopoietic stem cell transplantation: pre-transplant patient and donor factors: non-HLA genetics. Best Pract Res Clin Haematol. 2007;20:189–207. doi: 10.1016/j.beha.2006.10.004. [DOI] [PubMed] [Google Scholar]
- 826.Bochud P.Y., Chien J.W., Marr K.A. Toll-like receptor 4 polymorphisms and aspergillosis in stem-cell transplantation. N Engl J Med. 2008;359:1766–1777. doi: 10.1056/NEJMoa0802629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 827.Manning S.E., Rupprecht C.E., Fishbein D. Human rabies prevention—United States, 2008: recommendations of the Advisory Committee on Immunization Practices. MMWR Recomm Rep. 2008;57:1–28. [PubMed] [Google Scholar]
- 828.American Academy of Pediatrics. Rabies. In: LK Pickering, ed. Red Book: 2006 Report of the Committee on Infectious Diseases. Elk Grove Village, IL: American Academy of Pediatrics, 2006:552–559.
- 829.Fiore A.E., Wasley A., Bell B.P. Prevention of hepatitis A through active or passive immunization: recommendations of the Advisory Committee on Immunization Practices (ACIP) MMWR Recomm Rep. 2006;55:1–23. [PubMed] [Google Scholar]
- 830.Monto AS, Robinson DP, Herlocher ML, M. HJ, Jr., Elliott MJ, Crisp A. Zanamivir in the prevention of influenza among healthy adults: a randomized controlled trial. JAMA. 1999;282:31–35. [DOI] [PubMed]
- 831.Hayden F.G., Atmar R.L., Schilling M. Use of the selective oral neuraminidase inhibitor oseltamivir to prevent influenza. N Engl J Med. 1999;341:1336–1343. doi: 10.1056/NEJM199910283411802. [DOI] [PubMed] [Google Scholar]
- 832.Prevots D.R., Burr R.K., Sutter R.W., Murphy T.V. Poliomyelitis prevention in the United States. Updated recommendations of the Advisory Committee on Immunization Practices (ACIP) MMWR Recomm Rep. 2000;49:1–22. quiz CE1-7. [PubMed] [Google Scholar]
- 833.American Academy of Pediatrics. Poliovirus infections. In: LK Pickering, ed. Red Book: 2006 Report of the Committee on Infectious Diseases. Elk Grove Village, IL: American Academy of Pediatrics, 2006:542–547.
- 834.Burks A.W., Sampson H.A., Buckley R.H. Anaphylactic reactions after gamma globulin administration in patients with hypogammaglobulinemia. Detection of IgE antibodies to IgA. N Engl J Med. 1986;314:560–564. doi: 10.1056/NEJM198602273140907. [DOI] [PubMed] [Google Scholar]
- 835.Stiehm E.R. Human intravenous immunoglobulin in primary and secondary antibody deficiencies. Pediatr Infect Dis J. 1997;16:696–707. doi: 10.1097/00006454-199707000-00012. [DOI] [PubMed] [Google Scholar]
- 836.American Academy of Pediatrics. Passive Immunization. In: LK Pickering, ed. Red Book: 2006 Report of the Committee on Infectious Diseases. Elk Grove Village, IL: American Academy of Pediatrics, 2006:54–55.
- 837.Abe S., Miyamura K., Oba T. Oral ribavirin for severe adenovirus infection after allogeneic marrow transplantation. Bone Marrow Transplant. 2003;32:1107–1108. doi: 10.1038/sj.bmt.1704276. [DOI] [PubMed] [Google Scholar]
- 838.Cheng W.T., Li C.K., Leung T.F. Ribavirin for SARS in children. Clin.Pediatr. (Phila) 2004;43:193–196. doi: 10.1177/000992280404300211. [DOI] [PubMed] [Google Scholar]
- 839.Naquira C., Jimenez G., Guerra J.G. Ivermectin for human strongyloidiasis and other intestinal helminths. Am J Trop Med Hyg. 1989;40:304–309. doi: 10.4269/ajtmh.1989.40.304. [DOI] [PubMed] [Google Scholar]
- 840.Belay E.D., Bresee J.S., Holman R.C., Khan A.S., Shahriari A., Schonberger L.B. Reye's syndrome in the United States from 1981 through 1997. N Engl J Med. 1999;340:1377–1382. doi: 10.1056/NEJM199905063401801. [DOI] [PubMed] [Google Scholar]
- 841.Centers for Disease Control and Prevention (CDC) Prevention and treatment of tuberculosis among patients infected with human immunodeficiency virus: principles of therapy and revised recommendations. Centers for Disease Control and Prevention. MMWR Recomm Rep. 1998;47:1–58. [PubMed] [Google Scholar]
- 842.American Academy of Pediatrics . Tuberculosis. In: Pickering L.K., editor. Red Book: 2006 Report of the Committee on Infectious Diseases. American Academy of Pediatrics; Elk Grove Village, IL: 2006. pp. 678–698. [Google Scholar]